Abstract
Large-scale genomic studies focusing on the genetic contribution to human aging have mostly relied on cross-sectional data. With the release of longitudinally curated aging phenotypes by the UK Biobank (UKBB), it is now possible to study aging over time at genome-wide scale. In this work, we evaluated the suitability of competing models of change in realistic simulation settings, performed genome-wide association scans on simulation-validated measures of age-related deweekcline, and followed up with LD-score regression and Mendelian Randomization (MR) analyses. Focusing on global cognitive and physical function, we observed marked differences between baseline function (θ) and accelerated decline (Δ). Both outcomes showed distinct heritability levels (e.g., 31.38% \({h}_{\theta }^{2}\) versus 3.15% \({h}_{\Delta }^{2}\) for physical function) and different associated loci (e.g., DUSP6 specific to physical Δ). Further, we found little commonalities across the two dimensions of aging—while cognitive decline was largely driven by Alzheimer’s disease liability (standardized MR-effect, γ = 0.17), physical decline was mostly impacted by telomere length (γ = −0.05) and bone mineral density (γ = −0.05). Our work highlights the utility of longitudinal genomic efforts to scrutinize age-dependent genetic and environmental effects on physical and cognitive outcomes. Careful modelling and attention to participation characteristics are, however, crucial for valid inference.
Similar content being viewed by others
Introduction
Preserving cognitive and physical health in the global aging population is a major public health priority. Substantial resources are therefore invested into research scrutinizing modifiable risk factors of age-related decline1,2, ultimately aiming to optimize preventative interventions that can slow down aging and delay the onset of functional impairment in the elderly. To that end, the genetic contribution to human aging is increasingly studied, holding promise to directly (e.g., via molecular/pharmacological targets) or indirectly (e.g., via environmental targets) provide novel insights into treatment and prevention. The emergence of large-scale biobanks in particular has moved the field forward, advancing our understanding of individual differences underlying indexes of aging (e.g., longevity3, healthspan4, frailty5,6, biological aging7,8,9).
Although decline in cognitive and physical dimensions is considered part of normal aging, marked variation characterizes the rate and timing of age-related decline10. Yet, the genetic contribution to such heterogeneity remains poorly understood, possibly echoing the limited power for genome-wide discovery in existing prospective cohorts11,12,13,14, or potential biases affecting cross-sectional research on age-varying genetic effects (e.g., birth cohort effects15 or age-dependent study participation16). Large-scale biobank initiatives committed to longitudinal assessments therefore represent an unprecedented resource for the study of aging processes, enabling a move from cross-sectional (e.g., level of grip strength) genome-wide analyses to an estimation of longitudinal (e.g., loss in grip strength from that point) genetic effects. For that purpose, the UK Biobank (UKBB) has curated and released a rich set of prospectively ascertained aging phenotypes17, capturing key aspects of changes in physical (e.g., forced expiratory volume (FEV), fitness levels, grip strength) and cognitive (e.g., reaction time, fluid intelligence) dimensions. This resource now enables the investigation of prominent aging theories within a genetically informed framework, including common cause theories of aging18,19,20,21 or the role cognitive/physical reserve in age-related decline22,23,24,25,26,27,28 (c.f., ‘Research in context’ in the Supplement for further discussion).
Despite the promise as a powerful resource for the study of human aging, the analysis and interpretation of findings obtained from large prospective biobank samples present several challenges. First, selective participation already documented at the initial recruitment stage in the UKBB29,30,31 may be exacerbated in longitudinal samples due to selective attrition and survival, where prospectively ascertained individuals represent an even healthier subset of the initially healthy32. Assessing the robustness of findings to selective attrition therefore constitutes a necessary step when making inferences from non-representative prospective samples. Second, while the recruitment of individuals with at least two-wave data (i.e., baseline and one follow-up) is moving towards its goal of 100,000 individuals in the UKBB33, three-wave data (or more) is currently only available for a small fraction of the sample. As such, maximizing the number of individuals inevitably limits the number of follow-up waves, restricting longitudinal analyses to two-wave models of change that are not free of criticism34. While different change models have been proposed and applied in classic epidemiological35,36 and genetic37,38,39,40 studies, there is currently little consensus as to which approach (if any) is most appropriate for the discovery of longitudinal genetic effects. To minimize the risk of inferential errors, a deeper understanding of behaviours and limitations of commonly used two-wave models of change is therefore critical prior to their application in genome-wide scans.
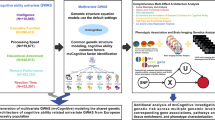
In summary, this work aims to exploit the prospective UKBB resource to scrutinize genetic and environmental contributions to physical and cognitive aging. To that end, we propose and apply an analytical framework (c.f., Figs. 1 and 2 for illustration) tailored to the analysis of change at genome-wide scale, while accounting for design-challenges inherent to prospective volunteer samples.
Analytical framework to study longitudinal genetic effects on phenotypes subject to age-related decline. The structural causal model is shown at the top, where the phenotype P at time point t (P0 = baseline, P1 = follow-up) varies as a function of time-invariant (baseline) genetics (G0), time-varying (longitudinal) genetics (GE) and the environment (E). Simulations are used (middle panel) to assess the suitability of three definitions of change (Δ) for longitudinal genome-wide analyses (Δ ~ γ ⋅ G), including absolute change (ΔDIFF, i.e., the absolute difference between the baseline phenotype, P0, and the follow-up phenotype, P1), relative change (ΔLOG, i.e., the difference between \(\log ({P}_{0})\) and \(\log ({P}_{1})\)) and conditional change (ΔRES, i.e., the difference between the observed P1 and the predicted \(\hat{{P}_{1}}\) phenotype). Genome-wide tests and downstream analyses (bottom panel) are performed on composite scores of cross-sectional (i.e., time-invariant) and longitudinal (i.e., time-varying) indexes of cognitive and physical aging.
The observed phenotype P* at time point t (0 = baseline, 1 = follow-up) was modelled as a function of time-invariant (baseline) genetics (G0), time-varying (longitudinal) genetics (GE) and the environment (E). All arrows labelled with Greek letters represent the structural coefficients of the underlying data-generating process, where α is the (main) baseline genetic effect, β the (main) environmental effect and γ the gene-by-environment effect. εt is the measurement error in the phenotype.
Results
Indexes of age-related decline in cognitive and physical function
We first assessed if the selected measures of cognitive and physical function showed the expected age-related decline, by estimating the effects of age on each of the 17 aging phenotypes in univariate linear regression analyses. For that, the physical and cognitive measures were transformed into standardized z-scores (\(\frac{{P}_{0}-{\mu }_{0}}{{\sigma }_{0}}\), where μ0 and σ0 are the mean and standard deviation of the baseline phenotype, P0, respectively). Thereby, a negative α-coefficient represents decline in standard deviations of a particular measure per additional year of age. As shown in Fig. 3, Supplementary Data 1 and Supplementary Fig. 1, all measures showed the expected age-related decline. However, the magnitude of age effects varied considerably across traits and UKBB sub-samples (mean α = −0.02, ranging from −0.06 to −0.003). For example, steeper age-related decline was, on average, present for physical (mean α = −0.03, SE = 0.005) than cognitive abilities (mean α = −0.02, SE = 0.005). The largest effect of age was observed for psychomotor abilities (symbol digit substitution test), where the test performance decreased by -0.06 SD on average per additional year of age. Of note, while these results are interpreted as age effects, alternative factors (e.g., birth cohort effects41) may also contribute to the observed between-subject differences across age groups. Further, when examining UKBB sub-samples with varying degrees of representativeness, we found that more selective samples exhibited a less pronounced decline. As illustrated in Fig. 3, selective participation resulted in attenuated age effects (Panel A/B), where attenuation bias aggravated with increasing rates of loss to follow-up and non-representativeness (Panel C). The implications of selective participation and attrition are further discussed in the Supplement (Supplementary Discussion).
