Abstract
Over geologic time, biocalcification – the process by which marine organisms make calcium carbonate (CaCO3) – has reshaped climates, ocean life, and seawater chemistry. In particular, the evolution of coccolithophores, the largest group of nannoplankton and today’s most productive calcifiers, transformed ocean environments and the carbon cycle. Their origins, however, remain enigmatic. This is partly because studying coccolithophore fossils traditionally requires CaCO3 preservation. Here, we bypass this limitation, searching for their ‘ghost’ fossils –imprints on organic matter. We present coccolithophores from ~241-million-year-old (Triassic) rocks, predating previous records by ~26 million years (myrs). The >100 ghost fossils, exceptionally preserved within zooplankton faeces, show that coccolithophores, nannoplankton, ‘modern’ eukaryotic phytoplankton, and planktonic biocalcification evolved earlier than previously thought. Coccolithophores now first appear alongside stony corals and other unrelated calcifiers, suggesting a diversification of a range of marine calcifying organisms following Earth’s deadliest mass extinction, the end-Permian event. These findings indicate that coccolithophore diversity remained remarkably low for ~50 myrs, until after the end-Triassic mass extinction, showing that both Triassic-bookending extinctions were critical in their evolution. Our discoveries elucidate the evolutionary origins of coccolithophores, but also highlight the role mass extinctions have played in shaping life on Earth.
Similar content being viewed by others
Introduction
A massive array of marine organisms, including corals, molluscs and crustaceans, build calcium carbonate (CaCO3) skeletons and shells through the process of biocalcification. Today, most marine biocalcification takes place in the open ocean water column (the pelagic realm) by calcifying plankton1, however, this was not always the case. Throughout the Palaeozoic, ~539–252 million years (myrs) ago, nearly all CaCO3 deposition occurred in shallow water environments2,3. It was not until the Triassic–Jurassic transition, ~201 myrs ago, that biocalcification became widespread in the pelagic realm, driven by the evolution of calcareous nannoplankton3. This development fundamentally changed seawater chemistry, long-term biogeochemical cycles and contributed to the formation of modern marine ecosystems; it therefore represents one of the most important evolutionary innovations in Earth’s history3,4,5. Among calcareous nannoplankton, coccolithophores—a group of pelagic single-celled photosynthetic eukaryotic algae—are overwhelmingly dominant6, representing the most productive calcifiers in today’s oceans7. They promote cloud formation8, play an important role in the carbon cycle7, and are a major contributor to marine primary production7. Traditionally, coccolithophores were thought to have evolved near to the end of the Triassic Period9, after calcareous dinoflagellates (a different group of calcareous nannoplankton) and nannoliths (minute CaCO3 remains of uncertain biological affinity)4,10. Indeed, to our knowledge, prior to this study, the earliest coccolithophore fossil was a ~ 215-myr-old indeterminate coccolith from the Norian Stage of the Late Triassic9. Given that coccolithophore fossils have been studied for almost 200 years11, with their fossils perhaps being the most numerous and stratigraphically complete of any group12, these views are widely accepted.
Until now, the early fossil record of coccolithophores has exclusively relied on the preservation of ‘body’ fossils—the CaCO3 remains of their ‘exoskeletal’ plates, called coccoliths6 (Fig. 1a, b). Unfortunately, even when conditions favour the deposition and burial of CaCO3 at the seafloor, CaCO3 can dissolve later due to heat, pressure, and acidic pore-water infiltration into rock strata13. Given that older rocks have experienced more time for CaCO3 dissolution to take effect, the preservation potential of CaCO3, and therefore body fossils, generally diminishes with age. Hence, older rocks that hold the answers to understanding the origins of coccolithophores are poorly positioned to do so from a taphonomic perspective. Here, we applied a methodological approach never before conducted on Triassic rocks, bypassing the need for CaCO3 body fossils. We primarily looked for their ‘ghost’, or imprint, fossils13,14 preserved on organic matter (Fig. 1c). We sampled Middle (Ladinian) and Upper (Norian) Triassic rocks from Switzerland and Austria, and where possible, selected sedimentary rocks interbedded with volcanic ash layers that have been radiometrically dated and thus have precise age estimates15 (Fig. 2, Supplementary Fig. 1 and Supplementary Data 1). After extracting organic matter from rocks, we searched for ghost fossils using a scanning electron microscope (see ‘Methods’ for details).
