Abstract
Thermoelectric (TE) materials directly convert temperature gradients into electrical potential. However, conventional rigid TE materials are limited by poor mechanical compliance, potential toxicity, and non-recyclability. Here, we present an ionic TE hydrogel that addresses these challenges through high stretchability, full recyclability, and non-toxic composition. The hydrogel can be recycled through an environmentally friendly process that generates no hazardous byproducts. Our material exhibits exceptional mechanical stretchability with 1400% strain capacity, 98% optical transparency, high electrical conductivity (1.9 mS cm-1), and a Seebeck coefficient (−1.05 mV/K). When encapsulated in recyclable polyurethane, the resulting devices enable stable dual-mode sensing through both TE and triboelectric mechanisms, allowing simultaneous temperature and pressure detection without complex signal processing. The devices maintain 96% of their electrical performance even after recycling and self-healing cycles. This innovative hydrogel design strategy aligns with circular economy principles for environment-friendly human-machine interface applications.
Similar content being viewed by others
Introduction
Thermal sensation is a key attribute of human skin, enabling the perception of temperature changes1,2. Consequently, integrating thermal sensors into human-machine interface (HMI) devices can significantly enhance their tactile sensing capabilities3,4. Various mechanisms5 can be employed for thermal sensing in HMI devices, including thermoelectric (TE)6, thermoresistive7, and transistor-based approaches8. Among these, TE materials offer a distinct advantage by directly converting thermal gradients into electrical signals without power consumption9,10,11,12,13,14. This active thermal sensing mechanism is more sustainable than passive sensing mechanisms, as it minimizes the need for external power sources, thereby enhancing device practicality15,16. Therefore, TE materials hold great and compelling potential for thermal sensing devices in HMI technologies.
Soft and non-hazardous electronics are preferred for HMI applications3,17 to provide compliant interfacing and safe usage. However, most TE materials are inherently rigid and/or hazardous. Traditional TE materials, such as gallium arsenide (GaAs), bismuth telluride (Bi2Te3), and antimony telluride (Sb2Te3), are rigid and toxic18. Current research on soft and flexible TE materials primarily focuses on three categories: conductive polymers and their derivatives, organic/inorganic hybrids, and inorganic thin films19,20. Conductive polymers with intrinsic flexibility, such as poly(3,4-ethylenedioxythiophene): poly(styrenesulfonate) (PEDOT:PSS)21,22, polyaniline (PANi)23, and polythiophene (PTH)24, have been extensively investigated for flexible TE applications. However, the intrinsic coloration of PEDOT:PSS, PTH, and PANi limits their optical transparency, restricting their viability in HMI devices requiring visual interfacing. Alternative conducting polymers like gelatin25, while potentially offering better optical transparency, typically exhibit inferior TE performance and require chemical doping for enhancement. This doping requirement introduces significant challenges, as most effective dopants are both hazardous and colored, potentially compromising both device safety and desired transparency.
Ionic liquids are widely used as dopants to enhance the performance and stability of TE materials, despite their inherent toxicity. For instance, the ionic liquid 1-ethyl-3-methylimidazolium ethyl sulfate ([EMIM][EtSO4]) has been incorporated into PANi to achieve higher TE power26. Various other ionic liquids, including 1-ethyl-3-methylimidazolium dicyanamide ([EMIM][DCA]), 1-ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide ([EMIM][TFSI]), and 1-butyl-3-methylimidazolium tetrafluoroborate ([BMIM][BF4]), have been extensively used to enhance the TE performance of poly(vinylidene fluoride-co-hexafluoropropylene) (PVDF-HFP)27,28. Beyond ionic liquids, redox couples in liquid electrolytes represent another widely explored class of dopants. Han et al. demonstrated substantial thermopower enhancement by incorporating the toxic redox couple ferro/ferricyanide [Fe(CN)64–/Fe(CN)63–] into non-hazardous gelatin25. Other commonly used redox couples, such as iron (II/III) (Fe2+/Fe3+) and cobalt (Co2+/Co3+), also raise toxicity concerns. The widespread reliance on these hazardous components in flexible TE materials remains a major barrier to their broader adoption in HMI applications.
The rapid proliferation of HMI devices in keyboards, touch panels, and robotics has significantly contributed to the growing electronic waste (E-waste)29,30, leading to environmental pollution and resource depletion31. This environmental challenge has driven an urgent demand for degradable and recyclable materials in HMI applications32,33. While recent research has advanced the development of degradable/recyclable materials and transient electronics34,35,36,37, the recyclability of TE materials remains a longstanding challenge. Early TE recycling methods primarily focus on recycling rare elements (e.g., bismuth) and precious metals (e.g., gold, silver, and palladium) from rigid TE devices38. These processes typically employ strong acids and harsh chemicals, which not only leave most components unrecycled but also generate hazardous residues that pose additional environmental risks. Current strategies for recyclable TE materials primarily involve encapsulating non-recyclable TE components within recyclable polymer matrices39,40,41. However, this approach only achieves partial recyclability, as the functional TE components remain non-recyclable39,40. While some flexible TE materials, such as those based on PVDF-HFP27, can be recycled, their recycling processes require hazardous solvents such as N,N-dimethyl acetamide and acetone42. These limitations underscore the pressing need for fully recyclable, eco-friendly TE materials to enable sustainable thermal sensing in next-generation HMI devices.
Here, we present a non-hazardous, fully recyclable, and optically transparent ionic TE hydrogel, termed ReCLEAR, as a sustainable solution for wearable HMI devices. The hydrogel consists of a highly stretchable polyacrylic acid (PAA)43 polymer network integrated with a non-hazardous, thermo-regulable salt, zinc trifluoromethanesulfonate (Zn(OTf)2)44, achieving excellent 98% optical transmittance and a thermodiffusion-based45 Seebeck coefficient of −1.05 mV/K. The material exhibits exceptional self-healing capability, anti-freezing properties, and mechanical durability (up to 1400% strain). Crucially, ReCLEAR achieves 100% degradability through an eco-friendly process using hydrogen peroxide (H2O2), generating no hazardous byproducts. When encapsulated with waterborne polyurethane (PU) films, the resulting devices enable dual-mode sensing via both TE and triboelectric (TENG) mechanisms, allowing simultaneous temperature and pressure detection without the need for complex signal decoupling. The devices maintain 96% of their electrical performance after recycling and self-healing cycles, demonstrating their robustness and sustainability. This innovative TE material design represents a significant advancement in sustainable HMI technologies, particularly for applications in tactile interfaces and robotic skins.
