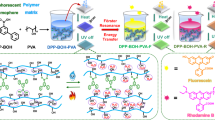
Abstract
Producing room temperature phosphorescent (RTP) materials from biomass resources using a solvent free method is essential but hard to achieve. Here, we discovered that lignin dissolved well in the liquid monomer, 2-hydroxyethyl acrylate (HEA), due to extensive hydrogen bonding and non-bonding interactions between lignin and HEA. Motivated by this discovery, we developed a solvent free system consisting of HEA and urethane dimethacrylate (UDMA) for converting lignin into RTP materials. With this design, lignin generated radicals upon UV irradiation, which initiated the polymerization of HEA (as monomer) and UDMA (as crosslinker). The as-obtained polymer network rigidifies lignin and activates the humidity/water-resistant RTP of lignin with a lifetime of 202.9 ms. Moreover, the afterglow color was successfully tuned to red after loading with RhB via energy transfer (TS-FRET). Using these properties, the as-developed material was used as photocured multiple-emission RTP inks, luminescent coatings and a smart anti-counterfeiting logo for a medicine bottle.
Similar content being viewed by others
Introduction
Lignin, the most abundant natural aromatic polymer, is produced as a byproduct in the pulping and paper industry. Where ~60–70 Mt of lignin is produced per year and ~95% is simply burnt1,2,3,4,5. As a result, utilizing technical lignin in a value-added manner represents a challenging global hot topic. From a chemical perspective, lignin is created through an enzyme-mediated dehydrogenative polymerization of these units including coumaryl alcohol, coniferyl alcohol and sinapyl alcohol6,7. The G, S and H units endow lignin with interesting optical properties, such as, UV blocking, fluorescence, photothermal conversion and room temperature phosphorescence (RTP)8,9,10,11,12,13,14.
Notably, RTP materials exhibit great potential for bioimaging, organic light-emitting diodes, X-ray scintillators and anti-counterfeiting applications15,16,17,18,19,20,21,22,23,24,25,26. Thus, converting lignin to RTP materials is a promising approach for creating value-added applications using lignin. The general method for processing lignin into RTP materials is to rigidify lignin using an external polymer matrix. As such, the triplet excitons of lignin are protected from quenching by external oxygen and humidity and can migrate to the ground state radiatively, which results in RTP emission. Using this strategy, valuable RTP fibers, films, papers and 3D printable inks can be obtained from lignin27,28,29,30,31,32,33.
Nevertheless, aqueous or ionic liquid solvent is required for processing lignin into RTP materials. Generally, to avoid compromising the RTP performance, aqueous solvents should be removed from the obtained RTP materials via evaporation at the cost of energy and time10,31,34 (Fig. 1). Notably, ionic liquids can be incorporated with the RTP materials because they do not interfere with the RTP performance. Thus, ionic liquid could be kept in the as-prepared RTP materials unlike other kinds of solvent., but the high price and possible biological toxicity hinders the practical applications of such RTP materials27,35. Moreover, RTP emission of the as-obtained materials via solvent processing can be easily quenched by humidity or water. Therefore, the ideal processing of lignin into robust RTP materials requires a solvent free system.
(Top) RTP materials from lignin via a solvent-processing method and (bottom) our solvent free method.
Motivated by this, we developed a solvent-free system consisting of HEA and UDMA for processing lignin into RTP materials (Fig. 1). With this design, lignin dissolved well in the HEA. Thus, no external solvent was required. Lignin generated radicals upon UV irradiation, which initiates the polymerization of HEA (as monomer) and UDMA (as crosslinker). Significantly, the as-obtained polymer network rigidifies lignin and activates RTP.
Results
The interaction between lignin and HEA
A homogeneous solution was obtained when the concentration of lignin increased from ~0.1 wt% to ~15 wt% (Fig. 2a and Supplementary Fig. S1) in HEA. To check the generality, a variety of technical lignin including alkaline lignin (AL) and sodium lignosulfonate (SL), were also explored and they both dissolved well in HEA (Supplementary Fig. S2). To understand the interaction, as a control liquid monomer, ethylene glycol and ethyl acrylate, were used. Lignin dissolved well in ethylene glycol but exhibited poor solubility in ethyl acrylate (Supplementary Fig. S3). The result indicated that the -OH group was crucial for dissolving lignin in HEA.
a Photographs of homogeneous solution of lignin dissolved in HEA at different concentrations, (the concentrations from left to right were 0.1 wt%, 0.2 wt%, 0.3 wt%, 0.4 wt% and 0.5 wt%). b 2D COS synchronous (left) and asynchronous (right) spectra generated from Supplementary Fig. S4. (In 2D COS spectra, the warm color (red) represents positive intensities, while cold color (blue) represents negative intensities). c Calculated interactions between lignin and HEA (up) and UDMA (down).
