Abstract
Linear carbon (Cn), an elusive sp-hybridized carbon allotrope, has long attracted interest for its remarkable properties and debated structures. Here, we report the synthesis of an uncapped linear C5 via tip-induced dehalogenation and ring-opening reaction of C5Br6, with its cumulenic structure confirmed by atomic force microscopy. We further demonstrate the tip-induced coupling of linear C5 to form longer chains (e.g., C10, C15). By applying higher voltage pulses to C5Br6 (also C6Br6), various carbon chains, including C9, C10, C13, C14, C15, C17, C18, C21, C23, could be synthesized. Even-numbered chains (C10, C14 and C18) adopt polyynic structures due to a Peierls transition on NaCl, whereas for odd-numbered chains, C9 adopts a cumulene-like structure, and longer ones exhibit hybrid structures with terminal triple bonds and cumulene-like interiors. Scanning tunneling spectroscopy reveals smaller transport gaps for odd- than even-numbered chains, consistent with Peierls theory, and decreasing gaps with increasing chain length.
Similar content being viewed by others
Introduction
Linear carbon (Cn), an elusive sp-hybridized carbon allotrope, has attracted much interests both experimentally and among theorists for decades due to remarkable properties and significant controversy1,2,3,4,5,6. Similar to recently popular sp-hybridized cyclo[n]carbon7,8,9,10,11,12,13, linear carbon may also have two distinct structural forms, i.e., cumulene for consecutive double bonds, or polyyne for alternating triple and single bonds. Peierls transition theory predicted that for infinite linear Cn, polyyne should be more stable than cumulene, and meanwhile opening a band gap at Fermi level14,15,16.
Due to its very high reactivity, synthesis and characterization of linear carbon are exceptionally challenging. Previous works have tried different methods17,18,19,20,21, especially heavy end-capping groups22,23,24 or confined into carbon nanotube25, to synthesize carbon chains, however, atomically precise synthesis of uncapped linear carbon, and real-space characterization of its detailed structure remains very rare13,26,27. Especially, its cumulenic form, is proved to be even harder to synthesize compared to polyynic one. In solution, till now, the longest cumulene known to date is n = 10 (also with protected group at both ends)28,29,30,31.
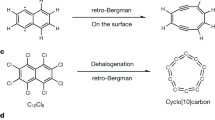
Previous theoretical works show that in the gas phase, short linear Cn (approximately n < 52) are all cumulenic32,33,34, and Peierls transition in odd-numbered Cn will be reached at larger number of carbon atoms than that in the even-numbered Cn34. Our work on the linear C613, however, reveals that it instead already adopts a polyynic structure, a result of the NaCl surface-induced Peierls transition (Fig. 1a). This finding motivated us to investigate the influence of number of carbon atoms and odevity effect on the structures of carbon chains. Linear C5, is thus considered to be a relevant candidate. Additionally, theoretical work also indicates that for n = 5, the linear form is expected to be much more stable than the cyclic form32,35,36,37,38,39, in contrast to the case of n = 613 (where the energy difference between its linear and cyclic forms is very small).
a Reaction scheme for the generation of linear C6. b Reaction scheme for the generation of linear C5. c I–c III, d I–d III AFM and Laplace-filtered AFM images of precursor, intermediate and product (linear C5). e Zoom-in AFM image of linear C5. f Calculated bond lengths (Å) of linear C5 in the gas phase and on the NaCl surface. g AFM image of linear C5 with NaCl lattice. Crossings of white lines mark Cl ion positions determined experimentally. h Side and top views of the calculated structure of linear C5 on the NaCl surface. The scale bar in (c I) applies to all experimental and Laplace-filtered AFM images except e. AFM tip offsets Δz: +1.5 Å, −1.0 Å, −1.4 Å, −0.5 Å, −1.5 Å for c I, c II, c III, e, g. Reference set points of Δz: I = 0.5 pA, V = 0.3 V for all AFM images.
