Abstract
Soft porous crystals, recognized as the third generation of smart porous materials, can undergo structural deformations in response to external stimuli, such as temperature, pressure, and guest molecules. Currently, the dynamic phase transformations of soft porous crystals are predominantly determined through quantitative modeling based on gas adsorption and powder X-ray diffraction. Herein, we investigate the single-crystal-to-single-crystal structural transformation of covalent organic soft porous crystal modeled on COF-300 and identified nine distinct conformational isomers induced by different guest molecules at room and high temperatures. Notably, COF-300 can maintain its single-crystal structure even at 280 °C and efficiently absorbs polycyclic aromatic hydrocarbons in their molten state. The kinetics of structural transformations among conformational isomers are investigated by combining PXRD and theoretical calculations. The structural transformation from a high-energy state to a low-energy state is a rapid, energetically favorable process, while the reverse transformation is a slow process driven by concentration gradients.
Similar content being viewed by others
Introduction
Soft porous crystals represent a class of adaptable and intelligent crystalline porous materials capable of lattice contraction, expansion, and deformation in response to external stimuli such as temperature, light, and guest molecules, which offer distinctive advantages in areas such as adsorption and separation, molecular recognition, sensing and catalysis1,2,3. Soft porous crystals have been one of the frontiers of research in porous materials since Kitagawa and co-workers introduced the concept of soft porous crystals as a third-generation porous material in 19981. Structural transformation of soft porous crystals means that the dynamic framework can remain long-range ordered between two or more crystalline phases without bond breaking or recombination, and the individual crystalline phases have identical topology but differ in cell parameters, symmetries, and porosities. The mechanisms of the structural transformation include the deformation of network nodes (breathing: successive open-close-open transitions with increasing guest loading), rotation of organic linkers (gate opening: guest-induced pore expansion), and displacement of interpenetrating networks3. So far, substantial research on soft porous crystals has focused on metal-organic frameworks (MOFs)4,5,6,7,8,9,10,11,12,13,14, whose flexible coordination bonds and diverse organic ligands provide feasibility for soft porous crystals, such as MIL-537 and MIL-8815,16 (MIL, Materials of Institut Lavoisier). Structural characterization of the conformational isomers during the structural transformation is a pervasive challenge, and quantitative modeling and speculation of these structural changes can usually only be achieved by a combination of gas adsorption and powder X-ray diffraction (PXRD) methods. This vague characterization typically yields two distinct states, close phase, and open phase, for most dynamic phase transitions in soft porous crystals. Single-crystal X-ray crystallography of conformational isomers continues to be one of the most precise characterization methods for the structural transformation of soft porous crystals.
Covalent organic frameworks (COFs) are emerging crystalline porous polymers with two- or three-dimensional structures assembled from geometrically organic building blocks17. Compared with MOFs, COFs exhibit greater covalent bonding energies, contributing to enhanced structural stabilities towards external stimuli or guest molecule inclusion and exclusion. As a result, COFs are deemed more appropriate for applications as soft porous crystals18,19,20,21,22,23,24,25,26,27,28,29,30,31. Since it is challenging to distinguish whether the guest molecules are adsorbed within the pores or confined between the interlayer lattice of 2D COFs, the dynamic 3D COFs are more suitable for investigating the characteristics of soft porous crystals. Firstly, most 3D COFs are constructed from tetratopic nodes (tetraphenylmethane, tetraphenylsilane, adamantane) and linear or planar linkers with dia or Pts topologies. The dia frameworks having large pores are susceptible to thermodynamic stabilization through interpenetration, and ‘breathing’ soft porous crystals are more likely to be achieved by the displacement of the interwoven subnetworks. Secondly, the carbon atoms, serving as tetrahedral nodes, often undergo geometric distortions, resulting in framework deformations that yield ‘breathing’ soft porous crystals. Finally, the rotation of C–C bonds within COFs can be induced by guest molecules, prompting soft porous crystals to exhibit ‘gate opening’ behaviors in response to external stimuli. Compared to conventional quantitative modeling methods21, a feasible strategy for studying structural transformations in soft porous crystals is to explore thermodynamically stable isomers through single-crystal-to-single-crystal transformations and to determine their well-defined structures and host-guest interactions at the atomic level32.
