Abstract
The entorhinal cortex (EC) is known to play important roles in spatial cognition of human behaviors. However, the functional contributions and the lateralization of EC are not well known. This paper proposed a new paradigm by employing non-invasive stimulation - Temporal interference (TI) - of human EC in left or right hemisphere under path integration (PI) tasks of sensory-driven and abstract processing. Five metrics were introduced to evaluate PI performance, with results revealing significant improvements after EC stimulation. Moreover, the left EC modulation achieved more enhanced performances of abstract processing, whereas the modulation of the right EC had better improvements in the sensory-driven condition. The results indicated that functional roles may differ in left and right EC under PI tasks. This study provided valuable insights into hemispheric specialization within EC and suggested potential pathways for developing early intervention strategies through the non-invasive modulation of EC.
Similar content being viewed by others
Introduction
Spatial cognition is a critical aspect of human intelligence, enabling individuals to perceive, interpret, and interact with their physical environment1. This cognitive process encompasses abilities such as spatial orientation, navigation, and the mental representation of spatial relationships2,3. Despite its significance, the neural mechanisms underlying spatial cognition are not yet fully understood. Research has identified key brain regions involved in spatial processing, including the entorhinal cortex (EC), located in the medial temporal lobe, which contains grid cells that provide a metric for spatial representation4,5,6. However, the precise interactions among these regions and the dynamics of their contributions to spatial cognition remain subjects of ongoing investigation.
Among all the spatial cognitive processes, path integration (PI) has been proven to be a fundamental cognitive process that enables individuals to navigate through continuously integrating sensory information regarding their movements and surroundings7. In a typical PI task, the current position is estimated in relation to an initial location, updating continuously as one moves through space8. It is particularly critical in environments with unfamiliar or absent landmarks9. Beyond navigation, PI also serves as a foundational mechanism for building cognitive maps by linking environmental cues to spatial location estimates. It plays a pivotal role in transferring spatial knowledge from cognitive maps10 to route-based knowledge and way-finding strategies11. Evidence from diverse animal studies7,12 as well as human research13 highlights the critical role of PI in spatial cognition.
In the existing literature, researchers have shed light on and addressed the importance of EC, in spatial processing, self-localization, and spatial mapping14,15,16, especially in PI tasks, which are thought to be subserved by EC12. For instance, disruptions to neural processing in the medial EC have been directly linked to impairments in PI behavior17. Human studies with fMRI analysis have also demonstrated that neural representations in EC correlate significantly with PI performance in virtual navigation tasks18. In addition, it has been documented that there is hemispheric asymmetry within EC, with volumetric studies showing that the left EC tends to be smaller than the right19, which underlies the potential functional differentiation in PI tasks. For instance, prior studies have reported greater involvement of the left EC in object encoding20, verbal learning19,21, and grid-like spatial coding22. Conversely, the right EC is preferentially associated with spatial encoding20, self-referential navigation23, and real-time spatial data integration24. However, the lateralized functional roles of the EC during PI tasks remain unclear.
Neuro-stimulation studies have provided further compelling evidence and pioneering methods, underscoring the significance of the EC’s functional contributions to PI processes in animal models8,25. However, there was limited research on human participants, with the causal relationship between EC and PI uncovered. It is notable that current mainstream neuromodulation, such as transcranial electric stimulation (tES) and conventional transcranial magnetic stimulation (TMS), are restricted to cortical regions due to the techniques per se26,27, making them ineffective for stimulating deep structures like the EC. Deep brain stimulation (DBS) has been applied in deep brain regions in humans19. However, its invasive nature limits its application to severely ill patients, such as those with Parkinson’s disease or Major Depressive Disorders. Thus, despite the availability of DBS for exploring the relationship between EC and PI tasks, it still raises concerns about the validity of causal inferences as conducted in patients. The neuro-modulation targeting ECs in healthy individuals can uncover their relationship with PI tasks. Nevertheless, no such research has been conducted to the best of our knowledge, possibly due to the limitations in current stimulation techniques.
The advent of temporal interference (TI) stimulation28 presented a promising solution to the challenge of non-invasively modulating deep brain regions such as EC. TI stimulation leverages the interference of two high-frequency alternating currents to create a low-frequency envelope field that can effectively stimulate deep neural structures while minimizing effects on overlying cortical areas28,29. This innovative approach can achieve a level of specificity and depth unattainable with traditional noninvasive methods, enabling precise modulation of deep brain regions. Previous studies have established the efficacy of TI stimulation across various animal models30,31,32,33. More recently, researchers have investigated TI stimulation in human participants, targeting diverse brain regions including the primary motor cortex (M1)34,35,36, right frontoparietal areas37, hippocampus38, right hippocampal-entorhinal regions39, striatum40,41, and the right globus pallidus internus42. Importantly, studies on humans have demonstrated the safety and tolerability of TI stimulation under specific parameters with only minimal, mild side effects and no significant adverse reactions38,43,44. Additionally, clinical studies employing TI stimulation in patients with Parkinson’s disease have also reported favorable tolerability profiles, underscoring the feasibility and clinical safety of TI as a therapeutic neuro-modulation approach42.
Therefore, this study aimed to investigate the functional contributions and the lateralization of EC in PI tasks. To this end, we developed a paradigm by employing TI stimulation to individually modulate the left or right EC in healthy participants as they performed two distinct PI tasks: a sensory-driven task (e.g., return-to-start task, characterized by first-person perspectives without spatial cues, mimicking egocentric navigation like blindfolded walking) and an abstract processing task (e.g., cognitive map task, presented from an aerial view with spatial cues, which inherently supports allocentric navigation and requires abstract spatial computation). Five metrics were designed to assess the effect on PI tasks, with Angle Deviation (Angle Deviation) and relative distance deviation (RDD) for sensory-driven tasks; average angle deviation (AAD), average relative distance deviation (ARDD), and overall distance deviation (ODD) for abstract processing tasks. Gleaned from the literature, we hypothesized that left EC stimulation would preferentially enhance abstract processing performance, while right EC stimulation would more selectively benefit sensory-driven PI tasks. In this case, enhancements would be observed more evident in the left EC stimulation group on AAD, ARDD, and ODD than right EC stimulation group in abstract processing tasks, whereas significant improvements would be observed more in the right EC stimulation group than left EC in sensory-driven tasks.
In summary, our findings successfully demonstrated a causal relationship between TI stimulation targeted at EC and PI abilities. Statistical results provided an indication that the left EC exhibited greater sensitivity to abstract processing tasks, while the right EC appeared more engaged in sensory-driven tasks. The present study provided evidence to systematically explore the functional role of the left and right EC in PI tasks. Although these results were observed in healthy adults, we believe this study opened new avenues for the development of early intervention strategies to mitigate PI impairments associated with mild cognitive impairment (MCI) and Alzheimer’s disease (AD) through TI modulation of the EC, thus holding great promise for future clinical applications.
Results
In this study, the effectiveness of TI stimulation on EC was verified through assessments of PI performance and MRI data analyses. Two experiments were designed to verify the effectiveness of TI stimulation on the EC regions during the PT tasks. First, PI performance improvement evaluations resulting from TI modulation of the EC were validated through designed behavioral tasks. Participants were administered to perform two PI tasks, that is, the return-to-start task and the cognitive map task, to evaluate their PI abilities. To quantitatively measure their performances, we developed five evaluation metrics that captured precisions of PI task execution from different aspects. Statistical analyses were conducted to test data homogeneity and to assess performance changes within and between groups, allowing us to determine the impact of TI stimulation on PI performance. Second, the accuracy of TI modulation on the EC regions was further validated by using electric field simulations and analysis of T1-weighted MRI data collected from participants. Additionally, we analyzed changes in functional MRI (fMRI) signals by computing connectivity matrices, which enabled us to evaluate the effectiveness of TI stimulation in modulating neural activities within the EC regions. By integrating behavioral assessments with neuroimaging analyses, we aimed to provide comprehensive results and evidence for the effectiveness of TI stimulation in enhancing PI performance and modulating EC activity. The results from both experiments contributed to our understanding of the potential of TI stimulation as a noninvasive method for targeting deep brain regions involved in PI tasks.
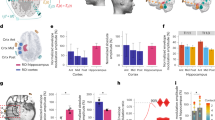
In behavioral experiments, sixty-three participants (35 female, 19–28 years old; M = 22.57, SD = 1.85) were randomly assigned into three distinct groups, namely, the left EC, the right EC stimulation, and the sham condition. In each experimental condition, participants were subjected to two PI tasks with five intermediate nodes separately. In each task, participants were required to return to the start cone in a 6 × 6 m from an egocentric point of view (Fig. 1a, b) which was identical to Howett et al.’s study13; in addition, participants were required to recall the missing cones (red cones in Fig. 2b) on a 6 × 6 m grid map based on two reference cones (grey cone in Fig. 2b) in which cognitive map was required. Angle deviation (Fig. 1c, with detailed descriptions in the “Methods” section (“Evaluation metrics”)) and RDD (Fig. 1d; see “Evaluation metrics” of “Methods” section) were calculated for each participant to measure the return-to-start task performance in each experimental condition. The cognitive map task performance was indexed by the AAD (Fig. 2c, with detailed descriptions in the “Methods” section (“Evaluation metrics”)), the ARDD (Fig. 2d), and the ODD (Fig. 2e). Mixed-design analysis of variance (ANOVA) was applied to these five metrics separately in each task with stimulation (pre- vs. post-stimulation) as a within-subject factor and stimulation type (left EC vs. right EC vs. sham) as a between-subject factor. The statistical results of return-to-start tasks and cognitive map tasks were separately demonstrated in the “Results” section (“PI performance in return-to-start tasks (sensory-driven)”, and “PI performance in cognitive map tasks (abstract processing)”). Both the left and right EC stimulation groups showed better performance after TI stimulation. The left EC stimulation group also demonstrated better performance in the cognitive map tasks, while participants receiving right EC stimulation exhibited enhanced performance more in the return-to-start tasks. The source data underlying Figs. 1 and 2 are provided in the Source Data file (Figure_1.xlsx and Figure_2.xlsx).
