Abstract
Conserving blue carbon ecosystems (BCEs) has gained international attention in climate change mitigation, reflected in United Nations policies and voluntary carbon-offset projects. These efforts assume significant and uniform losses of soil organic carbon (Corg) throughout the top meter following disturbances, yet this assumption lacks robust empirical support. Here, we synthesized 239 paired observations of intact and disturbed BCEs globally. Soil Corg stock losses in the top meters vary widely: from −68.4% (agricultural conversion, ±13.4%, 95% confidence interval) to +0.8% (harvesting, ±46.2%) in mangroves, −25.9% (climate/hydrological change, ± 30.7%) to +48.6% (grazing, ±78.7%) in saltmarshes, and −34.2% (vegetation cover damage, ±22.4%) to −27.4% (dredging, ±33.6%) in seagrasses. Extensive disturbances deplete Corg down to 50–200 cm, while limited disturbances impact only the top 10–30 cm or resulted in negligible losses. This refinement contributes to improved global inventories of greenhouse gas emissions from BCEs, supporting abatement policy settings for nationally determined contributions commitments.
Similar content being viewed by others
Introduction
Mangroves, saltmarshes, and seagrasses, the most “actionable” blue carbon ecosystems (BCEs) towards climate mitigation, occupy less than 0.2% of the ocean’s surface but store over 30 Pg of organic carbon (Corg) in the top meter of soil globally1,2. However, when these ecosystems are converted or degraded, an estimated 0.15–1.02 Pg of CO2 is released into the atmosphere annually3, accounting for 3–23% of annual emissions from global land use change over the past decade4. This release occurs primarily through the decomposition of soil Corg stock2,5,6. Although rates of BCE area decline are slowing down or even reversing in some regions7,8,9, the global annual loss rate remains high at 0.13% for mangroves10, 0.28% for saltmarshes11, and 1–2% for seagrasses12. As a result, conserving BCEs to avoid greenhouse gas (GHG) emissions has gained global recognition as a cost-effective climate action, supported by major international climate and biodiversity policies such as the Paris Agreement and the Kunming-Montreal Global Biodiversity Framework13,14.
Financial resources for the conservation of BCEs, aimed at preventing soil Corg stock loss and the resulting GHG emissions, can be effectively mobilized through compliance and voluntary carbon markets that recognize and assign carbon credits to blue carbon projects14,15,16. These markets require rigorous accounting and verification of GHG emission reductions to demonstrate “additionality”14,15,16. However, within the science and policy communities, calculations of GHG emission reduction from blue carbon projects often assume that the conversion and degradation of BCEs result in uniform Corg stock loss across the entire top meter of soil (e.g., refs. 3,11,17; Supplementary Table 1). This assumption, exemplified by the Intergovernmental Panel on Climate Change (IPCC) Tier 1 standard18, has been perpetuated across studies estimating of global, national, and regional GHG inventories without thorough examination or verification19, despite IPCC guidelines advocating for modifying the depth of top meter at higher tiers18.
Estimates of GHG emissions from soil Corg stock loss following BCEs disturbance have been assumed to range from 22.7% to 100% of the Corg stock present in the top meter of soil3,6,11,17,20,21 (Supplementary Table 1). However, growing empirical evidence suggests that the actual depth and amount of Corg loss can vary substantially across ecosystems and disturbance regimes22,23, indicating substantial uncertainties in GHG emission estimates that rely on untested assumptions. Such problematic GHG emission accounting methodologies risk inflating credit issuances, thereby undermining the true effectiveness of nature-based solutions within nationally determined contributions (NDCs). Indeed, the issuance of carbon credits for emission reductions through terrestrial forest conservation has faced scrutiny due to failure in demonstrating genuine GHG reductions, contributing to skepticism about the effectiveness of voluntary carbon markets24,25. Understanding how different ecosystems and disturbance regimes affect soil Corg stock loss is therefore critical, especially when estimating GHG emission reductions in carbon market transactions.
The various anthropogenic and climate change disturbances affecting BCEs can be broadly classified into two main categories: (1) those that physically remove or alter both the living biomass and the soil, such as conversion of BCEs for aquaculture or agriculture and dredging5,26,27; and (2) those that affect BCE plants without directly disturbing the soils, such as harvesting, degradation, or grazing5,28,29. For over a decade, it has been hypothesized that the trajectories of soil Corg stock loss in BCEs may vary substantially depending on the types of disturbance5,30 (Fig. 1). Disturbances that physically disrupt the soil are expected to cause greater Corg stock loss, penetrating deeper soil layers, while disturbances with minimal soil impact are likely to result in lower Corg stock loss, primarily from the shallower soil layers5,30. Soil Corg stock loss is also anticipated to last longer for disturbances that penetrate deep soil layers relative to those that minimally affect the soil post-disturbance5. A comprehensive examination of these hypotheses is essential to understand how soil Corg stock loss varies among BCEs subject to different disturbance regimes, which is crucial for accurately estimating GHG emission reductions in blue carbon projects.
The major disturbance drivers investigated for mangroves include agricultural reclamation for rice fields36,68,69,70,71, pastures37,72, coconut40,73,74, rubber40, or crop plantations75,76; aquaculture development for shrimp34,40,41,72,76,77,78,79,80,81,82,83,84,85,86,87,88, or fish22,88,89, along with salt ponds construction40 and sand mining90; the harvesting22,28,91,92,93,94, clearance40,95,96,97,98,99,100,101,102, or grazing of trees103; and climate impacts such as typhoons104,105,106,107,108, saltwater intrusion109, drought110,111,112,113, or strong El Niño-Southern Oscillation114, as well as hydrological changes like settlement construction115,116,117,118,119,120,121,122,123,124,125,126,127,128,129, sediment runoff130,131, or nutrient effluents from nearby aquaculture ponds87,132,133,134,135,136,137. For saltmarshes, the disturbance drivers include agricultural reclamation for rice48,49,138,139,140,141 or crop fields49,139,142,143,144,145,146,147; aquaculture development for shrimp41,88,148,149 or fish ponds41,49,147; climate impacts such as typhoons150, as well as hydrological changes like embankment construction120,148,151,152,153,154,155,156,157,158, or excessive flooding159,160; and the grazing29,161,162,163,164,165,166,167, coppicing168, shading169, or damage of plants170. In this study, aquaculture was combined with agriculture due to the limited and geographically biased data availability for aquaculture, with no significant difference observed between them (P = 0.217; see section “Methods”). For seagrasses, the disturbance drivers include unintentional dredging due to mooring51, boat grounding26,52, or clam harvesting171,172,173, and intentional dredging for sand174; and vegetation cover damage due to shading175,176,177, removal178,179, overgrazing180, loss23,181,182,183,184,185,186,187, or nutrient enrichment116,188,189,190,191,192,193. Created in BioRender. Fu, C. (2025) https://BioRender.com/q55g628.
Here, we address these knowledge gaps by examining the loss of soil Corg stocks across BCEs subject to different disturbance regimes. Our dataset includes paired reports of soil Corg stock from both disturbed and intact BCEs from 118 mangrove sites, 82 saltmarsh sites, and 39 seagrass sites worldwide (Supplementary Fig. 1). We categorized disturbances into those causing deep soil disruption—such as the conversion of mangroves and saltmarshes to agriculture or aquaculture and dredging of seagrass meadows—and those causing minimal soil disruption, including mangrove harvesting, saltmarsh grazing, climate/hydrological change in mangroves and saltmarshes, and vegetation cover damage in seagrass meadows (Fig. 1). We first analyzed how soil Corg stock loss varied across BCEs and with depth following disturbance to resolve the distribution of Corg stock loss both across ecosystems and along the soil depth profile under various disturbance regimes. We then examined the relationship between soil Corg stock loss and initial Corg stock, as well as its relationship with the duration of disturbance across different disturbance regimes. Our results provide empirical evidence that both soil depth and the extent of Corg stock loss differ among BCEs and are controlled by the type of disturbance regimes. The estimates of Corg stock loss reported here address a knowledge gap and enable more accurate and robust estimates of GHG reduction in blue carbon projects as a nature-based climate solution.
Results
Top-meter soil Corg stock loss under different disturbance regimes
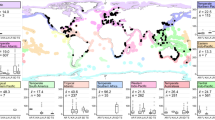
When extrapolating soil Corg stock to the top one meter globally, we found that disturbances in BCEs resulted in significantly higher losses of Corg stock in mangroves (−54.4 ± 8.7%, mean ± 95% confidence interval [CI]) and seagrasses (−36.4 ± 27.6%) relative to saltmarshes (−4.4 ± 36.3%, P < 0.001), though substantial variability was observed across all ecosystems. Soil Corg stock losses in mangroves were more pronounced and less variable in areas reported as mangrove loss hotspots, including Southeast Asia, and Central and South America10 (Fig. 2). In saltmarshes, soil Corg stock losses were similarly less variable and more prominent in China relative to Europe and USA, despite historical declines in saltmarsh area across these regions11,31. However, no clear geographic pattern of Corg stock loss was observed in seagrasses under disturbance. We could not detect an influence of plant species on soil Corg stock loss under disturbance across BCEs (P > 0.05; Supplementary Table 2), except in mangroves, where species interacted with disturbance regimes (disturbance × species, P = 0.003).
a The longitudinal distribution of soil Corg stock change (%) in BCEs; b global distribution of soil Corg stock change (%) in BCEs, c the latitudinal distribution of soil Corg stock change (%) in BCEs. Shapefile of the world obtained from www.naturalearthdata.com. Source data are provided as a Source Data file.
Disturbance regimes exerted significant control on soil Corg stock loss in mangroves (P < 0.001), but their effects were less pronounced in saltmarshes and seagrasses (P > 0.05). Major soil Corg loss in mangroves were caused by extensive soil disruption, in contrast to the variability in soil Corg stock loss under limited soil disturbance. Specifically, the conversion of mangroves to agriculture and aquaculture led to significant reductions in top one-meter soil Corg stocks, averaging −68.4 ± 13.4% and −65.4 ± 10.6%, respectively (Fig. 3). It should be noted, however, that the estimated Corg stock loss from converting mangroves to aquaculture might be conservative if soil excavation was involved during the initial “construction” phase18. In contrast, climate/hydrological change (−32.1 ± 23.0%) and harvesting activities (+0.8 ± 46.2%) in mangroves, which cause limited soil disturbance, result in less or nonsignificant Corg stock changes. In saltmarshes, no significant changes in soil Corg stock were detected in the top meter, regardless of the disturbance regime (agriculture, −1.1 ± 36.1%; climate/hydrological change, −25.9 ± 30.7%; grazing, +48.6 ± 78.7%). Similarly, in seagrass ecosystems, soil Corg stock losses driven by dredging (−27.4 ± 33.6%) were comparable to those from damage to the vegetation cover of seagrass meadows (−34.2 ± 22.4%).
