Abstract
Scaffold hopping is a key strategy in drug discovery. While one-to-one scaffold hopping strategies are thriving and evolving, one-to-multiple strategies remain challenging to design. We present here a distinct scaffold hopping strategy for the skeletal editing of pyrimidines into a wide range of heteroarenes through the addition of nucleophiles, ring-opening, fragmentation, and ring-closing (ANROFRC) processes. This method features the in situ generation of a vinamidinium salt intermediate, which serves as a unique N-C-C-C four-atom (A4) synthon that reacts with A1 and A2 synthons. Mechanistic studies reveal that C4-aryl substituents play a crucial role in stabilizing the vinamidinium salt intermediate. This work provides a powerful tool for the systematic construction and modification of nitrogen heterocycles, thereby expanding conventional molecular editing techniques.
Similar content being viewed by others
Introduction
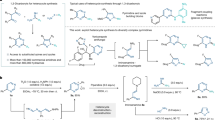
Nitrogen heterocycles are of importance in organic chemistry due to their prevalence in pharmaceuticals, agrochemicals, and materials1,2,3,4,5. For example, 82% of the top 200 FDA-approved new small molecule drugs from 2013 to 2023 contain at least one nitrogen heterocycle6. Strategic alteration to the nitrogen heterocycles of existing drug candidates can lead to changes in pharmacological properties and improve the efficacy of drug molecules, thus advancing the drug discovery process. For instance, a C-to-N exchange from imidazopyridazine to triazolopyridine significantly decreased the IC50 value from 130 to 69 nM of a PIM-1 kinase inhibitor7. Similarly, slight structural differences in azaheterocycles between Vardenafil and Sildenafil resulted in higher potency of Vardenafil as a phosphodiesterase5 inhibitor (Fig. 1A)8. However, the modification of azaheterocycles is deceptively easy, since a simple change in azaheterocycles in drug candidates often requires multi-step synthetic procedures and is time-consuming. To this end, developing methods capable of modifying existing nitrogen heterocycles in a single synthetic operation is highly desirable, as this could enable the efficient diversification of nitrogen- containing compounds. In addition to the conventional peripheral C–H or C–X bond functionalizations9,10, skeletal editing by altering the core framework to engineer molecules has emerged as a powerful platform for the modular modification of nitrogen heterocycles11,12,13,14,15,16,17.
A Selected examples of scaffold hopping in drug discovery. B ANROFRC process in editing pyrimidine. C Scaffold editing of pyrimidine via A4 synthon.
Skeletal editing of nitrogen heterocycles mainly falls into three strategies. The first strategy involves carbene or nitrene insertion-induced subsequent ring expansion or atom exchange reactions18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35, while the second strategy relies on the sequential dearomatization, cycloaddition, and rearomatization to realize atom exchange or functionalization of azaheterocycles36,37,38,39,40,41,42,43,44. Equally important, the third method to edit azaheterocycles consists of sequential addition of nucleophile, ring-opening, and ring-closing (ANRORC) reactions45. The ANRORC reaction is notable for its versatility and efficiency, and has been applied to achieve the atom exchange and functionalization of pyridines, as well as skeleton edit of pyrimidines46,47,48,49,50,51,52,53,54,55,56,57. Despite these advances, the vast majority of known skeletal editing approaches are limited to one-to-one scaffold hopping, converting one azaheterocycle into another specific azaheterocycle. Strategies are thus needed, to both achieve one-to-multiple transformations of azaarenes and to complement the-state-of-art of skeletal editing tactics.
In classical ANRORC reactions, the ring-opening step is typically followed directly by a ring-closing reaction, completing the reaction sequence. This prompted us to explore strategies to fragment the ring-opening intermediate, generating more reactive species before ring closure. The resulting intermediates can then undergo ring-closing process with various coupling reagents. Such a sequential process of addition of nucleophile, ring-opening, fragmentation, and ring-closing (ANROFRC) reactions may enable efficient one-to-multiple skeletal editing methods for azaarenes (Fig. 1B). In this context, Paton and McNally recently developed an elegant deconstruction-reconstruction protocol to engineer pyrimidines, which follows the ANROFRC process (Fig. 1B)58. The success of this method depends on whether the streptocyanine formed by the ring opening of the N-Tf pyrimidinium salt can undergo fragmentation to generate the key intermediate iminoenamine or vinamidinium salt. These in situ generated intermediates, acting as C-C-C three-atom A3 synthons with reactivity analogous to 1,3-dicarbonyl compounds, subsequently proceed to ring closure with a range of nitrogen-containing reagents to yield substituted pyrimidines, pyridines, and 1,2-azoles.
In this study, we present an alternative ANROFRC approach for scaffold editing of pyrimidines, wherein an N-C-C-C four-atom A4 synthon reacts with A1 and A2 synthons, for the modular synthesis of azaheterocycles, including pyrroles, pyridines, 15N labeled pyrimidines, and pyridones (Fig. 1C). The success of this strategy lies in the generation of an isolable iminium-enamine intermediate from pyrimidine ring cleavage, which functions as an N-C-C-C four-atom A4 synthon in the subsequent cyclization reaction. This method features broad substrate scope and functional group tolerance. Notably, this unique transformation of pyrimidines into a nitrogen-containing A4 synthon is significantly distinct from and complementary to the conventional C-C-C three-atom A3 synthons58.
