Abstract
β-Ag2Se holds a great promise for wearable thermoelectric generators due to its good mechanical properties and biocompatibility. However, optimizing its power factor through alterations in the Ag/Se stoichiometric ratio or incorporating secondary phases has reached the limitation. This work adopts a liquid-like sintering strategy, namely employing the migration and precipitation of silver ions from metallic silver to the silver selenide during the spark plasma sintering process to construct significantly sized and coherent Ag-rich nanoprecipitates within flexible Ag/β-Ag2Se composite films, thereby achieving an unprecedented power factor exceeding 4000 μWm−1K−2 at 303 K. The Ag-rich nanoprecipitates play a crucial role in elevating the carrier concentration, enhancing the density-of-states effective mass, and mitigating carrier scattering caused by phase interfaces and acoustic phonons. The generator containing 5 pieces of optimal films shows an normalized output power density of 10.89 μWcm−1K−2 under a temperature difference of 26.0 K. This study strongly suggests that the creation of Ag-rich nanoprecipitates is an effective avenue for improving the electrical performance of β-Ag2Se-based thermoelectric materials.
Similar content being viewed by others
Introduction
The rapid progress in flexible electronics has catalyzed an exigent need for power sources adaptable to the inherent flexibility of these systems1. Flexible thermoelectric films, capable of conforming to the skin and continuously converting temperature difference between the human body and the ambient environment into electrical energy for flexible electronics, are viewed as a highly promising flexible self-powered energy source2,3. Optimizing the power factor (PF) of the flexible thermoelectric film is the key to improving its output power density to meet the energy supply needs of as many electronic products as possible4.
Due to its better mechanical properties and lower toxicity compared to the traditional BiTeSe alloys, N-type silver selenide (β-Ag2Se at room temperature) has attracted sharply increasing attention in the field of flexible thermoelectric films5,6. However, although its Hall carrier mobility (μH) reaching up to 2000 cm2V−1s−1 at room temperature, the values of density-of-states effective mass (md) for carriers in the stoichiometric β-Ag2Se are far smaller than that of BiTeSe alloys7,8,9. Furthermore, owing to the doping limits of most elements (including Ag and Se) in β-Ag2Se being less than 0.1 mole%, the intrinsic small carrier concentration (nH) of stoichiometric β-Ag2Se can hardly be effectively elevated through doping or altering the Ag/Se ratio since the 1960s10,11,12,13. Thus, the state-of-art values of PF for stoichiometric β-Ag2Se, in both bulk and film forms, typically remain below 2700 μWm−1K−2 or even 2000 μWm−1K−2 at around room temperature for decades, which is markedly inferior to the exceeding 4000 μWm−1K−2 obtained by BiTeSe alloys4,14,15,16. On the other hand, despite incorporating metals or alloys as the second phase has been proven to enable the promotion of nH and enhancement of md in β-Ag2Se-based flexible films17,18, the values of PF for these films are still limited by the excessively high nH and additional carrier scattering at phase boundaries19,20,21,22.
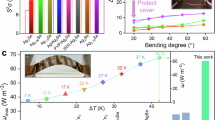
Very recently, we revealed that a previously overlooked microstructure, Ag-rich nanoprecipitates with a size of a few nanometers can be found in nominally stoichiometric β-Ag2Se sintered by spark plasma sintering (SPS)23. However, possibly because of their limited size and content, the nanoprecipitates did not exert a substantial influence on the thermoelectric properties of β-Ag2Se. In this work, addressing the mobility of Ag ions and its low solubility in β-Ag2Se, a liquid-like SPS approach is proposed, wherein Ag ions migrate from metallic silver to silver selenide during the sintering process, subsequently bonding partially with selenium atoms and precipitating out to form Ag-rich nanoprecipitates with sizes reaching several tens of nanometers (marked with red arrows in Fig. 1d) in flexible Ag/β-Ag2Se film (see “Methods”, Supplementary Fig. 1 and Fig. 1a–d). As shown in Fig. 1e, the boundary between the Ag-rich nanoprecipitate and β-Ag2Se matrix is completely coherent due to their identical crystal structure of orthorhombic Ag2Se. The electrons transferred from the Ag-rich nanoprecipitates and metallic Ag to the β-Ag2Se matrix collectively pushed up the nH of composite films (see Supplementary Fig. 2), and the Ag-rich nanoprecipitates also play a significant role in enhancing the md and diminishing carrier scattering caused by phase interfaces and acoustic phonons. Thus, as depicted in Fig. 1f, despite a high nH of 22.16 × 1018 cm−3, this optimal film (named as Ag2.3Se-SPS200 °C/5 min) maintains a moderate μH of 855.4 cm2V−1s−1. Meanwhile, because of the appropriate nH and md, the optimal film retains a Seebeck coefficient of −116.3 μVK−1, showing only a marginal decline compared to that of pristine β-Ag2Se (see Fig. 1g). As a result, via this novel strategy that can transform metallic Ag into the Ag-rich nanoprecipitates, an unprecedented PF exceeding 4000 μWm−1K−2 has been achieved in the optimal film, setting a new benchmark for the electrical performance of β-Ag2Se based thermoelectric materials (see Fig. 1h). In addition, regardless of whether the length of the film is taken into account, the normalized output power density (Pmd) of the thermoelectric generator formed by 5 pieces of optimal films (1 cm in length) significantly surpassing that of other β-Ag2Se based film-type thermoelectric generators (see Fig. 1i).