Bars around the means denote 95% CIs. The first and second numbers in parentheses indicate, respectively, the number of observations and the number of studies included in each calculation. Filled circles indicate significant effects of soil Corg stock loss, while circles indicate nonsignificant effects (95% CIs overlapping zero). Mangrove soil Corg stock changes resulted from agricultural and aquaculture conversions, climate/hydrological change, and harvesting; saltmarsh soil Corg stock changes resulting from agricultural conversions, climate/hydrological change, and grazing; seagrass soil Corg stock changes resulting from dredging and vegetation cover damage. Source data are provided as a Source Data file.
Depth patterns of soil Corg stock loss under different disturbance regimes
The depth patterns of soil Corg stock loss varied among different disturbance regimes. Extensive soil disruptions, such as the conversion of mangroves to agriculture or aquaculture and the dredging of seagrass meadows, led to significant Corg loss reaching soil depths of 100–200 cm and at least 50 cm, respectively (Fig. 4). Specifically, for mangroves converted to aquaculture, soil was excavated to a mean depth of 66 ± 24 cm (± 95% CI, Supplementary Fig. 2), potentially extending soil Corg stock loss to approximately 166 cm. In contrast, saltmarsh conversion to agriculture showed no significant soil Corg stock loss across depth increments (Fig. 4b). For disturbance regimes with limited soil impact, soil Corg stock loss was either negligible across the depth profiles, as observed in the harvesting of mangroves, or largely confined to top 10–30 cm layers (climate/hydrological change in mangroves and saltmarshes, grazing of saltmarshes, and vegetation cover damage in seagrass meadows). However, deeper soil data (>30 cm) for saltmarshes and seagrasses impacted by grazing and vegetation cover damage, respectively, were underrepresented in the literature, underscoring the need for further research. These divergent depth patterns of soil Corg stock loss nevertheless suggest that the current assumption of uniform Corg loss across the top meter of soil requires reconsideration.
a Mangrove soil Corg stock changes due to agricultural and aquaculture conversions, climate/hydrological change, and harvesting, b Saltmarsh soil Corg stock changes due to agricultural conversions, climate/hydrological change, and grazing, c Seagrass soil Corg stock changes due to dredging and vegetation cover damage. Bars around the means denote 95% CIs. The bars with arrows denote 95% CIs that extend beyond the X-axis range. The numbers alongside the arrows indicate the maximum values of the 95% CIs. The first and second numbers in parentheses indicate, respectively, the number of observations and the number of studies included in each calculation. Filled circles indicate significant effects of soil Corg stock loss, while circles indicate nonsignificant effects (95% CIs overlapping zero). Source data are provided as a Source Data file.
Relationship between soil Corg stock loss and initial Corg stock
Soil Corg stock losses resulting from the conversion of mangroves to agriculture and aquaculture were strongly dependent on the initial Corg stock (P < 0.001; Fig. 5 and Supplementary Fig. 3), indicating that mangroves with higher initial Corg stocks are more susceptible to greater Corg losses following disturbances. This dependence also held for the conversion of saltmarshes to agriculture. While converting saltmarsh to agriculture did not show significant soil Corg stock loss in the top meter overall, it did result in significant loss—reaching up to −60%—in saltmarshes with higher initial soil Corg stock levels (above ~75 Mg C ha−1, P < 0.001). In contrast, the conversion of saltmarshes to agriculture with lower initial Corg stocks could enhance, rather than reduce, Corg stock levels. Grazing of saltmarshes showed a notable gain of soil Corg stock, which positively increased with higher initial Corg stocks (P = 0.028). In seagrass meadows, both gains and losses of soil Corg stock were observed following dredging (P = 0.143) or vegetation cover damage (P = 0.109), independent of initial Corg stock. When compared to the GHG emission factor commonly used in previous studies (taking 25–75% of soil Corg stock loss as a conservative range; Supplementary Table 1), we found that as initial Corg stocks increased, losses driven by the conversion of mangroves and saltmarshes to agriculture and aquaculture were more aligned with or exceeded this range. However, as initial Corg stock increases, soil Corg stock losses resulting from seagrass disturbances were not well aligned with the range of the GHG emission factor.
a−c Top-meter soil Corg stock change (Mg C ha−1) in response to initial Corg stock, d–f Top-meter soil Corg stock change (%) in response to disturbance duration. Solid lines represent linear or logarithmic regression, with shading representing the 95% CI of regression fits. The carbon emission factor of 25–75% was taken as a conservative range commonly used in previous studies (Supplementary Table 1). Top-meter soil Corg stock change (%) in response to initial Corg stock are shown in (Supplementary Fig. 3). Source data are provided as a Source Data file.
Relationship between soil Corg stock loss and disturbance duration
Soil Corg stock losses generally increase with disturbance duration for activities that cause extensive soil disruptions, including the conversion of mangroves and saltmarshes to agriculture and aquaculture (P < 0.05), and the dredging of seagrass meadows (P = 0.035, Fig. 5). Specifically, losses in soil Corg stock after converting mangroves to agriculture and aquaculture asymptotically reached −68.2 ± 13.4% and -86.3 ± 14.0%, respectively, 30 years post-conversion. Reclamation of saltmarshes to agriculture resulted in an average soil Corg stock reduction of −49.9 ± 9.2% approximately 60 years post-conversion, despite showing both positive (net gain) and negative (net loss) changes at shorter intervals. Available reports indicate that dredging of seagrass meadows leads to a soil Corg stock loss of −77.1 ± 8.4% after 80 years since the disturbance. Overall, these patterns are consistent with estimates based on previous modeling, which calculated that cumulative GHG emissions from disturbed BCEs account for 70–80% of the initial top-meter soil Corg stocks over 40 years32. In contrast, for disturbances with minimal soil impact, the response of soil Corg stock loss to disturbance duration lacks a clear pattern.
Discussion
Within the latest NDCs under the Paris Agreement, 41 countries have prioritized ocean or BCEs as key sector for reducing GHG emissions33. However, the lack of empirical support for the long-held assumption that disturbance of BCEs leads to uniform loss of Corg stock across the top meter of the soil—ranging from 22.7% to 100%—poses a challenge in effectively informing and operationalizing NDCs34,35. Our synthesis and quantitative analysis of available evidence demonstrates that the widely used assumption that the top meter of soils in BCEs is uniformly susceptible to loss, regardless of disturbance regimes (e.g., ref. 3), should be rejected. The magnitude and dynamics of soil Corg loss in disturbed BCEs are highly dependent on ecosystem type, disturbance regimes, baseline soil Corg stock, and the time since disturbance. Furthermore, our estimations provide GHG emission factors for anthropogenic and natural disturbance drivers that were not previously considered by the IPCC (Table 1), such as climate/hydrological change in mangroves and saltmarshes, which increasingly lead to BCE losses10,11,12,13,14. This work marks a critical advancement in refining global and national inventories of GHG emissions from BCEs using the IPCC Tier 1 standards, providing robust support for GHG emissions accounting and abatement policy settings for future blue carbon projects and NDCs commitments.
Among the various disturbance drivers examined here, none strictly conformed to the assumption of significant soil Corg stock loss to the depth of one meter. For example, the conversion of mangroves to agriculture resulted in soil Corg loss up to 200 cm. This disturbance regime involves substantial hydrological manipulation and soil remobilization through drainage and dredging, which reverse hypoxic soil conditions and increase the exposure of Corg to aerobic decomposition, thereby extending Corg stock losses to deeper layers5,36,37,38. For instance, the drainage of mangrove forests in East Trinity Inlet, Australia, for agricultural production between 1976 and 2000 resulted in a soil elevation loss of 1.3 m—from 0.9 m above sea level to 0.4 meters below—driven by substantial soil Corg remineralization5. Thus, considering soil Corg stock loss only from the top meter may underestimate GHG emissions due to such disturbance. Furthermore, our results indicate that soil Corg stock loss from agricultural conversion persists over decades, albeit at a decreasing rate. Therefore, removing seawalls or dikes to reintroduce tidal flow—thereby inhibiting aerobic Corg decomposition—is key to prevent further Corg stock loss and enable cost-effective mangrove recovery14,39.
In contrast to agricultural conversions, converting mangroves to aquaculture typically involves more intensive soil excavation during pond construction, reaching down to depths of 50–250 cm, which substantially enhances Corg destabilization and loss under aerobic conditions5,38,40,41. We estimated that aquaculture conversions roughly excavated soil to depths, averaging ( ± 95% CI) 66 ± 24 cm, with additional soil Corg stock loss extending to 100 cm in post-excavated soil, culminating in soil Corg stock loss down to approximately 166 cm. This indicates that considering only Corg stock loss in the top meter of soils during the initial “construction”, without accounting for losses during the “use” and “discontinued” phases18, may substantially underestimate GHG emissions. Indeed, our results suggest that soil Corg stock during these subsequent phases may be completely depleted over 30 years. Overlooking soil Corg stock loss in the “use” and “discontinued” phases could result in the allocation of insufficient carbon credits to the project, thereby limiting the scale and ambition of blue carbon projects, as well as diminishing interest in funding these initiatives. Furthermore, our findings indicate that three out of 16 converted aquaculture ponds in our dataset of elevation changes exhibited gains in soil mass (Supplementary Fig. 2). This counterintuitive result may be attributed to the collapse of soil structures due to belowground Corg decomposition, combined with bottom leveling during pond construction and soil erosion from berms, or regional variance in soil depth across mangroves and aquaculture ponds42. Therefore, clarifying soil depth dynamics is essential to robustly estimate GHG emissions following mangrove conversion to aquaculture43.
Our findings further demonstrate that soil Corg stock losses due to agriculture and aquaculture conversions in mangroves are positively correlated with initial Corg stock. This likely reflects the role of hydrogeomorphic processes in shaping Corg levels in soils of BCEs, along with the mineral-dominant nature of soils with low Corg stock, which attenuates Corg remineralization after disturbance through mineral protection44. In contrast, soils with higher Corg stocks contain greater proportions of particulate organic matter with weaker mineral associations, making them more susceptible to Corg remineralization and increased GHG emissions following disturbance44,45. As such, conserving mangroves with high soil Corg stock against conversion to agriculture and aquaculture yields the greatest reduction in Corg loss and, therefore, potential GHG emissions. The variance in initial soil Corg stock is influenced by environmental factors, particularly hydrogeomorphic settings46. Estuarine interior mangroves generally have higher soil Corg stock than open-coast or fringe mangroves22 but are highly vulnerable to conversion for agriculture and aquaculture due to their fertile soils and flat landscapes, resulting in deep soil Corg stock loss47. Therefore, incorporating geomorphic context, either alongside or as a proxy for initial soil Corg stock can contribute to the development of robust conservation projects, enabling better prioritization of the most vulnerable areas and maximizing GHG emission reductions.
