Abstract
Giant negative thermal expansion (NTE), defined by volumetric expansion αV <ca. −50 × 10−6 K−1 (volume contraction (ΔV/V) < − 0.5% within ~100 K), is rarely observed at high temperatures. Here, we report a giant NTE persisting above 1000 K (ΔV/V = − 1.7 %, 900–1100 K) in stoichiometric PrMnO3 (PMON) with a peak coefficient αV = − 114 × 10−6 K−1 around 1000 K. Contrastingly, oxygen-rich PrMnO3+x (PMOA) exhibits only positive thermal expansion. The origin of NTE was uncovered via synchrotron X-ray total scattering, Cs-corrected STEM, and DFT calculations. Intriguingly, PMON uniquely hosts a local symmetry breaking featured by a 3D cross-arranged network of elongated Mn-O bonds, different from the 2D planar configurations in PMOA. By correlating atomic-scale symmetry breaking and thermally activated orbital reconfiguration to macroscopic thermal responses, we establish an unconventional paradigm for engineering giant NTE at elevated temperatures.
Similar content being viewed by others
Introduction
Materials with colossal negative thermal expansion (NTE) at elevated temperatures show exceptional promise in mitigating thermomechanical instability faced by high-temperature components, including engine turbine blades, thermal barrier coatings, and thermal management systems1,2. Nevertheless, materials demonstrating substantial lattice contraction with giant NTE coefficients remain scarce, particularly in high-temperature regimes (>500 K). NTE phenomenon arises from diverse physical mechanisms, such as geometric flexibility in framework compounds3,4,5, ferroelectric and magnetic phase transition6,7, charge (or orbital) ordering transition8,9,10,11, etc. These mechanisms counteract the positive thermal expansion (PTE) induced by high-frequency phonon modes12. As temperature increases, high-frequency phonon overwhelmingly prevails, resulting in severe constraints: most giant NTEs are confined to modest temperature ranges (typically <500 K) with limited magnitude of volume contraction.
The pursuit of giant NTE materials and their governing mechanisms remains an active frontier13,14,15,16,17. Electron-driven mechanisms, particularly those involving charge transfer, orbital reconfiguration, and ferroelectric/magnetic phase transitions, have emerged as critical pathways for achieving pronounced NTE. For instance, Bi0.95La0.05NiO3 demonstrates a − 2.9 % lattice volume shrinkage (ΔV/V) between 300–370 K with αV = − 413 × 10−6 K−1 via charge transfer8. (Sm, Y)S single crystal exhibits αV = − 195 × 10−6 K−1 (100–315 K)18. Pb0.7Bi0.3VO3 undergoes a − 7.9 % volume reduction (550–800 K) through ferroelectric phase transitions19. Orbital ordering (OO) induced NTE in Ca2RuO4-x yields a − 1 % volume reduction with αV = − 48 × 10−6 K−1 (135–345 K)9. Additional materials like Mn(Co, Cr)Ge (αV = − 155 × 10−6 K−1, 122–332 K)20, (Ni, Fe)S (αV = − 293 × 10−6 K−1, 240–290 K)21, La(Fe, Co, Si)13 (αV = − 78 × 10−6 K−1, 240–350 K)7 further underscore the prevalence of electron-driven NTE. By far, electron-driven type NTEs predominantly operate below 500 K, necessitating strategies to extend their operational windows to higher temperatures22,23. In contrast, low-frequency phonon driven NTE materials (e.g., framework compounds) exhibit a relatively wide temperature window that may eventually expand into the high-temperature range24, but suffer from diminished contraction magnitudes at elevated temperatures: ScF3 displays αV = − 14.1 × 10−6 K−1 (10–1100 K, ΔV/V ≈ − 1.5 %), yet its contraction attenuates to −0.05 % near 1000 K (αV = − 2.1 × 10−6 K−1, 760–1000 K)4. NaB(CN)4 shows αV degradation from −92 × 10−6 K−1 (100–200 K) to −36 × 10−6 K−1 at ~650 K25. Notably, as far as we know, no material has demonstrated giant NTE (αV < − 100 × 10−6 K−1) beyond 1000 K, highlighting a critical gap in high-temperature NTE engineering.
Perovskite-type RMnO3 (R = rare earth elements), characterized by the Jahn-Teller (JT) distortion of Mn3+ cations, have attracted multidisciplinary interest in catalysis, energy conversion/storage, magnetic recording, and electromagnetic wave absorption due to their unique electronic and magnetic functionalities26,27,28,29. Within this family, LaMnO3—the parent compound exhibiting colossal magnetoresistance—has been extensively studied through chemical doping at R/Mn sites and oxygen stoichiometry modulation30. These investigations focus on elucidating the interplay between crystal structure evolution, JT transitions, electronic configurations, and OO, aiming to decode the origins of its extraordinary properties31,32,33. Specifically, orbital order-disorder transitions (OO to OD) and localized JT distortions in LaMnO3 give clues to the origin of its −0.36 % volume collapse between 720–750 K34. Similarly, PrMnO3 demonstrates an anomalous −0.1 % lattice contraction near 200–300 K35. Notably, neutron diffraction investigation on PrMnO3 observed thermal shrinkage aligns with our NTE framework36, while established phase diagrams inform oxygen stoichiometry design strategies37. These observations highlight the potential of RMnO3 perovskites as candidates for high-T giant NTE.
