Abstract
“Hydrogenation by crude H2 - dehydrogenation to produce pure H2” strategy using liquid organic hydrogen carriers (LOHCs) can reduce the cost and shorten the process of hydrogen purification and utilization. The critical challenge is to eliminate catalyst poisoning by CO impurity in crude H2. Here, we develop a Pd/α-MoC catalyst that enables efficient hydrogenation of N-LOHCs under crude hydrogen feeds (CO concentration>5 vol%) below 150 °C, and has an activity 1-2 orders of magnitude higher than that of traditional Pd-based catalysts. The CO-resistant hydrogenation is also successfully conducted in the models of industrial crude H2 including CO, CO2 and CH4. Water, as solvent, contributes greatly to the hydrogenation activity against CO poisoning, since the utilization of low-temperature water gas shift (WGS) reaction. Moreover, the positive-charged Pd species hinder the combination of H* from WGS reaction and suppressed the undesirable H2 formation and release, which explains the substantial improvement in the performance of Pd/α-MoC compared to that of Pt/α-MoC.
Similar content being viewed by others
Introduction
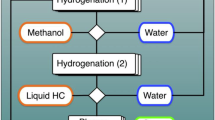
In the foreseeable future, hydrogen will play an essential role in the field of both energy and chemical industries, not only reducing CO2 emissions, but also establishing a cleaner and more sustainable industrial system1,2. Presently, large quantities of crude H2 containing CO, CH4, CO2 or other impurities, are produced from fossil fuel reforming or generated as a by-product of chemical industry3,4. However, downstream applications of H2, such as fuel cell and hydrogenation processes, usually have strict requirements for H2 purity, especially in terms of CO concentration5,6,7,8, since CO impurities can seriously poison the precious metal catalysts used4. The traditional method requires water-gas shift reaction and CO purification processes (preferential oxidation, pressure swing adsorption, palladium membrane stack, etc.) to remove CO impurities, which increases the cost of hydrogen utilization6,9. Not only that, the purified hydrogen gas needs to be compressed into special cylinders for storage and transportation, leading to a cumbersome and inefficient process (Fig. 1, up).
The production and transportation of crude H2 to high purity H2 conventionally require water-gas shift reaction (WGS) to remove most of CO impurity, coupled with deep purification techniques including preferential oxidation (PROX), pressure swing adsorption (PSA), palladium membrane stack, etc. Finally, the pure H2 is pressurized for transportation, storage and utilization. The novel route of “hydrogenation by crude H2 - dehydrogenation to produce pure H2” is proposed, which realizes a simple and efficient process that integrates crude hydrogen purification, hydrogen storage and hydrogen transportation.
The cyclic hydrogenation and dehydrogenation of liquid organic hydrogen carriers (LOHCs), are a potential approach for H2 storage and transportation10,11. Especially, the concept of “hydrogenation by crude H2 - dehydrogenation to produce pure H2” has been proposed and intensively attracted4,12,13. The main opinion is that if H0-LOHCs (H2-lean state, eg, quinoline) can be hydrogenated with crude H2 or even industrial waste H2 via catalytic CO-resistant hydrogenation, and pure H2 can be obtained by dehydrogenation of corresponding Hx-LOHCs (H2-rich state, eg, decahydroquinoline or tetrahydroquinoline), realizing a simple and efficient process that integrates crude hydrogen purification, hydrogen storage and hydrogen transportation (Fig. 1, bottom)4. Recently, Hoshimoto et al. discovered that frustrated Lewis pairs that are composed of Lewis bases (LBs) and triaryl boranes as the Lewis acids (LAs), called shelf-stable Bn-catalysts, could catalyze the CO-resistant hydrogenation of 2-methylquinoline9 and quinoline12 in crude H2 with CO concentration higher than 10%. The dehydrogenation of Hx-LOHCs can also be occurred on the same catalyst. It is a significant advance in CO-resistant hydrogenation and crude hydrogen purification in homogeneous catalysis. However, achieving CO-resistant hydrogen storage in H0-LOHCs under mild condition remains a formidable challenge for the heterogeneous catalytic systems. Jorschick et al. performed a pioneer work13 to extract pure H2 from inexpensive CO containing crude hydrogen source using CO-resistant catalytic hydrogenation of DBT (typical H0-LOHCs molecules14,15). Among Ru-, Rh- and Pt- based catalysts, the optimized Pd/Al2O3 catalyst showed the highest activity for the hydrogenation of H0-DBT with CO containing crude H2. However, when 0.5 vol%CO was introduced, the hydrogenation activity is less than 10% of the same catalyst under CO free condition at 240 °C. When the CO concentration increased to 5 vol%, 90% hydrogenation activity loss occurs even when the operation temperature was raised to 300 °C13. Another CO resistant hydrogenation concept is also realized by coupling CO methanation with toluene hydrogenation to remove CO poisons on a RuNi/TiO2 catalyst16. However, this catalyst is also unable to treat crude H2 with high CO concentration (1%) as the remaining CO impurities in the stream still causes poisoning and inhibits the hydrogenation. In addition, the incorporation of N into LOHCs (N-LOHCs) is beneficial to acquired pure H2 (with a purity of 99.9%) under mild reaction condition, as the dehydrogenation enthalpy of N-LOHCs are generally smaller than aromatic LOHCs17,18,19. Therefore, it is of great importance to develop heterogeneous catalysts that are capable of CO-resistant hydrogenation of N-LOHCs, and even adapt to different CO concentration under different conditions.
