Abstract
Relapsed or refractory osteosarcoma carries a poor prognosis after standard chemotherapy. We conducted a multicenter, randomized, phase II trial (NCT05277480) to compare apatinib plus ifosfamide/etoposide (IE) with IE alone in patients who had progressed following at least one prior line of chemotherapy. Patients were randomized 2:1 to receive apatinib (500 mg orally once daily) plus IE (ifosfamide 1.8 g/m²/day and etoposide 100 mg/m²/day, days 1–3 every 3 weeks) or IE alone (same doses, days 1–5 every 3 weeks). The primary endpoint was progression-free survival (PFS). Between April 14, 2022, and August 22, 2023, 81 patients were enrolled (53 in apatinib plus IE group and 28 in IE group). After a median follow-up of 19.9 months, the median PFS was 5.5 months (95% confidence interval [CI]: 3.9, 6.4) with apatinib plus IE compared with 3.4 months (95% CI: 1.4, 4.6) with IE (hazard ratio, 0.60; 95% CI: 0.37, 0.98; P = 0.0402). The trial met its pre-specified primary endpoint. These results suggest that apatinib plus IE may improve PFS in relapsed or refractory osteosarcoma, but as a randomized phase II study, the findings are exploratory and require confirmation in phase III trials.
Similar content being viewed by others
Introduction
The prognosis for patients with inoperable, metastatic osteosarcoma that is resistant to first-line chemotherapy remains poor. Previous studies have demonstrated that multi-targeted tyrosine kinase inhibitor (TKIs) can induce varying degrees of objective responses and prolong progression-free survival (PFS)1,2,3,4,5. However, the efficacy of TKIs is often compromised by factors such as large tumor volumes, poor performance status, and the axial location of lesions4,5. Additionally, clinical trials involving TKIs alone are typically with median PFS ranging from 4 to 6 months accompanied with inevitable development of secondary drug resistance1,2,3,4,5. Specifically, TKIs often exhibit limited and short-lived efficacy in controlling musculoskeletal lesions besides other metastasis outside the lung4,5,6, the underlying mechanism of which remains poorly understood. Osteosarcoma is characterized by significant genomic instability and genetic heterogeneity7, necessitating treatments that have traditionally focused on cytotoxic and cytostatic drugs. These treatments often require combination strategy to address the intra- and inter-tumoral heterogeneity effectively. In this context, classic second-line chemotherapy regimen, such as ifosfamide and etoposide (IE), might complement the activity of TKIs. It might be possible to achieve better overall disease control by combining IE with TKIs, especially for those with multiple metastatic lesions in different organs.
Apatinib, a multi-targeted TKI utilized in the treatment of gastric, non-small cell lung, and esophageal cancers8, has demonstrated efficacy in inhibiting the proliferation, migration, and invasion of osteosarcoma in both preclinical and clinical trials4,6 (Supplementary Table 1). IE are chemotherapeutic agents that induce DNA damage: ifosfamide acts as an alkylating agent, whereas etoposide inhibits topoisomerase II9. Since the 1990s, the combination of IE has been employed in multiple pediatric clinical settings, with a phase II trial reporting a response rate of 48% and acceptable toxicity in relapsed or refractory pediatric osteogenic sarcomas10. The rationale for combining TKIs with IE, including lenvatinib plus IE, was initially proposed by Gaspar et al.11,12,13 with the idea of inhibition of angiogenesis may increase uptake of IE into tumor tissue through vascular normalization. Based on our prior clinical observations, TKI monotherapy appeared insufficient to control extra-pulmonary lesions14. Furthermore, in heavily pretreated patients with inoperable lesions, managing adverse events (AEs) to a tolerable level is crucial since the therapy may need to be administered over a life-long period. By utilizing off-label doses for these combinations, we developed a suitable chemotherapy regimen in combination with apatinib in our previous study, allowing for a longer duration of chemotherapy compared to the OLIE/ITCC-082 study14,15.
Here, we show in a randomized phase II trial that apatinib combined with IE improves PFS compared with IE alone in patients with unresectable, recurrent, or refractory osteosarcoma, with an acceptable safety profile. These exploratory findings provide hypothesis-generating evidence that warrants confirmation in larger phase III studies.