In this study, we have systematically investigated the single-crystal-to-single-crystal structural transformations of covalent organic soft porous crystals modeled on COF-300 induced by guest molecules at room and high temperatures33. A total of nine discontinuous conformational isomers of COF-300 have been identified through single-crystal X-ray diffraction thanks to the high-quality single crystals32. Notably, the high-temperature single-crystal-to-single-crystal structural transformations of COF-300 triggered by the molten guest molecules were explored for the first time. Four representative polycyclic aromatic hydrocarbons (PAHs) as probes were successfully encapsulated within the channels of COF-300 at their melting temperature. It is particularly noteworthy that COF-300 can preserve a long-range ordered single-crystal structure even at 280 °C, enabling precise determination of the location of the guest molecules and the host-guest interactions through single-crystal X-ray diffraction. In addition, the energetic landscape of each conformational isomer is calculated by density functional theory (DFT), revealing that COF-300 (the initially synthesized) is the most stable configuration with the lowest energy20. Furthermore, we explored the kinetics of the structural transformations among the conformational isomers through a combination of PXRD and theoretical calculations, indicating that the transformations from a high-energy isomer to a low-energy one are an energetically favorable and rapid process, whereas the inverse transformation is a slow process driven by concentration gradients.
Results
Structural transformation at room temperature
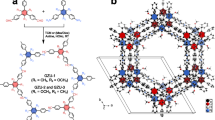
The structural transformation and dynamic nature of COF-300 were accurately elucidated benefiting from high-quality single crystals and single-crystal X-ray crystallography. A total of twenty-one solvents with different sizes and polarities were selected as probes to be exchanged into the channels of COF-300 and five distinct conformational isomers were uncovered (Fig. 1). The nascent COF-300 was synthesized according to the literature and extended the crystallization time (Fig. 2a)32, featuring a sevenfold-interpenetrated dia-c7 topology34,35 (space group I41/a, a = b = 27.015(3) Å, c = 7.3832(14) Å, V = 5388.2(16) Å3, Fig. 2b). In the structure of COF-300, the dimension of diamondoid unit is 26.19 Å × 26.19 Å × 53.13 Å3, in which the angles between the adjacent linkers of the tetratopic node are 88.98° and 120.59° (Fig. 2c). Seven symmetry-related diamondoid units assembled by interpenetration into a stable framework possessing one-dimensional channels along the crystallographic c-axis with dimensions of 13.1 × 13.1 Å2 (Fig. 2d). The thiophene guest molecules within the channels are arranged into four columns through host-guest van der Waals interactions. The total solvent-accessible void is calculated to be 2714.1 Å3, which is 52.0% of the unit cell volume.
The schematic representation of the structural transformation of COF-300 at room temperature.
a The optical microscopic image of single-crystal COF-300 with size ~ 200 μm. b The diamondoid unit and sevenfold-interpenetrated structure of COF-300. c The dimensions of diamondoid units and the angle between neighboring linkers. The channel sizes (d) and the simulated PXRD patterns (e) of COF-300-c, COF-300-ho, COF-300, COF-300-r and COF-300-o.
Notably, when water molecules serve as guest species, the framework of COF-300 undergoes a substantial contraction, evolving into a close phase attributed to the small size of water molecules and the strong hydrogen bonding interactions between the host and guest (COF-300-c, space group I41/a, a = b = 19.9605(11) Å, c = 8.8631(7) Å, V = 3531.2(5) Å3)32. Compared to COF-300, the diamondoid unit of COF-300-c has undergone significant tensile deformation, with dimensions of 19.96 Å × 19.96 Å × 62.04 Å3 (Fig. 2c). This deformation is due to the fact that the angle of two neighboring linkers in the tetrahedral node is reduced from the original 88.98° to 65.52°, while the other angle is increased from 120.59° to 135.00°, which makes the diamondoid cells taller and thinner. On the other hand, the rotation of phenyl rings in diimine linkers also influences the channel size. In COF-300, the phenyl ring of the diimine linker is almost parallel to the channel direction (dihedral angle: 15.1° Fig. 1), whereas in COF-300-c, the phenyl ring is nearly perpendicular to the channel direction (dihedral angle: 81.8° Fig. 1). The deformation of the skeleton and the rotation of phenyl ring reduce the channel size along the crystallographic c-axis from 13.1 Å to 5.8 Å, with a corresponding reduction in the solvent-accessible volume to 824.9 Å3, which is 23.4% of the unit cell volume.