Atomically dispersed Pt1 (or Pd1)-Ox- stabilized on a variety of supports20, such as silica21, titania22, was found to be highly active in WGS reactions, as the single-atom Pt sites have a cationic nature that results in weak CO adsorption. Although single-atom catalytic sites in Pt1 (or Pd1)-Ox-oxides possess the desired property of weaker CO adsorption23, they may not be stable enough under hydrogenation conditions24. Pt1Cu single atom alloy catalyst demonstrates improved CO resistant hydrogenation under realistic working conditions for H2 activation and acetylene hydrogenation reaction24, but the CO concentration is 200 ppm which is uncapable to adapt for the various crude H2 from industry. Atomically dispersed Pt over α-molybdenum carbide (α-MoC) can tolerate crude H2 with high CO concentration ( > 10%). Even using pure CO and H2O as hydrogen source, hydrogenation of nitrobenzene was still achievable. The weaker adsorption of CO on Ptδ+ sites and a new reaction pathway that H2O acted as H donor and promoted catalysis had been observed and investigated6,25,26. A H2-free highly selective hydrogenation of nitroarenes27 and acetylene28 processes are also achieved by directly using CO and water as the hydrogen source over Au/α-MoC catalyst, by using in situ-generated hydroxyl species from water as a hydrogen donor and low-cost CO as an oxygen acceptor. Although the substrates were limited to -NO2 and C ≡ C groups, it should be noted that MoC-based noble-metal catalysts have great potential in CO-resistant hydrogenation of LOHCs adapting to various crude H2 from different origins, especially in the presence of aqueous environments29. Notably, molybdenum-based carbide30,31,32 and nitride33,34 catalysts showed high catalytic activity for the reverse water-gas shift (RWGS) reaction, a competing reaction of LOHCs hydrogenation under relatively high reaction temperature. Therefore, a mild reaction condition is required for efficient LOHCs CO-resistant hydrogenation over MoC-based metal catalysts.
Here, we report Pd/α-MoC catalyst that can tolerate crude H2 with CO concentration up to 30 vol% and catalyze the CO-resistant hydrogenation of N-heterocycles, such as quinoline, 2-(6)-(Di)methylpyridine, enabling crude H2 storage in Hx-LOHCs below 150 °C. Combining reaction experiments, catalyst characterization and DFT calculation, the CO-resistant hydrogenation mechanism of Pd/α-MoC is demonstrated. On the one hand, the highly dispersed Pd modulated by α-MoC support is capable of activating H2 in the presence of CO under reaction conditions. On the other hand, H2O solvent plays an important role to remove the CO adsorbed on Pd by water-gas shift reaction (WGS, CO + H2O → CO2 + H2) which greatly improves hydrogenation performance. In addition, the isotopic experiments confirm that the H* generated by WGS on Pd/α-MoC catalyst participates in the CO-resistant hydrogenation of quinoline directly, while the higher formation energy of H2 from H*-Pd ensures that H species exist in the form of active Had rather than released H2.
Results
Pd/α-MoC as highly efficient CO-resistant catalyst
Hydrogenation of quinoline to decahydroquinoline (DHQ) or tetrahydroquinoline (THQ) in pure H2 is a well-investigated reaction usually catalyzed by noble metal catalysts such as Pt, Pd or Ir35,36,37,38. As shown in Table 1, Entry 1 and 4 (Supplementary Table 1, Entry 1 and 4), under the test conditions, traditional Pd/Al2O3 and Pd/CeO2 catalysts synthesized by impregnation method achieved an outstanding THQ yield of >95% at 100 °C. However, along with the addition of 5 vol% CO in the H2 gas, the hydrogenation activity of these catalysts was sharply suppressed (Table 1, Entry 2 and 5). On Pd/Al2O3 catalyst, no products were detected, indicating Pd particles on Al2O3 support was totally blocked by CO and losing the hydrogenation capability under the mild reaction condition. Pd particles loaded on CeO2 support showed certain CO-resistant hydrogenation ability, but the conversion dropped to only 10%. By contrast, both Pd/α-MoC and Pt/α-MoC still catalyzed the hydrogenation of quinoline efficiently under the same conditions of 5 vol% CO/H2 (Table 1, Entry 6–10), the conversion remained of 86% and 73%, respectively. And, the selectivity of THQ was nearly 100% as reported before36. When reaction time was extended, the conversion of quinoline could reach 100% (Supplementary Table 2). And similar to the case of nitrobenzene, when H2O was chosen as solvent, better CO-resistant hydrogenation performance would be acquired (Supplementary Fig. 1), which is an interesting phenomenon discussed later.
The metal-normalized hydrogenation activities of quinoline over 3%Pd/α-MoC, 3%Pt/α-MoC and 3%Pd/CeO2 were tested. As shown in Fig. 2a, the activities in pure H2 followed the sequence of Pd/α-MoC <Pt/α-MoC <Pd/CeO2. However, the sequence was reversed in crude H2 (5%CO/H2), the Pd/CeO2 was poisoned severely and its activity was lowered by more than one order of magnitude (from 612 h−1 to 22 h−1), but Pd/α-MoC kept considerable activity of 163 h−1 in the 5 vol% CO/H2 reaction condition, demonstrating better CO-tolerant ability than Pt/α-MoC and Pd/CeO2, and Pd/α-MoC catalysts with different Pd loadings were proved to be efficient in CO-resistant hydrogenation of quinoline (Supplementary Fig. 2 and Supplementary Table 3). To our surprise, in non-aqueous solvent such as cyclohexane and methanol, Pt/α-MoC and Pd/CeO2 were poisoned more severely by CO and yield of THQ was less than 1% (Fig. 2b), but the yield of THQ over Pd/α-MoC was around 10% at the same time, indicating that the intrinsic CO-tolerant ability of Pd/α-MoC was probably the strongest. Afterwards, the CO-resistant hydrogenation performance of Pd/α-MoC was further explored in crude H2 with multiple CO concentration. As shown in Fig. 2c, Pd/α-MoC was able to tolerate 1–30% CO in H2, and even when pure CO was utilized, quinoline still could be hydrogenated by H2 or active H* from WGS reaction in H2O solvent. In the model of industrial crude H2 including CO, CO2 and CH4, the CO-resistant hydrogenation of quinoline to THQ was still successfully conducted (Table 2 and Supplementary Table 4.), proving the practicality of Pd/α-MoC catalysts. Pd/α-MoC catalyst also showed excellent stability in a five-cycle stability test for CO-resistant hydrogenation (Fig. 2d and Supplementary Fig. 3).
