Abstract
Torpor, an adaptive hypometabolic state in response to fasting, is characterized by pronounced reductions in body temperature, heart rate, and thermogenesis. However, how the brain orchestrates these physiological changes to induce torpor and the relationships among them remain elusive. Inhibiting catecholaminergic (CA) neurons in the ventrolateral medulla (VLM) significantly impairs torpor in mice, while their activation reduces body temperature, heart rate, energy expenditure, physical activity, and thermogenesis. Importantly, the heart rate decline precedes body temperature reduction, resembling patterns observed in natural torpid animals. Moreover, a likely causal relationship exists between heart rate reduction and body temperature decline. VLM-CA neurons may regulate heart rate and thermogenesis through projections to the dorsal motor vagal nucleus and medial preoptic area, respectively. Additionally, these neurons are conserved in Daurian ground squirrels and become active before hibernation, indicating their potential role in hibernation. Here, we find that VLM-CA neurons play important roles in fasting-induced torpor.
Similar content being viewed by others
Introduction
Energy intake and expenditure are integral components of the energy balance equation, and their interaction is continuously evolving. When energy intake decreases, basal energy expenditure tends to decline in response. This adjustment in energy expenditure represents a conserved adaptive strategy utilized by various species, including humans, to cope with limited food resources. In the wild, many mammals adopt seasonal hibernation or periodic torpor to survive periods of food scarcity1,2,3. This intriguing natural phenomenon has sparked significant research interest because of its remarkable mechanisms in the regulation of energy homeostasis and potential for clinical and artificial human hibernation4,5. However, despite the long recognition of torpor and hibernation, the mechanisms underlying these states remain poorly understood.
Torpor is a hypometabolism state characterized by profound reductions in body temperature, heart rate, metabolic and respiratory rates, and thermogenesis6,7—an orchestrated cascade of physiological changes believed to be regulated by specialized neuronal circuits. Recent advancements have identified the preoptic area of the hypothalamus (POA), a vital brain region involved in regulating body temperature8,9,10,11,12, as contributing to torpor in mice. Activation of neurons within the POA expressing pituitary adenylate cyclase-activating polypeptide (Adcyap1), pyroglutamylated RFamide peptide (QRFP), and estrogen receptor alpha (ERα) induces torpor, while the disruption of synaptic transmission or the ablation of these neurons can variably inhibit torpor induced by fasting13,14,15. Despite progress in understanding the neural mechanisms of torpor, several key conceptual questions remain unanswered.
Firstly, torpor is not a passive decrease in body temperature, heart rate, and metabolism; instead, these physiological changes are actively-controlled processes with complex interrelationships7,16,17. The precise mechanisms by which the brain orchestrates these intricate physiological changes to induce torpor, and the causal relationships among these changes, remain unclear. Secondly, the autonomic nervous system is believed to play crucial roles in controlling the physiological changes during torpor18. However, how the thermoregulatory POA nucleus receives hunger signals and interfaces with the autonomic nervous system to coordinate various physiological changes is yet unknown. Finally, although torpor and hibernation share similarities as states of reduced metabolism, they also exhibit distinct differences. Our understanding of the similarities and disparities in the neural mechanisms governing these two states is limited.
Emerging evidence indicates that the brainstem plays significant roles in regulating energy intake and energy metabolism19,20,21. As a key node connecting the autonomic nervous system, it also plays pivotal roles in regulating both cardiovascular functions and thermogenesis22,23,24. Yet, it remains unknown whether and which brainstem neurons regulate torpor. Catecholaminergic (CA) neurons in the ventrolateral medulla (VLM) of the brainstem have been implicated in controlling autonomic outflows23,25,26,27, cardiovascular functions27,28,29, body temperature28, respiration27, feeding behavior30,31, and inflammatory responses25,32,33,34. Moreover, VLM-CA neurons are vital for mediating counterregulatory responses to glucose deficit35,36. Given the significance of these physiological changes in torpor, we hypothesized that VLM-CA neurons may be involved in the regulation of torpor.
Here, we explore the role of VLM-CA neurons in fasting-induced torpor and elucidate how these neurons coordinate various physiological changes during torpor.
Results
VLM-CA neurons are required for fasting-induced torpor
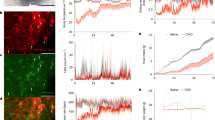
To study the neuronal mechanisms underlying the induction of torpor, we utilized a fasting-induced torpor model in mice. The animals underwent 24 h of food deprivation starting from dark onset while having unlimited access to water. A telemetry temperature sensor was surgically implanted in the abdominal cavity to continuously monitor the body temperature and physical activity of mice during fasting, under an ambient temperature range of 22–24 °C (Fig. 1a). Consistent with previous studies37, the core body temperature started to decline after ~7 h into fasting, eventually reaching a torpid state (defined as having a core temperature below 31 °C)15,38 (Fig. 1b). The lowest core body temperature was observed ~12 h post-food deprivation, after which it gradually recovered to baseline levels by the 24-h mark, even in the absence of refeeding. These observations suggest that fasting-induced torpor is an actively controlled physiological process.
a Experimental strategy for telemetric recording of core body temperature and locomotor activity. b The core body temperature (Tcore) and locomotor activity of a non-fasting mouse (upper panels) and a fasting mouse (bottom panels). c–e Fasting induced Fos (green) expression in tyrosine hydroxylase positive (TH+) VLM-CA neurons. c Representative images from mice at different time points during fasting along the rostral-caudal axis. Arrows indicate Fos positive (Fos+) CA neurons. Scale bars: 100 µm. d The percentages of Fos+ neurons among VLM-CA neurons at different time points. e The percentages of Fos+ neurons among VLM-CA neurons along the rostral-caudal axis. Control (n = 3 mice), 6 h (n = 5 mice), 9 h (n = 4 mice) and 15 h (n = 4 mice) in (d, e). f–h Chemogenetic inhibition of VLM-CA neurons. f The viral strategy. g Representative images of n = 12 mice showing the expression of hM4Di (red) in VLM-CA neurons. h Representative electrophysiological trace showing CNO (50 µM)-induced inhibition of hM4Di+ VLM-CA neurons. Changes in the core body temperature (Tcore) (i) and locomotor activity (j) of DbhVLM-hM4Di and DbhVLM-mCherry mice during 24-h food deprivation. k Quantitative analysis of the Tcore. Changes in the average Tcore before and after CNO injections (left), variance (middle), and lowest value (right) of the Tcore during fasting. l Total torpor time spent with Tcore ≤ 31 °C and number of torpor bouts with Tcore ≤ 31 °C during fasting. m Quantitative physical activity during the periods of 10–18 h and 10–24 h post food deprivation. The dashed lines in (i, j) denote the timing of the CNO injections. The gray bar indicates the dark phase (20:00 to 8:00). Two-tailed unpaired Student’s t-test in (d, k, l, m). Two-tailed paired Student’s t-test in (e). Two-way ANOVA analysis in (i). ns, not significant (P > 0.05). Data are presented as the means ± SEM. Source data are provided as a Source Data file. Figure 1a was created in BioRender. Zhan, C. (2025) https://BioRender.com/s14s282. Figure 1f was created in BioRender. Zhan, C. (2025) https://BioRender.com/fi02cic.
To determine the potential involvement of VLM-CA neurons in fasting-induced torpor, we used Fos immunostaining to assess their neuronal activity at 6, 9, and 15 h post-food deprivation. We observed a significant increase in the proportion of Fos+ VLM-CA neurons in the rostral and middle regions of the VLM in the fasting group compared to the non-fasting control group at different time points following food deprivation (Fig. 1c, d). Notably, over 95% of the Fos+ VLM-CA neurons activated by fasting were situated in the rostral and middle regions of the VLM, while less than 5% were found in the caudal VLM (Fig. 1e). These findings suggest that VLM-CA neurons, particularly those in the rostral and middle regions, become activated during fasting. Furthermore, the highest proportion of Fos+ VLM-CA neurons was observed at the 6-h time point post-food deprivation, just prior to the onset of core body temperature decline. These results suggest that VLM-CA neurons are likely involved in the induction of torpor.
To determine the role of VLM-CA neuronal activity in fasting-induced torpor, we used a chemogenetic inhibitory approach to silence VLM-CA neurons. Following bilateral injection of adeno-associated virus (AAV) conditionally expressing an inhibitory hM4Di (AAV-DIO-hM4Di-mCherry) into the VLM of dopamine-β-hydroxylase (Dbh)-Cre mice, hM4Di-mCherry was selectively expressed in VLM-CA neurons (referred to as DbhVLM-hM4Di mice) (Fig. 1f, g). Dbh is an enzyme that catalyzes the conversion of dopamine into noradrenaline. AAV expressing only mCherry (AAV-DIO-mCherry) was injected as a control (referred to as DbhVLM-mCherry mice). In-vitro electrophysiological recordings showed that Clozapine N-oxide (CNO) administration transiently silenced hM4Di/mCherry+ VLM-CA neurons (Fig. 1h), confirming the suitability of this chemogenetic method for the inhibition of VLM-CA neurons.
