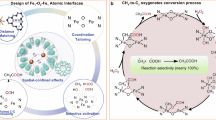
Abstract
Direct oxidation of methane (DOM) into high-value C2+ products using molecular oxygen (O2) is essential for the sustainable production of clean energy and bulk chemicals, but is still challenging due to the difficult C-H activation and uncontrollable C-C coupling process. Herein, we design and construct the Fe-(μ-O)-Zn dual-atom sites by supporting Fe and Zn atoms on ZSM-5 (Fe1-Zn1/ZSM-5), which achieves the DOM by O2 to acetic acid under ambient temperature and pressure. The Fe-(μ-O)-Zn dual-atom sites yield an acetic acid productivity of 3006 μmol•gcat−1•h−1 with 86.8% selectivity (total C2+ products selectivity of 93.0%) for at least 20 hours at 25 oC and atmospheric pressure. The mutual electronic modulation between Fe and Zn shifts the d-band center of Fe 3d in Fe-(μ-O)-Zn dual-atom sites upwards, which promotes the formation and stabilization of highly reactive Fe=O species through O2 photodissociation and thereby enhances the C-H bond activation of CH4. The Fe-(μ-O)-Zn dual-atom reaction sites (spatial distance of 2.7 Å) boost the C-C coupling of key CH3 and HCHO intermediate species, which steadily produce acetic acid and other C2+ oxygenates. This work would broaden the avenue towards the sustainable conversion of methane to value-added C2+ products under ambient temperature and pressure.
Similar content being viewed by others
Introduction
As crude oil resources become depleted, catalytic conversion of the naturally abundant methane (CH4) into high-value products is seen as an important alternative to the oil-based method for the production of energy and chemicals1, contributing to the establishment of a carbon-neutral society. Compared with traditional C1 products, the conversion of methane to acetic acid (CH3COOH) and other C2+ products is more attractive2. The current industrial process for methane to acetic acid is indirect and energy-intensive, involving methane reforming, methanol synthesis, and subsequent methanol carbonylation3,4. Although the direct routes for transforming methane into acetic acid have been explored using both homogeneous and heterogeneous catalysts, the indispensable use of corrosive acid inevitably leads to environmental issues and safety risks5, which pose great challenges for sustainable development.
Consequently, the direct oxidation of methane (DOM) into acetic acid (CH3COOH) and other C2+ oxygenates using molecular oxygen (O2) is of great importance for the sustainable production of clean energy and bulk chemicals6. However, the direct synthesis of C2+ products starting from CH4 often undergoes uncontrollable carbon-carbon coupling mechanisms, making the selectivity for specific products difficult. Furthermore, the direct use of O2 as an oxidant normally requires harsh conditions, e.g., high temperatures and strong oxidizing atmosphere, to activate oxygen because the cleavage of O2 to produce surface active oxygen species for CH4 activation is very difficult7. Moreover, the over-oxidation of initially formed oxygenated products into CO2 and H2O using the formed surface O* species is thermodynamically favored under the aforementioned harsh conditions, leading to the extremely low yield of value-added oxygenated products7,8. Therefore, various efforts have been devoted to the construction of binuclear or trinuclear iron or copper sites confined in zeolites or metal-organic framework (MOF) to mimic the active site structures of methane monooxygenase (MMO) for methane conversion with O2 under moderate temperatures9,10,11. So far, several catalysts have been reported to drive the CH4 conversion with O2 to C1 products at room temperature1,12, but an artificial process for CH4 to C2+ products by using O2 at ambient temperature and pressure remains a great challenge.
Herein, we report that the ZSM-5 supported Fe and Zn dual atoms (Fe1-Zn1/ZSM-5) as a concept of proof catalyst not only enable the stable formation of the reactive Fe=O oxygen species via the photodissociation of O2 under light irradiation at ambient temperature and pressure to efficiently activate C-H bond of methane but also provide dual-atom reaction sites to promote the C-C coupling reaction for DOM to acetic acid and other C2+ oxygenates. As a result, the synthesized Fe1-Zn1/ZSM-5 yields a C2+ productivity of 3420 μmol•gcat−1•h-1 with a C2 + selectivity of 93.0% in all products (acetic acid selectivity of 86.8%) at 25 oC and ambient pressure under light irradiation and sustains the original activity for at least 20 cycles (Supplementary Table S1).
Results
Synthesis and identification of Fe-(μ-O)-Zn reaction sites in Fe1-Zn1/ZSM-5
The Fe1-Zn1/ZSM-5 catalyst was synthesized via a modified co-adsorption method by finely tuning the adsorption parameters of metal precursors in the aqueous solution under the irradiation of ultraviolet light (details shown in Experimental Section)13,14,15,16,17,18. It has been reported that the introduction of UV light can promote the synthesis of heteronuclear dual-atom catalysts19,20,21. The Si/Al molar ratio of ZSM-5 is 12.5:1, and the actual loadings of Fe and Zn are 0.32 wt.% and 0.34 wt.%, determined by inductively coupled plasma-optical emission spectrometry (ICP-OES). Scanning electron microscope (SEM) back-scatter images indicate no nanoparticles or clusters of Fe and Zn species in Fe1-Zn1/ZSM-5, as shown in Supplementary Fig. S1. Furthermore, the TEM images with different magnifications also exhibit that there are no clusters or nanoparticles of Fe and Zn species in Fe1-Zn1/ZSM-5 (Supplementary Fig. S2). In addition, no characteristic peaks of Fe or Zn crystal can be observed from the X-ray diffraction pattern (XRD) of Fe1-Zn1/ZSM-5 (Supplementary Fig. S3), which further reveals that the Fe and Zn species are highly dispersed on ZSM-522,23. Besides, the HAADF-STEM images exhibit no nanoparticles or clusters of Fe and Zn species observed on Fe1-Zn1/ZSM-5 (Supplementary Fig. S4). The mapping results also show that Fe and Zn are evenly dispersed on the ZSM-5 (Supplementary Fig. S5). Additional atomic-scale HAADF-STEM images further verify the diatomic Fe1-Zn1 structure, as the average distance between Fe1-Zn1 dual atoms is around 2.7 Å (Supplementary Fig. S6 and Supplementary Fig. S7), which is also supported by the extended X-ray absorption fine structure (EXAFS) spectroscopy. Those results further confirm the atomically dispersed state of Fe and Zn species on Fe1-Zn1/ZSM-5.
To clarify the local coordination environment of Fe and Zn species in Fe1-Zn1/ZSM-5, extended X-ray absorption fine structure (EXAFS) spectroscopy is performed. For the Fe species, there is one Fe-O scattering at 1.5 Å in the first shell of Fe1-Zn1/ZSM-5 (Fig. 1a), without any Fe-Fe scattering that will be discussed later in the manuscript, which indicates that the Fe species are atomically dispersed instead of nanoparticles or clusters24,25. In addition, the peak at 2.4 Å higher than the Fe-Fe scattering in Fe foil (2.2 Å−1) but lower than the Fe-Fe scattering in FeO (2.5 Å−1) and Fe2O3 (2.5 Å−1), can be assigned to the Fe-Zn scattering in the second shell26, which is further confirmed by the DFT simulation and will be discussed later. This data verifies the presence of Zn species neighboring Fe species. For the Zn species, there is one Zn-O scattering at 1.6 Å in the first shell of Fe1-Zn1/ZSM-5 without any Zn-Zn scattering that will be discussed later, as shown in Fig. 1b, which also indicates that the Zn species are atomically dispersed instead of nanoparticles or clusters25,27. Besides, the peak at 2.4 Å higher than the Zn-Zn scattering in Zn foil (2.2 Å) but lower than the Zn-Zn scattering in ZnO (2.9 Å) can be assigned to the Zn-Fe scattering in the second shell26, which is further confirmed by the DFT simulation and will be discussed later. These results confirm the presence of Fe species neighboring Zn species, which is in good agreement with the Fe EXAFS data.
a k2-weighted μ(k) function of extended X-ray absorption fine structure (EXAFS) spectra of Fe spices. b EXAFS spectra of Zn spices. c Wavelet transform of the Fe K-edge EXAFS oscillations. d Wavelet transform of the Zn K-edge EXAFS oscillations; EXAFS fitting curve of (e) Fe and (f) Zn species in Fe1-Zn1/ZSM-5, the corresponding EXAFS K-space range used for the fitting is 2–12 Å−1; Inset, proposed coordination environment of Fe-(μ-O)-Zn entity.
To reveal the local coordination environment of Fe and Zn species in Fe1-Zn1/ZSM-5 more clearly, the wavelet transform (WT) of the Fe and Zn K-edge EXAFS oscillations is analyzed. As shown in Fig. 1c, the WT contour plots of Fe atoms in Fe1-Zn1/ZSM-5 present one intensity maximum at approximately 4.1 Å−1 that can be assigned to the Fe-O coordination, and no intensity maximum related to Fe-Fe coordination can be observed compared with that of the reference Fe foil, FeO, and Fe2O3 samples28. Meanwhile, due to the scattering effect of Zn on Fe29,30, there is a weak peak on Fe1-Zn1/ZSM-5 at approximately 5.3 Å−1, higher than that of Fe-Fe scattering in Fe foil (5.9 Å−1), FeO (5.4 Å−1), and Fe2O3 (6.1 Å−1), which can be assigned to the Fe-Zn scattering in WT. In addition, the WT contour plots of Zn species in Fe1-Zn1/ZSM-5 present one intensity maximum at approximately 4.2 Å−1 that can be assigned to the Zn-O coordination, and no intensity maximum related to Zn-Zn coordination can be observed compared with that of the reference Zn foil and ZnO samples31,32. Meanwhile, due to the scattering effect of Fe on Zn29,30, there is a weak peak on Fe1-Zn1/ZSM-5 at 5.4 Å−1, lower than that of the Zn-Zn scattering in Zn foil (6.0 Å-1) and ZnO (5.7 Å–1), which correspondingly can be assigned to Zn-Fe scattering in WT33,34. The WT contour plots of Zn species in Fe1-Zn1/ZSM-5 are consistent with the Fe spectrum and confirm that the Fe and Zn species exist as paired atoms on Fe1-Zn1/ZSM-5.