a The metal-normalized activity of quinoline hydrogenation of 3%Pd/α-MoC, 3%Pt/α-MoC and 3%Pd/CeO2 using pure H2 or crude H2 with 5%CO (reaction conditions: 5 mmol quinoline, 10 or 25 mg of catalysts, 15 mL of H2O, 100 °C, 2 MPa, 0.5 h). b THQ yield of these catalysts in different solvent (reaction conditions: 1 mmol quinoline, 25 mg of catalysts, 15 mL of solvent, 100 °C, 2 MPa 5%CO/H2, 2 h). c THQ yield of 3%Pd/α-MoC using crude H2 with different CO concentration (reaction conditions: 1 mmol quinoline, 25 mg of 3%Pd/α-MoC, 15 mL of H2O 100 °C, 2 MPa, 2 h). d Stability test of the 3%Pd/α-MoC catalyst for CO-resistant hydrogenation of quinoline (reaction conditions: 25 mg 3%Pd/α-MoC, 1 mmol quinoline, 15 mL H2O, 2 MPa of 5%CO/H2, 100 °C for each 2 h cycle).
Positive-charged Pd activating H2 against CO poisoning
To understand the observed outstanding CO-resistant hydrogenation performance of the Pd/α-MoC catalyst, we first examined the valence states of Pd species by quasi-in situ X-ray photoelectron spectra (Fig. 3a and Supplementary Fig. 4). The characteristic peaks of Pd 3 d over 3%Pd/α-MoC and 3%Pd/CeO2 were 335.9 and 335.6 eV, respectively, which can be assigned to positive charged palladium species (Pdδ+)39 indicating the electron transfer from Pd to the support. Compared to Pd-CeO2, Pd-α-MoC shows stronger metal-support interaction effect. From XRD patterns, no diffractions associated with Pd crystal were observed on 3%Pd/α-MoC catalyst (Supplementary Fig. 5), and no large Pd nanoparticles were identified by aberration-corrected scanning transmission electron microscopy (STEM) imaging with simultaneous energy dispersive X-ray spectroscopy (EDS) mapping and electron energy-loss spectroscopy (EELS) mapping (Fig. 3b and Supplementary Fig. 6), suggesting that Pd species were highly dispersed on α-MoC. Compared to Al2O3 and CeO2 supports, Pd atoms on α-MoC show higher dispersion (Supplementary Figs. 7-8). Since Pd has an atomic number close to Mo, it is hard to clearly visualize Pd single atoms on the α-MoC substrate in the Z-contrast HAADF-STEM images. Therefore, we further carried out DFT calculation as a supplement to experimental characterizations. As demonstrated in Fig. 3c, the binding energy between Pd single atom and α-MoC ranges from −4.22 ~ −4.69 eV, depending on the different Pd-Mo(C) coordination modes. Then, 10 optimized structures of Pd clusters from Pd4 to Pd13 were established and the corresponding cohesive energy of Pd clusters were displayed which ranges from −3.44 to −4.21 eV. The binding energy of Pd-Mo(C) is stronger than the cohesive energy of Pd clusters, in other words, the Pd clusters or small nanoparticles would be dispersed into single atoms or raft-like structure33 spontaneously under thermal driving on α-MoC support. Overall, it is speculated that the atomic dispersed Pd species were modulated by α-MoC in terms of structure and electronic properties, leading to an increase of H2 activation ability under CO atmosphere. In hydrogen/deuterium exchange temperature programed reaction (TPR) experiments (Fig. 4 and Supplementary Fig. 9), the production of HD indicated the dissociated adsorption and activation of hydrogen molecules, and HD appeared at lower temperature over Pd/α-MoC than Pd/CeO2 (32.5 °C vs 72 °C) when CO was co-fed. Based on the observed phenomena, we speculated that CO had a lower desorption energy barrier over the highly dispersed Pd on α-MoC, realizing CO-resistant H2 activation successfully. Besides, the same isotopic TPR experiments on pure α-MoC showed little production of HD (Supplementary Fig. 10), demonstrating the significance of Pd in H2 activation and further confirming the CO-tolerant catalytic properties of Pd species over α-MoC.
a Quasi in situ Pd 3 d X-ray photoelectron spectroscopy (XPS) spectra of the Pd catalysts. b HAADF-STEM images of 3%Pd/α-MoC, EDS elemental maps of Pd and EELS elemental maps of Mo. c The binding energy between Pd and α-MoC and the cohesive energy of Pd particles or clusters obtained from DFT calculation, and the model of Pd/α-MoC, bulk Pd and Pd clusters.