a Schematic diagram of the fabrication of nylon-supported flexible composite films. b–d EDS maps of Ag element (b) and Se element (c), and bright-field TEM image (d) for Ag2.3Se-SPS200 °C/5 min film (typical Ag-rich nanoprecipitates denoted with red arrows). e High-resolution transmission electron microscopy (HRTEM) image showing the coherent boundary (marked with red dotted circle) between the Ag-rich nanoprecipitate boundary and the β-Ag2Se matrix. All the lattice spaces marked with the white lines correspond to the planes of orthorhombic β-Ag2Se. f–h Comparison of electrical properties between the optimal film in this work and other β-Ag2Se based bulks and films. (1 ~ 10) Different β-Ag2Se based bulks and films include 1: Ag/Ag2Se film20, 2: Ag/Ag2Se film21, 3: Ag2Se film32, 4: Ag2Se bulk7, 5: Ag2.05Se film22, 6: Ag2Se1.01 bulk33, 7: Ag2Se1.06 bulk13, 8: (Ag0.998Cu0.02)2Se bulk14, 9: Ag2Se bulk34, 10: Ag1.8Se film15. i Comparison of normalized output power density between the thermoelectric generator in this work and other β-Ag2Se-based film-type thermoelectric generators15,17,18,19,21,26,32,35,36.
Results and discussion
By systematically investigating the composition and microstructure of composite films fabricated from composite powders of varying compositions and subjected to different sintering processes, the potential formation mechanism of Ag-rich nanoprecipitates can be deduced. According to the molar ratio of AgNO3 to Se powder in the solvothermal reaction, and the temperature and duration of SPS, the obtained thermoelectric films are named as Ag2.0Se-SPS200 °C/5 min, Ag2.1Se-SPS200 °C/5 min, Ag2.3Se-SPS140 °C/1 min, Ag2.3Se-SPS200 °C/5 min (the optimal film), Ag2.3Se-SPS200 °C/30 min, and Ag2.5Se-SPS200 °C/5 min.
Since the Ag-rich nanoprecipitates with notable size in this work have never been reported in the stoichiometric β-Ag2Se, it is speculated that the presence of sufficient metallic Ag in the composite powder before sintering is a prerequisite for the formation of Ag-rich nanoprecipitates. High-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) images in Supplementary Fig. 3 confirm this inference. In HAADF-STEM image of Ag2.1Se-SPS200 °C/5 min, hardly any notable Ag-rich nanoprecipitates can be observed, while HAADF-STEM images of Ag2.3Se-SPS200 °C/5 min and Ag2.5Se-SPS200 °C/5 min distinctly reveal the Ag-rich nanoprecipitates.
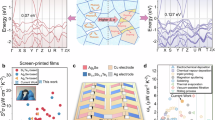
Composition and microstructure of films fabricated using Ag2.3Se powder have been investigated to illustrate the formation process of Ag-rich nanoprecipitates. For β-Ag2Se particles in the Ag2.3Se powder before sintering, the atomic fraction of Ag element is determined to be 67.69% by energy dispersive X-ray spectroscopy (EDS) maps (see Supplementary Fig. 4). As depicted in the dark-field TEM image presented in Fig. 2a, for Ag2.3Se-SPS140 °C/1 min film, numerous circular “bright” domains (typically marked with white arrows), primarily within 10 nm in size can be recognized by the strain field contrast. The result of EDS line scanning across a “bright” domain (see Supplementary Fig. 5) in Fig. 2d reveals that the average atomic fraction of Ag element in the “bright” domain (75.08%) is higher than that in the surrounding β-Ag2Se matrix (71.02%), namely, these domains are Ag-rich. Notably, the β-Ag2Se matrix also exhibits an elevated average atomic fraction of Ag element than that of the β-Ag2Se particles in the Ag2.3Se powder. For the composite films sintered at 200 °C for 5 min and 30 min, as shown in Fig. 2b, c, a large number of island-like nanoprecipitates spanning sizes from tens to approximately 100 nanometers can be observed. Determined by EDS maps of the typical nanoprecipitates (marked with red dashed lines) and β-Ag2Se matrix (marked with yellow dashed lines), for Ag2.3Se-SPS200 °C/5 min film, the average atomic fractions of Ag element in the nanoprecipitates and β-Ag2Se matrix are 69.42% and 67.15%, respectively. For clear comparison, the average atomic fractions of Ag element in the nanoprecipitates and β-Ag2Se matrix for all samples in this work are listed in Supplementary Table 1. As to Ag2.3Se-SPS200 °C/30 min film, the corresponding values increase to 73.49% and 68.81%, respectively. HRTEM images in Fig. 2e, f demonstrate that the boundaries between the Ag-rich nanoprecipitates and the surrounding matrix (indicated by red dashed lines) are coherent, and the Ag-rich nanoprecipitates exhibit pronounced Moiré fringes with varying spacings. The fast Fourier transform (FFT) patterns inserted in Fig. 2a, e, f, while displaying clear secondary diffraction features, can all be indexed to the orthorhombic phase of Ag2Se.
a Dark-field HRTEM image of Ag2.3Se-SPS140 °C/1 min film inserted with a FFT pattern. b, c HAADF-STEM images Ag2.3Se-SPS200 °C/5 min film and Ag2.3Se-SPS200 °C/30 min film. d Atomic fraction of Ag element revealed by EDS elemental line scan across a “bright” domain. e, f Dark-field HRTEM image of Ag2.3Se-SPS200 °C/5 min film and Ag2.3Se-SPS200 °C/30 min film inserted with the FFT patterns.
