Abstract
Green and sustainable 90Sr uptake is urgently needed for radionuclides remediation. Herein, we propose an efficient strategy based on electrochemically switched ion exchange (ESIX) method to enhance 90Sr uptake using electroactive ion exchange material. A titanate perovskite (Na2La2Ti3O10) with outstanding acid, irradiation resistance, and thermal stability can effectively capture Sr2+ and achieve remediation of actual acidic 90Sr-containing liquid waste (RSr > 99%). Na2La2Ti3O10 is prepared as working electrode C@NaLaTiO to enhance Sr2+ uptake via ESIX. Remarkably, the Sr2+ adsorption capacity increases (from 104.84 to 175.43 mg·g−1) and high selectivity for Sr2+ is maintained even under strongly acidic solutions. The Sr2+ adsorption-desorption can be controlled via facile potential modulation. Mechanism study indicates that efficient Sr2+ capture originates from the ion exchange between Sr2+ and interlayer Na+ in Na2La2Ti3O10 coupled with the electrochemical redox reaction between Ti4+/Ti3+ and the increase in oxygen vacancy. Density functional theory calculations support that ESIX enhances Sr2+ adsorption by increasing the binding energy of anionic [La2Ti3O10]n2n− layers towards Sr2+. This study offers a convenient and environmentally friendly way for the efficient 90Sr enrichment from radioactive waste liquids.
Similar content being viewed by others
Introduction
The sustainable development of the nuclear industry is closely linked to properly disposing of radioactive waste. As one of the most dangerous radionuclides in the 235U fission products, 90Sr (t1/2 ~ 28 years) exists in the form of ions (90Sr2+) in radioactive liquid waste and releases β rays1,2,3. Due to its chemical similarity to calcium, 90Sr2+ can accumulate in bones once entering the human body, which may lead to serious health problems (such as bone cancer and leukemia)4,5. Furthermore, the high solubility and easy environmental mobility make 90Sr2+ a significant threat to ecosystems1,4. For instance, after the Fukushima nuclear accident in 2011, the concentration of 90Sr was found to be four orders of magnitude higher than the environmental background level in the offshore waters, which aroused people’s concern about the safety of nuclear energy6. Therefore, the development of green and efficient methods to remove 90Sr from radioactive liquid waste is of great significance for environmental protection and human health.
Ion exchange method has been widely employed for the removal of Sr2+ from radioactive liquid waste2. However, traditional ion exchange materials such as resins, clays, and zeolites suffer from low adsorption capacity and poor selectivity for Sr2+, especially in acidic or high-salinity environments due to their poor chemical stability and interference from competitive ions7. The electrochemically switched ion exchange (ESIX) method has emerged as a promising approach for the efficient removal of radionuclides, which synergistically combines electrochemical adsorption with ion exchange8. This technique employs controlled electrical potentials to modulate redox states in electrode materials accompanied by the occurrence of ion exchange, enabling dynamic control over ion capture and release processes. Compared with the traditional ion exchange method, ESIX can significantly enhance the adsorption capacity by adjusting the voltage and minimize the formation of secondary pollutants in a more sustainable and environmentally friendly way9,10. The key of ESIX is to develop electrode materials with excellent structural stability that can dynamically respond to electric fields and meanwhile possess ion exchange properties. To date, hexacyanoferrates have been explored for ESIX applications due to their ion exchange performance and reversible redox reaction between Fe3+ and Fe2+, particularly for the removal of Cs+ ions11. For instance, nickel hexacyanoferrate/porous three-dimensional carbon felt (NiHCF/PTCF) and iron hexacyanoferrate/graphene/carbon fibers composite (PB/GN/CFs) have both demonstrated significant Cs+ uptake capacity through ESIX12,13. Other inorganic materials containing metals with variable valency have been used for Cs+ removal through ESIX, for example, Mo(Ⅵ)-based inorganic material Na1.5Mo6O1814. However, ESIX has not yet been used to treat Sr2+, which may originate from the fact that the large hydration radii and high valency of Sr²⁺ can lead to difficulty in ion mobility and reversible insertion15.
Perovskite-type oxides have emerged as a versatile class of materials with excellent water stability, tunable structures, and abundant active sites, making them ideal for applications in radionuclides adsorption16,17,18,19,20. Three-dimensional (3D) perovskite-type oxides such as CaAlOx, CaTiOx, and CaTiO3 have shown high adsorption capacities for U(VI) due to their large specific surface areas and accessible adsorption sites21,22. On the other hand, two-dimensional (2D) perovskite-type oxides offer superior tunability in composition and structure, which render them promising in radionuclides adsorption23. 2D perovskite-type oxides are categorized into three main types: R-P-type (Ruddlesden–Popper, A′2An-1BnO3n+1), D-J-type (Dion–Jacobson, A′An-1BnO3n+1), and AV-type (Aurivillius, (Bi2O2)An-1BnO3n+1)24. Among them, only D-J-type perovskite-type oxides (HCa2Nb3O10·1.5H2O and HLaNb2O7) have been reported for Cs+ removal25,26. By contrast, the R-P-type perovskite-type oxides with two interlayer cations per formula unit possess twice the anionic charge density of the perovskite-type oxides. Therefore, R-P-type perovskite-type oxides possess anionic layers with a higher negative charge, which will be beneficial to have higher ion exchange capacities27. To date, however, R-P-type perovskite-type oxides remain underexplored for radionuclide capture.
R-P-type titanate perovskites, including A2La2Ti3O10 (A = Li, Na, K, Rb, and H) and ALaTiO4 (A = Li, Na, K, and H), have been reported28,29,30. The presence of interlayer cations in their structures provides abundant exchangeable sites for adsorption. In addition, titanium can show a variety of valency states, including Ti2+, Ti3+, and Ti4+, which endows layered titanates with good redox activity. As a result, titanates have been well-suited for electrochemically controlled adsorption and desorption processes31,32. For example, under the applied electric field, the Cs+ adsorption capacity of Na2Ti3O7 nanotubes is greatly enhanced due to the valency change of Ti31. The redox-active nature of R-P-type titanate perovskites, combined with their tunable structures and high stability, offers great potential for selective radionuclide removal via ESIX. Therefore, it is anticipated that incorporating electroactive R-P-type titanate perovskites into the ESIX system should be a promising strategy for improving the capture of radioactive Sr2+.
