Abstract
An inflammatory process may increase the risk of arrhythmias after transcatheter aortic valve replacement (TAVR). In this single-centre, double-blind, placebo-controlled, randomized trial we investigated the efficacy of colchicine to reduce a composite of new-onset atrial fibrillation or atrioventricular conduction disturbances requiring the implantation of a permanent pacemaker at 30 days after TAVR. Between September 21, 2021 and April 25, 2024, 120 patients with aortic stenosis undergoing TAVR (mean age 80.6 ± 5 years, 64% male) were randomly allocated to treatment with colchicine (n = 60) or placebo (n = 60). The trial was prematurely stopped due to a higher rate of stroke in the experimental group in a pre-specified interim analysis (5 [8.3%] versus 0 at maximum available follow-up, p = 0.022). In the intention-to-treat population, the primary endpoint occurred in 6 patients (10%) in the colchicine group and in 15 patients (25%) in the placebo group (risk-difference −15.0%, 95% CI −28.3 to −1.7, p = 0.031). The prespecified imaging endpoint, subclinical leaflet thrombosis, was detected in 13 of 48 patients (27%) in the colchicine group versus 26 of 48 patients (54%) in the placebo group (risk difference −27.1%. 95% CI −46.0% to −8.2%, p = 0.007). Here, we show that periprocedural treatment with colchicine may reduce the incidence of new-onset arrhythmias and subclinical leaflet thrombosis after TAVR. However, given the premature termination of the trial due to an unexpected increase in the stroke rate among patients treated with colchicine, confirmatory trials are warranted to corroborate the effect of anti-inflammatory treatment on the incidence of arrhythmias and subclinical leaflet thrombosis after TAVR. The trial was an investigator-initiated study supported by dedicated grants from the Bangerter-Rhyner Foundation and the Swiss Life Foundation. ClinicalTrials.gov Identifier: NCT04870424.
Similar content being viewed by others
Introduction
Transcatheter aortic valve replacement (TAVR) evolved into a safe and effective treatment alternative to surgical aortic valve replacement (SAVR) in elderly patients with symptomatic severe aortic stenosis1. Technical refinements and procedural optimization resulted in a reduction in peri-procedural adverse events and paved the way for the expansion of TAVR to low-risk patients2.
Rhythm disturbances after TAVR continue to occur in up to one-half of all patients and affect therapeutic management and long-term prognosis1,3,4,5. New-onset atrial fibrillation after TAVR results from a combination of inflammation, atrial oxidative stress, and increased sympathetic drive, and has been associated with an increased risk of stroke and death within 30 days4,6,7,8. Mechanical injury, tissue oedema, and localized inflammation are important mechanisms in the development of atrioventricular conduction disturbances after TAVR. If persisting, patients require the implantation of a permanent pacemaker, which has been associated with an increased risk of heart failure hospitalization and death9,10,11. Strategies to minimize the risk of atrioventricular conduction disturbances after TAVR in patients without pre-existing pacemaker encompass type of transcatheter heart valve, and implantation technique, respectively12,13,14. Randomized trials in patients after cardiac surgery indicated that colchicine is safe and effective in the prevention of atrial fibrillation after cardiac surgery15,16. The anti-inflammatory effects of colchicine may furthermore mitigate the risk of atrioventricular conduction disturbances, and reduce the rate of permanent pacemaker implantation after TAVR17.
Subclinical prosthetic leaflet thrombosis emerged from four-dimensional cardiac computed tomography angiography (4D-CCTA) studies in patients with valve replacement and garnered interest due to an arguable association with cerebrovascular events and premature structural valve deterioration18,19. Strategies to prevent subclinical leaflet thrombosis after TAVR have focused on the final common pathway and investigated different antithrombotic regimens. Randomized clinical trials in patients without indication for oral anticoagulation indicated a reduction of subclinical leaflet thrombosis with non-vitamin K antagonist anticoagulants compared to antiplatelet regimens, which, however, came at the expense of an increased risk of bleeding20,21,22. Colchicine has anti-inflammatory effects, inhibits platelet activation, and mitigates endothelial dysfunction23. The combined effect thus targets several upstream mechanisms involved in thrombus formation and may prevent subclinical leaflet thrombosis after TAVR.
In this work, we present the primary endpoint results of the Colchicine for Patients with Aortic Stenosis Undergoing Transcatheter Aortic Valve Replacement (Co-STAR) trial, which demonstrate that periprocedural treatment with colchicine may reduce the incidence of new-onset arrhythmias and subclinical leaflet thrombosis in patients with severe aortic stenosis undergoing TAVR.
Results
Patient population
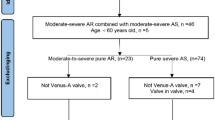
Between September 21, 2021 and April 25, 2024, a total of 381 patients were screened for participation in this study, 120 patients were enrolled and randomly assigned to treatment with colchicine (n = 60) or placebo (n = 60), and 117 patients underwent TAVR (Fig. 1). All patients started the study medication as randomly allocated; 53 participants (88%) in the colchicine group and 45 participants (75%) in the placebo group adhered to the study medication for at least 7 days (p = 0.097), and 36 (60%) and 37 (62%) completed the entire regimen (p = 1.0), respectively. The mean duration of continuous rhythm monitoring was 10.0 ± 2.7 days in the colchicine group and 8.5 ± 3.7 days in the placebo group (p = 0.011). The trial was prematurely terminated following the recommendation of the Data Safety Monitoring Board after review of the pre-specified interim analysis after complete follow-up of 120 patients over concerns of a higher rate of stroke in the colchicine compared to the placebo group at maximum available follow-up (5 [8.3%] versus 0; risk difference 8.2%, 95% CI 1.12 to 15.08, p = 0.022).
TAVR transcatheter aortic valve replacement.