Rietveld refined X-ray diffraction patterns have been employed to estimate the mass fraction of metallic Ag in the Ag2.3Se composite powder and the sintered films (see Supplementary Fig. 6 and Supplementary Table 2). All the diffraction peaks can be indexed to orthorhombic β-Ag2Se (JCPDF card number: 01-071-2410) and cubic metallic Ag (JCPDF card number: 03-065-2871), and Ag2.3Se composite powder contains approximately 8 wt% of metallic Ag. Nevertheless, as to the film sintered at 140 °C (slightly higher than the phase transition temperature (133 °C) of β-Ag2Se) for only 1 min, the Ag and Ag2Se particles quickly fused with each other to form bulks (see Supplementary Fig. 7), and the mass fraction of metallic Ag drops to about 4%. Further increasing the sintering temperature to 200 °C and extending the sintering durations to 5 and 30 min facilitated complete sintering of the composite films, which led to a reduction in the thickness of composite films and a shrinkage in size of the metallic Ag regions within the composite films (see Supplementary Figs. 8 and 9), but the mass fractions of metallic Ag in these films still remain around 4%. Hence, it is inferred that the excess Ag atoms in the Ag-rich nanoprecipitates stem from the metallic Ag, and the transfer of Ag atoms from metallic Ag to Ag2Se is completed during the phase transition.
Drawing from the above discussion, the presumed process underlying the formation of the Ag-rich nanoprecipitates during sintering is illustrated in Fig. 3a. In comparison to the Ag2.3Se powder, the mass fraction of metallic Ag in the Ag2.3Se-SPS140 °C/1 min film is diminished. Conversely, the atomic fraction of Ag element within the β-Ag2Se matrix is promoted, accompanied by the formation of Ag-rich domains. Therefore, during the phase transition of β-Ag2Se, part of Ag atoms migrates from the metallic Ag into the interstitial sites of Ag2Se, forming the nanosized Ag-rich domains in the β-Ag2Se matrix. This state is inherently unstable due to the extremely low solubility of silver atoms in Ag2Se. Consequently, the excess Ag atoms at interstitial sites tend to precipitate out, and the Ag-rich domains represent the early nucleation stage of the nanoprecipitates. As the sintering temperature and duration rise to 200 °C and 5 min, respectively, Ag atoms continuously precipitate out from the interstitial sites in Ag2Se and bond with Se atoms to form Ag-rich nanoprecipitates. This supposition accounts for the observed reduction in the atomic fractions of Ag elements within the β-Ag2Se matrix and Ag-rich nanoprecipitates when compared to the film sintered at 140 °C for 1 min. Meanwhile, the feasibility of this supposition has also been confirmed through theoretical models (see Fig. 3b). The model of perfect (11-2) plane of β-Ag2Se containing 72 atoms (Ag48Se24) was firstly created, then, 7 Ag atoms were inserted into the interstitial sites of this model (marked with the red dashed oval). After the relaxation of the model inserted with excess Ag atoms (Ag55Se24), the inserted Ag atoms moved from the interstitial sites to the surface of β-Ag2Se to form the Ag-rich nanoprecipitate (marked with the green dashed oval), which is coherent with the (11-2) plane of β-Ag2Se. Based on the models, the average values of Bader charge for Ag atoms in metallic Ag, β-Ag2Se, and Ag-rich nanoprecipitate are calculated to be 11.001, 10.772, and 10.844, respectively, suggesting that the Ag atoms in Ag-rich nanoprecipitate are partially bonded with the Se atoms. On the other hand, in Fig. 3a, with the extension of sintering duration to 30 min, the mass fraction of metallic Ag in the composite film remains at around 4 wt%, implying that the observed elevation in the atomic fraction of Ag element within the β-Ag2Se matrix and Ag-nanoprecipitates is not attributable to metallic Ag, but rather to Se loss resulting from volatilization. The volatilization of Se elements leads to a sharp increase in nH (50.4 × 1018 cm−3), causing a dramatic decline in the Seebeck coefficient and PF of the Ag2.3Se-SPS200 °C/30 min to −58.33 μVK−1 and 2338.96 μWm−1K−2, respectively. Thus, precise control of the SPS process plays a decisive role in regulating thermoelectric performance of Ag2Se-based materials.
a Schematic diagram revealing the possible microscopic reactions in the composite film at different stages of SPS process. b Theoretical models to verify the possible formation mechanism of Ag-rich nanoprecipitates. Blue balls represent Ag atoms and yellow balls represent Se atoms.
