Abstract
Discharge of wastewater containing nitrate (NO3−) disrupts aquatic ecosystems even at low concentrations. However, selective and rapid reduction of NO3− at low concentration to dinitrogen (N2) is technically challenging. Here, we present an electrified membrane (EM) loaded with Sn pair-atom catalysts for highly efficient NO3− reduction to N2 in a single-pass electrofiltration. The pair-atom design facilitates coupling of adsorbed N intermediates on adjacent Sn atoms to enhance N2 selectivity, which is challenging with conventional fully-isolated single-atom catalyst design. The EM ensures sufficient exposure of the catalysts and intensifies the catalyst interaction with NO3− through mass transfer enhancement to provide more N intermediates for N2 coupling. We further develop a reduced titanium dioxide EM as the anode to generate free chlorines for fully oxidizing the residual ammonia (<1 mg-N L−1) to N2. The sequential cathode-to-anode electrofiltration realizes near-complete removal of 10 mg-N L−1 NO3− and ~100% N2 selectivity with a water resident time on the order of seconds. Our findings advance the single-atom catalyst design for NO3− reduction and provide a practical solution for NO3− contamination at low concentrations.
Similar content being viewed by others
Introduction
Nitrate (NO3−) contamination resulting from the discharge of inadequately treated wastewater disrupts aquatic ecosystems through eutrophication followed by harmful algal blooms1. The algal blooms can be triggered by NO3− at a concentration as low as 1 mg-N L−1 2, therefore, underscoring the need for water treatment systems to remove NO3− with low concentrations3. However, near-complete removal of NO3− at low concentrations in complex water matrices is difficult and costly. Electrocatalytic NO3− reduction reaction (NO3RR) to dinitrogen (N2) or ammonia (NH3) offers a promising alternative to be applied for NO3− removal from water. Electrochemical processes offer advantages including rapid reaction kinetics, adaptability to various treatment scenarios, the potential to be modularized for decentralized application, and minimal generation of secondary wastes such as sludges or concentrates4,5.
Numerous electrocatalysts have been developed for NO3RR towards NH3 instead of N2 with the goal of resource recovery6,7,8,9. However, the practicability of NH3 recovery from low concentration NO3− remains questionable, considering the complexity and high cost of separating dilute NH3 from water10. It is noteworthy that NH3 can be even more toxic compared to NO3−11, posing higher environmental risks. Therefore, developing NO3RR systems that can completely convert low-concentration NO3− to N2 with close to 100% selectivity is yet to be developed. Conventionally, NO3− reduction to N2 requires a precious noble metal12,13 in combination with a promoter metal (e.g., Cu) to improve the adsorption and charge transfer of the reactants and intermediates. However, the limited availability and high cost of the noble metals restrict their application for NO3RR14.
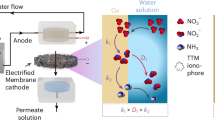
Single-atom catalysts (SACs) have emerged as a transformative alternative in catalyst material design. With close to 100% atomic efficiency, SACs expose more active surface sites than nanoparticle counterparts, offering unparalleled efficiency and selectivity in various catalytic schemes15. The strong interaction of SACs with the support via surface defects and N-atomic anchors also offers enhanced chemical stability16,17,18. Despite these advantages, the spatial isolation of catalytic sites in SAC architecture is not considered ideal for NO3RR toward N2. With fully isolated SACs, the *N intermediate generated on each SAC site cannot easily interact with another *N to undergo the *N-*N coupling step, which is critical for N2 generation8,19,20. This is the reason that most reported SACs are prone to promote the NH3 selectivity through *N-*H coupling21,22,23,24,25. (Fig. 1, left panel).
Schematic illustrating the design principles of incorporating single-atom catalysts in traditional plate electrode (left panel) or pair-atom catalysts in an electrified membrane for realizing near-complete conversion of low concentration nitrate to dinitrogen (right panel).
In this study, we propose a viable SAC architecture, pairs of tin (Sn) single atoms, to enhance the NO3RR selectivity toward N2 production (Fig. 1, right panel). Closely-spaced dual active sites intend to facilitate *N-*N coupling that are favored in multi-atom catalytic sites26. Sn is selected because of its rapid NO3RR kinetics and high N2 selectivity27,28. We incorporate Sn pair-atoms in an innovative electrified membrane (EM) to ensure their exposure in the membrane nanopores. The EM intensifies the catalyst interaction with NO3− during flow-through operation29,30,31,32, overcoming the mass transfer limitation of the conventional plate electrodes33,34 and providing more *N sources for N2 coupling35,36,37. We demonstrate highly efficient conversion of low concentration NO3− (10 mg-N L−1) to N2 in a single-pass filtration through the Sn pair-atom-loaded EM. We further incorporate a reduced titanium dioxide (TiO2-x) EM anodic bottom layer to generate free chlorines from chloride ion (Cl−) in water to fully oxidize the residual ammonia (NH3) downstream (<1 mg-N L−1) into N2.
Results
Synthesis and characterization of Sn single-atom catalysts
We loaded Sn single atoms on carbon black (CB) support using a ligand-mediated method (Fig. 2a)38. In brief, the Sn4+ complex with 1,10-phenanthroline (Phen) at a molar ratio of Sn4+:Phen = 1:3 was adsorbed onto CB at target Sn loading of 1 and 5 wt%. Subsequent annealing in Ar atmosphere at 600 °C for 3 h produced N-doped carbon black (NCB)-supported Sn SACs. Transmission electron microscopy (TEM) images showed the porous structure of NCB and confirmed the absence of nanoparticles (Fig. S1). Aberration-corrected high-angle annular dark field-scanning transmission electron microscopy (AC HAADF-STEM) images suggested the atomic dispersion of Sn atoms on NCB. At the target Sn loading of 5%, we predominantly observed pair-atom sites (i.e., two single atoms placed close to each other, denoted as Sn2/NCB, Fig. 2b). In contrast, fully-isolated Sn single-atom sites became more dominant over pair-atom sites at 1 wt% Sn loading (denoted as Sn1/NCB) (Fig. 2c). The formation of Sn pair-atoms may attribute to the generation of Phen-Sn4+ precursor binuclear complexes in a distorted trigonal bipyramidal configuration in the solution39,40,41. In addition, increasing the density of single atoms on the substrate to tune their site-to-site distance can create correlating atomic interactions between neighboring single atom sites42,43. When the dispersed single atoms are close enough in catalysts with a M1–N–C structure (M1 represents metal single atom), a strong interaction could occur between the two adjacent single atoms to form a double active center to improve the catalytic performance43. However, increasing only the density of single atoms to create pair-atom sites cannot precisely control the coordination configuration and distribution uniformity compared to methods such as the metal binuclear precursor deposition44,45. Therefore, both Sn pair-atom and single-atom sites were observed, yet the composition of the former was dominant with the increase of Sn loading. The energy dispersion spectroscopy (EDS) mapping of Sn2/NCB confirmed the uniform dispersion of Sn on the NCB support (Fig. 2d). Statistical image analysis determined the averaged distance between Sn atoms in Sn2/NCB at d = 4.8 ± 0.7 Å (Fig. 2e–g). We also confirmed the mass loading to be within 4% error from the target, using inductively coupled plasma-mass spectrometry (ICP-MS) analysis.
a Schematics of synthesis procedures of Sn2/NCB. b AC HAADF-STEM image of Sn2/NCB. c AC HAADF-STEM image of Sn1/NCB. d TEM EDS mapping images of Sn2/NCB. e Histogram of the distribution of distances between Sn pair-atoms. Three representative Sn pair-atoms sites: f enlarged AC HAADF-STEM images and surface intensity plots; g the variation in brightness along the Sn pair-atoms. Source data are provided as a Source Data file.