Remarkably, chemogenetic inhibition of VLM-CA neurons in mice with similar body weights significantly disrupted fasting-induced torpor. Following CNO injections, ~30% (five among fifteen) DbhVLM-hM4Di mice did not reach the torpor state, while all DbhVLM-mCherry control mice entered torpor (Supplementary Fig. 1). The reductions in core body temperature and physical activity, core body temperature variability, and duration of torpor were all significantly impaired by the inhibition of VLM-CA neurons (Fig. 1i–m). Furthermore, VLM-CA neuronal inhibition also delayed the entrance of fasting-induced torpor (i.e., 11.43 ± 0.74 h in DbhVLM-hM4Di mice vs 10.10 ± 0.31 h in control mice) (Fig. 1i). In contrast, the chemogenetic inhibition of VLM-CA neurons had no impact on the body temperature and activity in non-fasted mice (Supplementary Fig. 2a, b). Together, these results suggest that VLM-CA neuron activity is required for fasting-induced torpor.
VLM-CA neurons are sufficient to induce a torpor-like state
To directly examine whether VLM-CA neurons can induce torpor, we used a chemogenetic excitatory approach to activate VLM-CA neurons. Following bilateral injection of an AAV conditionally expressing excitatory hM3Dq and mCherry (AAV-DIO-hM3Dq-mCherry) into the VLM of Dbh-Cre mice (referred to as DbhVLM-hM3Dq mice), hM3Dq-mCherry was selectively expressed in VLM-CA neurons (Fig. 2a, b). In-vitro electrophysiological recordings showed that CNO administration transiently activated hM3Dq/mCherry+ VLM-CA neurons (Fig. 2c), confirming the suitability of this chemogenetic method for the activation of VLM-CA neurons.
a–c Chemogenetic activation of VLM-CA neurons. a The viral strategy. b Representative images of n = 5 mice showing the expression of hM3Dq (red) in TH+ VLM-CA neurons. Scale bars: 100 µm. c Representative electrophysiological trace showing CNO (10 µM)-induced activation of hM3Dq+ VLM-CA neurons. d–h Changes in the core body temperature (Tcore) (d, P < 0.0001) and physical activity (f, P = 0.0057) of DbhVLM-hM3Dq and DbhVLM-mCherry mice following CNO or saline injections. e Quantitative analysis of changes in the average Tcore (left), variance (middle) and lowest (right) Tcore after CNO or saline injections. g Quantitative physical activities. h Total torpor duration and number of torpor bouts. The dashed lines in (d, f) denote the timing of CNO or saline injection. Two-way ANOVA analysis in (d, f). Two-tailed paired Student’s t-test in (e, g, h). Data are presented as mean ± SEM. ns, not significant P > 0.05. Source data are provided as a Source Data file. Figure 2a was created in BioRender. Zhan, C. (2025) https://BioRender.com/fi02cic.
After intraperitoneal (IP) injection of CNO at an ambient temperature of 22–24 °C, non-fasted DbhVLM-hM3Dq mice exhibited a rapid reduction in body temperature and eventually entered a torpor-like state (Fig. 2d, e, h). The core body temperature of one tested mouse even decreased to below 25 °C, and lasted for more than 20 h (Supplementary Fig. 2c). Additionally, VLM-CA neuron activation significantly decreased physical activity (Fig. 2f, g, and Supplementary Fig. 2c). CNO injection into control mice did not alter body temperature or physical activity, suggesting that the observed effects were specifically due to the activation of VLM-CA neurons and not due to any off-target effects of CNO or its derivatives. Together, these results suggest that VLM-CA neurons are sufficient to profoundly decrease core body temperature and activity in non-fasted mice, mimicking the phenotypes observed in natural torpid mice.
VLM-CA neuron activation lowers metabolic rate and thermogenesis
In addition to lowering body temperature, torpor is characterized by a decreased metabolic rate and thermogenesis. These changes are active processes that occur before the decrease in body temperature, rather than simply being a result of it7,16,17. To determine how VLM-CA neurons induce torpor, we examined their impacts on metabolic rate and thermogenesis. We found that chemogenetic activation of VLM-CA neurons by CNO injections led to a significant reduction in O2 consumption, CO2 production, and calculated energy expenditure, with no effect observed in control mice (Fig. 3a–c). These results suggest that the activation of VLM-CA neurons significantly decreases the metabolic rate.
Oxygen consumption (a), carbon dioxide production (b), and calculated energy expenditure (c) in DbhVLM-hM3Dq and DbhVLM-mCherry mice following CNO or saline injections. Quantitative data for the 6 h following CNO or saline injections are shown in the bottom panels. d–f iBAT thermogenesis of DbhVLM-hM3Dq mice following CNO or saline injections. d Experimental setup. e Representative infrared images showing surface body temperature at 0, 30, 60, and 120 min after CNO or saline injections. f Quantitative analysis of the iBAT surface temperature. g–i iBAT sympathectomy. g Representative image of iBAT sympathectomy, involving cutting off five sympathetic nerve bundles. Noradrenaline (NA) concentration (h) and representative images (i, n = 3 mice) of sympathetic nerves (red, TH) in iBAT after sympathectomy or sham surgery. Scale bar: 500 µm. Relative mRNA expression of Ucp1 gene in iBAT (j), Tcore (k), iBAT temperature (l), and locomotion activity (m) of DbhVLM-hM3Dq mice before and after sympathectomy following CNO and saline injections. Two-way ANOVA analysis in (a, b, c, f, k, l, m). Two-tailed unpaired Student’s t-test and paired Student’s t-test in the bottom panels of (a, b, c, h), as appropriate. One-way ANOVA in (j). Data are presented as mean ± SEM. ns not significant P > 0.05. Source data are provided as a Source Data file. Figure 3d was created in BioRender. Zhan, C. (2025) https://BioRender.com/fi02cic.
Brown adipose tissue (BAT) is essential for thermogenesis, a process crucial for maintaining body temperature39. Infrared thermal imaging revealed significant decreases in interscapular BAT (iBAT) temperature and overall body surface temperature following the administration of CNO, but not saline, into DbhVLM-hM3Dq mice (Fig. 3d–f and Supplementary Movies 1 and 2). Sympathetic nervous system activation is a known driver of BAT-induced thermogenesis39. To investigate whether the observed reduction in iBAT thermogenesis was due to the altered sympathetic outflow into the iBAT, we performed bilateral sympathectomy of the iBAT (Fig. 3g). The success of sympathectomy was evidenced by the decreased noradrenaline content and the diminished sympathetic nerves (Fig. 3h, i). Sympathectomy reduced the basal levels of uncoupling protein 1 (Ucp1), a key thermogenic gene, in the iBAT and abolished the cold-induced increase in Ucp1 levels (Fig. 3j, and Supplementary Fig. 3a). Although the activation of VLM-CA neurons significantly decreased the Ucp1 expression in sympathetic-intact mice, it had no such effect in sympathectomized mice (Fig. 3j). However, the activation of VLM-CA neurons still significantly decreased the core body temperature, iBAT temperature, and locomotion activity in sympathectomized mice to a similar extent as in sympathetic-intact mice (Fig. 3k–m). Therefore, even though VLM-CA neurons cannot downregulate Ucp1 levels in sympathectomized mice, they are still capable of reducing the core body temperature and iBAT temperature, suggesting the existence of other peripheral effectors.
Sympathectomy had no impact on heart rate and core body temperature in mice with ad libitum access to food and water (Supplementary Fig. 3b). Moreover, sympathectomized mice were still capable of entering and recovering from fasting-induced torpor (Supplementary Fig. 3c, d). When compared with sympathetic-intact mice, they exhibited similar durations and bouts of fasting-induced torpor (Supplementary Fig. 3e). This suggests that the sympathetic innervation of iBAT may not be the determining factor for the induction and regulation of fasting-induced torpor. Instead, the data suggest the involvement of alternative regulatory mechanisms or compensatory thermogenic contributions from other tissues (e.g., other BAT depots or white adipose tissue). Although sympathectomy did not impede the VLM-CA neuronal activation-induced reduction in the core body temperature, sympathectomized mice displayed significantly lower average core body temperatures and higher body temperature variability during fasting compared to sympathetic-intact mice (Supplementary Fig. 3f). This indicates that the absence of sympathetic innervation into iBAT has a notable impact on the body’s ability to maintain a stable core body temperature under fasting conditions.
Activation of VLM-CA neurons increases heat dissipation
Under normal circumstances, endothermic animals typically have counteractive mechanisms to enhance heat production when their body temperature drops. In contrast, during torpor and hibernation, a decrease in body temperature does not lead to cold defense responses7. Instead, animals actively engage their thermoregulatory systems to lower body temperature, facilitating entry into torpor and hibernation. In rodents, the tail plays a crucial role in the thermoregulatory effector system40, aiding in heat dissipation through vasodilation or reducing it via vasoconstriction. Therefore, we examined the impact of VLM-CA neuronal activation on the thermoregulatory function of the tail.