Results
Design of the ReCLEAR hydrogel
The molecular structure of the non-hazardous and fully recyclable ionic TE hydrogel we designed for sustainable wearable HMI applications is illustrated in Fig. 1a. The polymer network of the ReCLEAR hydrogel is constructed by a cross-linked PAA matrix43 grafted with glycerol (denoted as PAA-Gly)46. Zinc cations (Zn2+) and triflate anions (OTf-) serve as the main freely mobile thermal charge carriers in the system to introduce the TE effect. The distinct mobilities of Zn2+ and OTf- within the hydrogel matrix generate potential differences in response to temperature gradients, a phenomenon known as the Soret effect47. In contrast to conventional colored and inherently toxic ionic TE pairs, such as Fe(CN)64–/Fe(CN)63–, Fe2+/Fe3+, and Co2+/Co3+ 25, the selected Zn(OTf)2 preserves the exceptional optical transparency and non-toxicity of the hydrogel system.
a Molecular structure of the thermoelectric hydrogel. H-bond: hydrogen bond. b Recycling process of the ReCLEAR hydrogel visualized using a self-healed transparent hydrogel containing a green-stained triangular segment. c Radar plot comparing key properties between the ReCLEAR hydrogel and contemporary thermoelectric materials. d Virtual typing interface enabled by ReCLEAR devices. e ReCLEAR-integrated robotic skin with dual-mode sensing for temperature and tactile inputs.
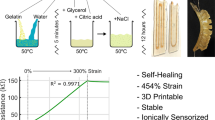
Figure 1b demonstrates the complete and environmentally sustainable recycling process of the ReCLEAR hydrogel, visualized using a dye-stained triangular section self-healed with pristine transparent hydrogel. The hydrogel achieves 100% degradation within 2–3 days through combined oxidative (3 wt% H2O2) and thermal (50-70 °C) conditions via free-radical-induced chain scission. H2O2 generates hydroxyl radicals (·OH), which attack the carbon–carbon backbone of the hydrogel, cleaving it into water-soluble low-molecular-weight oligomers (Supplementary Note 1). During degradation, the hydrogel becomes transparent as the dye undergoes oxidative decolorization. The degraded solution is thermally concentrated, followed by the reintroduction of acrylic acid monomers, crosslinker, and initiator. Thermal free-radical polymerization subsequently regenerates the hydrogel. The recycling mechanisms and performance are detailed in Supplementary Note 2.
Comparative analysis of current TE materials (Fig. 1c, Supplementary Table 3) highlights the distinct advantages of ReCLEAR, including full recyclability, environmental compatibility, optical transparency, self-healing capability, and mechanical stretchability. While its Seebeck coefficient is moderate compared to certain TE materials, this trade-off ensures environmental sustainability and optical transparency, achieved by selecting Zn(OTf)2 as a dopant (Supplementary Fig. 4). The synergistic multi-functionality of ReCLEAR facilitates its integration into wearable, sustainable HMI devices. Functional prototypes, such as virtual keyboards and robotic skins, enable simultaneous temperature and pressure sensing for realistic tactile interactions (Fig. 1d-e). These environmentally sustainable properties further establish ReCLEAR as a practical platform for next-generation wearable electronics.
Characterization of the ReCLEAR hydrogel
The ReCLEAR hydrogel matrix is based on cross-linked glycerol-grafted PAA chains. The addition of glycerol introduces abundant hydroxyl groups, which attract water molecules and induce extensive intra- or intermolecular hydrogen bonding both within carboxylic groups and between carboxylic and hydroxyl groups. This enhanced hydrogen bonding enables reliable anti-freezing properties, efficient self-healing, and exceptional durability.
As illustrated in Fig. 2a, glycerol-enhanced hydrogen bonding density within the hydrogel matrix simultaneously improves both low-temperature stability and self-healing efficiency. The left panel of Fig. 2b demonstrates the anti-freezing performance of the hydrogel at −20 °C after 1 week. ReCLEAR maintains mechanical flexibility, whereas the glycerol-free control sample exhibits immediate ice crystal formation and loss of mechanical compliance. The right panel of Fig. 2b provides optical validation of ReCLEAR’s self-healing efficiency through comparison of healed and cut samples.
a Mechanism underlying anti-freezing and self-healing behavior. H-bond/HB: hydrogen bond. b Demonstration of anti-freezing performance at −20 °C (left) and self-healing at room temperature (right). c Stress-strain curves of self-healed ReCLEAR hydrogels after different healing durations. SH: self-healed. d Optical transparency of ReCLEAR device components, with inset showing the ReCLEAR hydrogel placed over an NUS logo. e Fourier-transform infrared (FTIR) spectra of recycled and pristine ReCLEAR hydrogel showing identical characteristic peaks. f Schematic of the recycling process. g Weight changes and weight retention of the ReCLEAR hydrogel during the recycling process.
This PAA matrix offers high mechanical strength and elasticity to the hydrogel (Supplementary Fig. 5). Optimization of the ReCLEAR hydrogel focused on its mechanical properties, with Young’s modulus, stretchability, and fracture toughness evaluated across different glycerol concentrations using tensile testing. Increasing glycerol content reduces the Young’s modulus (Supplementary Fig. 7) and loss factor (Supplementary Fig. 8), as observed by dynamic mechanical analysis (DMA), because glycerol can soften polymer chains, enhancing segment fluidity and viscoelasticity. These findings suggest that glycerol both strengthens hydrogen bonds, and acts as a mechanical plasticizer, promoting chain mobility.
Fracture toughness, derived from stress-strain curves, revealed improved healing efficiency (from 20% to 60%) as glycerol content increased from 10 to 30 wt%. However, at 40 wt% glycerol, efficiency decreased to 29%, due to reduced modulus and compromised mechanical strength, which prevented free-standing hydrogel formation (Supplementary Fig. 9a). Thus, 30 wt% glycerol was identified as the optimal composition with balanced mechanical properties and healing performance (Supplementary Table 4).
With the optimized glycerol content of 30 wt%, we characterized the self-healing performance of the ReCLEAR hydrogel over various healing periods (Fig. 2c). The pristine hydrogel exhibited a strain of 1400%, which recovered to 550% with a toughness of 22.33 MJ m−3 (30.3% of the original) after one day. By Day 6, the self-healed hydrogel reached 1200% strain (85.7% of the original) and 43.64 MJ m-3 toughness (59.3% of the original). After 12 days, healing efficiency approached 90%, underscoring the excellent self-healing performance of the hydrogel (Supplementary Fig. 9b).