X-ray diffraction (XRD) patterns of Sn2/NCB and NCB showed similar broad peaks at 25° and 44° after pyrolysis (Fig. S2), indicating poor crystallinity of carbon. XRD analysis also confirmed the absence of metal nanoparticles. In Raman analysis, the relative intensity ratio between the D and G bands (ID/IG) of Sn2/NCB and NCB were 1.01 and 1.02 respectively, higher than the 0.99 of pure NCB (Fig. S3), consisting of the increased surface defects due to nitrogen doping46. X-ray photoelectron spectroscopy (XPS) of Sn2/NCB also confirmed the absence of characteristic Sn0 peak at ~485.0 eV47. XPS analysis further suggested that the binding energies of two Sn 3d peaks at 487.3 eV and 497.3 eV were close to the values reported for Sn4+ (Fig. S4a)47. The N 1s spectrum showed the presence of pyridinic N (399.0 eV), Sn–N (400.0 eV), pyrrolic N (400.8 eV), and graphitic N (401.5 eV) (Fig. S4b)48.
We further examined the oxidation state and coordination environment of Sn in Sn2/NCB using X-ray adsorption (XAS) spectroscopy. The X-ray absorption near edge structure (XANES) spectrum at Sn K-edge shows that the edge energy of Sn2/NCB was in between Sn foil and Sn(IV) phthalocyanine (SnPc) standards (Fig. 3a), suggesting that the oxidation state of Sn in Sn2/NCB (δ2+) was 0 < δ2+ < 4+. Note that there was a shift towards higher edge energy (eV) from Sn2/NCB to Sn1/NCB (Fig. 3b). The lower edge energy suggests that Sn pair-atoms would be in a more reduced state compared to single atoms (i.e., δ2+ < δ1+). A similar shift of electron distribution towards clustered single atoms due to the electronic interactions between short-distanced atoms has been observed with neighboring or ensemble single atoms49,50. The linear correlation analysis between the edge energy and oxidation state of Sn indicated that the oxidation states of Sn would be around δ2+ = 2.8 for Sn2/NCB and δ1+ = 3.6 for Sn1/NCB (Fig. 3c).
a, b Normalized XANES spectra of Sn K-edge. The arrow indicates the rise of oxidation state. c The linear correlation between the edge energy (eV) and oxidation state of Sn. d FT-EXAFS spectra. e FT-EXAFS fitting of Sn2/NCB. Inset: schematic diagram of the Sn-N4 structure. WT-XAFS spectra of (f) Sn foil, (g) SnPc, (h) Sn2/NCB, and (i) Sn1/NCB. Source data are provided as a Source Data file.
The Fourier transformed-extended X-ray absorption fine structure (FT-EXAFS) spectra of Sn2/NCB and Sn1/NCB exhibited major peaks at 1.57 Å and 1.58 Å, corresponding to the Sn-N coordination similar to the SnPc standard (Fig. 3d). The Sn2/NCB did not exhibit the peak of Sn-Sn coordination at 2.79 Å of Sn foil, indicating the absence of direct Sn-Sn interactions. The coordination number of Sn pair-atom in Sn2/NCB was estimated to be 4.1 ± 0.2 based on EXAFS fitting (Fig. 3e and Table S1), which suggested the formation of Sn-N4 structure for both pair-atoms. The k-space and R-space of Sn2/NCB, Sn1/NCB, SnPc, and Sn foil were additionally analyzed via wavelet transform (WT) to obtain 2D and 3D contour maps (Fig. 3f–i). The maximal peak near 4.9 Å−1 of Sn2/NCB was attributed to the Sn-N bond, which shifted slightly from the 5.2 Å−1 of Sn1/NCB. These indicated the differences in the Sn–N coordination in Sn2/NCB due to the coupling compared to fully isolated single-atom sites in Sn1/NCB.
Performance of Sn2/NCB in NO3RR and mechanistic insights
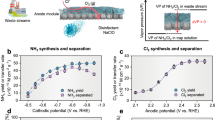
We first employed a batch cell to evaluate the NO3RR performance of Sn2/NCB. Sn2/NCB showed a higher current density compared to Sn1/NCB and NCB during linear sweep voltammetry (LSV) measurements, indicating its superior NO3RR activity than Sn1/NCB and NCB (Fig. 4a). The Sn2/NCB showed a comparable NO3− conversion rate (401.6 mg-N h−1 m−2) with Sn1/NCB (Fig. 4b). However, N2 selectivity was significantly higher with Sn2/NCB (86%) than Sn1/NCB (50%), consistent with our hypothesis that the Sn pair-atom architecture would be beneficial for enhancing the N2 generation over NH3. Sn2/NCB also exhibited higher NO3− conversion rate and N2 selectivity compared to other benchmark Sn materials such as nanocluster (1 wt% and 5 wt%), nanoparticle (10 wt% and 20 wt%), and bulk powder (Fig. S6). These results suggested that the pair-atom structure rather than the Sn loading played a critical role in enhancing the NO3RR performance. When applying Sn2/NCB for NO3RR under varied potentials, the NO3− conversion rate gradually increased with higher potentials, while the N2 selectivity started to decline above −1.2 V vs. RHE (Fig. 4c).
a LSV curves of Sn2/NCB with and without NO3−, Sn1/NCB with NO3−, and NCB with NO3−. The voltages were not iR corrected. b NO3− conversion rates and N2 selectivity of Sn2/NCB, Sn1/NCB, and NCB. c NO3− conversion rates and N2 selectivity of Sn2/NCB under varied potentials. The voltages were not iR corrected (electrode surface area = 1 cm2, resistance value = 3 Ω). The data in (b) and (c) are presented as mean values ± s.d. (n = 3). In situ ATR-SEIRAS and the corresponding contour maps of (d) Sn1/NCB and (e) Sn2/NCB during NO3RR. f The charge density difference plots of NO3− on Sn2/NCB and Sn1/NCB. g The adsorption energies of NO3− on CB, NCB, Sn1/NCB, and Sn2/NCB. h Gibbs free energy curves of NO3− reduction on Sn2/NCB. The inset molecular structures represent the most thermodynamically favorable N2 generation path on Sn2/NCB. Transition state energy diagrams of (i) *H and (j) *N spillover on Sn2/NCB and Sn1/NCB. Red, blue, white, and grey atoms represent O, N, H, and C elements respectively. Source data are provided as a Source Data file.
In situ attenuated total reflectance surface-enhanced infrared absorption spectroscopy (ATR-SEIRAS) was applied to identify the key N intermediates during NO3RR for Sn2/NCB and Sn1/NCB (Fig. 4d, e). Both groups displayed a negative peak at 1224 cm−1 corresponding to the Si–O stretching vibration51 and a bending vibration mode of H2O at 1640 cm−1 52. The peaks at 1330 cm−1, 1190 cm−1, and 1560 cm−1 were assigned to *NO3‒, *NO2‒, and *NO, respectively53. For Sn1/NCB, a clear peak at 1460 cm−1 from the stretching vibration of the N−H bond in *NH4+ was observed, along with the intermediates including *NH2 at 1140 cm−1 and *NH2OH at 1110 cm−1 52. A peak at 1030 cm−1 corresponded to the generation of *N254,55,56. In contrast, for Sn2/NCB, the peak intensity of *NH4+ at 1460 cm−1 along with other intermediates for NH3 production decreased significantly compared to Sn1/NCB, which was also observed from the corresponding contour maps. Further, the peak intensity of *N2 at 1030 cm−1 was more prominent, indicating that Sn2/NCB promoted the conversion of NO3− to N2 over NH3 compared to Sn1/NCB.