10 mL·min−1 H2 and 10 mL·min−1 10%D2/Ar were co-fed over 3%Pd/α-MoC (a) and 3%Pd/CeO2 (c). 10 mL·min−1 5%CO/H2 and 10 mL·min−1 10%D2/Ar were co-fed over 3%Pd/α-MoC (b) and 3%Pd/CeO2 (d). The temperature was raised at a rate of 5 °C·min−1, and the mass spectrometry signals of H2 (m/z = 2), HD (m/z = 3), D2 (m/z = 4) and CO (m/z = 28) were recorded.
Significant role of H2O in CO-resistant hydrogenation
In addition, it was observed that H2O solvent improved CO-resistant hydrogenation greatly (Fig. 2b). Given that noble metal loaded on α-MoC is an outstanding WGS catalyst40,41, WGS reaction would occur during CO-resistant hydrogenation in H2O solvent. To further understand its influence, we measured the hydrogenation rates and WGS rates of 3%Pd/α-MoC, 3%Pt/α-MoC and 3%Pd/CeO2 catalysts. As shown in Fig. 5a, Pt/α-MoC had the highest WGS activity, and its hydrogenation activity in pure H2 was also higher than Pd/α-MoC (Fig. 2a), however, the best CO-resistant hydrogenation catalyst was Pd/α-MoC, indicating clearly that CO-resistant hydrogenation was not equivalent to WGS reaction coupling hydrogenation. Besides, in a reaction under low temperature and longtime condition (Fig. 5b), we observed that the CO concentration did not decrease, mainly due to the consumption of hydrogen while the THQ yield increased. These experiments pointed out that the CO-resistant hydrogenation of quinoline over Pd/α-MoC did not reduce the concentration of CO, and the coexistence of high WGS activity and high hydrogenation activity did not lead to efficient CO-resistant hydrogenation necessarily. Referring to literature of hydrogen transfer reaction6,42,43,44, it is speculated that the active H* species generated from WGS on catalyst surface played an important role in CO-resistant hydrogenation of quinoline. In other words, H2O participated in the hydrogenation directly through the WGS route, leading to higher activity than in non-aqueous solvent. In order to prove this hypothesis, isotopic experiments were carried out. As demonstrated in Fig. 5c and Supplementary Table 5, stronger kinetic isotope effect (rH/rD ≈ 1.4) was obtained in 5%CO/H2 than in pure H2 (rH/rD ≈ 1.1), and the proportion of deuterated THQ in reaction with 5%CO/H2 was also higher (Supplementary Figs. 11-15, Supplementary Tables 6-7.), suggesting that the dissociation of H2O is an essential step that the active H* generated participates in hydrogenation of quinoline under CO atmosphere. Thus, the affinity of H* on the metal (M-H) and formation energy of H2 from H* is important to ensure it existing as active Had rather than released as H2. Based on the DFT calculations, as shown in Fig. 5d and Supplementary Fig. 16, the binding energy of H* on Pd atom is 1.19 eV, about 0.18 eV higher than Pt. Especially, the barrier for H2 formation on Pd1/α-MoC is 1.37 eV, about 0.39 eV higher than that on Pt1/α-MoC which is known as a metal catalyst with excellent hydrogen evolution capability45. This explains why the CO-resistant hydrogenation of Pd/α-MoC is better than Pt/α-MoC catalyst, even if Pt/α-MoC shows the better WGS activity.
a The performance of quinoline hydrogenation and activity of water-gas shift (WGS) reaction under different conditions on 3%Pd/α-MoC, 3%Pt/α-MoC and 3%Pd/CeO2 (reaction conditions: 25 mg of catalysts, 15 mL H2O, 1 mmol of quinoline, 2 MPa of 10%CO/H2 or pure CO, 2 h; no quinoline was added when WGS rates were tested). b The time-dependent quinoline hydrogenation performance and CO concentration (reaction conditions: 10 mg of 3%Pd/α-MoC, 1 mmol of quinoline, 80 °C, 2 MPa of 5%CO/H2). c Activity of quinoline hydrogenation in H2O and D2O under different atmosphere (reaction conditions: 5 mmol of quinoline, 10 or 25 mg of catalysts, 15 mL of H2O or D2O, 100 °C, 2 MPa of gas, 0.5 h, the conversion of quinoline was <15%, the error bars show the deviation of TOF based on three repeated experiments). d The energy profiles for the H2 formation on the Pd1/α-MoC and Pt1/α-MoC; in the inserted structures of the corresponding intermediates, the Mo, Pd, Pt, C and H atoms are given in cyan, orange, blue, gray and white, respectively.
In general, Pd/α-MoC catalysts are active in the CO-resistant hydrogenation reaction in both aqueous and non-aqueous solvents (Fig. 6). The CO-resistant hydrogenation activity under aqueous reaction condition is 18 times higher than that in non-aqueous solvents, as H2O acts not only as a solvent but also as a reactant. The dissociated *OH species promotes the conversion of CO* adsorbed on Pd into CO2, and at the same time, the generated active H* atoms are effectively used for quinoline hydrogenation. The removal of CO and the H* adsorbed on Pd synergistically facilitates the hydrogenation reaction in crude hydrogen.
Benefiting from the high activity of Pd/α-MoC catalyst for WGS reaction, it exhibits excellent quinoline anti-CO hydrogenation in aqueous solution compared with non-aqueous solvent.