To accurately reveal the local coordination environment of Fe and Zn species in Fe1-Zn1/ZSM-5, various crystal models built by DFT simulation are used to fit the EXAFS data, but only the Z[Fe-(μ-O)-Zn] model fits the original EXAFS data well. Furthermore, the Z[Fe-(μ-O)-Zn] model also fits the original EXAFS data at K-space well (Supplementary Fig. S8), and the fitting details are listed in Supplementary Figs. S9–11 and Supplementary Tables S2, 3. The Z[Fe-(μ-O)-Zn] model fits the original EXAFS data of Fe and Zn species well, as shown in Fig. 1e, f and Supplementary Tables S2, 3, ensuring that the fitting results are reliable and enable an accurate demonstration of the local structure of isolated Fe and Zn atoms in the Fe1-Zn1/ZSM-5. The detailed fitting data show that the Fe atom is coordinated with one bridge oxygen (OBG) and two framework oxygens (OFM), respectively. In detail, for Fe species, the bond length of the Fe atom and the bridge oxygen (Fe-OBG) is 2.0 Å, and the average bond length of the Fe atom and the framework oxygen (Fe-OFM) is 2.1 Å. Furthermore, the fitting data of Fe species on the second shell indicates that the distance between Fe and Zn atoms is 2.7 Å, which is in line with the original EXAFS data (Fig. 1a, b) and the Fe-Zn path in Z[Fe-(μ-O)-Zn] model (Supplementary Fig. S12). For Zn species, Zn atoms are coordinated to one OBG and two framework oxygens (OFM), respectively. The bond length of the Zn atom to the bridge oxygen (Zn-OBG) is 2.1 Å, and the average bond length of the Zn atom to the framework oxygen (Zn-OFM) is 2.2 Å. Furthermore, the fitting data of Zn species on the second shell indicates that the distance between Fe and Zn atoms is 2.7 Å, which is in line with the fitting data for Fe species (2.7 Å) and the Zn-Fe path in Z[Fe-(μ-O)-Zn] model (Supplementary Fig. S12). The results further ensure the reliability of the geometric configurations of the Z[Fe-(μ-O)-Zn] site.
In addition, for Fe1-Zn1/ZSM-5 sample, five extra possible models (Z[Fe-(μ-O)2-Zn], Z[Fe1-Zn2], Z[Fe-Zn], Z[Fe2-Zn3], and Z[Fe2-Zn1]) built for DFT calculation have been compared to fit the structure of active sites, but the significant deviation between the fitting curve and the original data excludes the possibility of Fe and Zn atoms existing as Z[Fe-(μ-O)2-Zn] and Z[Fe-Zn], and also exclude the possibility of the small metal cluster species e.g., Z[Fe1-Zn2], Z[Fe2-Zn3], and Z[Fe2-Zn1]. Those results further verify the accuracy of the fitted Z[Fe-(μ-O)-Zn] structure in Fe1-Zn1/ZSM-5 (Supplementary Figs. S9–11, and Supplementary Tables S2, 3).
Fe-(μ-O)-Zn dual-atom sites mutually modulating the electronic states of Fe and Zn species
The electronic states of Fe and Zn species in Fe1-Zn1/ZSM-5 are further investigated by X-ray absorption near-edge structure (XANES). For Fe species, the absorption edge of Fe1-Zn1/ZSM-5 is higher than that of FeO but lower than that of Fe2O3 (Fig. 2a), indicating that the valence state of the Fe species is between Fe2+ and Fe3+ 24,35. For Zn species, the absorption edge of Fe1-Zn1/ZSM-5 is higher than that of Zn foil but lower than that of ZnO (Fig. 2b), s state of the Zn atoms is between Zn0 and Zn2+ 34. The edge energy of the XANES further validates the above conclusion (Supplementary Fig. S13 and Supplementary Table S4). Furthermore, for the Zn species in Zn1/ZSM-5, the electron paramagnetic resonance (EPR) spectrum has a signal at g = 1.998, which is associated with the Zn+ species36. However, such a characteristic Zn+ peak cannot be observed for the Fe1-Zn1/ZSM-5 (Supplementary Fig. S14), which reveals that the presence of Fe atoms regulates the electronic state of Zn species.
a Fe K-edge X-ray absorption near-edge structure (XANES) spectra. Inset, pre-edge of the K-edge region of XANES. b Zn K-edge XANES spectra. Inset, pre-edge of the K-edge region of XANES. c XPS of Fe 2p for Fe1-Zn1/ZSM-5 and Fe1/ZSM-5. d XPS of Zn 2p for Fe1-Zn1/ZSM-5 and Zn1/ZSM-5. e Bader charge analysis of Fe in FeO bulk, Z[Fe-(μ-O)-Zn]2+, and Fe2O3 bulk. f Bader charge analysis of Zn in Z[Fe-(μ-O)-Zn]2+ and ZnO bulk.
Furthermore, the X-ray photoelectron spectroscopy (XPS) was used to probe the electronic state of Fe and Zn species (Fig. 2c, d). For Fe1/ZSM-5, there is one peak centered at 710.2 eV in the Fe 2p3/2, which can be assigned to the Fe3+37,38. For the Fe1-Zn1/ZSM-5 sample, there is one peak centered at 709.8 eV in Fe 2p3/2, slightly shifting to a lower energy (0.4 eV) compared to that in Fe1/ZSM-5, which is correspondingly assigned to the Feδ+ (2<δ < 3) (Fig. 2c)39. For Zn1/ZSM-5, the sole peak centered at 1021.8 eV in Zn 2p3/2 can be assigned to the Zn+ species40,41, which is in line with the EPR result (Supplementary Fig. S14). However, for Fe1-Zn1/ZSM-5 sample, the sole peak centered at 1022.0 eV in Zn 2p3/2 is slightly shifted to a higher energy compared to that in Zn1/ZSM-5 (1021.8 eV) but slightly lower than the Zn2+ (1022.2 eV), which suggests that the Zn species possesses the unique oxidation state of Znσ+ (1 < σ < 2)41,42 (Fig. 2d). These results further imply that the electronic states of Fe and Zn have been mutually modulated in the Fe-(μ-O)-Zn dual-atom sites, which is in line with the XANES and EPR results.
To further understand the electronic state of Fe and Zn species in the as-synthesized samples, the experiments of diffuse reflectance infrared Fourier transform spectroscopy using NO as a probe molecule (NO-DRIFTs) were performed on the Fe1/ZSM-5, Zn1/ZSM-5, and Fe1-Zn1/ZSM-5 samples (Supplementary Fig. S15)14,43,44. Before each measurement, all the samples are preheated at 423 K for 2 h to remove the possible surface residual adsorbates. Due to the weak chemical interaction between Zn and NO species, no peaks associated with Zn-NO can be observed, which is in line with the open literature45. But for Fe species, there is only one peak centered at 1723 cm−1 in Fe1/ZSM-5, which is associated with the NO linear adsorption on the Fe3+ sites (Fe3+-NO)46,47. For the Fe1-Zn1/ZSM-5, the NO linear adsorption on Fe species red-shifting to 1715 cm−1 (Feδ+-NO) compared with that on the Fe1/ZSM-5, indicates that the oxidation state of Fe is slightly decreased48, which confirms that the Zn atoms regulate the electronic state of Fe species. Additionally, the electron paramagnetic resonance (EPR) of the Fe center (Supplementary Fig. S16) further indicates the electronic states of Fe have been significantly modulated in the Fe-(μ-O)-Zn dual-atom sites, which is in line with the XANES, XPS, and NO-DRIFTs results. The UV-Vis diffuse reflectance spectra of Fe1/ZSM-5, Zn1/ZSM-5, and Fe1-Zn1/ZSM-5 (Supplementary Fig. S17) also verify the strong electronic interaction between neighboring Fe and Zn atoms49,50.
To further explore the unique electronic states of Fe and Zn in Fe1-Zn1/ZSM-5, the Bader charge analysis and charge density difference calculations were performed using DFT. In Fig. 2e, the Bader charge of Fe in Z[Fe-(μ-O)-Zn]2+ (1.17 |e|) is higher than that of Fe in FeO bulk (1.01 |e|) but lower than that in Fe2O3 bulk (1.42 |e|). This confirms that the Fe species in Fe1-Zn1/ZSM-5 exhibit the unique electronic state of Feδ+ (2 < δ < 3), corroborating the XANES, XPS, and NO-DRIFTs data. For Zn species, the Bader charge of Zn in Z[Fe-(μ-O)-Zn]2+ (1.11 |e|) is slightly lower than that in ZnO (1.21 |e|) (Fig. 2f), verifying that the Zn species in Fe1-Zn1/ZSM-5 exhibit the unique electronic state of Znσ+ (1 < σ < 2). Furthermore, the Bader charges of Fe1/ZSM-5 and Zn1/ZSM-5 were calculated as shown in Supplementary Fig. S18. The Bader charge of Fe in Z[Fe-(μ-O)-Zn]2+ (1.17 |e|) is lower than that of Fe in Z[Fe-(OH)2]+ (1.39 |e|), which suggests that the Fe in Z[Fe-(μ-O)-Zn]2+ gains electrons compared to the Fe in Z[Fe-(OH)2]+. The Bader charge of Zn in Z[Fe-(μ-O)-Zn]2+ (1.11 |e|) is higher than that of Zn in Z[Zn] (0.72 |e|), which indicates that the Zn in Z[Fe-(μ-O)-Zn]2+ loses electrons compared with the Zn in Z[Zn]+. Therefore, the electronic states of Fe and Zn are mutually modulated. The charge density difference plot of Fe1-Zn1/ZSM-5 further confirms the above conclusion, as shown in Supplementary Fig. S19. Moreover, the p-DOS analysis results (Supplementary Fig. S20) show that the Fe 3d orbital in Fe1-Zn1/ZSM-5 exhibits a d-band center position (upshift 2.15 eV) closer to the Fermi energy (Ef) compared with Fe1/ZSM-5. These results further reveal that the introduction of Zn atoms regulates the electronic state of Fe species, which is in good agreement with the XANES, EPR, XPS, and NO-DRIFTs results. As the d-band center of Fe 3d shifts closer to the Ef in Fe₁-Zn₁/ZSM-5, it significantly promotes the directional and stable adsorption of O2 on the Fe atom with a unilateral adsorption configuration (Z[O2-Fe-(μ-O)-Zn]2+), which correspondingly enhances the O2 activation to form active Fe=O species under light irradiation. The active Fe=O oxygen species play the determining role in activating the C-H bond of CH4, which will be thoroughly discussed later.