Additionally, FT–IR analysis exhibits intense and broad signals at ~3415 cm−1 (belonging to the O–H stretching vibration) with increased concentration of lignin, indicating increased hydrogen bonding between lignin and HEA (Supplementary Fig. S4)36. 2D COS spectra were further generated from Supplementary Fig. S4 to discern the sequential response for different O-H species. According to the Noda’s judging rule, both signals at ~3580 cm−1 and ~3210 cm−1 responded earlier than the signal at ~3415 cm−1 according to the synchronous and asynchronous spectra upon increasing the concentration of lignin (Fig. 2b). These results indicate that the intermolecular hydrogen bonds between lignin and HEA formed and became more intense for concentrated lignin. Significantly, such interactions are particularly beneficial for dissolving lignin in HEA. Theoretical calculations also indicated that lignin interacts strongly with HEA with an intermolecular binding energy value of −60.62 eV. While the intermolecular binding energy value between lignin and UDMA was −49.31 eV, apparently weaker than the former (Fig. 2c and Supplementary Table S1). These calculations agreed well with the experimental results. Since, the experimental results indicated that lignin dissolved well in HEA, but was poorly soluble in UDMA (Supplementary Fig. S5).
To further determine the interaction between lignin and HEA upon UV irradiation, which was crucial for the preparation of RTP materials in the next step, 2D HSQC NMR analysis of the reaction systems consisting of lignin and HEA was conducted before and after UV irradiation (Supplementary Fig. S6). Both the typical signals of lignin and polymerized HEA (PHEA) were observed from 2D HSQC NMR spectra, suggesting the polymerization of HEA occurred in the presence of lignin after UV irradiation. To further understand the reaction, 1H NMR analysis of the reaction system was then conducted before and after UV irradiation (Supplementary Fig. S7). The result indicated that the signals of the double bond was decreased, indicating that the polymerization of HEA had occurred. Moreover, a new signal, assigned as H of –O–CH2– (located at 5.3 ppm), appeared. Meanwhile, the 31P NMR suggested that the signals of phenolic hydroxyl moieties from lignin almost disappeared after the reaction (Supplementary Fig. S8). This result suggested that phenolic moieties of lignin reacted with the double bond of HEA during the reaction and polymerization of HEA. Further FT-IR analysis also confirmed the reaction. Signals at 1242 cm−1, assigned as –O–CH2– increased after reaction (Supplementary Fig. S9)37. Additionally, the GPC trace also confirmed that the as-obtained products exhibited higher molecular weight than raw lignin, indicating chemical reaction had occurred between the lignin and the polymerized HEA (Supplementary Fig. S10). All these results indicated that HEA could be polymerized and linked with lignin via –O–CH2– bonds upon UV irradiation.
Preparation and RTP properties of Lig-Poly
To obtain RTP materials, lignin was dissolved in a mixture of HEA and UDMA with a ratio of 0.1 wt% and 3:7 (w/w). Upon UV irradiation the generated OH photoradicals of lignin, as determined by ESR spectroscopy, ~80% double bond conversion was achieved (Fig. 3a and Supplementary Fig. S11)38,39. This method was then used to prepare Lig-Poly. The mechanical performance of Lig-Poly was then investigated. Lig-Poly exhibited a tensile strength of 52.8 MPa and elongation at break of 9.8% (Supplementary Fig. S12). As a comparison, cured samples (Poly) in the absence of lignin exhibited 34.7 MPa and 9.4%. These results indicate that lignin is beneficial for the mechanical enhancement of the polymers. Further dynamic mechanical analysis (DMA) of Lig-Poly indicated that the glass transition temperature was 68 oC, confirming that it can be thermally processed when the temperature is above 68 °C (Supplementary Fig. S13). As a control, no photocuring was observed when DMPO a radical scavenger was added into the reaction mixtures (Supplementary Fig. S14). Also, the formula in the absence of lignin was not cured upon UV excitation (Supplementary Fig. S15). All these results confirm photoradical-induced polymerization catalyzed by lignin. After that, the mechanical performance of the cured sample was measured. The hardness and Young’s modulus of the sample were 203.57 MPa and 3.93 GPa, respectively (Supplementary Fig. S16).
