Abstract
Precise organization of organic molecules into homochiral double-helix remains a challenge due to the difficulty in controlling both self-assembly process and chirality transfer across length scales. Here, we report that a type of bisnaphthalene bisurea molecule could assemble into chirality-controlled nanoscale double-helices by a supramolecular rosette-intermediated hierarchical self-assembly mechanism. A solvent-mixing self-assembly protocol is adopted to direct bisnaphthalene bisurea cyclization into chiral discrete rosettes through cooperative intramolecular and intermolecular hydrogen bonds. Controlled hexagonal packing of rosettes at higher concentrations gives one-dimensional single-stranded nanofibers, which intertwine into well-defined double-helix nanostructures with preferred chirality that depends on the absolute configurations of bisnaphthalene bisurea. The hierarchical organization of bisnaphthalene bisurea molecules enables effective excitation energy delocalization within the double-helix, which contributes to near-unity energy transfer from double-helix to adsorbed acceptor dyes even in donor/acceptor ratios over 1000, leading to bright circularly polarized luminescence from the originally achiral acceptor. The experimental and theoretical simulation results not only provide a hierarchical strategy to fabricate homochiral double-helix but also bring insights in understanding the high-efficiency light-harvesting process in photosystem II.
Similar content being viewed by others
Introduction
Helical structures widely exist in nature as the basis for transferring chiral information and realizing complex biological functions1,2. For example, DNA double-helix and protein α-helix play indispensable roles in gene replication and directional peptide folding3,4,5. The synthesis of artificial helical structures can not only meet people’s pursuit of aesthetics originating from these nature models but also provide a structural foundation for understanding the complexity of chirality and chiroptical functions6,7,8,9,10,11,12,13,14,15,16,17,18,19. Inspired by the hydrogen-bond-driven exquisite double-helix of DNA, scientists have explored the self-assembly of various building blocks into double-helices by non-covalent interactions20,21. One pioneering methodology in this field was the preparation of unique coordination compounds from metal ions and several linear polydentate ligands, which were termed by Lehn22 as ‘helicates’ in 1987. Since then, the creation of a variety of helicates has witnessed flourishing decades, which significantly puts forward the research field of helical supramolecular structures23,24,25,26,27. The helical assembly of the ligands around the metal coordination sphere also brought the question of stereocontrol, i.e., the control of left- or right-helicity, which was one of the major challenges in the helicate field until the discovery of Scott and co-workers28 in 2012. They successfully synthesized a type of asymmetric helicate with enantiomerical purity by using carefully selected directional chiral ligands.
Compared with coordination-guided assembly to form colorful achiral and chiral helicates, the self-assembly of organic small molecules into helical structures, especially double-stranded ones16,20,27,28,29,30,31,32,33,34,35,36,37, is more difficult due to the challenge of helical twist wrapping of molecules around each other in the absence of templates such as metal ions. Besides, in contrast to significant advances in the fabrication of double-helix structures, the functionalization of these double-helix systems was backward. Here, by adopting hierarchical self-assembly of chiral bisnaphthalene bisurea (NU) molecules, we constructed handedness-controlled nanoscale double-helices from supramolecular rosette intermediates and realized high-efficiency chiral light-harvesting function. Discrete supramolecular rosettes, i.e., rosette-like cyclic hexamers38,39,40,41,42,43,44, were firstly prepared by an anti-solvent self-assembly protocol in dilute solution and fully characterized with collective data including diffusion-ordered spectroscopy, high-resolution mass spectroscopy, cryo transmission electron microscope, and density functional theory calculations. Further rosette stacking and intertwining in a hexagonal mode at a higher concentration led to the formation of well-defined nanoscale double-helices, which were employed to adsorb achiral dyes to establish a chiral artificial light-harvesting antenna (LHA)45,46,47,48,49,50,51,52,53,54. Excitation of donor chromophores within the LHA led to efficient Förster resonance energy transfer with impressive energy transfer efficiency (ΦET up to 96.1%) in particular in the cases where donor/acceptor (D/A) ratios ≥1000, which represented an elegant example that overcame the trade-off between ΦET value and D/A ratio in artificial LHAs55,56,57,58,59,60,61,62. Together with induced chirality from the double-helix63,64,65,66,67,68, the achiral dye after excitation energy transfer could emit a strong cyan CPL signal with a quantum yield of up to 87%.
