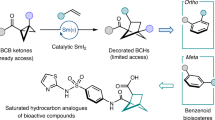
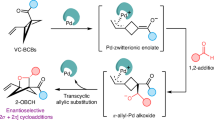
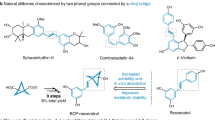
Abstract
Bicyclo[2.1.1]hexanes (BCHs), three-dimensional benzene bioisosteres characterized by high sp3-carbon content, hold great promise for diverse applications in medicinal chemistry. Although significant advances have been made in the synthesis of racemic BCHs, highly enantioselective approaches remain comparatively rare. Here we report a mild, secondary amine–catalyzed asymmetric [2π + 2σ] cycloaddition of bicyclo[1.1.0]butanes (BCBs) with α,β-unsaturated aldehydes, which overcomes key limitations of existing metal-catalyzed and photochemical methods. The protocol operates under ambient air and tolerates a wide range of BCB and aldehyde substrates bearing diverse functional groups, affording BCH scaffolds in yields of up to 84% under Supramolecular Iminium Catalysis with excellent enantioselectivity (up to 99% ee) and high diastereoselectivity (>20:1 dr). The mild conditions and operational simplicity underscore the potential of this transformation for stereoselective manufacturing of BCHs at scale. Mechanistic experiments and DFT studies support an acid-promoted dual activation of both substrates, followed by an enamine–iminium tandem catalytic process that delivers the enantioenriched products.
Similar content being viewed by others
Introduction
Benzene rings represent the most frequently encountered structural motifs in pharmacopeia, serving either as primary bioactive cores or as topological anchors for pharmacophore alignment. Despite their ubiquity, recent studies have highlighted that the indiscriminate incorporation of phenyl rings often imparts suboptimal physicochemical properties to advanced molecules, ultimately limiting their development into effective therapeutic agents1. To overcome these drawbacks, the concept of “escaping from flatland” has emerged, emphasizing the design of F(sp³)-rich scaffolds. Increased carbon saturation enhances molecular solubility and three-dimensionality, thereby improving receptor binding selectivity, reducing off-target interactions, and raising the probability of clinical success2,3. For example, Stepan and Mykhailiuk independently demonstrated that substituting phenyl rings with saturated three-dimensional bioisosteres can significantly improve drug-like properties4,5,6,7. Among such bioisosteres, bicyclo[2.1.1]hexanes (BCHs) have emerged as privileged replacements for aromatic rings (Fig. 1a). They offer unique opportunities to simultaneously optimize pharmacological performance and expand intellectual property space, which has spurred strong interest in developing efficient synthetic routes8,9,10,11,12. One of the most powerful strategies relies on the [2π + 2σ] cycloaddition of bicyclo[1.1.0]butanes (BCBs) with π-systems, a transformation that delivers BCHs with excellent atom economy and synthetic efficiency13,14,15,16,17.
a BCHs as 3D benzene bioisosteres. b Previous works focused on construction of BCHs. c Previous work: Enantioselective [2π + 2σ] cycloaddition via metal or photocatalysis. d This work: Organocatalytic enantioselective [2π + 2σ] cycloaddition.
Significant progress has been made toward the synthesis of racemic BCHs, notably via Lewis acid catalysis18,19,20,21,22,23,24,25,26,27,28,29, radical-mediated strategies30,31,32,33,34,35,36,37,38, and triplet-energy-transfer-enabled [2π + 2σ] cycloadditions (Fig. 1b)39,40,41,42. However, studies by Baran and co-workers have shown that enantiomerically distinct BCH analogs can exhibit dramatically different biological activities when serving as phenyl bioisosteres43. This underscores the critical importance of developing stereoselective methods for BCH synthesis. Early contributions include Bach’s pioneering work, which achieved enantioselective additions of BCBs to quinolones using a chiral template (two equivalents) combined with triplet-energy transfer under 366 nm irradiation at –60 °C44. Subsequently, Jiang’s group reported a bifunctional chiral phosphoric acid–catalyzed process between BCBs and vinylazaarenes under blue LED light at –40 °C45. More recently, Hong and Liu independently realized asymmetric [2π + 2σ] cycloadditions of BCBs with α,β-unsaturated ketones and coumarins using Lewis acid catalysis with chiral ligands (Fig. 1c)46,47. In parallel, Zheng and Zi independently reported palladium-catalyzed enantioselective additions of vinyl–carbonyl–BCBs to alkenes48,49. Photocatalytic asymmetric cycloadditions of BCBs with α,β-unsaturated ketones, vinyl azides, and naphthalenes have also been disclosed by Yoon, Zheng, and You using chiral Lewis or Brønsted acid systems (see Supplementary Fig. S1)50,51,52.
