Abstract
Quantifying climate sensitivity is essential for future climate projections, yet it varies with major Earth system changes. We present a glacial CO₂ reconstruction using paleosols from the Chinese Loess Plateau, covering 2580 to 800 thousand years ago. A stepwise decline in glacial CO₂ levels from ~300 ppm to <200 ppm is observed. Our paleosol-based CO₂ estimates support the key role of atmospheric CO₂ in driving major climate transitions during the Pleistocene, such as the long-term global cooling and the amplification of the glacial cycles. Based on compiled glacial and interglacial CO2 records, Earth system sensitivity, defined as the global temperature change for a doubling of CO2 once the whole Earth system has reached equilibrium, is estimated to be ~6.2–7.4 K (3.2–12.0 K, 95% confidence). Equilibrium climate sensitivity, after accounting for the different efficacy between ice-sheet and CO2 forcing and other slow feedbacks, is estimated to be 3.3 K (2.1–6.3 K, 95% confidence) and 3.7 K (1.7–6.3 K, 95% confidence), respectively. The lack of a significant difference between these values suggests no apparent state-dependency of climate sensitivity between glacial and interglacial climate states.
Similar content being viewed by others
Introduction
Equilibrium climate sensitivity (ECS) refers to the global mean surface temperature response to changes in radiative forcing caused by a doubling of atmospheric CO2 concentration when the responses to fast feedbacks reach equilibrium1. ECS is a useful measure of the impacts of greenhouse gas emissions on global temperatures and provides a critical reference for future policymaking regarding climate mitigation2. Paleoclimate studies offer a “deep-time” perspective on ECS as they capture long-term Earth system responses under large ranges of CO2 forcing, which helps evaluate the feedback processes and improve climate models3. The Pleistocene epoch experienced long-term cooling marked by the expansion of bipolar ice sheets4 and the amplification of glacial cycles from 400 ky during 2580–1250 thousand years ago (ka) (the “40k world”) to 100 ky since 700 ka (the “100k world”)5,6, with this climatic transition often referred to as the Mid-Pleistocene Transition (MPT)6. As such, the Pleistocene represents a natural experiment from which empirical estimates of ECS can be derived7,8. However, studies of the early Pleistocene ECS are rare, with high uncertainty8, which largely results from the lack of high-resolution, high-fidelity CO2 records.
Early Pleistocene CO2 levels are mainly estimated by proxies from a variety of geological archives9,10,11. Recent work on the Antarctic blue ice managed to extend direct CO2 measurements to ~3000 ka12,13 (Fig. 1b). Nonetheless, the blue-ice data provide limited snapshots of the early Pleistocene, which may fail to capture overall CO2 variations12,13. The δ11B values of planktic foraminifera and δ13C values of algal biomarkers such as alkenones provide extensive Pleistocene CO2 estimates, yet none of the existing CO2 records cover the entire Pleistocene with ample resolution9,14,15,16,17. Moreover, due to various limitations related to each proxy (e.g., biological “vital” effects and post-burial diagenesis), it is necessary to reconstruct paleo-CO2 from a multi-proxy perspective18. Notably, since ~2000 ka, the available CO2 data from blue-ice measurements12 and proxy-based estimates (δ11B and paleosols)9,10,17 show a constant maximum CO2 of 280–300 ppm (Fig. 1b and Supplementary Fig. S1), in contrast to the long-term cooling as suggested by marine benthic δ18O19,20 (Fig. 1a). Our previous work based on the paleosols from the Chinese Loess Plateau also suggests CO2 was nearly constant among Pleistocene interglacials10. Although a long-term glacial CO2 record is absent, studies on the MPT suggest that glacial CO2 dropped by 24–38 ppm from the 40 k to the 100 k world, which might have played a key role in the long-term global cooling9,12,17,21, a supposition supported by recent conceptual modeling, which calculated changes in global ice-sheet volume in response to different forcing scenarios22. This has motivated us to establish a Pleistocene glacial CO2 record, aiming to elucidate the relationship between long-term CO2 evolution and changes in the climate system.
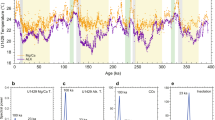
a Benthic-δ18O stack19. Shaded areas correspond to glacial periods. b Ice-core CO2 record over the past 800 ky78 and early Pleistocene blue-ice CO2 snapshots (circles)12. c Glacial paleosol-based CO2 estimates from this study, compared with the ice-core record. Error bars denote 1σ errors from Monte Carlo simulations. Blue curve denotes the LOESS smoothed curve with a span of 0.5 (50% of all the data that are nearest to a given age were included in the local regression fit). Horizontal dashed lines highlight the maximum and minimum CO2 from the Antarctic ice.
The paleosol-CO2 proxy is a key method for reconstructing paleo-CO2 from the terrestrial perspective23. The carbon isotope composition (δ13C) of pedogenic carbonate records the δ13C of soil CO2 that consists of two endmembers with distinct δ13C signals—CO2 from soil respiration and atmospheric CO224. Pedogenic carbonate thus documents the concentration of atmospheric CO2, provided that several input parameters can be constrained25,26 (Materials and Methods). However, soil-respired CO2 concentration at soil depth z of pedogenic carbonate formation (S(z)), a key variable that underpins this methodology, remains poorly constrained and introduces considerable uncertainty to the paleosol-CO2 proxy26. Moreover, due to the lack of continuous occurrence of paleosols in sediment strata as well as precise dating methods, the paleosol-CO2 method also suffers from low time resolution.
In this study, we leverage the continuous, well-dated paleosols from the Chinese Loess Plateau (CLP), where eolian deposits experienced soil development under the influence of the East Asian summer monsoon27. The Quaternary deposits on the CLP, known as the loess-paleosol sequences, are characterized by alternating light yellow ′loess′ units (Entisols, poorly developed paleosols) and dark brown ′paleosol′ units (Aridisols/Mollisols, stronger pedogenesis) formed during glacial and interglacial episodes, respectively. The chronological framework of the loess-paleosol sequences is built on magnetostratigraphy and other direct dating methods27, enabling widespread comparison with other high-resolution archives such as marine sediments and ice cores28. We reconstructed CO2 levels during glacial periods (as identified by the deposition of weakly pedogenically modified loess, which have been correlated with Marine Isotope Stage glacials) of the entire early Pleistocene (2580–800 ka), using paleosols collected from two sections, Fuxian and Zhaojiachuan, situated in the central CLP (Materials and Methods, Supplementary Fig. S2). Owing to the nearly continuous eolian deposition and seasonal wet-dry cycles, pedogenic carbonates grew throughout the CLP eolian deposits, making them excellent materials for continuous CO2 reconstruction.
Our previous work reconstructed interglacial CO2 levels using finely disseminated calcites from the paleosol units10. In this study, we overcame several principal uncertainties (i.e., detrital contamination, ages, magnitude of S(z)) that are particularly relevant to weakly developed paleosols within CLP loess units to reconstruct glacial CO2 levels. The newly reported glacial CO2 was used in tandem with published CO2 records and various global climate records to assess climate sensitivity across the Pleistocene epoch.
Results and discussion
Pedogenic carbonates documenting glacial CO2 levels
Before using soil carbonates for glacial-CO2 reconstructions, two prerequisites need to be met: 1) ensuring that these carbonates were formed during pedogenesis and are free of detrital (originating from soil parent materials) carbonates, and 2) confirming the absence of significant illuviation process, which would cause substantial offsets between the depositional ages of loess and the formation ages of pedogenic carbonates.
