Abstract
There is an urgent need to identify those who may benefit from immunotherapy-based chemoradiotherapy (CRT) for patients with locally advanced rectal cancer (LARC) with proficient mismatch repair (pMMR). This single-arm, phase-II trial (NCT05450029), enrolled 46 treatment-naïve patients with histologically confirmed T3–4N0M0 or T1–4N+M0 LARC with intermediate or high Immunoscore. Patients received 6 cycles of mFOLFOX6 and long-course radiotherapy (50 Gy in 25 fractions) followed by surgery. Sintilimab was added during CRT (2nd-6th cycle). The primary endpoint, pathologic complete response (pCR) rate, was 65.2% [30/46, 95%CI: 49.7–78.6], with 85.7% (6/7) in high and 61.5% (24/39) in intermediate Immunoscore, meeting the pre-specified primary endpoint. Secondary endpoints included R0 resection rate (97.8%), the clinical tumor response (ORR 93.5%), the complication rate and safety, 3-year event-free survival rate, and 3-year overall survival rate (immature). The most common treatment related adverse event (TRAE) was leukopenia (69.6%, 32/46). The TRAE of Grade 3 occurred in 7 patients (15.2%). Four patients had postoperative complications (all grade ≤2). Here, we showed that PD-1 blockade combined with long-course CRT yielded promising therapeutic effects with a favorable pCR rate and acceptable safety profile among patients with intermediate/high-Immunoscore pMMR LARC.
Similar content being viewed by others
Introduction
Deficient mismatch repair (dMMR) or microsatellite instability-high (MSI-H) has been recognized as particularly promising indicators of responsiveness to immune checkpoint inhibitors (ICIs) across different types of cancers, including colorectal cancers (CRC)1. Recent studies have shown that in MSI-H and dMMR locally advanced rectal cancer (LARC), the application of immunotherapy has achieved a pCR rate as high as 60–88%2,3. This heralds a promising future for implementing organ-preservation strategies after neoadjuvant immunotherapy4,5.
However, the overall clinical response rate to ICIs in MSI CRC tumors is only 60–70%2,3. In other words, MSI-H might not be able to fully represent the immune-active status within tumor tissues. The Immunoscore test is the first internationally validated standardized assay for quantifying the immune infiltrate6,7, providing a reliable evaluation of the natural immune reaction at the tumor site. It is a digital pathology-based assay on the basis of the quantification of CD3+ and CD8+ lymphocytes at both the invasive margin and the core of the tumor8. This method quantifies the densities of both cell types in both regions, establishing a scoring system ranging from low to high immune cell densities9. Notably, an Immunoscore biopsy (ISB) performed at diagnosis has been indicated useful to predict the response to chemoradiotherapy (CRT) in LARC patients, independent of MSI status10,11. An intermediate or high Immunoscore is associated with a more favorable response to CRT, compared to a low one11. Recently, ISB has been further validated as a biomarker to predict both local regrowth and distant metastasis, indicating its clinical utility in selecting LARC patients eligible for a Watch-and-Wait strategy12.
On the other hand, only a modest proportion (approximately 5–10%) of rectal adenocarcinomas exhibit dMMR13, leaving a larger proportion of rectal cancer as mismatch-repair proficient (pMMR) or microsatellite stability (MSS) and insensitive to immunotherapy2,14. The efficacy of monotherapy with PD-1 inhibitors has been proved negligible for pMMR LARC14. Even with the introduction of total neoadjuvant therapy, a pathological complete response (pCR) was observed in less than 30% of patients, as reported in two large phase III trials15,16. The combination of immunotherapy and CRT represents a promising strategy. However, clinical studies focusing on PD-1 and CRT in LARC patients with pMMR status yielded inconsistent efficacy outcomes, with unimpressive pCR rates17,18,19,20. This underscores a significant unmet need in identifying patients who could derive substantial benefits from immunotherapy-based CRT for pMMR LARC. A subgroup of MSS or pMMR LARC patients has been shown to have a dense immune infiltration and a high Immunoscore and thus might respond better to immunotherapy21. Collectively, Immunoscore might be of great value to identify subsets of patients who are sensitive to immunotherapy-based CRT for pMMR LARC.