A Each dot represents the mean phenotype score assessed at baseline (y-axis) per 2 year age bin (x-axis) across UKBB samples with varying levels of representativeness. Age effects (c.f., labels to the right) on cognitive and physical measures assessed at baseline were obtained from linear regression models, applied in (a) the unweighted UKBB baseline sample (‘baseline sample, 1-wave’), (b) the inverse probability weighted UKBB baseline sample (‘weighted 1-wave’), (c) the UKBB follow-up sample with complete data in at least one follow-up assessment (‘follow-up sample, 2-waves’) and (d) the UKBB follow-up sample with complete data in at least two follow-up assessments (‘follow-up sample, 3-waves’). Individual plots for all cognitive and physical decline phenotypes are included in Supplement Supplement Tables 2--3. B Summary of age effects (with 95% confidence intervals) on cognitive and physical baseline phenotypes, obtained from the four UKBB sub-samples (a-d). Phenotypes shown in this figure include those with little missing data when assessed at baseline (i.e., at least 450,000 UKBB participants). The negative αSTD-coefficient represents decline in standard deviations in the outcome per additional year of age. C plots the differences in age effects (y-axis) obtained from the baseline UKBB sample (α0, unweighted baseline sample in grey, weighted baseline sample in black) and the UKBB follow-up sample (α1) for the phenotypes shown in (B). The x-axis indicates the attrition rate per phenotype. The two plotted lines are the lines of best fit. Source data are provided in Supplementary Data 1.
Slopes of global cognitive and physical decline were derived from 8 aging phenotypes, covering between 1 and 18 years of follow-up (mean = 7.8) (c.f., Supplementary Figs. 2–3 and Supplementary Data 2 for phenotype-specific characteristics). Complete data on global cognitive and physical decline was available for 123,194 and 85,502 individuals, respectively. Supplementary Fig. 4 shows the correlations between the slopes of decline in cognitive and physical measures. While most slopes were positively correlated across the different aging phenotypes, the coefficients were substantially smaller compared to the correlations among the cross-sectional phenotype pairs (Supplementary Fig. 5).
Based on our simulation work (c.f., Supplementary Figs. 6–7 and Supplementary Discussion), we found that baseline-adjusted change (ΔRES) introduces bias by falsely associating cross-sectional (time-invariant) genetic effects with change. Conversely, models capturing absolute (ΔDIFF) and relative change (ΔLOG) are more robust in distinguishing cross-sectional from longitudinal genetic effects under various realistic conditions. In subsequent analyses, we therefore prioritized definitions of absolute and relative change when scrutinizing longitudinal genetic effects.
Longitudinal and cross-sectional genetic variant effects on cognitive and physical function
Longitudinal (ΔLOG, ΔDIFF) and cross-sectional (P0) genome-wide tests were performed on the composite scores indexing global physical and cognitive function, in addition to the 8 individual aging phenotypes used to derive the scores. In total, 10,338 independent genome-wide variants were identified, including 7 associated with longitudinal decline and 10,331 with cross-sectional function.
The top LD-independent variant associated with relative and absolute cognitive decline (N = 103,938) included a missense variant in APOE (rs429358), where each additional copy of the T allele showed protective effects on cognitive decline (e.g., βLOG = −0.03, P = 4.3e-19). The same variant also reached genome-wide significance in tests on baseline function (N = 404,449), albeit smaller effect magnitude (increasing cognitive function, with \({\beta }_{{P}_{0}}=\) 0.02, P = 3.2e-11) (Fig. 4). rs117041440 (closest gene: KLF4) showed more Δ-specific effects, associating with reduced decline in fluid intelligence (βLOG = −0.07, P = 1.9e-08) but not baseline fluid intelligence at genome-wide significance (βP0 = −0.02, P = 0.036) (Supplementary Fig. 8).
Effects estimates of variants reaching genome-wide significance in association tests on either cross-sectional physical/cognitive functioning (x-axis) or cognitive/physical decline (y-axis). The colour scheme highlights variants associated with either cross-sectional (P0 in red), longitudinal (ΔLOG in orange, ΔDIFF in blue) or both (in black) outcomes. The dashed slope (line of best fit, obtained from βΔ ~ βP0) represents the association between the cross-sectional and longitudinal SNP effects. The SNP-heritability estimates (h2, with 95% confidence intervals) obtained from longitudinal and cross-sectional genome-wide analyses are shown to the right of each panel. Source data are provided in Supplementary Data 3.
One variant (rs113645269, closest gene: DUSP6) was identified for global (relative) physical decline, increasing physical decline by βLOG = 0.09 standard deviations per additional copy of the G allele (P = 2.5e-08, N = 72,220). This variant was specific to decline and did not impact baseline physical function (P = 0.6, N = 405,979). Similarly, rs190141474 (closest gene: MNX1) associated with less relative decline in FEV (βLOG = −0.14, P = 2.3e-09, N = 44,048) but not baseline FEV (P = 0.32, N = 373,397). Both variants have no documented genome-wide associations with previously studied traits (c.f., PheWAS plots, Supplementary Fig. 9).
Among the six phenotypes assessed in interaction testing (cross-sectional physical and cognitive function, absolute cognitive and physical decline, and relative cognitive and physical decline), two genetic variants exhibited sex-specific effects: rs13141641 (nearest gene: HHIP) and rs9748016 (RFLNB), both of which were identified in the genome-wide tests on baseline physical function. Mapping these variants to prior phenotype-genotype associations revealed that these sex-dependent genetic effects mostly linked to indices of physical health, such as lung function and heel bone mineral density (Supplementary Fig. 10).
We found only negligible h2-estimates on measures of decline, ranging from 0.03% to 1.2% for measures of cognitive decline and 0.98% to 3.15% for measures of physical decline. Significant h2-estimates (P < 0.05) on change were present for global physical decline (\({h}_{DIFF}^{2}=\) 3.2% and \({h}_{LOG}^{2}=\) 2.7%), decline in height (\({h}_{DIFF}^{2}=\) 2.4% and \({h}_{LOG}^{2}=\) 2.3%), decline in grip strength (\({h}_{DIFF}^{2}=\) 1.4%), decline in FEV (\({h}_{DIFF}^{2}=\) 2.2%) and decline in fluid intelligence (\({h}_{DIFF}^{2}=\) 0.9%). Heritability estimates obtained for measures of baseline functioning were larger in all instances (Fig. 4), ranging from 13.5% – 52.7% for physical function, and from 6.6% – 20.9% for cognitive function.
In line with our simulation results, longitudinal genetic estimates obtained from baseline-adjusted change (ΔRES) falsely captured substantial parts of the baseline genetic effects, leading to an inflation in genomic signal (Supplementary Fig. 8, in grey). Here, the impact of adjustment-bias was particularly prominent for global cognitive decline, evident by the excess in genome-wide identified variants (kRES = 6 versus kLOG = 1) and SNP-heritability (\({h}_{RES}^{2}=\) 6.3% versus \({h}_{LOG}^{2}=\) 0.2%). A complete summary of the genome-wide results is included in Supplementart Data 3.