Guided by this strategy, the R-P-type titanate perovskite Na2La2Ti(Ⅳ)3O10 with high acid, thermal, and irradiation stability is screened out. It exhibits high adsorption capacity (104.84 mg·g−1) and fast kinetics (30 min) for Sr2+ adsorption. Na2La2Ti3O10 demonstrates excellent treatment capability for actual acidic 90Sr-liquid-waste generated during industrial production. When Na2La2Ti3O10 is prepared as the working electrode C@NaLaTiO for Sr2+ capture, under ESIX, the Sr2+ adsorption capacity increases significantly to 175.43 mg·g−1. Impressively, the selectivity for Sr2+ in highly acidic conditions (pH = 1) is improved by nearly two orders of magnitude (56.30 mL·g−1 to 2.23 × 103 mL·g−1). The Sr2+ adsorption mechanism is revealed and the efficient Sr2+ capture is attributed to the ion exchange between Sr2+ and interlayered Na+ in Na2La2Ti3O10 and electrochemical adsorption. By employing an external potential field, Ti4+ is reduced to Ti3+, causing the increase in oxygen vacancy and promoting Sr2+ adsorption in the interlayer space33. On the contrary, when Ti3+ is oxidized to Ti4+ under a reverse electric field, the material can achieve the desorption of Sr2+. DFT calculations show that introducing ESIX enhances the binding energy of the anionic [La2Ti3O10]n2n- layers for Sr2+, indicating enhanced interaction between the anionic layer [La2Ti3O10]n2n- and Sr2+ ions under applied voltage. This work not only advances the development of the ESIX system for Sr2+ uptake but also provides a sustainable approach for managing radioactive strontium contamination.
Results
Characterizations
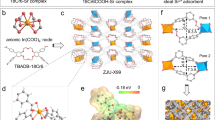
The structure of Na2La2Ti3O10 features the anionic triple perovskite slabs of [La2Ti3O10]n2n- with interlayer Na+ ions (Supplementary Fig. 1a)34. The triple perovskite slabs of [La2Ti3O10]n2n- are built up of [TiO6] octahedra in corner-sharing mode, while La3+ ions are cavitied in the intralyer spaces. The adjacent slabs are stacked with a displacement of 0.5 along the a-axis24. High-valency Ti4+ in the structure provides the redox condition for the treatment of Sr2+ by ESIX.
The Sr2+ adsorption product of Na2La2Ti3O10 is denoted as Na2La2Ti3O10-Sr. Energy dispersive spectroscopy (EDS) analysis and elemental distribution mapping confirm the successful adsorption of Sr2+ and its uniform distribution in the Na2La2Ti3O10-Sr sample (Supplementary Fig. 1b–d). The optical adsorption edges of Na2La2Ti3O10 and Na2La2Ti3O10-Sr are 3.21 eV and 3.34 eV, respectively (Supplementary Fig. 2a, b). By comparing the powder X-ray diffraction (PXRD) patterns of Na2La2Ti3O10-Sr with the simulated one of Na2La2Ti3O10, it can be found that the layered structure of Na2La2Ti3O10 is maintained before and after the adsorption of Sr2+ (Supplementary Fig. 2c, d). Thermogravimetric analysis (TGA) results show that there is almost no weight loss of Na2La2Ti3O10 from 25 to 1200 °C (Supplementary Fig. 2e). Due to the loss of adsorbed water molecules, Na2La2Ti3O10-Sr has a weight loss of 0.70% from 25 to 160 °C (Supplementary Fig. 2e). PXRD analyses confirm that Na2La2Ti3O10-Sr can maintain the original skeleton without collapsing after heating to 1200 °C. These results indicate excellent thermal stability of the current titanate perovskite (Supplementary Fig. 2f). Furthermore, by comparing the scanning electron microscope (SEM) images of Na2La2Ti3O10 and Na2La2Ti3O10-Sr, it is evident that Na2La2Ti3O10 exhibits a sheet-like morphology, whereas Na2La2Ti3O10-Sr maintains a similar sheet-like structure, albeit with a slight tendency toward pulverization (Supplementary Fig. 3a, b).
To confirm the acid stability of Na2La2Ti3O10, Na2La2Ti3O10 samples were placed in various solutions (pH = 7 to 4 mol·L−1 HNO3) at room temperature (RT) with soaking for 12 h, respectively. In the pH range of 4 to 7, the PXRD patterns of the immersed materials are consistent with that of the original Na2La2Ti3O10 (Supplementary Fig. 3c). As the acidity increases from pH of 3 to 2 mol·L−1 HNO3, the immersed samples transform into the H2La2Ti3O10 according to the PXRD results, whereas the layered framework remains stable (Supplementary Fig. 3c). More importantly, the leaching rates of La3+ and Ti4+ for Na2La2Ti3O10 in the pH range from 1 to 7 and even in 1–2 mol·L−1 HNO3 solutions are less than 6.17% and 5.51%, respectively (Supplementary Fig. 3d and Table 1).
Further, Na2La2Ti3O10 was combined with carbon black and polyvinylidene fluoride, and then coated on the surface of carbon cloth to prepare the working electrode C@NaLaTiO for Sr2+ capture (Fig. 1a and Supplementary Fig. 4a, b). The adsorption product of C@NaLaTiO is denoted as C@NaLaTiO-Sr. EDS analysis confirms the successful Sr2+ adsorption and uniform distribution in C@NaLaTiO-Sr (Supplementary Fig. 4c, d). Cyclic voltammograms of C@NaLaTiO in the solutions (50 mg·L−1 SrCl2·6H2O) at different scanning rates of 0.02–0.1 V·s−1 were tested (Fig. 1b). It can be seen that the redox peak is very wide, indicating that C@NaLaTiO has good electrochemical activity. As the scanning rate increases, there is a peak shift, but the overall shape of the redox peak remains, which means that the capacity of C@NaLaTiO is still reversible even at high scanning rates. The diffusion-controlled contribution reaches 66.83% at 0.08 V·s−1, suggesting that the partial current generated by the insertion and release of Sr2+ in the interlayer space has a significant effect on the total current (Supplementary Table 2). In addition, when the scanning rate increases from 0.02 V·s−1 to 0.1 V·s−1, the capacitance contributions increase from 50.29% to 69.23%. Therefore, the kinetics of the capacitance contribution on the surface is a rate-determining process rather than a diffusion-controlled process9. Furthermore, after 100 times of the electrochemical redox process, the CV curves do not change significantly, which proves that C@NaLaTiO has good stability during the electrochemical process (Fig. 1c). Additionally, there is no obvious change in the CV curves after 100 electrochemical redox processes in the voltage range of –4 V to 4 V, demonstrating the exceptional cycling stability of C@NaLaTiO under high-voltage operation (Supplementary Fig. 4e). The SEM images of C@NaLaTiO and C@NaLaTiO-Sr reveal a characteristic stacked plate-like morphology, which facilitates efficient ion transport pathways and provides abundantly accessible sites for Sr2+ adsorption (Supplementary Fig. 4f, g).
a Diagram of electrochemical adsorption and desorption process of Sr2+ on the C@NaLaTiO electrode. b CV curves of C@NaLaTiO at different scan rates in 50 mg·L-1 SrCl2·6H2O solutions. c CV curves of C@NaLaTiO from the 1st to 100th cycle at 0.08 V·s-1 in 50 mg·L-1 SrCl2·6H2O solutions. Source data are provided as a Source Data file.