Results
Optimization studies
We began our investigation by exploring the scaffold editing of pyrimidines into other azaheterocycles, as pyrimidine motifs are widely present in biologically active compounds (Fig. 2). The initial attempts to activate pyrimidine using N-2,4-dinitrophenyl chloride, based on our previous strategy for activating pyridines59, were not successful. We then turned to trifluoromethanesulfonic anhydride for the activation of 4-phenylpyrimidine (1a), inspired by the works of Sarpong49 and Toscano60. When 1a was activated by trifluoromethanesulfonic anhydride in dichloromethane at −78 °C for 30 min, the subsequent ring-opening and fragmentation occurred upon the addition of secondary amines as nucleophiles, leading to the reactive synthons. These synthons then reacted with the A1 synthon, trimethylsulfoxonium chloride in the presence of base, resulting in the formation of 2-phenylpyrrole (2a). Through systematic screening of secondary amine nucleophiles, N-methylaniline was found to produce 2-phenylpyrrole in 51% yield. All reactions needed to be conducted with aluminum foil protection due to the light-sensitive nature of pyrrole products. In the experiments attempting to synthesize 2-trifluoromethylpyridine, it was found that piperidine, as the nucleophilic ring-opening reagent, led to a more favorable yield. In addition to the ring-opening reagent, the base, concentration, and solvent were all found to significantly affect the reaction yields during the optimization studies (Supplementary Tables 1–12). Intensive optimizations eventually revealed the optimal reaction conditions for both pyrimidine skeleton editing protocols: (1) 2-phenylpyrrole was synthesized in 87% yield through Tf₂O activation and N-methylaniline-mediated ring-opening at −78 °C, followed by reaction with trimethylsulfoxonium chloride using KOH as base in THF at 80 °C for 16 h under light-protected conditions; (2) 2-trifluoromethylpyridine was obtained in 75% yield via Tf₂O activation and piperidine-induced ring-opening at 0 °C, then followed by reaction with trifluoroacetone in 1,4-dioxane at 120 °C for 16 h using KOtBu as base.
A Optimizations for the formation of 2a. B Optimizations for the formation of 3a. All reactions were performed on a 0.2 mmol scale. Isolated yields are reported unless otherwise specified.
Substrates scope
With the optimal conditions in hand, we then explored the substrate scope for pyrimidine skeleton editing (Fig. 3). In addition to the model substrate 4-phenylpyrimidine (1a), the effect of substituents on the phenyl ring was first investigated. The para-substituted 4-phenylpyrimidines, bearing both electron-donating and electron-withdrawing groups, were compatible with the reaction conditions, yielding the corresponding 2-arylpyrroles (2b–2 g) in moderate yields. The meta-substituted 4-phenylpyrimidines can also be converted to 2-arylpyrrole (2h–2j) in moderate yields. Likewise, ortho-substituted 4-phenylpyrimidines proved to be suitable substrates, yielding 2k and 2 l in 55% and 51% yield, respectively. Replacing the phenyl group with polyaryl and heteroaryl groups, including naphthyl (2 m), benzothienyl (2n), benzofuranyl (2o), and thiophenyl (2p), at the 4-position of pyrimidine generally resulted in the formation of the corresponding pyrroles in moderate to good yields. Notably, disubstituted pyrimidines were also transformed into corresponding 2,3-disubstituted pyrroles (2q and 2r), albeit with lower yields. To further demonstrate the generality of this protocol, the contraction of complex molecules containing pyrimidine was also examined. For instance, the sterically hindered pyrimidines bearing isoxazole reacted smoothly to form 2 s and 2t in 48% and 51% yield, respectively, while pyrrole 2 u was obtained in 41% yield. For disubstituted and structurally complex pyrimidines, the reaction efficiency was notably diminished, with significant amounts of unreacted starting material observed. For product 2r, we attribute the low yield to the poor solubility of the pyrimidine substrates, which may hinder the efficient formation of pyrimidinium salts, thus leading to lower yields. The low yield for product 2 u is likely due to the unique structure of the substrate, which results in incomplete triflylation. Subsequent evaluation of a bioactive pyrimidine-containing derivative afforded the desired pyrrole product (2 v) in merely 21% yield, with 45% of the starting material remaining unreacted. The diminished yield is attributed to competitive triflation by Tf₂O at multiple nucleophilic nitrogen centers in the substrate, thereby lowering the activation efficiency of the pyrimidine ring.