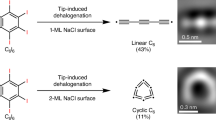
We employed the scanning tunneling microscopy (STM) tip-induced26,40,41 dehalogenation method to generate C5. We chose a fully halogenated cyclopenta-1,3-diene (perbromocyclopenta-1,3-diene, C5Br6) as the precursor, and through tip-induced entire dehalogenation and ring-opening reaction, linear C5 was generated on both 1 ML and 2 ML NaCl/Au(111) surfaces with 100 % yield (81 precursors were tested in the experiments), and no cyclic form was observed (Fig. 1b). Moreover, importantly, bond-resolved AFM images represent a nearly uniform feature resembling the cumulenic structures reported before9. This naturally indicates a cumulenic structure for linear C5, in accordance with the relaxed structure of C5 on the NaCl surface. To further explore the structures and their related electronic states of linear Cn with different number of carbon atoms, we achieved tip-induced coupling reactions of linear C5, and successfully generated various carbon chains including C9, C10, C13, C14, C15, C17, C18, C21 on the NaCl surface, together with characterization of their structures and measurement of electronic states.
Results and discussion
To generate linear C5, C5Br6 molecules were introduced on the cold NaCl/Au(111) held at ~6 K. All experiments were conducted at 4.7 K. The AFM and Laplace-filtered AFM images in Fig. 1c I, d I show C5Br6 molecule adopts a non-planer geometry, with only the topmost Br atom visible as a round protrusion. To trigger dehalogenation reactions, the tip was initially positioned on a single C5Br6 molecule, and retracted by ~6–8 Å from a setpoint (typically I = 0.5 pA, V = 0.3 V), after that, ~4 V pulses were applied on the molecule with currents on the order of hundreds of fA. This procedure typically resulted in the removal of one bromine atom, forming a C5Br5 intermediate (Fig. 1c II, d II, with one bromine atom aside). Subsequent voltage pulses applied to the C5Br5 intermediates can induce further dehalogenations and accompanied ring-opening reaction, resulting in the final product of linear C5 (Fig. 1c III, d III, with six bromine atoms aside). Note that cyclic C5 was not generated on either 1 ML or 2 ML NaCl surface, different from the case of C613.
AFM images of linear C5 (Fig. 1c III, d III, e and Supplementary Fig. 1) clearly reveal a uniform contrast, indicating linear C5 has a cumulenic structure, different from the polyynic linear C6 on the NaCl surface. Previous theoretical studies predicted that linear C5 adopts a cumulenic structure as the lowest energy geometry in the gas phase32,33,34 (see also the bond length calculations in Fig. 1f at the ωB97XD/def2-TZVP level). When relaxing the cumulenic linear C5 on the NaCl surface with the experimentally determined registry (Fig. 1g), it was found that linear C5 still maintains the cumulenic structure (Fig. 1f, h), consistent with the experimental results.
Interestingly, when applying voltage pulses ( ~ 4 V) between two isolated linear C5, we observed that they could couple together and lead to the formation of a linear C10 (Fig. 2a, b I, b II). Moreover, it is also feasible to couple a linear C10 and a linear C5 to generate a linear C15 (Fig. 2a, c I, c II).
a Reaction scheme for the formations of linear C10 and C15 via tip-induced coupling reaction of linear C5. b I AFM image of two isolated linear C5. b II After applying voltage pulses, a linear C10 was generated. c I AFM image of isolated linear C10 and C5. c II After applying voltage pulses, a linear C15 was generated. The scale bar in (b I) applies to all AFM images. AFM tip offsets Δz: −1.5 Å, −1.3 Å, −1.4 Å, −0.7 Å for b I, b II, c I, c II. Reference set points of Δz: I = 0.5 pA, V = 0.3 V for all AFM images.
