Abstract
Cellulose-based ionogels are promising for flexible electronics and energy devices, yet their performance is often constrained by the trade-offs among mechanical robustness, ionic conductivity, and thermal stability. Here, we propose a synergistic strategy that integrates dual-ions complexation with crystallization-induced molecular assembly to fabricate a cellulose ionogel. This strategy results in a comprehensive ionogel (noted as Cry-gel) with high mechanical strength (2.3 MPa in tension and 5.3 MPa in compression) and high ionic conductivity (96.8 mS cm−1). Moreover, the Cry-gel can maintain impressive structural stability across a temperature range of −40 to 80 °C. Flexible thermoelectric devices and smart sensors derived from Cry-gels demonstrate a voltage of 0.28 V at a temperature gradient of 60 K, an impressive Seebeck coefficient of 6 mV K−1, and high sensitivity to pressure, temperature, touch, and human pulse. This work provides a paradigm for creating multifunctional sustainable materials, effectively bridging the gap between high-performance ionogels and their applications in cutting-edge bioelectronics and energy harvesting systems.
Similar content being viewed by others
Introduction
Ionogels have emerged as a class of advanced materials known for their exceptional flexibility, electrochemical properties, and biocompatibility1,2,3, making them promising candidates for applications in sensors4, energy storage5, and bioelectronics6,7,8. However, the widespread adoption of ionogels is hindered by a critical challenge: the intrinsic trade-offs between mechanical robustness, ionic conductivity, and environmental adaptability9,10. To address these challenges, various strategies have been explored to enhance the performance of ionogels11,12,13. Early approaches primarily involved simply mixing polymers with ionic liquids or salts14, which improved the ionic conductivity of ionogels but compromised their mechanical strength. Subsequently, the introduction of cross-linked networks15 and the incorporation of organic solvent systems with nano-reinforced phases16,17 were utilized to create multi-scale network structures that could improve the mechanical performance of ionogels. Despite these advances, preserving the structural integrity, stability, and reliable mechanical properties of ionogels across a wide temperature range continues to pose significant challenges.
Recent advances highlight the potential of ion complexation and crystallization to tailor material architectures and performance18,19,20,21. For instance, metal-ion coordination can reinforce polymer networks to improve mechanical performance22,23,24,25,26, while crystallization dynamics offer pathways to optimize ion transport and supramolecular structural integrity by guiding the arrangement of molecular chains27. However, the dynamic interplay between ion clustering, hydrogen bonding (H-bonding), and crystallization kinetics remains unexplored in cellulose ionogel systems. Understanding how to effectively orchestrate these processes within cellulose ionogel is crucial for achieving simultaneous improvements in mechanical, electrical, and thermal properties.
In this study, we present a robust and highly conductive cellulose ionogel, formulated through crystallization-induced molecular assembly, which effectively decouples the trade-off among mechanical properties, ionic conductivity, and thermal stability. Unlike conventional methods, our approach utilizes crystallization to initiate the self-assembly of cellulose molecular chains, promoting a more compact structure while enhancing temperature stability and mechanical strength through ion complexation that binds water and cellulose molecules. The resulting cellulose ionogel (Cry-gel) demonstrates a tensile strength of 2.3 MPa, an ionic conductivity of 96.8 mS cm−1, and a reliable structural performance within a wide temperature range of −40 to 80 °C. Therefore, the Cry-gel outperforms existing ionogels in balanced properties, achieving conductivity comparable to liquid electrolytes while maintaining robustness under extreme conditions. Furthermore, this Cry-gel exhibits a high Seebeck coefficient of 6 mV K−1, an output thermal conductivity of 0.4 W m−1 K−1, and a power density of 0.1 mW m−2 K−2. These performance advantages indicate promising potential of Cry-gel for applications in flexible thermoelectric devices, self-powered sensors, and medical health technologies.
Results
Fabrication design and characterizations of Cry-gel
Among the various non-covalent interactions, metal coordination bonds typically exhibit relatively high binding strengths28. Lithium bonds (Li-bonds), analogous to hydrogen bonds (H-bonds), are anticipated to exhibit similar characteristics and functionalities29,30,31. Notably, Li-bonds are neither fully saturated nor strictly directional, allowing for fine-tuning of the coordination environment by varying the proportions of Li+ ions and functional groups. This adjustability provides a design for manipulating mechanical properties, ionic conductivity, and other characteristics within gel materials.
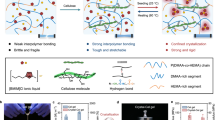
Based on this understanding, we utilized lithium bromide (LiBr) and zinc chloride (ZnCl2) in combination with water to create a double-ion salt system that facilitates the assembly adjustment of cellulose molecules, resulting in the production of a crystallization-induced ionogel (Fig. 1a and Supplementary Fig. 1). Through heat treatment at 100 °C, cellulose fibers with the degree of polymerization (DP) of approximately 830 were dissolved in this double-ion solution, forming a homogeneous cellulose molecular system containing Li+, Zn2+, Br⁻, and Cl⁻ ions, along with cellulose molecules with a DP of around 450. As the temperature falls to a room temperature of approximately 25 °C, the multiple ions within the cellulose molecular system aggregate and initiate crystallization, which is driven by the coordination interactions among these metal ions. During the crystal growth process, the cellulose molecular chains become more compact through crystallization, resulting in a denser and more convergent cellulose network with microscale directional aggregation.
a Schematic diagram illustrating the ion complexation and crystallization-induced construction strategy for Cry-gel. b Optical image of the resulting Cry-gel, highlighting its flexibility, mechanical robustness, anti-temperature, and high ionic conductivity. c Radar chart comparing the mechanical compressive strength, tensile strength, anti-temperature, ionic conductivity, frost resistance, and self-healing ability of Cry-gel with other gel materials. d Conceptual illustration of potential applications for Cry-gel in portable, sustainable flexible electronic devices. Each element in (a) and (d) was conceived by the authors and created using Adobe Photoshop CC 2019 and 3ds Max 2018 software.