The parent material of the CLP paleosols, composed of eolian dust from inland Asia, contains detrital carbonates such as dolomite and calcite29, the δ13C values of which are distinct from pedogenic carbonates30. During interglacial periods, strong pedogenesis dissolved all the detrital carbonates in some paleosol units30. During glacial periods, pedogenesis was weaker and therefore all the loess units contain certain amounts of detrital carbonates30. To ensure our glacial period carbonate samples are pedogenic and not detrital, we isolated carbonate in the clay-sized fractions (<2 μm) from bulk paleosols (Materials and Methods). Scanning electron microscopy imaging confirms that the clay-sized fractions are predominantly nanoscale needle fiber calcite (NFC, Supplementary Fig. S3)–a typical form of pedogenic carbonate31. Recent efforts have focused on understanding the formation and geochemistry of NFC in these paleosols, as well as utilizing them for paleoclimate reconstructions10,32. Radiocarbon ages of clay-sized carbonates formed during the last glacial period closely align with those of coeval soil organic matter and are younger than the depositional ages of the loess from corresponding stratigraphic levels, as expected for pedogenic materials (Materials and Methods, Supplementary Fig. S4), suggesting minimal contribution from radiocarbon-dead detrital carbonates.
During interglacial episodes, increased rainfall promotes intensive leaching of carbonate minerals and deep carbonate accumulation that could penetrate the underlying glacial units10. Therefore, the age determination of pedogenic carbonates in the upper part of the glacial units becomes complicated. To circumvent this issue, our samples were taken exclusively from the lower sections of the glacial (loess) units. Because our carbonate samples are dominated by NFC that grows close to the soil-air interface33, it is unlikely that they were formed during interglacial periods. Moreover, the radiocarbon ages of these clay-sized carbonates from the top of the L1 glacial unit, where the accumulation of younger carbonates during the Holocene is likely most problematic, indicate they are at most 10-ky younger than the depositional ages of the loess in which they reside (Supplementary Fig. S4), suggesting that the clay-sized carbonates lower in the glacial units (and therefore presumably older) were formed during glacial periods. Taken together, all evidence supports our carbonate samples in the clay-sized fractions as valuable archives for documenting glacial paleoenvironments, including atmospheric CO2 levels. Notably, since we specifically sampled the lower part of the glacial units, our records are likely biased towards the beginning of glacials. Nonetheless, because early Pleistocene glacial cycles do not have the “saw-tooth” pattern evident in late Pleistocene, sampling the bottom half of the glacial units should provide a good estimate of mean glacial conditions.
δ18Oc as a proxy for glacial S(z)
The accuracy and precision of the paleosol-CO2 method hinge on the constraints on S(z)26. Determining S(z) in paleosols has been challenging due to its high seasonal and spatial variations34, which introduces considerable uncertainty to the resulting CO2 estimates26. In our previous work on interglacial CO2, S(z) was estimated through soil magnetic susceptibility, as both parameters respond to monsoonal rainfall10. However, this method loses its sensitivity in less-weathered glacial loess units due to a relatively high rainfall threshold for magnetite growth and increased interference with detrital magnetite from the parent material35.
We therefore investigated the δ18O values of the clay-sized pedogenic carbonates (δ18Oc) as a proxy for glacial S(z). A negative relationship between δ18Oc and S(z) is expected from a process perspective because they are influenced by similar factors. Soil water δ18O values (the principal control on δ18Oc) inherit rainfall δ18O values, which in monsoon climates such as the one studied here, tend to decrease as rainfall amount increases36. Conversely, S(z) values increase with rainfall amount because they increase with soil respiration rate and the depth of carbonate accumulation, both of which increase with rainfall37,38. Thus, S(z) should increase as δ18Oc decreases. Moreover, we expect that this negative relationship is accentuated by gas transport affecting both evaporation and the escape of respired CO2 from soils. Evaporation and respired CO2 escape from soil by net diffusion from the soil pore spaces to the atmosphere. When pathways for gas diffusion are restricted, the gases are retained in the soil, minimizing evaporation and elevating S(z). Because evaporation increases the δ18O value of residual soil water, the negative relationship should be enhanced by this factor. Taken together, the primary factors (rainfall, carbonate accumulation depth, and soil gas transport) affecting both δ18Oc and S(z) should be captured by the inverse δ18Oc-S(z) relationship (Supplementary Fig. S5). δ18Oc is thus expected to serve as a reasonable empirical proxy for S(z).
To examine the relationship between δ18Oc and S(z), we back-calculated S(z) using the clay-sized carbonate fraction of the glacial paleosol samples spanning the last 800 ky (Eq. 3 in Materials and Methods), during which CO2 levels are known independently from the Antarctic ice cores. To propagate uncertainties from all the inputs (i.e., S(z), δ13C of atmospheric CO2, soil-respired CO2, pedogenic carbonate, and carbonate formation temperature), we adopted a Monte Carlo random sampling approach similar to the PBUQ code26, which generates S(z) distributions through 10,000 iterations. During each run, input variables were randomly chosen from their normal distributions based on the mean and uncertainty of each parameter. The medians, 14th and 84th percentiles of the resulting S(z) distributions were used to represent the best estimates and the 1σ error.
Glacial S(z) estimates over the past 800 ky range between 224–542 ppm with a mean 1σ of ±86 ppm. Glacial S(z) levels are generally lower than the corresponding interglacial S(z) levels (356–815 ppm)10, in line with the overall colder and drier climate and lower soil productivity during glacial than interglacial episodes27. The S(z) range is also consistent with modern S(z) measurements (572 ± 273 ppm) on the CLP during the warm, dry periods when pedogenic carbonates likely grow39. Then, an exponential correlation between δ18Oc and S(z) was identified (Supplementary Fig. S6):
We next applied a bootstrap resampling technique to our samples spanning the last 800 ky, which confirmed the robustness of the empirical equation in predicting glacial S(z) (Materials and Methods).
Notably, the Zhaojiachuan data do not significantly overlap with the Fuxian data (Supplementary Fig. S6). Such a difference is anticipated as modern observations suggest lower rainfall and higher aridity in Zhaojiachuan than Fuxian, from which we would expect lower S(z) and higher δ18Oc40. However, considering both δ18Oc and S(z) are primarily controlled by rainfall amount across the CLP10,40, and given the close proximity (<150 km) of the two sections (Supplementary Fig. S2), it is unlikely that their δ18Oc—S(z) relationships differ significantly. We also calculated S(z) using δ18Oc—S(z) empirical equations derived from individual sections, and the resulting CO2 estimates are within the 1σ error of those obtained from the uniform equation (Supplementary Fig. S7).