We found that the efficiency of the tip-induced coupling reaction can be significantly improved by applying voltage pulses up to 4.5 V and reducing the tip height with currents up to about 10–50 pA. In addition, an appropriate molecular coverage (e.g., 0.3 ML) also helps to increase the chances of molecules encountering each other, thereby facilitating the formation of various carbon chains (Fig. 3a). Note that in these processes carbon-carbon bonds may also be broken. By optimizing the parameters, additional coupling-reaction products, including linear C9, C13, C14, C17, C18, C21 can also be generated (Fig. 3b–i). The assignments of these carbon chains are detailed in Supplementary Figs. 2 to 4.
a I, a II STM images before (a I) and after (a II) voltage pulses. (a III) Statistical analysis. (b I to b V, c I to c V, d I to d V, e I to e V, f I to f V, g I to g V, h I to h V, i I to i V) AFM images, Laplace-filtered AFM images at far and close tip height together with superimposed model of linear C9, C10, C13, C14, C15, C17, C18, C21. The arrows in c IV, e IV, h IV indicate the carbon-carbon triple bonds of linear C10, C14, C18, respectively, and the blue arrows indicate the invisible triple bonds at the ends. The scale bar in (b I) applies to all experimental and Laplace-filtered AFM images. AFM tip offsets Δz: +0.7 Å, +0.5 Å, −1.5 Å, −1.7 Å, +0.5 Å, +0.3 Å, −1.0 Å, −1.2 Å, −0.7 Å, −1.2 Å, +0.7 Å, +0.6 Å, +0.8 Å, +0.6 Å, +0.7 Å, +0.4 Å for b I, b III, c I, c III, d I, d III, e I, e III, f I, f III, g I, g III, h I, h III, i I and i III. Reference set points of Δz: I = 5 pA, V = 0.3 V for e I and e III, I = 0.5 pA, V = 0.3 V for all AFM images except e I and e III.
Our calculations and literature32,33,34 show that in the gas phase, these carbon chains illustrated in Fig. 3 are all cumulenic (Supplementary Fig. 5). While, on the NaCl surface, for even-numbered linear C10, AFM images at far and close tip heights reveal three and five distinct characteristic bright features, which can be assigned to the five triple bonds12,26, indicating that linear C10 adopts a polyynic structure. At a larger tip height (e.g., Fig. 3c I and c II), two triple bonds at both ends are less distinct, likely due to the non-planer adsorption configuration of linear C10 on the NaCl surface (see the relaxed structure in Supplementary Fig. 2b I to b III). For the longer linear C14 and C18, AFM images at both far and close tip heights can only reveal five and seven distinct characteristic bright features for triple bonds, and another two triple bonds at both ends for C14 and C18 are invisible (indicated by blue arrows in Fig. 3e IV and h IV). These experimental results and calculations (see Supplementary Fig. 2b I to b III, d I to d III, g I to g III) show that linear C10, C14 and C18 undergo surface-induced Peierls transitions (i.e., cumulenic one in the gas phase and polyynic one on the surface), similar to the case of linear C613.
For the odd-numbered linear C9, a nearly uniform contrast is observed (Fig. 3b), similar to C5, and our calculations show that C9 has rather small bond length alternations (Supplementary Fig. 2a I to a III), we thus assign it a cumulene-like structure9 (see the model in Fig. 3b V). Note that the observed apparent length of C9 is shorter than the model because of the two invisible bonds at both ends. While, for other longer ones, i.e., C13, C15, C17, and C21, AFM images at larger tip height normally show fainted dots (but different from the characteristic features of triple bonds), and at closer tip height, nearly uniform contrasts are observed together with two less distinct dots at both ends (similar to the even-numbered chains). In accordance with our calculations (Supplementary Fig. 2c I to c III, e I to e III, f I to f III, h I to h III), for example, the relaxed structure of linear C15 on the NaCl surface reveals that the BLAs are significantly larger at the two ends compared to the middle, but overall, smaller than the even-numbered ones. We tentatively assign hybrid structures for the odd-numbered linear C13, C15, C17, and C21, in which the both ends exhibit triple bond characteristics and the remaining moieties are still cumulene-like ones (see the models in Fig. 3d V, f V, g V, i V).