a Complete coccolithophore exoskeleton, made of coccoliths (individual plates); composed of calcite. b Coccolith ‘body’ fossil; composed of calcite. c Coccolith ‘ghost’ fossil; imprint of coccolith on organic matter after calcite dissolution (see Fig. 3 for photographed examples); note the hole in the centre of the imprint formed from the spine of the coccolith.
a Study site locations. b Global palaeogeographic map showing the location of the Ladinian study site. c Global palaeogeographic map showing the location of Norian study sites. d Age of samples and fossil occurrences. Palaeogeographic maps modified from ref. 47. For further sampling details, see ‘Methods’, Supplementary Fig. 1 and Supplementary Data 1.
Results and discussion
Ghost and body fossils
Ghost fossils of coccoliths were present in five samples (1, 2, 5, 6, 10) (Figs. 2 and 3 and Supplementary Figs. 1–3). To our knowledge, these are now the earliest known coccoliths in the fossil record, with the oldest predating the previous earliest coccolith by ~26 myrs9; they represent the first record of coccoliths from the Ladinian Stage and the Middle Triassic, and the first ghost fossils from the Triassic. Our discovery of Ladinian coccoliths also implies that coccolithophores must have existed throughout the Carnian, despite no previous reports of coccoliths from that stage. The ghost fossils are small, with coccoliths averaging 2.55 µm in length (n = 50). This partly explains their lack of discovery until now, but also reveals that in their early history, coccoliths started small, increasing in size with time.
a Indeterminate coccolith, sample 1 (Cava superiore 1; S206000-03); along with other coccoliths from this sample, to our knowledge this is now the earliest coccolith in the fossil record. b Crucirhabdus primulus, sample 6 (Val Mara D1; S206003-03). c, d Several C. primulus (arrows), sample 6 (Val Mara D1; S206003-03). e C. primulus, sample 6 (Val_Mara_D1ii; S206003-05). f ?Archaeozygodiscus koessenensis, sample 6 (Val_Mara_D1ii; S206003-05). Each sample was surveyed for 4 h. Blue images are inverted to better visualise the original coccoliths. Note that, compared to body fossils, imprinted and inverted images need to be interpreted with additional care, especially regarding features such as clockwise versus anticlockwise imbrication direction. Scale bars, 1 µm.
All identifiable ghost fossils belonged to the coccolith species, Crucirhabdus primulus (e.g. Fig. 3b), except for one specimen of ?Archaeozygodiscus koessenensis (Fig. 3f). The oldest sample (1), collected 23 cm below a volcanic ash bed 241.07 ± 0.13 myrs old (Ladinian)15, yielded indeterminate coccoliths (e.g. Fig. 3a) and C. primulus (Supplementary Fig. 2), which to our knowledge now represent the earliest records of this species by ~32 myrs9. Samples 2 and 5 only yielded indeterminate coccoliths (Supplementary Fig. 2), but sample 6, taken 10 m below a volcanic ash layer 239.51 ± 0.15 myrs old15 (Ladinian; Supplementary Fig. 1), was the most productive sample. This yielded >70 C. primulus specimens, one ?A. koessenensis and several indeterminate coccoliths (Fig. 3b–f, Supplementary Figs. 2 and 3). The ?A. koessenensis is now the earliest specimen of this species to our knowledge, predating previous records by ~27 myrs. Preservation in sample 6 was exquisite, with imprinted coccolith spines, central area structures and rim elements (Fig. 3). Sample 10, the youngest productive ghost fossil sample, yielded two C. primulus, and several indeterminate, specimens (Supplementary Fig. 3); although these are the youngest C. primulus here, they are still ~7 myrs older than the previous earliest records of this species9.
