Abstract
Polymer dielectrics with enhanced thermal stability and electrical insulation are urgently needed for capacitive energy storage applications in electric power systems. There is a persistent challenge to break the contradictory correlation between high heat resistance and low electrical conduction in polymers. Here, we employ benzyl-induced crosslinking to rearrange short-range structural units in polyimide chains, reducing electrical conduction loss. The designed polymer exhibits an electrical conductivity more than 3 orders of magnitude lower than that of commercial heat-resistant polymers, while its glass transition temperature (Tg) increases from 236.31 °C (for polyetherimide) to 289.72 °C. Consequently, a discharged energy densities of 6.38 J cm−3 and 3.04 J cm−3, with charge-discharge efficiencies above 90%, are achieved at 200 °C and 250 °C, respectively, demonstrating among the best in all-organic dielectric polymers. This work presents a feasible approach to break the adverse correlation between thermal stability and electrical insulation in polyimide materials.
Similar content being viewed by others
Introduction
Dielectric capacitors, known for their characteristics of fast charge-discharge speeds and high operating voltage, are prevalent in modern electronic devices and power systems1,2,3. Dielectric polymers have shown great potential in the field of electrostatic energy storage due to their high breakdown strength, light weight, and facile processability4,5. In recent decades, there has been a growing demand for high-performance dielectrics that can operate stably under extreme conditions ranging from 150 °C to 250 °C, especially in the fields of clean energy, aerospace, and oil/gas explorations6,7,8. However, most polymers experience degraded electrical insulation at elevated operating temperatures. For instance, the charge-discharge efficiency of commercial biaxially oriented polypropylene (BOPP) remains at 95% at room temperature when the applied electric field is close to the breakdown limit, but sharply drops to below 65% at 120 °C9.
Heat-resistant dielectric polymers with high glass transition temperatures (Tg) and melting points (Tm), such as polyimide (PI) and fluorene polyester (FPE), contain numerous highly conjugated structures that facilitate the movement of electrons, leading to a rapid increase in electrical conductivity under applied thermal and electric fields10,11,12,13,14. To suppress the migration of electrons in heat-resistant polymers, insulated inorganic fillers or molecular semiconductors are normally selected to be added into the host polymers to improve their high-temperature capacitive performance15,16,17. Additionally, cyclic-olefin polymers and fully alicyclic polymers with non-conjugated and non-planar structures have been synthesized and reported, aiming to increase their forbidden bandwidth (Eg) to achieve desirable electrical insulation18,19. However, film dielectrics modified by these strategies show little improvement in thermal stability, which results in a significant deterioration of capacitive performance for these polymers at operating temperatures between 200 and 250 °C (higher than their Tg)20,21,22.
In aromatic polyimides, intramolecular and intermolecular charge transfer complexes (CTC) can be easily formed between the donor (aromatic diamine) and acceptor (aromatic dianhydride) segments23,24. Although the strong CTC effect gives rise to an increase in thermal stability, it significantly enhances the excitation and migration of electrons under elevated electric fields and high temperatures, ultimately leading to a sharp decrease in electrical insulation. Recently, a series of alicyclic polyimides has been reported to weaken the CTC effect of polymer chains25,26. However, the synthetic process of the alicyclic polyimides is relatively complicated and immature, which hinders the application prospects of alicyclic polyimides in the market. Therefore, it is urgent to develop simple and commercially feasible methods to address the problem of increased electrical conduction loss in PI due to the CTC effect.
In this study, we reported a facile and practical strategy employing benzyl-induced crosslinking to interfere with the rearrangement of monomeric units within polymer chains. This approach leads to the formation of a preferred layer packing (PLP) structure in polymer dielectrics, thereby effectively mitigating the intermolecular CTC effect. The strategy simultaneously achieves suppressed high-temperature electrical conductivity and improved heat resistance in polymer dielectrics. Additionally, the experimental results demonstrate that the PLP structure is beneficial for the enhancement of the polymer modulus under high temperatures, thereby impeding the occurrence of electromechanical breakdown. Consequently, the resultant film exhibits a high discharged energy density (Ue) of 6.38 J cm−3 at 200 °C and 3.04 J cm−3 at 250 °C, with an η exceeding 90%. This work successfully demonstrates the feasibility of rearranging short-range structural units within polymer chains with excellent capacitive energy storage performance in large-scale films under harsh environments.
Results and discussion
Structure design and characterization
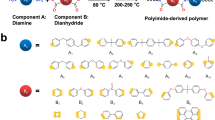
A schematic of the two polymers, PEI and TPEI, synthesized from the commercial dianhydride monomer 4,4′-(4,4′-isopropylidenediphenoxy) diphthalic anhydride (BPADA) and the diamine monomers phenylenediamine (PDA) and 2,3,5,6-tetramethyl-1,4-phenylenediamine (TPD), is shown in Fig. 1a, with the fabrication process detailed in the experimental methods section. The chemical structure of prepolymers was confirmed by 1HNMR (Fig. S1, Supporting Information). These polymer films were obtained through thermal imidization within a temperature range of 70 °C–290 °C in air, during which the polymer film containing benzyl functional groups underwent a thermo-oxidation crosslinking reaction. The crosslinking degree of the obtained TPEI film was measured using UV-vis spectroscopy (Fig. S2, Supporting Information), and the result was 57.41%. The crosslinked TPEI film cannot be dissolved in N-methyl-2-pyrrolidone (NMP), whereas it exhibits swelling behavior with a gel content of 82.25% when immersed in NMP (Fig. S3 and Table S1, Supporting Information). Additionally, the quality of the polymer films was examined using atomic force microscopy (AFM) and scanning electron microscopy (SEM) (Figs. S4, and S5, Supporting Information). The images reveal that polymer films are flat and free of apparent defects. As shown in Fig. 1b, the adjacent stacking of electron-donating diamine moieties and electron-accepting dianhydride moieties in traditional aromatic polyimide chains, known as mixed layer packing (MLP) structure, facilitates the formation of intermolecular charge transfer complexes (CTC) effect. In contrast, the electron-donating diamine moieties and electron-accepting dianhydride moieties in polyimide chains are stacked individually, forming a structure known as preferred layer packing (PLP), which results in minimal intermolecular charge transfer effects (Fig. 1c). This benzyl-induced crosslinking aims to interfere with the rearrangement of electron-donating diamine and electron-accepting dianhydride units, creating the PLP structure in polymer dielectrics, which thereby enhances their thermal stability and capacitive energy storage performance.