The mean age of the study population was 80.6 ± 5 years and 64% of the participants were male. The mean Society of Thoracic Surgeons Predicted Risk of Mortality score was 2.5 ± 1.5%. There was no difference in the prevalence of pre-existing right bundle branch block. Two-thirds of patients were treated with balloon-expandable transcatheter heart valves and one-third with self-expanding transcatheter heart valves with equal distribution between groups. Extent of valve oversizing and need for post-dilation were well balanced between groups. Baseline characteristics and procedural characteristics are summarized in Table 1.
A total of 96 patients (48 in the colchicine group and 48 in the placebo group) underwent 4D-CCTA at a mean of 37 ± 7 days after TAVR (Supplemental Figure S1). Of these, forty-five patients in the colchicine group (94%) and 40 patients in the placebo group (83%) took the study medication for ≥7 days (p = 0.199). Baseline and procedural characteristics of patients who underwent 4D-CCTA at 30 days after TAVR can be found in Supplemental Tables S3/S5. There were no differences in treatment with Vitamin K (1[2%] versus 0, p = 1.0) and non-Vitamin K (5[10%] versus 1[2%], p = 0.204) oral anticoagulants between patients in the colchicine as compared to the placebo group, respectively (Supplemental Table S9).
Primary endpoint
Twenty-three primary endpoint events occurred within 30 days after TAVR. In the intention-to-treat analysis, the primary composite end point of new-onset atrial fibrillation or atrioventricular conduction disturbances requiring the implantation of a permanent pacemaker occurred in 6 patients (10%) in the colchicine group and in 15 patients (25%) in the placebo group (risk difference −15%, 95% CI −23.3 to −1.7, p = 0.031) at 30 days (Table 2, Fig. 2). In the as-treated analysis, the primary endpoint occurred in 6 patients in the colchicine group (11.1%) and in 15 patients (28.8%) in the placebo group (risk difference −17.7%, 95% CI −32.6% to −2.8%, p = 0.022). In a post-hoc stratified analysis of the primary endpoint across subgroups of sex, pre-existing complete right bundle branch block, transcatheter valve design, native valve calcification, and pre- and post-dilatation, we found no significant interaction with the treatment effect (Supplemental Figure S2).
Shown are the percentages of patients who had new-onset atrial fibrillation or atrioventricular conduction disturbance requiring the implantation of a permanent pacemaker in the colchicine group (n = 60 patients) and the placebo group (n = 60 patients) in the intention-to-treat analysis. The two-sided P value and the risk difference with 95% confidence intervals from the Mantel-Haenszel chi-square test for the comparison of the primary endpoint between the two treatment groups is shown. Data are presented as percentage of patients in which the primary endpoint occurred +/− the 95% confidence intervals (error bars). Source data are provided as a Source Data file. CI confidence interval.
Secondary endpoints
New-onset atrial fibrillation was recorded in 3 patients in the colchicine group (5.0%) and in 4 patients in the placebo group (6.7%) (risk difference −1.7%, 95% CI −10.1 to 6.7, p = 0.697). Conduction disturbances requiring the implantation of a permanent pacemaker occurred in 5 patients in the colchicine group (8.3%) and in 11 patients (18.3%) in the placebo group (risk difference −10.0%, 95% CI −22.0 to 2.0, p = 0.107). There were no differences in the occurrence of first-, second-, or third-degree atrioventricular block between groups. Nine patients in the placebo group and 2 patients in the colchicine group had a pacemaker rhythm in the electrocardiogram at 30 days (p = 0.029). Recorded rhythm disorders after TAVR are detailed in Table 2.
Prespecified imaging analysis
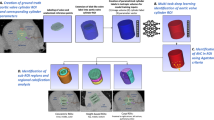
The incidence of at least one prosthetic leaflet with >50% motion reduction or thickening amounted to 41% at 30 days. At least one leaflet with >50% motion reduction or thickening was detected in 13 patients (27%) in the colchicine group versus 26 patients (54%) in the placebo group (risk difference −27.1%. 95% CI −46.0% to −8.2%, p = 0.007, Fig. 3). The mean transvalvular gradient was 11 ± 5 mmHg in the colchicine versus 12 ± 6 mmHg in the placebo group (p = 0.531). Four patients in the colchicine group (8%) and 3 patients in the placebo group (6%) had a transvalvular mean gradient ≥20 mmHg (p = 1.00). The findings in the as-treated cohort were consistent with the main analysis (Table 3, Figs. 3, 4, Supplemental Table S13). In a post-hoc stratified analysis across subgroups of sex, obesity, transcatheter valve size, left ventricular function, and oral anticoagulants, no significant interaction with the treatment effect was observed (Supplemental Figure S3).
Shown are the percentages of patients who had at least one prosthetic valve leaflet with >50% reduced motion or thickening in the colchicine group (n = 48 patients) and the placebo group (n = 48 patients) in the intention-to-treat analysis. The two-sided P-value and the risk difference with 95% confidence intervals from the Mantel-Haenszel chi-square test for the comparison between the two treatment groups is shown. Data are presented as percentage of patients in which the prespecified imaging endpoint occurred +/− the 95% confidence intervals (error bars). Source data are provided as a Source Data file. CI confidence interval.
Shown are examples of patients with normal leaflet morphology and function, leaflet thickening, and reduced leaflet motion. Created in BioRender. Ryffel, C. (2025) https://BioRender.com/0pgcy54.
Clinical events and safety outcomes
At 30 days, there were no differences in major adverse clinical events between groups (Tables 2, 3). There were 3 strokes (5.0%) in the colchicine group and none in the placebo group (risk difference 5.0%, 95% CI −1 to 11, p = 0.244). Safety outcomes such as kidney injury, bleeding, gastrointestinal side effects, and infections were comparable between patients in the colchicine and the placebo group. Thrombocytopenia was more common in patients in the placebo as compared to the colchicine group (49 [81.7%] versus 37 [61.7%], risk difference −20.0%, 95% CI −36 to −4, p = 0.025).