The context-dependent effect of initial Corg stock on soil Corg stock loss is particularly pronounced in conversion of saltmarshes to agriculture. The assumption of significant soil Corg stock loss in the top one meter was only met in saltmarshes with initially higher soil Corg stocks. In contrast, reclamation of saltmarshes with lower initial soil Corg stock to agriculture leads to the net gain in soil Corg stock, likely due to the application of organic or chemical fertilizers that promote the accumulation of plant-derived organic material in soils48,49. This tendency is particularly conspicuous in China, where the newly formed saltmarshes, created after long-term sequential reclamation, have low soil Corg stock31,50. Consequently, the reclamation of these saltmarshes has enhanced their soil Corg stock after converting them to paddy or crop lands31,49. Similarly, aquaculture production in converted saltmarshes with low initial soil Corg stock may introduce substantial organic inputs, such as excessive fodder and feces from cultured fish or shrimp, contributing to soil Corg accumulation. Therefore, conservation projects for saltmarshes that do not account for background soil Corg stocks may not achieve the anticipated avoidance of GHG emissions. Our results further show that soil Corg stock losses following conversion of saltmarshes to agriculture are sustained over centuries. Hence, rapid deployment of restoration efforts where conversion of saltmarshes with high soil Corg stock to agriculture already occurred is critical for maximizing GHG emission reduction, while restoring Corg sequestration.
Soil Corg stock loss due to dredging of seagrass meadows can penetrate to depths of at least 50 cm, aligning with observations that dredging—whether unintentional or intentional—disturbs soil to depths ranging from 10 to 160 cm26,51,52. However, significant Corg stock loss was not observed when extrapolating to the top meter of soil, owing to the variable response of soil Corg stock following dredging. This variability may be attributed to constant seawater inundation of disturbed seagrass soil, which limits oxygen penetration and Corg decomposition1,5,32. Nutrient release from soil disturbance can also stimulate phytoplankton and bacterial blooms53,54, which may either lead to the accumulation of labile Corg or enhanced Corg decomposition through the priming effect55,56. Further research is needed to delineate the impact of soil Corg stock loss due to dredging, with a particular focus on sampling deeper soil layers in seagrass meadows.
In contrast to disturbance regimes that cause extensive soil disruption, our results show that climate/hydrological changes in mangroves and saltmarshes, harvesting of mangroves, grazing of saltmarshes, and vegetation cover damage in seagrass meadows result in Corg stock loss primarily within the top 10–30 cm, or lead to minimal Corg stock losses or even net gains due to limited soil disruption. Climate/hydrological changes in mangroves and saltmarshes and vegetation cover damage in seagrass meadows lead to the cessation of new organic material accumulation, while the root decay may trigger a priming effect, enhancing the decomposition of surface soil Corg and/or soil erosion55,57. Harvesting of mangroves and grazing on saltmarshes substantially reduce aboveground biomass and disturb the surface soil via trampling28,29. However, surface soil Corg stock losses may be partially offset by increased belowground biomass production29 and ecosystem recovery post-disturbance, which re-promotes Corg accumulation22,28. Trampling may also compact surface soil, enhancing the development of anaerobic conditions that weaken Corg mineralization58. Consequently, conservation efforts targeting these disturbances may not achieve the anticipated avoidance of GHG emissions. These findings underscore that overestimated emission factors, based on unverified assumptions perpetuated across studies3,11,17,20,21, may have led to the issuance of excessive carbon credits to blue carbon projects achieving additionality through avoidance of these impacts. Failure to achieve the committed GHG emission avoidance undermines the credibility of nature-based solutions, including blue carbon projects, despite their important role in contributing to climate change mitigation efforts.
While the avoided GHG emissions from conservation projects focusing on BCEs affected by disturbances causing limited soil disruption are modest, these efforts can still be motivated by the broader benefits of the ecosystem services they provide, such as biodiversity enhancement and coastal protection14,15,16. Management regimes that include sustainably managed harvesting practices, such as logging of mangroves and grazing of saltmarshes, can minimize GHG emissions while supporting provision services that benefit communities14,29. Incentivizing such sustainable management practices can achieve modest GHG avoidance while supporting co-benefits, representing a new dimension of blue carbon projects beyond those focused on conservation and/or restoration that deserves consideration.
While our global synthesis provides a compelling analysis of soil Corg dynamics across different disturbance regimes, it is restricted by data scarcity for some regions, such as South America and Africa, particularly for saltmarshes and seagrasses. To further reduce uncertainty, there is a need to expand empirical evidence on Corg dynamics over long time scales and across a broader range of disturbance regimes. Nevertheless, the disturbance regimes explored in this study can serve as approximations for unstudied disturbance drivers, based on their potential effects on ecosystems and their convergence with studied drivers. For example, disturbances leading to plant die-off and loss due to insects or diseases59,60,61 could be approximated using climate/hydrological change category for mangroves and saltmarshes, as well as the vegetation cover damage category for seagrasses, as described in this study. Notably, due to the lack of paired data reporting Corg dynamics, we could not explore coastal erosion as a disturbance driver increasingly causing BCE loss, such as reported in Bangladesh, Brazil, USA and Russia10,11. Moreover, losses of soil Corg are not necessarily equivalent to GHG emissions but provide a useful upper ceiling to resulting GHG emissions. Available evidence suggests that some of the eroded Corg from saltmarshes and terrestrial sources delivered to estuaries may be reburied elsewhere in the coastal ocean62,63. Therefore, further assessment of the fate of eroded Corg will be key to constrain the GHG emission driven by direct or indirect erosion.
The results presented provide robust, empirically validated estimates across BCEs, allowing for the development of improved emission factors and the assessment of potential GHG emission reductions from actions that protect BCEs at risk from various disturbances. Major soil Corg stock losses due to the conversion of mangroves and saltmarshes to agriculture or aquaculture, and the dredging of seagrass meadows, highlight the urgent need for preventing such disturbances, particularly in regions with higher Corg stocks, which can be achieved through including habitats highly vulnerable to such disturbances into marine protected areas14. Given the enduring nature of Corg loss after disturbances, restoration efforts underpinned by major global instruments should prioritize projects in converted areas, improving land tenure and providing economic alternatives for affected communities to prevent further GHG emissions and restore Corg sequestration14,16,64. Climate/hydrological change results in relatively lower soil Corg stock loss due to lesser soil disturbance, imparting optimism that restoring degraded BCEs can recover Corg stock while preventing further losses. Furthermore, sustainable, non-destructive use of BCEs, such as selective harvesting of mangroves, can sustain human benefits without impairing soil Corg storage. These findings contribute to strengthening blue carbon projects as a robust and effective nature-based solutions, supporting the inclusion of BCEs as an important asset in the NDCs’ more ambitious commitments to address the growing magnitude and pace of climate change.
Methods
Literature search and screening
We systematically searched all peer-reviewed literature using the Web of Science (http://apps.webofknowledge.com) with the keywords listed in Supplementary Table 3 published before 20 November 2023, following the PRISMA protocol65, to systemically investigate the effects of disturbance on soil Corg stock loss in BCEs. A follow-up search was conducted on 25 December 2024 using the same keywords to include recent studies published after 20 November 2023. To prevent bias in selection from the 4886 potentially relevant articles obtained from the two systematical searches, we first removed a significant number of articles through title screening, leaving 1943 articles for further inspection (Supplementary Fig. 4). The title, abstract, and full text were screened based on the following criteria: first, they must report the anthropogenic or natural driver of soil Corg stock loss in BCEs, such as, agricultural reclamation, aquaculture development, dredging, clearance, and climate/hydrological change; second, they must contain reference sites for the disturbed sites, such as an undisturbed, natural, or controlled plots without apparent human and/or climate change impacts, sharing the same parent material and environmental conditions, including at least one undisturbed site if multiple disturbed sites were present; third, they must report at least the soil Corg (or soil organic matter) content for the paired sites; and fourth, they must report the sampled soil depth to determine soil Corg content. If an article included multiple sites or different habitat types, each was treated as a separate entry in the database. We excluded studies that did not specify a clear disturbance driver, reported multiple disturbance drivers affecting the same site, or investigated soil Corg stock losses based on area changes of BCEs, modeling with scaled‐up data (i.e., GIS and remote sensing modelling), or laboratory manipulation experiments. We also excluded studies that investigated soil Corg stock changes due to ecological invasion (e.g., the Spartina alterniflora invasion in Chinese BCEs66), encroachment between BCEs (e.g., mangroves encroached into saltmarshes near the poleward limits67), given that the loss of the original habitats is compensated by the gain of another BCE.
All data for the analysis were derived from paired intact and disturbed BCEs, from 140 research articles encompassing 118 mangrove sites, 82 saltmarsh sites, and 39 seagrass sites worldwide (Supplementary Fig. 1 and Supplementary Fig. 4). The major disturbance drivers investigated in these studies were categorized based on two key principles: (1) the purpose of the disturbance to BCEs and the extent of soil disruption, and (2) the availability of sufficient reports to perform statistical analysis. When the number of studies was insufficient, disturbance drivers were combined according to the first principle. For mangroves, the disturbance drivers include: (1) agricultural reclamation for rice fields36,68,69,70,71, pastures37,72, coconut40,73,74, rubber40, or crop plantations75,76; (2) aquaculture development for shrimp34,40,41,72,76,77,78,79,80,81,82,83,84,85,86,87,88, or fish22,88,89, along with salt pond construction40 and sand mining90; (3) harvesting22,28,91,92,93,94, clearance40,95,96,97,98,99,100,101,102, or grazing of trees103; and (4) climate impacts such as typhoons104,105,106,107,108, saltwater intrusion109, drought110,111,112,113, or strong El Niño-Southern Oscillation114, as well as anthropogenic activities like settlement construction115,116,117,118,119,120,121,122,123,124,125,126,127,128,129, sediment runoff130,131, or nutrient effluents from aquaculture ponds87,132,133,134,135,136,137. For saltmarshes, the disturbance drivers included: (1) agricultural reclamation for rice48,49,138,139,140,141 or crop fields49,139,142,143,144,145,146,147; (2) aquaculture development for shrimp41,88,148,149 or fish ponds41,49,147; (3) climate impacts such as typhoons150, as well as hydrological changes like embankment construction120,148,151,152,153,154,155,156,157,158 or excessive flooding159,160; and (4) the grazing29,161,162,163,164,165,166,167, coppicing168, shading169, or damage of plants170. We combined (1) and (2) due to only one study reporting soil Corg dynamics to one meter soil depth due to fish aquaculture conversion148. Furthermore, all reports on converting saltmarshes to aquaculture come exclusively from China41,49,88,147,148,149, and no significant difference was observed between (1) and (2) (P = 0.217). We have categorized the combined disturbance driver as agriculture as the data is dominating by agricultural conversions. For seagrasses, the disturbance drivers included: (1) unintentional dredging due to mooring51, boat grounding26,52, or clam harvesting171,172,173, and intentional dredging for sand174; and (2) vegetation cover damage due to shading175,176,177, removal178,179, overgrazing180, loss23,181,182,183,184,185,186,187, or nutrient enrichment116,188,189,190,191,192,193.