We further performed density functional theory (DFT) calculations to scrutinize the mechanisms of the enhanced N2 selectivity on Sn pair-atoms over single-atoms. We first evaluated the adsorption strength and charge transfer of NO3‒ on different substrates. The adsorption configurations of NO3‒ on Sn1/NCB and Sn2/NCB were optimized and shown in Fig. S7. Instead of the conventional vertical adsorption configuration on Sn single-atom, on Sn2/NCB, NO3‒ interacted with two adjacent Sn pair-atoms, with an inclined omega angle (N-Sn-O2-O1) of 87.1°. The coordinates of all atoms in the NO3‒ adsorbed Sn1/NCB and Sn2/NCB models were presented in Supplementary Data 1. The charge density difference plots of NO3‒ on NCB, Sn1/NCB, and Sn2/NCB (Figs. 4f, S8) indicated that the charge transfer from Sn pair-atoms to NO3‒ (1.02 e‒) was more significant than Sn single-atoms (0.93 e‒). The adsorption of NO3‒ on Sn2/NCB and Sn1/NCB was predominantly chemical adsorption, with adsorption energies of ‒1.54 eV and ‒1.89 eV, respectively (Fig. 4g), confirming the adsorption of NO3‒ on Sn2/NCB was stronger than that on Sn1/NCB. The projected density of states (PDOS) diagram showed that after Sn1 introduction, the band gap decreased from 0.68 eV of NCB to 0.23 eV of Sn1/NCB (Fig. S9). After further adding Sn atom to form Sn2/NCB, the contribution of the electronic peak was more obvious, and the band gap was further reduced to 0.14 eV, which would enhance the charge transfer across the catalyst. The Sn2/NCB possessed a slightly more positive d-band center and consequently a stronger adsorption strength than that of Sn1/NCB.
We further compared the lowest energy NO3RR pathways for Sn2/NCB versus Sn1/NCB (Fig. 4h and Fig. S10) and other control materials (Fig. S11). The reduction of NO3‒ to *NO followed the NO3 → *NO3 → *NO3H → *NO2 → *NO2H → *NO hydrogenation route. For NCB, the weak adsorption of NO3‒ was inconducive to the occurrence of the reaction. Further hydrogenation of *NO followed three pathways, generating *NOH, *ONH, and *N2O2H, respectively. On Sn1/NCB, the production of *NOH was inhibited by a positive ΔGmax of 1.0 eV. The N2 generation occurred through energetically favorable pathway of *NO → *N2O2H → *N2O → *N2 + OH → *N2. In contrast, the path toward NH3 (*NO → *ONH → *ONH2 → *OHNH2 → *NH2 → *NH3) only had a small energy barrier of ΔGmax of 0.1 eV from *NO to *ONH. Therefore, both two paths could happen, while the N2 generation was slightly more favorable. This was consistent with the selectivity of 50% for N2 and the selectivity of 38% for NH3 in batch NO3RR tests.
In contrast, the adsorbed *NO3 and *NO3H interacted with two Sn pair-atoms on Sn2/NCB. Consequently, their free energies were more negative than those in the case of Sn1/NCB. This indicates that the pair-atom interaction is more conducive to the initiation of the NO3RR. For the *NOH, *ONH, and *N2O2H pathways, the most thermodynamically favorable route was *NO → *NOH → *N → *N2. The Sn pair-atom design altered the dominant intermediate of N2 generation to *NOH, which contrasts from *N2O2H on Sn single-atoms. In comparison, the *ONH path had a ΔGmax of 0.9 eV of *ONH2 → *OHNH2, while the *N2O2H path had a ΔGmax of 0.7 eV of *N2O2H → *N2O, both inhibiting these pathways. The above calculations collectively suggest that Sn pair-atoms would preferentially lead to the production of N2 compared with NH3, consistent with the experimental results.
Finally, we examined the energetics of transition state involving *H and *N spillover, preceding N2 formation. The energy barriers for water dissociation on both Sn2/NCB and Sn1/NCB were similar (Fig. S12). For Sn2/NCB, *H formed on a Sn site could easily migrate and attack the *NO on the adjacent Sn site (Fig. 4i). In contrast, for Sn1/NCB, *H on a Sn atom needed to spillover onto and migrate across NCB support with an energy barrier of >0.8 eV to react with *NO on another distant Sn atom site. This difference accounts for more effective hydrogenation of *NO with Sn2/NCB than Sn1/NCB. The simulation also suggested that the energy barrier of coupling two *N atoms on Sn pair-atoms to form N2 was 0.8 eV (Fig. 4j), which was lower than the diffusion (spillover) barrier (0.9 eV) of *N from a Sn single-atoms to NCB support. These results confirm that efficient N2 formation through *N coupling is due to close proximity of *N atoms placed on neighboring Sn pair-atoms.
Design principle, fabrication, and characterization of bilayer electrified membrane
Based on N2-selective NO3RR demonstrated in the batch cell, we incorporated the Sn2/NCB onto a bilayer electrified membrane (bilayer-EM) to further improve the NO3RR kinetics and selectivity. As shown in Fig. 5a, the cathodic layer was placed on the top of the flow cell (i.e., toward the influent side) to selectively reduce NO3− to N2. The mass transfer of NO3− to catalytic sites would be enhanced due to convection across the porous electrode. Through the anodic bottom layer (i.e., placed below the cathode), free chlorines generated in situ would oxidize the small amount of NH3 generated from the cathode to N257,58. This bilayer-EM was designed to achieve sequential cathode-to-anode electrofiltration and to realize near-complete removal of low-concentration NO3− with ~100% N2 selectivity.
a Schematic showing the concept of applying the bilayer-EM with sequential cathode-to-anode electrofiltration for realizing near-complete NO3−-to-N2 reduction with ~100% selectivity. b Photograph and SEM images of (c) the top and (d) the cross-sectional views of the top-layer bilayer-EM (i.e., Sn2/NCB-EM). e Photograph, (f) top view SEM images, and (g) XPS of the bottom-layer bilayer-EM (i.e., TiO2-x-EM). h Pore size distribution and the average value of the Sn2/NCB-EM and the CNT-EM. i Water flux of the Sn2/NCB-EM, the CNT-EM, and the TiO2-x-EM. Water permeabilities of the membranes are indicated by the fitted slopes. j EIS spectra of the Sn2/NCB-EM, the CNT-EM, and the TiO2-x-EM over a frequency range of 1–106 Hz in 10 mM Na2SO4 solution. Source data are provided as a Source Data file.
We prepared a free-standing electrified membrane (Sn2/NCB-EM) that serves as the top layer of the bilayer-EM by coating the Sn2/NCB on the CNT interwoven framework. The membrane was morphologically uniform across the effective membrane area of 12.6 cm2 and mechanically robust as a flow-through electrode (Fig. 5b). The comparison of the top view SEM images between the catalyst-loaded membrane (Fig. 5c) and a control CNT electrified membrane (CNT-EM, Fig. S13) indicated that the CNT interwoven framework was well-coated with the Sn2/NCB. The cross-section SEM images showed a lamellar structure of the membrane with a thickness of 0.15 mm (Fig. 5d). We also prepared a TiO2-x ceramic electrified membrane (TiO2-x-EM) that serves as the bottom anodic layer of the bilayer-EM by reducing a TiO2 ceramic membrane in pure H2 atmosphere at 1100 °C (see Fig. S14a for photos of the ceramic membrane before and after reduction). The TiO2-x-EM had the same effective membrane area as the Sn2/NCB-EM (Fig. 5e) but exhibited a much rougher surface (Fig. 5f). XPS spectra demonstrated the slight reduction of the TiO2 ceramic membrane to TiO2-x (x = 0.11) with the appearance of Ti3+ (Figs. 5g, S14b). We finally placed a thin macroporous polyester fabric as a spacer (photo see Fig. S15) between the top and bottom layers to construct the bilayer-EM.