Discussion
In this double-blind, randomized, placebo-controlled trial investigating the efficacy of colchicine in reducing procedure-related arrhythmias in patients undergoing TAVR for severe aortic stenosis, colchicine was associated with a reduced rate of new-onset atrial fibrillation and conduction disturbances requiring the implantation of a permanent pacemaker at 30 days as compared to placebo. In the pre-specified imaging analysis, peri-procedural treatment with colchicine up to 14 days after TAVR was associated with a 50% reduction in the incidence of subclinical leaflet thrombosis compared to placebo at 30 days.
The etiopathology of TAVR-related arrhythmias is a complex interplay of local and systemic factors in which inflammatory processes are considered to be an important trigger. A systemic inflammatory response syndrome peaking between 24 and 48 h after TAVR has been documented in 39 to 56% of patients, and may be causal in the development of atrial fibrillation24,25,26. Inflammation generates atrial oxidative stress leading to electrical remodelling, affecting calcium homeostasis, modulating conduction barriers, and facilitating ectopic activity7. Moreover, systemic inflammation may promote atrial fibrillation by the activation of the sympathetic nervous system7,8. In contrast, the anatomical proximity of the device landing zone and the conduction system accounts for the development of atrioventricular conduction disturbances. Local inflammation and oedema contribute to direct mechanical trauma induced by the stent frame of the transcatheter heart valve and represent an important pathomechanism leading to permanent pacemaker implantation. A number of cohort studies indicated that atrioventricular conduction disturbances after TAVR are often temporary. Less than half of all patients are pacemaker-dependent during extended follow-up depending on valve design, balloon post-dilatation, and degree of atrioventricular block. A majority of patients show complete recovery of the conduction system within a couple of weeks27,28,29.
The inflammatory pathway of cardiac arrhythmias after TAVR offers a therapeutic target during peri-procedural management. Observational data yielded conflicting results on the effectiveness of anti-inflammatory treatment30,31. An exploratory study suggested that systemic corticosteroid exposure reduced the risk of permanent pacemaker implantation after TAVR30. The findings were however not corroborated in an analysis of 87 propensity-score matched pairs32. Colchicine is an anti-inflammatory drug that has been extensively studied in the prevention of post-operative atrial fibrillation in patients undergoing cardiac surgery15,16. In the double-blind randomized Colchicine for prevention of post-pericardiotomy syndrome (COPPS) study, administration of colchicine was associated with a 45% relative risk reduction of postoperative atrial fibrillation33. In contrast, no reduction in postoperative atrial fibrillation was found in the COPPS-2 study including 360 patients undergoing cardiac surgery at 11 centers34. Recent meta-analyses of randomized trials reported a consistent reduction of postoperative atrial fibrillation proving the effectiveness of colchicine in surgical patients15,16.
The Co-STAR trial built on the available evidence from the surgical literature and consistently showed a reduction in arrhythmias after TAVR both in the intention-to-treat and the as-treated populations. Numerical differences in the individual components of the primary endpoint did not reach statistical significance, and did not translate into differences in hard clinical endpoints. Factors known to be associated with the occurrence of atrioventricular block after TAVR such as pre-existing right bundle branch block, prosthetic valve design, oversizing, and post-dilatation were well balanced between the two treatment groups.
Subclinical leaflet thrombosis was detected in 41% of patients 30 days after TAVR. None of the study participants had clinically manifest valve thrombosis. The incidence of subclinical leaflet thrombosis was higher in the present analysis compared to previous studies ranging from 9 to 32%20,21,22,35. Timing of 4D-CCTA differed significantly between studies; the time interval from TAVR to 4D-CCTA was 3 to 6 months in the GALILEO 4D, ATLANTIS-4D-CT and ADAPT-TAVR trials compared to 1 month in the present study20,21,22,36,37. The recorded incidence of subclinical leaflet thrombosis in the Co-STAR trial is consistent with the reported incidence of 40% in the PORTICO IDE trial, in which CT scans were performed after a median of 32 days (IQR 28–37) after TAVR38. Subclinical leaflet thrombosis is closely linked to the antithrombotic regimen after TAVR. Patients with atrial fibrillation were not eligible for inclusion in the Co-STAR trial. Hence, only 7 patients (7%) were treated with oral anticoagulation at baseline. Two patients in the colchicine arm and 2 patients in the placebo arm were started on oral anticoagulation after detection of new-onset atrial fibrillation (p = 1.000) after TAVR. Conversely, a majority of patients in the present study were on single antiplatelet treatment. Previous studies compared different antithrombotic regimens to reduce the occurrence of subclinical leaflet thrombosis. In GALILEO 4D treatment with rivaroxaban in combination with aspirin reduced leaflet thickening and leaflet motion as compared to dual antiplatelet treatment at 3 months but was associated with a higher risk of death or thromboembolic events, and a higher risk of life-threatening, disabling, or major bleeding in the main trial39. In the ATLANTIS 4D-CT study, apixaban was associated with a lower risk of reduced valve leaflet motion or leaflet thickening with no differences in ischemic or bleeding events between groups22. In the ADAPT-TAVR trial, there was a trend toward a lower incidence of leaflet thrombosis in patients treated with edoxaban as compared to dual antiplatelet therapy with no difference in any bleeding events21. As a result, long-term OAC in patients without indication for OAC is currently not recommended40.