Here, we report the primary results from a single-arm, single-center, phase 2 trial (NCT05450029) to evaluate the efficacy and safety of PD-1 blockades (sintilimab) plus CRT as neoadjuvant in pMMR LARC patients with intermediate or high Immunoscore.
Results
Patient characteristics
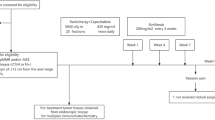
From July 2022 to September 2023, a total of 69 patients underwent a prospective Immunoscore assessment. Twelve patients with low Immunoscore were excluded, while 57 patients demonstrating intermediate/high Immunoscore proceeded to full eligibility assessment. Ultimately, 46 patients met all inclusion criteria and were enrolled in the trial (Fig. 1A). The schedules and timelines of treatments are schematically shown in Fig. 1B. The median follow-up from study enrollment to the clinical data cutoff date (September 1, 2024) was 19.2 months (95%CI: 17.3–21.1 months; range, 12.0–26.0 months). As shown in Table 1, the median age was 59 years (range, 35–74 years), and the median distance of the tumor from the anal verge was 4.8 cm (range, 2.1–9.7 cm). Thirty-nine (84.8%) patients had intermediate Immunoscore, and 7 (15.2%) had high Immunoscore. More than half of the patients had at least one high-risk factor, including cT4 disease in 34.8% (16/46), cN2 disease in 43.5% (20/46), mesorectal fascia margin positive in 56.5% (26/46), extramural vascular invasion in 54.3% (25/46) and tumors located within <5 cm from the anal verge in 52.2% (24/46). All patients were pMMR (Table 1).
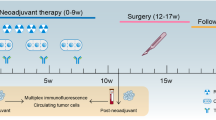
A Study flowchart.* The two dMMR/MSI-H patients were excluded from the study. One had lung metastasis at the baseline, and another had a history of breast cancer within the past 5 years. B Study design. Patients with T3–4N0M0 or T1–4N+M0 received 6 cycles of mFOLFOX6 and long course radiotherapy (50 Gy/25f, 2 Gy/f, 5 days/week) followed by surgery. Sintilimab (3 mg/kg, iv.gtt, 2nd–6th cycle) was added during chemoradiotherapy.
Efficacy results
Among the 46 patients enrolled (the intention-to-treat population), one patient died from a stroke during CRT, which was deemed unrelated to the study treatments. The remaining 45 patients completed the entire course of CRT and immunotherapy and underwent further radical surgery (Fig. 1A). All patients undergoing surgical resection achieved R0 resection with sphincter preservation.
The pCR rate was 65.2% [30/46, 95% confidence interval (CI): 49.7–78.6] (Table 2). The major pathologic response (MPR) and pathologic partial response rates were 26.1% (12/46, 95%CI: 14.3–41.1) and 6.52% (3/46, 95%CI: 1.40–17.9), respectively (Fig. 2). A total of 61.5% (24/39, 95%CI: 44.6–76.6) of patients with intermediate Immunoscore reached pCR, with an MPR rate of 28.2% (11/39, 95%CI: 15.0–44.9). Among the seven participants with high Immunoscore, the pCR rate was 85.7% (6/7, 95%CI: 42.1–99.6), while the MPR rate was 14.3% (1/7, 95%CI: 0.40–57.9), including one patient with ypT3N0 (Table 2). Among those who underwent radical surgery, 13 achieved a complete response, evaluated by both pathological and clinical standards (Fig. 2 and Supplementary Fig. 1). Representative radiological or pathologic images for patients with pCR are shown in Supplementary Fig. 2.
Waterfall plot depicting tumor response following neoadjuvant chemoradiotherapy combined with sintilimab. One patient death occurred during chemoradiotherapy and was conservatively categorized as non-pCR in the intention-to-treat analysis. Source data are provided as a Source Data file.
