Abstract
Reactive capture integrates CO2 capture and electrochemical conversion into CO — a key building block in the synthesis of industrial chemicals and fuels — avoiding costly regeneration steps and improving efficiency. Amino acid salt solutions, which offer rapid CO2 capture, facile CO2 release, O2 tolerance, and low toxicity, are promising sorbents for reactive capture. However, we find that amino acids can adsorb to common CO-producing catalysts, covering the active sites and deactivating the catalyst, and that they bind less to nickel phthalocyanine (NiPc). Still, when tested for reactive capture systems — where CO2 supply is inherently limited — NiPc’s performance is constrained by its weak CO2 adsorption and activation. Here we develop a nickel molecular catalyst supported on carbon nanotubes with a conjugated NiPc framework that resists amino acid adsorption and a coordinatively unsaturated Ni-N3 structure that promotes CO2 adsorption and enhances CO selectivity. As a result, we achieve 94% CO Faradaic efficiency at 100 mA cm–2 with an energy efficiency of 42% and an energy cost of 25 GJ tCO–1.
Similar content being viewed by others
Introduction
The industrial synthesis of long-chain hydrocarbons, aldehydes, and alcohols relies on carbon monoxide (CO) as the starting material1. Efficient conversion of carbon dioxide (CO2) into CO can therefore unlock sustainable synthesis pathways broadly—from chemicals to aviation fuels2. The electrochemical CO2 reduction reaction (CO2RR), powered by renewable electricity, offers a promising route for converting CO2 to CO. However, conventional CO2RR requires high-purity CO2, typically sourced through capture processes that rely on energy- and capital-intensive, fossil-fuel-powered thermal regeneration3.
Electrochemical CO2 reactive capture bypasses CO2 regeneration and offers a more efficient pathway to produce CO (Fig. 1a). In a reactive capture system, absorbed CO2 in the capture solution is released through a pH swing induced by protons generated at the anode; this regenerated CO2 is then converted into CO at the cathode (Fig. 1b). An ideal capture solution should combine a high CO2 absorption rate, facile CO2 desorption to supply sufficient CO2 for conversion, tolerance to oxygen (O2), recyclability, and low toxicity.
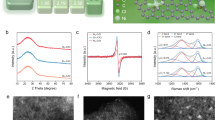
a Schematic of the upstream and downstream processes to transform CO2 into gaseous product using gas-phase CO2 electrolysis and reactive capture. b Schematic of the amino-acid-based reactive capture system. c A radar plot comparison of different capture solution systems. CO2/e represents the number of CO2 produced per electron. Source data are provided as a Source data file.
Alkaline hydroxide-based solutions absorb CO2 with rapid sorption kinetics, forming carbonate, which can then be fed into a reactive capture process to generate CO4,5. However, carbonate-to-CO2 release via a pH swing consumes two protons per CO2 molecule, limiting the local CO2 availability at the catalyst and lowering electron selectivity towards CO (Supplementary Tables 1–4). This fundamental limitation restricts the Faradaic efficiency (FE) to less than 50% towards CO from carbonate solutions. Further purging down carbonate with CO2 generates bicarbonate, which reduces proton demand to one proton per CO2 molecule and improves local CO2 availability; however, the inherently slow capture rate of carbonate renders this approach impractical for capturing diluted CO2 streams6.
Amine-captured CO2 solutions exist as bicarbonate and carbamate, each requiring only one proton to regenerate each CO2 molecule7,8,9. This enables higher local CO2 concentration at the cathode (Supplementary Fig. 1), offering the potential to achieve higher FE towards CO. However, conventional amines are volatile and prone to O2 degradation, making them unsuitable for capturing CO2 from O2-rich streams10, such as air.
Amino acid salt solutions exhibit high CO2 absorption rates and capacities comparable to or greater than conventional amine sorbents, while offering O2 tolerance, non-corrosiveness and environmental friendliness (Fig. 1c and Supplementary Table 5)11,12,13,14. Amino acids are also priced comparably to common capture reagents, such as monoethanolamine15. These advantages have led to the large-scale use of amino acid salts in CO2 capture from flue gas16,17,18 and air15. Like conventional amines, amino acid salts, such as potassium glycinate (K-Gly), capture CO2 to generate bicarbonate and carbamate (Supplementary Note 1, Supplementary Figs. 2–4, and Supplementary Table 6). These advantages make them promising candidates for high CO selectivity in reactive capture systems13. Indeed, a preliminary study combining amino acid sorbents with reactive capture achieved CO FEs of up to 53% at ambient conditions and 64% at elevated temperature and pressure13. However, further performance gains are required for practical implementation.
In this work, we identify a key factor underlying prior performance limitations: strong interactions between amino acids and common CO-producing catalysts can block active sites and suppress activity. Guided by density functional theory (DFT) and supported by X-ray photoelectron spectroscopy (XPS) and electrochemical measurements, we find that amino acids bind far less strongly to nickel phthalocyanine (NiPc). However, when tested for reactive capture, NiPc’s performance remained constrained by its weak CO2 adsorption and activation. To address this, we retained the NiPc framework for amino acid tolerance and incorporated a coordinatively unsaturated Ni-N3 structure to promote CO2 binding. As a result, the catalyst achieves 94% CO FE at 100 mA cm–2 with an energy efficiency (EE) of 42%, and maintains performance when directly capturing and converting contaminant-bearing flue gas, demonstrating its practical applicability for industrial reactive capture.
Results and discussion
Amino acid adsorption in reactive capture
We explored amino acid salts for reactive capture, selecting K-Gly as a representative capture fluid. We began by testing a silver (Ag) catalyst—a well-established catalyst known for efficiently converting CO2 to CO—to evaluate how amino acid influences its catalytic selectivity during reactive capture (Supplementary Fig. 5). Using 0.5 M KHCO3 as the catholyte, the Ag catalyst demonstrated a CO FE of 66.4% at 100 mA cm–1, comparable to previous report19. However, upon adding 0.1M and 0.3M K-Gly to the electrolyte, CO FE decreased to 46.7% and 34.4%, respectively. We attributed this decline to glycine adsorption onto the Ag catalyst, blocking active sites for CO2 conversion to CO20,21.