Subsequently, the crystallized system is exposed to room temperature to facilitate the adsorption of H2O molecules. The presence of H2O not only disrupts the coordination interactions between metal ions, thereby releasing the constraints on cellulose molecules, but also promotes metal ions and themselves (H2O) with cellulose molecules to form complexation and H-bonding interactions, respectively (right of Fig. 1a). The multi-ion complexation strengthens the H-bond network associated with cellulose molecules, resulting in Cry-gel with a H2O content of around 43.6 wt% (Fig. 1b). Compared to previously reported gel materials32,33,34, the Cry-gel, derived from double-ion salt system with the optimal mass ratio of LiBr:ZnCl₂:H₂O at 65:4:35 (Supplementary Fig. 2), demonstrates superiority in several performance aspects including mechanical performance, self-healing, ionic conductivity, and anti-temperature (Fig. 1c). Due to these beneficial features, the Cry-gel exhibits significant potential for applications in portable, sustainable flexible electronics such as smart sensors, bioelectronics, and thermoelectric devices (Fig. 1d).
Mechanism of crystallization-induced assembly of Cry-gel
To further study the properties of Cry-gel, we investigated the cellulose molecular assembly behavior during crystallization induction as well as the intrinsic mechanisms involved in gelation. As shown in Fig. 2a, with the cooling effect of surroundings on cellulose molecular system, crystal growth gradually becomes apparent. Moreover, the crystallization behavior of the dual salts LiBr/ZnCl2 in the Cry-gel system demonstrates a directional quality (Supplementary Fig. 3), which acts as a driving force promoting the alignment and proximity of cellulose molecules. This suggests that the crystallization strategy preserves the original network structure of the cellulose while directing its assembly, thereby enhancing the density and achieving a directional morphological structure in the cellulose molecular network.
a Macro photograph showing the crystallization-induced formation of Cry-gel. b Comparison of FTIR spectra between crystalline state and gel state. Insets show the enlarged data graphics of the shaded portions. c SAXS spectra of LiBr gel and Cry-gel. d 2D scattering SAXS patterns of LiBr gel. e 2D scattering SAXS patterns of Cry-gel. f Matrix-assisted laser desorption time-of-flight mass spectrometer (TOF-MS) analysis of LiBr gel and Cry-gel. g Snapshot of LiBr gel from MD simulations. h Snapshot of Cry-gel from MD simulations. i Comparison of the radial distribution function, g(r), between ions (Li+, Br−, Zn2+, and Cl−) and H2O molecules within Cry-gel.
After a three-hour crystallization period, the bottle cap was removed, exposing the contents to a room temperature environment. This exposure caused the adsorbed H2O molecules to form hydrates with the ions in the crystalline cellulose system, facilitating a transition from a crystalline state to a gel state and leading to the formation of Cry-gel (Fig. 2a). During this transition, the ionic crystals, which previously immobilized the cellulose molecules, now release them. These liberated cellulose molecules can then establish a new molecular network through hydrogen bonding. Moreover, small and polar H2O molecules penetrate this molecular network, forming hydrogen bonds with the cellulose molecules’ hydroxyl groups (-OH). This process enhances ion complexation between the hydrated ions and the cellulose molecules, thereby strengthening the molecular network within the Cry-gel.
Fourier transform infrared spectroscopy (FTIR) analysis, as illustrated in Fig. 2b, reveals a redshift at 1620 cm−1, corresponding to the bending vibration of water, and a shift in the range of 3000–3700 cm−1, attributed to the stretching vibrations of the -OH groups of cellulose. These findings support the aforementioned observations. Small-angle X-ray scattering (SAXS) analysis presented in Fig. 2c indicates a peak shift in the Cry-gel compared to the LiBr gel, with the cellulose interchain distance decreasing from 1.48 nm to 1.43 nm. This shift signifies a reduction in the distance between cellulose molecules due to crystallization induction35,36. In contrast to the LiBr gel (Fig. 2d), Cry-gel exhibits a distinctive two-dimensional SAXS scattering pattern (Fig. 2e), indicative of a denser microstructure and a more uniform ion distribution (Supplementary Figs.4 and 5).
Time-of-flight mass spectrometry (TOF-MS) analysis (Fig. 2f) reveals the primary ion cluster peaks of Cry-gel at m/z 172.06 and 85.01, while the LiBr gel exhibits peak clusters at m/z 202.05 and 590.11. These observations suggest that the introduction of Zn2+ ions disrupts the Li+ clusters, facilitating the formation of stable Zn2+ and Li+ ion-complexation with cellulose molecules. Solid-state 7Li nuclear magnetic resonance (NMR) analysis shows that the chemical shift of Li+ in the LiBr/ZnCl₂ solvent is −0.66 ppm, while in the Cry-gel, this shift changes to 0.29 ppm (Supplementary Fig. 6). This variation further corroborates the complexation interactions between Li+ ions and cellulose molecules within Cry-gel. The Raman spectra demonstrate a distinct split peak in the region of 3000–3700 cm−1, attributable to the –OH vibrations resulting from the complexation interactions between the Zn2+/Li+ ions and cellulose molecules (Supplementary Fig. 7a, b).