In this work, we report a breakthrough in achieving giant NTE above 1000 K through oxygen stoichiometry engineering in PrMnO3. Specifically, stoichiometric PrMnO3 (PMON) exhibits a colossal volume contraction exceeding 1000 K (ΔV/V = − 1.7 %, 900–1100 K) with a peak volumetric thermal expansion coefficient αV = − 114 × 10−6 K−1 around 1000 K. In contrast, oxygen-excess PrMnO3+x (PMOA) displays conventional PTE. The electronic structure analysis was performed via X-ray absorption spectroscopy (Mn K-, O K-, and Mn L-edges). Temperature-dependent synchrotron X-ray diffraction (SXRD) unveiled dynamic MnO6 octahedral geometry changes and JT distortion evolution. Pair distribution function analysis (PDF) and aberration-corrected scanning transmission electron microscopy (STEM) revealed a unique local structural distortion in PMON, characterized by three-dimensional (3D) cross-arrangement of elongated Mn–O bonds and zigzag atomic configurations. Conversely, PMOA adopts a two-dimensional planar distribution of elongated Mn–O bond. The first-principles density functional theory (DFT) calculations give electronic orbital patterns and JT distortion in degree of PMON-derived structural models, revealing condition-dependent orbital evolution pathways. This study establishes the highest-temperature giant NTE reported to date and pioneers a strategic pathway for tailoring anomalous thermal expansion through manipulating oxygen stoichiometry.
Results and discussion
Electronic structure analysis and NTE
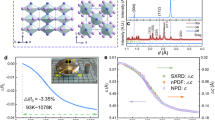
PrMnO3 with excess lattice oxygen (denoted as PMOA) was synthesized via a solid-state reaction under the ambient atmosphere. Stoichiometric PrMnO3 (designated as PMON) was obtained through citrate precursor decomposition followed by nitrogen-atmosphere annealing to remove oxygen excess. Figure 1a displays synchrotron X-ray diffraction (SXRD) patterns of PMOA and PMON at 800 K, with characteristic reflections indexed to the orthorhombic Pnma structure.
a SXRD patterns of PMOA and PMON at 800 K with indexed diffraction peaks. b The O K-edge, and (c) Mn L-edge XAS spectrum of PMOA, PMON, reference samples (Mn2O3, MnO2). d Contour plots of typical diffraction peaks (110)/(002) and (111) of PMOA and PMON. e The relative lattice volume change ΔV/V, along with error bars from the standard error of lattice volumes. The green/orange colored background represents a gradual change in temperature from low to high. f The relative lattice volume shrinkage −ΔV/V versus the maximum working temperature Tmax of giant NTE materials induced by charge transfer (CT), magnetic order (MO), phonons (PH), and orbital ordering (OO). Detailed giant NTE data are listed in Table S1, Supplementary Information. Source data are provided as a Source Data file.
To probe the electronic structures of PMON and PMOA, we present O K-edge and Mn L-edge XAS spectra for both compounds alongside reference standards (Mn2O3 and MnO2) in Fig. 1b, c. Three characteristic features (denoted A, B, C) emerge in PMON and PMOA, ascribed to hybridized states of O 2p-Mn 3 d, O 2p-Pr 5 d, and O 2p-Mn 4sp respectively38. The O K-edge spectra of both materials exhibit close resemblance to Mn2O3 but distinct divergence from MnO2. Critically, the pre-edge feature (A) below 535 eV corresponds to unoccupied O 2p-Mn 3 d hybridized states39, whose intensity correlates directly with Mn valence—a relationship established in analogous systems like (La, Sr)MnO3 where enhanced pre-edge intensity accompanies hole doping and elevated Mn oxidation states40. Notably, the markedly intensified pre-edge feature in PMOA (indicated by the black arrow, Fig. 1b) relative to PMON provides direct spectroscopic evidence of a higher Mn valence in PMOA.
The Mn L-edge XAS spectra exhibit characteristic L3 (2p3/2) and L2 (2p1/2) peaks arising from spin-orbit coupling, as shown in Fig. 1c. PMON displays peak profiles and positions closely resembling Mn2O3 (Mn3+), particularly evident in the identical L3-edge maximum (black bar). While PMOA maintains this L3-edge maximum, a distinct high-energy shoulder (violet dotted line) aligns with the L3-edge peak of MnO2, suggesting an average Mn valence exceeding +3. The dominant Mn3+ valence state, admixed with minor Mn4+, aligns with X-ray photoelectron spectroscopy (XPS) data (Fig. S2). Further supporting this, the L2-edge maximum for PMON occurs at 652.8 eV–identical to Mn2O3–whereas PMOA shows a slight shift to 652.9 eV. Both values remain significantly lower than MnO2’s 653.4 eV. This systematic L2-edge blueshift is analogous to observations in La1-xSrxMnO3 systems, where valence increases from +3 (652.2 eV) to +3.9 (653.0 eV)40. Collectively, these features indicate a + 3 valence state for PMON, analogous to Mn2O3, while PMOA exhibits a slightly elevated valence consistent with charge compensation via oxygen incorporation. Moreover, we systematically analyzed the first derivatives of the Mn K-edge X-ray absorption near-edge structure spectra (Fig. S1b), yielding estimated valences of +3 for PMON and +3.16 for PMOA. These values align with XPS and Raman spectroscopy analyses (Figs. S2, 3). Charge compensation considerations further indicate an oxygen stoichiometry of PrMnO3.08 for PMOA.