To assess glycine adsorption, we performed XPS, observing a 0.3 eV decrease in the oxidation state of Ag for the glycine-treated catalyst compared to the untreated Ag catalyst (Supplementary Fig. 6a). We further measured the electrochemical active surface area-normalized CO partial current density and found that it reduced upon adding K-Gly, suggesting a decrease in its intrinsic activity due to glycine adsorption (Supplementary Note 2 and Supplementary Fig. 8m). Since changes in the catholyte composition may also affect local CO2 availability during reactive capture, we conducted gas-phase CO2RR experiments in a flow cell supplied with excess CO2 to rule out the possibility that reduced CO production stemmed from limited local CO2. A similar trend emerged: the Ag catalyst achieved over 90% CO FE when using 1M KCl as the catholyte, but this FE decreased when the catholyte was replaced with 1M K-Gly (Fig. 2a). These results exhibited that glycine adsorption, rather than changes in CO2 availability, caused the observed performance deterioration. While previous study showed that small, submonolayer amounts of amino acids may enhance CO2RR22, the presence of amino acids in bulk solution, and continuous exposure can impair performance.
CO2RR performance comparisons of Ag nanoparticles (a), NiPc–CNT (b) and NiN3–Pc–CNT (c) using different catholytes and CO2 concentrations. Schematic illustration of postulated mechanisms for K-Gly-based reactive capture using glycine-absorbing catalyst (d), glycine-free catalyst with weak CO2 adsorption (e) and glycine-free catalyst with moderate CO2 adsorption (f). Source data are provided as a Source data file.
In search of catalysts that are tolerant to amino acids, we performed DFT calculations to study glycine binding energies across various CO-producing catalysts (Supplementary Fig. 9). Atomically dispersed catalysts exhibited much lower binding energy compared to nanoparticles. Among these, NiPc showed the weakest binding energy with glycine, suggesting its potential for amino-acid-based reactive capture. XPS analysis further showed minimal glycine adsorption on NiPc, as evidenced by negligible changes in the Ni 2p peak (Supplementary Fig. 6b).
However, initial experiments employing NiPc alone in K-Gly-based reactive capture resulted in hydrogen production exclusively (Supplementary Fig. 10a), likely due to NiPc aggregation (Supplementary Fig. 10b, c). To prevent aggregation, we dispersed NiPc onto carbon nanotube (CNT), generating a NiPc–CNT catalyst. Yet, the CO FE remained low (~30% at 50 mA cm−2) and shifted further towards hydrogen production at higher current densities (Supplementary Fig. 10d–i).
To determine why NiPc–CNT—despite minimal adsorptionṣ—still exhibited limited CO FE, we performed gas-phase CO2RR in a flow cell, isolating the effect of amino acid adsorption from local CO2 availability. We obtained CO FE > 95% with and without K-Gly in the catholyte (Fig. 2b), supporting its amino acid tolerance. We thus hypothesized that the poor selectivity observed during reactive capture resulted from limited local CO2 availability near the catalyst, combined with the inherently weak CO2 adsorption of NiPc–CNT23.
To simulate low local CO2 conditions in reactive capture24, we tested NiPc–CNT in a gas-phase flow cell with diluted CO2 (15 and 25 vol% CO2, diluted by Ar). The CO FE decreased with lower CO2 concentrations, with the decline becoming more pronounced at higher current densities, indicating that the poor selectivity of NiPc–CNT in reactive capture stemmed from the limited local CO2 supply.
Therefore, our results point to two distinct failure modes: conventional catalysts, like Ag, adsorb glycine and become blocked and inactivated (Fig. 2d), while saturated single atom catalysts, such as NiPc–CNT, although tolerant to glycine, exhibit weak CO2 adsorption and thus fail to convert CO2 (Fig. 2e). Thus, the ideal catalyst for amino-acid-based reactive capture should combine amino acid tolerance with moderate CO2 adsorption for high CO FE under low CO2 concentrations (Fig. 2f).
Unsaturated nickel molecular catalyst
While NiPc—with a saturated Ni-N4 structure—is amino-acid-tolerant, it exhibits unfavorable CO2 adsorption and activation23. Adjusting coordination number in molecular catalysts modulates local electronic densities around metal centers, influencing CO2 adsorption and catalytic activity25,26,27. We hypothesized that introducing an unsaturated nickel coordination environment into NiPc could enhance CO2 adsorption and activation while preserving amino acid tolerance. DFT calculations further supported our hypothesis (Supplementary Note 3 and Supplementary figs. 11–14), revealing that the coordinatively unsaturated NiPc (NiN3–Pc) exhibits a more favorable Gibbs energy for *COOH adsorption than conventional NiPc.
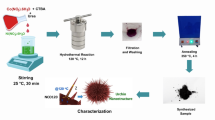
We therefore aimed to synthesize NiN3–Pc via a solvothermal method28,29 (Fig. 3a, synthesis details in the “Methods”). This solvothermal treatment induced partial 1,2,4,5-tetracyanobenzene (TCNB) polymerization with Ni2+ ions to form the conjugated NiPc structure on CNT (NiN3–Pc–CNT). Structural characterizations showed uniformly distributed Ni atoms on CNTs (Fig. 3b) without agglomeration (Fig. 3c, Supplementary Note 4 and Supplementary Fig. 15). Powder X-ray diffraction (XRD) patterns exhibited two broad peaks corresponding to graphite (002) and (101) planes without metallic Ni signals (Fig. 3d). UV–vis spectroscopy revealed NiN3–Pc–CNT has longer wavelength at Q band than NiPc–CNT (Fig. 3e), suggesting the formation of a polymeric phthalocyanine network in NiN3–Pc–CNT30. Raman and FTIR spectra showed that NiN3–Pc–CNT retained characteristic signals of NiPc, indicating the successful polymerization of TCNB during the preparation process (Supplementary Fig. 16g, h). Ni K-edge X-ray absorption near-edge structure (XANES) results indicated a slightly lower oxidation state for Ni atoms in NiN3–Pc–CNT relative to Ni2+ in NiPc (Fig. 3f and Supplementary Note 5). Extended X-ray absorption fine-structure (EXAFS) fitting further showed Ni coordination with three adjacent N atoms, validating successful formation of an unsaturated Ni–N3 structure (Fig. 3g, Supplementary Fig. 17, Supplementary Table 8, and Supplementary Data 1).
