Abstract
Estimation of net CO2 consumption by weathering in orogen is complicated as high erosion rate promotes competing processes of CO2 consumption (silicate weathering) and releasing (sulfuric acid (H2SO4) dissolution of carbonate). Quantification of H2SO4 disturbing on weathering is missing in the Tibetan Plateau, hindering the understanding of Himalayan orogenesis impact on global carbon cycle. Here we calculate the riverine solute contributions from both carbonic and sulfuric acid mediated weathering, and their weathering fluxes with major river geochemistry dataset from the Tibetan Plateau. We find that silicate weathering is not anomalous, while carbonate weathering flux is 2.09% of the global value with 1.01% drainage area. Over 80% H2SO4 originated from pyrite oxidation is consumed by carbonate weathering, which counteracts ~58% of the CO2 consumption flux by silicate weathering. The refined weathering CO2 budget in this work provides quantitative modern evidence for pyrite weathering in orogen serving as negative feedback on atmospheric pCO2.
Similar content being viewed by others
Introduction
The formation of the Tibetan Plateau exerts profound impacts on Earth's surface environment evolution in the Cenozoic. Particularly, it has been a hotspot for continental weathering studies since the proposing of “uplift-weathering hypothesis”1,2, which ascribes the atmospheric pCO2 drawdown and global cooling during the Cenozoic to the enhanced silicate weathering (SW) and CO2 consumption in the Himalayan-Tibetan orogeny. However, later studies questioned it3,4,5,6 and proposed that the impact of plateau formation on continental weathering and atmospheric pCO2 level was limited7,8,9. Key controversies lie in the negative feedback mechanism of the Uplifting-Weathering Hypothesis. Sulfuric acid (H2SO4) generated in oxidative weathering of pyrite (OWP) serves as a proton provider in carbonate weathering, which has been argued as an important CO2 source counteracting CO2 consumption by SW at different scales recently10,11,12,13,14,15,16. The high erosion rate on the Tibetan Plateau resulted from uplifting, and the largest distribution area of low-altitude glaciers makes it a hotspot for H2SO4 generation by OWP, and thus a CO2 source counteracting CO2 consumption by silicate weathering, which might serve as an important negative feedback mechanism of atmospheric pCO2 in Cenozoic. However, quantitative and systematic evidence, especially for the relative importance of different weathering pathways and the functions of different acid agents, is missing.
Hydro-geochemistry of river water is informative of weathering process, fluxes, and CO2 budgets as it integrates the weathering products over the whole drainage area of the river networks. Regional case studies on basin weathering have been conducted for the Tibetan Plateau, revealing that the solute sources and weathering fluxes of Himalayan–Tibetan river basins vary considerably through the plateau17,18,19,20,21,22. The river solute source identification and quantification for large rivers in the Tibetan Plateau are not well constrained as significant spatial variations exist on both weathering bedrock and weathering acid agents, and systematic datasets to calculate the riverine ion contribution from different weathering reactions are lacking. Investigation of the fluvial geochemistry is needed to gain a quantitative estimation of the chemical weathering and associated CO2 fluxes for the plateau as a whole. A knowledge gap exists when evaluating the effect of H2SO4 on chemical weathering as sulfate contributions are complicated from atmosphere deposition, evaporite, hot spring, and OWP in the plateau23. In particular, OWP-sourced H2SO4, which is a strong competition acid agent with carbonic acid in chemical weathering reactions, has not been quantitively estimated yet, thus obstructing the estimation of its disturbance on the CO2 budget of plateau weathering.
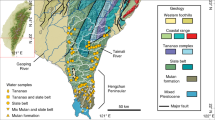
We systematically sampled all the major river networks on the Tibetan Plateau (Fig. 1, detailed in Supplementary Text A), including the main channels and tributaries of Nu Jiang River (upper reaches of Salween River), Yarlung Tsangpo River (upper reaches of Brahmaputra River), the upper Yellow River, the upper Yangtze River and Lantsang Jiang River (upper reaches of Mekong River) in the high flow period. Riverine hydrochemistry and multiple isotope systems (87Sr/86Sr, δ13CDIC, δ34SSO4, and δ18OSO4) have been analyzed to partition riverine solute sources and to estimate the carbonic and sulfuric acid agent supply in chemical weathering reactions of different rock types and their CO2 effects. This work quantifies the net weathering CO2 sequestration fluxes considering the roles of both carbonic and sulfuric acid and reveals the strong modulating effect of the non-carbonic acid on weathering CO2 budget of the Tibetan Plateau.
The panel on the upper left corner shows the location of the studied area, and the main panel presents the major river basins on the Tibetan Plateau and the sampling sites. The blue lines represent the river channels, and the red lines represent the basin boundaries. Solid red dots show the sampling locations and purple triangles noted the hydrological stations. Light blue dots with black center noted the major cities in the studied area.