In contrast, the inclusion of mesitylene within the channels induces a significant distortion of the skeleton, transforming to a half-open phase with a change of the space group from I41/a to C2/c (COF-300-ho, a = 34.340(2) Å, b = 7.9814(5) Å, c = 23.0868(15) Å, β = 130.440(3)°, V = 4815.9(5) Å3). Apart from the deformation of the diamondoid units, the most important structural feature of COF-300-ho is that half of the phenyl rings of the diimine linker are parallel to the channel direction (dihedral angle: 11.5° Fig. 1), while the other half are perpendicular to the channel direction (dihedral angle: 77.9° Fig. 1), thus forming a half-open phase. Another structural feature of COF-300-ho is that due to the increased size of the guest molecules, only two columns of mesitylene guest molecules can be accommodated in the channel (Fig. 1). The channel dimension is 8.7 × 11.5 Å2 with the total solvent-accessible void is calculated to be 2134.3 Å3, which is 44.3% of the unit cell volume.
When guest molecules turned to 1,2,4-trimethylbenzene or 1-methylnaphthalene, the geometry of the COF-300 channels was deformed from square to rectangular, accompanied by a shift in the space group from I41/a to I4. (COF-300-r, a = b = 26.900(3) Å, c = 7.4006(13) Å, V = 5355.0(15) Å3). The deformation of channels is mainly due to the different sizes and arrangement of the guest molecules, and each rectangular channel can only accommodate two columns of guest molecules (Supplementary Figs. S6, S8). The introduction of cyclohexane as a guest molecule produced a noticeable expansion of the COF-300 channels, with the space group changing to P4n2 (COF-300-o, a = b = 28.020(6) Å, c = 7.035(3) Å, V = 5523(3) Å3). The COF-300-o has the largest channel size (14.1 × 14.1 Å2) and the largest solvent-accessible volume (2953.6 Å3, 53.5% of the unit cell volume). It is essential to note that for the majority of solvents examined, including dichloromethane, trichloromethane, ethanol, acetonitrile, acetone, N, N-dimethylformamide, dimethyl sulfoxide, n-hexane, tetrahydrofuran, thiophene, toluene, chlorobenzene, o-xylene, m-xylene, p-xylene, and benzyl alcohol, COF-300 retains its initially synthesized conformation without substantial deformation of the skeleton (Supplementary Table S1). All observed structural transformation is induced by disparities in the size and arrangement of the guest molecules within the channels. Despite variations in cell parameters and space groups, the whole framework remains unchanged in terms of the same connectivity and degree of interpenetration.