a Schematic illustration of atomically dispersed nickel molecular catalyst preparation. b High-angle annular dark-field scanning TEM (HAADF-STEM) image and energy dispersive X-ray spectroscopy (EDS) maps (C, red; N, green; Ni, pink) of NiN3–Pc–CNT. c HAADF-STEM image of NiN3–Pc–CNT. d X-ray diffraction patterns of NiN3–Pc–CNT, NiPc–CNT, CNT and NiPc. The JCPDS profiles of graphite (blue) and metallic nickel (dark yellow) are displayed for reference. e UV–vis spectra of NiN3–Pc–CNT and NiPc–CNT. f, g Ni K-edge XANES spectra, along with marked positions for the comparison of oxidation state (these positions were determined by the first inflection point in the edge region) (f) and Ni K-edge EXAFS spectra (g) of NiN3–Pc–CNT, NiPc–CNT and Ni foil. Voltages are not iR-corrected. Source data are provided as a Source data file.
We further assessed NiN3–Pc–CNT using XPS to verify its amino-acid tolerance. The negligible change in Ni 2p binding energy upon glycine treatment suggested minimal glycine adsorption (Supplementary Fig. 6c). The amino-acid-tolerant property of NiN3–Pc–CNT was further verified by its unchanged CO FE in CO2RR with and without K-Gly ( > 95%) (Fig. 2c and Supplementary Fig. 7c). Notably, even under diluted CO2 conditions (15 and 25 vol% CO2)—simulating low local CO2 availability in reactive capture—NiN3–Pc–CNT retained CO FE > 95% across a broad range of current densities, highlighting its robust catalytic performance.
Reactive capture performance
We evaluated the electrochemical performance of NiN3–Pc–CNT in a membrane electrode assembly using 1M K-Gly as the capture solution. By optimizing catalyst loading, ionomer content and nickel loading, we achieved a maximum FE towards CO of 70% at 100 mA cm−2 (Supplementary Note 6 and Supplementary Fig. 18). Recognizing that the catholyte flow rate influences CO2 mass transfer to the catalyst surface31, we further optimized it, obtaining a peak CO FE of 94% with an EE of 42% at 100 mL min−1 (Fig. 4a and Supplementary Fig. 19).
a FE(CO)s using NiN3–Pc–CNT at different catholyte flowrates with 1M post-capture K-Gly. b FE(CO)s of NiN3–Pc–CNT, NiPc–CNT and Ag using 1M post-capture K-Gly at 80 mL min−1. c EE(CO)s and full cell voltage of NiN3–Pc–CNT and NiPc–CNT using 1 M post-capture K-Gly at 80 mL min−1. d Comparison of the maximum FE(CO) and current density over NiN3–Pc–CNT and the start-of-the-art catalysts used in reactive capture. NiN3–Pc–CNT shows distinct CO FE in reactive capture (yellow star, at 100 mA cm−2; orange star, at 200 mA cm−2). Hydroxide-based systems are reported in refs. 4,5, amine-based systems are reported in refs. 7,8,9,13. e FE(CO)s comparison using different amino-acid-salt solutions at 80 mL min−1. f FE(CO)s using 1M post-capture K-Gly from simulated flue gas (pH 8.3) and air (pH 9.6) at 80 mL min−1. The error bars indicate one standard deviation based on three measurements. Source data are provided as a Source data file.
NiN3–Pc–CNT showed competitive performance compared with NiPc–CNT and Ag catalysts in K-Gly-based reactive capture (Fig. 4b and Supplementary Fig. 20a–c), achieving a CO EE of 40% at 80 mL min−1 (Fig. 4c). We further investigated the impact of catholyte pH (Supplementary Fig. 20d), finding that lower pH improved partial current densities towards CO2 conversion, which we attributed to the enhanced local CO2 availability at the catalyst surface. NiN3–Pc–CNT catalyst also achieved over 95% CO FE across a wide range of current densities when 3M KHCO3 was used as the capture solution (Supplementary Fig. 20e). The NiN3–Pc–CNT catalyst surpasses previous literature benchmarks in both FE and EE for CO production (Fig. 4d and Supplementary Tables 9 and 10).
We performed a 10 h-stability test with NiN3–Pc–CNT at 100 mA cm−2 (Supplementary Fig. 21a), during which both cell voltage and CO FE remained stable. Structural characterizations after the stability test revealed that most Ni remained atomically dispersed but small Ni clusters appeared (Supplementary Fig. 21b, c), suggesting site aggregation may be a degradation mechanism. Analysis of bicarbonate and glycine carbamate concentrations in post-capture solution (pH 8.1) and post-electrolysis solution (pH 8.45) indicated that bicarbonate may be the primary in situ CO2 source, decreasing from 0.34 to 0.20 molCO2 molK-Gly−1, while glycine carbamate concentrations slightly increased from 0.25 to 0.29 molCO2 molK-Gly−1 (Supplementary Fig. 4a, b and Supplementary Table 6). Relative to the post-capture solution (pH 8.1), the post-electrolysis K-Gly solution showed only bicarbonate/carbamate redistribution with no new products (Supplementary Fig. 4c, d), indicating that the amino acid was neither degraded nor electroreduced. A control electrolysis in fresh 1M K-Gly without captured CO2 produced only H2 (Supplementary Fig. 21d), further supporting that direct amino acid electroreduction is unlikely.
We then assessed the versatility of NiN3–Pc–CNT by employing other amino acid salt solutions, including K-sarcosine and K-L-proline, as capture fluids (Fig. 4e and Supplementary Fig. 22a, b). We achieved CO FE over 75% in each case, demonstrating the catalyst’s board applicability. To demonstrate industrial relevance, we used amino acid salt solutions to capture diluted CO2 from flue gas and air (details in the “Methods”). Purging 1M K-Gly with (i) diluted CO2 (15% CO2, balance N2), (ii) simulated flue gas containing common contaminants (15% CO2, 10% O2, 500 ppm NO and 500 SO2), and (iii) air yielded solution pH values of 8.3, 8.3 and 9.6 (Supplementary Fig. 22c), and achieved CO FE of 83%, 83% and 34% at 100 mA cm−2, respectively (Fig. 4f). Although O2 and other flue-gas impurities dissolve in the aqueous capture solution and could, in principle, be reduced at the cathode (e.g., oxygen reduction reaction), their inherently low solubility (for O2, 1.2 mmol L−1 at 1 atm, 298 K) and concentrations limit the reaction32. The CO selectivity obtained with simulated flue gas containing common contaminates (15% CO2, 10% O2, 500 ppm NO and 500 ppm SO2) matched that under pure CO2 (Supplementary Fig. 22c–f). This result aligns with a recent study of Ni-based catalysts in bicarbonate electrolytes, which likewise reported negligible impact of SO2 and NO on CO production33.