Temperature-dependent SXRD profiles of PMOA and PMON were analyzed via Rietveld refinement using the orthorhombic Pnma structural model (Fig. S2). Figure 1d illustrates the thermal evolution of characteristic diffraction peaks. PMON exhibits merging of (110) and (002) reflections near 1000 K, corresponding to an orthorhombic-to-orthorhombic structural phase transition. Distinct thermal responses are observed: PMOA peaks systematically shift to lower 2θ angles with heating, whereas PMON peaks above 1000 K migrate toward higher angles, reflecting divergent thermal expansion properties in PMOA and PMON. Indeed, quantitative analysis (Fig. 1e) demonstrates conventional PTE in PMOA(blue hexagon), with an average volumetric coefficient αᴠ = 32 × 10−6 K−1 (800–1100 K). Lattice parameters a, b, and c monotonically increase with temperature (Fig. S3a–c). In stark contrast, PMON displays giant NTE, achieving the peak αᴠ = −114 × 10−6 K−1 around 1000 K. A − 1.7 % volume contraction occurs between 900–1100 K (average αᴠ = −85 × 10−6 K−1), surpassing the reported colossal NTE systems mediated by charge transfer (CT), magnetic ordering (MO), phonon (PH), or orbital-ordering (OO) mechanisms (Fig. 1f). Notably, PMON demonstrates operational stability for colossal NTE, maintaining functionality above 1000 K—the highest temperature window reported to date.
The reversibility and reproducibility of the giant NTE and PTE in PMON and PMOA are rigorously confirmed by SXRD and laboratory-based XRD measurements, as illustrated in Fig. S6–9. Moreover, the Mn L-edge XAS spectra of PMON and PMOA, which were after high-temperature X-ray experiment, retain spectral features identical to the reference Mn2O3, as detailed in Fig. S10. This spectral invariance effectively excludes manganese oxidation as a contributory mechanism for the observed lattice volume contraction. The observed difference in peak maxima of the Mn L edge of Mn2O3 between Figs. 1c, S10 aligns with well-established knowledge of systematic energy shifts in XAS spectra across beamlines. A detailed discussion of this phenomenon is provided in the text immediately following Fig. S10. Moreover, multiple batches of samples were synthesized, with XRD results demonstrating consistent lattice constants (Fig. S12, Table S2). We further characterized the magnetism, electrical resistance, and thermal conductivity of PMON and PMOA, as presented in Figs. S13–15.
Structural evolution and JT distortion
As mentioned above, the progressive evolution of SXRD patterns (Fig. 1e, S2a–d) confirms PMON undergoes a structural phase transition near 1000 K, characterized by the coexistence of two orthorhombic Pnma phases (Phase1 and Phase2) above this temperature. Lattice contraction in PMON arises primarily from compression along the b-axis, while a- and b-dimensions converge in Phase2 to form a metrically quasi-tetragonal structure (Fig. S5d, e).
First, we focus on the JT distortion of MnO6 octahedra. Figure 2a illustrates the MnO6 octahedral substructure in PMON, annotated with critical bond lengths (longest dl, intermediate dm, shortest ds) and Mn-O-Mn bond angles (γ, β). Both PMOA and PMON exhibit significant dl–ds disparities below 900 K (Fig. 2b, c), indicative of strong JT distortions. At 800 K, PMOA demonstrates dl = 2.14 Å / ds = 1.89 Å compared to PMON’s dl = 2.20 Å / ds = 1.91 Å, with PMON displaying greater JT distortion magnitude in this regime. Quantitative JT distortion analysis (δ = [dl – ds]/[2(dl + ds)]) reveals distinct thermal responses (Fig. 2f). PMOA maintains persistent distortion (δ ≈ 3.1%) up to 1100 K, while PMON shows dramatic δ reduction from 3.5 % at 900 K (Phase 1) to 1.4 % at 920 K, stabilizing at ca. 1.1 % in Phase 2 (920–1100 K).
a The substructure of an orthorhombic PMON with the longest, mediate, and shortest Mn-O bonds as indicated by dl, dm, and ds, respectively. The Mn-O-Mn bond angles are marked by γ and β. b The Mn-O-Mn bond lengths dl, dm, and ds of PMOA. c The Mn-O-Mn bond lengths of PMON (Phase1 and Phase2). d The Mn-O-Mn bond angles β. e The Mn-O-Mn bond angles γ. f JT distortion δ. Phase 1 of PMON experienced a jump in β and γ and a sudden drop in δ beyond 900 K. Mn atoms (large size) in purple, O atoms (small size) in blue/orange. The green/orange colored background represents a gradual change in temperature from low to high. Source data are provided as a Source Data file.
Next, we focus on the relative distortion between the neighboring MnO6 octahedron, assessed by the bond angles γ and β. Bond angle evolution provides further mechanistic insight: PMOA retains constant γ and β values across temperatures (Fig. 2d), reflecting rigid octahedral connectivity. Conversely, PMON exhibits abrupt increases in γ and β at 920 K (Fig. 2e), signaling reduced octahedral tilting. This contrast suggests PMOA possesses “frozen” MnO6 octahedra, while PMON enables enhanced structural flexibility. Note that lattice contraction accompanied by increased octahedral rotational degree of freedom was previously observed in LaMnO331. The PMON volume contraction initiating at 900 K correlates with bond length convergence and planar octahedral alignment, marking an OO to OD transition between 900–920 K. In contrast, PMOA maintains strong JT distortions and preserved OO state across the whole temperature range.
Local structure investigation
In addition to the average structural evolution through SXRD data refinement, we notice that local structural features, distinct from the long-range periodic atomic arrangement, critically influence local electron and orbital configurations5,13,32,41,42. To elucidate these structural disparities between PMOA and PMON, synchrotron X-ray pair distribution function (PDF) analysis was implemented.