We also searched for body fossils from the same, plus several more CaCO3-rich rock samples (Supplementary Fig. 1), finding one Crucirhabdus minutus (not found as a ghost fossil) in sample 3, and one A. koessenensis in sample 7 (Supplementary Fig. 4). To our knowledge, the A. koessenensis is now the earliest body fossil of its species and the C. minutus is the earliest body or ghost fossil of its species, predating previous records by ~27 myrs (Supplementary Data 2). The C. minutus specimen is now to our knowledge also the earliest coccolith body fossil in the entire fossil record, by ~25 myrs.
Taken alone, our body fossil finds would have substantially pushed back the initial occurrence of coccolithophores, however, our oldest ghost fossils push back these records further still. Furthermore, only two body fossils were found compared to >100 ghost fossils, which include numerous well preserved specimens (Fig. 3). No samples yielded both ghost and body fossils, and of the three species documented here, only A. koessenensis (one ?A. koessenensis ghost fossil and one A. koessenensis body fossil) was found in both records (Fig. 3f and Supplementary Fig. 4). Our results clearly demonstrate the importance of ghost fossil analysis, but the differences between ghost and body fossil records show that each record is selectively capturing certain species, highlighting the utility of a combined approach.
Zooplankton predation
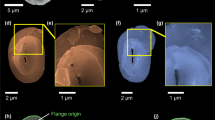
The ghost fossils were all found on a type of ‘smooth’ amorphous organic matter (AOM), compared to a more abundant ‘angular’ type (Supplementary Fig. 5 and Supplementary Movie 1). Although the origins of the angular AOM are unclear, the smooth fragments resemble zooplankton faecal pellets16. Zooplankton ingestion increases the likelihood of coccolith fossilisation, as faecal pellets provide protection and additional ballast, enabling coccoliths to sink to the seafloor6,17. Clustered and overlapping imprints (Fig. 3c, d) represent recently (at the time of fossilisation) collapsed complete coccolithophore exoskeletons. This suggests that the coccolithophores were eaten whilst alive; if they had died before ingestion, their articulated exoskeletons would have likely dispersed, breaking apart in the water column and degrading, or preserving as isolated imprints. Faecal pellets containing coccoliths also demonstrate that, by the Middle Triassic, coccolithophores were acting as primary producers at the base of marine food chains, transferring energy to higher trophic levels.
Crucirhabdus—the common ancestor of coccoliths
Our results reveal a hidden early fossil record of coccoliths, demonstrating that the three most ancient species—C. primulus, C. minutus and A. koessenensis—have substantially longer ranges than previously reported (Fig. 4). In particular, C. primulus now has an exceptionally long range of >58 myrs13. Before this study, the initial occurrences of C. minutus and A. koessenensis were in the late Norian, and C. primulus was in the early Rhaetian9. Based partly on these initial occurrences, C. minutus was thought to be ancestral to C. primulus9,18,19,20,21. Our records reveal a different initial occurrence order of: (1) C. primulus, (2) C. minutus, and (3) A. koessenensis. At face value, this suggests that C. primulus was ancestral to C. minutus, but we emphasise that comparing these initial occurrences is imperfect, as C. primulus is restricted to ghost, and C. minutus is restricted to body, fossil records here. However, since the genus Crucirhabdus now predates all other coccolith genera in both ghost and body fossil records, our results suggest that this genus is ancestral to all known coccoliths.
EPME end-Permian mass extinction, ETME end-Triassic mass extinction. See Supplementary Data 2 for precise range details.
Phytoplankton evolution and marine calcifying revolution
Among the eukaryotic phytoplankton that produce readily fossilisable remains, today, three groups—dinoflagellates, coccolithophores and diatoms—known as the ‘modern’ eukaryotic phytoplankton, carry out the vast majority of the export flux of organic matter to the ocean interior and higher trophic levels22. Traditionally, dinoflagellates were thought to be the first of these groups to evolve22. However, our records challenge this view, showing that coccolith ghost fossils, to our knowledge, are now the earliest unequivocal fossils of modern eukaryotic phytoplankton, predating the earliest dinoflagellate fossils23,24. Our results therefore rewrite the appearance order of modern eukaryotic phytoplankton to: (1) coccolithophores, (2) dinoflagellates, and (3) diatoms. Our findings also overturn the paradigm that calcareous dinoflagellates and nannoliths10,12 were the first calcifying plankton, which until this study, predated coccolithophores by ~21 myrs9. Instead, our results indicate that coccolithophores were the first pelagic plankton to develop biocalcification, a process that transformed marine environments and the carbon cycle.