We performed a system energy analysis including the energy cost for CO2 capture, conversion and regeneration (Supplementary Note 7, Supplementary Table 11 and Supplementary Fig. 23). The analysis estimated an energy cost of 24.6 GJ ton−1 for K-Gly-based reactive capture to produce CO, which is over 50% less than that of gas-phase CO2RR34.
Mechanistic insights in reactive capture
To investigate the origin of the improved activity towards CO production, we used in situ X-ray absorption spectroscopy (XAS) measurements to assess the chemical states of the catalysts during reaction. The XANES spectrum of NiPc–CNT during reaction closely matched the open-circuit potential (OCP) state (Supplementary Fig. 24). In contrast, the Ni K-edge positions for NiN3–Pc–CNT shifted by ~2 eV to lower energies during operation and returned to the OCP state afterward (Fig. 5a). This reversible shift suggested that the reduced Ni sites during reaction—likely in the metallic or monovalent state—are responsible for improved CO formation, consistent with the previous report35. EXAFS analysis revealed an increase in the Ni–N bond length from 1.38 Å (at OCP) to 1.41 Å under electrolysis conditions at 50 and 100 mA cm−2 (Fig. 5b), likely resulting from the distortion of Ni atoms out of the carbon plane36. This structure facilitates electron transfer from Ni sites to CO2 molecules, promoting CO2 activation and aligning with our earlier DFT findings (Supplementary Fig. 14).
Normalized operando Ni K-edge XANES spectra (a) and the corresponding k2-weighted Fourier transforms of NiN3–Pc–CNT at different current densities (b). OCP, open-circuit potential; OCP-2, return to open-circuit potential after all measurements. Source data are provided as a Source data file.
In conclusion, we developed a design principle for reactive capture electrocatalysis whereby the adsorption of amino acid is minimized to avoid catalyst deactivation. By utilizing the partial polymerization of TCNB during the catalyst preparation, we developed an unsaturated NiPc molecular catalyst (NiN3–Pc–CNT). We found that nickel single-atom catalysts, such as NiPc and NiN3–Pc–CNT, were amino-acid-tolerant yet exhibited different performance in amino-acid-based reactive capture. NiN3–Pc–CNT has a conjugated NiPc network with Ni-N3 structure, which achieves amino acid tolerance and improved CO2 adsorption and activation. This catalyst enabled the efficient electrolysis of an amino-acid-based capture medium, achieving a CO FE of 94% at 100 mA cm−2, demonstrating competitive performance with both amine- and hydroxide-based reactive capture systems. The integrated system achieved a 42% EE, requiring only 24.61 GJ of energy per ton CO produced, compared to 64 GJ for gaseous CO2 electrolysis and 56 GJ for hydroxide-based reactive capture. This study offers a pathway to designing CO2 capture and utilization systems that lower energy costs and simplifies process flows, making reactive capture competitive with sequential capture and conversion approaches.
Methods
Synthesis of carbon nanotube supported nickel molecular electrocatalysts
A solvothermal method was used to synthesize of NiN3–Pc–CNT. 5 mg of nickel (II) chloride (98%, Sigma-Aldrich) and 22.5 mg of 1,2,4,5-TCNB (97%, Sigma-Aldrich) were dissolved into 20 mL of ethanol to form a transparent solution. Subsequently, 87.5 mg of CNT (98% carbon basis, Sigma-Aldrich) were added, and the mixture was ultrasonicated for 30 min. The obtained mixture was heated at 180 °C for 8 h after transferring into a 50 mL Teflon-lined stainless-steel autoclave. The as-prepared sample of CNTs supported conjugated NiPc (NiN3–Pc–CNT) was washed with 1M hydrogen chloride solution, ethanol, and water several times, followed with freeze-drying for 48 h. The CNT supported Pc without nickel atoms (Pc–CNT) was prepared in a similar fashion to NiN3–Pc–CNT, except nickel (II) chloride was not added during the initial step. NiN3–Pc was also prepared in a similar fashion to NiN3–Pc–CNT, except adding CNT during the preparation.
Carbon-nanotube-supported nickel phthalocyanine (NiPc–CNT) was prepared by mixing NiPc (95%, Fisher Scientific) with CNT in N, N-dimethylformamide (DMF). Briefly, NiPc (5 mg) was added in 30 mL DMF, and CNTs (20 mg) were then added. The mixed solution underwent ultrasonic treatment for 1 h and then stirred at room temperature (25 °C) for 48 h. Finally, NiPc–CNT was collected by filtration, washed by DMF and water, followed with freeze-drying for 48 h.
Material characterization
The high-resolution TEM and the HAADF-STEM images were captured via Thermo Fisher Scientific Spectra Ultra equipped with a probe spherical aberration corrector and a high-brightness Schottky field emitter gun with ultra-stable monochromator source at 300 kV. EDS maps were acquired using the Ultra-X EDX system at a dwell time of 20 µs/pixel. Post-filtering of the EDS maps was done by averaging every 3 pixels using Velox software (version 3.10.0.1130-5d766716c0). X-ray powder diffractometer measurements were performed in a Philips XRD system with a PW 1830 HT generator, a PW 1050 goniometer, and PW3710 control electronics. The metal content of the catalysts was measured by inductively coupled plasma optical emission spectroscopy (Thermo Scientific iCAP Pro ICP OES). UV–vis absorption experiments were performed on a Lambda 365 UV/Vis spectrometer. Fourier-transform infrared spectroscopy was conducted in PerkinElmer Spectrum Two. XPS measurements were performed in a Thermo Scientific K-Alpha spectrophotometer with the monochromated Al Kα X-ray radiation source. For the XPS measurements to check the glycine adsorption of fresh and glycine-treated catalysts, we initially dispersed the catalyst into 1M K-Gly and stirred for 24 h. After being thoroughly washed by DI water and ethanol, the samples were dried in the oven before the measurement. FTIR spectra were recorded using a PerkinElmer Spectrum Two spectrometer. Raman spectra were collected on a Renishaw inVia Reflex spectrometer with a 633 nm excitation laser, 20 s exposure time, five accumulations, and 5% laser power.