The bilayer-EM was overall hydrophilic (Fig. S16) with the water contact angle of the bottom TiO2-x-EM layer of only 9.0°. The Sn2/NCB coat on the CNT framework improved the hydrophilicity of the top Sn2/NCB-EM layer to a low water contact angle of 30.3°. The coating structure reduced the average pore size and the water permeability of the membrane from 31.3 nm and 148 L m−2 h−1 bar−1 (CNT-EM) to 24.9 nm and 77 L m−2 h−1 bar−1 (Sn2/NCB-EM, Fig. 5h, i), respectively. The smaller pore size and lower water permeability demonstrated a dense interwoven nature of the membrane, which was essential to intensify the utilization of the catalysts and the mass transfer of low concentration NO3− during electrofiltration. We noted that the water flux of the bilayer-EM was mainly determined by the top Sn2/NCB-EM layer due to its much smaller water permeability compared with the bottom TiO2-x-EM layer (Fig. 5i). Additionally, the electrochemical impedance spectroscopy (EIS) amplitudes of the semicircles for both the Sn2/NCB-EM and the TiO2-x-EM were only 23 Ω in 10 mM Na2SO4 solution (Fig. 5j). These results demonstrated the high conductivity of the bilayer-EM for sufficient electron transfer under a relatively low electrolyte concentration.
Electrochemical NO3 −-to-N2 reduction using bilayer electrified membrane
We evaluated the performance of the bilayer-EM for removing low-concentration NO3− (10 mg-N L−1) by electrofiltration of neutral feed solutions containing either 10 mM Na2SO4 or 10 mM NaCl + 5 mM Na2SO4 (Fig. S17). The loading amount of Sn2/NCB per membrane was first optimized based on NO3− removal efficiency in 10 mM Na2SO4 (Fig. S18). Under a current density of 2.5 mA cm−2 and a permeate flow rate of 1.2 mL min−1, NO3− removal reached 95.3% at 50 mg of Sn2/NCB. Further increase of loading to 100 mg only slightly increased the removal to 96.3%. Therefore, we used 50 mg catalyst loading for further experiments. We compared the NO3− reduction performance of a single-layer Sn2/NCB-EM in flow-through mode (Fig. 6a, middle panel) with that of a Sn2/NCB-loaded carbon paper electrode in flow-by mode (Fig. 6a, left panel). Under the same current density of 2.5 mA cm−2, the N2 selectivity was similar between flow-through (90.1%) and flow-by (89.3%). However, the flow-through mode showed a 3-fold enhancement of NO3− conversion rate (546.1 mg-N h−1 m−2 and 95.3% removal efficiency) compared to that of the flow-by mode (186.3 mg-N h−1 m−2 and 31.1% removal efficiency), suggesting the significant role of the flow-through system in stimulating mass transfer and reaction kinetics.
a Effect of electrified operation modes, including flow-by (left, grey shading), single-layer flow-through (middle, blue shading), and bilayer flow-through (right, pink shading) modes, on the distribution of nitrogen species in the permeate (left axis) and N2 selectivity (right axis) using the membrane. Black bars indicate the residual nitrogen concentration (i.e., the sum of NO3−, NO2−, and NH3) in the permeate. The voltages were not iR corrected (membrane electrode surface area = 12.6 cm2, resistance value = 23 Ω). CFD simulating the NO3− concentration distributions under (b1) flow-through and (b2) flow-by modes and (b3) the concentration changes with flow distance. c Generation of free chlorine by the TiO2-x-EM as a function of current density using a feed solution with 5 mM Na2SO4 and 10 mM NaCl under a permeate flow rate of 1.2 mL min−1. d NO3− removal performance of the bilayer-EM when treating different feed solutions containing 10 mg-N L−1 NaNO3 in 10 mM Na2SO4, 1 mM Na2SO4, or simulated surface water (containing 2 mM Cl−, other constituents listed in Table S2). The data in (a) and (b) are presented as mean values ± s.d. (n = 3). e NO3− removal efficiency and loss of Sn of the bilayer-EM as a function of filtration cycles. After operating for 1.5 h, the membrane was rinsed and dried and then used for the next cycle. f Sn FT-EXAFS spectra of the Sn2/NCB-EM before and after long-term operation. Source data are provided as a Source Data file.
We performed computational fluid dynamics (CFD) simulations to examine how flow-through operation enabled more efficient mass transfer and NO3− conversion than flow-by mode (Fig. 6b). Flow-through and flow-by modes were simulated considering water flow over a nanofiber interwoven framework and a flat plate, respectively (Fig. S19). The simulated velocity fields showed that the flow of water was significantly intensified inside the nanofiber framework (Fig. S20). Catalysts coated on the framework could increase the thickness of the nanofibers to further enhance the local velocity, resulting in a 12-fold difference in the average velocity fluctuation compared to the plate. This is crucial for improving the utilization efficiency of the highly exposed catalysts coated on the framework. When considering a constant NO3− reduction rate at the exposed surface, the NO3− concentration decreased rapidly under flow-through mode (Fig. 6b1), whereas the NO3− concentration decrease under flow-by mode was moderate (Fig. 6b2). This resulted in a three-time difference in the amount of NO3− removal between the flow-through and flow-by modes (Fig. 6b3).
We then applied sequential cathode-to-anode electrofiltration using the bilayer-EM to determine whether the existence of Cl− could further enhance the selectivity of N2 generation. In the absence of Cl−, similar NO3− removal efficiency and N2 selectivity were observed under single-layer (Fig. 6a, middle panel) and bilayer modes (Fig. 6a, right panel). However, when 10 mM Cl− was added to the feed, the N2 selectivity increased significantly to 97.7% under the bilayer mode (Fig. 6a, right panel). The free chlorine was efficiently generated from electrofiltration of Cl− through the TiO2-x-EM (e.g., 6.7 mg-Cl2 L−1 at 2.5 mA cm−2, Fig. 6c). The free chlorine or hypochlorous acid (HClO) from chlorine dissolution would oxidize the small amount of NH3 released from the top Sn2/NCB-EM layer into N257,58, which resulted in a low residual total nitrogen concentration (the sum of NO3−, NO2−, and NH3) of only 0.5 mg-N L−1 in the permeate even under the high water treatment capacity of 72 mL h−1. We note that our proposed method is suitable only for removing NH3 with low concentrations (<1.5 mg-N L−1), considering that free chlorine (dosage of 6–15 mg-Cl2 L−1) is generally added in the treated municipal water for disinfection59,60. Applying chlorination to oxidize high-concentration NH3 can significantly increase the potential for harmful chlorinated by-product formation.