Batch adsorption experiments of Na2La2Ti3O10
The adsorption capacity of Na2La2Ti3O10 for Sr2+ at RT has been measured by static batch adsorption experiments. The Sr2+ adsorption equilibrium curve of Na2La2Ti3O10 has been fitted with the Langmuir, Freundlich, and Langmuir-Freundlich adsorption isotherm models (Fig. 2a and Supplementary Table 3)35. It is found that the isotherm curve can be better fitted with the Langmuir-Freundlich isotherm model with a higher correlation coefficient of 0.99028 (Supplementary Table 4). According to the Langmuir-Freundlich adsorption isotherm model, the maximum Sr2+ adsorption capacity (qmSr) of Na2La2Ti3O10 is 104.84 mg·g−1 (Supplementary Table 4). Compared with Na2TinO2n+1 (49.6 mg·g−1), titanate nanotube (66.72 mg·g−1), K2Ti6O13/SiO2 (14.631 mg·g−1), Fe3O4-titanate fiber magnetic composite (37.1 mg·g−1), and other titanate materials, Na2La2Ti3O10 has a higher Sr2+ adsorption capacity36,37,38,39. The qmSr of Na2La2Ti3O10 is also higher than that of traditional adsorption materials such as ZrO2-Sb2O5 (22.21 mg·g−1), thorium tungsten phosphate (57 mg·g−1), Sb(III)/Sb2O5 (25.7 mg·g−1), and hex-WO3 (28 mg·g−1)40,41,42,43.
a The Sr2+ adsorption equilibrium data of Na2La2Ti3O10 fitted with the Langmuir, Freundlich, and Langmuir-Freundlich isotherm models. b The kinetics curve of Na2La2Ti3O10 for Sr2+ adsorption. c Kd (mL·g-1) and R (%) of Na2La2Ti3O10 for Sr2+ and Cs+ removal under different Cs/Sr molar ratios. d The treatment capacity of Na2La2Ti3O10 for 5 mL acidic (pH = 3.75) 90Sr-liquid-waste plotted as radioactivity concentration and RSr (%) vs the mass of adsorbent (mg), respectively. Source data are provided as a Source Data file.
It is important that a material can rapidly capture the target radioactive ions in solutions44,45. Thus, the kinetic adsorption experiment was carried out to evaluate the Sr2+ capture ability of Na2La2Ti3O10 (Fig. 2b). The removal rate R (%) of Sr2+ quickly reaches more than 90% within 30 min (Supplementary Tables 5 and 6). The kinetic data of Na2La2Ti3O10 for Sr2+ capture can be fitted better with the pseudo-second-order kinetics (R2 = 0.98423) rather than the pseudo-first-order kinetics model (R2 = 0.93519), which proves that the adsorption process of Sr2+ is chemical sorption46.
The pH values of solutions have a strong influence on the adsorption due to the competition of protons and the influence of the material’s acid stability47. Therefore, the Sr2+ adsorption experiment of Na2La2Ti3O10 under a wide pH range has been carried out (Supplementary Fig. 5b and Table 7). In the pH range of 4–12, Na2La2Ti3O10 maintains high distribution coefficients (KdSr > 3.68 × 103 mL·g−1). In particular, it can remove more than 81% of Sr2+ ions with the KdSr of 4.31 × 103 mL·g−1 at pH of 4. Even under strongly alkaline conditions (pH = 12), Na2La2Ti3O10 can maintain high removal performance (KdSr = 7.37 × 104 mL·g−1). However, in the pH range of 1–3, KdSr decrease to lower than 103.56 mL·g−1. The significant decrease of KdSr is attributed to the competitive adsorption of protons, because H+ intercalation leads to the formation of the H2La2Ti3O10 phase (Supplementary Fig. 3c). The above pH-dependent experiment demonstrates that Na2La2Ti3O10 can maintain excellent Sr2+ removal capacity in the pH range of 4 to 12.
Sr2+ adsorption performances of Na2La2Ti3O10 before and after 100 and 200 kGy (kilogray) γ irradiations were studied (Supplementary Table 8). After γ irradiation, the parent structure of Na2La2Ti3O10 can remain unchanged, which is confirmed by comparing the PXRD patterns of the pristine and irradiated samples (Supplementary Fig. 5c). The RSr of the pristine Na2La2Ti3O10 is 96.37%, while samples after 100 and 200 kGy γ irradiation can still retain high RSr (more than 95.12%) (Supplementary Fig. 5d). These results demonstrate that Na2La2Ti3O10 has excellent irradiation stability.
Selectivity is one of the important indexes of adsorption materials48,49,50. Therefore, the Sr2+ capture performance of Na2La2Ti3O10 in the presence of individual Cs+, K+, and Ca2+ was investigated. When Cs/Sr molar ratios vary from 10.36 to 938.43, the KdSr can be maintained in the range from 6.83 × 103 to 1.94 × 104 mL·g−1, corresponding to the RSr greater than 87%, whereas the KdCs are only in the range of 1.28 × 102 to 2.25 × 102 mL·g−1 (Fig. 2c and Supplementary Table 9). Even at very high concentration of Cs+ (Cs/Sr molar ratio up to 4574.04), the KdSr of Na2La2Ti3O10 still reaches 1.58 × 103 mL·g−1, whereas the KdCs is lower than 20 mL·g−1. Similarly, under K/Sr molar ratios of 17.57 to 2633.29, the KdSr values of Na2La2Ti3O10 can be maintained in the range from 5.3 × 103 to 2.26 × 104 mL·g−1, while the KdK only vary from 93.50 to 2.44 × 102 mL·g−1 (Supplementary Table 10). Apparently, Na2La2Ti3O10 can selectively capture Sr2+ in the presence of excess Cs+ and K+. In addition, KdSr range from 5.7 × 103 to 1.86 × 104 mL·g−1 (RSr > 85%) when Ca/Sr molar ratios are in the range of 6.52 to 637.01 (Supplementary Table 11). At the same time, KdCa are all lower than 5.13 × 102 mL·g−1. This indicates that in the presence of interfering Ca2+ ions, Na2La2Ti3O10 can still maintain high Sr2+ removal performance.
Further, the selective Sr2+ capture ability of Na2La2Ti3O10 under the coexistence of K/Ca/Na/Mg/Sr/Cs/Eu/Zr was also investigated. When the mass concentrations of competing ions (K+, Na+, Cs+, Ca2+, and Mg2+) are close to that of Sr2+, the KdSr remains higher than 7.24 × 103 mL·g−1, whereas Kd of competing ions (K+, Na+, Cs+, Ca2+, and Mg2+) are all less than 5.56 × 102 mL·g−1 (Supplementary Table 12). Even if the mass concentrations of competing ions are five times higher than that of Sr2+ under the coexistence of K+/Ca2+/Na+/Mg2+/Sr2+/Cs+, the KdSr still reaches 3.66 × 103 mL·g−1 (Supplementary Table 13). Moreover, the KdSr is higher than 2.95 × 104 mL·g−1 with the RSr more than 96.73% when Sr2+ coexists with equal mass concentrations of Mn+ ions (Mn+ = Cs+, Eu3+, and Zr4+), while the KdM are lower than 2.66 × 103 mL·g−1 (Supplementary Table 14). Despite the mass concentrations of Mn+ being five times higher than that of Sr2+ under the coexistence of Sr2+/Cs+/Eu3+/Zr4+, the KdSr remains above 2.49 × 104 mL·g−1 (Supplementary Table 15). Moreover, to evaluate the feasibility of removing Sr2+ in actual water systems, the adsorption experiment of Na2La2Ti3O10 was further carried out in Sr2+-contaminated actual water samples (Supplementary Table 16). Na2La2Ti3O10 shows good removal efficiency of Sr2+ in lake water, river water, and tap water with KdSr of 3.65 × 103 mL·g−1, 4.24 × 103 mL·g−1, and 6.18 × 103 mL·g−1, respectively.