These disturbance drivers cover all common disturbances occurring in BCEs10,12,14,194 and were categorized based on their potential impact on soils, following the framework proposed by Lovelock et al.5. Consequently, the drivers for mangrove disturbance were divided into four categories: agriculture, aquaculture (extensive soil disturbance), climate/hydrological change, and harvesting (limited soil disturbance). For saltmarsh disturbance, the drivers were divided into three categories: agriculture (extensive soil disturbance), climate/hydrological change, and grazing (limited soil disturbance). For seagrass disturbance, the drivers were divided into two categories: dredging (extensive soil disturbance), and vegetation cover damage (limited soil disturbance). All the studies were conducted in paired sites using the “space for time” approach27. Given that soil Corg may take several years or decades to reach a new equilibrium32,195, there were limited studies with time series data extending back to pre-disturbance conditions. Consequently, for each paired site, it was assumed that soil conditions were similar before the disturbance. For chronosequences, data from all time points were utilized in this study.
Data extraction and standardization
Data on latitude and longitude, mean annual temperature (MAT), mean annual precipitation (MAP), sampling depth (and intervals), sampling number, soil Corg (or soil organic matter) content, dry bulk density (DBD), and duration of disturbance were extracted from the eligible literature. For those data not provided numerically but graphed, we determined values from figures with Web Plot Digitizer (https://automeris.io/). For studies reported soil organic matter, we used the conversion factor reported for mangroves (soil Corg = 0.21 × soil organic matter ∨ 1.12, ref. 196), saltmarshes (soil Corg = 0.52 × soil organic matter −0.21, ref. 196), and seagrasses (soil Corg = 0.40 × soil organic matter −1.17, ref. 197) to recalculate soil Corg content to facilitate the calculation of soil Corg stock. For studies providing only age intervals (e.g., 10–25 years or >66 years), we averaged the range (e.g., 17.5 years) or use the lower range if the upper range was not reported (e.g., 66 years). If a publication presented data for multiple control sites, we combined those control sites and recalculated their mean, standard deviation and sample size.
Despite its importance for determination of soil Corg stock, DBD was reported in only 55% of the included studies (34/77 for mangroves, 19/39 for saltmarshes, and 10/28 for seagrasses). We therefore used a random forest algorithm to estimate the missing DBD for each soil depth which reported soil Corg data, using all available predictor variables, including soil Corg content, BCE species (only for intact sites), middle depth of each sampling depth interval, MAT, MAP, disturbance driver (only for disturbed sites) and disturbance duration (only for disturbed sites), in a manner analogous to a pedotransfer function198 (Supplementary Fig. 5). For observations where MAT and MAP were not reported, we filled in the missing data using the WorldClim 2.0 dataset (spatial resolution: 30 s, https://www.worldclim.org/data/worldclim21.html) by averaging values within a 1 km buffer of each site’s longitude and latitude using ArcGIS 10.8 (ESRI, Redlands, CA). Random forest model combines a large number of regression trees, trained using bootstrap aggregation, to build a robust predictive model resistant to noise in the data199. The DBDs predicted using the random forest algorithm show significant linear agreement with the observed DBDs for both intact and disturbed BCEs, with an overall R2 of 0.89 (range: 0.73–0.99; P < 0.001) and a root mean square error of 0.11 ± 0.02 g cm−3 (mean ± standard error; range: 0.05–0.16 g cm−3; Supplementary Fig. 6), supporting the validity of this imputation method.
Standardization of soil depth was necessary due to the depth pattern of soil Corg stock loss being a core focus of this study, but there was considerable variation in soil depth and intervals reported, with depth classes ranging from 5 to 300 cm across eligible studies. To ensure comparability, we identified the most common sample depths for each ecosystem: 0–15, 15–30, 30–50, 50–100, 100–200, and 200–300 cm for mangroves; 0–10, 10–20, 20–30, 30–40, 40–60, 60–80, and 80–100 cm for saltmarshes; and 0–5, 5–10, 10–15, 15–20, 20–25, 25–30, 30–40, 40–50, 50–60, 60–70, 70–80, 80–90, 90–100, 100–120, 120–140, 140–160, and 160–180 cm for seagrasses. Data from the included research articles that reported sample depths not aligning with these distinct ranges were standardized using two methods198,200, depending on the maximum depth intervals reported. First, if at least five depth intervals were reported in the study, we fitted the soil Corg content and DBD to soil depth for each profile using various regression models, including linear regression, second- and third-order polynomial regressions, exponential function, loess regression, and spline regression200. The midpoint of each soil layer (e.g., 7.5 cm for the 0–15 cm soil layer of mangroves) was used as the representative depth. The best-fit model, determined by the smallest residual standard error, was then used to predict soil Corg content and DBD within the sampled profile200. The average residual standard error of the best-fit model for DBD in intact and disturbed BCEs are 0.05 ± 0.004 (range: 0.002–0.16) g cm-3 and 0.05 ± 0.004 (range: 0.002–0.22) g cm−3, respectively. Meanwhile, the average residual standard error of the best-fit model for soil Corg in intact and disturbed BCEs are 0.58 ± 0.08 (range: 0.01–3.02) % and 0.48 ± 0.07 (range: 0.002–4.41) %, respectively (Supplementary Fig. 7).
It is worth noting that for mangrove soils, studies often report soil Corg content and DBD for the entire 100–300 cm depth range based on a published protocol201. However, the actual sampling depth is typically just 5 cm in the middle of this range, specifically from 197.5 cm to 202.5 cm. To avoid introducing uncertainty in assessing Corg stock changes for the 200–300 cm depth range, we regarded this data as representing the 100–200 cm depth. Therefore, only data specifically sampled from the 200–300 cm range were included for this depth category.
Second, if fewer than five depth intervals were reported, we assigned soil Corg content and DBD to specific depth categories by first finding the median of the reported depth increment and then determining the appropriate depth category as best as possible198. For studies reporting multiple soil Corg content and DBD measurements within one depth category, a single value was calculated using a weighted average198. The weighted average for soil Corg (Corg,weighted, g C kg-1) was calculated by using the normalized depth intervals as the weighting factor for Corg of each layer (Corg,i and Corg,i+1). The depth intervals were normalized by dividing each depth interval (Ti and Ti+1) by the sum of the depth intervals, ensuring that the sum of the weighting factors equaled one (formula 1). The same approach was applied to DBD (g cm−3).
Soil Corg stock calculation and extrapolation
Converting mangroves to aquaculture ponds typically involves soil excavation, but the actual depth of excavation is seldomly reported in literature43. To estimate the potential soil excavation depth, we filtered soil depth data from our database for paired intact mangroves and aquaculture ponds reported to have been sampled to the bedrock or parent material. Studies that did not sample to these depths were excluded from this analysis due to inability to accurately capture soil depth changes. We first calculated the soil mass to the bedrock or parent material of intact mangroves (Mintact, Mg ha−1) and aquaculture ponds (Maquaculture, Mg ha−1) by multiplying DBD, the thickness of the corresponding soil layer (Ti, m), and a unit conversion factor (104 m2 ha-1) (formula 2). Then, we calculated changes in soil mass and estimated the potential excavation depth (Texcavation, m) by dividing the soil mass loss by the average DBD of the intact mangroves (formula 3).
We caution against directly comparing the soil Corg stock at the original depth for intact mangroves with that at excavated soil depths in aquaculture ponds, as this may introduce uncertainty in estimating soil Corg stock loss. However, our investigation into the depth pattern of soil Corg density in intact mangroves using a linear mixed model revealed that soil Corg density did not vary significantly across different soil depths (Supplementary Fig. 8; P = 0.104). This supports the direct comparison of soil Corg stock between intact mangroves and aquaculture across soil profiles without correction.
We recalculated soil Corg stocks in disturbed BCEs using a soil mass equivalent approach rather than fixed depth and general soil Corg stock differences22,27,39. This soil mass equivalent approach compares soil Corg stock between intact and disturbed sites on an equivalent soil mass basis for each soil layer, reducing uncertainties associated with DBD variations. This method can address Corg density changes caused by processes such as soil compaction, subsidence, drainage, or flooding, which are often significant during or following disturbances22,27,37,202. Briefly, we first calculated the dry soil mass (Mi, Mg ha−1) of each layer by multiplying DBD, the thickness of the corresponding soil layer (Ti, m), and a unit conversion factor (104 m2 ha−1) (formula 4). Then, we calculated the areal Corg stock (Ci,fix, kg C ha−1) of each layer to a fixed depth by multiplying Corg content (Corg,i, g C kg−1) and Mi (formula 5). Next, we calculated the soil mass difference between the intact and disturbance sites for each soil layer by subtracting Mi from the selected minimum soil mass (Mi,equiv, Mg ha−1) (formula 6). Finally, the equivalent Corg mass (kg C ha−1) in a soil layer was calculated by subtracting the added Corg mass from top soil layer and adding Corg mass from the bottom soil layer (formula 7).
We extrapolated soil Corg stock to the top one meter to allow the comparison with the current paradigm and literature values. For studies that reported soil Corg stock to depth less than 100 cm depth, we assumed that the Corg density of the unmeasured layer was the same as that of the deepest measured layer39. For example, if the deepest measured soil layer was 30–50 cm, the Corg density in the 50–100 cm layer was assumed to be the same as that in 30–50 cm. This approach, however, might overestimate soil Corg density in the deeper layers given that top soil section contains typically more Corg than the older bottom section31,197. Nonetheless, our compiled soil Corg content and DBD data showed limited depth trend from 30 cm to 100 cm (Supplementary Fig. 9), supporting the plausibility of this approach. Moreover, to further reduce uncertainty, we did not include studies that only measured surface soil Corg stock (<30 cm) when calculating top-meter soil Corg stock. In addition, if studies reported total soil Corg stock to a depth less than 100 cm, we projected Corg stock in the top meter by multiplying the Corg stock by the reciprocal of the ratio to 100, to extrapolate the Corg stocks to 100 cm depth54. To estimate emission factors for soil layers associated with significant Corg stock losses, we used only the measured or depth-standardized values without extrapolation to minimize uncertainty.
Data synthesis of disturbance on soil Corg stock
The natural log-transformed response ratio (lnRR) was employed to quantify the effect sizes of disturbance on soil Corg stock across different soil depths (formula 8; ref. 203):
where Cdisturbed and Cintact are the soil Corg stock in the disturbed and intact BCEs, respectively.
However, in our dataset, standard deviation or the standard error of soil Corg stock was not reported in 96 of the 140 articles, preventing us from estimating the variance of the effect size based on the standard deviations, sample numbers and mean values of the disturbed and intact groups. Therefore, we employed the number of replications for weighting (formula 9; refs. 204,205,206):
where Wr is the weight for each observation, Ndisturbed and Nintact are the numbers of replicates in the disturbed and corresponding intact BCEs, respectively.