The applicability of the bilayer-EM for removing low concentration NO3− under different electrolyte conditions was further evaluated. Under the single-layer mode at neutral pH, when altering the electrolyte from 10 mM Na2SO4 to lower ionic strength (1 mM Na2SO4) and simulated surface water (see Table S2 for the composition), similar NO3− removal performances were achieved (Fig. 6d). Under the bilayer mode when treating the simulated surface water, the N2 selectivity further increased to 98.1% as the surface water contained 2 mM Cl−. The simultaneous achievement of high NO3− removal efficiency and N2 selectivity of Sn2/NCB bilayer-EM with only ~8 s resident time was superior when compared to the previous NO3RR system for NO3− decontamination to N2 (Table S3). When treating water with NO3− concentration at municipal wastewater level of 50 mg-N L−1 (Fig. S21), the bilayer-EM also achieved high removal efficiency (86%), conversion rate (2457.6 mg-N h−1 m−2, a 6-fold enhancement compared to the batch system), and Faradaic efficiency (91%). Notably, the bilayer-EM maintained a stable NO3− removal efficiency of ~96% with negligible dissolution of the Sn (<0.05%) throughout a long-term testing (6 repeated cycles which corresponds to 9 h of operation, Fig. 6e). The Sn FT-EXAFS spectra for the Sn2/NCB-EM before and after the long-term operation showed that the Sn maintained the Sn-N coordination structure without aggregation of Sn (Fig. 6f). The Sn XANES spectra for the Sn2/NCB-EM before and after reaction indicated that the oxidation state of Sn was also maintained (Fig. S22). These results collectively demonstrate the high stability of Sn pair-atoms in the flow-through operation, which was ascribed to the strong binding through the nitrogen coordination61,62. The robustness of the Sn pair-atom loaded membrane for removing low concentration NO3− at realistic water conditions further confirms its superiority over previous nanoparticle-based NO3RR systems.
Discussion
In this work, we developed a bilayer electrified membrane incorporating Sn pair-atoms as the NO3RR catalysts. We successfully demonstrated that the membrane could realize superior removal efficiency and N2 selectivity for converting NO3− with low concentrations in single-pass electrofiltration. Our design principles on the catalysts and the electrofiltration system present three major advancements. First, without using noble metals, the Sn pair-atoms enhanced NO3− adsorption and electron transfer and promoted *N-*N coupling to result in a high N2 selectivity via facilitating *N spillover, which would be difficult to achieve via fully-isolated single-atom sites. Second, coating the pair-atom catalysts with high N2 selectivity on the interwoven framework of the electrified membrane enhanced the exposure and utilization of the catalysts during flow-through operation to facilitate the NO3RR compared to directly functionalizing the catalysts on a plate electrode for conventional flow-by operation. Third, the sequential cathode-to-anode NO3− reduction and residual NH3 oxidation during the single-pass electrofiltration achieved a near 100% N2 selectivity with near zero total dissolved N in the effluent when treating water with an low NO3− concentration (10 mg-N L−1). Further efforts should focus on the mitigation of side reactions when applying electrofiltration to remove contaminants at low concentrations from complex water matrices63,64. The findings of this study not only advance the NO3RR catalyst design but also pave the way for scalable treatment of water containing NO3− with low concentrations using electrified membranes.
Methods
Chemicals and materials
Tin chloride (SnCl4), tin powder, sodium nitrate (KNO3), 1,10-phenanthroline (1,10-phen), carbon nanotubes (CNT, multi-walled), Nafion solution, isopropanol, N,N-dimethylformamide (DMF), polyacrylonitrile (PAN), and sodium sulfate (Na2SO4) were purchased from Sigma-Aldrich. Carbon black was obtained from Cabot Corporation (CB, EMPEROR 2000). All chemicals used in the experiments were reagent grade or higher and used as received without further purification. Experimental solutions were prepared using deionized water (DI, >18.2 MΩ·cm) from the Milli-Q system.
Synthesis of Sn2/NCB and Sn1/NCB
The synthesis step first involved mixing SnCl4 and 1,10-phen at a molar ratio of 1:3 in ethanol. The CB was further added into the mixed solution for impregnation. The nominal loading of Sn was 5% and 1% for Sn2/NCB and Sn1/NCB respectively. The suspensions were dried at 80 °C for 24 h. The catalysts were then calcined at 600 °C for 3 h in a tube furnace (STF1200, Across International) under N2 at a flow rate of 250 sccm.
Preparation of Sn loaded electrodes
5 mg of Sn2/NCB (or NCB, Sn1/NCB, Sn NC-1%, Sn NC-5%, Sn NP-10%, Sn NP-20%, and Sn metal powder) and 20 µL of 5 wt% Nafion solution were dispersed 1 mL isopropanol, followed by sonication for 1 h to prepare a catalyst ink. 200 µL of the ink was drop-casted on carbon fiber paper with an effective working area of 1 × 1 cm2, giving a mass loading of 1 mg cm−2.
Fabrication of Sn2/NCB-EM
The as-received CNTs were pre-treated in a concentrated HCl solution (36.5–38.0%) at 90 °C under refluxing for 12 h, followed by washing with DI water until neutral and drying at 60 °C for 12 h for preparing the pristine CNTs. A certain amount of Sn2/NCB (10 to 100 mg) and 50 mg of pristine CNTs were dispersed in 40 mL DMF containing 0.1 wt% PAN, followed by sonication for 20 min using an ultrasonic probe. We applied layer-by-layer assembly by vacuum filtering 2 mL of the as-prepared suspension onto a ceramic membrane (0.45 μm, Sterlitech). The prepared membrane with 20 layers in total was then rinsed with DI water followed by drying at 90 °C for 12 h. The Sn2/NCB-FEM was obtained by directly peeling the whole carbonaceous layer off from the ceramic membrane after drying.
Fabrication of TiO2-x-EM
The TiO2-x-EM was obtained by calcining a pristine TiO2 membrane (Sterlitech) under pure H2 atmosphere at 1100 °C for 12 h using the tube furnace at a heating rate of 5 °C min−1.
Preparation of bilayer-EM
The bilayer-EM was obtained by stacking the free-standing Sn2/NCB-EM on the TiO2-x-EM with a thin macroporous polyester fabric as the spacer in between to separate the membranes.
Characterizations of catalysts
The Sn loaded onto the catalyst was measured using inductively-coupled plasma mass spectrometry (ICP-MS). The morphology of synthesized Sn2/NCB catalysts was characterized using transmission electron microscopy (TEM, Tecnai Osiris 200 kV, FEI) and aberration-corrected high-angle annular dark-field scanning transmission electron microscopy (AC-HAADF-STEM) at 300 kV. X-ray Diffraction (XRD, Rigaku SmartLab) using Cu Kα radiation (λ = 1.542 Å) was carried out to identify the crystallinity of carbon and the existence of metal phases. Raman spectroscopy (LabRAM HR Evolution, Horiba) was carried out to observe the characteristic D and G bands of the catalysts. The Sn 3d and N 1 s were measured by X-ray photoelectron spectroscopy (XPS) with a Versa Probe II scanning XPS microprobe (PhysicalElectronics) using monochromatic Al Kαradiation (1486.6 eV).
XAFS measurement of Sn2/NCB
To characterize the coordination environment of Sn, X-ray absorption spectra (XAS) at Sn K-edge were recorded at the ISS beamline of the National Synchrotron Light Source II at Brookhaven National Laboratory65, using a Si(111) double-crystal monochromator and a passivated implanted planar silicon (PIPS) fluorescence detector. Near-edge X-ray absorption spectroscopy (XANES) and extended X-ray absorption fine structure (EXAFS) data were collected at room temperature, with energy calibrated using a Sn foil. EXAFS data was fitted by using Demeter 0.9.25 software package. Athena software was used to calibrate energy and normalize the data. The edge energy was determined using the maximum of the first peak in the first derivative of the XANES spectra66. EXAFS spectra were fit using a least-squares fit in R-space of k2 weighted Fourier transform (FT) to determine the coordination number (CN) and bond distances (R) between Pd and Cu using the Artemis software66. The first shell was used to fit the EXAFS spectra. Least-squares fit in R-space of the k2-weighted Fourier transform data from 3.0 to 12.0 Å−1 was used to obtain the EXAFS coordination parameters. The amplitude reduction factor (S02) was determined as 0.78 by fitting a reference spectrum of the Pd foil, and then it was used for fitting all the other EXAFS spectra.