Clinical efficacy was assessed according to RECIST v1.1. After the neoadjuvant treatment, 25 (54.3%) exhibited a complete response, 18 (39.1%) exhibited a partial response and 3 (6.52%) had stable disease status; none of the patients experienced a progression of the disease (Fig. 2 and Supplementary Table 1). A tumor shrinkage of 30% or greater was identified in 43 of 46 patients (93.5%) (Fig. 2). Meanwhile, 89.1% (41/46) patients achieved a downstage of the clinical T category (Supplementary Fig. 3A), while 91.3% (42/46) achieved downstaging of the clinical N category after the CRT plus sintilimab regimen (Supplementary Fig. 3B).
The 3-year event-free survival rate and 3-year overall survival rate were predefined as secondary outcomes of this study. However, these outcomes are not reported herein, primarily due to the current insufficiency of follow-up duration. The corresponding data will be presented in subsequent follow-up analyses.
Safety results
Treatment-related adverse events (TRAEs) and immune-related AEs were summarized in Table 3. The most common TRAEs of any grade during the treatment were leukopenia (69.6%, 32/46), nausea (65.2%, 30/46) and radiation proctitis (54.3%, 25/46). The most common Grade 3 AEs were leukopenia (6.52%, 3/46) and neutropenia (4.35%, 2/46). No grade-4 or -5 TRAEs were recorded during the study. Two patients experienced immune-related AEs: one patient had prolonged hospitalization due to grade-3 drug rash and one had diarrhea and was treated with an anti-diarrheal agent.
Among the 45 patients who underwent radical surgery, four developed postoperative complications, including anastomotic leakage (Grade 2), anastomotic bleeding (Grade 2), incisional infection (Grade 1) and intestinal obstruction (Grade 2). No postoperative complications with Grade 3 or above occurred (Supplementary Table 2). The median interval from surgery to hospital discharge was 8 days (range, 6–15 days). No recurrence or death was observed during follow-up.
Discussion
The SILAR study unveils the efficacy and safety of CRT combined with PD-1 blockade in pMMR/MSS LARC patients with an intermediate or high Immunoscore. Our results indicated that the addition of sintilimab combined with a long course of CRT achieved a pCR rate of 65.2% (30/46), a CR rate of 54.3% and an ORR of 93.5% with no cases of PD. The pre-defined primary endpoint was achieved, signifying the success of the SILAR clinical trial. In addition, the combination was well tolerated, without new or unexpected safety concerns.
Preoperative radiotherapy combined with chemotherapy has been found to be effective for patients with rectal cancer, with studies demonstrating tumor downstaging, reduced local recurrence and acceptable toxicity16,22,23. However, the pCR rate in these studies varied from 17 to 30% in patients with rectal cancer16,22,23. In this study, the pCR rate of 65.2% is promising, indicating that our therapeutic approach could potentially offer more opportunities for sphincter-preserving surgery. Our results also hint at the possibility that more patients with LARC, especially those with a high Immunoscore, might achieve a clinical complete response and benefit from organ preservation through a watch-and-wait strategy or local excision. These improvements might be largely attributed to the enhancement in patient screening, and the effectiveness of this approach remains to be confirmed in future randomized trials.
Previous studies have indicated that chemotherapy and radiotherapy have the potential to promote lymphocyte infiltration, enhance antitumor immunity and thus could achieve a synergistic effect combined with ICIs for pMMR tumors24. In fact, recent studies have explored the combination of immunotherapy strategies and CRT as neoadjuvant in pMMR LARC12,17,20,25,26,27,28. The NRG-GI002 study reported a pCR rate of 31.9% (22/69) in pMMR LARC patients receiving preoperative CRT and sequential intravenous pembrolizumab, with grade-3 to -4 adverse events of 48.2% (39/81)20. A recent single-arm phase II trial from China reported patients receiving total neoadjuvant therapy (TNT) with induction chemoimmunotherapy followed by long-course chemoradiation, achieving a pCR rate of 33.3% (7/21)17. From another study by Lin et al., 46.2% (12/26) patients with pMMR LARC achieved pCR after receiving preoperative SCRT combined with subsequent CAPOX and camrelizumab25. Nevertheless, 73.3% (22/30) of patients experienced an immune-related AE of reactive cutaneous capillary endothelial proliferation25. In this study, we enrolled pMMR LARC patients with intermediate or high Immunoscore and adopted a combination of CRT and PD-1 blockade. It is interesting that a tumor shrinkage of 30% or greater was identified in 93.5% (43/46) of patients. Although cross-study comparisons should be made with caution, the pCR rate of 65.2% (30/46) was higher than that in the EPOC 1405 VOLTAGE study26 (29.7%, 11/37), Averectal27 study (37.5%, 15/40), NECTAR28 study (40%, 20/50) and the studies mentioned above17,20,25. And it is worth noting that the regimen was generally well tolerated, without an increase in the incidence of immune-related AEs.
