Abstract
Long-persistent afterglows based on organic donor-acceptor systems feature ultra-long duration reaching hours, leading to the advantages in long-time-range display and bio/medical applications. However, the understanding of this optical phenomenon is insufficient. Herein, dibenzothiophene-phosphine oxide hybrids named nDBTxPO with different room temperature phosphorescence characteristics are used as acceptor matrixes. It shows that after doping N,N,N′,N′-tetramethylbenzidine (TMB) as donor, afterglow intensities and durations of nDBTxPO:1% TMB are strongly correlated to the stabilized triplet state (Tn*) properties of nDBTxPO. Compared to other congeners, high-population and high-lying Tn* state of 28DBTDPO matrix supports the positive and efficient energy transfer to nCT* states of 28DBTDPO-TMB local dipoles in the doped film. This energy relay between two long-persistent Tn* and nCT* states is the key determinant resulting in the longest afterglow of 28DBTDPO:1% TMB. This work provides clear insight into energy transfer for lightly-doping donor-acceptor systems, therefore will promote the accurate system design for practical applications.
Similar content being viewed by others
Introduction
Compared to inorganic afterglow materials, pure-organic molecules with long-persistent luminescence in solid states emerged rapidly in recent decades, owing to their advantages in low-temperature processability, low cost, good sustainability, widely tunable gamut, and so on1,2,3,4. The flexible and diverse structures and clear and tunable excited-state characteristics of organic molecules were utilized to achieve desired long-persistent luminescent performance for versatile applications, e.g., information display and anti-counterfeiting encryption5,6,7, biological imaging8,9, X-ray scintillation10,11, and light-emitting devices12,13. Since long-persistence afterglow is through a prolonged photon release from excited states after excitation source removal, emission duration and efficiency are two key determinants, which are in proportion to excited-state stability and radiation probability, respectively14,15,16.
Obviously, one prerequisite is effectively suppressing non-radiative deactivation of excited states. It was verified that molecular vibrations and rotations can be restrained by crystallization17,18, aggregation with p–π and π–π interactions19,20,21,22, self-assembly by hydrogen bonds23,24,25 and fixation in rigid matrixes26, which effectively reduce phonon relaxation-induced non-radiation, and therefore stabilize long-lifetime triplet states. Heavy atom substitution enhances spin−orbital coupling, and thereby facilitates intersystem crossing (ISC) to further increase triplet population27,28,29,30. Despite room-temperature phosphorescence achieved by these organic systems, the energy transfer between their excited states was still small, rendering emission duration time commonly limited to 10 s. In contrast, multi-component donor–acceptor systems have an advantage in emission duration, whose ultra-long afterglows can persist dozens of minutes and even several hours31,32,33,34,35. Therefore, these donor–acceptor systems are more advantageous for applications in large time scales33,36,37,38.
It is believed that long-persistent luminescence from donor–acceptor doping systems followed two mechanisms: (i) energy transfer: Förster and/or Dexter energy transfer from long-persistent emissive donors to acceptors can give rise to afterglows from the latter, making lifetimes further elongated and multi-color emissions feasible39; (ii) charge trapping: electron transfer from the highest occupied molecular orbital (HOMO) of donor dopants to the lowest unoccupied molecular orbital (LUMO) of acceptor matrixes generates charge-transfer excited states (nCT*, n = 1 and 3 for singlet and triplet states, respectively)40. The long-range charge separation and trapping/de-trapping postpones charge recombination and radiative transitions, therefore elongating emission lifetimes41.
For these multicomponent systems, low doping concentration is crucial for increasing emission duration, which can not only reduce the rate and extend the time for energy transfer, but also increase the distances of charge delocalization. Nonetheless, even at the same doping concentrations, e.g., 1.0 wt%, charge trapping-featured donor–acceptor systems can achieve afterglows persisting for several hours40,41,42,43, which were more than one order of magnitude longer than those of energy transfer-based systems with lifetimes of dozens minutes44 or even several seconds33,45. Obviously, the understanding of the accurate mechanism of excited-state evolution for doping-induced afterglow is still unclear and challenging. It is known that charge and energy transfer processes are often associated, although their differences in time scale may shorten emission duration. Actually, energy transfer from nCT* states of charge-trapping systems to locally excited states of fluorescent dopants can give rise to the afterglows of the latter46. So, charge and energy transfer can be integrated and mutually synergistic in luminescent process, but should occur sequentially to avoid competition in de-activation.