Finally, we performed weighted genome-wide association analyses to correct for possible bias resulting from selective follow-up participation. Supplementary Fig. 11 shows that bias was present in both directions, leading to over-estimation (e.g., APOE-effects on cognitive-Δ) and under-estimation (e.g., DUSP6-effects on physical-Δ) of variant effects. Across all tested variants, we did not, however, find evidence of altered direction of effects.
Risk factors of longitudinal and cross-sectional cognitive and physical function
Mendelian Randomization (MR) analysis was used to identify causal factors involved in age-related decline. Contrasting results from phenotypic association tests to MR-estimates (c.f., Supplementary Fig. 12) indicated that many factors reaching significance in phenotypic analyses (133 out of 228 exposure-outcome associations with P < 0.05) showed smaller and non-significant effects when tested in MR, possibly reflecting the influence of confounders and reduced statistical power. More specifically, only few exposures showed substantial causal effects on decline (e.g., shorter parental lifespan), and more common were influences of smaller magnitude that were specific to either cognitive or physical decline (Fig. 5). For example, while cognitive decline was mostly predicted (at nominal significance level) by Alzheimer’s disease, lipid traits (e.g., Apolipoprotein A and B) and behaviours potentially altering those (e.g., vegetable intake), there was little overlap with risks identified for physical decline (e.g., shorter telomere length, higher bone mineral density, basal metabolic rate, poor sleep). 10 exposures on cognitive/physical decline survived suggestive Bonferroni correction for multiple testing (P < 0.05/ 11 = 0.005, were 11 is the number of independent exposure dimensions) (c.f., triangle shapes in Fig. 5), of which 6 (highlighted in rectangular shapes) remained significant following stringent Bonferroni correction (P < 0.05/ 106 = 0.0005, were 106 is the number of exposures tested in MR). Of note, most of the stringently identified risk factors pointed towards scale-dependent conclusions. For example, higher baseline physical function predicted steeper physical decline when assessed in absolute (γ = 0.1, P = 8.1e-14) but not relative (γ = 0.02, P = 0.16) terms, a pattern consistent with the presence of non-linear change. Further, parental lifespan (PLS) on cognitive decline was flagged as possibly biased based on a number of MR sensitivity analyses, given the level of heterogeneity (Q-test P = 3.2e-08), the significant MR-Egger intercept term (P = 1.5e-05 for PLS → ΔLOG) and the non-significant MR-PRESSO effects (e.g., P = 0.23). The complete set of MR-results, including the exposure-outcome associations when performing MR on the individual aging phenotypes, is included in Supplementary Data 4 and Supplementary Figs. 13–14.
Standardized Mendelian Randomization effect estimates (βSTD, with 95% confidence intervals) of exposure effects on cross-sectional outcomes (P0 in red, with positive coefficients indexing higher levels of function) and longitudinal outcomes (ΔDIFF in blue and ΔLOG in orange, with positive coefficients indexing larger decline). Filled points, triangles and diamonds highlight significant effects (at P < 0.05, P < 0.05/11 and P < 0.05/106, respectively). Circles highlight non-significant (P > 0.05) effects. Source data are provided in Supplementary Data 4.
Further, we found little evidence of common underpinnings shared by (cross-sectional) level of functioning and (longitudinal) decline in cognitive and physical outcomes, implicated by the lack of association between cross-sectional MR-effects (γP0) and longitudinal MR-effects when modelled as relative change [Pearson correlation coefficient r(γP0, γLOG) = −0.06, P = 0.56] or absolute change [r(γP0, γDIFF) = 0.03, P = 0.72]. Shared liabilities were, however, observed across cognitive and physical domains within cross-sectional analyses, notably for risks relating to lifestyles (e.g., time spent watching television), brain health (e.g., volume of thalamus) and social factors (e.g., income, education). Other exposures appeared more specific with respect to the outcome dimension, where risk factors tapping into mental health (e.g., ADHD, bipolar disorder, schizophrenia) were more specific to cognitive function, and biomarker traits (e.g., creatinine levels, sex hormone binding globulin, insulin-like growth factor 1) mostly to specific physical function.
Discussion
Large-scale longitudinal biobank samples such as the UK Biobank (UKBB) have the potential to advance our understanding of the genetic and environmental contributions to aging. In this work, we exploited the prospectively ascertained UKBB sample to separate cross-sectional from longitudinal cognitive and physical function within a genome-wide framework, while evaluating the impact of design-constraints inherent to longitudinal biobank schemes. A more comprehensive discussion complementing the findings described below is available in the section ‘Research in Context’ in the Supplement.
We first evaluated three commonly used definitions of change for two-wave prospective data (i.e., absolute, conditional, relative change). Simulation work implicated that relative and absolute change can robustly distinguish longitudinal from cross-sectional genetic effects in realistic simulation settings, without showing susceptibility to biases affecting baseline-adjusted change scores. Exacerbation of false-positives via baseline adjustment occurs in situations where a genetic variant links to the baseline phenotype, resulting in bias that is proportional to the baseline genetic effects and the measurement error in the phenotype. Our recommendations are therefore in line with existing discussions37,38,39 in discouraging the use of baseline-adjusted change for longitudinal genetic effect estimation. With respect to absolute/relative change, inconsistent results (e.g., male sex linking to increased absolute but decreased relative physical decline) can occur in situations where change is non-linear (e.g., exponential). The choice between modelling absolute vs relative change will therefore alter the interpretation of the results: If the priority lies in isolating change from possible baseline dependencies, approaches of relative change can help to account for non-linear change. Alternatively, absolute change may be preferred if a quantification of change on the original scale is of particular relevance (e.g., variant effects on kg-change following intervention42,43). More comprehensively, researchers may choose to model both absolute and relative change, to facilitate the interpretation of identified variant effects and identify possible scale-dependencies.
Moving forward with models of relative/absolute change, we performed genome-wide tests to assess the genetic contribution to cross-sectional and longitudinal phenotypes of aging. APOE was identified as the top gene associated with accelerated cognitive decline and, to a lesser extend, with lower levels of cognitive function. Since APOE has already been linked to numerous measures of age-related change and cross-sectional functioning (e.g., cognitive ability44,45,46,47/decline14,44,45,47,48, brain levels/change49, Alzheimer’s disease risk/progression50,51, BMI levels/change52,53, biomarker levels/change38,54), together these findings highlight that APOE is neither trait nor state specific. Together with evidence from MR implicating lipid traits as causal factors involved in cognitive decline (further discussed below), focusing further on lipids may therefore prove a promising avenue for future research into cognitive health55,56,57. DUSP6 (Dual Specificity Phosphatase 6) and MNX1 (Motor neuron and pancreas homeobox 1) were implicated in different proxies of physical decline. Both genes are commonly studied in the context of cancer progression (e.g., pancreatic58, lung59, colorectal60, leucemia61) and immune system responses62, suggesting that these variants may increase sensitivity to environmental stressors. More broadly, the identified variant effects may represent gene-environment interactions, where the genetic effects differ across changing environments as individuals age. While gene-by-environment studies typically examine interactions of a genotype with defined environmental exposures (e.g., pharmacological intervention38, lifestyles63,64,65, environmental toxicants64), here we capture (any) environmental change occurring during the aging process. Leveraging prospective data to examine gene-by-age effects in this context offers a particularly robust solution, which avoids common sources of bias (e.g., birth cohort effects) that complicate the interpretation of cross-sectional interaction effects.