XAS measurement
The experiment utilized a three-electrode configuration with working electrodes of NiPc–CNT or NiN3–Pc–CNT, each loaded at 1 mg cm−2. A platinum plate served as the counter electrode, and a saturated calomel electrode was used as the reference electrode. The electrolyte solution was CO2-saturated 1M K-glycine with a pH of 8.1. The electrolytes were prepared shortly before electrolysis experiments. XAS of XANES and EXAFS was performed in total-fluorescence-yield mode using a silicon drift detector at the BL32A beamline of TPS in National Synchrotron Radiation Research Centre, Taiwan. The baseline before the pre-edge was subtracted, and the spectra were normalized using the post-edge. EXAFS analysis included performing a Fourier transform on k2-weighted EXAFS oscillations to assess the contributions of each bonded pair to the peak in the Fourier transform. The position that was used to check the oxidation state of the catalyst was determined by the first inflection point in the edge region.
Electrochemical measurements of reactive capture
All electrochemical experiments were conducted using a membrane electrode assembly with data collected using a Autolab PGSTAT204 potentiostat. Post-capture solution consisting of 1M K-glycine (or 1M K-Sarcosine and 1M K-L-Proline) and 0.1M KH2PO4 as the supporting electrolyte was circulated as the catholyte. A 1M K-glycine solution was prepared by dissolving glycine and potassium hydroxide in deionized water at a 1:1 molar ratio, followed by dilution to the desired volume. We utilized 0.1M KH2PO4 to improve electrolyte conductivity, stabilize the pH in the CO2 diffusion layer and maintain stable cell voltage. In a typical experiment, 75 mL of the capture solution was purged with 100 sccm of pure CO2 until a pH of 8.1 was reached. The electrolytes were prepared shortly before electrolysis experiments. NMR analysis showed that after purging the 1M K-Gly solution (pH 10.8), the post-capture solution (pH 8.1) contained 0.59 mol glycine zwitterion, 0.16 mol glycinate, 0.25 mol glycine carbamate, and 0.34 mol bicarbonate (Supplementary Fig. 4 and Supplementary Table 6). The anolyte was 0.05M H2SO4 solution. During electrolyzer operation, the catholyte was constantly purged with 30 sccm Argon. Unless otherwise specified, the cathode ran at flowrate of 80 mL min−1 and anode at 20 mL min−1.
For the electrodes tested in reactive capture system, the cathode electrode was fabricated by spray-coating a nickel catalyst ink onto hydrophilic carbon paper (AvCarb MGL190, Fuel Cell Store). The catalyst ink for a 6.25 cm2 substrate was prepared by dispersing 8 mg of Nickel catalyst and 100 µL Nafion dispersion (5 wt%, Ion Power) in 4 mL of ethanol and sonicating until a homogeneous dispersion is formed. A catalyst loading of ~1 mg cm−2 was achieved. Pure NiPc electrode was also prepared via spraying the mixture of NiPc and 20 wt% Nafion (5 wt%, Ion Power) onto the electrode to achieve 1 mg cm−2. For the Ag-based electrode, the Ag ink was prepared by dispersing 50 mg of Ag nanoparticles (99.99%, 20 nm, US Research Nanomaterials) and 50 mg of Nafion dispersion in 2 mL of methanol and sonicating for 1 h before spray-coating. A catalyst loading of 2.5 mg cm−2 was achieved. The prepared catalyst was used as the cathode electrode with an exposed area of 1 cm2. Titanium supported iridium oxide (Magneto Special Anodes, Evoqua Water Technologies) was used as the anode electrode. The cathode and anode electrodes were separated by a Nafion membrane (N117, 183 mm, Ion Power), with an additional layer of mixed cellulose ester membrane filter (8.0 μm pore size, Millipore Sigma) added on the top of cathode. Nafion membranes were activated via the following procedure: 1 h in 90 °C 3 wt% H2O2, 1 h in 90 °C DI water, 1 h in 90 °C 5 wt% H2SO4, and 1 h in 90 °C DI water again. The activated Nafion membranes were stored in DI water.
Electrochemical measurements of CO2RR
CO2RR measurements were conducted in a flow-cell system under galvanostatic mode. In a typical flow-cell system, the catalyst-deposited gas-diffusion electrode (Freudenberg H23C3) was used as the working electrode (cathode), while an Ag/AgCl electrode served as the reference electrode and platinum mesh was used as the counter-electrode (anode). A cation-exchange membrane (Nafion 117) was used to separate the cathodic and anodic compartments. The catholyte (1M KCl or 1M post-capture K-Gly) was circulated, and 0.05M H2SO4 solution was circulated as the anolyte, while dilute CO2 (15 or 25 vol%) was supplied behind the gas-diffusion electrode. All the electrochemical measurements were conducted using an electrochemical workstation (Autolab PGSTAT204). For the electrode preparation in the flow cell, 4 mg of atomically dispersed nickel sample, 50 μL of Nafion dispersion (5 wt%), and 2 mL of ethanol were mixed and sonicated to prepare the catalyst ink. The well-mixed catalyst ink was spray-coated onto gas-diffusion electrode until 1 mg cm−2 loading was achieved. For the Ag-based electrode, the Ag ink was prepared by dispersing 50 mg of Ag and 50 mg of Nafion dispersion in 2 mL of methanol and sonicating for 1 h before spray-coating, to achieve 1 mg cm−2 loading.