a The illustrative scene: participants were instructed to navigate from the starting cone zero to five sequential intermediate nodes. b The real VR scene: the VR view through the head-mounted display. The location marker was tagged with a label on top. c, d Schematic of the evaluation metrics for measuring the return-to-start task performance. c Angle deviation (AD): Angle A was formed by the real direction vector node zero and the vector from node four to node five. Angle B was formed by the participants' estimated position of node zero. ∣A–B∣ represents the calculation formula of angle deviation. d Relative distance deviation (RDD): The absolute distance \(| D-D{\prime} |\) between the actual distance (D) and the predicted distance (D') represents the relative distance deviation. e–g Behavioral results for the angle deviation (AD). 63 participants were recruited and randomly assigned to three groups with 21 replicates each. Statistical analysis was performed using one-way ANOVA followed by Bonferroni’s post hoc multiple comparisons test for (e) and (g). All tests were two-sided. Data are presented as mean values +/− SD. e Behavioral performance before TI stimulation (baseline) between the left, right EC, and sham group. f Comparisons between the pre- and post-stimulation stimulation in each stimulation group with two-sided paired t-tests (left group: t(20) = 3.077, p = 0.006, right group: t(20) = 3.105, p = 0.006). g The differences were calculated for each participant by subtracting the AD score obtained in post-stimulation from that obtained in pre-stimulation in the sham, left-, and right-EC stimulation group (two-sided ANOVA followed by Bonferroni’s post hoc comparisons, f(2, 60) = 4.155, p = 0.020, with post-hoc p = 0.034 between sham and right group). h–j The corresponding behavioral results for the relative distance deviation (RDD), with all settings similar to (e–g). i Comparisons between pre- and post-stimulation (left group: t(20) = 4.295, p < 0.001, right group: t(20) = 3.827, p = 0.001). j RDD differences between stimulation groups (f(2, 60) = 6.479, p = 0.003, post-hoc: sham to left: p = 0.003, sham to right: p = 0.031). Note: *≤ 0.05, **≤ 0.01, and ***≤ 0.001.
a The illustrative scene: A 6 × 6 grid presenting the cones from a top-down perspective. Participants were required to mark the remaining missing cones on the grid with two remaining reference cones. b The real VR scene: A control panel where participants received prompts to place missing cones from cone 1 to 5 in sequence. c–e Schematic of evaluation metrics for cognitive map task performance. 63 participants were recruited and randomly assigned to three groups with 21 replicates each. c Average angle deviation (AAD): The true node, estimated node, and two reference nodes formed angles A1 and A2. The average of these angles across all missing cones represented the AAD. d Average relative distance deviation (ARDD): The actual and estimated distances between the real and reference cones were averaged to calculate the ARDD. e Overall distance deviation (ODD): The Euclidean distances between the real cones and the remaining cones were compared with the estimated distances. The sum of these differences represented the ODD. f–h The corresponding behavioral results for the average angle deviation. Statistical analysis was performed using one-way ANOVA followed by Bonferroni’s post hoc multiple comparisons test for (f) and (h). All tests were two-sided. Data are presented as mean values +/− SD. e Behavioral performances before TI stimulation (baseline) in the left, right EC, and sham group with two-sided paired t-tests. f The pre- and post-stimulation behavioral performances in each stimulation group. g The differences calculated by subtracting the AAD performances obtained in post-stimulation from those in pre-stimulation across all stimulation groups. i–k The corresponding behavioral results for the average relative distance deviation, with all settings similar to (f–h). j Comparisons between pre- and post-stimulation (left group: t(20) = 3.147, p = 0.005, right group: t(20) = 2.739, p = 0.013). k ARDD differences between stimulation groups (f(2, 60) = 3.024, p = 0.056, post-hoc: sham to left: p = 0.052). l–n The corresponding behavioral results for the overall distance deviation, with all settings similar to (f–h). m Comparisons between pre- and post-stimulation (left group: t(20) = 2.838, p = 0.010, right group: t(20) = 2.939, p = 0.008). Note: *≤ 0.05, **≤ 0.01, and ***≤ 0.001.
PI performance in return-to-start tasks (sensory-driven)
Angle deviation and RDD were computed to evaluate the precision of sensory-driven PIs in return-to-start tasks. Angle deviation was calculated as the angle between two vectors: one extending from the endpoint to the actual starting point, and the other from the endpoint to the participant’s perceived starting point (see Fig. 1c). RDD was determined by calculating the ratio of the difference in length between these two vectors to the actual distance between the endpoint and the starting point (see Fig. 1d). The detailed computation methods for both evaluation metrics were provided in the “Methods” section (“Evaluation metrics”).
Angle deviation: A significant stimulation status main effect was observed (f = 6.01, p = 0.017, η2 = 0.09) with a significantly smaller angle deviation (AD, M = 21.65°, SD = 11.13) detected after stimulation as compared to that in the pre-test (M = 26.52°, SD = 12.39). However, there was no main effect of stimulation, with f = 2.14, p = 0.127, η2 = 0.07. Comparable performance were observed in the left EC (M = 21.90°, SD = 10.64), right EC stimulation condition (M = 23.35°, SD = 11.25) as well as the sham condition (M = 26.99°, SD = 13.56). Interestingly, stimulation status × stimulation type interaction was found on angle deviation, f = 4.16, p = 0.020, η2 = 0.12. As shown in Fig. 1e, one-way ANOVA was conducted on the pre-test and revealed no stimulation type simple effect, with f = 0.27, p = 0.763. This suggested the homogeneity of participants assigned in the left (M = 26.04°, SD = 12.94), right EC stimulation group (M = 28.12°, SD = 12.87) as well as in the sham condition (M = 25.39°, SD = 11.77). Paired t-test was further conducted, illustrated in Fig. 1f and revealed the systematical difference between stimulation types: the angle deviation participants can detect was significantly smaller after TI stimulation on either left (M = 17.76°, SD = 5.37) or right EC (M = 18.59°, SD = 6.80) as compared to their pre-stimulation performance (left EC: M = 26.04°, SD = 12.94; right EC: M = 28.12°, SD = 12.87), both ts ≥ 3.08, ps ≤ 0.006. However, such stimulation effect was not evident in the sham condition, showing comparable performance in pre- (M = 25.39°, SD = 11.77) and post-stimulation phases (M = 28.59°, SD = 15.26), t = −0.74, p = 0.470. To further characterize the modulation effect of EC on PI via TI, the difference of angle deviation was calculated for each participant by subtracting the performance from post- by pre-stimulation, as illustrated in Fig. 1g. One-way ANOVA was again applied and revealed main effects of stimulation type, f = 4.16, p = 0.020, with significantly larger improvements observed in the right EC stimulation (M = −9.53°, SD = 14.07) than sham condition (M = 3.20°, SD = 19.92), p = 0.034. Though the improvements were numerically larger after the stimulation on the left EC (M = −8.28°, SD = 12.33) than the sham condition, this did not reach the statistical significance level, p = 0.065. No statistical difference was observed between right and left EC stimulation groups as well, p = 1.000.
Relative distance deviation: The RDD was significantly reduced after TI stimulation (M = 0.20 m, SD = 0.09) compared to the pre-test values (M = 0.23 m, SD = 0.08), indicating a considerable main effect of the stimulation status (f = 11.64, p = 0.001, η2 = 0.16). However, no significant differences were found among different types of stimulation (f = 2.09, p = 0.132, η2 = 0.07), with comparable performance in the left EC stimulation (M = 0.20 m, SD = 0.07), right EC stimulation (M = 0.20 m, SD = 0.07), and the sham conditions (M = 0.24 m, SD = 0.11). Notably, a significant interaction between stimulation status and stimulation type (f = 6.48, p = 0.003, η2 = 0.18) was detected for RDD. As illustrated in Fig. 1h, the application of one-way ANOVA revealed the homogeneity of the groups in the baseline condition (before TI stimulation), with f = 0.03, p = 0.971. The averaged RDD was around 0.20 m in all the three groups (left-EC: M = 0.20 m, SD = 0.07; right-EC: M = 0.20 m, SD = 0.07; sham: M = 0.24 m, SD = 0.11). Interestingly, when paired t-tests were carried out on RDD before and after TI stimulation in each group, systematic differences were detected under different stimulation types. As shown in Fig. 1i, dramatic enhanced performance was detected after TI stimulation on both left- (M = 0.16 m, SD = 0.04) and right-EC (M = 0.18 m, SD = 0.07) than their corresponding baseline performance (left EC: M = 0.23 m, SD = 0.07; right EC: M = 0.23 m, SD = 0.08), both with ts ≥ 3.83, ps < 0.001. However, no significant change was observed in the sham condition, t = −0.71, p = 0.486, with similar performance in the pre- (M = 0.23 m, SD = 0.12) and post-stimulation phases (M = 0.25 m, SD = 0.11). In addition, a one-way ANOVA was employed on the difference performance with stimulation type as between-subject factor (left-, right-EC and sham condition) and revealed a main effect of stimulation type, f = 6.48, p = 0.003, as illustrated in Fig. 1j. Statistically significant improvements were observed in the left- (M = −0.07m, SD = 0.08) and the right-EC stimulation group (M = −0.05 m, SD = 0.06) compared to the sham condition (M = 0.02 m, SD = 0.10), both with ps ≤ 0.031, while no statistical difference was detected between the left and right EC stimulation groups (p = 1.000), further confirming the effect of EC modulation on PI performance.
PI performance in cognitive map tasks (abstract processing)
To evaluate the precision of abstract processing PIs in cognitive map tasks, we computed three evaluation metrics as follows, whose schematics were illustrated in Fig. 2a, b. The AAD was computed as the mean angle between the actual cones and the two reference cones, as well as the estimated true cones and the reference cones. The ARDD was calculated as the mean of the RDDs for all these lines. The ODD was determined by averaging the differences between the true cones and all remaining true cones, as well as the estimated cones and all remaining true cones. The detailed computations for these evaluation metrics were demonstrated in the “Methods” section (“Evaluation metrics”). The baseline values across the three stimulation groups, along with their respective pre- and post-stimulation measurements and the corresponding change scores (post minus pre), were illustrated in Fig. 2f–n, respectively.
Average angle deviation: A significant main effect of the stimulation status was observed (f = 11.64, p < 0.001, η2 = 0.16). As illustrated in Fig. 2g, participants can detect a significantly smaller AAD (M = 22.84°, SD = 11.14) after stimulation as compared to that in the pre-test (M = 28.35°, SD = 11.60). However, there were no main effect of stimulation type, f = 0.00, p = 1.000, η2 = 0.00, with comparable performance observed in the left EC (M = 25.57°, SD = 13.79), right EC stimulation condition (M = 25.60°, SD = 11.75), as well as sham condition (M = 25.61°, SD = 9.28). In addition, no significant interaction between stimulation status and stimulation type was found in AAD, f = 0.88, p = 0.420, η2 = 0.03.