While these elegant studies provide viable strategies for accessing enantioenriched BCHs, they generally suffer from several drawbacks: (i) reliance on cryogenic conditions under inert atmospheres, (ii) requirement for specialized photochemical setups, and (iii) use of potentially toxic transition metals, which diminishes their suitability for pharmaceutical applications. To overcome these limitations, we envisioned a mechanistically distinct approach—leveraging secondary amine catalysis under ambient air, at room temperature, and under operationally simple conditions. This strategy not only expands the synthetic toolbox for BCHs but also addresses key challenges in developing practical, sustainable, and pharmaceutically compatible methods.
Results
Condition optimization
To evaluate the feasibility of our design, we first examined the reaction between cinnamaldehyde (1a) and pyrazole amide–substituted bicyclo[1.1.0]butane (2a) as model substrates (Table 1). A range of secondary amines were screened. With imidazolinone 4a as the catalyst, the desired product 3a was obtained in 75% yield but with only 8% ee. Seeking improved enantioselectivity, we tested sterically hindered catalyst 4b; however, the outcome was unsatisfactory. Seeking improved enantioselectivity, we tested sterically hindered catalyst 4b; however, the outcome was unsatisfactory. In contrast, the use of diarylprolinol silyl ether 4c enhanced both yield and enantioselectivity. Encouraged by this improvement, we further tested the more reactive and sterically demanding catalyst 4d. Although the yield decreased slightly, product 3a was obtained with very high enantioselectivity. We next screened solvents and found acetone to be the optimal medium. To further enhance efficiency, we incorporated hydrogen-bonding catalyst 5 based on our previously established supramolecular iminium ion catalysis concept, followed by comprehensive screening and optimization (see Supplementary Table S1)53,54,55,56. Ultimately, the use of auxiliary catalyst 5a afforded 3a in 82% yield and 99% ee within 24 h.
Substrate scope
With optimal conditions established, we explored the substrate scope of aldehydes (Fig. 2). Substituents such as methyl (3b), methoxy (3c), or halides (3d–3f) at the para-position of cinnamaldehyde maintained excellent enantioselectivity, though yields were slightly reduced. Similar trends were observed for meta-substitution (3m–3r), highlighting sensitivity to electronic effects. Aldehydes bearing a bulky tert-butyl group (3i) also gave excellent enantioselectivity. Importantly, substrates featuring synthetically versatile groups—including nitro (3g), cyano (3h), ester (3j), trifluoromethylthio, and biphenyl moieties (3k, 3l)—afforded the desired products in moderate yields with consistently high enantioselectivity. Beyond substituted phenyl groups, fused aryl and heteroaryl aldehydes proved compatible, delivering products 3s–3y with excellent enantioselectivity. However, ortho-substituted substrates (3za–3zc) suffered from steric hindrance that impeded reactivity. Interestingly, employing a naphthyl-substituted aldehyde not only improved reaction yield but also facilitated product isolation. Thus, substrate 1s was selected as the representative aldehyde for exploring the scope of BCBs. Substituted BCBs exhibited varied reactivity. Halogen or electron-donating groups at the para- or meta-positions of the phenyl ring afforded good results (3ze–3zi). However, para-electron-donating substituents led to competitive self-ring-opening under acidic conditions, reducing the yield to 42% (3zd). In contrast, strongly electron-withdrawing groups such as para-trifluoromethyl suppressed BCB activation, producing only trace product (3zj). Furthermore, BCBs bearing groups other than acyl pyrazole proved unstable under the reaction conditions, leading to low yields and facile decomposition57. By comparison, the acyl-pyrazole–substituted BCB exhibited superior stability, underscoring its unique suitability for this transformation (see Supplementary Fig. S4 for unsuccessful substrates).
Standard reaction conditions: aldehyde (0.1 mmol), BCB (0.15 mmol), cat. 4 d (20 mol%), cat. 5a (20 mol%), TFA (40 mol%) and acetone (2 mL).