Histopathological analyses suggested inflammation and endothelial dysfunction as pathological mechanisms involved in thrombus formation41. At variance with antithrombotic regimens, colchicine may have an upstream effect on the incidence of leaflet thrombosis by modulating the inflammatory response, endothelial activation, and thrombus formation. Colchicine attenuates the inflammatory response by inhibiting the production of cytokines and interleukins, and affecting the function of neutrophils23. In addition, colchicine preserves endothelial integrity and inhibits the adhesion and aggregation of platelets23. The observed reduction of subclinical leaflet thrombosis with colchicine was consistent in an as-treated analysis and a sensitivity analysis excluding patients treated with oral anticoagulation. Several factors have been associated with the risk of subclinical leaflet thrombosis such as male sex, obesity, small annulus, and reduced left ventricular function42. All of these factors were well-balanced between treatment groups in the present study.
This study needs to be interpreted in light of several limitations. First, the trial included only 120 out of 200 anticipated participants and was prematurely terminated after crossing the stopping boundary for stroke in the pre-specified interim analysis. Two out of a total of 5 strokes occurred however well after completion of colchicine intake. Moreover, due to a play of chance, patients in the colchicine group more often had a history of prior stroke at baseline compared to patients in the placebo group (5 [8.3%] versus 0). Prior cerebrovascular events have consistently been reported to be associated with an increased risk of stroke after TAVR43. To the best of our knowledge, there is no evidence of an increased stroke risk with colchicine treatment. In contrary, a meta-analysis found that colchicine significantly reduces the risk of stroke in patients with coronary artery disease44. There was no difference in any of the other safety outcomes or reported medication side effects. As the study was prematurely stopped and the event rates of the primary outcome were lower than anticipated, the trial was underpowered, the observed treatment effect may be overestimated and chance findings cannot be excluded. Second, not all participants complied with study medication intake throughout the entire course of the trial. However, adherence to study drug intake was monitored by use of a patient diary and pill count. Furthermore, the findings in the as-treated population were consistent with the intention-to-treat analysis. Third, a difference in monitoring time after TAVR between the two treatment arms may have introduced surveillance bias. However, the monitoring duration was longer in the colchicine group as compared to the placebo group thus favoring underreporting of rhythm disturbances in the placebo group. In addition, follow-up was limited to 30 days. However, the majority of rhythm disturbances occurred within 3 days (83%) after TAVR, and only 1 event (4.3%) occurred beyond 10 days of follow-up. Also, since the study medication was discontinued 12 days after TAVR, the follow-up window would have allowed to record a potential catch-up phenomenon of arrhythmias occurring late after discontinuation of colchicine. Fourth, 4D-CCTA was performed at a single time point 30 days after TAVR. It is possible, that colchicine does not prevent but rather defer the occurrence of subclinical leaflet thrombosis. Serial imaging would be required to rule out a catch-up effect of thrombus formation. Fifth, only 31% of all patients assessed for eligibility could be enrolled into this trial (Fig. 1); the study population was highly selected by excluding patients with a history of atrial fibrillation, permanent pacemaker, and severe renal failure. Conversely, the study sample represents a low-surgical risk population (mean Society of Thoracic Surgeons Predicted Risk of Mortality score 2.5 ± 1.5%), which has recently come into focus with the expansion of TAVR to younger patients. And finally, the clinical relevance of subclinical leaflet thrombosis is a matter of ongoing debate. It remains to be determined whether differences in the incidence of leaflet thickening or reduced leaflet motion detected early after TAVR translate into differences in adverse clinical events.
In summary, in patients with severe aortic stenosis undergoing TAVR, treatment with colchicine was associated with a lower risk of new-onset atrial fibrillation or atrioventricular conduction disturbances requiring the implantation of a permanent pacemaker at 30 days. In addition, a pre-specified imaging analysis demonstrated a significant reduction in subclinical leaflet thrombosis in patients treated with colchicine as compared to placebo. The trial was however prematurely halted due to a higher rate of stroke in patients treated with colchicine. Confirmatory trials with colchicine or other inflammatory agents, i.e. monoclonal antibodies targeting interleukin1-β or interleukin-6 are warranted to corroborate the effect of anti-inflammatory treatment on the incidence of arrhythmias and subclinical leaflet thrombosis after TAVR.
Methods
Study design and participants
The Co-STAR study was an investigator-initiated, single-centre, double-blind randomized controlled trial investigating the efficacy of peri-procedural treatment with colchicine compared to placebo to reduce new-onset arrhythmias in patients with severe aortic stenosis undergoing TAVR. Patients ≥65 years of age undergoing transfemoral TAVR for symptomatic severe aortic stenosis defined by an aortic valve area (AVA) ≤ 1.0 cm2 or an AVA indexed to body surface area of <0.6 cm2/m2 were eligible for participation in this trial. Patients who already had a permanent pacemaker and patients with a history of atrial fibrillation were excluded from participation. A detailed list of inclusion and exclusion criteria is provided in the supplemental appendix. The study complied with the declaration of Helsinki and was approved by the Cantonal Ethics Committee in Bern, Switzerland (REC number 2021-00399). All participants provided written informed consent for participation in this trial before randomization. The study was registered with clinicaltrials.gov (NCT04870424). The full trial protocol is provided in the Supplementary Information.
Randomization and masking
Study participants were randomly allocated in a 1:1 ratio to treatment with colchicine or placebo by use of sequentially numbered boxes of study medication. Placebo and colchicine capsules were specifically produced for this trial by the Department of Pharmacy at Bern University Hospital, Switzerland, and were identical in appearance, colour, smell, and taste, as well as packaging and labelling. The allocation schedule was based on computer-generated block randomization (block size of 8) and stratified by the presence or absence of complete right bundle branch block. Study participants, treating physicians, outcome assessors, and data analysts were blinded to treatment allocation.