In addition, processes affecting the δ18Oc (e.g., moisture source, soil water evaporation)40 and S(z) (e.g., soil respiration, gas diffusion controlled by soil texture)34 might vary across the Pleistocene glacial cycles, leading to potential changes in the empirical δ18Oc—S(z) relation. However, temperature and precipitation reconstructions from the CLP show that variations during the early Pleistocene glacial cycles are well encapsulated in those of the last 800 ky41,42. Moreover, the parent material of the loess-paleosol sequence remained relatively invariant over time27, and C3 plants persistently dominated the CLP (as suggested by the δ13C of soil organic matter, Zhaojiachuan: −24.9‰ ± 0.9‰, Fuxian: −23.6‰ ± 0.7‰). These facts mean that the edaphic and biological factors affecting S(z) likely changed little among glacial periods throughout the Pleistocene. In this case, we expect the environmental controls on δ18Oc and S(z) to remain relatively stable. Further, the δ18Oc during the past 800 ky (−11.0‰ ~ −8.7‰) that are used to generate the empirical δ18Oc—S(z) equation generally overlap with those during the early Pleistocene (−11.2‰ ~ −8.1‰). It is thus reasonable to apply the empirical model to early Pleistocene glacial samples, assuming static relationships among environmental factors (e.g., temperature, rainfall, soil texture), soil respiration, and rainfall-δ18O through time.
Applying the δ18Oc-S(z) empirical scaling relation (Eq. 1) to early Pleistocene samples yields glacial S(z) ranging between 241–551 ppm, with a mean 1σ error of ±203 ppm. S(z) estimates from both sections slightly increase from 317 ± 35 ppm (mean ± SD) in Zhaojiachuan and 355 ± 56 ppm in Fuxian in the 40k world to 355 ± 67 ppm and 411 ± 58 ppm in the 100k world, respectively (Supplementary Fig. S8). The long-term increase in S(z) indicates enhanced soil respiration, which is consistent with paleoenvironmental reconstructions from the CLP that suggest increased land surface temperature42 and near-constant precipitation41 during the glacials across the early Pleistocene.
The long-term glacial CO2 decline
We compute early Pleistocene glacial CO2 levels based on carbonate samples in the clay-sized fractions (n = 141). CO2 uncertainties are quantified by propagating uncertainties sourced from the input variables (i.e., δ13C, temperature, and S(z)) using the PBUQ code based on Monte Carlo random sampling26 (Materials and Methods). Best CO2 estimates indicate a long-term decline of ~30–40 ppm, from an average of 256 ppm (SD = ± 45 ppm, n = 103) in the 40k world to 221 ppm (SD = ± 40 ppm, n = 38) during 1250–800 ka (Fig. 1c). For comparison, δ11B-based glacial CO2, which are overall higher, decreased from 272 ± 62 ppm (n = 81) to 251 ± 31 ppm (n = 79) (Supplementary Fig. S1). The decline in minimum CO2 (on an orbital timescale) is consistent with direct measurements from the Antarctic ice cores12 (Fig. 1b).
Notably, samples from Zhaojiachuan show slightly higher atmospheric CO2 estimates than those from Fuxian (Fig. 1c), possibly biased by merging samples from two sections together to build a statistically significant δ18Oc-S(z) empirical equation. Nonetheless, the CO2 variation trends in the Fuxian and Zhaojiachuan samples are identical throughout the early Pleistocene (Fig. 1c), and the differences between the two sections (~40 ppm) are much smaller compared to errors associated with individual estimates (average 1σ = ± 140 ppm). It should be noted that the paleosol-based glacial and interglacial CO2 estimates are indistinguishable from each other10 (Supplementary Fig. S9). This is likely due to a combination of high uncertainties and incomplete documentation. Specifically, given that the CO2 uncertainty is higher than the full amplitude of CO2 variations (~120 ppm in the 100k world, probably less pronounced in the 40k world), our paleosols cannot resolve the glacial-interglacial CO2 difference. Moreover, because of the dissolution and reprecipitation of pedogenic carbonates, it is unlikely that our paleosol samples recorded the full glacial-interglacial CO2 cycle10.
Coupled glacial CO2 drawdown and global cooling
The paleosol-derived glacial CO2 record allows us to assess the long-term dynamics between atmospheric CO2 and global climate. We selected three types of marine-proxy-based records that capture the large-scale variability of Earth’s climate system with sufficient resolution to resolve orbital cycles: the benthic δ18O stack19, bottom water temperature (BWT) based on benthic foraminifera Mg/Ca from DSDP Site 607 in the North Atlantic43, and sea surface temperature (SST) reconstructions based on alkenone thermometry (\({{{{\rm{U}}}}}_{37}^{{{{\rm{k}}}}\hbox{'}}\)). The benthic δ18O represents a composite signal of global ice volume and deep ocean water temperature, which is spatially more uniform compared to global SST22. Combined with independent BWT reconstructions, these proxies provide insights into the co-evolution of ice volume and ocean temperature. It is important to acknowledge that the Mg/Ca-BWT proxy is complicated by diminished sensitivity at low temperatures and the potential for nonthermal influences44. Nonetheless, given the current lack of a direct, high-fidelity recorder of bottom water temperature45, we proceed with Mg/Ca-BWT record while recognizing its limitations. For SST, we first focus on the ODP Site 1143 in the Western Pacific Warm Pool (WPWP)46, which exhibits a relatively simple response to radiative forcing. As the warmest surface ocean water on Earth, the WPWP temperature affects both the meridional and zonal thermal gradients, exerting significant influences on regional and global climate patterns, thereby representing a critical component of the global climate system47. Unlike their Pliocene counterparts, Pleistocene \({{{{\rm{U}}}}}_{37}^{{{{\rm{k}}}}\hbox{'}}\) data from Site 1143 exhibit higher, orbitally resolved resolution, and the proxy is not saturated (when SST is at ~28 °C, \({{{{\rm{U}}}}}_{37}^{{{{\rm{k}}}}\hbox{'}}\) approaches 1). For comparison, we also included the latest global mean surface temperature (GMST) record based on a compilation of published SST records and climate model simulations48.
Each record was divided into two subgroups (glacial vs interglacial), and the data from each subgroup were binned to a 300-ky time window to allow for direct data comparisons at the same resolution. The 300-ky time window was chosen to ensure enough data coverage (at least 20 glacial/interglacial CO2 estimates within each time interval) such that the mean is representative, but also to allow comparison of long-term variations among individual records. Splitting the data into larger time windows does not change the following conclusions regarding the long-term trends and climate sensitivity (Supplementary Fig. 10).
Atmospheric CO2 levels over the last 800 ky were obtained from ice-core data. Beyond 800 ka, we compiled direct CO2 measurements from blue ice12 as well as proxy-based CO2 records based on paleosols10 and the boron isotope of marine carbonates9,14,15,16,17,49. The alkenone-based CO2 estimates were considered questionable due to the lack of constraints on the associated parameters and thus not included here50,51. A recent work used leaf-wax δ13C from ocean sediments to reconstruct CO2 spanning the last 1.5 Ma52. However, given the complex responses of leaf-wax δ13C to multiple environmental parameters other than atmospheric CO2 levels (e.g., the relative contribution of C3 and C4 plants, temperature, water availability)53,54,55, which were not sufficiently addressed in the original paper, we decided not to include this record. Given the age uncertainty of the blue-ice CO2 data, the upper and lower halves of the measurements were used to represent interglacial and glacial CO2, respectively. The Mg/Ca-based BWT record was recalculated to account for changes in seawater Mg/Ca56 (Materials and Methods). The alkenone-based SST record was calculated using the BAYSPLINE model that better accounts for the non-linearity of the \({{{{\rm{U}}}}}_{37}^{{{{\rm{k}}}}'}\)—SST relationship57 (Materials and Methods).