Results
Baseline characteristics of patients
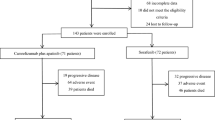
Between April 14, 2022, and August 22, 2023, 89 patients were assessed for eligibility, of whom 81 were enrolled. Of these, 53 were randomized to the apatinib plus IE (Apa+IE) group and 28 to the IE group. All enrolled patients received treatment and were included in both the full analysis set (FAS) and the safety analysis set (SS) (Fig. 1).
FAS, full analysis set, SS, safety analysis set. Source data are provided as a Source Data file.
As shown in Table 1, baseline characteristics were generally balanced between the two groups. The proportion of participants younger than 18 was comparable, accounting for 54.7% in the Apa+IE group and 53.6% in the IE group. The median time since initial diagnosis was slightly shorter in the Apa+IE group at 12.1 months (interquartile range [IQR] 5.4-18.8) compared to 15.1 months (IQR 8.6-22.7) in the IE group. Approximately 30% of patients had an Eastern Cooperative Oncology Group performance status (ECOG PS) of 0. Lung-only metastasis was observed in 66.0% of patients in the Apa+IE group and 71.4% in the IE group. All patients had received prior anthracyclines, while 88.7% of the Apa+IE group and 92.9% of the IE group had received prior ifosfamide treatment in combination with other chemo-agents. 71.7% of patients in the Apa+IE group had undergone prior one surgery, compared to 64.3% in the IE group, while 22.6% and 32.1% respectively had two or more surgeries. Seven patients in the Apa+IE group and four in the IE group received local therapy during the study period. Details of these interventions, including their timing, intent (curative vs. palliative), and associated responses, are summarized in Supplementary Table 2 and visually represented in the swimmer plot (Fig. 2), which also indicates each patient’s baseline resectability status and treatment timeline. Among these, five patients received curative local therapy following tumor shrinkage, while six patients underwent palliative local therapy for pain relief or the prevention of complications.
Treatment exposure and duration of response in the (A) Apa+IE group and (B) IE group. CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease; NE, not estimable. Source data are provided as a Source Data file.
Efficacy
By the cut-off date of October 8, 2024, median follow-up was 19.9 months (95% confidence interval [CI]: 19.1, 21.8). Treatment was ongoing for three patients in the Apa+IE group and one patient in the IE group with last follow-up.
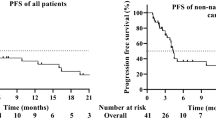
The primary outcome, PFS, was 5.5 months (95% CI, 3.9–6.4) in the Apa+IE group compared with 3.4 months (95% CI, 1.4–4.6) in the IE group (hazard ratio [HR], 0.60; 95% CI, 0.37–0.98; P = 0.0402) (Fig. 3A). The 4-month and 6-month PFS rates were 65.8% (95% CI: 50.3, 77.5) and 36.7% (95% CI: 22.5, 50.9) in the Apa+IE group, versus 41.6% (95% CI: 22.4, 59.8) and 15.9% (95% CI: 4.2, 34.3) in the IE group, respectively. When local therapy was counted as an event rather than censored, the median event-free survival (EFS) was 5.5 months (95% CI: 3.9, 5.9) for the Apa+IE group, compared to 3.4 months (95% CI: 1.4, 4.6) for the IE group (HR, 0.64; 95% CI: 0.40, 1.02, nominal P = 0.0598) (Supplementary Fig. 1). The median overall survival (OS) was 18.1 months (95% CI: 13.7, 25.2) for the Apa+IE group, compared to 22.9 months (95% CI: 19.3, not estimated [NE]) for the IE group (HR, 1.48; 95% CI: 0.76, 2.89; nominal P = 0.2493) (Fig. 3B).
Kaplan-Meier curves of (A) progression-free survival and (B) overall survival. Survival outcomes were estimated using the Kaplan-Meier method and compared between groups with the two-sided log-rank test. Hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated using the Cox proportional hazards model. NE, not estimable. Source data are provided as a Source Data file.