a Double bond conversion of Lig-Poly during polymerization, inset: images of Lig-Poly before (left) and after (right) irradiation with UV light. b RTP lifetimes of Lig-Poly with different irradiation times during polymerization, inset: images of Lig-Poly in daylight (left), under excitation of UV light (middle) and after turning off the UV light (right). c RTP lifetimes of Lig-Poly under different humidity (red line), inset: images of Lig-Poly soaking in water in daylight (left), under the excitation of UV light (middle) and after turning off the UV light (right). (Conditions for the images: The samples were immersed into water for 24 h. After that, the digital images of the sample were taken under bright field, UV field and after switching off the UV light sources). d RTP lifetimes of Lig-Poly before (gray) and after (colored) when soaked in different solvents. e Fluorescence (black line) and RTP emission (red line) spectra of Lig-Poly/RhB excited by 320 nm light, inset: images of Lig-Poly/RhB in daylight (left), under excitation of UV light (middle) and after turning off the UV light (right). f RTP decay curve of Lig-Poly/RhB.
Interestingly, exposure of the precursors to UV light sources led to an enhancement of both the RTP intensity and lifetime (Fig. 3b and Supplementary Fig. S17). After 30 min, the RTP performance of Lig-Poly reached equilibrium and exhibited long-lasting emission for 202.9 ms (Supplementary Fig. S18). As a control, only very weak RTP emission, attributed to the clustering-induced emission, was observed for the Poly sample in the absence of lignin (Supplementary Fig. S19). This result illustrates that lignin was the main chromophore of Lig-Poly. To understand the RTP emission from Lig-Poly, theoretical calculations were conducted. The result indicted that lignin exhibits stronger interactions with the polymerized network than the monomers (Supplementary Fig. S20 and Supplementary Table S1). Such interactions restrict the molecular motion, resulting in RTP emission40,41,42. Additionally, mechanical analysis indicated that increasing the exposure time of the formula with UV light sources led to an enhanced tensile strengthen and reduced strain ratios (Supplementary Fig. S21). This result indicates that the rigidity of the as-formed Lig-Poly increased with extended photocuring. Thus, exposure of the precursors to UV light sources led to an increased polymerization degree. The enhanced polymerization degree induced a higher crosslinking density, contributing to a rigidified environment and promotion of the RTP performance of Lig-Poly43,44. This helps explain the enhanced intensity and lifetime of Lig-Poly with an extended curing time. Notably, the RTP performance of Lig-Poly was dependent on the concentration of lignin. Increasing the concentration of lignin from 0.01% to 0.1% enhanced the RTP performance (Supplementary Fig. S22). However, further increasing the concentration of lignin decreased the RTP emission and lifetime because of the π–π stacking of aromatic units in lignin. Additionally, the samples were prepared using different types of lignin including sodium lignosulfonate (SL) and alkaline lignin (AL) and all these materials exhibited RTP emission, suggesting the generalization of the method (Supplementary Fig. S23). Moreover, all the samples prepared using different crosslinkers and HEA in the presence of lignin exhibited RTP emission, further indicating the generalization of the strategy (Supplementary Fig. S24).
Lig-Poly exhibited humidity/water-resistant RTP emission, which has rarely been observed for similar systems. Specifically, the RTP lifetime did not decrease when it was treated with 90% humidity or immersed in water for 24 h (Fig. 3c). To further understand the phenomenon, the surface tension and contact angle of Lig-Poly were measured (Supplementary Fig. S25). The values of surface tension and contact angle were 65.53 mN/m and 41.6°, respectively. Interestingly, the surface of Lig-Poly exhibited hydrophilicity rather than hydrophobicity. This was because the water molecules form hydrogen bonds with the surface hydroxyl moieties from the polymerized HEA of Lig-Poly45. Such interactions induced formation of dense molecular networks preventing the water entering the network. As a result, the triplet excitons were well protected in the Lig-Poly and RTP emission was stable when the sample was immersed into water.