Results
The formation and characterization of rosettes
Inspired by the chemical structure characteristics of concentrating pigments and cyclic assembly geometry of the natural LHA69, a new type of chiral building block consisting of two naphthalene units, chiral centers, and hydrogen bonding moieties, (R, R)-NU and (S, S)-NU enantiomers, was designed and synthesized from o-phenylenediamine and chiral naphthalene isocyanate with a yield of about 80% (Fig. 1a and Supplementary Fig. 1). NU enantiomers were characterized by 1H, 13C nuclear magnetic resonance (NMR) and high-resolution mass spectroscopy. The hydrogen atoms on NU were assigned according to 1H-NMR coupling constant analysis, correlated spectroscopy (COSY), and nuclear Overhauser effect spectroscopy (NOESY) (Supplementary Figs. 1 and 2). DFT-optimized NU structure indicated that two urea groups adopted opposite directions using the benzene ring as a reference plane and formed an intramolecular hydrogen bond (Fig. 1b and Supplementary Fig. 3a). This unique conformation brought significant impacts on naphthalene orientation and interchromophoric interaction as revealed by excimer emission and bisignate Cotton effect in dilute N, N-dimethylformamide (DMF) solution of NU (Supplementary Fig. 4). The electrostatic potential (ESP) map supported an asymmetric electron distribution with negative electron density on one urea and positive electron density on the other (Fig. 1c), which ensured the formation possibility of head-to-tail intermolecular H-bond array. We therefore added water as a poor solvent to promote the self-assembly of NU monomers dissolved in DMF while keeping the total concentration constant at 0.1 mM. For NMR experiments, NU was firstly dissolved in DMF-d7 by heating, and then deuterated water was added after cooling to R.T. to promote self-assembly. The signals of aromatic protons in NMR measurements showed an obvious shift upon increasing the deuterated water fractions (fw) from 0 to 30% (Fig. 2a), which was a typical characteristic of urea hydrogen bond formation and chromophore self-assembly. A plot of chemical shifts (proton a) against water fractions showed a linear fitting relationship, indicating the bulky solvent effect of added water in promoting the self-assembly (Supplementary Fig. 6). Diffusion-ordered spectroscopy (DOSY) measurement of the assemblies at fw = 30% proved the formation of a single NU oligomer with a diffusion coefficient of D ≈ 1.50 × 10−6 cm2 · s−1 (Fig. 2b). According to the Stocks-Einstein equation, D = kT/3πηd, where k, T, η and d represent Boltzmann constant, temperature, solvent viscosity and particle hydrodynamic diameter, respectively, the hydrodynamic diameter of NU oligomer was then calculated to be about d = 3.0 ± 0.1 nm (Supplementary Equation 1) upon three repeated measurements (Supplementary Fig. 7). High-resolution electrospray ionization mass spectroscopy (HR-ESI-MS) showed the absence of 7mer, 8mer and larger species in the region of m/z > 3000 (Supplementary Fig. 8) and confirmed the existence of a NU hexamer, which clearly showed m/z values corresponding to NU hexamers and fragmented NU dimers, trimers, tetramers and pentamers (Fig. 2c and Supplementary Table 1). Considering the opposite orientation of urea groups and aggregate size, a rosette-like cyclic hexamer of NU molecules was deduced (Fig. 1d). The DFT-optimized rosette hexamer displayed a well-defined cyclic geometry with alternate intramolecular and intermolecular H-bonds among urea groups and a geometrical diameter of about 3.0 nm (Fig. 2f, g and Supplementary Fig. 9a), which was in good agreement with the experimental hydrodynamic diameter (~3.0 nm) from the DOSY results. Cryogenic capture of NU oligomers at fw = 30% by cryo transmission electron microscope (cryo-TEM) showed nanostructures with an averaged diameter of 3.0 ± 0.1 nm (Fig. 2d, e, Supplementary Fig. 10), which therefore directly confirmed the formation of NU rosette. The extracted urea groups from the DFT-optimized rosette were visualized in space-filling mode and showed a cyclic H-bond array with six free N–H sites, contributing to further hierarchical self-assembly and guest-binding abilities (Fig. 2h and Supplementary Fig. 9b).
a The chemical structures of (R, R)-NU and (S, S)-NU enantiomers. b DFT-optimized structure of (R, R)-NU showed opposite urea directions using the benzene ring as a reference plane. c DFT-optimized electrostatic potential map of (R, R)-NU. Scale bar: arbitrary unit (arb. units). d The chemical structure of a rosette held together by intermolecular (black dashed lines) and intramolecular (red dashed lines) hydrogen bonds. R = ethyl naphthalene. e Schematic illustration of rosette stacking and intertwining into double-helix nanostructures, which adsorb achiral CBS dye to perform chiral light-harvesting, leading to circularly polarized luminescence from CBS. CPL: circularly polarized luminescence. CBS: 2,2′-([1,1′-biphenyl]-4,4′-diyldi-2,1-ethenediyl)bis-benzenesulfonic acid disodium salt. D/A ratios: donor/acceptor ratios. ΦET: excitation energy transfer efficiency.
a Stacked 1H-NMR spectra of (R, R)-NU in DMF-d7 with increasing deuterated water fractions (fw). The peaks between 7.9 and 8.2 ppm are ascribed to the solvent residual of DMF. [NU] = 0.1 mM. The labels of a to m at the bottom refer to the aromatic protons of NU, whose chemical structure is inserted in (2b). b Diffusion-ordered spectroscopy of (R, R)-NU in DMF-d7/D2O mixed solvent, fw = 30%, [NU] = 0.1 mM. D: diffusion coefficient, d: hydrodynamic diameter. c High-resolution electrospray ionization mass spectroscopy of (R, R)-NU in acetonitrile. The inserted image represents experimental results (upper) and calculated results (bottom) of isotopically resolved m/z signals of a NU hexamer [6NU+Na]+. d The cryo-TEM image of NU assemblies in DMF/H2O mixed solvents, fw = 30%, [NU] = 0.1 mM. The inserted image is an enlarged micrograph of the observed nanostructure. e The statistical particle size distribution of nanostructures in (2 d). f, g DFT-optimized structure of a rosette by Gaussian 09 program at B3LYP 6-31 G* level. The red, blue, white, and gray balls represent oxygen, nitrogen, hydrogen, and carbon atoms, respectively. Except for hydrogen atoms in urea groups, other hydrogen atoms are omitted for clarity. h The space-filling mode of the cyclic hydrogen bond array extracted from a DFT-optimized rosette.
Rosettes stacking and intertwining into nanoscale double-helices
By increasing water fractions, we investigated the subsequent assembly of rosettes. Increasing fw to 40% caused the decrease of proton integrals (Fig. 2a, [NU] = 0.1 mM), implying the formation of NMR-invisible large aggregates, which was also verified by obvious Tyndall effects (Supplementary Fig. 11). SEM images showed that short nanofibers were formed at fw = 40%, which gradually got longer and intertwined into double-helices upon increasing fw values and finally led to the formation of double-helix nanofibers with length scale over several micrometers (Supplementary Fig. 12). We have measured the CD spectra of NU assemblies at different water fractions at the concentration of 0.1 mM. For NU assemblies under water fractions ≤40% (Supplementary Fig. 13a), the shape and major peaks of CD spectra were similar to the NU monomers (Supplementary Fig. 4c), which meant that the molecular organization into rosettes and short nanofibers did not change the chiroptical properties of NU. In sharp contrast, the double-helices obtained at water fractions ≥50% gave inversed CD signals to NU monomers (Supplementary Fig. 13). Therefore, the intertwining of short nanofibers into long double-helices significantly changed the supramolecular chirality, which represents a good example for the presentation of chirality transfer across length scales.