The objective response rate (ORR) was 32.1% in the Apa+IE group and 25.0% in the IE group. The disease control rate (DCR) was observed at 90.6% in the Apa+IE group, while the IE group had a DCR of 60.7%. The median time to progression (TTP) was 5.1 months (95% CI: 3.7, 5.7) in the Apa+IE group and 2.5 months (95% CI: 1.4, 4.1) in the IE group (Table 2). However, the median time to response (TTR) was almost the same, with 1.3 months (95% CI: 1.1, 2.6) in the Apa+IE group while 1.5 months (95% CI: 1.3, NE) in the IE group. The duration of response (DoR) was also nearly the same between these two groups, with 3.9 months (95% CI: 1.9, 7.8) for the Apa+IE group and 4.1 months (95% CI: 1.7, NE) for the IE group. The percentage changes from baseline in target lesions for all patients are shown in Supplementary Fig. 2. One patient with non-extremity primary osteosarcoma, ECOG PS of 1, and a single metastatic site achieved a complete response (CR) following Apa+IE treatment. The patient demonstrated a partial response (PR) after 1 month of therapy, reached CR at 4 months, but subsequently experienced disease progression at 5 months.
In the Apa+IE group, 30.2% had no further treatment (OS events) compared to 18.9% in the IE group. The most common subsequent therapies in the Apa+IE group were multiple other chemotherapy plus TKIs (18.9%), chemotherapy alone (17.0%), and other TKI monotherapy (9.4%). In the IE group, chemotherapy alone (21.4%), chemotherapy plus TKI (14.3%) and antibody-drug conjugates (14.3%) were primarily used (Supplementary Table 3).
Safety profiles
Treatment-related adverse events (TRAEs) were reported in 98.1% of patients in the Apa+IE group and 89.3% of patients in the IE group. Grade 3 or higher TRAEs occurred in 69.8% of the Apa+IE group compared to 64.3% in the IE group. Serious adverse events (SAEs) were observed in 11.3% of patients receiving Apa+IE and 10.7% of those in the IE group. For Apa+IE group, indeed more toxicities occurred, such as pneumothorax. However, neutropenia and thrombocytopenia did not seem to be more severe in the Apa+IE group because the dose of the chemotherapy had been reduced in the combination group (Table 3). In the Apa+IE group, there were no dose interruptions or reductions with chemotherapy, whereas in the IE group, one patient (3.6%) discontinued treatment because of severe myelosuppression. For apatinib, dose interruptions occurred in seven patients (13.2%), dose reductions in 16 patients (30.2%), and discontinuation in one patient (1.9%) within the Apa+IE group (Supplementary Table 4).
Quality of life
Throughout the treatment course, patients in the Apa+IE group reported a statistically significant improvement in global health status, with the mean score increasing from 47.12 at baseline to 56.67 by cycle 9 (nominal P = 0.042). Conversely, the IE group did not exhibit a statistically significant change in Quality of Life (QoL) over the course of treatment (Supplementary Table 5).
Discussion
This randomized, multicenter phase II trial met its primary endpoint, demonstrating a longer median PFS for Apa+IE compared with IE alone. However, unlike our previous retrospective study in which local therapy was permitted14, the current trial did not achieve the anticipated median PFS of approximately 11 months; instead, the observed median PFS was 5.5 months in the Apa+IE group and 3.4 months in the IE group. For patients with unresectable or refractory osteosarcoma following first-line chemotherapy, prognosis remains poor, with 5-year post-relapse survival rates below 20%16,17. Although multi-targeted TKIs can induce measurable responses, durable disease control continues to be a major challenge1,4,5,6,13,14,18. In the present trial, the combination of apatinib with IE improved PFS relative to IE alone, particularly among patients with multiple metastatic sites and high tumor burden (Supplementary Fig. 3). Nevertheless, controlling extrapulmonary lesions remains difficult, which may be partly attributable to the lower daily dose of IE used. In real-world settings, we have explored higher IE doses, similar to those used in the OLIE/ITCC-082 trial; however, most patients (who were heavily pretreated with first-line chemotherapy for one year or longer) could not tolerate more than five cycles. Consequently, although the addition of apatinib to IE provided a modest benefit in PFS, it did not fully overcome rapid secondary resistance, and the extent of improvement was less than initially anticipated.