Despite the relatively large sample size, only few genetic variants showed significant age-dependent effects. While this could reflect insufficient power to detect small effects, our findings point towards a lack of major genetic influences on within-individual decline. For example, cross-sectional cognitive and physical function showed orders of magnitude higher levels of SNP-heritability compared to age-related decline, consistent with what would be expected based on previous genome-wide works on within-individual change (e.g., h2 < 5% for BMI52,53, brain function49, drug response38). As such, our results suggest that unmodelled gene-by-age interactions are unlikely to account for substantial portions of missing trait-heritability and population heterogeneity among cognitive and physical measures of aging. Of note, while indexes of decline likely contain more measurement error than the baseline measures from which they are derived66, thereby inducing downward bias in h2-estimates67, this attenuation bias is unlikely to fully explain the stark differences in variance components (e.g., h2 for baseline height = 52.71% versus \({h}_{\Delta }^{2}\) for reductions in height = 2.35%). Instead, our observations imply that environmental factors may play a more significant role in driving within-person aging processes, in line with twin-study evidence attributing mostly non-shared environmental sources to variations in the rate of change68,69.
Mendelian Randomization was used to further disentangle risk factors of accelerated decline, defined as a more rapid deterioration in physical and cognitive function over time, yielding three main insights: First, we observed little commonalities across the two dimensions of decline - while Alzheimer’s disease liability and shorter parental lifespan constituted the main risk factors of accelerated cognitive decline, physical decline was predicted by a number of biological factors amenable to lifestyle and/or environmental interventions (e.g., basal metabolic rate, telomere length, bone mineral density). This contrasts findings from cross-sectional MR, where levels of cognitive and physical function showed shared aetiologies with overall larger exposure effects. Second, our findings implicate that high baseline function in a given trait (e.g., cognition), or factors shaping that trait (e.g., educational attainment), may not serve as a buffer against age-related decline in that trait. In other words, while high cognitive or physical reserve can delay the onset of functional impairment, it is unlikely to provide additional protective effects by slowing the rate of decline, in line with previous observations70,71,72,73,74,75,76.
The presented results should be interpreted in light of a number of limitations (see also Supplement for an extended discussion on the study limitations). First, age-related change was assessed using data from only two time points per individual, which reduces measurement precision compared to approaches utilizing more intensive longitudinal data. Second, despite the large sample size, statistical power likely remains an issue. Genetic interaction effects are inherently small and harder to detect compared to marginal effects77, which may have hindered the identification of genetic variants and causal factors of decline. Third, the predictability of age-related decline was generally low, given the number and effect sizes of the exposures identified in MR. Since our genetically informed framework only tests for lifetime risk factors, non-instrumentable time-varying environmental exposures previously implicated in age-related decline are therefore not captured in this work, examples of which include life events (e.g., loss of spouse78), age-related biological changes (menopausal status79), toxic environmental exposures (e.g., air pollution80) or changes in medication use81. As such, while barriers to risk-identification may reflect insufficient power to detect small exposure effects in MR, an alternative scenario is that unmodelled time-varying environmental factors play an important role in aging processes. Finally, implementing strategies designed to increase sample representativeness (Inverse Probability Weighting, IPW), we explored the impact and direction of bias resulting from selective participation. The results implicated that selective participation influenced both phenotypic and genotypic estimates (c.f., Supplementary Discussion). Therefore, boosting retention rates in future follow-up assessments will be crucial to minimize existing attrition biases. This is particularly important as statistical tools designed to probe the robustness of findings (e.g., IPW) reach a bottleneck in highly non-representative sample, where genome-wide discovery is hampered by the substantial loss in (effective) sample size when performing bias-corrected genome-wide tests.
In summary, our findings suggest that largely distinct genetic and environmental mechanisms characterize levels of function and decline in indexes of physical and cognitive function. Combining cross-sectional with longitudinal genomic efforts therefore holds promise for discovery of additional preventative targets that can help push back the age at which functional impairment begins. Importantly, design and analytical limitations of longitudinal genomic efforts can threaten the validity of findings, such as inferential errors resulting from inappropriate modelling of change or bias induced by selective attrition. While these constraints should ideally be addressed at the design stage of a study, here we provide a conceptual and analytical framework to guide longitudinal genomic initiatives when these conditions are not met. We conclude that large-scale prospective biobank data represent a powerful resource that comes with unique opportunities and challenges when studying the genetic basis of dynamic processes.
Methods
UK Biobank longitudinal assessments and slopes of age-related decline
The methodological framework of this work is illustrated in Fig. 1. We used data from the UK Biobank (UKBB), a large population-based cohort of >500,000 adults aged between 40–69 at baseline. The UKBB was approved by the National Health Service North-West Center Research Ethics Committee (REC No. 16/NW/0274) and this research was conducted under application number 16389. Between 2006 and 2010, UKBB participants completed a battery of cognitive and physical tests when attending the baseline assessment in one of the 22 research centres located across the United Kingdom. Since then, most tests were re-administered at varying follow-up intervals and in different subsets of the UKBB. The first repeat assessment centre visit took place (on average) in 2013 and included around 20,000 participants living within 35 km of the Stockport Biobank coordinating centre (21% response rate to the email/letter invitation82). The second repeat assessment centre visit (ongoing) started in 2014 and includes the first brain magnetic resonance imaging assessment, inviting back around 100,000 of the original volunteers83. The most recent assessment centre visit (ongoing) aims to re-invite 60,000 individuals with existing imaging data to take part in repeat imaging between 2022 and 202884.
Supplementary Fig. 15 lists the 17 aging phenotypes initially selected for longitudinal analyses in this work, plotting the number of available follow-up assessments per measure, the environment in which it was obtained (assessment centre versus online) and the number of participants with complete multi-wave data. Of note, despite the availability of a number of self-report measures relevant for aging (e.g., walking pace, physical activity), we only included objectively ascertained phenotypes. As self-report measures are prone to (age-related) misreporting and measurement error67,85,86, which reduces power for gene discovery and creates challenges for models of change87, these measures were not included in this work. In total, we selected 6 physical measures and 11 cognitive measures (c.f., Supplement for a more detailed description). Supplementary Fig. 15 and Supplementary Data 5 list the number of participants with non-missing longitudinal data per phenotype. While up to four waves of data (i.e., baseline and three follow-up assessments) are collected, complete four-wave data was only available for a small fraction of individuals (between 19 to 18,917 participants, mean = 2654) and was therefore not considered for longitudinal genome-wide analyses. Similarly, since the number of individuals with complete three-wave data was small (between 527 and 69,801 participants, mean = 16,350), we restricted all longitudinal analyses to individuals with non-missing two-wave data (up to 161,821 individuals, mean = 66,655). Of note, as three-wave data for fluid intelligence was available for a relatively large subset (69,801 participants), we explored possible gains resulting from adding one more time point (c.f., Supplementary Discussion).