To further investigate the molecular network properties of Cry-gel, we conducted molecular dynamics (MD) simulations to analyze the H-bonds and complexation interactions between cellulose molecules and ions (see Supplementary Methods for details). The complexation pattern within the LiBr gel is primarily characterized by interactions between Li+ ions and H2O molecules, referred to as Li+ ion-complexation behavior (Fig. 2g). Subsequently, the Li⁺-hydrated complexes form H-bonds and complexation interactions with the –OH groups on cellulose molecules. In contrast, Cry-gel exhibits two distinct types of complexation patterns: one involving Zn2+ ion-complexation behavior and the other involving Li+ ion-complexation behavior (Fig. 2h). Analysis of the radial distribution function reveals that the radial peaks of Zn2+ ions and H2O molecules are sharper and more pronounced (Fig. 2i), indicating that Zn2+ ions can establish more intense complexation interactions with H2O molecules. Consistently, MD simulation results demonstrate that Cry-gel exhibits stronger cellulose–cellulose and cellulose–solvent interactions than those of LiBr gel, as evidenced by lower interaction energies (Supplementary Table 1 and Supplementary Fig. 7c) and slightly higher hydrogen bond numbers (Supplementary Table 2 and Supplementary Fig. 7d). The end-to-end distance and radius of gyration (Rg) of cellulose molecular chains were assessed (Supplementary Fig. 8a, b). The results indicate that the Cry-gel exhibits shorter end-to-end distances and smaller Rg values compared to the LiBr gel. This suggests a more coiled chain conformation, which can be attributed to strong intermolecular interactions within the Cry-gel system. The inter-chain distances between cellulose molecular chains were further calculated for all 15 pairwise combinations of six cellulose chains (as shown in Supplementary Fig. 9a–o). In the Cry-gel system, most chain pairs exhibit shorter distances than those in the LiBr gel. The averaged inter-chain distance, as summarized in Supplementary Fig. 9p, decreases from 1.56 nm in the LiBr gel to 1.21 nm in the Cry-gel. These findings further substantiate the enhanced intermolecular associations and compact molecular organization in Cry-gel. Snapshots from MD simulations illustrate that this multi-modal ion complexation behavior facilitates the self-assembly of cellulose molecules, resulting in a more compact molecular network structure (Fig. 2h). The synergistic effects arising from hydrogen bonding and complexation interactions enhance the structural stability of the Cry-gel network, imparting it with robust mechanical properties as well as enhanced electrical performance and temperature resistance.
Thermal stability and self-healing properties of Cry-gel
Thermal stability and phase transition behavior are critical attributes for the performance of ionogel materials. Rheological analyses (Fig. 3a, b) reveal that ZnCl2 gel predominantly exhibits fluid behavior from 25 to 100 °C, characterized by a loss modulus (G″) that exceeds the storage modulus (G′); LiBr gel undergoes a phase transition at 65 °C, transitioning into a viscoelastic flow state with reduced viscosity (Fig. 3a). In comparison, the Cry-gel maintains its elasticity and flexibility even at temperatures up to 95 °C, as characterized by a G′ exceeding the G″, demonstrating mechanical behavior that permits bending and twisting (Fig. 3b). These results indicate that the Cry-gel possesses superior thermal stability and resistance to high temperatures.
a Rheological behavior of LiBr gel and ZnCl2 gel during heating and cooling cycles. b Rheological behavior of Cry-gel as a function of temperature. Inset shows the photograph illustrating the mechanical flexibility of Cry-gel. c DSC behaviors between LiBr gel and Cry-gel. d Photographs and size changes of Cry-gel exposed to temperature: heated at 80 °C for 50 h and cooled at −40 °C for 6 days. e The dimensional retention of Cry-gel during exposure at 80 °C for 50 h. f The dimensional retention of Cry-gel during exposure at −40 °C for 6 days. g Schematic diagram of the thermal reversibility and the reconstruction of the network structure of Cry-gel. Each inset image was conceived by the authors and created using Adobe Photoshop CC 2019 software. h Photograph showing the self-healing behavior of Cry-gel.
Differential scanning calorimetry (DSC) analysis (Fig. 3c and Supplementary Fig. 10) indicates distinct phase transition behaviors across the Cry-gel, LiBr gel, and ZnCl₂ gel. Notably, Cry-gel exhibits a significantly lower crystallization temperature of −71.59 °C, in contrast to −22.72 °C for the LiBr gel, which suggests enhanced stability and resilience at low temperatures. Additionally, Cry-gel displays lower temperatures for both cold crystallization and melting peaks, indicating faster phase transitions and recovery. These findings underscore the crucial role of Zn2+ in stabilizing the cellulose molecular network through ion-water complexation interactions, which confers enhanced temperature resistance to the Cry-gel.