Average relative distance deviation: There was a notable main effect of stimulation status being identified (f = 14.57, p < 0.001, η2 = 0.20), revealing a significantly enhanced performance as indexed by ARDD after stimulation (M = 0.14 m, SD = 0.05) than that in the pre-test (M = 0.18 m, SD = 0.05). Furthermore, a significant main effect of stimulation type was evident on ARDD (f = 4.00, p = 0.023, η2 = 0.12). The ARDD performance was significantly better in the left-EC stimulation group (M = 0.15 m, SD = 0.07) than in the sham condition (M = 0.18 m, SD = 0.04), p = 0.041. The ARDD performance was also better in the right-EC stimulation group (M = 0.15 m, SD = 0.05) than the sham group, p = 0.067, whereas no such group difference appeared in the right-EC and the left-EC stimulation groups. Additionally, a marginal interaction between stimulation type and stimulation status was present on ARDD (f = 3.02, p = 0.056, η2 = 0.09). As depicted in Fig. 2i, no main effect of stimulation type was observed among the base values (f = 0.19, p = 0.825), confirming the homogeneity among participants in the left EC (M = 0.18 m, SD = 0.08), right EC (M = 0.17 m, SD = 0.04), and sham conditions (M = 0.18 m, SD = 0.04). The following paired t-test demonstrated systematic differences between pre- and post-stimulation performances under different stimulation types. After TI stimulation, the performance remarkably increased as indexed by ARDD for both the left EC (M = 0.12 m, SD = 0.03) and the right EC group (M = 0.13 m, SD = 0.05) as compared to baseline performance, both ts ≥ 2.74, ps < 0.013, as depicted in Fig. 2j. In contrast, similar levels were observed in pre- (M = 0.18 m, SD = 0.04) and post-stimulation performance (M = 0.17 m, SD = 0.41) in sham condition, t = 0.43, p = 0.674. Moreover, as depicted in Fig. 2k, main effects of stimulation type (f = 3.02, p = 0.056) existed when considering the performance difference between pre- and post-stimulation in each group (right-, left-EC and sham condition). Participants showed significant improvements noted in the left EC stimulation (M = −0.06 m, SD = 0.08) group than the sham condition (M = −0.01 m, SD = 0.06), p = 0.052. Similar performance was also observed in the right EC group exhibiting numerically larger improvements (M = −0.04 m, SD = 0.06) than the sham condition, while did not reach a statistically significant difference level, p = 0.459. Other than that, no statistical difference was observed between the right and left EC stimulation groups, p = 0.967.
Overall distance deviation: A significantly enhanced performance was detected after stimulation (M = 1.91 m, SD = 0.58) than that in baseline (M = 2.31 m, SD = 0.60) as indexed by the ODD, f = 13.98, p < 0.001, η2 = 0.19. In addition, main effects of stimulation type emerged as a marginal significance on ODD, f = 3.27, p = 0.045, η2 = 0.10, while revealing no statistical difference between each two groups, all ps ≥ 0.077. The interaction effect of stimulation status × type appeared borderline significance in ODD, f = 3.10, p = 0.057, η2 = 0.09. As depicted in Fig. 2l, no main effect of stimulation type was observed in baseline performance among left- (M = 2.31 m, SD = 0.72), right- EC stimulation group (M = 2.32 m, SD = 0.56) as well as the sham condition (M = 2.29 m, SD = 0.54), f = 0.00, p = 0.990, suggesting the potential homogeneity of participants. Succeeding paired t-tests, illustrated as Fig. 2m, further revealed systematic differences between pre- and post-stimulation performances in each stimulation group. The performance was significantly enhanced after TI stimulation on either left (M = 1.71 m, SD = 0.53) or right EC stimulation groups (M = 1.74 m, SD = 0.56) as compared to their corresponding baseline performance, both with ts ≥ 2.84, p < 0.010. However, this effect was not significant in the sham condition, showing comparable performance in pre- (M = 2.29 m, SD = 0.54) and post-stimulation phases (M = 2.26 m, SD = 0.49), t = 0.20, p = 0.842. To further explore the modulation effect of EC on PI performance, one-way ANOVA was again applied to the difference in performance between pre- and post-stimulation, considering stimulation type as a between-subject factor (left-, right-EC, and sham condition). No main effects of stimulation type were presented on ODD difference, as illustrated in Fig. 2n, f = 3.01, p = 0.057. Participants did not show significant improvements in the left EC stimulation (M = −0.60 m, SD = 0.97) group compared to the sham condition (M = −0.03 m, SD = 0.66), p = 0.102. Similar performance was also detected between the right EC (M = −0.57 m, SD = 0.89) group and the sham stimulation groups, p = 0.126. Moreover, the left and right EC stimulation groups also revealed no statistical differences, p = 1.000.
The electric field estimates for TI stimulation of the ECs
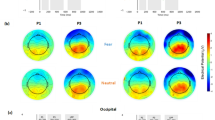
To further observe the accuracy and efficacy of EC-targeted stimulation, another 32 healthy participants (20 females; 19–30 years; M = 23.63, SD = 3.17) were recruited in the present experiment (see the “Methods” section (“Participants”)). To validate the effectiveness of the TI stimulation protocols, participants underwent structural T1-weighted magnetic resonance imaging (MRI) scans. Electric field simulations were calculated on the T1-weighted images for each participant (see the “Methods” section (“MRI Acquisition”)). Comparisons were made to explore the simulated amplitude envelope between the target stimulation area (EC) and the overlying cortical areas, as well as the average envelope electric field values (see the “Methods” section (“TI modulation ECs via electric field simulation”)). As shown in Fig. 3e and h, significantly higher amplitude envelopes were detected in the ROI than in the corresponding overlying cortical areas. The source data are provided in the Source Data file (Figure_3.xlsx).
a Schematic of TI stimulation. Two electrode pairs with each sharing the same current and frequency but with opposite poles. Frequencies f1 and f2 were assigned to currents I1 and I2, with a difference Δf = f2–f1. b Electrode placements for the left EC stimulation protocol. 1A, 1B and 2A, 2B respectively represented the two electrode pairs. The placement followed the international 10–10 system, with positions at C5, CP5, FT10, and F8. c Electrode placement for the right EC stimulation protocol with positions at F9, FT9, CP4, and C6. d–f Envelope simulation results for left EC stimulation protocol with 32 participants recruited. d Axial view with two cortex ROIs under electrodes (C5, CP5) in the left hemisphere. e Average absolute envelope intensities comparisons between the left EC and cortex ROI1 (t(30) = 2.193, p = 0.036), cortex ROI2 (t(30) = 2.879, p = 0.007), and the whole brain average (t(30) = 3.454, p = 0.002). All statistical comparision were done through two-sided paired t-tests with no adjustments. Data are presented as mean values +/− SD. f Group-averaged voxel-wise values of the absolute envelope values. g–i Results under right EC stimulation protocol with all settings similar to (d–f). h Envelope intensities comparisons between the right EC and cortex ROI1 (t(30) = 8.039, p < 0.001), cortex ROI2 (t(30) = 9.072, p < 0.001), and the whole brain average (t(30) = 5.425, p < 0.001). j–k Resting-state fMRI results under left stimulation protocol. The sham group with n = 9 participants, left group n = 11, and right group, n = 12. j FC comparisons between the left stimulation group and sham group. The alternations from before to during stimulation of FCs between left EC and other ROIs, together with the alternations of FCs between right EC and other ROIs. k Classification accuracy of FC differences between left EC and sham condition, calculated by subtracting data before TI stimulation from during stimulation. Box plots indicate median (middle line), 25th, 75th percentile (box) and min to max (whiskers) as well as all data points. l–m Resting-state fMRI results under the right EC stimulation protocols, with all settings similar to (j–k). Note: *≤ 0.05, **≤ 0.01, and ***≤ 0.001.
Individualized head models were constructed using the T1-weighted MRI data collected from all the participants. Simulation software was utilized to model the magnitude of the amplitude modulation of the electric fields across the whole brain for each subject under the specific stimulation protocols. Each participant underwent simulations for both the left EC stimulation and right EC stimulation protocols, resulting in two separate simulation outputs. Using the Brainnetome Atlas45, ROI-based signal extraction was performed on the simulation results to quantify the electric field intensity in the target and overlying cortex regions. Additionally, two overlying cortex regions near the electrodes, located in the same hemisphere as the target stimulation ROI, were selected for each stimulation protocol. For the left EC stimulation protocol, as depicted in Fig. 3d, ROI 145 (cortex ROI 1; light blue area) and ROI 141 (cortex ROI 2; dark blue area) underneath the electrodes (C5, CP5; dark blue) were selected as the overlying cortex, in which simulated envelope results was extracted and compared with that in the left-EC (the target ROI; purple area). For the right EC protocol (see Fig. 3g), ROI 146 (cortex ROI 1; light blue area) and ROI 142 (cortex ROI 2; dark blue area) underneath the pair of electrodes (CP4, C6; dark blue) were selected as the cortex ROI to compare the envelope value with that in right-EC (target ROI; purple area).
The absolute envelope electric field intensities at the two cortex ROIs and the targeted ROI were calculated for each participant, respectively. Repeated-measure ANOVA was applied on the envelope intensities with cortex as a within-subject factor (cortex ROI 1 vs. cortex ROI 2 vs. target ROI) for left- and right-EC protocol, respectively. As illustrated in Fig. 3e and h, the main effects of cortex were evident in both left- and right-EC protocols, both fs ≥ 6.64, ps ≤ 0.002. As revealed by post-hoc analysis, for left-EC protocol (Fig. 3e), the absolute envelope field intensity in target ROI (purple bar; M = 0.09; SD = 0.02) was significantly higher than those in both cortex ROI 1 (dark blue bar: M = 0.08, SD = 0.02), t = 2.88, p = 0.007, and ROI 2 (light blue bar: M = 0.08, SD = 0.02), t = 2.19, p = 0.036. Also, the average envelope values across the whole brain (green bar: M = 0.07, SD = 0.01) were calculated, and paired t-test analysis was applied on the average envelope value between the whole brain average and the targeted ROI with t = 4.37, p < 0.001. Similar observation was also observed in the right-EC protocol (Fig. 3h), with significantly higher envelope intensity in target ROI (purple bar: M = 0.10, SD = 0.03) than the two cortex ROI (ROI 1: dark blue bar; M = 0.05; SD = 0.01; ROI 2: light blue bar; M = 0.06; SD = 0.01), and the whole brain average (green bar: M = 0.08, SD = 0.01) all with ts ≥ 7.73, ps < 0.001. These findings provide evidence that the TI stimulation protocols, whether targeting the left or right EC regions, were effective in delivering focused stimulation to the intended deep brain regions while minimizing the spread of the envelope field to overlying cortical areas. The rendered left and right EC regions with voxel-wise envelope electric field values were demonstrated in Fig. 3f and i accordingly.
Functional connectivity changes via resting fMRI data analysis
Participants recruited for electric simulation phases (32 healthy participants, 20 females; 19–30 years; M = 23.63, SD = 3.17)) also underwent resting-state fMRI scans and were randomly assigned to three groups, left-EC group (12 participants; 8 female; 19–28 years; M = 23.80, SD = 3.13), right-EC (11 participants; 6 female; 19–30 years; M = 23.85, SD = 3.05) and sham group (9 participants; 6 female; 20–28 years; M = 23.71, SD = 3.13). As our interest area, two distinct TI stimulation protocols were applied to target the left and right EC regions, respectively for left-EC and right-EC groups. For the sham group, a frequency of 2000 Hz and a current of 1.5 mA were applied to both pairs of electrodes. Participants were randomly assigned such that half received electrode placements according to the left stimulation scheme, while the other half followed the right stimulation protocol. For all three groups, resting-state scans were acquired before and during the TI stimulation (see the fourth subsection (“MRI Acquisition”) of the “Methods” section).