Fe-(μ-O)-Zn dual-atom sites achieving DOM to acetic acid under ambient temperature and pressure
To explore the catalytic performance of Fe1-Zn1/ZSM-5, DOM is performed in a photoreactor under the reaction condition of 50 mL/min CH4 and 25 mL/min O2 for 1 h, and H2O as the reaction medium (details shown in Experimental Section in the Supplementary Information), which is out of the explosive limits of the CH4/O2 mixture system. The detections of gas mixture and liquid products are shown in Supplementary Figs. S21–23. As shown in Fig. 3a, no products can be observed after DOM over Fe1-Zn1/ZSM-5 under dark conditions, indicating that the DOM is driven by light irradiation. When introducing the light irradiation with a high-pressure mercury lamp (~220 to 1100 nm, the effective light intensity is 18.1 mW/cm2 in the wavelength range of 220 and 240 nm) into the reaction over Fe1-Zn1/ZSM-5 for DOM, the yield of C2+ oxygenates is 3420 ± 137 μmol•gcat−1•h−1 with a selectivity of 93.0% in all products and the yield of acetic acid is 3006 ± 109 μmol•gcat−1•h−1 with a selectivity of 86.8% in all liquid products at 25 oC. The average CH4 conversion rate over Fe1-Zn1/ZSM-5 is 0.13 ± 0.01%. The mass balance of carbon between the converted CH4 and formed carbonaceous products is 98.4%, which secures that the measured data is reliable (Supplementary Fig. S24). Furthermore, when changing the methane or oxygen concentration, the Fe1-Zn1/ZSM-5 still shows excellent yield and selectivity of C2 + oxygenates, as shown in Supplementary Fig. S25 and Supplementary Fig. S26. Considering the heating effect of light irradiation, the ice and water mixture was used to sustain the set reaction temperature. Those results further indicate that the light irradiation plays a crucial role in the DOM in our reaction system. For DOM over pure H-ZSM-5 under the same reaction condition with light irradiation, the major product is CO2 (57.7%) and the C2 + oxygenates productivity is only 44 ± 6 μmol•gcat−1•h−1, around 78 times lower than that of Fe1-Zn1/ZSM-5, which indicates that the Fe-(μ-O)-Zn dual-atom sites are the main active sites (Fig. 3b). For DOM over Fe1/ZSM-5, the yield of C1 oxygenates is 816 ± 82 μmol•gcat−1•h−1 (selectivity of 56.6%) and the yield of C2 + oxygenates is 524 ± 33 μmol•gcat−1•h−1 (selectivity of 32.4%), which indicates that the Zn sites play the crucial role in determining the coupling conversion of DOM. For DOM over Zn1/ZSM-5, the major products are C2 + oxygenates (C2 + selectivity of 81.1%), and the yield is 1182 ± 83 μmol•gcat−1•h−1, around 3 times lower than that of Fe1-Zn1/ZSM-5, which indicates that the Fe sites play the crucial role in boosting the catalytic activity for DOM. Meanwhile, the detailed control experiments by measuring the catalytic activity with similar loading content of metal sites over Fe1/ZSM-5, Zn1/ZSM-5, and Fe1-Zn1/ZSM-5 (the actual metal loadings measured by ICP-OES as shown in Supplementary Table S5) were conducted to exclude the influence of the density of metal sites (Supplementary Fig. S27). In addition, the yield of C2+ oxygenates for the physical mixtures of Fe1/ZSM-5 and Zn1/ZSM-5 with similar metal loading as that of Fe1-Zn1/ZSM-5 is only 1265 ± 94 μmol•gcat−1•h−1 with a selectivity of 59.8% in all products, around 2.7 times lower than that of Fe1-Zn1/ZSM-5, which clearly confirms the synergistic effects between Fe-(μ-O)-Zn dual-atom sites in Fe1-Zn1/ZSM-5 (Supplementary Fig. S28). By tuning the molar ratio of Fe/Zn, the yield of Fe1-Zn1/ZSM-5 with different Fe/Zn ratios for C2+ oxygenates is much higher than that of the sole Fe/ZSM-5 or sole Zn/ZSM-5, as shown in Supplementary Fig. S29. Moreover, by altering the loading content of Fe or Zn, the catalytic performance of Fe1-Zn1/ZSM-5 with similar Fe/Zn content (0.32 wt.% for Fe and 0.34 wt.% for Zn) for C2+ oxygenates is much better than that of the more or less Fe and Zn content, as shown in Supplementary Fig. S30. Moreover, the NO-DRIFTs results also reveal that altering the Fe/Zn ratio can notably regulate the oxidation state of Fe, and the excessive Fe atoms mainly exist in an isolated status similar to that of Fe1-ZSM-5, which essentially exhibits higher oxidation states and benefits the formation of C1 oxygenates (Supplementary Fig. S31). Those results clearly indicate that the Fe-(μ-O)-Zn dual-atom sites in Fe1-Zn1/ZSM-5 not only promote the selectivity of C2+ oxygenates but also boost the catalytic activity for DOM.
a Comparative experiments for DOM on Fe1-Zn1/ZSM-5 under light or dark conditions. b Yields and selectivity of C2+ on H-ZSM-5, Fe1/ZSM-5, Zn1/ZSM-5 and Fe1-Zn1/ZSM-5. c Yields and selectivity of C2+ of Fe1-Zn1/ZSM-5 on different flow rate ratios of CH4 to O2. d Reaction cycles of DOM over Fe1-Zn1/ZSM-5. e Yields and selectivity of C2+ of Fe1-Zn1/ZSM-5 on different temperatures. Reaction Condition (a): 20 mg catalysts dispersed in 100 mL deionized water, 50 mL/min CH4 and 25 mL/min O2 for 1 h at 25 oC; Reaction Condition (b) and (d): 20 mg catalysts dispersed in 100 mL deionized water, Hg lamp, 50 mL/min CH4 and 25 mL/min O2 for 1 h at 25 oC; Reaction Condition (c): 20 mg catalysts dispersed in 100 mL deionized water, Hg lamp for 1 h at 25 oC; Reaction Condition (e): 20 mg catalysts dispersed in 100 mL deionized water, Hg lamp, 50 mL/min CH4 and 25 mL/min O2 for 1 h. In (a–c) and (e), all data points were tested for three times to obtain the error bar.
To further identify the catalytic role of Fe-(μ-O)-Zn dual-atom sites in DOM, several control experiments have been performed. The catalytic yield of DOM over Fe1-Zn1/ZSM-5 is first investigated under the reaction atmosphere of different CH4 and O2 flow rates (Fig. 3c). An optimal flow ratio of CH4 to O2 is reached at 2:1. Over high or low ratio will decrease the productivity and selectivity of C2 + oxygenates, which indicates that the synergistic activation of CH4 and O2 plays a crucial role in the formation of C2+ oxygenates. In the absence of O2, no products can be observed, which suggests that O2 is the oxidant rather than the H2O (reaction medium) for DOM. In addition, the isotopic experiments (18O2) exhibit that the significant oxygenated products containing 18O, e.g., CH3C18O18OH, can be detected after the DOM when using 18O2 as the oxidant (Supplementary Fig. S32). Moreover, the significant oxygenated products containing 16O, e.g., CH3C16O16OH, can be detected after the DOM when using H218O as the solvent (Supplementary Fig. S32). These isotopic experiment results indicate that it is the O2 rather than the H2O as the oxygen source to drive DOM in our catalyst system. In particular, DFT calculations also indicate that water did not serve as a cocatalyst or the oxidizing agents to assist methane activation (Supplementary Fig. S33). In the absence of CH4, no products can be observed, which indicates that the carbon source is from the methane (Supplementary Fig. S34). Furthermore, the reaction order with respect to CH4 (0.47) is higher than that of O2 (0.16) as shown in Supplementary Fig. S35, indicating that the CH4 activation for DOM on Fe1-Zn1/ZSM-5 is critical to the reaction kinetics51, which is in line with the DFT simulation and will be discussed later.
To explore the stability of DOM over Fe1-Zn1/ZSM-5, we first probed the recycling stability. The obtained results reveal that there is no significant loss in the catalytic yield, methane conversion rate, and selectivity of C2+ oxygenates for at least 20 h (one hour for each reaction cycle, Fig. 3d and Supplementary Fig. S36). Then the stability was further analyzed by prolonging the reaction time. The total productivity of C2+ oxygenates is increased to 9744 ± 482 μmol•gcat−1 after 4 h and 23,211 μmol•gcat−1 after 24 h, as shown in Supplementary Fig. S37. Those results indicate that the Fe1-Zn1/ZSM-5 is stable during the DOM and enables to inhibition of the overoxidation of the formed C2+ oxygenates in the reaction atmosphere. The conditions used to regenerate the spent catalyst in each recycling measurement are listed in the Experimental Section. The SEM back-scatter images, XRD, and ICP-OES data of the used Fe1-Zn1/ZSM-5 sample after 20 cycles verify the leaching resistance ability of the Fe1-Zn1/ZSM-5 catalyst (Supplementary Figs. S38, 39 and SupplementaryTable S6).