Results and discussion
Solute geochemistry and weathering source quantification of the Tibetan Plateau rivers
The field parameter, ion content, and isotopic compositions of river waters are tabulated in Table S1. Total dissolved solid (TDS) of large rivers in the Tibetan Plateau averages at 215.8 mg L−1, doubled of the global mean (100 mg L−1, ref. 24). The ionic composition is dominated by Ca2+, Mg2+, and HCO3− (Fig. S1), and the concentrations of which are distinctively higher than the rivers on the southern slope of Himalayas, such as Indus River, Ganges and Brahmaputra river system25,26. The frequency distributions of Sr concentrations and 87Sr/86Sr ratios for the studied rivers and surrounding area are presented in Fig. S2. The plateau rivers are with significantly higher Sr concentration and similar 87Sr/86Sr range (averaging at 3.13 μmol L−1 and 0.7111 for the mainstream samples) compared with the global mean (0.89 μmol L−1 and 0.71253, refs. 24,27), slightly higher than the value range of carbonate (0.708–0.710, refs. 24,28), but significantly lower than the well documented normal silicate endmember value range (0.72–0.73, refs. 28,29,30). The plots of 87Sr/86Sr and cation ratio index in Fig. S3 showed that most samples are distributed around the carbonate dissolution end-members, suggesting a highlighted carbonate-dominated weathering regime on the plateau.
The rivers are characterized with both higher δ13CDIC and lower δ34SSO4 values (averaging at −8.4‰ and 2.6‰, respectively) than the global average (−11.6‰ and 4.4‰; refs. 13,31,32). Meanwhile, the headwaters of the Yellow, Jinsha, Lancang and Nu rivers in the arid interior region of the plateau, are with more positive δ13CDIC and δ34SSO4 values, accompanying with higher Na+, Cl−, Ca2+ and SO42− concentration, which is the typical feature of river water chemistry resulted from halite and gypsum dissolution. The δ34SSO4 shows a generally decreasing trend down the mainstream of the studied rivers (Table S1), suggesting an increasing portion of OWP originated SO42− with a more negative δ34SSO4 (endmember value discussion detailed in the Supporting Text B III section). Among the plateau large rivers, the Yarlung-Tsangpo River flowing in the south has the highest δ13CDIC and lowest δ34SSO4 mean value at −7.1‰ and +0.11‰, respectively, which implicates potential enhancement of OWP originated H2SO4 participation in weathering.
Quantifying the contributions of different riverine solute sources is pre-requisite before deriving chemical weathering rates and associated CO2 consumption at the basin scale24,26,33,34,35,36. A forward model based on mass budget equations of cations from different sources is conducted to quantify the solute contribution (calculation procedures and results detailed in Supplementary Text B). We thoroughly consider the end members in the plateau (atmospheric precipitation, anthropogenic input, carbonate and silicate rock weathering, evaporite dissolution, and sulfide mineral weathering). The results indicate that carbonate weathering (CW) and evaporite dissolution contributions to the riverine solutes are highly variable, with an average portion at 72.8% (ranging from 6.5 to 97.2%) and 8.9% (ranging from 0 to 92.9%) of the total cationic loads in the river systems on the plateau (Table S3). In comparison, the contribution from SW is much lower and less variable. Specifically, 93% of the sampling site has SW contribution of the total cationic loads lower than 30%, with an averaging value at 17.0% (Table S3). Calculated contributions for riverine cations and sulfate from different reservoirs for the main channels are presented in Fig. 2. A new end member of sulfide mineral weathering has been brought to evaluate the sulfuric acid involvement in weathering, which has not been systematically quantified for the plateau as a whole previously. Considering both the highlighted evaporites and OWP input of riverine SO42− in the plateau, it is critical to distinguish their contribution respectively when estimating basin weathering flux and carbon effects, as evaporites dissolution is carbon neutral while OWP generates H2SO4, which dissolves carbonate and releases CO2. Riverine sulfate origins are calculated with SO42− concentration and δ34SSO4 value (Table S3, calculation detailed in the Supporting Text B III). We estimate that about 25.0 to 92.1% (averaging at 56.0%) riverine sulfate originate from OWP, which is 11% higher than the estimation for global river (45%, ref. 12). The higher fpyrite (the fraction of riverine sulfate originated from OWP) values for the studied river systems indicate significant sulfuric acid involvements in the plateau weathering.
a The total cationic loads (the sum of Na+, K+, Ca2+, and Mg2+); b riverine sulfate (SO42−) for the main channels of the large rivers draining in the Tibetan Plateau. The bars represent sampling locations downward the main channels from left to right within a river.