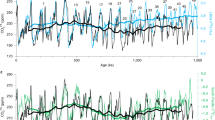
Several key features emerge when comparing interglacial and glacial CO2 separately with global climate records. First, despite an overall cooling trend, key climate conditions during interglacial maxima varied little across the Pleistocene (Fig. 2f–j), especially for benthic δ18O and WPWP-SST records. Likewise, interglacial CO2 show no apparent long-term trend, with median CO2 fluctuating between 220 and 282 ppm except for the Plio-Pleistocene transition (2.6–2.4 Ma) (Fig. 2j). On the other hand, glacial period benthic δ18O, WPWP-SST, and GMST records show long-term, stepwise cooling trends across the early Pleistocene (Fig. 2a–c), which align with the secular decrease of glacial CO2 (Fig. 2d). This exercise underscores the tight coupling between atmospheric CO2 and global climate, as well as the role of a gradually decreasing glacial CO2 in driving the Pleistocene global cooling and the MPT.
All the records were separated into glacial (a–e on the left) and interglacial (f–j on the right) subsets and binned to a 0.3-million-year (300-ky) time window. CO2 data beyond 800 ka were compiled paleosol- and boron-based estimates9,10,14,15,16,17,49 and blue-ice measurements12,78. The medians are highlighted by horizontal lines. Gray dashed lines connect the maximum and minimum values of each time window. The long-term decline in glacial CO2, along with the relatively stable interglacial CO2 maxima, is broadly consistent with global temperature trends.
Climate sensitivities among glacials and interglacials
The composite CO2 record allows us to evaluate the sensitivities of the global climate in response to CO2 forcing among the cold glacials and warm interglacials separately. The SST-based GMST record was used here for climate sensitivity analyses (Fig. 2d, i, Supplementary Fig. S11). In paleoclimate studies, the long-term global temperature response to changes in CO2 forcing is referred to as Earth system sensitivity (ESS)58, which is also equivalent to the specific climate sensitivity term SCO2. ESS accounts for both fast and slow feedbacks (e.g., ice sheets, vegetation, non-CO2 greenhouse gases) and is different from ECS, which only includes fast-feedback processes. For close-to-equilibrium changes in the geologic past, because the annual mean global insolation changes are negligible, radiative perturbations from slow and fast feedbacks must be almost balanced (ΔFslow + ΔFfast ≈ 0). ECS can therefore be estimated from paleoclimate data by quantifying radiative forcings from all slow-feedback processes and removing their influences from the calculated climate sensitivity (Materials and Methods)59. The most important slow-feedback forcing during glacial cycles is ice-sheet albedo (ΔFLI), which is commonly used in paleoclimate studies to approximate ECS2.
In this study, we first calculated ESS based on the linear regression slope (SCO2) in the ΔGMST–ΔFCO2 space (Fig. 3a, Materials and Methods, Eqs. 6–7). The linear regression of interglacial records yields higher p values than those of the glacial records, due to the relatively higher CO2 uncertainty and smaller variabilities of both CO2 and GMST estimates (Fig. 3a–c). The median value of interglacial SCO2 is 1.7 K W−2 m−1, slightly lower than the glacial value of 2.0 K W−2 m−1 (Fig. 3a). The higher glacial SCO2 is consistent with previous work, indicating a larger land-ice-albedo feedback affecting changes in climate among glacial than among interglacial periods9,16. These SCO2 values are also in line with those (~1–2 K W−2 m−1) during the Pliocene-Pleistocene transition calculated from ice-core and boron-derived CO2 data16. Multiplied by a radiative forcing of ~3.7 W m−2 resulting from CO2 doubling, our SCO2 results in ESS estimates of 6.2-7.4 K (3.2–12.0 K, 95% confidence).
a–c ΔGMST48 plotted against changes in different radiative forcings, including CO2 forcing alone (ΔFCO2), combined CO2 and ice-sheet forcings before (ΔFCO2,LI) and after (ΔFCO2,LI-cal) applying the efficacy factor ε. The linear regression slopes represent climate sensitivities. Glacial (blue) and interglacial (orange) records were analyzed separately. The data points and error bars represent the means and standard deviations of all the data within each 300-ky time window. Dashed lines and shades denote linear regression lines and 95% confidence intervals. d–f Probability density distributions of climate sensitivities based on Monte Carlo random sampling (Materials and Methods). d, e correspond to (a, b), whereas f shows Stotal accounting for all slow feedbacks, which approximates ECS. Solid and dashed lines denote the medians and 95% confidence intervals. Cohen’s d and p values are used to evaluate the difference between glacial and interglacial sensitivities, with d > 0.3 and p < 0.001 indicating a significant difference.
To allow comparison with published values, we next calculated specific climate sensitivity using the combined ΔFCO2 and ΔFLI (Materials and Methods, Eq. 8), the latter of which were from published literature based on the deconvolution of benthic δ18O stack with three-dimensional ice-sheet models, in combination with the latitudinal changes in incoming insolation8 (Supplementary Fig. S11). An efficacy factor ε (0.45+0.34/−0.2, mean ± 1σ) was then applied on ΔFLI to account for the smaller contribution of land-ice changes to global temperature change per unit radiative forcing (K W−2 m−1) compared to atmospheric CO260. The corresponding specific climate sensitivity, SCO2,LI-cal, was calculated as ΔT/(ΔFCO2 + ε ΔFLI). Finally, to approximate ECS, the calculated SCO2,LI-cal (Fig. 3c) was multiplied by a conversion factor φ (0.64 ± 0.07, mean±1σ) to account for other slow feedbacks such as vegetation and non-CO2 greenhouse gases (Stotal)3.
Accounting for slow-feedback processes improves the linear correlation between ΔGMST and ΔF (Fig. 3b, c). In particular, median values of SCO2,LI are 0.9 and 1.3 K W−2 m−1 for interglacial and glacial periods, respectively (Fig. 3e). It should be noted that the glacial SCO2,LI remains higher than the interglacial one after considering the ice-sheet albedo feedback, indicating that the radiative response to non-ice-sheet feedbacks, including both fast (e.g., aerosol, sea ice) and/or slow feedbacks (e.g., vegetation) were probably stronger among interglacial periods compared to glacials. The SCO2,LI translate to interglacial and glacial ESS estimates of 3.3 K (1.8–5.5 K, 95% confidence) and 4.8 K (3.0–7.6 K, 95% confidence), respectively, which are consistent with previously reported ESS based on SCO2,LI from the Pliocene-Pleistocene glacial cycles (3.5-5.7 K)8,16,61.
The difference between glacial and interglacial ΔGMST - ΔFtotal regression slopes (Stotal) is negligible after considering all slow-feedback processes, with median Stotal values of 0.9 and 1.0 K W−2 m−1 for glacial and interglacial periods, respectively (Fig. 3f). The corresponding ECS estimates are 3.3 K (2.1–6.3 K, 95% confidence) and 3.7 K (1.7–6.3 K, 95% confidence), which aligns well with the commonly accepted ECS range of 2.6–4 K based on instrumental records and process understanding62. Notably, evidence from both model and data indicates state-dependency of ECS, which refers to changes in the efficacy of feedback processes under different background climate conditions (e.g., temperature, plate configuration, ice sheets), leading to a non-linear relationship between global temperature and CO2 forcing63. For instance, ECS estimates based on observations from glacial-interglacial cycles found lower sensitivity to forcing (smaller ECS) during colder periods compared to warmer periods61,64. However, ECS estimates based on our compiled CO2 records, after accounting for different efficacies of ice-sheet and CO2 forcing as well as other slow-feedback processes, remained consistent among glacial and interglacial periods, indicating no apparent state-dependency. The lack of state-dependency suggests that fast feedbacks maintained relatively constant behavior despite the contrasting climate states of cold glacial and warm interglacial periods.
