Abstract
Pt/CeO2 ensemble catalysts are promising for propylene (C3H6) oxidation in vehicle exhaust, yet identifying the intrinsic active sites and understanding how the metal-support interface evolves at varying reaction temperatures remains contentious. Herein, we demonstrate that H2-activated Pt/CeO2 ensemble catalysts feature metallic Pt ensembles as intrinsic active sites, lowering the 50% conversion temperature by 120 °C after hydrogen activation. Various operando characterization techniques reveal an approximately 170 °C threshold temperature for the dynamic change of the reaction models. Meanwhile, kinetics and theoretical analysis illustrates that oxygen-facilitated dehydrogenation of sp3 C-H bonds is the rate-determining step. At low temperatures, both C3H6 and O2 adsorb and activate on metallic Pt, without CeO2 involvement. Once the temperature exceeds threshold, C3H6 fully covers Pt sites, while O2 activates over Pt-O-Ce interfaces and participates in dehydrogenation. This study highlights the dynamic nature of oxygen activation, leading to distinct reaction temperature regimes during C3H6 oxidation.
Similar content being viewed by others
Introduction
Pt is recognized as one of the most active components for diesel oxidation catalysts and three-way catalysts in vehicle emission control1,2. CeO2 has been commonly used as the supporting materials for Pt-based emission control catalysts, due to the decent oxygen storage capacity and excellent reducibility1. Wherein, the defect sites and sufficient active oxygen species at the interfacial sites contributed to anchoring Pt active species and promoting low-temperature oxidation activity3,4. In the past decade, atomically dispersed Pt catalysts received much attention owing to the maximum utilization of precious metals. Especially, Pt single-atom catalysts (Pt SACs) exhibited outstanding reactivity towards CO oxidation5,6. However, the absence of Pt ensemble sites (Pte) still constrains the catalytic activity towards alkene (such as C3H6) oxidation7. Therefore, great efforts have been devoted to fabricating Pt ensemble catalysts that could effectively accomplish the cleavage of C–H methyl or C = C double bonds during C3H6 oxidation8,9. It is still challenging to elucidate the intrinsic active species of Pte/CeO2 ensemble catalysts and clarify the specific reaction pathways for the catalytic oxidation of C3H6.
The diversity in valence states (electronic properties) was one of the decisive aspects determining the reactivity of Pte catalysts, besides considering the geometry and size as critical factors. So far, it remains debated the oxidation state in charge of the intrinsic activity of atomically dispersed Pt catalysts. It was reported that Pt0 or Ptᵟ+ were the intrinsic active sites for catalytic oxidation of C3H6 from the previous literature10,11,12, which might be strongly affected by the catalyst support and promoters. The contentious issue of the oxidation state of Pt catalysts has also been investigated with CO oxidation, with intrinsic similarity to C3H6 oxidation. For example, Maurer et al. indicated that \({\mathrm{Pt}}_{{\rm{X}}}^{{\rm{\delta }}+}\) forming during catalytic oxidation of CO were the exclusive active sites13. Ding et al., however, discovered that Pt0 was the sole active phase and Ptᵟ+ was the spectator for catalytic oxidation of CO14. Therefore, complementary investigation of intrinsic Pte species with specific electronic properties could provide a guide for the rational design of highly efficient Pte catalysts.
Regarding the reaction pathway of catalytic oxidation of C3H6, either the reactive oxygen species (superoxide and peroxide) transformed from gaseous O2 or lattice oxygen from interfacial sites might engage in catalytic oxidation of C3H6. On one hand, C3H6 could adsorb over the catalyst surface, and the reaction took place between the C3H6 molecules and reactive oxygen species to form intermediates10,15. On the other hand, C3H6 oxidation might follow the Mars-van Krevelen mechanism, in which C3H6 oxidation could react with lattice oxygen, causing the appearance of anion vacancies, followed by the re-oxidation of catalysts by gaseous oxygen in a separate step16. Yet, it might be subjective to directly conclude the catalytic oxidation reaction model without considering the reaction temperatures. For instance, the dynamic toluene oxidation mechanism variation was triggered by the activation of the different active oxygen species17,18. In the low-temperature regime, gaseous oxygen molecules were directly converted to adsorbed oxygen species to facilitate the toluene oxidation17,18. With the increased temperature, the lattice oxygen from the bulk phase of CeO2 supports gradually migrated to the interface and acted as the active surface lattice oxygen species to drive toluene oxidation17,18. A similar trend was detected for a dynamic transition of reaction mechanism from Langmuir-Hinshelwood to Mars-van Krevelen for toluene oxidation with rising temperature19. Furthermore, Li et al. found that the oxygen vacancy at the interfacial sites between Pt ensembles and CeO2 was inactive in the low-temperature domain for the water-gas shift reaction. Once the temperature exceeded 180 °C, oxygen vacancy-Ce3+ sites were stimulated and imitated to convert and supply active oxygen species to the interface20. Based on the above research, there might be a threshold temperature determining the oxygen activation to participate in the C3H6 oxidation reactions over Pte/CeO2 catalysts. It is valuable to investigate the dynamic evolution properties to explicit the possible change in the reaction mechanism during the different temperature ranges. The results will benefit the understanding of the specific catalytic roles of Pte catalysts in emission control applications.
Herein, Pt ensembles were loaded over the CeO2 supports (Pte/CeO2) via incipient wetness impregnation, which was further activated by H2 reduction to improve the catalytic performance for C3H6 oxidation. High-angle annular darkfield scanning transmission electron microscopy (HAADF-STEM), extended X-ray adsorption fine structure (EXAFS), X-ray photoelectron spectroscopy (XPS), and catalytic performance tests unraveled that H2 activation constructed metallic Pt ensembles locating at upper tiers of CeO2 serving as the intrinsic active sites. In situ Raman spectra, near ambient pressure X-ray photoelectron spectroscopy (NAP-XPS), and Electron energy loss spectroscopy (EELS) results demonstrated that gaseous oxygen was activated at Pt-O-Ce interfacial sites to promote C3H6 oxidation above 170 °C, acting as the threshold temperature. In situ Diffuse Reflectance Infrared Fourier Transform Spectroscopy (DRIFTS), rigorous kinetic studies, and Density Functional Theory (DFT) calculations affirmed that the C3H6 coverage change and oxygen activation at the interfacial sites caused the dynamic transformation of the reaction models. The results will guide the precise design of Pte catalysts and be also helpful for the understanding of their catalytic roles under varying reaction temperatures.
Results
Evaluation of the catalytic performance of Pte/CeO2 catalysts
The study first tried to measure C3H6 oxidation light-off performance over H2-activated Pte/CeO2 catalysts, which significantly boosted the catalytic oxidation performance and could help explore the transformation of Pt ensemble structure and size during H2 activation at different temperatures (Supplementary Fig. 1). As shown in Fig. 1a, Pte barely reached 50% conversion of C3H6 (T50) at approximately 282 °C, Meanwhile, Pte-300A (Pte after H2 reduction pretreatment at 300 °C) could significantly shift T50 to low temperatures around 160 °C. According to Fig. 1b, H2 activation caused a decline of the apparent activation energies from 138.0 to 111.5 kJ/mol for Pte and Pte-300A, respectively. It was noteworthy that Pte-300A catalysts achieved an exceptional catalytic consumption rate for C3H6 oxidation compared to other Pt-based catalysts as shown in Supplementary Table 1, proving the considerable activity of activated Pte-300A catalysts (Fig. 1c). A similar promoting trend of H2 activation was mirrored for the catalytic oxidation activities of C3H6 and/or CO, and the apparent activation energies of CO oxidation also dropped from 67.3 to 41.5 kJ/mol (Supplementary Figs. 2 and 3). Furthermore, the as-synthesized Pte-300A catalysts showed the same order of magnitude in CO consumption rate as the previously reported Pt/CeO2 catalysts from Supplementary Table 2. The above results indicated that the H2 activation could successfully fabricate efficient Pte/CeO2 oxidation catalysts.
a C3H6 oxidation light-off curves. Reaction condition: 1000 ppm C3H6, and 10% O2 in N2 balance with a WHSV of 240,000 mL g−1 h−1; b Arrhenius plots of C3H6 oxidation; c Comparison of the reactivity of C3H6 oxidation between Pte-300A and previously reported Pt-based catalysts. For details for comparison, see Supplementary Table 1.