To our knowledge, the Immunoscore test is the first internationally validated standardized assay for quantifying the immune infiltrate24. As for rectal cancer, a recent study showed that patients with dMMR LARC achieved a 100% CR rate after PD-1 blockade, which indicated that the in situ immune response boosted by the immunotherapy could eradicate the tumor and prevent recurrences5. The Immunoscore biopsy (ISB) has emerged as a valuable tool for predicting both prognosis and treatment response in LARC11,12. Specifically, high ISB has been associated with prolonged disease-free survival, improved histologic response to neoadjuvant therapy, and successful outcomes in watch-and-wait strategies, while low ISB serves as an indicator of elevated recurrence risk29. Building on these findings, Zhao et al. further demonstrated that integrating ISB with radiomics and pathomics could enhance the prediction of distant metastasis30. However, despite these promising advances, important challenges remain, particularly regarding biopsy heterogeneity and the need for further validation of functional outcomes in organ-preservation approaches31. In the current study, ISB was also utilized to enroll patients with intermediate or high Immunoscore. A pCR rate of 65.2% was achieved, indicating that a majority of LARC patients in this study might have the opportunity to benefit from organ preservation. Interestingly, among the seven participants with high Immunoscore, the pCR rate was 85.7% (6/7), while the MPR rate was 14.3% (1/7). These results may have significant implications in selecting potential candidates for organ preservation, especially those with a high ISB. Notably, the presence of a suppressive microenvironment in pMMR patients and treatment resistance guided by independent tumor-intrinsic factors should not be ignored, since not all patients achieve a good response to our combination therapy strategy.
Our study had some limitations. This was a single-arm trial with a relatively small sample size and a short postoperative follow-up, and no control group was included. The absence of a low-Immunoscore control group posed a limitation to directly validate the predictive value of Immunoscore. Therefore, our efficacy results might be subject to selection bias and should be interpreted as preliminary. Although this prospective study showed promising short-term outcomes, such as pCR rate, the long-term efficacy of the combination perioperative therapy was not assessed. Larger prospective studies stratified by Immunoscore levels with long-term follow-up are necessary to clarify whether this biomarker can predict the efficacy of neoadjuvant immune checkpoint inhibitors.
Overall, this single-arm trial provides preliminary evidence that sintilimab combined with CRT shows promising efficacy and an acceptable safety profile for pMMR LARC patients with an intermediate or high Immunoscore. This strategy may contribute to high CR and increased opportunities for organ preservation in this selected population. A large clinical multi-center randomized trial of all comers stratified by Immunoscore is required.
Methods
Study design and participants
This investigator-initiated, open-label, single-arm, phase II study was undertaken at the Sixth Affiliated Hospital of Sun Yat-sen University in Guangzhou, China, with a specific focus on patients diagnosed with LARC. The eligible participants, aged between 18 and 75 years, had a diagnosis of T3–4N0M0 or T1–4N+M0 rectal adenocarcinoma with an intermediate or high Immunoscore, with an inferior margin of ≤12 cm from the anal verge. In addition to these criteria, participants were required to have an Eastern Cooperative Oncology Group (ECOG) performance status score of 0–1 and adequate anal function. Key exclusion criteria included recurrent rectal cancer, complications such as active bleeding, perforation, or the need for emergency surgery, pre-existing or coexisting other malignant tumors (excluding those curatively treated and disease-free for more than 5 years), any previous systemic anticancer therapy for rectal cancer and active or historical autoimmune disease or systemic immune suppression. Patients with low Immunoscore were excluded and received standard-of-care treatment per NCCN guidelines (neoadjuvant CRT with mFOLFOX6 followed by total mesorectal excision). This trial was registered at https://www.clinicaltrials.gov before participant enrollment (NCT05450029). The study protocol received approval from the Institutional Review Board (IRB) of Sun Yat-sen University and adhered to the principles of the Declaration of Helsinki. All patients provided written informed consent. For the full study design, refer to the research protocol in the Supplementary file.