The rationale for combining apatinib with IE in this study is grounded by the hypothesis that integrating traditional, effective second-line chemotherapy can enhance the overall disease control for both pulmonary and extrapulmonary lesions, which, especially the later, are often poorly controlled by apatinib alone1,2,3,4,5. Nathalie Gaspar et al. previously explored the combination of lenvatinib with IE in patients with refractory or relapsed osteosarcoma, achieving a median PFS of 8.7 months (95% CI 4.5–12.0) in the phase I/II study of ITCC-05012. Their approach was predicated on the synergistic mechanism of cytotoxic agents and VEGF pathway inhibition, which can slow tumor growth through angiogenesis suppression and indue more cytotoxic reactions11. However, results from the phase II randomized controlled study of OLIE/ITCC-082 did not demonstrate statistical superior survival benefit, even for PFS only (6.5 months vs. 5.5 months, respectively)13. Our study, by contrast, has shown that the combination of apatinib with IE prolongs PFS in patients progressed upon chemotherapy, which might mostly be due to a higher percentage of participants in our study had progressed following ifosfamide-containing combination chemo-protocol (90.1% compared to 46.9% in the OLIE/ITCC-082 study)13, which likely weakened the efficacy of the control group. Although single IE intensified the intensity and density of ifosfamide than first-line high-dose methotrexate, doxorubicin, cisplatin and ifosfamide (MAP+I), which could induce some responses or control disease for some time in our study, it was less effective in the ifosfamide-pretreated population other than in the ifosfamide-naïve population. Thus, unlike the OLIE/ITCC-082 trial, our control arm was less effective, which may have contributed to the improved outcomes observed in our Apa+IE group. This factor may account for the numerically shorter median PFS observed in both groups of our study compared to OLIE/ITCC-082.
In the meanwhile, there are some other differences between the OLIE/ITCC-082 and present study13. Most notably, local therapy, which could be performed after 18 weeks of treatment in the lenvatinib plus IE, was not permitted in this study, which might partly explain why the median PFS seems to be longer in the OLIE/ITCC-082 than in our trial. For patients with advanced osteosarcoma, even when lesions are not fully resectable at baseline, local therapies such as radiotherapy for large tumor burdens can significantly delay secondary drug resistance and prolong EFS. This is exemplified in our retrospective study14, where despite patients being in more advanced stages of their disease, the median EFS could reach 11 months. The restriction on localized treatment in the current study created a complex scenario. Although the trial targeted patients with initially inoperable osteosarcomas, treatment led to sufficient tumor downstaging in five patients after 3-4 cycles, enabling them to receive curative-intent local therapy based on investigator assessment. However, eligibility for local interventions was not confirmed by centralized imaging review. Thus, when we calculated all those censoring data to compare the median EFS between the two groups, it was interesting to notice that the P value seemed not to be so significant by 0.0598, which might also be deemed as a confounding factor in OLIE/ITCC-082. While not recommended within the study protocol, this real-world adjustment highlights the ethical dilemmas faced in balancing trial guidelines with patient best interests. As a result, the tumor burden in this study was expectedly higher compared to other trials where local therapies allowed, as we included a larger proportion of patients with late-stage disease, further complicating analysis and comparability of these trials.
Furthermore, the two trials employed different TKIs, making it challenging to directly compare the efficacy of lenvatinib and apatinib given the varied patient populations. Additionally, there were discrepancies in the daily dose and duration of the IE chemotherapy between the trials. Our study utilized a lower daily dose of ifosfamide (1.8 g/m²/day compared to 3 g/m²/day in ITCC-050 and OLIE/ITCC-082) but extended the chemotherapy regimen to 10 cycles, compared to five cycles in the ITCC-050 and OLIE/ITCC-082 studies12,13. This approach was informed by our retrospective findings indicating that Asian patients experienced significant myelosuppression after 3–4 cycles of the higher ifosfamide dose14, and almost all progressions in the ITCC-050 study occurred after the cessation of IE chemotherapy12. Therefore, to balance toxicity and efficacy, we reduced the daily dose and prolonged the duration of chemotherapy. Further investigations should be tried and conducted by managing the toxicities without compromising the effectiveness of the chemotherapy in these kinds of combinations. To facilitate interpretation of cross-trial comparisons, the key characteristics and efficacy outcomes of our study and prior trials investigating TKI plus IE regimens are summarized in Supplementary Table 6.