Chemical weathering rates and fluxes of the plateau
The chemical weathering fluxes and rates for individual river basins are calculated with the results of solute source quantification above, the basin area, and the discharge of different hydrology stations on the main channel of individual large rivers studied (Table 1 and Fig. 3)12,24,25,26,28,37,38,39,40,41,42,43,44. The area-averaged chemical weathering rate of the Tibetan Plateau is calculated at 41.7 t km−2 a−1. As the upper reaches of the major rivers draining on the Asian continent, the large river basins in the plateau doubled the average total weathering rates of the global values (21 t km−2 a−1, estimated by ref. 45, and 24 t km−2 a−1 estimated by ref. 24). However, the enhanced weathering rate in the plateau is discriminated for CW and SW.
a The silicate weathering rates (SWR, red bar) and carbonate weathering rates (CWR, blue bar). b The CO2 fluxes of silicate weathering (SW, red bar), carbonate weathering (CW, blue bar), carbonate weathering by sulfuric acid (CW by H2SO4, green bar), and the net value (purple bar). Results and raw data for river downstream: refs. 37,38 for the downstream Huang River; ref. 28 for the Yangtze River; refs. 39,40,41 for the Mekong River, Salween River, and Irrawaddy River; ref. 26 and refs. 42,43 for the Brahmaputra River and Ganges River; ref. 44 for the Red River; refs. 24,25 for Indus river, respectively.
The SW flux of the studied river basins is 0.71% of the global value, and their area percentage is 1% of the global river basin area. The two values are 10.85% and 6.6%, respectively, when the downstream basin area is included (Table 1). The large spatial comparison indicates that the plateau river basins have significantly lower SWR (silicate weathering rate), which is only 21–55% of their lower reaches (Upper Huang, Jinsha, Nu, Lancang and Yarlung Tsangpo River, Table 1 and Fig. 3). Therefore, our calculations show that the Tibetan Plateau does not have an obvious enhancement of SWR as previous thought. Instead, the silicate materials on the plateau probably only experience initial chemical alterations with high erosion rates, relative short water and material residential time there, while longer materials residential time together with the monsoonal climate with higher mean annual temperature (MAT) and precipitation (MAP) downstream would facilitate the chemical weathering of silicate materials in the downward floodplain. We speculate that the uplifting of the plateau acts as a factory of fresh silicate materials by high erosion rates, which constantly supply sediments to the floodplain locating in the prevailing area of Asian monsoon and the enhancement of SW happens in the downstream plain area of large river basins originated from the plateau, instead of on the plateau in-situ.
Different from SW, the calculation shows that the plateau has disproportionately higher CW fluxes (2.09% of the global CW fluxes with 1.0% drainage area, Table 1). The CWR (carbonate weathering rate) of river basins on the plateau are comparable with or even higher than the value of their downstream for Huang, Changjiang and Mekong River (Table 1 and Fig. 3). It is worthwhile to be noted that the Palong Tsangpo river basin (a major tributaries of the Brahmaputra River at the upper reach) with MAT and MAP at 8.6 °C and 871 mm, respectively,46,47 on the eastern syntaxis of the Himalaya has the highest CWR which is comparable with the downstream Brahmaputra River at the Indian plain with much higher MAT and MAP at 17.3 °C and 1543 mm (ref. 48), respectively (47.2 ton km−2 a−1 vs. 47 ton km−2 a−1). We propose that the direct impact of high physical erosion rates on the plateau drives a more significant enhancement on CWR than SWR, while the plateau river basins cannot be considered anomalous in terms of SW in comparison to other major rivers globally. Carbonate and sulfide minerals dissolve up to three orders of magnitude faster than silicate minerals13. The high erosion rate promotes the fresh surface generation of both carbonate and silicate minerals on the plateau basins, but the much more intensely enhanced CWR compared to SWR on the plateau is assumed to be the result of interplays between their different reacting rate with weathering fluids. Considering the different CO2 effects of carbonate and silicate weathering with sulfuric acid, it is obligated to further evaluate the CO2 budget of these weathering processes.
Sulfuric acid participation and its effects on weathering CO2 consumption budgets of the Tibetan Plateau
The Tibetan Plateau is characterized by both the enhanced OWP exposure and sulfuric acid generation due to high erosion rates resulting from tectonic and glacier activity16,49,50, and a disproportionately strengthening of carbonate weathering revealed by river water geochemistry as discussed above. The plateau riverine DIC is dominantly contributed by carbonic and sulfuric acid weathering of carbonate as illustrated in the δ13CDIC and typical ionic ratios (Fig. 4 and Supplementary Text B)16,26,51,52,53,54,55, indicating that OWP-originated sulfuric acid has significantly regulated the riverine DIC source and generating pathway. Strong heterogeneity of sulfuric acid disturbing on chemical weathering and CO2 budget has been observed in previous studies at different catchment scale in the Tibetan Plateau14,16,22. Partitioning the riverine sulfate sources is the premise, but a knowledge gap currently exists in the plateau to estimate the portion of protons originating from OWP and to discriminate cations released by carbonic and sulfuric acid weathering, which obstructs the evaluation of net CO2 consumption fluxes for the plateau weathering as a whole. The flux of sulfate derived from OWP of the river networks on the Tibetan Plateau is calculated at 54.3 × 109 mol a−1 (Table 1), accounting for 4.17% of the global flux of riverine sulfate sourced from OWP (1300 × 109 mol a−1, ref. 12) with a total drainage area of 1.0%. As a region with intense tectonic activity and the widest distribution of low-latitude glaciers, the Tibetan Plateau serves as one of the hotspots for pyrite exposure, which leads to the disproportionately high OWP-originated sulfate flux in the rivers there. Furthermore, we employed the simultaneous equations of δ34SSO4 and solutes contribution from different weathering pathways to quantify sulfuric acid consumed by carbonate and silicate weathering, respectively (Supplementary Text B). The calculations show that over 80% of sulfuric acid from OWP has been consumed by carbonate weathering, and the same amount of CO2 would be released to the atmosphere.