Molecular clock predictions have placed the origin of coccolithophores between ~270–200 myrs ago25,26, however, recalibration has refined this to ~243 myrs ago27 (Fig. 4). Given that molecular clocks regularly predict origination dates significantly earlier than the earliest known fossils of many groups28, it is notable to find fossils here that agree with, and even predate some, clock-based estimates. In this respect, coccolithophores are unusual, as unlike most present-day highly diverse clades29, their crown group diversified remarkably slowly in their early history. Indeed, the scarcity of Triassic coccolith-derived limestones and chalks30,31 indicates that, although present, coccolithophores remained a somewhat subsidiary component of marine ecosystems until after this period. It was not until the Jurassic, ~50 myrs after their inferred Early–Middle Triassic origin, that coccolithophores speciated more rapidly32,33, suggesting that the end-Triassic mass extinction (ETME; ~201 myrs ago) was critical in giving rise to the requisite ecological and/or environmental changes that enabled their subsequent diversification.
The initial occurrence of coccolithophores is now ~10‒11 myrs after the onset of Earth’s deadliest event34, the end-Permian mass extinction (EPME; ~252 myrs ago). Due to the severity of the EPME, marine ecosystems took ~5‒10 myrs to recover35,36,37, thus our coccolithophore initial occurrence coincides with the upper estimates of this recovery phase. Several other highly diverse extant clades first appear in the aftermath of mass extinctions, including famously, the placental mammals38 and modern birds39, which both appeared after the end-Cretaceous mass extinction (~66 myrs ago). Our ghost fossil record now demonstrates that coccolithophores also follow this pattern. Perhaps most significantly however, we note that their initial occurrence also coincides with the initial occurrences, and post-EPME Lazarus taxa reoccurrences, of several unrelated marine calcifiers, including the stony (scleractinian) corals40, calcareous dinoflagellates10, heavily calcified sponges, and calcifying green algae41 (Fig. 4). Given that coccolithophores are today’s most productive marine calcifiers1, and stony corals are the most productive in coastal seas42—and incidentally, are the most important for maintaining marine biodiversity by constructing reefs43—these evolutionary developments would go on to overhaul ocean life, reshaping near- and off-shore environments. Here, we propose this as the early Mesozoic marine calcifying revolution (Fig. 4). Since the various groups involved are phylogenetically unrelated but biochemically similar, we infer that extrinsic factors related to the reorganisation of marine ecosystems and changes to ocean chemistry after the EPME were central to their contemporaneous emergence. Specifically, the extinction of numerous Permian calcifiers44 and the disappearance of complex reefs45, which were the principal zones of marine biocalcification through the Palaeozoic46, removed competitors and increased the availability of carbonate ions in Early–Middle Triassic oceans. Surviving lineages, including the soft (i.e. they lacked a hard CaCO3 exoskeleton) precursors to coccolithophores, were then able to exploit this resource, giving rise to a roughly simultaneous diversification event across multiple calcifying lineages (Fig. 4).
Our findings show that coccolithophores originated much earlier than previously thought. Their post-EPME emergence and post-ETME diversification indicate that both Triassic-bookending extinctions were pivotal in their evolution, but also reveal how these now ubiquitous plankton persisted at low diversity for a substantial portion of their early history. Although it is surprising that these ancient coccoliths have eluded discovery until now, all prior attempts to find the earliest forms have only looked for their CaCO3 body fossil remains. Our discoveries, which were made possible using the unconventional approach of ghost fossil analysis, demonstrate the potential of this method. More broadly, our study highlights that even intensively studied fossil groups, such as coccoliths, still have much to be discovered, but that innovative methodological approaches are needed to reveal such hidden records.