Condition a: Pyrimidine (0.2 mmol), Tf2O (1.1 equiv.), N-methylaniline (4.0– 6.0 equiv., 0.5 M in DCM), trimethyloxosulfonium chloride (0.4 mmol), KOH (3.0 equiv.), THF (0.05 M), Aluminum foil light shielding. Condition b: Pyrimidine (0.2 mmol), Tf2O (1.5 equiv.), piperidine (4.0–6.0 equiv., 0.5 M in DCM), 1,1,1-trifluoroacetone (1.0 mmol), KOtBu (0.8 mmol), 1,4-dioxane (0.1 M). aIsolated yields. bYields were determined by 19F NMR using fluorobenzene as an internal standard. cN-methylaniline (6.0 equiv., 0.5 M in DCM). dN-methylaniline (4.0 equiv., 0.5 M in DCM). ePiperidine (4.0 equiv., 0.5 M in DCM). fPiperidine (6.0 equiv., 0.5 M in DCM). gYields were determined by ¹H NMR using 1,2-dichloroethane as an internal standard. hThe pyrimidine was scaled up to 7.0 mmol (1.09 g) in gram-scale experiments.
Subsequently, the scope of pyrimidine skeletal modification using the A2 synthon, trifluoroacetone, to form trifluoromethylated pyridines was also explored. Both electron-rich and electron-poor substituents on the phenyl ring of 4-phenylpyrimidine were well tolerated, leading to the formation of the corresponding 2-aryl-6-(trifluoromethyl)pyridines (3a–3 h) in good yields. 4-Naphthyl and heteroaryl-substituted pyrimidines also acted as effective A4 synthons, yielding reasonable to moderate results, as exemplified by pyridines 3i–3 m. The 4,5-disubstituted pyrimidines were also converted to the corresponding trifluoromethylpyridines (3n and 3o), albeit with lower yields and significant amounts of unreacted starting material. Similarly, structurally more complex pyrimidines smoothly underwent the skeletal modification, yielding 3p–3r in moderate yields. Unfortunately, these C4 alkyl-substituted pyrimidines and unsubstituted pyrimidine did not undergo successful conversion to the corresponding pyrrole and pyridine products (Supplementary Fig. 6). The scalability of this transformation was successfully demonstrated through 7.0 mmol scale reactions, with both the pyrrole and ortho-trifluoromethylpyridine derivatives being obtained in good isolated yields of 70% and 72%, respectively.
In addition to trifluoroacetone, other ketones could also serve as A2 synthons, undergoing this ANROFRC process with 1a under slightly modified reaction conditions with appropriate amines as ring-opening reagents (Fig. 4). Both acyclic (4-6) and cyclic (7, 8) alkyl-alkyl ketones proved to be effective A2 synthons, yielding disubstituted or trisubstituted pyridines in 33–55% yield. The reactions of pinacolone (6) and L-menthone (8) proceeded in low yields due to steric hindrance, with 34% and 31% of the corresponding open-chain intermediates remaining unreacted, respectively. The retention of the parent stereochemical configuration was observed in the formation of pyridine 8. Moreover, aryl-alkyl ketones also participated effectively in the ANROFRC process with 4-phenylpyrimidine, yielding the corresponding pyridines (9–21) in moderate to good yields. The low yield observed for pyridine 19 was again attributed to steric effects. The method was further extended to heteroaryl-alkyl ketones, with similar success. Pyridinyl (22), pyrimidinyl (23), pyrazinyl (24), imidazolyl (25), furanyl (26), thiophenyl (27), benzofuranyl (28), and benzothiophenyl (29) groups were all well tolerated, demonstrating the broad compatibility of this approach. Additionally, 2-phenylacetaldehyde successfully underwent this ANROFRC process, affording 2,5-diphenylpyridine (30) in 55% yield.
Condition: Pyrimidine (0.2 mmol), Tf2O (2.0 equiv.), Morpholine (5.0 equiv.), ketones (1.0 mmol), KOtBu (0.8 mmol), THF (0.1 M). aIsolated yield. bMorpholine (5.0 equiv., 0.5 M in DCM). cMorpholine (5.0 equiv., 0.4 M in DCM). dBase is replaced by NaH.
Mechanistic studies
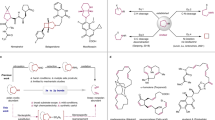
To identify the reactive intermediates generated upon the fragmentation of pyrimidines, compound 1a was subjected to the reaction with Tf2O at -78 °C for 30 min, initiating the activation process (Fig. 5A). N-Methylaniline was then added as the ring-opening reagent, and the reaction was allowed to proceed for an additional 30 min. Notably, this one-pot reaction eventually resulted in the formation of vinamidinium salt 31 in 85% isolated yield. This salt underwent efficient cyclization with trimethylsulfoxonium chloride, yielding the desired 2-phenylpyrrole (2a) in 80% yield. In a parallel experiment, piperidine instead of N-methylaniline as nucleophile led to the isolation of intermediate 32 in an excellent yield of 95%, as confirmed by X-ray single-crystal diffraction analysis. Similarly, vinamidinium salt 32 was able to react with trifluoroacetone to generate pyridine 3a in 85% yield. These results indicated that vinamidinium salts might be the key reactive fragments, acting as N-C-C-C four-atom A4 synthons in the skeletal editing of pyrimidines.
A Identification of reactive intermediates. B Proposed pathway for vinamidinium salts formation. C Transformation of 5-phenylpyrimidine. D Real-time monitoring of reactive species of 4-phenylpyrimidine and 5-phenylpyrimidine.