Initially, the average orthorhombic structural model (Pnma symmetry with constrained unit cell parameters and atomic positions) derived from SXRD Rietveld refinement was applied to fit the short-range PDF data (Fig. 3a, upper panel). The relatively poor fit quality (Rw = 28 %) revealed significant local structural deviations from the average periodic structure, as evidenced by mismatches between experimental and calculated PDF profiles in the 1.5–6 Å r-range (indicated by red arrows). Subsequently, PDF refinement with relaxed Pnma symmetry constraints (unrestricted unit cell parameters and atomic positions) substantially improved the fit (Rw = 16.3 %, Fig. 3a middle panel), yielding a 2D model characterized by ab-plane confinement of the longest Mn-O bonds (Fig. 3d).
a Fit of three models to the PDF data by using (i) average structure model extracted from the SXRD Rietveld refinement (Pnma symmetry, green line), (ii) 2D model (Pnma symmetry, yellow line, discussed in the text), and (iii) 3D model (P21/c symmetry, red line). The red arrows highlight the discrepancy between the experimental and calculated PDF(ΔG(r)). Rw is the goodness of fit. b Substructure of 3D model. The longest Mn–O bonds in different MnO6 octahedra (Mn atoms in Royal and purple, O atoms in orange) are shown by arrows. c Overall viewing of the longest Mn-O bond orientation in the 3D model. d The longest Mn-O bond orientation in the 2D model. Only Mn networks are presented. The longest Mn–O bonds of the 3D model are distributed in the horizontal and vertical planes. In contrast, the 2D model has the longest Mn-O bonds, approximately confined within the horizontal planes, i.e., ab planes. Mn atoms in royal/purple, O atoms in orange. Source data are provided as a Source Data file.
Ultimately, a monoclinic P21/c symmetry model demonstrated superior PDF fitting (Rw = 10.5 %, Fig. 3a lower panel), revealing three-dimensional orientation of the longest Mn-O bonds rather than the planar restriction observed in the 2D model (as discussed later in the text). This energetically favorable 3D configuration exhibited enhanced agreement with experimental data, corroborated by minimized residual differences in ΔG(r) profiles (purple curve, lower panel). These findings indicate a short-range symmetry breakdown in PMON from orthorhombic to monoclinic structure, highlighted by 3D-ordered Mn-O bond elongation. Contrastingly, in the case of PMOA, fits of Pnma and P21/c symmetry to the PDF data did not show a significant difference, suggesting no such symmetry breakdown occurred (Fig. S15).
In the PDF fitting of PMON, we can derive two types of models (2D and 3D models) with different arrangements of the longest Mn–O bonds. The 3D model’s octahedral substructure reveals two distinct MnO6 units (royal/purple in Fig. 3b) with alternating corner connectivity. The longest Mn-O bond (d1 = 2.24 Å vs d2 = 2.20 Å) in the two different octahedra creates crosswise alignment in three-dimensional space (Fig. 3c). This contrasts sharply with the equatorial plane (i.e., the ab plane) restriction observed in the 2D model (Fig. 3d). In PMOA, only a 2D model was derived, whether utilizing Pnma or P21/c symmetries in the fitting (Fig. S15). This unique orbital configuration of PMON possibly enables the flexibility of MnO6 octahedra and facilitates OD through modified Mn-3d/O-2p hybridization, potentially correlating with the colossal NTE observed in PMON. The PDF fitting procedure is detailed in the Methods section. Theoretical investigations of orbital configuration-NTE relationships will be discussed later in the text.
Atomic-scale microstructure
To further elucidate the local atomic structure, we conducted atomic-resolution aberration-corrected scanning transmission electron microscopy (Cs-corrected STEM) studies. Figure 4a presents an annular bright-field (ABF) image of PMON along the [110] zone axis, and Fig. 4b displays its corresponding geometric phase analysis (GPA), revealing pronounced local strain field fluctuations. High-magnification high-angle annular dark-field (HAADF) imaging along [100] (Fig. 4c) resolves alternating Pr and Mn atomic columns, further corroborated by energy-dispersive spectroscopy (EDS) elemental mapping. Figures 4d, e compare cation ordering in PMON and PMOA, respectively. In PMON (Fig. 4d, e), HAADF and ABF imaging consistently reveal a zigzag (Z-shaped) cation arrangement. This configuration aligns with the 3D structural model derived from PDF refinement under P21/c symmetry (Fig. 4h, left panel), contrasting sharply with the quasi-linear cation alignment predicted by the orthorhombic Pnma 2D model (Fig. 4h, right panel). The observed Z-type ordering directly validates the local symmetry reduction (orthorhombic to monoclinic) identified through PDF analysis. Concerning PMOA, both HAADF and ABF images show a quasi-linear cation arrangement (Fig. 4f, g), consistent with its 2D structural models refined under either Pnma or P21/c symmetry (Fig. 4i). This structural invariance under differing symmetry constraints confirms the absence of significant local symmetry breaking in PMOA.
a Atomic-resolution STEM-ABF image of PMON along [110] zone axis with FFT image inset. b The corresponding GPA strain analysis of (a) using 200 and 0–20 reflections. c The STEM-HAADF image and atomic EDS mapping of PMON along [100] zone axis. The HAADF and ABF images of (d) PMON along [100] zone axis, and (f) of PMOA along [001], respectively. e, g Enlarged images of the white-rectangle regions in (d) and (f) show the cation (Pr and Mn) network arrangements. h, i Structural models derived from PDF fit of PMON and PMOA. The yellow and blue arrows indicate the slight static movements of Pr atoms. For PMON, the cation network in (e) shows a zigzag arrangement, consistent with the 3D model shown in (h). Meanwhile, the quasi-linear cation arrangement in the 2D model ((h), right panel) is inconsistent with the experimental image of (e). Regarding PMOA, only the 2D model could be generated, whether using P21/c or Pnma. This 2D model agrees with the atomic arrangement in (g). The scale bar in (a) indicates 5 nm, and the other scale bars indicate 0.5 nm. Pr atoms in green, Mn atoms in purple, O atoms in orange.