Product derivatization
To assess the practicality of this protocol, we carried out a gram-scale synthesis of product 3s (3 mmol). The reaction maintained excellent enantioselectivity, with only a slight decrease in yield (–4%). The exposed aldehyde group of 3s proved highly versatile for downstream transformations. Under NaH conditions, it underwent a Wittig reaction to afford alkene 6 in 65% yield with complete retention of enantioselectivity. Reduction with NaBH4 produced alcohol 7 in 86% yield and 98% ee. The amide–pyrazole group could be smoothly converted into a methyl ester in 99% yield under DBU/MeOH conditions, while aldehyde protection with ethylene glycol afforded acetal 9, enabling single-crystal X-ray analysis (CCDC 2441435). Moreover, the aldehyde functionality was transformed into a terminal alkyne in 96% yield using the Bestmann–Ohira reagent. This alkyne intermediate served as a versatile handle for further diversification: it participated in a Yamanaka–Sakamoto–Sonogashira indole synthesis to furnish indole 11 (92% yield, 94% ee), underwent Sonogashira coupling with aryl iodides to provide 12 (79% yield, 93% ee), and engaged in CuAAC “click” chemistry to yield triazole 13 (85% yield, 91% ee) (Fig. 3).
Scale-up synthesis and post-synthetic modifications.
We further demonstrated the utility of 3s by oxidizing the aldehyde group to carboxylic acid 14 via Pinnick oxidation (Fig. 4). This transformation greatly expanded opportunities for late-stage functionalization with bioactive molecules. Through amidation or esterification, we successfully conjugated 14 with amine-containing compounds—including tryptamine (16), rivaroxaban (17), fluvoxamine (18), oseltamivir (20), dehydroabietylamine (21), and linagliptin (25)—as well as alcohol-containing molecules such as geraniol (15), cholesterol (19), diacetone-D-glucose (22), pregnenolone (23), L-menthol (24), and testosterone (26). These reactions furnished a diverse library of biologically relevant BCH analogs in good to excellent yields (58–98%).
Late-stage functionalization of the BCH scaffold for bioactive molecule diversification.
Mechanistic investigation
To gain insight into the reaction pathway, we performed a series of control experiments (Fig. 5a). The reaction did not proceed in the absence of either the secondary amine catalyst or the acid, demonstrating that both components are essential. To clarify the role of the acid, we systematically evaluated a range of acids. Neither weakly acidic benzoic acid (BA) nor strongly acidic p-toluenesulfonic acid (TsOH) delivered the target product (Fig. 5b). 1H NMR studies (Supplementary Fig. S5) confirmed that BA failed to promote condensation between the aldehyde and secondary amine, thereby preventing iminium ion formation and halting the reaction. In contrast, TsOH successfully promoted iminium ion generation but its excessive acidity induced rapid ring-opening of the BCB substrate, leading to nearly quantitative formation of ring-opened byproducts and preventing product formation. When 1a was treated with acid alone, we isolated product 27, confirming that BCB ring-opening can occur under acidic conditions in the absence of the amine catalyst.
a Control experiments. b Reaction with different acid. c Proposed catalytic cycle.
Based on these results, we propose the mechanism outlined in Fig. 5c. The secondary amine catalyst 4d and hydrogen-bonding catalyst 5a undergo acid-promoted condensation with aldehyde 1a to form supramolecular iminium intermediate A. The acid-activated BCB then attacks the methylene carbon of A, generating carbocation intermediate D, which subsequently undergoes cyclization with the enamine. The tandem enamine–imine catalytic cycle ultimately regenerates the secondary amine catalyst, affording the target product 3.
DFT calculations
To further support this proposal, density functional theory (DFT) calculations were carried out (Fig. 6). The transition state ts-a-II, formed from 2a in the presence of TFA, was stabilized by 5.6 kcal/mol compared with the TFA-free system ts-a, with subsequent steps also exhibiting reduced energy barriers. These results corroborate the role of TFA in activating the BCB moiety. Additionally, we evaluated the effect of catalyst 5a on the iminium ion pair. Prior to its introduction, the N–O distance measured 2.92 Å. Incorporation of 5a led to a 1.6 kcal/mol stabilization and increased the N–O distance to 3.20 Å. This indicates that 5a weakens electrostatic interactions between the trifluoroacetate anion and the iminium cation, thereby stabilizing the ion pair and improving reactivity. Notably, catalyst 5a also enhances the electrophilicity of the iminium double bond, particularly at the methylene carbon, facilitating nucleophilic attack by the BCB-derived carbanion. These computational insights strongly support the cooperative catalytic role of 5a in the reaction.
a Investigated whether TFA assists in the ring-opening of BCBs through DFT calculations. b Investigated the influence of hydrogen-bonding catalysts on iminium ion pairs through DFT calculations.