A plausible pathway was therefore proposed to elucidate the transformation of pyrimidine into the vinamidinium salt, based on literature precedents and our experimental observations (Fig. 5B)49,58,61. The proposed pathway begins with the nucleophilic attack of a secondary amine at the C6 position of Tf-activated pyrimidine (I), initiating ring-opening to form an aza-Zincke imine intermediate (II). The subsequent amine exchange yields an aza-streptocyanine intermediate (III), which then undergoes hydrolysis mediated by adventitious moisture in the system to generate a vinamidinium salt (32) (Supplementary Fig. 5). The intermediates II and III were observed in high-resolution mass spectrometry (HRMS, see Supplementary Information, Section 3.9), while the amide VI was often isolated as a by-product.
Interestingly, the skeletal editing of 5-phenylpyrimidine to pyrrole and pyridine was unsuccessful, with the reaction ending up with the formation of isolable vinamidinium salt 35 (Fig. 5C). However, the use of NH4OAc as an external nitrogen source facilitated the formation of the desired pyridine 36 in moderate yields, both from 5-phenylpyrimidine and intermediate 35, consistent with the previous report by Marcoux62 and McNally58. To explain the different reactivity of 4-phenylpyrimidine and 5-phenylpyrimidine, we propose the following hypothesis. In both cases, the N-C-C-C A4 synthons (32 and 34) will firstly be formed. As for 4-phenylpyrimidine, the steric hindrance of the phenyl group at the C4 position prevents nucleophilic attack by piperidine, leading to the formation of the key intermediate 32, which acts as the N-C-C-C A4 synthon. In contrast, the A4 synthon 34 derived from 5-phenylpyrimidine can easily undergo an amine exchange with piperidine, resulting in the formation of the A3 synthon 35. We subsequently conducted a real-time monitoring of reactive species for 4-phenylpyrimidine and 5-phenylpyrimidine (Fig. 5D). The results showed that 4-phenylpyrimidine gradually converted into N-C-C-C A4 synthon 32, while intermediate 33 remained at a consistently low concentration, indicating its limited reactivity. In contrast, the kinetic profile of 5-phenylpyrimidine revealed rapid formation of intermediate 34, which subsequently converted into the major C-C-C A3 synthon 35. These findings validate our hypothesis and highlight the different reactivity and mechanistic differences between 4-phenylpyrimidine and 5-phenylpyrimidine, thereby explaining their varying efficiencies in skeletal editing.
To elucidate the distinct reactivity patterns between 4-phenyl- and 5-phenylpyrimidine-derived vinamidinium salts, density functional theory (DFT) calculations were performed at the M062X-D3/6-311 + G(d,p) level with SMD dichloromethane solvation (Fig. 6). The computational results revealed that the A4-to-A3 conversion occurs via three consecutive steps: (i) C-N bond formation, (ii) hydrogen transfer, and (iii) NH3 elimination. Kinetic analysis identified the hydrogen transfer as the rate-determining step, exhibiting a 2.2 kcal/mol higher activation barrier for the 4-phenylpyrimidinium salt (ΔG‡ = 23.2 kcal/mol) compared to its 5-phenyl counterpart (ΔG‡ = 21.0 kcal/mol). This energy difference rationalizes the experimentally observed higher conversion efficiency of 5-phenylpyrimidinium salts to A3 synthons.
DFT computational study comparing the reactivity profiles of 4- and 5-phenylpyrimidine-derived vinamidinium salts (free energies in kcal/mol).
Transformations of vinamidinium salt
After identifying the A4 synthon nature of 32, we proceeded to expand its scope of application (Fig. 7). Cyclization with various dialkyl malonates—including diethyl, dimethyl, dipropyl, diisopropyl, and dibenzyl malonates—afforded the corresponding pyridone products in moderate yields (37–41). In addition, the reaction of benzamide with 32 yielded diphenylpyrimidine (42) in 44% yield. The formation of 42 was also observed when benzothioamide was used as an A2 synthon (see Supplementary Information, Section 3.7). Moreover, the reaction employing 15N labeled formamide also proceed smoothly to afford 43 in 48% yield, achieving selective 15N labeling at the N1 position of 4-phenylpyrimidine. The use of acetonitrile derivatives, such as malononitrile, phenylacetonitrile, and ethyl 2-cyanoacetate, as A2 synthons results in the formation of mutlifunctionalized 2-aminopyridines (44–46) in moderate yields. It is worth noting that vinamidinium salt 32 can also function as a C-C-C A3 synthon for nitrogen heterocycle transformations58,62. For instance, the reaction of 32 with phenylamidine gave diphenylpyrimidine 42 in 70% yield, whereas reactions with hydroxylamines and phenylhydrazine generated the five-membered isoxazoles 47, 48 and pyrazole 49, respectively, in moderate yields. The dual nature of vinamidinium salts, acting as both A4 and A3 synthons, may provide a distinct platform for the skeletal editing of pyrimidine scaffolds into diverse nitrogen heterocycles.
All reactions were performed on a 0.2 mmol scale. Isolated yields are reported unless otherwise specified. The counter anion of crystal structure is omitted for clarity. Detailed reaction conditions are provided in the Supplementary Information.