Discussion
In summary, we demonstrate that Pd/α-MoC is an exceptional catalyst for CO-resistant hydrogenation of quinoline and (di)methylpyridine, with CO concentration as high as 30 vol%, realizing crude H2 storage in N-LOHCs. Structural characterization shows that Pd species were highly dispersed on α-MoC and are positively charged. DFT calculation further confirmed that due to the strong interaction between Pd and α-MoC support, the Pd clusters or small nanoparticles would spontaneously dispersed into single atoms or raft-like structure on α-MoC support under thermal driving. In hydrogen/deuterium exchange temperature programed experiments, we recognized the CO-resistant hydrogenation mechanism that the Pd/α-MoC with unique electronic properties were capable of activating H2 in the presence of CO at room temperature. Especially in aqueous solution, the CO-resistant hydrogenation improved greatly. Based on isotopic experiments of H2O/D2O, a stronger kinetic isotope effect and a higher proportion of deuterated py-THQ in 5%CO/H2 (rH/rD ≈ 1.4) than in pure H2 (rH/rD ≈ 1.1) were observed, suggesting that the active H* generated from CO and H2O participates in hydrogenation of quinoline. Thus, ensuring it exists as active Had* rather than released H2 is important for the enhanced CO-resistant hydrogenation activity. DFT calculation showed that the binding energy of H* and the barrier of H2 formation on Pd1/α-MoC are both higher than that on Pt1/α-MoC, which are 0.18 and 0.39 eV, respectively, explaining why the CO-resistant hydrogenation of Pd/α-MoC is better than Pt/α-MoC catalyst, even if Pt/α-MoC shows the better WGS activity. These developments and knowledge are expected to shed light on new hydrogen storage and transportation systems utilizing crude H2.
Methods
Materials
Analytical-grade chemicals including palladium chloride (PdCl2, 99%), chloroplatinic acid hexahydrate (H2PtCl6·6H2O, 99%), ceric oxide (CeO2, 99.5%), ammonium molybdate tetrahydrate ((NH4)6Mo7O24·4H2O, 99%), aluminum oxide (Al2O3, 99.5%), quinoline (98%), m-xylene (99%), cyclohexane (99.7%) and methanol (99.7%) were purchased from Sinopharm Chemical Reagent Co., Ltd. Gas (5%CO/H2, pure H2) was purchased from Shanghai YouJiaLi Liquid Helium Co., Ltd.
Catalyst preparation
Preparation of α-MoC
MoO3 was prepared by calcination of (NH4)6Mo7O24·4H2O in air atmosphere at 500 °C for 4 hours. The obtained MoO3 was heated to 700 °C (5 °C·min−1) in flowing NH3 (160 mL·min−1) and held at 700 °C for 2 h, cooled down to room temperature and then treated in flowing 20%CH4/H2 (60 mL·min−1) at 700 °C (heating rate: 5 °C·min−1) for 2 h to obtain α-MoC. The carbonized sample was passivated in 0.5%O2/Ar mixture (60 mL·min−1) at room temperature overnight.
Preparation of Pd/α-MoC
Pd/α-MoC catalysts with different Pd content were synthesized via a wet-impregnation method using PdCl2 as the precursor. Taking 3 wt% Pd/α-MoC as an example, 0.3 g of α-MoC was dispersed in 25 mL of deionized water in a round-bottom flask, and the oxygen in the flask was removed by blowing N2. Appropriate amount of PdCl2 solution containing 9 mg of Pd was added under vigorous stirring. The obtained mixture was stirred for 3 hours at room temperature under an N2 atmosphere. The water in the mixture was evaporated using a rotary evaporator at 58 °C to obtain a solid sample which was then frozen and dried overnight. Before catalytic evaluation, the sample was activated in flowing 15%CH4/H2 at 590 °C for 2 hours.
Preparation of Other catalysts
The Pt/α-MoC was prepared using H2PtCl6·6H2O as the precursor. The Pd/CeO2 and Pd/Al2O3 catalysts were prepared using CeO2 and Al2O3 as support respectively instead of α-MoC. 0.3 g of support was dispersed in 25 mL of deionized water in a round-bottom flask, and the oxygen in the flask was removed by blowing N2. Appropriate amount of metal solution was added under vigorous stirring. The obtained mixture was stirred for 3 hours at room temperature under an N2 atmosphere. The water in the mixture was evaporated using a rotary evaporator at 58 °C to obtain a solid sample which was then frozen and dried overnight. Before catalytic evaluation, Pt/α-MoC was activated in flowing 15%CH4/H2 at 590 °C for 2 hours, while Pd/CeO2 and Pd/Al2O3 were reduced in flowing 10%H2/N2 at 200 °C for 2 h.
Catalyst characterization
X-Ray diffraction (XRD)
X-ray diffraction patterns were recorded using a PANalytical X’Pert PRO powder diffractometer using Cu Kα radiation (λ = 0.1541 nm). The working voltage and current were 40 kV and 40 mA. The patterns were collected over a 2θ range of 10° to 80° with a step of 0.0167°.
X-ray photoelectron spectroscopy (XPS)
Quasi in situ XPS spectra were collected using an Axis Ultra imaging photoelectron spectrometer (Kratos Analytical) equipped with Al Kα (1486.7 eV) quartz monochromator source. Prior to the measurement, the samples were reduced in a fixed-bed reactor with 15% CH4/H2 or 10% H2/N2 as mentioned above. After it was cooled to atmospheric temperature, the reactor was sealed and transferred into the glove box without exposure to air. The freshly prepared samples were clamped on a copper sample holder and transferred into the chamber of the XPS spectrometer under argon protection. Graphite carbon C1s signal of binding energy at 284.8 eV was used as the reference. The XPS spectra were processed using CasaXPS software.
Inductively coupled plasma optical emission spectroscopy (ICP-OES)
The actual content of supported Pd in catalysts was determined using inductively coupled plasma optical emission spectroscopy (ICP-OES, Varian 720).