3D surface maps and height profiles of the hydrogel at different self-healing stages within 24 h are shown in Supplementary Fig. 10, demonstrating rapid self-healing. To evaluate the durability of the self-healing capability, repeated self-healing experiments were conducted (Supplementary Fig. 11). The performance remained stable during the first two cycles but decreased by ~20%, 30%, and 50% in the third, fourth, and fifth cycles, respectively. This decline is likely due to cumulative surface contamination, minor material loss, and incomplete chain diffusion during repeated healing. Overall, the hydrogel maintains efficient self-healing in the initial cycles but shows reduced efficiency with multiple repetitions.
The incorporation of glycerol also substantially improved durability. The ReCLEAR hydrogel maintained stable electrical performance over 6 weeks under ambient and cryogenic conditions (Supplementary Fig. 12), whereas glycerol-free hydrogels degraded within 6 days. This enhancement in temperature range and long-term stability highlights the ReCLEAR hydrogel’s potential for practical applications.
The colorless and environmentally friendly TE salt, Zn(OTf)2, we selected preserves both the high transparency and sustainability of the hydrogel while imparting TE functionality. According to the UV-visible spectra (Fig. 2d), the optical transmittance of the ReCLEAR hydrogel maintained >98% between 500 to 800 nm. Even after encapsulation with PU films, the optical transmittance of the ReCLEAR device decreased only slightly and still remained above 90%, demonstrating its excellent optical transparency. An optimal Zn(OTf)2 concentration of 1 mol/L was identified, providing high ionic conductivity while minimizing salt content (Supplementary Tables 5 and 6).
Fourier-transform infrared (FTIR) spectroscopy was utilized to investigate the molecular interactions within the ReCLEAR hydrogel (Fig. 2e). The nearly identical characteristic peaks observed in the FTIR spectra for pristine and recycled hydrogels confirm that the recycling process does not significantly alter the molecular structure. Supplementary Fig. 13 displays FTIR spectra of the hydrogel with different glycerol and Zn(OTf)2 concentrations. At 1704 cm-1, the addition of Zn(OTf)2 weakens the C = O stretching vibrations of PAA and glycerol. OTf- contribute S = O stretching at 1245 cm-1, and C-F stretching of the -CF3 groups at 1174 cm-1. The strong peak at 1029 cm-1 is associated with Zn2+, indicating that Zn(OTf)2 slightly retards carboxyl group vibrations, influencing the self-healing performance of the ReCLEAR hydrogel.
To promote sustainable usage, we developed an environmentally friendly recycling method for the ReCLEAR hydrogel as mentioned in Fig. 1b. The full recycling process is detailed in Supplementary Fig. 14 and Supplementary Movie 1. In this recycling process, the hydrogel undergoes degradation followed by reformation (Fig. 2f). During degradation, the PAA-based hydrogel undergoes a combination of oxidative and thermal breakdown, with carboxyl groups on the PAA chains degrading into smaller oligomers48,49,50. Notably, both ReCLEAR hydrogels, with or without Zn2+, degrade effectively in low-concentration (3 wt%) H2O2 solution without requiring catalysts such as iron or copper ions. The hydrogel mass change during a single recycling cycle is shown in Fig. 2g.
The ReCLEAR hydrogel demonstrates consistent TE performance over five recycling cycles, with only minor performance loss attributed to slight material loss during transfer (Supplementary Fig. 3). However, after five cycles, the hydrogel forms more bubbles and becomes softer, which may begin to affect its mechanical stability and TE output in extended recycling. This eco-friendly approach minimizes hazardous byproduct generation and reduces the risk of secondary pollution.
TE and TENG performance of the ReCLEAR device
Moreover, the ReCLEAR hydrogel is encapsulated between two layers of PU films to form a freestanding ReCLEAR HMI device (Fig. 3a). PU, known for its stretchability and self-healability51, exhibits high optical transparency (Fig. 2d), and can be stretched to up to 1000% and recover to 200% strain and 11.2% toughness after 3 days of healing (Supplementary Fig. 15). Additionally, PU films act as effective insulating layers for the conductive ionic hydrogel (Supplementary Fig. 16). Notably, PU is recyclable52 using non-hazardous deionized (DI) water, with its recycling process demonstrated at both small and large scales in Supplementary Figs. 17 and 18. Consequently, this fully recyclable and non-hazardous design ensures high transparency, stretchability, and self-healing capability, making the ReCLEAR device a promising candidate for sustainable HMI applications.
a Schematic of the device structure. b Thermoelectric (TE) sensing mechanism of ReCLEAR devices. c Triboelectric (TENG) sensing mechanism of ReCLEAR devices. d Real-time TE voltage outputs of pristine, self-healed, and recycled ReCLEAR hydrogels during temperature cycling (10 to 60 °C) using Peltier heating, creating –8 °C to +5 °C temperature difference across electrodes. e Extracted thermal voltages from the curves in (d). f TENG responses of pristine, self-healed, and recycled ReCLEAR hydrogels under 10 N applied force. g TE-dominated long-press sensing mechanism (H: hold; R: release). h TENG-dominated short-press sensing mechanism (T: tap; R: release). Input intensity: long press: 3–5 N at 27–28 °C; short press: 1–2 N at 24–25 °C.
Interestingly, the PU films bring TENG properties to the ReCLEAR device, enabling sensitive tactile motion detection. The device achieves simultaneous multimodal sensing of temperature (via the TE effect) and tactile stimuli (via the TENG effect) without requiring complex signal decoupling. By mimicking the functions of thermoreceptors and mechanoreceptors in biological skin, the ReCLEAR device transduces temperature and tactile stimuli into electrical signals for efficient processing. These self-powered properties enhance adaptability and improve interaction experiences in HMI applications.
As shown in Fig. 3b, a temperature gradient within the hydrogel affects the molecular kinetics of dissociated TE carriers, leading to unbalanced charge accumulation and a resulting potential difference. The underlying mechanism is discussed in Supplementary Note 3. The ionic Seebeck coefficient6,53, which quantifies TE voltage responses to temperature changes, is defined as \({S}_{i}=-\frac{{V}_{h}-{V}_{c}}{{T}_{h}-{T}_{c}}\,(1)\), where \({V}_{h}\) and \({V}_{c}\) are the voltages at the hot and cold electrodes, and \({T}_{h}\) and \({T}_{c}\) are the temperatures of the hot zone and cold zone, respectively. Additionally, contact-separation motions between the fingertip and PU encapsulation, produce a TENG spike. The TENG mechanism is shown in Fig. 3c. Together, these two mechanisms enable simultaneous detection of temperature variations (via TE effect) and tactile inputs (via TENG effect).