Investigational product and study procedures
Colchicine is an alkaloid interfering with the intracellular assembly of the inflammasome complex present in neutrophils and monocytes that mediates activation of interleukin-1β. The anti-inflammatory effect of colchicine peaks 24 to 48 h after administration based on intra-leukocyte accumulation45. Colchicine is indicated for the treatment of acute gout flares and familial Mediterranean fever, recommended for long-term management after acute coronary syndrome, and has been shown to reduce recurrent pericarditis, post-pericardiotomy syndrome, and atrial fibrillation in patients after cardiac surgery15,34,46,47.
Randomization took place the day before TAVR. Colchicine treatment was initiated with a loading dose of 1 mg at the day before TAVR and the day of the intervention, respectively, followed by a maintenance dose of 0.5 mg once daily for 12 days after TAVR. To ascertain adherence to the study drug, participants completed a diary to document pill intake and returned the boxes after the end of treatment for pill count. All patients were monitored during hospitalization and underwent continuous electrocardiogram (ECG) holter monitoring until 7 days post-discharge. Clinical follow-up was performed at 30 days including blood tests for complete blood count, hepatic and renal function, 12-lead electrocardiogram, transthoracic echocardiography, and 4D-CCTA.
Endpoints and definitions
The primary outcome was a composite of new-onset atrial fibrillation or atrioventricular conduction disturbances requiring the implantation of a permanent pacemaker as assessed 30 days after TAVR. The management of atrioventricular conduction disturbances post-TAVR and the indication for permanent pacemaker implantation followed a proposed algorithm outlined in an expert consensus statement48. All recorded rhythm disturbances were independently adjudicated by two electrophysiologists blinded to the participants and the treatment assignment. Discrepancies were resolved by joint review and mutual consensus.
The prespecified imaging endpoint of the study was the incidence of subclinical leaflet thrombosis at 30 days, defined as at least one prosthetic leaflet with >50% motion reduction or thickening as assessed by 4D-CCTA (Fig. 4). 4D-CCTA was performed using multiple detector CT and contrast-enhanced retrospective ECG gated throughout an entire cardiac cycle (i.e., 0-100% RR-interval). Images were reconstructed in 5% increments with a 0.75 mm slice thickness. The extent of leaflet thickening of the prosthetic valve leaflets, identifiable in at least two multiplanar reconstructed planes (double-oblique axial and sagittal oblique of the aortic valve) in 4D-CCTA, present in at least one valve. They were semi-quantitatively graded using a grading scale from “no leaflet thickening”, “<50% leaflet thickening”, and “>50% leaflet thickening”. Reduced systolic leaflet motion excursion was classified as unrestricted, “<50% reduced motion”, “>50% reduced motion” and “largely immobile”. Evaluations of 4D-CCTA scans were performed by two independent cardiovascular imaging specialists that were blinded to treatment allocation. In case of discrepancies, a third independent expert was consulted to reach a consensus.
Secondary endpoints included the individual components of the primary endpoint, the incidence of other arrhythmias or conduction disturbances, and major adverse cardiac events such as all-cause mortality, stroke, transient ischemic attack, bleeding, kidney injury, systemic embolism, myocardial infarction, infections, and clinical valve thromboses. Clinical events were adjudicated according to Valve Academic Research Consortium (VARC)−3 definitions49. Sex was determined based on self-reporting.
The primary endpoint and the imaging endpoint were assessed in the intention-to-treat population; confirmatory analyses were conducted in the as-treated population. The as-treated population was defined as all patients who complied with the study medication for at least 7 days or terminated intake earlier due to the occurrence of the primary endpoint. Complete intake of study medication from day −1 to day 5 after TAVR was deemed critical, as this is the most vulnerable period for the occurrence of rhythm disturbances.
All data were recorded by a dedicated research team in an electronic data capture system REDCap (version 15.0.11) held at the Clinical Trials Unit Bern (University of Bern, Switzerland)50.
Statistical analysis
The trial was designed as a superiority study for the primary endpoint at 30 days. Based on available evidence, we estimated an incidence of the primary endpoint in the control arm of 42% at 30 days3,51,52. Randomized trials in patients undergoing cardiac surgery indicated a relative risk reduction in the incidence of new-onset atrial fibrillation ranging between 20 and 50% in patients treated with colchicine as opposed to no treatment or placebo15. Assuming a relative risk reduction in the colchicine arm of 45%, we estimated a cumulative incidence of the primary endpoint in the colchicine arm of 23.1%. Enrolment of 200 patients was calculated to provide 80% power to establish superiority of colchicine compared to placebo at a two-tailed significance level of α = 0.05 and an attrition rate of 4%. An interim analysis was planned after the enrolment of 120 patients and reviewed by a Data Safety Monitoring Board. Pocock-type boundaries for premature termination of the trial were pre-specified in the study protocol for harm ( < 0.029 for mortality, stroke, or infection requiring prolonged index hospitalization or re-hospitalization), and Peto boundaries for benefit with respect to the primary endpoint. The interim analysis did not result in any changes to the statistical analysis plan (e.g., significance level of analyses).
The main measure of effect was the Mantel-Haenszel risk difference with 95% confidence intervals (CI) comparing colchicine versus placebo for the primary outcome at 30 days; the same procedure applied for binary outcomes including the components of the primary outcome, the prespecified imaging endpoint, and secondary outcomes at 30 days. Risk ratios with 95% CIs were also provided. For the pre-specified subgroup analyses, Mantel-Haenszel stratified by the subgroup factor was used and the approximate heterogeneity chi-square test reported to evaluate an interaction (modifying) effect on the risk difference comparing colchicine vs placebo. All other post-randomization comparisons of colchicine versus placebo were conducted using Fisher’s test (for 2 × 2 comparisons of counts), chi-square tests (for n x n comparisons not covered by Fisher’s test), and ANOVA F-tests (for continuous variables). Tables show counts (with % and sample sizes) and means with standard deviations (±, with sample sizes). All statistical analyses were performed by the Clinical Trials Unit Bern (University of Bern, Switzerland) using Stata version 18 (StataCorp, College Station, Texas).