To minimize the interference of stress responses caused by animal handling and CNO injection on vascular contraction and relaxation, we used stabilized step function opsin (SSFO) as an optogenetic activator to stimulate VLM-CA neurons. SSFOs induce rapid and long-lasting neuronal activation (~30 min) in response to brief light stimulation41, enabling quick manipulation of VLM-CA neurons and for an extended period while minimizing heat-related injury. Following bilateral injection of an AAV conditionally encoding SSFO (AAV-DIO-SSFO-eYFP) into the VLM of Dbh-Cre mice (referred to as DbhVLM-SSFO mice), SSFO-YFP was selectively expressed in VLM-CA neurons (Fig. 4a, b). AAV expressing only GFP (AAV-DIO-GFP) was injected as a control. In-vitro electrophysiological recordings showed that brief light stimulation produced rapid and long-lasting activation in VLM-CA neurons expressing SSFO (Fig. 4c).
a–c Optogenetic activation of VLM-CA neurons. a The viral strategy. b Representative images showing the expression of SSFO (green) in TH+ VLM-CA neurons. Scale bar: 100 µm. c Representative electrophysiological trace illustrating the activation of SSFO+ VLM-CA neurons by a blue light pulse (2 s, blue bar). d–g The body temperature of DbhVLM-SSFO and DbhVLM-GFP control mice before and after light stimulation (indicated by blue bars, 20 Hz, 30 s). Representative infrared images (d), quantitative analysis of tail (e) and iBAT (f) surface temperature, and core body temperature (g, P = 0.0033). h–j Thermal gradient assay schematic with heatmaps depicting the cumulative positions of a representative DbhVLM-hM3Dq mouse on a thermal gradient for 50 min following saline or CNO injection. The average gradient position (0 = 50 °C, 1 = 15 °C) (i, P = 0.0386) and median gradient position (j, P = 0.0003, each point represents one mice) during the 50-min period. The dashed lines in (i) denote the timing of CNO or saline injection. Two-way ANOVA analysis in (e, f, g, i). Two-tailed paired Student’s t-test in (j). Data are presented as mean ± SEM. *P < 0.05; ***P < 0.001. Source data are provided as a Source Data file. Figure 4a was in created in BioRender. Zhan, C. (2025) https://BioRender.com/k4e71f1. Figure 4h includes a mouse illustration created in BioRender. Zhan, C. (2025) https://BioRender.com/k4e71f1.
Following optogenetic activation of VLM-CA neurons, there was a significant increase in tail skin temperature within 2 min, followed by a rapid reduction (Fig. 4d, e), indicating an initial vasodilatory response and subsequent vasoconstriction response. Consistent with chemogenetic activation, optogenetic activation of VLM-CA neurons also decreased iBAT thermogenesis and core body temperature (Fig. 4f, g). Light stimulation had no effect on tail or iBAT temperature in control mice. These observations suggest that the activation of VLM-CA neurons rapidly increases heat dissipation, thereby facilitating a decrease in body temperature.
Activation of VLM-CA neurons drives cool preference
Behavior is another crucial component of the thermoregulatory system. We conducted a thermal gradient assay to assess the impact of VLM-CA neuronal activation on temperature preference. DbhVLM-hM3Dq mice were allowed to explore a linear track with varying temperature (15 °C to 50 °C) (Fig. 4h). At baseline, mice showed a consistent preference for the thermoneutral zone following saline injection, typically at ~30 °C. Upon CNO injection-induced neuronal activation, the mice actively chose to stay in the cooler zone rather than in the warmer zone (Fig. 4h–j), despite the fact that VLM-CA neuronal activation actually decreased their core body temperature. One possible explanation for this “cool-preference” behavior is that the “balance-point” or “set-point” of body temperature falls below the core body temperature, a characteristic also observed during natural entry into torpor and hibernation42,43.
Heart rate drop precedes body temperature drop
Torpid animals typically exhibit a profound decrease in heart rate, another key feature of torpor and hibernation. We conducted electrocardiogram (ECG) recordings to examine the impact of VLM-CA neuronal activation on heart rate in freely behaving mice (Fig. 5a). Remarkably, following CNO administration, the heart rate of DbhVLM-hM3Dq mice decreased significantly from a baseline of 730 ± 19 (mean ± SEM) bpm to 345 ± 26 bpm (Fig. 5b). Additionally, sinus arrhythmia (beat-to-beat heart rate variability) and skipped beats were frequently observed (Fig. 5c). These observations are consistent with the known effect of enhanced cardiac vagal tone44, a characteristic feature of the cardiovascular profile of natural torpor in mammals18,45. This pronounced reduction in heart rate was not observed following saline injections. These results suggest that the activation of VLM-CA neurons induces profound cardiovascular suppression, resembling the state observed in natural torpor.
a–c ECG recordings of heart rate in freely behaving DbhVLM-hM3Dq mice. a Experimental setup for ECG recordings. b Heart rate of freely behaving DbhVLM-hM3Dq mice after CNO or saline injection (P < 0.0001). c Representative ECG traces in mice 1 h after saline or CNO injections. Red arrows indicate skipped heartbeats. d Dynamic changes in heart rate and Tcore in a WT mouse during fasting-induced torpor. The bottom panel shows the four-phase fluctuations during the period indicated by the dashed box in the upper panel. e–i Simultaneous recordings of heart rate and Tcore in DbhVLM-SSFO mice following light stimulation (indicated by blue bars, 20 Hz, 30 s). e Experimental setup (left panel) and representative ECG traces (right panel). The red arrow indicates skipped heartbeats. f Changes in heart rate and Tcore of DbhVLM-SSFO mice. g Latency to decrease (upper panel) and latency to the lowest points (bottom panel) of heart rate and Tcore (n = 6 mice, each point represents one mice). h, i Representative traces of a DbhVLM-SSFO mouse showing the dynamic changes in heart rate and Tcore after light stimulation. i The four-phase fluctuations during the period indicated by the dashed box in (h). Two-way ANOVA analysis in (b). Two-tailed paired student’s t-test in (g). Data are presented as mean ± SEM. ****P < 0.0001. Source data are provided as a Source Data file. Figure 5a, e were created in BioRender. Zhan, C. (2025) https://BioRender.com/fi02cic.
Consistent with previous observations in naturally torpid animals46,47,48, we observed a complex relationship between core body temperature and heart rate in fasting mice, which can be divided into four phases. In phase (a), heart rate rapidly decreased from baseline to ~400 bpm before a significant drop in core body temperature (Fig. 5d). As the core body temperature continued to decline during the onset of torpor in phase (b), the heart rate further slowed to its lowest point. Subsequently, in phase (c), the heart rate rapidly increased, with no appreciable change in core body temperature. During the arousal phase (d) from torpor, as the core body temperature increased, the heart rate gradually returned to its baseline level. Furthermore, at any given core body temperature, the heart rate was slower upon entry into torpor compared to the rate during the arousal phase.
To investigate whether the VLM-CA neuronal activation could induce a complex relationship between core body temperature and heart rate similar to that observed in natural torpid animals, we conducted simultaneous recordings of core body temperature and heart rate in freely behaving mice (Fig. 5e). Upon initiating brief light stimulation (30 s, 2 Hz), the heart rate of DbhVLM-SSFO mice rapidly decreased within seconds (2.2 ± 1.2 s, mean ± SEM) and reached its minimum value after ~1 min (0.89 ± 0.21 min, mean ± SEM) (Fig. 5f, g). In contrast, the body temperature gradually decreased over minutes (2.41 ± 0.51 min, mean ± SEM) and reached its lowest point after more than 10 min (14.08 ± 1.28 min, mean ± SEM). Consistent with the findings of chemogenetic activation experiments (Fig. 5c), sinus arrhythmia and skipped beats were frequently observed following light stimulation (Fig. 5e). These results demonstrate that the decrease in heart rate induced by VLM-CA neuronal activation precedes reductions in core body temperature, which is consistent with previous observations in natural torpid animals7. Interestingly, the relationship between these two variables in VLM-CA neuron-activated mice can also be divided into four phases (Fig. 5h, i, and Supplementary Fig. 4), mirroring observations in natural torpid animals. Overall, these findings suggest that the pattern of changes in heart rate and core body temperature induced by VLM-CA neuronal activation is similar to that during natural torpor.
Downstream targets of VLM-CA neurons
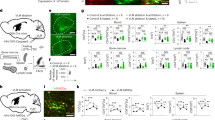
To investigate the downstream circuitry mechanisms by which VLM-CA neurons induce a torpor-like state, we conducted virus-based anterograde tracing experiments by injecting an AAV conditionally encoding mGFP (AAV-DIO-mGFP) into the VLM of Dbh-Cre mice (Fig. 6a). VLM-CA neurons project to multiple brain areas implicated in thermoregulation and autonomic outflow control, including the brainstem dorsal vagal complex (DVC), dorsomedial hypothalamus (DMH), paraventricular hypothalamus (PVN), and POA (Fig. 6b and Supplementary Fig. 5). The projections of VLM-CA neurons extensively cover the entire POA, with particularly dense innervations in the medial preoptic area (MPA) (Fig. 6b). Within the DVC, the projections span three specific brain regions: the area postrema (AP), nucleus of the solitary tract (NTS), and dorsal motor nucleus of the vagus (DMV), with heaviest innervations to the DMV region (Fig. 6b).
a, b Anterograde tracing of VLM-CA neurons. a Schematic of anterograde tracing (upper) and labeled VLM-CA neurons (bottom). b Representative images of VLM-derived projections in DVC, DMH, MPA, and PVN (n = 4 regions per 7 independent biological replicate). Scale bars: 200 µm. c Representative images of Fos signals in DVC, DMH, MPA, and PVN of DbhVLM-hM3Dq (bottom panels, n = 3 animals) and DbhVLM-mCherry control (upper panels, n = 2 animals) mice. Fos immunostaining was performed 2 h after CNO injections. Scale bars: 100 µm. Schematic of the Fos-Triggered Downstream Neuronal Activation (FTDNA) strategy (d) and representative results (e) of selective expression of SSFO in VLM-activated neurons (Fos+) in the MPA (n = 521 cells in 3 MPA area sections from 3 animals). FTDNA-based optogenetic activation (indicated by blue bars, 20 Hz, 30 s) of the VLMM3 → PVNSSFO (f, P = 0.0028), VLMM3 → DMHSSFO (g, P = 0.0012), VLMM3 → MPASSFO (h, P = 0.0042), and VLMM3 → DVCSSFO (i, P = 0.0019) neural circuits, respectively. The left panels show SSFO expression and fiber implantation, and the right panels show Tcore. Two-way ANOVA analysis in (f, g, h, i). Data are presented as mean ± SEM. *P < 0.05; **P ≤ 0.01. Source data are provided as a Source Data file.