Thermovoltages of the ReCLEAR hydrogel were characterized using a customized Peltier station (Supplementary Fig. 19). Figure 3d shows the real-time TE response of pristine, self-healed, and recycled ReCLEAR hydrogels under cyclic temperature changes between 10 °C and 60 °C, producing a temperature difference of approximately –8 °C to +5 °C across the electrodes using a Peltier station. To evaluate response speed, TE measurements were performed under thermal inputs at varying ramp rates (Supplementary Fig. 20), where the hydrogel exhibited fast and sensitive responses. Additionally, clear and measurable TE outputs were also obtained under minor temperature gradients of 1–3 K (Supplementary Fig. 21), confirming the high thermal sensitivity and suitability of the hydrogel for dynamic and wearable applications. To further enhance the response speed, reducing the hydrogel thickness and minimizing the electrode spacing can help by shortening the thermal diffusion path and ionic migration distance. Supplementary Fig. 22 presents thermovoltage outputs and calculated Seebeck coefficients for all three forms. The hydrogels exhibited nearly identical Seebeck coefficients: pristine (-1.048 ± 0.008 mV K-1), self-healed (-1.005 ± 0.009 mV k-1), and recycled (-1.005 ± 0.009 mV k-1) at 70% relative humidity (RH). The closely aligned linear fitting curves in Fig. 3e indicate that self-healing and recycling processes have negligible impact on the TE performance of the ReCLEAR hydrogel.
The TE energy conversion capacity of the ReCLEAR hydrogel is detailed in Supplementary Note 4. Based on data from Supplementary Figs. 23-25 and Supplementary Tables 6-7, our ReCLEAR hydrogel exhibits an ionic conductivity of 1.9 mS cm−1, and a thermal conductivity of 0.48 W/m·K. The power factor (PFi) of ionic TE materials can be expressed as, \({{PF}}_{i}={{S}_{i}}^{2}\sigma \,(2)\), where \({S}_{i}\) is the ionic Seebeck coefficient and \(\sigma\) is the electrical conductivity. For the ReCLEAR hydrogel, PFi is calculated as 0.2 μWm-1K-2.
Meanwhile, the PU encapsulation induces TENG performance54 for detecting tactile stimuli (Fig. 3f). The PU layer alone can generate TENG voltages of ~200 mV and 4–6 mV when combined with the hydrogel. The reduction in TENG output is mainly due to the electron transfer from the conductive hydrogel surface and the decreased induction capacitance caused by ionic movements. TENG performance of the ReCLEAR device at 10 °C, 23 °C, and 40 °C under varying loading forces is presented in Supplementary Fig. 26. Importantly, self-healed and recycled PU layers also displayed comparable voltage outputs to the pristine ones (Fig. 3f), indicating that the entire ReCLEAR device can be mechanically and electrically recovered after self-healing or recycling processes.
Given the good stretchability of ReCLEAR devices, we investigated the TE and TENG performances under varying stretch conditions. The TE responses of the ReCLEAR hydrogel under different strains are shown in Supplementary Fig. 27a. Only slight fluctuations were observed in the corresponding Seebeck coefficients across a large strain range (Supplementary Fig. 27b), indicating that strains have minimal impact on TE performance. Similarly, TENG signals of the PU layer under different strains showed negligible deviations, even up to 900% strain (Supplementary Fig. 28). These results confirm that both TE and TENG performances of the entire ReCLEAR device remain highly stable under extreme strains.
Mechanisms of four signal configurations
The distinctive sensing mechanisms of TE and TENG effects enable effective decoupling of electrical outputs from thermal and tactile stimuli, owing to their differing response times and signal profiles. TENG signals respond rapidly to contact-separation motions, producing sharp millisecond-scale spikes. In contrast, TE signals, governed by ionic thermal diffusion, respond over longer durations ranging from seconds to minutes, depending on the temperature gradient and encapsulation thickness. This dual-response capability simplifies signal processing by allowing simultaneous recognition of temperature and tactile inputs in a single output. The ReCLEAR device leverages this self-decoupling feature, eliminating the need for multiplexed electrodes or complex decoupling calculations, thereby streamlining multimodal sensing in flexible bifunctional devices.
The ReCLEAR device features two electrodes connected to the positive and ground terminals of a multimeter. Under identical tactile stimuli, the two electrodes exhibit opposite electrical responses. By combining TE and TENG effects, we identified four distinct signal configurations corresponding to holding on the right/left electrode (> 0.5 s, TE-dominated), and tapping on the right/left electrode (< 0.5 s, TENG-dominated).
For long press responses (> 0.5 s), dominated by the TE effect, Fig. 3g illustrates the mechanism through four stages. In stage 1, when a heat source approaches the positive electrode, the hydrogel temperature rises. OTf-, a highly diffusive and weakly coordinating organic anion, migrates toward the cold end via thermodiffusion55,56. Zn2+, being strongly coordinating and slower to diffuse, lags behind, generating a positive voltage. Upon release (stage 2), the anions diffuse back, restoring the baseline signal. Conversely, when the heat source approaches the ground electrode (stage 3), anions gather at the other electrode, producing a negative voltage, which also returns to baseline after release (stage 4). Opposite responses occur with cold sources, as they drive ion migration in reverse directions (Supplementary Fig. 29). ReCLEAR devices also exhibit reversed responses when electrode connections are swapped.
For short presses (< 0.5 s), dominated by the TENG effect, two opposite sharp voltage spikes are generated when pressing the ReCLEAR device’s electrodes (Fig. 3h). Based on the TENG series, PU has low electronegativity, inducing positive charges on its surface and inducing negative charges on the contacting objects during rapid tapping. The response process proceeds in four stages: At stage (i), when an object touches the positive electrode, positive surface charges on the PU cause anions to migrate away, producing a sharp negative spike. Upon release (ii), capacitance decreases as the object moves away, allowing anions to return, gradually restoring the signal. Similarly, (iii) tapping the ground electrode causes anions to migrate away, generating a positive spike, and release (iv) restores the signal. This mechanism generates opposite-polarity spikes on the two electrodes during tapping.