A geometrical analysis of the framework reveals that the distortion of tetrahedral nodes significantly influences structural deformations. For instance, as shown in Fig. 2c, the angles between adjacent connectors of tetrahedral nodes in the diamondoid unit of COF-300-c are measured at 65.52° and 135.00°, respectively. This configuration results in an elongated diamondoid cell measuring 19.96 Å × 19.96 Å × 62.04 Å, which constrains the channel size to 5.8 Å along the crystallographic c-axis. As the angle increases from 65.52 (COF-300-c) to 97.32° (COF-300-o), the height of the diamondoid unit gradually reduced from 62.04 Å to 49.62 Å, while the width expands from 19.96 to 28.20 Å. Concurrently, the unit cell volume increases from 3531.2(5) Å3 (COF-300-c) to 5523(3) Å3 (COF-300-o), accompanied by an expansion in channel dimensions from 5.8 Å to 14.1 Å, resulting in a 3.5-fold increase in solvent-accessible volume from 824.9 Å3 to 2953.6 Å3 (calculated by the PLATON program)36. This observation points to a ‘swelling’ phenomenon recognized within the structural transformation of soft porous crystals16. In addition, the channel’s openness is also influenced by the rotational behavior of the diimine linkers. The dihedral angle between benzene rings and channel wall progressively decreases from 81.8° (COF-300-c) to 15.1° (COF-300), ultimately reaching 12.7° (COF-300-o), which initiates a ‘gate opening’ mechanism within soft porous crystals, resulting in the channels closing and opening (Fig. 1). Finally, the length of diimine linkers connecting the tetrahedral nodes further contributes to the structural deformation; as the length of diimine linker increases from 18.44 Å (COF-300-c) to 18.78 Å (COF-300-o), the resultant unit-cell volumes and channel dimensions for COF-300 progressively increased. The phase of the COF-300 conformational isomer can also be clearly distinguished by the characteristic diffraction peaks of PXRD patterns. The distinct diffraction peak at 8.86° of COF-300-c corresponds to the (200) plane reflection, whereas the characteristic peaks of (200) plane reflections for COF-300, COF-300-r, and COF-300-o are at 6.54°, 6.57° and 6.30°, respectively. For COF-300-ho, the two characteristic diffraction peaks at 6.76° and 7.66° represent the (200) and (202) plane reflections, respectively (Fig. 2e).
Structural transformation at high temperature
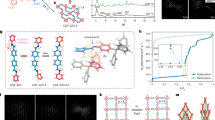
The structural integrity and stability of soft porous crystals are important for their potential applications5. In our work, four representative PAHs with different melting points, naphthalene (NA) 82 °C, anthracene (AN) 216 °C, pyrene (PY) 148 °C, and perylene (PE) 280 °C, were selected as guest molecules to be exchanged into the channels of COF-300 in their molten state. Remarkably, COF-300 still maintained a highly ordered single-crystal structure at 280 °C even after absorbing molten PAHs and undergoing structural transformations. The structures of four high-temperature conformational isomers were precisely determined through single-crystal X-ray crystallography, elucidating the positions of the PAHs guest molecules and the interactions between the host and guest (Fig. 3). The structure of COF-300-NA is similar to COF-300, featuring comparable unit cell parameters and the same space group I41/a, where naphthalene molecules are arranged into four columns through van der Waals interactions between N atoms of the host framework and H atoms of naphthalene molecules. When anthracene serves as guest molecules, their arrangement was optimized into two columns to minimize intermolecular repulsions, consequently transforming the channel geometry from square to rectangular so as to better accommodate anthracene molecules. This alteration results in the space group of COF-300-AN shifting to P4. For the inclusion of larger pyrene and perylene, the channel of COF-300 can accommodate only a single column of guest molecules, leading to slight deformation of the channel to adapt to the guest molecules. Although both COF-300-PY and COF-300-PE crystallize in the identical I4 space group, the channel of COF-300-PY exhibits a configuration more akin to a square shape, contrasting with the elongated rectangular shape channels of COF-300-PE. Furthermore, in the high-temperature conformational isomers, the distortion of the tetrahedral nodes is crucial for structural deformation, and the angles between adjacent connectors are 91.91°, 91.48°, 86.19°, and 89.75°, which are comparable to those of COF-300 and COF-300-r, respectively (Supplementary Fig. S69). To accommodate the large guest molecules, all four conformational isomers demonstrated ‘gate-opening’ states with dihedral angles of 19.3°, 29.3°, 15.0°, and 12.8° between the diimine linker and channel wall, and the solvent accessible volumes of them are 52.6% (naphthalene), 51.2% (anthracene), 49.7% (pyrene), and 51.2% (perylene), respectively. The photoluminescence properties of COF-300, PAHs, and COF-300-PAHs have been investigated. As shown in Supplementary Fig. S70, COF-300-NA shows obviously red-shifted and broader emission spectra compared with solid naphthalene, whereas COF-300-AN and COF-300-PY display similar emission spectra compared to the solid anthracene and pyrene, respectively. This difference can be attributed to the generation of more obviously intermolecular charge transfer excited state (CT*) from COF-300 to PAHs, resulting in a reduced excited state energy level in COF-300-naphthalene, which further induces the observed red-shifted emission. The excitation spectra of COF-300-PAHs showed similar excitation peaks to PAHs (Supplementary Fig. S71). The lifetimes of COF-300-PAHs showed fluorescence emissive features with a decay lifetime of nanoseconds compared with PAHs (Supplementary Fig. S72), indicating that the encapsulation process induces luminescence quenching, thereby reducing the lifetime. These results indicate that the emission properties of COF-300 could be effectively turned by the PAHs-encapsulated process, which may promote the functionalization and application of host-guest COF crystalline material.