Moving forward with two-wave models of change to derive the individual indexes of decline, we evaluated (c.f., simulation work in the Supplement), applied and compared the results obtained from three competing definitions of change38, including (1) difference scores (ΔDIFF), (2) residual change scores (ΔRES) and (3) log-difference scores (ΔLOG). All change scores were adjusted for the follow-up duration (details below) and computed such that higher values represent increased age-related decline:
-
(A)
Absolute change, using difference scores (ΔDIFF), derived by subtracting the follow-up phenotype from the baseline phenotype (ΔDIFF = P0 − P1)
-
(B)
Conditional change, using residual change scores (ΔRES), derived by regressing the baseline phenotype out of ΔDIFF, thereby indexing absolute change that is no longer predictable by the observed baseline scores.
-
(C)
Relative change, using log-difference scores (ΔLOG), computed as the difference between the natural log-transformed baseline and follow-up phenotype (ΔLOG = log(P0) − log(P1)).
Indexes of decline (Δ) were generated for all aging phenotypes meeting the criteria for inclusion: First, we included only phenotypes with at least 40,000 non-missing longitudinal (i.e., two-wave) observations. Second, to avoid problems resulting from poor measurement reliability, for example as documented for some of the cognitive measures88 in the UKBB, we also discarded phenotypes showing poor consistency across time (i.e., r < 0.4, where r is the average correlation across measurement occasions per phenotype, c.f., Supplementary Figs. 16–17). The box and and scatter plots of the selected cognitive and physical measures are shown in Supplementary Figs. 2–3. For all selected phenotypes, we then extracted the residuals from a model regressing change (Δ, defined as either ΔDIFF, ΔRES or ΔLOG) on age and the follow-up (FU) duration (\(\Delta={{\mbox{age}}}_{0}+{\mbox{FU}}+{{\mbox{age}}}_{0}\times {\mbox{FU}}+{{\mbox{age}}}_{0}^{2}\)). FU was defined as FU = age1 − age0, where age0 and age1 correspond to the age at which P0 and P1 were obtained, respectively. \({\,{\mbox{age}}}_{0}^{2}\) was included in our models to account for possible non-linear rates of change, i.e., situations where the effect of age on within-individual change increases over time. In the last step, the composite scores for global physical and cognitive decline were computed by averaging the standardized Δ-scores (mean = 0 and s.d. = 1), where we used row-wise mean imputation to address missing values.
To compare longitudinal to cross-sectional findings, we also derived composite scores of global cognitive and physical baseline functioning, using the same protocol as described above. In brief, we included the same set of aging phenotypes, extracted the baseline data of each phenotype and averaged the standardized baseline-scores (residualized for age and age2). The resulting composite scores for global physical and cognitive function (P0) and decline (Δ) were used as the primary outcomes in subsequent phenotypic, genome-wide and downstream analyses.
We performed a series of simulation analyses to assess risk of bias when using the three change definitions described above, including absolute (ΔDIFF), conditional (ΔRES) and relative change (ΔLOG). We simulated data according to the structural causal model shown in Fig. 2. As illustrated, the longitudinal (i.e., age-dependent) effects were conceptualized as gene × environment interactions, where the genetic effect differs across age-varying environments. The observed phenotype P* at time point t (0 = baseline, 1 = follow-up) was modelled as follows:
where Pt is the phenotype free of measurement error, εt the measurement error in the phenotype, λ the intercept, α is the time-invariant (cross-sectional) genetic effect, G0 the time-invariant (cross-sectional) genetics, β the environmental effect, E the environment, γ the gene × environment effect, GE the time-varying (longitudinal) genetics. A detailed description of the simulation approach is included in the Supplement.
Statistics & Reproducibility
No statistical method was used to predetermine sample size. Details regarding the sample selection process for the cross-sectional and longitudinal analyses are provided in Supplementary Fig. 18. This analyses are reproducible from the publicly available analytical scripts.
Longitudinal and cross-sectional genome-wide scans and downstream analyses
We then performed longitudinal genome-wide association (GWA) tests on indexes of cognitive and physical decline and compared the results to GWA findings obtained on baseline (cross-sectional) physical and cognitive function. Tests were performed on the cognitive and physical decline composite scores, as well as the individual indicators used to derive the composite scores. In total, we tested four models per aging phenotype:
-
(1)
Longitudinal genetic effects (difference scores): ΔDIFF = β ⋅ G + ε
-
(2)
Longitudinal genetic effects (residual change scores): ΔRES = β ⋅ G + ε
-
(3)
Longitudinal genetic effects (log-difference scores): ΔLOG = β ⋅ G + ε
-
(4)
Cross-sectional genetic effect (baseline phenotype, P0): P0 = β ⋅ G + ε.
All models were adjusted for the first 10 genetic principal components (PCs), genotyping array, and sex. Age was not included as a covariate as all outcomes were residualized for age and age2 prior to inclusion in genome-wide analyses. Genome-wide scans were performed in REGENIE (v3.2.6)89, which proceeds in two main steps: Step 1 fits a whole-genome regression model via ridge regression to account for local LD structures and relatedness. QC-filtered SNPs were included in step 1 (minor allele frequency (MAF) ≥ 1%, Hardy-Weinberg equilibrium P-value ≥1 × 0−15, genotyping rate ≥99%, not involved in inter-chromosomal LD and passing LD pruning at R2-threshold of 0.9 with a window size of 1000 markers and a step size of 100 markers89). In step 2, the association test is carried out by fitting linear regression models conditioning on the predictions derived in the first step. This step uses the imputed UKBB genotypes (version 3, imputed using the Haplotype Reference Consortium) with MAF > 1%. The sample was restricted to individuals of European genetic ancestry and individuals with high missing rate and/or high heterozygosity on autosomes (as determined by the UKBB90) were excluded. As age-related change in cognitive and physical function have been reported to differ between sexes91,92, we also estimated sex-dependent genome-wide effects. For that, we extended our models on change and baseline level of function by including a gene-by-sex interaction term (Δ = β1 ⋅ G + β2 ⋅ G ⋅ SEX + ε and P0 = β1 ⋅ G + β2 ⋅ G ⋅ SEX + ε, respectively).
LD-independent SNPs reaching genome-wide significance (P < 5 × 10−8) were selected via clumping in plink93 (–clump-kb 250 –clump-r2 0.1). Lead SNPs were annotated to the nearest gene and mapped to previously associated phenotypes using the Open Target Genetics R package otargen94. The same software was used to map lead SNPs to previously studied phenotypes, by drawing statistical summary data from public databases such as UKBB (www.nealelab.is), FINNGEN and GWAS Catalogue (https://www.ebi.ac.uk/gwas/). SNP-heritabiliy (h2) estimates were obtained using LD-score regression95 as implemented in GenomicSEM96.