a Synthetic chemical structures of PEI and TPEI with benzyl-induced crosslinking. b A detailed schematic design of the PLP structure transformed from the MLP structure. c Schematic of electron transport within MLP and PLP structure.
To understand the formation of the PLP and MLP structure and their impacts on the electronic transitions in polymer films, we performed density functional theory (DFT) calculations to analyze the total energy variation and the electrostatic potential distribution among polymer chains. As shown in Fig. 2a, b1, in the PEI film, compared to the total energy of two molecular chains arranged in a PLP structure, the total energy of the two molecular chains arranged in a MLP structure reaches its lowest point at a distance of 5.25 Å. This indicates that the arrangement of PEI polymer chains primarily follows the MLP structure, which enhances electrostatic interactions among the polymer chains, resulting in a dramatic drop in energy storage performance. Conversely, there is no minimum total energy value for the arrangement of polymer chains in a PLP structure, indicating that the PLP structure is unstable. A benzyl-induced crosslinking method is utilized to create the PLP structure within polymer chains, significantly diminishing electrostatic interactions among them (Fig. 2c1–d1). Additionally, the local interchain spacing in the polymer chains is increased by leveraging the electrostatic repulsion at the same positions between molecular chains, leading to difficulty in the transport of charge carriers within the polymer. The molecular dynamics (MD) simulations reveal that TPEI exhibits a higher fractional free volume (FFV = 23.48%) and greater interchain distance (5.24 Å) compared to those (FFV = 6.03%; interchain distance = 4.15 Å) of PEI (Figs. S6 and 7).
a The total energy of the MLP and PLP of the PEI molecular chain as a function of scan distance (Inset: partial enlarged detail of energy scanning). b1/c1/d1 Schematic chemical structures and electrostatic potential distributions, and b2/c2/d2 using the interfragment CT (IFCT) method to evaluate amount of electron transfer between the donor (diamine) and acceptor (dianhydride) during charge-transfer (CT) excitation, and Chole-Cele distribution map of (b) PEI, (c) TPEI with one crosslink point, and d TPEI with two crosslink points. The marks of color bars indicate the difference in electrostatic potential between the two polymer chains.
The interfragment CT (IFCT) method in Multiwfn27 revealed that 0.92 electrons are transferred from the donor (diamine) to the acceptor (dianhydride) during charge-transfer (CT) excitation of PEI (Fig. 2b2, the electronic excitation analysis of polymer units is shown in the Supporting Information). Notably, in the PLP structure induced by benzyl-induced crosslinking, the transfer of electrons from the diamine to the dianhydride during CT excitation is significantly diminished, with the amount of charge transfer nearly approaching 0 electrons between the two cross-linked molecular chains (Fig. 2c2–d2). Moreover, the electron migration amount in TPEI containing two crosslinking points is significantly lower than that in TPEI with a crosslinking point. This difference arises from the enhanced face-to-face stacking of diamine units, which forms a more pronounced PLP structure. In contrast, TPEI with a crosslinking point exhibits partially misaligned stacking between diamine units, inducing a stacking more resembling MLP structure that weakens the suppression of electron migration (Fig. S8, Supporting Information). Moreover, the optical bandgap (2.9 eV) of TPEI is narrower than that (3.1 eV) of PEI (Fig. S9, Supporting Information). This result contrasts with conventional observations in dielectric polymers where a wider bandgap typically correlates with better electrical insulation26,28. This strongly supports the idea that PLP structure effectively restricts electronic transport between polymer dielectrics. Furthermore, the substituent methyl groups on the diamine benzene rings increase the dihedral angles at the imide ring position of TPEI, inducing twisting at the donor-acceptor junctions within the molecular chains and thereby diminishing the electrostatic interactions within the molecular chains11 (Figs. S10 and S11, Supporting Information). Based on the fluorescence emission spectra of PEI and TPEI, TPEI exhibits a significantly enhanced fluorescence signal with a blue-shifted peak compared to PEI, which proves that the benzyl-induced PLP structure simultaneously inhibits both interchain and intrachain charge transfer (Fig. S12, Supporting Information)29,30.
The chemical structure of PEI and TPEI films was analyzed using FT-IR spectroscopy, as shown in Fig. 3a. The characteristic peaks observed at wavenumbers of 1780 cm⁻¹, 1720 cm⁻¹, 1350 cm⁻¹, and 740 cm⁻¹ are attributed to the vibrational modes associated with the imide rings. Additionally, the absorption bands at 1850 cm⁻¹ and 1110 cm⁻¹ correspond to the vibrations of -CO-O-CO- and aliphatic -C-O-C- groups, respectively, indicating the presence of benzyl-induced crosslinked structure. Moreover, the results of X-ray photoelectron spectroscopy (XPS) for TPEI and uncross-linked TPEI (TPEIu) films also demonstrate the formation of aliphatic -C-O-C- groups at the crosslinked positions (Fig. S13, Supporting Information). The signals at 533.3 eV in the O1s spectrum and 286.6 eV in the C1s spectrum are assigned to the -C-O-C- groups, with a greater percentage observed in TPEI compared with TPEIu. The critical role of oxygen (O2) in the thermal-oxidative degradation and benzyl-induced crosslinking of polymers containing benzyl groups is confirmed by TGA (Fig. 3b)31,32,33. Compared with the TGA curve of the TPEIu measured in an inert atmosphere (N2), the TGA curve of the TPEIu tested in air shows a significant increase in residual weight within a temperature range of 280 °C–380 °C. This phenomenon further indicates the correlation of the thermos-oxidative crosslinking reaction with O2 and benzyl groups.