To study the reusability of Na2La2Ti3O10, adsorption-desorption experiments were carried out. The adsorption-desorption efficiency of Na2La2Ti3O10 was systematically investigated for four adsorption-desorption cycles. It is found that Na2La2Ti3O10 can still maintain the stability of its layered skeleton after four adsorption-desorption cycles, and RSr can still reach more than 98% (Supplementary Table 17 and Fig. 5e).
Na2La2Ti3O10 was also used for the treatment of acidic (pH = 3.75) 90Sr-liquid-waste generated from the production of radioactive sources at CNNC HTA Co., Ltd (Fig. 2d). 90Sr-liquid-waste contains 60Co2+ and Na+ ions and so on in addition to 90Sr2+. The results show that only 5 mg of Na2La2Ti3O10 can remove 98.80% of 90Sr from 5 mL acidic 90Sr-liquid-waste, and the 90Sr activity concentration of wastewater is reduced by two orders of magnitude (Supplementary Table 18). When 40 mg of Na2La2Ti3O10 is used (solid-liquid ratio m/V = 8 mg·mL−1), the RSr reaches 99.18%. Therefore, a small amount of Na2La2Ti3O10 can significantly reduce the radioactivity of acidic radioactive 90Sr wastewater. Na2La2Ti3O10 exhibits excellent 90Sr removal performance under actual working conditions, highlighting the current layered titanate-based perovskite as a 90Sr scavenger with practical application potential.
Electrochemical adsorption experiments of C@NaLaTiO
Electrochemical adsorption experiments were carried out in a three-electrode system, in which C@NaLaTiO was the working electrode, carbon cloth was the counter electrode, and the saturated calomel electrode (Hg/Hg2Cl2/KCl saturated solution) was the reference electrode. The electrochemical adsorption experiment was performed by applying a potential of 0 to –4 V in 50 mL SrCl2·6H2O solution (C0Sr = 57.89 mg·L−1) to determine the effect of the voltage on the Sr2+ adsorption (Fig. 3a and Supplementary Table 19). In the electrochemically controlled adsorption process, the adsorption capacity remains almost stable in the voltage range of 0 to –1 V. When the negative potential is up from –2 to –4 V, the adsorption capacity begins to increase significantly and finally reaches 175.43 mg·g−1. Compared with the batch adsorption experiment, the adsorption capacity increases from 104.84 mg·g−1 to 175.43 mg·g−1. The Sr2+ adsorption capacity of C@NaLaTiO is more than that of some Sr2+ adsorbents such as FZU-1 (62.84 mg·g−1), SSAC hydrogel (134.40 mg·g−1), and SA-PA-H (151.70 mg·g−1)51,52,53,54. Meanwhile, it is found that the applied voltage has a great influence on the electrochemical adsorption performance; thus, the applied voltage of –4 V was used in the subsequent experiments.
a Sr2+ adsorption capacity by C@NaLaTiO at different potentials. b KdSr (mL·g-1) of C@NaLaTiO at various pH values. c Kd (mL·g-1) of various metal ions by C@NaLaTiO in the coexistence of K+, Na+, Cs+, Ca2+, Sr2+, and Mg2+ under neutral conditions. d Kd (mL·g-1) of various metal ions by C@NaLaTiO in the coexistence of K+, Na+, Cs+, Ca2+, Sr2+, and Mg2+ at pH of 3. Error bars present the standard deviation of the mean of three experiments. Source data are provided as a Source Data file.
The kinetic study was carried out to evaluate the capture ability of C@NaLaTiO toward Sr2+ ions (Supplementary Fig. 6a and Table 20). The concentration of Sr2+ decreases rapidly in the neutral aqueous solution as the contact proceeds. After 240 min, the Sr2+ adsorption of C@NaLaTiO reaches equilibrium, and the adsorption capacity of Sr2+ rapidly achieves 164.29 mg·g−1. Kinetic data of the electrochemical experiment can be well fitted by the pseudo-second-order kinetic model with a high correlation coefficient R2 of 0.9899 (Supplementary Fig. 6b and Table 21).
In the pH-dependent experiments, C@NaLaTiO shows satisfactory removal rates on Sr2+ in a wide pH range. The KdSr of C@NaLaTiO reaches 5.90 × 103 mL·g−1 at pH of 7 (Fig. 3b and Supplementary Table 22). Importantly, KdSr can still be maintained above 103 mL·g−1 under acidic conditions (pH < 4). Even at pH of 1, C@NaLaTiO can retain efficient adsorption capacity for Sr2+ (KdSr = 2.23 × 103 mL·g−1).
The Sr2+ removal performance of C@NaLaTiO in solutions containing K+/Na+/Cs+/Sr2+/Ca2+/Mg2+ was studied. C@NaLaTiO exhibits selective Sr²⁺ capture in complex neutral and acidic solutions with the coexistence of K+/Na+/Cs+/Sr2+/Ca2+/Mg2+. Its KdSr reaches 4.23 × 103 mL·g−1 in neutral solutions, whereas the KdM (Mn+ = K+, Na+, Cs+, Ca2+, and Mg2+) are all lower than 2.53 × 103 mL·g−1 (Fig. 3c; KdNa and KdK are shown in Supplementary Tables 23–25). Remarkably, C@NaLaTiO can maintain good Sr²⁺ adsorption capacity under acidic conditions (KdSr = 2.95 × 103mL·g−1 at pH of 3; KdSr = 7.56 × 102 mL·g−1 at pH of 1) (Fig. 3d and Supplementary Fig. 6c, Tables 24 and 25).
To further test the complete electrochemical adsorption and desorption cycle, four cyclic reuse experiments of C@NaLaTiO were carried out. The adsorption process of Sr2+ was studied in solutions containing 67.70 mg·L−1 Sr2+ at –4 V, and the desorption process was carried out in 0.5 mol·L−1 NaCl solution at 4 V (Supplementary Fig. 6d and Table 26). The experimental results show that C@NaLaTiO maintains excellent cycling stability and the adsorption capacity remains above 170.32 mg·g−1.