In this phase II, hypothesis-generating trial, the primary endpoint of PFS reached statistical significance; however, the observed median PFS in both arms was lower than anticipated. This discrepancy likely reflects design features (notably the exclusion of protocol-disallowed local therapies) and the inclusion of a heavily pretreated population. Crucially, PFS was analyzed with censoring at the time of local therapy, whereas EFS counted initiation of subsequent anti-tumor therapy, including local therapy, as an event; under this definition, the between-group difference in EFS did not reach statistical significance (HR 0.64; 95% CI 0.40–1.02; nominal P = 0.0598). Moreover, no OS benefit was observed, potentially influenced by post-progression treatments and AEs related to local interventions. Taken together, these results should be interpreted with appropriate caution: apatinib plus IE may offer benefit for patients with relapsed or refractory osteosarcoma, but the data are exploratory, and confirmation in larger, prospective phase III studies is needed to establish comparative efficacy and optimize patient selection.
In our previous retrospective study, the most prominent AE associated with the combination therapy was ifosfamide-induced encephalopathy, occurring more frequently and with greater severity than typically reported14, partly due to apatinib-induced hypoalbuminemia and hypertension15. However, in the current trial, with improved patient selection and proactive preconditioning measures, no cases of ifosfamide-induced encephalopathy were observed. Another significant AE impacting patients’ QoL was pneumothorax, likely due to necrosis within pulmonary lesions. All patients with pneumothorax were managed by chemical or mechanical pleurodesis in this trial. This procedure, though typically arduous and accompanied by chest pain and fevers, allowed for the continuation of treatment without interruption15.
This study has several limitations. Firstly, we did not include any biomarker analysis, which could have provided insights into the mechanisms underlying the efficacy of the combination. Secondly while our combination cohort demonstrated superior efficacy compared to IE alone, we did not evaluate the benefits of Apa+IE against apatinib monotherapy or apatinib combined with anti-PD-1 therapy. This omission leaves an open question regarding which strategy might provide the most benefit for heavily pretreated populations. Additionally, the open-label design and the apparent differences in chemotherapy dose intensity between treatment groups may have introduced bias into patient-reported QoL assessments, potentially influencing the reliability of these results.
In summary, for patients with unresectable or refractory osteosarcomas that have progressed following chemotherapy, the combination of apatinib and IE demonstrated superior PFS compared to IE alone, with an acceptable toxicity profile. However, no OS benefit was observed, and the discrepancy between assumed and observed PFS estimates, together with the lack of statistical significance for EFS, underscore the exploratory nature of these findings. Future phase III trials are required to confirm these findings and optimize patient selection.
Methods
Study design and patients
This multicenter, randomized phase II trial (OAIE/PKUPH-sarcoma 11) was conducted across four centers in China under the auspices of the Chinese Sarcoma Study Group. The study protocol was approved by the Ethics Committee of the Peking University People’s Hospital; Jinling Hospital, Medical School, Nanjing University; Shanghai General Hospital, Shanghai Jiao Tong University School of Medicine; and Shanghai Jiao Tong University Affiliated Sixth People’s Hospital. It was conducted in accordance with the Declaration of Helsinki and applicable local regulatory requirements and laws. Written informed consents were obtained from all participants, with minors providing assent alongside consents from their legal guardians. The trial is registered on ClinicalTrials.gov (NCT05277480). Patients were provided with the study treatment for free.
The study enrolled adolescents and adults aged 12–65 years, with body surface areas exceeding 1.0 m². Participants were required to have histologically or pathologically confirmed locally advanced or metastatic high-grade osteosarcoma that had either progressed following at least one line of prior chemotherapy (including neoadjuvant or adjuvant chemotherapy). Enrollment criteria mandated that the disease be measurable according to the Response Evaluation Criteria in Solid Tumors (RECIST 1.1)19. Eligible patients needed to have an ECOG PS of 0-1, an expected survival of more than three months, and satisfactory organ function. Exclusion criteria included prior treatment with any TKIs (e.g., apatinib, anlotinib, regorafenib, cabozantinib, sorafenib) or those treated solely with IE chemotherapy. Additionally, patients with symptomatic central nervous system metastasis or uncontrolled comorbid conditions that could interfere with protocol adherence were excluded. Detailed inclusion and exclusion criteria are provided in the study protocol.