As depicted in Fig. 3d−f, Cry-gel demonstrates notable structural stability and dimensional consistency, showing negligible size changes after 50 h at 80 °C and no measurable variation after 6 days at −40 °C with encouraging mechanical flexibility (Supplementary Fig. 11) and relatively stable H2O content (Supplementary Fig. 12). This performance is not achievable with LiBr gel and ZnCl2 gel. Beyond 130 °C, the Cry-gel transitions into a viscoelastic flow state; this transformation is likely attributable to the disruption of network structure between cellulose molecules, H2O, Zn2+ and Li− ions under high thermal energy (Fig. 3g). Upon cooling, the complexation interactions between cellulose molecules and Zn2+/Li− ion-hydrates are reestablished, with some of the absorbed energy stored within the bonds and the remainder dissipated as heat. Simultaneously, the H-bonding networks between cellulose molecules and between cellulose and H2O molecules are restructured, presenting the mechanical robustness for Cry-gel. This thermally reversible nature endows the Cry-gel with unique self-healing properties (Fig. 3h). This feature not only reduces material waste but also enhances the potential for sustainable use and recycling of ionogel materials.
Mechanical and electrical properties of Cry-gel
The mechanical properties of Cry-gel are firstly evaluated using nanoindentation tests, as shown in Fig. 4a. Under the same applied force, Cry-gel exhibits a shallower indentation depth compared to the LiBr gel, indicating the robust mechanical performance of Cry-gel. Quantitative assessment of the mechanical properties was achieved through tensile stress-strain tests. As presented in Fig. 4b, Cry-gel achieves a maximum tensile stress of 2.3 MPa at a fracture strain of 150%, which is triple the elongation at break and tensile strength compared to those of the LiBr gel (Fig. 4b). As shown in Supplementary Figs. 13 and 14, ZnCl₂ gel exhibits poor mechanical properties, shattering upon even the slightest contact, whereas LiBr gel fractures at a strain of just 50%. In contrast, Cry-gel remains intact even at 50% strain, demonstrating enhanced toughness and elongation. Furthermore, Cry-gel can recover a tensile strength of 1 MPa even after multiple cuts and self-healing processes (Supplementary Fig. 15). Notably, the mechanical properties remain robust and reliable even after Cry-gel is subjected to extreme temperatures of 80 °C and −40 °C for 4 h (Fig. 4c). This indicates the Cry-gel’s enhanced thermo-mechanical performance. Additionally, even in high-humidity conditions (relative humidity (RH) of 90%), Cry-gel still maintains impressive mechanical properties, exhibiting tensile strength values nearing 2 MPa (Supplementary Fig. 16).
a Nanoindentation load-displacement curves for LiBr gel and Cry-gel. b Tensile stress-strain curves for LiBr gel and Cry-gel at 25 °C. c Tensile stress-strain curves of LiBr gel and Cry-gel when exposed to −40 °C and 80 °C for 4 h, respectively. d Compressive stress-strain curves for LiBr gel and Cry-gel at 25 °C. e Compressive behaviors between LiBr gel and Cry-gel exposed to −40 °C and 80 °C for 4 h, respectively. f Comparison of compressive strength and tensile strength between reported gels and Cry-gel. g The Cry-gel (3 mm thick, 5 mm wide) lifting a 1 kg weight. h Photograph of Cry-gel supporting an adult with the weight of 47 kg. i Investigating the LED brightness using Cry-gel as a flexible conductor in different temperature environments. j Electrochemical impedance spectroscopy (EIS) curves for Cry-gel and LiBr gel in a room temperature environment. k EIS curves of LiBr gel and Cry-gel when exposed to −40 °C and 80 °C for 4 h, respectively. l Comparison of ionic conductivity between Cry-gel and previously reported ionogels. Data are presented as mean values ± SD, n = 3 independent samples.
While enhancing the compressive strength of gel materials generally presents more challenges than improving tensile strength, Cry-gel showcases exceptional compressive resistance (Supplementary Fig. 17). Compression stress–strain tests show that Cry-gel exhibits a maximum compressive stress of 5.3 MPa (Fig. 4d), which is five times higher than that of LiBr gel and 75 times higher than that of ZnCl₂ gel. Moreover, this superior performance is maintained under extreme temperatures of 80 °C and −40 °C, respectively (Fig. 4e). For example, after exposure to 80 °C and −40 °C for 4 h, Cry-gel maintains a compressive strength exceeding 3 MPa. Compared to those reported gel materials32,37,38,39,40,41, Cry-gel displays high tensile and compressive strength (Fig. 4f), easily lifting a 1 kg weight (Fig. 4g) and supporting an adult with the weight of 47 kg (Fig. 4h). In contrast, LiBr gel exhibits failure and damage behavior upon attempting to lift a 200 g weight (Supplementary Fig. 18).
The robust mechanical properties of Cry-gel can be attributed to network densification induced by cooling crystallization. We systematically investigated the mechanical performance of Cry-gels prepared under three distinct cooling conditions: rapid cooling at −20 °C, normal cooling at room temperature (25 °C), and slow cooling at 50 °C. As shown in Supplementary Fig. 19, the Cry-gel produced through rapid cooling at −20 °C demonstrates enhanced extensibility and flexibility. Conversely, the Cry-gel obtained via slow cooling at 50 °C exhibits superior mechanical strength and toughness. Notably, the Cry-gel prepared at room temperature of 25 °C, employed in this work, effectively combines the benefits of extensibility, flexibility, and pronounced mechanical strength and toughness. Additionally, Cry-gel displays a similar trend in compressive mechanical properties as it did in tensile properties across the three different cooling methods (Supplementary Fig. 19b). This indicates that cooling at room temperature is more conducive to crystalline growth and facilitates the assembly of cellulose molecules into a dense network within Cry-gel.