Capture mechanism
X-ray photoelectron spectroscopy (XPS) spectra of Na2La2Ti3O10 and Na2La2Ti3O10-Sr were tested (Supplementary Fig. 7a and Fig. 4a, b). By comparing the XPS spectra of Na+, it is found that the characteristic peak of Na 1s near 1072 eV is significantly weakened after the adsorption of Sr2+ (Fig. 4a)55. In XPS of Na2La2Ti3O10-Sr, Sr 3d5/2 and Sr 3d3/2 characteristic peaks of Sr 3d appear at 133.5 eV and 135.1 eV, respectively (Fig. 4b)55,56. The above results verify that the Sr2+ adsorption of Na2La2Ti3O10 is due to the ion exchange between Sr2+ and interlayer Na+ in Na2La2Ti3O10.
a XPS spectra of Na 1s for Na2La2Ti3O10 and Na2La2Ti3O10-Sr. b The XPS spectrum of Sr 3d for Na2La2Ti3O10-Sr. c XPS spectra of Na 1s for C@NaLaTiO and C@NaLaTiO-Sr. d XPS spectra of Ti 2p for C@NaLaTiO and C@NaLaTiO-Sr. e The XPS spectrum of Ti 2p for C@NaLaTiO-Sr. f EPR spectra of C@NaLaTiO and C@NaLaTiO-Sr. Source data are provided as a Source Data file.
Moreover, in the above electrochemical adsorption experiments, we found that the adsorption capacity of C@NaLaTiO exceeded the theoretical exchange capacity of Na2La2Ti3O10 (139.66 mg·g−1), and the selectivity under acidic conditions was also improved. Therefore, we speculate that in addition to ion exchange, there are redox reactions in C@NaLaTiO that promote the Sr2+ adsorption. To verify this point, XPS spectra of C@NaLaTiO and C@NaLaTiO-Sr were tested (Supplementary Fig. 7b). After Sr2+ adsorption, the XPS peak intensity of Na 1s decreases, indicating that the content of Na+ reduces (Fig. 4c)57. Peaks of Ti 2p3/2 and Ti 2p1/2 at 459.6 eV and 465.6 eV can be observed in XPS of C@NaLaTiO, showing the presence of Ti4+ (Fig. 4d)58. In addition, after the adsorption of Sr2+, the Ti 2p peak shifts to the low energy compared with C@NaLaTiO, suggesting that C@NaLaTiO-Sr obtains many electrons, and Ti4+ is partially reduced (Fig. 4e)59. The distinct Sr 3d3/2 and Sr 3d5/2 characteristic peaks in the survey spectrum of C@NaLaTiO-Sr are observed at 135.6 and 133.8 eV, respectively (Supplementary Fig. 7c)47. Importantly, compared with Na2La2Ti3O10-Sr, the relative intensity of the characteristic peaks of Sr 3d in the XPS spectrum of C@NaLaTiO-Sr is stronger, which indicates that C@NaLaTiO has high adsorption capacity for Sr2+.
To investigate the structural evolution during Sr2+ adsorption, synchrotron X-ray diffraction (SXRD) patterns of Na2La2Ti3O10, Na2La2Ti3O10-Sr, and C@NaLaTiO-Sr were collected (Supplementary Fig. 8). Rietveld refinement confirmed the phase purity of the pristine Na2La2Ti3O10 (Supplementary Fig. 8a). Upon Sr2+ adsorption, both Na2La2Ti3O10-Sr and C@NaLaTiO-Sr exhibited peak splitting, particularly in the 004 and 006 reflections (Supplementary Fig. 8b). This splitting may indicate a symmetry lowering, structural distortion, or interlayer displacement induced by Sr2+ intercalation. Notably, the relative intensity of the split peaks is higher in C@NaLaTiO-Sr compared with Na2La2Ti3O10-Sr, suggesting the higher Sr2+ uptake capacity of C@NaLaTiO-Sr.
Since the peak shift of Ti 2p is observed in the XPS spectrum of C@NaLaTiO-Sr, the defect structures of C@NaLaTiO and C@NaLaTiO-Sr are further characterized by electron paramagnetic resonance (EPR) (Fig. 4f). Compared with C@NaLaTiO, the EPR spectrum of C@NaLaTiO-Sr shows that the intensity at 2.003 is gradually getting stronger, which is caused by the rise of oxygen vacancy concentration in C@NaLaTiO-Sr33. And in the XPS spectrum of C@NaLaTiO-Sr, the Ti 2p3/2 and Ti 2p1/2 of Ti4+ are located at 458.9 eV and 465.1 eV, respectively, and two relatively weak peaks appear at 457.9 eV and 463.5 eV, corresponding to Ti 2p3/2 and Ti 2p1/2 of Ti3+, respectively60. The reason is the partial reduction of Ti4+ to Ti3+ under the applied voltage induction, which increases the electron cloud density of oxygen atoms and promotes the formation of oxygen vacancies. When Ti3+ is transformed to Ti4+ after applying the opposite voltage, two Ti 2p peaks in C@NaLaTiO-Sr XPS appear at 459.4 eV (Ti 2p3/2) and 465.2 eV (Ti 2p1/2), respectively, and the oxygen electron cloud density decreases (Supplementary Fig. 7d). Consequently, Sr2+ is successfully released (Supplementary Fig. 7e). Thus, the electrochemical adsorption and desorption of Sr2+ is achieved. Therefore, the XPS, SXRD, and EPR confirm that the capture mechanism of C@NaLaTiO for Sr2+ is the ion exchange between Sr2+ and interlayer Na+ in Na2La2Ti3O10 and electrochemical reduction of Ti4+ to Ti3+ under –4 V potential, which generates oxygen vacancies and promotes Sr2+ adsorption. Conversely, when the negative voltage (4 V) is applied to the electrode, the reverse oxidation of Ti3+ to Ti4+ reduces the oxygen vacancies and facilitates the desorption of Sr2+ (Fig. 5a).
a Schematic diagram of the Sr2+ adsorption/desorption process of C@NaLaTiO ([Ti4+O6] octahedra and [Ti3+O6] octahedra are presented in light blue and dark blue colors, respectively). The calculation on binding energies of the [La2Ti3O10]n2n- layer to Sr2+ ions before (b) and after applying voltage (c).
Density functional theory calculations
In order to further elucidate the mechanism of introducing ESIX to increase the adsorption capacity of Sr2+, density functional theory (DFT) calculations on the interaction of the [La2Ti3O10]n2n- layer with Sr2+ ion before and after applying the voltage (–4 V) were performed. The DFT results reveal that the binding energy of the anionic [La2Ti3O10]n2n- layer to Sr2+ decreases from –0.376 eV to –0.998 eV under the action of the electric field (Fig. 5b, c). The binding energy of Sr2+ to the anionic layer [La2Ti3O10]n2n- during electrochemical adsorption is more negative than that of batch adsorption, suggesting a stronger interaction between the anionic layer [La2Ti3O10]n2n- and the Sr2+ ions under the influence of the applied voltage. And it is easier to replace Na+ ions with Sr2+ ions in the interlayer when the voltage is applied. DFT results indicate that through using the method of ESIX, as the voltage is applied, Na2La2Ti3O10 shows a much higher adsorption capacity for Sr2+ from the energy perspective, providing a more efficient way to remove specific ions from solutions.