The input distributions of finger temperature and pressing force during short and long presses are shown in Supplementary Fig. 30. The differing time scales of long presses (hold) and short presses (tap) enable the ReCLEAR device to generate four unique electrical signals within a single system. This capability offers significant advantages for designing multifunctional control panels using a single device.
Virtual typing controller for patients with limited mobility
Paralyzed patients are facing numerous inconveniences due to limited finger movements and slow reaction times with non-dexterous fingers when operating touch-based HMI devices, such as smartphones or keyboards. The ReCLEAR device offers four configurations of active signals, which can be encoded into designated actions to control electronic devices using merely a single finger with ultra-short movements. Here, we demonstrated a virtual keyboard assembled by a single ReCLEAR device. This keyboard requires only minor movements between two electrodes, less than 1 cm, with two achievable operations, tapping and holding. The right electrode is connected to the positive terminal of an analog-to-digital converter (ADC), with the left electrode grounded. An Arduino-based microcontroller was further developed to process the sampled signals from the ADC for recognition and to achieve virtual typing (Fig. 4a). This innovative approach leverages the unique abilities of the ReCLEAR device to deliver a user-friendly and accessible virtual keyboard solution for individuals with limited finger mobility and slow reactions.
a Workflow of the virtual typing system, from mechanical operation and signal transduction to algorithmic processing and functional demonstration. b Signal-function correlation matrix quantifying the relationship between four distinct finger configurations and their corresponding typing operations. c Time-resolved relative response curves and corresponding triggered outputs for four signal configurations. Finger input intensity: long press, 3–5 N at 27–28 °C; short press, 1–2 N at 24–25 °C. d Real-time response signals and corresponding triggered outputs during typing of the word NUS with error correction demonstration.
Using the ReCLEAR device, four functions, including auto-change columns, auto-change rows, enter, and delete, are designed for convenient alphabet selection (Fig. 4b). Configured signals trigger distinct spikes for each action. An infinite impulse response (IIR) filter captures the signal baseline, while dynamic thresholds distinguish between short presses and long presses. A short press on the right electrode generates a negative red spike for auto-changing columns, while a left short press triggers a negative green spike for auto-changing rows. Similarly, a left long press encodes the enter function (positive red spike), and a right long press encodes delete (positive green spike) (Fig. 4c). Because the measured voltage depends on the input impedance of the acquisition system57, the ADC we used has an input impedance of ~10 MΩ, which is not directly comparable to that from a high-impedance meter such as the Keithley DMM6500. For consistent visualization, we therefore present the detected voltages in arbitrary units.
During typing, navigate across columns and rows to select the desired letters, confirming inputs with the enter action or correcting errors using the delete action (Supplementary Movie 2). This intuitive and accessible interface facilitates an inclusive virtual typing experience, particularly suited for users with limited mobility or slower reaction speeds.
The time interval between columns and rows switching can be customized based on the response speed of individual users. Currently, the time interval for changing is set to 0.5 s in Supplementary Movie 2. Even though the overall operation time increased, it provided sufficient time for sluggish or paralyzed patients to react. Figure 4d shows the real-time signals when typing the word NUS, and correcting typographical errors, demonstrating the robust functionality of the virtual keyboard based on our proposed ReCLEAR HMI device. This system offers a practical and user-centric solution for patients or elderly individuals with reduced mobility, empowering them to interact with electronic devices more effectively and independently.
Robotic skins with proximity and tactile sensing
To enhance robotic sensory systems, the ReCLEAR devices with skin-like, highly compacted multilayer structures were utilized as transparent, self-healable, and recyclable robotic skins. Their thermal sensing capability facilitates proximity sensing58 for robots, preventing damage from extreme temperatures and enabling smart operations59. For instance, when a robotic hand approaches a heated object, the ReCLEAR device detects thermovoltage changes in real time, allowing the system to determine the object’s temperature before contact (Fig. 5a). This pre-contact temperature detection enhances safety, as users may be injured by high temperatures, while cool and cold objects are generally safe to handle. The current proximity distance between the hand and the objects is ~1 cm.
a Schematic of proximity-based thermal detection and algorithmic decision framework. b−d Real-time thermovoltage responses of pristine, self-healed, and recycled ReCLEAR devices during proximity detection with thermal stimuli at (b) 60 °C, (c) 25 °C, and (d) 10 °C. Critical proximity sensing regions are highlighted by red dashed boundaries. e Optical transparency demonstration showing light transmission through ReCLEAR device. f Robotic skins implementation using pristine, self-healed, and recycled ReCLEAR devices. g Sequential degradation of ReCLEAR devices by hydrogen peroxide.
In the case of grasping hot objects, the voltage signals from the ReCLEAR skin increase rapidly as the robotic hand approaches. Consequently, the robotic hand decides to withdraw from the hot object after receiving the warning from the ReCLEAR skin. Then, the thermovoltage signals decrease as the robotic hand retreats. The withdrawal process occurs at ~60 s, with the slight delay attributed to the programmed response speed of the robotic hand, which can be adjusted for faster operation. Remarkably, the self-healed and recycled ReCLEAR devices exhibited similar performance to the pristine devices (Fig. 5b).
For cool (room-temperature) objects, only minor signal changes appear as the robotic hand approaches, allowing it to grasp the object without hesitation. Rapid signal changes are induced by tactile contact and release (Fig. 5c). In cold conditions, there is a rapid voltage drop as the robotic hand approaches the object (Fig. 5d). The real-time responses of the robotic skins in these three conditions are recorded in Supplementary Movie 3. Notably, the self-healed and recycled ReCLEAR devices performed similarly in the cool and cold conditions, validating that the sustainable ReCLEAR devices maintain stable electrical performance even after self-healing and recycling processes.
Furthermore, the high optical transparency of the ReCLEAR device allows it to be applied to various surfaces of robots without causing any visual blockage, such as signal lights (Fig. 5e). The robotic hand assembled with pristine, self-healed, and recycled ReCLEAR devices is shown in Fig. 5f. Importantly, an eco-friendly solvent, H2O2, is utilized to degrade the entire ReCLEAR device at 70 oC. The complete degradation process is shown in Fig. 5g. The transparent ReCLEAR hydrogel initially extends and then gradually degrades over time, fully degrading within 12 h. The white compounds observed are the oxidized PU residuals, which require a longer time for degradation compared to the hydrogel. The ability to fully degrade using a non-hazardous solvent further underscores the potential for environmentally friendly applications and end-of-life management. To enable complete device recycling, the ReCLEAR hydrogel and PU layers can be separated by immersion in DI water, since the hydrogen bonding at the PAA surface weakens in the water environment, reducing viscosity and allowing the layers to detach.