These results suggest that on the NaCl surface Peierls transition completely occurs for the even-numbered linear C10, C14, C18, while, only partly occurs for the odd-numbered linear C13, C15, C17, C21. Cumulene-like linear C9 may exhibit a Peierls-transition intermediate between cumulenic linear C5 and partly polyynic linear C13.
To further investigate the relationship between the structures and related electronic properties of the carbon chains above, we performed scanning tunneling spectroscopy (STS) measurements. The dI/dV spectra, represented by the blue and red lines, were acquired over the triple and single bonds of linear C10, C14 and C18, respectively. For the odd-numbered ones, the blue and red lines are acquired at the dot and between the dots, respectively.
STS measurements reveal that the transport gaps of the odd-numbered chains are dramatically smaller than that of the adjacent even-numbered chains, e.g., C9 vs C10, C13 vs C14, C17 vs C18 (Supplementary Fig. 6). As shown in Fig. 4, for linear C9, the dI/dV spectra show two prominent peaks at approximately V = −1.75 V and 1.46 V, yielding a transport gap of about 3.21 eV. STM images obtained at these bias voltages correspond to the positive ion resonance (PIR) and negative ion resonance (NIR) states. For both PIR and NIR, the STM images show five distinct lobes. For linear C10, the spectra show two prominent peaks at approximately V = −2.0 V and 3.3 V, yielding a transport gap of about 5.3 eV. For PIR, the STM image shows five lobes, while, for NIR, the lobes numbered 2, 3, 4 cannot be separately resolved.
a Scanning tunneling spectroscopy (STS) of linear C9, C10, C13, C14, C15, C17, C18, C21 conducted on the NaCl surface, in which linear C10 and C14 were done on the 1 ML NaCl surface, and the others were done on the 2 ML NaCl surface. The differential conductance (dI/dV) signals show two peaks that can be attributed to the positive ion resonance (PIR) and negative ion resonance (NIR) states. b AFM images, in-gap STM images (0.3 V), STM images (PIR), and STM images (NIR) of linear C9, C10, C13, C14, C15, C17, C18, C21. The blue and red lines in a are conducted at the positions on the blue and red crosses on AFM images in b. The numbers on STM images (PIR and NIR) indicate the number of lobes. The STS spectra and STM images are all acquired with CO-terminated tip. The scale bar in (b) applies to all STM and AFM images.
For linear C13, the dI/dV spectra show two prominent peaks at approximately V = −1.65 V and 1.3 V, yielding a transport gap of about 2.95 eV. STM images obtained at PIR and NIR show seven distinct lobes. For linear C14, the dI/dV spectra show two prominent peaks at approximately V = −2.8 V and 2.5 V, yielding a transport gap of about 5.3 eV. For PIR, the STM image shows seven lobes, while, for NIR, the lobes numbered from 2 to 6 cannot be separately resolved.
For linear C17, the dI/dV spectra show two prominent peaks at approximately V = −1.63 V and 1.16 V, yielding a transport gap of about 2.79 eV. STM images obtained at PIR and NIR show nine distinct lobes. For linear C18, the dI/dV spectra show two prominent peaks at approximately V = −2.75 V and 2.0 V, yielding a transport gap of about 4.75 eV. STM images obtained at PIR and NIR show nine and ten distinct lobes, respectively. For the other two even-numbered linear C10 and C14, it is rather challenging to measure the entire dI/dV spectra on a 2 ML NaCl surface due to quite large transport gaps, i.e., at larger bias voltages, the molecular carbons normally diffuse around on the surface.