To elucidate the factors contributing to the record-high PF observed in Ag2.3Se-SPS200 °C/5 min film, the carrier transport characteristics of this film have been thoroughly analyzed and compared with that of other β-Ag2Se-based materials. Firstly, the higher nH observed in Ag2.3Se-SPS200 °C/5 min film compared to single-phase β-Ag2Se-based materials at or near stoichiometry is reasonable. As per the theoretical models outlined in Fig. 3b, the Ag-rich nanoprecipitates can be fundamentally conceptualized as β-Ag2Se with Se vacancies, naturally exhibiting a higher concentration of electron compared to stoichiometric β-Ag2Se. Moreover, the computed charge density differences suggest that both the Ag-rich nanoprecipitate and metallic Ag are capable of electron donation to β-Ag2Se, hence, enhancing the abundance of Ag-rich nanoprecipitates and metallic Ag within the composite films in this work can bolster the nH. However, an excessive presence of Ag-rich nanoprecipitates alongside metallic Ag would lead to a remarkably heightened nH and a notable decline in the PF (see Supplementary Note 1 and Supplementary Tables 1–5). Secondly, in order to figure out why Ag2.3Se-SPS200 °C/5 min maintains a high nH while preserving a μH close to 1000 cm2V−1s−1, the values of activation energy (Eb) characterizing the barrier height of grain boundaries and phase interfaces in the β-Ag2Se-based materials were estimated using the equation \({\mu }_{H}=\frac{{el}}{\sqrt{8{k}_{B}T\pi {m}_{d}}}\exp ({-E}_{b}/{k}_{B}T)\) (see Fig. 4a), where e is the elementary charge, l is the average grain size (set as 100 μm for the calculation), kB is the Boltzmann constant and T is the absolute temperature (set as 300 K for the calculation)24. Clearly, due to the existence of interfacial barrier in the composites, the composites show larger values of Eb than the single-phase β-Ag2Se-based materials at or near stoichiometry. Of particular interest is the observation that the Ag2.3Se-SPS200 °C/5 min film in this work exhibits Eb akin to those observed in single-phase β-Ag2Se-based materials, unlike other composites. This suggests that the coherent grain boundaries between the Ag-rich nanoprecipitate and β-Ag2Se have negligible effects on carrier scattering, and the carriers are also not significantly scattered by the interfaces between metallic Ag and β-Ag2Se, because of the relatively small mass fraction (about 4%) of metallic Ag in the film. Thus, to circumvent excessive interfacial scattering, precise regulation of the quantity of secondary phases in β-Ag2Se-based materials is crucial. Further, the values of deformation potential, indicative of the interaction strength between acoustic phonons and carriers, were evaluated for the Ag2.3Se-SPS200 °C/5 min film in this work and other Ag2Se-based materials with similar scattering mechanism using the single parabolic band (SPB) model. Based on the values of md and Edef, the nH dependent theoretical μH and PF were calculated and plotted (see “Methods” and Fig. 4b, c). As shown in Fig. 4b, compared to single-phase β-Ag2Se-based materials, Ag2.3Se-SPS200 °C/5 min film despite having the maximum value of md (0.510 m0), shows only two-third to half of the Edef for single-phase β-Ag2Se-based materials, indicating minimal scattering of carriers by acoustic phonons in it. Thus, it can be summarized that even with the high nH and large md, Ag2.3Se-SPS200 °C/5 min film maintains a moderate μH for two reasons, namely the mitigation of carriers scattering at phase interfaces, and the reduction in scattering of carriers by acoustic phonons. The plots in Fig. 4c reveal that irrespective of adjustments to nH, the maximum theoretical PF at 300 K of the reported single-phase β-Ag2Se-based materials is limited at approximately 3000 μWm−1K−2, which is predominantly attributed to the relatively large Edef. On the contrast, leveraging the diminished Edef, the PF of Ag2.3Se-SPS200 °C/5 min film holds promise to exceed 4500 μWm−1K−2 if further optimization of nH is achieved. In fact, some of the representative Ag2.3Se-SPS200 °C/5 min films and bulks in this work indeed have realized such a large PF (see Supplementary Fig. 10), and the overall relative standard deviation of the PF values for these samples is calculated to be 5.13% (see Supplementary Table 6), indicating good repeatability in performance. To illuminate the reasons behind the larger md and smaller Edef of the Ag2.3Se-SPS200 °C/5 min film compared to single-phase β-Ag2Se-based materials, based on the theoretical models (see Supplementary Fig. 11), the electronic band structure and theoretical Edef for stoichiometric β-Ag2Se and Ag-rich nanoprecipitate have been obtained (see “Methods”, Fig. 4d, e, f). As revealed by Fig. 4d, e, the bottom of conduction band for β-Ag2Se appears relatively sharp, and the corresponding band-edge effective mass (mb) is 0.265 m0, while the Ag-rich nanoprecipitate displays a markedly flattened bottom of conduction band, with the mb of 0.414 m0. Additionally, by calculating the strain-dependent variations of the conduction band minimum (CBM) in Fig. 4f, the values of theoretic Edef for stoichiometric β-Ag2Se and Ag-rich nanoprecipitate are 11.49 eV and 5.15 eV, respectively. Therefore, the formation of Ag-rich nanoprecipitates in the composite films in this work leads to the larger md and smaller Edef.
a Barrier height of grain boundaries and phase interfaces for the optimal film in this work and other β-Ag2Se based bulks and films. (1–10) Different β-Ag2Se based bulks and films include 1: (1–10) Different β-Ag2Se based bulks and films include 1: Ag2Se1.01 bulk33, 2: Ag2Se bulk7, 3: (Ag0.998 Cu0.02)2Se bulk14, 4: Ag2Se bulk34, 5: Ag2Se1.06 bulk13, 6: Ag2Se film32, 7: Ag1.8Se film15, 8: Ag/Ag2Se film21, 9: Ag2.05Se film22, 10: Ag/Ag2Se film20. b, c Calculated nH dependent theoretical μH and PF plots for the optimal film in this work and other β-Ag2Se based bulks and films with similar scattering mechanism. The values of md and Edef include 1: md = 0.211 me, Edef = 7.605 eV, 2: md = 0.186 me, Edef = 10.733 eV, 3: md = 0.221 me, Edef = 9.120 eV, 4: md = 0.219 me, Edef = 9.393 eV, 5: md = 0.298 me, Edef = 6.653 eV, 6: md = 0.276 me, Edef = 7.541 eV, 7: md = 0.248 me, Edef = 9.071 eV. d–f Calculated electronic band structures and strain-dependent variations in the conduction band minimum of β-Ag2Se matrix and Ag-rich nanoprecipitate.