The synergistic interaction between Pt ensembles and CeO2 was confirmed by comparing the catalytic performance between Pt/CeO2 and Pt/γ-Al2O3 catalysts. To explore the interface effect of Pt metals and CeO2 support, the catalytic performance of Pt/γ-Al2O3 with inert supports was also examined. Supplementary Fig. 4 exhibited that H2-activated Pte-300A had better catalytic reactivity at a low-temperature regime than Pte/γ-Al2O3-300A catalysts. It indicated that the synergistic interaction between Pt and reducible CeO2 supports was important in governing catalytic oxidation activity over Pte-300A catalysts. It is noteworthy that the physically mixed Pte/γ-Al2O3&CeO2-300A catalysts obtained comparable activity toward C3H6 and CO oxidation to Pte/γ-Al2O3-300A catalyst. It implied that the proximity mattered for Pte/CeO2 catalysts, and the synergistic interactions only occurred at the Pt-O-Ce interfacial sites. Moreover, H2 activation had little effect on the catalytic oxidation activity of bare CeO2 supports for C3H6 oxidation (Supplementary Fig. 5). The results revealed the transformation of Pt active sites after H2 activation and synergistic interactions over Pt-O-Ce interfacial sites were two key factors in elevating oxidation performance of Pt/CeO2 catalysts.
Water vapor is a common component as a key element influencing the performance and longevity of Pt-based emission control catalysts21,22. The co-feeding of 5% H2O did not reduce the light-off performance of C3H6 oxidation over Pte-300A (Supplementary Fig. 6). The catalytic activity remained stable in the presence of 5% H2O, highlighting the promise of Pte-300A in practical applications for vehicle emission control (Supplementary Fig. 7). Meanwhile, Pte-300A catalysts was pretty stable without any change of Pt valence states during the stability tests, based on the XPS spectra of Pt 4f core-level analysis (Supplementary Fig. 8). No deactivation was observed for Pte-300A samples after cycling tests, also confirming the thermal stable properties of H2-activated catalysts (Supplementary Fig. 9).
HAADF-STEM was carried out to analyze the geometry of Pt species on pristine and activated catalysts, which might be undergoing a dramatic reconstruction after H2 activation. A few Pt single atoms and a single-layer Pt ensemble with a mean diameter of 0.45 nm could be detected on pristine Pte catalysts (Fig. 2a and Supplementary Fig. 10a, b). H2 activation caused a transformation of Pt species to form multilayer Pt ensembles, with a mean diameter of 0.84 nm (Fig. 2b and Supplementary Fig. 10c, d). In situ DRIFTS experiments of CO adsorption were further performed to investigate the chemical states of surface Pt species. As shown in Fig. 2c, three major peaks at 2094, 2079, and 2036 cm−1 were deconvoluted from the IR bands on Pte catalysts, which could be ascribed to linearly bound CO adsorbed on Pt single atoms, the well-coordinated terrace sites, and the under-coordinated corner sites on Ptᵟ+ ensembles, respectively23,24,25. For Pte-300A catalysts with hydrogen activation, an intense band with two sub-peaks at approximately 2051 and 2033 cm-1 appeared, corresponding to the linear CO adsorption at the well-coordinated terrace sites and under-coordinated edge sites within Pt0 ensembles, respectively26,27,28. The extra peak at 1983 cm−1 could be attributed to the bridging bound CO adsorbed on Pt ensembles29, affirming an enlarged Pt ensembles after H2 activation30. Moreover, the band at 2072 cm−1 remained on Pte-300A as the CO linearly bound on unreduced oxidized Pt species. These newly created bands illustrated that the Pt ensembles over Pte-300A catalysts were composed of both Pt0 and Ptᵟ+ sites.
HAADF-STEM images of a Pte and b Pte-300A (yellow cycle: Pt single atoms; red dashed cycle: planner single-layer Pt ensembles; red solid cycle: multilayer Pt ensembles); c in situ DRIFTS spectra of CO adsorption at 30 °C on Pte and Pte-300A; d normalized Pt L3-edge XANES and e Fourier-transformed k2-weighted EXAFS spectra in R space for Pte and Pte-300A; f wavelet transform plot of Pt L3-edge EXAFS spectra for Pt foil, PtO2, Pte, and Pte-300A samples.
The XAS data were further collected to elucidate the changes in the oxidation states and specific local coordination environments of Pt species after H2 activation. Figure 2d compared the X-ray absorption near edge structure (XANES) spectra between Pte and Pte-300A catalysts using Pt foil and PtO2 as the reference samples. The edge position within Pt L3-edge XANES data of Pte was close to that of PtO2, suggesting the single-layer Pt ensembles mainly existed in the highly oxidized states. Furthermore, the edge position shifted to lower energy after H2 activation, suggesting a more reduced Pt state over Pte-300A versus as-prepared Pte samples. As displayed in Fig. 2e, the EXAFS spectra were plotted in R space to investigate the local structures. Pte catalysts only demonstrated the first Pt-O coordination shell at 2.00 Å, implying that Pt ensembles were predominantly bound with the surface oxygen on CeO2 (100) facets. The fitting model of the Pte-300A catalysts included both metallic Pt–Pt and Pt–O bonds, which indicated the formation of Pt0 ensemble sites over the top layer of Ptᵟ+ planar (Supplementary Figs. 11 and 12). Meanwhile, the shorter Pt-Pt bond distance over Pte−300A (2.74 Å), compared to the Pt foil (2.76 Å), could be ascribed to the increment of the local electron density between two adjacent metal atoms triggered by the rehybridization of the spd orbitals in metal clusters31,32. This phenomenon usually occurs in the small metal nanoclusters33, coinciding with the relatively small average diameter of the Pt ensembles over Pte−300A catalysts. Additionally, Fig. 2f showed the wavelet transform analysis based on Pt L3-edge EXAFS oscillations, where the Pte-300A plots comprised Pt–O and Pt–Pt bonds. In contrast, only Pt-O bonds could be identified over Pte catalysts, revealing that H2 activation created metallic Pt ensembles as the major species, accompanied by partially oxidized Pt. Additional XRD and N2 physisorption results proved that H2 activation did not significantly affect the textural properties of CeO2 supports (Supplementary Figs. 13, 14).
XPS experiments were performed to measure the chemical states of the supported Pt species. As shown in Supplementary Fig. 15 and Supplementary Table 3, only Pt2+ and Pt4+ were detected on Pte samples, suggesting that highly oxidized Pt species exclusively survived on CeO2. Metallic Pt was formed after H2 activation, the deconvoluted doublets shifted to 71.8, 72.7, 75.1, and 76.0 eV, corresponding to Pt0 or Pt2+ in Pt 4f7/2 spectra and Pt 4f5/2 spectra, respectively34,35. H2-TPR profiles (Supplementary Fig. 16) further demonstrated a significant declination of the relative area in the range of 100–250 °C on Pte-300A catalysts, indicating the disappearance of oxidized Pt species such as PtOx36, which agreed with the lowered coordination number of Pt–O bond between Pte and Pte-300A (4.2 ± 0.2 vs. 1.5 ± 0.1) from Supplementary Table 4.