Characterization of membranes
SEM (SU8230, Hitachi) was employed to investigate the surface and cross-section morphologies of the membrane. Membrane pore size distribution was estimated by analyzing the surface SEM images using Nano Measurer software. Water contact angles were measured by the sessile drop method using a contact angle goniometer (OneAttension, Biolin Scientific). Water flux was calculated by dividing the permeate flow rate by the effective membrane area (i.e., 12.6 cm2). Membrane pore volume was determined by the weight difference between a wet and dry membrane. Water residence time was calculated by dividing the pore volume by the permeate flow rate.
Electrochemical measurements
Electrochemical measurements were performed by an electrochemical workstation (CHI 660E, CH Instruments) in a typical three-electrode batch cell containing the working electrode, a mixed metal oxide (MMO) electrode as the counter electrode, and an Ag/AgCl electrode as the reference electrode. All potentials were expressed relative to the RHE scale and were calculated using the following equation: E (vs RHE) = E (vs Ag/AgCl) + 0.197 + 0.0591 × pH. LSV curves were collected at a scan rate of 20 mV s−1 in 50 mM Na2SO4 solution with or without 50 mg-N L−1 NaNO3 addition. EIS was conducted by applying frequencies ranging from 1 to 106 Hz in a 10 mM Na2SO4 solution at open circuit voltage. The electrolyte was prepared before each test.
Batch NO3RR experiments
The NO3− reduction performance of the catalysts was investigated through batch experiments. The batch system consisted of a reactor with the catalyst coated electrode and an MMO mesh electrode with a spacing of 1 cm. Batch experiments were performed for treating 20 mL feed solution with 50 mg-N L−1 NaNO3 and 50 mM Na2SO4 for 2 h at a string rate of 500 rpm. The reason for choosing 50 mg-N L−1 instead of 10 mg-N L−1 was that the low mass transfer limitations in the batch system would result in no visible effect among different catalysts.
Electrofiltration NO3RR experiments
Electrofiltration experiments were performed using a cross-flow membrane filtration system. The membrane and a counter MMO mesh electrode were placed in an electrofiltration cell with 1 cm spacing. Electrolytes containing 10 mg-N L−1 NaNO3 in 10 mM Na2SO4 or in 10 mM NaCl + 5 mM Na2SO4 at neutral pH were used as the feed solution. The feed was circulated at a flow rate of 200 mL min−1 by a peristaltic pump and the permeate flow rate was controlled to 1.2 mL min−1.
Nitrogen speciation and free chlorine measurement
NO3−, NO2−, NH4+, and free chlorine concentrations in both the permeate and the feed were quantified after 1 h of operation. The concentrations of NO3−, NO2−, NH4+, and free chlorine were determined according to the cadmium reduction method, the diazotization method, the Nessler method, and the N,N-diethyl-1,4-phenylenediamine sulfate method respectively, using assay kits (HI93728 for NO3−, HI93707 for NO2−, HI93715 for NH4+, and HI97710 for free chlorine, Hanna Instruments). The standard curves used for the colorimetric quantification of NO3−, NO2− and NH4+ are shown in Fig. S5. Ion chromatography (IC, 930 Compact, Metrohm) was also employed to double-check the concentrations of the nitrogen ions. The N2 selectivity was calculated by the following equation, where C represents the concentration of a certain ion:
The Faradaic efficiency (FE) calculation
The FE for the conversion of NO3− to NO2−, NH4+/NH3, and N2 were calculated according to the following equation:
where n is the number of charge equivalents required to produce 1 mole of product (n = 2 for NO2−, n = 8 for NH4+/NH3, n = 5 for N2). F is the Faraday’s constant (96485 C/mol), \({C}_{{product}}\) is the concentration of product (g/L), V is the volume of the electrolyte (0.04 L), MWproduct is the molecular weight of products (MWNH3 = 17 g mol−1, MWNO2 = 46 g mol−1, MWN2 = 28 g mol−1), i is the operating current density, and t is the electrolysis time.
In situ attenuated total reflectance surface-enhanced infrared absorption spectroscopy
ATR-SEIRAS was performed using a Nicolet iS50 FTIR spectrometer equipped with an MCT detector and a reflectance unit for the electrochemical cell at an incidence angle of 60°. The in situ electrochemical cell consisted of two chambers with three electrodes and the chambers were filled with 10 mL of 50 mg-N L−1 NaNO3 and 50 mM Na2SO4 electrolyte. Platinum sheet (1 cm2) and Ag/AgCl were used as the counter electrode and reference electrode, respectively. For the working electrode, 30 μL of catalyst ink (5 mg mL−1) was dropped onto a silicon crystal and dried in air before testing. The detector was cooled with liquid nitrogen for at least 30 min before testing to maintain a stable signal. NO3RR testing was performed using the chronoamperometry method, and the spectral signal was collected after 1 min of reaction. Scans were conducted from the open circuit potential to −1.0 V (vs. RHE). All spectra were given by absorbance with a spectral resolution of 4 cm−1 for each curve.
Density functional theory calculations
Spin-polarized first-principles calculations were performed by using the Perdew-Burke-Ernzerhof (PBE) exchange-correlation functional as implemented in the Vienna ab initio simulation package (VASP). The projector augmented wave (PAW) method and the corresponding pseudopotentials were employed. A cutoff energy of 500 eV was set for the plane wave basis set. The adsorption configurations of NO3‒ on Sn1/NCB and Sn2/NCB and the structures of reaction intermediates were optimized by controlling the convergence thresholds for the energy and force to be 10−5 eV and 0.02 eV/Å, respectively. During the relaxation, all atomic positions were allowed to relax. The Grimme’s DFT-D3 correction method was included to describe the weak dispersion interactions during surface adsorption. A 2 × 2 × 1 Monkhorst-Pack k-point sampling was set for all models. The corresponding adsorption Gibbs free energies were then calculated as
where ΔZPE and ΔS are the changes in zero point energy and in entropy, respectively, between the adsorbed state and free state, which can be obtained from the vibrational frequencies calculations (with adsorbates relaxed and substrates fixed) and standard thermodynamic data. T is the temperature. The adsorption energy of species on the surface was calculated as:
where Ex/surface, Ex, and Esurface are the energies of the slab with adsorbed species, the adsorbed species calculated in a cubic cell, and the energies of the slab, respectively.
Computational fluid dynamics simulation
CFD was performed for flow field simulation inside the membrane using COMSOL Multiphysics software. The geometry was constructed with a cube fluid domain with a side length of 400 nm. The cube was swept with random rotation and movement multiple times to obtain multiple gaps and the geometric structures outside the fluid domain were removed. Three models were built inside the fluid domain, including an interwoven framework with thick fibers, an interwoven framework with thin fibers, and a flat plate control. The simulations were solved based on the Navier-Stokes equations using laminar flow physics. On the basis of the convection velocity calculation, the conservation calculation of convective mass transfer was performed. The calculations meet the dilute species transfer interface under different pore conditions and the mass transfer was simulated through the diffusion equation:
where D is diffusivity (m2/s), c is concentration (mol/m3), u is fluid velocity vector (m/s), and R is the rate expression of matter (mol/m3·s). Considering that the reaction happens mainly on the surface rather than in the body of the model, the R term here is zero and the overall reaction satisfies Fick’s diffusion law. The inlet flow rate at the entrance of the mass transfer region was set to 2.4 μm/s and the surface reaction rate was set to 2 × 10−4 m/s as the field boundaries.