In addition to the thermal stability and mechanical strength, Cry-gel exhibits noteworthy ionic conductivity. As shown in Fig. 4i, Cry-gel can function as a flexible conductor in a closed electronic circuit even at the temperatures of 80 °C and −40 °C. From the electrochemical impedance spectroscopy (EIS) curves (Fig. 4j), the calculated ionic conductivity of Cry-gel is as high as 96.8 mS cm−1, exceeding the 63 mS cm−1 of LiBr gel. Additionally, the ionic electrical properties of Cry-gel demonstrate notable advantages in both high and low temperature environments, such as 80 °C and −40 °C (Fig. 4k), with ionic conductivity reaching 217 mS cm−1 and 43 mS cm−1 (Supplementary Fig. 20). Meanwhile, even exposed to RH of 90% for 12 h, Cry-gel still maintains high ionic conductivity of close to 80 mS cm−1 (Supplementary Fig. 16b). In comparison to other reported gels42,43,44,45,46, Cry-gel demonstrates its superior ionic conductivity (Fig. 4l).
Thermoelectric devices and self-powered sensors applications
Cry-gel exhibits high mechanical properties, self-healing abilities, high ionic conductivity, and an extensive operational temperature range, establishing itself as a promising candidate for applications in low-grade heat harvesting, smart sensors, and various flexible electronics. To assess the thermoelectric potential of Cry-gel, we conducted characterization experiments utilizing a custom-designed temperature gradient platform. When one terminal of Cry-gel is heated to temperatures of 314 K, 324 K, and 354 K, while the opposing terminal is maintained at 294 K, we observed stable output voltages of 0.13 V, 0.18 V, and 0.28 V, respectively (Fig. 5a). The corresponding currents measured were 2.6 µA, 3.1 µA, and 4.7 µA for the Cry-gel configuration (Fig. 5b). As shown in Supplementary Fig. 21, Cry-gel demonstrates real-time thermo-electrical responsiveness to dynamic variations in temperature difference. Additionally, even in environments with high RH of 90%, Cry-gel demonstrates impressive thermoelectric performance (Supplementary Fig. 16c, d). In comparison to other reported materials47,48,49,50,51, Cry-gel demonstrates significant advantages in thermoelectric performance, achieving a Seebeck coefficient of up to 6 mV K−1 (Fig. 5c). Furthermore, this Cry-gel could reproduce its thermoelectric properties through self-healing behavior (Supplementary Fig. 22), even if it becomes damaged during use.
a Voltage-time curves showing the thermoelectric response of Cry-gel at different temperature differences (ΔT). b Current-time curves showing the thermoelectric response of Cry-gel at different temperature differences (ΔT). c Comparison of the Seebeck coefficient between reported gel materials and Cry-gel. Data are presented as mean values ± SD, n = 3 independent samples. d Real-time infrared images of Cry-gel at ΔT = 130 K. e Investigating the power density of Cry-gel with different temperature differences. f Comparison of the ionic conductivity of Cry-gel at different temperatures. Data are presented as mean values ± SD, n = 3 independent samples. g Investigating the thermal conductivity of Cry-gel at different temperatures. Data are presented as mean values ± SD, n = 3 independent samples. h Temperature-time curves showing the temperature response of Cry-gel exposed to different solar radiation intensities. i Application potential of Cry-gel-based thermoelectric devices in smart buildings. The schematic diagram was conceived by the authors and created using Adobe Photoshop CC 2019 and 3ds Max 2018 software.
Infrared thermal imaging of Cry-gel under a temperature gradient indicates a ΔT of up to 130 K (Fig. 5d). Furthermore, its normalized power density exceeds 0.1 mW m−2 K−2, even at a ΔT of 60 K (Fig. 5e). In comparison to LiBr gel derived from a single-ion salt system, Cry-gel demonstrates enhanced electrical properties that improve with increasing temperature, as evidenced by the rise in ionic conductivity (Fig. 5f), which is particularly advantageous for thermoelectric devices. Thermal conductivity assessments of Cry-gel show a low value of 0.4 W m−1 K−1 (Fig. 5g). Moreover, Cry-gel exhibits thermal stability, maintaining its structural integrity across significant temperature gradients without shrinkage or melting, thereby ensuring consistent electrical output. Under Xenon plasma irradiation, Cry-gel sustains a stable temperature gradient at solar intensities of 1 and 2.4 (Fig. 5h), highlighting its potential for integration into thermoelectric systems in smart buildings (Fig. 5i).
In addition to its thermoelectric properties and derived applications, we also demonstrated the potential of Cry-gel in self-powered sensor technology. As illustrated in Fig. 6a, the Cry-gel-based sensor can simultaneously detect multiple stimuli while generating discernible electrical signal feedback. Even during substantial bending movements at the human wrist (Fig. 6b), the sensor maintains seamless interaction with human skin tissue, ensuring reliable and accurate signal feedback. Figure 6c−e further demonstrate that the self-powered sensor provides corresponding electronic signal feedback in response to varying bending amplitudes, mechanical stretching, and pressure.
a The diagram of self-powered sensors for signal detection. b Photographs of Cry-gel on the human arm under different bending amplitudes. c−i Current waveforms of Cry-gel sensing bending, stretching, pressure, breathing, touch, temperature, and human pulse, respectively. Each element in a and c−i was conceived by the authors and created using Adobe Photoshop CC 2019 software.