Methods
Sampling and chronology
We collected paleosol samples from two loess-paleosol sequences located in the central CLP (Fig. S2). For the Zhaojiachuan section (N 35°46′38″ E 107°47′25″), soil samples were collected from the outcrops along the hillside at 10 cm intervals. We trenched the soil profiles 2 m deep before sampling to avoid regolith contamination. For the Fuxian section, we drilled a 206-m core from the highest terrace near Fuxian County (N 36°9′33″ E 109°27′6″), with the upper 161.7 m being the Quaternary loess-paleosol sequence. Soil deposition ages of the two sections are established by MS chronostratigraphy with respect to the Lingtai section, the chronology of which was determined by paleomagnetic polarity and astronomical tuning65.
The ages of pedogenic carbonates in this study were determined by the deposition ages of paleosol layers within which they are embedded. Notably, this age assignment represents their maximum ages as pedogenic carbonate grows below the soil surface. Comparison between loess deposition ages and the radiocarbon ages of clay-sized carbonates formed during the last glacial period suggests that the carbonates were formed at 0-50 cm depth, which corresponds to a maximum age difference of 10-ky (Fig. S4). Such an age offset is substantially smaller than the time span of each glacial period (100 ky). To avoid pedogenic carbonates formed during interglacial periods that were translocated into the underlying glacial units, samples were selected from the bottom part of each glacial unit.
Particle size separation
To avoid contamination of detrital carbonates, we target calcites within the clay-sized fractions (<2 μm) of bulk paleosols. This approach has been successfully applied to extract pedogenic carbonates from paleosols in previous studies40. Due to the preferential dissolution and precipitation of fine-grained carbonate by soil solution, pedogenic carbonate dominates the clay-sized fractions of loess-parented soils40. The clay-sized particles were extracted through centrifugal sedimentation following Stocks’ law. Specifically, a sample split of ~10 g was ultrasonically dispersed in deionized water and then centrifuged at 2000 rpm for 3 minutes. The upper turbid solution was transferred and centrifuged again at 5000 rpm for 15 minutes, with the bottom precipitation being the clay-sized fraction. We repeated the above steps multiple times until little suspension existed in the solution. The precipitates were oven-dried at 60 °C and then ground into powder with an agate mortar and pestle for subsequent analyses. The micromorphology and geochemistry of our samples corroborate a minor contribution from detrital carbonates. Specifically, scanning electron microscopy imaging reveals that the clay-sized fractions are dominated by nanoscale NFC (Supplementary Fig. S3). Moreover, the radiocarbon ages of clay-sized carbonates are close to those of coeval soil organic matter, and the depositional ages of loess from the corresponding layer (Supplementary Fig. S4), indicating a minor contribution of the radiocarbon-dead, detrital carbonates.
Stable isotope analyses
We selected 181 paleosol samples from the two loess sections on the CLP (96 from Fuxian and 85 from Zhaojiachuan) for CO2 reconstruction. For δ13C and δ18O analyses, sample splits of 50 mg were first treated with 10% H2O2 to remove soil organic matter. The residues were then oven-dried and ground into powder for homogenization. The samples were measured on a Thermo-Finnigan MAT 253 IRMS attached with a Kiel IV carbonate device (75 °C reactions in 100% H3PO4). The isotopic values are reported in the δ notation (normalized to the VPDB standard) with permil (‰) unit. Replicates of samples (n = 5) were analyzed, yielding a standard deviation of 0.03‰ for both δ13C and δ18O. For δ13Co analyses, sample splits of 200 mg were reacted with 1 N HCl overnight to remove carbonates. HCl was added multiple times until no bubbles were produced to ensure the complete removal of inorganic carbon. The samples were subsequently rinsed with deionized water repeatedly until pH reached neutral, oven-dried, and ground into powder. 25–30 mg of sample was sealed in a tin capsule and analyzed using a Costech 4010 Elemental Analyzer coupled to a Thermo Delta V IRMS system. δ13Co values were corrected to the VPDB scale with an average standard deviation of 0.2‰ from replicate analyses of samples (n = 3).
Radiocarbon analyses
To examine the potential contamination of detrital carbonate and the illuviation of pedogenic carbonate, we performed radiocarbon analysis on both the carbonate (n = 9) and soil organic matter (n = 4) in our clay-sized samples from the loess-paleosol unit corresponding to the Holocene and last glacial period (Supplementary Fig. S4). The measurement was carried out at the Keck-Carbon Cycle AMS facility at the University of California, Irvine. There, sample splits were combusted to CO2, cryogenically purified, graphitized, and measured on an NEC Compact (1.5 SDH) Accelerator Mass Spectrometer. Radiocarbon activities are reported in Δ14C (‰), which was calculated from fraction modern carbon (Fm) and corrected for the decay of the radiocarbon standard since 195066. Radiocarbon ages were converted to calendar ages using the INTCAL20 calibration curve67.
The paleosol-CO2 method
The paleosol-CO2 proxy is based on the steady-state diffusion reaction model25. Specifically, the δ13C of CO2 in soil pore space (δ13Cs) is controlled by two endmembers—CO2 sourced from the respiration of plant roots and soil microbes, and atmospheric CO2 penetrating the soil–air interface through diffusion24. The equation is as follows:
where δ13Ca and δ13Cr∗ represent the δ13C of atmospheric CO2 and soil-derived CO2 (including both autotrophic and heterotrophic respiration), respectively; [CO2]atm and [CO2]soil are atmospheric CO2 and total-soil CO2 concentrations, respectively; S(z) is the concentration of soil-respired CO2 at the depth z of pedogenic carbonate growth. At steady-state diffusion, the δ13C of CO2 in the soil pore space sourced from the respiration (δ13Cr∗) can be calculated from the δ13C of respired CO2 (δ13Cr) via diffusion coefficients25, and we get the paleosol-CO2 equation by rearranging Eq. 2:
where 1.0044 and 4.4 are diffusion coefficients resulting from the different diffusivity of 12CO2 and 13CO2 in soil pores. The R values, which can be solved by the three δ13C parameters, represent the ratio of atmospheric CO2 and soil-respired CO2 in the soil pore space during pedogenic carbonate growth. Under surficial climatic conditions, pedogenic carbonates formed in isotopic equilibrium with soil CO2, and the fractionation between calcite and CO2 is temperature-sensitive68. δ13Cs can thus be solved by the δ13C of pedogenic carbonate (δ13Cc) with calcite crystallization temperature (T).