In summary, we have devised an efficient protocol that enables 4-arylpyrimidines to undergo skeletal editing via the ANROFRC reaction, facilitating scaffold hopping into a broad spectrum of nitrogen-containing heterocycles. This approach encompasses the formal denitrogenative formation of pyrroles, as well as a 6-to-6 ring transformation that yields pyridines decorated with diverse functional groups, demonstrating the versatility of this method. Moreover, this strategy enables site-specific 15N labeling of pyrimidines. The key to this successful approach lies in the in situ transformation of 4-arylpyrimidines into a distinct vinamidinium salt intermediate, which serves as versatile N-C-C-C A4 synthons. Control experiments disclosed the different reactivity of 4-arylpyrimidines and 5-arylpyrimidines, providing mechanistic insights for the future design of skeletal editing methods. The realization of vinamidinium salt intermediate as a A4 synthon, paves the way for a myriad of possibilities in the synthesis of nitrogen-containing heterocycles. The ongoing development of additional pyrimidine skeletal editing methodologies that utilize this vinamidinium salt as the intermediate is being actively pursued in our laboratory.
Methods
General procedure for the synthesis of pyrimidines
Procedure A
An oven-dried 80 mL pressure tube with a stir bar was charged with the corresponding NH4OAc (20.0 mmol, 1.0 equiv.), NH4I (1.0 mmol). The reaction tube was purged with argon three times. Then, acetophenone (10.0 mmol) and N, N-dimethylformamide dimethyl acetal (30.0 mmol) were added by a syringe. The reaction tube was stirred in an oil bath at 120 °C for 6 h. After cooling to room temperature, the reaction was diluted with DCM and water, then extracted with DCM (3x). The organic extract was dried over MgSO₄, filtered, and concentrated. The crude material was then purified by flash column chromatography.
General procedure for the synthesis of pyrroles
Procedure B
A flame-dried 25 mL Schlenk tube equipped with a stir bar was charged with the corresponding pyrimidine (0.2 mmol, 1.0 equiv.). DCM (0.5 mL) was added, and the reaction vessel was cooled to −78 °C. Then, Tf₂O (0.22 mmol, 1.1 equiv.) was added dropwise, and the reaction mixture was stirred for 30 min. Next, N-methylaniline (4.0–6.0 equiv.), dissolved in DCM (0.5 M), was added slowly (1 drop every 2 s). The reaction was stirred for an additional 30 minutes at −78 °C. The cooling bath was then removed, and the solvent was evaporated under reduced pressure. Trimethyloxosulfonium chloride (0.4 mmol, 2.0 equiv), KOH (0.6 mmol, 3.0 equiv), and THF (0.05 M) were added. The reaction was heated to 80 °C and protected from light using aluminum foil, and stirred for 16 h. After cooling to room temperature, the reaction was diluted with DCM and water, then extracted with DCM (3x). The organic extract was dried over MgSO₄, filtered, and concentrated. The crude material was then purified by preparative thin layer chromatography to give pure product.
General procedure for the synthesis of trifluoromethylpyridines
Procedure C
A flame-dried 25 mL Schlenk tube equipped with a magnetic stir bar was charged with the corresponding pyrimidine substrate (0.2 mmol, 1.0 equiv.). DCM (0.5 mL) was added, and the mixture was cooled to 0 °C. Tf₂O (0.3 mmol, 1.5 equiv.) was added dropwise, and the reaction was stirred for 30 minutes. Piperidine (4.0–6.0 equiv.), dissolved in DCM (0.5 M), was added slowly (1 drop every 2 s), and the reaction was stirred at 0 °C for an additional 30 min. The cooling bath was removed, and the solvent was evaporated under reduced pressure. Next, trifluoroacetone (1.0 mmol, 5.0 equiv.), KOtBu (0.8 mmol, 4.0 equiv.), and 1,4-dioxane (0.1 M) were added. The reaction was heated to 120 °C for 16 h. After cooling to room temperature, the reaction was diluted with DCM and water, then extracted with DCM (3 x). The organic extract was dried over MgSO₄, filtered, and concentrated. The crude material was then purified by preparative thin layer chromatography to give pure product.
Procedure for the conversion of Pyrimidines to substituted Pyridines
Procedure D
A flame-dried 25 mL Schlenk tube equipped with a magnetic stir bar was charged with the 4-phenylpyrimidine (0.2 mmol, 1.0 equiv.). DCM (0.5 mL) was added, and the mixture was cooled to 0 °C. Tf₂O (0.4 mmol, 2.0 equiv.) was added dropwise, and the reaction was stirred for 30 min. Morpholine (5.0 equiv.), dissolved in DCM (0.4–0.5 M), was added slowly (1 drop every 2 s), and the reaction was stirred at 0 °C for an additional 30 min. The cooling bath was removed, and the solvent was evaporated under reduced pressure. Next, ketones (1.0 mmol, 5.0 equiv.), KOtBu (0.8 mmol, 4.0 equiv.), and THF (0.1 M) were added. The reaction was heated to 120 °C for 16 h. After cooling to room temperature, the reaction was diluted with DCM and water, then extracted with DCM (3x). The organic extract was dried over MgSO₄, filtered, and concentrated. The crude material was then purified by preparative thin layer chromatography to give pure product.