Data availability
All data needed to evaluate the conclusions in the paper are present in the paper and/or the Supplementary Information. Source data are provided with this paper.
References
Spalding, R. F. & Exner, M. E. Occurrence of nitrate in groundwater—a review. J. Environ. Qual. 22, 392–402 (1993).
Xu, H., Paerl, H. W., Qin, B., Zhu, G. & Gaoa, G. Nitrogen and phosphorus inputs control phytoplankton growth in eutrophic Lake Taihu, China. Limnol. Oceanogr. 55, 420–432 (2010).
Kim, K. et al. Nitrogen stimulates Microcystis-dominated blooms more than phosphorus in river conditions that favor non-nitrogen-fixing genera. Environ. Sci. Technol. 54, 7185–7193 (2020).
Duca, M. & Koper, M. T. Powering denitrification: the perspectives of electrocatalytic nitrate reduction. Energy Environ. Sci. 5, 9726–9742 (2012).
Xu, H., Ma, Y., Chen, J., Zhang, W.-X. & Yang, J. Electrocatalytic reduction of nitrate–a step towards a sustainable nitrogen cycle. Chem. Soc. Rev. 51, 2710–2758 (2022).
Li, J. et al. Atomically dispersed Fe atoms anchored on S and N–codoped carbon for efficient electrochemical denitrification. Proc. Natl. Acad. Sci. 118, e2105628118 (2021).
Li, P. et al. A multifunctional copper single-atom electrocatalyst aerogel for smart sensing and producing ammonia from nitrate. Proc. Natl. Acad. Sci. 120, e2305489120 (2023).
Wu, Z.-Y. et al. Electrochemical ammonia synthesis via nitrate reduction on Fe single atom catalyst. Nature Comm 12, 2870 (2021).
Zhu, T. et al. Single-atom Cu catalysts for enhanced electrocatalytic nitrate reduction with significant alleviation of nitrite production. Small 16, 2004526 (2020).
Wang, X. et al. Free-standing membrane incorporating single-atom catalysts for ultrafast electroreduction of low-concentration nitrate. Proc. Natl. Acad. Sci. 120, e2217703120 (2023).
Schuytema, G. S. & Nebeker, A. V. Comparative toxicity of ammonium and nitrate compounds to Pacific treefrog and African clawed frog tadpoles. Environ. Toxicol. Chem. 18, 2251–2257 (1999).
Gootzen, J. et al. The electrocatalytic reduction of NO3− on Pt, Pd and Pt+ Pd electrodes activated with Ge. J. Electroanal. Chem. 434, 171–183 (1997).
Kato, M., Okui, M., Taguchi, S. & Yagi, I. Electrocatalytic nitrate reduction on well-defined surfaces of tin-modified platinum, palladium and platinum-palladium single crystalline electrodes in acidic and neutral media. J. Electroanal. Chem. 800, 46–53 (2017).
Wang, Y., Wang, C., Li, M., Yu, Y. & Zhang, B. Nitrate electroreduction: mechanism insight, in situ characterization, performance evaluation, and challenges. Chem. Soc. Rev. 50, 6720–6733 (2021).
Yang, X.-F. et al. Single-atom catalysts: a new frontier in heterogeneous catalysis. Acc. Chem. Res. 46, 1740–1748 (2013).
Li, J. et al. Highly active and stable metal single-atom catalysts achieved by strong electronic metal–support interactions. J. Am. Chem. Soc. 141, 14515–14519 (2019).
Zhuo, H.-Y. et al. Theoretical understandings of graphene-based metal single-atom catalysts: stability and catalytic performance. Chem. Rev. 120, 12315–12341 (2020).
Liu, J. Catalysis by supported single metal atoms. ACS Catal 7, 34–59 (2017).
Murphy, E. et al. Elucidating electrochemical nitrate and nitrite reduction over atomically-dispersed transition metal sites. Nature Comm 14, 4554 (2023).
Wang, S. et al. High-throughput identification of highly active and selective single-atom catalysts for electrochemical ammonia synthesis through nitrate reduction. Nano Energy 100, 107517 (2022).
Xue, Y. et al. Electrocatalytic hydrogenation boosts reduction of nitrate to ammonia over single-atom Cu with Cu(I)-N3C1 sites. Environ. Sci. Technol. 56, 14797–14807 (2022).
Yin, H., Peng, Y. & Li, J. Electrocatalytic reduction of nitrate to ammonia via a Au/Cu single atom alloy catalyst. Environ. Sci. Technol. 57, 3134–3144 (2023).
Cheng, X. F. et al. Coordination symmetry breaking of single‐atom catalysts for robust and efficient nitrate electroreduction to ammonia. Adv. Mater. 34, 2205767 (2022).
Li, J. et al. Boosted ammonium production by single cobalt atom catalysts with high Faradic efficiencies. Proc. Natl. Acad. Sci. 119, e2123450119 (2022).
Liu, H. et al. Efficient electrochemical nitrate reduction to ammonia with copper‐supported rhodium cluster and single‐atom catalysts. Angew. Chem. Inter. Edit. 61, e202202556 (2022).
Li, R. & Wang, D. Superiority of dual‐atom catalysts in electrocatalysis: one step further than single‐atom catalysts. Adv. Ener. Mater. 12, 2103564 (2022).
Fajardo, A. S., Westerhoff, P., Sánchez-Sánchez, C. M. & Garcia-Segura, S. Earth-abundant elements a sustainable solution for electrocatalytic reduction of nitrate. Appl. Catal. B Environ. 281, 119465 (2021).
Garcia-Segura, S., Lanzarini-Lopes, M., Hristovski, K. & Westerhoff, P. Electrocatalytic reduction of nitrate: Fundamentals to full-scale water treatment applications. Appl. Catal. B Environ. 236, 546–568 (2018).
Ma, W. et al. Catalytic membrane with copper single-atom catalysts for effective hydrogen peroxide activation and pollutant destruction. Environ. Sci. Technol. 56, 8733–8745 (2022).
Meng, C. et al. Angstrom-confined catalytic water purification within Co-TiOx laminar membrane nanochannels. Nature Comm 13, 4010 (2022).
Wu, X. et al. Single-atom cobalt incorporated in a 2D graphene oxide membrane for catalytic pollutant degradation. Environ. Sci. Technol. 56, 1341–1351 (2021).
Zuo, K. et al. Electrified water treatment: fundamentals and roles of electrode materials. Nature Rev. Mater. 8, 472–490 (2023).
Sun, M. et al. Electrified membranes for water treatment applications. ACS EST Engg 1, 725–752 (2021).
Zhao, Y. et al. Emerging challenges and opportunities for electrified membranes to enhance water treatment. Environ. Sci. Technol. 56, 3832–3835 (2022).
Gayen, P. et al. Electrocatalytic reduction of nitrate using magnéli phase TiO2 reactive electrochemical membranes doped with Pd-based catalysts. Environ. Sci. Technol. 52, 9370–9379 (2018).