In line with the anatomical projections, Fos signals were increased in the DVC, DMH, PVN, and MPA 2 h after CNO administration into DbhVLM-hM3Dq mice compared to DbhVLM-mCherry control mice (Fos+ cell numbers per brain slice, DbhVLM-hM3Dq vs control: DVC 48 ± 39 vs 11 ± 4, DMH 99 ± 88 vs 26 ± 12, PVN 187 ± 135 vs 16 ± 10, MPA 50 ± 30 vs 18 ± 12. Mean ± SEM) (Fig. 6c). These results suggest functional connectivity between VLM-CA neurons and these downstream brain regions.
To avoid the potential non-specific downstream activation caused by back-propagating optogenetic action potentials during terminal stimulation, we developed a targeted downstream neuronal activation method. We crossbred Dbh-Cre or Dbh-Flp mice with mice harboring inducible iCre recombination driven by Fos expression to generate Dbh-Cre::Fos2A-iCre or Dbh-Flp::Fos2A-iCre mice. AAV-DIO-hM3Dq-mCherry or AAV-fDIO-hM3Dq-mCherry was injected into the VLM, and AAV-DIO-SSFO-eYFP was injected into downstream sites (e.g., the MPA) (Fig. 6d). Dbh-Cre/Flp or Fos2A-iCre negative littermates were used as control. After 2 weeks of viral expression, CNO was administered to activate VLM-CA neurons and induce Fos expression in their downstream neurons. Concurrently, 4-OHT was given to prompt downstream neuronal expression of Cre, leading to selective expression of SSFO in those downstream neurons (Fig. 6d, e). Pre-implanted optical fibers delivered blue light to selectively activate SSFO-expressing neurons at downstream sites. Through this Fos-Triggered Downstream Neuronal Activation (FTDNA) strategy, we selectively activated VLM-CA’s downstream neurons in the PVN, DMH, MPA, and DVC, respectively. Our results showed that the activation of downstream neurons in the PVN and DMH did not lead to a reduction in body temperature; instead, it resulted in an increase in body temperature (Fig. 6f, g). In contrast, the activation of downstream neurons in the MPA and DVC significantly decreased core body temperature (Fig. 6h, i). These results suggest that the VLMDbh → DVC and VLMDbh → MPA neural circuits rather than the other two are sufficient to decrease body temperature.
Leveraging the FTDNA strategy, we further examined the impacts of the VLMDbh → DVC and VLMDbh → MPA neural circuits on heart rate and thermogenesis. Selective activation of the downstream neurons in the DVC, similar to the activation of VLM-CA neurons, caused a rapid and significant reduction in heart rate, accompanied by a relatively slow decrease in core body temperature, iBAT temperature, and tail temperature (Supplementary Fig. 6a–e). Different from the VLMDbh → DVC neural circuit, selective activation of MPA downstream neurons led to a relatively slow and moderate reduction in the heart rate (Supplementary Fig. 6f, g). The reductions in core body temperature, iBAT temperature, and tail surface temperature were comparable between these two neural circuits (Supplementary Fig. 6c–e and h–j).
The VLM → MPA neural circuit
We observed that the body temperature decrease induced by FTDNA-based activation was relatively small, which was likely due to the limitations of this strategy (refer to the “Discussion” section for details). To further confirm the regulatory effects of the VLMDbh → MPA and VLMDbh → DVC neural circuits on body temperature and heart rate, we selectively activated them by directly infusing CNO into the DVC or MPA of DbhVLM-hM3Dq mice, respectively (Fig. 7a, b). Direct infusion of CNO into either the MPA led to significant reductions in core body temperature by ~2 °C, as well as in iBAT temperature (Fig. 7c, e). However, the VLMDbh → MPA circuit had no significant impacts on heart rate (Fig. 7d). These data suggest that the VLMDbh → MPA circuit mainly contributes to the regulation of core body temperature and thermogenesis rather than heart rate.
a, b Selective activation of the VLMDbh → DVC or VLMDbh → MPA neural circuits by direct infusion of CNO (1 µl, 0.5 µg/µl dissolved in saline) into the DVC or MPA through pre-implanted cannulas. a Experimental schematic. b Representative images of cannula positions in the DVC and MPA. The effects of selective activation of the VLMDbh → MPA neural circuit on core body temperature (c, P = 0.0461), heart rate (d, P = 0.0315), and iBAT thermogenesis (e, P = 0.0095). The effects of selective activation of the VLMDbh → DVC neural circuit on core body temperature (f, P = 0.0195), heart rate (g, P = 0.0084), and iBAT thermogenesis (h, P = 0.0122). i In vitro whole-cell recording of DMV neurons during optogenetic activation of VLM-CA neuronal projections (blue bar, 5 ms) in the DVC of DbhVLM-ChR2 mice. Representative traces (middle panel) of EPSCs following administering: (1) artificial cerebrospinal fluid (ACSF), (2) picrotoxinin (PTX), (3) 6,7-dinitroquinoxaline-2,3-dione (DNQX), and (4) after a washout period. Group data (right panel, n = 9 neurons). All recordings were performed during the application of TTX (10 µM) and 4-AP (1 mM). The effects of saline or isoproterenol on the heart rate (j) and Tcore (k). l–r The impacts of selective inhibition of the MPA or DVC on the body temperature and heart rate. l Schematic of the FTDNI strategy. Changes in core body temperature (m, P = 0.1453), heart rate (n, P = 0.8122) and iBAT temperature (o, P = 0.3244) when selectively inhibiting MPA neurons. Changes in core body temperature (p, P = 0.7215), heart rate (q, P = 0.7967) and iBAT temperature (r, P = 0.9174) when selectively inhibiting DVC neurons. Two-way ANOVA analysis in (c–h, m–r). Two-tailed paired Student’s t-test in (i). Data are presented as mean ± SEM. ns, not significant P > 0.05; *P < 0.05; **P ≤ 0.01. Source data are provided as a Source Data file. Figure 7a includes a gavage tube illustration created with BioRender.com. Zhan, C. (2025) https://BioRender.com/k4e71f1. Figure 7i was created in BioRender. Zhan, C. (2025) https://BioRender.com/fi02cic. Figure 7l were manually drawn based on anatomical features using Microsoft PowerPoint.
The MPA consists of many types of neurons, including glutamatergic neurons, GABAergic neurons, Adcyap1-positive neurons, ERα-positive neurons among others. Many of these neuron subtypes have been implicated in the regulation of thermogenesis and body temperature11,12,15. To determine which neuron types in the MPA may be recruited by VLM-CA neurons for controlling thermogenesis and body temperature, we conducted in situ hybridization along with FOS immunostaining in the MPA following CNO injections into DbhVLM-hM3Dq mice (Supplementary Fig. 7a). Among MPA Fos+ neurons induced by the VLM-CA neuronal activation, ~30% were Vgat+ neurons, 20% were Vglut2+, 15% were Adcyap1+ neurons, and 10% were Erα+ neurons (Supplementary Fig. 7b, c). These results suggest that VLM-CA neurons might recruit multiple neuronal subtypes in the MPA to regulate thermogenesis and body temperature.
The VLM → DVC neural circuit
Direct infusion of CNO into the DVC of DbhVLM-hM3Dq mice led to significant reductions in core body temperature and iBAT temperature (Fig. 7f, h). Moreover, the reduction in core body temperature induced by the VLMDbh → DVC circuit appears to be more significant and last longer than that induced by the VLMDbh → MPA circuit (Fig. 7c, f). Notably, selective activation of VLMDbh → DVC circuit significantly decreased the heart rate (Fig. 7g), suggesting its important role in heart rate regulation.
The DVC consists of the DMV, NTS, and AP, and the DMV provides vagal efferents controlling cardiac function49,50,51. To determine the specific DVC subnucleus responsible for the heart rate-lowering effect, we injected AAV-DIO-ChR2-mCherry into the VLM of Dbh-Cre mice (referred to as DbhVLM-ChR2 mice) and conducted in-vitro patch clamp electrophysiological recordings in the DMV, NTS, and AP. Our results showed that optogenetic activation of VLM-CA neuronal projections in the DVC exclusively evoked excitatory postsynaptic currents (EPSC) in DMV neurons (Fig. 7i), while no such response was observed in NTS and AP neurons (Supplementary Fig. 8). These results indicate that only DMV neurons receive excitatory monosynaptic inputs from VLM-CA neurons.
We next explored the relationship between heart rate and core body temperature using isoproterenol, a drug commonly used to treat bradycardia. In wild-type mice, treatment with isoproterenol (30 mg/kg, IP) increased heart rate without affecting core body temperature when compared to saline treatment (Supplementary Fig. 9). Importantly, in DbhVLM-SSFO mice, isoproterenol treatment not only counteracted the reduction in heart rate but also that in core body temperature induced by the VLM-CA neuronal activation (Fig. 7j, k). These results suggest that the reduction in heart rate may be the major peripheral mediator in the body temperature decline induced by the VLM-CA neuronal activation.