Discussion
In summary, we have developed a secondary amine–catalyzed enantioselective [2π + 2σ] cycloaddition of BCBs with α,β-unsaturated aldehydes. The reaction proceeds efficiently under ambient conditions without the need for an inert atmosphere, displaying broad functional group tolerance and delivering products with outstanding enantioselectivity (up to 99% ee). Beyond the direct transformation, the resulting products proved highly versatile, undergoing a variety of downstream modifications to furnish structurally and functionally diverse derivatives. Mechanistic investigations, supported by control experiments and DFT studies, confirmed an acid-promoted secondary amine catalytic pathway. Overall, this work establishes a robust and practical platform for accessing chiral BCH scaffolds and offers a valuable complement to the limited existing methods for asymmetric BCB cycloadditions.
Methods
General procedure for enantioselective cycloaddition of and α,β-unsaturated aldehydes and bicyclo[1.1.0]butanes
To a 10 mL Schlenk tube equipped with a magnetic stir bar was added cat. 4d (0.02 mmol, 0.2 eq.), cat. 5a (0.02 mmol, 0.2 eq.), aldehydes 1 (0.1 mmol, 1.0 eq.), BCBs 2 (0.15 mmol, 1.5 eq.) and TFA (0.04 mmol, 0.4 eq.), then acetone (2 mL) was added. The resulting mixture was stirred at 10 °C for 24 h. Upon completion of the reaction, the reaction mixture was concentrated under reduced pressure, and the resulting crude mixture was purified by silica gel column chromatography to afford the pure product 3.
Data availability
The data that support the findings of this study are available within the main text and its Supplementary Information. Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2441435 (9). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. All data are available from the corresponding author upon request. Source data are provided with this paper.
Change history
16 March 2026
A Correction to this paper has been published: https://doi.org/10.1038/s41467-026-69148-y
References
Meanwell, N. A. & Subbaiah, M. A. M. Bioisosteres of the phenyl ring: recent strategic applications in lead optimization and drug design. J. Med. Chem. 6, 14046–14128 (2021).
Lovering, F., Bikker, J. & Humblet, C. Escape from flatland: increasing saturation as an approach to improving clinical success. J. Med. Chem. 52, 6752–6756 (2009).
Lovering, F. Escape from flatland 2: complexity and promiscuity. Med. Chem. Commun. 4, 515–519 (2013).
Stepan, A. F. et al. Application of the bicyclo[1.1.1]pentane motif as a nonclassical phenyl ring bioisostere in the design of a potent and orally active γ-secretase inhibitor. J. Med. Chem. 55, 3414–3424 (2012).
Mykhailiuk, P. K. Saturated bioisosteres of benzene: where to go next?. Org. Biomol. Chem. 1, 2839–2849 (2019).
Denisenko, A., Garbuz, P., Shishkina, S. V., Voloshchuk, N. M. & Mykhailiuk, P. K. Saturated bioisosteres of ortho-substituted benzenes. Angew. Chem. Int. Ed. 5, 20515–20521 (2020).
Levterov, V. V., Panasyuk, Y., Pivnytska, V. O. & Mykhailiuk, P. K. Water-soluble non-classical benzene mimetics. Angew. Chem. Int. Ed. 59, 7161–7167 (2020).
Rigotti, T. & Bach, T. Bicyclo[2.1.1]hexanes by visible light-driven intramolecular crossed [2 + 2] photocycloadditions. Org. Lett. 24, 8821–8825 (2022).
Hsu, C.-W., Lu, Y.-T., Lin, C.-P. & Yoo, W.-J. Synthesis of bicyclo[2.1.1]hexan-5-ones via a sequential Simmons-Smith cyclopropanation and an acid-catalyzed pinacol rearrangement of α-hydroxy silyl enol ethers. Adv. Synth. Catal. 365, 3082–3087 (2023).