The stability of Lig-Poly was further investigated in organic solvents. The results indicated that the RTP lifetime of Lig-Poly was not compromised when it was immersed in different organic solvents, such as, ethanol, methanol, ethyl acetate, tetrahydrofuran, dichloromethane and acetonitrile for 24 h (Fig. 3d). However, the RTP of Lig-Poly was sensitive to temperature. When the temperature decreased, both the intensity and lifetime of Lig-Poly increased, attributed to enhanced radiative migration of triplet excitons (Supplementary Fig. S26).
Notably, spectral analysis revealed a significant overlap between the RTP emission band of Lig-Poly and the absorption spectrum of RhB (Supplementary Fig. S27), suggesting the potential for efficient energy transfer between these two components. As such we successfully incorporated RhB into the Lig-Poly matrix to construct a novel Lig-Poly/RhB composite system. Remarkably, the resulting Lig-Poly/RhB complex demonstrated distinct red afterglow emission properties, with a measured lifetime of 167.7 ms, as clearly illustrated in Fig. 3e and f. To further understand the energy transfer, the lifetime of Lig-Poly before and after loading with RhB was measured. Time-resolved photoluminescence analysis of lignin phosphorescence at 500 nm (λex = 320 nm) revealed a significant reduction in the average lifetime from 203.2 ms to 141.5 ms upon incorporation of RhB into Lig-Poly (Supplementary Fig. S28). This lifetime reduction provides compelling evidence for non-radiative Förster resonance energy transfer (FRET) from the triplet state of lignin donors to the singlet state of RhB acceptors in the Lig-Poly/RhB system, specifically through a triplet-to-singlet FRET (TS-FRET) mechanism. The observed lifetime change effectively rules out the possibility of a simple emission-reabsorption process, since for that the donor lifetime would not change, which is consistent with previous reports31,46,47. Further evidence supporting the TS-FRET mechanism was obtained through selective excitation experiments. When Lig-Poly/RhB was excited at 500 nm (acceptor excitation), no emission was detected in the time-gated spectra. In contrast, distinct emission was observed upon excitation at 320 nm (donor excitation) (Supplementary Fig. S28). These results unequivocally confirm that the long-lived triplet excitons of lignin donors serve as the exclusive source for populating the singlet state of RhB acceptors through an efficient TS-FRET process, generating persistent delayed fluorescence. Notably, the energy transfer efficiency between lignin and RhB in the Lig-Poly matrix exhibited a strong dependence on RhB concentration, suggesting the potential for tunable photophysical properties in this system. The energy transfer efficiency increased from 0% to 30.4% when the concentration ratio of RhB to lignin increased from 0:1 to 0.04:1 (w/w) (Supplementary Table S2).
Applications
Using the photocuring of Lig-Poly different shapes were easily obtained using a template and the in situ exposure of the precursors to UV light sources (Fig. 4a). In addition, 3D materials with red phosphorescence were prepared by exposure of the precursors of Lig-Poly/RhB to UV light (Fig. 4b). Additionally, Lig-Poly and Lig-Poly/RhB were easily combined to generate shapes with two colors of phosphorescence (Fig. 4c). Attributed to the inherent properties of the photocured materials, Lig-Poly/RhB can also be used for anti-counterfeiting coatings (Fig. 4d). To further demonstrate the potential, Lig-Poly/RhB was processed into a QR code and tag, which can be used on a medicine bottle for anti-counterfeiting purposes. All these systems mentioned above-exhibited afterglow RTP emission after removing the excitation sources. Moreover, the Lig-Poly was used as a photo-cured coating for twine (Fig. 4e). The obtained RTP twines were then processed into different shapes. Using thermal processing, Lig-Poly and Lig-Poly/RhB were easily processed into different shapes with RTP emission (Supplementary Fig. S29). Interestingly, Lig-Poly and Lig-Poly/RhB returned to their initial shapes with RTP emission due to inherent shape-memory properties. The whole process was reversible several times. The effect of thermal processing on RTP performance of Lig-Poly and Lig-Poly/RhB was quantitively determined. Significantly, both the RTP intensity and lifetime of Lig-Poly and Lig-Poly/RhB were not compromised after thermal processing. Moreover, they did not significantly change after being recycled 5 times by thermal processing (Supplementary Fig. S30).