The single-crystal-to-single-crystal structural transformation of COF-300 at melting temperature of polycyclic aromatic hydrocarbon molecules.
Kinetics of structural transformation
The kinetics of structural transformation between the conformational isomers of COF-300 were investigated by a combination of PXRD and single-crystal X-ray crystallography. Three representative conformational isomers, COF-300, COF-300-c, and COF-300-ho were selected as the initial states for the dynamic structural transformation. Notably, the structural transformations among these conformational isomers demonstrate two distinct processes. For instance, the transformation from COF-300-ho to COF-300 is a rapid process involving guest molecule exchange accompanied by skeleton deformation. The PXRD pattern, recorded within ten minutes of solvent addition confirmed this observation. As shown in Fig. 4a, the characteristic diffraction peaks of COF-300-ho disappeared, replaced by a well-defined diffraction pattern corresponding to COF-300. A similar process was observed during the structural transformation from COF-300-c to COF-300 (Fig. 4b). In contrast, the reverse structural transformation from COF-300 to COF-300-ho, or from COF-300-c to COF-300-ho is a slow process that can be accomplished within 24 h, as evidenced by PXRD patterns (Fig. 4c, d). Furthermore, single-crystal X-ray diffraction data confirmed the feasibility of structural transformations from COF-300-ho to COF-300-PAHs at high temperatures, as well as from COF-300-PAHs to COF-300 at room temperature. Based on the above information, a schematic representation showing the structural transformations among these conformational isomers of COF-300 is given in Fig. 4e.
a The PXRD patterns of structural transformation from COF-300-ho to COF-300. b From COF-300-c to COF-300. c From COF-300 to COF-300-ho. d From COF-300-c to COF-300-ho. e Schematic representation of the structural transformations.
To comprehend the fundamental mechanisms underlying the structural transformation of conformational isomers, density functional theory (DFT) calculations are employed to evaluate the energetic landscape of each conformational isomer. The deformation energy (Edef) was calculated to evaluate the energy of the empty conformational isomer considering the contribution of the distortion of tetrahedral nodes, the rotational of the diimine linkers, and the connector lengths. The interaction energy (Eint) represents the energy of guest-host interactions, and the total anergy (Etot) is the sum of Edef and Eint3,19. To correct the basis set superposition error (BSSE), the counterpoise (CP) procedure has been used for calculating the interaction energies qualitatively. The Eint is calculated by the following formula: Eint(AB) = E(AB)AB – [E(A)AB + E(B)AB]. The free energy landscape analysis indicates that COF-300, the initially synthesized phase, exhibits the lowest energy and represents the thermodynamically stable state20. In the case of COF-300-c (guest: H2O), the Edef value of 158.6 Kcal mol–1 represents the energy difference between COF-300-c and COF-300. The Eint value of the guest-host is – 10.2 Kcal mol–1 (two water molecules have hydrogen bond interactions with the model, Supplementary Fig. S72c) and the calculated Etot value is 148.4 Kcal mol–1. Based on the same modeling and calculation methods, the Etot of each conformational isomer can be obtained (Supplementary Table S28). The stability order of the COF-300 conformational isomers is COF-300, COF-300-r, COF-300-ho, COF-300-c, and COF-300-o. These results contribute to elucidating the intrinsic mechanisms underlying the structural transformations of conformational isomers from an energetic perspective.