Electron microscopy (EM)
The samples were sparsely dispersed in ethanol and subsequently deposited onto copper grids coated with holey carbon films, followed by desiccation for TEM observations. The Pd/α-MoC samples were characterized by STEM on the JEOL ARM300F2 Grand ARM2 operating at 200 kV with a convergence angle of around 32 mrad. The EDS and EELS mapping was acquired under the same setting. Pd/CeO2 and Pd/Al2O3 catalysts were characterized by TEM using a FEG-TEM instrument (Tecnai G2 F30 S-Twin) operating at 300 kV.
H-D exchange TPR
The H2-D2 exchange TPR experiments were conducted in a fixed-bed flow reactor. 20 mg catalysts (diluted with 80 mg SiO2) were placed in a quartz tube and reduced in 20 vol% H2/Ar (flow rate = 30 mL·min−1) at 400 °C for 2 h. After cooling to room temperature, the gas feed was switched to 10 % D2/Ar (10 mL·min−1), H2 (10 mL·min−1) or 5%CO/H2 (10 mL·min−1) or CO/H2/H2O (5%CO/H2 10 mL·min−1) until signals were stable. The H-D exchange behavior was recorded during a temperature-programed heating process with a ramp rate of 5 °C·min−1 from 25 °C to 250 °C. Mass spectrometry (OMNI Star™ Gas Analysis System) was used to analyze the signal of m/z = 2 (H2), 3 (HD), 4 (D2), 18 (H2O), 44 (CO2), 28 (CO), 16 (CH4), 40 (Ar). All the signals were normalized in respect to m/z = 40 to eliminate the variations of response in mass spectrometry.
Evaluation of catalytic performance
Hydrogenation of quinoline
Quinoline hydrogenation reactions were carried out in a stainless-steel autoclave (Laibei AE100) equipped with a pressure control system and an automatic temperature controller. In a typical process, 25 mg catalyst, 1 mmol quinoline, 1 mmol m-xylene as the internal standard, and 15 mL H2O were added to the autoclave. The stainless-steel autoclave was purged with nitrogen three times to remove residual air. After that, the autoclave was purged with mixed gas (5%CO/H2) three times to remove residual nitrogen and then pressurized to 2 MPa. The resulting mixture was vigorously stirred with a stirring rate of 800 rpm at a specified temperature for a set reaction time. At the end of the experiment, the autoclave was cooled to room temperature with a water bath. After reaction, 15 mL toluene containing 103.4 mg m-xylene internal standard was added into the autoclave, and then stirred for another 10 minutes to extract the residual substrate and products from water. The sample composition was analyzed by gas chromatography-mass spectrometry (GC-MS) and was quantified Agilent 8860 gas chromatograph, all are equipped with HP-5 column. The quantitative analysis was carried out with m-xylene as the internal standard, and the standard curves were established for the substances that may appear in the reaction (quinoline, 1,2,3,4-tetrahydroquinoline (py-THQ), 5,6,7,8-tetrahydroquinoline (bz-THQ), decahydroquinoline (DHQ)). According to the chromatographic analysis, the peak area of each product and the known internal standard m-xylene were substituted into the standard curve to obtain the product content. The carbon balance of the experimental results is in the range of 95–102 %.
The gas and liquid products were analyzed by gas chromatography (Agilent 8860) equipped with a flame ionization detector (FID) connecting to an HP-5 capillary column (0.32 mm in diameter, 30 m in length) and a thermal conductivity detector (TCD) connecting to Porapak Q and 5 A molecular sieve columns. Gas-phase products were analyzed by GC and detected by TCD. The liquid mixture was filtered using nylon membranes (0.22 μm), and the obtained liquid products were analyzed by GC and detected by FID.
The conversion of quinoline (χQ) and yield of the expected product THQ were determined by:
where mCat., wPd and MPd represent the mass of the catalyst (gcat), weight percentage of Pd (gPdgcat−1) and relative atomic mass of Pd (gPd molPd −1), respectively.
The calculation of theoretical CO concentration in Fig. 4b:
where nH2(before reaction), nH2(after reaction) and nCO(after reaction) could be acquired by GC-MS, while nTHQ could be detected in the analysis of liquid products as mentioned above.
Kinetic test
10 or 25 mg catalyst, 5 mmol quinoline, the corresponding molar amount of m-xylene (as the internal standard) and 15 mL H2O were mixed in the autoclave filled with 2 MPa of H2 or mixed gas (5%CO/H2) at a specified temperature. The reaction conversion was controlled less than 10% to guarantee the test was conducted in the kinetic regime. After reaction, the TOF would be calculated.
Stability test of the 3 wt%Pd/α-MoC catalyst for CO-resistant hydrogenation of quinoline
The stability test was carried out using the following conditions: 25 mg 3 wt%Pd/α-MoC, 1 mmol quinoline, 15 mL H2O, and 1 mmol m-xylene as the internal standard, 2 MPa of 5%CO/H2, 100 °C, 2 h. After each reaction, the catalyst was collected, washed with ethanol five times and subsequently dried in a vacuum freeze-dryer. And then, the used catalyst was activated and later used for the reaction again.
DFT
Calculation methods
All calculations were performed using the Vienna Ab Initio simulation package (VASP). The electron-ion interaction was described with the projector augmented wave (PAW) method46,47. The electron exchange and correlation energies were treated within the generalized gradient approximation in the Perdew-Burke-Ernzerhof formalism (GGA-PBE)48,49. The energy cutoff of plane wave basis was set to 450 eV, and the MP k-point sampling was utilized. According to the Methfessel-Paxton technique, the electron smearing width of σ = 0.2 eV was employed. For the transition states, the Climbing Image Nudged Elastic Band (CI-NEB) method was used4, and the stretching frequency analysis was performed to estimate whether a transition state associated with a solely imaginary frequency.