From the comparison above, it is shown that the transport gaps for the even-numbered carbon chains (with polyynic structures) are dramatically larger than the odd-numbered ones (with partly cumulenic structures), which is in a good agreement with Peierls transition theory14,15,16.
Additionally, for the linear C15, the dI/dV spectra show two prominent peaks at approximately V = −1.6 V and 1.2 V, yielding a transport gap of about 2.8 eV. STM images obtained at PIR and NIR show eight distinct lobes. For linear C21, the dI/dV spectra show two prominent peaks at approximately V = −1.6 V and 1.15 V, yielding a transport gap of about 2.75 eV. STM images obtained at PIR and NIR show eleven distinct lobes. We have also calculated the frontier orbitals of the carbon chains that reasonably matched well with the experimental results (cf. Supplementary Fig. 7 to S10). In above, the transport gaps for both odd-and even-numbered carbon chains gradually decrease with increasing number of carbon atoms.
To demonstrate the generality, linear C6 was considered as the initial point for further coupling reactions. To do so, C6Br6 precursors were introduced on the cold NaCl/Au(111) held at ~6 K (Fig. 5a). To induce the coupling reactions, similarly, voltage pulses up to 4.5 V were applied with currents up to about 10–50 pA. As expected, we find that the linear C6 can also fuse together and form longer carbon chains (Fig. 5b). The upper one in Fig. 5b was further characterized and assigned to a linear C23. The AFM image (Fig. 5d) shows similar characteristics to those odd-numbered ones, like linear C13, C15, C17, C21. The dI/dV spectra show two prominent peaks at approximately V = −1.1 V and 0.9 V, yielding a transport gap of about 2.0 eV (Fig. 5c). For both PIR and NIR, the STM images show twelve distinct lobes (Fig. 5d), confirming the assignment of the linear C23.
STM images (0.3 V) before (a) and after (b) voltage pulses. Two carbon chains were generated after applying voltage pulses. Inset: AFM image of a C6Br6. c STS of the linear C23 (the upper one in b). d AFM image, in-gap STM image (0.3 V), STM image (PIR, −1.1 V), and STM image (NIR, 0.9 V) of the linear C23. The numbers on the STM images (PIR and NIR) indicate the number of lobes. The STS spectra and STM images are all acquired with CO-terminated tip. The scale bar in (d) applies to all STM and AFM images in (d).
Moreover, we find that some longer carbon chains have shown the flexibility with curved geometries, for example, the generated linear C30 demonstrates the possibility as the precursor to forming the corresponding cyclocarbon (Supplementary Fig. 11).
In conclusion, we have demonstrated the feasibility of generating various carbon chains via tip-induced coupling reactions of linear C5 or C6. This enables to experimentally identify that on the NaCl surface, Peierls transition completely occurs for even-numbered carbon chains, leading to the formation of polyynic structure, while, for odd-numbered ones, the both ends exhibit triple bond characteristics and the remaining moieties are still cumulene-like. Such a structural difference between odd- and even-numbered carbon chains was reflected by STS measurements that the electronic transport gaps of the odd-numbered chains are smaller than that of the adjacent even-numbered chains due to Peierls transition. This study offers an approach for atomically precise synthesis of a series of uncapped carbon chains, and may also provide a new strategy to synthesize their corresponding isomers, cyclocarbons.