Discussion
We have developed a fully recyclable, non-hazardous ionic TE hydrogel that marks a significant advance in sustainable HMI devices and robotic technologies. Our ReCLEAR devices uniquely combine transparency, non-toxicity, stretchability, self-healing ability, and complete recyclability. The devices achieve simultaneous temperature and tactile sensing through integrated TE and TENG effects, enabling multimodal detection without complex signal decoupling while maintaining consistent electrical performance through recycling and self-healing cycles.
The practical applications of this work are compelling. A virtual keyboard designed for individuals with limited mobility demonstrates its potential in assistive technologies, while its integration as a robotic skin with non-contact thermal sensing enhances robotic safety and intelligence. We believe that the ReCLEAR hydrogel establishes a promising platform for next-generation wearable devices across sustainable electronics, assistive technologies, and smart robotics. Future developments in hydrogel optimization, advanced signal processing with machine learning, and expanded sensing capabilities will further advance the impact of these devices in HMI and robotic applications.
Methods
Materials and chemicals
Acrylic acid (AA), anhydrous glycerol, zinc trifluoromethanesulfonate (Zn(OTf)2), hydrogen peroxide (3 wt%, H2O2), N,N’-methylenebisacrylamide (MBAA), and ammonium persulfate (APS) were purchased from Sigma-Aldrich. Water-dispersible polyurethane dispersion U3251 (PU) was obtained from Alberdingk®. Copper films were obtained from 3M. All materials and chemicals were used as received without further purification.
Synthesis of the ReCLEAR hydrogel
4.0 g of AA monomer, 6.0 g of solvent with certain ratios of deionized (DI) water to glycerol, and 1 mol/L Zn(OTf)2 (Supplementary Tables 4 and 5) were mixed by magnetic stirring. After that, 2.0 mg MBAA and 30.0 mg APS were added to the mixed solution as cross-linkers and initiators, respectively, and stirred until fully dissolved. The fully mixed solution was poured into a glass petri dish and heated in an oven at 70 °C for 1 h for radical polymerization. After polymerization, the as-synthesized hydrogel was cooled down to room temperature and stored in a 5 °C freezer for usage.
Fabrication of ReCLEAR devices
20.0 g of the water-dispersed PU dispersion was poured into a petri dish with a diameter of 10 cm and evaporated in an oven at 70 °C for a PU film. PDMS was cast in the petri dish first to facilitate the separation of the PU film. Copper films with a width of 5 mm were utilized as electrodes. Two copper electrodes were placed on top of the hydrogel layer, and PU films were used to encapsulate both sides of the hydrogel and electrodes as an integrated device with a size of 20 mm × 5 mm.
Materials characterization
The ReCLEAR samples were cut equivalently into dumbbell shapes following ASTM D1708. Self-healing experiments were performed at room temperature after bifurcating the samples and contacting the cut regions for healing. An Instron Tensile Testing System (INSTRON, 68SC-1) was utilized to characterize the mechanical properties of the hydrogel and PU film, testing at a rate of 50 mm/min, including stretchability and fracture toughness. The viscoelasticity of the hydrogel was investigated by dynamic mechanical analysis (DMA, TA Instruments, ElectroForce 3200 series) under compression mode. The optical transparency of the materials was analyzed by a UV-visible spectrometer (Shimadzu UV-1800). Additionally, an ATR-FTIR (Vertex 70 spectrometer) was employed to study the chemical bonding of the hydrogel from 400 to 4000 cm-1. The samples were imaged using an optical microscope (KEYENCE, digital microscope) or an SEM (Zeiss Sigma 300 with SmartEDS system). The molecular weight of ReCLEAR hydrogels was characterized using gel permeation chromatography (Agilent, PL-GPC 220). The surface topography and height profiles of self-healing hydrogel samples were analyzed by a 3D laser scanning confocal microscope (KEYENCE, VK-X1000K).
Thermoelectric performance
A testing platform was built for TE tests. A Peltier station (Ferrotec, FTA951) was used to change the surrounding temperature. Half of the ReCLEAR hydrogel was placed on the Peltier station, and the other half was placed on a room-temperature platform with the same height. Two thermocouples were utilized to collect the real temperature near the electrodes of the ReCLEAR hydrogel. The TE voltages were collected by the Keithley multimeter (DMM6500). A Palmsens4 Multitrace electrochemical workstation was used to characterize electrical conductivity. AC frequency was swept between 1 Hz and 1 MHz. A thermal conductivity tester (HNB-DR32, Xiamen Senbei Technology Co., Ltd.) was used for thermal conductivity measurements.
Hydrogel stability tests
ReCLEAR hydrogel samples were stored separately in three distinct environments: an ambient environment without additional encapsulation (20 °C, 70% RH), a dry environment (20 °C, 20% RH), and a freezer set to -5 °C. The TE outputs were measured over multiple days to assess their stability performance.
Recycling progress and degradation progress
For the recycled test, the ReCLEAR hydrogel was immersed in a 3 wt% H2O2 solution and heated in an oven at 50–70 °C. Higher temperatures facilitated a faster degradation rate of the hydrogel. After complete degradation, the solvent was evaporated at 50 °C to ~ 20 g, transferred to a smaller petri dish, and baked at 70-90 °C into a dry film corresponding to 60%-70% of the initial hydrogel weight. The film was rehydrated with 6 g DI water and heated at 70 °C for 30 min to yield ~ 15 g of low-viscosity solution. Subsequently, 1-2 g AA monomer and 2-3 g DI water were added and mixed by magnetic stirring, with the AA and water amounts used to tune the mechanical strength of the recycled hydrogel. For hydrogel without Zn(OTf)2, 30 mg APS and 2 mg MBAA were added, while for hydrogel with Zn(OTf)2, 200 mg APS and 10 mg MBAA were added to the mixture for repolymerization at 70 °C for 60 min. The higher APS content compensates for the coordination effect of zinc cations. The hydrogel mass during recycling was measured by weighing the residual free-standing gel transferred to a pre-weighed container. The PU films were recycled by dissolving them in DI water and evaporating the resulting solution to recycle the films. For the whole ReCLEAR device, it can be fully degraded by immersing it in a 3 wt% H2O2 solution and heating it in an oven at 50 °C for several days. Alternatively, the device can be immersed in DI water for 30 min, allowing the separation of multiple layers, and then recycled individually.