Defining input parameters
The δ13Ca is estimated by the δ13C of marine carbonates, which records the δ13C of ocean dissolved inorganic carbon that is sensitive to changes in δ13Ca69. We do not have direct T measurements in this study. Nonetheless, the paleosol-CO2 method is not sensitive to T compared to other inputs (e.g., S(z), δ13Cr), with 5 °C uncertainty contributing to less than 10% of the total uncertainty of the resulting CO2 estimate70. Soil surface temperatures reconstructed from biomarkers on the CLP reveal a small, gradual warming trend of ~1.1 °C across the Pleistocene accompanied by large fluctuations42. Notably, due to increased vegetation coverage during interglacial periods, there are no apparent soil temperature differences between glacial and interglacial periods. In this case, considering the dampening of soil temperature with depth, we assigned a uniform T using modern mean annual soil temperatures at 20 cm depth from each sampling site (http://www.data.cma.cn). The 1σ error was set to ±3 °C based on the variations of reconstructed soil surface temperatures on the CLP over the Pleistocene glacial cycles42,71. Importantly, as both the late Pleistocene S(z) estimates used to calibrate the δ18O-S(z) relationship and the early Pleistocene CO2 estimates were calculated using the same temperature, if the actual Ts are systematically different from our assumed values, their influences are inherently canceled in our approach. The δ13Cr is approximated by measuring the δ13C of soil organic matter (δ13Co) from the same sediment layer where pedogenic carbonates were collected. Notably, studies on modern soil and paleosol profiles indicate 13C/12C fractionation accompanying organic matter decomposition72,73. We applied a −1 ± 0.5‰ (mean±1σ) correction on δ13Co data to account for the decomposition-related fractionation based on the difference between δ13C of soil organic matter occluded in calcite nodules and those in bulk paleosols, the former of which are considered resistant to decomposition73. The δ18Oc - S(z) relation (Eq. 1) was used to estimate S(z), with the 1σ error propagated through Monte Carlo random sampling.
A MATLAB-PBUQ code based on Monte Carlo simulations26 was used to calculate CO2 and propagate errors associated with input variables. The medians, 16th and 84th, and 2.5th and 97.5th of the resulting CO2 distributions were reported as the best estimates with 1σ and 2σ errors. The time series of both the raw input parameters (δ13Cc, δ18Oc, δ13Co, δ13Ca) and intermediate parameters (S(z), δ13Cs) used for calculating CO2 were provided in Supplementary Figs. S12 and S8, respectively.
Bootstrap sampling
We divided the 800-ky samples (n = 40) into two groups—one training group used to generate the δ18Oc—S(z) empirical scaling relation and one test group that calculated CO2 with S(z) estimated from the δ18Oc—S(z) relation established using the training group data. For a given training group size n, we randomly drew n samples from the population, calculated CO2 for the remaining (40–n) samples, and examined the mean relative difference (χ) between estimated CO2 and the corresponding ice-core CO2:
The bootstrap sampling was repeated 10,000 times to obtain the corresponding χ values. The χ distributions cluster around −0.05 regardless of the training group size (n = [10,30]), with 95% of the data points falling within ~−20/ + 10% difference (Supplementary Fig. S13). It is noteworthy that there is a negative skew for the χ distributions. However, such deviations are minor, as the mean χ of −0.05 translates to 9–11 ppm when CO2 levels are 174 to 218 ppm (highest and lowest ice-core CO2 values used for comparison). Therefore, this test demonstrates the robustness of the δ18Oc—S(z) scaling approach, lending support for applying this approach to resolve S(z).
Recalculating BWT and SST records
The original BWT record from North Atlantic43 assumed a constant seawater Mg/Ca throughout the Pleistocene. Although both Mg and Ca have long residence time in the ocean, recent studies suggest a non-negligible long-term increase in seawater Mg/Ca over the past 30 million years due to changes in hydrothermal activity, weathering, and carbonate deposition56,74. We used the following equation to recalculate BWT accounting for changes in seawater Mg/Ca based on the calibration equation56 and the original equation43:
Where [Mg/Ca]sw,0, [Mg/Ca]sw,t, [Mg/Ca]foram are the Mg/Ca ratios of modern seawater, seawater of the time of interest, and foraminifera, respectively. [Mg/Ca]sw,0 is set to 5.2 mmol/mol75. [Mg/Ca]sw,t is based on a fourth-order polynomial curve fit through compiled [Mg/Ca]sw proxy records56.
A global compilation of core-top samples shows strong seasonal production of alkenones and significant attenuation of the \({{{{\rm{U}}}}}_{37}^{{{{\rm{k}}}}\hbox{'}}\) response to higher temperature as it reaches saturation57. To account for these observations, we recalculated the \({{{{\rm{U}}}}}_{37}^{{{{\rm{k}}}}\hbox{'}}\)-based SSTs by applying the original \({{{{\rm{U}}}}}_{37}^{{{{\rm{k}}}}\hbox{'}}\) data to a Bayesian B-spline regression model, BAYSPLINE57.
Climate sensitivity analyses
The ESS, or SCO2, is defined as the long-term temperature response (ΔT) to a given change in CO2 forcing (ΔFCO2)58:
where both ΔT and ΔFCO2 refer to changes relative to the pre-industrial condition. Here we applied the latest global mean surface temperature record (ΔGMST), which is based on a compilation of SST records48. The ΔFCO2 can be related to changes in atmospheric CO2 levels by accounting for the logarithmic dependence of the infrared radiation absorption on CO2:
where Co is the pre-industrial level of 278 ppm. To account for radiative forcings resulting from all slow-feedback processes and estimate ECS, we used the following equation derived from Stap et al.60:
where ΔFLI is the ice-sheet forcing obtained from Köhler et al.8, ε is the efficacy factor (0.45+0.34/−0.2, mean±1σ) that accounts for the smaller radiative efficacy of ice-sheet feedback compared to atmospheric CO260, and φ is a conversion factor (0.64 ± 0.07, mean ± 1σ) that represents radiative forcings from other slow feedbacks3. ΔF2xCO2 is the radiative forcing resulting from a doubling CO2 (~3.7 W/m2)76. To generate the probability density distributions of the calculated climate sensitivities, a value was randomly sampled from normal distributions defined by the mean value and standard deviation of each input parameter (ΔGMST, CO2, ΔFLI, φ, ε) for each 300-ky time window, forming the basis for generating a linear regression slope between ΔGMST and ΔF. We performed 100,000 iterations and retained the slopes with p values below 0.01. The linear regression slopes between ΔGMST and ΔF (change relative to the pre-industrial conditions) indicate climate sensitivities, with steeper slopes indicating higher sensitivity.
Data availability
The data generated in this study have been deposited in the Figshare database (https://doi.org/10.6084/m9.figshare.22134656.v4). Data and codes used for statistical analyses and plotting figures have been deposited in GitHub (https://github.com/da-jiawei/Glacial_CO2)77.
Change history
18 August 2025
A Correction to this paper has been published: https://doi.org/10.1038/s41467-025-62940-2
References
Charney, J. G. et al. Carbon Dioxide and Climate: A Scientific Assessment. (National Academy of Sciences, Washington, DC, 1979).
IPCC. Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. In Press (Cambridge University Press, USA, 2021).
Rohling, E. J. et al. Making sense of palaeoclimate sensitivity. Nature 491, 683–691 (2012).
Deconto, R. M. et al. Thresholds for Cenozoic bipolar glaciation. Nature 455, 652 (2008).
Imbrie, J. et al. On the structure and origin of major glaciation cycles 1. Linear responses to Milankovitch forcing. Paleoceanography 7, 701–738 (1992).
Berends, C. J., Köhler, P., Lourens, L. J. & van de Wal, R. S. W. On the Cause of the Mid-Pleistocene Transition. Rev. Geophys. 59, e2020RG000727 (2021).
Friedrich, T., Timmermann, A., Tigchelaar, M., Elison Timm, O. & Ganopolski, A. Nonlinear climate sensitivity and its implications for future greenhouse warming. Sci. Adv. 2, e1501923 (2016).