Methods
Experimental details for STM and AFM measurements
STM and AFM measurements were carried out in a commercial (Createc) low-temperature system operated at 4.7 K with a base pressure better than 1 × 10−10 mbar. The single crystalline Au(111) surface was cleaned by several sputtering and annealing cycles. The NaCl films were obtained by thermally evaporating NaCl crystals onto a clean Au(111) surface at room temperature, resulting in islands of one and two monolayer (ML) thickness. The perbromocyclopenta-1,3-diene (C5Br6, purchased from Shaanxi Yikunte Pharmaceutical Technology Co., Ltd., 98 %) and hexabromobenzene (C6Br6, purchased from Adamas-beta, 98 %) molecules were separately deposited on cold NaCl/Au(111) surface by thermal sublimation from a molecular evaporator (sublimation temperature: 44˚C for C5Br6, 85 ˚C for C6Br6). CO molecules for tip modification42 were dosed onto the cold sample via a leak valve. We used qPlus sensors43 with a resonance frequency f0 = 29.49 kHz, quality factor Q ≈ 45,000 and a spring constant k ≈ 1800 N/m operating in frequency-modulation mode44. The bias voltage V was applied to the sample with respect to the tip. AFM images were acquired in constant-height mode at V = 0 V and an oscillation amplitude of A = 1 Å. The tip-height offsets Δz for constant-height AFM images are defined as the offset in tip-sample distance relative to the STM set point at the NaCl surface. The positive (negative) values of Δz correspond to the tip-sample distance increased (decreased) with respect to a STM set point. The charge-state determination of linear carbons on NaCl was shown in Supplementary Fig. 12.
Density functional theory calculations
Density functional theory (DFT) calculations were carried out in the gas phase using Gaussian 16 program package45. ωB97XD exchange-correlation functional46 in conjunction with def2-TZVP47 basis sets was used for linear carbons calculations in gas phase.
The Vienna ab initio simulation package (VASP)48,49 was used to perform the DFT calculations on NaCl surface. For describing the interaction between electrons and ions, the projector-augmented wave method50,51 was used, and the Perdew−Burke−Ernzerhof generalized gradient approximation exchange−correlation functional was employed52. Van der Waals corrections to the PBE density functional were also included using the DFT-D3 method of Grimme53. The kinetic energy cutoff was set to 400 eV. We used a 1 ML NaCl(001) slab on top of Au(111) substrate modeled by a three-layer slab, and the bottom layer of the Au(111) was fixed. The atomic structures were relaxed until the atomic forces were less than 0.03 eV/Å. Simulated STM images were obtained based on the Tersoff−Hamann method54,55.
Data availability
All data are available from the corresponding author upon request. Source data are provided with this paper.
References
Baughman, R. H. Dangerously seeking linear carbon. Science 312, 1009–1110 (2006).
Kavan, L. Electrochemical carbon. Chem. Rev. 97, 3061–3082 (1997).
Heimann, R. B., Evsyukov, S. E. & Kavan, L. Carbyne and Carbynoid Structures Vol. 21 (Springer Science & Business Media, 1999).
Liu, M., Artyukhov, V. I., Lee, H., Xu, F. & Yakobson, B. I. Carbyne from first principles: Chain of C atoms, a nanorod or a nanorope. ACS Nano 7, 10075–10082 (2013).
El Goresy, A. & Donnay, G. A new allotropic form of carbon from the Ries crater. Science 161, 363–364 (1968).
Pu, L., Zhang, Z., King, R. B. & Allen, W. D. Most favorable cumulenic structures in iron-capped linear carbon chains are short singlet odd-carbon dications: a theoretical view. Phys. Chem. Chem. Phys. 20, 15496–15506 (2018).
Kaiser, K. et al. An sp-hybridized molecular carbon allotrope, cyclo[18]carbon. Science 365, 1299–1301 (2019).
Scriven, L. M. et al. Synthesis of cyclo[18]carbon via debromination of C18Br6. J. Am. Chem. Soc. 142, 12921–12924 (2020).
Sun, L. et al. On-surface synthesis of aromatic cyclo[10]carbon and cyclo[14]carbon. Nature 623, 972–976 (2023).
Gao, Y. et al. On-surface synthesis of a doubly anti-aromatic carbon allotrope. Nature 623, 977–981 (2023).
Albrecht, F. et al. The odd-number cyclo[13]carbon and its dimer, cyclo[26]carbon. Science 384, 677–682 (2024).