CO2RR was also conducted in an H-type cell, using a sealed two-compartment cell separated by a Nafion 117 membrane. An Ag/AgCl electrode (saturated KCl solution) and a platinum mesh were employed as the reference and counter electrodes, respectively. All potentials reported in this work were converted to the reversible hydrogen electrode (RHE) scale using the equation ERHE = EAg/AgCl + 0.0591 × pH + 0.197 V, unless otherwise specified. Potential measured is non-iR corrected voltage. Linear sweep voltammetry was conducted in CO2-saturated 0.5M KHCO3 aqueous solution with varying K-Gly amount at a scan rate of 20 mV s−1. Electrochemical active surface area was determined by scanning cyclic voltammetry curves at the non-faradaic region (−0.45 to −0.65 VRHE) using varied scan rates (20–100 mV s−1). Prior to electrolysis, the catholyte was purged with CO2 at 50 mL min−1 for 30 min. CO2RR performance at different potentials was evaluated via chronoamperometry in CO2-saturated 0.5M KHCO3 solution with rapid stirring. For the electrode preparation, 250 μL of catalyst ink (4 mg nickel atomic catalyst and 50 μL Nafion dispersion in 2 mL ethanol) was deposited onto hydrophobic carbon paper, resulting in a catalyst loading of 0.5 mg cm−2. The Ag-based electrode was prepared by drop-casting 125 μL of Ag catalyst ink (12.5 mg Ag and 12.5 mg Nafion dispersion in 2 mL methanol) onto hydrophobic carbon paper, with a catalyst loading of 2.5 mg cm−2.
Gas and liquid analysis
The gas products were collected in 1 mL volume using gas-tight syringes (Hamilton chromatography syringe) and then injected into an offline gas chromatography (PerkinElmer Clarus 590) with an argon carrier gas for analysis. A thermal conductivity detector and flame ionization detector were used to analyse gas products. FE was calculated using the following equation:
where \({z}_{{{{\rm{i}}}}}\) represents the number of electrons needed to produce the product \({{{\rm{i}}}}\), \(F\) is the Faraday constant, \(P\) represents the pressure at the outlet of the system where the sample is collected (atmospheric), \(R\) is the ideal gas constant, \(T\) represents the temperature at the outlet of the system where the sample is collected (room temperature, 25 °C), \(\nu\) represents the gas flow rate of product \(i\), and \(I\) represents the total current.
The EE toward CO was calculated using the following equation:
where \({E}_{{{\mathrm{Cell}}}}^{0}\) represents the thermodynamic cell potential to produce CO (\({E}_{{{\mathrm{Cell}}}}^{0}=\) 1.33 V); and \({E}_{{{\mathrm{cell}}}}\) is the measured non-iR corrected full cell voltage.
CO2 utilization is measured by the gas composition of the cathode product gas stream, and calculated using the following equation:
Proton nuclear magnetic resonance (1H NMR) in water suppression mode was used to analyse electrochemical liquid products. NMR was performed on an Agilent DD2 600 spectrometer in D2O with trimethylsilylpropanoic acid as the internal standard.
Flue gas and direct air capture
For flue gas capture, 100 sccm of a mixed gas stream was bubbled through the capture solution. The simulated flue gas (15% CO2, 10% O2, N2 balanced) was composed of 15 sccm CO2, 48 sccm compressed air, and 37 sccm N2. For simulated flue gas containing common contaminates, the gas stream (15% CO2, 10% O2, 500 ppm SO2, 500 ppm NO, N2 balanced) contained 15 sccm CO2, 10 sccm O2, 50 sccm of a mixed gas (1000 ppm SO2, 1000 ppm NO, balanced by N2) and 25 sccm N2. For direct air capture, the capture solution was circulated in a humidifier (Envion Four Seasons), which acted as an air contactor37. DI water was added periodically at 76 mL min−1 with a timed pump to compensate for water evaporation and maintain the capture solution reservoir at 2 L. The pH of the capture solution was recorded using a pH probe placed inside the reservoir. The pH of the air capture solution was recorded until a plateau was reached.
Multiphysics simulations of carbonate/bicarbonate electrolysis
We simulate the carbonate and bicarbonate electrolysis system in 1D with COMSOL Multiphysics package. The CO32− (HCO3−) are set to be 2M (0.5M) for carbonate (bicarbonate) electrolysis system at catholyte. The H+ is set to be 0.1M for both carbonate and bicarbonate system at anolyte. The concentration of CO2(aq), CO32−, HCO3− and pH were calculated by the following homogeneous reactions.
The following equations are the electrochemical reactions within the cathode CL domain
The following equation is the electrochemical reactions within the anode Ti supported IrO2 domain
The balance of species at the steady state is given by
In Eq. 13, \({C}_{{{{\rm{i}}}}}\) is the concentration of the \(i{{\mathrm{th}}}\) species, \({J}_{{{{\rm{i}}}}}\) is the flux for the \(i{{\mathrm{th}}}\) species, \({R}_{{{{\rm{i}}}}}\) is the rate of production of the \(i{{\mathrm{th}}}\) species due to the homogeneous reaction, \({R}_{{{\mathrm{PT}}},{{{\rm{i}}}}}\) is the phase transform rates of \(i{{\mathrm{th}}}\) species (here is CO2) from aqueous to gas.
The flux of species (\({J}_{{i}}\)) is defined by
In Eq. 14, \({D}_{{{{\rm{i}}}}}\) is the diffusion constant of the \(i{{\mathrm{th}}}\) species, \({a}_{{{{\rm{i}}}}}\) is the solvation size of the \(i{{\mathrm{th}}}\) species, \({z}_{{{{\rm{i}}}}}\) is the valence charge of the \(i{{\mathrm{th}}}\) species, \(F\) is the Faradaic constant, \(R\) is the ideal gas constant, \(T\) is the temperature, \({N}_{{{{\rm{A}}}}}\) is the Avogadro constant, and \(\phi\) is the space potential in the electrolyte.
The transport of species in porous medium is corrected by adopting effective transport coefficients as below:
\({\varepsilon }_{{{{\rm{l}}}}}\) is the electrolyte volume fraction.
The diffusion coefficients of anionic species in CEM are an order of magnitude lower than in the electrolyte to simulate limited transport.
The rate of each species \({R}_{{{{\rm{i}}}}}\), can be broken into carbonate equilibria as follows:
The phase transform rates from aqueous CO2 to gaseous CO2 (\({R}_{{{\mathrm{PT}}},{{\mathrm{CO}}}2}\)) are calculated via
The values of the rate constants for reactions 16–20 are given in Supplementary Table 3.