a FT-IR spectra, b TGA and DTG curves (Inset: partial enlarged detail of TGA), c DSC curves, (d) In-plane thermal expansion change curves (Inset: CET values) for PEI and TPEI films. Exploded view of the XRD pattern for (e) PEI film and f TPEI film. g The WAXS patterns for TPEIu and TPEI films. The 2D GIWAXS patterns of (h) TPEIu and i TPEI films.
The glass transition temperature (Tg) increases from 236.31 °C for PEI to 289.72 °C for TPEI, suggesting that the movement of polymer chains is restrained by benzyl-induced crosslinking networks (Fig. 3c). In addition, the variation in specific heat capacity (ΔCp) during the glass transition, caused by changes in the conformation of polymer chains, was obtained from DSC data. ΔCp is significantly reduced from 0.205 J g−1 °C−¹ for PEI to 0.119 J g−1 °C−¹ for TPEI, indicating that the polymer chains in the crosslinked films are more stretched compared to those in PEI, thereby minimizing voids and defects in the polymer films17,34. Moreover, the results of the in-plane thermal dimension change curves show a lower coefficient of thermal expansion (CTE) in TPEI films compared to that of PEI films in a specific temperature range (Fig. 3d). These results indicate that benzyl-induced crosslinking network effectively restricts the movement of polymer chains, thereby improving the thermal stability of the polymer films. Furthermore, the TGA curve of the TPEI film exhibits only 5% weight loss at 420 °C, further confirming its improved structural stability (Fig. S14, Supporting Information).
As shown in Fig. 3e, f, the exploded views of the XRD patterns of the films indicate the presence of ch-pack ordered domains observed in TPEI compared with PEI35. The ch-pack packing structure of polymer chains, similar to the PLP structure, originates from liquid-crystalline-like ordered domains. Hence, the PLP structure formed by benzyl-induced crosslinking predominates in TPEI film, indicating an efficient reduction in electrical conduction loss. In contrast, the main ordered packing structure in PEI is a π-stack formation, which represents the π-π stacking of imide and phenyl rings in ordered domains, thereby largely promoting charge carrier transport in polymers. Notably, the variation in interchain spacing of polymer chains was calculated from the XRD data of PEI and TPEI films, which is consistent with the theoretical charge results from DFT calculations and MD simulations (Fig. S15, Supporting Information). The WAXS patterns reveal significantly intensified and broadened diffraction peaks in TPEI compared to TPEIu, confirming the structural rearrangement into ch-pack packing structure (PLP structure) induced by benzyl crosslinking (Fig. 3g)36. Furthermore, the 2D GIWAXS patterns reveal that TPEI exhibits broad and isotropic amorphous halos, contrasting sharply with the poorly defined amorphous scattering features observed in TPEIu (Figs. 3h, i and S16 in the Supporting Information). This stark difference indicates substantial alterations in molecular packing and arrangement induced by benzyl crosslinking, resulting in a heterogeneous microstructure comprising coexisting ordered domains and amorphous regions in TPEI. Furthermore, Analysis of one-dimensional intensity versus scattering vector (I-q) profiles derived from the 2D GIWAXS data reveals two characteristic scattering peaks for TPEI. The scattering peak of TPEI at a scattering vector q of 1.01–1.35 Å−¹ is attributed to the ordered ch-pack stacking structure (PLP structure; Fig. S17). Additionally, the scattering peak of TPEI at q = 1.35–1.83 Å−¹ is ascribed to π-pack interaction between imide rings and benzene rings in adjacent polymer chains.
Mechanical strengthening and electrical insulation mechanisms
The high-field electrical insulation properties of polymeric materials at elevated temperatures are closely linked to the electromechanical breakdown process. As shown in Fig. S18, the thickness of polymer films under an external electric field (E) decreases due to the increasing electrostatic stress (\(=\frac{1}{2}{\varepsilon }_{0}{\varepsilon }_{r}{E}^{2}\), where \({\varepsilon }_{0}\) and \({\varepsilon }_{r}\) denote the dielectric permittivity of a vacuum and the dielectric constant of dielectrics, respectively), which is counterbalanced by the mechanical stress arising from elastic deformation (\(={Ye}\), where \(Y\) is the modulus and \(e\) represents the elastic strain of the dielectric)7,37,38. The increased breakdown strength of the polymer films can be attributed to their greater modulus. The storage modulus of TPEI (for example, 4.46 GPa at 150 °C, 3.97 GPa at 200 °C and 3.42 GPa at 250 °C) is greater than that of PEI (for example, 2.82 GPa at 150 °C, 2.46 GPa at 200 °C and 0 GPa at 250 °C), throughout a temperature range of 35 °C to 250 °C (Fig. 4a–c). Notably, TPEI exhibits a tensile strength of 80 MPa and an elongation at break of 3.4% (Fig. S19, Supporting Information).
DMA curves for (a) PEI film and b TPEI film. c Comparison of the storage modulus of PEI and TPEI films. d Leakage current density of polymer films as a function of electric field at 150 °C and e 200 °C. f Electrical conductivity of TPEI, PEI, FPE, Kapton, and PEI-PSU films at various temperatures. The conductivity values represent the average of multiple measurements.