Discussion
We develop an efficient strategy for Sr2+ capture through an environmentally friendly ESIX method using an electroactive titanate-based perovskite (Na2La2Ti(Ⅳ)3O10) with exceptional acid and irradiation resistance, and thermal stability. It demonstrates an excellent treatment capability for actual acidic 90Sr-liquid-waste generated during industrial production. Comprehensive characterizations (SXRD, EDS, XPS, EPR) reveal that Sr²⁺ capture and release occur through Na+/Sr2+ ion exchange and electrochemically controlled Ti3+/Ti4+ redox reactions, enabling efficient electrochemical adsorption and desorption. DFT calculations further confirm that the introduction of ESIX significantly enhances the Sr2+ adsorption capacity of Na2La2Ti3O10. By synergistically combining ion exchange with electrochemical adsorption, the current strategy provides a sustainable approach to Sr2+ removal and lays the foundation for the development of more efficient materials for radiostrontium treatment. Ultimately, this work expands the potential of electroactive titanate-based perovskites for Sr²⁺ capture via ESIX, supporting the safe and sustainable advancement of the nuclear industry.
Methods
Materials
Na2CO3 (AR, Shanghai Hongguang Chemical Plant Co., Ltd), TiO2 (RG, Acros), La2O3 (99.99%, Sinopharm Chemical Reagent Co., Ltd), HNO3 (65~68%, Sinopharm Chemical Reagent Co., Ltd), NaOH (AR, Tianjin Guangfu Reagent Co., Ltd), CsCl (99.99%, Shanghai Longjin Metallic Material Co., Ltd), KCl (AR, Tianjin Fuchen Chemical Reagent Co., Ltd), NaCl (AR, Sinopharm Chemical Reagent Co., Ltd), CaCl2·2H2O (74%, Shanghai Sili Chemical Plant), MgCl2 (AR, Adamas Reagent Co., Ltd), SrCl2·6H2O (AR, Tianjin Guangfu Reagent Co., Ltd), EuCl3·6H2O (AR, RUIKE State Engineering Research Center of Rare Earth Metallurgy and Functional Materials Co., Ltd), ZrOCl2·8H2O (98%, Adamas Reagent Co., Ltd), carbon black (CP, Qingdao Chenyang Graphite Co., Ltd), polyvinylidene fluoride (PVDF) (99%, Shanghai McLean Biochemical Technology Co., Ltd), N-methylpyrrolidone (NMP) (AR, Sinopharm Chemical Reagent Co., Ltd), carbon cloth (Suzhou Zhengtairong New Material Co., Ltd). All the chemicals were used without further purification. For economic and safety reasons, we used non-radioactive strontium instead of radionuclide strontium in the batch adsorption and electrochemical adsorption experiments. Radionuclide strontium was only used in the actual treatment of acidic 90Sr-liquid-waste generated from the production of radioactive sources at CNNC HTA Co., Ltd.
Synthesis of Na2La2Ti3O10 and C@NaLaTiO
According to the previously reported synthesis method, Na2La2Ti3O10 was prepared by the solid phase reaction34. Na2CO3 (2.8 mmol, 0.2968 g), TiO2 (6 mmol, 0.4792 g), and La2O3 (2 mmol, 0.6516 g) were mixed stoichiometrically and ground well. The mixture was placed in a 20 mL round-bottomed alumina crucible and kept at 900 °C for 24 h, followed by cooling slowly for 48 h to RT. Products were rinsed several times with deionized water and ethanol, and they were separated by filtration and finally dried in an oven at 80 °C overnight to obtain white powder Na2La2Ti3O10 as pure phase. About 1.1936 g of Na2La2Ti3O10 could be obtained with a yield of 95.12% (based on Ti), which is stable in water and air. The synthesized Na2La2Ti3O10, carbon black, and PVDF were dispersed into NMP at a mass ratio of 8:1:1 at RT and dried in an oven at 60 °C overnight. The prepared slurry was coated on the carbon cloth (1.0 × 1.0 cm2) as the working electrode (C@NaLaTiO).
Characterizations
PXRD patterns were obtained at RT using a Miniflex II diffractometer with CuKα (λ = 1.54178 Å) at 30 kV and 15 mA within the angular range of 2θ = 5–65°. Elemental analysis and mapping of the samples were performed using a JEOL JSM-6700F SEM and energy-dispersive X-ray spectroscopy (EDS). Solid-state optical diffuse reflectance spectra were recorded on a Shimadzu 2600 UV/vis spectrometer at RT in the range of 800–200 nm. TGA was performed on a NETZSCH STA 449F3 DTA-TG analyzer under a N2 atmosphere from RT to 1200 °C with a ramp rate of 10 °C·min−1. XPS was performed on the ThermoFisher ESCALAB 250Xi XPS spectrometer system, with all binding energies referenced to the C1s peak of adventitious carbon at 284.8 eV. EPR was recorded using an EPR spectrometer (Elexsys E500, Bruker, Germany). Metal ion concentrations in solutions were determined by inductively coupled plasma mass spectrometry (ICP-MS) or inductively coupled plasma optical emission spectrometry (ICP-OES), utilizing an XSeries II and a Thermo 7400 instrument, respectively. Powder SXRD data were collected at SPring-8’s BL02B2 beamline (Japan) using high-energy radiation (λ = 0.42 Å). The samples were encapsulated in borosilicate capillaries before measurement. Structural refinement was subsequently performed using GSAS software. The pH of all solutions was measured with Shanghai Lexmark Electronics 201F. The supernatant was separated using the G16-WS centrifuge. Electrochemical tests were conducted using an electrochemical workstation (CHI660E, CH Instruments, China). Samples were irradiated with γ-rays at a total dose of 100 kGy (1.2 kGy·h−1 for 89 h) and 200 kGy (1.2 kGy·h−1 for 175 h) using a 60Co irradiation source (2 million curies) provided by the Detection Center of Suzhou CNNC Huadong Radiation Co., Ltd, China.
Density functional theory calculations
All the calculations are performed in the framework of the DFT with the projector augmented plane-wave method, as implemented in the Vienna ab initio simulation package (VASP, version number 5.4.4)61. The generalized gradient approximation proposed by Perdew, Burke, and Ernzerhof is selected for the exchange-correlation potential62. The long-range van der Waals interaction is described by the DFT-D3 approach63. The cut-off energy for the plane wave is set to 500 eV. The energy criterion is set to 10−6 eV in the iterative solution of the Kohn–Sham equation. The K-mesh resolved in real space is 0.04 2π·Å−1. All the structures are relaxed until the residual forces on the atoms have declined to less than 0.02 eV·Å−1.
Data availability
The ion exchange data generated in this study are provided in the Supplementary Information/Source Data file. Source data are provided in this paper. Additional information can be obtained from the corresponding author upon request. Source data are provided with this paper.