Since two different Pt sites, including Pt0 and Ptᵟ+, have been detected on Pt ensembles, it is unavoidable to directly compare the reactivity on these sites regarding C3H6 oxidation quantitatively, which would further validate the rationality of metallic Pt ensembles as the unique intrinsic active sites. On one hand, DFT calculations were first conducted to precisely analyze the free energy changes of the oxygen-facilitated dehydrogenation of the sp3 hybrid C-H bonds, which was recognized as the rate-determining step (RDS) for catalytic oxidation of propylene in the following section. As shown in Supplementary Figs. 17 and 18, Pt0 sites on the top layers obtained a much lower energy barrier (1.53 eV) than both Ptδ+ at the bottom sites (2.41 eV) and single-layer Pt ensembles on pre-activated Pte catalysts (1.68 eV) for the dehydrogenation process, which led to the much better catalytic activity of C3H6 oxidation on metallic Pt sites formed during the H2-trigged structural evolution. Additionally, it was consistent that the abstraction of the C-H bonds on the methyl group over metallic Pt ensemble sites exhibited a similar magnitude of activation energy barrier, compared to the identical process that took place on Pd/Cu55 clusters (1.43 eV)37 and Pt2Sn/Pt(111) surface (1.63 eV) catalysts38. Supplementary Fig. 19 demonstrated density of state (DOS) results based on the d-orbital of Pt ensembles for different sites, where the d-band of Pt0 was centered at a higher energy (−1.98 eV) compared to Ptᵟ+ (−2.58 eV), indicating an increment of the adsorption reactivity accompanied with the facilitation of C3H6 adsorption on metallic Pt sites39. As illustrated in Supplementary Fig. 20, the Bader charges of the upper-tier Pt0 and bottom-layer Ptᵟ+ over CeO2 surface were calculated to be −0.13e and +0.30e, respectively. It revealed that metallic Pt ensemble sites could provide many more electrons for the C3H6 adsorbed molecules. Moreover, the difference in charge density of the oxygen-facilitated dehydrogenation step illustrated a more frequent electron transfer over Pt0 than Ptᵟ+ sites, where the correlated results were affirmed by the charge density difference of 1.06e and 0.89e for −CH3 activation with the assistance of oxygen over Pt0 and Ptᵟ+ sites, respectively. Furthermore, DOS calculations for C3H6 adsorbed at Pt0 and Ptᵟ+ sites revealed distinct electronic interactions shown in Supplementary Fig. 21. At the Pt0 site, a significant orbital hybridization between C and Pt occurred within the energy range from −5 to −10 eV. The broad overlap across multiple energy levels indicated strong electron cloud interactions. The C-Pt orbital hybridization was also present over the Ptᵟ+ site, yet the hybridized peaks exhibited markedly reduced intensity and narrower energy distribution. Meanwhile, strong hybridization at the Pt0 site shifted the d-band center to lower energies to −3.02 eV, in comparison to −2.07 eV at the Ptᵟ+ site. Collectively, these results demonstrated more stable C3H6 adsorption at top adsorption sites and stronger interfacial interactions, thereby facilitating subsequent C3H6 activation. On the other hand, the comparison of inherent catalytic oxidation activity between Pt0 and Ptᵟ+ sites was also elucidated by FTIR tests, where CO was used as the titration gas to measure the reactivity of different Pt sites. The transient reactions were conducted between the saturated adsorbed CO and the flowing O2 (Supplementary Fig. 22). Metallic Pt exhibited a rapid CO consumption rate over Pte-300A catalysts, confirming its role served as the sole intrinsic active site for catalytic oxidation, demonstrating significantly better reactivity than Ptᵟ+. The universality of the metallic Pt ensembles was further examined on Pte/γ-Al2O3 catalysts. Supplementary Figs. 23–25 additionally confirmed a positive correlation between the increased catalytic oxidation reactivity and the rising ratio of metallic Pt sites regarding Pte/γ-Al2O3 catalysts with H2 activation, validating that metallic Pt functioned as the intrinsic active sites on Pt ensemble clusters supported by γ-Al2O3.
Investigation of interfacial property by in situ characterization techniques
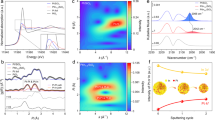
The dynamic change of the reaction model was studied using in situ characterization methods to uncover the mystery of C3H6 oxidation over Pte-300A at different temperatures. Firstly, in situ Raman spectra were measured using the identical reaction conditions as light-off tests. Figure 3a, b demonstrated two prominent bands at 458 and 588 cm−1, symbolizing the F2g symmetry mode of the CeO2 fluorite structure and the defect-induced mode, respectively40,41. Meanwhile, the band at 859 cm−1 could be assigned to peroxide species (O22−)40, and the bands at 1059 and 1166 cm−1 could be assigned to the in-plane bend of C–H species19. The intensity ratio of ID/IF2g remained constant around 23.0% as the temperature rose from 100 to 160 °C, revealing that the concentration of oxygen vacancies was relatively stable. Once the reaction temperature was further elevated and exceeded 170 °C, the oxygen vacancy defects were gradually annihilated due to the adsorption and activation of adsorbed oxygen molecules. At the same time, the concentrations of peroxide species were also decreased above 170 °C, which was in line with their catalytic roles in C3H6 oxidation. Moreover, an equivalent phenomenon was detected over CO oxidation in Supplementary Fig. 26, where the ID/IF2g ratio abruptly dropped above 170 °C, confirming a similar oxygen activation process stimulated and participated in CO oxidation. Secondly, NAP-XPS spectra of Ce3d were displayed in Fig. 3c and Supplementary Fig. 27 to trace the transformation in chemical valence during the heating process. From 100 to 160 °C, the fraction of Ce3+ species was relatively stable at approximately 25%. When the reaction temperature increased above 170 °C, the Ce3+ concentration gradually declined. It illustrated that the ratio of Ce3+/(Ce3++Ce4+) showed a decreased trend with ramping reaction temperatures, implying that the oxygen vacancy defects over Pt-O-Ce interfacial sites were gradually replenished during the reaction at high temperatures. Lastly, the Ce3+ ratio at the interfacial sites after catalytic reactions below and above the threshold temperature of approximately 170 °C was probed by EELS to unambiguously verify the change of surface vacancies. As shown in Fig. 3d, e, the interfacial section of Pte-300A catalysts exhibited a more pronounced yellowish hue in the color bar after the C3H6 oxidation reaction at 162 °C compared to that at 188 °C, indicating a higher Ce3+ ratio at the lower temperature. This observation suggests that oxygen vacancy defects were filled by activated oxygen during C3H6 oxidation with ramping temperature and cause a greater proportion of Ce4+ formation. By combining in situ Raman spectra, NAP-XPS, and EELS results, it could be concluded that 170 °C was the threshold temperature for C3H6 oxidation on Pte-300A catalyst by different reaction pathways.
a In situ Raman spectra of C3H6 oxidation over Pte-300A from 100 to 250 °C; b Variation of peroxide ratio and ID/IF2g ratio in the range of 100 to 225 °C; c NAP-XPS of Ce 3d spectra at different temperatures; d, e EELS result of Pte-300A after catalytic reactions at 162 °C and 188 °C, respectively.