Procedure
Eligible patients were randomized in a 2:1 ratio to receive either the combination of apatinib and IE (Apa+IE group) or IE alone (IE group). Participants in the Apa+IE group received 500 mg of apatinib orally once daily, combined with ifosfamide (1.8 g/m²/day d1-3) and etoposide (100 mg/m²/day d1-3), administered every three weeks (Q3W). The IE group received the same daily dose of ifosfamide and etoposide, albeit on an extended schedule (d1-5 Q3W)14. Treatment continued until disease progression, unacceptable toxicity, withdrawal of consent, initiation of non-protocol cancer therapies, or death. Additionally, apatinib was administered for a maximum of one year, while IE was administered for up to 10 cycles; patients who were judged by investigators to be deriving sustained clinical benefit from apatinib at one year were permitted to continue treatment at the physician’s discretion. Dose adjustments were permitted for toxicity management according to protocol; however, if treatment was postponed for over four weeks beyond a planned cycle, discontinuation was mandated.
Both groups underwent tumor evaluations using computed tomography or magnetic resonance imaging at baseline and subsequently every two cycles according to RECIST 1.1 criteria. AEs were recorded and graded using the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE), version 5.0. Furthermore, all participants completed the European Organization for Research and Treatment of Cancer (EORTC) 30-item core Quality of Life (QoL) questionnaire (QLQ-C30)20 prior to initiating treatment, with follow-up QoL assessments conducted every two cycles.
Endpoints
In this study, the primary endpoint was PFS, as assessed by the investigators using RECIST 1.1 criteria. PFS was defined as the time from randomization to disease progression or death from any cause, whichever occurred first. Secondary endpoints included 4-month PFS rate; 6-month PFS rate; OS, measured from randomization to death from any cause; ORR, defined as the proportion of patients achieving a CR or PR; DCR, which encompassed the percentage of patients with either CR, PR, or stable disease (SD); DoR, calculated from the first occurrence of CR or PR to progressive disease (PD) or death; TTR, the interval from randomization to the first CR or PR; TTP, defined as the time from randomization to disease progression; and safety assessments throughout the study duration. The exploratory endpoints included QoL, assessed using the EORTC QLQ-C30 questionnaire, and EFS. During treatment, some patients received local therapies (e.g., surgery or radiotherapy) that were not permitted by protocol. For the PFS analysis, patients were censored at the time of initiation of local therapy. For the EFS analysis, defined as the time from randomization to the first occurrence of disease progression, local or distant recurrence, initiation of subsequent anti-tumor therapy, or death, initiation of local therapy was counted as an event.
Statistical analysis
Assuming a median PFS of 5.0 months in the IE group and 11.0 months in the Apa+IE group14, detecting a HR of 0.45 with 80% power at a two-sided significance level of 0.05 required 53 PFS events. Considering a 2:1 randomization ratio and anticipating an 18-month enrollment period followed by a 12-month follow-up, the study aimed to enroll 46 patients in the Apa+IE group and 23 patients in the IE group to observe sufficient PFS events by the study’s conclusion. Factoring in a 10% dropout rate, the total required sample size was adjusted to 52 patients in the Apa+IE group and 26 patients in the IE group, culminating in a total enrollment of 78 patients.