To examine the influence of ecosystem or disturbance regimes on top-meter soil Corg stock, a linear mixed model was employed with ecosystem or disturbance denoted as fixed factors, while study as a random factor to account for the autocorrelation among observations within each study (formula 10). These analyses were conducted using restricted maximum likelihood estimation (REML) with the lme4 (version 1.1.29) and emmeans (version 1.7.5) packages with Wr as the weight for each corresponding observation in R (version 4.0.4, http://www.r-project.org/).
where β1 is the coefficient to be estimated; πstudy is the random effect factor of study; ɛ is sampling error.
Similarly, to examine the influence of disturbance regimes on soil Corg stock across the depth profile, we employed depth interval denoted as fixed factors for each disturbance driver, while study was treated as a random factor as before (formula 11).
For ease of interpretation, LnRR and its corresponding 95% CIs were further transformed into percent soil Corg stock change (ΔCorg,%; formula 12). If the 95% CI of ΔCorg does not cover zero, the effect of individual disturbances was significant (positive or negative, at P = 0.05) in affecting soil Corg stock.
To explore the response of soil Corg stock loss to initial Corg stock and disturbance duration, we first statistically compared the linear and logarithmic functions with the factor of interest as the fixed effect, and ‘site’ as the random effect, using the Akaike information criterion (AIC). We found that the logarithmic functions resulted in lower or similar AIC values for the response of soil Corg stock change (%) to initial soil Corg stock and disturbance duration (Supplementary Table 4), while linear functions resulted in lower or similar AIC values for the response of soil Corg stock change (Mg C ha−1) to initial soil Corg stock (Supplementary Table 5). Therefore, we used logarithmic or linear functions to construct the response of soil Corg stock change to initial soil Corg stock and disturbance duration and visualized with ggplot2 (version 3.3.6) packages in R.
Data availability
The source data supporting Figs. 2–5 are available in the Source Data file. The complete database is deposited in Figshare database (https://doi.org/10.6084/m9.figshare.27074590). Source data are provided with this paper.
Code availability
Codes for creating each figure are available at Figshare database (https://doi.org/10.6084/m9.figshare.27074590).
References
Duarte, C. M., Losada, I. J., Hendriks, I. E., Mazarrasa, I. & Marbà, N. The role of coastal plant communities for climate change mitigation and adaptation. Nat. Clim. Change 3, 961–968 (2013).
Macreadie, P. I. et al. Blue carbon as a natural climate solution. Nat. Rev. Earth Environ. 2, 826–839 (2021).
Pendleton, L. et al. Estimating global “blue carbon” emissions from conversion and degradation of vegetated coastal ecosystems. PLoS ONE 7, e43542 (2012).
Friedlingstein, P. et al. Global carbon budget 2022. Earth Syst. Sci. Data 14, 4811–4900 (2022).
Lovelock, C. E. et al. Assessing the risk of carbon dioxide emissions from blue carbon ecosystems. Front. Ecol. Environ. 15, 257–265 (2017).
Adame, M. F. et al. Future carbon emissions from global mangrove forest loss. Glob. Change Biol. 27, 2856–2866 (2021).
de Los Santos, C. B. et al. Recent trend reversal for declining European seagrass meadows. Nat. Commun. 10, 1–8 (2019).
Duarte, C. M. et al. Rebuilding marine life. Nature 580, 39–51 (2020).
Wang, X. et al. Rebound in China’s coastal wetlands following conservation and restoration. Nat. Sustain. 4, 1076–1083 (2021).
Goldberg, L., Lagomasino, D., Thomas, N. & Fatoyinbo, T. Global declines in human‐driven mangrove loss. Glob. Chang. Biol. 26, 5844–5855 (2020).
Campbell, A. D., Fatoyinbo, L., Goldberg, L. & Lagomasino, D. Global hotspots of salt marsh change and carbon emissions. Nature 612, 701–706 (2022).
Dunic, J. C., Brown, C. J., Connolly, R. M., Turschwell, M. P. & Côté, I. M. Long‐term declines and recovery of meadow area across the world’s seagrass bioregions. Glob. Change Biol. 27, 4096–4109 (2021).
Buelow, C. A. et al. Ambitious global targets for mangrove and seagrass recovery. Curr. Biol. 32, 1641–1649 (2022).
Fu, C., Steckbauer, A., Mann, F. & Duarte, C. M. Achieving the Kunming-Montreal Biodiversity targets for blue carbon ecosystems. Nat. Rev. Earth Environ. 5, 538–552 (2024).
Canning, A. D. et al. Financial incentives for large-scale wetland restoration: Beyond markets to common asset trusts. One Earth 4, 937–950 (2021).
Macreadie, P. I. et al. Operationalizing marketable blue carbon. One Earth 5, 485–492 (2022).
Kelleway, J. J. et al. A national approach to greenhouse gas abatement through blue carbon management. Global Environ. Change 63, 102083 (2020).
Hiraishi, T. et al. 2013 Supplement to the 2006 IPCC Guidelines for National Greenhouse Gas Inventories: Wetlands (eds Hiraishi, T. et al.) (IPCC, 2014).
Macreadie, P. I. et al. The future of Blue Carbon science. Nat. Commun. 10, 1–13 (2019).
Donato, D. C. et al. Mangroves among the most carbon-rich forests in the tropics. Nat. Geosci. 4, 293–297 (2011).
Siikamäki, J., Sanchirico, J. N. & Jardine, S. L. Global economic potential for reducing carbon dioxide emissions from mangrove loss. Proc. Natl Acad. Sci. USA 109, 14369–14374 (2012).
Sasmito, S. D. et al. Mangrove blue carbon stocks and dynamics are controlled by hydrogeomorphic settings and land‐use change. Glob. Change Biol. 26, 3028–3039 (2020).
Salinas, C. et al. Seagrass losses since mid‐20th century fuelled CO2 emissions from soil carbon stocks. Glob. Change Biol. 26, 4772–4784 (2020).
Pande, R. Can the market in voluntary carbon credits help reduce global emissions in line with Paris Agreement targets? Science 384, eadp5223 (2024).
Probst, B. S. et al. Systematic assessment of the achieved emission reductions of carbon crediting projects. Nat. Commun. 15, 9562 (2024).
Bourque, A. S., Kenworthy, W. J. & Fourqurean, J. W. Impacts of physical disturbance on ecosystem structure in subtropical seagrass meadows. Mar. Ecol. Prog. Ser. 540, 27–41 (2015).
Sasmito, S. D. et al. Effect of land-use and land-cover change on mangrove blue carbon: a systematic review. Glob. Change Biol. 25, 4291–4302 (2019).
Murdiyarso, D., Sasmito, S. D., Sillanpää, M., MacKenzie, R. & Gaveau, D. Mangrove selective logging sustains biomass carbon recovery, soil carbon, and sediment. Sci. Rep. 11, 12325 (2021).
Graversen, A. E. L., Banta, G. T., Masque, P. & Krause‐Jensen, D. Carbon sequestration is not inhibited by livestock grazing in Danish salt marshes. Limnol. Oceanogr. 67, S19–S35 (2022).
Murray, B., Pendleton, L., Jenkins, W. & Sifleet, S. Green Payments for Blue Carbon: Economic Incentives for Protecting Threatened Coastal Habitats (Nicholas Institute for Environmental Policy Solutions, Duke University, 2011).
Fu, C. et al. Stocks and losses of soil organic carbon from Chinese vegetated coastal habitats. Glob. Change Biol. 27, 202–214 (2021).
Lovelock, C. E., Fourqurean, J. W. & Morris, J. T. Modeled CO2 emissions from coastal wetland transitions to other land uses: tidal marshes, mangrove forests, and seagrass beds. Front. Mar. Sci. 4, 143 (2017).
United Nations Framework Convention on Climate Change (UNFCCC). Nationally determined contributions under the Paris Agreement-Synthesis report by the secretariat (November 2024). https://unfccc.int/documents/632334.
Bernardino, A. F. et al. The inclusion of Amazon mangroves in Brazil’s REDD+ program. Nat. Commun. 15, 1549 (2024).
Herr, D., & Landis, E. Coastal blue carbon ecosystems. Opportunities for Nationally Determined Contributions. Policy Brief (International Union for Conservation of Nature and The Nature Conservancy, 2016).
D’Amico, M. E., Barbieri, M., Khair, D. A. E. & Comolli, R. Mangrove rice productivity and pedogenic trends in Guinea Bissau, West Africa. J. Soil. Sediment. 24, 244–258 (2024).
Kauffman, J. B. et al. Carbon stocks of mangroves and losses arising from their conversion to cattle pastures in the Pantanos de Centla, Mexico. Wetl. Ecol. Manag. 24, 203–216 (2016).
Friess, D. A. et al. Mangrove blue carbon in the face of deforestation, climate change, and restoration. Annu. Rev. Plant Biol. 3, 427–456 (2020).
Song, S. et al. Mangrove reforestation provides greater blue carbon benefit than afforestation for mitigating global climate change. Nat. Commun. 14, 756 (2023).
Castillo, J. A. A., Apan, A. A., Maraseni, T. N. & Salmo, S. G. III Soil C quantities of mangrove forests, their competing land uses, and their spatial distribution in the coast of Honda Bay, Philippines. Geoderma 293, 82–90 (2017).
Lin, S. et al. Losses and destabilization of soil organic carbon stocks in coastal wetlands converted into aquaculture ponds. Glob. Change Biol. 30, e17480 (2024).
Food And Agriculture Organization of The United Nations (FAO). Malaysia Coastal Aquaculture Development (FAO, 1984).
Murdiyarso, D., Swails, E., Hergoualc’h, K., Bhomia, R. & Sasmito, S. D. Refining greenhouse gas emission factors for Indonesian peatlands and mangroves to meet ambitious climate targets. Proc. Natl. Acad. Sci. USA 121, e2307219121 (2024).
Spivak, A. C., Sanderman, J., Bowen, J. L., Canuel, E. A. & Hopkinson, C. S. Global-change controls on soil-carbon accumulation and loss in coastal vegetated ecosystems. Nat. Geosci. 12, 685–692 (2019).
Li, Y. et al. Increased mineral‐associated organic carbon and persistent molecules in allochthonous blue carbon ecosystems. Glob. Change Biol. 31, e70019 (2025).
Rovai, A. S. et al. Global controls on carbon storage in mangrove soils. Nat. Clim. Change 8, 534–538 (2018).
Kauffman, J. B. et al. Total ecosystem carbon stocks of mangroves across broad global environmental and physical gradients. Ecol. Monogr. 90, e01405 (2020).
Cui, J. et al. Long-term changes in topsoil chemical properties under centuries of cultivation after reclamation of coastal wetlands in the Yangtze Estuary, China. Soil Tillage Res. 123, 50–60 (2012).
Zhang, H. et al. Dynamics and driving factors of the organic carbon fractions in agricultural land reclaimed from coastal wetlands in eastern China. Ecol. Indic. 89, 639–647 (2018).
Li, Y. et al. Soil carbon, nitrogen, and phosphorus stoichiometry and fractions in blue carbon ecosystems: Implications for carbon accumulation in allochthonous-dominated habitats. Environ. Sci. Technol. 57, 5913–5923 (2023).