Evaluation of C3H6 oxidation mechanism by varying reaction temperatures
In situ DRIFTS experiments of steady-state reactions at various temperatures were further conducted over Pte and Pte-300A catalysts to elucidate different surface intermediates during C3H6 oxidation, and the detailed assignment of IR spectra was ascribed to Supplementary Table 5. As shown in Supplementary Fig. 28, the bands at 1660, 1274, and 1622 cm−1 were observed at 30 °C on Pte, which could be attributed to C = C stretching, CH2 deformation of gaseous C3H6, and C = C stretching of the adsorbed C3H6 molecules, respectively42,43,44. Once the temperature was increased to 150 °C, gaseous C3H6 completely vanished, suggesting C3H6 activation at high temperatures. The characteristic bands at 1240 and 1284 cm−1 appeared when the reaction temperature reached 200 °C, corresponding to the generation of surface acrolein and acrylate45,46. These species would be further converted to acetate (1463 and 1405 cm−1)46. As shown in Fig. 4a, b, the IR spectra of Pte-300A demonstrated complex adsorbed species at 30 °C. The bands at 1496 cm−1 and 1435 cm−1 were attributed to π-allylic intermediates, which were generated by the hydrogen abstraction from the weak methyl group with sp3 hybridization47,48. Once the temperature reached 100 °C, gaseous C3H6 still presented at ca. 1658 and 1265 cm−1, while acrolein at 1268 cm−1 was generated as the successive intermediates46. Acrylate exhibited intensive bands at approximately 1640 and 1288 cm−1, while acetate could be observed at 1459 and 1395 cm−1 above 200 °C49. The intensity of acetate was gradually increased with further ramping temperature, suggesting the facilitation of acetate generation at high temperatures. It should be noted that formate species might also be formed accompanying acetate generation, due to the destructive oxidation of C3H6 by breaking C = C bonds. Yet, no obvious formate species were detected in the IR spectra, probably due to the thermal decomposition above 200 °C. Moreover, compared to the situation on Pte sample, many distinctive bands of acetate were observed at corresponding temperatures, while the characteristic peaks of CO2 were only detected on the surface of Pte-300A. Therefore, it suggested that the metallic Pt ensemble sites on H2-activated catalysts stimulated the oxidation process of acrylate to acetate, which could be recognized as a prior step in producing the final products of CO2 and H2O.
a In situ DRIFTS spectra of steady-state C3H6 and O2 co-adsorption; b Contour graphs for C3H6 oxidation; c, d Effects of C3H6 and O2 partial pressures on C3H6 consumption rate over Pte-300A at 162 °C and 188 °C, respectively; e DFT calculations of C3H6 oxidation mechanisms and energy barriers with detailed transition states and free energy change.
Rigorous kinetic studies were further conducted to evaluate the elementary reaction steps of C3H6 oxidation over Pte-300A catalysts. Figure 4c exhibited a sub-linear dependence between the C3H6 consumption rate on the partial pressures of C3H6 and O2 at 162 °C, which revealed classic Langmuir-Hinshelwood models on Pte-300A catalysts. Based on the kinetics data at 162 °C, the elementary steps of C3H6 oxidation could be deduced and summarized in Supplementary Fig. 29. Initially, the gaseous O2 was reversibly adsorbed on the vacancy sites (*) over metallic Pt ensemble sites (step 1), and then dissociated to produce the adsorbed O* atoms (step 2). Meanwhile, the quasi-equilibrium adsorption of C3H6 molecules occurred on the same sites and generated C3H6* (step 3). Subsequently, the dissociated O* kinetically activated the C-H bond by irreversibly coupling with the C3H6* to facilitate dehydrogenation and form surface-adsorbed C3H5* and OH* (step 4), which was recognized as RDS in the whole reaction process. The kinetics models of oxygen-facilitated dehydrogenation were similar to the catalytic oxidation of C3H6 over Ag/Al2O3 cluster catalysts50. The following process was induced by the quasi-equilibrated interaction between surface C3H5* and O*, generating adsorbed CO2* and OH* (step 5). Ultimately, the final product of CO2 was desorbed from the catalyst surface (step 6), and H2O was generated from the reaction between OH* molecules and then desorbed by leaving the vacancy sites (steps 7 and 8). The derivation of Supplementary Equation (1) was mainly based on the assumption of the pseudo-steady state for the kinetically observable O2*, O*, and C3H6* species, accompanied by the quasi-equilibrium for steps 1–3 previous to the RDS. Supplementary Table 6 summarized the kinetic parameters derived by regressing the kinetics data to Supplementary Equation (1) while minimizing the residuals. Generally, C3H6 oxidation pathways started from the adsorbed and activation of C3H6 and O2. Then, the reactions underwent C–H scission with O* to form an allyl intermediate (C3H5*), which was considered as kinetically-relevant step for the whole reaction.
DFT calculations were further conducted to explore the change of energies in the specific reaction route based on the elementary steps, and Supplementary Table 7 summarizes the potential energies in every reaction step. As shown in Fig. 4e, the reaction was initiated by the adsorption of O2 on metallic Pt sites and then dissociated to the adsorbed O* molecules with a low dissociation energy of 0.10 eV (TS-1). The adsorbed O* was subsequently reacted with C3H6* adsorbed on metallic Pt ensembles with the activation energy of 1.53 eV (TS-2). Subsequently, the dehydrogenated C3H5* was further activated by O* to generate C3H4* and OH* with an energy barrier of 0.69 eV (TS-3). After the formed OH* desorbed from the surface of metallic Pt ensembles with the energy of 1.01 eV (TS-4), the dissociated O* would interact with C3H4* to generate the surface-adsorbed acrolein (C3H4O*) with the reaction energy calculated as 0.83 eV (TS-5). Afterward, the second O2 dissociation process occurred, with an identical activation energy to TS-1, to generate more O* species (TS-6), which was then coupled with C3H4O* to form C3H3O* intermediates with the reaction energy of 0.71 eV (TS-7). Next, C3H3O* acted as the crucial precursor of the carboxylates and was further coupled with the dissociated O* to generate activated acrylate species (C3H3O2*) with an activation energy of 1.05 eV (TS-8). Lastly, CO2* and H2O* were produced and desorbed as the final products with a total free energy of −18.21 eV.
It was demonstrated that the reaction pathways of C3H6 oxidation followed classic Langmuir-Hinshelwood models based on the above kinetics and DFT results. Notably, the kinetic model of C3H6 oxidation might be changed by elevating the reaction temperature to 188 °C. Figure 4d revealed a zero-reaction order for C3H6 partial pressure, while a dependence with the half-reaction order of 0.53 was observed for O2 partial pressure. The reaction order of approximately 0.5 regarding O2 partial pressure was also observed for the kinetic studies of CO oxidation over Pt1/CeO251 and Rh/CeO252 catalysts, which might prefer to occur on the supported catalysts with small metal clusters. It suggested that C3H6 molecules adsorbed stronger than O2 and fully covered the surface of metallic Pt ensemble sites at 188 °C. Meanwhile, the adsorption and dissociation of O2 took place at the vacant sites of the Pt-O-Ce interface (Pt-Ov-Ce). Supplementary Fig. 30 listed the sequence of fundamental steps consistent with the observed kinetics data of C3H6 oxidation once the reaction elevated to 188 °C. Compared to Supplementary Fig. 29, the major difference was O2 adsorption and activation at the unoccupied oxygen vacancy (#) over Pt-O-Ce interfacial sites without competition with C3H6. Subsequently, the dissociated O# at the interfacial sites reacted with the adsorbed C3H6* at the Pt ensembles to finish the dehydrogenation of the sp3 hybrid C-H bond and generate adsorbed C3H5* and OH#, which was irreversible and determined to be the RDS for the whole reaction. Supplementary Equation (2) could accurately describe the kinetics data of the reaction rates of C3H6 oxidation at 188 °C. Furthermore, the parity plots further verified the accuracy of the rate equation by comparing experimental and calculated data (Supplementary Fig. 31). The fractional coverages of θ(C3H6*) could be further calculated based on the obtained kinetics data and related partial pressures of the reactants. It was found that the fractional coverages were higher than 0.99 for θ(C3H6*) at temperatures of 188 °C, while the values would be only 0.20–0.55 for the same reaction at 162 °C. The results implied that the surface coverage of C3H6 over Pt0 active sites was much more significant with increasing reaction temperatures, while oxygen would be adsorbed and activated at the Pt-O-Ce interfacial sites without competition with surface C3H6. Based on the above results, it was reasonable to deduce that there should be a threshold temperature affecting the reaction model during C3H6 oxidation. According to the findings, the dynamic reaction models based on different reaction temperature ranges were summarized and illustrated in Fig. 5.