Additionally, this self-powered sensor is adept at detecting stimuli pertinent to human skin feedback, including respiration (Fig. 6f), touch (Fig. 6g), temperature (Fig. 6h), and pulse (Fig. 6i). Even when damaged, the self-powered sensor can regain its intelligent sensing capabilities through self-healing properties (Supplementary Fig. 23). Even at high temperature (80 °C) and low temperature (−40 °C), the Cry-gel sensor can still output stable bending stimulation signals (Supplementary Fig. 24). These characteristics underscore its potential for applications in wearable sensors, health monitoring, and smart electronic skin technologies.
Discussion
In this study, we demonstrate a significant advancement in cellulose ionogel design through dual-ion synergy and crystallization-guided assembly, effectively decoupling the classical trade-offs among mechanical robustness, ionic conductivity, and thermal stability. The Cry-gel achieves strong tensile strength (2.3 MPa), compressive strength (5.3 MPa), and ionic conductivity (96.8 mS cm−1), alongside operational stability across a wide temperature range (−40 to 80 °C). Mechanistic investigations reveal that Zn2+ disrupts Li+ clusters to form stable ion-cellulose-water complexes, while crystallization-induced densification enhances H-bonding, collectively reinforcing the hierarchical network, thereby enhancing the mechanical strength of Cry-gel. Beyond fundamental insights, Cry-gel exhibits notable thermoelectric performance (Seebeck coefficient: 6 mV K−1) and self-powered sensing capabilities, enabling applications in wearable electronics and low-grade heat harvesting. Furthermore, even if the Cry-gels and their flexible devices experience damage during use, their self-healing properties can be utilized to restore functionality.
This work not only provides a universal strategy for balancing multifunctionality in sustainable materials but also bridges the gap between laboratory innovation and real-world applications. Future studies could explore diverse ion combinations to generalize this design principle and investigate long-term cyclic stability for industrial scalability. By resolving the conflict between material stability and performance, our findings pave the way for cellulose-based systems to the frontiers of flexible and energy-efficient bioelectronics.
Methods
Materials
Cellulose is extracted from poplar flour with an average polymerization degree of around 830, a crystallinity of 64%, and a diameter of 50 − 250 µm52,53,54,55. Zinc chloride (99.7%) and lithium bromide (99%) were purchased from Aladdin (China).
Preparation of double ion salt solution
The 250 mL three-necked flask was thoroughly washed and dried prior to synthesis. LiBr, ZnCl2, and H2O were then added at a mass ratio of 65:4:35 and the mixture was stirred in the flask at 60 °C until the solution became crystal clear.
Preparation of LiBr salt solution and ZnCl2 salt solution
For the LiBr salt solution, LiBr and H2O at a mass ratio of 65:35 were mixed and stirred in a flask at 60 °C until the solution became crystal clear. Similarly, for ZnCl2 salt solution, ZnCl2 and H2O at a mass ratio of 65:35 were stirred in a flask at 60 °C until the solution became crystal clear.
Preparation of Cry-gel, LiBr gel and ZnCl2 gel
For Cry-gel, 3.5 g cellulose and 100 g double ion salt solution were mixed in a three-neck flask and stirred at 500 rpm at 100 °C for 3 h to form a homogeneous cellulose dissolving solution. The solution was then quickly poured onto a glass plate preheated to 100 °C and allowed to undergo cooling, gelation, crystallization, and moisture absorption to form Cry-gel (with H2O content of around 43.6 wt%). For LiBr gel, 3.5 g cellulose and 100 g LiBr salt solution were mixed in a three-neck flask and stirred at 130 °C for 40 min to form a homogeneous cellulose dissolving solution. The solution was poured onto a glass plate preheated to 100 °C and allowed to undergo cooling, gelation, crystallization, and moisture absorption to form LiBr gel. Similarly, 3.5 g cellulose and 100 g ZnCl2 salt solution were mixed in a three-neck flask and stirred at 100 °C for 40 min to form a homogeneous cellulose dissolving solution. Then, ZnCl2 gel was prepared following the procedures described above.
Ionic conductivity measurement
A sample disc 12 mm in diameter was sandwiched between two pieces of 16 mm stainless-steel sheets to assemble a device. The device was connected to the CHI660e electrochemical workstation (Chenhua Instruments, Shanghai, China) to test the electrochemical impedance spectrum. The ionic conductivity was calculated using the equation:
Where L (cm) is the thickness of sample in the button cell, A (cm2) is the effective cross-sectional area of the sample and Rb (Ω) is the bulk resistance of the sample.
Data availability
All data needed to evaluate the conclusions in the paper are present in the paper and Supplementary Information. All data are available from the corresponding author upon request, or on Figshare (https://doi.org/10.6084/m9.figshare.29632208).
References
Fan, X. et al. Ionogels: recent advances in design, material properties and emerging biomedical applications. Chem. Soc. Rev. 52, 2497–2527 (2023).
Long, Q., Jiang, G., Zhou, J., Zhao, D. & Yu, H. A cellulose ionogel with rubber-like stretchability for low-grade heat harvesting. Research 7, 0533 (2024).