Hsu, C.-W., Wu, C.-F., Lee, Y.-C. & Yoo, W.-J. Synthesis of 2-substituted bicyclo[2.1.1]Hexan-1-Ols via SmI2-mediated reductive cyclization reactions. Adv. Synth. Catal. 366, 4747–4754 (2024).
Posz, J. M. et al. Synthesis of borylated carbocycles by [2 + 2]-cycloadditions and photo-ene reactions. J. Am. Chem. Soc. 146, 10142–10149 (2024).
Harmata, A. S., Tatunashvili, E., Chang, A., Wang, T. & Stephenson, C. R. J. Bicyclo[2.1.1]hexanes via intramolecular formal (3+2)-cycloaddition. Angew. Chem. Int. Ed. 64, e202413695 (2025).
Kelly, C. B., Milligan, J. A., Tilley, L. J. & Sodano, T. M. Bicyclo-butanes: from curiosities to versatile reagents and covalent warheads. Chem. Sci. 13, 11721–11737 (2022).
Tyler, J. L. & Aggarwal, V. K. Synthesis and applications of bicyclo[1.1.0]butyl and azabicyclo[1.1.0]butyl organometallics. Chem. Eur. J. 29, e202300008 (2023).
Golfmann, M. & Walker, J. C. L. Bicyclobutanes as unusual building blocks for complexity generation in organic synthesis. Commun. Chem. 6, 9 (2023).
Cuadros, S. et al. Light-driven synthesis and functionalization of bicycloalkanes, cubanes and related bioisosteres. Angew. Chem. Int. Ed. 63, e202317333 (2024).
Yang, X.-C., Wang, J.-J., Xiao, Y.-J. & Feng, J.-J. Catalytic asymmetric synthesis of chiral caged hydrocarbons as arenes bioisosteres. Angew. Chem. Int. Ed. 64, e202505803 (2025).
Woelk, K. J., Dhake, K., Schley, N. D. & Leitch, D. C. Enolate addition to bicyclobutanes enables expedient access to 2-oxo-bicyclohexane scaffolds. Chem. Commun. 59, 13847–13850 (2023).
Ni, D. et al. Intermolecular formal cycloaddition of indoles with bicyclo[1.1.0]butanes by Lewis acid catalysis. Angew. Chem. Int. Ed. 62, e202308606 (2023).
Radhoff, N., Daniliuc, C. G. & Studer, A. Lewis acid catalyzed formal (3+2)-cycloaddition of bicyclo[1.1.0]butanes with ketenes. Angew. Chem. Int. Ed. 62, e202304771 (2023).
Tang, L. et al. Silver-catalyzed dearomative [2π+2σ] cycloadditions of indoles with bicyclobutanes: access to indoline fused bicyclo[2.1.1]hexanes. Angew. Chem. Int. Ed. 62, e202310066 (2023).
Lin, Z., Ren, H., Lin, X., Yu, X. & Zheng, J. Synthesis of azabicyclo[3.1.1]heptenes enabled by catalyst-controlled annulations of bicyclo[1.1.0]butanes with vinyl azides. J. Am. Chem. Soc. 146, 18565–18575 (2024).
Hu, S., Pan, Y., Ni, D. & Deng, L. Facile access to bicyclo[2.1.1]hexanes by Lewis acid-catalyzed formal cycloaddition between silyl enol ethers and bicyclo[1.1.0]butanes. Nat. Commun. 15, 6128 (2024).
Zhu, S., Tian, X. & Li, S.-W. Intermolecular formal [2π + 2σ] cycloaddition of enol silyl ethers with bicyclo[1.1.0]butanes promoted by Lewis acids. Org. Lett. 26, 6309–6313 (2024).
Su, J.-Y. et al. Access to spiro-bicyclo[2.1.1]hexanes via BF3·Et2O-catalyzed formal [2π + 2σ] cycloaddition of bicyclo[1.1.0]butanes with benzofuran-derived Oxa(Aza)dienes. Org. Chem. Front. 11, 4539–4545 (2024).
Hu, S., Gao, Y., Pan, Y., Ni, D. & Deng, L. Modular synthesis of azidobicyclo[2.1.1]hexanes via (3 + 2) annulation of α-substituted vinyl azides and bicyclo[1.1.0]butanes. J. Org. Chem. 89, 15151–15157 (2024).