Role of the funding source
The trial was an investigator-initiated study supported by a dedicated grant from the Bangerter-Rhyner Foundation and the Swiss Life Foundation. The funding source had no role in the design of the study, data collection, data monitoring, data analysis, data interpretation, or writing of the report. The first (CR) and last (TP) author had full access to all the data in the study and assumed final responsibility for the decision to submit for publication.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Source data are provided with this paper. Individual participant data that underline the results in this article are available to researchers who provide a methodologically sound proposal. Proposals should be directed to thomas.pilgrim@insel.ch. To gain access to the data, applicants will need to sign a data access agreement. Proposals may be submitted up to 36 months following article publication. Source data are provided with this paper.
References
Siontis, G. C. M. et al. Transcatheter aortic valve implantation vs. surgical aortic valve replacement for treatment of symptomatic severe aortic stenosis: an updated meta-analysis. Eur. Heart J. 40, 3143–3153 (2019).
Pilgrim, T., Siontis, G. C. M. & Windecker, S. Transcatheter aortic valve implantation: a blueprint for evidence-based evaluation of technological innovation. Eur. Heart J. 44, 853–855 (2023).
Nozica, N. et al. Assessment of New Onset Arrhythmias After Transcatheter Aortic Valve Implantation Using an Implantable Cardiac Monitor. Front. Cardiovasc. Med. 9, 876546 (2022).
Ryan, T. et al. New-Onset Atrial Fibrillation After Transcatheter Aortic Valve Replacement: A Systematic Review and Meta-Analysis. JACC Cardiovasc. Interv. 15, 603–613 (2022).
Bruno, F. et al. Predictors of pacemaker implantation after transcatheter aortic valve implantation according to kind of prosthesis and risk profile: a systematic review and contemporary meta-analysis. Eur. Heart J. Qual. Care Clin. Outcomes 7, 143–153 (2021).
Stähli, B. E. et al. Assessment of inflammatory response to transfemoral transcatheter aortic valve implantation compared to transapical and surgical procedures: a pilot study. J. Invasive Cardiol. 24, 407–411 (2012).
Tarantini, G., Mojoli, M., Urena, M. & Vahanian, A. Atrial fibrillation in patients undergoing transcatheter aortic valve implantation: epidemiology, timing, predictors, and outcome. Eur. Heart J. 38, 1285–1293 (2017).
Chen, P.-S. & Tan, A. Y. Autonomic nerve activity and atrial fibrillation. Heart Rhythm 4, S61–S64 (2007).
Mangieri, A. et al. TAVI and Post Procedural Cardiac Conduction Abnormalities. Front. Cardiovasc. Med. 5, 85 (2018).
Auffret, V. et al. Clinical effects of permanent pacemaker implantation after transcatheter aortic valve implantation: Insights from the nationwide FRANCE-TAVI registry. Arch. Cardiovasc. Dis. 117, 213–223 (2024).
Lee, M. Y. et al. Mechanisms of Heart Block after Transcatheter Aortic Valve Replacement – Cardiac Anatomy, Clinical Predictors and Mechanical Factors that Contribute to Permanent Pacemaker Implantation. Arrhythmia Electrophysiol. Rev. 04, 81 (2015).
Koniari, I. et al. Incidence, pathophysiology, predictive factors and prognostic implications of new onset atrial fibrillation following transcatheter aortic valve implantation. J. Geriatr. Cardiol. JGC 15, 50–54 (2018).
Wienemann, H. et al. Cusp overlap versus standard three-cusp technique for self-expanding Evolut transcatheter aortic valves. EuroIntervention 19, e176–e187 (2023).
Siontis, G. C. M. et al. Transcatheter aortic valve implantation vs. surgical aortic valve replacement for treatment of severe aortic stenosis: a meta-analysis of randomized trials. Eur. Heart J. 37, 3503–3512 (2016).
Lennerz, C., Barman, M., Tantawy, M., Sopher, M. & Whittaker, P. Colchicine for primary prevention of atrial fibrillation after open-heart surgery: Systematic review and meta-analysis. Int. J. Cardiol. 249, 127–137 (2017).
Salih, M. et al. Colchicine for prevention of post-cardiac procedure atrial fibrillation: Meta-analysis of randomized controlled trials. Int. J. Cardiol. 243, 258–262 (2017).
Gasparyan, A. Y., Ayvazyan, L., Yessirkepov, M. & Kitas, G. D. Colchicine as an anti-inflammatory and cardioprotective agent. Expert Opin. Drug Metab. Toxicol. 11, 1781–1794 (2015).
Bogyi, M. et al. Subclinical Leaflet Thrombosis After Transcatheter Aortic Valve Replacement: A Meta-Analysis. JACC Cardiovasc. Interv. 14, 2643–2656 (2021).
Hein, M. et al. Long-Term Follow-Up of Hypoattenuated Leaflet Thickening After Transcatheter Aortic Valve Replacement. JACC Cardiovasc. Interv. 15, 1113–1122 (2022).
De Backer, O. et al. Reduced Leaflet Motion after Transcatheter Aortic-Valve Replacement. N. Engl. J. Med. 382, 130–139 (2020).
Park, D.-W. et al. Edoxaban Versus Dual Antiplatelet Therapy for Leaflet Thrombosis and Cerebral Thromboembolism After TAVR: The ADAPT-TAVR Randomized Clinical Trial. Circulation 146, 466–479 (2022).
Montalescot, G. et al. Apixaban and Valve Thrombosis After Transcatheter Aortic Valve Replacement: The ATLANTIS-4D-CT Randomized Clinical Trial Substudy. JACC Cardiovasc. Interv. 15, 1794–1804 (2022).
Moras, E. et al. Role of Colchicine in Cardiovascular Disorders. Cardiol. Rev. (2024) https://doi.org/10.1097/CRD.0000000000000723.