Virtual typing
The ReCLEAR device was connected to a 24-bit ADC converter (Texas Instruments ADS1220) with an input impedance of ~10 MΩ and a programmable gain amplifier (PGA) configured at 16×, which was interfaced and configured via an Arduino Nano 33 IoT. The sensor voltage was calculated using the standard ADC transfer function with a 2.048 V reference, according to the equation: \(V=\,\frac{{ADC\; Code}}{{2}^{23}-1}*\left(\frac{{V}_{R{EF}}}{{PG}{A}_{{Gain}}}\right)(3)\). No additional amplification or filtering was applied. The TENG signal was sampled and accompanied by a 3.3 V digital trigger output for timing correlation. In addition, the Arduino was emulated as a USB keyboard, sending dedicated keystrokes corresponding to the acquired signal pattern.
Robotic skins
The pristine, self-healed, and recycled ReCLEAR devices were fixed on a robotic hand (Arm: UNIVERSAL ROBOTS, UR 10; Hand: Shadow Robot, Shadow Dexterous Hand) as robotic skins. Adhesive transfer tape (3M, 9460PC) was used to attach the devices to the robotic hand. A thermal camera (Fluke, Ti480) was utilized to record the heat flows during the robotic operations. A Keithley multimeter (DMM6500) was used to monitor the real-time thermovoltage changes.
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its supplementary information files. All other additional data are available from the corresponding author upon request.
References
Yoo, S. et al. Responsive materials and mechanisms as thermal safety systems for skin-interfaced electronic devices. Nat. Commun. 14, 1024 (2023).
Chen, L., Chang, X., Wang, H., Chen, J. & Zhu, Y. Stretchable and transparent multimodal electronic-skin sensors in detecting strain, temperature, and humidity. Nano Energy 96, 107077 (2022).
Yin, R., Wang, D., Zhao, S., Lou, Z. & Shen, G. Wearable sensors-enabled human–machine interaction systems: from design to application. Adv. Funct. Mater. 31, 2008936 (2021).
Han, Y. et al. Ultrasensitive flexible thermal sensor arrays based on high-thermopower ionic thermoelectric hydrogel. Adv. Sci. 10, e2302685 (2023).
Cai, J., Du, M. & Li, Z. Flexible temperature sensors constructed with fiber materials. Adv. Mater. Technol. 7, 2101182 (2022).
Massetti, M. et al. Unconventional thermoelectric materials for energy harvesting and sensing applications. Chem. Rev. 121, 12465–12547 (2021).
Dinh, T. et al. Thermoresistive effect for advanced thermal sensors: fundamentals, design considerations, and applications. J. Microelectromech. Syst. 26, 966–986 (2017).
Reverter, F. A tutorial on thermal sensors in the 200th anniversary of the seebeck effect. IEEE Sens. J. 21, 22122–22132 (2021).
Yin, Y. et al. A flexible dual parameter sensor with hierarchical porous structure for fully decoupled pressure–temperature sensing. Chem. Eng. J. 430, 133158 (2022).
Li, M. et al. Large-area, wearable, self-powered pressure–temperature sensor based on 3D thermoelectric spacer fabric. ACS Sens 5, 2545–2554 (2020).
He, X. et al. Stretchable thermoelectric-based self-powered dual-parameter sensors with decoupled temperature and strain sensing. ACS Appl. Mater. Interfaces 13, 60498–60507 (2021).
Yang, Q. et al. Flexible thermoelectrics based on ductile semiconductors. Science 377, 854–858 (2022).
Hong, S. et al. Wearable thermoelectrics for personalized thermoregulation. Sci. Adv. 5, eaaw0536 (2019).
Zhang, F., Zang, Y., Huang, D., Di, C. & Zhu, D. Flexible and self-powered temperature–pressure dual-parameter sensors using microstructure-frame-supported organic thermoelectric materials. Nat. Commun. 6, 8356 (2015).
Dong, B. et al. Technology evolution from self-powered sensors to AIoT enabled smart homes. Nano Energy 79, 105414 (2021).
Sun, Z., Zhu, M., Shan, X. & Lee, C. Augmented tactile-perception and haptic-feedback rings as human-machine interfaces aiming for immersive interactions. Nat. Commun. 13, 5224 (2022).
Rao, Z. et al. Soft electronics for the skin: from health monitors to human–machine interfaces. Adv. Mater. Technol. 5, 2000233 (2020).
Sootsman, J. R., Chung, D. Y. & Kanatzidis, M. G. New and old concepts in thermoelectric materials. Angew. Chem. Int. Ed. 48, 8616–8639 (2009).
Du, Y., Xu, J., Paul, B. & Eklund, P. Flexible thermoelectric materials and devices. Appl. Mater. Today 12, 366–388 (2018).
Zhang, L., Shi, X.-L., Yang, Y.-L. & Chen, Z.-G. Flexible thermoelectric materials and devices: from materials to applications. Mater. Today 46, 62–108 (2021).
Fan, Z. & Ouyang, J. Thermoelectric properties of PEDOT:PSS. Adv. Electron. Mater. 5, 1800769 (2019).
Kim, D.-H., Akbar, Z. A., Malik, Y. T., Jeon, J.-W. & Jang, S.-Y. Self-healable polymer complex with a giant ionic thermoelectric effect. Nat. Commun. 14, 3246 (2023).
Liu, S., Li, H., Li, P., Liu, Y. & He, C. Recent advances in polyaniline-based thermoelectric composites. CCS Chem. 3, 2547–2560 (2021).
Yao, C.-J., Zhang, H.-L. & Zhang, Q. Recent progress in thermoelectric materials based on conjugated polymers. Polymers 11, 107 (2019).
Han, C.-G. et al. Giant thermopower of ionic gelatin near room temperature. Science 368, 1091–1098 (2020).
Yoo, D., Lee, J. J., Park, C., Choi, H. H. & Kim, J.-H. N-type organic thermoelectric materials based on polyaniline doped with the aprotic ionic liquid 1-ethyl-3-methylimidazolium ethyl sulfate. RSC Adv. 6, 37130–37135 (2016).
Cheng, H., He, X., Fan, Z. & Ouyang, J. Flexible quasi-solid state ionogels with remarkable seebeck coefficient and high thermoelectric properties. Adv. Energy Mater. 9, 1901085 (2019).
Le, Q., Chen, Z., Cheng, H. & Ouyang, J. Giant thermoelectric performance of n-type ionogels by synergistic dopings of cations and anions. Adv. Energy Mater. 13, 2302472 (2023).