The binding energies of the metallic Pd and Pt atom, as well as the adsorption energies of the gaseous H2 molecule were calculated according to Eb = EX/slab – [Eslab + EX], where EX/slab is the total energy of the slab with adsorbates of the metallic atom or the gaseous molecule in its optimization configuration, Eslab is the total energy of the bare slab, and EX is the total energy of the free adsorbates in the gas phase. Therefore, the more negative the Eads, the stronger the adsorption. The energy barrier (Ea) was calculated according to Ea = ETS – EIS where EIS and ETS are the total energy of the corresponding initial state (IS) and transition state (TS), respectively.
Calculation models
The calculated lattice constant of a cubic MoC cell (fcc) is 4.332 Å, and the Mo-Mo bond length is 3.063 Å. For the top and side views of Pd1/α-MoC and Pt1/α-MoC models (Supplementary Fig. 16), the unit cell p(4×4) and the 3×3×1 k-point sampling was used to treat the models. In total, Pd1/α-MoC and Pt1/α-MoC has 1 Pd or Pt atom, 48 molybdenum and 48 carbon atoms, in which 16 molybdenum atoms and 32 carbon atoms are fixed. Compared with Pd1/α-MoC, there are x carbon atoms are adsorbed around the Pd atom for the Pd1Cx/α-MoC models.
Data availability
The data that support the plots within this paper and another finding of this study are available from the corresponding author upon reasonable request. Source data are provided with this paper.
References
Abe, J. O. et al. Hydrogen energy, economy and storage: Review and recommendation. Int. J. Hydrog. Energy 44, 15072–15086 (2019).
Staffell, I. et al. The role of hydrogen and fuel cells in the global energy system. Energy Environ. Sci. 12, 463–491 (2019).
Dincer, I. et al. Review and evaluation of hydrogen production methods for better sustainability. Int. J. Hydrog. Energy 40, 11094–11111 (2015).
Li, S. et al. Direct utilization of crude and waste H2 via CO-tolerant hydrogenation. Innovation 4, 100353 (2023).
Cao, L. et al. Atomically dispersed iron hydroxide anchored on Pt for preferential oxidation of CO in H2. Nature 565, 631–635 (2019).
Lin, L. et al. A highly CO-tolerant atomically dispersed Pt catalyst for chemoselective hydrogenation. Nat. Nanotechnol. 14, 354–361 (2019).
Cai, B. et al. Advanced Progress for Promoting Anodic Hydrogen Oxidation Activity and Anti-CO Poisoning in Fuel Cells. ACS Catal. 14, 13602–13629 (2024).
Byeon, J. et al. CO-tolerant electrocatalysts for hydrogen fuel cells: Fundamental study-based design and real-life applications. Chem. Eng. J. 493, 152626 (2024).
Hashimoto T., et al. Main group catalysis for H2 purification based on liquid organic hydrogen carriers. Sci. Adv. 8, eade0189 (2022).
Preuster, P. et al. Liquid organic hydrogen carriers (LOHCs): toward a hydrogen-free hydrogen economy. Acc. Chem. Res. 50, 74–85 (2016).
He, T. et al. Liquid organic hydrogen carriers. J. Energy Chem. 24, 587–594 (2015).
Sakuraba, M. et al. Recent Trends in Triarylborane Chemistry: Diversification of Structures and Reactivity via meta-Substitution of the Aryl Groups. Synthesis 56, 3421–3430 (2024).
Jorschick, H. et al. Hydrogenation of liquid organic hydrogen carrier systems using multicomponent gas mixtures. Int. J. Hydrog. Energy 44, 31172–31182 (2019).
Jeong, K. et al. Benzyl-methylbenzyl-benzene: Improving hydrogen storage and release performance of dibenzyltoluene based liquid organic hydrogen carrier. Chem. Eng. J. 488, 150927 (2024).
Zhang, X. et al. Regulating the d-band center of Pt for highly effective H2 storage through toluene hydrogenation at low temperatures. J. Mater. Chem. A 12, 23392–23397 (2024).
Wang, Z. et al. CO-tolerant RuNi/TiO2 catalyst for the storage and purification of crude hydrogen. Nat. Commun. 13, 4404 (2022).
Teichmann, D. et al. Energy storage in residential and commercial buildings via Liquid Organic Hydrogen Carriers (LOHC). Energy Environ. Sci. 5, 9044–9054 (2012).
Yao, J. et al. Pt/Al2O3 as efficient catalyst for the dehydrogenation of Dodecahydro-N-ethylcarbazole. Chem. Eng. J. 491, 152100 (2024).
Naveen, K. et al. MOF-derived Co/Zn single-atom catalysts for reversible hydrogenation and dehydrogenation of quinoline hydrogen carrier. Appl. Catal., B 328, 122482 (2023).
Zhang, W. et al. Ambient-condition acetylene hydrogenation to ethylene over WS2-confined atomic Pd sites. Nat. Commun. 15, 9457 (2024).
Zhai, Y. et al. Alkali-Stabilized Pt-OHx Species Catalyze Low-Temperature Water-Gas Shift Reactions. Science 329, 1633–1636 (2010).
Parvez, S. et al. Substoichiometric Hydroxynonenylation of a Single Protein Recapitulates Whole-Cell-Stimulated Antioxidant Response. J. Am. Chem. Soc. 137, 10–13 (2015).