Serrano, O. et al. Impact of mooring activities on carbon stocks in seagrass meadows. Sci. Rep. 6, 23193 (2016).
Arney, R. N., Shepherd, A. K., Alexander, H. D. & Rahman, A. F. Soil carbon and nitrogen storage in natural and prop-scarred Thalassia testudinum seagrass meadows. Estuar. Coast. 44, 178–188 (2021).
Arias-Ortiz, A. et al. A marine heatwave drives massive losses from the world’s largest seagrass carbon stocks. Nat. Clim. Change 8, 338–344 (2018).
Fu, C. et al. Substantial blue carbon sequestration in the world’s largest seagrass meadow. Commun. Earth Environ. 4, 474 (2023).
Trevathan-Tackett, S. M., Thomson, A. C., Ralph, P. J. & Macreadie, P. I. Fresh carbon inputs to seagrass sediments induce variable microbial priming responses. Sci. Total Environ. 621, 663–669 (2018).
Macreadie, P. I. et al. Vulnerability of seagrass blue carbon to microbial attack following exposure to warming and oxygen. Sci. Total Environ. 686, 264–275 (2019).
Rietl, A. J., Megonigal, J. P., Herbert, E. R. & Kirwan, M. L. Vegetation type and decomposition priming mediate brackish marsh carbon accumulation under interacting facets of global change. Geophys. Res. Lett. 48, e2020GL092051 (2021).
Tan, L. et al. Land use and land cover changes in coastal and inland wetlands cause soil carbon and nitrogen loss. Glob. Ecol. Biogeogr. 31, 2541–2563 (2022).
Lu, W. et al. Insect outbreaks have transient effects on carbon fluxes and vegetative growth but longer-term impacts on reproductive growth in a mangrove forest. Agric. For. Meteorol. 279, 107747 (2019).
Sippo, J. Z., Lovelock, C. E., Santos, I. R., Sanders, C. J. & Maher, D. T. Mangrove mortality in a changing climate: An overview. Estuar. Coast. Shelf Sci. 215, 241–249 (2018).
Sullivan, B. K., Sherman, T. D., Damare, V. S., Lilje, O. & Gleason, F. H. Potential roles of Labyrinthula spp. in global seagrass population declines. Fungal Ecol 6, 328–338 (2013).
Blair, N. E. & Aller, R. C. The fate of terrestrial organic carbon in the marine environment. Annu. Rev. Mar. Sci. 4, 401–423 (2012).
Sapkota, Y. & White, J. R. Long-term fate of rapidly eroding carbon stock soil profiles in coastal wetlands. Sci. Total Environ. 753, 141913 (2021).
Lovelock, C. E., Barbier, E. & Duarte, C. M. Tackling the mangrove restoration challenge. PLoS Biol 20, e3001836 (2022).
Page, M. J. et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Syst. Rev. 10, 1–11 (2021).
Zhang, J. et al. Spartina alterniflora invasion benefits blue carbon sequestration in China. Sci. Bull. 69, 1991–2000 (2024).
Kelleway, J. J. et al. Review of the ecosystem service implications of mangrove encroachment into salt marshes. Glob. Chang. Biol. 23, 3967–3983 (2017).
Andreetta, A., Huertas, A. D., Lotti, M. & Cerise, S. Land use changes affecting soil organic carbon storage along a mangrove swamp rice chronosequence in the Cacheu and Oio regions (northern Guinea-Bissau). Agric. Ecosyst. Environ. 216, 314–321 (2016).
Chauhan, R., Datta, A., Ramanathan, A. L. & Adhya, T. K. Whether conversion of mangrove forest to rice cropland is environmentally and economically viable? Agric. Ecosyst. Environ. 246, 38–47 (2017).
Padhy, S. R., Bhattacharyya, P., Nayak, S. K., Dash, P. K. & Mohapatra, T. A unique bacterial and archaeal diversity make mangrove a green production system compared to rice in wetland ecology: a metagenomic approach. Sci. Total Environ. 781, 146713 (2021).
Tripathi, R. et al. Soil quality in mangrove ecosystem deteriorates due to rice cultivation. Ecol. Eng. 90, 163–169 (2016).
Ruiz, F. et al. Iron’s role in soil organic carbon (de) stabilization in mangroves under land use change. Nat. Commun. 15, 10433 (2024).
Bournazel, J. et al. The impacts of shrimp farming on land-use and carbon storage around Puttalam lagoon, Sri Lanka. Ocean Coast. Manag. 113, 18–28 (2015).
Dinesh, R. & Chaudhuri, S. G. Soil biochemical/microbial indices as ecological indicators of land use change in mangrove forests. Ecol. Indic. 32, 253–258 (2013).
Liu, C. et al. Effect of land use conversion on the soil aggregate-associated microbial necromass carbon in estuarine wetland of the Pearl River in China. Catena 236, 107761 (2024).
Wang, Q. et al. Effect of land-use and land-cover change on mangrove soil carbon fraction and metal pollution risk in Zhangjiang Estuary. China. Sci. Total Environ. 807, 150973 (2022).
Arifanti, V. B., Kauffman, J. B., Hadriyanto, D., Murdiyarso, D. & Diana, R. Carbon dynamics and land use carbon footprints in mangrove-converted aquaculture: The case of the Mahakam Delta, Indonesia. For. Ecol. Manag. 432, 17–29 (2019).
Bhomia, R. K., MacKenzie, R. A., Murdiyarso, D., Sasmito, S. D. & Purbopuspito, J. Impacts of land use on Indian mangrove forest carbon stocks: implications for conservation and management. Ecol. Appl. 26, 1396–1408 (2016).
Cameron, C., Hutley, L. B., Friess, D. A. & Brown, B. Community structure dynamics and carbon stock change of rehabilitated mangrove forests in Sulawesi, Indonesia. Ecol. Appl. 29, e01810 (2019).
Cameron, C. et al. Impact of an extreme monsoon on CO2 and CH4 fluxes from mangrove soils of the Ayeyarwady Delta, Myanmar. Sci. Total Environ. 760, 143422 (2021).
Duncan, C. et al. Rehabilitating mangrove ecosystem services: a case study on the relative benefits of abandoned pond reversion from Panay Island, Philippines. Mar. Pollut. Bull. 109, 772–782 (2016).
Eid, E. M. et al. Effect of the conversion of mangroves into shrimp farms on carbon stock in the sediment along the southern Red Sea coast, Saudi Arabia. Environ. Res. 176, 108536 (2019).
Elwin, A., Bukoski, J. J., Jintana, V., Robinson, E. J. & Clark, J. M. Preservation and recovery of mangrove ecosystem carbon stocks in abandoned shrimp ponds. Sci. Rep. 9, 1–10 (2019).
Hao, L. & Huang, J. Effect of aquaculture reclamation on sediment nitrates reduction processes in mangrove wetland. J. Mar. Sci. Eng. 10, 857 (2022).
Kauffman, J. B. et al. Shrimp ponds lead to massive loss of soil carbon and greenhouse gas emissions in northeastern Brazilian mangroves. Ecol. Evol. 8, 5530–5540 (2018).
Kauffman, J. B., Heider, C., Norfolk, J. & Payton, F. Carbon stocks of intact mangroves and carbon emissions arising from their conversion in the Dominican Republic. Ecol. Appl. 24, 518–527 (2014).
Santos-Andrade, M., Hatje, V., Arias-Ortiz, A., Patire, V. F. & da Silva, L. A. Human disturbance drives loss of soil organic matter and changes its stability and sources in mangroves. Environ. Res. 202, 111663 (2021).
Wang, M. et al. Conversion of coastal marsh to aquaculture ponds decreased the potential of methane production by altering soil chemical properties and methanogenic archaea community structure. Water Res 268, 122608 (2025).
Royna, M. et al. Carbon stocks and effluxes in mangroves converted into aquaculture: a case study from Banten province, Indonesia. Front. Ecol. Evol. 12, 1340531 (2024).
Slamet, N. S., Dargusch, P., Aziz, A. A. & Wadley, D. Mangrove vulnerability and potential carbon stock loss from land reclamation in Jakarta Bay, Indonesia. Ocean Coast. Manag. 195, 105283 (2020).
Hanggara, B. B. et al. Effects of diverse mangrove management practices on forest structure, carbon dynamics and sedimentation in North Sumatra, Indonesia. Estuar. Coast. Shelf Sci. 259, 107467 (2021).
Hapsari, K. A. et al. Structure and carbon stocks of accessible mangroves under different conservation status in the Colombian Caribbean. For. Ecol. Manag. 564, 121984 (2024).
Gillis, L. G., Belshe, E. F. & Narayan, G. R. Deforested mangroves affect the potential for carbon linkages between connected ecosystems. Estuar. Coast. 40, 1207–1213 (2017).
Zanvo, S. M., Salako, K. V., Mensah, S. & Kakaï, R. G. Impacts of harvesting intensity on carbon allocation to species, size classes and pools in mangrove forests, and the relationships with stand structural attributes. Ecol. Indic. 155, 111037 (2023).
Adame, M. F. et al. Loss and recovery of carbon and nitrogen after mangrove clearing. Ocean Coast. Manag. 161, 117–126 (2018).
Arias-Ortiz, A. et al. Losses of soil organic carbon with deforestation in mangroves of Madagascar. Ecosystems 24, 1–19 (2021).
Grellier, S. et al. Changes in soil characteristics and C dynamics after mangrove clearing (Vietnam). Sci. Total Environ. 593, 654–663 (2017).
Bastakoti, U., Robertson, J., Marchand, C. & Alfaro, A. C. Mangrove removal: effects on trace metal concentrations in temperate estuarine sediments. Mar. Chem. 216, 103688 (2019).
Bernardino, A. F., de Oliveira Gomes, L. E., Hadlich, H. L., Andrades, R. & Correa, L. B. Mangrove clearing impacts on macrofaunal assemblages and benthic food webs in a tropical estuary. Mar. Pollut. Bull. 126, 228–235 (2018).
Bulmer, R. H., Lundquist, C. J. & Schwendenmann, L. Sediment properties and CO2 efflux from intact and cleared temperate mangrove forests. Biogeosciences 12, 6169–6180 (2015).
Sharma, S. et al. The impacts of degradation, deforestation and restoration on mangrove ecosystem carbon stocks across Cambodia. Sci. Total Environ. 706, 135416 (2020).
Sweetman, A. K. et al. Impacts of exotic mangrove forests and mangrove deforestation on carbon remineralization and ecosystem functioning in marine sediments. Biogeosciences 7, 2129–2145 (2010).
Dajam, A. S. et al. Mangrove (Avicennia marina) conservation contributed to a higher carbon sequestration rate at protected sites compared to overgrazed mangrove forests. J. Soil Sci. Plant Nutr. 24, 4868–4879 (2024).
Dung, L. V., Tue, N. T., Nhuan, M. T. & Omori, K. Carbon storage in a restored mangrove forest in Can Gio Mangrove Forest Park, Mekong Delta, Vietnam. For. Ecol. Manag. 380, 31–40 (2016).