It is convincing that through rationally combining both “energy transfer” and “charge trapping” mechanisms, ultra-long persistent luminescence can be realized through the room temperature phosphorescence acceptor matrixes lightly doped by donors, in which singlet and triplet energy levels of donors and acceptors are comparable, leading to the formation of local bimolecular dipoles similar to donor–acceptor exciplexes with nCT* states (Fig. 1a). In this sense, considering donor–acceptor dipoles of local bimolecular couples as a unit, the systems can be designated as dipole-doped room temperature phosphorescence matrixes. Thus, step-by-step energy transfer from the single-molecular first triplet states (T1), over multi-molecule stabilized triplet states (Tn*) of room temperature phosphorescence matrixes to nCT* states of dipoles can give rise to ultra-long afterglow47. Obviously, nCT* set up the basis of long-persistent luminescence, but Tn* states play the crucial role in excited-state energy relay from T1 to nCT*, whose incorporation can further postpone nCT* formation, finally resulting in ultra-long afterglow. However, the understanding of this energy relay process and its key influencing factors is still insufficient and unclear. Regarding to energy level relationship, if Tn* states were energetically higher than nCT* to make energy transfer exothermic, the energy relay would have thermodynamic advantage, thus be inevitable and efficient; in the opposite case, energy transfer from lower Tn* to higher nCT* is endothermic but time-taking, reducing possibility of energy relay in spite of longer duration.
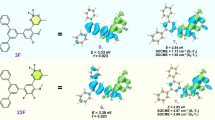
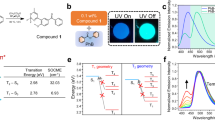
a Energy transfer diagram of room temperature phosphorescence (RTP) acceptor locally doped by donor with the compatible first singlet (S1) and triplet (T1) energy levels. Donor–acceptor (D-A) electronic coupling generates local dipoles featuring the low-lying singlet (1CT*) and triplet charge transfer (3CT*) states. The stabilized triplet states (Tn*) of RTP acceptors can serve as the intermediate states for energy relay to D-A dipoles, which are influenced by the energy-level order of Tn* and nCT* (n = 1 or 3) states. ISC and RISC refer to forward and reverse intersystem crossing. The positive energy transfer based on “Tn* > nCT*” with thermodynamic predominance to the endothermal case of “Tn* < nCT*”. b Chemical structures of RTP acceptors nDBTxPO and donor TMB, and the corresponding highest occupied (HOMO) and lowest unoccupied (LUMO) molecular orbitals. nDBTxPO-TMB dipoles are highlighted with a dashed circle. c Photos of melt films based on nDBTxPO and nDBTxPO:1% TMB excited with UV light at 365 nm and after UV excitation for 10 s and 30 min, corresponding to time ranges for RTP enhanced by aggregation and long-persistent afterglow prolonged by D-A dipoles, respectively.
In this work, different to previous reports focused on nCT* formation, a series of dibenzothiophene-phosphine oxide (PO) hybrids collectively named nDBTxPO, including two monophosphine oxides (2DBTSPO and 4DBTSPO) and two biphosphine oxides (28DBTDPO and 46DBTDPO), are used as acceptor matrixes 1% doped with N,N,N’,N’-tetramethylbenzidine (TMB) as donor to figure out the whole energy relay process (Fig. 1b and Supplementary Fig. 1). The LUMOs and HOMOs of PO matrixes are 0.5−1.5 eV shallower than those of TMB (Supplementary Fig. 2), rendering high enough driving forces of donor–acceptor electronic coupling to enhance intermolecular charge transfer in local and discrete PO:TMB dipoles. It was believed that charge trapping by dipoles induced afterglow from 28DBTDPO:1% TMB melt films40,48,49. However, compared to melt films of pure monophosphine oxides having weak room-temperature phosphorescence, pure biphosphine oxide melt films reveal efficient room-temperature phosphorescence in aggregation states, with a duration of dozens of seconds and quantum efficiencies of 10−14%, respectively (Fig. 1c and Supplementary Table 1). Notably, 1% TMB doping endows 2DBTSPO featuring negligible room temperature phosphorescence with bright afterglow, whose duration is even comparable to those of 4DBTSPO/46DBTDPO:1% TMB melt films, despite its quantum efficiency of ~6% is only one quater of those of the latter (22% and 29%). Moreover, although the room temperature phosphorescence of 46DBTDPO melt film is longer and more efficient than that of 4DBTSPO, 1% TMB doping induces their nearly identical afterglow properties. 28DBTDPO:1% TMB melt film displays the longest afterglow duration of several hours and the highest and threefold-increased quantum efficiency reaching 42% (Supplementary Table 2). Obviously, different to 2DBTSPO, 4DBTSPO and 46DBTDPO, 28DBTDPO matrix supports the desired long-persistent afterglow based on the accumulation of its aggregation-stablized Tn* state and dipole-attributed nCT* states in the whole afterglow process. Since the excited state charateristics of PO acceptors can be controllably modulated by facile chemical modification, the understanding of this energy relay mechanism provide deep insight into the determinants of ultralong afterglow and the feasible and effective approaches for desired performance optimization.