At low temperatures (<170 °C), the reactions followed the classic Langmuir-Hinshelwood model, where both C3H6 and O2 were adsorbed over metallic Pt ensembles. In contrast, at high temperatures (>170 °C), metallic Pt ensembles were fully covered by adsorbed C3H6, where O2 was adsorbed and activated at vacant sites of Pt-O-Ce interfaces without competition with C3H6.
It should be noted that the C3H6 oxidation might not follow the Mars-van Krevelen model at 188 °C in this study. On one hand, the Mars-van Krevelen model typically initiated the reaction between C3H6 and activation of lattice oxygen, followed by the activated oxygen (O*) filling into the oxygen vacancies. The reaction order of approximately 0.5 regarding O2 partial pressure suggested the assumption that the elementary step of oxygen filling right after O* formation was the RDS. Yet, the fractions of Ce3+ would gradually decrease with ramping reaction temperatures and caused an increase of Ce4+ concentrations as evidenced by EELS tests and also in situ Raman and NAP-XPS study, suggesting that the oxidation of Ce3+ was accelerated in the high temperature ranges. It meant that the oxygen activation and filling into vacancies was a fast reaction at high temperatures, and would not govern the whole catalytic reaction rate. On the other hand, theoretical results simulated that oxygen was adsorbed and activated at the top layer and interfacial Pt-O-Ce sites, which exhibited the energy barriers of 0.10 and 0.22 eV, respectively (Supplementary Table 8). Meanwhile, the energy barrier of dehydrogenation facilitated by interfacial oxygen was observed as 1.53 and 1.68 eV for the corresponding sites, respectively. It suggested that the oxygen-facilitated dehydrogenation was still the RDS step for C3H6 oxidation.
Discussion
A highly efficient Pt/CeO2 ensemble catalyst was obtained through a facile H2 activation, which exhibited potential application toward vehicle emission control. HAADF-STEM and EXAFS results suggested the reconstruction from Pt single-layer planar (0.45 nm) to multilayer ensemble (0.84 nm) with the top layers composed of Pt0 sites by hydrogen activation. In situ Raman spectra, NAP-XPS, and EELS experiments revealed that Ce3+ defects and dioxygen intermediates were activated once the reaction temperature exceeded 170 °C. Based on the complementary experiments and theoretical results, Pt0 ensemble sites demonstrated a low activation barrier for the dehydrogenation of sp3 hybrid C-H with the assistance of oxygen, identified as the RDS, compared to Ptᵟ+ sites. The kinetic results revealed that the C3H6 oxidation experienced a dynamic transformation of the reaction models, where the adsorption and dissociation of O2 occurred at interfacial Pt-Ov-Ce sites after the temperature surpassed the threshold at approximately 170 °C. Combining the experimental phenomena, it was deduced that the involvement of interfacial Pt-Ov-Ce sites was the key to triggering the dynamic evolution of the reaction models. Generally, this research not only successfully clarified that the metallic Pt ensembles were the intrinsic active sites for the highly efficient Pt/CeO2 catalysts regarding C3H6 oxidation, but also elucidated the dynamic change of the interfacial properties and the reaction model using in situ characterization methods. This work will guide the precise design and optimization of effective atomically dispersed platinum-based catalysts for emission control applications.
Methods
Catalyst preparation
Materials
All the chemicals are analytical reagent (AR) grades unless otherwise stated. Cerium nitrate hexahydrate (Ce(NO3)3·6H2O) and sodium hydroxide (NaOH) powder were purchased from Sinopharm Chemical Reagent Co., Ltd. The noble metal precursor of tetraammineplatinum (II) nitrate (Pt(NH3)4(NO3)2) with 99.995% trace metals basis was obtained from Sigma-Aldrich. The γ-Al2O3 was bought from Sasol Chemical LLC. The deionized water was directly produced and collected via a Milli-Q ® water purification machine from Merck KGaA.
Preparation of CeO2 supports
A hydrothermal method was applied to synthesize supporting CeO2 nanocubes53. 16.88 g NaOH and 1.96 g Ce(NO3)3·6H2O were dissolved in 30 mL and 40 mL deionized water, respectively. After magnetic stirring for 15 min, the NaOH solution was added dropwise to Ce(NO3)3·6H2O solution under vigorous stirring for another 30 min. Subsequently, the mixed solution was transferred to a 100 mL Teflon bottle, which was then tightly sealed in a stainless-steel vessel autoclave and hydrothermally treated at 180 °C for 24 h. After cooling to room temperature, the white precipitate was washed and collected by centrifuging with deionized water at least three times and vacuum dried at 80 °C for 12 h. Finally, the yellowish products were calcined at 500 °C for 4 h with a ramping rate of 1 °C min−1 to obtain CeO2.
Preparation of Pte/CeO2 ensemble catalysts
0.5 wt.% Pt species were loaded on CeO2 nanocubes via an incipient wetness impregnation method. 71 μL Pt(NH3)4(NO3)2 solution ([Pt] = 25 g L−1) was applied as the Pt precursor and diluted in 0.9 mL deionized water. The aqueous solution was sonicated and slowly dripped onto 0.25 g ground CeO2 powder. Subsequently, the mixture was evaporated at 60 °C for 1 h and oven-dried at 110 °C for 8 h. The resulting powder was calcined in static air at 500 °C for 6 h with a temperature ramp of 1 °C min−1 to produce Pt ensemble nanoclusters denoting as Pte. For the hydrogen activation process, Pte was pretreated in 10% H2/N2 reducing flow under different temperatures for 1 h. The pretreated samples were denoted as Pte-XA, where X represented the activation temperature. Pte catalysts were pretreated at 100, 200, 300, or 400 °C to identify the optimal activation condition. As evidenced in Supplementary Fig. 1, Pte-300A exhibited the highest catalytic activity towards C3H6 oxidation among the series of Pte-XA catalysts.
Preparation of Pte/γ-Al2O3 ensemble catalysts
To conduct a comparison study, 0.5 wt% Pte/γ-Al2O3 catalysts were prepared by the identical procedure to Pte catalysts. Prior to the synthesis, the commercial γ-Al2O3 was pretreated in a reducing flow of 10% H2 with N2 as the carrier gas at 350 °C for 2 h, leading to the formation of unsaturated Al3+penta sites54,55.
Catalytic activity evaluation
Light-off tests
Reactant gases including 1% C3H6/N2 (99.999%), 5% CO/N2 (99.999%) high-purified O2 (99.999%), and high-purified N2 (99.999%) were purchased from Shanghai Weichuang Standard Gas Analytical Technology Co., Ltd. The gas flow was precisely manipulated via mass flow controllers from Beijing Sevenstar Electronics Co., Ltd.
The catalytic light-off performance of C3H6 oxidation was measured in a fixed-bed quartz tube reactor with an internal diameter of 5.0 mm. Two thermocouples located upstream and downstream of the catalyst bed were utilized to monitor and regulate the temperature in the reactor. During each light-off test, the furnace reached the target temperature with a ramp rate of 2 °C min−1. 50 mg granulated catalysts with mesh sizes of 180–250 μm were diluted by the same size quartz sands, which was pre-calcined at 800 °C for 6 h in static air before mixing, to minimize the heat effect. The total flow rate was maintained at 200 mL min−1 for all reactions under atmospheric pressure, which corresponded to a weight hourly space velocity (WHSV) of 240,000 mL g−1 h−1. For C3H6 oxidation activity test, a feeding gas flow composed of 0.1% C3H6, 0.4% CO (when used), 5% H2O (when used), 10% O2 balanced with N2 passed through the tube reactor. The reactant and product gas concentrations were collected and analyzed by a Fourier-transform infrared (IR) spectrometer (Antaris IGS Gas Analyzer, Thermo Fisher Scientific Inc.). A high-pressure syringe pump was applied to precisely manipulate the injection rate of deionized water, which was completely vaporized in a gasifier isothermal at 150 °C before pumping into the reactor system. The C3H6 and CO conversions (\(X\)) were calculated by the following equations:
where Cin and Cout represent the inlet and outlet concentrations of the reactants, respectively.