Griffiths, L. N. & Mitsch, W. J. Estimating the effects of a hurricane on carbon storage in mangrove wetlands in Southwest Florida. Plants 10, 1749 (2021).
Ouyang, X., Guo, F. & Lee, S. Y. The impact of super-typhoon Mangkhut on sediment nutrient density and fluxes in a mangrove forest in Hong Kong. Sci. Total Environ. 766, 142637 (2021).
Salmo, S. G., Lovelock, C. E. & Duke, N. C. Assessment of vegetation and soil conditions in restored mangroves interrupted by severe tropical typhoon ‘Chan-hom’in the Philippines. Hydrobiologia 733, 85–102 (2014).
Sippo, J. Z. et al. Coastal carbon cycle changes following mangrove loss. Limnol. Oceanogr. 65, 2642–2656 (2020).
Chowdhury, A., Naz, A., Sharma, S. B. & Dasgupta, R. Changes in salinity, mangrove community ecology, and organic blue carbon stock in response to cyclones at Indian Sundarbans. Life 13, 1539 (2023).
de Oliveira Gomes, L. E. et al. Ecosystem carbon losses following a climate-induced mangrove mortality in Brazil. J. Environ. Manag. 297, 113381 (2021).
Harada, Y. et al. Stable isotopes track the ecological and biogeochemical legacy of mass mangrove forest dieback in the Gulf of Carpentaria, Australia. Biogeosciences 17, 5599–5613 (2020).
Perdomo-Trujillo, L. V., Mancera-Pineda, J. E., Medina-Calderón, J. H., Sánchez-Núñez, D. A. & Schnetter, M. L. Effect of Restoration actions on organic carbon pools in the lagoon—delta Ciénaga Grande de Santa Marta, Colombian Caribbean. Water 13, 1297 (2021).
Queiroz, H. M. et al. Changes in soil iron biogeochemistry in response to mangrove dieback. Biogeochemistry 158, 357–372 (2022).
Otero, X. L. et al. High fragility of the soil organic C pools in mangrove forests. Mar. Pollut. Bull. 119, 460–464 (2017).
Amiri, F. & Shabani, M. The effects of heavy metal concentration in two healthy and damaged stands of Mangrove (Avicennia marina) ecosystem in Nai Band National Park, South coast of Iran. J. Coast. Conserv. 27, 54 (2023).
Analuddin, K. et al. Blue carbon stock in sediments of mangroves and seagrass ecosystems at Southeast Sulawesi, Indonesia. Ecol. Res. 38, 508–520 (2023).
Costa, G. M. D. et al. Effects of degradation on microbial communities of an Amazonian mangrove. Microorganisms 11, 1389 (2023).
Gutiérrez, J. C. S. et al. Comparison of the mangrove soil with different levels of disturbance in tropical Agua Brava Lagoon, Mexican Pacific. Appl. Ecol. Environ. Res. 14, 45–57 (2016).
Hemati, Z., Hossain, M., Emenike, C. U. & Rozainah, M. Z. Rate of carbon storage in soil of natural and degraded mangrove forest in Peninsular Malaysia. Clean–Sediment. Air, Water 43, 614–619 (2015).
Howe, A. J., Rodríguez, J. F. & Saco, P. M. Surface evolution and carbon sequestration in disturbed and undisturbed wetland soils of the Hunter estuary, southeast Australia. Estuar. Coast. Shelf Sci. 84, 75–83 (2009).
Huang, X. et al. Responses of depth-dependence of C: N: P stoichiometry to check dam in mangrove wetlands. J. Environ. Manag. 371, 123341 (2024).
Marchio, D. A. Jr, Savarese, M., Bovard, B. & Mitsch, W. J. Carbon sequestration and sedimentation in mangrove swamps influenced by hydrogeomorphic conditions and urbanization in Southwest Florida. Forests 7, 116 (2016).
MacKenzie, R. A. et al. Sedimentation and belowground carbon accumulation rates in mangrove forests that differ in diversity and land use: a tale of two mangroves. Wetl. Ecol. Manag. 24, 245–261 (2016).
Nsombo, E. N. et al. Effects of vegetation’s degradation on carbon stock, morphological, physical and chemical characteristics of soils within the mangrove forest of the Rio del Rey Estuary: case study–Bamusso (South-West Cameroon). Afr. J. Environ. Sci. Technol. 10, 58–66 (2016).
Ochoa-Gómez, J. G. et al. Mangrove wetland productivity and carbon stocks in an arid zone of the Gulf of California (La Paz Bay, Mexico). For. Ecol. Manag. 442, 135–147 (2019).
Pérez, A. et al. Changes in organic carbon accumulation driven by mangrove expansion and deforestation in a New Zealand estuary. Estuar. Coast. Shelf Sci. 192, 108–116 (2017).
Trujillo, L. P., Mancera-Pineda, J. E., Medina-Calderon, J. H., Zimmer, M. & Schnetter, M. L. Massive loss of aboveground biomass and its effect on sediment organic carbon concentration: Less mangrove, more carbon? Estuar. Coast. Shelf Sci. 248, 106888 (2021).
Vovides, A. G., Bashan, Y., López‐Portillo, J. A. & Guevara, R. Nitrogen fixation in preserved, reforested, naturally regenerated and impaired mangroves as an indicator of functional restoration in mangroves in an arid region of Mexico. Restor. Ecol. 19, 236–244 (2011).
Zakaria, R. et al. Carbon stock of disturbed and undisturbed mangrove ecosystems in Klang Straits, Malaysia. J. Sea Res. 176, 102113 (2021).
Hong, L. C., Hemati, Z. & Zakaria, R. Carbon stock evaluation of selected mangrove forests in peninsular Malaysia and its potential market value. J. Environ. Sci. Manag. 20-2, 77–87 (2017).
Senger, D. F. et al. Impacts of wetland dieback on carbon dynamics: A comparison between intact and degraded mangroves. Sci. Total Environ. 753, 141817 (2021).
Chen, J. et a. Dredging wastewater discharge from shrimp ponds affects mangrove soil physical-chemical properties and enzyme activities. Sci. Total Environ. 926, 171916 (2024).
Nóbrega, G. N., Ferreira, T. O., Romero, R. E., Marques, A. G. B. & Otero, X. L. Iron and sulfur geochemistry in semi-arid mangrove soils (Ceará, Brazil) in relation to seasonal changes and shrimp farming effluents. Environ. Monit. Assess. 185, 7393–7407 (2013).
Queiroz, H. M. et al. Hidden contribution of shrimp farming effluents to greenhouse gas emissions from mangrove soils. Estuar. Coast. Shelf Sci. 221, 8–14 (2019).
Rodrigues, J. V., Cotovicz, L. C. Jr, Beloto, N., Gmach, M. R. & Bezerra, L. E. A. Historical land use changes lead to massive loss of soil carbon stocks in a recovering, semiarid mangrove. Mar. Pollut. Bull. 208, 116980 (2024).
Suárez-Abelenda, M. et al. The effect of nutrient-rich effluents from shrimp farming on mangrove soil carbon storage and geochemistry under semi-arid climate conditions in northern Brazil. Geoderma 213, 551–559 (2014).
Tian, Y., Chen, G., Lu, H., Zhu, H. & Ye, Y. Effects of shrimp pond effluents on stocks of organic carbon, nitrogen and phosphorus in soils of Kandelia obovata forests along Jiulong River Estuary. Mar. Pollut. Bull. 149, 110657 (2019).
Bu, N. S. et al. Reclamation of coastal salt marshes promoted carbon loss from previously-sequestered soil carbon pool. Ecol. Eng. 81, 335–339 (2015).
Cui, J. et al. Bacterial succession during 500 years of soil development under agricultural use. Ecol. Res. 27, 793–807 (2012).
Li, J. et al. Effect of reclamation on soil organic carbon pools in coastal areas of eastern China. Front. Earth Sci. 12, 339–348 (2018).
Wang, P. et al. Long-term rice cultivation stabilizes soil organic carbon and promotes soil microbial activity in a salt marsh derived soil chronosequence. Sci. Rep. 5, 1–13 (2015).
Bai, J. et al. Soil organic carbon as affected by land use in young and old reclaimed regions of a coastal estuary wetland, China. Soil Use Manag. 29, 57–64 (2013).
Santín, C. et al. Effects of reclamation and regeneration processes on organic matter from estuarine soils and sediments. Org. Geochem. 40, 931–941 (2009).
Van de Broek, M., Baert, L., Temmerman, S. & Govers, G. Soil organic carbon stocks in a tidal marsh landscape are dominated by human marsh embankment and subsequent marsh progradation. Eur. J. Soil Sci. 70, 338–349 (2019).
Wang, Y., Wang, Z. L., Feng, X., Guo, C. & Chen, Q. Long-term effect of agricultural reclamation on soil chemical properties of a coastal saline marsh in Bohai Rim, Northern China. PLoS ONE 9, e93727 (2014).
Xu, C., Pu, L., Li, B., Xu, Y. & Kong, F. Coupling mechanism of C: N: P stoichiometry between plant and soil after long-term reclamation in the coastal area, eastern China. Reg. Stud. Mar. Sci. 68, 103178 (2023).
Yang, W. et al. Shift in soil organic carbon and nitrogen pools in different reclaimed lands following intensive coastal reclamation on the coasts of eastern China. Sci. Rep. 9, 5921 (2019).
Wan, S., Liu, X. & Mu, X. Vertical distribution characteristics of carbon and nitrogen contents in soils of 4 types of wetlands in Shuangtai River Estuary. Wetland Sci 15, 629–634 (2017).
Yang, P. et al. Soil organic nitrogen mineralization and N2O production driven by changes in coastal wetlands. Glob. Biogeochem. Cycles 38, e2024GB008154 (2024).
Li, T. et al. Harvested Spartina area performs better than native Scirpus in sedimentation and carbon preservation under storm surge. Ocean Coast. Manag. 249, 107002 (2024).
Cai, T. et al. Saltmarsh carbon stock changes under combined effects of vegetation succession and reclamation. Ecosyst. Health Sustain. 9, 0114 (2023).
Feng, H. et al. The responses of soil bacterial and archaeal communities to coastal embankments in three typical salt marshes of Eastern China. Plant Soil 477, 439–459 (2022).
Hussain, S. et al. Composition and assembly mechanisms of prokaryotic communities in wetlands, and their relationships with different vegetation and reclamation methods. Sci. Total Environ. 897, 166190 (2023).
Lewis, C. J. E. et al. Impacts of land reclamation on tidal marsh ‘blue carbon’ stocks. Sci. Total Environ. 672, 427–437 (2019).
Li, J. et al. The impact of sea embankment reclamation on greenhouse gas GHG fluxes and stocks in invasive Spartina alterniflora and native phragmites australis wetland marshes of East China. Sustainability 13, 12740 (2021).