We employed Mendelian Randomization (MR) analysis to evaluate causality of selected exposures with cross-sectional and longitudinal cognitive/physical outcomes. MR uses genetic instrumental variables (IV) as proxies for the exposure of interest (e.g., genetically predicted exposure to smoking), to estimate the causal effect of the exposure on an outcome97. We included data from 144 genome-wide studies to extract the genetic instruments, tapping into 11 exposure dimensions, such as aging-markers (e.g., telomere length), biomarkers (e.g., lipid measures), brain phenotypes (e.g., grey matter volume), other indexes of health (e.g., diabetes), lifestyles (e.g., physical activity, diet), mental health (e.g., schizophrenia), social variables (e.g., education, income), as well as the cognitive and physical measures used in this work to derive rates of decline. The list of genome-wide summary statistic files used in this work is included in Supplementary Data 6. Two-sample MR was performed using the R-Package TwoSampleMR98,99. As genetic instruments, we selected LD-independent SNPs (–clump-kb 10,000 –clump-r2 0.001) and MR was performed for all exposures with at least five genetic instruments reaching genome-wide significance (P < 5 × 10−8). The causal effects on the outcome were estimated using inverse-variance weighted (IVW) estimator. To assess the robustness of MR findings, we obtained a number of test diagnostics, including (a) the IVW F-statistic to evaluate the instrument strength (with an F-statistic of >10 indicating that risk of weak instrument bias is likely to be low100), (b) the IVW Q-statistic to assess heterogeneity (with a Q-test P-value < 0.05 indicating heterogeneity across instruments101), (c) the intercept term in MR-Egger regression to assess possible directional horizontal pleiotropy102, and (d) MR-PRESSO103 to assess the robustness against outliers. The summary of MR results includes the standardized causal estimates, the corresponding 95% Confidence intervals and the nominal and Bonferroni-corrected P-values, adjusting for the number of exposure dimensions tested (suggestive P-value threshold = P < 0.05/ 11 = 0.005) and the total number of exposures tested in MR (conservative P-value threshold = P < 0.05/ 106 = 0.0005). All SNP estimates and corresponding standard errors were standardized as follows prior to inclusion in MR: \({\gamma }_{STD}(SN{P}_{j})=\frac{\gamma (SN{P}_{j})/SE(SN{P}_{j})}{\sqrt{{N}_{j}}}\) and \(S{E}_{STD}(SN{P}_{j})=1/\sqrt{{N}_{j}}\), where γ(SNPj) is the unstandardized effect and SE(SNPj) the standard error of SNP j on the phenotype and Nj is the sample size per SNPj.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The genome-wide summary statistics generated as part of this work are deposited in the GWAS Catalogue under accession codes GCST90565836-GCST90565865.
Code availability
The following software was used to run the analyses:
• REGENIE (https://github.com/rgcgithub/regenie)
• TwoSampleMR (https://mrcieu.github.io/TwoSampleMR/)
• GenomicSEM (https://github.com/GenomicSEM/GenomicSEM).
• otargen (https://amirfeizi.github.io/otargen/)
• LDAK (https://dougspeed.com/).
All analytical scripts are available at https://github.com/TabeaSchoeler/TS2024_UKBBlongitudinal104.
References
Beard, J. R. et al. The world report on ageing and health: a policy framework for healthy ageing. Lancet 387, 2145–2154 (2016).
Plassman, B. L., Williams, J.W. Jr., Burke, J. R., Holsinger, T. & Benjamin, S. Systematic review: factors associated with risk for and possible prevention of cognitive decline in later life. Ann. Intern. Med. 153, 182–193 (2010).
Deelen, J. et al. A meta-analysis of genome-wide association studies identifies multiple longevity genes. Nat. Commun. 10, 3669 (2019).
Zenin, A. et al. Identification of 12 genetic loci associated with human healthspan. Commun. Biol. 2, 41 (2019).
Atkins, J. L. et al. A genome-wide association study of the frailty index highlights brain pathways in ageing. Aging Cell 20, e13459 (2021).
Foote, I. F. et al. Uncovering the multivariate genetic architecture of frailty with genomic structural equation modelling. medRxiv https://doi.org/10.1101/2024.07.24.24310923 (2024).
McCartney, D. L. et al. Genome-wide association studies identify 137 genetic loci for DNA methylation biomarkers of aging. Genome Biol. 22, 1–25 (2021).
Wen, J. et al. The genetic architecture of biological age in nine human organ systems. Nat. Aging 4, 1290–1307 (2024).
Rosoff, D. B. et al. Multivariate genome-wide analysis of aging-related traits identifies novel loci and new drug targets for healthy aging. Nat. aging 3, 1020–1035 (2023).
Belsky, D. W. et al. Quantification of biological aging in young adults. Proc. Natl Acad. Sci. USA 112, E4104–E4110 (2015).
Kamboh, M. I. et al. Population-based genome-wide association study of cognitive decline in older adults free of dementia: Identification of a novel locus for the attention domain. Neurobiol. Aging 84, 239–e15 (2019).
Wendel, B. et al. A genome-wide association study of the longitudinal course of executive functions. Transl. Psychiatry 11, 386 (2021).
Mahedy, L. et al. Investigation of genetic determinants of cognitive change in later life. Transl. Psychiatry 14, 31 (2024).
Arpawong, T. E. et al. Genetic variants specific to aging-related verbal memory: Insights from GWASs in a population-based cohort. PLoS ONE 12, e0182448 (2017).
Zelinski, E. M., Kennison, R. F., Watts, A. & Lewis, K. L. Convergence between cross-sectional and longitudinal studies: cohort matters. In Aging and Cognition: Research Methodologies and Empirical Advances (eds. Bosworth, H. B. & Hertzog, C.) 101–118 (American Psychological Association, 2009).
Baltes, P. B. Longitudinal and cross-sectional sequences in the study of age and generation effects. Hum. Dev. 11, 145–171 (1968).
Allen, N. E. et al. Prospective study design and data analysis in UK Biobank. Sci. Transl. Med. 16, eadf4428 (2024).
Clouston, S. A. et al. The dynamic relationship between physical function and cognition in longitudinal aging cohorts. Epidemiol. Rev. 35, 33–50 (2013).
Christensen, H. et al. Are changes in sensory disability, reaction time, and grip strength associated with changes in memory and crystallized intelligence? A longitudinal analysis in an elderly community sample. Gerontology 46, 276–292 (2000).
Baltes, P. B. & Lindenberger, U. Emergence of a powerful connection between sensory and cognitive functions across the adult life span: a new window to the study of cognitive aging? Psychol. Aging 12, 12 (1997).
Zammit, A. R., Robitaille, A., Piccinin, A. M., Muniz-Terrera, G. & Hofer, S. M. Associations between aging-related changes in grip strength and cognitive function in older adults: a systematic review. J. Gerontol. Ser. A 74, 519–527 (2019).
Gow, A. J. et al. Is age kinder to the initially more able? Yes, and no. Intelligence 40, 49–59 (2012).
Christensen, H. & Henderson, A. Is age kinder to the initially more able? A study of eminent scientists and academics. Psychol. Med. 21, 935–946 (1991).
Deary, I. J., Starr, J. M. & MacLennan, W. J. Is age kinder to the initially more able? Differential ageing of a verbal ability in the healthy old people in edinburgh study. Intelligence 26, 357–375 (1998).
Stern, Y. What is cognitive reserve? Theory and research application of the reserve concept. J. Int. Neuropsychol. Soc. 8, 448–460 (2002).
Stern, Y. et al. Whitepaper: Defining and investigating cognitive reserve, brain reserve, and brain maintenance. Alzheimer’s. Dement. 16, 1305–1311 (2020).
Kuh, D., Karunananthan, S., Bergman, H. & Cooper, R. A life-course approach to healthy ageing: Maintaining physical capability. Proc. Nutr. Soc. 73, 237–248 (2014).
Salthouse, T. A. Mental exercise and mental aging: evaluating the validity of the ‘use it or lose it’ hypothesis. Perspect. Psychol. Sci. 1, 68–87 (2006).
Fry, A. et al. Comparison of sociodemographic and health-related characteristics of UK biobank participants with those of the general population. Am. J. Epidemiol. 186, 1026–1034 (2017).
van Alten, S., Domingue, B. W., Faul, J., Galama, T. & Marees, A. T. Reweighting UK biobank corrects for pervasive selection bias due to volunteering. Int. J. Epidemiol. 53, dyae054 (2024).