Further experimental details are provided in the Supplementary Information.
Data availability
The details of experimental procedures and the data about the findings of this study are available within the article and its supplementary information. The X-ray crystallographic coordinates for structures reported in this study have been deposited at the Cambridge Crystallographic Data Centre (CCDC), under deposition number 2410082. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif. Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre, under deposition number CCDC 2410082 (32). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. All data are available from the corresponding authors upon request. Source data are provided with this paper.
References
Bur, S. K. & Padwa, A. The pummerer reaction: methodology and strategy for the synthesis of heterocyclic compounds. Chem. Rev. 104, 2401–2432 (2004).
Vitaku, E., Smith, D. T. & Njardarson, J. T. Analysis of the Structural diversity, substitution patterns, and frequency of nitrogen heterocycles among U.S. FDA Approved Pharmaceuticals: Miniperspective. J. Med. Chem. 57, 10257–10274 (2014).
Heravi, M. M. & Zadsirjan, V. Prescribed drugs containing nitrogen heterocycles: an overview. RSC Adv. 10, 44247–44311 (2020).
Kerru, N., Gummidi, L., Maddila, S., Gangu, K. K. & Jonnalagadda, S. B. A review on recent advances in nitrogen-containing molecules and their biological applications. Molecules 25, 1909 (2020).
Shearer, J., Castro, J. L., Lawson, A. D. G., MacCoss, M. & Taylor, R. D. Rings in clinical trials and drugs: present and future. J. Med. Chem. 65, 8699–8712 (2022).
Marshall, C. M., Federice, J. G., Bell, C. N., Cox, P. B. & Njardarson, J. T. An update on the nitrogen heterocycle compositions and properties of U.S. FDA-Approved Pharmaceuticals (2013–2023). J. Med. Chem. 67, 11622–11655 (2024).
Hu, Y., Stumpfe, D. & Bajorath, J. Recent advances in Scaffold hopping: Miniperspective. J. Med. Chem. 60, 1238–1246 (2017).
Ahmed, W. S., Geethakumari, A. M. & Biswas, K. H. Phosphodiesterase 5 (PDE5): structure-function regulation and therapeutic applications of inhibitors. Biomed. Pharmacother. 134, 111–128 (2021).
Lam, N. Y. S., Wu, K. & Yu, J. Advancing the logic of chemical synthesis: C−H activation as strategic and tactical disconnections for C−C bond construction. Angew. Chem. Int. Ed. 60, 15767–15790 (2021).
Korch, K. M. & Watson, D. A. Cross-coupling of heteroatomic electrophiles. Chem. Rev. 119, 8192–8228 (2019).
Joynson, B. W. & Ball, L. T. Skeletal Editing: Interconversion of Arenes and Heteroarenes. Helv. Chim. Acta 106, e202200182 (2023).
Li, E. Q., Lindsley, C. W., Chang, J. & Yu, B. Molecular skeleton editing for new drug discovery. J. Med. Chem. 67, 13509–13511 (2024).
Zhang, P., Hua, L., Takahashi, T., Jin, S. & Wang, Q. Recent advances in the dearomative skeletal editing of Mono- azaarenes. Synthesis 56, 55–70 (2024).
Li, X. & Xu, Z. Skeletal editing: ring insertion for direct access to heterocycles. Molecules 29, 1920 (2024).
Zhang, R., Wen, K. & Dong, G. Downsizing Lactams via Rh-Catalyzed C–C Activation. Chem. https://doi.org/10.1016/j.chempr.2025.102622 (2025).
Peng, Y., Wang, G., Klare, H. F. T. & Oestreich, M. Ring contraction of saturated cyclic amines and rearrangement of acyclic amines through their corresponding hydroxylamines. Angew. Chem. Int. Ed. 63, e202410483 (2024).
Liu, Z., Zhang, X., Sivaguru, P. & Bi, X. Triftosylhydrazone in single-atom skeletal editing. Acc. Chem. Res. 58, 130–149 (2025).
Xu, M.-M., Cao, W.-B., Xu, X.-P. & Ji, S.-J. Efficient synthesis of 2-arylquinazolin-4-amines via a copper-catalyzed diazidation and ring expansion cascade of 2-arylindoles. Chem. Commun. 54, 12602–12605 (2018).
Dherange, B. D., Kelly, P. Q., Liles, J. P., Sigman, M. S. & Levin, M. D. Carbon Atom insertion into pyrroles and indoles promoted by chlorodiazirines. J. Am. Chem. Soc. 143, 11337–11344 (2021).
Ma, D., Martin, B. S., Gallagher, K. S., Saito, T. & Dai, M. One-carbon insertion and polarity inversion enabled a pyrrole strategy to the total syntheses of pyridine-containing lycopodium alkaloids: complanadine A and Lycodine. J. Am. Chem. Soc. 143, 16383–16387 (2021).