Methods
Study sites
We sampled three localities: (1) Monte San Giorgio, in the Southern Alps, Switzerland, and (2) Hahntennjoch and (3) Seefeld, in the Seefeld Basin, Northern Calcareous Alps, Austria. See Supplementary Fig. 1 and Supplementary Data 1 for sampling details. Samples in Austria were taken near roads in the public domain. No authorisation to sample and to export was necessary. The Monte San Giorgio area is a protected UNESCO site. Authorisation to sample and export was obtained from Dr. Rudolf Stockar, Cantonal Museum of Natural History, Lugano, Switzerland.
Ghost fossil analysis
Eight rock samples of ~20 g were dissolved in 40% hydrochloric and 40% hydrofluoric acids to remove carbonates and silicates, respectively, in order to isolate organic matter. These methods are standard processing techniques in the field of palynology, i.e. to extract spores, pollen and marine organic-walled microfossils from sedimentary rocks. Residues were then sieved at 10 µm, and the resultant material was pipetted directly onto aluminium SEM stubs. Residues were left to dry for 24 h and gold-coated for 180 s. Examination of organic matter was conducted using an ESEM FEI Quanta FEG 650 SEM at the Swedish Museum of Natural History, Stockholm. Organic matter was observed in traverses, between ×5000 and ×10,000 magnifications to search for ghost fossils. Photographs were taken using higher magnifications. Each sample was surveyed for 4 h.
Body fossil analysis
Twelve samples were prepared and examined for body fossils. Fresh rock surfaces of 1 square cm’s were cut and polished with green silicon carbide powder at 800-mesh per inch using distillate water. The blocks were etched for 15 s in 0.1% hydrochloric acid and briefly cleaned in an ultrasonic bath with distilled water. Samples were then dried overnight at 50 °C and coated with a 1 nm (20 s) layer of platinum/palladium using Cressington Sputter Coater 208HR. SEM observations were conducted with a TESCAN MIRA 3 SEM at Lund University, Sweden, and each sample was examined for 4 h.
Organic matter (palynofacies) analysis
To determine the predominant types of substrates onto which ghost fossils preserve, the proportions of different types of organic matter were counted using a light microscope (LM). Surplus organic matter residue from ghost fossil analysis was strewn across LM coverslips and mounted onto glass slides with epoxy resin. A minimum of 500 organic particles were counted per sample; see Supplementary Data 3 for organic matter categories; see Supplementary Note 1 for further details of organic matter (palynofacies) analysis.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All data generated or analysed during this study are included in this published article and its Supplementary Information files. All materials are housed in the collections of the Department of Palaeobiology at the Swedish Museum of Natural History, Stockholm (further details, including sample numbers are provided in Supplementary Data 1); contact the corresponding author for access.
References
Riebesell, U. et al. Reduced calcification of marine plankton in response to increased atmospheric CO2. Nature 407, 364–367 (2000).
Knoll, A. H. Biomineralization and evolutionary history. Rev. Mineral. Geochem. 54, 329–356 (2003).
Hull, P. M. Emergence of modern marine ecosystems. Curr. Biol. 27, R466–R469 (2017).
Erba, E. The first 150 million years history of calcareous nannoplankton: biosphere–geosphere interactions. Palaeogeogr. Palaeoclimatol. Palaeoecol. 232, 237–250 (2006).
Hohn, S., Dal Corso, J., Hoke, K. L., Thoms, S. & Merico, A. Environmental and physiological conditions that led to the rise of calcifying nannoplankton in the Late Triassic. Front. Earth Sci. 11, 747059 (2023).
Bown, P. R. & Young, J. R. In Calcareous Nannofossil Biostratigraphy (ed. Bown, P. R.) 1–15 (Springer Science+Business Media, 1998).
Rost, B. & Riebesell, U. In Coccolithophores: Molecular Processes to Global Impact (eds Thierstein, H. R. & Young, J. R.) 99–125 (Springer, 2004).
Alcolombri, U. et al. Identification of the algal dimethyl sulfide–releasing enzyme: a missing link in the marine sulfur cycle. Science 348, 1466–1469 (2015).
Demangel, I. et al. Development of early calcareous nannoplankton in the Late Triassic (Northern Calcareous Alps, Austria). Glob. Planet. Chang. 193, 103254 (2020).