Another advantage of the TPEI film is its excellent electrical insulation performance at high temperatures. According to the tested results of leakage current density of the polymer films (Figs. 4d, e and S20–S22 in the Supporting Information), TPEI exhibits over an order of magnitude lower leakage current density than that of PEI and other heat-resistant polymers at high electric fields and elevated temperatures. For instance, at 200 °C and 300 MV m−1, the leakage current density decreases from 1.19 × 10−5 A cm−2 for PEI to 4.46 × 10−7 A cm−2 for TPEI, indicating that the PLP structure in TPEI polymer can significantly suppress the conduction loss from the injected and excited charge carriers under extreme conditions. Simultaneously, the hopping distance (λ, derived from the hopping conduction model, detailed in the Supporting Information) decreases from 1.983 nm for PEI films to 1.17 nm for TPEI, indicating a higher charge trap density and a deeper charge trap depth in the TPEI film. Notably, at 250 °C, the leakage current density of TPEI is 2.35 × 10⁻⁶ A cm⁻² at 300 MV m−1 (Fig. S22, Supporting Information), which is significantly lower than that of existing heat-resistant polymers. Additionally, TPEI exhibits the lowest electrical conductivity over a temperature range from 50 °C to 250 °C (Fig. 4f), which is several orders of magnitude lower than that of other heat-resistant polymers, including PEI, FPE, Kapton, and copolymer of PEI with polysulfone (PEI-PSU). For instance, the electrical conductivity of TPEI is 4.77 × 10⁻¹³ S m−¹, whereas the value of the PEI-PSU is 5.43 × 10⁻9 S m⁻¹ at 250 °C.
Dielectric properties and capacitive energy storage performance
According to the equation (\({U}_{e}=\frac{1}{2}{\varepsilon }_{0}{{\varepsilon }_{r}E}_{b}^{2}\), where Eb denotes the breakdown strength of dielectrics), a high dielectric constant (\({\varepsilon }_{r}\)) and a high intrinsic Eb are crucial for achieving an enhanced discharged energy density (Ue) in a linear dielectric. The dielectric constant (\({\varepsilon }_{r}\)) and dielectric loss (tan δ) of PEI and TPEI were tested as functions of frequencies and temperatures (see Fig. 5a, b). A higher \({\varepsilon }_{r}\) of 3.85 is observed for TPEI, compared with 3.43 for PEI, at 1 kHz and room temperature. The enhancement of \({\varepsilon }_{r}\) in TPEI is likely due to the formation of flexible aliphatic ether linkages (-C-O-C-) among adjacent polymer chains through benzyl-induced crosslinking. This acts as “lubricating agents” to weaken dipole-dipole interactions, thereby improving the dipole polarization capability. Moreover, when the temperatures close to the Tg of PEI, the local motions of the polymer chains become more pronounced, leading to a significant increase in \({\varepsilon }_{r}\) and tan δ. In contrast, the dielectric properties of TPEI are highly stable within a wide temperature range from 30 °C to 250 °C.
a Dielectric frequency spectrum and b dielectric temperature spectrum of PEI and TPEI films. Weibull distribution of the characteristic breakdown strength of PEI and TPEI polymers at (c) 150 °C and d 200 °C. e Weibull distribution of characteristic breakdown strength for TPEI samples at 250 °C. f Breakdown strength of PEI and TPEI films at different temperatures.
Next, the breakdown strength (Eb) and the shape parameter (β) of PEI and TPEI at high temperatures were estimated using a two-parameter Weibull statistic method (details are shown in the Supporting Information). A higher value of β indicates better stability of the polymer dielectric. As shown in Fig. 5c, d, the breakdown strength (Eb) of TPEI is 734.27 MV m−1 at 150 °C and 661.87 MV m−1 at 200 °C, both of which surpass the reported values of most heat-resistant polymers. For instance, at 150 °C, the Eb of PEI is 513.22 MV m−1, while that of polytetrafluoroethylene (PTFE) is 396 MV m−1. At 200 °C, the values are 428.84 MV m−1 for PEI and 368 MV m−1 for PTFE. Notably, TPEI exhibits a higher Eb of 570.8 MV m−1 at 250 °C (Fig. 5e, f), surpassing that of existing dielectric polymers under the same conditions. For instance, the breakdown strength (Eb) of Al-1 PI and c-BCB is 541 MV m−1 and 262 MV m−1, respectively, at 250 °C10,26. Furthermore, a notable increase of the β is observed in TPEI, for example, with a value of 24.1 for TPEI compared with 10.98 for PEI at 200 °C. This suggests that TPEI shows improved dielectric reliability at elevated temperatures. These improvements can be attributed to the significantly enhanced storage modulus and reduced electrical conductivity resulting from the PLP structure formed by benzyl-induced crosslinking in the polymers.
The capacitive energy storage performance of benzyl-induced crosslinking polymers, with varying quantity of benzyl, and PEI was tested at 150 °C and 200 °C (Fig. S23–26, Supporting Information). It has been found that TPEI (with diamine monomers containing 4 benzyl groups) exhibits the best capacitive performance, for example, a discharged energy density (Ue) of 8.85 J cm−3 at 760 MV m−1 and 150 °C, as well as a Ue of 7.16 J cm−3 at 660 MV m−1 and 200 °C (Fig. 6a, b). Notably, Figs. 6a–e and S17–29 demonstrate that TPEI exhibits significant advantages compared with state-of-the-art commercial heat-resistant polymers such as PEI, Kapton, FPE, and PEI-PSU. Moreover, as shown in the comprehensive performance radar chart (Fig. 6f), TPEI outperforms PEI in multiple critical performance parameters, including a maximum discharged energy density (Uemax = 4.72 J cm−3), Ue at η above 90% (Ue90), Eb at 250 °C, as well as higher Tg and εr. Additionally, at η ≥ 90%, TPEI exhibits a Ue of 6.38 J cm−3 at 200 °C and 3.04 J cm−3 at 250 °C, surpassing that of most reported works (Fig. 6g, h). Moreover, TPEI can continuously undergo charge-discharge cycling for 100,000 cycles at 200 °C and 200 MV m−1 (Fig. S30, Supporting Information), indicating that it possesses excellent long-term stability in energy storage performance at high temperatures. Next, the fast discharge capability of TPEI at 200 °C and BOPP at 120 °C was measured (Fig. 6i). The results show that the discharge time (τ95, defined as the time needed to discharge 95% of the charged energy density) for TPEI is equivalent to that of BOPP at 120 °C. However, the power density of TPEI (0.16 MW cm−3 at 200 °C) is higher than that of BOPP (0.10 MW cm−3 at 120 °C) under the same electric field, suggesting a significantly enhanced discharge capability of TPEI films.