Functional connectivity (FC) differences were calculated by subtracting FCs obtained during TI stimulation from those before TI stimulation. These differences in the sham group were further subtracted from the true stimulation group for further exploration. Fig. 3j depicted the FC differences between the left stimulation group and sham group under the left EC stimulation protocol. FCs between left EC and other brain regions (left) and those between right EC and other brain regions (Middle) were depicted. Fig. 3l illustrated the FC differences between the right stimulation group and sham group under the right EC stimulation protocol. The strength of connectivity between the EC and other brain regions was indicated by the thickness and color intensity of the connecting lines, where thicker lines and more intense green coloration represented stronger FC. As depicted in Fig. 3j and l, compared with sham group, FC values between EC and other brain areas were enriched by TI stimulation. Additionally, when stimulating the left EC, the FC of the left EC was enhanced more than the right EC, and vice versa. To further substantiate the effectiveness of the stimulation protocols, machine learning techniques were employed to classify the FC differences between the left-EC and sham group, and so as between the right-EC and sham group. A Random Forest classifier was utilized with a fivefold cross-validation approach to assess whether the stimulation-induced discernible changes in neural activity patterns. As shown in Fig. 3k, the classification accuracy between the left EC stimulation and sham stimulation conditions was 52.5% ± 0.018 (mean ACC ± SEM). Also, the accuracy between the right EC stimulation and sham stimulation conditions was 52.8% ± 0.014, as depicted in Fig. 3m. Both of these values exceed the 50% threshold, suggesting that the Random Forest classifier could distinguish between the stimulated and sham conditions. These findings implied that the TI stimulation protocols, targeting either the left or right EC, produced alterations in resting-state FC patterns. Perhaps, it provided further evidence that TI stimulation can modulate FC in a specific manner, such that TI stimulation to the left EC modulated left connectivity more than right and vice versa.
Discussion
The present study aimed to explore the causal relationship between left- or right-EC and spatial cognition in humans via an advanced non-invasive neuromodulation technology—TI stimulation. Our findings contribute to this area by providing empirical evidence that the left and right EC may not have identical roles in PI tasks and lay the foundation for further research in humans. Following methodologies similar to those of Zhu et al.35 and Violanie et al.38, we recruited healthy young adults for TI stimulation targeted at EC in each hemisphere. First, the effectiveness and accuracy of TI stimulation on EC were verified through electric field simulation and the resting-state fMRI signals comparison before and during TI stimulation. Similar to Howett’s paradigm13, participants were asked to complete two tasks in PI: the return-to-start tasks (the sensory-driven reference frame) and the cognitive map tasks (the abstract processing reference frame) in which the real path was recorded and five metrics were calculated for each participant. In our study, we compared behavioral performances in the sham group, left or right EC stimulation group both before and after TI stimulation to analyze the effect of left- or right-EC stimulation via TI on PI performances. Angle deviation, RDD, AAD, average distance deviation, and ODD were calculated for each participant in which angle deviation and RDD were indices for the return-to-start task, whereas AAD, average distance deviation, and ODD were indices for the cognitive map task. To further explore the differences caused by the TI intervention, the values were calculated by subtracting the signals after TI stimulation by the signals before TI stimulation for each index. Comparisons were conducted on these index differences between the sham, left, and right EC stimulation groups.
Conventional TMS techniques46 can only stimulate the cortical area with a depth of 0.7–2.5 cm47. While some TMS with Hesed coils (H-coils)48 could reach deeper brain regions with a depth of 4–6 cm49 as they reported, no practical experiments have been conducted to modulate EC. Besides, direct TMS of targets at depths of 4 cm or more can result in superficial stimulation strength beyond safety guidelines49,50. Compared to TMS, TI stimulation can provide effective modulation of deeper structures while substantially reducing unintended cortical stimulation, thereby enhancing the potential for more focused interventions of deep brain regions, such as EC.
Consistent with previous studies highlighting the role of the right EC in PI51,52, our findings provide additional evidence supporting its critical function in this domain. Specifically, in the return-to-start task, similar to the results reported by Newton et al.53 and Howett et al.13, the experiment group receiving TI stimulation on the right EC exhibited significant performance enhancements compared to the sham group without TI stimulation. Angle deviation and RDD showed statistically significant improvements, indicating that TI stimulation effectively modulated the right EC to enhance PI abilities. However, when considering the cognitive map task, which could be evolved from PI system2,54, the results were more nuanced. Although there was an increase in performance after stimulation in two of the three evaluation metrics, these improvements did not reach statistical significance when compared to the sham condition. This suggested that while TI stimulation of the right EC can enhance certain aspects of PI performance, its effect on tasks requiring cognitive mapping may be limited or require further investigation. These findings underscore a high degree of causality between the modulation of the right EC and PI tasks, particularly in sensory-driven navigation tasks, as also indicated by previous research55.
Furthermore, we investigated the modulation effects of TI stimulation on the left EC, which has also been associated with PI processes18,56. Remarkably, all PI performance metrics—except for AAD—showed significant enhancements following TI stimulation of the left EC. Additionally, comparisons between the left EC stimulation group and the sham group revealed significant improvements in the cognitive map tasks. This indicated that TI stimulation can effectively enhance PI performance related to the left EC, suggesting its involvement in abstract processing PI tasks. It is noteworthy that the left and right EC are thought to have different functions57, with lateralization effects influencing PI strategies. Bierbrauer et al.18 reported that only the right posterior medial EC (pmEC) exhibited significant hexadirectional symmetry of pattern similarity. However, few studies have directly confirmed similar lateralized functions in humans.
Despite the promising results of the present study, there are several limitations of TI stimulation. First, TI stimulation exhibits inherent precision constraints because the envelope amplitude area is often much larger than the intended target region. Second, TI stimulation can also elicit off-target effects, producing BOLD increases or decreases in brain regions that are both adjacent to and distant from the intended target. (off-target areas, see Supplementary Fig. 4)43. These effects arise because the stimulation strategy optimizes for maximum intensity in the target region without explicitly minimizing stimulation in surrounding neural structures. To alleviate these limitations, there are two possible strategies for enhancement. One strategy is to introduce more pairs of electrodes to refine the focality. As shown in Lee et al.58, their simulation of the envelope amplitude under multipair transcranial TI stimulation revealed that introducing electrode pairs could achieve more focalized stimulation of deep brain regions. Another strategy is to use the focused ultrasound (FUS), characterized by its non-invasiveness, high spatial precision, and deep-tissue penetration to enhance the spatial specificity of TI stimulation. Integrating FUS with TI could also provide more precise neuromodulation of targeted brain regions. Future studies could be conducted to further validate these possible solutions in practice.
Regarding the modulation of the EC, the results of this study cannot be directly interpreted as indicating changes in EC activity. There are two plausible mechanisms through which our stimulation protocol might influence PI performance. One possibility is that the 40 Hz neural oscillations induced by stimulation directly modulated the EC, resulting in observed cognitive enhancements. Alternatively, stimulation could have induced changes in neural oscillations across multiple brain regions, which in turn led to the observed changes in PI tasks. Although these two possibilities do not contradict each other, we cannot rule out the existence of the second possibility based on the present evidence in this study.
Additionally, the sample size in this study remains a limiting factor. The relatively small sample size means that the findings may not be fully generalizable and could introduce bias. However, the observed behavioral changes in this sample offer valuable insights into the potential effects of TI stimulation on spatial perception. These results lay the groundwork for future studies with larger sample sizes, which will be essential to further validate and expand upon these initial findings. As such, the small sample size is not seen as a critical drawback, but rather as an opportunity to guide future research aimed at refining the effects of TI on cognitive function.
Overall, TI stimulation has been proven to be well tolerated and safe to be applied in humans, with minor side effects reported after stimulation34,38,41. The bilateral ECs play a crucial role in the spatial-related abilities, especially in PI tasks25,59, not only in rodents60 but in humans as well13,23,61. Dysfunctions in the EC are highly correlated with neurodegenerative conditions such as AD and MCI62. Utilizing TI to stimulate either left or right EC provides a new potential method for modulating PI behavior and presents promising prospects for the diagnosis and treatment of MCI in the future. Future studies could extend the application from healthy participants, as presented in this paper, to patient populations for direct observation. Such research could facilitate early diagnosis of AD and contribute to the development of effective therapeutic interventions targeting either the left or right EC regions.
Methods
Participants
Sixty-three participants (35 female, 19–28 years old; M = 22.57, SD = 1.85) were recruited for the behavioral experiments validating the improvements in PI performances after TI stimulation. All participants were accounted for in the analysis. All participants were right-handed, with education year ≥ 12, and had no self-reported history of neurological or psychiatric disorders. None of the participants had heart, lung, or brain metal implants, and all exhibited unimpaired basic cognitive abilities, such as hearing and vision. Additionally, none of the participants suffered from vertigo63 or Meniere’s syndrome64. All participants were informed of potential risks and signed the informed consent form. No participants reported any adverse reactions. Sample sizes were not predetermined using statistical methods.
Besides, thirty-two participants (20 female; 19–30 years old; mean age ± s.d. 23.63 ± 3.17) were recruited for the MRI experiments to ensure the effectiveness and accuracy of TI stimulation on EC regions. All participants were right-handed and with education year ≥ 12. They reported no history of neurological or psychiatric disorders. Additionally, all participants confirmed that they had not worked in metal environments, did not suffer from claustrophobia, nephritis, or other kidney disease, had no history of surgery or head trauma, did not have Meniere’s syndrome or eye injuries, were not pregnant or breastfeeding, and had no history of stroke, fainting, epilepsy, or immune deficiencies that would increase infection risk. To avoid imaging artifacts, participants were screened to ensure they did not have any metallic or electronic implants, such as pacemakers, heart stents, cochlear implants, joint replacements, or tattoos. They also confirmed they were not carrying any magnetic substances like watches, mobile phones, keys, or jewelry, and were advised not to wear clothing with metal accessories or use contact lenses, false eyelashes, or other similar items. Before the MRI scan, all participants signed the informed consent form, a safety checklist, and provided informed consent, indicating they had been fully informed about the study’s purpose, procedures, potential risks and side effects, and benefits and costs. Only two participants assigned to the sham condition reported sensations of a strong electrical current. All the participants confirmed that they had received satisfactory answers to their questions and were willing to participate in this study. According to the exclusion criteria of the nuclear magnetic experiment, no participants were excluded from data analysis. Also, no participants were excluded under the head movement exclusion criteria when processing data collected.
All data used in this study were collected under ethical approval from the Shanghai Mental Health Center, Ethics Committee of Shanghai Mental Health Center (Approval Number: 2021-55C2). Additionally, all participants provided written informed consent and received monetary compensation for their participation. This study is in line with the Declaration of Helsinki. All data generated in this study are provided in the Supplementary Information (source data.zip).