Discussion
It has been acknowledged that elucidating the precise atomic-level structure of conformational isomers and the kinetics of structural transformations of soft porous crystals remains a persistent challenge. In our work, the single-crystal-to-single-crystal structural transformations of covalent organic soft porous crystal modeled on COF-300 induced by guest molecules are systematically investigated at room and high temperatures. Twenty-one solvents at room temperature and four PAHs at high temperature were selected as guest molecular probes to investigate the structural transformation and skeleton deformation of COF-300. Nine discontinuous conformational isomers of COF-300 were uncovered associated with different guest molecules. As indicated by PXRD patterns and energetic landscapes of the conformational isomers, the structural transformation from a high-energy state to a low-energy state is a rapid, energetically favorable process, while the reverse transformation is a slow process driven by concentration gradients. The fact that COF-300 maintains an intact single-crystal structure even after absorbing molten PAHs at 280 °C renders it a promising candidate as a crystalline sponge for selective adsorption and separation of gases and fluids under high-temperature conditions.
Methods
Materials and instruments
Tetrakis(4-aminophenyl)methane (TAM, purity ≥ 98%), Benzene-1,4-dicarboxaldehyde (BDA, purity ≥ 99%) were purchased from Jilin Chinese Academy of Sciences-Yanshen Technology Co., Ltd. Mesitylene, 1,2,4-trimethylbenzene or 1-methyl naphthalene, cyclohexane, dichloromethane, trichloromethane, methanol, ethanol, acetonitrile, acetone, N, N-dimethylformamide, dimethyl sulfoxide, n-hexane, tetrahydrofuran, thiophene, toluene, chlorobenzene, o-xylene, m-xylene, p-xylene, and benzyl alcohol were purchased from Aladdin Chemical Reagents Co., Ltd., China.
Single-crystal X-ray diffraction (SXRD) data were collected on a Bruker D8 VENTURE CMOS photon 100 diffractometer with helios mx multilayer monochromator Cu Kα radiation (λ = 1.5418 Å) at 173 K and 293 K. Crystal data and details of the structure refinement are given in Supplementary Tables S3–S27. Powder X-ray diffraction (PXRD) data were collected on a Rigaku-DMAX 2500 diffractometer with Cu Kα radiation (λ = 1.5418 Å) at a scanning rate of 2° /min for 2θ ranging from 5° to 30° (Supplementary Fig. S51–S66). The emission spectra and excitation spectra were collected on Hitachi F-4600 fluorescence spectrophotometer (Supplementary Figs. S70, S71). The lifetimes were collected on an Edinburgh FLS920P fluorescence spectrometer equipped with a 450 W xenon lamp as an excitation source (Supplementary Fig. S72). The morphology of COF-300 was examined by Hitachi SU-8010 field-emission scanning electron microscope (Supplementary Fig. S74).
Solvent-induced structural transformation at room temperature
Single crystals of COF-300 were immersed in various solvents for 24 h at room temperature, including water, mesitylene, 1,2,4-trimethylbenzene, 1-methyl naphthalene, cyclohexane, dichloromethane, trichloromethane, ethanol, acetonitrile, acetone, N, N-dimethylformamide, dimethyl sulfoxide, n-hexane, tetrahydrofuran, thiophene, toluene, chlorobenzene, o-xylene, m-xylene, p-xylene, and benzyl alcohol. The single crystals were then sealed in a capillary tube to collect single-crystal X-ray crystallography.
Structural transformation at high temperature
Single crystals of COF-300 and PAHs (naphthalene, anthracene, pyrene, and perylene) were mixed well and then placed in a quartz tube and heated to the melting temperature of PAHs for 6 h, then cooled down to room temperature (melting points: naphthalene 82 °C, anthracene 216 °C, pyrene 148 °C, and perylene 280 °C). Solid PAHs were dissolved with ethanol and COF-300 single crystals were collected for single-crystal X-ray crystallography.
Structural modeling and density functional theory calculations
Based on the single-crystal data, we selected a complete edge of the square channel as a model to calculate the single-point energy, including the deformation energy (Edef), the interaction energy (Eint), and the total energy of the framework (Etot) by using M06-2X function and 6-311 G(d) basis set. All calculations were conducted by using the Gaussian 16 program package37. In the case of COF-300-c (guest: H2O), the asymmetric unit contains only one-quarter of a tetrakis(4-aminophenyl)methane unit and one-half of a terephthalaldehyde unit, as COF-300-c crystallizes in the highly symmetric I41/a space group (Supplementary Fig. S73a). Given that the seven-fold interpenetrating dia networks are generated from the asymmetric unit through symmetry operations, a complete edge of the square channel is selected for modeling to investigate the free energy landscape (Supplementary Fig. S73b).