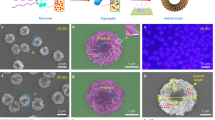
Further increasing concentrations ([NU] = 10 mM, fw = 70%) were capable of triggering the cross-linking of double-helices, leading to the formation of an organohydrogel material within several seconds, which was confirmed by both vial inversion test and rheological measurement (Supplementary Fig. 14). The microscopic morphology of NU gel was carefully studied by electron microscope techniques including cryo-TEM, scanning electron microscope (SEM) and atomic force microscope (AFM). Well-defined double-helix nanostructures were observed from cryo-TEM micrographs with different zooms of the gel samples (Fig. 3a, b, and Supplementary Fig. 15), which intertwined to maintain the gel network (Fig. 3c, d). The width of the single-stranded nanofiber of the double-helix nanostructures was measured to be about 8.8 ± 0.3 nm (Fig. 3a, b, and Supplementary Figs. 16 and 17), which was approximately three times to rosette diameter. The helicity directions of these self-assembled double-helix nanostructures were apparently determined by the absolute configurations of NU molecules from micrograph observation. Right-handed and left-handed double-helices were prepared from (R, R)-NU and (S, S)-NU monomers, respectively (Fig. 3c, d). AFM measurements and morphological analysis revealed that the chiral double-helices had a uniform helical pitch of 37.2 ± 1.7 nm for (R, R)-NU and 37.4 ± 2.5 nm for (S, S)-NU (Fig. 3e, f and Supplementary Figs. 18, 19). Further increasing water fractions to fw = 90% brought little effect on the height and Cotton effects of double-helices (Supplementary Fig. 20). The statistical height of the double-helix at fw = 90% gave a value of 10.0 ± 0.6 nm, which is very similar to the result obtained at fw = 70%. In contrast to the self-assembly at R.T., thermodynamically controlled self-assembly gave totally different morphology. After heating the same amount of NU in co-solvent (DMF/H2O = 3/7, v/v) to a transparent solution and then naturally cooling to R.T., only white precipitates were obtained, whose morphology showed nanorods from SEM observation (Supplementary Fig. 21b). Besides, increasing the NU concentration to 30 mM in pure DMF could also obtain a gel by applying a heating-cooling assembly protocol. The SEM images of the resulting gel sample also showed helix structures but with less double-helices (Supplementary Fig. 22). These results indicated that the double-helical nanostructures could only be prepared in a kinetically controlled approach by a solvent-mixing strategy.
Cryo-TEM micrographs of a (R, R)-NU and b (S, S)-NU gels exhibited double-helix nanostructures. SEM micrographs of c (R, R)-NU and d (S, S)-NU xerogels. P: right-handedness, M: left-handedness. The inserted images at the left bottom are cartoons of corresponding double-helices. e AFM micrograph of (R, R)-NU. The numbers denote the selected double-helix sections for pitch analysis (for details, please see Supplementary Fig. 18). f Statistical analysis of helical pitch distribution of double-helices in (e). g 2D-GIWAXS patterns of NU gel. h Top view (left) and side view (right) of a hexagonally packed model of dozens of rosettes obtained by MD simulations. i Normalized absorption and fluorescence spectra of (R, R)-NU gel. j CD and k CPL spectra of (R, R)-NU (cyan lines) and (S, S)-NU gels (orange lines). Unless otherwise noted, [NU] = 10 mM in DMF/H2O mixed solvent, fw = 70%.
2D grazing incident wide-angle X-ray scattering (2D-GIWAXS) patterns and powder X-ray diffraction (PXRD) were applied to clarify the molecular packing mode of the double-helix nanostructures. The major d-spacing values of 1.59, 1.17, 0.91, 0.79, 0.72, 0.69, 0.60, 0.58, 0.51 and 0.49 nm were corresponding to Miller Indices (hkl) of (010), (014), (111), (020), (024), (025), (210), (123), (303) and (305), which were in consistency with theoretical hkl values of a typical hexagonal system (Fig. 3g, Supplementary Fig. 23 and Supplementary Table 2)70. The d-spacing value of 1.59 nm corresponding to the first diffraction peak was exactly equal to the DFT-optimized molecular width of NU (Supplementary Fig. 3b). Therefore, the hexagonal packing diffraction peaks most likely originated from the stacked rosettes (with a length of side about 1.5 nm). Atomic MD simulations were applied to provide insight into the understanding of the self-assembly mechanism. By adopting a hexagonal packing mode, 7 × 12 pairs of DFT-optimized rosettes were aligned as one block of cluster with a shoulder-to-shoulder rosette distance of ~1.5 nm (Supplementary Fig. 24), which was then solvated in DMF/H2O mixture (fw = 70%) and equilibrated through MD simulations under the constant pressure/temperature (NPT) ensemble for 40 ns. The equilibrium configuration showed that both the rosettes and their hexagonal packing structure remained almost unchanged. The length of the rosette side was measured to be about 1.5 nm and the diameter of the whole structure was 9.1 nm, which was consistent with the d-spacing value (~1.6 nm) and statistical nanofiber width (8.8 ± 0.3 nm) from cryo-TEM micrographs, respectively. Based on these experimental and computational results, a possible self-assembly mechanism was proposed (Fig. 1d, e). Six NU molecules first formed a cyclic intermediate as a rosette, which then self-assembled into 1-D fibrous structures along the perpendicular direction of the rosette plane by adopting a hexagonal packing mode. Two 1-D nanofibers subsequently intertwined together into a nanoscale double-helix, which was confirmed by a double-helix nanostructure with two unwound single-stranded nanofibers at one terminus (Supplementary Fig. 15b). Therefore, the measured size of the single-stranded nanofiber cross-section (~8.8 nm) should be about three times to the rosette diameter (~3 × 3 nm = 9 nm).