C3H6 and CO consumption rates (mmol molPt−1 s−1) were collected at the constant temperature points and calculated by the following equation:
Where \(v\) represents the flow rate of the reactants (mL s−1); \({M}_{\mathrm{Pt}}\) is the atomic mass of platinum; \({m}_{\mathrm{Pt}}\) is the mass of the platinum in the catalysts and determined by ICP-OES methods; \({V}_{{\rm{m}}}\) is the molar volume of gas equals 24.5 L mol−1 at room temperature.
Apparent activation energy and kinetic evaluation
Arrhenius plots were constructed in a differential reactor by testing the C3H6 consumption rate at various temperature points following the identical reaction gas composition and WHSV to the light-off tests. All the catalytic conversions were strictly restricted below 15% to exclude the heat and mass transfer limitations. Meanwhile, the reactor was ramped to each target temperature at 2 °C min−1 and held for at least 45 min to achieve a steady state. The apparent activation energy (Ea) was calculated by the Arrhenius plot.
For the kinetic experiments, the C3H6 oxidation rate was measured at a constant temperature by altering the partial pressure of the reactant gas without any back pressure in the reactor system. Heat and mass transfer limitations have been ruled out based on the theoretical results (Supplementary Table 9). 20 mg fine-ground catalysts with mesh sizes of 180–250 μm were mixed with 50 mg identical-size quartz sands, and packed into the fixed-bed tube reactor. Most catalytic conversions were restrained below 12%, maximizing at approximately 18%, which ensured that the kinetic evaluation could presume the differential reaction conditions. The mean partial pressure was determined as the average pressure between the inlet and outlet flows using the following equations:
The mean partial pressure of C3H6 and O2 ranged from 45.52 to 195.04 Pa and 2.51 to 12.64 kPa, respectively.
Inductively coupled plasma optical emission spectroscopy (ICP-OES)
The ICP-OES experiments were conducted on Avio 550 (PerkinElmer Inc.) to detect the actual Pt content of different catalysts.
Scanning transmission electron microscopy (STEM)
The aberration-corrected scanning transmission electron microscopy was conducted on a Thermo Fisher Themis Z transmission electron microscope to analyze the morphology and elemental distribution of the catalysts. This instrument was operated at a working voltage of 300 kV and equipped with two aberration correctors. 4 in-column Super-X detectors were applied to conduct Energy-dispersive X-ray spectroscopy analysis. High angle annular dark-field (HAADF) STEM images were captured using a camera length of 115 mm on HAADF detectors with inner and outer collection angles of 47 and 200 mrad, respectively. EELS data were acquired using a Gatan Enfinium ER (model 977) EELS spectrometer with a dual EELS function.
X-ray absorption spectroscopy (XAS)
The XAS scans were performed for Pte and Pte-300A on 21 A X-ray nanodiffraction beamline (4-bounce channel-cut Si (111) monochromator) of Taiwan Photon Source (TPS) at the National Synchrotron Radiation Research Center (NSRRC). The measurements for the Pt-L3 edge were carried out in the fluorescence mode in an energy range from 6 to 27 eV corresponding to the photon flux between 1 × 1011 ~ 3 × 109 photon/s. The end-station is equipped with three ionization chambers and a Lytle/SDD detector after the focusing position of the KB mirror to collect data. The XANES data and EXAFS data were analyzed and fitted using the Athena and Artemis software from the Demeter software package, respectively.
X-ray diffraction (XRD)
The XRD experiments were carried out on a LabX XRD-6100 instrument from Shimadzu Corporation, which operated at 40 mA and 40 kV with Cu Kα radiation (λ = 0.15406 nm) under an ambient condition. The scanning range of 2θ angle was recorded from 5 to 90° in a step rate of 3.33° min−1 to investigate the phase structure.
N2 adsorption and desorption isotherm
N2 physisorption isotherm experiments were carried out on a Micromeritics ASAP 2020 analyzer. Prior to each measurement, the samples were firstly degassed under 300 °C for 8 h. The Barrett–Joyner–Halenda method was applied to determine the pore size distribution using the data of nitrogen desorption isotherm. The specific surface area was calculated using the Brunauer–Emmett–Teller equation.
X-ray photoelectron spectroscopy (XPS)
The XPS spectra were collected on a Kratos AXIS Ultra DLD instrument (Shimadzu Corporation) operating at a working current and voltage of 8 mA and 14 kV, respectively, utilizing a monochromatic Al source. The binding energies of all tested elements were calibrated on the base of the standard C1s line at 284.8 eV.
H2 temperature-programmed reduction (H2-TPR)
H2-TPR experiments were performed on the AutoChem II 2920 chemisorption analyzer from Micromeritics to investigate the redox properties of the catalysts. 40 mg catalysts were loaded into the U-shape quartz tube, which was pretreated in a 50 mL min−1 Ar flow at 300 °C for 30 min and then cooled down to room temperature. Once the baseline was stable at room temperature, the reactor was heated to 1000 °C at a ramp rate of 10 °C min−1 under a reducing gas stream of 5% H2/N2 flow (50 mL min−1). The signal was monitored and recorded with a thermal conductivity detector. For Pte-300A samples, an additional activation process was conducted after the Ar purge at 300 °C, and the catalyst was pretreated in a 5% H2/Ar at the target temperature for 1 h prior to cooling to room temperature.
In situ diffuse reflectance infrared Fourier transform spectroscopy (in situ DRIFTS)
In situ DRIFTS experiments were conducted on a Nicolet 6700 FTIR equipped with a mercury-cadmium-telluride detector cooling by liquid nitrogen. The fine-ground catalyst powder was loaded into a high-temperature reaction chamber with three Ba2F windows on the dome of the cell. The IR spectra resulted from averaging 64 scans at a resolution of 4 cm−1.
Pte and Pte-300A samples were pretreated in the N2 flow (100 mL min−1) and 10% H2/N2 (100 mL min−1) at 300 °C for 1 h, respectively. Subsequently, the reaction chamber was cooled down to the target temperatures under N2 gas flow. The background spectrum was recorded at each desired temperature and subtracted from the sample spectrum.
For the CO adsorption and O2 purging experiments, 1% CO/N2 in a total flowrate of 100 mL min−1 was passed through the reaction chamber at the desired temperature. Once the CO adsorption saturated on the surface of catalysts, the system was purged by 100 mL min−1 N2 flow until the spectra became unchanged to remove the weakly adsorbed CO molecules. Finally, 10% O2 was introduced into the reactor to investigate the reactivity of different active sites. DRIFTS spectra were recorded throughout the entire reaction process. For the C3H6 oxidation, a mixture gas flow composed of 0.4% C3H6 and 10% O2 in N2 balance (100 mL min−1) was introduced into the reaction cell. The DRIFTS spectra were recorded at each temperature for at least 30 min.
In situ Raman spectroscopy
A Horiba LabRam HR spectrometer using visible laser excitation with a wavelength of 514 nm emitted by a He-Cd laser was utilized for the in situ Raman experiments. A confocal microscope (Olympus BX-30-LWD) paired with a 50x long working distance objective was applied to focus the laser on the sample. The scattered photons were concentrated onto a single-stage monochromator and monitored using a UV-sensitive liquid nitrogen-cooled charge-coupled device (CCD) detector (Horiba CCD-3000 V). Pte-300A samples were pretreated in 10% H2 at the tube reactor before the test. For C3H6 oxidation, the experiments were performed under the flowing reactant composed of 1000 ppm C3H6 and 10% O2 balanced with N2 at a total flowrate of 200 mL min−1. For CO oxidation, the experiments were conducted under the flowing reactant composed of 4000 ppm CO and 10% O2 balanced with N2 at a total flowrate of 50 mL min−1. Every spectrum was collected after the reaction for 20 min at each temperature point from 50 to 250 °C.