Janofske, D. In Nannoplankton Research, Proceedings of the 4th INA Conference, Prague 1991 Vol. 1 (eds Hamrsmid, B. & Young, J. R.), 14a, 87–109 (Knihovnicka ZPN, 1992).
Ehrenberg, C. G. Bemerkungen über feste mikroskopische anorganische Formen in den erdigen und derben Mineralien. 84–85 (Abhandlungen der Königlich Preussischen Akademie der Wissenschaften Berlin, 1836).
Bown, P. R., Lees, J. A. & Young, J. R. In Coccolithophores: From Molecular Processes to Global Impact (eds Thierstein, H. R. & Young, J. R.) 481–508 (Springer-Verlag, 2004).
Slater, S. M., Bown, P., Twitchett, R. J., Danise, S. & Vajda, V. Global record of “ghost” nannofossils reveals plankton resilience to high CO2 and warming. Science 376, 853–856 (2022).
Slater, S. M., Bown, P. R. & Jardine, P. E. Nannofossil imprints across the Paleocene–Eocene thermal maximum. Geology 52, 271–275 (2024).
Stockar, R., Baumgartner, P. O. & Condon, D. Integrated Ladinian bio-chronostratigraphy and geochronology of Monte San Giorgio (Southern Alps, Switzerland). Swiss J. Geosci. 105, 85–108 (2012).
Beaumont, K. L., Nash, G. V. & Davidson, A. T. Ultrastructure, morphology and flux of microzooplankton faecal pellets in an east Antarctic fjord. Mar. Ecol. Prog. Ser. 245, 133–148 (2002).
Honjo, S. Coccoliths: Production, transportation and sedimentation. Mar. Micropaleontol. 1, 65–79 (1976).
Gardin, S., Krystyn, L., Richoz, S., Bartolini, A. & Galbrun, B. Where and when the earliest coccolithophores? Lethaia 45, 507–523 (2012).
Thierstein, H. R. Mesozoic calcareous nannoplankton biostratigraphy of marine sediments. Mar. Micropaleontol. 1, 325–362 (1976).
Jafar, S. A. Significance of Late Triassic calcareous nannoplankton from Austria and Southern Germany. Neues Jahrb. für Geol. Paläontol. Abh. 166, 218–259 (1983).
Bown, P. R. Taxonomy, evolution, and biostratigraphy of late Triassic–early Jurassic calcareous nannofossils. Spec. Pap. Palaeontol. 38, 1–118 (1987).
Falkowski, P. G. et al. The evolution of modern eukaryotic phytoplankton. Science 305, 354–360 (2004).
Riding, J. B., Mantle, D. J. & Backhouse, J. A review of the chronostratigraphical ages of Middle Triassic to Late Jurassic dinoflagellate cyst biozones of the northwest shelf of Australia. Rev. Palaeobot. Palynol. 162, 543–575 (2010).
Mangerud, G., Paterson, N. W. & Riding, J. B. The temporal and spatial distribution of Triassic dinoflagellate cysts. Rev. Palaeobot. Palynol. 261, 53–66 (2019).
De Vargas, C., Aubry, M. P., Probert, I. & Young, J. In Evolution of Aquatic Photoautotrophs (eds Falkowski, P. G. & Knoll, A. H.) 251–285 (Elsevier, 2007).
Medlin, L. K., Sáez, A. G. & Young, J. R. A molecular clock for coccolithophores and implications for selectivity of phytoplankton extinctions across the K/T boundary. Mar. Micropaleontol. 67, 69–86 (2008).
Liu, H., Aris-Brosou, S., Probert, I. & de Vargas, C. A timeline of the environmental genetics of the haptophytes. Mol. Biol. Evol. 27, 161–176 (2010).
Cunningham, J. A., Liu, A. G., Bengtson, S. & Donoghue, P. C. J. The origin of animals: can molecular clocks and the fossil record be reconciled? Bioessays 39, 1600120 (2016).
Budd, G. E. & Mann, R. P. Evolutionary Tempo, Supertaxa, and Living Fossils. Syst. Biol. syaf020, 1–18 (2025).