The apparent activation energy (Ea) of the synthesized Fe1-Zn1/ZSM-5 catalysts was investigated to better understand the catalytic behavior of Fe1-Zn1/ZSM-5 (Supplementary Fig. S40). A similar method has been reported in previous literature52. The Fe1-Zn1/ZSM-5 exhibits the lowest Ea (25.7 kJ/mol) compared with that of Fe1/ZSM-5 (34.1 kJ/mol) and Zn1/ZSM-5 (30.4 kJ/mol), which indicates that the catalytic process of DOM is intrinsically boosted by Fe-(μ-O)-Zn dual-atom sites in Fe1-Zn1/ZSM-5 compared with the sole Fe or Zn single atoms. Moreover, the selectivity of C2+ oxygenates at 20 oC is 90.5%, which is close to that at 25 oC, although the productivity of C2+ oxygenates is only 2939 ± 153 μmol•gcat−1•h−1 (Fig. 3e). When further decreasing the reaction temperatures, the productivity of C2+ oxygenates is 2486 ± 77 μmol•gcat−1•h−1 with a selectivity of 89.1% at 15 oC and 1771 ± 130 μmol•gcat−1•h−1 with a selectivity of 84.8% at 10 oC, which suggests that the Fe-(μ-O)-Zn dual-atom sites possess excellent catalytic capability for DOM even below room temperature.
CH4 activation promoted by active Fe = O oxygen species formed via O2 photodissociation on Fe-(μ-O)-Zn dual-atom sites
To gain insights into the nature of the efficient promotion on CH4 activation over Fe1-Zn1/ZSM-5 catalyst, density functional theory (DFT) calculations were further performed (Fig. 4a). The high-spin state was utilized for the Fe-Zn dual-atom site to perform the DFT calculation (Supplementary Table S7). We constructed the catalyst model according to the characterization results discussed in Section 2.1. On the basis of the HAADF-STEM, EXAFS, and WT data of Fe1-Zn1/ZSM-5, the active site is a hetero-binuclear structure. Based on the above characterization data, several model active sites have been searched (Supplementary Figs. S9 and S10) but only the Z[Fe-(μ-O)-Zn]2+ is the most reliable reaction site, where the distance of neighboring dual Fe and Zn atoms is 2.7 Å and the three-coordinated Fe and Zn atom located at the γ-8MR of ZSM-5 zeolite is connected by one bridged oxygen. Each dual-atom pair with three-fold coordination is connected by one bridged oxygen with neighboring Fe and Zn atoms. The detailed bond length and coordination structure of Fe/Zn species in the model active site fit the original EXAFS data well, which confirms the reliability of the model used in DFT calculations (Fig. 1e, f, Supplementary Table S2, and S3). Moreover, the previous calculation suggested that the framework Al3+ replacing the original Si4+ at T12 and T7 sites in the sinusoidal channel γ-8MR of ZSM-5 zeolite is the most energetically stable one to anchor Fe-(μ-O)-Zn atoms13, which was further proved by pyridine-IR data (Supplementary Fig. S41 and Supplementary Table S8). In short, these results verify the reliability of the simulated Fe-(μ-O)-Zn dual-atom sites. The specific geometry structure of Z[Fe-(μ-O)-Zn]2+ is listed in Supplementary Fig. S42.
The unilateral state structure of Fe=O species at Z[O=Fe-(μ-O)2-Zn]2+ site (a); The formation of Fe=O species captured by in situ Raman spectra under different conditions (b). The formation of Fe=O species captured by in situ FT-IR spectra under different conditions (c). The comparison on the CH4 first C-H bond activation energy over Fe1-Zn1/ZSM-5 under light and dark conditions (d). The CH4 activation and O-O coupling process at Z[O=Fe-(μ-O)2-Zn]2+ site under irradiation (e). The geometry structures of transition states and intermediate species for CH4 activation and O-O coupling process at Z[O=Fe-(μ-O)2-Zn]2+ site (f). The transition state structure and the energy barrier to activate the first C-H bond of CH4 for DOM over Fe1-Zn1/ZSM-5 (g). The process of Fe=O species activating CH4 molecules captured by in situ DRIFTs (h and i). The Gibbs energy profile was calculated under the reaction conditions of 0.66 bar CH4, 0.33 bar O2 at 298 K. The H, C, O, Si, Al, Fe, and Zn atoms of the reaction sites are displayed in white, gray, red, yellow, pink, cerulean, and coral in the ball and stick style, respectively.
Our DFT simulation results indicate that the bridging oxygen (μ-O) between Fe and Zn at the initial Z[Fe-(μ-O)-Zn]2+ site is a divalent oxygen anion (Supplementary Fig. S9), which is difficult to directly abstract a hydrogen atom and activate the C-H bond of methane (barrier of 1.06 eV). Therefore, the cleavage of O2 molecules by UV irradiation to generate the O* species adsorbed at the Z[Fe-(μ-O)-Zn]2+ site is the first step to produce reactive oxygen species for DOM. Our comparative experiments with different photo energies confirm that the O2 molecule was dissociated via a photochemistry rather than a photo-catalysis process in our reaction system (Supplementary Fig. S43). At first, O2 is readily chemisorbed at the Fe site of Z[Fe-(μ-O)-Zn]2+, which is exothermic by 1.50 eV. With the assistance of the photo, the O-O bond could be broken to simultaneously form Fe=O and another bridging O, thereby transforming the sites to Z[O=Fe-(μ-O)2-Zn]2+ (Supplementary Fig. S44). Specifically, in the range of the Herzberg continuum (from 218 to 239 nm) that was provided by the high-pressure mercury lamp in our reaction system, the O2 enables to assimilation of light energy and excites the ground state oxygen O2\(({\scriptstyle{3}\atop}{\sum }_{g}^{-})\) to the excited states \(A({\scriptstyle{3}\atop}{\sum }_{u}^{+})\), \(c({\scriptstyle{1}\atop}{\sum }_{u}^{-})\), and A’(3Δu)53 Subsequently, the above excited states facilitate the cleavage of the O=O bond to generate O atoms, i.e., O(3P2) + O(3P2), mainly through spin-orbit and orbit-rotation interactions54,55. Notably, due to the higher oxophilicity of Fe2+ compared with Zn2+ species56, the oxygen atoms prefer to directionally adsorb over the Fe2+ site rather than the Zn2+ site at Z[Fe-(μ-O)-Zn]2+. Thus, one of the O(3P2) atoms coordinates with Fe and Zn atoms to form the bridging oxygen (μ-O), while the other O(3P2) atom is stably anchored on the Fe site to generate the active oxygen species (Fe = O) at Z[O = Fe-(μ-O)2-Zn]2+ site (Fig. 4a). To sum up, the catalytic activation of methane using O2 as oxidant at Z[Fe-(μ-O)-Zn]2+ begins with the formation of Fe=O active species by photo-dissociation of O2, then followed by the Fe=O species activating methane via a radical-like mechanism57,58,59 (barrier of only 0.40 eV), which will be discussed later. In addition, the initial structure of the transition state with an energy barrier of 0.40 eV corresponds to a methane molecule located at the intersection between the straight channel and the sinusoidal channel of ZSM-5 (Supplementary Fig. S45). And if the physical adsorption of methane is considered, the activation energy barrier would be 0.44 eV.
To experimentally identify the reactive oxygen species (Fe=O) formed by O2, the in situ Raman and DRIFTs were performed (Fig. 4b, c). When the O2 and light irradiation were both introduced, the peak at 775 cm-1 associated with the Fe=O species was observed in the Raman spectra60, which is in line with DFT calculation (IM1). Furthermore, when 16O2 was replaced by 18O2, a redshift of the characteristic peak of Fe=O species could be observed (Supplementary Fig. S46), further indicating the generation of Fe=O species61. Moreover, when the O2 and light irradiation were both introduced, the same Fe=O species was observed at 843 cm−1 in the IR spectrum48,62,63, which is consistent with the Raman data and DFT calculation. However, when the reaction conditions were changed into Ar with light off, Ar with light on, or O2 with light off, none of the corresponding signals were detected in the Raman and IR spectra (Fig. 4b, c), which indicates that the O2 and light irradiation are necessary to generate active Fe=O species. In addition, our control experiments suggest that no products can be observed after the DOM in the absence of light irradiation or O2 (Fig. 3a). These results provide solid evidence that the Fe=O species is the active oxygen species for CH4 activation, which is consistent with the DFT simulation.
Importantly, the free energy barrier of activating the first C-H bond of CH4 by the produced Z[O=Fe-(μ-O)2-Zn]2+ sites with the assistance of light irradiation is only 0.40 eV, which is around 5 times lower than that on the Z[O2-Fe-(μ-O)-Zn]2+ without light irradiation (2.23 eV, Fig. 4d). These results suggest that the Fe=O species, which are generated by Fe-(μ-O)-Zn dual-atom active sites with the assistance of O2 photo-dissociation, can significantly boost the methane activation.