Results
Donor–acceptor system design
Steric hindrance of diphenylphosphine oxide group is used to adjust the intermolecular interactions and packing modes on dibenzothiophene chromophores of the acceptor matrixes through changing substitution positions, which would influence the properties of Tn* states (Supplementary Figs. 3−8). The interruptions from impurities are negligible, since the high purities of matrixes and doped films (Supplementary Figs. 9, 10). Compared to unsymmetrical monophosphine oxides with weaker π–π interactions, biphosphine oxides are more effective in triplet stabilization by intermolecular π–π stacking, in accord with the high decomposition temperatures of the latter (Supplementary Fig. 11). Nonetheless, all the neat and doped films are basically amorphous (Supplementary Fig. 12). Furthermore, dibenzothiophene in 46DBTDPO is exposed at the opposite side of diphenylphosphine oxide groups, facilitating ordered aggregation in quasi-crystal structures50. The formation of intermolecular charge transfer between nDBTxPO and TMB is evidenced by the red-shifted and markedly broadened emissions of nDBTxPO:1% TMB solutions, as well as elongated emission lifetimes, in comparison to emission spectra of nDBTxPO and TMB in solutions (Supplementary Figs. 13−15). The first singlet (S1) and T1 energy levels of nDBTxPO and TMB are comparable, making the generation of nCT* states by singlet and triplet coupling feasible.
Luminescence properties
All the PO molecules have the identical electronic absorption, fluorescence and phosphorescence spectra, attributed to their same dibenzothiophene chromophore based locally excited states (Fig. 1a and Supplementary Figs. 16−18). Therefore, owing to insulating effect of P = O, the first singlet and triplet energy levels of PO molecules are almost equal. Aggregation of PO molecules in their as-prepared powders and melt films induces emission red-shifts by ~80 nm of steady-state photoluminescence spectra (Fig. 2a and Supplementary Fig. 19). Different to melt films of its congeners with near-ultraviolet fluorescence as primary emissions, the main peak of 28DBTDPO melt film reveals dual emission bands respectively centered at about 360 and 500 nm. More importantly, its green emission band is predominant and thoroughly overlapped with its room-temperature phosphorescence, indicating the efficient Tn* state population of 28DBTDPO matrix (Supplementary Fig. 20a). It is noted that the T1 - Tn* energy gap of 28DBTDPO matrix is only 0.1 eV, evidenced by its similar single-molecular phosphorescence and room-temperature phosphorescence spectra, which benefits triplet energy transfer. Furthermore, Tn* states can also support effective energy transfer to nCT* states, owing to their similar intermolecular excited characteristics. However, the Tn* energy levels of 4DBTSPO and 46DBTDPO matrixes markedly decrease to 2.6 eV, in comparison to 3.0 eV of 28DBTDPO matrix.
Electronic absorption (Abs.), fluorescence (FL) and phosphorescence (PH) spectra in dilute dichloromethane (DCM), and photoluminescence (PL) and RTP/afterglow spectra of (a) nDBTxPO and (b) nDBTxPO:1% TMB. PH spectra in solution were measured at 77 K after a delay of 3 ms; RTP and afterglow spectra were recorded after a delay of 30 ms. All the emission spectra were measured with excitation at 280 nm. The energy levels of Tn* and 3CT* states were estimated according to edges of RTP and afterglow spectra, respectively; (c) Comparison on the different energy transfer processes in melt films of 28DBTDPO:1% TMB and 46DBTDPO:1% TMB, in which higher Tn* energy level of 28DBTDPO matrix supports exothermal energy transfer to nCT* (n = 1 and 3 for singlet and triplet, respectively) energy levels of 28DBTDPO-TMB dipoles, on the contrary to endothermal energy transfer in the latter. d Excitation−emission spectral mapping of melt films based on nDBTxPO and nDBTxPO:1% TMB at room temperature. The correlations between the excitations of RTP and afterglow from neat and 1% TMB doped nDBTxPO films, respectively, are indicated by dash lines.
All PO:1% TMB melt films reveal the similar photoluminescence spectra, which are also identical to their afterglows (Fig. 2b, Supplementary Fig. 20b and Supplementary Table 2). Time decays of the doped reveal evolution from exponential components in the range of 0−10 s to power-law components after 10 s, respectively, originated from Tn* and nCT* states doped films, which directly evidences the energy transfer from Tn* to nCT* states (Supplementary Figs. 20c, d). The power-law decays of PO:1% TMB reflect typical single-photon processes through charge separation, transfer and recombination, which are comparable, in accord with the similar LUMO energy levels and electron transporting abilities of nDBTxPO (Supplementary Fig. 21).
It indicates the profiles of emissions from these doped melt films are identical to those of exciplex emissions from PO:TMB mixtures in dilute toluene solution (Supplementary Fig. 22). It is convincing that emission origins of PO:1% TMB melt films are the same as nCT* states of local PO-TMB dipoles with energy levels of ~2.7 eV, which are based on the near-zero singlet-triplet splitting energies and the balance of intersystem crossing and reverse intersystem crossing (Supplementary Fig. 23). The charge separation in PO:1% TMB melt films is further demonstrated by the absorption peaks in their electron paramagnetic resonance spectra (Supplementary Fig. 24). Oxygen can also markedly shorten emission durations of all the doped films, due to the quenching effects on triplet states and radicals involved in afterglow processes (Supplementary Fig. 25). These results are in accordance with the time-depenent density functional theory simulations on the short intermolecualr distances and the charge transfer featured singlet and triplet excitations of nDBTxPO-TMB dipoles (Supplementary Figs. 26 and 27). The emission spectra of TMB doped 28DBTDPO or 46DBTDPO melt films are almost independent on TMB concentrations, but afterglow durations are marked decreased at high TMB concentrations, which is in accord to nCT* predominant “charge trapping” mechanism (Supplementary Figs. 28 and 29). Then, doping nDBTxPO-TMB systems in rigid polymethylmethacrylate (PMMA) makes afterglow vanished, since charge transfer and recombination are blocked by electrically inertia PMMA matrix (Supplementary Fig. 30).