Jiang, G., Xu, G., Xia, Q., Yu, H. & Zhao, D. Cellulose gels: functional design and promising smart applications. Small 21, 2412538 (2025).
Yang, M. et al. Robust mechanically interlocked network ionogels. Angew. Chem. Int. Ed. 64, e202423847 (2025).
Saifi, S. et al. An ultraflexible energy harvesting-storage system for wearable applications. Nat. Commun. 15, 6546 (2024).
Shin, Y. et al. Low-impedance tissue-device interface using homogeneously conductive hydrogels chemically bonded to stretchable bioelectronics. Sci. Adv. 10, eadi7724 (2024).
Li, P. et al. N-type semiconducting hydrogel. Science 384, 557–563 (2024).
Shi, L. et al. Highly stretchable and transparent ionic conducting elastomers. Nat. Commun. 9, 2630 (2018).
Yin, J. et al. Recent advances in self-powered sensors based on ionic hydrogels. Research 8, 0571 (2025).
Xiang, H., Li, X., Wu, B., Sun, S. & Wu, P. Highly damping and self-healable ionic elastomer from dynamic phase separation of sticky fluorinated polymers. Adv. Mater. 35, 2209581 (2023).
Kong, W. et al. Muscle-inspired highly anisotropic, strong, ion-conductive hydrogels. Adv. Mater. 30, 1801934 (2018).
Ye, Y. et al. Cellulose-based ionic conductor: an emerging material toward sustainable devices. Chem. Rev. 123, 9204–9264 (2023).
Yang, F., Cholewinski, A., Yu, L., Rivers, G. & Zhao, B. A hybrid material that reversibly switches between two stable solid states. Nat. Mater. 18, 874–882 (2019).
Picchio, M. L. et al. Mixed ionic and electronic conducting eutectogels for 3D-printable wearable sensors and bioelectrodes. Adv. Mater. Technol. 7, 2101680 (2022).
Li, X. et al. Stimulation-reinforced cellulose–protein ionogels with superior mechanical strength and temperature resistance. Adv. Funct. Mater. 34, 2408160 (2024).
Rong, Q., Lei, W., Huang, J. & Liu, M. Low temperature tolerant organohydrogel electrolytes for flexible solid-state supercapacitors. Adv. Energy Mater. 8, 1801967 (2018).
Tang, H. et al. High-strength, multi-mode processable bamboo molecular bioplastic enabled by solvent-shaping regulation. Nat. Commun. 16, 8729 (2025).
Yan, C. C. et al. Ionogels: preparation, properties and applications. Adv. Funct. Mater. 34, 2314408 (2024).
Wu, M. et al. A high-performance hydroxide exchange membrane enabled by Cu2+-crosslinked chitosan. Nat. Nanotechnol. 17, 629–636 (2022).
Zhang, J., Zhang, M., Wan, H., Zhou, J. & Lu, A. Coordinatively stiffen and toughen polymeric gels via the synergy of crystal-domain cross-linking and chelation cross-linking. Nat. Commun. 16, 320 (2025).
Liu, Q., Fang, Y., Xiong, X., Xu, W. & Cui, J. Ostwald ripening for designing time-dependent crystal hydrogels. Angew. Chem. 136, e202320095 (2024).
Zhang, X. F. et al. Inorganic salts induce thermally reversible and anti-freezing cellulose hydrogels. Angew. Chem. Int. Ed. 58, 7366–7370 (2019).
Tong, Z. et al. Super-robust cellulose rayon filaments engineered via molecular orientation-cross-linking assembly. Nano Lett. 25, 14489–14496 (2025).
Zhao, D. et al. A stiffness-switchable, biomimetic smart material enabled by supramolecular reconfiguration. Adv. Mater. 34, 2107857 (2022).
Jiang, G. et al. A scalable bacterial cellulose ionogel for multisensory electronic skin. Research 2022, 9814767 (2022).
Meng, J. et al. Nanocarbon catalysts with co-active S−P−C sites enhance metal-free direct oxidation of alcohols. SusMat 4, e221 (2024).
Yu, X. et al. Tunable glycyrrhizic acid supramolecular hydrogels via metal ion complexation. Giant 17, 100240 (2024).
Li, L. et al. Ultra-tough and recyclable ionogels constructed by coordinated supramolecular solvents. Angew. Chem. Int. Ed. 61, e202212512 (2022).
Chen, X., Bai, Y. K., Zhao, C. Z., Shen, X. & Zhang, Q. Lithium bonds in lithium batteries. Angew. Chem. Int. Ed. 59, 11192–11195 (2020).
Hou, T. Z. et al. Lithium bond chemistry in lithium–sulfur batteries. Angew. Chem. Int. Ed. 56, 8178–8182 (2017).
Tian, Z., Hou, L., Feng, D., Jiao, Y. & Wu, P. Modulating the coordination environment of lithium bonds for high performance polymer electrolyte batteries. ACS Nano 17, 3786–3796 (2023).
Wang, S. et al. Strong, tough, ionic conductive, and freezing-tolerant all-natural hydrogel enabled by cellulose-bentonite coordination interactions. Nat. Commun. 13, 3408 (2022).
Han, L. et al. Mussel-inspired adhesive and conductive hydrogel with long-lasting moisture and extreme temperature tolerance. Adv. Funct. Mater. 28, 1704195 (2018).