For the potential application of flexible thermoelectric films, besides the PF, the length of thermoelectric film should also be optimized to maximize the output power density. In this work, the length of a single Ag2.3Se-SPS200 °C/5 min film with width of 5 mm and thickness of 30 μm is optimized by numerical simulation. The model for simulation is shown in Fig. 5a, and the parameters for simulation are listed in Supplementary Table 7. Additionally, the contact resistance (Rc) between the electrodes (silver paste) and Ag2.3Se-SPS200 °C/5 min film is measured to be 3.92 mΩ (See Supplementary Fig. 12). The ambient temperature (Ta) is 298.15 K, and the heat exchange coefficient between the film and air is set as 4 W m−2 K−1. The length of the silver electrodes on both ends of the film is 2 mm. The thermal conductivity (κ) of Ag2.3Se-SPS200 °C/5 min film (1.77 Wm−1K−1) is calculated as κ = ρDCp, where ρ is the density (8198.4 kgm−3), D is the thermal diffusivity and Cp is the specific heat capacity (see Supplementary Fig. 13). Therefore, ZT of this film at 303 K is calculated to be 0.7, which is comparable to the commonly reported ZT of Ag2Se based bulk materials. The electrical and thermal properties of nylon substrate and silver electrodes are obtained from the software (Comsol Multiphysics 5.6) for simulation and the thickness of nylon substrate is 35 μm. The temperature gradient and electrical potential field have been simulated for the film with temperature of hot side (Th) set as 308.15 K and 318.15 K, respectively. Figure 5b shows the simulated results for the film with Th set as 308.15 K. The temperature of the cold side (Tc) decreases from 306.45 K to 298.23 K as the length of the film increases from 1 mm to 18 mm, and the open circuit voltage (Uoc) increases from 0.19 mV to 1.14 mV. The maximum output power (Pm) is expressed as\(\,{P}_{{\rm{m}}}=\,\frac{{U}_{{\rm{oc}}}^{2}}{4{\times }({R}_{{\rm{f}}}+{2\times} ({R}_{{\rm{c}}}+{R}_{{\rm{e}}}))}\), where Rf and Re the theoretical resistance of the Ag2.3Se-SPS200 °C/5 min film and silver paste. The simulated highest Pm of 1.40 μW is realized in the film with a length of 6 mm, and as displayed in Fig. 5c, the value of Uoc for this film is 8.86 × 10−4 V. Likewise, in Fig. 5d, with the increasing length of film, the Tc drops from 314.76 K to 298.31 K for the Th of 318.15 K. In addition, the simulated Uoc climbs from 0.39 mV to 2.28 mV and a peak value for Pm of 5.58 μW is realized in the film. In Fig. 5e, the corresponding value of Uoc is 1.77 × 10−3 V. Moreover, the theoretical values of energy conversion efficiency (ηT) of the single-film generator with Th of 308.15 K and 318.15 K are 0.36% and 0.71%, respectively (see Supplementary Note 2 and Supplementary Fig. 14).
a Model of single Ag2.3Se-SPS200 °C/5 min film for numerical simulation. b–e Simulated results and potential distribution for the film with Th set as 308.15 K (b and c) and 318.15 K (d and e). f, g Experimentally measured output voltage (f) and power (g) of the thermoelectric generator composed of 5 pieces of optimal films.
It is noteworthy that, although the natural convection condition at the cold end represents the most probable practical scenario for film-type wearable thermoelectric generators, currently, the output performance of flexible film-type thermoelectric generators are predominantly measured under conditions of fixed values of ΔT. In this work, in order to facilitate and ensure the utmost accuracy in the preparation and measurement of the film-type generator, as well as to enable an objective comparison with the output performance reported in other works, 5 pieces of Ag2.3Se-SPS200 °C/5 min film with a length of 1 cm were connected in series to form a thermoelectric generator (see Supplementary Fig. 15), and the output performance of this generator was then tested with different values of ΔT. The output voltage and power of this generator are shown in Fig. 5f, g, respectively. Both the open-circuit voltage and maximum output power increase with the rise in the ΔT across the generator. For the ΔT of 26.0 K and 57.0 K, the measured Pm attains 55.23 μW and 204.78 μW, respectively. In addition, depending on whether the length of the film (L) is taken into account, there are two methods for the calculation of normalized output power density, namely \({P}_{{\rm{md}}}=\,\frac{{P}_{{\rm{m}}}}{{A}_{{\rm{TEG}}}{\Delta T}^{2}}\)18,25, or \({P}_{{\rm{md}}}=\,\frac{{P}_{{\rm{m}}}L}{{A}_{{\rm{TEG}}}{\Delta T}^{2}}\)26,27, where ATEG is the cross-sectional area of the thermoelectric generator. According to these two expressions, the highest Pmd this work is calculated of 10.89 (μWcm−2K−2 or μWcm−1K−2) with ΔT = 26.0 K.