The higher Tn* energy level of 28DBTDPO matrix leads to exothermic energy transfer to local dipoles in 28DBTDPO:1% TMB melt film; meanwhile, the suitable Tn*-nCT* energy gap of 0.3 eV supports positive energy transfer with limited energy loss. On the contrary, in 1% TMB doped 4DBTSPO and 46DBTDPO melt films, energy transfer from the lower Tn* energy levels of PO matrixes to nCT* states is endothermic (Fig. 2c). Consistently, excitation wavelengths of afterglow for 28DBTDPO:1% TMB are nearly equal to those of room-temperature phosphorescence for the matrix rather than TMB, indicating the same origins of Tn* and nCT* states (Fig. 2d and Supplementary Figs. 31−33). In contrast, 4DBTSPO and 46DBTDPO matrixes show absorption and excitation wavelengths of room-temperature phosphorescence even longer than those of afterglows for their 1% TMB doped melt films (Supplementary Fig. 34).
Compared to as-prepared powders, room temperature phosphorescence durations of nDBTxPO melt films are markedly improved, owing to improved molecular orders and environment rigidity in melt films (Supplementary Fig. 35). Furthermore, although room temperature phosphorescence lifetimes of 4DBTSPO are only one-fifth of those of 46DBTDPO, afterglow lifetimes of their 1% TMB doped films are nearly equal, which are even comparable to those of film based on 2DBTSPO matrix with negligible room temperature phosphorescence (Fig. 3a, Supplementary Fig 20c and Supplementary Table 1). Thus, Tn* characteristics of 2DBTSPO, 4DBTSPO and 46DBTDPO matrixes only slightly influence afterglow durations of their doped films (Supplementary Fig. 20d and Supplementary Table 2). Time-resolved emission spectra of PO matrixes verify that their room-temperature phosphorescence occurs through the typical mechanism of T1 → Tn* energy transfer (Fig. 3b). Tn* populations are in direct proportion to efficiencies and lifetimes of room-temperature phosphorescence, namely 28DBTDPO » 46DBTDPO > 4DBTSPO » 2DBTSPO. It shows that emission bands ascribed to T1 and Tn* states of 2DBTSPO and 28DBTDPO and nCT* states of dipoles sequentially occur (Fig. 3c). In contrast, Tn* and nCT* attributed bands of 4DBTSPO/46DBTDPO:1% TMB melt films are simultaneously observed, and the latter is markedly stronger.
a Time decays of RTP and afterglow from melt films of nDBTxPO and nDBTxPO:1% TMB; Time-resolved emission spectra (TRES) of (b) nDBTxPO and (c) nDBTxPO:1% TMB melt films in 0−200 µs, and the corresponding sliced TRES in the same time range. Energy transfer processes are indicated with arrows. Two different energy transfer modes of nDBTxPO:1% TMB melt films are further illustrated in two insets, respectively. For 2DBTSPO/28DBTDPO:1% TMB, successive triplet population evolution can be recognized, and based on the energy relay of T1 → Tn* → CT* states, in which the profiles of T1 and Tn* attributed intermediate bands are highlighted with dash lines. While, for 4DBTSPO/46DBTDPO:1% TMB, reverse energy transfers of Tn* → CT* states are indicated by narrowed emission profiles; (d) Temperature-dependent TRES (left) and sliced PL spectra at the first 200 µs (right) of 28DBTDPO:1% TMB and 46DBTDPO:1% TMB melt films in the range of 20 K and room temperature. The TRES variation tendencies and emission-band attributions are highlighted with arrows.
Afterglow mechanism
Beyond simple “charge trapping” mechanim, the whole energy transfer process in 28DBTDPO:1% TMB melt film should be involved both two long-persistent Tn* state of 28DBTDPO matrix and nCT* state of 28DBTDPO-TMB dipoles: S1 → T1 → Tn* → nCT*, giving rise to longer duration of afterglow (insets in Fig. 3c and Supplementary Fig. 36). Since Tn* population of 2DBTSPO matrix is the weakest, the afterglow of its 1% TMB doped melt film is mainly attributed to direct energy trapping by local dipoles, namely S1 → T1 → nCT*. Since Tn* energy levels of 4DBTSPO and 46DBTDPO matrixes are lower than nCT* energy levels, these two energy transfer processes can simultaneously contribute to afterglows of 4DBTSPO/46DBTDPO:1% TMB melt films: (i) direct way similar to 2DBTSPO-based analogs, in which Tn* states are excluded from energy transfer; and (ii) reverse way, in which triplet energy transfer is intermediated by Tn* states like the case of 28DBTDPO hosted analogs, but based on an upconversion rather than downconversion to nCT* states. It is known that compared to endothermic process, exothermic energy transfer would have larger transition possibility, since it does not need activation energy to overcome the energy barrier. Therefore, exothermic energy transfer can increase the contribution of long-lived nCT* states to final alfterglow. In addition, the Tn* lifetime of 28DBTDPO matrix is the longest among the phosphine oxide matrixes (Supplementary Table 1). As a result, 28DBTDPO:1% TMB melt film achieves the longest afterglow duration and efficiency, and its highest afterglow rate constant further demonstrates the synergistic effects between Tn* and nCT* states (Supplementary Table 2).