Ma, M., Shang, Y., Shen, H., Li, W. & Wang, Q. Highly transparent conductive ionohydrogel for all-climate wireless human-motion sensor. Chem. Eng. J. 420, 129865 (2021).
Cheng, W. et al. A general synthesis method for patterning PEDOT toward wearable electronics and bioelectronics. Research 7, 0383 (2024).
Li, W. et al. Supramolecular ionogels tougher than metals. Adv. Mater. 35, 2301383 (2023).
Xing, L. et al. A mussel-inspired semi-interpenetrating structure hydrogel with superior stretchability, self-adhesive properties, and pH sensitivity for smart wearable electronics. J. Mater. Chem. C. 11, 13376–13386 (2023).
Qian, Y. et al. Wide-temperature flexible supercapacitor from an organohydrogel electrolyte and its combined electrode. Chem. Eur. J. 29, e202300123 (2023).
Li, S.-N. et al. Environmentally stable, mechanically flexible, self-adhesive, and electrically conductive Ti3C2TX MXene hydrogels for wide-temperature strain sensing. Nano Energy 90, 106502 (2021).
Huang, S. et al. Molecular crowding agent modified polyanionic gel electrolyte for zinc ion batteries operating at 100 °C. Adv. Funct. Mater. 35, 2419153 (2025).
Bui, K. et al. Stretchable, healable, and weldable vitrimer ionogel for ionotronic applications. Chem. Eng. J. 474, 145533 (2023).
Zheng, Z. et al. Ethanol vapor-induced synthesis of robust, high-efficiency zinc ion gel electrolytes for flexible Zn-ion batteries. Small Struct. 5, 2400180 (2024).
Jiang, H. et al. Mechanically robust, highly conductive, wide-voltage cellulose ionogels enabled by molecular network reconstruction. Adv. Funct. Mater. 35, 2503512 (2025).
Ma, G. et al. A flexible supercapacitor based on deep eutectic solvent/[EMIM][TFSI] ionogel with high energy density and wide temperature range. ACS Appl. Electron. Mater. 6, 1434–1443 (2024).
Luo, Z., Li, W., Yan, J. & Sun, J. Roles of ionic liquids in adjusting nature of ionogels: a mini review. Adv. Funct. Mater. 32, 2203988 (2022).
Zhao, B. et al. Bioinspired conductive enhanced polyurethane ionic skin as reliable multifunctional sensors. Adv. Sci. 10, 2300857 (2023).
Shin, G. et al. Mechanically adaptable high-performance p(SBMA-MMA) copolymer hydrogel with iron (II/III) perchlorate for wearable thermocell applications. Adv. Funct. Mater. 35, 2412524 (2025).
Liu, Y. et al. Advanced wearable thermocells for body heat harvesting. Adv. Energy Mater. 10, 2002539 (2020).
Li, J. et al. High performance bacterial cellulose organogel-based thermoelectrochemical cells by organic solvent-driven crystallization for body heat harvest and self-powered wearable strain sensors. Adv. Funct. Mater. 33, 2306509 (2023).
Hu, Q. et al. Strong tough ionic organohydrogels with negative-thermopower via the synergy of coordination interaction and Hofmeister effect. Adv. Funct. Mater. 34, 2406968 (2024).
Zhao, W. et al. Metal-halogen interactions inducing phase separation for self-healing and tough ionogels with tunable thermoelectric performance. Adv. Mater. 36, 2402386 (2024).
Zhao, D. et al. A dynamic gel with reversible and tunable topological networks and performances. Matter 2, 390–403 (2020).
Zhu, Y. et al. A general strategy for synthesizing biomacromolecular ionogel membranes via solvent-induced self-assembly. Nat. Synth. 2, 864–872 (2023).
Zhang, Y. et al. All-cellulose hydrogel with ultrahigh stretchability exceeding 40000%. Mater. Today 74, 67–76 (2024).
Cao, K. et al. Bio-inspired multiscale design for strong and tough biological ionogels. Adv. Sci. 10, 2207233 (2023).
Acknowledgements
H.Y. acknowledges the support by the National Key Research and Development Program of China (No. 2023YFD2200504) and the National Natural Science Foundation of China (No. 32330072). D.Z. acknowledges the National Natural Science Foundation of China (No. 32171720 and 32371823), the Liaoning Province Xingliao Talents Leading Talent Program (No. XLYC2402043), and the project from Key Laboratory of Bio-based Material Science & Technology of Ministry of Education (Northeast Forestry University) (No. DLSWZ-01).
Author information
Authors and Affiliations
Contributions
H.Y. and D.Z. supervise the project. X.L., D.Z., and Z.T. designed the experiments. X.L. and Y.Q. carried out most of the experiments. Z.Z. participated in the experiments and contributed to the microscopic morphology analysis. S.Z. contributed to the analysis of MD and DFT simulation. X.L., D.Z., and H.Y. collectively analyzed all data and wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Informed consent
The authors affirm that human research participants provided informed consent for publication of the images in Figs. 1b, 4h, and 6b. Written informed consent was obtained from all volunteers who participated in this study.
Peer review
Peer review information
Nature Communications thanks Ferran Feixas and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, X., Tong, Z., Qiu, Y. et al. Balancing mechanical-thermal-electrical properties in cellulose ionogels via crystallization-induced molecular assembly. Nat Commun 16, 9970 (2025). https://doi.org/10.1038/s41467-025-64966-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-64966-y