NAP-XPS
The NAP-XPS were acquired using a SPECS-AU190069 instrument. The instrument features a multi-stage differential pumping system and a static voltage lens, making it suitable for usage in ultra-high vacuum (1 × 10−9 mbar) with gases ranging from 0 to 5 mbar. The spectra were obtained using monochromatized Al Kα irradiation (1486.6 eV), generated by 50 W of excitation source power in an Al anode (SPECS XR-50). The X-ray spot was approximately 0.3 mm in diameter and located near the nozzle’s opening. A pressure-reducing valve maintained a reaction pressure of 1 mbar. The powder sample was flattened into a smooth sheet and placed on a specially designed sample table that may be heated during the reaction. An electron flood cannon was used to correct for the charging of catalysts during tests.
Computational method
The DFT calculations were performed by the Vienna Ab initio Simulation Package (VASP 5.4.1)56 with the projector augmented wave method57. The exchange-functional is treated using the generalized gradient approximation with Perdew-Burke-Emzerhof 58 functional. The energy cutoff for the plane wave basis expansion was set to 450 eV. Partial occupancies of the Kohn−Sham orbitals were allowed using the Gaussian smearing method and a width of 0.2 eV. The Brillouin zone was sampled with the Monkhorst–Pack k-point of 2 × 2 × 1 was applied for all the calculations for surface structures. The CeO2(100) support was modeled as a four-layer slab, with the top two layers fully relaxed and the bottom two layers constrained. The pre-activated surface was represented by a supported single-layer Pt7 cluster, while the post-activated state was modeled by a multi-layer Pt17 cluster. The self-consistent calculations apply a convergence energy threshold of 10−5 eV, and the force convergency was set to 0.05 eV/Å. The free energy corrections were calculated by the following equation:
where ΔE, ΔGZPE, ΔGU, and ΔS refer to the DFT calculated energy change, the correction from zero-point energy, the correction from inner energy, and the correction from entropy59. The transition state was located via constrained optimization based on a process of varying the target reaction coordinate while relaxing all other degrees of freedom. The optimized structure was subsequently validated by a vibrational frequency analysis, confirming the presence of exactly only one imaginary frequency60,61.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data generated in this study are provided in the Supplementary Information/Source Data file. Data are available from the corresponding authors upon request. Source data are provided with this paper.
References
Gandhi, H. S., Graham, G. W. & McCabe, R. W. Automotive exhaust catalysis. J. Catal. 216, 433–442 (2003).
Russell, A. & Epling, W. S. Diesel oxidation catalysts. Catal. Rev. 53, 337–423 (2011).
Wang, F. et al. Identification of direct anchoring sites for monoatomic dispersion of precious metals (Pt, Pd, Ag) on CeO2 support. Angew. Chem. Int. Ed. 63, e202318492 (2024).
Liu, X. et al. Activation of subnanometric Pt on Cu-modified CeO2 via redox-coupled atomic layer deposition for CO oxidation. Nat. Commun. 11, 4240 (2020).
Qiao, B. et al. Single-atom catalysis of CO oxidation using Pt1/FeOx. Nat. Chem. 3, 634–641 (2011).
Zhang, Z. et al. Thermally stable single atom Pt/m-Al2O3 for selective hydrogenation and CO oxidation. Nat. Commun. 8, 16100 (2017).
Jeong, H. et al. Fully dispersed Rh ensemble catalyst to enhance low-temperature activity. J. Am. Chem. Soc. 140, 9558–9565 (2018).
Gabelnick, A. M., Capitano, A. T., Kane, S. M., Gland, J. L. & Fischer, D. A. Propylene oxidation mechanisms and intermediates using in situ soft X-ray fluorescence methods on the Pt(111) surface. J. Am. Chem. Soc. 122, 143–149 (2000).
Jeong, H., Shin, S. & Lee, H. Heterogeneous atomic catalysts overcoming the limitations of single-atom catalysts. ACS Nano 14, 14355–14374 (2020).
Fang, Y. et al. Oxygen vacancy-governed opposite catalytic performance for C3H6 and C3H8 combustion: the effect of the Pt electronic structure and chemisorbed oxygen species. Environ. Sci. Technol. 56, 3245–3257 (2022).
Liang, Y. et al. Improved low-temperature catalytic oxidation performance of Pt-based catalysts by modulating the electronic and size effects. New J. Chem. 44, 10500–10506 (2020).
Gu, L. et al. Propene and CO oxidation on Pt/Ce-Zr-SO42– diesel oxidation catalysts: effect of sulfate on activity and stability. Chin. J. Catal. 38, 607–615 (2017).
Maurer, F. et al. Tracking the formation, fate and consequence for catalytic activity of Pt single sites on CeO2. Nat. Catal. 3, 824–833 (2020).
Ding, K. et al. Identification of active sites in CO oxidation and water-gas shift over supported Pt catalysts. Science 350, 189–192 (2015).
Lu, Y. et al. Interaction between noble metals (Pt, Pd, Rh, Ir, Ag) and defect-enriched TiO2 and its application in toluene and propene catalytic oxidation. Appl. Surf. Sci. 606, 154834 (2022).
Bratan, V. et al. In situ electrical conductivity study of Pt-impregnated VOx/γ-Al2O3 catalysts in propene deep oxidation. J. Mater. Sci. 55, 10466–10481 (2020).
Su, Z. et al. Probing the actual role and activity of oxygen vacancies in toluene catalytic oxidation: evidence from in situ XPS/NEXAFS and DFT+U calculation. ACS Catal. 13, 3444–3455 (2023).
Su, Z. et al. Roles of oxygen vacancies in the bulk and surface of CeO2 for toluene catalytic combustion. Environ. Sci. Technol. 54, 12684–12692 (2020).
Wang, Q. et al. Operando investigation of toluene oxidation over 1D Pt@CeO2 derived from Pt cluster-containing MOF. J. Am. Chem. Soc. 143, 196–205 (2021).
Li, Y. et al. Dynamic structure of active sites in ceria-supported Pt catalysts for the water gas shift reaction. Nat. Commun. 12, 914 (2021).
Wang, C. et al. Water-mediated Mars–Van Krevelen mechanism for CO oxidation on ceria-supported single-atom Pt1 catalyst. ACS Catal. 7, 887–891 (2017).
Nie, L. et al. Activation of surface lattice oxygen in single-atom Pt/CeO2 for low-temperature CO oxidation. Science 358, 1419–1423 (2017).
Ke, J. et al. Strong local coordination structure effects on subnanometer PtOx custers over CeO2 nanowires probed by low-temperature CO oxidation. ACS Catal. 5, 5164–5173 (2015).
Avanesian, T. et al. Quantitative and atomic-scale view of CO-induced Pt nanoparticle surface reconstruction at saturation coverage via DFT calculations coupled with in situ TEM and IR. J. Am. Chem. Soc. 139, 4551–4558 (2017).
Jin, T., Zhou, Y., Mains, G. J. & White, J. M. Infrared and X-ray photoelectron spectroscopy study of carbon monoxide and carbon dioxide on platinum/ceria. J. Phys. Chem. 91, 5931–5937 (1987).
Xie, S. et al. Structure-activity relationship of Pt catalyst on engineered ceria-alumina support for CO oxidation. J. Catal. 405, 236–248 (2022).
Tan, W. et al. Transformation of highly stable Pt single sites on defect engineered ceria into robust Pt clusters for vehicle emission control. Environ. Sci. Technol. 55, 12607–12618 (2021).
Pozdnyakova, O. et al. Preferential CO oxidation in hydrogen (PROX) on ceria-supported catalysts, part I: oxidation state and surface species on Pt/CeO2 under reaction conditions. J. Catal. 237, 1–16 (2006).
Kappers, M. J., Miller, J. T. & Koningsberger, D. C. Deconvolution and curve fitting of IR spectra for CO adsorbed on Pt/K-LTL: potassium promoter effect and adsorption site distribution. J. Phys. Chem. 100, 3227–3236 (1996).