To examine the necessity of the MPA and DVC circuits in mediating the torpor-like state induced by the VLM-CA neuronal activation, we employed a Fos-Triggered Downstream Neuronal Inhibition (FTDNI) strategy to silence the downstream neurons in the MPA or DVC separately. Similar to the FTDNA strategy, we injected AAV-fDIO-hM3Dq-mCherry into the VLM of Dbh-Flp::Fos2A-iCre mice, and the chemogenetic inhibitory AAV-DIO-hM4Di-GFP into MPA or DVC (Fig. 7l). As control groups, AAV-fDIO-mCherry and AAV-DIO-GFP were injected into the VLM and downstream areas respectively. After 2 weeks of viral expression, CNO was administered to activate VLM-CA neurons and induce hM4Di expression in their downstream neurons. Through this strategy, CNO injections allowed us to inhibit downstream neurons in the MPA or DVC while activating VLM-CA neurons. We found that chemogenetic inhibition of either MPA or DVC neurons failed to prevent the reductions in core body temperature, heart rate, and iBAT temperature induced by the VLM-CA neuronal activation (Fig. 7m–r). Simultaneous inhibition of the MPA even led to a more pronounced decrease in body temperature (Fig. 7m), although it did not reach a statistical significance. The inhibition of downstream neurons in the MPA or DVC alone had no impact on body temperature and heart rate. These data suggest that VLM-CA neurons do not necessarily rely on the MPA and DVC to induce the torpor-like state, indicating that other downstream pathways may also play a role.
VLM-CA neuronal activity in Daurian ground squirrels
To determine the potential roles of VLM-CA neurons in hibernation, we examined their presence in the Daurian ground squirrel (Spermophilus dauricus), a known hibernator, by performing immunostaining with the TH antibody. In northern regions of China, Daurian ground squirrels usually prepare for hibernation during the late summer and early autumn months, with hibernation typically commencing around October52. They hibernate from approximately October to next March to endure the winter season. Similar to what is observed in mice, many CA neurons are present in the VLM of the Daurian ground squirrel along the rostral-caudal axis (Supplementary Fig. 10a). Using Fos immunostaining, we detected VLM-CA neuronal activity in ground squirrels across different seasons. Interestingly, ground squirrels just before hibernation onset (in October in North China, with an ambient temperature of ~15 °C) exhibited significantly more Fos+ VLM-CA neurons than did those in non-hibernation seasons (in May in North China, with an ambient temperature of ~25 °C) and during hibernation (in November or December in North China, with an ambient temperature of ~5 °C) (Supplementary Fig. 10b, c). There was no significant difference observed among the animals in the hibernation and non-hibernation seasons. These results suggest that VLM-CA neurons are activated immediately before the onset of hibernation rather than during non-hibernation or hibernation seasons.
Discussion
Our study shows that fasting-activated brainstem VLM-CA neurons are required for fasting-induced torpor and can also induce a torpor-like state. Inhibiting these neurons significantly impairs torpor initiation and duration, while activating them mimics natural torpor’s physiological and metabolic changes, including the reductions in energy expenditure, iBAT thermogenesis, physical activity, body temperature, heart rate, and the body temperature “balance-points” (Supplementary Fig. 11).
The physiological changes induced by VLM-CA neuron activation meet the three criteria for torpor7,53,54: (1) a decrease in body temperature with active thermoregulation; (2) controlled cardiovascular depression; and (3) vasoconstriction, which distinguishes torpor from heat-defense responses. However, VLM-CA neurons are likely not the only ones involved in fasting-induced torpor, as fasting activates other neurons and inhibiting VLM-CA neurons does not fully abolish torpor. There are differences between the VLM-CA-induced torpor-like state and natural torpor. For example, tail vasodilation was initially observed after VLM-CA activation, followed by vasoconstriction, indicating a dynamic vascular response not typically reported in natural torpor. Activating VLM-CA neurons increased heat dissipation and made mice prefer cooler environments, perhaps due to a lower body temperature “balance-point”. Although some naturally torpid animals also like cooler places55, increased heat dissipation during natural torpor hasn’t been reported. This may be because the body temperature “balance-point” drops slowly in natural torpor, making it hard to notice a quick rise in heat dissipation. Moreover, while sympathectomy of iBAT does not affect the ability of mice to enter and exit torpor, it does impact the temperature fluctuations and the lowest temperature during fasting-induced torpor. This is also different from its lack of effect on the body temperature decrease induced by the VLM-CA neuron activation.
The VLM-CA neurons probably induce torpor through different downstream neural circuits. They control both the sympathetic and parasympathetic arms of the autonomic nervous system25,26,27, and the VLM → DVC circuit likely influences heart rate and body temperature by regulating parasympathetic activity. Notably, VLM-CA activation triggers a heart rate decline that precedes the drop in body temperature, and isoproterenol blocks both reductions, suggesting a causal role for cardiac modulation in thermoregulation. While isoproterenol did not increase iBAT temperature in fed mice (data not shown), we cannot exclude the possibility that its effects on core temperature involve indirect BAT stimulation. Future studies should employ parasympathetic blockers or β1/β2-specific agonists to selectively decouple heart rate changes from thermogenic responses. Although heart rate and body temperature often fluctuate together, such as during sleep or exercise, the magnitudes of the decreases in heart rate and body temperature induced by VLM-CA neuron activation are relatively large. This could be because mice are small animals with a higher susceptibility to fluctuations, or because VLM-CA neuron activation decreases body temperature “balance point”, facilitating the temperature decrease. Previous studies have implicated multiple MPA neuronal populations in controlling fasting-induced torpor13,14,15, and our results show that the VLM → MPA neural circuit may reduce thermogenesis and body temperature by activating multiple neuron subtypes in the MPA, with little impact on heart rate. Surprisingly, inhibiting the VLM → DVC and VLM → MPA circuits failed to alter the body temperature and heart rate decreases induced by VLM-CA neuron activation. This finding, combined with the fact that VLM-CA neurons can still reduce iBAT thermogenesis and body temperature even after sympathectomy, indicates the involvement of complex neural circuits. Additionally, the long-lasting hypothermia induced by VLM-CA neuron activation, which persists far beyond the decay time of CNO, suggests the existence of an unknown mechanism for maintaining this hypothermic state.
It must be acknowledged that the FTDNA and FTDNI methods we employed to manipulate downstream neurons have limitations. Firstly, we cannot confirm that the targeted neurons are directly downstream of VLM-CA neurons; they could be several synapses away. Secondly, these methods may inadvertently modulate neurons outside the intended downstream circuit. Moreover, the TRAP system used in these methods has poor temporal control. Consequently, the TRAPed neurons might be activated in response to downstream physiological changes. Therefore, advanced techniques such as anterograde trans-synaptic viruses (e.g., HSV) are required to explore the central-to-peripheral circuits.
We observed the presence of VLM-CA neurons in the Daurian ground squirrel and noted their activation just before the onset of seasonal hibernation, suggesting these neurons may contribute to the induction of hibernation. Further exploration of the role of VLM-CA neurons in hibernation will help us better understand the similarities and differences in the mechanisms underlying torpor and hibernation. Additionally, VLM-CA neurons are highly conserved across various species56,57, including humans. Although humans do not enter torpor or hibernation, we do experience reduced resting energy expenditure during fasting, accompanied by slight but noticeable decreases in core body temperature and heart rate58. Exploring the manipulation of VLM-CA neurons in larger animals and potentially even in humans to induce artificial hypothermia in future studies could offer valuable insights into their potential applications in clinical settings, such as organ transplantation, and even in the context of space travel4,59,60.
Methods
Animals
All mice were housed at 22–24 °C with a 12 h light/12 h dark cycle and provided with standard pellet chow (SPF-F02, SiPeiFu) and water ad libitum. All transgenic mice used in this study were on the C57BL/6J background. All experiments were performed on adult mice (8–16 weeks old, both male and female). Dbh-Cre mice (RRID: MMRRC_036734-UCD) and Dbh-Flp mice (RRID: MMRRC_041575-UCD) were obtained from the MMRRC. Similarly, Dbh-Cre::Fos2A-iCre mice were generated by crossing Dbh-Cre mice with Fos2A-iCre mice. Dbh-Flp::Fos2A-iCre mice were generated by crossing Dbh-Flp mice with Fos2A-iCre mice. Animal care and procedures were approved by the University of Science and Technology of China in compliance with institutional guidelines for the ethical care and use of animals.
Daurian ground squirrels (S. dauricus) utilized in this study were captured in Shenyang, Liaoning Province, China (123 °E, 42 °N), in May 2021. The animals were transported to and housed in the laboratory of Shenyang Normal University. Each squirrel was individually housed in plastic cages (48 × 35 × 20 cm) with sawdust bedding. All squirrels were kept under natural temperature and light cycles. Water and food (commercial rat chow from Shenyang QianMin Feed Co.) were provided ad libitum. Prior to the experiment, all animals (1–2 years old, both male and female) were acclimated to these conditions for a minimum of 2 weeks. The physiological states of squirrels were determined through observation, the season, the animals’ body weight, and their reactions when handled. For the ground squirrels sacrificed in May (non-hibernation season), their body weight remained relatively stable at around 250 g, and they showed obvious resistance when handled. For the ground squirrels sacrificed in October (before hibernation onset), their body weight increased rapidly to around 400 g, and they resisted handling but not strongly. In November or December (hibernation season), they mostly stopped feeding, and their body weight decreased significantly, ranging from about 200 g to 300 g. The use of Daurian ground squirrels was approved by the Animal Care and Use Committee of Shenyang Normal University (CLS― XingX-2023-1).