Schoeler, T. et al. Participation bias in the UK Biobank distorts genetic associations and downstream analyses. Nat. Hum. Behav. 7, 1216–1227 (2023).
Lyall, D. M. et al. Quantifying bias in psychological and physical health in the UK biobank imaging sub-sample. Brain Commun. 4, fcac119 (2022).
Littlejohns, T. J. et al. The UK biobank imaging enhancement of 100,000 participants: rationale, data collection, management and future directions. Nat. Commun. 11, 2624 (2020).
Tennant, P. W., Arnold, K. F., Ellison, G. T. & Gilthorpe, M. S. Analyses of ‘change scores’ do not estimate causal effects in observational data. Int. J. Epidemiol. 51, 1604–1615 (2022).
Cronbach, L. J. & Furby, L. How we should measure “change”: or should we? Psychol. Bull. 74, 68 (1970).
Glymour, M. M., Weuve, J., Berkman, L. F., Kawachi, I. & Robins, J. M. When is baseline adjustment useful in analyses of change? An example with education and cognitive change. Am. J. Epidemiol. 162, 267–278 (2005).
Oni-Orisan, A. et al. The impact of adjusting for baseline in pharmacogenomic genome-wide association studies of quantitative change. NPJ Genom. Med. 5, 1 (2020).
Sadler, M. C. et al. Leveraging large-scale biobank EHRs to enhance pharmacogenetics of cardiometabolic disease medications. medRxiv https://doi.org/10.1101/2024.04.06.24305415 (2024).
McArdle, P. & Whitcomb, B. Improper adjustment for baseline in genetic association studies of change in phenotype. Hum. Heredity 67, 176–182 (2009).
Zhang, H., Chhibber, A., Shaw, P. M., Mehrotra, D. V. & Shen, J. A statistical perspective on baseline adjustment in pharmacogenomic genome-wide association studies of quantitative change. NPJ Genom. Med. 7, 33 (2022).
Glenn, N. D. Cohort analysts’ futile quest: statistical attempts to separate age, period and cohort effects. Am. Sociol. Rev. 41, 900–904 (1976).
Aasbrenn, M. et al. Genetic markers of abdominal obesity and weight loss after gastric bypass surgery. PLoS ONE 16, e0252525 (2021).
Heitkamp, M. et al. Obesity genes and weight loss during lifestyle intervention in children with obesity. JAMA Pediatr. 175, e205142–e205142 (2021).
Batterham, P. J., Bunce, D., Cherbuin, N. & Christensen, H. Apolipoprotein e ε4 and later-life decline in cognitive function and grip strength. Am. J. Geriatr. Psychiatry 21, 1010–1019 (2013).
Ritchie, S. J. et al. Predictors of ageing-related decline across multiple cognitive functions. Intelligence 59, 115–126 (2016).
Wisdom, N. M., Callahan, J. L. & Hawkins, K. A. The effects of apolipoprotein e on non-impaired cognitive functioning: a meta-analysis. Neurobiol. Aging 32, 63–74 (2011).
Laukka, E. J., Köhncke, Y., Papenberg, G., Fratiglioni, L. & Bäckman, L. Combined genetic influences on episodic memory decline in older adults without dementia. Neuropsychology 34, 654 (2020).
Sun, J. et al. APOE ε4 allele accelerates age-related multi-cognitive decline and white matter damage in non-demented elderly. Aging 12, 12019 (2020).
Brouwer, R. M. et al. Genetic variants associated with longitudinal changes in brain structure across the lifespan. Nat. Neurosci. 25, 421–432 (2022).
Venkatraghavan, V. et al. Analyzing the effect of APOE on alzheimer’s disease progression using an event-based model for stratified populations. Neuroimage 227, 117646 (2021).
Strittmatter, W. J. & Roses, A. D. Apolipoprotein e and alzheimer disease. Proc. Natl Acad. Sci. USA 92, 4725–4727 (1995).
Venkatesh, S. S. et al. Characterising the genetic architecture of changes in adiposity during adulthood using electronic health records. Nat. Commun. 15, 5801 (2024).
Kemper, K. E. et al. Genetic influence on within-person longitudinal change in anthropometric traits in the UK biobank. Nat. Commun. 15, 3776 (2024).
Ferguson, A. C. et al. Alzheimer’s disease susceptibility gene apolipoprotein e (APOE) and blood biomarkers in UK Biobank (n = 395,769). J. Alzheimer’s. Dis. 76, 1541–1551 (2020).
Lewis, T. L. et al. Overexpression of human apolipoprotein AI preserves cognitive function and attenuates neuroinflammation and cerebral amyloid angiopathy in a mouse model of alzheimer disease. J. Biol. Chem. 285, 36958–36968 (2010).
Livingston, G. et al. Dementia prevention, intervention, and care: 2024 report of the Lancet standing commission. Lancet 404, 572–628 (2024).
Löffler, T. et al. Impact of ApoB-100 expression on cognition and brain pathology in wild-type and hAPPsl mice. Neurobiol. aging 34, 2379–2388 (2013).
Bailey, P. et al. Genomic analyses identify molecular subtypes of pancreatic cancer. Nature 531, 47–52 (2016).
Wu, J. et al. MNX1 facilitates the malignant progress of lung adenocarcinoma through transcriptionally upregulating CCDC34. Oncol. Lett. 26, 1–12 (2023).
Yang, X. et al. MNX1 promotes cell proliferation and activates wnt/β-catenin signaling in colorectal cancer. Cell Biol. Int. 43, 402–408 (2019).
Kong, T. et al. DUSP6 mediates resistance to JAK2 inhibition and drives leukemic progression. Nat. Cancer 4, 108–127 (2023).
Lang, R. & Raffi, F. A. Dual-specificity phosphatases in immunity and infection: an update. Int. J. Mol. Sci. 20, 2710 (2019).
Zhu, X. et al. An approach to identify gene-environment interactions and reveal new biological insight in complex traits. Nat. Commun. 15, 3385 (2024).
Ritz, B. R. et al. Lessons learned from past gene-environment interaction successes. Am. J. Epidemiol. 186, 778–786 (2017).
Tyrrell, J. et al. Gene–obesogenic environment interactions in the UK biobank study. Int. J. Epidemiol. 46, 559–575 (2017).
Keller, M. B. et al. The longitudinal interval follow-up evaluation: a comprehensive method for assessing outcome in prospective longitudinal studies. Arch. Gen. psychiatry 44, 540–548 (1987).
Schoeler, T., Pingault, J.-B. & Kutalik, Z. The impact of self-report inaccuracy in the UK biobank and its interplay with selective participation. Nat. Hum. Behav. 9, 584–594 (2024).
Lyons, M. J. et al. A longitudinal twin study of general cognitive ability over four decades. Dev. Psychol. 53, 1170 (2017).
Reynolds, C. A., Finkel, D., Gatz, M. & Pedersen, N. L. Sources of influence on rate of cognitive change over time in swedish twins: an application of latent growth models. Exp. Aging Res. 28, 407–433 (2002).
Lövdén, M., Fratiglioni, L., Glymour, M. M., Lindenberger, U. & Tucker-Drob, E. M. Education and cognitive functioning across the life span. Psychol. Sci. Public Interest 21, 6–41 (2020).