Discharge energy density and efficiency versus electric field of PEI and TPEI samples at (a) 150 °C and b 200 °C. Discharge energy density and efficiency versus electric field for TPEI and other polymers (Kapton, FPE, PEI-PSU) at (c) 150 °C and BOPP at 120 °C, (d) 200 °C, and e 250 °C. f Radar chart comparing the key properties of PEI and TPEI. gComparison of discharge energy density (efficiency > 90%) at 200 °C11,12,18,19,25,26,28,48,49,50,51,52,53,54,55,56,57,58 for the TPEI and all-organic polymer dielectrics. h Comparison of maximum discharge energy density at 250 °C11,26,54,58,59 for the TPEI and all-organic polymers. i Fast discharge testing of TPEI (200 °C) and BOPP (120 °C) at 200 MV m−1 and 20 kΩ.
This work offers an effective strategy for the synthesis of heat-resistant insulative dielectric polymers. The preferred layer packing (PLP) structure, formed by benzyl-induced crosslinking methods within intricately arranged polymer chains, significantly enhances the thermal stability and electrical insulation of polymer dielectrics. The lowest electrical conductivity of the designed homopolymer, in comparison with existing polymeric materials, is achieved at elevated temperatures. Therefore, an enhanced discharged energy density (η > 90%) of 6.38 J cm−3 (200 °C) and 3.04 J cm−3 (250 °C) is achieved in TPEI, which is higher than that of most reported works. This study presents a simple method for large-scale production of dielectric films to address the challenge of insufficient capacitive energy storage performance under extreme conditions in heat-resistant polymer dielectrics.
Methods
Materials
4,4′-(4,4′-isopropylidenediphenoxy) diphthalic anhydride (BPADA), 1,4-phenylenediamine (PDA), 2,5-diaminotoluene (DNT), 2,5-dimethyl-1,4-phenylenediamine (DPD), 2,3,5,6-tetramethyl-1,4-phenylenediamine (TPD) were purchased from Tokyo Chemical Industry. The pyromellitic dianhydride (PMDA) and 4,4′-oxydianiline (ODA) were purchased from Shanghai Aladdin Biochemical Technology Co., Ltd Among which, all exhibit a monomer purity of 98%. Copolymer of PEI with polysulfone (PEI-PSU) particles (Tg ≈ 265 °C), fluorene polyester (FPE) particles (Tg = 330 °C), and commercial biaxially oriented polypropylene (BOPP) films with a thickness of 5.8 μm were provided by Polyk Technologies Company. N-methyl-2-pyrrolidinone (NMP) of analytical reagent grade was obtained from Sinopharm Chemical Reagent Co., Ltd. All materials were used as received without further purification.
Synthesis of polymer films
A certain molarity of diamine monomer was dissolved in the NMP solution. Then, the same molar amount of dianhydride monomer was added into the mixed solution, and stirred at room temperature for 36 h to synthesize a viscous poly(amic acid) solution. The reaction solution was drop-cast on a clean glass slide and baked in a circulation oven at 85 °C for 2 h, 110 °C for 1 h, 160 °C for 1 h, and 220 °C for 1 h to remove the substantial solvent and form imide rings. Then, the glass slide was kept at 260 °C for 1 h, and then at 290 °C for 1.5 h to induce benzyl crosslinking and remove the residual solvent. The polymer films were peeled off from the glass slides in deionized water and dried in a vacuum oven at 110 °C for 24 h to remove the residual moisture. The polymer films named PEI, 1CH3-PEI, 2CH3-PEI, and TPEI are synthesized from BPADA and PDA, DNT, DPD, and TPD, respectively, following the preparation process outlined above. In addition, the uncross-linked TPEI (labeled TPEIu) was synthesized using a similar preparation method, with the exception of the heat treatment conditions, where a vacuum oven was employed. Kapton film, which has the same structure as commercial Kapton polyimide, is obtained through a reaction between PDMA and ODA. The curing reaction of Kapton films proceeded according to the following process: 80 °C/3 h, 100 °C/1 h, 150 °C/1 h, 200 °C/1 h, 250 °C/1 h, 300 °C/1 h, and 350 °C/2 h. PEI-PSU and FPE polymer films were prepared as comparison samples using the solution casting method. PEI-PSU particles and FPE particles were dissolved in NMP, stirring at 70 °C for 24 h. Next, the viscous polymer solution was cast and baked at 70 °C for 2 h, and then the sample films were dried in a vacuum oven at 200 °C for 24 h.