The FAS included all participants who received at least one dose of the study treatment and served as the primary efficacy analysis dataset. The SS encompassed all patients who received at least one dose of the study treatment. The Kaplan-Meier method was utilized to estimate the median PFS for each treatment group, and the 95% CIs of median PFS were calculated using the Brookmeyer and Crowley method. Differences in PFS between the Apa+IE group and the IE group were evaluated using an unstratified log-rank test. Additionally, an unstratified Cox proportional hazards regression model estimated the HR and its associated 95% CI. Subgroup analyses were performed according to baseline factors. The median values for OS, DoR, TTP, and TTR for each group were also estimated using the Kaplan-Meier method, with 95% CIs calculated by the Brookmeyer and Crowley method. The 95% CIs for the 4- and 6-month PFS rate, ORR and DCR were estimated using the Clopper-Pearson method. QoL assessments, as measured by the subscales of the EORTC QLQ-C30, were scored according to the EORTC scoring manual. Each scale was transformed so that scores ranged from 0 to 100, with higher scores indicating better functioning and lower scores indicating worse symptoms. Scale scores were summarized as continuous variables for baseline and each scheduled post-baseline visit by treatment group, with mean values and standard deviations reported for each time point. The Friedman test was used to assess significant changes in QoL scores over time. All statistical analyses were performed using SAS version 9.3 or higher. A P value < 0.05 was considered statistically significant. Data collection was performed using Bioknow electronic data capture system.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
De-identified individual participant data underlying the results of this study will be made available to qualified researchers for academic purposes upon request. Requests should be directed to the corresponding author (email: tang15877@126.com) and will require approval from the study sponsor. Data access will be granted following submission of a methodologically sound proposal and completion of a data use agreement. Applicants can expect a response within 4 weeks. Data will become available 24 months after study completion and will remain accessible for 5 years. The study protocol and statistical analysis plan are provided in the Supplementary Information (Supplementary Notes 1 and 2). Processed data underlying the figures and tables are provided in the accompanying Source Data file. Due to patient privacy and data protection regulations, raw patient-level data cannot be deposited in a public repository. Source data are provided with this paper.
References
Grignani, G. et al. A phase II trial of sorafenib in relapsed and unresectable high-grade osteosarcoma after failure of standard multimodal therapy: an Italian Sarcoma Group study. Ann. Oncol. 23, 508–516 (2012).
Davis, L. E. et al. Randomised double-blind phase II study of regorafenib in patients with metastatic osteosarcoma. J. Clin. Oncol. 37, 1424–1431 (2019).
Duffaud, F. et al. Efficacy and safety of regorafenib in adult patients with metastatic osteosarcoma: a non-comparative, randomised, double-blind, placebo-controlled, phase 2 study. Lancet Oncol. 20, 120–133 (2019).
Xie, L. et al. Apatinib for advanced osteosarcoma after failure of standard multimodal therapy: an open label phase II clinical trial. Oncologist 24, e542–e550 (2019).
Gaspar, N. et al. Phase I/II study of single-agent lenvatinib in children and adolescents with refractory or relapsed solid malignancies and young adults with osteosarcoma (ITCC-050). ESMO Open 6, 100250 (2021).
Xie L. et al. Apatinib plus camrelizumab (anti-PD1 therapy, SHR-1210) for advanced osteosarcoma (APFAO) progressing after chemotherapy: a single-arm, open-label, phase 2 trial. J Immunother Cancer. 8, e000798 (2020).
Schott, C., Shah, A. T. & Sweet-Cordero, E. A. Genomic complexity of osteosarcoma and its implication for preclinical and clinical targeted therapies. Adv. Exp. Med Biol. 1258, 1–19 (2020).
Scott, L. J. Apatinib: a review in advanced gastric cancer and other advanced cancers. Drugs 78, 747–758 (2018).
Blackledge, G. et al. Experience with ifosfamide in the EORTC soft tissue and bone sarcoma group. Semin Oncol. 19, 14–18 (1992).
Gentet, J. C. et al. Ifosfamide and etoposide in childhood osteosarcoma. A phase II study of the French Society of Paediatric Oncology. Eur. J. Cancer 33, 232–237 (1997).
Gaspar, N., Campbell-Hewson, Q., Huang, J., Okpara, C. E. & Bautista, F. OLIE, ITCC-082: a phase II trial of lenvatinib plus ifosfamide and etoposide in relapsed/refractory osteosarcoma. Future Oncol. 17, 4249–4261 (2021).
Gaspar, N. et al. Lenvatinib with etoposide plus ifosfamide in patients with refractory or relapsed osteosarcoma (ITCC-050): a multicentre, open-label, multicohort, phase 1/2 study. Lancet Oncol. 22, 1312–1321 (2021).