Torpor induction
To induce torpor through fasting, the mice were individually housed at ambient temperature (22–24 °C). Food was removed from the cages at the beginning of the dark phase. After 24 h of fasting, food was provided to the mice. Torpor was defined as the state in which the core body temperature equaled or fell below 31 °C. To ensure consistency in our experiment, female mice weighing between 18 and 25 grams and male mice weighing between 25–30 grams were selected. To induce torpor through neuronal activation, the mice were individually housed at an ambient temperature of 22–24 °C and provided unrestricted access to food and water.
Viral injections
Mice were anesthetized with 2,2,2-tribromoethanol (240 mg/kg) dissolved in 2.5% 2-methyl-2-butanol and placed in a stereotaxic holder (RWD Life Science, China). Viral vectors were injected into the target brain areas with pressure at a rate of 50 nL/minute (Nanoliter 2000 Injector, WPI). For chemogenetic inhibition or activation, AAV2/9-DIO-hM4Di-mCherry (Cat#: AAV-DR039, PackGene Biotech. 2 × 1012 V. G/ml, 300 nl for each side) or AAV2/9-DIO-hM3Dq-mCherry (Cat#: 44361, Addgene, 2 × 1012 V/G/ml, 300 nl for each side) was bilaterally injected into the VLM (VLM coordinates AP/ML/DV: −6.9/±1.2/−5.5 mm) of Dbh-Cre mice. For optogenetic activation, AAV2/9-DIO-SSFO-eYFP (Cat#: 35503, Addgene. 2 × 1012 V. G/ml, 300 nl for each side) or AAV-DIO-ChR2-mCherry (Cat#: 55645, Addgene, 2 × 1012 V. G/ml, 300 nl) was bilaterally injected into the VLM of Dbh-Cre mice. For anterograde tracing, AAV2/9-DIO-mGFP (Cat#: S0276-9, Tailtool. 2 × 1012 V. G/ml, 300 nl for each side) was bilaterally injected into the VLM of Dbh-Cre mice. To selectively activate downstream targets of VLM-CA neurons, AAV2/9-DIO-hM3Dq-mCherry (Cat#: 44361, Addgene. 2 × 1012 V. G/ml, 300 nl for each side) and AAV2/9-DIO-SSFO-eYFP (Cat#: 35503, Addgene. 2 × 1012 V. G/ml, 300 nl for each side) were bilaterally injected into the VLM and its downstream areas of Dbh-Cre::Fos2A-iCre mice, respectively. AAV2/9-DIO-mCherry or AAV2/9-DIO-mGFP was injected as a control.
Fos TRAPing
Dbh-Cre::Fos2A-iCre mice or Dbh-Flp::Fos2A-iCre mice were used to trap downstreams of VLM-CA neurons. Two weeks after these mice received AAV virus injections, 4-OHT (H6278, Sigma‒Aldrich) was administered intraperitoneally (IP) following CNO injection. 4-OHT was dissolved in ethanol at a concentration of 20 mg/mL, a double volume of a 1:4 mixture of castor oil:sunflower oil was added, and the mixture was vortexed. The ethanol was evaporated via vacuum centrifugation, and the final concentration was 10 mg/mL. Each mouse was administered a dose of 50 mg/kg.
Optogenetic stimulation
For the delivery of the laser, optical fibers (200 μm in diameter, NA = 0.39, Cat#: FT200UMT, Thorlabs) were inserted into the targeted brain areas and affixed to the skull with dental cement. A blue laser (473 nm, SDL-473-100T, DreamLaser, Shanghai) was used to generate blue laser pulses (30 s, 2 Hz, 10 mW) for optogenetic stimulation.
Chemogenetic manipulation
CNO (Cat#: BML-NS105-0025, ENZO) was dissolved in saline. For chemogenetic activation of VLM-CA neurons, 2 mg/kg CNO was administered (IP). For chemogenetic inhibition of VLM-CA neurons, CNO was administered (IP) at a dose of 3 mg/kg. For the selective chemogenetic activation of the VLM → MPA or VLM → DVC neural circuits, cannulas were inserted into the targeting brain areas. CNO (1 µl, 0.5 µg/µl dissolved in saline) was delivered into the MPA or DVC through the pre-implanted cannulas. Post-histology examinations were conducted to exclude mice that did not show viral expression in the VLM or had inaccurately implanted cannulas (Deviation exceeding 500 microns) from further data analysis.
Telemetric recording of core body temperature and locomotor activity
Mice were singly housed and implanted abdominally with telemetric temperature and activity probes (Starr Life Sciences E-Mitter-G2). After a recovery period of at least 1 week, the mice were recorded in standard cages placed onto a radiofrequency receiver (Starr Life Sciences ER4000). Core body temperature and locomotor activity were continuously monitored in freely behaving mice every 3 min or 10 s.
Heart rate recording and analysis
To record the heart rate (electrocardiogram [ECG]), two electrodes were sutured to the muscles of the chest wall. The electrode wires were subcutaneously guided to the top of the head through a midline incision extending over the skull to the base of the neck. Dental cement and Super-Bond C&B were applied to affix ECG electrodes to the skull and cover the exposed skull completely. After a recovery period of at least 1 week, ECG signals were recorded (Amplifier Model 1700, A-M System, USA), bandpass filtered (10–5000 Hz), and digitized at 250 Hz with Intracept Chat software. The ECG electrodes were connected to flexible recording cables via a slip ring to prevent entanglement during animal movement. The heart rate was calculated from ECG traces using a custom-written MATLAB code.
Thermographic analysis
Mice were placed in experimental cages (30 × 30 × 38 cm), and their surface body temperature was monitored using an infrared thermal imaging camera (Fotric 225S, IRtek, Shanghai). To ensure accurate surface temperature readings, the back hair of the mice was carefully removed with hair clippers 1 day before the experiment commenced. Data from the thermograms were collected at a frequency of 1 Hz and subsequently analyzed using AnalyzIR software (IRtek, Shanghai).
Thermal gradient assay
A custom thermal gradient was constructed using an 8.7 × 8.7 × 140 cm square aluminum tube. Mice were allowed to explore the center region measuring 8.7 × 8.7 × 120 cm. The cold end of the tube was covered in a bespoke box and maintained at 0 °C using ice, whereas the warm end was positioned on a heated surface that was set to 150 °C. Before the start of the experiment, the temperature of the gradient was allowed to stabilize for an hour, and temperature measurements were checked regularly at set position intervals. The corresponding temperatures for each gradient position were measured from readings taken after each experiment. During the experiment, mouse movement traces were recorded using a Logitech video camera and subsequently analyzed using a custom-written MATLAB code. Prior to the administration of saline or CNO, the mice were given 1 h to explore the pre-stabilized temperature gradient and establish their preferred temperature.
Measurements of O2 consumption, CO2 production, and energy expenditure
Oxygen consumption and CO2 production were recorded using a combined indirect gas calorimetry system (PhenoMaster, TSE, Germany). Following 1 day of adaptation in test cages, the mice received a dose of CNO. Oxygen expenditure and CO2 production data were normalized to mouse body weight. Energy expenditure was determined by analyzing indirect calorimetry data and was normalized to body weight using the following Weir equation:\({EE}=\left(3.94{V}_{O2}+1.1{V}_{{CO}2}\right)\times 1440\).
Measurement of noradrenaline concentration in iBAT
After euthanasia by cervical dislocation, intrascapular brown adipose tissue (iBAT) was dissected, weighed, and stored in liquid nitrogen. The tissue was homogenized in 0.1 M perchloric acid (1:4, w/v) at 4 °C, followed by centrifugation at 21,300 × g at 4 °C. The middle layer was collected and subjected to centrifugal filtration (Amicon® Ultra, 30 kDa). The filtrate was diluted 2-fold and then prepared for subsequent mass spectrometry analysis. The noradrenaline concentration was measured using an Ultra-High-Performance Liquid Chromatography (UHPLC) system (Waters, Milford, MA, USA) coupled with a Triple Quadrupole 7500 mass spectrometer (SCIEX, Framingham, MA, USA). Chromatographic separation was achieved using a Waters Acquity Premier HSS T3 column (2.1 × 100 mm, 1.8 µm). The column temperature was maintained at 40 °C. The mobile phases consisted of A (0.1% formic acid in water) and B (0.1% formic acid in water/acetonitrile (ACN) mixture). A linear gradient elution program was applied, starting with 100% A (0.0–2 min), then transitioning to 100% A to 5% A (2.0–7.0 min), maintaining 5% A (7.0–9.0 min), and further shifting to 0% A (9.0–9.1 min). The gradient was finalized by holding at 100% B for 1.9 min. The flow rate was set to 0.3 mL/min, and the sample injection volume was 5 µL. The electrospray ionization (ESI) source parameters were as follows: spray voltage, 1500 V, in positive ion mode.