Data availability
The X-ray crystallographic coordinates for structures reported in this article have been deposited at the Cambridge Crystallographic Data Center (CCDC), under deposition numbers CCDC 2375329 to 2375353. These data can be obtained free of charge from the CCDC via www.ccdc.cam.ac.uk/data_request/cif. Source data are provided in this paper.
References
Kitagawa, S. & Kondo, M. Functional micropore chemistry of crystalline metal complex-assembled compounds. Bull. Chem. Soc. Jpn. 71, 1739–1753 (1998).
Horike, S., Shimomura, S. & Kitagawa, S. Soft porous crystals. Nat. Chem. 1, 695–704 (2009).
Krause, S., Hosono, N. & Kitagawa, S. Chemistry of soft porous crystals: Structural dynamics and gas adsorption properties. Angew. Chem. Int. Ed. 59, 15325–15341 (2020).
Carrington, E. J. et al. Solvent-switchable continuous-breathing behaviour in a diamondoid metal–organic framework and its influence on CO2 versus CH4 selectivity. Nat. Chem. 9, 882–889 (2017).
Vanduyfhuys, L. et al. Thermodynamic insight into stimuli-responsive behaviour of soft porous crystals. Nat. Commun. 9, 204 (2018).
Gu, Y. et al. Structural-deformation-energy-modulation strategy in a soft porous coordination polymer with an interpenetrated framework. Angew. Chem. Int. Ed. 59, 15517–15521 (2020).
Bourrelly, S. et al. Different adsorption behaviors of methane and carbon dioxide in the isotypic nanoporous metal terephthalates MIL-53 and MIL-47. J. Am. Chem. Soc. 127, 13519–13521 (2005).
Krause, S. et al. Engineering micromechanics of soft porous crystals for negative gas adsorption. Chem. Sci. 11, 9468–9479 (2020).
Lama, P. & Barbour, L. J. Distinctive three-step hysteretic sorption of ethane with in situ crystallographic visualization of the pore forms in a soft porous crystal. J. Am. Chem. Soc. 140, 2145–2150 (2018).
Rogge, S. M. J., Waroquier, M. & Van Speybroeck, V. Unraveling the thermodynamic criteria for size-dependent spontaneous phase separation in soft porous crystals. Nat. Commun. 10, 4842 (2019).
Krause, S. et al. Cooperative light-induced breathing of soft porous crystals via azobenzene buckling. Nat. Commun. 13, 1951 (2022).
Nikolayenko, V. I. et al. Reversible transformations between the non-porous phases of a flexible coordination network enabled by transient porosity. Nat. Chem. 15, 542–549 (2023).
Henke, S., Li, W. & Cheetham, A. K. Guest-dependent mechanical anisotropy in pillared-layered soft porous crystals – a nanoindentation study. Chem. Sci. 5, 2392–2397 (2014).
Ortiz, A. U., Boutin, A., Fuchs, A. H. & Coudert, F.-X. Anisotropic elastic properties of flexible metal-organic frameworks: How soft are soft porous crystals? Phys. Rev. Lett. 109, 195502 (2012).
Mellot-Draznieks, C., Serre, C., Surblé, S., Audebrand, N. & Férey, G. Very large swelling in hybrid frameworks: A combined computational and powder diffraction study. J. Am. Chem. Soc. 127, 16273–16278 (2005).
Serre, C. et al. Role of solvent-host interactions that lead to very large swelling of hybrid frameworks. Science 315, 1828–1831 (2007).
El-Kaderi, H. M. et al. Designed synthesis of 3D covalent organic frameworks. Science 316, 268–272 (2007).
Auras, F. et al. Dynamic two-dimensional covalent organic frameworks. Nat. Chem. 16, 1373–1380 (2024).
Kang, C. et al. Covalent organic framework atropisomers with multiple gas-triggered structural flexibilities. Nat. Mater. 22, 636–643 (2023).