Gaspar, N. et al. Lenvatinib plus ifosfamide and etoposide in children and young adults with relapsed osteosarcoma: a phase 2 randomized clinical trial. JAMA Oncol. 10, 1645–1653 (2024).
Xie, L. et al. Apatinib plus ifosfamide and etoposide for relapsed or refractory osteosarcoma: a retrospective study in two centres. Oncol. Lett. 22, 552 (2021).
Xie, L. et al. Management of apatinib-related adverse events in patients with advanced osteosarcoma from four prospective trials: Chinese sarcoma study group experience. Front. Oncol. 11, 696865 (2021).
Lagmay, J. P. et al. Outcome of patients with recurrent osteosarcoma enrolled in seven phase II trials through Children’s Cancer Group, Pediatric Oncology Group, and Children’s Oncology Group: learning from the past to move forward. J. Clin. Oncol. 34, 3031–3038 (2016).
van Ewijk, R. et al. A systematic review of recent phase-II trials in refractory or recurrent osteosarcoma: Can we inform future trial design?. Cancer Treat. Rev. 120, 102625 (2023).
Schulte, B. et al. Phase II study of pazopanib with oral topotecan in patients with metastatic and non-resectable soft tissue and bone sarcomas. Br. J. Cancer 125, 528–533 (2021).
Eisenhauer, E. A. et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur. J. Cancer 45, 228–247 (2009).
Aaronson, N. K. et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J. Natl. Cancer Inst. 85, 365–376 (1993).
Acknowledgements
OAIE/PKUPH-sarcoma 11 was funded by Noncommunicable Chronic Diseases-National Science and Technology Major Project (No. 2024ZD0525800) and Jiangsu Hengrui Pharmaceuticals Co, Ltd. Apatinib as well as ifosfamide and etoposide in this trial were all provided free of charge by Jiangsu Hengrui Pharmaceuticals Co, Ltd. The first author L.X. also received Research and Development Fund of Peking University People’s Hospital (Clinical research incubation program, No. RDL2022-37) and Beijing Natural Science Foundation-Changping Innovation Collaborative Fund (No. L234041) to support this trial. The funder of the study was involved in the study design, data collection, data analysis, data interpretation, and writing of the report. All authors had full access to all the data in this study and take final responsibility for the decision to submit the manuscript for publication. We extend our deepest gratitude to the patients and their families for their invaluable participation. We also acknowledge the dedicated efforts of all investigators and study staff who contributed to the execution of this trial. We thank Silu Wang (Department of Medical Affairs, Jiangsu Hengrui Pharmaceuticals) for data interpretation.
Author information
Authors and Affiliations
Contributions
W.G., X.T., and L.X. conceived and designed the study. L.X. and X.T. obtained financial support, and L.X. provided administrative support. L.X., X.S., J.X., G.Z., S.W., Y.H., H.H., Y.Y., and T.J. provided study materials or patients. L.X., J.X., X.S., K.L., K.S., Y.L., R.L., D.W., S.S., Z.C., Z.P., G.Z., S.W., Y.H., H.H., W.R., Y.Y., T.J., W.G., and X.T. collected, analyzed and interpreted the data. L.X., J.X., X.S., K.L., K.S., Y.L., R.L., D.W., S.S., Z.C., Z.P., G.Z., S.W., Y.H., H.H., W.R., Y.Y., T.J., W.G., and X.T. drafted and critically revised the manuscript. All authors approved the final version of the manuscript and agree to be accountable for all aspects of the work.
Corresponding authors
Ethics declarations
Competing interests
D.W., S.S., Z.C. and Z.P. are employees of Jiangsu Hengrui Pharmaceuticals. The other authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Vivien Yin, Nathalie Gaspar, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xie, L., Xu, J., Sun, X. et al. Apatinib plus ifosfamide and etoposide versus ifosfamide and etoposide in patients with advanced osteosarcomas (OAIE/PKUPH-sarcoma 11): a randomized phase II study. Nat Commun 16, 10473 (2025). https://doi.org/10.1038/s41467-025-65467-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-65467-8