The mechanical properties of the Ag2.3Se-SPS200 °C/5 min materials have also been investigated. After 800 bending cycles (the radius for bending is 2.5 mm), an increase in resistivity of the bent film (ρb) is observed, amounting to approximately a 10% elevation compared to its pristine resistivity (ρ0), and then keep almost constant during the subsequent bending cycles. On the other hand, throughout the entire flexibility test, the variation in the Seebeck coefficient of the bent film (Sb) remained within ±4% of its original value (S0), suggesting that the bending cycles can hardly affect the Seebeck coefficient of the film (see Supplementary Fig. 16a). Moreover, the bending strength of Ag2.3Se-SPS200 °C/5min-based bulk is measured to be 128.69 MPa (see Supplementary Fig. 16b), which is very close to that of β-Ag2Se reported by Pei’s group and significantly exceeds that of (Ag0.998Cu0.02)2Se composite and Ag2Se/CNTs composite6,14,28.
Via a liquid-like SPS process, Ag-rich nanoprecipitates with notable sizes and proper composition have been created in the flexible Ag/β-Ag2Se composite films. The formation of the Ag-rich nanoprecipitates is speculated to arise from the migration of Ag atoms from metallic Ag to the interstitial sites of the Ag2Se lattice, where they bond with Se atoms and precipitate out. Ag-rich nanoprecipitates can positively influence the thermoelectric performance of composite films by not only increasing the nH and improving the md, but also reducing carrier scattering from phonons and phase boundaries. An unprecedented and stable power factor exceeding 4000 μWm−1K−2 at room temperature has been realized in the optimal film. Moreover, the thermoelectric generator composed of 5 pieces of optimal films achieved an normalized output power density up to 10.89 μWcm−1K−2. This work has presented an innovative research direction for further enhancing the thermoelectric performance of β-Ag2Se-based materials.
Methods
Materials
Silver nitrate (AgNO3, ≥99.8%) was purchased from XiLong Scientific Co., Ltd. Selenium powder (Se, ≥99.9%) and ethylenediamine (C2H8N2, 99%) were purchased from Aladdin Chemical Co., Ltd. Ethylene glycol and anhydrous ethanol were bought from Sinopharm Chemical Reagent Co., Ltd. The glass-fiber sheets were purchased from Yuyan (Shanghai) Chemical Co. and the nylon membrane was produced by Yibo (Zhejiang) Filtration Equipment Co., Ltd. All these materials were used without further purification.
Synthesis of Ag/β-Ag2Se powders
At room temperature, a certain amount of Se powder and AgNO3 were dissolved in 12.5 mL and 5 mL of ethylenediamine, respectively. Then these two solutions were mixed and maintained at 180 °C for 5 h in a sealed 25 mL Teflon-lined stainless-steel autoclave. After the autoclave was naturally cooled to room temperature, the obtained dark grey powder was collected by centrifugation and washed with ethanol and deionized water for three times. The total amount of the AgNO3 and Se powder for synthesis of all samples is 0.4187 g and molar ratio of AgNO3 and Se varies from 2.0 to 2.5 to adjust the mass fraction of metallic Ag in the resulted composite powders.
Fabrication of composite films and bulks
The resulted powder was uniformly dispersed in 10 mL ethylene glycol and 1.2 mL of the dispersion was drop-casted on a piece of glass-fiber sheet with size of 5 mm × 20 mm. After the glass-fiber sheet coated with composite powder was dried in vacuum at 80 °C for 10 h, it was sandwiched between 2 pieces of nylon membrane and sintered at 30 MPa. During the sintering process, the composite powder was insulated from the pulse current of SPS equipment by the nylon membrane, avoiding the migration of Ag ions caused by the pulse current. The powder was transformed into the nylon-supported dense composite film during the sintering process, and the debris of glass-fiber sheet could be easily removed with a brush. To demonstrate that the nylon substrate has no influence on the thermoelectric properties of the films, some composite bulk materials were also prepared by sintered the composite powder in the conditions same with the fabrication of films, but without using the glass-fiber membrane and nylon film.
Material characterizations and property measurements
The phase and composition of composite powders and films were analyzed by X-ray diffractometer using Cu Ka radiation (Rigaku, SmartLab). The mass fractions of Ag element and Se element in samples were acquired by inductively coupled plasma optical emission spectrometry (ICP-OES, Agilent 720). The morphology and thickness of composite powders and films were revealed via field emission scanning electron microscopy (Hitachi S-4800). The microstructures were observed by transmission electron microscopy (FEI TalosF200X), with an Oxford energy dispersive X-ray spectrometer (EDS) to analyze the elemental compositions and distributions of the samples. The simultaneous measurement of the Seebeck coefficient and electrical conductivity at room temperature were carried out in an Ulvac-Riko ZEM-3 system. The measurement was conducted in argon atmosphere with temperature gradients of 20, 30, and 40 °C. The carrier mobility and carrier concentration were collected using a Hall effect system (Lake Shore Cryotronics 8404). A laser flash diffusivity apparatus (Netzsch LFA 467) and a differential scanning calorimeter (Netzsch 404 F3) were applied to determine the thermal diffusivity and specific heat capacity, respectively. The density was measured via the Archimedes’ method. Since the measured thermal diffusivity of composite film is dramatically lower than the reasonable value, pellets were sintered using Ag/β-Ag2Se powder in the conditions same with the fabrication of films for the measurement of thermal diffusivity and density. Each pellet is circular, with a thickness of 0.5 mm and a diameter of 12.7 mm. The sound velocity was measured via a sound velocimeter (JSR, DPR300). The hot and cold side temperatures of single composite film were measured employing a multiplex temperature meter (DCUU, DC5508U), with the cold side of film suspending in the air. The spot welding was used to connect Pt-Rh wires with the films, and the output properties of the thermoelectric generator composed of 5 pieces of films were collected by a multimeter (Agilent 34970A) and a constant current source (Agilent B2901A)29. The bending test was conducted via a hand-cranked tensile testing machine (AIGU, ZP-1000).