References
Zhang, F. L., Wang, J. L., Bi, Q. Q. & Du, J. Z. 90Sr in seawater of the East China Sea: Inventory, new potential source, and environmental implications. Sci. Total Environ. 764, 144266 (2021).
Inan, S. Inorganic ion exchangers for strontium removal from radioactive waste: a review. J. Radioanal. Nucl. Chem. 331, 1137–1154 (2022).
Tang, J. H., Sun, H. Y., Ma, W., Feng, M. L. & Huang, X. Y. Recent progress in developing crystalline ion exchange materials for the removal of radioactive ions. Chin. J. Struct. Chem. 39, 2157–2171 (2020).
Zhang, J. R. et al. Distinctive two-step intercalation of Sr2+ into a coordination polymer with record high 90Sr uptake capabilities. Chem 5, 977–994 (2019).
Wang, K. T. et al. One-pot preparation of NaA Zeolite microspheres for highly selective and continuous removal of Sr(II) from aqueous solution. ACS Sustain. Chem. Eng. 7, 2459–2470 (2019).
Casacuberta, N., Masqué, P., Garcia-Orellana, J., Garcia-Tenorio, R. & Buesseler, K. O. 90Sr and 89Sr in seawater off Japan as a consequence of the Fukushima Dai-ichi nuclear accident. Biogeosciences 10, 3649–3659 (2013).
Rahman, R. O. A., Ibrahium, H. A. & Hung, Y. T. Liquid radioactive wastes treatment: a review. Water 3, 551–565 (2011).
Ding, D. H., Zhang, Z. Y., Lei, Z. F., Yang, Y. N. & Cai, T. M. Remediation of radiocesium-contaminated liquid waste, soil, and ash: a mini review since the Fukushima Daiichi Nuclear Power Plant accident. Environ. Sci. Pollut. Res. 23, 2249–2263 (2016).
Bian, Y. Y. et al. Highly selective separation of Cs+ from brines using an electroactive layered composite of vanadosilicate. ACS Sustain. Chem. Eng. 11, 10884–10894 (2023).
Wang, B. Y. et al. Electrochemical adsorption of cesium using a nickel hexacyanoferrate-doped porous carbon electrode. J. Radioanal. Nucl. Chem. 332, 2589–2600 (2023).
Liao, S. L. et al. Simultaneous separation of iodide and cesium ions from dilute wastewater based on PPy/PTCF and NiHCF/PTCF electrodes using electrochemically switched ion exchange method. Sep Purif. Technol. 139, 63–69 (2015).
Su, J. Y. et al. The characterization and application of prussian blue at graphene coated carbon fibers in a separated adsorption and electrically switched ion exchange desorption processes of cesium. Electrochim. Acta 230, 399–406 (2017).
Sun, B. et al. Separation of low concentration of cesium ion from wastewater by electrochemically switched ion exchange method: Experimental adsorption kinetics analysis. J. Hazard Mater. 233, 177–183 (2012).
Bian, Y. Y. et al. Highly selective capture of cesium using an electrochemically renewable molybdenum-based crystalline adsorbent. J. Environ. Chem. Eng. 12, 111931 (2024).
Liu, X. J., Wu, J. L. & Wang, J. L. Electro-adsorption of Sr(II) from aqueous solution by activated carbon cloth/nickel hexacyanoferrate composite electrode through capacitive deionization. J. Clean. Prod. 380, 135075 (2022).
Choi, H., Do, K., Park, S., Yu, J. S. & Ko, J. Efficient hole transporting materials with two or four N, N-Di(4-methoxyphenyl)aminophenyl arms on an ethene unit for perovskite solar cells. Chem. Eur. J. 21, 15919–15923 (2015).
Huang, L. et al. Research progresses on the application of perovskite in adsorption and photocatalytic removal of water pollutants. J. Hazard Mater. 442, 130024 (2023).
Wang, W., Tadé, M. O. & Shao, Z. P. Research progress of perovskite materials in photocatalysis- and photovoltaics-related energy conversion and environmental treatment. Chem. Soc. Rev. 44, 5371–5408 (2015).
Hsu, Y. C., Chang, S. H., Chung, W. C. & Chang, M. B. Photocatalytic removal of trichloroethylene from water with LaFeO3. Environ. Sci. Pollut. Res. 26, 26276–26285 (2019).
Zhang, J. B. et al. Nitrogen-doped perovskite as a bifunctional cathode catalyst for rechargeable lithium oxygen batteries. ACS Appl. Mater. Interfaces 10, 5543–5550 (2018).
Hu, Y. Z. et al. Superior sorption capacities of Ca-Ti and Ca-Al bimetallic oxides for U(VI) from aqueous solutions. Chem. Eng. J. 316, 419–428 (2017).
Lu, S. H. et al. Influence of carbonate on sequestration of U(VI) on perovskite. J. Hazard Mater. 364, 100–107 (2019).
Chitrakar, R. et al. HCa2Nb3O10•1.5H2O as an ion exchanger for NH4+ ion removal. Ind. Eng. Chem. Res. 47, 176–179 (2008).
Li, X. T., Hoffman, J. M. & Kanatzidis, M. G. The 2D halide perovskite rulebook: How the spacer influences everything from the structure to optoelectronic device efficiency. Chem. Rev. 121, 2230–2291 (2021).
Chitrakar, R., Makita, Y. & Sonoda, A. Cesium ion decontamination by layered perovskite HCa2Nb3O10 center dot 1.5H2O. Bull. Chem. Soc. Jpn 86, 1419–1425 (2013).
Sun, H. Y. et al. Boosting selective Cs+ uptake through the modulation of stacking modes in layered niobate-based perovskites. Nat. Commun. 15, 8681 (2024).
Gopalakrishnan, J. & Bhat, V. A2Ln2Ti3O10 (A = potassium or rubidium; Ln = lanthanum or rare earth): a new series of layered perovskites exhibiting ion exchange. Inorg. Chem. 26, 4299–4301 (1987).
Nozaki, R. et al. Vibrational study of layered perovskites M2La2Ti3O10 (M = Li, Na, K, Rb): Raman spectra and normal mode analysis. J. Phys. Chem. B 105, 7950–7953 (2001).
Toda, K., Kameo, Y., Kurita, S. & Sato, M. Crystal structure determination and ionic conductivity of layered perovskite compounds NaLnTiO4 (Ln=rare earth). J. Alloy. Compd. 234, 19–25 (1996).
Kawashima, K. et al. Understanding the effect of partial N3−-to-O2- substitution and H+-to-K+ exchange on photocatalytic water reduction activity of Ruddlesden-Popper layered perovskite KLaTiO4. Mol. Catal. 432, 250–258 (2017).
Li, M. C. et al. Enhanced adsorption of cesium ions by electrochemically switched ion exchange method: based on surface-synthetic Na2Ti3O7 nanotubes. Colloids Surf. A 579, 123712 (2019).
McCafferty, E. & Wightman, J. P. Determination of the concentration of surface hydroxyl groups on metal oxide films by a quantitative XPS method. Surf. Interface Anal. 26, 549–564 (1998).