a Images of Lig-Poly in daylight (left), Lig-Poly upon excitation of UV light (middle) and Lig-Poly after switching off the UV light (right) (scale bar = 5 mm). b Images of Lig-Poly/RhB in daylight (left), Lig-Poly/RhB upon excitation of UV light (middle) and Lig-Poly/RhB after switching off the UV light (right) (scale bar = 5 mm). c Images of Lig-Poly and Lig-Poly/RhB in daylight (left), Lig-Poly and Lig-Poly/RhB upon excitation of UV light (middle) and Lig-Poly and Lig-Poly/RhB after switching off the UV light (right) (scale bar = 5 mm). d Image of a medicine bottle with 2D code on the cap and peony made for the bottle (scale bar = 3 mm). e Images of single twine treated by Lig-Poly and knitting produced using twine treated with Lig-Poly (scale bar = 5 mm).
Discussion
In summary, we have developed a solvent-free RTP system consisting of lignin, HEA and UDMA. The strong hydrogen bonding interaction between HEA and lignin facilitates the dissolution of lignin in a mixture of HEA and UDMA. Then, dissolved lignin generated photoradicals, triggering the polymerization of HEA and UDMA. The as-formed rigid polymer network confined lignin and triggered green RTP emission from lignin with a lifetime of 202.9 ms. Moreover, the afterglow color was successfully converted to red after loading with RhB using a TS-FRET strategy. Using these properties, the solvent-free RTP system was used to produce functional luminescent materials for coatings and anti-counterfeiting applications. Considering the sustainability, convenience, inexpensive and energy saving properties, this research will enable the synthesis of practical RTP materials from renewable resources on a large scale. Moreover, considering the simplicity in fabrication, high transparency and hardness, the as-obtained Lig-Poly exhibited potential to be used as RTP glass, which has great potential in optical displays, wearable devices, and portable optoelectronics48.
Methods
Preparation of Lig-Poly
One milligram of EL (SL or AL) was added in 300 mg HEA and dissolved completely by assistance of ultrasonic, and then 700 mg UDMA (TEGDA, TPGDA or TMPTA) was added into the solution to obtain the homogeneous solution by stirring and ultrasound. After that, the solution was put into mold and exposed under UV light sources (365 nm, 70 mW cm-2) for 20 min for photocuring.
Preparation of Poly
The control sample was prepared as following: 1 mg of AIBN was added in 300 mg HEA and dissolved completely by assistance of ultrasonic, and then 700 mg UDMA was added into the solution to obtain the homogeneous solution by stirring and ultrasound. After that, the solution was put into mold and exposed under UV light sources (365 nm, 70 mW cm-2) for 20 min for photocuring.
Data availability
All relevant data are included in this article and its Supplementary Information file. All data are available from the corresponding author (Zhijun Chen) upon request. Source data are provided with this paper.
References
Ragauskas, A. J. et al. Lignin valorization: improving lignin processing in the biorefinery. Science 344, 1246843 (2014).
Meng, Q. et al. Sustainable production of benzene from lignin. Nat. Commun. 12, 4534 (2021).
Xia, Z. et al. Processing and valorization of cellulose, lignin and lignocellulose using ionic liquids. J. Bioresour. Bioprod. 5, 79–95 (2020).
Zhang, Y. & Naebe, M. Lignin: a review on structure, properties, and applications as a light-colored UV absorber. ACS Sustainable Chem. Eng. 9, 1427–1442 (2021).
Balakshin, M. Y. et al. New opportunities in the valorization of technical lignins. ChemSusChem. 14, 1016–1036 (2021).
Chio, C., Sain, M. & Qin, W. Lignin utilization: a review of lignin depolymerization from various aspects. Renew. Sus. Energ. Rev. 107, 232–249 (2019).
Sun, R. C. Lignin source and structural characterization. ChemSusChem 13, 4385–4393 (2020).
Wan, K. et al. Structural materials with afterglow room temperature phosphorescence activated by lignin oxidation. Nat. Commun. 13, 5508 (2022).
Zhao, X. et al. Melanin-inspired design: preparing sustainable photothermal materials from lignin for energy Generation. ACS Appl. Mater. Interfaces 13, 7600–7607 (2021).
Yuan, J. et al. Sustainable afterglow materials from lignin inspired by wood phosphorescence. Cell Rep. Phys. Sci. 2, 100542 (2021).