The long-term net CO2 consumption budget for the major river basins on the plateau could be calculated by deducting the CO2-releasing fluxes of H2SO4-carbonate reaction from CO2 consumption fluxes of SW (detailed in the method section). The net CO2 consumption rate (ФCO2net) is the highest at 105.1 × 103 mol km−2 a−1 in the Dadu river basin at the furthest east edge of the plateau and decreases consistently towards the interior plateau basins, dropping to 11.4 × 103 mol km-2 a-1 at Nu River basin (Table 1). The Yarlung Tsangpo river basin with the highest altitude located at the inner area of the plateau serves as a weak CO2 sink with its average net CO2 consumption rate as low as 1.5 × 103 mol km−2 a−1 (Table 1). The decreasing trend of ФCO2net is ascribed to the generally moderative SWR accompanied with accelerated carbonate dissolution by sulfuric acid derived from OWP from the east edge to the interior of the plateau. The pattern for SWR and associated CO2 consumption is assumed to be mainly controlled by climate (temperature and water accessibility) as silicate weathering is more kinetic-limited within the background high erosion rate, while the CO2-releasing rate by sulfuric acid reaction with carbonate is primarily dominated by erosion rates. It is worth noting that the Palong Tsangpo river basin, locating at the Eastern Himalayan syntaxis, one of the fastest exhuming regions on Earth since ~10 Ma56, serves as a CO2 source with the highest CWR and an average net CO2 consumption flux at −0.07 × 109 mol a−1 (Table 1), suggesting intense CO2 contracting effects by H2SO4-carbonate reaction. Based on our calculation, the whole plateau serves as a long-term CO2 sink with a consumption flux at 33.2 × 109 mol a−1. In contrast, the value for CO2 sequestration flux is 79.4 × 109 mol a−1 if the sulfuric acid weathering effect is ignored. Therefore, although sulfide oxidation involved weathering in a single catchment in the Tibetan Plateau have been reported either net CO2 source or sink16,49,50, which is all reasonable, depending on their sulfide oxidation rates and the proportion of carbonate and silicate rock exposure, chemical weathering is still an important net CO2 sink from the comprehensive results of the large river basins in the Tibetan Plateau as a whole. According to our estimation, the sulfuric acid dissolution of carbonate has counteracted about 58% of the CO2 consumption flux by SW on the plateau. Our study, based on multiple isotope and solute composition observation for major river systems draining in the plateau, for the first time gains the accurate estimation of net CO2 consumption fluxes of the whole Tibetan Plateau by chemical weathering considering the involvement of sulfuric acid.
The refined CW and SW rates and associated CO2 budgets calculations in our study show that: (1) the sustained addition of sulfuric acid from OWP could exert a comparable magnitude of CO2 source flux in the Tibetan Plateau as SW consumption, modified the net CO2 budgets of plateau weathering. Our work quantitatively shows that the plateau formation of Himalayan orogenesis releases and consumes CO2 at the same magnitude by weathering and might lead to a weaker disturbance on atmospheric-ocean CO2 reservoir than previously thought. Therefore, the strong climate-driven feedback needed to offset the orogenic perturbations on the global carbon cycle could be relaxed and should be reconsidered in both modeling and geological record studies. However, attention should be paid to the sampling limitation. First, the ideal procedure is to do the calculation with high-frequency samples through a whole hydrological year, which is challenging due to field logistics and the lack of discharge data in the Tibetan Plateau. The samples in this study are from the high flow season, and average annual discharge data has been used for the flux estimation. Based on the daily dataset obtained in Nujiang22, it has been estimated that the silicate weathering flux calculated from high flow season sampling is on average 48% lower than the result calculated by the time serials dataset. Therefore, the silicate weathering flux and the associated CO2 consumption calculation are assumed to be a lower limit. Meanwhile, the sulfuric acid involvement is recognized highest in the high flow season within a hydrology year22. Therefore, the flux estimation in this work represents a lower limit of CO2 sequestration capability for the plateau weathering. Second, it is worthwhile to notice that the weathering signals and CO2 budgets observed and calculated in our study may represent a non-steady state resulted by increased OWP due to continental glaciations in the Pleistocene and intense glacial activity in the background of modern global change57, which would highlight the OWP and its effect on chemical weathering10,16, as thus it is a maximum estimation of H2SO4 disturbing in weathering and CO2 effect in the Tibetan Plateau weathering during its formation. Considering its tectonic and glacial activity variation, the long-term significance of H2SO4 could be less, and more work is needed to understand the situation in the deep time; (2) weathering sensitivities of different rock type to landscape variation15 are quantitatively illustrated in our study from the perspective of large river hydro-geochemistry at continent scale. The orogenesis and plateau formation exert a significantly discriminatory impact on carbonate and silicate weathering, as well as their associated CO2 effect. CW is significantly enhanced in the plateau, while SW is not anomalous. The formation of the Himalayas Mountain range and the Tibetan Plateau supplies the largest river runoff volume and sediment fluxes globally58 on the one hand and serves as a critical factor for the formation of strong South Asian monsoon on the other hand59,60,61, both of which will facilitate the subsequent SW in the lower reach for the sediments transported from the plateau by the river networks originated from the plateau. Thus, the CO2 effect of CW and SW should be considered separately for the large river basins spanning orogenic areas and alluvial plains when exploring and modeling the global carbon cycling of the orogenic events in the Cenozoic. The findings of this work propose that different sensitivities of silicate and carbonate weathering mediated by carbonate and non-carbonic acid in different landscapes need to be considered in current or future geologic carbon cycling models as they will bring about significant CO2 sink and source transition of chemical weathering at continental scale, and thus modulate the atmospheric CO2 effects of plateau uplifting event in Cenozoic era.