At 20−298 K, photoluminescence spectra of 28DBTDPO:1% TMB melt film only consist of emission bands from nCT* states, which are unchanged in the first 200 µs after excitation removal (Fig. 3d). This temperature-independent feature and exclusion of the T1 and Tn* states for neat 28DBTDPO melt film at low temperatures are in accord with the positive and endothermic energy transfer from room-temperature phosphorescence matrix to dipoles (Supplementary Fig. 37a)51,52. However, for 46DBTDPO:1% TMB melt film, room-temperature phosphorescence bands from Tn* states of 46DBTDPO matrixes can be always distinguished in the first 200 µs (Supplementary Fig. 37b), whose intensity is in reverse proportion to temperature, and always comparable to 46DBTDPO-TMB dipole emission band at 20−200 K, due to exothermic feature of Tn* → nCT* upconversion. The afterglow mechanism of 4DBTSPO:1% TMB melt film is identical (Supplementary Fig. 38). Obviously, such reverse energy transfer from low-triplet-energy PO matrixes to dipoles can not facilitate effective long-persistent afterglow. At the same time, because of reduced triplet quenching and transfer at low temperature, the T1 emission bands from 4DBTSPO and 46DBTDPO molecules also appear at 20−200 K. The situation of 2DBTSPO:1% TMB melt film is similar, since only the T1 state of 2DBTSPO molecule is predominant in energy transfer to nCT* states (Supplementary Fig. 39). Since 2DBTSPO, 4DBTSPO and 46DBTDPO matrixes with markedly different Tn* populations support the comparable afterglow durations to their doped films, the direct energy trapping mechanism with minor contributions from Tn* states should be predominant for their 1% TMB doped melt films, namely S1 → T1 → nCT*, different to energy relay mechanism for 28DBTDPO:1% TMB melt films (Supplementary Fig. 40).
Therefore, it is convincing that for donor–acceptor afterglow systems, the energy relay of two long-lived excited states would be beneficial to improve efficiencies and durations of long-persistent luminescence, provided the matrixes had stabilized excited states and energy levels of frontier molecular orbitals and excited states matching with dopants.
Application demonstration
It shows that luminescence persistence can be purposefully tuned through rationally using PO matrixes with suitable Tn* characteristics. Based on different duration times of room temperature phosphorescence and afterglows from neat and 1% TMB doped PO melt films, long-time-range and multi-level information display can be realized (Fig. 4). The ground powders of 28DBTDPO and PO:1% TMB materials filled the grooves on glass substrates, which were prepared by HF etching of patterns in paraffin coating (Fig. 4a). After annealing, melt films formed Chinese characters of Heilongjiang University and English abbreviation of organic long-persistent luminescence (OLPL), respectively. When turning off UV lamp, all the characters gave out bright afterglows, and then disappeared successively, in accord with luminescence lifetimes of the melt films. The patterning of the melt films can be more conveniently realized by mosaic (Fig. 4b). Through spreading material powders on square quartz substrates and then annealing, five different mosaic bricks respectively covered with three neat and two 1% TMB doped PO melt films were prepared to form five leaves of a pentapetalous pattern. Due to their different emission durations, after excitation removal, emissions from the leaves sequentially faded away.
a Preparation process of anti-counterfeiting patterns containing Chinese calligraphy “黑龙江大学” of Heilongjiang University (above) and acronym “OLPL” of organic long-persistent luminescence (below), and emission variations of melt nDBTxPO:1% TMB patterns with afterglow under 365 nm excitation and at selected stages at 100 ms and 1−8 min after UV turn-off. b Image mosaicing based on melt films prepared with nDBTxPO and nDBTxPO:1% TMB. Pentapetalous pattern is prepared with five kinds of quartz plates covered by melt films with different emission durations, leading to sequentially fading away. c Time-encoded security application of a mosaicing panel based on nDBTxPO, 46DBTDPO:1% TMB and 28DBTDPO:1% TMB, whose afterglow is vanished in turn, corresponding to a number sequence within 5 min. 2DBTSPO melt film is incorporated as a check code, whose position indicates the correct digital number of password from the sequence. d Erasable optical memory based on 28DBTDPO melt film. Using dichloromethane solution of TMB as ink, a regular “P” letter with yellow afterglow can be written on the film, which can be further erased with ethanol. The recovered film can be further used for writing another bold “P” letter.