Bown, P. R., Burnett, J. A. & Gallagher, L. T. Critical events in the evolutionary history of calcareous nannoplankton. Hist. Biol. 5, 279–290 (1991).
Demangel, I. et al. Fate of calcareous nannofossils during the Rhaetian (Late Triassic): evidence from the Northern Calcareous Alps, Austria. Lethaia 56, 1–24 (2023).
Monteiro, F. M. et al. Why marine phytoplankton calcify. Sci. Adv. 2, e1501822 (2016).
Suchéras-Marx, B. et al. The colonization of the oceans by calcifying pelagic algae. Biogeosciences 16, 2501–2510 (2019).
Raup, D. M. Size of the Permo-Triassic bottleneck and its evolutionary implications. Science 206, 217–218 (1979).
Erwin, D. H. Lessons from the past: biotic recoveries from mass extinctions. Proc. Natl. Acad. Sci. 98, 5399–5403 (2001).
Benton, M. J. & Twitchett, R. J. How to kill (almost) all life: the end-Permian extinction event. Trends Ecol. Evol. 18, 358–365 (2003).
Friesenbichler, E., Hautmann, M. & Bucher, H. The main stage of recovery after the end-Permian mass extinction: taxonomic rediversification and ecologic reorganization of marine level-bottom communities during the Middle Triassic. PeerJ 9, e11654 (2021).
O’Leary, M. A. et al. The placental mammal ancestor and the post–K-Pg radiation of placentals. Science 339, 662–667 (2013).
Brusatte, S. L., O’Connor, J. K. & Jarvis, E. D. The origin and diversification of birds. Curr. Biol. 25, R888–R898 (2015).
Stanley, G. D. Jr The evolution of modern corals and their early history. Earth Sci. Rev. 60, 195–225 (2003).
Payne, J. L. et al. Large perturbations of the carbon cycle during recovery from the end-Permian extinction. Science 305, 506–509 (2004).
Milliman, J. D. Production and accumulation of calcium carbonate in the ocean: budget of a nonsteady state. Glob. Biogeochem. Cycles 7, 927–957 (1993).
Knowlton, N. et al. Coral reef biodiversity. In Life in the World’s Oceans: Diversity, Distribution, and Abundance (ed. McIntyre, A. D.) 65–77 (Wiley-Blackwell, 2010).
Dal Corso, J. et al. Environmental crises at the Permian–Triassic mass extinction. Nat. Rev. Earth Environ. 3, 197–214 (2022).
Pruss, S. B. & Bottjer, D. J. The reorganization of reef communities following the end-Permian mass extinction. C. R. Palevol 4, 553–568 (2005).
Kiessling, W., Simpson, C. & Foote, M. Reefs as cradles of evolution and sources of biodiversity in the Phanerozoic. Science 327, 196–198 (2010).
Kocsis, Á. T. & Scotese, C. R. Mapping paleocoastlines and continental flooding during the Phanerozoic. Earth Sci. Rev. 213, 103463 (2021).
Acknowledgements
This research was funded by the Swedish Research Council (2019-04524 [S.M.S.], 2023-03330 [S.M.S.], 2022-04969 [S.R.]), Formas (Swedish Research Council for Sustainable Development; 2023-00984 [S.M.S.]), the Austrian National Committee for the International Geoscience Programme (Austrian Academy of Sciences [S.R.]) and the European Research Council under the European Union’s Horizon 2020 Research and Innovation Program (833454 DEVOCEAN [Conley]). S.R. is grateful to R. Brandner, T.J. Algeo and R. Stockar for their help during sampling.
Funding
Open access funding provided by Swedish Museum of Natural History.
Author information
Authors and Affiliations
Contributions
S.M.S. and S.R. conceived the research. S.R. conducted the fieldwork. S.M.S. and I.D. performed the experiments and collected the data. All authors analysed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Jeremy Young, Isabella Raffi and the other anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Slater, S.M., Demangel, I. & Richoz, S. ‘Ghost’ fossils of early coccolithophores point to a Triassic diversification of marine calcifying organisms. Nat Commun 16, 9283 (2025). https://doi.org/10.1038/s41467-025-65116-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-65116-0