Procedures
In this trial, participants underwent preoperative treatment, which included six cycles of mFOLFOX6. This regimen comprised intravenous oxaliplatin (85 mg/m2 on day 1), intravenous leucovorin (400 mg/m2 on day 1) and intravenous bolus fluorouracil (400 mg/m2 on day 1, followed by continuous infusion of 1200 mg/m2/day for 2 days). Patients received one 14-day cycle of mFOLFOX6, followed by sintilimab (3 mg/kg intravenously over 30 min) on day 1 of each subsequent 14-day cycle. Concurrent radiotherapy was delivered at 2.0 Gy per fraction once daily (Monday to Friday) during cycles 2–4, totaling 50 Gy in 25 fractions over 5 weeks. Surgical intervention, adhering to the principles of total mesorectal excision, was scheduled 4 weeks after chemotherapy ends and 7 weeks after the completion of radiotherapy.
The patients underwent a comprehensive baseline assessment, which involved the collection of demographic information, medical history, and disease characteristics before enrollment. Systematic physical examinations, coupled with relevant laboratory and imaging tests, were conducted both before and after treatment. Radiological evaluations of tumors, utilizing enhanced thoracoabdominal CT scans and pelvic MRI, were carried out at baseline and before surgery according to the 8th edition of the American Joint Committee on Cancer (AJCC) tumor-node-metastasis (TNM) classification for rectal cancer32. The criteria for tumor staging were defined as follows: tumors invading through the muscularis propria into pericolorectal tissues were categorized as T3-stage, while tumors invading the visceral peritoneum or directly adhering to adjacent organs or structures were classified as T4-stage. Regional lymph nodes were deemed metastatic if they exhibited a short-axis diameter exceeding 10 mm and displayed a round shape or if they had a short axis of 5–9 mm and met at least two of the following criteria: having a round shape, having an irregular border, or exhibiting a heterogeneous signal intensity on CT or MRI.
Pretreatment tumor biopsies were obtained during colonoscopies and subjected to an Immunoscore test. The Immunoscore assessment, based on the Immunoscore scoring system (HalioDx, Marseille, France) and validated in the international Immunoscore Consortium study6, categorized all patients as having an intermediate or high Immunoscore. Briefly, 4-μm formalin-fixed paraffin-embedded tumor sections underwent automated CD3/CD8 immunohistochemical staining. T-cell densities in the tumor region were quantified and converted to percentiles relative to the reference cohort. The Immunoscore was subsequently categorized into three groups: low [Lo] (mean percentile 0%–25%), intermediate [Int] (>25%–70%) and high [Hi] (>70%–100%). Resected tumors underwent a thorough examination, and the regression of resected tumors was assessed by estimating the percentage of residual viable tumor in the macroscopically identifiable tumor bed, as identified through routine hematoxylin and eosin (H&E) staining33. All tissue selection and quality control were performed by our institutional Quality Control Committee (3 pathologists and 2 surgeons), with unanimous agreement required for each case. Patients received postoperative surveillance with quarterly clinical evaluations, including carcinoembryonic antigen testing. Thoracoabdominal CT scans were performed every 3 months throughout follow-up. Additionally, colonoscopy was conducted every 6 months during the first 3 postoperative years.