Zhao, X. et al. Breaking the Trade-Off between CO Tolerance and Intrinsic Activity in Hydrogenation on Metal Oxide-Supported Noble Metal Single Atoms through Coordination Environment Engineering. ACS Catal. 14, 15325–15335 (2024).
Liu, J. et al. Tackling CO Poisoning with Single-Atom Alloy Catalysts. J. Am. Chem. Soc. 138, 6396–6399 (2016).
Briquet, L. G. V. et al. Platinum Group Metal Adsorption on Clean and Hydroxylated Corundum Surfaces. J. Phys. Chem. C. 113, 16747–16756 (2009).
Lin, L. et al. Low-temperature hydrogen production from water and methanol using Pt/α-MoC catalysts. Nature 544, 80–83 (2017).
Wei, Y. et al. Hydrogenation of Nitroarenes by Onsite-Generated Surface Hydroxyl from Water. ACS Catal. 13, 15824–15832 (2023).
Huang, R. et al. Acetylene hydrogenation to ethylene by water at low temperature on a Au/α-MoC catalyst. Nat. Catal. 6, 1005–1015 (2023).
Lin, L. et al. Heterogeneous catalysis in water. JACS Au 1, 1834–1848 (2021).
Ma, Y. et al. Molybdenum carbide clusters for thermal conversion of CO2 to CO via reverse water-gas shift reaction. J. Energy Chem. 50, 37–43 (2020).
Cao, S. et al. Synergizing Mon Clusters and Mo2C Nanoparticles on Oxidized Carbon Nanotubes Boosting the CO2 Reduction Activity. ACS Catal. 14, 10939–10950 (2024).
Ahmadi Khoshooei, M. et al. An active, stable cubic molybdenum carbide catalyst for the high-temperature reverse water-gas shift reaction. Science 384, 540–546 (2024).
Lin, L. et al. Reversing sintering effect of Ni particles on γ-Mo2N via strong metal support interaction. Nat. Commun. 12, 6978 (2021).
Liu, H.-X. et al. Ptn–Ov synergistic sites on MoOx/γ-Mo2N heterostructure for low-temperature reverse water–gas shift reaction. Nat. Commun. 13, 5800 (2022).
Bi, Q. et al. One-Step High-Temperature-Synthesized Single-Atom Platinum Catalyst for Efficient Selective Hydrogenation. Research 2020, 1–10 (2020).
Li, S. et al. Atomically dispersed Ir/alpha-MoC catalyst with high metal loading and thermal stability for water-promoted hydrogenation reaction. Natl Sci. Rev. 9, nwab026 (2022).
Hu, X. et al. Pd-Supported N/S-Codoped Graphene-Like Carbons Boost Quinoline Hydrogenation Activity. ACS Sustain. Chem. Eng. 7, 11369–11376 (2019).
Lu, Y. et al. Unsupported nanoporous palladium-catalyzed chemoselective hydrogenation of quinolines: Heterolytic cleavage of H2 molecule. Chin. J. Catal. 39, 1746–1752 (2018).
Chastain, J. et al. Handbook of X-ray photoelectron spectroscopy. Perkin-Elmer Corporation 40, 221 (1992).
Yao, S. et al. Atomic-layered Au clusters on alpha-MoC as catalysts for the low-temperature water-gas shift reaction. Science 357, 389–393 (2017).
Zhang, X. et al. A stable low-temperature H2-production catalyst by crowding Pt on alpha-MoC. Nature 589, 396–401 (2021).
Deng, Y. et al. Solvent tunes the selectivity of hydrogenation reaction over alpha-MoC catalyst. J. Am. Chem. Soc. 140, 14481–14489 (2018).
Lin, L. et al. Atomically Dispersed Ni/α-MoC Catalyst for Hydrogen Production from Methanol/Water. J. Am. Chem. Soc. 143, 309–317 (2021).
Li, S.-S. et al. Efficient and exceptionally selective semireduction of alkynes using a supported gold catalyst under a CO atmosphere. Chem. Commun. 50, 5626–5628 (2014).
Messmer, R. P. et al. The interaction of atomic hydrogen with Ni, Pd, and Pt clusters. Chem. Phys. Lett. 51, 84–89 (1977).
Kresse, G. et al. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Kresse, G. et al. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Kresse, G. et al. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Acknowledgements
This work was financially supported by the Natural Science Foundation of China (No. 22278367 and 22172083), Zhejiang Provincial Natural Science Foundation of China (LR22B030003), and Natural Science Foundation of Inner Mongolia Autonomous Region of China (No. 22172083). We acknowledge the Analytical Instrumentation Center of Peking University for quasi-situ XPS measurement.
Author information
Authors and Affiliations
Contributions
D.M. and L.L. designed the study. X.J. and X.L. performed most of the reactions and did most of the analysis. R.G. did the DFT calculations. C.L. did the measurement and analysis of XPS. S.Y. and G.S. carried out the electron microscopy analysis. X.Y. did the analysis of GC-MS. C.S. did the reaction mechanism analysis. L.L. and X.L. wrote the paper. All authors performed certain experiments, discussed and revised the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liang, X., Jin, X., Yu, S. et al. CO-resistant hydrogenation over noble metal/α-MoC catalyst. Nat Commun 16, 4159 (2025). https://doi.org/10.1038/s41467-025-59321-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-59321-0
This article is cited by
-
Advances in hydrogenation catalysis by porous materials supported palladium nanoparticles
Frontiers of Materials Science (2026)