Methods
Sampling and measurements of river water samples
River water samples were collected near the middle of a channel from bridges using high-density polyethylene containers and filtered through a 0.22 μm Millipore membrane filter. One portion of filtrate was acidified to pH <2 with 6 M double sub-boiling distilled HNO3 for cation, trace elements, and Sr isotopic ratio (87Sr/86Sr) analysis, and another portion was stored for anion analysis. All containers were previously washed with high-purity HNO3, rinsed with Milli-Q 18.2 MΩ water, and then rinsed with filtrate three times in the field. Samples for dissolved inorganic carbon isotope (δ13C) measurements were preserved with HgCl2 to prevent biological activity in 50 ml polyethylene bottles with air-tight caps and were refrigerated until analysis. For sulfur and oxygen isotopic analysis of SO42−, the dissolved sulfate in the filtered water samples was precipitated as BaSO4 by adding excess amount of BaCl2 solution after the water was filtered and acidified to a pH value of ~2. This precipitation was filtered, washed, and dried in the laboratory for isotopic analysis.
The temperature (T), pH, and electrical conductivity (EC) were measured with a portable EC/pH meter. The HCO3− was determined in the field using the titration method. Anions (F−, Cl−, SO42−, and NO3−) were measured by the ion chromatography (Dionex 120) with a precision of ±5%. Cations (K+, Na+, Ca2+, and Mg2+) were determined by an Atomic Absorption Spectrometry with a precision of ±5%. Dissolved silica (SiO2) concentrations were determined by spectrophotometry using the molybdate blue method, with a precision of ±5%.
The 87Sr/86Sr was measured by a Multiple Collector Inductively Coupled Plasma Mass Spectrometry (MC-ICP-MS) after purifying using AG 50W-X8 cation exchange resin in the clean laboratory. The total procedure blank was ~100 pg Sr. The value of the NBS987 standard was 0.710232 ± 0.000020.
For the stable carbon isotope composition of river-dissolved inorganic carbon (DIC) measurements, the filtered samples were injected into glass bottles with phosphoric acid. The CO2 was then extracted and cryogenically purified using a high vacuum line. The δ13C isotopic ratios were analyzed on Finnigen MAT-252 stable isotope mass spectrometer and the results were expressed relative to VPDB (Vienna Pee Dee Belemnite) standard as δ13CDIC = (Rsample/Rstandard −1) × 1000, where δ13CDIC is the inorganic carbon isotope composition, and Rsample and Rstandard are the 13C/12C ratio in water sample and standard, respectively. The analyzing precision for international standard NBS-19 and the samples is better than 0.2‰.
δ34SSO4 and δ18OSO4 for dissolved SO42− were determined using the elemental analysis-isotope ratio mass spectrometry (EA-IRMS) and were reported in the δ notation relative to the Vienna Canyon Diablo Troilite reference (V-CDT) and Vienna Standard Mean Ocean Water reference (V-SMOW) in permil, respectively. The analytical precision for δ34SSO4 and δ18OSO4 values of NBS127 and samples was generally better than 0.2‰ and 0.5‰, respectively.
Calculations for the CO2 consumption rates by silicate and carbonate weathering considering the sulfuric acid involvement
The participation of sulfuric acid involved in carbonate and silicate weathering could be calculated by the simultaneous equations of concentration and isotopic composition of sulfate from different sources (Eqs. 1–3), with the assumption that silicate and carbonate weathering by sulfuric acid are in the same ratio as they are weathered by carbonic acid44.