TI protocol
TI stimulation was delivered using the TI stimulation device made by Soterix Medical (New York, USA), which was designed to generate kHz currents through two electrically isolated current sources. The bus electrode was configured with four electrode positions, namely e1, e2, e3, and e4. Electrode pair e1 and e2, and electrode pair e3 and e4, were two balanced pairs of current sources and were driven in opposite phases, providing two sinusoidal waveforms (2 kHz and 2.040 kHz). High Definition—Interferential Stimulation (HD-IFS) System was utilized with HD electrodes held in HD1 electrode holders embedded in a 10–1065 HD-Cap (Soterix Medical, USA). Each HD electrode holder was fully filled with conductive gel (HD-GEL, Soterix Medical, USA), ensuring that the entire available scalp contact area was utilized. Two pairs of stimulating electrodes were placed in rounded HD-tES MRI Electrode Holders with a base diameter of 25 mm and a height of 10.5 mm. These holders were positioned on the participants’ heads. Participants wore a brain cap with pre-defined electrode position holes according to the international 10–10 system65, plus an additional 10 positions, totaling 74 electrode positions. Conductive pastes were applied between the electrodes and the corresponding scalp area to reduce impedance and enhance stimulation efficacy.
Two different electrical stimulation schemes were introduced, targeting the left and the right EC separately. The stimulation protocols were initially obtained by the HD-Targets software (Soterix Medical, USA, https://www.soterixmedical.com/research/software/hd-targets) to determine the electrode locations for the TI protocol. Then, the electrode locations were optimized using the strategy and the optimization function in ref. 66. In the left EC stimulation protocol, the electrodes were placed at positions C5 and CP5, and positions FT10 and F8, corresponding to e1, e2, e3, and e4. The e1 and e2 shared one pair of electrodes driven in opposite phases and the same for e3 and e4. Both electrode pairs (e1 and e2, e3 and e4) received a current of 1.5 mA (peak-to-peak), with 2000 Hz applied to pair 1 and 2040 Hz to pair 2. Impedance checks were performed both before and during TI stimulation to ensure that the impedance was within a safe range. In the right EC stimulation protocol, electrodes were positioned at F9 and FT9, and CP4 and C6. 2000 Hz was applied to electrode pair 1 (F9, FT9) and 2040 Hz to pair 2 (CP4, C6), creating a frequency difference of 40 Hz. Due to the utilization of alternating current, the polarity within each electrode pair was not differentiated. In both scenarios, stimulation lasted for 20 min, with a 30-s ramp-up to 1.5 mA at the start and a 30-s ramp-down at the end. To provide a realistic experience for participants, a sham stimulation protocol was also implemented. Unlike the true stimulation, both electrode pairs in the sham setup received a 2000 Hz current, simulating TI stimulation conditions. However, after ramping up to 1.5 mA, the current immediately ramped down, avoiding continuous stimulation.
In the MRI tasks, participants received TI stimulation while being scanned in a magnetic resonance chamber. For HD-IFS, we used the same electrodes as HD-tES—both outside and inside the MRI. Stimulation duration was consistent across all subjects. The start and end of the stimulation were controlled by a trigger. After scanning all field maps and collecting pre-stimulation structural and fMRI data, the MRI engineer manually started the TI device and began fMRI data collection under stimulation conditions upon receiving the completion signal. Uniform operational procedures were used for all subjects to avoid noise introduced by differing methods. The TI protocols in MRI tasks were identical to those in behavioral tasks, using the same stimulation schemes for both the left EC and right EC. The TI stimulation device was placed outside the magnetic resonance chamber and connected via a CSOP-D5 (Corning Submarine Optical Fiber Cable D5) output cable and a ground lead. The electrode pairs remained the same as in behavioral tasks, driven in opposite phases. It should be noted that all the electrodes utilized during MRI data acquisition were MRI-compatible. Also, impedance checks were performed both before and during TI stimulation. The waveforms were set at frequencies of 2 kHz and 2.04 kHz through these electrode pairs, with the ground lead carrying any imbalance current. Participants wore the brain cap with pre-defined 10–10 system electrode position holes. HD-tES MRI electrode holders were used to contain the electrode bars, filled with conductive paste to enhance conductivity. Consistent with behavioural experiments, the left EC stimulation scheme involved placing electrodes at C5 and CP5, and FT10 and F8, while the right EC stimulation scheme placed electrodes at F9 and FT9, and CP4 and C6. True stimulation lasted for 10 min, including a 30-s ramp-up, a sustained stimulation period, and a 30-s ramp-down. In contrast, the sham stimulation did not have a sustained stimulation period. In the sham setup, whose electrode placements were totally the same as those in the behavioural experiments, both electrode pairs received a 2000 Hz, 1.5 mA current. The duration of stimulation was consistent across all subjects. In the MRI tasks, participants underwent online TI stimulation while scanning the resting fMRI data. The duration of stimulation was consistently maintained across all subjects. To further investigate potential differences between the two sham conditions—one involving zero current and the other involving zero frequency differences—we conducted additional behavioral comparisons between these two conditions. This analysis aimed to clarify whether the presence of electrical currents alone could account for observed stimulation effects. Detailed results of these comparisons were provided in the supplementary materials (see Supplementary Fig. 2, with source data provided in the source data file). Additionally, since previous studies have applied different frequency bands in TI stimulation on humans, such as hippocampus38 (5 Hz) and striatum41 (80 Hz), we conducted another pilot study on behavioral performances with different frequency bands (5, 40, and 80 Hz). The results in the supplementary materials (Supplementary Fig. 3, with source data provided in the source data file) demonstrated that the 40 Hz condition achieves better performance enhancement, which supported the chosen frequency.
Path integration tasks
The Virtual reality (VR) setup included a VR headset, four carbon fiber brackets, and two VR hand controllers—one responsible for controlling the viewpoint within the VR scene and the other for selecting and confirming decisions. Specifically, in the return-to-start tasks, when participants thought they reached the right place of the starting cone, they could press the button to make confirmation. Also, in the cognitive map tasks, participants could also press the button on the left joystick to set the missing cones that needed to be placed. The right joystick allowed participants to control the view of the grid chessboard. The knob on the right joystick enabled the adjustment of the view, allowing for a dynamic change in perspective during the task. The PI task was designed within a 6 × 6 m space for participants to walk. Consequently, the study was conducted in a spacious classroom, exceeding 6 × 6 m in size, free from reflective surfaces or obstructions that could affect the VR experience. Once participants ventured out of the 6 × 6 m space, a warning would alert them to stop walking further. Additionally, researchers remained nearby to ensure participants did not move beyond the test area.
The VR program was developed using the Unity game engine, programmed to meet the required specifications. After the development phase, the programme was compiled and exported as an executable file compatible with the Windows operating system, ensuring seamless deployment and user accessibility.
Upon donning the VR headset and launching the VR software, participants found themselves in an open field projected to infinity with grassy terrain and distant hills. The VR environments were meticulously designed according to a strict protocol that omitted any prominent landmarks, both within and outside the task area. This design prevented the use of egocentric beaconing strategies, ensuring that participants’ decisions relied solely on their spatial perception capabilities, thus providing a uniform experience across all participants. The direct alignment between real-world and virtual movements successfully prevented vestibular mismatch and significantly reduced discomfort associated with VR experiences.
Before commencing the experiment, participants would go through a comprehensive tutorial phase to become acquainted with the VR setup and controls. All the participants were required to complete the return-to-start tasks, followed by the cognitive map tasks in both tutorial and formal experiments. Initially, participants received an introduction to the whole trial process, accompanied by both visual and auditory instructions. The entire experimental process comprised three parts: the speed control part, the tutorial part, and the formal experiment part. In the speed control part, participants were required to maintain a specific speed over a designated distance, which they must sustain to complete the subsequent experiment. This step was designed to roughly control the time taken by all participants. Upon receiving an instruction, participants were navigated to the starting cone zero and followed the instructions to cone one. Notably, moving too fast or too slow would result in the restart of this part. After completing the speed control part, participants proceeded to the tutorial part. At the beginning of each tutorial, participants were instructed on the procedures they needed to follow during the formal experiment. Two types of spatial diagrams were designed to minimize potential ceiling effects in task performance, one involving three intermediate points and another involving five intermediate points. The tutorial phase focused exclusively on learning the procedures with the three-point diagrams, with the formal procedures consisting of five intermediate cones operating similarly. After completing the two teaching tasks in the tutorial part, they proceeded to the formal experiment. In the formal phase, participants consistently completed the tasks with three-point diagrams, followed by the five-point diagrams. The results for the return-to-start and cognitive map tasks in the three-point diagrams were presented in the first supplementary materials (Supplementary Fig. 1, with source data provided in the source data file).
The formal experiment consisted of eight distinct maps in three-point diagrams and eight distinct maps in five-point diagrams. The positions of the cones were pseudo-randomly assigned, providing a comprehensive assessment of PI performances. Each participant completed the same set of eight distinct configurations (three-point diagrams and five-point diagrams) but in a random order. Initially, participants undertook one round of VR tasks, which included return-to-start tasks followed by the cognitive map tasks in each trial. During the rest interval, they received the TI stimulation while watching a 20-min clip from the movie Coco. After the rest period, they began the second round of VR tasks. In the formal procedure, each participant was required to complete sensory-driven and abstract processing conditions. Each condition consisted of two difficulty levels: three points and five points with eight trials each. Therefore, individual participants underwent 32 trials in either pre- or post-stimulation with two experimental conditions (sensory-driven and abstract processing), with 16 trials in the sensory-driven condition and 16 in the abstract processing condition.
Return-to-start tasks (sensory-driven)
Similar to Newton et al.53’s paradigm, participants were instructed to walk through several intermediate landmarks, each identified by inverted cones positioned at head height and sequentially numbered. At the base of each cone, a cross was placed to precisely indicate its position. Additionally, a yellow circle expanded outward from the center of the cross to further emphasize and highlight the exact location of the cone. Only one cone was visible at any given time and would vanish upon the participant reaching it. Subsequently, the next cone in the sequence would then become visible. Additionally, arrow-like hints hovering on the grassy ground indicated the direction the participant was facing. These arrows, which were adjusted based on the participants’ movements, appeared 0.75 m ahead and 0.25 m behind the participant. The arrows hovered slightly above the grass, measuring 0.2 m in length, with an interaction distance of 0.05 m between them.
Different from the previous paradigm53, since all participants recruited were healthy adults, diagrams with five intermediate cones were introduced in this study to increase difficulties for participants. They were required to finish two tasks: the return-to-start and the cognitive map task. The return-to-start task was identical to Howett’s description13 in which participants were required to navigate back to the starting cone based on their memory. This task followed a sequence where participants were first instructed to visit several intermediate cones and ultimately reach a final cone before attempting to return to the starting cone. The positions of all these cones were randomly assigned, but maintaining a fixed Euclidean distance of 2 m between adjacent cones. Each cone would appear independently in sequence and be accompanied by an auditory cue. Upon reaching the final cone, a message would appear in the virtual scene with an auditory cue instructing participants to return to the location of the start cone (cone zero). They would press a trigger on the hand controller to place a tag marking the starting cone once reached their perceived starting cone. The true location of cone zero would be displayed as feedback. They had five seconds to compare the relative position of the real starting cone with the one they had marked.