The NU gel composed of double-helices had a maximum absorption peak centered at 290 nm, which was ascribed to naphthalene HOMO-LUMO transition as demonstrated by DFT-calculated molecular orbitals and UV-Vis absorption spectrum (Supplementary Figs. 25 and 26). The fluorescence spectrum showed a strong emission band at 352 nm with a shoulder peak near 425 nm (Fig. 3i, k). Time-resolved fluorescence measurements monitored at 425 nm gave a lifetime value (τ) of about 29.8 ns, which was four times longer than the τ value (7.2 ns) of NU naphthalene in dilute solution (Supplementary Fig. 27). The long fluorescence lifetime most likely originated from closely assembled naphthalene chromophores within the double-helix network49. The presence of intermolecular naphthalene interactions also significantly affected the chiroptical properties71. The first positive Cotton effect centered at 292 nm corresponding to naphthalene HOMO-LUMO transition of (R, R)-NU in dilute solution moved to 330 nm after forming double-helices (Fig. 3j, cyan line), accompanied with the emergence of a positive CPL signal at 365 nm (Fig. 3k, cyan line). Besides, mirror-imaged CD and CPL signals were observed for (R, R)-NU and (S, S)-NU double-helices (Fig. 3j, k, and Supplementary Fig. 28 for luminescent dissymmetry factors glum), respectively, indicating effective chirality transfer from the molecular to supramolecular level72.
Chiral light-harvesting function of nano double-helices
The nanometer-sized double-helices are similar to the helical subunits of natural LHA in terms of topology and chirality, which encourages us to utilize the assembled chiral double-helices as an artificial LHA. A CBS dye (for chemical structure, see Fig. 1e) with a high fluorescence quantum yield of 62% in solution was selected as an energy acceptor due to its spectral overlap and appropriate binding ability with donor NU. The emission curve of NU almost completely overlapped with the absorption band of CBS (Fig. 4a, red solid line, and blue dotted line), ensuring the possibility of an efficient FRET process. A series of 1H-NMR and UV-Vis titration experiments were conducted to clarify the binding behaviors between NU and CBS. Upon increasing the CBS ratios, the signals of two urea NH protons gradually shifted downfield (Supplementary Fig. 29), indicating the presence of a binding event between urea groups and sulfonate anions on CBS. Job’s plot analysis based on UV-Vis measurements disclosed that the donor and acceptor interacted with each other in a 1:1 complex ratio in the solution (Supplementary Fig. 30). NMR titration experiments also supported the 1:1 complexation model with a binding constant Ka of 177.6 ± 2.0 M−1 (Supplementary Fig. 31) in DMF-d7. In the presence of D2O, the binding constant slightly increased to Ka of 216.1 ± 14.3 M−1 (Supplementary Figs. 32 and33). The moderate binding constant could ensure effective adsorption of CBS dye on the double-helix surfaces without damaging the ordered nanostructures.
a Normalized absorption and emission spectra of NU gel and CBS solution, in which the overlapped region was marked with gradient color. b Emission spectra, and c ФET values of the LHA system with different NU/CBS ratios. d Fluorescence decay curves of NU gel without (blue line) and with (orange line) CBS acceptor. The emission was monitored at 350 nm with an excitation wavelength of 300 nm. e CD spectra of (R, R)-NU/CBS (red line) and (S, S)-NU/CBS (blue line) co-gels. f CPL spectra of (R, R)-NU/CBS (solid lines) and (S, S)-NU/CBS (dashed lines) co-gels by excitation on NU (λex = 290 nm, red lines) or CBS (λex = 350 nm, blue lines). g The changes of helical pitch and height profile of double-helices by AFM images under different NU/CBS ratios. The blue and yellow dashed lines indicate error bars, which represent standard errors from AFM height profiles. h Proposed CBS binding mode based on NMR and DFT-calculated results. Left image shows two rows of MD-optimized stacked rosettes and two adsorbed CBS on the surface. Right image shows the DFT-optimized binding mode of one NU and one CBS.
Both excitation energy transfer and chirality transfer among donor and acceptor within the double-helix structures were explored to evaluate their chiral light-harvesting capability. Co-assembling trace amount of CBS into NU gel (NU/CBS = 10000:1 in molar ratio) initiated a FRET process with a ФET value of about 51.5% according to the equation of ФET = 1-IDA/ID, where IDA and ID represented the donor emission intensity in the presence and absence of CBS acceptor, respectively (Fig. 4b, c). Changing the D/A ratios from 10000:1 to 1000:1 led to a significant emission decline of NU donor, while the CBS emission peak located at 430 nm gradually increased, affording impressive ФET values up to 96.1%, which were among the largest ones at the same level of D/A ratios to the best of our knowledge. The high energy transfer efficiency was also supported by changes in donor fluorescence lifetime before (29.82 ns) and after (2.34 ns) the FRET process (Fig. 4d and Supplementary Fig. 34). An antenna effect value of 6.0 was calculated at a D/A ratio of 1000:1 (Supplementary Fig. 35). Further increasing the CBS loading ratio to 100%, i.e., D/A ratio = 1:1, brought quantitative excitation energy transfer with an enhanced acceptor fluorescence quantum yield of 87%. CD spectra of (R, R)-NU/CBS co-gel (molar ratio kept at 1000:1) showed positive Cotton effects corresponding to the CBS absorption band in the wavelength range of 325–425 nm (Fig. 4e, red line) due to induced chirality from the double-helices., which was in agreement with the positive Cotton effects of NU gels in their major absorption bands. A CD stack plot of (R, R)-NU gel and (R, R)-NU/CBS co-gel (molar ratio kept at 1000:1) showed intensity change near 250 and 300 nm, which was probably caused by the CD signal superposition of NU host and CBS guest (Supplementary Fig. 36). The combination of effective excitation energy transfer and chirality transfer laid the foundation for chiral light-harvesting process as revealed by the CPL studies. Excitation on NU resulted in CPL from CBS dyes with a luminescent dissymmetry factor |glum| of 4.5 × 10−3 (Fig. 4f, red lines), which was eight times larger than that obtained upon direct excitation on CBS (Fig. 4f, blue lines), indicating the unique role of chiral light-harvesting in amplifying CPL.