Sun, Z. et al. Lignin powered versatile bioelastomer: a universal medium for smart photothermal conversion. Adv. Funct. Mater. 34, 2405130 (2024).
Ma, Z. et al. Seeking brightness from nature: J-aggregation-induced emission in cellulolytic enzyme lignin nanoparticles. ACS Sustain. Chem. Eng. 6, 3169–3175 (2018).
Pan, X. et al. Tough supramolecular hydrogels crafted via lignin-induced self-assembly. Adv. Mater. 36, 2406671 (2024).
Zhao, S. et al. Lignin-based carbon quantum dots with high fluorescence performance prepared by supercritical catalysis and solvothermal treatment for tumor-targeted labeling. Adv. Compos. Hybrid Mater. 6, 73 (2023).
Cao, M. et al. Biobased and biodegradable films exhibiting circularly polarized room temperature phosphorescence. Nat. Commun. 15, 2375 (2024).
Zhou, W.-L. et al. Ultralong purely organic aqueous phosphorescence supramolecular polymer for targeted tumor cell imaging. Nat. Commun. 11, 4655 (2020).
Zhang, Y. et al. Large-area, flexible, transparent, and long-lived polymer-based phosphorescence films. J. Am. Chem. Soc. 143, 13675–13685 (2021).
He, Z. et al. Achieving persistent, efficient, and robust room-temperature phosphorescence from pure organics for versatile applications. Adv. Mater. 31, 1807222 (2019).
Chen, Q. et al. Long lifetimes white afterglow in slightly crosslinked polymer systems. Nat. Commun. 15, 2947 (2024).
Davydova, M. P. et al. Strong magnetically-responsive circularly polarized phosphorescence and X-ray scintillation in ultrarobust Mn(II)-organic helical chains. Adv. Mater. 35, 2303611 (2023).
Zheng, H., Zhang, Z., Cai, S., An, Z. & Huang, W. Enhancing purely organic room temperature phosphorescence via supramolecular self-assembly. Adv. Mater. 36, 2311922 (2024).
Peng, X. et al. Purely organic room-temperature phosphorescence molecule for high-performance non-doped organic light-emitting diodes. Angew. Chem. Int. Ed. 63, e202405418 (2024).
Zhan, L. et al. Organic molecules with dual triplet-harvesting channels enable efficient X-ray scintillation and imaging. Aggregate 5, e485 (2024).
Zhang, J. et al. Stimuli-responsive deep-blue organic ultralong phosphorescence with lifetime over 5 s for reversible water-jet anti-counterfeiting printing. Angew. Chem. Int. Ed. 60, 17094–17101 (2021).
Zhou, J., Li, J., Zhang, K. Y., Liu, S. & Zhao, Q. Phosphorescent iridium(III) complexes as lifetime-based biological sensors for photoluminescence lifetime imaging microscopy. Coordin. Chem. Rev. 453, 214334 (2022).
Wu, P. et al. 3D Printed room temperature phosphorescence materials enabled by edible natural konjac glucomannan. Adv. Mater. 36, 2402666 (2024).
Guo, H. et al. Photocured room temperature phosphorescent materials from lignosulfonate. Nat. Commun. 15, 1590 (2024).
Zhai, Y. et al. Room temperature phosphorescence from natural wood activated by external chloride anion treatment. Nat. Commun. 14, 2614 (2023).
Yang, M. et al. Repurposing lignin to generate functional afterglow paper. Cell Rep. Phys. Sci. 3, 100867 (2022).
Liu, R. et al. Producing a room temperature phosphorescent film from natural wood using a top-down approach. Adv. Funct. Mater. 34, 2312254 (2023).
Cao, M. et al. Producing naturally degradable room-temperature phosphorescent materials by covalently attaching lignin to natural polymers. Cell Rep. Phys. Sci. 5, 101811 (2024).
Yin, W.-M. et al. Producing sustainable room temperature phosphorescent materials using natural wood and sucrose. Cell Rep. Phys. Sci. 5, 101792 (2024).
Zhou, J. et al. Photoactivated room temperature phosphorescence from lignin. Nat. Commun. 15, 7198 (2024).
Nie, F. & Yan, D. Macroscopic assembly of chiral hydrogen-bonded metal-free supramolecular glasses for enhanced color-tunable ultralong room temperature phosphorescence. Angew. Chem. Int. Ed. 62, e202302751 (2023).