Endpoints and assessments
The primary endpoint in this study was the pCR rate. pCR was specifically characterized as the absence of any remaining viable cancer cells in both the resected primary tumor specimen and all sampled regional lymph nodes (ypT0N0). All histological slides of enrolled patients were submitted to two expert pathologists (Prof. Huang and Prof. Wang) for independent central review to confirm pCR. Additional secondary endpoints included the R0 resection rate, defined as the rate of achieving negative margins microscopically, with CRM distance >1 mm from the tumor to the mesorectal fascia, the 3-year event-free survival rate, defined as the percentage of patients without disease recurrence, progression, or death due to any cause at the 3-year follow-up, the 3-year overall survival rate, defined as the percentage of patients alive at the 3-year follow-up, the clinical tumor response, the complication rate and safety. The clinical tumor response was assessed pre-operation according to Response Evaluation Criteria in Solid Tumors (RECIST) v1.1 through MRI34. Partial response (PR) rate plus complete response (CR) rate was the objective response rate (ORR). Safety was evaluated by assessing treatment-related adverse events (TRAEs), recorded from the time of informed consent until 30 days after surgery. Adverse events (AEs) were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events, version 5.0 (CTCAE; version 5.0).
Statistical analysis
The trial adopted the optimal version of Simon’s two-stage design, which provides a fixed sample size framework suitable for assessing binary endpoints while maintaining statistical rigor. The null hypothesis for our study was a pCR rate set at 30%, based on our clinical experience and the previously published pCR rate of 27.5% in the FOWARC study22. The alternative hypothesis was a pCR rate of ≥50%. According to the Simon two-stage design, a minimum of 15 patients were required in stage I, and if at least 6 of them achieved pCR, an additional 31 patients were required to be enrolled in stage II. With 46 patients in this stage, a cumulative total of 19 or more responses over the two stages would indicate that this regimen is efficacious and warrants further investigation. This design would have 80% power to reject the null hypothesis at a significance level of one-sided 0.05. We did all efficacy and safety analyses in the intention-to-treat population.
Continuous variables were presented as the median (interquartile range), while categorical variables were reported as frequencies (percentages). Descriptive statistics were applied for the analysis of efficacy and safety, and the two-sided 95% confidence interval (CI) of the proportion parameter was calculated using the exact (Clopper–Pearson) method. The median follow-up time was estimated using the inverse Kaplan-Meier method. Statistical analyses were performed using SPSS (version 25.0, IBM) and R (version 4.1.3).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The Clinical raw data are not publicly available due to involving patient privacy, but can be accessed on request from the corresponding author for 10 years. Any request should be sent to the corresponding author, via email at kangl@mail.sysu.edu.cn, along with a detailed description of your research protocol; individual deidentified participant data will be shared. Please allow 1 month for a response to requests. The corresponding author and the Sixth Affiliated Hospital of Sun Yat-sen University will evaluate the reasonability of the request for our data and reserve the right to decide whether to share the data or not. Data may only be used for research purposes. All data shared will be de-identified and will be available for 1 year after access is granted. The study protocol is available as Supplementary Note in the Supplementary Information file. Source data are provided with this paper.
References
Holder, A. M. et al. Defining clinically useful biomarkers of immune checkpoint inhibitors in solid tumours. Nat. Rev. Cancer 24, 498–512 (2024).
Chalabi, M. et al. Neoadjuvant immunotherapy leads to pathological responses in MMR-proficient and MMR-deficient early-stage colon cancers. Nat. Med. 26, 566–576 (2020).
Hu, H. et al. Neoadjuvant PD-1 blockade with toripalimab, with or without celecoxib, in mismatch repair-deficient or microsatellite instability-high, locally advanced, colorectal cancer (PICC): a single-centre, parallel-group, non-comparative, randomised, phase 2 trial. Lancet Gastroenterol. Hepatol. 7, 38–48 (2022).
Xiao, B. Y. et al. Neoadjuvant immunotherapy leads to major response and low recurrence in localized mismatch repair-deficient colorectal cancer. J. Natl Compr. Cancer Netw. 21, 60–66.e65 (2023).
Cercek, A. et al. PD-1 blockade in mismatch repair-deficient, locally advanced rectal cancer. N. Engl. J. Med. 386, 2363–2376 (2022).