First-principles calculations
This work utilized periodic slab models for all DFT calculations, conducted via the Vienna ab initio simulation package, including DFT-D3 empirical van der Waals corrections. The HSE exchange-correlation functional was applied within the generalized gradient approximation GGA30. Electron-ion interactions were described using the projector-augmented wave method, employing a plane-wave basis set with a cutoff energy of 500 eV. A 3 × 4 × 3 Monkhorst-Pack k-point mesh for perfect bulk β-Ag2Se with lattice constants a = 9.17 Å, b = 7.04 Å, c = 7.58 Å. The coverage criteria for energies and forces were set to 10−5 eV and 0.02 eV/Å. The first order Methfessel–Paxton method with a smearing width of 0.05 eV was used. The model for the Ag-rich nanoprecipitate was constructed by removing one Se atom from the structure of Ag16Se8. For the (11-2) crystal plane of β-Ag2Se and its derived models, a 3 × 2 × 1 Monkhorst-Pack k-point mesh was applied. To calculate the deformation potential, the positions of the CBM for the structures with strain magnitudes ranging from −1% to 1% were calculated in increments of 0.5%. The energy difference between the CBM and the core energy level yields ECBM. The slope of the variation of ECBM with strain represents the deformation potential of the material.
Numerical simulation
The software for numerical simulation is Comsol Multiphysics 5.6. The surrounding air around the film was set to be natural convection, and the thermal resistance between the composite film and silver electrodes was ignored.
Single parabolic band (SPB) model for carrier transport characteristics
For the SPB model, the transport coefficients are expressed below:
Density-of-states effective mass (md):
Deformation potential (Edef):
Seebeck coefficient (S):
Hall Carrier concentration (nH):
Hall Carrier mobility (μH):
Electrical conductivity (σ):
Power factor (PF):
In the above equations, the integral F [n, η] is defined by: \(F[n,\eta ]={\int }_{0}^{\infty }[\frac{{x}^{n}}{1+{\rm{Exp}}(x-\eta )}]\). h, me kB, e, and r are the Planck constant, the mass of free electron, the Boltzmann constant, the charge the electron, the scattering factor (r = −0.5 for acoustic phonon scattering). vl is the longitudinal speed of sound of β-Ag2Se (measured to be around 2540 m/s in this work), d is·the density of sample (the theoretical density of 8.24 g cm−3 was applied for calculation).
Data availability
The data generated in this study have been deposited in figshare database with the DOI of https://doi.org/10.6084/m9.figshare.2925686031.
References
Xu, C., Song, Y., Han, M. & Zhang, H. Portable and wearable self-powered systems based on emerging energy harvesting technology. Microsyst. Nanoeng. 7, 25 (2021).
Shi, X. et al. Room-temperature ductile inorganic semiconductor. Nat. Mater. 17, 421–426 (2018).
Li, X., Cai, K., Gao, M., Du, Y. & Shen, S. Recent advances in flexible thermoelectric films and devices. Nano Energy 89, 106309 (2021).
Cha, J., Zhou, C., Cho, S.-P., Park, S. H. & Chung, I. Ultrahigh power factor and electron mobility in n-type Bi2Te3–x%Cu stabilized under excess Te condition. ACS Appl. Mater. Interfaces 11, 30999–31008 (2019).
Wu, H., Shi, X.-L., Duan, J., Liu, Q. & Chen, Z.-G. Advances in Ag2Se-based thermoelectrics from materials to applications. Energy Environ. Sci. 16, 1870–1906 (2023).
Liu, M., Zhang, X., Zhang, S. & Pei, Y. Ag2Se as a tougher alternative to n-type Bi2Te3 thermoelectrics. Nat. Commun. 15, 6580 (2024).
Jin, M. et al. Investigation on low-temperature thermoelectric properties of Ag2Se polycrystal fabricated by using zone-melting method. J. Phys. Chem. Lett. 12, 8246–8255 (2021).
Zhu, B. et al. Realizing record high performance in n-type Bi2Te3-based thermoelectric materials. Energy Environ. Sci. 13, 2106–2114 (2020).
Perez-Taborda, J. A., Caballero-Calero, O., Vera-Londono, L., Briones, F. & Martin-Gonzalez, M. High thermoelectric zT in n-type silver selenide films at room temperature. Adv. Energy Mater. 8, 1702024 (2018).
Wei, T.-R., Qiu, P., Zhao, K., Shi, X. & Chen, L. Ag2Q-Based (Q = S, Se, Te) silver chalcogenide thermoelectric materials. Adv. Mater. 35, 2110236 (2023).
Conn, J. B. & Taylor, R. C. Thermoelectric and crystallographic properties of Ag2Se. J. Electrochem. Soc. 107, 977 (1960).
Simon, R., Bourke, R. C. & Lougher, E. H. Preparation and thermoelectric properties of β-Ag2Se. Adv. Energy Convers. 3, 481–505 (1963).
Mi, W. et al. Thermoelectric transport of Se-rich Ag2Se in normal phases and phase transitions. Appl. Phys. Lett. 104, 133903 (2014).
Chen, J. et al. Simultaneously enhanced strength and plasticity of Ag2Se-based thermoelectric materials endowed by nano-twinned CuAgSe secondary phase. Acta Mater. 220, 117335 (2021).