Moreover, the stability of Fe=O species is also investigated to probe the possibility of the Fe=O species transforming back into O2 via the O-O coupling. By comparing the O-O coupling the free energy barrier of two O* and the free energy barrier of the first C-H bond cleavage in CH4 (Fig. 4e, f), we can better understand the importance of Z[O=Fe-(μ-O)2-Zn]2+ for CH4 activation. For Z[O=Fe-(μ-O)2-Zn]2+ (IM1), the O-O coupling free energy barrier (1.03 eV) of two O* is much higher than the free energy barrier of the first C-H bond cleavage in CH4 (0.40 eV, TS1, Fig. 4g), so the Fe=O in Z[O=Fe-(μ-O)2-Zn]2+ (IM1) would preferentially catalyze the CH4 activation rather than the recombination of two O* back into O2. Conversely, for Z[(O)2-Fe-(OH)2]+, the O-O coupling free energy barrier (0.19 eV) of two O* is lower than the first C-H bond cleavage in CH4 (0.28 eV), indicating that two O* at Z[(O)2-Fe-(OH)2]+ would be readily coupled to transform into Z[(O2)-Fe-(OH)2]+ for mononuclear Fe1/ZSM-5 (Supplementary Fig. S47). However, once Z[(O2)-Fe-(OH)2]+ is formed, the free energy barrier of methane C-H bond activation surges to 1.36 eV, indicating that mononuclear Fe1/ZSM-5 is deactivated. These results indicate that the mononuclear Fe1/ZSM-5 is unable to effectively activate CH4 compared with that of Fe1-Zn1/ZSM-5 binuclear sites. For the Z[O=Fe-(μ-O)2-Fe]2+, the O-O coupling free energy barrier (2.74 eV) of two O* is also higher than the first C-H bond cleavage in CH4 (0.96 eV), but its free energy barrier to cleave the first C-H bond in CH4 is much higher than that of Z[O=Fe-(μ-O)2-Zn]2+ (0.40 eV). Therefore, the catalytic capability of Z[O=Fe-(μ-O)2-Fe]2+ to activate methane is lower than that of Z[O=Fe-(μ-O)2-Zn]2+ (IM1). Consequently, comparing the C-H bond activation and O-O coupling over hetero-binuclear Fe1-Zn1/ZSM-5, homo-binuclear Fe1-Fe1/ZSM-5, and mononuclear Fe1/ZSM-5 (Supplementary Fig. S48), we could find that the synergistic effects between Fe-(μ-O)-Zn dual-atom sites not only stabilize the reactive oxygen species, but also exhibit better capability for the activation of C-H bond in CH4. Furthermore, we have computationally compared the first C-H bond activation of the CH4 free energy barrier of hetero binuclear Z[O = Fe(μ-O)2-Zn]2+, Z[O = Fe(μ-O)2-Cu]2+, and Z[O = Fe(μ-O)2-Ni]2+ (Supplementary Fig. S49a). The order of the free energy barriers for the C-H bond activation follows as Z[O=Fe-(μ-O)2-Zn]2+ (0.40 eV) < Z[O=Fe-(μ-O)2-Cu]2+ (0.51 eV) < Z[O=Fe-(μ-O)2-Ni]2+ (0.54 eV), which indicates the Fe-(μ-O)-Zn dual-atom sites enhance performance, facilitate the C-H bond activation (Supplementary Fig. S49b). Meanwhile, for DOM over Fe1-Ni1/ZSM-5 and Fe1-Cu1/ZSM-5, the yield of C2+ oxygenates is 1993 ± 86 μmol•gcat−1•h–1 (C2+ oxygenates selectivity of 81.9%) on Fe1-Ni1/ZSM-5 and 2488 ± 150 μmol•gcat−1•h−1 (C2 + oxygenates selectivity of 92.3%) on Fe1-Cu1/ZSM-5, which are lower than the yield and selectivity of C2 + oxygenates on Fe1-Zn1/ZSM-5 (3420 ± 137 μmol•gcat−1•h−1 with a selectivity of 93.0%). Those results verify that the synergistic effects of Fe-(μ-O)-Zn dual-atom sites enable to enhance the performance for DOM to C2+ oxygenates (Supplementary Fig. S49c).
The process of Fe=O species activating CH4 molecules was further validated by the in situ DRIFTs. When both CH4 and O2 were introduced under light irradiation, the peak associated with the Fe=O stretch vibration is gradually redshifted as the reaction proceeds (Fig. 4h) and the C-H stretch vibration of CH4 on Fe1-Zn1/ZSM-5 is also gradually redshifted (Fig. 4i), which confirms the methane activation is triggered by the Fe=O species for DOM on Fe1-Zn1/ZSM-5. Alternatively, only when sole O2 or sole CH4 was introduced under light irradiation, there is no shifting on Fe=O species and C-H stretch vibration of CH4 (Supplementary Figs. S50, S51). Furthermore, the in situ O2-DRIFTs under light irradiation and switch to CH4-DRIFTs under dark indicate that Fe=O active species are formed by light-driven O=O bond dissociation, and the C-H activation in CH4 is triggered by the formed Fe=O species (Supplementary Fig. S52). Those results confirm that the methane activation is triggered by the Fe=O species for DOM on Fe1-Zn1/ZSM-5, which is in line with the DFT calculation.
In addition, H2O2 is also observed in the liquid determined by the potassium titanium oxalate method64 after DOM, and the concentration is 231 ± 9.2 μM, as shown in Supplementary Fig. S53. Some previous works reported that H2O2 or its derived •OH radicals are the reactive oxygen species for the DOM65,66. To identify whether H2O2 is the reactive oxygen species for the DOM in our reaction system, the DOM is conducted by externally adding H2O2 rather than O2. Only a trace amount of methanol (14 μmol•gcat−1•h−1) is observed in the liquid after DOM when the externally added H2O2 is used as an oxidant with a similar concentration (~ 231 μM, Supplementary Fig. S54). These results indicate that the H2O2 species that existed in the solution is not the reactive oxygen species for DOM on our catalyst system. In addition, to identify whether the hydroxyl radicals (•OH) formed by the dissociation of H2O2 are the active oxygen species for the DOM, Na2S was added as a •OH scavenger23 to conduct the comparison experiment for DOM. The comparison experiments exhibit a similar yield of the C2+ products (3,413 ± 179 μmol•gcat−1•h−1, Supplementary Fig. S55), which indicates that the •OH produced by H2O2 is not the reactive oxygen species for DOM as well. These results further confirm that only the Fe=O species in Z[O=Fe-(μ-O)2-Zn]2+ (IM1) is the reactive oxygen species for DOM.
Fe-(μ-O)-Zn dual-atom sites boosting C-C coupling for DOM toward acetic acid via synergistic-tandem mechanism
To further clarify the reaction pathway of DOM over Fe1-Zn1/ZSM-5 catalyst, the reaction mechanism for the production of CH3COOH, which is the main C2+ product on Z[O = Fe-(μ-O)2-Zn]2+ site, was computationally investigated (Fig. 5a). The models for key intermediate species confined in the channels of the Fe1-Zn1/ZSM-5 by DFT calculation are also shown in Fig. 5a, b. After the first C-H bond in CH4 is activated by Fe=O species through the radical-like mechanism (IM1 and IM2 shown in Fig. 4f), the generated methyl radicals can be readily captured by the bridge oxygen at Z[(HO)-Fe-(μ-O)2-Zn]2+ (IM2 in Fig. 4f), which is energetically lowered by 2.33 eV. Then, the second C-H bond of CH4 can be activated by the hydroxyl to generate Z[(H2O)(HCHO)-Fe-(μ-O)-Zn]2+ (IM4 in Fig. 5b), which needs to overcome a free energy barrier of 0.67 eV (TS2). The water molecules at Z[(H2O)(HCHO)-Fe-(μ-O)-Zn]2+ (IM4) are desorbed from Fe atoms to generate Z[HCHO-Fe-(μ-O)-Zn]2+ (IM5), and the desorption energy is 0.84 eV. In addition, there is a possible side reaction to generate HCOOH, which is carried out by dehydrogenation of formyl aldehyde (HCHO*) of IM5 to μ-O (Supplementary Fig. S56). But for Z[HCHO-Fe-(μ-O)-Zn]2+ (IM5), the branch to generate formic acid is energetically less favorable than the acetic acid branch, starting with the migration of HCHO* from the Fe site to the Zn site that only necessitates the energy input of 0.36 eV. It indicates that the acetic acid would be easier to produce than formic acid, which is consistent with our experimental results. Then, the chemisorbed formyl aldehyde (HCHO*) at the Fe site can easily migrate to the Zn site, with a free energy barrier of 0.36 eV (Supplementary Fig. S57), which is exothermic by 1.13 eV and correspondingly generates Z[Fe-(μ-O)-Zn-(HCHO)]2+ (IM6) site. Then, a second Fe=O species can be produced again by photo-assisted O2 dissociation to generate Z[O=Fe-(μ-O)2-Zn-(HCHO)]2+ (IM7) as discussed in Section 2.4. In addition, compared with the Fe-Fe diatomic site, the desorption free energy (0.83 eV) of the formyl aldehyde at the Z[O=Fe-(μ-O)2-Zn-(HCHO)]2+ (IM7) site is higher than that at the Z[O=Fe-(μ-O)2-Fe-(HCHO)]2+ site (0.43 eV) (Supplementary Fig. S58). This indicates that the Fe-(μ-O)-Zn dual-atom site can stabilize the intermediates generated by methane, thereby promoting the C-C coupling of the intermediates to generate C2 + products.
Reaction pathways for DOM to CH3COOH over Fe1-Zn1/ZSM-5 (a). The geometric structures of the key transition states for CH3COOH formation and oxidative dehydrogenation (b). The experimentally captured intermediates by FT-IR spectra of CH4 + O2 under light irradiation over Fe1-Zn1/ZSM-5 (c). Schematic illustration of the Fe-(μ-O)-Zn active sites catalyzing DOM to CH3COOH (d). The Gibbs energy profile was calculated under the reaction conditions of 0.66 bar CH4, 0.33 bar O2 at 298 K. The H, C, O, Si, Al, Fe, and Zn atoms of the reaction sites are displayed in white, gray, red, yellow, pink, cerulean, and coral in the ball and stick style, respectively.