Xia, Z. et al. Eco-friendly fractionation of natural straws: sustainable ultralong room-temperature phosphorescence and super anti-ultraviolet materials. Sci. China Chem. 67, 2373–2381 (2024).
Li, Z., Gong, Y., Li, H., Jiang, M. & Dong, L. Hydrogen bond reorganization-enabled dielectric pulsing effects in polar polymers. Adv. Funct. Mater. 34, 2309157 (2023).
Zong, E. et al. Graft polymerization of acrylic monomers onto lignin with CaCl2-H2O2 as initiator: preparation, mechanism, characterization, and application in poly(lactic acid). ACS Sustain. Chem. Eng. 6, 337–348 (2017).
Guo, X. et al. A sustainable wood-based iron photocatalyst for multiple uses with sunlight: water treatment and radical photopolymerization. Angew. Chem. Int. Ed. 62, e202301242 (2023).
Gan, D. et al. Plant-inspired adhesive and tough hydrogel based on Ag-lignin nanoparticles-triggered dynamic redox catechol chemistry. Nat. Commun. 10, 1487 (2019).
Yu, Y. et al. Room-temperature-phosphorescence-based dissolved oxygen detection by core-shell polymer nanoparticles containing metal-free organic phosphors. Angew. Chem. Int. Ed. 56, 16207–16211 (2017).
Kwon, M. S. et al. Suppressing molecular motions for enhanced room-temperature phosphorescence of metal-free organic materials. Nat. Commun. 6, 8947 (2015).
Tian, R., Gao, S., Li, K. & Lu, C. Design of mechanical-robust phosphorescence materials through covalent click reaction. Nat. Commun. 14, 4720 (2023).
Liu, X. W. et al. Photo-thermo-induced room-temperature phosphorescence through solid-state molecular motion. Nat. Commun. 13, 3887 (2022).
Chen, K. et al. Twofold rigidity activates ultralong organic high-temperature phosphorescence. Nat. Commun. 15, 1269 (2024).
Kang, W., Bao, Y., Wang, H., Cao, N. & Cui, S. Force-induced enhancement of hydrophilicity of individual polymethyl methacrylate chain. Chin. J. Chem. 41, 2289–2295 (2023).
Kuila, S. & George, S. J. Phosphorescence energy transfer: ambient afterglow fluorescence from water-processable and purely organic dyes via delayed sensitization. Angew. Chem. Int. Ed. 59, 9393–9397 (2020).
Dang, Q. et al. Room-temperature phosphorescence resonance energy transfer for construction of near-infrared afterglow imaging agents. Adv. Mater. 32, 2006752 (2020).
Nie, F. & Yan, D. Bio-sourced flexible supramolecular glasses for dynamic and full-color phosphorescence. Nat. Commun. 15, 9491 (2024).
Acknowledgements
Z.J.C. wishes to thank the National Natural Science Foundation of China (32471803) and Fundamental Research Funds for the Central Universities (2572022CG02). T.D.J. wishes to thank the University of Bath and the Open Research Fund of the School of Chemistry and Chemical Engineering, Henan Normal University (2020ZD01) for support.
Author information
Authors and Affiliations
Contributions
Conceptualization: Z.C., S. Li., and T.D.J.; Methodology: M.W. and W.Y.; Investigation: M.W., Y.Z., and J.Z.; Visualization: M.W., S. Liu., J.L., and S. Li.; Supervision: Z.C., S. Li., and T.D.J.; Writing-original draft: All authors; Writing-review and editing: All authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, M., Yin, WM., Zhai, Y. et al. Solvent-free processing of lignin into robust room temperature phosphorescent materials. Nat Commun 16, 2455 (2025). https://doi.org/10.1038/s41467-025-57712-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-57712-x
This article is cited by
-
Multiple hydrogen bonding enables large-area doped phosphorescent glasses with robust stability and high-temperature afterglow
Nature Communications (2026)
-
Advancing lignin analytics via elucidation of linkage progressions in lignin populations
Communications Chemistry (2025)
-
Up-recycling of waste wood into value-added room temperature phosphorescent materials
Nature Communications (2025)
-
Lignin-derived room temperature phosphorescent materials
Science China Chemistry (2025)