Characterization
1H nuclear magnetic resonance (NMR) spectra of the prepolymers were recorded using a JNM-ECZ600R spectrometer in DMSO-d6. The crosslinking degree of the films was determined by calculating the absorbance of the polymer solutions, which was recorded using a UV-vis spectrophotometer (UV-2600i, Island Ferry, Japan). The morphologies of the polymer films were observed by a scanning electron microscope (SEM, TESCAN) and an atomic force microscope (AFM, NanomanTM VS, USA) operated in tapping mode. X-ray photoelectron spectroscopy (XPS) analysis was carried out by a Thermo Fisher K-Alpha spectroscopy with an Al source. Solid-state UV-vis absorption spectra were recorded using a Shimadzu UV-3600 spectrophotometer. The steady-state/transient photoluminescence (PL) spectra of the polymer films were recorded using an Edinburgh FS5 spectrometer. Fourier transform infrared (FTIR) spectra of the samples were used to confirm their characteristic functional groups by a Nicolet iS50 FTIR spectrometer. X-ray diffraction (XRD) analysis was conducted with Cu radiation by a PANalytical Empyren diffractometer. The wide-angle X-ray scattering (WAXS) measurements were performed using a copper-target X-ray generator (Anton Paar) with operational parameters strictly controlled at ≤40 kV accelerating voltage and ≤50 mA tube current. Grazing incidence wide-angle X-ray scattering (GIWAXS) measurements were performed on a Rigaku Homelab system (Japan) equipped with a Cu Kα X-ray source (8.05 keV, λ = 1.54 Å) and a HyPix-6000 photon-counting detector. The sample-detector distance (SDD) was accurately calibrated using silicon standard sample data. The detector configuration included a pixel size of 100 μm × 100 μm, with an exposure time of 120 s per measurement. Thin films for GIWAXS analysis were prepared on a glass substrate by spin-coating. Thermogravimetric analysis (TGA) curves were obtained from a NETZSCH TGA209F1 thermogravimetry under nitrogen (air) atmosphere at a heating rate of 10 °C min−1, ranging from 30 °C to 800 °C. Differential scanning calorimetry (DSC) was performed on a TA Q10 DSC at a scan rate of 10 °C min−1, ranging from 30 °C to 300 °C in a nitrogen atmosphere. In DSC measurements, the glass transition temperature (Tg) of the polymer films was determined from the inflection point of the second heating curve after an initial heating-cooling cycle, a protocol designed to eliminate thermal history and enhance data reliability. The in-plane thermal expansion change curves of the films were recorded by a TMA Q400 from TA Instruments, with a force of 0.02 N, under nitrogen flow at a heating rate of 5 °C min−1, ranging from 30 °C to 350 °C. Dynamic mechanical analysis (DMA) of samples was conducted in tensile mode using a DMA 850 (TA Instruments) at a load frequency of 1 Hz, with a displacement amplitude of 20 μm, a temperature range from 50 to 350 °C, a heating rate of 5 °C min−¹, and under nitrogen flow. The stress-strain behavior of the crosslinked film was evaluated in tensile mode using a DMA 850 (TA Instruments, USA) with a preload force of 0.1 N and a displacement ramp rate of 0.05 mm min-1. Gold electrodes with diameters of 3 mm and 8 mm were coated onto both sides of the films using a GVC-2000 magnetron ion sputtering instrument for electrical testing under high electric field (D-E loop, breakdown strength, and leakage current) and low electric field (dielectric spectra), respectively. The dielectric frequency spectrum and dielectric temperature spectrum of the samples were measured using a precision impedance analyzer (Agilent 4294 A, Germany) and a DMS-500 dielectric temperature spectroscopy measurement system (DMS-500, China), respectively. The high-temperature-resistivity of the films was tested using a Tonghui TH2690A insulation resistance meter. The displacement-electric field hysteresis loop (D-E loop), breakdown strength, and cyclic charge-discharge performance of the samples were measured by a TF Analyzer 3000 series (AixACT, Germany) at 100 Hz. The leakage current density of sample films was measured using a TF Analyzer 3000 series (AixACT, Germany). A stepwise electric field increment mode was used, in which the electric field strength was increased by 10 MV m−1 per step (each step maintained for 2 s) until reaching the preset electric field strength. The fast charge-discharge tests were performed using a DCQ-20A measurement system (Polyk Technologies) with a load resistor (RL) of 10 kΩ.
DFT calculations
The ground-state geometry optimizations and energy scanning of the polymer structural units were performed by Gaussian 16 with the B3LYP hybrid function and 6–31 G (d) basis function. The structure construction and the visualization of electrostatic potential distribution of polymer units were conducted by GaussView 6.1 and Multiwfn 3.839,40,41. The model construction of polymer structural units for energy scanning was completed by dimerscan and xyz2QC42. The first 20 excited states of polymer structural units were calculated using time-dependent density functional theory (TDDFT) combined with the CAM-B3LYP/6-31 G functional43,44. The interfragment charge-transfer (IFCT) method was implemented using the Multiwfn 3.8 program to analyze the electronic excitation behavior of the polymer molecule (see the Supporting Information for detailed analysis)27,45,46.
Molecular dynamics (MD) simulation
Unit cells of PEI and TPEI were constructed using Materials Studio 20 software, both comprising 10 molecular chains with 20 repeat units per chain. Notably, 9 molecular chains in the TPEI system underwent crosslinking. Electrostatic interactions were handled by the Ewald summation technique, and van der Waals interactions were computed using an atom-based approach. A Nose-Hoover thermostat was used to control temperature, and pressure regulation was achieved through a Berendsen barostat. We use progressive relaxation and annealing methods to eliminate initial structural biases, enabling the system to reach thermodynamic equilibrium47. The fractional free volume (FFV) of polymer is determined by the equation:
Where \({V}_{c}\) corresponds to the unoccupied volume and \({V}_{o}\) represents the volume occupied by polymer chains.
Data availability
The data supporting the findings of this study are available in this article and its Supplementary Information files. Source data are provided at https://doi.org/10.6084/m9.figshare.29327069.v1. All data are available from the corresponding author upon request.