[SO4]SCW and [SO4]SSW represent the fractions of sulfuric acid consumed by carbonate and silicate weathering, respectively. A is the ratio of carbonic acid consumed by silicate weathering to carbonic acid consumed by carbonate weathering, which could be calculated with the cation partition between different end-members gained in section Supplementary Text Information B. [SO4]pyrite has been calculated in section Supplementary Text Information B.
The long-term net CO2 consumption budget considering the sulfuric acid involvement in the plateau could be calculated by deducting the CO2 releasing fluxes in carbonate weathering by sulfuric acid from the CO2 consumption fluxes by silicate weathering (Eq. 4) based on the riverine solute sourcing in Supplementary Text Information B and the fractions of sulfuric acid consumed by carbonate and silicate weathering calculated above:
ФCO2sil* and ФCO2SCW indicate yield (mol km−2 a−1) of atmospheric CO2 consumption/releasing of silicate weathering and carbonate weathering, respectively, with consideration of sulfuric acid involvements. The subscripts of “sil” denote cations originating from silicate weathering.
Data availability
All data needed to evaluate the conclusion in the paper are present in the paper and the Supplementary Materials. Source data generated in this study have been deposited in the Figshare database (https://doi.org/10.6084/m9.figshare.28092671). Source data are provided with this paper. The digital elevation model (DEM) used in this study is derived from the Shuttle Radar Topography Mission (SRTM) 3 arc-second (90 m) dataset62, openly available from NASA’s Earthdata portal (https://earthdata.nasa.gov/).
References
Raymo, M. E. & Ruddiman, W. F. Tectonic forcing of late Cenozoic climate. Nature 359, 117–122 (1992).
Raymo, M. E., Ruddiman, W. F. & Froelich, P. N. Influence of late Cenozoic Mountain building on ocean geochemical cycles. Geology 16, 649–653 (1988).
Quade, J., Roe, L., DeCelles, P. G. & Ojha, T. P. The late Neogene 87Sr/86Sr record of lowland Himalayan rivers. Science 276, 1828–1831 (1997).
Blum, J. D., Gazis, C. A., Jacobson, A. D. & Page Chamberlain, C. Carbonate versus silicate weathering in the Raikhot watershed within the High Himalayan Crystalline Series. Geology 26, 411–414 (1998).
Willenbring, J. & von Blanckenburg, F. Long-term stability of global erosion rates and weathering during late-Cenozoic cooling. Nature 465, 211–214 (2010).
Caves Rugenstein, J. K., Ibarra, D. E. & von Blanckenburg, F. Neogene cooling driven by land surface reactivity rather than increased weathering fluxes. Nature 571, 99–102 (2019).
Galy, V. et al. Efficient organic carbon burial in the Bengal fan sustained by the Himalayan erosional system. Nature 450, 407–410 (1997).
Louvat, P. & Allègre, C. J. Present denudation rates on the island of Réunion determined by river geochemistry: basalt weathering and mass budget between chemical and mechanical erosions. Geochim. Cosmochim. Acta 61, 3645–3669 (1997).
West, A. J., Bickle, M. J., Collins, R. & Brasington, J. Small-catchment perspective on Himalayan weathering fluxes. Geology 30, 355–358 (2002).
Torres, M. A. et al. Glacial weathering, sulfide oxidation, and global carbon cycle feedbacks. Proc. Natl. Acad. Sci. USA 114, 8716–8721 (2017).
Torres, M. A., West, A. J. & Li, G. Sulphide oxidation and carbonate dissolution as a source of CO2 over geological timescales. Nature 507, 346–349 (2014).
Burke, A. et al. Sulfur isotopes in rivers: Insights into global weathering budgets, pyrite oxidation, and the modern sulfur cycle. Earth Planet. Sc. Lett. 496, 168–177 (2018).
Bufe, A. et al. Co-variation of silicate, carbonate, and sulfide weathering drives CO2 release with erosion. Nat. Geosci. 14, 211–216 (2021).
Bufe, A. et al. The effect of lithology on the relationship between denudation rate and chemical weathering pathways–evidence from the eastern Tibetan Plateau. Earth Surf. Dynam. 10, 513–530 (2022).
Bufe, A., Rugenstein, J. & Hovius, N. CO2 drawdown from weathering is maximized at moderate erosion rates. Science 383, 1075–1080 (2024).
Liu, W. J. et al. Lithological and glacial controls on sulfide weathering and the associated CO2 budgets in the Tibetan Plateau: new constraints from small catchments. Geochim. Cosmochim. Acta 343, 341–352 (2023).
Tipper, E. T., Galy, A. & Bickle, M. J. Riverine evidence for a fractionated reservoir of Ca and Mg on the continents: implications for the oceanic Ca cycle. Earth Planet. Sci. Lett. 247, 267–279 (2006).
Rai, S. K. & Singh, S. K. Temporal variation in Sr and 87Sr/86Sr of the Brahmaputra: implications for annual fluxes and tracking flash floods through chemical and isotope composition. Geochem. Geophys. Geosyst. 8, Q08008 (2007).