Syryca, F. et al. Systemic inflammatory response syndrome in patients undergoing transcatheter aortic valve implantation. Cardiovasc. Revascularization Med. Mol. Interv. 62, 27–33 (2024).
Sinning, J.-M. et al. Systemic inflammatory response syndrome predicts increased mortality in patients after transcatheter aortic valve implantation. Eur. Heart J. 33, 1459–1468 (2012).
Schwietz, T. et al. Occurrence and prognostic impact of systemic inflammatory response syndrome in transfemoral and transapical aortic valve implantation with balloon- and self-expandable valves. EuroIntervention J. Eur. Collab. Work. Group Interv. Cardiol. Eur. Soc. Cardiol. 10, 1468–1473 (2015).
Kaplan, R. M. et al. Conduction recovery following pacemaker implantation after transcatheter aortic valve replacement. Pacing Clin. Electrophysiol. PACE 42, 146–152 (2019).
Raelson, C. A. et al. Recovery of atrioventricular conduction in patients with heart block after transcatheter aortic valve replacement. J. Cardiovasc. Electrophysiol. 28, 1196–1202 (2017).
Ghannam, M. et al. Atrioventricular conduction in patients undergoing pacemaker implant following self-expandable transcatheter aortic valve replacement. Pacing Clin. Electrophysiol. PACE 42, 980–988 (2019).
Oestreich, B. et al. Exposure to glucocorticoids prior to transcatheter aortic valve replacement is associated with reduced incidence of high-degree AV block and pacemaker. Cardiovasc. Revascularization Med. Mol. Interv. 20, 328–331 (2019).
Havakuk, O. et al. Steroid therapy and conduction disturbances after transcatheter aortic valve implantation. Cardiovasc. Ther. 34, 325–329 (2016).
Bernhard, B. et al. Systemic Corticosteroid Exposure and Atrioventricular Conductance Delays After Transcatheter Aortic Valve Implantation. Cardiovasc. Revascularization Med. Mol. Interv. 37, 1–6 (2022).
Imazio, M. et al. Colchicine reduces postoperative atrial fibrillation: results of the Colchicine for the Prevention of the Postpericardiotomy Syndrome (COPPS) atrial fibrillation substudy. Circulation 124, 2290–2295 (2011).
Imazio, M. et al. Colchicine for prevention of postpericardiotomy syndrome and postoperative atrial fibrillation: the COPPS-2 randomized clinical trial. JAMA 312, 1016–1023 (2014).
Chakravarty, T. et al. Subclinical leaflet thrombosis in surgical and transcatheter bioprosthetic aortic valves: an observational study. Lancet Lond. Engl. 389, 2383–2392 (2017).
Makkar, R. R. et al. Subclinical Leaflet Thrombosis in Transcatheter and Surgical Bioprosthetic Valves: PARTNER 3 Cardiac Computed Tomography Substudy. J. Am. Coll. Cardiol. 75, 3003–3015 (2020).
Blanke, P. et al. Bioprosthetic Aortic Valve Leaflet Thickening in the Evolut Low Risk Sub-Study. J. Am. Coll. Cardiol. 75, 2430–2442 (2020).
Makkar, R. R. et al. Possible Subclinical Leaflet Thrombosis in Bioprosthetic Aortic Valves. N. Engl. J. Med. 373, 2015–2024 (2015).
Dangas, G. D. et al. A Controlled Trial of Rivaroxaban after Transcatheter Aortic-Valve Replacement. N. Engl. J. Med. 382, 120–129 (2020).
Vahanian, A. et al. 2021 ESC/EACTS Guidelines for the management of valvular heart disease. Eur. Heart J. 43, 561–632 (2022).
Sellers, S. L. et al. Transcatheter Aortic Heart Valves: Histological Analysis Providing Insight to Leaflet Thickening and Structural Valve Degeneration. JACC Cardiovasc. Imaging 12, 135–145 (2019).
Giuliani, C. et al. Subclinical valve leaflet thrombosis following bioprosthetic aortic valve replacement. Curr. Opin. Cardiol. (2024) https://doi.org/10.1097/HCO.0000000000001161.
Vlastra, W. et al. Predictors, Incidence, and Outcomes of Patients Undergoing Transfemoral Transcatheter Aortic Valve Implantation Complicated by Stroke. Circ. Cardiovasc. Interv. 12, e007546 (2019).
Ma, Z., Chen, J., Jin, K. & Chen, X. Colchicine and coronary heart disease risks: A meta-analysis of randomized controlled clinical trials. Front. Cardiovasc. Med. 9, 947959 (2022).
Slobodnick, A., Shah, B., Pillinger, M. H. & Krasnokutsky, S. Colchicine: old and new. Am. J. Med. 128, 461–470 (2015).
Imazio, M. et al. Efficacy and safety of colchicine for treatment of multiple recurrences of pericarditis (CORP-2): a multicentre, double-blind, placebo-controlled, randomised trial. Lancet 383, 2232–2237 (2014).
Byrne, R. A. et al. 2023 ESC Guidelines for the management of acute coronary syndromes. Eur. Heart J. 44, 3720–3826 (2023).
Rodés-Cabau, J. et al. Management of Conduction Disturbances Associated With Transcatheter Aortic Valve Replacement: JACC Scientific Expert Panel. J. Am. Coll. Cardiol. 74, 1086–1106 (2019).
VARC-3 WRITING COMMITTEE et al. Valve Academic Research Consortium 3: Updated Endpoint Definitions for Aortic Valve Clinical Research. J. Am. Coll. Cardiol. 77, 2717–2746 (2021).
Harris, P. A. et al. The REDCap consortium: Building an international community of software platform partners. J. Biomed. Inform. 95, 103208 (2019).