Li, Y., Ma, J., Waite, T. D., Hoffmann, M. R. & Wang, Z. Development of a mechanically flexible 2D-MXene membrane cathode for selective electrochemical reduction of nitrate to N2: mechanisms and implications. Environ. Sci. Technol. 55, 10695–10703 (2021).
Ma, J. et al. Electrochemical reduction of nitrate in a catalytic carbon membrane nano-reactor. Water Res. 208, 117862 (2022).
Yang, H. et al. A universal ligand mediated method for large scale synthesis of transition metal single atom catalysts. Nat. Commun. 10, 4585 (2019).
Accorsi, G., Listorti, A., Yoosaf, K. & Armaroli, N. 1, 10-Phenanthrolines: versatile building blocks for luminescent molecules, materials and metal complexes. Chem. Soc. Rev. 38, 1690–1700 (2009).
Fox, G. A., Bhattacharya, S. & Pierpont, C. G. Structural and electrochemical properties of binuclear complexes containing 1, 10-phenanthroline-5, 6-diolate as a bridging ligand. Inorg. Chem. 30, 2895–2899 (1991).
Wang, Y. Y., Wang, X., Shi, Q. Z. & Gao, Y. C. A novel binuclear copper (II) complex with fumarate and 1, 10-phenanthroline. Transit. Met. Chem. 27, 481–484 (2002).
Chai, Y. et al. Dual-atom catalyst with N-colligated Zn1Co1 species as dominant active sites for propane dehydrogenation. J. Am. Chem. Soc. 146, 263–273 (2023).
Shan, J. et al. Metal-metal interactions in correlated single-atom catalysts. Sci. Adv. 8, eabo0762 (2022).
Zhang, Y.-X. et al. General synthesis of a diatomic catalyst library via a macrocyclic precursor-mediated approach. J. Am. Chem. Soc. 145, 4819–4827 (2023).
Zhao, X. et al. Highly efficient electrochemical CO2 reduction on a precise homonuclear diatomic Fe–Fe catalyst. ACS Catal. 12, 11412–11420 (2022).
Reddy, A. L. M. et al. Synthesis of nitrogen-doped graphene films for lithium battery application. ACS Nano 4, 6337–6342 (2010).
Zhu, H. et al. Ni–M–O (M= Sn, Ti, W) catalysts prepared by a dry mixing method for oxidative dehydrogenation of ethane. ACS Catal. 6, 2852–2866 (2016).
Guo, J. et al. Control over electrochemical CO2 reduction selectivity by coordination engineering of tin single‐atom catalysts. Adv. Sci. 8, 2102884 (2021).
Maurer, F. et al. Tracking the formation, fate and consequence for catalytic activity of Pt single sites on CeO2. Nat. Catal. 3, 824–833 (2020).
Guo, Y., Wang, M., Zhu, Q., Xiao, D. & Ma, D. Ensemble effect for single-atom, small cluster and nanoparticle catalysts. Nat. Catal. 5, 766–776 (2022).
Cheng, X. et al. Unveiling structural evolution of Fe single atom catalyst in nitrate reduction for enhanced electrocatalytic ammonia synthesis. Nano Res. 17, 6826–6832 (2024).
Liu, Y. et al. Shear‐strained Pd single‐atom electrocatalysts for nitrate reduction to ammonia. Angew. Chem. Inter. Edit. 63, e202411396 (2024).
Zhang, S. et al. Oxygen-coordinated single Mn sites for efficient electrocatalytic nitrate reduction to ammonia. Nano-Micro Lett. 16, 9 (2024).
Clarke, O. J., Rowley, A., Fox, R. V., Atifi, A. & Burgess, I. J. Diamonds in the rough: direct surface enhanced infrared spectroscopic evidence of nitrogen reduction on boron-doped diamond supported metal catalysts. Anal. Chem. 95, 10476–10480 (2023).
Li, T. et al. A spectroscopic study on nitrogen electrooxidation to nitrate. Angew. Chem. Inter. Edit. 135, e202217411 (2023).
Li, X. et al. Unique two-electron transfer pathway of Bismuth nanocrystal for enhanced N2 electroreduction revealed by in situ infrared spectroscopy. Appl. Catal. B Environ. 342, 123365 (2024).
Liu, Z. et al. Electrochemically mediated nitrate reduction on nanoconfined zerovalent iron: properties and mechanism. Water Res. 173, 115596 (2020).
Zhang, C., He, D., Ma, J. & Waite, T. D. Active chlorine mediated ammonia oxidation revisited: Reaction mechanism, kinetic modelling and implications. Water Res. 145, 220–230 (2018).
Huo, Z.-Y. et al. Synergistic nanowire-enhanced electroporation and electrochlorination for highly efficient water disinfection. Environ. Sci. Technol. 56, 10925–10934 (2022).
Li, Q., Liu, G.-h, Qi, L., Wang, H. & Xian, G. Chlorine-mediated electrochemical advanced oxidation process for ammonia removal: Mechanisms, characteristics and expectation. Sci. Total Environ. 896, 165169 (2023).
Chen, C. et al. Local reaction environment in electrocatalysis. Chem. Soc. Rev. 53, 2022–2055 (2024).
Zhang, Y. et al. The effect of coordination environment on the activity and selectivity of single-atom catalysts. Coord. Chem. Rev. 461, 214493 (2022).
Alkhadra, M. A. et al. Electrochemical methods for water purification, ion separations, and energy conversion. Chem. Rev. 122, 13547–13635 (2022).
Gao, X. et al. Boosting urea electrooxidation on oxyanion-engineered nickel sites via inhibited water oxidation. Nat. Commun. 14, 5842 (2023).
Leshchev, D. et al. The Inner Shell Spectroscopy beamline at NSLS-II: a facility for in situ and operando X-ray absorption spectroscopy for materials research. J. Synchrotron Radiat. 29, 1095–1106 (2022).
Ravel, B. & Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Radiat. 12, 537–541 (2005).
Acknowledgements
This study was supported by National Natural Science Foundation of China (nos. 52470044 and 52400098), NSF Division of Chemical, Bioengineering, Environmental, and Transport Systems (CBET) Grant #1955793, the NSF Nanosystems Engineering Research Center for Nanotechnology-Enabled Water Treatment (ERC-1449500), Guangdong Basic and Applied Basic Research Foundation (2024A1515012461), Shenzhen Key Laboratory of Advanced Technology for Marine Ecology (ZDSYS20230626091459009). We acknowledge Dr. Qingxiao Wang at the Imaging and Characterization Core Lab at King Abdullah University of Science and Technology for his assistance in HAADF-STEM measurements. This research used ISS beamline (8-ID) of the National Synchrotron Light Source II, a U.S. Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by Brookhaven National Laboratory under Contract No. DE-SC0012704.
Author information
Authors and Affiliations
Contributions
X. Wu, X. Wang, and J.-H. K. designed research; X. Wu and X. Wang performed research; Y. Wu, H. X., Z. L., R. H., and K. R. analyzed data; X. Wu, X. Wang, Z. W., and J.-H. K wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Dong-Hee Lim, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wu, X., Wang, X., Wu, Y. et al. Bilayer electrified-membrane with pair-atom tin catalysts for near-complete conversion of low concentration nitrate to dinitrogen. Nat Commun 16, 1122 (2025). https://doi.org/10.1038/s41467-025-56102-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-56102-7
This article is cited by
-
Optimization and Application of Biological-Ion Exchange-Electrocatalysis-Biological Process for Treatment of Nitrate in Actual Wastewater
Water, Air, & Soil Pollution (2026)
-
Pursuing spatiotemporal coordination in electrocatalysis
Nature Reviews Chemistry (2025)