Wang, J., Lu, H., He, Y., Jing, C. & Wei, H. Cobalt-catalyzed nitrogen atom insertion in arylcycloalkenes. J. Am. Chem. Soc. 144, 22433–22439 (2022).
Patel, S. C. & Burns, N. Z. Conversion of aryl azides to aminopyridines. J. Am. Chem. Soc. 144, 17797–17802 (2022).
Liu, S. & Cheng, X. Insertion of ammonia into alkenes to build aromatic N-heterocycles. Nat. Commun. 13, 425 (2022).
Reisenbauer, J. C., Green, O., Franchino, A., Finkelstein, P. & Morandi, B. Late-stage diversification of indole skeletons through nitrogen atom insertion. Science 377, 1104–1109 (2022).
Hyland, E. E., Kelly, P. Q., McKillop, A. M., Dherange, B. D. & Levin, M. D. Unified access to Pyrimidines and Quinazolines Enabled by N–N Cleaving Carbon Atom Insertion. J. Am. Chem. Soc. 144, 19258–19264 (2022).
Joynson, B. W., Cumming, G. R. & Ball, L. T. Photochemically mediated ring expansion of indoles and pyrroles with chlorodiazirines: synthetic methodology and thermal hazard assessment. Angew. Chem. Int. Ed. 62, e202305081 (2023).
Pearson, T. J. et al. Aromatic nitrogen scanning by ipso -selective nitrene internalization. Science 381, 1474–1479 (2023).
Li, L. et al. Dearomative insertion of fluoroalkyl carbenes into azoles leading to fluoroalkyl heterocycles with a quaternary center. Angew. Chem. Int. Ed. 63, e202313807 (2024).
Boudry, E., Bourdreux, F., Marrot, J., Moreau, X. & Ghiazza, C. Dearomatization of pyridines: photochemical skeletal enlargement for the synthesis of 1,2-Diazepines. J. Am. Chem. Soc. 146, 2845–2854 (2024).
Luo, J., Zhou, Q., Xu, Z., Houk, K. N. & Zheng, K. Photochemical skeletal editing of pyridines to bicyclic pyrazolines and pyrazoles. J. Am. Chem. Soc. 146, 21389–21400 (2024).
Li, C. et al. C-F bond insertion into indoles with CHBr2F: an efficient method to synthesize fluorinated quinolines and quinolones. Chin. J. Chem. 42, 1128–1132 (2024).
Zhang, X. et al. Asymmetric dearomative single-atom skeletal editing of indoles and pyrroles. Nat. Chem. https://doi.org/10.1038/s41557-024-01680-0 (2024).
Ghosh, B. et al. Sulfenylnitrene-mediated nitrogen-atom insertion for late-stage skeletal editing of N-heterocycles. Science 387, 102–107 (2025).
Kafle, P., Herndon, D. & Sharma, I. Sulfenylcarbene-Mediated Carbon Atom Insertion for the Late-Stage Functionalization of N-Heterocycles. J. Am. Chem. Soc. https://doi.org/10.1021/jacs.5c02012 (2025).
Liu, L. et al. Indole-quinoline transmutation enabled by a formal rhodium carbynoid. Angew. Chem. Int. Ed. e202501966 https://doi.org/10.1002/anie.202501966 (2025).
Chen, Z. & Trudell, M. L. Chemistry of 7-Azabicyclo [2.2.1]hepta-2,5-dienes, 7-Azabicyclo[2.2.1]hept-2-enes, and 7-Azabicyclo[2.2.1]heptanes. Chem. Rev. 96, 1179–1194 (1996).
Blackman, M. L., Royzen, M. & Fox, J. M. Tetrazine ligation: fast bioconjugation based on inverse-electron-demand diels−alder reactivity. J. Am. Chem. Soc. 130, 13518–13519 (2008).
Cabrera-Pardo, J. R., Chai, D. I., Liu, S., Mrksich, M. & Kozmin, S. A. Label-assisted mass spectrometry for the acceleration of reaction discovery and optimization. Nat. Chem. 5, 423–427 (2013).
Lee, S., Jena, R. & Odom, A. L. Substituted pyridines from isoxazoles: scope and mechanism. Org. Biomol. Chem. 20, 6630–6636 (2022).
Cheng, Q. et al. Skeletal editing of pyridines through atom-pair swap from CN to CC. Nat. Chem. 16, 741–748 (2024).
Xiao, W. et al. Molecular editing of Pyrroles to Benzenes/Naphthalenes by N2O Deletion. Angew. Chem. Int. Ed. 63, e202411166 (2024).
Wang, T. et al. Skeletal editing of Pyridine and Quinoline N-Oxides Through Nitrogen to Carbon Single Atom Swap. CCS Chem. https://doi.org/10.31635/ccschem.024.202404133 (2024).
Xu, P. & Studer, A. Skeletal editing through cycloaddition and subsequent cycloreversion reactions. Acc. Chem. Res. 58, 647–658 (2025).
Tian, D. et al. Switchable skeletal editing of quinolines enabled by cyclizative sequential rearrangements. Nat. Chem. https://doi.org/10.1038/s41557-025-01793-0 (2025).