The excellent chiral light-harvesting ability was supposed to be closely related to the rosette stacking and guest binding on double-helix surfaces, which was further unambiguously proven by a series of characterizations and morphological observation. First, the macroscopic gel state kept unchanged upon doping varied amount of CBS (D/A ratios ranging from 1000:1 to 1:1, Supplementary Fig. 37). Second, the helical pitches of microscopic double-helix structures displayed no significant change while the fiber height increased from 10 to 29 nm as observed in SEM, AFM, and TEM images (Fig. 4g and Supplementary Figs. 38–45). Third, the PXRD and FT-IR peaks of NU/CBS co-gels at different D/A ratios had similar patterns (Supplementary Fig. 46). These results indicated that CBS molecules were adsorbed on the helix surfaces instead of intercalating into NU aggregates, which led to the enhancement of fiber height while maintaining the helical pitch and gel structure. This inference was also verified by the following control experiment. After the formation of NU gel, a certain amount of CBS (D/A ratio = 1000:1) in aqueous solution was carefully placed on the top of the gel to allow gradient adsorption (Supplementary Fig. 47a). The NU/CBS co-gel obtained through the post-assembly adsorption method showed similar chiroptical signals and double-helix nanostructures to those of the co-assembled gels (Supplementary Figs. 47 and48). MD simulation and DFT calculation results showed that the face-to-face rosette distance was measured to be about 1.7 nm, which was in the same length scale as the sulfonate group distance (~1.8 nm) of the CBS acceptor (Fig. 4h, left image). Therefore, one CBS dye could anchor two NU molecules in neighboring rosettes through urea-sulfonate H-bond as revealed by DFT calculations (Fig. 4h, right image), which contributed to efficient energy transfer and chirality transfer from aggregated NU donor to CBS acceptor. We have compared the chiral light-harvesting efficiency of double-helices and nanorods obtained by different self-assembly protocols (Supplementary Fig. 49). The nanorods showed a much lower efficiency at the same level of NU/CBS ratios. As shown in Supplementary Fig. 49, a FRET efficiency of 40.1% at the condition of NU/CBS = 1000:1 was obtained. To ensure a complete FRET process, the NU/CBS ratio needs to be reduced to 100:1 for the nanorod system, highlighting the unique role of double-helical topology in directing high-efficiency FRET.
Discussion
In summary, a hierarchically self-assembled double-helix nanostructure from supramolecular rosette intermediates was prepared and utilized to build a chiral LHA model, which could realize almost quantitative energy transfer at a significantly high D/A ratio and emit unique circular polarized luminescence (CPL) from acceptor after light-harvesting. The uniform-sized chiral double-helix nanostructures were prepared by hierarchical self-assembly of NU molecules, which firstly aggregated into rosette-like cyclic hexamers and then hexagonally packed into nanoscale double-helices as revealed by collective experimental and computational data. Non-covalent adsorption of an achiral CBS acceptor on the double-helix surface through urea-sulfonate interactions brought negligible influence on the size of helical pitch, which contributed to the robustness of the co-assembled chiral LHA. Excitation of the aggregated naphthalene chromophores led to an efficient FRET process among the D/A dyad with impressive ΦET values (up to 96.1%) in particular in the cases where D/A ratios ≥1000. We believe that this work will deepen our understanding of natural LHA with chirality as a key characteristic and provide a powerful methodology to develop chiroptical materials with double-helical topology.
Methods
Materials
All commercial chemicals were used as received without further purification. Milli-Q water (18.2 MΩ·cm) was used in all cases. 1-[(1S)-1-isocyanatoethyl] naphthalene (purity >95%), 1-[(1 R)-1-isocyanatoethyl] naphthalene (purity >95%), 1,2-diaminobenzene (purity >98%) were purchased from TCI. Triethylamine (purity >99%) was purchased from J&K. The reactants 1,2-diaminobenzene, 1-[(1S)-1-isocyanatoethyl] naphthalene (or 1-[(1 R)-1-isocyanatoethyl] naphthalene), trimethylamine were dispersed and stirred in dry dichloromethane at r.t. to obtain the crude products (S, S)-NU (or (R, R)-NU). The products were purified by recrystallization and characterized by 1H NMR, 13C NMR, and MALDI-FTICR-MS. The detailed synthetic procedures of (R, R)-NU and (S, S)-NU are listed in Supplementary Fig. 1.
Self-assembly protocol
NU (0.01 mmol) was dissolved in DMF (300 μL) upon heating. After cooling to room temperature for 10 min, 700 μL distilled water was added to the above DMF solution. A colorless transparent gel was obtained immediately and the sample was allowed to age for 24 h before further testing.
Co-assembly protocol
(R, R)-/(S, S)-NU (0.01 mmol) and dye CBS with different molar ratios were dissolved in DMF (300 uL) upon heating. The same procedures were then adopted as those in the self-assembly protocol.
Ultraviolet-Visible spectroscopy
UV-Vis spectra were recorded on SHIMADZU UV-2600 spectrometers. The monomer samples were prepared for testing in quartz cuvettes with a light path of 10 mm. The scanning speed was 500 nm/min.
Fluorescence spectra
The fluorescence spectra were recorded on a Hitachi F-4600 fluorescence spectrophotometer. The solution samples were measured in quartz cuvettes with a light path of 10 mm, while gel samples were measured in quartz cuvettes with a light path of 0.1 mm. The testing voltage was 400 V, the width of the excitation and emission slits was 5 nm, the scanning speed was 1200 nm/min. Fluorescence quantum yields were measured by absolute method on a FluoroMax+ (HORIBA) instrument by using an integrating sphere. Fluorescence lifetime curves were recorded on FLS 980 (Edinburgh Instruments).
Circular dichroism (CD) spectra
CD spectra were measured with a JASCO 1500 spectrophotometer. The solution samples were measured in quartz cuvettes with a light path of 10 mm, while gel samples were measured in quartz cuvettes with a light path of 0.1 mm. The scanning wavelength range was from 200 to 700 nm, and the scanning speed was 500 nm/min.