Borgmans, S., Rogge, S. M. J., De Vos, J. S., Van Der Voort, P. & Van Speybroeck, V. Exploring the phase stability in interpenetrated diamondoid covalent organic frameworks. Commun. Chem. 6, 5 (2023).
Chen, Y. et al. Guest-dependent dynamics in a 3D covalent organic framework. J. Am. Chem. Soc. 141, 3298–3303 (2019).
Li, L. et al. Emission-tunable soft porous organic crystal based on squaraine for single-crystal analysis of guest-induced gate-opening transformation. J. Am. Chem. Soc. 143, 3856–3864 (2021).
Zeng, T. et al. Atomic observation and structural evolution of covalent organic framework rotamers. Proc. Nat. Acad. Sci. USA 121, e2320237121 (2024).
Thakur, S. & Giri, A. Reversible and high-contrast thermal conductivity switching in a flexible covalent organic framework possessing negative Poisson’s ratio. Mater. Horiz. 10, 5484–5491 (2023).
Liu, X. et al. A crystalline three-dimensional covalent organic framework with flexible building blocks. J. Am. Chem. Soc. 143, 2123–2129 (2021).
Liu, X. et al. Gas-triggered gate-opening in a flexible three-dimensional covalent organic framework. J. Am. Chem. Soc. 146, 11411–11417 (2024).
Ma, Y.-X. et al. A dynamic three-dimensional covalent organic framework. J. Am. Chem. Soc. 139, 4995–4998 (2017).
Zhu, Q. et al. 3D Cage COFs: A dynamic three-dimensional covalent organic framework with high-connectivity organic cage nodes. J. Am. Chem. Soc. 142, 16842–16848 (2020).
Yu, B. et al. Linkage conversions in single-crystalline covalent organic frameworks. Nat. Chem. 16, 114–121 (2024).
Yi, L., Gao, Y., Luo, S., Wang, T. & Deng, H. Structure evolution of 2D covalent organic frameworks unveiled by single-crystal X-ray diffraction. J. Am. Chem. Soc. 146, 19643–19648 (2024).
Zhou, Z. et al. Linker-guided growth of single-crystal covalent organic frameworks. J. Am. Chem. Soc. 146, 3449–3457 (2024).
Han, J. et al. Fast growth of single-crystal covalent organic frameworks for laboratory x-ray diffraction. Science 383, 1014–1019 (2024).
Uribe-Romo, F. J. et al. A crystalline imine-linked 3-D porous covalent organic framework. J. Am. Chem. Soc. 131, 4570–4571 (2009).
Ma, T. et al. Single-crystal x-ray diffraction structures of covalent organic frameworks. Science 361, 48–52 (2018).
Ma, T. et al. Observation of interpenetration isomerism in covalent organic frameworks. J. Am. Chem. Soc. 140, 6763–6766 (2018).
Spek, A. PLATON SQUEEZE: a tool for the calculation of the disordered solvent contribution to the calculated structure factors. Acta. Crystallogr. Sect. C 71, 9–18 (2015).
Frisch, M. J., et al. Gaussian 16, Revision C.01, Gaussian Inc, Wallingford CT (2019).
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (no. 22371032).
Author information
Authors and Affiliations
Contributions
C.S. and X.W. conceived the idea. A.Y. conducted the synthesis and crystal growth of COF-300. H.X. conducted the theoretical calculation. K.S. carried out the crystallographic studies. C.Q., X.W., and Z.S. interpreted the results and prepared the manuscript. All the authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yao, A., Xu, H., Shao, K. et al. Guest-induced structural transformation of single-crystal 3D covalent organic framework at room and high temperatures. Nat Commun 16, 1385 (2025). https://doi.org/10.1038/s41467-025-56750-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-56750-9
This article is cited by
-
Enhanced proton conductivity in azole-functionalized three-dimensional crystalline covalent organic frameworks
Nature Communications (2026)
-
Epitaxial growth of high-nuclearity zirconium oxo clusters from Zr16 to Zr20 and subsequent ligand exchange
Science China Chemistry (2026)