Yang, W. et al. The impact of sea embankment reclamation on soil organic carbon and nitrogen pools in invasive Spartina alterniflora and native Suaeda salsa salt marshes in eastern China. Ecol. Eng. 97, 582–592 (2016).
Yang, W. et al. Seawall construction alters soil carbon and nitrogen dynamics and soil microbial biomass in an invasive Spartina alterniflora salt marsh in eastern China. App. Soil Ecol. 110, 1–11 (2017).
Zhou, S. & Bi, X. Seawall effects in a coastal wetland landscape: spatial changes in soil carbon and nitrogen pools. J. Coast. Conserv. 24, 11 (2020).
Day, J. W. et al. Vegetation death and rapid loss of surface elevation in two contrasting Mississippi delta salt marshes: The role of sedimentation, autocompaction and sea-level rise. Ecol. Eng. 37, 229–240 (2011).
Schoolmaster, D. R. Jr. et al. A model of the spatiotemporal dynamics of soil carbon following coastal wetland loss applied to a Louisiana salt marsh in the Mississippi River Deltaic Plain. J. Geophys. Res. Biogeosci. 127, e2022JG006807 (2022).
Ford, H., Garbutt, A., Jones, L. & Jones, D. L. Methane, carbon dioxide and nitrous oxide fluxes from a temperate salt marsh: Grazing management does not alter Global Warming Potential. Estuar. Coast. Shelf Sci. 113, 182–191 (2012).
Harvey, R. J., Garbutt, A., Hawkins, S. J. & Skov, M. W. No detectable broad-scale effect of livestock grazing on soil blue-carbon stock in salt marshes. Front. Ecol. Evol. 7, 151 (2019).
Olsen, Y. S. et al. Cattle grazing drives nitrogen and carbon cycling in a temperate salt marsh. Soil Biol. Biochem. 43, 531–541 (2011).
Persico, E. P., Sharp, S. J. & Angelini, C. Feral hog disturbance alters carbon dynamics in southeastern US salt marshes. Mar. Ecol. Prog. Ser. 580, 57–68 (2017).
Sharp, S. J. et al. Large grazers suppress a foundational plant and reduce soil carbon concentration in eastern US saltmarshes. J. Ecol. 112, 2624–2637 (2024).
Yang, Z., Nolte, S. & Wu, J. Tidal flooding diminishes the effects of livestock grazing on soil micro-food webs in a coastal saltmarsh. Agric. Ecosyst. Environ. 236, 177–186 (2017).
Zhang, P., Yang, Z. & Wu, J. Livestock grazing promotes ecosystem multifunctionality of a coastal salt marsh. J. Appl. Ecol. 58, 2124–2134 (2021).
Xie, L. et al. Impacts of coppicing on Tamarix chinensis growth and carbon stocks in coastal wetlands in northern China. Ecol. Eng. 147, 105760 (2020).
Macreadie, P. I., Hughes, A. R. & Kimbro, D. L. Loss of ‘blue carbon’ from coastal salt marshes following habitat disturbance. PloS ONE 8, e69244 (2013).
Howard, R. J., Wells, C. J., Michot, T. C. & Johnson, D. J. Effects of disturbance associated with seismic exploration for oil and gas reserves in coastal marshes. Environ. Manag. 54, 30–50 (2014).
Barañano, C., Fernández, E. & Méndez, G. Clam harvesting decreases the sedimentary carbon stock of a Zostera marina meadow. Aquat. Bot. 146, 48–57 (2018).
Branco, J. et al. Natural recovery of Zostera noltii seagrass beds and benthic nematode assemblage responses to physical disturbance caused by traditional harvesting activities. J. Exp. Mar. Bio. Ecol. 502, 191–202 (2018).
Román, M. et al. Loss of surficial sedimentary carbon stocks in seagrass meadows subjected to intensive clam harvesting. Mar. Environ. Res. 175, 105570 (2022).
Do, V. T., de Montaudouin, X., Blanchet, H. & Lavesque, N. Seagrass burial by dredged sediments: Benthic community alteration, secondary production loss, biotic index reaction and recovery possibility. Mar. Pollut. Bull. 64, 2340–2350 (2012).
Dahl, M. et al. Effects of shading and simulated grazing on carbon sequestration in a tropical seagrass meadow. J. Ecol. 104, 654–664 (2016).
Fu, M., Song, Y., Wang, Y., Fu, G. & Zhang, X. Effects of Shading on the Growth and Carbon Storage of Enhalus acoroides. Appl. Sci. 13, 6035 (2023).
Trevathan‐Tackett, S. M. et al. Effects of small‐scale, shading‐induced seagrass loss on blue carbon storage: Implications for management of degraded seagrass ecosystems. J. Appl. Ecol. 55, 1351–1359 (2018).
Githaiga, M. N., Frouws, A. M., Kairo, J. G. & Huxham, M. Seagrass removal leads to rapid changes in fauna and loss of carbon. Front. Ecol. Evol. 7, 62 (2019).
Macreadie, P. I. et al. No detectable impact of small-scale disturbances on ‘blue carbon’ within seagrass beds. Mar. Biol. 161, 2939–2944 (2014).
Carnell, P. E., Ierodiaconou, D., Atwood, T. B. & Macreadie, P. I. Overgrazing of seagrass by sea urchins diminishes blue carbon stocks. Ecosystems 23, 1437–1448 (2020).
Aoki, L. R. et al. Seagrass recovery following marine heat wave influences sediment carbon stocks. Front. Mar. Sci. 7, 576784 (2021).
Casal-Porras, I. et al. Sedimentary organic carbon and nitrogen stocks of intertidal seagrass meadows in a dynamic and impacted wetland: Effects of coastal infrastructure constructions and meadow establishment time. J. Environ. Manag. 322, 115841 (2022).
Marbà, N. et al. Impact of seagrass loss and subsequent revegetation on carbon sequestration and stocks. J. Ecol. 103, 296–302 (2015).
Macreadie, P. I. et al. Losses and recovery of organic carbon from a seagrass ecosystem following disturbance. Proc. R. Soc. B Biol. Sci. 282, 20151537 (2015).
Moksnes, P. O. et al. Major impacts and societal costs of seagrass loss on sediment carbon and nitrogen stocks. Ecosphere 12, e03658 (2021).
Piñeiro-Juncal, N. et al. Cover loss in a seagrass Posidonia oceanica meadow accelerates soil organic matter turnover and alters soil prokaryotic communities. Org. Geochem. 151, 104140 (2021).
Yue, S. et al. The super typhoon Lekima (2019) resulted in massive losses in large seagrass (Zostera japonica) meadows, soil organic carbon and nitrogen pools in the intertidal Yellow River Delta, China. Sci. Total Environ. 793, 148398 (2021).
Chen, S. et al. Implication of macroalgal bloom to soil organic carbon stock in seagrass meadows-a case study in South Hainan, China. Front. Mar. Sci. 9, 870228 (2022).
Jiang, Z. et al. Eutrophication indirectly reduced carbon sequestration in a tropical seagrass bed. Plant Soil 426, 135–152 (2018).
Reyes, A. G. B., Vergara, M. C. S., Blanco, A. C. & Salmo, S. G. III Seagrass biomass and sediment carbon in conserved and disturbed seascape. Ecol. Res. 37, 67–79 (2022).
Stankovic, M., Tantipisanuh, N. & Prathep, A. Carbon storage in seagrass ecosystems along the Andaman coast of Thailand. Bot. Mar. 61, 429–440 (2018).
Thorhaug, A., Poulos, H. M., López-Portillo, J., Ku, T. C. & Berlyn, G. P. Seagrass blue carbon dynamics in the Gulf of Mexico: Stocks, losses from anthropogenic disturbance, and gains through seagrass restoration. Sci. Total Environ. 605, 626–636 (2017).
Rahayu, Y. P. et al. Sedimentary seagrass carbon stock and sources of organic carbon across contrasting seagrass meadows in Indonesia. Environ. Sci. Pollut. Res. 30, 97754–97764 (2023).
Gedan, K. B., Silliman, B. R. & Bertness, M. D. Centuries of human-driven change in salt marsh ecosystems. Annu. Rev. Mar. Sci. 1, 117–141 (2009).
Goldstein, A. et al. Protecting irrecoverable carbon in Earth’s ecosystems. Nat. Clim. Change 10, 287–295 (2020).
Ouyang, X. & Lee, S. Y. Improved estimates on global carbon stock and carbon pools in tidal wetlands. Nat. Commun. 11, 317 (2020).
Fourqurean, J. W. et al. Seagrass ecosystems as a globally significant carbon stock. Nat. Geosci. 5, 505–509 (2012).
Emde, D., Hannam, K. D., Most, I., Nelson, L. M. & Jones, M. D. Soil organic carbon in irrigated agricultural systems: A meta‐analysis. Glob. Change Biol. 27, 3898–3910 (2021).
Breiman, L. Random forests. Mach. Learn. 45, 5–32 (2001).
Zhou, Y. et al. Soil carbon in tropical savannas mostly derived from grasses. Nat. Geosci. 16, 710–716 (2023).
Kauffman, J. B. & Donato, D. C. Protocols for the Measurement, Monitoring and Reporting of Structure, Biomass and Carbon Stocks in Mangrove Forests (Center for International Forestry Research (CIFOR), 2012).
Lee, J., Hopmans, J. W., Rolston, D. E., Baer, S. G. & Six, J. Determining soil carbon stock changes: simple bulk density corrections fail. Agric. Ecosyst. Environ. 134, 251–256 (2009).
Hedges, L. V., Gurevitch, J. & Curtis, P. S. The meta-analysis of response ratios in experimental ecology. Ecology 80, 1150–1156 (1999).
Park, S. & Beretvas, S. N. Using total sample size weights in meta-analysis of log-odds ratios. J. Exp. Educ. 87, 400–414 (2019).
Chen, X. & Chen, H. Y. Plant mixture balances terrestrial ecosystem C: N: P stoichiometry. Nat. Commun. 12, 4562 (2021).
Wang, Y. et al. Effects of land clearing for agriculture on soil organic carbon stocks in drylands: a meta‐analysis. Glob. Change Biol. 29, 547–562 (2023).
Acknowledgements
This research was funded by Convex Seascape Survey with funding provided to C.M.D. (RGC/3/5372-01-01). We thank D. Emde, Y. Zhou, and Y. Wang for their help in developing code for soil organic carbon and dry bulk density modeling and for weighting the effect size in the data synthesis.
Author information
Authors and Affiliations
Contributions
C.M.D. conceived this study. C.F., S.G.K. and J.B. performed the paper searching and screening. C.F. collected data and conducted the analyses with the support from S.G.K., K.K.L. and A.S. C.F. and C.M.D. wrote the original manuscript. All authors contributed to reviewing and editing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Chaofan Li, Guanghui Lin and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Fu, C., Klein, S.G., Breavington, J. et al. Nonuniform organic carbon stock loss in soils across disturbed blue carbon ecosystems. Nat Commun 16, 4370 (2025). https://doi.org/10.1038/s41467-025-59752-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-59752-9