Circularly polarized luminescence (CPL) spectra
CPL spectra were measured with a JASCO CPL-300 spectrophotometer, the gel sample was prepared for testing in quartz cuvettes with a light path of 0.1 mm, and the scanning speed was 500 nm/min. The cuvette samples were tested in different directions and angles to ensure the reliability of the CPL signals.
NMR and mass spectra
1H-NMR, 13C NMR, COSY, and NOESY spectra were recorded on Bruker ADVANCE III 400 and 700 (1H: 400 MHz and 700 MHz, 13C: 700 MHz) spectrometers. Mass spectrum data were obtained by using a BIFLEIII matrix-assisted laser desorption/ionization-time of flight mass spectrometry (MALDI-FTICR-MS) instrument.
Scanning electron microscopy (SEM)
SEM images were recorded on Hitachi S-4800 with an accelerating voltage of 10 kV and a working current of 10 μA. All samples for morphology measurement were prepared on a single-crystal silicon wafer, dried under vacuum, and coated with a thin layer of Pt to increase the contrast.
Atomic force microscope (AFM)
The atomic force microscopy image was recorded on a Dimension FastScan (Bruker Nano) instrument with a silicon tip on silicon nitride cantilevers in the ScanAsyst-Air mode under ambient conditions (115 μm length with typical resonant frequencies of 70 kHz and spring constant of 0.4 N·m−1).
Transmission electron microscopy (TEM)
Cryo-TEM was performed on a Themis300 (Thermo Scientific) instrument with an accelerating voltage of 300 kV.
Fourier transform infrared (FT-IR)
FT-IR spectra were recorded on a VERTEX 70 v (Bruker) FT-IR instrument. All gel samples were dried and further dispersed in a KBr pellet and submitted for FT-IR spectra measurement.
X-ray diffraction (XRD) measurements
PXRD analysis was performed on a Rigaku D/Max-2500 X-ray diffractometer with Cu/Kα radiation (λ = 1.5406 Å), which was operated at a voltage of 40 kV and a current of 200 mA. Samples were prepared on single-crystal silicon wafers and vacuum-dried for PXRD testing.
2D grazing incident wide-angle X-ray scattering (2D-GIWAXS)
2D-GIWAXS was recorded on Xeuss 2.0 equipment (Xenocs, France) with a sample-to-detector distance of 134 mm and an X-ray radiation wavelength of 1.5418 Å. The NU gel sample was wrapped with polyimide film for in-situ testing, and the background of the polyimide film was deducted. The final data were background-calibrated and fitted using SasView software. 1-D intensity curves could be generated by integrating the background-calibrated 2D-GIWAXS patterns.
Density functional theory (DFT) computation
DFT and time-dependent DFT calculations were performed by the Gaussian 09 program at B3LYP 6-31 G* level.
Molecular dynamic (MD) simulation
The molecular dynamic (MD) simulation was implemented with the GROMACS 2023.2 program and the General Amber Force-Field. These original structures were firstly geometry optimized using Gaussian 16. Rosette with a pre-set of 15 Å d-spacings was located in a hexagonal stacking in a box filled with DMF and water (fw = 70%). The cut-off distance for non-bonded interactions was set as 1.0 nm. The steepest descent mode was used for the energy minimization process. Thousands of steps of energy minimization using the steepest descent algorithm and NPT ensemble were conducted before the molecular dynamic simulations. MD simulations for all systems were carried out for 40 ns with a time step of 1 fs per integration step under the ensemble conditions of T = 298 K.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All relevant data generated during and/or analyzed during the current study are available from the corresponding author. Supplementary Information is available in the online version of the paper. Source data are provided with this paper.
References
Ruzmaikin, A., Sokoloff, D. & Shukurov, A. Magnetism of spiral galaxies. Nature 336, 341–347 (1988).
Prive, G. G. et al. Helix geometry, hydration, and G·A mismatch in a B-DNA decamer. Science 238, 498–504 (1987).
Ferry, G. The structure of DNA. Nature 575, 35–36 (2019).
Choi, H.-K. et al. Watching helical membrane proteins fold reveals a common N-to-C-terminal folding pathway. Science 366, 1150–1156 (2019).
Shen, H. et al. De novo design of self-assembling helical protein filaments. Science 362, 705–709 (2018).
Yashima, E. et al. Supramolecular helical systems: helical assemblies of small molecules, foldamers, and polymers with chiral amplification and their functions. Chem. Rev. 116, 13752–13990 (2016).
Su, L. et al. Dilution-induced gel-sol-gel-sol transitions by competitive supramolecular pathways in water. Science 377, 213–218 (2022).
Slęczkowski, M. L., Mabesoone, M. F. J., Slęczkowski, P., Palmans, A. R. A. & Meijer, E. W. Competition between chiral solvents and chiral monomers in the helical bias of supramolecular polymers. Nat. Chem. 13, 200–207 (2021).
Du, C., Li, Z., Zhu, X., Ouyang, G. & Liu, M. Hierarchically self-assembled homochiral helical microtoroids. Nat. Nanotechnol. 17, 1294–1302 (2022).
Liu, J., Zhou, H., Yang, W. & Ariga, K. Soft nanoarchitectonics for enantioselective biosensing. Acc. Chem. Res. 53, 644–653 (2020).
Xu, L. et al. Enantiomer-dependent immunological response to chiral nanoparticles. Nature 601, 366–373 (2022).
Banerjee-Ghosh, K. et al. Separation of enantiomers by their enantiospecific interaction with achiral magnetic substrates. Science 360, 1331–1334 (2018).
Yuan, C. et al. Hierarchically oriented organization in supramolecular peptide crystals. Nat. Rev. Chem. 3, 567–588 (2019).
De, S. et al. Designing cooperatively folded abiotic uni- and multimolecular helix bundles. Nat. Chem. 10, 51–57 (2018).