Wang, J.-J. et al. Switching between the [2π+2σ] and hetero-[4π+2σ] cycloaddition reactivity of bicyclobutanes with Lewis acid catalysts enables the synthesis of spirocycles and bridged heterocycles. Angew. Chem. Int. Ed. 63, e202405222 (2024).
Hu, Q.-Q. et al. Lewis acid catalyzed cycloaddition of bicyclobutanes with ynamides for the synthesis of polysubstituted 2-amino-bicyclo[2.1.1]hexenes. Angew. Chem. Int. Ed. 63, e202405781 (2024).
Hu, Q.-Q., Geng, Z.-X., Bai, X., Chen, J. & Zhou, L. Lewis acid catalyzed divergent reaction of bicyclo[1.1.0]butanes with quinones for the synthesis of diverse polycyclic molecules. Angew. Chem. Int. Ed. 64, e202506228 (2025).
Xu, M. et al. Diboron(4)-catalyzed remote [3+2] cycloaddition of cyclopropanes via dearomative/rearomative radical transmission through pyridine. Angew. Chem. Int. Ed. 61, e202214507 (2022).
Agasti, S. et al. A catalytic alkene insertion approach to bicyclo[2.1.1]hexane bioisosteres. Nat. Chem. 15, 535–541 (2023).
Liu, Y. et al. Pyridine-boryl radical-catalyzed [2π + 2σ] cycloaddition of bicyclo[1.1.0]butanes with alkenes. ACS Catal. 13, 5096–5103 (2023).
Yan, H., Liu, Y., Feng, X. & Shi, L. Hantzsch esters enabled [2π+2σ] cycloadditions of bicyclo [1.1.0] butanes and alkenes under photo conditions. Org. Lett. 25, 8116–8120 (2023).
Dutta, S. et al. Photoredox-enabled dearomative [2π + 2σ] cycloaddition of phenols. J. Am. Chem. Soc. 146, 2789–2797 (2024).
Liu, Y. et al. Titanium catalyzed [2σ + 2π] cycloaddition of bicyclo[1.0]butanes with 1,3-dienes for efficient synthesis of stilbene bioisosteres. Nat. Commun. 1, 4374 (2024).
Tyler, J. L. et al. Bicyclo[1.1.0]butyl radical cations: synthesis and application to [2π + 2σ] cycloaddition reactions. J. Am. Chem. Soc. 146, 16237–16247 (2024).
Golfmann, M. et al. Photocatalytic oxidative activation of bicyclo[1.1.0]butanes for formal [2σ+2π] cycloadditions. ACS Catal. 14, 13987–13998 (2024).
Fu, Z., Cheng, J., Li, X.-X., Li, X. & Yu, S. Gem-difluorobicyclo[2.1.1]hexanes via photochemical [2π + 2σ] cycloaddition initiated by oxidative activation of gem-difluorodienes. Org. Lett. 26, 9961–9966 (2024).
Guo, R. et al. Strain-release [2π + 2σ] cycloadditions for the synthesis of bicyclo[2.1.1]hexanes initiated by energy transfer. J. Am. Chem. Soc. 144, 7988–7994 (2022).
Kleinmans, R. et al. Intermolecular [2π+2σ]-photocycloaddition enabled by triplet energy transfer. Nature 605, 477–482 (2022).
Kleinmans, R. et al. Ortho-selective dearomative [2π + 2σ] photocycloadditions of bicyclic aza-arenes. J. Am. Chem. Soc. 145, 12324–12332 (2023).
Dutta, S. et al. Double strain-release [2π+2σ]-photocycloaddition. J. Am. Chem. Soc. 146, 5232–5241 (2024).
Zhao, J.-X. et al. 1,2-Difunctionalized bicyclo[1.1.1]pentanes: long–sought-after mimetics for ortho/meta-substituted arenes. PANS 118, e2108881118 (2021).
de Robichon, M. et al. Enantioselective, intermolecular [π2 + σ2] photocycloaddition reactions of 2(1h)-quinolones and bicyclo[1.1.0]butanes. J. Am. Chem. Soc. 145, 24466–24470 (2023).
Fu, Q. et al. Enantioselective [2π + 2σ] cycloadditions of bicyclo[1.1.0]butanes with vinylazaarenes through asymmetric photoredox catalysis. J. Am. Chem. Soc. 146, 8372–8380 (2024).