This “mosaic” design also makes information encryption with these melt films feasible (Fig. 4c). As a proof, four neat and two 1% TMB doped PO melt films were used to prepare six different kinds of mosaic bricks. A password panel was form by nine bricks, corresponding to numbers of 1 to 9, four of which were covered with 28DBTDPO:1% TMB; while, the rest five were based on other melt films. After excitation removal, afterglows of the bricks disappeared in turn, corresponding to time-correlated digital sequences, which were 4, 49, 497 and 4971 in this example. 2DBTSPO-based brick as the check code indicated the correct sequential number. Thus, the final correct password was 497. The different afterglow durations of neat and light-doped films establish an effective approach to realize reversible information storage (Fig. 4d). A 28DBTDPO melt film covered a quartz substrate was adopted as a draw panel. A “P” character was written on it by using a dichloromethane solution of TMB (1 molar per liter) as ink, whose yellow afterglow can be clearly distinguished from the substrate at 1 s after excitation removal. It is ascribed to the surface TMB doping. Therefore, the panel can be cleaned by ethanol washing to erase the original pattern, and then rewritten to display the second “P” character. Obviously, owing to their large difference in ethanol solubility, the writing-erasing process is repeatable.
Discussion
We use a series of dibenzothiophene-based PO acceptor matrixes with different Tn* characteristics to figure out the optimal mechanism and determinants for realizing ultra-long afterglows in donor–acceptor systems. It shows that the 28DBTDPO matrix has the high-population and high-lying Tn* state in comparison to nCT* states of its 1% TMB doped film, giving rise to a step-by-step energy transfer for 28DBTDPO:1% TMB involving in both of aggregation-stabilized Tn* and low-radiative-rate nCT*: S1 → T1 → Tn* → nCT*. This energy relay from room temperature phosphorescence matrix to local dipole results in long-persistent afterglow with duration over dozens of minutes with the naked eyes. In the case of PO matrixes with non-stabilized triplet states or Tn* energitically lower than nCT*, direct energy transfer from the T1 states of PO molecules to nCT* states becomes predominant, rendering the markedly shorter afterglow duration for the doped films. This understanding provides a convenient and effective approach to modulate long-persistent luminescent properties of donor–acceptor doping systems, therefore paves a way of purposeful system design for long-time-range and multi-level time-resolution optical applications in bio-imaging, medicine, information storage, anti-counterfeiting, and so on.
Methods
Photophysical measurement
Absorption spectra were measured using a SHIMADZU UV-3150 spectrophotometer. Photoluminescence spectra were measured with an Edinburgh FPLS 1000 fluorescence spectrophotometer. The time decay spectra was measured using Time-Correlated Single Photon Counting (TCSPC) method with a picosecond hydrogen lamp for 100 ps−10 μs and a microsecond pulsed Xenon light source for 1 μs−10 s lifetime measurement, the synchronization photomultiplier for signal collection and the Multi-Channel Scaling Mode of the PCS900 fast counter PC plug-in card for data processing. The long-persistent emission decay were measured using the kinetic mode of the FLS1000, and the curves were acquired after 90 s of excitation with a Xe lamp at 365 nm. Lifetime values were simulated by single exponential fitting function in Fluoracle software. For anti-counterfeiting and encryption applications, a circular ultraviolet flashlight (GET-104) was used as the excitation source with a power of 3 W and the peak wavelength at 365 nm, whose spot diameter is about 2 cm at the distance of 10 cm above the samples.
General procedure for film preparation
(i) Platemaking: glass substrate is sealed with paraffin, and etched with hydrofluoric acid according to the customized pattern (Fig. 4a).
(ii) Materials and Film Preparation: all phosphine oxides were prepared through a typical two-step reaction of lithiation and phosphorylation (Supplementary Fig. 1), whose chemical structures were confirmed by nuclear magnetic resonance spectra (Supplementary Figs. 2−5). The neat or doped powders were put uniformly on the specific positions of glass substrates, heated to the melting points, and cooled down to form melt films (Fig. 4a). Through melting process, the regular aggregation in the films can be improved, which enhances the stability of Tn* state, and simultaneously facilitates charge diffusion, therefore elongating the charge migration time for hole-electron recombination on nCT* states (Supplementary Fig. 6). The purities of the films were confirmed by high-performance liquid chromatography (HPLC, Supplementary Figs. 7 and 8). Quartz plates were covered with the powders directly for forming melt films on the top of the “mosaic” bricks (Fig. 4b−d). A dilute dichloromethane solution of TMB (1 M) and ethanol were used as ink and erasing liquid, respectively.
Data availability
The authors declare that the data generated in this study are provided in Supplementary Information. Data are available from the corresponding author on request. Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Center, under deposition numbers 2455009 (2DBTSPO) [https://doi.org/10.5517/ccdc.csd.cc2ndmvk], 2455375 (28DBTDPO) [https://doi.org/10.5517/ccdc.csd.cc2nf0ns], 842938 (4DBTSPO) [https://doi.org/10.5517/ccx94kw] and 946475 (46DBTDPO) [https://doi.org/10.5517/cc10rwg3], respectively. Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/.
References
Li, Y., Gecevicius, M. & Qiu, J. Long persistent phosphors—from fundamentals to applications. Chem. Soc. Rev. 45, 2090–2136 (2016).