Gan, Q. et al. Helix-rod host-guest complexes with shuttling rates much faster than disassembly. Science 331, 1172–1175 (2011).
Jones, C. D. et al. Braiding, branching and chiral amplification of nanofibres in supramolecular gels. Nat. Chem. 11, 375–381 (2019).
Sun, B. et al. Homochiral porous nanosheets for enantiomer sieving. Nat. Mater. 17, 599–604 (2018).
Clarke, D. E., Wu, G., Wu, C. & Scherman, O. A. Host–guest induced peptide folding with sequence-specific structural chirality. J. Am. Chem. Soc. 143, 6323–6327 (2021).
Tantakitti, F. et al. Energy landscapes and functions of supramolecular systems. Nat. Mater. 15, 469–476 (2016).
Zhao, D., van Leeuwen, T., Cheng, J. & Feringa, B. L. Dynamic control of chirality and self-assembly of double-stranded helicates with light. Nat. Chem. 9, 250–256 (2017).
Wang, Y., Niu, D., Ouyang, G. & Liu, M. Double helical π-aggregate nanoarchitectonics for amplified circularly polarized luminescence. Nat. Commun. 13, 1710 (2022).
Lehn, J. M. et al. Spontaneous assembly of double-stranded helicates from oligobipyridine ligands and copper(I) cations: structure of an inorganic double helix. Proc. Natl Acad. Sci. USA 84, 2565–2569 (1987).
Nitschke, J. R. Construction, substitution, and sorting of metallo-organic structures via subcomponent self-assembly. Acc. Chem. Res. 40, 103–112 (2007).
Piguet, C., Bernardinelli, G. & Hopfgartner, G. Helicates as versatile supramolecular complexes. Chem. Rev. 97, 2005–2062 (1997).
Zhao, Y. et al. Supramolecular adhesive hydrogels for tissue engineering applications. Chem. Rev. 122, 5604–5640 (2022).
Ayme, J.-F. et al. A synthetic molecular pentafoil knot. Nat. Chem. 4, 15–20 (2012).
Miwa, K., Furusho, Y. & Yashima, E. Ion-triggered spring-like motion of a double helicate accompanied by anisotropic twisting. Nat. Chem. 2, 444–449 (2010).
Howson, S. E. et al. Optically pure, water-stable metallo-helical ‘flexicate’ assemblies with antibiotic activity. Nat. Chem. 4, 31–36 (2012).
Ariga, K., Mori, T., Kitao, T. & Uemura, T. Supramolecular chiral nanoarchitectonics. Adv. Mater. 32, 1905657 (2020).
Hirschberg, J. H. K. K. et al. Helical self-assembled polymers from cooperative stacking of hydrogen-bonded pairs. Nature 407, 167–170 (2000).
Li, Y. et al. Double-helical assembly of heterodimeric nanoclusters into supercrystals. Nature 594, 380–384 (2021).
Hasell, T. et al. Triply interlocked covalent organic cages. Nat. Chem. 2, 750–755 (2010).
Wood, C. S., Ronson, T. K., Belenguer, A. M., Holstein, J. J. & Nitschke, J. R. Two-stage directed self-assembly of a cyclic [3]catenane. Nat. Chem. 7, 354–358 (2015).
Yao, G. et al. Meta-DNA structures. Nat. Chem. 12, 1067–1075 (2020).
Lu, J. et al. Enhanced optical asymmetry in supramolecular chiroplasmonic assemblies with long-range order. Science 371, 1368–1374 (2021).
Berl, V., Huc, I., Khoury, R. G., Krische, M. J. & Lehn, J.-M. Interconversion of single and double helices formed from synthetic molecular strands. Nature 407, 720–723 (2000).
Tanaka, K., Tengeiji, A., Kato, T., Toyama, N. & Shionoya, M. A discrete self-assembled metal array in artificial DNA. Science 299, 1212–1213 (2003).
Vreekamp, R. H., van Duynhoven, J. P. M., Hubert, M., Verboom, W. & Reinhoudt, D. N. Molecular boxes based on calix[4]arene double rosettes. Angew. Chem. Int. Ed. 35, 1215–1218 (1996).
Johnson, R. S., Yamazaki, T., Kovalenko, A. & Fenniri, H. Molecular basis for water-promoted supramolecular chirality inversion in helical rosette nanotubes. J. Am. Chem. Soc. 129, 5735–5743 (2007).
Datta, S. et al. Self-assembled poly-catenanes from supramolecular toroidal building blocks. Nature 583, 400–405 (2020).
Huang, Z. et al. Pulsating tubules from noncovalent macrocycles. Science 337, 1521–1526 (2012).
Aratsu, K. et al. Supramolecular copolymerization driven by integrative self-sorting of hydrogen-bonded rosettes. Nat. Commun. 11, 1623 (2020).
Yagai, S. et al. Supramolecular engineering of oligothiophene nanorods without insulators: hierarchical association of rosettes and photovoltaic properties. Chem. Eur. J. 20, 16128–16137 (2014).
Jonkheijm, P. et al. π-conjugated oligo-(p-phenylenevinylene) rosettes and their tubular self-assembly. Angew. Chem. Int. Ed. 43, 74–78 (2004).
Ji, L. et al. Cooperative chirality and sequential energy transfer in a supramolecular light-harvesting nanotube. Angew. Chem. Int. Ed. 58, 844–848 (2019).
Wade, J. et al. 500-fold amplification of small molecule circularly polarised luminescence through circularly polarised FRET. Angew. Chem. Int. Ed. 60, 222–227 (2021).
Cheng, Q., Hao, A. & Xing, P. A chemosensor-based chiral coassembly with switchable circularly polarized luminescence. Nat. Commun. 12, 6320 (2021).
DuBose, J. T. & Kamat, P. V. Directing energy transfer in halide perovskite–chromophore hybrid assemblies. J. Am. Chem. Soc. 143, 19214–19223 (2021).