Theoretical investigation
The JT distortion in MnO6 octahedra promotes preferential electron occupancy in the Mn 3 d orbitals, stabilizing OO. The reduction of JT distortion, concomitant with the onset of OD, triggers a redistribution of 3 d electrons, thereby inducing anisotropic Coulomb interactions across crystallographic directions11. This directional electronic reorganization drives lattice parameter adjustments and NTE. Unlike conventional NTE mechanisms involving low-frequency phonons or magnetic order, the dynamic evolution of JT distortion governs the giant NTE observed in PMON. To elucidate the interplay between orbital configuration, JT distortion, and NTE, we performed density functional theory (DFT) calculations on PMON-derived structural models under varying conditions.
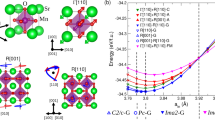
DFT simulations compared two PMON configurations: Structure I (room temperature, RT) and Structure II (high temperature, HT), both with constrained oxygen positions to preserve specific Mn-O bond distances and JT distortion. Figure 5a reveals alternating 3 d orbital patterns at neighboring Mn sites in Structure I, hallmarking robust OO. Conversely, Structure II (Fig. 5b) exhibits a distorted-cross orbital morphology indicative of OD. The observed orbital occupancy patterns at room temperature (RT) and high temperature (HT) accurately reflect the electronic configuration characteristics of PMON as it undergoes the critical transition from OO to OD. To probe defect-mediated orbital modulation, we constructed derivative models from Structure I: oxygen-unconstrained (III), single/double oxygen-intercalated (IV/V), and hole-doped (VI) systems. Structures III-V retained OO (Fig. 5c-e), mirroring experimental observations in oxygen-rich PMOA. Notably, only hole-doped Structure VI (Fig. 5f) exhibited OD, underscoring hole-doping-specific orbital destabilization.
a–f PCD (upper panel) and PCD map projected along [100] direction (lower panel) of structure I-VI: a, b I and II structures with fixed oxygen at room temperature (RT) and high temperature (HT); c III: RT structure with unfixed oxygen; d IV: structure with inserting one oxygen; e V: structure with inserting two oxygens; f VI: structures with hole doping. The blue arrow indicates the existence of interstitial oxygen. Scale bar: charge intensity. g A summary of JT distortion, δ. Experimental δ derives from structures I’-III’ of PMON at 900 K and 920 K. Calculated δ is extracted based on the calculated structures I-VI of PMON. The red dashed line represents the threshold value of δ, below which the specific structural configurations exhibit OD. Source data are provided as a Source Data file.
OO is positively correlated with JT distortion magnitude (denoted as δ, see Section 3.1). A large δ value indicates significant disparities in Mn–O bond lengths and preferential electron occupancy in the 3 d orbitals. Conversely, a smaller δ value suggests more uniform Mn–O bond lengths and a randomized 3 d electron distribution. Figure 5g summarizes the JT distortion derived from both experimental and calculated structures. Experimental PMON data at 900 K (δ = 3.47%, Structure I’) confirm OO dominance, while HT phases at 920 K (Structures II’/III’: δ = 1.50% / 1.14%) reflect OD onset, coinciding with NTE emergence. This suggests that changes in 3 d electron occupancy, closely linked to Coulomb forces in different directions, drive the anomalous lattice volume changes with temperature. In addition, calculated Structure I (RT, fixed oxygen) yielded δ = 3.68% (OO), whereas Structure II (HT, fixed oxygen) showed δ = 1.28% (OD). Oxygen-intercalated models to mimic PMOA (Structure IV/V, δ > 3%) maintained OO. This indicates that interstitial oxygen insertion in PrMnO3 does not disrupt the OO state, contrasting with the interstitial-oxygen-driven OO destabilization found in Ca2RuO411. Strikingly, hole doping (Structure VI) minimized δ to 0.06% (OD, Fig. 5g), highlighting its possibility to affect the preferred occupancy of 3 d electrons.
In conclusion, we present, to the best of our knowledge, the first observation of giant NTE exceeding 1000 K (αV = − 85 × 10−6 K−1, 900–1100 K) in PMON, with a peak αV = − 114 × 10−6 K−1, representing the highest operational temperature range among known giant NTE materials. Intriguingly, oxygen-rich PMOA shows a complete suppression of NTE behavior and instead transitions to conventional PTE. Comprehensive structural analysis indicates that the anomalous NTE occurs along with the temperature-dependent evolution of JT distortions in MnO6 octahedra and an OO to OD transition. Notably, a local structural distortion characterized by 3D cross-arranged Mn-O bond elongation was identified in PMON through PDF analysis, in contrast to the 2D planar configurations observed in PMOA. This 3D structural motif is further confirmed by Cs-corrected high-resolution TEM imaging, which reveals characteristic zigzag alignments of Pr atomic columns. First-principles calculations provide direct evidence of oxygen-stabilized OO and demonstrate that hole doping effectively induces OD. These findings broaden the scope of high-temperature giant NTE materials characterized by unique local structural distortions, atomic configurations, and OD phenomena, offering potential applications for engineering components in operation.