Singh, S. K., Rai, S. K. & Krishnaswami, S. Sr and Nd isotopes in river sediments from the Ganga Basin: sediment provenance and spatial variability in physical erosion. J. Geophys. Res. Atmos. 113, F03006 (2008).
Manaka, T. et al. Downstream and seasonal changes of lithium isotope ratios in the Ganges- Brahmaputra River system. Geochem. Geophys. Geosyst. 18, 3003–3015 (2017).
Zhong, L. et al. Using zircon U-Pb ages to constrain the provenance and transport of heavy minerals within the northwestern shelf of the South China Sea. J. Asian Earth Sci. 134, 176–190 (2017).
Liu, W. et al. Hydrochemistry and carbon isotope characteristics of Nujiang River water: Implications for CO2 budgets of rock weathering in the Tibetan Plateau. Sci. China Earth Sci. 66, 2953–2970 (2023).
Huh, Y. Estimation of atmospheric CO2 uptake by silicate weathering in the Himalayas and the Tibetan Plateau: a review of existing fluvial geochemical data. Geo. Soc. Lond. Spec. Pub. 342, 129–151 (2010).
Gaillardet, J., Dupré, B., Louvat, P. & Allègre, C. J. Global silicate weathering and CO2 consumption rates deduced from the chemistry of large rivers. Chem. Geol. 159, 3–30 (1999).
Pande, K. et al. The Indus river system (India-Pakistan): major-ion chemistry, uranium and strontium isotopes. Chem. Geol. 116, 245–259 (1994).
Galy, A. & France-Lanord, C. Weathering processes in the Ganges–Brahmaputra basin and the riverine alkalinity budget. Chem. Geol. 159, 31–60 (1999).
Palmer, M. R. & Edmond, J. M. The strontium isotope budget of the modern ocean. Earth Planet. Sci. Lett. 92, 11–26 (1992).
Chetelat, B. et al. Geochemistry of the dissolved load of the Changjiang Basin rivers: anthropogenic impacts and chemical weathering. Geochim. Cosmochim. Acta 72, 4254–4277 (2008).
Wang, Z. L., Zhang, J. & Liu, C. Q. Strontium isotopic compositions of dissolved and suspended loads from the main channel of the Yangtze River. Chemosphere 69, 1081–1088 (2007).
Oliver, L. et al. Silicate weathering rates decoupled from the 87Sr/86Sr ratio of the dissolved load during Himalayan erosion. Chem. Geol. 201, 119–139 (2003).
Shan, S. et al. Carbon isotopic constrains on the sources and controls of the terrestrial carbon transported in the four large rivers in China. Adv. Earth Sci. 35, 948–961 (2020).
Marwick, T. R. et al. The age of river-transported carbon: a global perspective. Glob. Biogeochem. Cycles 29, 122–137 (2015).
Meybeck, M. Global chemical weathering of surficial rocks estimated from river dissolved loads. Am. J. Sci. 287, 401–428 (1987).
Négrel, P., Allègre, C. J., Dupré, B. & Lewin, E. Erosion sources determined by inversion of major and trace element ratios and strontium isotopic ratios in river water: the Congo Basin case. Earth Planet. Sci. Lett. 120, 59–76 (1993).
Xu, Z. F. & Liu, C. Q. Chemical weathering in the upper reaches of Xijiang River draining the Yunnan -Guizhou Plateau, Southwest China. Chem. Geol. 239, 83–95 (2007).
Liu, W. J. et al. Geochemistry of the dissolved loads during high-flow season of rivers in the southeastern coastal region of China: anthropogenic impact on chemical weathering and carbon sequestration. Biogeosciences 15, 4955–4971 (2018).
Fan, B. L. et al. Characteristics of carbonate, evaporite and silicate weathering in Huanghe River basin: a comparison among the upstream, midstream and downstream. J. Asian Earth Sci. 96, 17–26 (2014).
Wang, B. et al. Distinct patterns of chemical weathering in the drainage basins of the Huanghe and Xijiang River, China: evidence from chemical and Sr-isotopic compositions. J. Asian Earth Sci. 59, 219–230 (2012).
Li, S., Lu, X. X. & Bush, R. T. Chemical weathering and CO2 consumption in the Lower Mekong River. Sci. Total Environ. 472, 162–177 (2014).
Manaka, T. et al. Chemical weathering and long-term CO2 consumption in the Ayeyarwady and Mekong river basins in the Himalayas. J. Geophys. Res. Biogeosci. 120, 1165–1175 (2015).
Chapman, H., Bickle, M., Thaw, S. & Thiam, H. Chemical fluxes from time series sampling of the Irrawaddy and Salween Rivers, Myanmar. Chem. Geol. 401, 15–27 (2015).
Galy, A., France-Lanord, C. & Derry, L. A. The strontium isotopic budget of Himalayan rivers in Nepal and Bangladesh. Geochim. Cosmochim. Acta 63, 1905–1925 (1999).