The second CH4 could still be readily activated by Fe=O species at the Z[O=Fe-(μ-O)2-Zn-(HCHO)]2+ (IM7) site through the radical-like mechanism to produce a methyl radical and Z[OH-Fe-(μ-O)2-Zn-(HCHO)]2+ (IM8) site, which requires a free energy barrier of 0.46 eV (TS3). The efficient methane activation by alpha oxygen of Fe=O through the radical-like mechanism enables the barrierless C-C bond coupling between the formed methyl radical and HCHO* to generate Z[(OH)(O)-Fe(μ-O)-Zn-(CH3CH2O)]2+ (IM9), which is exothermic by 0.60 eV. Thus, the alpha hydrogen of open-shell CH3CH2O* at Z[(OH)(O) -Fe(μ-O)-Zn-(CH3CH2O)]2+ (IM9) site could be readily abstracted by the oxygen adsorbed at Fe site, and then the Z[(OH)-Fe(μ-O)(μ-OH)-Zn-(CH3CHO)]2+ (IM10) site is generated, which only needs to overcome a free energy barrier of 0.17 eV (TS4). The process of IM10 → IM11 (Z[(OH)-Fe(μ-O)(μ-OH)-Zn-(CH3CHO)]2+) involves two steps: C-H bond cleavage of CH3CHO* and Zn-O bond cleavage for the switch of adsorption moiety of CH3CO* from carbonyl O to C (Supplementary Fig. S59). For IM10, CH3CHO* could be dehydrogenated by the μ-O to generate CH3CO* that is chemisorbed atop the Zn site through the carbonyl oxygen of CH3CO*, with a free energy barrier of 0.61 eV. Then, the adsorption moiety of CH3CO* would be readily transformed from carbonyl oxygen to carbonyl carbon, together with the Zn-O bond cleavage and C-Zn bond formation, which only needs to overcome a free energy barrier of 0.17 eV. Then the hydrogen atom transfer from the bridged hydroxyl to the non-bridge hydroxyl will generate Z[(H2O)-Fe-(μ-OH)(μ-O)-Zn-(CH3CO)]2+ (IM12), which only requires a free energy barrier of 0.69 eV (TS6). Subsequently, the water molecule can desorb from the Fe atom to generate Z[(OH)-Fe-(μ-O)-Zn-(CH3CO)]2+ (IM13), and the desorption free energy is only 0.53 eV. IM13 → IM14 undergoes the transformation of the adsorption configuration of CH3CHO*, followed by the C-O coupling for acetic acid formation (Supplementary Fig. S60). The transformation of the adsorption configuration of CH3CO* from carbonyl carbon to carbonyl oxygen requires a free energy barrier of 0.51 eV. Then, CH3CO* can couple with OH* to generate CH3COOH* by overcoming a free energy barrier of 0.85 eV, which is significantly exothermic by 2.56 eV. Then, the CH3COOH can desorb from the Z[Fe-(μ-O)-Zn-(CH3COOH)]2+ (IM14) site to regenerate the active site of Z[Fe-(μ-O)-Zn]2+(IM15), requiring an energy input of 1.02 eV.
To experimentally verify the proposed reaction pathway, the in situ DRIFTs of CH4 + O2 on Fe1-Zn1/ZSM-5 under light irradiation were conducted. As shown in Fig. 5c, the peaks at 1054 cm−1 and 1107 cm−1 are associated with the stretching vibration of C-O in the adsorbed CH3O* (IM3) and HCHO* (IM6) species, respectively67,68, which are in line with our DFT simulation. Meanwhile, the externally added formaldehyde was conducted to further verify the catalytic role of the HCHO* intermediate. The yield of acetic acid significantly increased with the enhancement of externally added formaldehyde (Supplementary Fig. S61 and S62). However, when CH4 was removed and only HCHO and O2 were added, no CH3COOH was detected (Supplementary Fig. S61). These results suggest that HCHO* and intermediates formed by CH4 play a pivotal role in the formation of acetic acid. Under dark conditions, no CH3COOH was detected, which indicates that the formation of acetic acid is driven by light irradiation (Supplementary Fig. S63). The peak at 1342 cm−1 is associated with the bending vibrations of C-H in the adsorbed CH3CH2O* (IM9) species69, which is the key intermediate to C2+ after the C-C coupling reaction on Fe-(μ-O)-Zn dual-atom sites. The peaks at 1652 cm-1 and 1457 cm-1 are ascribed to the stretching vibrations of C=O of the adsorbed CH3CO* (IM11) and CH3COOH* (IM14) species, respectively70,71, which are the intermediates generated by the further oxidation of CH3CH2O* (IM9) after C-C coupling on Fe-(μ-O)-Zn dual-atom sites. Meanwhile, no signals can be observed from the in situ DRIFTs when there is no oxygen or light irradiation, which further indicates that the photo-assisted O2 activation plays a crucial role in the DOM (Supplementary Fig. S64 and S65). The significant redshifts of C-O/C-H vibration frequency acquired by isotope-labeling 13CH4 + O2 DRIFTS further verify the accuracy of the assignment of the CH3O*, HCHO*, and CH3CH2O* intermediates experimentally captured by FT-IR spectra of 12CH4 + O2 under light irradiation over Fe1-Zn1/ZSM-5 (Supplementary Fig. S66). These intermediates observed from in situ DRIFTs experimentally verify the reliability of the DFT calculation for the reaction pathway of DOM (Fig. 5a). The tandem catalysis process triggered by Fe=O species under light irradiation was further verified by the stepwise reaction process, as shown in Supplementary Fig. S67.
Hence, according to the above DFT calculation, in situ characterization results and formaldehyde-as-reactant control experiments, the most possible reaction pathway of DOM to acetic acid on Fe1-Zn1/ZSM-5 is proposed as steps 1-9 in Fig. 5d, and the comprehensive reaction cycles including 14 steps are listed in Supplementary Fig. S68.
Those results indicate the Fe-(μ-O)-Zn dual-atom sites have significantly promoted DOM toward acetic acid via the synergistic-tandem mechanism.
Discussion
In summary, we designed and constructed the Fe-(μ-O)-Zn dual-atom sites by supporting Fe and Zn atoms on ZSM-5 (Fe1-Zn1/ZSM-5), which achieved the DOM by O2 to acetic acid under ambient temperature and pressure. The Fe1-Zn1/ZSM-5 yields a C2+ productivity of 3420 ± 137 μmol•gcat−1•h−1 and a C2+ selectivity of 93.0% in all products (the selectivity of acetic acid is 86.8%) and sustains the original activity for at least 20 h (one hour for one reaction cycle) at 25 oC and ambient pressure under light irradiation. The comprehensive characterization results reveal that with the assistance of the ultraviolet light, the Fe-(μ-O)-Zn active sites boost the stable formation of highly reactive Fe=O species and provide dual reaction sites, which promote the activation of the C-H bond of CH4 and the C-C coupling reaction. Although further refinement in optimizing the reaction conditions and other factors may improve the catalytic performance, this work should broaden the avenue towards the sustainable conversion of methane to value-added C2+ products under ambient temperature and pressure.
Methods
Synthesis of catalysts
The modified adsorption methods were used to prepare the H-ZSM-5 supported Fe and Zn dual atoms (Fe1-Zn1/ZSM-5) via finely tuning the adsorption parameters in the aqueous solution13,14,15,16,17,18. Typically, H-ZSM-5 (The Si/Al molar ratio of ZSM-5 is 12.5:1, the Brunauer-Emmett-Teller (BET) surface area of ca. 310 m2/g; Tianjin Yuanli Chemical Co.) was dispersed in deionized water (18.2 MΩ cm−1), and the pH of the H-ZSM-5 solution was finely tuned from 4.5 to 3.0 by using HCl aqueous. The Fe & Zn precursor solutions (HFeCl4 is prepared by FeCl3 and HCl72. Typically, dissolving 135.10 g of FeCl₃·6H₂O in a small amount of water, and prepare 542 mL of 12 mol/L HCl solution. Then, transferring both solutions to a 1 L volumetric flask and dilute to the mark with water to synthetize HFeCl4. FeCl3 and ZnCl2 is purchased from Sinopharm Chemical Reagent Co., China), were simultaneously pumped into the H-ZSM-5 suspension with a speed of ~ 0.2 ml/min under stirring, and the solution underwent irradiation treatment with ultraviolet light (5 W LED, wavelength 254 nm) for 4 h at 5 ± 2 oC. After aging for 4 h at 5 ± 2 oC, the sample was centrifuged, washed, and then dried at 40 oC in a vacuum and calcined at 350 oC for 4 h in Ar flow. It has been reported that the introduction of UV light can promote the synthesis of heteronuclear dual-atom catalysts since the second metal atoms (M2) are prone to navigation and positioning to the first metal atoms (M1) after reacting with photogenerated electrons near the M1 site19,20,21. Similarly, under UV irradiation, the Fe3+ from HFeCl4 and Zn2+ from ZnCl2 form Fe1-Zn1 dual-atom sites. The actual loadings of Fe (0.324 wt.%) and Zn (0.344 wt.%) were measured by the inductively coupled plasma optical emission spectroscopy (ICP-OES) method. The other Fe1-Ni1/ZSM-5, Fe1-Cu1/ZSM-5, were prepared in a similar manner by using NiCl₂·6H2O, CuCl2·2H2O, and FeCl3 (Sinopharm Chemical Reagent Co., China) as metal precursors.
Evaluation of catalytic performance
DOM was performed in a 240 mL photoreactor equipped with a quartz window. The catalyst (typically 20 mg) was dispersed in 100 mL deionized water (18.2 MΩ cm−1). The photoreactor was sealed and completely degassed through a vacuum pump to remove the possible dissolved air in the deionized water. Then the mixture gas of methane (typically 50 ml/min) and oxygen (typically 25 ml/min) was introduced into the solution by inserting a gas pipeline below the liquid surface and stirred vigorously at ca. 1200 rpm. Subsequently, the reactor was irradiated by a 500 W high-pressure mercury lamp (~220 to 1100 nm, the effective light intensity is 18.1 mW/cm2 in the wavelength range of 220 and 240 nm measured by OHSP-350UVS) for 1 h. During the reaction, the temperature of the liquid was maintained at the desired reaction temperature (typically 25 oC) by the ice and water mixture. The reactor was connected with gas chromatography (GC 2060, Shanghai Ruimin Instrument Co., Ltd), and then the gaseous composition was directly analyzed by GC (column type: TDX-01) equipped with a methanizer unit and FID detector. Only CO, CO2, and CH4 could be detected in a typical GC of a gas mixture after DOM (Supplementary Fig. S21). Oxygenates in the liquid were analyzed by 1H NMR spectra on a Bruker 400 MHz NMR. Typically, 0.7 mL of product solution was mixed with 0.1 mL D2O (with 0.1012 μmol DSS) in the tube to conduct NMR measurement. 3-(trimethylsilyl)-1-propanesulfonic acid sodium salt (DSS, purchased from TCI) was used as a calibration standard. The solvent suppression technique was run to suppress the dominant H2O signal during NMR measurement. The experimental detailed for 1H-NMR were as follows: a pulse sequence of noesygppr1d was used with a spectral width of 15 ppm, 64 k data points, an acquisition time of 8.19 seconds, 32 scans, a pre-scan delay of 6.50 µs, delays of 30 s, and a transmitter frequency offset of 4.699 ppm. The 1H NMR spectra of liquid products over Fe1-Zn1/ZSM-5 after DOM are shown in Supplementary Fig. S22. The identified oxygenated products in liquid were acetic acid (δ = 2.07 ppm), acetone (δ = 2.22 ppm), methanol (δ = 3.34 ppm), and formic acid (δ = 8.28 ppm). To quantify the products accurately, their standard curves were established, respectively (Supplementary Fig. S23). Considering that CH4 is a flammable gas, it should be mentioned that all the CH4 concentrations (64.2 ~ 83.3 vol.%) we used in this work are out of the explosive limits of CH4/O2 mixtures (5.4 ~ 59.2 vol.%).