References
Sarjeant, W. J., Clelland, I. W. & Price, R. A. Capacitive components for power electronics. Proc. IEEE 89, 846–855 (2001).
Yang, L. et al. Perovskite lead-free dielectrics for energy storage applications. Prog. Mater. Sci. 102, 72–108 (2019).
Zhang, M. et al. Ultrahigh energy storage in high-entropy ceramic capacitors with polymorphic relaxor phase. Science 384, 185–189 (2024).
Chu, B. et al. A dielectric polymer with high electric energy density and fast discharge speed. Science 313, 334–336 (2006).
Luo, H. et al. Progress on polymer dielectrics for electrostatic capacitors application. Adv. Sci. 9, 2202438 (2022).
Watson, J. & Castro, G. High-temperature electronics pose design and reliability challenges. Analog Dialogue 46, 3–9 (2012).
Li, Q. et al. High-temperature dielectric materials for electrical energy storage. Ann. Rev. Mater. Res. 48, 219–243 (2018).
Wan, Y. et al. Ultrahigh energy density of scalable thermal-crosslinked polyetherimides at 250 °C. Adv. Funct. Mater. n/a, 2506635.
Zhou, Y. et al. A scalable, high-throughput, and environmentally benign approach to polymer dielectrics exhibiting significantly improved capacitive performance at high temperatures. Adv. Mater. 30, 1805672 (2018).
Li, Q. et al. Flexible high-temperature dielectric materials from polymer nanocomposites. Nature 523, 576–579 (2015).
Wang, R. et al. Designing tailored combinations of structural units in polymer dielectrics for high-temperature capacitive energy storage. Nat. Commun. 14, 2406 (2023).
Dong, J. et al. Scalable polyimide-organosilicate hybrid films for high-temperature capacitive energy storage. Adv. Mater. 35, 2211487 (2023).
Yan, C. et al. Improved capacitive energy storage at high temperature via constructing physical cross-link and electron–hole pairs based on p-type semiconductive polymer filler. Adv. Funct. Mater. 34, 2312238 (2023).
Yang, M. et al. Surface ion-activated polymer composite dielectrics for superior high-temperature capacitive energy storage. Energy Environ. Sci. 17, 1592–1602 (2024).
Yuan, C. et al. Polymer/molecular semiconductor all-organic composites for high-temperature dielectric energy storage. Nat. Commun. 11, 3919 (2020).
Dai, Z. et al. Scalable polyimide-poly(amic acid) copolymer based nanocomposites for high-temperature capacitive energy storage. Adv. Mater. 34, 2101976 (2021).
Yang, M. et al. Roll-to-roll fabricated polymer composites filled with subnanosheets exhibiting high energy density and cyclic stability at 200 °C. Nat. Energy 9, 143–153 (2024).
Deshmukh, A. A. et al. Flexible polyolefin dielectric by strategic design of organic modules for harsh condition electrification. Energy Environ. Sci. 15, 1307–1314 (2022).
Chen, J. et al. Ladderphane copolymers for high-temperature capacitive energy storage. Nature 615, 62–66 (2023).
Wu, C. et al. Rational design of all-organic flexible high-temperature polymer dielectrics. Matter 5, 2615–2623 (2022).
Chen, J. et al. Aromatic-free polymers based all-organic dielectrics with breakdown self-healing for high-temperature capacitive energy storage. Adv. Mater. 35, 2306562 (2023).
Pei, Z. et al. Wide bandgap heterostructured dielectric polymers by rapid photo-crosslinking for high-temperature capacitive energy storage. Adv. Funct. Mater. 34, 2307639 (2023).
Hasegawa, M. & Horie, K. Photophysics, hotochemistry, and optical properties of polyimides. Prog. Polym. Sci. 26, 259–335 (2001).
Zhuang, Y., Seong, J. G. & Lee, Y. M. Polyimides containing aliphatic/alicyclic segments in the main chains. Prog. Polym. Sci. 92, 35–88 (2019).
Song, J. et al. Alicyclic polyimides with large band gaps exhibit superior high-temperature capacitive energy storage. Mater. Horiz. 10, 2139–2148 (2023).
Ren, W. et al. Metallized stacked polymer film capacitors for high-temperature capacitive energy storage. Energy Storage Mater. 65, 103095 (2024).
Lu, T. & Chen, F. Multiwfn: a multifunctional wavefunction analyzer. J. Comput Chem. 33, 580–592 (2012).
Huang, W. et al. Alicyclic polyimide with multiple breakdown self-healing based on gas-condensation phase validation for high temperature capacitive energy storage. Adv. Mater. 36, 2410927 (2024).
Wang, R. et al. Dielectric polymers with mechanical bonds for high-temperature capacitive energy storage. Nat. Mater. https://doi.org/10.1038/s41563-025-02130-z (2025).
Chen, L. et al. Tuning second near-infrared fluorescence activation by regulating the excited-state charge transfer dynamics change ratio. J. Am. Chem. Soc. https://doi.org/10.1021/jacs.5c03763 (2025).
Song, Q. et al. Controlled thermal oxidative crosslinking of polymers of intrinsic microporosity towards tunable molecular sieve membranes. Nat. Commun. 5, 4813 (2014).
Zhu, S. et al. Benzyl-induced crosslinking of polymer membranes for highly selective CO2/CH4 separation with enhanced stability. Macromolecules 55, 6890–6900 (2022).
Wang, F. et al. Development of phenolphthalein-based copolyimides and their derivative cross-linked and thermally rearranged polymers for gas separation. Macromolecules 57, 1370–1382 (2024).
Yang, M., Zhou, L., Li, X., Ren, W. & Shen, Y. Polyimides physically crosslinked by aromatic molecules exhibit ultrahigh energy density at 200 °C. Adv. Mater. 35, 2302392 (2023).