Methods
Materials synthesis
Pr6O11 (99.99%, Macklin), Mn2O3 (99.9%, Aladdin), HNO3 (65–68%, Sinopharm), and citric acid ( > 99.5%, Aladdin) were used as starting materials. The PMOA containing excess oxygen was prepared using the solid-state reaction method, which involves carefully mixing and grinding the Pr6O11 and Mn2O3 according to the nominal stoichiometric ratio. The mixed powder was annealed in the atmosphere at 1200 °C for 12 h. The PMON was prepared by a citrate decomposition method. Specifically, the Pr6O11 and Mn2O3 with a nominal stoichiometric ratio were dissolved in citrate and nitrate solutions, followed by evaporation at 120 °C and decomposed at 200 °C, leading to a fragile precursor. The precursor was ground into powder and annealed at 700 °C for 12 h in air to remove the organics and nitrates, and loaded into the tube furnace and heated at a ramping rate of 5 °C min−1 to 1100 °C, held at this temperature point for 12 h, and subsequently cooled in the furnace. The entire process was under the protection of high-purity nitrogen (99.99%) gas with a flow rate of 50 mL min-1.
SXRD, total scattering and XRD
Synchrotron X-ray experiments were conducted at the 11-ID-C and 17BM beamlines of Argonne National Laboratory. PMON and PMOA powders were loaded into Hilgenberg quartz glass capillaries within a glove box maintained at O2 and H2O levels below 0.1 ppm. After temporary sealing with plasticine, the capillaries were sealed using a hydrogen pilot flame to preserve the sample under inert conditions. These sealed capillaries were then subjected to high-temperature X-ray measurements in transmission mode. Prior to heating, careful temperature calibration was performed. For heating, hot air was applied to the capillaries, with measurement parameters set as follows: exposure time at 90 s, waiting time at 120 s, and ramping rate at 20 K min−1.
Synchrotron X-ray total scattering measurements were conducted by positioning the detector approximately 350 mm from the sample. Powder samples were sealed into 2 cm-long, 1 mm-diameter Kapton tubes, with both ends secured using plasticine. For data acquisition, the exposure time was set to 4 s per frame, with 30 consecutive frames collected to generate a single data image. To enhance data quality, twenty such data images were acquired and averaged. Prior to sample measurement, an empty Kapton tube was measured to record the background scattering signal. Total scattering data were processed using PDFgetx2, which involved corrections, background subtraction, and S(Q) normalization to yield high-quality structural information43.
High-temperature XRD patterns were acquired using a Rigaku powder X-ray diffractometer equipped with a Cu target, operating at 40 kV and 40 mA. Powder samples were uniformly dispersed on the Pt pad and transferred into the measurement chamber under vacuum conditions. The exposure time, wait time, and ramping rate were set to 45 min, 30 min and 10 K min−1, respectively.
XAS measurements
XAS for the O K-, Mn L-, and Mn K-edges were collected at multiple synchrotron facilities, including BL08U1A and BL14W1 of the Shanghai Synchrotron Radiation Facility (SSRF), as well as ID07 and ID46 of the High Energy Photon Source (HEPS). For the Mn L- and O K-edge measurements, samples were analyzed in total electron yield mode under high-vacuum conditions. To acquire high-quality Mn K-edge data, suitable amounts of samples were first mixed and ground with boron nitride powder, then pressed into 1 mm-thick pellets. Transmission mode was employed for data collection to ensure an edge jump of approximately 1, optimizing the signal-to-noise ratio for accurate spectral analysis.
PDF analysis
The PDF G(r) function was refined using the small-box method in PDFgui44 with a Q resolution of 25 Å−1. Structural models were constructed based on Pnma and P21/c space groups. Initial parameters for the average structural model were sourced directly from Rietveld refinement of the SXRD patterns. For both 2D and 3D models, refinement included variable parameters such as scale factors, δ1, Q_broad, Q_damp, atomic coordinates, and isotropic atomic displacement parameters (ADPs). The starting value of Q_damp was set to 0.03, which was determined by calibration of the CeO2 reference sample. In the 3D model based on P21/c symmetry, the α angle was allowed to relax. Additionally, the ADPs of O1 and O2 in the 3D model were constrained to be equivalent, ensuring consistency in the number of refined parameters with the 2D model. Notably, the P21/c symmetry introduces two distinct O2 sites (O2 and O2’), but to avoid unnecessary parameter expansion, the symmetry relationship between O2 (x, y, z) and O2’ (1−x, y + 0.5, z) was enforced. Both PMON and PMOA were refined against both 2D and 3D models. For PMON, the 3D model yielded a significantly better fit (Rw = 10.5%) compared to the 2D model (Rw = 16.3%), whereas for PMOA, the fits of the 3D and 2D models were highly comparable. This discrepancy underscores the presence of local structural distortions in PMON, characterized by 3D-ordered Mn-O bond elongation.
Cs-corrected STEM measurements
The high-angle annular dark-field aberration-correlated scanning transmission electron microscopy (HAADF-STEM, Titan 80–300 FEI) was employed to study the atomic structure of PrMnO3. The specimen was cut finely by a focused ion beam (FIB) method (Helios 450, FEI).
DFT calculations
The first-principles density functional theory (DFT) calculations were employed by the Vienna Ab initio Simulation Package (VASP)45,46. The exchange-correlation functional was selected to be the revised Perdew-Burke-Ernzerhof variant for solids (PBEsol) of the generalized gradient approximation (GGA)47. The plane wave basis set and a cutoff energy of 450 eV. The electronic and force convergence criteria were chosen as 10−5 eV and 10−3 eV Å−1 , respectively. To identify the partial charge density and density of states in PMON which was inserted into two oxygens, a 2 × 2 × 1 supercell was constructed. The Brillouin zone was sampled using a 6 × 6 × 4 and 3 × 3 × 4 Gamma mesh for the PMON, and PMON inserted two oxygens. The spin polarization of Pr was taken into consideration by setting the ferromagnetic order according to the previously reported magnetic geometry48. The effect of electronic corrections on the Pr 4 f shell was considered using the GGA + U method with U = 7.0 eV and J = 7.0 eV(for Pr), and U = 4.0 eV, J = 0.87 eV for the 3 d orbital of the Mn49.