Theoretical calculation methods
To explore the mechanism of direct oxidation of methane to acetic acid over Fe1-Zn1/ZSM-5, periodic density functional theory (DFT) calculations were performed using the Vienna Ab-initio Simulation Package (VASP5.4.1)73. The projector augmented wave (PAW) method was used to describe the interaction between the atomic nucleus and electrons, with a cutoff energy for the plane wave set to 450 eV74. We used the generalized gradient approximation (GGA) Perdew-Burke-Ernzerhof (PBE) functional to describe the exchange-correlation interactions75. The Key van der Waals interactions in molecular sieves were accounted for using the DFT-D3 (BJ) method76. The convergence criterion for the maximum force on all relaxed atoms was set to 0.05 eV/Å. The transition states were located using the constraint minimization method with the same force convergence criterion77. We carefully compared all the possible spin configurations and utilized the most stable spin configuration in the manuscript. As shown in Supplementary Table S7, the ferromagnetic S = 2/2 state of high-spin and the S = 0 state of low-spin status could be finally located for [O=Fe-(μ-O)2-Zn]2+. Hence, the most stable ferromagnetic S = 2/2 state of high spin was used for Fe-Zn dual-atom sites. For Z[O = Fe-(μ-O)2-Fe]2+, four spin configurations were found, among which the ferromagnetic S = 6/2 of the high spin was the most stable one. The energy difference between S = 6/2 and metastable S = 4/2 is trivial. Therefore, the high-spin state as the method was utilized for Fe-Zn and Fe-Fe dual-atom site to perform the DFT calculation. Several groups have reported that the molecular entropy loss confined within the pores of ZSM-5 zeolite is 38% relative to the gas phase78. Therefore, the entropy of small molecules in ZSM-5 zeolite was corrected by this value. Details on the Gibbs free energy calculations, including zero-point energy, internal energy, and entropy contributions, can be found in our previous work and Supplementary Information15,22.
Data availability
The data supporting the findings of this study are included within the article and its Supplementary Information files. The data generated in this study are provided in the Source Data file. Source data are provided in this paper. All data are available upon request from the corresponding authors.
References
Mao, J. et al. Direct conversion of methane with O2 at room temperature over edge-rich MoS2. Nat. Catal. 6, 1052–1061 (2023).
Ding, J. et al. A tin-based tandem electrocatalyst for CO2 reduction to ehanol with 80% selectivity. Nat. Energy 8, 1386–1394 (2023).
Lin, M. & Sen, A. Direct catalytic conversion of methane to acetic acid in an aqueous medium. Nature 368, 613–615 (1994).
Forster, D. Mechanistic pathways in the catalytic carbonylation of methanol by rhodium and iridium complexes. Adv. Organomet. Chem. 17, 255–267 (1979).
Periana, R. A., Mironov, O., Taube, D., Bhalla, G. & Jones, C. Catalytic, oxidative condensation of CH4 to CH3COOH in one step via C-H activation. Science 301, 814–818 (2003).
Zheng, K. et al. Room-temperature photooxidation of CH4 to CH3OH with nearly 100% selectivity over hetero-ZnO/Fe2O3 porous nanosheets. J. Am. Chem. Soc. 144, 12357–12366 (2022).
Qi, G. et al. Au-ZSM-5 Catalyses the selective oxidation of CH4 to CH3OH and CH3COOH using O2. Nat. Catal. 5, 45–54 (2022).
Mohamedali, M., Ayodele, O. & Ibrahim, H. Challenges and prospects for the photocatalytic liquefaction of methane into oxygenated hydrocarbons. Renew. Sust. Energ. Rev. 131, 110024 (2020).
Hammond, C. et al. Direct catalytic conversion of methane to methanol in an aqueous medium by using copper-promoted Fe-ZSM-5. Angew. Chem. Int. Ed. 51, 5129–5133 (2012).
Snyder, B. E. R. et al. The active site of low-temperature methane hydroxylation in iron-containing zeolites. Nature 536, 317–321 (2016).
Sushkevich, V. L., Palagin, D., Ranocchiari, M. & van Bokhoven, J. A. Selective anaerobic oxidation of methane enables direct synthesis of methanol. Science 356, 523–527 (2017).
Lieberman, R. L. & Rosenzweig, A. C. Crystal structure of a membrane-bound metalloenzyme that catalyses the biological oxidation of methane. Nature 434, 177–182 (2005).
Yu, B. et al. Silver and copper dual single atoms boosting direct oxidation of methane to methanol via synergistic catalysis. Adv. Sci. 10, 2302143 (2023).
Zhang, Q. et al. Establishing metal-nonoxygen bonds to improve thermal stability of Pt1/CeO2 via coating boron nitride. Chem. Eng. Sci. 302, 120856 (2025).
Ni, L. et al. Thiourea modification promoting the activity on Pt/CeO2 for CO oxidation by weakening the metal−support interaction. Ind. Eng. Chem. Res. 64, 4835−4844 (2025)
Yu, B. et al. Surface hydroxyl group dominating aerobic oxidation of methane below room temperature. Energy Environ. Sci. 17, 8127–8139 (2024).
Zhao, Y. et al. Steric-confinement Rh2/MoS2 dual-atom catalyst directionally modulating adsorption configuration of ester group to boost ethanol synthesis. Chem 10, 3342–3363 (2024).
Shi, Y. et al. Local In-O-Pd lewis acid-base pair boosting CO2 selective hydrogenation to methanol. Chem. Eng. J. 485, 150093 (2024).
Zhou, P. et al. Synergetic interaction between neighboring platinum and ruthenium monomers boosts CO oxidation. Chem. Sci. 10, 5898–5905 (2019).
Zhao, Q.-P. et al. Photo-induced synthesis of heteronuclear dual-atom catalysts. Nat. Synth. 3, 497–506 (2024).
Chen, J. et al. Photoinduced precise synthesis of diatomic Ir1Pd1-In2O3 for CO2 hydrogenation to methanol via angstrom-scale-distance dependent synergistic catalysis. Angew. Chem. Int. Ed. 63, e202401168 (2024).
Zeng, M. et al. ZSM-5-confined Cr1–O4 active sites boost methane direct oxidation to C1 oxygenates under mild conditions. EES Catal. 1, 153–161 (2023).
Wang, S. et al. H2-reduced Phosphomolybdate promotes room-temperature aerobic oxidation of methane to methanol. Nat. Catal. 6, 895–905 (2023).
Zhao, Y. et al. Vacancy defects inductive effect of asymmetrically coordinated single-atom Fe─N3S1 active sites for robust electrocatalytic oxygen reduction with high turnover frequency and mass activity. Adv. Mater. 36, 2308243 (2024).
Ou, H. et al. Carbon nitride photocatalysts with integrated oxidation and reduction atomic active centers for improved CO2 conversion. Angew. Chem. Int. Ed. 61, e202206579 (2022).
Wang, G. et al. Bonding interaction of adjacent Pt and Ag single-atom pairs on carbon nitride efficiently promotes photocatalytic H2 production. CCS Chem. 6, 1523–1534 (2024).
An, Z. et al. Highly active, Ultra-low loading single-atom iron catalysts for catalytic transfer hydrogenation. Nat. Commun. 14, 6666 (2023).
Cui, J. et al. Regulating the metal–support interaction: double jump to reach the efficiency apex of the Fe–N4-catalyzed fenton-like reaction. ACS Catal. 12, 14954–14963 (2022).
Zhang, X. et al. Identifying and tailoring C–N coupling site for efficient urea synthesis over diatomic Fe–Ni catalyst. Nat. Commun. 13, 5337 (2022).
Zhang, L. et al. Water activation triggered by Cu-Co double-atom catalyst for silane oxidation. Angew. Chem. Int. Ed. 62, e202313343 (2023).
Shu, X., Tan, D., Wang, Y., Ma, J. & Zhang, J. Bimetal-bridging nitrogen coordination in carbon-based electrocatalysts for pH-universal oxygen Reduction. Angew. Chem. Int. Ed. 63, e202316005 (2024).
Yang, Y. et al. Integrating enrichment, reduction, and oxidation sites in one system for artificial photosynthetic diluted CO2 reduction. Adv. Mater. 35, 2304170 (2023).
Su, H. et al. Flexibility tuning of dual-metal S-Fe-Co-N5 catalysts with O-axial ligand structure for electrocatalytic water splitting. Adv. Energy Mater. 13, 2301547 (2023).
Li, Y. et al. Synergistic effect of atomically dispersed Ni–Zn pair sites for enhanced CO2 electroreduction. Adv. Mater. 33, 2102212 (2021).
Sun, X. et al. Isolated Fe-Co heteronuclear diatomic sites as efficient bifunctional catalysts for high-performance lithium-sulfur batteries. Nat. Commun. 14, 291 (2023).