The CO2 saturation concentration was calculated using Henry’s law as given by
Where \({K}_{{{{\rm{H}}}}}^{0}\) is Henry’s constant and is given as a function of temperature T by Eq. 22:
where T is the temperature and is assumed to be 298.15 K in this study. The saturated concentration of CO2 in an electrolyte is then given by Eq. 23:
In Eq. 24, \({C}_{{{{\rm{S}}}}}\) is the molar concentration of the electrolyte and \({K}_{{{{\rm{S}}}}}\) is the Sechenov’s constant and can be calculated by the Eq. 25:
Values of h for all species can be found in Supplementary Table 4.
Theoretical and computational methods
DFT calculations were conducted to examine glycine adsorption and the electrochemical reduction of CO2 to CO on different metal catalysts. These calculations were performed using the Vienna Ab Initio Simulation Package38,39,40 with a plane wave pseudo-potential implementation41. The exchange-correlation functional was described by the spin-polarized generalized gradient approximation of Perdew–Burker–Ernzerhof41 and the electron-ion interactions was described by projector augmented wave potentials42,43. The kinetic cut-off energy of 450 eV was used for the plane-wave expansion. The 3 × 3 × 1 and 1 × 1 × 1 Monkhorst-Pack k-point44 meshes were used for periodic (i.e., metal surfaces, Fe–N4, and Ni–N4) and nonperiodic geometries (i.e., NiPc and Co–N4), respectively.
Metal surfaces such as Ag (111) and Au (111) were modeled using a (3 × 3) supercell structure with four atomic layers, comprising 36 metal atoms. The Fe–N4 (Ni–N4) surface was created by substituting 6 carbon atoms with 4 nitrogen atoms and 1 iron atom (or 4 nitrogen atoms and 1 nickel atom) in a graphene surface that originally contained 72 carbon atoms. A 15 Å vacuum layer was included in the z-direction to prevent interactions between periodic surfaces.
The implicit solvent model, as implemented in VASPsol45,46, was utilized in all DFT calculations. This method involves placing ionic counter-charges implicitly at the interface to screen the net dipole resulting from the addition or removal of electrons. The Debye length was set to 3 Å, corresponding to a 1M electrolyte concentration, and the relative permittivity of the bulk solvent was set to that of water (78.4).
The Gibbs free energy was calculated using the computational hydrogen electrode (CHE) model, which involved adjusting the electronic energies obtained from DFT calculations with zero-point energies and entropies derived from the harmonic oscillator approximation at 298.15 K. Details on the contributions to Gibbs free energies for each gas species and adsorbate are provided in Supplementary Table 7.
Data availability
All necessary data supporting the findings of this study are available in the article and its Supplementary Information. Source data are provided with this paper.
References
Xiang, Y. & Kruse, N. Tuning the catalytic CO hydrogenation to straight-and long-chain aldehydes/alcohols and olefins/paraffins. Nat. Commun. 7, 13058 (2016).
Hirunsit, P., Senocrate, A., Gómez-Camacho, C. E. & Kiefer, F. From CO2 to sustainable aviation fuel: navigating the technology landscape. ACS Sustain. Chem. Eng. 12, 12143–12160 (2024).
Li, M., Irtem, E., Iglesias van Montfort, H.-P., Abdinejad, M. & Burdyny, T. Energy comparison of sequential and integrated CO2 capture and electrochemical conversion. Nat. Commun. 13, 5398 (2022).
Li, Y. C. et al. CO2 electroreduction from carbonate electrolyte. ACS Energy Lett. 4, 1427–1431 (2019).
Xiao, Y. C. et al. Direct carbonate electrolysis into pure syngas. EES Catal. 1, 54–61 (2023).
Smith, K. et al. Demonstration of a concentrated potassium carbonate process for CO2 capture. Energy Fuels 28, 299–306 (2014).
Lee, G. et al. Electrochemical upgrade of CO2 from amine capture solution. Nat. Energy 6, 46–53 (2021).
Kim, J. H. et al. The insensitive cation effect on a single atom Ni catalyst allows selective electrochemical conversion of captured CO2 in universal media. Energy Environ. Sci. 15, 4301–4312 (2022).
Langie, K. M. G. et al. Toward economical application of carbon capture and utilization technology with near-zero carbon emission. Nat. Commun. 13, 7482 (2022).
Wang, T., Lackner, K. S. & Wright, A. Moisture swing sorbent for carbon dioxide capture from ambient air. Environ. Sci. Technol. 45, 6670–6675 (2011).
Barzagli, F., Giorgi, C., Mani, F. & Peruzzini, M. Screening study of different amine-based solutions as sorbents for direct CO2 capture from air. ACS Sustain. Chem. Eng. 8, 14013–14021 (2020).
Ramezani, R., Mazinani, S. & Di Felice, R. State-of-the-art of CO2 capture with amino acid salt solutions. Rev. Chem. Eng. 38, 273–299 (2022).
Xiao, Y. C. et al. Reactive capture of CO2 via amino acid. Nat. Commun. 15, 7849 (2024).
Knuutila, H., Aronu, U. E., Kvamsdal, H. M. & Chikukwa, A. Post combustion CO2 capture with an amino acid salt. Energy Procedia 4, 1550–1557 (2011).
Brethomé, F. M., Williams, N. J., Seipp, C. A., Kidder, M. K. & Custelcean, R. Direct air capture of CO2 via aqueous-phase absorption and crystalline-phase release using concentrated solar power. Nat. Energy 3, 553–559 (2018).
Garrabrant, K. A. et al. Energy-efficient CO2 capture from flue gas by absorption with amino acids and crystallization with a bis-iminoguanidine. Ind. Eng. Chem. Res. 58, 10510–10515 (2019).
Jockenhövel, T. & Schneider, R. Towards commercial application of a second-generation post-combustion capture technology—Pilot plant validation of the siemens capture process and implementation of a first demonstration case. Energy Procedia 4, 1451–1458 (2011).
Reichl, A. E., Schneider, R., Ohligschläger, A., Rogalinski, T. & Hauke, S. Process development and scale-up for post combustion carbon capture-validation with pilot plant operation. Energy Procedia 63, 6379–6392 (2014).