Pagès, F. et al. International validation of the consensus immunoscore for the classification of colon cancer: a prognostic and accuracy study. Lancet 391, 2128–2139 (2018).
Marliot, F. et al. Analytical validation of the immunoscore and its associated prognostic value in patients with colon cancer. J. Immunother. Cancer 8, e000272 (2020).
Galon, J. et al. Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313, 1960–1964 (2006).
Bruni, D., Angell, H. K. & Galon, J. The immune contexture and immunoscore in cancer prognosis and therapeutic efficacy. Nat. Rev. Cancer 20, 662–680 (2020).
Anitei, M. G. et al. Prognostic and predictive values of the immunoscore in patients with rectal cancer. Clin. Cancer Res. 20, 1891–1899 (2014).
El Sissy, C. et al. A diagnostic biopsy-adapted immunoscore predicts response to neoadjuvant treatment and selects patients with rectal cancer eligible for a watch-and-wait strategy. Clin. Cancer Res. 26, 5198–5207 (2020).
El Sissy, C. et al. International validation of the immunoscore biopsy in patients with rectal cancer managed by a watch-and-wait strategy. J. Clin. Oncol. 42, 70–80 (2024).
Cercek, A. et al. Mismatch repair-deficient rectal cancer and resistance to neoadjuvant chemotherapy. Clin. Cancer Res. 26, 3271–3279 (2020).
Le, D. T. et al. PD-1 blockade in tumors with mismatch-repair deficiency. N. Engl. J. Med. 372, 2509–2520 (2015).
Conroy, T. et al. Neoadjuvant chemotherapy with FOLFIRINOX and preoperative chemoradiotherapy for patients with locally advanced rectal cancer (UNICANCER-PRODIGE 23): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 22, 702–715 (2021).
Bahadoer, R. R. et al. Short-course radiotherapy followed by chemotherapy before total mesorectal excision (TME) versus preoperative chemoradiotherapy, TME, and optional adjuvant chemotherapy in locally advanced rectal cancer (RAPIDO): a randomised, open-label, phase 3 trial. Lancet Oncol. 22, 29–42 (2021).
Li, Y. et al. Total neoadjuvant therapy with PD-1 blockade for high-risk proficient mismatch repair rectal cancer. JAMA Surg. 159, 529–537 (2024).
Xia, F. et al. Randomized phase II trial of immunotherapy-based total neoadjuvant therapy for proficient mismatch repair or microsatellite stable locally advanced rectal cancer (TORCH). J. Clin. Oncol. 42, Jco2302261 (2024).
Xiao, W. W. et al. Effect of neoadjuvant chemoradiotherapy with or without PD-1 antibody sintilimab in pMMR locally advanced rectal cancer: a randomized clinical trial. Cancer Cell 42, 1570–1581.e4 (2024).
Rahma, O. E. et al. Use of total neoadjuvant therapy for locally advanced rectal cancer: initial results from the pembrolizumab arm of a phase 2 randomized clinical trial. JAMA Oncol. 7, 1225–1230 (2021).
Oaknin, A. et al. Safety, efficacy, and biomarker analyses of dostarlimab in patients with endometrial cancer: interim results of the phase I GARNET study. Clin. Cancer Res. 29, 4564–4574 (2023).
Deng, Y. et al. Modified FOLFOX6 with or without radiation versus fluorouracil and leucovorin with radiation in neoadjuvant treatment of locally advanced rectal cancer: initial results of the Chinese FOWARC multicenter, open-label, randomized three-arm phase III trial. J. Clin. Oncol. 34, 3300–3307 (2016).
Fokas, E. et al. Randomized phase II trial of chemoradiotherapy plus induction or consolidation chemotherapy as total neoadjuvant therapy for locally advanced rectal cancer: CAO/ARO/AIO-12. J. Clin. Oncol. 37, 3212–3222 (2019).
Heinhuis, K. M. et al. Enhancing antitumor response by combining immune checkpoint inhibitors with chemotherapy in solid tumors. Ann. Oncol. 30, 219–235 (2019).