Sun, L. et al. On-surface synthesis and characterization of anti-aromatic cyclo[12]carbon and cyclo[20]carbon. Nat. Commun. 15, 7649 (2024).
Sun, L., Guo, Y., Xiang, W. & Xu, W. On-surface synthesis and characterization of linear and cyclic C6. Nat. Synth. 4, 940–946 (2025).
Tommasini, M. et al. Intramolecular vibrational force fields for linear carbon chains through an adaptative linear scaling scheme. J. Phys. Chem. A 111, 11645–11651 (2007).
Jacquemin, D. & Champagne, B. Optimizing the geometry of stereoregular polymers. III. Polyyne and the basis set quasi-linear dependence. Int. J. Quant. Chem. 80, 863–870 (2000).
Peierls, R. Quantum Theory of Solids (Oxford University Press, 1955).
Cataldo, F. Submerged electric arc between graphite electrodes: A one-pot tool for the synthesis of long-chain polyynes in solution. Tetrahedron Lett. 45, 141–144 (2004).
Matsutani, R. et al. Preparation of polyynes up to C22H2 by liquid-phase laser ablation and their immobilization into SiO2 gel. Carbon 47, 1659–1663 (2009).
Matsutani, R. et al. Preparation of long-chain polyynes C18H2 and C20H2 by laser ablation of pellets of graphite and perylene derivative in liquid phase. Carbon 46, 1103–1106 (2008).
Pan, B. et al. Carbyne with finite length: The one-dimensional sp carbon. Sci. Adv. 1, e1500857 (2015).
Jin, C., Lan, H., Peng, L., Suenaga, K. & Iijima, S. Deriving carbon atomic chains from graphene. Phys. Rev. Lett. 102, 205501 (2009).
Patrick, C. W. et al. Masked alkynes for synthesis of threaded carbon chains. Nat. Chem. 16, 193–200 (2023).
Gao, Y. et al. The loss of endgroup effects in long pyridyl-endcapped oligoynes on the way to carbyne. Nat. Chem. 12, 1143–1149 (2020).
Chalifoux, W. A. & Tykwinski, R. R. Synthesis of polyynes to model the sp-carbon allotrope carbyne. Nat. Chem. 2, 967–971 (2010).
Shi, L. et al. Confined linear carbon chains as a route to bulk carbyne. Nat. Mater. 15, 634–639 (2016).
Pavliček, N. et al. Polyyne formation via skeletal rearrangement induced by atomic manipulation. Nat. Chem. 10, 853–858 (2018).
Gao, W. et al. On-surface synthesis and characterization of polyynic carbon chains. Natl Sci. Rev. 11, nwae031 (2024).
Januszewski, J. A. & Tykwinski, R. R. Synthesis and properties of long [n]cumulenes (n ≥ 5). Chem. Soc. Rev. 43, 3184–3203 (2014).
Krause, N. Science of Synthesis Vol. 44 (Thieme Chemistry, 2008).
Wendinger, D. & Tykwinski, R. R. Odd [n]cumulenes (n = 3, 5, 7, 9): Synthesis, characterization, and reactivity. Acc. Chem. Res. 50, 1468–1479 (2017).
Franz, M. et al. Cumulene rotaxanes: stabilization and study of [9]cumulenes. Angew. Chem. Int. Ed. 54, 6645–6649 (2015).
Van Orden, A. & Saykally, R. J. Small carbon clusters: spectroscopy, structure, and energetics. Chem. Rev. 98, 2313–2357 (1998).
Giuffreda, M. G., Deleuze, M. S., François, J. P. & Trofimov, A. B. Theoretical study of the vertical electron excitation of linear carbon clusters C3, C5, and C7. Int. J. Quantum Chem. 85, 475–491 (2001).
Yang, S. & Kertesz, M. Linear Cn clusters: are they acetylenic or cumulenic?. J. Phys. Chem. A 112, 146–151 (2007).