Cognitive map tasks (abstract processing)
The cognitive map task was designed to evaluate the abstract processing spatial representation ability. In this task, participants would view a 6 × 6 m grid map on the VR screen with all cones marked except two missing ones. Participants’ tasks were to recall the relative positions of the missing cones from a top-down view and place a marker icon on the map, indicating where they thought the missing cones should be. Participants could mark the missing cones using the left-hand controller and rotate the entire map, excluding the cones and hint text, by turning the button on the right-hand controller. The missing cones were to be marked and withdrawn in sequential order. Similar to the return-to-start task, once participants confirmed the positions they had marked, the screen would display the correct answers as feedback. The cognitive map tasks allowed a response time of 60 s for the participants. A countdown timer was displayed at the top right corner of the screen as a reminder. When the countdown timer expired, the actual location of the real cone was displayed directly, and any incomplete answer would be treated as the final response.
Evaluation metrics
To quantitatively assess the participants’ performance, five evaluation metrics were introduced. The five metrics were angle deviation, RDD, AAD, ARDD, and ODD.
In the return-to-start task, the actual experimentally designated location was defined as the true cone, and the location reached by the participant as the estimated cone. The five-point diagram was designed to consist of five consecutive cones after the starting cone. The starting cone was labeled as cone zero, while the final cone was labeled as cone five, representing the last location to which participants were guided. The participant’s estimated path was the straight segment from the final cone (cone five) to the participant’s perceived starting cone (their estimated position of cone zero). By contrast, the actual path was the straight segment from the final cone (cone five) to the true starting cone (the actual location of cone zero). By contrast, the actual path was measured from the final cone (cone five) to the true starting cone (the actual location of cone zero). In the cognitive map task, participants were provided with two cones as reference cones and were required to recall the positions of the remaining missing cones based on the process from the return-to-start task. The actual cone locations were defined as true cones, whereas the locations marked by participants were defined as estimated cones.
Specially, angle deviation measures the angular difference between the participant’s estimated path and the actual path while RDD quantifies the discrepancy between the participant’s estimated distances and the actual distances between cones. AAD computes the mean angular deviation across multiple cones, ARDD calculates the mean RDD across multiple cones, and ODD is a comprehensive metric that combines both angular and distance deviations to reflect overall task performance accuracy. The formulas used to compute these metrics are elaborated below.
Angle deviation: Angle deviation (AD) was a critical metric for assessing the precision of PI tasks. This index quantified the accuracy of a participant’s rotational movement from cone five back to cone zero. Considering PI tasks that incorporate five intermediate nodes, the angle deviation was determined by the discrepancy between Atrue and Aestimated, where Atrue represents the actual angle from cone five to cone zero, and Aestimated denotes the angle from cone five to the estimated final position, referred to as cone zero estimated. The angle deviation can be computed as follows.
where v1 signifies the directional vector from cone four to cone five, v2 represents the true trajectory vector from cone five to cone zero, and \({{{{{\bf{v}}}}}_{{{{\bf{2}}}}}}^{{\prime} }\) indicates the estimated trajectory vector towards cone zero. The function atan2d computes the angle between two vectors by taking their cross and dot products and returns the result in degrees, ranging from −180° and 180°. A positive value indicates an underturning from an allocentric viewpoint, whereas a negative value suggests an overturning. Consequently, the closer the absolute value of θangle_deviation is to zero, the more accurately the participant has executed the PI task as designed.
Relative distance deviation: RDD served as another essential measure for evaluating the accuracy of PI tasks. This metric gauged the precision between the final cone and the starting cone. This deviation was quantified by the discrepancy between Dtrue and Destimated. Here, Dtrue indicates the actual Euclidean distance between the final and starting cones, while Destimated represents the Euclidean distance from the final cone to the estimated starting cone. The RDD can be calculated using the following formula.
In this context, D is determined by the formula \(D=\sqrt{{({x}_{1}-{x}_{2})}^{2}+{({y}_{1}-{y}_{2})}^{2}}\) for any two cones X and Y, where the coordinates for cone X are (x1, y1) and those for cone Y are (x2, y2). A smaller value of drelative_distance_deviation indicates a higher level of accuracy in the participant’s execution of the PI task as designed.
Average angle deviation: The AAD was a crucial metric in cognitive map tasks. It quantified the angular accuracy between an estimated cone and a given reference or datum cone. The deviation was calculated by computing the angle between the trajectory formed by the line from the real cone to the reference cone with the trajectory formed by the line from the estimated cone to the reference cone. Notably, our cognitive map task included four datum cones, necessitating the computation of four angles for each estimated cone by the participant. The AAD was computed as the average of all eight angles derived from the task. It is mathematically expressed as follows.
where N refers to the number of cones missing, i indexes the nodes that participants were tasked to mark, and j represents the two datum cones provided. The vij denotes the trajectory from the datum cones to the true position of a missing cone, while wij denotes the trajectory from the datum cones to the estimated missing cone. The function atan2d is the same as the one in computing the angle deviation, which computes the four-quadrant inverse tangent of the ratio of the sine to the cosine of the angles defined by these vectors. A smaller value of θaverage_angle_deviation indicates greater accuracy in the participant’s execution of the missing cones recalling task as designed.
Average relative distance deviation: Similar to the computation of RDD, the ARDD was introduced to gauge the accuracy of the cognitive map task. This metric measures the distance accuracy between an estimated cone and a given reference or datum cone. The deviation was calculated by comparing the trajectory from the real cone to the reference cone with the trajectory from the estimated cone to the reference cone. This deviation was quantified by the difference between Dtrue and Destimated, where Dtrue represented the actual Euclidean distance between the true position of a missing cone and the datum cone while Destimated indicated the Euclidean distance from the estimated position of a missing cone to the datum cone. This required computing two distances for each estimated cone by the participant. The ARDD was then calculated as the average of all such discrepancies across the task. The formula for this calculation is as follows.
In this context, N is the number of cones missing, D is determined by the Euclidean distance formula \(D=\sqrt{{({x}_{1}-{x}_{2})}^{2}+{({y}_{1}-{y}_{2})}^{2}}\) for any two cones X and Y, where the coordinates for cone X are (x1, y1) and for cone Y are (x2, y2). A smaller value of drelative_distance_deviation indicates a higher level of accuracy in the participant’s execution of the missing cone recalling task as designed.
Overall distance deviation: The consideration of both angle and distance deviations in cognitive map tasks highlighted the inherent sensitivities of these metrics to positional and distance changes, respectively. To address the limitations associated with these metrics when both distance and positional accuracies were critical, a comprehensive metric called the ODD was introduced. This metric incorporated both angular and distance properties to provide a more holistic evaluation of spatial estimation accuracy. Let us consider the estimated cone as cone zero and another truth cone one. To ensure that the predicted distance from the estimated cone to the truth cone matches the actual distance, the estimated cone zero should ideally lie on a circle centered at cone one with a radius equal to the true distance between cone one and the truth cone. Introducing a second truth cone, cone five, requires that the estimated cone zero also lies on a circle centered at cone five with an appropriate radius. The exact position of the estimated cone zero, for perfect accuracy, would be one of the intersections of these circles. With additional circles drawn from more data cones, the feasible locations for the predicted cone narrow, increasingly refining the estimation accuracy. This concept can be quantitatively assessed through the ODD, which aggregates the squared discrepancies between true and estimated distances across all estimated cones and the remaining respective truth cones. The formula can be given by the following equation.
where N is the cones missing, M is the total number of cones, \({D}_{tru{e}_{ij}}\) represents the actual distance between the actual position of the ith missing cone and the jth true cone, while \({D}_{estimate{d}_{ij}}\) denotes the estimated distance between the estimated ith datum cone and the jth true cone. By squaring the differences and summing them across all datum cones and their paired estimations, this metric provides a robust measure of accuracy that effectively captures discrepancies in both distance and angular alignment. The resultant value of doverall_distance_deviation serves as an aggregate indicator of precision, with lower values indicating higher accuracy in the positioning of estimated cones relative to their true locations.
As mentioned above, both PI tasks contained eight different maps. All five metrics were calculated separately in the return-to-start (AD and RDD) and cognitive map tasks (ADD, ARDD, and ODD) for each participant, respectively. ANOVA and its post-hoc analyses with Bonferroni corrections were applied on each metric with stimulation type (left-, right-EC stimulation, and sham condition) as between-subject factor and stimulation (pre- vs. post-stimulation) as within-subject factor. To fully characterize the stimulation effect of left or right EC on PI performance, three additional post-hoc analyses were conducted whenever stimulation type × stimulation interaction was observed. First, group homogeneity was explored via one-way ANOVA with stimulation type as a between-subject factor (left-EC vs. right-EC stimulation vs. sham), though participants were randomly assigned to each group. Then paired t-test was carried out on pre- and post-stimulation metrics in each stimulation group separately to see whether performance was enhanced after stimulation. To further characterize the modulation effect of EC stimulation, the difference between pre- and post-stimulation was calculated for each metric and participant. One-way ANOVA was again applied to these metric differences with stimulation type (left-EC vs. right-EC vs. sham) as a between-subject factor.
MRI acquisition
Scanning was performed using United Imaging Healthcare’s 3T uMR 890, using a 64-channel head coil. The protocol included standard T1-weighted structural images, resting fMRI images, field maps, and single-band images. The T1 imaging protocol utilized a voxel size of 0.8 × 0.8 × 0.8 mm, resulting in a total scan time of 3 min and 46 s. The acquisition matrix of the MRI was 208 × 300 × 320. The specific sequence employed was a gradient echo sequence (gre-fsp). Key imaging parameters included sagittal orientation, 3D acquisition, a bandwidth of 260 Hz/pixel, a repetition time (TR) of 6.7 ms, an echo time (TE) of 2.3 ms, a flip angle (FA) of 8°, and a field of view of 256 mm. The scan was performed with an acceleration factor of 2, using IR-Non.SS recovery mode without fat suppression. Field map scans were acquired to correct the echoplanar imaging images for signal distortion (TR = 641.6 ms, TE = 4.92 ms, FA = 60°). Single-band images were acquired to simplify the imaging procedure and reduce artifacts (TR = 4332 ms, TE = 1 ms, FA = 60°). The resting-state fMRI images were obtained with a voxel size of 2.4 × 2.4 × 2.4 mm, a 2.40 mm3 isotropic voxel, TR of 750 ms, TE of 30 ms, FA of 60°, field of view of 216 mm, 500 slices, and an acceleration factor of 2. The acquisition matrix for fMRI was 90 × 90 × 66. The scanning time was consistent for all participants. The resting-state fMRI images were scanned both before and during the TI stimulation.