Real-time PCR
Two hours after the administration of CNO or saline, mice were euthanized using carbon dioxide inhalation, and intrascapular brown adipose tissue (iBAT) was rapidly dissected and stored in liquid nitrogen. Total RNA was extracted using TRIzol reagent (Catalog No. 15596-026, Life Technologies). One microgram of total RNA was reverse transcribed into cDNA using PrimeScript RT Master Mix (Perfect Real Time, Catalog No. RR036A, Takara). qPCR was performed using a SYBR Green PCR kit (Cat#: 1725121, Bio-Rad. Gene expression levels were analyzed using a Bio-Rad CFX96 real-time PCR detection system (Bio-Rad Laboratories), and Ucp1 gene expression levels were normalized to Gapdh RNA expression.
Electrophysiological recordings
Mice were anesthetized via intraperitoneal injection of 2,2,2-tribromoethanol (240 mg/kg). After confirming the depth of anesthesia, the brains were quickly removed and placed in ice-cold, oxygenated artificial cerebrospinal fluid (aCSF). Brain sections (250 µm) were immersed in and perfused with oxygenated artificial cerebrospinal fluid (aCSF) containing 125 mM NaCl, 2 mM CaCl2, 2.5 mM KCl, 1.3 mM MgCl2, 1.3 mM NaH2PO4, 1.3 mM Na-ascorbate, 0.6 mM Na-pyruvate, 25 mM NaHCO3, and 20 mM glucose. Neurons were visualized and recorded using an Olympus BX51WI microscope equipped with an infrared (IR) differential interference contrast (DIC) and a charge-coupled device (CCD) camera (IR-2000, DAGE-MTI). Patch pipettes were pulled from borosilicate glass capillary tubes (OD = 1.5 mm, ID = 0.84, Sutter Instrument) using a Model P-1000 puller (Sutter Instrument) to achieve a final tip resistance of 4–7 MΩ. The pipettes were filled with a solution containing 126 mM K-gluconate, 10 mM HEPES, 4 mM KCl, 4 mM Mg-adenosine triphosphate (ATP), 0.3 mM Na4-guanosine triphosphate (GTP), 0.05 mM EGTA, and 10 mM phosphocreatine for recording action potentials (current clamp). All recordings were performed using an EPC 10 USB amplifier (HEKA Elektronik), and electrophysiological data were filtered at 2.9 kHz and sampled at 20 kHz using Patchmaster software.
For optogenetic stimulation, a brief pulse of blue light (470 nm, 2 Hz, 30 s) from a light-emitting diode (LED) source was applied to brain sections through an Olympus 40× water-immersion lens to activate SSFO+ or ChR2+ neurons. An EPC 10 USB amplifier was used to control the pulse onset, duration, and frequency of light stimulation. For chemogenetic manipulation, brain sections were perfused with CNO (10 µM for neuronal activation and 50 µM for neuronal inhibition) for ~10 min.
Bilateral iBAT sympathectomy
For bilateral iBAT sympathectomy, the iBAT was exposed by making cuts through the overlaying skin and adjacent muscles under a microscope. Next, five sympathetic nerve bundles that supplied each lobe of the iBAT were carefully identified, lifted with forceps, and cut. A similar procedure was carried out in the sham-operated groups, except that no sympathetic nerve incision was made in the iBAT. After the surgery, the mice were allowed to recover for 1 week before any further experiments were conducted.
Histology, immunostaining, in situ hybridization, and imaging
Mice or squirrels were deeply anesthetized with 2,2,2-tribromoethanol (300 mg/kg). Then, they underwent transcardial perfusion with cold 1× PBS solution, followed by 4% PFA in PBS solution. The brains and iBAT tissues were subsequently extracted and post-fixed in 4% PFA overnight, and then dehydrated in 30% sucrose. Coronal brain sections (40 µm) and iBAT sections (50 µm) were obtained using a freezing cryostat (Leica CM 1950). These sections were either collected in PBS or mounted on glass slides. The tissue sections were incubated overnight at 4 °C with primary antibodies against TH (1:1000, AB152, Millipore) or Fos (1:1000, 2250S, Cell Signaling Technology). After that, they were incubated with secondary antibodies for 2–4 h at room temperature. The secondary antibodies used were Alexa 488-conjugated goat anti-rabbit (1:1000, 111-545-003, Jackson ImmunoResearch) or Alexa 647-conjugated goat anti-rabbit (1:500, 111-605-003, Jackson ImmunoResearch).
Regarding Fluorescence in situ hybridization (FISH), the brain sections underwent hybridization with digoxigenin-labeled antisense cRNA probes for Vgat (952 bp sequence amplified by primers GCCATTCAGGGCATGTTC and AGCAGCGTGAAGACCACC), Vglut2 (580 bp amplified by primers CCAAATCTTACGGTGCTACCTC and TAGCCATCTTTCCTGTTCCACT), Adcyap1 (861 bp sequence amplified by primers ACGCCCTTTACTATCCAGCG and TTGAAGAAGCGGCAGTAG), and ERα (886 bp sequence amplified by primers CAGTCTCTGGGCGACATTCT and TCTTTCCGTATGCCGCCTTT). Following hybridization, brain sections were incubated with anti-digoxigenin-POD (1:500, 11207733910, Roche) at 4 °C for 30 h. A TSA Plus Cyanine 3 kit (1:100, NEL744001KT, PerkinElmer) was used to detect the primary antibody.
The mounting medium used was glycerol with DAPI. Montage images were acquired and stitched using either a confocal microscope (LSM 800, Zeiss) or an automated slide scanner (VS200, Olympus).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Source Data are provided with this paper. Any additional information required to reanalyze the data reported in this work paper is available from the lead contact upon request.
References
Overton, J. M. & Williams, T. D. Behavioral and physiologic responses to caloric restriction in mice. Physiol. Behav. 81, 749–754 (2004).
Knight, W. D., Witte, M. M., Parsons, A. D., Gierach, M. & Overton, J. M. Long-term caloric restriction reduces metabolic rate and heart rate under cool and thermoneutral conditions in FBNF1 rats. Mech. Ageing Dev. 132, 220–229 (2011).
Zimmer, M. B. & Milsom, W. K. Effects of changing ambient temperature on metabolic, heart, and ventilation rates during steady state hibernation in golden-mantled ground squirrels (Spermophilus lateralis). Physiol. Biochem. Zool. 74, 714–723 (2001).
Ou, J. et al. iPSCs from a hibernator provide a platform for studying cold adaptation and its potential medical applications. Cell 173, 851–863.e816 (2018).
Cerri, M., Hitrec, T., Luppi, M. & Amici, R. Be cool to be far: exploiting hibernation for space exploration. Neurosci. Biobehav. Rev. 128, 218–232 (2021).
Carey, H. V., Andrews, M. T. & Martin, S. L. Mammalian hibernation: cellular and molecular responses to depressed metabolism and low temperature. Physiol. Rev. 83, 1153–1181 (2003).
Ambler, M., Hitrec, T. & Pickering, A. Turn it off and on again: characteristics and control of torpor. Wellcome Open Res. 6, 313 (2021).
Madden, C. J. & Morrison, S. F. Central nervous system circuits that control body temperature. Neurosci. Lett. 696, 225–232 (2019).
Tan, C. L. & Knight, Z. A. Regulation of body temperature by the nervous system. Neuron 98, 31–48 (2018).
Wang, T. A. et al. Thermoregulation via temperature-dependent PGD(2) production in mouse preoptic area. Neuron 103, 309–322.e307 (2019).
Tan, C. L. et al. Warm-sensitive neurons that control body temperature. Cell 167, 47–59.e15 (2016).
Zhao, Z. D. et al. A hypothalamic circuit that controls body temperature. Proc. Natl. Acad. Sci. USA 114, 2042–2047 (2017).
Hrvatin, S. et al. Neurons that regulate mouse torpor. Nature 583, 115–121 (2020).
Takahashi, T. M. et al. A discrete neuronal circuit induces a hibernation-like state in rodents. Nature 583, 109–114 (2020).
Zhang, Z. et al. Estrogen-sensitive medial preoptic area neurons coordinate torpor in mice. Nat. Commun. 11, 6378 (2020).
Withers, P. C. Metabolic, respiratory and haematological adjustments of the little pocket mouse to circadian torpor cycles. Respir. Physiol. 31, 295–307 (1977).
Geiser, F. Reduction of metabolism during hibernation and daily torpor in mammals and birds: temperature effect or physiological inhibition?. J. Comp. Physiol. B 158, 25–37 (1988).
Lyman, C. P. & O’Brien, R. C. Autonomic control of circulation during the hibernating cycle in ground squirrels. J. Physiol. 168, 477–499 (1963).
Nectow, A. R. et al. Identification of a brainstem circuit controlling feeding. Cell 170, 429–442.e411 (2017).
Schneeberger, M. et al. Regulation of energy expenditure by brainstem GABA neurons. Cell 178, 672–685.e612 (2019).
Bai, L. et al. Genetic identification of vagal sensory neurons that control feeding. Cell 179, 1129–1143.e1123 (2019).
Cao, W. H., Madden, C. J. & Morrison, S. F. Inhibition of brown adipose tissue thermogenesis by neurons in the ventrolateral medulla and in the nucleus tractus solitarius. Am. J. Physiol. Regul. Integr. Comp. Physiol. 299, R277–R290 (2010).
Morrison, S. F. RVLM and raphe differentially regulate sympathetic outflows to splanchnic and brown adipose tissue. Am. J. Physiol. 276, R962–R973 (1999).