Hou, S. et al. High performance wearable thermoelectric generators using Ag2Se films with large carrier mobility. Nano Energy 87, 106223 (2021).
Lu, T. et al. Synergistically enhanced thermoelectric and mechanical performance of Bi2Te3 via industrial scalable hot extrusion method for cooling and power generation applications. Mater. Today Phys. 32, 101035 (2023).
Li, Y. et al. Exceptionally high power factor Ag2Se/Se/polypyrrole composite films for flexible thermoelectric generators. Adv. Funct. Mater. 32, 2106902 (2022).
Li, X. et al. Exceptional power factor of flexible Ag/Ag2Se thermoelectric composite films. Chem. Eng. J. 434, 134739 (2022).
Lu, Y. et al. Ultrahigh power factor and flexible silver selenide-based composite film for thermoelectric devices. Energy Environ. Sci. 13, 1240–1249 (2020).
Gao, Q. et al. High power factor Ag/Ag2Se composite films for flexible thermoelectric generators. ACS Appl. Mater. Interfaces 13, 14327–14333 (2021).
Gao, J. et al. Thermoelectric flexible silver selenide films: compositional and length optimization. iScience 23, 100753 (2020).
Zheng, Z.-H. et al. Achieving ultrahigh power factor in n-type Ag2Se thin films by carrier engineering. Mater. Today Energy 24, 100933 (2022).
Huang, S. et al. Thermoelectric Ag2Se: imperfection, homogeneity, and reproducibility. ACS Appl. Mater. Interfaces 13, 60192–60199 (2021).
Zhao, D., Ning, J., Li, S. & Zuo, M. Synthesis and thermoelectric properties of C60/Cu2GeSe3 composites. J. Nanomater. 2016, 5923975 (2016).
Yang, Q. et al. Flexible thermoelectrics based on ductile semiconductors. Science 377, 854–858 (2022).
Liu, Y. et al. High performance Ag2Se films by a one-pot method for a flexible thermoelectric generator. J. Mater. Chem. A 10, 25644–25651 (2022).
Lu, Y. et al. Staggered-layer-boosted flexible Bi2Te3 films with high thermoelectric performance. Nat. Nanotechnol. 18, 1881–1288 (2023).
Wang, H. et al. Constructing n-type Ag2Se/CNTs composites toward synergistically enhanced thermoelectric and mechanical performance. Acta Mater. 223, 117502 (2022).
Naumov, P., Barkalov, O., Mirhosseini, H., Felser, C. & Medvedev, S. A. Atomic and electronic structures evolution of the narrow band gap semiconductor Ag2Se under high pressure. J. Phys.: Condens. Matter 28, 385801 (2016).
Liang, J. et al. Modulation of the morphotropic phase boundary for high-performance ductile thermoelectric materials. Nat. Commun. 14, 8442 (2023).
Gao, J. et al. Coherent Ag-rich nanoprecipitates/β-Ag2Se flexible film with unprecedented thermoelectric performance by liquid-like sintering. Figshare https://doi.org/10.6084/m9.figshare.29256860 (2025).
Lei, Y. et al. Microstructurally tailored thin β-Ag2Se films toward commercial flexible thermoelectrics. Adv. Mater. 34, 2104786 (2022).
Jood, P., Chetty, R. & Ohta, M. Structural stability enables high thermoelectric performance in room temperature Ag2Se. J. Mater. Chem. A 8, 13024–13037 (2020).
Chen, J. et al. Hierarchical structures advance thermoelectric properties of porous n-type β-Ag2Se. ACS Appl. Mater. Interfaces 12, 51523–51529 (2020).
Liu, Y. et al. Nanoengineering approach toward high power factor Ag2Se/Se composite films for flexible thermoelectric generators. ACS Appl. Mater. Interfaces 15, 36587–36593 (2023).
Wang, Z., Liu, Y., Li, J., Huang, C. & Cai, K. High-performance Ag2Se film by a template method for flexible thermoelectric generator. Mater. Today Phys. 36, 101147 (2023).
Acknowledgements
This work was supported by the Natural Science Foundation of Guangxi, China (Grant No. 2024GXNSFAA010366), the Guangxi Key Laboratory of Information Materials (Guangxi Science and Technology Program AD25069070), the National Natural Science Foundation of China (Grant No. U21A2054), the National Natural Science Foundation of China (Grant No. 51961011, 52273285, 52232010). This work was supported by Sinoma Institute of Materials Research (Guang Zhou) Co., Ltd (SIMR) for assisting SEM&TEM characterizations.
Author information
Authors and Affiliations
Contributions
J.G. conceived the idea. J.L., J.G., L.J., J.C.G., T.Y., J.L.C., X.W., and E.N. prepared the samples and performed the characterizations and tests. J.G. and J.L. post-processed the data. J.G. performed the DFT calculations and numerical simulation. J.G., L.M., X.S., and P.Q. wrote and edited the manuscript. P.Q., S.Z., X.Y., and T.S. contributed to the fabrication and measurement of the thermoelectric generator. L.M. and X.S. supervised this research and promoted the manuscript as corresponding authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Priyanshu Banerjee, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gao, J., Li, J., Miao, L. et al. Coherent Ag-rich nanoprecipitates/β-Ag2Se flexible film with unprecedented thermoelectric performance by liquid-like sintering. Nat Commun 16, 6010 (2025). https://doi.org/10.1038/s41467-025-61079-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-61079-4