Nowruzi, S., Shokouhyar, S., Dehghan, O., Nezafati, N. & Shokoohyar, S. A human-machine interaction framework for identifying factors influential consumer participation in e-waste treatment schemes. Int. J. Comput. Integr. Manuf. 0, 1–25 (2023).
Vishwakarma, S. et al. E-waste in information and communication technology sector: existing scenario, management schemes and initiatives. Environ. Technol. Innov. 27, 102797 (2022).
Xia, Q. et al. A strong, biodegradable and recyclable lignocellulosic bioplastic. Nat. Sustain. 4, 627–635 (2021).
Zarei, M., Lee, G., Lee, S. G. & Cho, K. Advances in biodegradable electronic skin: material progress and recent applications in sensing, robotics, and human–machine interfaces. Adv. Mater. 35, 2203193 (2023).
Dong, L. et al. Fully biofriendly, biodegradable and recyclable hydrogels based on covalent-like hydrogen bond engineering towards multimodal transient electronics. Chem. Eng. J. 457, 141276 (2023).
Shieh, P. et al. Cleavable comonomers enable degradable, recyclable thermoset plastics. Nature 583, 542–547 (2020).
Morado, E. G. et al. End-of-life upcycling of polyurethanes using a room temperature, mechanism-based degradation. Nat. Chem. 15, 569–577 (2023).
McCulloch, I., Chabinyc, M., Brabec, C., Nielsen, C. B. & Watkins, S. E. Sustainability considerations for organic electronic products. Nat. Mater. 22, 1304–1310 (2023).
Reeder, J. T. et al. Soft, bioresorbable coolers for reversible conduction block of peripheral nerves. Science 377, 109–115 (2022).
Bahrami, A., Schierning, G. & Nielsch, K. Waste recycling in thermoelectric materials. Adv. Energy Mater. 10, 1904159 (2020).
Ren, W. et al. High-performance wearable thermoelectric generator with self-healing, recycling, and Lego-like reconfiguring capabilities. Sci. Adv. 7, eabe0586 (2021).
Zheng, J. et al. Integrating recyclable polymers into thermoelectric devices for green electronics. J. Mater. Chem. A 10, 19787–19796 (2022).
Zhu, P. et al. Recyclable, healable, and stretchable high-power thermoelectric generator. Adv. Energy Mater. 11, 2100920 (2021).
Cao, Y. et al. Self-healing electronic skins for aquatic environments. Nat. Electron. 2, 75–82 (2019).
Wei, Z. et al. Autonomous self-healing of poly(acrylic acid) hydrogels induced by the migration of ferric ions. Polym. Chem. 4, 4601 (2013).
Jin, X. et al. A self-healing zinc ion battery under −20 °C. Energy Storage Mater. 44, 517–526 (2022).
Chen, B. et al. Giant negative thermopower of ionic hydrogel by synergistic coordination and hydration interactions. Sci. Adv. 7, eabi7233 (2021).
Ge, G. et al. Muscle-inspired self-healing hydrogels for strain and temperature sensor. ACS Nano 14, 218–228 (2020).
Wu, X., Gao, N., Jia, H. & Wang, Y. Thermoelectric converters based on ionic conductors. Chem. – Asian J. 16, 129–141 (2021).
Lindén, L. -åke & Rabek, J. F. Structures and mechanisms of formation of poly(acrylic acid)-iron (II and III) chloride gels in water and hydrogen peroxide. J. Appl. Polym. Sci. 50, 1331–1341 (1993).
Neira, A., Tarraga, M. & Catalan, R. Degradation of polyacrylic acid by fenton’s reagent. J. Chil. Chem. Soc. 52, 1314–1317 (2007).
Ullah, N., Guziejewski, D., Barton, B. & Mirceski, V. Electrocatalytic reduction of hydrogen peroxide in solution in the presence of polyacrylic acid and copper ions: application for gaseous hydrogen peroxide detection. J. Electroanal. Chem. 951, 117918 (2023).
Xu, J. et al. Highly stretchable PU Ionogels with self-healing capability for a flexible thermoelectric generator. ACS Appl. Mater. Interfaces 13, 20427–20434 (2021).
Xue, Y. et al. Stretchable, ultratough, and intrinsically self-extinguishing elastomers with desirable recyclability. Adv. Sci. 10, e2207268 (2023).
Chi, C. et al. Selectively tuning ionic thermopower in all-solid-state flexible polymer composites for thermal sensing. Nat. Commun. 13, 221 (2022).
Ghatak, B. et al. Design of a self-powered triboelectric face mask. Nano Energy 79, 105387 (2021).
Würger, A. Thermoelectric ratchet effect for charge carriers with hopping dynamics. Phys. Rev. Lett. 126, 068001 (2021).
Fu, M., Sun, Z., Yuan, Y. & Yue, K. Ionic thermoelectric materials based on the thermodiffusion effect: mechanism, advancements, and applications. Macromol. Chem. Phys. 226, 2400358 (2025).
Cheng, T., Shao, J. & Wang, Z. L. Triboelectric nanogenerators. Nat. Rev. Methods Prim. 3, 39 (2023).
Guo, H. et al. Artificially innervated self-healing foams as synthetic piezo-impedance sensor skins. Nat. Commun. 11, 5747 (2020).
Liu, W. et al. Touchless interactive teaching of soft robots through flexible bimodal sensory interfaces. Nat. Commun. 13, 5030 (2022).
Acknowledgements
B.C.-K.T. acknowledges funding support from the Agency for Science, Technology and Research Singapore (A*STAR) grants M23NBK0090, A18A8b0059, and the NUS iHealthtech Institute.
Author information
Authors and Affiliations
Contributions
B.C.-K.T., J.Y., and Z.W. conceived and designed the experiments. B.C.-K.T. supervised the project. J.Y. and Z.W. carried out experiments and collected the data. J.Y. designed the two demonstrations. Z.Y. developed the electrical circuits for virtual typing. M.L. worked on the transparency and FTIR tests of the hydrogel. D.L.-K.H. contributed to the operation of the robotic hand. K.B. contributed to the recycling experiments. J.O. suggested improvements to the experiment setups. J.Y. wrote the paper and designed illustrations. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Tao Xiang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yang, J., Wang, Z., Yang, Z. et al. Non-hazardous and fully recyclable ionic thermoelectrics for sustainable human–machine interfaces. Nat Commun 16, 10619 (2025). https://doi.org/10.1038/s41467-025-65377-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-65377-9