Wei, J. et al. Full-color persistent room temperature phosphorescent elastomers with robust optical properties. Nat. Commun. 14, 4839 (2023).
Zhang, Y. et al. Ultraviolet irradiation-responsive dynamic ultralong organic phosphorescence in polymeric systems. Nat. Commun. 12, 2297 (2021).
Li, Z., Yue, Q., Zhang, H. & Zhao, Y. Methodologies for constructing multi-color room temperature phosphorescent systems. Mater. Today 78, 209–230 (2024).
Li, G. et al. Organic supramolecular zippers with ultralong organic phosphorescence by a dexter energy transfer mechanism. Angew. Chem. Int. Ed. 61, e202113425 (2022).
Su, Y. et al. Ultralong room temperature phosphorescence from amorphous organic materials toward confidential information encryption and decryption. Sci. Adv. 4, eaas9732 (2018).
Zhou, Q., Yang, C. & Zhao, Y. Dynamic organic room-temperature phosphorescent systems. Chem 9, 2446–2480 (2023).
Zhen, X. et al. Ultralong phosphorescence of water-soluble organic nanoparticles for in vivo afterglow imaging. Adv. Mater. 29, 1606665 (2017).
Liu, Y. et al. Ratiometric afterglow luminescent nanoplatform enables reliable quantification and molecular imaging. Nat. Commun. 13, 2216 (2022).
Ou, X. et al. High-resolution X-ray luminescence extension imaging. Nature 590, 410–415 (2021).
Pei, P. et al. X-ray-activated persistent luminescence nanomaterials for NIR-II imaging. Nat. Nanotech. 16, 1011–1018 (2021).
Qiu, W. et al. Afterglow OLEDs incorporating bright closely stacked molecular dimers with ultra-long thermally activated delayed fluorescence. Matter 6, 1231–1248 (2023).
Tan, S., Jinnai, K., Kabe, R. & Adachi, C. Long-persistent luminescence from an exciplex-based organic light-emitting diode. Adv. Mater. 33, 2008844 (2021).
Xu, S. et al. Design of highly efficient deep-blue organic afterglow through guest sensitization and matrices rigidification. Nat. Commun. 11, 4802 (2020).
Lu, G. et al. Delayed room temperature phosphorescence enabled by phosphines. Nat. Commun. 15, 3705 (2024).
Zhao, W., He, Z. & Tang, B. Z. Room-temperature phosphorescence from organic aggregates. Nat. Rev. Mater. 5, 869–885 (2020).
Gu, L. et al. Colour-tunable ultra-long organic phosphorescence of a single-component molecular crystal. Nat. Photon. 13, 406–411 (2019).
Gong, Y. et al. Achieving persistent room temperature phosphorescence and remarkable mechanochromism from pure organic luminogens. Adv. Mater. 27, 6195–6201 (2015).
Song, X. et al. Phosphine-manipulated p–π and π–π synergy enables efficient ultralong organic room-temperature phosphorescence. Angew. Chem. Int. Ed. 62, e202300980 (2023).
Yang, J. et al. The influence of the molecular packing on the room temperature phosphorescence of purely organic luminogens. Nat. Commun. 9, 840 (2018).
He, Z. et al. White light emission from a single organic molecule with dual phosphorescence at room temperature. Nat. Commun. 8, 416 (2017).
Zhang, Y. et al. π–π interaction-induced organic long-wavelength room-temperature phosphorescence for in vivo atherosclerotic plaque imaging. Angew. Chem. Int. Ed. 63, e202313890 (2024).
Li, D. et al. Completely aqueous processable stimulus responsive organic room temperature phosphorescence materials with tunable afterglow color. Nat. Commun. 13, 347 (2022).
Guo, J., Yang, C. & Zhao, Y. Long-lived organic room-temperature phosphorescence from amorphous polymer systems. Acc. Chem. Res. 55, 1160–1170 (2022).
Yang, Z. et al. Boosting the quantum efficiency of ultralong organic phosphorescence up to 52 % via intramolecular halogen bonding. Angew. Chem. Int. Ed. 59, 17451–17455 (2020).
Bian, L. et al. Simultaneously enhancing efficiency and lifetime of ultralong organic phosphorescence materials by molecular self-assembly. J. Am. Chem. Soc. 140, 10734–10739 (2018).
Alam, P. et al. Organic3 long-persistent luminescence from a single-component aggregate. J. Am. Chem. Soc. 144, 3050–3062 (2022).
Ma, L., Sun, S., Ding, B., Ma, X. & Tian, H. Highly efficient room-temperature phosphorescence based on single-benzene structure molecules and photoactivated luminescence with afterglow. Adv. Funct. Mater. 31, 2010659 (2021).
Pan, Z., Lu, Y.-Y. & Liu, F. Sunlight-activated long-persistent luminescence in the near-infrared from Cr3+-doped zinc gallogermanates. Nat. Mater. 11, 58–63 (2012).
Cai, S. et al. Enhancing ultralong organic phosphorescence by effective π-type halogen bonding. Adv. Funct. Mater. 28, 1705045 (2018).