Köhler, P., de Boer, B., von der Heydt, A. S., Stap, L. B. & van de Wal, R. S. W. On the state dependency of the equilibrium climate sensitivity during the last 5 million years. Climate 11, 1801–1823 (2015).
Chalk, T. B. et al. Causes of ice age intensification across the Mid-Pleistocene transition. Proc. Natl. Acad. Sci. USA 114, 13114 (2017).
Da, J., Zhang, Y. G., Li, G., Meng, X. & Ji, J. Low CO2 levels of the entire Pleistocene epoch. Nat. Commun. 10, 4342 (2019).
Zhang, Y. G., Pagani, M., Liu, Z., Bohaty, S. M. & Deconto, R. A 40-million-year history of atmospheric CO2. Philos. Trans. R. Soc. A: Math. Phys. Eng. Sci. 371, 20130096 (2013).
Yan, Y. et al. Two-million-year-old snapshots of atmospheric gases from Antarctic ice. Nature 574, 663–666 (2019).
Peterson, J. M. et al. Ice cores from the Allan Hills, Antarctica show relatively stable atmospheric CO2 and CH4 levels over the last 3 million years. Preprint at https://doi.org/10.21203/rs.3.rs-5610566/v1 (2024).
Hönisch, B., Hemming, N. G., Archer, D., Siddall, M. & McManus, J. F. Atmospheric carbon dioxide concentration across the mid-Pleistocene transition. Science 324, 1551–1554 (2009).
Bartoli, G., Hönisch, B. & Zeebe, R. E. Atmospheric CO2 decline during the Pliocene intensification of Northern Hemisphere glaciations. Paleoceanography 26 (2011).
Martinez-Boti, M. A. et al. Plio-Pleistocene climate sensitivity evaluated using high-resolution CO2 records. Nature 518, 49–54 (2015).
Dyez, K. A., Hönisch, B. & Schmidt, G. A. Early Pleistocene obliquity-scale pCO2 variability at ~1.5 million years ago. Paleoceanogr. Paleoclimatol. 33, 1270–1291 (2018).
The Cenozoic Co2 Proxy Integration Project (CENCO2PIP) Consortium Toward a Cenozoic history of atmospheric CO2. Science 382, eadi5177 (2023).
Lisiecki, L. E. & Raymo, M. E. A plio-Pleistocene stack of 57 globally distributed benthic δ 18 O records. Paleoceanography 20, 1–17 (2005).
Herbert, T. D., Peterson, L. C., Lawrence, K. T. & Liu, Z. Tropical ocean temperatures over the past 3.5 million years. Science 328, 1530–1534 (2010).
Willeit, M., Ganopolski, A., Calov, R. & Brovkin, V. Mid-Pleistocene transition in glacial cycles explained by declining CO2 and regolith removal. Sci. Adv. 5, eaav7337 (2019).
Legrain, E., Parrenin, F. & Capron, E. A gradual change is more likely to have caused the mid-Pleistocene Transition than an abrupt event. Commun. Earth Environ. 4, 1–10 (2023).
Beerling, D. J. & Royer, D. L. Convergent cenozoic CO2 history. Nat. Geosci. 4, 418–420 (2011).
Cerling, T. E. The stable isotopic composition of modern soil carbonate and its relationship to climate. Earth Planet. Sci. Lett. 71, 229–240 (1984).
Cerling, T. E. Use of carbon isotopes in paleosols as an indicator of the P(CO2) of the paleoatmosphere. Glob. Biogeochem. Cycles 6, 307–314 (1992).
Breecker, D. O. Quantifying and understanding the uncertainty of atmospheric CO2 concentrations determined from calcic paleosols. Geochem. Geophys. Geosyst. 14, 3210–3220 (2013).
An, Z. et al. Late Cenozoic climate change in monsoon-arid Asia and global changes. In Late Cenozoic Climate Change in Asia, 491–581 (Springer, 2014).
Cohen, K. M. & Gibbard, P. L. Global chronostratigraphical correlation table for the last 2.7 million years, version 2019 QI-500. Quat. Int. 500, 20–31 (2019).
Meng, X. et al. Dolomite abundance in Chinese loess deposits: a new proxy of monsoon precipitation intensity. Geophys. Res. Lett. 42, 10391–10398 (2015).
Meng, X. et al. Mineralogical evidence of reduced East Asian summer monsoon rainfall on the Chinese loess plateau during the early Pleistocene interglacials. Earth Planet. Sci. Lett. 486, 61–69 (2018).
Milliere, L. et al. Stable carbon and oxygen isotope signatures of pedogenic needle fibre calcite. Geoderma 161, 74–87 (2011).
Wang, K. et al. Enhanced soil respiration, vegetation and monsoon precipitation at Lantian, East Asia during Pliocene warmth. Clim. Dyn. 59, 2683–2697 (2022).
Barta, G. Secondary carbonates in loess-paleosoil sequences: a general review. Open Geosci. 3, 129–146 (2011).
Breecker, D. O., Sharp, Z. D. & McFadden, L. D. Seasonal bias in the formation and stable isotopic composition of pedogenic carbonate in modern soils from central New Mexico, USA. Geol. Soc. Am. Bull. 121, 630–640 (2009).
Balsam, W., Ji, J. & Chen, J. Climatic interpretation of the Luochuan and Lingtai loess sections, China, based on changing iron oxide mineralogy and magnetic susceptibility. Earth Planet. Sci. Lett. 223, 335–348 (2004).
Wang, Y. et al. Millennial- and orbital-scale changes in the East Asian monsoon over the past 224,000 years. Nature 451, 1090–1093 (2008).
Retallack, G. J. Pedogenic carbonate proxies for amount and seasonality of precipitation in paleosols. Geology 33, 333–336 (2005).
Cotton, J. M. & Sheldon, N. D. New constraints on using paleosols to reconstruct atmospheric pCO2. Geol. Soc. Am. Bull. 124, 1411–1423 (2012).
Ji, S. et al. Quantifying soil-respired CO2 on the Chinese Loess Plateau. Palaeogeogr. Palaeoclimatol. Palaeoecol. 562, 110158 (2021).
Da, J., Li, G. K. & Ji, J. Seasonal changes in the formation time of pedogenic carbonates on the Chinese Loess Plateau during quaternary glacial cycles. Quat. Sci. Rev. 305, 108008 (2023).
Cai, J. et al. Pleistocene global cooling did not weaken the East Asian summer monsoon. J. Geophys. Res. Atmos. 130, e2024JD042746 (2025).
Lu, H. et al. Decoupled land and ocean temperature trends in the early-middle Pleistocene. Geophys. Res. Lett. 49, e2022GL099520 (2022).
Sosdian, S. & Rosenthal, Y. Deep-sea temperature and ice volume changes across the pliocene-pleistocene climate transitions. Science 325, 306–310 (2009).
Yu, J. & Broecker, W. S. Comment on “deep-sea temperature and ice volume changes across the pliocene-pleistocene climate transitions”. Science 328, 1480–1480 (2010).
Herbert, T. D. The mid-Pleistocene climate transition. Annu. Rev. Earth Planet. Sci. 51, 389–418 (2023).
Li, L. et al. A 4-Ma record of thermal evolution in the tropical western Pacific and its implications on climate change. Earth Planet. Sci. Lett. 309, 10–20 (2011).