Data availability
The data supporting the findings of this study are available from the article and the supplementary materials file. All the raw data are available from the corresponding authors upon request. Source data are provided with this paper.
References
Takenaka, K. Negative thermal expansion materials: technological key for control of thermal expansion. Sci. Technol. Adv. Mat. 13, 11 (2012).
Zhang, Y. et al. Thermal-expansion offset for high-performance fuel cell cathodes. Nature 591, 246–251 (2021).
Mary, T. A., Evans, J. S. O., Vogt, T. & Sleight, A. W. Negative thermal expansion from 0.3 to 1050 Kelvin in ZrW2O8. Science 272, 90–92 (1996).
Greve, B. K. et al. Pronounced negative thermal expansion from a simple structure: cubic ScF3. J. Am. Chem. Soc. 132, 15496–15498 (2010).
Hu, L. et al. Localized symmetry breaking for tuning thermal expansion in ScF3 nanoscale frameworks. J. Am. Chem. Soc. 140, 4477–4480 (2018).
Song, Y. Z. et al. Zero thermal expansion in magnetic and metallic Tb(Co,Fe)2 intermetallic compounds. J. Am. Chem. Soc. 140, 602–605 (2018).
Huang, R. J. et al. Giant negative thermal expansion in NaZn13-type La(Fe, Si, Co)13 compounds. J. Am. Chem. Soc. 135, 11469–11472 (2013).
Azuma, M. et al. Colossal negative thermal expansion in BiNiO3 induced by intermetallic charge transfer. Nat. Commun. 2, 347 (2011).
Takenaka, K., Okamoto, Y., Shinoda, T., Katayama, N. & Sakai, Y. Colossal negative thermal expansion in reduced layered ruthenate. Nat. Commun. 8, 14102 (2017).
Pachoud, E., Cumby, J., Lithgow, C. T. & Attfield, J. P. Charge order and negative thermal expansion in V2OPO4. J. Am. Chem. Soc. 140, 636–641 (2018).
Hu, L. et al. Origin and absence of giant negative thermal expansion in reduced and oxidized Ca2RuO4. Chem. Mater. 33, 7665–7674 (2021).
Agne MT, Anand S, Snyder GJ. Inherent anharmonicity of harmonic solids. Research-China 2022, (2022).
Iikubo, S. et al. Local lattice distortion in the giant negative thermal expansion material Mn3Cu1-xGexN. Phys. Rev. Lett. 101, 205901 (2008).
Zheng, X. G. et al. Giant negative thermal expansion in magnetic nanocrystals. Nat. Nanotechnol. 3, 724–726 (2008).
Gao, Q. L. et al. Switching between giant positive and negative thermal expansions of a YFe(CN)6-based prussian blue analogue induced by guest species. Angew. Chem. Int Ed. 56, 9023–9028 (2017).
Sun, H. Y. et al. Colossal anisotropic thermal expansion through coupling spin crossover and rhombus deformation in a hexanuclear {FeIII4FeII2} compound. Angew. Chem. Int Ed. 62, 8 (2023).
Xu, M. et al. Giant uniaxial negative thermal expansion in FeZr alloy over a wide temperature range. Nat. Commun. 14, 4439 (2023).
Mazzone, D. G. et al. Kondo-induced giant isotropic negative thermal expansion. Phys. Rev. Lett. 124, 125701 (2020).
Yamamoto, H., Imai, T., Sakai, Y. & Azuma, M. Colossal negative thermal expansion in electron-doped PbVO3 perovskites. Angew. Chem. Int Ed. 57, 8170–8173 (2018).
Zhao, Y. Y. et al. Giant negative thermal expansion in bonded MnCoGe-based compounds with Ni2In-type hexagonal structure. J. Am. Chem. Soc. 137, 1746–1749 (2015).
Lin, J. C. et al. Giant room-temperature barocaloric effect at the electronic phase transition in Ni1-xFexS. Mater. Horiz. 7, 2690–2695 (2020).
Yu, C. Y. et al. An isotropic zero thermal expansion alloy with super-high toughness. Nat. Commun. 15, 2252 (2024).
Cao, Y. L. et al. Ultrawide Temperature Range Super-Invar Behavior of R2(Fe,Co)17 Materials (R = Rare Earth). Phys. Rev. Lett. 127, 055501 (2021).
Liang, Z. L. et al. Uniaxial Negative Thermal Expansion in γ-LiBO with a Closed-Framework Diamond-Like Structure. Chem. Mater. 36, 7299–7305 (2024).
Gao, Q. L. et al. Giant Negative Thermal Expansion in Ultralight NaB(CN)4. Angew. Chem. Int Ed. 63, 6 (2024).
Spaldin, N. A. & Ramesh, R. Advances in magnetoelectric multiferroics. Nat. Mater. 18, 203–212 (2019).
Tokura, Y. & Nagaosa, N. Orbital physics in transition-metal oxides. Science 288, 462–468 (2000).
Mefford, J. T., Hardin, W. G., Dai, S., Johnston, K. P. & Stevenson, K. J. Anion charge storage through oxygen intercalation in LaMnO perovskite pseudocapacitor electrodes. Nat. Mater. 13, 726–732 (2014).