MRI pre-processing
The resting fMRI data were preprocessed using the Data Processing & Analysis for Brain Imaging (DPABI) toolbox67. For each subject, the first 10 volumes were discarded to allow for the stabilization of the gradient magnetic field, as initial scans may contain unstable and noisy signals that could adversely affect subsequent analyses. Initial skull stripping was performed using DPABI, while those with residual non-brain tissues were manually processed with the Brain Extraction Tool68 from the FMRIB Software Library69. However, due to parameter configurations, certain subjects’ data may retain excessive extracranial tissue or erroneously remove parts of the intracranial region. To address this, manual adjustments and corrections were performed, thereby ensuring good data quality throughout preprocessing. All skull-stripped images were consolidated into a common directory, ensuring consistency for subsequent preprocessing steps. Subsequently, the BOLD signals were segmented into grey matter, white matter, and cerebrospinal fluid components. Spatial smoothing was applied using an isotropic Gaussian kernel with a full width at half maximum of 6 mm in all three spatial dimensions (6 × 6 × 6 mm). Temporal filtering was performed with a band-pass filter set to 0.01–0.1 Hz to reduce low-frequency drift and high-frequency physiological noise. Finally, the brain was parcellated into 246 ROIs according to the Brainnetome atlas. To ensure consistency in data processing, both pre-stimulation data and data during stimulation were organized within the same directory and underwent pre-processing simultaneously using the established pipeline.
To further provide a more intuitive visualization of the brain regions targeted by TI stimulation, electric field simulation modeling was performed for each specific subject. Three-dimensional (3D) modeling was conducted on the T1-weighted MR images of the subjects using QDLab software (a commercial simulation software developed by QD Technology Co., designed specifically for modeling and simulating TI stimulation effects based on a subject’s T1-weighted MRI data, http://www.neurodome-med.com/), enabling the calculation of the electric field distribution at each position under the specified TI stimulation protocol. The input T1 images were initially converted into right-anterior-superior space70 to ensure compatibility with subsequent registration processes. Given that the original data collection used a resolution of 0.8 mm, and QDLab only supports calculations at a 1 mm resolution, the T1 data were resampled to a voxel size of 1 × 1 × 1 mm. Furthermore, the differences in head sizes may result in incomplete MRI data with missing part of brain data, which may lead to errors for QDLab in building head models and thus causing the electrodes to extend beyond the image area during electrode registration, potentially compromising the accuracy of the simulation. To mitigate this issue, a zero-padding operation was applied to the edges of the T1 data, effectively extending the image boundaries and ensuring that all the electrode positions remained within the image domain.
TI modulation ECs via electric field simulation
In our study, the commercial software QDLab was used to estimate the TI, which is based on the magnitude of the amplitude modulation of the electric fields, as shown in Fig. 3a. In this platform, the electric fields from the two pairs of electrodes were simulated. Six tissue conductivities (scalp, skull, cerebrospinal fluid, white matter, gray matter, and air cavities) were used for the estimation of the electric fields. The interference pattern of the two electric fields was calculated according to the TI electric field coupling principle28. The interference pattern was then applied to each participant based on their head model and resulted in the TI amplitude envelope intensity files. Finally, the participants’ MRI and envelope data were registered to the Brainnetome template45 to extract the amplitude envelope in targeted regions. Specifically, four files were generated for each given subject by QDLab software, including the T1 (b1), the electrode position mask, the electrode position mask file in accordance with the given TI protocol, as well as the simulated envelope amplitude file (e1). Since the envelope simulations were computed based on T1 MRI images that included extracranial tissue, these simulations were unsuitable for template matching. To address this, the envelope file (e1) underwent a series of pre-processing steps, including skull removal, linear registration, and ROI extraction. As direct manipulation of the electric field simulation files was not feasible, Python’s pyrobex toolkit was employed to perform skull removal on the pre-processed T1 image (b1). This operation resulted in the creation of a skull-removed file (b2). Subsequently, a brain mask (bm) was generated from the skull-removed data (b2). This mask was then applied to the envelope field file (e1), producing a skull-removed envelope file (e2). Next, the skull-removed T1 data (b2) were registered to the Brainnetome Atlas, and the corresponding mapping matrix (br) was synchronously generated and saved. This mapping matrix (br) was then applied to the skull-removed envelope file (e2), yielding a registered envelope file (e3) that was fully aligned with the Brainnetome Atlas. To extract the simulated envelope field results for specific ROIs, ROI masks were correspondingly created based on the ROI labels provided in the Brainnetome Atlas, resulting in a series of ROI masks m1,m2,...,m246. A dot multiplication operation was performed on the registered envelope file (e3) using the ROI mask, allowing for the extraction of envelope data for each ROI. The mean value of the non-zero regions was calculated to determine the electric field intensity within each ROI for each subject.
To verify that TI stimulation effectively targeted deeper brain regions - specifically the EC - rather than the overlying cortex, the simulated envelope electric field signals were extracted from both the target regions and the superficial cortical regions near the electrode placements in the same hemisphere. Those overlying cortical regions were defined as control ROIs. For the left EC stimulation protocol, the target region was the left EC (ROI 115). The control ROIs were ROIs 141 and 145, which were located directly beneath the electrodes placed at positions FT10 and F8, respectively. These cortical ROIs, along with the target EC ROI 115, were included in the statistical analysis. The voxel-wise amplitude envelope values in the corresponding brain ROI were averaged. A repeated measures ANOVA and the subsequent post-hoc paired t-tests were conducted to compare the envelope values, i.e., magnitude of the amplitude modulation of the electric fields among these regions. This approach allowed for the assessment of whether the stimulation predominantly affected the deep EC region compared to the superficial cortical areas. A similar methodology was applied to the right EC stimulation protocol. The target region was the right EC (ROI 116). The control ROIs selected were ROIs 142 and 146, located directly beneath the electrodes placed at positions CP4 and C6 on the right hemisphere. These ROIs were also included in the repeated measures ANOVA and paired t-test analyses to evaluate differences between the targeted deep brain region and the overlying cortex. In addition, to further compare the differences in amplitude envelope, the mean envelope field was compared across the whole brain with that of the targeted brain regions. By comparing the amplitude envelope intensities across these brain regions formed by the interference patterns of TI stimulation, the analysis aimed to demonstrate that TI stimulation selectively activates the deep EC regions while minimizing stimulation of the superficial cortical areas near the electrodes. A three-dimensional visualization of the targeted area was also constructed, and the targeted EC region was rendered with the MATLAB toolkit and overlapped with the envelope distribution. This approach allowed us to more accurately reflect the stimulation effects on the target brain region under a specific stimulation protocol, highlighting the localized distribution of the envelope generated.
Resting fMRI data analysis
The data processed through DPABI were categorized based on the recorded information during data collection into three stimulation groups: sham condition, left EC stimulation, and right EC stimulation. Within each stimulation group, data were further classified into pre-stimulation and in-stimulation conditions. The FC matrix, denoted as \(fc\in {{\mathbb{R}}}^{n\times n},n=246\), was computed on the time series by Pearson correlation between each pair of ROIs. In this experiment, the pre-stimulation signal was treated as the baseline across all stimulation groups (sham, left stimulation, right stimulation). The baseline signal was subtracted from the signal during stimulation, and the resulting FC matrix was used as the input signal for subsequent statistical analysis. To validate the left EC stimulation scheme, the FC values between the left EC corresponding ROI (ROI 115) and all other ROIs were extracted. A Random Forest classification was then conducted between the left EC stimulation and sham condition, with five cross-validations employed. Given the challenges posed by small sample size and substantial inter-subject variability, 490 time slices obtained from each subject were divided into 10 sub-sets, each containing 49 slices, denoted as s1,s2,...,s10. Each subject’s data was further divided into fivefolds, with each fold containing two subsets in sequential order. To prevent data leakage, the sequence of slices drawn from each subject remained consistent. In the Random Forest calculation process, each fold’s data consisted of all the corresponding data sets from each subject with that fold. Additionally, during both the training and testing phases, the data within the training and test sets were randomly shuffled to prevent the model from memorizing the sequence and ensure better generalization. Subsequently, an accuracy (ACC) value was calculated for each fold, and the average ACC and standard error of the mean (SEM) were computed across the fivefold data. The same procedure was applied to validate the right EC stimulus scheme. If effective activation of the EC region was achieved under TI stimulation, the classification accuracy of the Random Forest model distinguishing between the sham and the left stimulation conditions was expected to exceed the baseline of 50%, and so was the sham and right stimulation conditions.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The majority of the code developed for this study has been made available on the Ocean Code platform (https://doi.org/10.24433/CO.7152423.v1). Due to the integration of commercially licensed software in certain aspects of the results, the complete codebase cannot be open-sourced. Source data are provided with this paper.
References
Hart, R. A. & Moore, G. T. The development of spatial cognition: a review. In Downs, R.M. & Stea, D. Eds. Image & Environment: Cognitive Mapping and Spatial Behavior. 246–288 (Aldine Publishing Company, Chicago, 1973).
Epstein, R. A., Patai, E. Z., Julian, J. B. & Spiers, H. J. The cognitive map in humans: spatial navigation and beyond. Nat. Neurosci. 20, 1504–1513 (2017).
Ekstrom, A. D. et al. Cellular networks underlying human spatial navigation. Nature 425, 184–188 (2003).
Hafting, T., Fyhn, M., Molden, S., Moser, M.-B. & Moser, E. I. Microstructure of a spatial map in the entorhinal cortex. Nature 436, 801–806 (2005).
Gu, Y. et al. A map-like micro-organization of grid cells in the medial entorhinal cortex. Cell 175, 736–750 (2018).
Fyhn, M., Hafting, T., Treves, A., Moser, M.-B. & Moser, E. I. Hippocampal remapping and grid realignment in entorhinal cortex. Nature 446, 190–194 (2007).
Etienne, A. S. & Jeffery, K. J. Path integration in mammals. Hippocampus 14, 180–192 (2004).
Fukawa, A., Aizawa, T., Yamakawa, H. & Eguchi Yairi, I. Identifying core regions for path integration on medial entorhinal cortex of hippocampal formation. Brain Sci. 10, 28 (2020).
Najafian Jazi, M. et al. Hippocampal firing fields anchored to a moving object predict homing direction during path-integration-based behavior. Nat. Commun. 14, 7373 (2023).
Buzsáki, G. & Moser, E. I. Memory, navigation and theta rhythm in the hippocampal-entorhinal system. Nat. Neurosci. 16, 130–138 (2013).
Stangl, M., Kanitscheider, I., Riemer, M., Fiete, I. & Wolbers, T. Sources of path integration error in young and aging humans. Nat. Commun. 11, 2626 (2020).
Segen, V., Ying, J., Morgan, E., Brandon, M. & Wolbers, T. Path integration in normal aging and Alzheimer’s disease. Trends Cogn. Sci. 26, 142–158 (2022).
Howett, D. et al. Differentiation of mild cognitive impairment using an entorhinal cortex-based test of virtual reality navigation. Brain 142, 1751–1766 (2019).
Sargolini, F. et al. Conjunctive representation of position, direction, and velocity in entorhinal cortex. Science 312, 758–762 (2006).