Noble, S. et al. Comparison of procedural and clinical outcomes with Evolut R versus Medtronic CoreValve: a Swiss TAVI registry analysis. EuroIntervention J. Eur. Collab. Work. Group Interv. Cardiol. Eur. Soc. Cardiol. 12, e2170–e2176 (2017).
Lanz, J. et al. Safety and efficacy of a self-expanding versus a balloon-expandable bioprosthesis for transcatheter aortic valve replacement in patients with symptomatic severe aortic stenosis: a randomised non-inferiority trial. Lancet Lond. Engl. 394, 1619–1628 (2019).
Acknowledgements
The study has been supported by dedicated grants from the Bangerter-Rhyner Foundation (received by C.R., T.P. and J.L.) and the Swiss Life Foundation (received by C.R., T.P. and J.L.). We would like to thank Dr. Laura Morf, Dr. André Frenk, Dr. Kathrin Zürcher and Ivana Ummel for the organizational and administrative support for this study.
Author information
Authors and Affiliations
Contributions
All authors made significant contributions to this research, fulfilling the criteria for authorship. C.R., J.L., S.W. and T.P. designed the study. N.G., D.R. and D.S. recruited participants. M.B., A.A.P., D.T. and C.G. performed imaging analyses. M.F. and S.S. performed follow-up visits. D.H. performed statistical analyses. N.G. and C.R. were responsible for data collection. T.R. were responsible for rhythm-monitoring analyses. T.P. supervised the study. T.P. and C.R prepared the first draft of the manuscript. All authors reviewed, provided feedback on, and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
Jonas Lanz reports speaker fees from Edwards Lifesciences and Abbott to the institution. Daryoush Samim has received funding for an online course from Edwards Lifesciences. Stefan Stortecky reports research grants to the institution from Edwards Lifesciences, Medtronic, Abbott, Boston Scientific, and consultancy fees from BTG / Boston Scientific, and Teleflex outside the submitted work. Dik Heg is employed by the DCR, University of Bern, which has a staff policy of not accepting honoraria or consultancy fees. However, DCR is involved in design, conduct, or analysis of clinical studies funded by not-for-profit and for-profit organizations. In particular, pharmaceutical and medical device companies provide direct funding to some of these studies. For an up-to-date list of our conflicts of interest see https://www.ctu.unibe.ch/research_projects/declaration_of_interest/index_eng.html. David Reineke reports travel expenses from Abbott, Edwards Lifesciences and Medtronic and has proctor and consulting contracts with Abbott and Medtronic. Tobias Reichlin: Research grants from the Swiss National Science Foundation, the Swiss Heart Foundation, the sitem insel support funds, Biotronik, Boston Scientific and Medtronic, all for work outside the submitted study. Speaker/consulting honoraria or travel support from Abbott/SJM, Bayer, Biosense-Webster, Biotronik, Boston-Scientific, Farapulse, Medtronic, Pfizer-BMS, all for work outside the submitted study. Support for his institution’s fellowship program from Abbott/SJM, Biosense-Webster, Biotronik, Boston-Scientific and Medtronic for work outside the submitted study. Christoph Gräni has received funding support from the Swiss National Science Foundation, InnoSuisse, the CAIM foundation, the GAMBIT foundation, and the Novartis Biomedical Research Foundation. Stephan Windecker reports research, travel or educational grants to the institution from Abbott, Abiomed, Amgen, Astra Zeneca, Bayer, Bbraun, Biotronik, Boehringer Ingelheim, Boston Scientific, Bristol Myers Squibb, Cardinal Health, CardioValve, Cleerly Inc., Cordis Medical, Corflow Therapeutics, CSL Behring, Daiichi Sankyo, Edwards Lifesciences, Farapulse Inc. Fumedica, GE Medical Systems, Gebro Pharma, Guerbet, Idorsia, Inari Medical, InfraRedx, Janssen-Cilag, Johnson & Johnson, Medalliance, Medicure, Medtronic, Merck Sharp & Dohm, Miracor Medical, Neucomed, Novartis, Novo Nordisk, Organon, OrPha Suisse, Pharming Tech, Pfizer, Philips AG, Polares, Regeneron, Sanofi-Aventis, Servier, Siemens Healthcare, Sinomed, SMT Sahajanand Medical Technologies, Terumo, Vifor, V-Wave, Zoll Medical. Stephan Windecker served as advisory board member and/or member of the steering/executive group of trials funded by Abbott, Abiomed, Amgen, Astra Zeneca, Bayer, Boston Scientific, Biotronik, Bristol Myers Squibb, Edwards Lifesciences, MedAlliance, Medtronic, Novartis, Polares, Recardio, Sinomed, Terumo, and V-Wave with payments to the institution but no personal payments. He is also member of the steering/executive committee group of several investigator-initiated trials that receive funding by industry without impact on his personal remuneration. Thomas Pilgrim reports research grants from the Swiss National Science Foundation, the Swiss Heart Foundation, the Swiss Polar Institute, the Bangerter-Rhyner Foundation, the Mach-Gaensslen Foundation, and the Monsol Foundation. Research, travel or educational grants to the institution without personal remuneration from Biotronik, Boston Scientific, Edwards Lifesciences, and ATSens; speaker fees and consultancy fees to the institution from Biotronik, Boston Scientific, Edwards Lifesciences, Abbott, Medtronic, Biosensors, and Highlife. All other authors have reported that they have no relationships relevant to the contents of this paper to disclose.
Peer review
Peer review information
Nature Communications thanks Tao Chen, and the other, anonymous, reviewer for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ryffel, C., Lanz, J., Guntli, N. et al. Colchicine in patients with aortic stenosis undergoing transcatheter aortic valve replacement: a double-blind randomized trial. Nat Commun 16, 6501 (2025). https://doi.org/10.1038/s41467-025-61916-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-61916-6