Frischmann, P. D., Mahata, K. & Würthner, F. Powering the future of molecular artificial photosynthesis with light-harvesting metallosupramolecular dye assemblies. Chem. Soc. Rev. 42, 1847–1870 (2013).
Huang, Z. et al. Light-responsive circularly polarized luminescence polymers with INHIBIT logic function. Adv. Opt. Mater. 9, 2100135 (2021).
Li, M., Wang, M.-Y., Wang, Y.-F., Feng, L. & Chen, C.-F. High-efficiency circularly polarized electroluminescence from TADF-sensitized fluorescent enantiomers. Angew. Chem. Int. Ed. 60, 20728–20733 (2021).
Lian, Z. et al. [2,2] Paracyclophanes-based double helicates for constructing artificial light-harvesting systems and white LED device. Nat. Commun. 14, 2752 (2023).
Wu, Y. et al. Circularly polarized fluorescence resonance energy transfer (C-FRET) for efficient chirality transmission within an intermolecular system. Angew. Chem. Int. Ed. 60, 24549–24557 (2021).
Yao, K. et al. Ultrastrong red circularly polarized luminescence promoted from chiral transfer and intermolecular Förster resonance energy transfer in ternary chiral emissive nematic liquid crystals. J. Phys. Chem. Lett. 12, 598–603 (2021).
Qin, X., Suga, M., Kuang, T. & Shen, J.-R. Structural basis for energy transfer pathways in the plant PSI-LHCI supercomplex. Science 348, 989–995 (2015).
Chen, X.-M. et al. Self-assembled supramolecular artificial light-harvesting nanosystems: construction, modulation, and applications. Nanoscale Adv. 5, 1830–1852 (2023).
Chen, P.-Z. et al. Light-harvesting systems based on organic nanocrystals to mimic chlorosomes. Angew. Chem. Int. Ed. 55, 2759–2763 (2016).
Yuan, Y.-X. et al. Fluorescent TPE macrocycle relayed light-harvesting system for bright customized-color circularly polarized luminescence. J. Am. Chem. Soc. 144, 5389–5399 (2020).
Chen, X.-M. et al. An efficient near-infrared emissive artificial supramolecular light-harvesting system for imaging in the Golgi apparatus. Angew. Chem. Int. Ed. 59, 10493–10497 (2020).
Teng, K.-X., An, Z.-P., Niu, L.-Y. & Yang, Q.-Z. A supramolecular artificial light-harvesting system with excitation energy and electron transfer. ACS Mater. Lett. 6, 290–297 (2024).
Rajak, A. & Das, A. Crystallization-driven controlled two-dimensional (2D) assemblies from chromophore-appended poly(L-lactide)s: highly efficient energy transfer on a 2D surface. Angew. Chem. Int. Ed. 61, e202116572 (2022).
Rajak, A. & Das, A. Cascade energy transfer and white-light emission in chirality-controlled crystallization-driven two-dimensional co-assemblies from donor and acceptor dye-conjugated polylactides. Angew. Chem. Int. Ed. 62, e202314290 (2023).
Goto, H., Furusho, Y. & Yashima, E. Double helical oligoresorcinols specifically recognize oligosaccharides via heteroduplex formation through noncovalent interactions in water. J. Am. Chem. Soc. 129, 9168–9174 (2007).
Gan, Q. et al. Quadruple and double helices of 8-fluoroquinoline oligoamides. Angew. Chem. Int. Ed. 47, 1715–1718 (2008).
Kato, K. et al. Double-helix supramolecular nanofibers assembled from negatively curved nanographenes. J. Am. Chem. Soc. 143, 5465–5469 (2021).
Mamula, O., Zelewsky, Avon, Bark, T. & Bernardinelli, G. Stereoselective synthesis of coordination compounds: self-assembly of a polymeric double helix with controlled chirality. Angew. Chem. Int. Ed. 38, 2945–2948 (1999).
Samanta, A. et al. Supramolecular double-helix formation by diastereoisomeric conformations of configurationally enantiomeric macrocycles. J. Am. Chem. Soc. 138, 14469–14480 (2016).
Li, F., Li, X., Wang, Y. & Zhang, X. Trismaleimide dendrimers: helix-to-superhelix supramolecular transition accompanied by white-light emission. Angew. Chem. Int. Ed. 58, 17994–18002 (2019).
Scholes, G. D., Fleming, G. R., Olaya-Castro, A. & Grondelle, R. V. Lessons from nature about solar light harvesting. Nat. Chem. 3, 763–774 (2011).
Marsh, D. Lateral order in gel, subgel and crystalline phases of lipid membranes: wide-angle X-ray scattering. Chem. Phys. Lipids 165, 59–76 (2012).
Young, R. M. & Wasielewski, M. R. Mixed electronic states in molecular dimers: connecting singlet fission, excimer formation, and symmetry-breaking charge transfer. Acc. Chem. Res. 53, 1957–1968 (2020).
Li, Z. et al. Photoresponsive supramolecular coordination polyelectrolyte as smart anticounterfeiting inks. Nat. Commun. 12, 1363 (2021).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (22322207 and 92356307 for G.O., 52321006 for M.L.), the National Key Research and Development Program of China (2022YFA1204402 for M.L. and G.O.), the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB0520201 for M.L.) and Beijing Nova Program (20220484071 and 20230484423 for G.O.).
Author information
Authors and Affiliations
Contributions
M.L. conceived the idea and supervised the project. G.O. and T.L. designed the experiments, T.L. carried out most of the experiments, and D.N. made part of the experiments. G.O. and L.J. made the computational simulation, and Q.L., B.G., and H.W. helped with measurements and discussions. G.O. wrote the manuscript with inputs from T.L., and M.L. revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Takeharu Haino and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, T., Niu, D., Ji, L. et al. Supramolecular rosette intermediated homochiral double helix. Nat Commun 16, 1698 (2025). https://doi.org/10.1038/s41467-025-57059-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-57059-3
This article is cited by
-
Dynamic regulation of hydrogen bond-induced fluorescent chiral MOFs with photoexcited activity
Science China Chemistry (2025)