Jeong, J. et al. Divergent enantioselective access to diverse chiral compounds from bicyclo[1.1.0]butanes and α,β-unsaturated ketones under catalyst control. J. Am. Chem. Soc. 146, 27830–27842 (2024).
Li, Y.-J. et al. Catalytic intermolecular asymmetric [2π + 2σ] cycloadditions of bicyclo[1.1.0]butanes: practical synthesis of enantioenriched highly substituted bicyclo[2.1.1]hexanes. J. Am. Chem. Soc. 146, 34427–3444 (2024).
Li, T. et al. Zwitterionic π-Allyl-Pd species enabled [2σ+2π] cycloaddition reactions of vinylbicyclo[1.1.0]butanes (VBCBs) with alkenes, carbonyls, and imines. ACS Catal. 14, 18799–18809 (2024).
Qin, T. & Zi, W. Palladium-catalyzed enantioselective [2σ + 2π] cycloadditions of vinyl-carbonyl-bicyclo[1.1.0]butanes with arylidenemalononitriles. Chin. Chem. Lett. https://doi.org/10.1016/j.cclet.2025.111072 (2025).
Plachinski, E. F. et al. Enantioselective [2π + 2σ] photocycloaddition enabled by brønsted acid catalyzed chromophore activation. J. Am. Chem. Soc. 146, 31400–31404 (2024).
Ren, H.-S. ; et al. Lewis Acid-catalyzed asymmetric [2σ + 2π] cycloaddition reactions of bicyclo[1.1.0]butanes and vinyl azido/diazo compounds. ACS Catal. 15, 4634–4643 (2025).
Shen, W.-J., Zou, X.-X., Li, M., Cheng, Y.-Z. & You, S.-L. Enantioselective dearomative [2π + 2σ] photocycloaddition of naphthalene derivatives with bicyclo[1.1.0]butanes enabled by gd(iii) catalysis. J. Am. Chem. Soc. 147, 11667–11674 (2025).
Wang, Y., Yu, T.-Y., Zhang, H.-B., Luo, Y.-C. & Xu, P.-F. Hydrogen-bond-mediated supramolecular iminium ion catalysis. Angew. Chem. Int. Ed. 51, 12339–12342 (2012).
Gu, Y., Wang, Y., Yu, T.-Y., Liang, Y.-M. & Xu, P.-F. Rationally designed multifunctional supramolecular iminium catalysis: direct vinylogous Michael addition of unmodified linear dienol substrates. Angew. Chem. Int. Ed. 53, 14128–14131 (2014).
Jia, Z.-L., Wang, Y., Zhao, C.-G., Zhang, X.-H. & Xu, P.-F. Highly enantioselective construction of Hajos–Wiechert ketone skeletons via an organocatalytic vinylogous Michael/Stetter relay sequence. Org. Lett. 19, 2130–2133 (2017).
Zhao, C.-G. et al. Highly enantioselective construction of strained spiro[2,3]hexanesthrough a Michael addition/ring expansion/cyclization cascade. Angew. Chem. Int. Ed. 59, 3058–3062 (2020).
Liang, Y., Paulus, F., Daniliuc, C. G. & Glorius, F. Catalytic formal [2π+2σ] cycloaddition of aldehydes with bicyclobutanes: expedient access to polysubstituted 2-oxabicyclo[2.1.1]hexanes. Angew. Chem. Int. Ed. 62, e202305043 (2023).
Acknowledgements
We are grateful to the NSFC (U22A20390 and 22371098), supported by the Xinjiang Uygur Autonomous Region Science and Technology Department’s project (No. Xincaihang [2023-211]), the Science and Technology Major Program of Gansu Province of China (22ZD6FA006, 23ZDFA015, 23JRRA1512) and the “111” program from the MOE of PR China.
Author information
Authors and Affiliations
Contributions
Y.-X.G., T.-F.X. and M.-M.L. carried out the experiments and data analysis work. P.-P.Z. and D.X. performed the DFT calculations. G.-Q.X. and P.-F.X. designed the reaction and directed the project. The paper was written by G.-Q.X. and P.-F.X. All authors contributed to discussions.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Geng, YX., Xiao, TF., Xie, D. et al. Organocatalytic enantioselective [2π + 2σ] cycloaddition reactions of bicyclo[1.1.0]butanes with α,β-unsaturated aldehydes. Nat Commun 16, 9361 (2025). https://doi.org/10.1038/s41467-025-64399-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-64399-7