ANRORC Rearrangement: (Degenerate Ring Transformation of Heterocycles). in Comprehensive Organic Name Reactions and Reagents 87–90 (Wiley,). https://doi.org/10.1002/9780470638859.conrr020. (2010).
Morofuji, T., Kinoshita, H. & Kano, N. Connecting a carbonyl and a π-conjugated group through a p-phenylene linker by (5+1) benzene ring formation. Chem. Commun. 55, 8575–8578 (2019).
Xu, K. et al. Synthesis of 2-Formylpyrroles from Pyridinium Iodide Salts. Org. Lett. 22, 6107–6111 (2020).
Morofuji, T., Inagawa, K. & Kano, N. Sequential ring-opening and ring-closing reactions for converting para-substituted pyridines into meta -Substituted Anilines. Org. Lett. 23, 6126–6130 (2021).
Bartholomew, G. L., Carpaneto, F. & Sarpong, R. Skeletal editing of pyrimidines to pyrazoles by formal carbon deletion. J. Am. Chem. Soc. 144, 22309–22315 (2022).
Woo, J. et al. Scaffold hopping by net photochemical carbon deletion of azaarenes. Science 376, 527–532 (2022).
Woo, J., Stein, C., Christian, A. H. & Levin, M. D. Carbon-to-nitrogen single-atom transmutation of azaarenes. Nature 623, 77–82 (2023).
Morofuji, T., Nagai, S., Watanabe, A., Inagawa, K. & Kano, N. Streptocyanine as an activation mode of amine catalysis for the conversion of pyridine rings to benzene rings. Chem. Sci. 14, 485–490 (2023).
Tang, J. et al. Tandem Ring-Contraction/Regioselective C–H Iodination Reaction of Pyridinium Salts. J. Org. Chem. 88, 2809–2821 (2023).
Wu, F. P. et al. Nitrogen-to-functionalized carbon atom transmutation of pyridine. Chem. Sci. 15, 15205–15211 (2024).
Conboy, A. & Greaney, M. F. Synthesis of benzenes from pyridines via N to C switch. Chem 10, 1940–1949 (2024).
Du, X. et al. Skeletal editing of pyrimidines: new approaches to diverse high energy density materials. Mater. Chem. Front. 9, 894–909 (2025).
Shi, W. et al. Pyrimidines to Pyridines: two atom swap skeletal editing. CCS Chem. 1–7 https://doi.org/10.31635/ccschem.025.202405166 (2025).
Uhlenbruck, B. J. H., Josephitis, C. M., De Lescure, L., Paton, R. S. & McNally, A. A deconstruction–reconstruction strategy for pyrimidine diversification. Nature 631, 87–93 (2024).
Li, S. et al. C3 Selective chalcogenation and fluorination of pyridine using classic Zincke imine intermediates. Nat. Commun. 15, 7420 (2024).
Toscano, R. A. et al. Nucleophilic Reactions on 1-Trifluoromethanesulfonylpyridinium Trifluoromethanesulfonate (Trifylpyridinium Triflate, TPT). Ring-Opening and “Unexpected” 1,4-Dihydropyridine Reaction Products. Chem. Pharm. Bull. 45, 957–961 (1997).
Bartholomew, G. L. et al. 14N to 15N isotopic exchange of nitrogen heteroaromatics through skeletal editing. J. Am. Chem. Soc. 146, 2950–2958 (2024).
Marcoux, J. F. et al. A general preparation of pyridines and pyridones via the annulation of ketones and esters. J. Org. Chem. 66, 4194–4199 (2001).
Acknowledgements
We acknowledge the financial support from the National Natural Science Foundation of China (22072099), the Sichuan Science and Technology Program (2024YFFK0016; 2024ZYD0099), the Fundamental Research Funds for the Central Universities (1082204112I96; 0082604151502), and Leshan Normal University Scientific Research Start-up Project for Introduction High-level Talents (RC202516, RC2025018). We also thank Chunchun Zhang from the Centre of Analysis & Testing, Dongyan Deng, Jing Li from the College of Chemistry, Sichuan University for NMR and HRMS measurements.
Author information
Authors and Affiliations
Contributions
S.L. and Y.S. initiated and conducted the primary experiments, analyzed data, and contributed to the drafting of the manuscript; J.T., M.Y., S.H., T.D., S.W., and X.J. contributed to experiments and characterization of products; Z.S. contributed to DFT calculation.; W.J., J.X., X.Z., and R.L. contributed to the discussion on the study; H.F., W.X. and H.C. supervised the research and provided critical revisions to the manuscript. All authors contributed to this research project through in-depth discussions, data analysis, and result interpretation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Xue-Jing Zhang and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, S., Shi, Y., Tang, J. et al. Skeletal editing of 4-arylpyrimidines into diverse nitrogen heteroaromatics via four-atom synthons. Nat Commun 16, 7112 (2025). https://doi.org/10.1038/s41467-025-62547-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-62547-7
This article is cited by
-
Chiral hypervalent iodine catalyzed stereoselective skeletal editing of pyrimidine fused heterocycles
Nature Communications (2025)