Vannice, M. A. & Twu, C. C. Extinction coefficients and integrated intensities for linear- and bridged-bonded CO on platinum. J. Chem. Phys. 75, 5944–5948 (1981).
Delley, B., Ellis, D. E., Freeman, A. J., Baerends, E. J. & Post, D. Binding energy and electronic structure of small copper particles. Phys. Rev. B 27, 2132–2144 (1983).
Miller, J. T. et al. The effect of gold particle size on AuAu bond length and reactivity toward oxygen in supported catalysts. J. Catal. 240, 222–234 (2006).
Gatla, S. et al. Room-temperature CO oxidation catalyst: low-temperature metal–support interaction between platinum nanoparticles and nanosized Ceria. ACS Catal. 6, 6151–6155 (2016).
Hüfner, S. & Wertheim, G. K. Core-line asymmetries in the X-ray-photoemission spectra of metals. Phys. Rev. B 11, 678–683 (1975).
Tan, Y. et al. Facile fabrication of robust hydrogen evolution electrodes under high current densities via Pt@Cu interactions. Adv. Funct. Mater. 31, 2105579 (2021).
Trovarelli, A. Catalytic properties of ceria and CeO2-containing materials. Catal. Rev. 38, 439–520 (1996).
Cao, X., Ji, Y. & Luo, Y. Dehydrogenation of propane to propylene by a Pd/Cu single-atom catalyst: insight from first-principles calculations. J. Phys. Chem. C. 119, 1016–1023 (2015).
Yang, M.-L., Zhu, Y.-A., Zhou, X.-G., Sui, Z.-J. & Chen, D. First-principles calculations of propane dehydrogenation over PtSn catalysts. ACS Catal. 2, 1247–1258 (2012).
Nørskov, J. K., Abild-Pedersen, F., Studt, F. & Bligaard, T. Density functional theory in surface chemistry and catalysis. Proc. Natl. Acad. Sci. USA 108, 937–943 (2011).
Pushkarev, V. V., Kovalchuk, V. I. & d’Itri, J. L. Probing defect sites on the CeO2 surface with dioxygen. J. Phys. Chem. B 108, 5341–5348 (2004).
Wu, Z., Li, M., Howe, J., Meyer, H. M. & Overbury, S. H. Probing defect sites on CeO2 nanocrystals with well-defined surface planes by Raman spectroscopy and O2 ddsorption. Langmuir 26, 16595–16606 (2010).
Zaera, F. & Chrysostomou, D. Propylene on Pt(111). Surf. Sci. 457, 71–88 (2000).
Driscoll, D. M. et al. Binding sites, geometry, and energetics of propene at nanoparticulate Au/TiO2. J. Phys. Chem. C. 121, 1683–1689 (2017).
Lord, R. C. & Venkateswarlu, P. The infrared spectra of propylene and propylene-d6*. J. Opt. Soc. Am. 43, 1079–1085 (1953).
Finocchio, E., Busca, G., Lorenzelli, V. & Willey, R. J. FTIR studies on the selective oxidation and combustion of light hydrocarbns at metal oxide surfaces. Propane and propene oxidation on MgCr2O4. J. Chem. Soc. Faraday Trans. 90, 3347–3356 (1994).
Finocchio, E., Busca, G., Lorenzelli, V. & Escribano, V. S. V. S. FTIR studies on the selective oxidation and combustion of light hydrocarbons at metal oxide surfaces. Part 2.—propane and propene oxidation on Co3O4. J. Chem. Soc. Faraday Trans. 92, 1587–1593 (1996).
Finocchio, E., Busca, G., Lorenzelli, V. & Willey, R. J. The activation of hydrocarbon CH bonds over transition metal oxide catalysts: a FTIR study of hydrocarbon catalytic combustion over MgCr2O4. J. Catal. 151, 204–215 (1995).
Concepción, P., Botella, P. & Nieto, J. M. L. Catalytic and FT-IR study on the reaction pathway for oxidation of propane and propylene on V- or Mo–V-based catalysts. Appl. Catal. A 278, 45–56 (2004).
Wan, J., Ran, R., Li, M., Wu, X. & Weng, D. Effect of acid and base modification on the catalytic activity of Pt/Al2O3 for propene oxidation. J. Mol. Catal. 383-384, 194–202 (2014).
Lachkov, P. T. & Chin, Y.-H. Catalytic consequences of reactive oxygen species during C3H6 oxidation on Ag clusters. J. Catal. 366, 127–138 (2018).
Wang, H. et al. Surpassing the single-atom catalytic activity limit through paired Pt-O-Pt ensemble built from isolated Pt1 atoms. Nat. Commun. 10, 3808 (2019).
Bunluesin, T., Cordatos, H. & Gorte, R. J. Study of CO oxidation kinetics on Rh/Ceria. J. Catal. 157, 222–226 (1995).
Mai, H.-X. et al. Shape-selective synthesis and oxygen storage behavior of ceria nanopolyhedra, nanorods, and nanocubes. J. Phys. Chem. B 109, 24380–24385 (2005).
Kwak, J. H. et al. Coordinatively unsaturated Al3+ centers as binding sites for active catalyst phases of platinum on γ-Al2O3. Science 325, 1670–1673 (2009).
Jeong, H. et al. Highly durable metal ensemble catalysts with full dispersion for automotive applications beyond single-atom catalysts. Nat. Catal. 3, 368–375 (2020).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Perdew, J. P. et al. Atoms, molecules, solids, and surfaces: applications of the generalized gradient approximation for exchange and correlation. Phys. Rev. B 46, 6671–6687 (1992).
Cao, X.-M., Burch, R., Hardacre, C. & Hu, P. An understanding of chemoselective hydrogenation on crotonaldehyde over Pt(111) in the free energy landscape: the microkinetics study based on first-principles calculations. Catal. Today 165, 71–79 (2011).
Liu, Z.-P. & Hu, P. General rules for predicting where a catalytic reaction should occur on metal surfaces: a density functional theory study of C–H and C–O bond breaking/making on flat, stepped, and kinked metal surfaces. J. Am. Chem. Soc. 125, 1958–1967 (2003).
Alavi, A., Hu, P., Deutsch, T., Silvestrelli, P. L. & Hutter, J. CO oxidation on Pt (111): an ab initio density functional theory study. Phys. Rev. Lett. 80, 3650 (1998).
Acknowledgements
L.M. acknowledges the financial support by the National Key Research and Development Program of China (2024YFB4105100), the National Natural Science Foundation of China (22176122), the Shanghai Pujiang Program (20PJ1407000), the Oceanic Interdisciplinary Program of Shanghai Jiao Tong University (SL2022ZD104), and the Zhejiang Key Laboratory of Low-carbon Control Technology for Industrial Pollution, Zhejiang University of Technology (2025DTZL01 and 2025ZY01076). S.D. was supported by the National Natural Science Foundation of China (22376062) and the Science and Technology Commission of Shanghai Municipality (24DX1400200 and 22ZR1415700). H.C. was supported by the National Natural Science Foundation of China (22176217) and the National Engineering Laboratory for Mobile Source Emission Control Technology (NELMS2018A12). F.L. thanks the Startup Fund from the University of California, Riverside (UCR).
Author information
Authors and Affiliations
Contributions
L.M. conceived and designed experiments. Z.-H.L. prepared samples, performed characterization, conducted catalytic performance tests; Y.L. and S.D. performed TEM tests; X.C., H.C., Z.-G.L. and K.Y. conducted research, discussed results and commented on the manuscript. Z.-H.L., F.L. and L.M. discussed results, wrote and revised the manuscript. N.Y. supervised the research. All the authors have given approval to the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, Z., Chen, X., Lv, Y. et al. Temperature-driven mechanistic transition in propylene oxidation over Pt/CeO2 ensemble catalysts. Nat Commun 16, 9199 (2025). https://doi.org/10.1038/s41467-025-64243-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-64243-y