Høydal, Ø. A., Skytøen, E. R., Andersson, S. O., Moser, M.-B. & Moser, E. I. Object-vector coding in the medial entorhinal cortex. Nature 568, 400–404 (2019).
Bigus, E. R., Lee, H.-W., Bowler, J. C., Shi, J. & Heys, J. G. Medial entorhinal cortex mediates learning of context-dependent interval timing behavior. Nat. Neurosci. 27, 1–12 (2024).
Gil, M. et al. Impaired path integration in mice with disrupted grid cell firing. Nat. Neurosci. 21, 81–91 (2018).
Bierbrauer, A. et al. Unmasking selective path integration deficits in Alzheimer’s disease risk carriers. Sci. Adv. 6, eaba1394 (2020).
Suthana, N. et al. Memory enhancement and deep-brain stimulation of the entorhinal area. N. Engl. J. Med. 366, 502–510 (2012).
Bellgowan, P. S., Buffalo, E. A., Bodurka, J. & Martin, A. Lateralized spatial and object memory encoding in entorhinal and perirhinal cortices. Learn. Mem. 16, 433–438 (2009).
Frisk, V. & Milner, B. The relationship of working memory to the immediate recall of stories following unilateral temporal or frontal lobectomy. Neuropsychologia 28, 121–135 (1990).
Wagner, I. C. et al. Entorhinal grid-like codes and time-locked network dynamics track others navigating through space. Nat. Commun. 14, 231 (2023).
Howard, L. R. et al. The hippocampus and entorhinal cortex encode the path and Euclidean distances to goals during navigation. Curr. Biol. 24, 1331–1340 (2014).
Meister, M. L. & Buffalo, E. A. Neurons in primate entorhinal cortex represent gaze position in multiple spatial reference frames. J. Neurosci. 38, 2430–2441 (2018).
Schmidt-Hieber, C. & Häusser, M. Cellular mechanisms of spatial navigation in the medial entorhinal cortex. Nat. Neurosci. 16, 325–331 (2013).
Jacobs, J. et al. Direct electrical stimulation of the human entorhinal region and hippocampus impairs memory. Neuron 92, 983–990 (2016).
Lefaucheur, J.-P. et al. Evidence-based guidelines on the therapeutic use of repetitive transcranial magnetic stimulation (rTMS). Clin. Neurophysiol. 125, 2150–2206 (2014).
Grossman, N. et al. Noninvasive deep brain stimulation via temporally interfering electric fields. Cell 169, 1029–1041 (2017).
Mirzakhalili, E., Barra, B., Capogrosso, M. & Lempka, S. F. Biophysics of temporal interference stimulation. Cell Syst. 11, 557–572 (2020).
Acerbo, E. et al. Focal non-invasive deep-brain stimulation with temporal interference for the suppression of epileptic biomarkers. Front. Neurosci. 16, 945221 (2022).
Mojiri, Z., Akhavan, A., Rouhani, E. & Zahabi, S. J. Quantitative analysis of noninvasive deep temporal interference stimulation: a simulation and experimental study. Heliyon 10, e29482 (2024).
Missey, F. et al. Orientation of temporal interference for non-invasive deep brain stimulation in epilepsy. Front. Neurosci. 15, 633988 (2021).
Song, X., Zhao, X., Li, X., Liu, S. & Ming, D. Multi-channel transcranial temporally interfering stimulation (TTIS): application to living mice brain. J. Neural Eng. 18, 036003 (2021).
Ma, R. et al. High gamma and beta temporal interference stimulation in the human motor cortex improves motor functions. Front. Neurosci. 15, 800436 (2022).
Zhu, Z. et al. Temporal interference (ti) stimulation boosts functional connectivity in human motor cortex: a comparison study with transcranial direct current stimulation (tdcs). Neural Plast. 2022, 7605046 (2022).
Zheng, S. et al. Repetitive temporal interference stimulation improves jump performance but not the postural stability in young healthy males: a randomized controlled trial. J. Neuroeng. Rehabil. 21, 38 (2024).
Zhang, Y. et al. Temporal interference stimulation targeting right frontoparietal areas enhances working memory in healthy individuals. Front. Hum. Neurosci. 16, 918470 (2022).
Violante, I. R. et al. Non-invasive temporal interference electrical stimulation of the human hippocampus. Nat. Neurosci. 26, 1994–2004 (2023).
Beanato, E. et al. Noninvasive modulation of the hippocampal-entorhinal complex during spatial navigation in humans. Sci. Adv. 10, eado4103 (2024).
Wessel, M. J. et al. Noninvasive theta-burst stimulation of the human striatum enhances striatal activity and motor skill learning. Nat. Neurosci. 26, 2005–2016 (2023).
Vassiliadis, P. et al. Non-invasive stimulation of the human striatum disrupts reinforcement learning of motor skills. Nat. Hum. Behav. 8, 1–18 (2024).
Yang, C. et al. Transcranial temporal interference stimulation of the right globus pallidus in parkinson’s disease. Mov. Disord. 40, 1061–1069 (2024).
Modak, P. et al. Temporal interference electrical neurostimulation at 20 Hz beat frequency leads to increased fMRI BOLD activation in orbitofrontal cortex in humans. Brain Stimul. 17, 867–875 (2024).
Vassiliadis, P. et al. Safety, tolerability and blinding efficiency of non-invasive deep transcranial temporal interference stimulation: first experience from more than 250 sessions. J. Neural Eng. 21, 024001 (2024).
Fan, L. et al. The human brainnetome atlas: a new brain atlas based on connectional architecture. Cereb. Cortex 26, 3508–3526 (2016).
Hallett, M. Transcranial magnetic stimulation and the human brain. Nature 406, 147–150 (2000).
Deng, Z.-D., Lisanby, S. H. & Peterchev, A. V. Electric field depth–focality tradeoff in transcranial magnetic stimulation: simulation comparison of 50 coil designs. Brain Stimul. 6, 1–13 (2013).
Zangen, A., Roth, Y., Voller, B. & Hallett, M. Transcranial magnetic stimulation of deep brain regions: evidence for efficacy of the h-coil. Clin. Neurophysiol. 116, 775–779 (2005).
Deng, Z.-D., Lisanby, S. H. & Peterchev, A. V. Coil design considerations for deep transcranial magnetic stimulation. Clin. Neurophysiol. 125, 1202–1212 (2014).
Rossi, S. et al. Safety, ethical considerations, and application guidelines for the use of transcranial magnetic stimulation in clinical practice and research. Clin. Neurophysiol. 120, 2008–2039 (2009).
Tacikowski, P., Kalender, G., Ciliberti, D. & Fried, I. Human hippocampal and entorhinal neurons encode the temporal structure of experience. Nature 635, 1–8 (2024).
Killian, N. J., Jutras, M. J. & Buffalo, E. A. A map of visual space in the primate entorhinal cortex. Nature 491, 761–764 (2012).
Newton, C. et al. Entorhinal-based path integration selectively predicts midlife risk of Alzheimer’s disease. Alzheimer’s. Dement. 20, 2779–2793 (2024).
Bostelmann, M., Lavenex, P. & Lavenex, P. B. Children five-to-nine years old can use path integration to build a cognitive map without vision. Cogn. Psychol. 121, 101307 (2020).
Kunz, L. et al. A neural code for egocentric spatial maps in the human medial temporal lobe. Neuron 109, 2781–2796 (2021).
Mallory, C. S. et al. Mouse entorhinal cortex encodes a diverse repertoire of self-motion signals. Nat. Commun. 12, 671 (2021).
Devanand, D. P. et al. MRI hippocampal and entorhinal cortex mapping in predicting conversion to Alzheimer’s disease. Neuroimage 60, 1622–1629 (2012).
Lee, S., Park, J., Lee, C. & Im, C.-H. et al. Multipair transcranial temporal interference stimulation for improved focalized stimulation of deep brain regions: a simulation study. Comput. Biol. Med. 143, 105337 (2022).
Wang, C. et al. Egocentric coding of external items in the lateral entorhinal cortex. Science 362, 945–949 (2018).
Oswald, C. & Good, M. The effects of combined lesions of the subicular complex and the entorhinal cortex on two forms of spatial navigation in the water maze. Behav. Neurosci. 114, 211 (2000).
Miller, J. F., Fried, I., Suthana, N. & Jacobs, J. Repeating spatial activations in human entorhinal cortex. Curr. Biol. 25, 1080–1085 (2015).
Pennanen, C. et al. Hippocampus and entorhinal cortex in mild cognitive impairment and early AD. Neurobiol. Aging 25, 303–310 (2004).
Baloh, R. W. Vertigo. Lancet 352, 1841–1846 (1998).
Sajjadi, H. & Paparella, M. M. Meniere’s disease. Lancet 372, 406–414 (2008).
Jurcak, V., Tsuzuki, D. & Dan, I. 10/20, 10/10, and 10/5 systems revisited: their validity as relative head-surface-based positioning systems. Neuroimage 34, 1600–1611 (2007).
Wang, M., Lou, K., Liu, Z., Wei, P. & Liu, Q. Multi-objective optimization via evolutionary algorithm (movea) for high-definition transcranial electrical stimulation of the human brain. Neuroimage 280, 120331 (2023).
Yan, C.-G., Wang, X.-D., Zuo, X.-N. & Zang, Y.-F. Dpabi: data processing & analysis for (resting-state) brain imaging. Neuroinformatics 14, 339–351 (2016).
Smith, S. M. Fast robust automated brain extraction. Hum. Brain Mapp. 17, 143–155 (2002).
Jenkinson, M., Beckmann, C. F., Behrens, T. E., Woolrich, M. W. & Smith, S. M. Fsl. Neuroimage 62, 782–790 (2012).
on Slicer.org. C. S.http://www.slicer.org/slicerWiki/index.php/Coordinate_systems.
Acknowledgements
This work was supported in part by the National Natural Science Foundation of China(62171283 to M.L., BC1200274 to B.Z.), the Fundamental Research Funds for the Central Universities (YG2024LC11, YG2024QNA57, YG2024QNA58) to M.L., Shanghai Municipal Science and Technology Commission (BS1200001) to B.Z., and Shanghai Jiao Tong University 2030 Initiative (AF0300546, AH1200003) to B.Z. Shanghai Municipal Science and Technology Major Project(2021SHZDZX0102) to M.L. This research greatly benefited from the grammatical review and expert editing provided by Mr. Darrel Li. The authors gratefully acknowledge his contribution to the clarity and accuracy of this manuscript.
Author information
Authors and Affiliations
Contributions
B.Z. and M.L. conceived the idea. B.Z., H.Z., and J.H. developed the method. H.Z. and J.H. implemented the software and performed the experiments. H.Z., B.Z., and M.L. wrote and revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Priyamvada Modak, Zoltan Nadasdy and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhao, H., Zhao, B., He, J. et al. Distinctive roles of left and right entorhinal cortex in path integration via a non-invasive stimulation study. Nat Commun 16, 10214 (2025). https://doi.org/10.1038/s41467-025-64986-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-64986-8