Junkins, M. S., Bagriantsev, S. N. & Gracheva, E. O. Towards understanding the neural origins of hibernation. J. Exp. Biol. 225, jeb229542 (2022).
Abe, C. et al. C1 neurons mediate a stress-induced anti-inflammatory reflex in mice. Nat. Neurosci. 20, 700–707 (2017).
Schreihofer, A. M. & Guyenet, P. G. Identification of C1 presympathetic neurons in rat rostral ventrolateral medulla by juxtacellular labeling in vivo. J. Comp. Neurol. 387, 524–536 (1997).
Abbott, S. B. et al. Selective optogenetic activation of rostral ventrolateral medullary catecholaminergic neurons produces cardiorespiratory stimulation in conscious mice. J. Neurosci. 33, 3164–3177 (2013).
Souza, G., Stornetta, R. L., Stornetta, D. S., Guyenet, P. G. & Abbott, S. B. G. Adrenergic C1 neurons monitor arterial blood pressure and determine the sympathetic response to hemorrhage. Cell Rep. 38, 110480 (2022).
Burke, P. G., Coates, A. bbottS. B., Viar, M. B., Stornetta, K. E. & Guyenet, R. L. PG. Optogenetic stimulation of adrenergic C1 neurons causes sleep state-dependent cardiorespiratory stimulation and arousal with sighs in rats. Am. J. Respir. Crit. Care Med. 190, 1301–1310 (2014).
Sofia Beas, B. et al. A ventrolateral medulla-midline thalamic circuit for hypoglycemic feeding. Nat. Commun. 11, 6218 (2020).
Li, A. J., Wang, Q. & Ritter, S. Selective pharmacogenetic activation of catecholamine subgroups in the ventrolateral medulla elicits key glucoregulatory responses. Endocrinology 159, 341–355 (2018).
Abe, C. & Inoue, T. Role of C1 neurons in anti-inflammatory reflex: mediation between afferents and efferents. Neurosci. Res 136, 6–12 (2018).
Abe, C. et al. Repeated activation of C1 neurons in medulla oblongata decreases anti-inflammatory effect via the hypofunction of the adrenal gland adrenergic response. Brain Behav. Immun. 111, 138–150 (2023).
Wang, L. et al. Fasting-activated ventrolateral medulla neurons regulate T cell homing and suppress autoimmune disease in mice. Nat. Neurosci. 27, 462–470 (2024).
Ritter, S., Li, A. J., Wang, Q. & Dinh, T. T. Minireview: the value of looking backward: the essential role of the hindbrain in counterregulatory responses to glucose deficit. Endocrinology 152, 4019–4032 (2011).
Ritter, S., Dinh, T. T. & Li, A. J. Hindbrain catecholamine neurons control multiple glucoregulatory responses. Physiol. Behav. 89, 490–500 (2006).
Swoap, S. J., Gutilla, M. J., Liles, L. C., Smith, R. O. & Weinshenker, D. The full expression of fasting-induced torpor requires beta 3-adrenergic receptor signaling. J. Neurosci. 26, 241–245 (2006).
Jensen, T. L., Kiersgaard, M. K., Sorensen, D. B. & Mikkelsen, L. F. Fasting of mice: a review. Lab Anim. 47, 225–240 (2013).
Morrison, S. F., Madden, C. J. & Tupone, D. Central control of brown adipose tissue thermogenesis. Front. Endocrinol. 3, 5 (2012).
Rand, R. P., Burton, A. C. & Ing, T. The tail of the rat, in temperature regulation and acclimatization. Can. J. Physiol. Pharm. 43, 257–267 (1965).
Stujenske, J. M., Spellman, T. & Gordon, J. A. Modeling the spatiotemporal dynamics of light and heat propagation for in vivo optogenetics. Cell Rep. 12, 525–534 (2015).
Craig Heller, H., Colliver, G. W. & Beard, J. Thermoregulation during entrance into hibernation. Pflug. Arch. 369, 55–59 (1977).
Geiser F. Hibernation: endotherms. In Encyclopedia of Life Sciences (John Wiley & Sons Ltd, 2011).
Grossman, P., Wilhelm, F. H. & Spoerle, M. Respiratory sinus arrhythmia, cardiac vagal control, and daily activity. Am. J. Physiol. Heart Circ. Physiol. 287, H728–H734 (2004).
Milsom, W. K., Zimmer, M. B. & Harris, M. B. Regulation of cardiac rhythm in hibernating mammals. Comp. Biochem Physiol. A Mol. Integr. Physiol. 124, 383–391 (1999).
Swoap, S. J. & Gutilla, M. J. Cardiovascular changes during daily torpor in the laboratory mouse. Am. J. Physiol. Regul. Integr. Comp. Physiol. 297, R769–R774 (2009).
Mertens, A., Stiedl, O., Steinlechner, S. & Meyer, M. Cardiac dynamics during daily torpor in the Djungarian hamster (Phodopus sungorus). Am. J. Physiol. Regul. Integr. Comp. Physiol. 294, R639–R650 (2008).
Strumwasser, F. Thermoregulatory, brain and behavioral mechanisms during entrance into hibernation in the squirrel, Citellus beecheyi. Am. J. Physiol. 196, 15–22 (1959).
Machhada, A. et al. Control of ventricular excitability by neurons of the dorsal motor nucleus of the vagus nerve. Heart Rhythm 12, 2285–2293 (2015).
Gourine, A. V., Machhada, A., Trapp, S. & Spyer, K. M. Cardiac vagal preganglionic neurones: an update. Auton. Neurosci. 199, 24–28 (2016).
Falvey, A. et al. Electrical stimulation of the dorsal motor nucleus of the vagus in male mice can regulate inflammation without affecting the heart rate. Brain Behavior and Immunity. 120, 630–639 (2024).
Ren, Y., Song, S., Liu, X. & Yang, M. Phenotypic changes in the metabolic profile and adiponectin activity during seasonal fattening and hibernation in female Daurian ground squirrels (Spermophilus dauricus). Integr. Zool. 17, 297–310 (2022).
Song, X., Körtner, G. & Geiser, F. Reduction of metabolic rate and thermoregulation during daily torpor. J. Comp. Physiol. B 165, 291–297 (1995).
Himms-Hagen, J. Food restriction increases torpor and improves brown adipose tissue thermogenesis in ob/ob mice. Am. J. Physiol. 248, E531–E539 (1985).
Gumma, M. R., South, F. E. & Allen, J. N. Temperature preference in golden hamsters. Anim. Behav. 15, 534–537 (1967).
Arango, V. et al. Catecholaminergic neurons in the ventrolateral medulla and nucleus of the solitary tract in the human. J. Comp. Neurol. 273, 224–240 (1988).
Sawchenko, P. E. & Swanson, L. W. Central noradrenergic pathways for the integration of hypothalamic neuroendocrine and autonomic responses. Science 214, 685–687 (1981).
Muller, M. J. et al. Metabolic adaptation to caloric restriction and subsequent refeeding: the Minnesota Starvation Experiment revisited. Am. J. Clin. Nutr. 102, 807–819 (2015).
Stanzani, G., Tidswell, R. & Singer, M. Do critical care patients hibernate? Theoretical support for less is more. Intensive Care Med. 46, 495–497 (2020).
Aslami, H. & Juffermans, N. P. Induction of a hypometabolic state during critical illness—a new concept in the ICU?. Neth. J. Med. 68, 190–198 (2010).
Acknowledgements
We thank Dr. Yan-Chuan Shi (Garvan Institute) for teaching us iBAT bilateral sympathectomy; Dr. Haohong Li (Zhejiang University) for help with techniques and instruments; and Dr. Shiqiang Wang (Peking University) and the “Neuroscience Pioneer Club” for critical reading and discussion of the manuscript. C.Z. is supported by grants from the Strategic Priority Research Program of the Chinese Academy of Sciences (grant no. XDB0940000), the National Natural Science Foundation of China (82495183, 31822026, 32271063, 31500860), the Research Funds of the Center for Advanced Interdisciplinary Science and Biomedicine of IHM (QYPY20220018), and the National Science and Technology Innovation 2030 Major Project of China (2021ZD0203900).
Author information
Authors and Affiliations
Contributions
M. Cheng, M. Wang, L. Wang, and W. Gao were responsible for conducting chemogenetic and optogenetic experiments, as well as measuring body temperature, locomotion activity, energy expenditure, and ECG recordings. M. Cheng and L. Wang conducted immunostaining and real-time PCR experiments. F. Yin and Y. Fan designed and built custom systems for ECG recording and thermal gradient assays and conducted related data analysis. J. Shen and Z. Liu performed brain slice electrophysiological recordings. M. Cheng and M. Wang analyzed the data. X. Xing, M. Cheng, and M. Wang conducted experiments on ground squirrels. Y. Shi, P. Wu, and P. Cao assisted in manuscript preparation. C. Zhan supervised the study and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Wei Li and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
41467_2025_61179_MOESM3_ESM.mp4
Supplementary Movie 1
41467_2025_61179_MOESM4_ESM.mp4
Supplementary Movie 2
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Cheng, M., Wang, M., Wang, L. et al. Brainstem catecholaminergic neurons induce torpor during fasting by orchestrating cardiovascular and thermoregulation changes. Nat Commun 16, 5954 (2025). https://doi.org/10.1038/s41467-025-61179-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-61179-1