Zhang, Z. et al. Porous metal electrodes enable efficient electrolysis of carbon capture solutions. Energy Environ. Sci. 15, 705–713 (2022).
Pakiari, A. & Jamshidi, Z. Interaction of amino acids with gold and silver clusters. J. Phys. Chem. A 111, 4391–4396 (2007).
Buglak, A. A., Ramazanov, R. R. & Kononov, A. I. Silver cluster–amino acid interactions: a quantum-chemical study. Amino Acids 51, 855–864 (2019).
Li, Y. et al. Enhancing local CO2 adsorption by L-histidine incorporation for selective formate production over the wide potential window. Angew. Chem. 135, e202313522 (2023).
Zhang, X. et al. Molecular engineering of dispersed nickel phthalocyanines on carbon nanotubes for selective CO2 reduction. Nat. Energy 5, 684–692 (2020).
Lee, G. et al. CO2 electroreduction to multicarbon products from carbonate capture liquid. Joule 7, 1277–1288 (2023).
Fei, H. et al. Single atom electrocatalysts supported on graphene or graphene-like carbons. Chem. Soc. Rev. 48, 5207–5241 (2019).
Zhang, Y., Jiao, L., Yang, W., Xie, C. & Jiang, H. L. Rational fabrication of low-coordinate single-atom Ni electrocatalysts by MOFs for highly selective CO2 reduction. Angew. Chem. Int. Ed. 60, 7607–7611 (2021).
Yan, C. et al. Coordinatively unsaturated nickel–nitrogen sites towards selective and high-rate CO2 electroreduction. Energy Environ. Sci. 11, 1204–1210 (2018).
Abel, M., Clair, S., Ourdjini, O., Mossoyan, M. & Porte, L. Single layer of polymeric Fe-phthalocyanine: an organometallic sheet on metal and thin insulating film. J. Am. Chem. Soc. 133, 1203–1205 (2011).
Nardi, E., Koudia, M., Kezilebieke, S., Bucher, J.-P. & Abel, M. On-Surface Synthesis. In Proc. International Workshop On-Surface Synthesis, École des Houches, Les Houches 25–30 May 2014 115–129 (Springer, 2016).
Han, N. et al. Supported cobalt polyphthalocyanine for high-performance electrocatalytic CO2 reduction. Chem. 3, 652–664 (2017).
Shen, M. et al. Hierarchical design enables sufficient activated CO2 for efficient electrolysis of bicarbonate to CO. Joule 8, 1999–2015 (2024).
Pimlott, D. J., Jewlal, A., Kim, Y. & Berlinguette, C. P. Oxygen-resistant CO2 reduction enabled by electrolysis of liquid feedstocks. J. Am. Chem. Soc. 145, 25933–25937 (2023).
He, D., Ma, X., Zhou, H., Zhang, Y. & Wu, Y. Continuous conversion of flue gas into syngas by a bipolar membrane-integrated single-cell cyclic system. Joule 9, 101806 (2025).
Pan, B. et al. Close to 90% single-pass conversion efficiency for CO2 electroreduction in an acid-fed membrane electrode assembly. ACS Energy Lett. 7, 4224–4231 (2022).
Kim, H. et al. Identification of single-atom Ni site active toward electrochemical CO2 conversion to CO. J. Am. Chem. Soc. 143, 925–933 (2021).
Yang, H. B. et al. Atomically dispersed Ni (i) as the active site for electrochemical CO2 reduction. Nat. energy 3, 140–147 (2018).
Stamberga, D., Einkauf, J. D., Liu, M. & Custelcean, R. Direct air capture of CO2 via reactive crystallization. Cryst. Growth Des. 24, 4556–4562 (2024).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B 47, 558 (1993).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169 (1996).
Payne, M. C., Teter, M. P., Allan, D. C., Arias, T. & Joannopoulos, A. J. Iterative minimization techniques for ab initio total-energy calculations: molecular dynamics and conjugate gradients. Rev. Mod. Phys. 64, 1045 (1992).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953 (1994).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758 (1999).
Monkhorst, H. J. & Pack, J. D. Special points for Brillouin-zone integrations. Phys. Rev. B 13, 5188 (1976).
Mathew, K., Kolluru, V., Mula, S., Steinmann, S. N. & Hennig, R. G. Implicit self-consistent electrolyte model in plane-wave density-functional theory. J. Chem. Phys. 151, 234101 (2019).
Mathew, K., Sundararaman, R., Letchworth-Weaver, K., Arias, T. & Hennig, R. G. Implicit solvation model for density-functional study of nanocrystal surfaces and reaction pathways. J Chem. Phys. 140, 084106 (2014).
Acknowledgements
The authors gratefully acknowledge support from Shell Global Solutions International B.V., the Canada Research Chairs Program, and the Natural Sciences and Engineering Research Council of Canada (NSERC) through the Alliance Program. The authors also thank Yu Yan for assisting with the XRD test, and Roham Dorakhan and Sungjin Park for the XAS measurements. Electron microscopy characterization was conducted at the Canadian Centre for Electron Microscopy at McMaster University. We acknowledge the support of the Government of Canada’s New Frontiers in Research Fund (NFRF), CANSTOREnergy project NFRFT-2022-00197.
Author information
Authors and Affiliations
Contributions
Z.M.G. conceived and designed the project, carried out all experiments and wrote the manuscript. F.L. performed COMSOL and DFT simulations and wrote the manuscript. Y.C.X. and R.K.M. designed the cell system, analysed the data, and wrote the manuscript. S.F.H., Y.R.L., K.P. and Y.L. performed and analysed the in situ and ex situ XAS measurements. A.F. and D.H. performed and analysed (S)TEM-EDS measurements. S.L. performed UV–vis test. J.L., S.S.S., Y.C., Q.W., M.L., C.W., Y.L., M.F., Z.A.D., P.P., Y.W., A.S.Z., K.H. and P.C. contributed to data analysis and manuscript polishing. D.S. and R.K.M. supervised the project. All authors discussed the results and assisted during manuscript preparation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Yun Jeong Hwang and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Guo, Z., Li, F., Xiao, Y.C. et al. Efficient amino-acid-based reactive capture of CO2 via nickel molecular catalyst. Nat Commun 16, 10373 (2025). https://doi.org/10.1038/s41467-025-65331-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-65331-9