Singh, S. K., Kumar, A. & France-Lanord, C. Sr and 87Sr/86Sr in waters and sediments of the Brahmaputra river system: Silicate weathering, CO2 consumption and Sr flux. Chem. Geol. 234, 308–320 (2006).
Moon, S., Huh, Y., Qin, J. & Nguyen, V. P. Chemical weathering in the Hong (Red) River basin: rates of silicate weathering and their controlling factors. Geochim. Cosmochim. Acta 71, 1411–1430 (2007).
Berner, E. K.& Berner R. A. Global Environment: Water, Air, and Geochemical Cycles (Prentice Hall, 1996).
Zeng, X. et al. Characteristics and Genesis of low frequency debris flow along ParlungZangbo River Zone-take ChaoBulongba gully as an example. Sci. Tech. Eng. 19, 103–107 (2019).
Zhang, G. The controlling effect of structure and climate on the distribution of debris flow in Palong Zangbu Basin 1-69 (Xizang Univ., 2023).
Yatagai, A. et al. A 44-year daily gridded precipitation dataset for Asia based on a dense network of rain gauges. Sola 5, 137–140 (2009).
Cao, Y. et al. Chemical weathering in glacial catchment acting as a net carbon source. Sci. Total Environ. 901, 165842 (2023).
Zhang, F. et al. Chemical weathering and CO2 consumption in the glaciated Karuxung River catchment, Tibetan Plateau. Hydrol. Process. 35, e14330 (2021).
Spence, J. & Telmer, K. The role of sulfur in chemical weathering and atmospheric CO2 fluxes: evidence from major ions, δ13CDIC, and δ34SSO4 in rivers of the Canadian Cordillera. Geochim. Cosmochim. Acta 69, 5441–5458 (2005).
Das, A., Krishnaswami, S., Sarin, M. M. & Pande, K. Chemical weathering in the Krishna Basin and Western Ghats of the Deccan Traps, India: rates of basalt weathering and their controls. Geochim. Cosmochim. Acta 69, 2067–2084 (2005).
Singh, S. K., Sarin, M. M. & France-Lanord, C. Chemical erosion in the eastern Himalaya: major ion composition of the Brahmaputra and δ13C of dissolved inorganic carbon. Geochim. Cosmochim. Acta 69, 3573–3588 (2005).
Li, S. L. et al. Sulfuric acid as an agent of carbonate weathering constrained by δ13CDIC: examples from southwest China. Earth Planet. Sci. Lett. 270, 189–199 (2008).
Li, C. & Ji, H. B. Chemical weathering and the role of sulfuric and nitric acids in carbonate weathering: Isotopes (13C, 15N, 34S, and 18O) and chemical constraints. J. Geophys. Res. Biogeosci. 121, 1288–1305 (2016).
Yang, J. et al. Erosion-driven isostatic flow and crustal diapirism: analytical and numerical models with implications for the evolution of the Eastern Himalayan Syntaxis, Southern Tibet. Tectonics 42, e2022TC007717 (2023).
Yao, T. D. et al. Different glacier status with atmospheric circulations in Tibetan Plateau and surroundings. Nat. Clim. Chang. 2, 663–667 (2012).
Gleick, P. H. In Water in crisis: a guide to the world’s fresh water resources (Oxford Univ. Press, 1993).
Ruddiman, W. F. & Kutzbach, J. E. Forcing of late Cenozoic northern hemisphere climate by plateau uplift in southern Asia and the American west. J. Geophys. Res. Atmos. 94, 18409–18427 (1989).
Ruddiman, W. F., Prell, W. L. & Raymo, M. E. Late Cenozoic uplift in southern Asia and the American West: rationale for general circulation modeling experiments. J. Geophys. Res. Atmos. 94, 18379–18391 (1989).
Prell, W. L. & Kutzbach, J. E. Sensitivity of the Indian monsoon to forcing parameters and implications for its evolution. Nature 360, 647–652 (1992).
Farr, T. G. et al. The shuttle radar topography mission. Rev. Geophys. 45, RG2004 (2007).
Acknowledgements
This work was supported by the National Key Research and Development Program of China (No. 2020YFA0607700), the National Natural Science Foundation of China (Nos. 42422303, 42488201 and 41730857), and the Key Research Program of the Institute of Geology & Geophysics, CAS (No. IGGCAS−202204, 202201). Wenjing Liu acknowledges support from the Youth Innovation Promotion Association CAS (Y2023014).
Author information
Authors and Affiliations
Contributions
W.L., Z.X., and Z.G. designed the project and investigation; W.L., H.S., M.Z., and Y.X. conducted the field work and data analysis and performed the research; W.L. drafted the original manuscript; Z.X., H.S., M.Z., and Z.G. edited and revised the draft. All the coauthors contributed new information and interpretations of the findings during the manuscript writing process.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Fei Zhang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, W., Xu, Z., Sun, H. et al. Refined weathering CO2 budget of the Tibetan Plateau strongly modulated by sulphide oxidation. Nat Commun 16, 2741 (2025). https://doi.org/10.1038/s41467-025-58046-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-58046-4