Wakita, J., Jin, S., Shin, T. J., Ree, M. & Ando, S. Analysis of molecular aggregation structures of fully aromatic and semialiphatic polyimide films with synchrotron grazing incidence wide-angle X-ray scattering. Macromolecules 43, 1930–1941 (2010).
Bai, L. et al. Thermal expansion behavior of poly(amide-imide) films with ultrahigh tensile strength and ultralow CTE. Chin. J. Polym. Sci. 38, 748–758 (2019).
Stark, K. H. & Garton, C. G. Electric strength of irradiated polythene. Nature 176, 1225–1226 (1955).
Shi, Y. et al. A processable, high-performance dielectric elastomer and multilayering process. Science 377, 228–232 (2022).
Manzetti, S. & Lu, T. The geometry and electronic structure of Aristolochic acid: possible implications for a frozen resonance. J. Phys. Org. Chem. 26, 473–483 (2013).
Zhang, J. & Lu, T. Efficient evaluation of electrostatic potential with computerized optimized code. Phys. Chem. Chem. Phys. 23, 20323–20328 (2021).
Lu, T. & Manzetti, S. Wavefunction and reactivity study of benzo[a]pyrene diol epoxide and its enantiomeric forms. Struct. Chem. 25, 1521–1533 (2014).
Lu T. Molclus program, Version 1.12. http://www.keinsci.com/research/molclus.html (accessed 23, Aug. 2023).
Gross, E. K. U. & Kohn, W. Local density-functional theory of frequency-dependent linear response. Phys. Rev. Lett. 55, 2850–2852 (1985).
Yanai, T., Tew, D. P. & Handy, N. C. A new hybrid exchange–correlation functional using the Coulomb-attenuating method (CAM-B3LYP). Chem. Phys. Lett. 393, 51–57 (2004).
Liu, Z., Lu, T. & Chen, Q. An sp-hybridized all-carboatomic ring, cyclo[18]carbon: electronic structure, electronic spectrum, and optical nonlinearity. Carbon 165, 461–467 (2020).
Lu, T. A comprehensive electron wavefunction analysis toolbox for chemists, Multiwfn. J. Chem. Phys. 161, 082503 (2024).
Lei, H. et al. DFT and molecular dynamic simulation for the dielectric property analysis of polyimides. Chem. Phys. Lett. 786, 139131 (2022).
Dong, J. et al. Scalable high-permittivity polyimide copolymer with ultrahigh high-temperature capacitive performance enabled by molecular engineering. Adv. Energy Mater. 14, 2303732 (2023).
Liu, M. et al. Significant enhancement in dielectric properties of polyimide alloys through a two-phase interlocking structure. Adv. Funct. Mater. 34, 2313258 (2024).
Shukla, S. et al. Pendant group functionalization of cyclic olefin for high temperature and high-density energy storage. Adv Mater. 36, 2402133 (2024).
Pei, J. Y. et al. Flexible high-temperature polymer dielectrics induced by ultraviolet radiation for high efficient energy storage. Adv. Funct. Mater. 34, 2316869 (2024).
Xu, W. et al. Anisotropic semicrystalline homopolymer dielectrics for high-temperature capacitive energy storage. Angew. Chem. Int. Ed. 63, e202319766 (2024).
Zou, B. et al. Optimizing the charge transfer complex structure of polyimides with fluorinated side biphenyl for superior high-temperature capacitive performance. Adv. Funct. Mater. 2505254. https://doi.org/10.1002/adfm.202505254 (2025).
Xu, W. et al. Self-healing polymer dielectric exhibiting ultrahigh capacitive energy storage performance at 250 °C. Energy Environ. Sci. 17, 8866–8873 (2024).
Gurnani, R. et al. AI-assisted discovery of high-temperature dielectrics for energy storage. Nat. Commun. 15, 6107 (2024).
Qin, H. et al. Stereoisomerically enhanced polynorbornene-imide dielectric energy storage performance. Chem. Eng. J. 483, 149244 (2024).
Li, H. et al. Machine learning-accelerated discovery of heat-resistant polysulfates for electrostatic energy storage. Nat. Energy 10, 90–100 (2024).
He, G. et al. Enhanced high-temperature energy storage in semi-aromatic polyimides via dual regulation of short-range ordered and crosslinked architectures. Energy Environ. Sci. 18, 2405–2414 (2025).
Tian, Y. et al. Intrinsic-designed polyimide dielectric materials with large energy storage density and discharge efficiency at harsh ultra-high temperatures. Mater. Horiz. 10, 5835–5846 (2023).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (52172265 (H.L)), Excellent Youth Science Foundation of Hunan Province (2022JJ20067 (H.L)), The Science and Technology Innovation Program of Hunan Province (2022RC1074 (H.L)), Central South University Innovation-Driven Research Program (2023CXQD010 (H.L)), and the State Key Laboratory of Powder Metallurgy, Central South University, Changsha, China.
Author information
Authors and Affiliations
Contributions
Y.W. and H.L conceived the idea and designed the research. Y.W. carried out experiments, analyzed the data. Y.W., H.L., Z.Y., S.S., J.P., X.L., G.H., D.Z., and J.Z. analyzed the data and participated in helpful discussions. Y.W., H.L., D.Z., and J.Z. wrote the first draft of the manuscript, with input from all authors.
Corresponding authors
Ethics declarations
Competing interests
All authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Potao Sun and Peiyuan Zuo for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wan, Y., Luo, H., Yan, Z. et al. Decoupling thermal stability and insulation in dielectric polymers via donor-acceptor rearrangement. Nat Commun 16, 6242 (2025). https://doi.org/10.1038/s41467-025-61539-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-61539-x
This article is cited by
-
Correlating the dielectric properties with chain packing density of polar functionalities in hyperbranched polyimides
Science China Materials (2026)