Zhang, Y. G., Pagani, M. & Liu, Z. A 12-million-year temperature history of the Tropical Pacific Ocean. Science 344, 84–87 (2014).
Clark, P. U., Shakun, J. D., Rosenthal, Y., Köhler, P. & Bartlein, P. J. Global and regional temperature change over the past 4.5 million years. Science 383, 884–890 (2024).
Seki, O. et al. Alkenone and boron-based Pliocene pCO2 records. Earth Planet. Sci. Lett. 292, 201–211 (2010).
Köhler, P. Atmospheric CO2 Concentration Based On Boron Isotopes Versus Simulations Of The Global Carbon Cycle During The Plio-Pleistocene. Paleoceanogr. Paleoclimatol.38, e2022PA004439 (2023).
Zhang, Y. G. et al. Refining the alkenone-pCO2 method I: Lessons from the Quaternary glacial cycles. Geochim. Cosmochim. Acta 260, 177–191 (2019).
Yamamoto, M. et al. Increased interglacial atmospheric CO2 levels followed the mid-Pleistocene Transition. Nat. Geosci. 15, 307–313 (2022).
Zhou, H., Helliker, B. R., Huber, M., Dicks, A. & Akçay, E. C4 photosynthesis and climate through the lens of optimality. Proc. Natl. Acad. Sci. 115, 12057–12062 (2018).
Franks, P. J. et al. New constraints on atmospheric CO2 concentration for the Phanerozoic. Geophys. Res. Lett. 41, 4685–4694 (2014).
Schubert, B. A. & Jahren, A. H. The effect of atmospheric CO2 concentration on carbon isotope fractionation in C3 land plants. Geochim. Cosmochim. Acta 96, 29–43 (2012).
Sosdian, S. M. & Lear, C. H. Initiation of the Western Pacific Warm Pool at the Middle Miocene climate transition?. Paleoceanogr. Paleoclimatol. 35, e2020PA003920 (2020).
Tierney, J. E. & Tingley, M. P. BAYSPLINE: a new calibration for the alkenone paleothermometer. Paleoceanogr. Paleoclimatol. 33, 281–301 (2018).
Lunt, D. J. et al. Earth system sensitivity inferred from Pliocene modelling and data. Nat. Geosci. 3, 60–64 (2010).
Rohling, E. J. et al. Comparing climate sensitivity, past and present. Annu. Rev. Mar. Sci. 10, 261–288 (2018).
Stap, L. B., Köhler, P. & Lohmann, G. Including the efficacy of land ice changes in deriving climate sensitivity from paleodata. Earth Syst. Dyn. 10, 333–345 (2019).
von der Heydt, A. S., Köhler, P., van de Wal, R. S. W. & Dijkstra, H. A. On the state dependency of fast feedback processes in (paleo) climate sensitivity. Geophys. Res. Lett. 41, 6484–6492 (2014).
Sherwood, S. C. et al. An assessment of earth’s climate sensitivity using multiple lines of evidence. Rev. Geophys. 58, e2019RG000678 (2020).
Caballero, R. & Huber, M. State-dependent climate sensitivity in past warm climates and its implications for future climate projections. Proc. Natl. Acad. Sci. 110, 14162–14167 (2013).
Köhler, P. et al. A State-Dependent Quantification of Climate Sensitivity Based on Paleodata of the Last 2.1 Million Years. Paleoceanography 32, 1102–1114 (2017).
Sun, Y., Clemens, S. C., An, Z. & Yu, Z. Astronomical timescale and palaeoclimatic implication of stacked 3.6-Myr monsoon records from the Chinese Loess Plateau. Quat. Sci. Rev. 25, 33–48 (2006).
Stuiver, M. & Polach, H. A. Discussion Reporting of 14C Data. Radiocarbon 19, 355–363 (1977).
Reimer, P. J. et al. The IntCal20 Northern Hemisphere radiocarbon age calibration curve (0–55 cal kBP). Radiocarbon 62, 725–757 (2020).
Romanek, C. hristopherS., Grossman, E. thanL. & Morse, &J. ohnW. Carbon isotopic fractionation in synthetic aragonite and calcite: Effects of temperature and precipitation rate. Geochim. Cosmochim. Acta 56, 419–430 (1992).
Tipple, B. J., Meyers, S. R. & Pagani, M. Carbon isotope ratio of Cenozoic CO2: a comparative evaluation of available geochemical proxies. Paleoceanography 25, PA001851 (2010).
Breecker, D. O. Improving paleosol carbonate based estimates of ancient atmospheric CO2. Geochem. News 144 (2010).
Lu, H. et al. 800-kyr land temperature variations modulated by vegetation changes on Chinese Loess Plateau. Nat. Commun. 10, 1958 (2019).
Wynn, J. G. Carbon isotope fractionation during decomposition of organic matter in soils and paleosols: Implications for paleoecological interpretations of paleosols. Palaeogeogr. Palaeoclimatol. Palaeoecol. 251, 437–448 (2007).
Da, J., Li, G. K. & Ji, J. Overestimate of C4 plant abundance caused by soil degradation-induced carbon isotope fractionation. Geophys. Res. Lett. 48, e2021GL093407 (2021).
Evans, D. & Müller, W. Deep time foraminifera Mg/Ca paleothermometry: nonlinear correction for secular change in seawater Mg/Ca. Paleoceanography 27 (2012).
Broecker, W. S. & Peng, T. H. Tracers in the Sea. (Palisades, New York: Lamont-Doherty Geological Observatory, Columbia University, 1982).
Myhre, G., Highwood, E. J., Shine, K. P. & Stordal, F. New estimates of radiative forcing due to well mixed greenhouse gases. Geophys. Res. Lett. 25, 2715–2718 (1998).
Da, J. No apparent state-dependency of equilibrium climate sensitivity between the Pleistocene glacial and interglacial climate states. GitHub https://doi.org/10.5281/zenodo.15706451 (2025).
Bereiter, B. et al. Revision of the EPICA Dome C CO2 record from 800 to 600 kyr before present. Geophys. Res. Lett. 42, 542–549 (2015).
Acknowledgements
The authors acknowledge Yilan Liu, Haiyan Shen, and Xiaomei Xu for supervising stable isotope and radiocarbon analyses. We appreciate the assistance of Y.S., Wenwen Zhen, Xianqiang Meng, Rui Bao, and Zhibing Zheng in field sampling. We sincerely thank the two anonymous reviewers for their insightful and constructive comments. This work was supported by the National Natural Science Foundation of China (42030503) to J.J., by a UCSB Regent’s Junior Faculty Fellowship to G.K.L. and by NSF FRES grant (2121325) D.O.B.
Author information
Authors and Affiliations
Contributions
J.D. contributed study design, data acquisition, data analysis, writing, editing, and funding acquisition. Y.G.Z. contributed study design, data analysis, and editing. X.L. contributed to data acquisition, editing. G.K.L. contributed data analysis, editing. D.O.B. contributed data analysis, editing, and funding acquisition. T.C. contributed data analysis, editing. J.J. contributed study design, supervision, data analysis, editing, and funding acquisition.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Hui Tang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Da, J., Zhang, Y.G., Liu, X. et al. No apparent state-dependency of equilibrium climate sensitivity between the Pleistocene glacial and interglacial climate states. Nat Commun 16, 6608 (2025). https://doi.org/10.1038/s41467-025-61941-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-61941-5