Li, L. et al. Efficient sunlight-driven dehydrogenative coupling of methane to ethane over a Zn+-modified zeolite. Angew. Chem. Int. Ed. 50, 8299–8303 (2011).
Cheng, M. et al. Constructing charge transfer channel between dopants and oxygen vacancies for enhanced visible-light-driven water oxidation. Nano. Res. 14, 3365–3371 (2021).
Yan, Y., Jiang, S., Zhang, H. & Zhang, X. Preparation of novel Fe-ZSM-5 zeolite membrane catalysts for catalytic wet peroxide oxidation of phenol in a membrane reactor. Chem. Eng. J. 259, 243–251 (2015).
Yasin, G. et al. Simultaneously engineering the synergistic-effects and coordination-environment of dual-single-atomic iron/cobalt-sites as a bifunctional oxygen electrocatalyst for rechargeable zinc-air batteries. ACS Catal. 13, 2313–2325 (2023).
Lu, W. et al. Preparation of ZnO films with variable electric field-assisted atomic layer deposition technique. Appl. Surf. Sci. 303, 111–117 (2014).
Kong, Y. et al. Atomically dispersed Zinc(I) active sites to accelerate nitrogen reduction kinetics for ammonia electrosynthesis. Adv. Mater. 34, 2103548 (2022).
Deng, D. et al. Engineering electronic density and coordination environment of Mn-Nx sites via Zn cooperation for quasi-solid-state zinc-air batteries. Renewables 1, 362–372 (2023).
Qiao, B. et al. Highly efficient catalysis of preferential oxidation of CO in H2-Rich stream by gold single-atom catalysts. ACS Catal. 5, 6249–6254 (2015).
Tsukamoto, D. et al. Photocatalytic H2O2 production from ethanol/O2 system using TiO2 loaded with Au–Ag bimetallic alloy nanoparticles. ACS Catal. 2, 599–603 (2012).
Ruschel, G. K., Nemetz, T. M. & Ball, D. W. Matrix isolation and density functional studies of novel transition metal complexes. NO + Fe, Co, Ni, Cu, and Zn in argon matrices. J. Mol. Struct. 384, 101–114 (1996).
Sun, J., Gao, W., Fei, H. & Zhao, G. Efficient and selective electrochemical reduction of nitrate to N2 by relay catalytic effects of Fe-Ni bimetallic sites on MOF-derived structure. Appl. Catal. B Environ. 301, 120829 (2022).
Wang, L. et al. In situ DRIFTS study of the NO + CO reaction on Fe–Co binary metal oxides over activated semi-coke supports. RSC Adv. 7, 7695–7710 (2017).
Snyder, B. E. R. et al. Mechanism of selective benzene hydroxylation catalyzed by iron-containing zeolites. Proc. Natl. Acad. Sci. USA 115, 12124–12129 (2018).
Kumar, M. S., Schwidder, M., Grünert, W. & Brückner, A. On the nature of different iron sites and their catalytic role in Fe-ZSM-5 DeNOx catalysts: new insights by a combined EPR and UV/VIS spectroscopic approach. J. Catal. 227, 384–397 (2004).
Yu, T. et al. Highly selective oxidation of methane into methanol over Cu-promoted monomeric Fe/ZSM-5. ACS Catal. 11, 6684–6691 (2021).
Wang, H. et al. Facilitating the dry reforming of methane with interfacial synergistic catalysis in an Ir@CeO2-x catalyst. Nat. Commun. 15, 3765 (2024).
Li, M. et al. Single-step selective oxidation of methane to methanol in the aqueous phase on iridium-based catalysts. Appl. Catal. B Environ. 292, 120124 (2021).
Alexander, A. J., Kim, Z. H. & Zare, R. N. Photodissociation of O2 via the herzberg continuum: measurements of O-atom alignment and orientation. J. Chem. Phys. 118, 10566–10574 (2003).
Buijsse, B. et al. Angular distributions for photodissociation of O2 in the herzberg continuum. J. Chem. Phys. 108, 7229–7243 (1998).
Zare, R. N. & Herschbach, D. R. Doppler line shape of atomic fluorescence excited by molecular photodissociation. P. IEEE 51, 173–182 (1963).
Kepp, K. P. A quantitative scale of oxophilicity and thiophilicity. Inorg. Chem. 55, 9461–9470 (2016).
Xu, J., Cao, X.-M. & Hu, P. Improved prediction for the methane activation mechanism on rutile metal oxides by a machine learning model with geometrical descriptors. J. Phys. Chem. C. 123, 28802–28810 (2019).
Aljama, H., Nørskov, J. K. & Abild-Pedersen, F. Theoretical insights into methane C–H bond activation on alkaline metal oxides. J. Phys. Chem. C. 121, 16440–16446 (2017).
Szecsenyi, A., Li, G., Gascon, J. & Pidko, E. A. Mechanistic complexity of methane oxidation with H2O2 by single-site Fe/ZSM-5 catalyst. ACS Catal. 8, 7961–7972 (2018).
Van Heuvelen, K. M. et al. One-electron oxidation of an oxoiron(IV) complex to form an [O=FeV=NR]+ center. Proc. Natl. Acad. Sci. USA 109, 11933–11938 (2012).
Nakamoto, K. Resonance raman spectra and biological significance of high-valent Iron (IV, V) porphyrins. Coord. Chem. Rev. 226, 153–165 (2002).
Snyder, B. E. R. et al. Structural characterization of a non-heme iron active site in zeolites that hydroxylates methane. Proc. Natl. Acad. Sci. USA 115, 4565–4570 (2018).
Hou, K. et al. Reactive high-spin iron(IV)-oxo sites through dioxygen activation in a metal–organic framework. Science 382, 547–553 (2023).
Zhang, Y. et al. H2O2 Generation from O2 and H2O on a near-infrared absorbing porphyrin supramolecular photocatalyst. Nat. Energy 8, 361–371 (2023).
Jin, Z. et al. Hydrophobic zeolite modification for in situ peroxide formation in methane oxidation to methanol. Science 367, 193–197 (2020).
Xu, W. et al. Metal-oxo electronic tuning via in situ CO decoration for promoting methane conversion to oxygenates over single-atom catalysts. Angew. Chem. Int. Ed. 63, e202315343 (2024).
Xie, J. et al. Stable photocatalytic coupling of methane to ethane with water vapor using TiO2 supported ultralow loading AuPd nanoparticles. Acta Phys. Sin.-Ch. Ed. 39, 2306037 (2023).
Wu, W. et al. CO2 Hydrogenation over copper/ZnO single-atom catalysts: water-promoted transient synthesis of methanol. Angew. Chem. Int. Ed. 61, e202213024 (2022).
Ding, L. et al. Over 70 % faradaic efficiency for CO2 electroreduction to ethanol enabled by potassium dopant-tuned interaction between copper sites and intermediates. Angew. Chem. Int. Ed. 61, e202209268 (2022).
Li, F.-M., Xia, C., Fang, W., Chen, Y. & Xia, B. Y. RhCuBi Trimetallenes with composition segregation coupled crystalline-amorphous heterostructure toward ethanol electrooxidation. Adv. Energy Mater. 14, 2400112 (2024).
Jia, G. et al. Asymmetric coupled dual-atom sites for selective photoreduction of carbon dioxide to acetic acid. Adv. Funct. Mater. 32, 2206817 (2022).
Liu, Y., Xue, D., Li, W., Li, C. & Wan, B. A simple method for the precise determination of multi-elements in pyrite and magnetite by ICP-MS and ICP-OES with matrix removal. Microchem. J. 158, 105221 (2020).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B 47, 558–561 (1993).
Perdew, J. P. et al. Atoms, molecules, solids, and surfaces: applications of the generalized gradient approximation for exchange and correlation. Phys. Rev. B 46, 6671–6687 (1992).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Grimme, S., Ehrlich, S. & Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 32, 1456–1465 (2011).
Cheng, Q. et al. Maximizing active Fe species in ZSM-5 zeolite using organic-template-freesynthesis for efficient selective methane oxidation. J. Am. Chem. Soc. 145, 5888–5898 (2023).
Dauenhauer, P. J. & Abdelrahman, O. A. A universal descriptor for the entropy of adsorbed molecules in confined Spaces. ACS Cent. Sci. 4, 1235–1243 (2018).
Acknowledgements
This project was supported financially by the National Key R&D Program of China (2021YFB3501900, 2023YFA1507601), National Natural Science Foundation of China (U21A20326, 22172065, 22306063, and 22022302), Natural Science Foundation of Jiangsu Province (BK20211239), and Jiangsu Specially-Appointed Professor (1046010241211400). We thank the Center for High Performance Computing at Shanghai Jiao Tong University for providing the computing resources of the Siyuan-1 cluster. We acknowledge support from Lixia Yan (Analysis and Testing Center of Jiangnan University) for the help with the HRTEM/STEM test.
Author information
Authors and Affiliations
Contributions
Baiyang Yu: Data curation, Investigation, Formal analysis, Writing-original draft. Wenlong Li: DFT Data curation, Investigation, Formal analysis, Writing-original draft. Xuan Tang: Data curation, Investigation, Formal analysis. Lu Cheng: DFT Data curation, Investigation, Formal analysis. Fudong Liu: Data curation, Investigation, Formal analysis. Jiawei Zhang: Formal analysis. Shuobing Yang: Formal analysis. Jing Xu: Formal analysis. Ying Zhang: Investigation. Chengsi Pan: Formal analysis. Xiaoming Cao: DFT Data curation, Supervision, Writing-review & editing. Yongfa Zhu: Supervision. Yang Lou: Conceptualization, Methodology, Data curation, Funding acquisition, Writing-original draft, review & editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yu, B., Li, W., Tang, X. et al. Fe-(μ-O)-Zn dual-atom boosting C-C coupling for direct oxidation of methane to acetic acid using O2. Nat Commun 16, 9471 (2025). https://doi.org/10.1038/s41467-025-64505-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-64505-9