Lin, Z. et al. Phase II, single-arm trial of preoperative short-course radiotherapy followed by chemotherapy and camrelizumab in locally advanced rectal cancer. J. Immunother. Cancer 9, e003554 (2021).
Bando, H. et al. Preoperative chemoradiotherapy plus nivolumab before surgery in patients with microsatellite stable and microsatellite instability-high locally advanced rectal cancer. Clin. Cancer Res. 28, 1136–1146 (2022).
Shamseddine, A. et al. Short-course radiation followed by mFOLFOX-6 plus avelumab for locally-advanced microsatellite stable rectal adenocarcinoma: the Averectal study. Eur. J. Cancer 222, 115428 (2025).
Yang, Z. et al. Efficacy and safety of PD-1 blockade plus long-course chemoradiotherapy in locally advanced rectal cancer (NECTAR): a multi-center phase 2 study. Signal Transduct. Target. Ther. 9, 56 (2024).
Zeng, Q. et al. Prognostic and predictive value of a modified diagnostic biopsy-adapted immunoscore in patients with rectal cancer after neoadjuvant treatment: a translational study from the STELLAR trial. Int. J. Radiat. Oncol. Biol. Phys. 121, 640–650 (2025).
Zhao, R. et al. Integrating radiomics, pathomics, and biopsy-adapted immunoscore for predicting distant metastasis in locally advanced rectal cancer. ESMO Open 10, 104102 (2025).
Gao, Z., Zhu, Z. & Li, K. Immunoscore biopsy in patients with rectal cancer managed by a watch-and-wait strategy. J. Clin. Oncol. 42, 1088 (2024).
Weiser, M. R. AJCC 8th edition: colorectal cancer. Ann. Surg. Oncol. 25, 1454–1455 (2018).
Quirke, P., Durdey, P., Dixon, M. F. & Williams, N. S. Local recurrence of rectal adenocarcinoma due to inadequate surgical resection. Histopathological study of lateral tumour spread and surgical excision. Lancet 2, 996–999 (1986).
Hodi, F. S. et al. Evaluation of immune-related response criteria and RECIST v1.1 in patients with advanced melanoma treated with pembrolizumab. J. Clin. Oncol. 34, 1510–1517 (2016).
Acknowledgements
We thank the patients who participated in this study and their families, as well as the nursing and research staff at the study center. We are also grateful for the time and efforts of Genecast Biotechnology Co., Ltd. and Innovent Biologics. This work is supported by the National Key Clinical Discipline, National Natural Science Foundation of China (82470696, X.Z.; 82473451, H.L.; 82200569, H.L.; 82472930, L.K.), Guangdong Basic and Applied Basic Research Foundation (2022A1515012498, X.Z.; 2024A1515010956, X.Z.), the program of Guangdong Provincial Clinical Research Center for Digestive Diseases (2020B1111170004), Science and Technology Projects in Guangzhou (2024A04J6400, X.Z.), Sun Yat-sen University Clinical Research 5010 Program (2016005, L.K.), and the Innovative Clinical Technique of Guangzhou.
Author information
Authors and Affiliations
Contributions
L.K., Y.L. and L.H. contributed equally to this trial. X.Z., H.L., L.S. and Z.Z. were responsible for the conception and design of the study. X.Z., H.L., L.S., Z.Z., X.Z., S.L., Y.C., Z.L., Z.L., Y.H., Z.Y., X.H., J.K., L.H., Y.L. and L.K. contributed to the collection of data. L.S. was responsible for the statistical analysis and interpretation and the data review. X.Z., H.L., L.S., Z.Z., Y.L. and L.K. accessed and verified the data. All authors read and approved the final draft of the article.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Matthew Strickland and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zheng, X., Liu, H., Shi, L. et al. Neoadjuvant chemoradiotherapy plus sintilimab in proficient mismatch repair locally advanced rectal cancer with intermediate/high-immunoscore (SILAR): a single-arm phase II trial. Nat Commun 17, 772 (2026). https://doi.org/10.1038/s41467-025-65162-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-65162-8