Meng, W. J., Dang, Z. Z., Li, D. S. & Jiang, L. Long-cycle-life sodium-ion battery fabrication via a unique chemical bonding interface mechanism. Adv. Mater. 35, 2301376 (2023).
Toda, K., Kameo, Y., Fujimoto, M. & Sato, M. Crystal-structure and ionic-conductivity of a layered perovskite, Na2La2Ti3O10. J. Ceram. Soc. Jpn 102, 737–741 (1994).
Duong D. D. Adsorption Analysis: Equilibria and Kinetics (Journal of the Ceramic Society of Japan, 1998).
Chen, Z. et al. Selective removal of Sr2+ by cation exchange using silica-based titanate adsorbents. J. Solid State Chem. 312, 123247 (2022).
Li, C. M. et al. Studies on the separation and in-situ sintering solidification of strontium by a highly-efficient titanate-based adsorbent. Microporous Mesoporous Mater. 288, 109607 (2019).
Kasap, S., Piskin, S. & Tel, H. Titanate nanotubes: preparation, characterization and application in adsorption of strontium ion from aqueous solution. Radiochim. Acta 100, 925–929 (2012).
Yi, R., Ye, G. & Chen, J. Synthesis of core-shell magnetic titanate nanofibers composite for the efficient removal of Sr2+. RSC Adv. 9, 27242–27249 (2019).
Igarashi, S., Sato, S., Takashima, T. & Ogawa, M. Preparation of finite particles of layered niobate (KCa2Nb3O10) for improved materials performance. Ind. Eng. Chem. Res. 52, 3329–3333 (2013).
Yavari, R., Khanchi, A. R., Maragheh, M. G. & Waqif-Husain, S. Sorption of radionuclides on thorium tungstophosphate: a new inorganic ion-exchanger. J. Radioanal. Nucl. Chem. 267, 685–690 (2006).
Zhang, L. et al. Strontium(II) adsorption on Sb(III)/Sb2O5. Chem. Eng. J. 267, 245–252 (2015).
Li, X. L. et al. Adsorption kinetic, isotherm and thermodynamic studies of Sr2+ onto hexagonal tungsten oxide. J. Radioanal. Nucl. Chem. 298, 47–53 (2013).
Tai, B. et al. Flexible interdigitated layered framework with multiple accessible active sites for high-performance CH3I capture. Sci. China Chem. 67, 1569–1577 (2024).
Xie, Y. H. et al. Recent progress of radionuclides separation by porous materials. Sci. China Chem. 67, 3515–3577 (2024).
Ho, Y. S. Review of second-order models for adsorption systems. J. Hazard Mater. 136, 681–689 (2006).
Guo, Y. L. et al. Efficient removal of Sr2+ ions by a one-dimensional potassium phosphatoantimonate. Chem. Eng. J. 460, 141697 (2023).
Lv, C. et al. Efficient uranium adsorption of low-cost and eco-friendly MQ2-collagen fibers (M = Mo, W; Q = S, Se) composite materials prepared by co-ball milling. Natl Sci. Open 4, 20240029 (2024).
Tang, J. H., Feng, M. L.& Huang, X. Y. Metal chalcogenides as ion-exchange materials for the efficient removal of key radionuclides: a review. Fundam. Res. https://doi.org/10.1016/j.fmre.2023.1010.1022 (2023).
Zhang, X. et al. Emerging MOFs, COFs, and their derivatives for energy and environmental applications. Innovation 6, 100778 (2025).
Zheng, B. T. et al. Inverse vulcanization-induced self-assembly of polysulfide into responsive micelles and their templated hydrogel adsorbent for Sr2+ removal. Langmuir 41, 11614–11629 (2025).
Liu, Y. X. et al. Site differentiation strategy for selective strontium uptake and elution within an all-inorganic polyoxoniobate framework. Nat. Commun. 15, 8896 (2024).
Li, L. et al. A negatively-charged supramolecular trap for precisely catching strontium ion. Nat. Commun. 16, 2606 (2025).
Zheng, B. T. et al. Thiol-rich and ion-imprinted alginate hydrogel as a highly adsorptive and recyclable filtration membrane for rapid and selective Sr(II) removal. Chem. Eng. J. 465, 142752 (2023).
Moulder, J. F., Chastain, J. & King, R. C. Handbook of X-ray photoelectron spectroscopy: a reference book of standard spectra for identification and interpretation of XPS data. Chem. Phys. Lett. 220, 7–10 (1992).
Yuan, Z. et al. Synthesis and properties of Sr2+ doping alpha-tricalcium phosphate at low temperature. J. Appl Biomater. Funct. Mater. 19, 1–8 (2021).
Zhao, Z. W. et al. XPS and FTIR studies of sodium arsenate vitrification by cullet. J. Non-Cryst. Solids 502, 254–254 (2018).
Ouyang, J. L., Sun, X. T., Chen, X. S., Chen, J. Y. & Zhuang, X. M. Preparation of layered bioceramic hydroxyapatite/sodium titanate coatings on titanium substrates using a hybrid technique of alkali-heat treatment and electrochemical deposition. J. Mater. 49, 1882–1892 (2014).
Ni, J. F. et al. Superior sodium storage in Na2Ti3O7 nanotube arrays through surface engineering. Adv. Energy Mater. 6, 1502568 (2016).
Fu, S. D., Ni, J. F., Xu, Y., Zhang, Q. & Li, L. Hydrogenation driven conductive Na2Ti3O7 nanoarrays as robust binder-free anodes for sodium-ion batteries. Nano Lett. 16, 4544–4551 (2016).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Acknowledgements
We greatly thank the financial support from the National Natural Science Foundation of China, the Natural Science Foundation of Fujian Province, and the Strategic Priority Research Program of the Chinese Academy of Sciences. M.F. was supported by the National Natural Science Foundation of China (Nos. 22325605 and U21A20296) and the Strategic Priority Research Program of the Chinese Academy of Sciences (No. XDB1170000). Z.L. was supported by the Major Program of the National Natural Science Foundation of China (Nos. 22494680 and 22494684) and the Key Program of the National Natural Science Foundation of China (No. 22336006). H.S. was supported by the National Natural Science Foundation of China (No. 22406185) and the Natural Science Foundation of Fujian Province (No. 2024J08105).
Author information
Authors and Affiliations
Contributions
The manuscript was written through the contributions of all authors. Z.C., X.H., M.F., and Z.L. conceived the project. Z.C. designed and performed most of the experiments and analyzed the data. F.D., S.J., H.S., S.L., and J.T. offered help in the partial experiments. Z.C. prepared the manuscript. M.F., Z.L., F.D., and X.H. revised the manuscript. M.F. and Z.L. supervised the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Yubing Sun, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, ZH., Ding, FH., Jia, SQ. et al. Enhancing radiostrontium uptake by a layered titanate perovskite via sustainable electrochemically switched ion exchange. Nat Commun 16, 9452 (2025). https://doi.org/10.1038/s41467-025-64537-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-64537-1