Hirata, S. et al. Efficient persistent room temperature phosphorescence in organic amorphous materials under ambient conditions. Adv. Funct. Mater. 23, 3386–3397 (2013).
Liang, X., Zheng, Y.-X. & Zuo, J.-L. Two-Photon Ionization Induced Stable White Organic Long Persistent Luminescence. Angew. Chem. Int. Ed. 60, 16984–16988 (2021).
Man, Z. et al. Host surface-induced excitation wavelength-dependent organic afterglow. J. Am. Chem. Soc. 145, 13392–13399 (2023).
Yang, Y. et al. Efficient and color-tunable dual-mode afterglow from large-area and flexible polymer-based transparent films for anti-counterfeiting and information encryption. Angew. Chem. Int. Ed. 61, e202201820 (2022).
Li, W. et al. Organic long-persistent luminescence from a thermally activated delayed fluorescence compound. Adv. Mater. 32, 2003911 (2020).
Xu, S., Chen, R., Zheng, C. & Huang, W. Excited state modulation for organic afterglow: materials and applications. Adv. Mater. 28, 9920–9940 (2016).
Jin, P. et al. Stepwise charge/energy transfer in MR-TADF molecule-doped exciplex for ultralong persistent luminescence activated with visible light. Adv. Mater. 36, 2400158 (2024).
Wang, Z. H. et al. Color-tunable polymeric long-persistent luminescence based on polyphosphazenes. Adv. Mater. 32, 1907355 (2020).
Song, J., Ma, L., Sun, S., Tian, H. & Ma, X. Reversible multilevel stimuli-responsiveness and multicolor room-temperature phosphorescence emission based on a single-component system. Angew. Chem. Int. Ed. 61, e202206157 (2022).
Kabe, R. & Adachi, C. Organic long persistent luminescence. Nature 550, 384–387 (2017).
Jinnai, K., Kabe, R., Lin, Z. & Adachi, C. Organic long-persistent luminescence stimulated by visible light in p-type systems based on organic photoredox catalyst dopants. Nat. Mater. 21, 338–344 (2022).
Lin, Z., Kabe, R., Wang, K. & Adachi, C. Influence of energy gap between charge-transfer and locally excited states on organic long persistence luminescence. Nat. Commun. 11, 191 (2020).
Nishimura, N., Lin, Z., Jinnai, K., Kabe, R. & Adachi, C. Many exciplex systems exhibit organic long-persistent luminescence. Adv. Funct. Mater. 30, 2000795 (2020).
Wang, Y. et al. High Performance of simple organic phosphorescence host–guest materials and their application in time-resolved bioimaging. Adv. Mater. 33, 2007811 (2021).
Zhang, X., Chong, K. C., Xie, Z. & Liu, B. Color-tunable dual-mode organic afterglow for white-light emission and information encryption based on carbazole doping. Angew. Chem. Int. Ed. 62, e202310335 (2023).
Jinnai, K., Kabe, R. & Adachi, C. Wide-range tuning and enhancement of organic long-persistent luminescence using emitter dopants. Adv. Mater. 30, 1800365 (2018).
Han, C. et al. Ladder-like energy-relaying exciplex enables 100% internal quantum efficiency of white TADF-based diodes in a single emissive layer. Nat. Commun. 12, 3640 (2021).
Yin, C.-R. et al. Hindrance-functionalized π-stacked polymer host materials of the cardo-type carbazole–fluorene hybrid for solution-processable blue electrophosphorescent devices. Macromolecules 44, 4589–4595 (2011).
Park, S. K. et al. Tailor-made highly luminescent and ambipolar transporting organic mixed stacked charge-transfer crystals: an isometric donor–acceptor approach. J. Am. Chem. Soc. 135, 4757–4764 (2013).
Han, C. et al. Short-axis substitution approach selectively optimizes electrical properties of dibenzothiophene-based phosphine oxide hosts. J. Am. Chem. Soc. 134, 19179–19188 (2012).
Turro, N. J. Energy transfer processes. Pure Appl. Chem. 49, 405–429 (1977).
Lai, R. et al. Shallow distance-dependent triplet energy migration mediated by endothermic charge-transfer. Nat. Commun. 12, 1532 (2021).
Acknowledgments
The authors thank the support by the National Natural Science Foundation of China (22375057, W2412073, 52273173, 22325502, 92461304, 22305073).
Author information
Authors and Affiliations
Contributions
H.X. conceived the projects. C.Z., Z.W., Y.Z., and C.D. performed the experiments. H.X., C.Z., G.L., Y.M., J.Z., and C.H. analyzed the data. H.X. and C.Z. wrote the paper. All authors commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Dmytro Volyniuk, who co-reviewed with Matas Gužauskas; and the other, anonymous, reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, C., Lu, G., Wang, Z. et al. Ultralong afterglow enabled by energy relay from room-temperature phosphorescent matrixes to local dipoles. Nat Commun 16, 5935 (2025). https://doi.org/10.1038/s41467-025-60863-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-60863-6
This article is cited by
-
Ionic bonding stimulated organic afterglow fluids for smart adhesives
Science China Chemistry (2026)