Suntivich, J. et al. Design principles for oxygen-reduction activity on perovskite oxide catalysts for fuel cells and metal-air batteries. Nat. Chem. 3, 546–550 (2011).
Uehara, M., Mori, S., Chen, C. H. & Cheong, S. W. Percolative phase separation underlies colossal magnetoresistance in mixed-valent manganites. Nature 399, 560–563 (1999).
Qiu, X. Y., Proffen, T., Mitchell, J. F. & Billinge, S. J. L. Orbital correlations in the pseudocubic and rhombohedral phases of LaMnO3. Phys. Rev. Lett. 94, 177203 (2005).
Thygesen, P. M. M. et al. Local structure study of the orbital order/disorder transition in LaMnO3. Phys. Rev. B 95, 174107 (2017).
Sánchez, M. C., Subías, G., García, J. & Blasco, J. Cooperative Jahn-Teller phase transition in LaMnO3 studied by X-ray absorption spectroscopy. Phys. Rev. Lett. 90, 045503 (2003).
Chatterji, T., Fauth, F., Ouladdiaf, B., Mandal, P. & Ghosh, B. Volume collapse in LaMnO3 caused by an orbital order-disorder transition. Phys. Rev. B 68, 052406 (2003).
Tikkanen, J., Kauhala, S., Huhtinen, H. & Paturi, P. Anomalous thermal expansion in (Pr, Ca)MnO3 due to orbital ordering. Physcs Proc. 75, 475–481 (2015).
Sánchez D, Alonso JA, Martínez-Lope MJ. Neutron-diffraction study of the Jahn-Teller transition in PrMnO3. J. Chem. Soc. Dalton, 4422-4425 (2002).
Vedmid’, L. B., Yankin, A. M., Fedorova, O. M. & Kozin, V. M. Phase diagram of the Pr-Mn-O system in composition-temperature-oxygen pressure coordinates. Russ. J. Phys. Chem. 90, 977–982 (2016).
Ulyanov, A. N. et al. Electron structure and magnetic properties of self-doped Pr1-xMnO3+δ manganites: XANES and EXAFS study. Comparing with A- and B-site doped perovskites. Solid State Sci. 118, 106653 (2021).
Frati, F., Hunault, M. O. J. Y. & de Groot, F. M. F. Oxygen K-edge X-ray absorption spectra. Chem. Rev. 120, 4056–4110 (2020).
Abbate, M. et al. Controlled-valence properties of La1-xSrxFeO3 and La1-xSrxMnO3 studied by soft-X-ray absorption spectroscopy. Phys. Rev. B 46, 4511–4519 (1992).
Cao, D., Bridges, F., Kowach, G. R. & Ramirez, A. P. Correlated atomic motions in the negative thermal expansion material ZrW2O8: a local structure study. Phys. Rev. B 68, 014303 (2003).
Qin, F. Y. et al. Integrating abnormal thermal expansion and ultralow thermal conductivity into (Cd,Ni)2Re2O7 via synergy of local structure distortion and soft acoustic phonons. Acta Mater. 264, 119544 (2024).
Qiu, X., Thompson, J. W. & Billinge, S. J. PDFgetX2: a GUI-driven Program to Obtain the Pair Distribution Function from X-ray Powder Diffraction Data. J. Appl. Crystallogr. 37, 678 (2004).
Farrow, C. L. et al. PDFfit2 and PDFgui: computer programs for studying nanostructure in crystals. J. Phys.-Condens Mat. 19, 335219 (2007).
Kresse, G. & Furthmuller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Kresse, G. & Furthmuller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Perdew, J. P. et al. Restoring the density-gradient expansion for exchange in solids and surfaces. Phys. Rev. Lett. 100, 136406 (2008).
Mishra, S. K., Gupta, M. K., Mittal, R., Kolesnikov, A. I. & Chaplot, S. L. Spin-phonon coupling and high-pressure phase transitions of RMnO3 (R = Ca and Pr): An inelastic neutron scattering and first-principles study. Phys. Rev. B 93, 214306 (2016).
Bouadjemi, B., Bentata, S., Abbad, A., Benstaali, W. & Bouhafs, B. Half-metallic ferromagnetism in PrMnO3 perovskite from first principles calculations. Solid State Commun. 168, 6–10 (2013).
Acknowledgments
We acknowledge funding from Science Fund Program for Distinguished Young Scholars (Overseas), National Natural Science Foundation of China (No. 22205171, 52472076, 22405202), Key Research and Development Program of Shaanxi Province (No. 2024CY2-GJHX-86 and 2024PT-ZCK-41), International Postdoctoral Exchange Fellowship Program (Talent-Introduction Program), China Postdoctoral Science Foundation (No. 2023M732754), Postdoctoral Research Project of Shaanxi Province (No. 31271000000040), and Natural Science Basic Research Program General Project–Youth Project of Shaanxi Province (2024JC-YBQN-0117).
Author information
Authors and Affiliations
Contributions
L.H. and F.Q. conceived the idea of the work. F.Q. carried out the main experiments, data processed, and prepared the manuscript. P.C., D.W., X.B., Y.F., J.W., and P.Z. carried out the measurement and contributed to calculations. L. H., X. D., and J.S. supervised the project. All the authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Qin, F., Bai, X., Fang, YW. et al. Giant negative thermal expansion exceeding 1000 K in PrMnO3 via synergy of local structure distortion and orbital disordering. Nat Commun 16, 9977 (2025). https://doi.org/10.1038/s41467-025-64925-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-64925-7