Hutter, J., Lüthi, H. P. & Diederich, F. Structures and vibrational frequencies of the carbon molecules C2-C18 calculated by density functional theory. J. Am. Chem. Soc. 116, 750–756 (1994).
Martin, J. M. L. & Taylor, P. R. Structure and vibrations of small carbon clusters from coupled-cluster calculations. J. Phys. Chem. 100, 6047–6056 (1996).
Yen, T. W. & Lai, S. K. Use of density functional theory method to calculate structures of neutral carbon clusters Cn (3 ≤ n ≤ 24) and study their variability of structural forms. J. Chem. Phys. 142, 084313 (2015).
Brito, B. G. A., Hai, G. Q. & Cândido, L. Quantum Monte Carlo study on the structures and energetics of cyclic and linear carbon clusters Cn (n = 1,…,10). Phys. Rev. A 98, 062508 (2018).
Parasuk, V. & Almlöf, J. The electronic and molecular structure of C6: Complete active space self-consistent-field and multireference configuration interaction. J. Chem. Phys. 91, 1137–1141 (1989).
Pavliček, N. et al. On-surface generation and imaging of arynes by atomic force microscopy. Nat. Chem. 7, 623–628 (2015).
Zhong, Q. et al. Constructing covalent organic nanoarchitectures molecule by molecule via scanning probe manipulation. Nat. Chem. 13, 1133–1139 (2021).
Gross, L., Mohn, F., Moll, N., Liljeroth, P. & Meyer, G. The chemical structure of a molecule resolved by atomic force microscopy. Science 325, 1110–1114 (2009).
Giessibl, F. J. High-speed force sensor for force microscopy and profilometry utilizing a quartz tuning fork. Appl. Phys. Lett. 73, 3956–3958 (1998).
Albrecht, T. R., Grütter, P., Horne, D. & Rugar, D. Frequency modulation detection using high-Q cantilevers for enhanced force microscope sensitivity. J. Appl. Phys. 69, 668–673 (1991).
Frisch, M. J. et al. Gaussian 16 Rev. C.01; Gaussian, Inc. (Wallingford, CT., 2016).
Chai, J. D. & Head-Gordon, M. Long-range corrected hybrid density functionals with damped atom-atom dispersion corrections. Phys. Chem. Chem. Phys. 10, 6615–6620 (2008).
Weigend, F. & Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy. Phys. Chem. Chem. Phys. 7, 3297–3305 (2005).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for open-shell transition metals. Phys. Rev. B 48, 13115–13118 (1993).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Vanpoucke, D. E. P. & Brocks, G. Formation of Pt-induced Ge atomic nanowires on Pt/Ge(001): A density functional theory study. Phys. Rev. B 77, 241308 (2008).
Tersoff, J. & Hamann, D. R. Theory of the scanning tunneling microscope. Phys. Rev. B 31, 805–813 (1985).
Acknowledgements
The authors acknowledge the financial support from the National Natural Science Foundation of China (22125203 to W.Xu., 22402149 to L.S.), the National Key R&D Program of China (2023YFE0101900 to W.Xu.), the Ministry of Science and Technology of the People’s Republic of China, and the Shanghai Science and Technology Program (24ZR1470000 to L.S.).
Author information
Authors and Affiliations
Contributions
W.Xu. conceived the research; L.S., Y.G, W.Xiang, and M.Z. performed the STM/AFM experiments, L.S. and Y.G. carried out the DFT calculations, all authors contributed to writing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Sun, L., Guo, Y., Xiang, W. et al. The cumulenic linear C5 and its coupling-reaction products. Nat Commun 16, 9222 (2025). https://doi.org/10.1038/s41467-025-64310-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-64310-4
This article is cited by
-
On-surface synthesis of cyclo[20]carbon and cyclo[30]carbon from cyclo[10]carbon
Nature Communications (2025)