Deary, I. J. et al. Losing one’s grip: a bivariate growth curve model of grip strength and nonverbal reasoning from age 79 to 87 years in the lothian birth cohort 1921. J. Gerontol. Ser. B Psychol. Sci. Soc. Sci. 66, 699–707 (2011).
Fjell, A. M. et al. Reevaluating the role of education in cognitive decline and brain aging: Insights from large-scale longitudinal cohorts across 33 countries. medRxiv https://doi.org/10.1101/2025.01.29.25321305 (2025).
Ritchie, S. J., Tucker-Drob, E. M., Starr, J. M. & Deary, I. J. Do cognitive and physical functions age in concert from age 70 to 76? evidence from the lothian birth cohort 1936. Span. J. Psychol. 19, E90 (2016).
Zahodne, L. B. et al. Education does not slow cognitive decline with aging: 12 year evidence from the victoria longitudinal study. J. Int. Neuropsychol. Soc. 17, 1039–1046 (2011).
Singh-Manoux, A. et al. Does cognitive reserve shape cognitive decline? Ann. Neurol. 70, 296–304 (2011).
Christensen, H. et al. Age is no kinder to the better educated: absence of an association investigated using latent growth techniques in a community sample. Psychol. Med. 31, 15–28 (2001).
Aschard, H. A perspective on interaction effects in genetic association studies. Genet. Epidemiol. 40, 678–688 (2016).
Hanes, D. W. & Clouston, S. A. Cognitive decline after divorce and widowhood: Is marital loss always a loss? Innov. Aging 8, igae033 (2024).
Triebner, K. et al. Menopause is associated with accelerated lung function decline. Am. J. Respir. Crit. Care Med. 195, 1058–1065 (2017).
Yao, Y. et al. The effect of China’s clean air act on cognitive function in older adults: a population-based, quasi-experimental study. Lancet Healthy Longev. 3, e98–e108 (2022).
Hajjar, I. et al. Cross-sectional and longitudinal association between antihypertensive medications and cognitive impairment in an elderly population. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 60, 67–73 (2005).
Lyall, D. M. et al. Cognitive test scores in UK biobank: data reduction in 480,416 participants and longitudinal stability in 20,346 participants. PLoS ONE 11, e0154222 (2016).
Miller, K. L. et al. Multimodal population brain imaging in the UK biobank prospective epidemiological study. Nat. Neurosci. 19, 1523–1536 (2016).
Foster, P. J. et al. Cohort profile: rationale and methods of UK biobank repeat imaging study eye measures to study dementia. BMJ Open 13, e069258 (2023).
Tsimpida, D., Kontopantelis, E., Ashcroft, D. & Panagioti, M. Comparison of self-reported measures of hearing with an objective audiometric measure in adults in the english longitudinal study of ageing. JAMA Netw. Open 3, e2015009–e2015009 (2020).
Folley, S., Zhou, A. & Hyppönen, E. Information bias in measures of self-reported physical activity. Int. J. Obes. 42, 2062–2063 (2018).
Ghisletta, P. et al. On the use of growth models to study normal cognitive aging. Int. J. Behav. Dev. 44, 88–96 (2020).
Fawns-Ritchie, C. & Deary, I. J. Reliability and validity of the UK biobank cognitive tests. PLoS ONE 15, e0231627 (2020).
Mbatchou, J. et al. Computationally efficient whole-genome regression for quantitative and binary traits. Nat. Genet. 53, 1097–1103 (2021).
Biobank, U. Genotyping And Quality Control Of Uk Biobank, A Large-scale, Extensively Phenotyped Prospective Resource. https://biobank.ctsu.ox.ac.uk/crystal/crystal/docs/genotyping_qc.pdf (2015).
Okabe, T. et al. Sex differences in age-related physical changes among community-dwelling adults. J. Clin. Med. 10, 4800 (2021).
McCarrey, A. C., An, Y., Kitner-Triolo, M. H., Ferrucci, L. & Resnick, S. M. Sex differences in cognitive trajectories in clinically normal older adults. Psychol. Aging 31, 166 (2016).
Purcell, S. et al. PLINK: A tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007).
Feizi, A. & Ray, K. Otargen: GraphQL-based r package for tidy data accessing and processing from open targets genetics. Bioinformatics 39, btad441 (2023).
Bulik-Sullivan, B. K. et al. LD score regression distinguishes confounding from polygenicity in genome-wide association studies. Nat. Genet. 47, 291–295 (2015).
Grotzinger, A. D. et al. Genomic structural equation modelling provides insights into the multivariate genetic architecture of complex traits. Nat. Hum. Behav. 3, 513–525 (2019).
Davey Smith, G. & Ebrahim, S. ‘Mendelian randomization’: can genetic epidemiology contribute to understanding environmental determinants of disease? Int. J. Epidemiol. 32, 1–22 (2003).
Hemani, G. et al. The MR-base platform supports systematic causal inference across the human phenome. eLife 7, e34408 (2018).
Hemani, G., Tilling, K. & Davey Smith, G. Orienting the causal relationship between imprecisely measured traits using GWAS summary data. PLoS Genet. 13, e1007081 (2017).
Burgess, S., Thompson, S. G. & Collaboration, C. C. G. Avoiding bias from weak instruments in Mendelian randomization studies. Int. J. Epidemiol. 40, 755–764 (2011).
Greco M, F. D., Minelli, C., Sheehan, N. A. & Thompson, J. R. Detecting pleiotropy in Mendelian randomisation studies with summary data and a continuous outcome. Stat. Med. 34, 2926–2940 (2015).
Bowden, J., Davey Smith, G. & Burgess, S. Mendelian randomization with invalid instruments: Effect estimation and bias detection through egger regression. Int. J. Epidemiol. 44, 512–525 (2015).
Verbanck, M., Chen, C.-Y., Neale, B. & Do, R. Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat. Genet. 50, 693–698 (2018).
Schoeler, T., Pingault, J.-B. & Kutalik, Z. Combining cross-sectional and longitudinal genomic approaches to identify determinants of cognitive and physical decline. Zenodo https://doi.org/10.5281/zenodo.15011326 (2025).
Acknowledgements
This research has been conducted with the UK Biobank Resource under application number 16389; we thank all biobank participants for sharing their data. We thank all participants involved in the Health Survey England and we thank the Office for National Statistics for granting access to the data. This study would not have possible without the use of publicly available genome-wide summary data and software tools. We acknowledge these resources and thank the research participants, research teams and institutions that have contributed to this research. The computations were performed on the HPC cluster of the Lausanne University Hospital. Z.K. was funded by the Swiss National Science Foundation (# 310030-189147). T.S. was funded by a Wellcome Trust Sir Henry Wellcome fellowship (grant 218641/Z/19/Z). JB.P. has received funding from the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation programme (grant agreement No. 863981).
Author information
Authors and Affiliations
Contributions
Z.K. and T.S. conceptualized the study. T.S. performed the statistical analyses. Z.K., T.S. and J.-B.P. discussed the results and provided comments on the paper. All authors critically reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Joris Deelen, Julian Mutz and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schoeler, T., Pingault, JB. & Kutalik, Z. Combining cross-sectional and longitudinal genomic approaches to identify determinants of cognitive and physical decline. Nat Commun 16, 4524 (2025). https://doi.org/10.1038/s41467-025-59383-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-59383-0
This article is cited by
-
The genetics of cannabis lifetime use
Neuropsychopharmacology (2026)