Abstract
Organocatalytic allylic substitution of Morita-Baylis-Hillman (MBH) adducts is widely regarded as one of the most powerful transformations in organic synthesis. A range of activated carbon nucleophiles have been successfully employed in this reaction, enabling the incorporation of diverse functional moieties. Despite its potential, the use of inert C–H nucleophiles—critical for broadening the reaction’s versatility and synthetic utility—remains a significant challenge in the field. Direct α-C–H functionalization of benzyl amines with MBH adducts offers a promising route to form a new C–C bond while simultaneously establishing a chiral amine moiety, a feature highly attractive from the perspective of organic synthesis. However, this transformation is particularly challenging due to the inherent inertness of the α-C(sp³)–H bonds, significant nucleophilic interference from the NH₂ group, and the complexity of selectivity control. Herein, we have successfully achieved an asymmetric direct α-C–H allylic alkylation of NH₂-unprotected benzylamines with MBH adducts using a bifunctional chiral pyridoxal catalyst, producing biologically important chiral γ-amino acid derivatives in good yields with excellent diastereo- and enantioselectivities. The reaction offers a distinct strategy for synthesizing multiply functionalized compounds from readily available starting materials, significantly expanding access to complex chiral architectures.
Similar content being viewed by others
Introduction
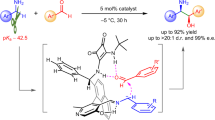
Organocatalytic allylic substitution of Morita-Baylis-Hillman (MBH) adducts has emerged as a highly versatile synthetic transformation1,2,3,4,5,6,7, allowing the introduction of a wide diversity of nucleophiles into multifunctional molecular frameworks. While activated carbon-nucleophiles have been widely utilized8,9,10,11,12,13,14,15,16,17,18,19,20,21,22, the use of inert C–H nucleophiles, essential for expanding the reaction’s versatility and synthetic utility, remains a major challenge23. Benzylamines are a class of readily accessible primary amines possessing two inert α C(sp3)‒H bonds24. Direct asymmetric organocatalytic α-C(sp3)‒H allylic alkylation of benzylamines with MBH adducts offers a promising strategy for simultaneous formation of a C–C bond and establishment of chiral amine functionality (Fig. 1a), providing an efficient and appealing approach to construct chiral substituted γ-aminobutyric acid (GABA) scaffolds that are prevailing core structures of numerous bioactive natural products and pharmaceutical molecules (Fig. 1b)25,26,27,28,29. Moreover, through simple cyclization, γ-aminobutyric acids can be easily converted into another type of biologically important γ-lactam analogs (Fig. 1b)30,31,32. In spite of its potential benefits, this transformation remains undeveloped and is a significant challenge in organic chemistry, even when employing NH2-protected benzylamine derivatives. The difficulties can be attributed to the following two factors (Fig. 1a). First, deprotonating the α-amino C–H bonds to produce active carbanions for initiating the addition is rather difficult due to the extremely low acidity of α-amino C−H bonds (pKa ~ 42.5)24. Second, owing to the high nucleophilicity of the NH₂ group, the classical N-substitution33,34 usually disrupts the desired α-C alkylation, resulting in the predominant formation of the N-alkylated products 4 and/or 5 (Fig. 1a).
a N-Alkylation vs α-C alkylation (SN2’-SN2’) of NH2-unprotected benzylamines. b Representative bioactive γ-aminobutyric acid derivatives. c This work: chiral pyridoxal catalyzed direct asymmetric α C(sp3)‒H alkylation of NH2-unprotected benzylamines with MBH acetates. Ar, aromatic group; Ph, phenyl; SN2’, bimolecular allylic nucleophilic substitution; DBU, 1,8-diazabicyclo[5.4.0]undec-7-ene; DCM, dichloromethane; dr, diastereomeric ratio; ee, enantiomeric excess.
Carbonyl catalysis35,36,37,38,39,40 has become an effective strategy for direct α C‒H functionalization of NH2-unprotected primary amines41,42,43,44,45,46,47,48,49,50. With chiral pyridoxals as the carbonyl catalysts, a series of enantioselective transformations of activated primary amines, such as α-amino esters with diverse electrophiles, have been successfully accomplished, affording diverse chiral amino acid derivatives41,42,43. Nevertheless, direct asymmetric α-functionalization of primary amines containing inert α-amino C–H bonds24,44,45, for instance, benzylamines, presents greater difficulties. Till now, for the carbonyl-catalyzed reaction of benzylamines, only the asymmetric addition to aldehydes has been achieved24. Allylic alkylation of benzylamines with MBH acetates is more difficult to achieve, primarily due to the strong interference from the highly nucleophilic amino group and the more complicated functional group compatibility of MBH acetates. Herein, we would like to disclose our success on the direct asymmetric α C−H alkylation of benzylamines 1 with MBH acetates 2, with a switched nucleophilic selectivity from N to α-C of benzylamines 1, enabled by the chiral bifunctional pyridoxal catalysts 641 bearing an amide side chain attached to the C3 position of the naphthyl ring. This reaction produced a wide range of chiral polysubstituted γ-amino acid esters 3 with excellent diastereo- and enantioselectivities (up to >20:1 dr, 99% ee) (Fig. 1c).
Results
Reaction optimization
Our studies commenced with the investigation of the direct asymmetric α C−H alkylation of benzylamine (1a) with MBH acetate 2a (Fig. 2, Supplementary Table S1). To our delight, with pyridoxal (S,S)-6a as the catalyst and DBU as the base, the reaction proceeded smoothly for 24 h as anticipated to afford chiral γ-amino acid ester 3a in a 58% yield with >20:1 diastereomeric ratio (dr) and 99% ee for the major diastereomer accompanied by the formation of some N-alkylated by-products (Fig. 2, Supplementary Table S1, entry 1). The pyridoxal catalyst is crucial for this reaction, as no desired α-C allylic alkylation but only N-alkylation can be observed in its absence (Supplementary Table S1, entries 2 and 3), demonstrating that the pyridoxal catalyst is capable of switching the nucleophilic selectivity of benzylamine 1a from N to α-C without protecting the NH₂ group. Extending the reaction time to 72 h can effectively improve the yield of the reaction to 71% while maintaining the excellent diastereo- and enantioselectivities ( > 20:1 dr, 99% ee) (Fig. 2, Supplementary Table S1, entry 4). The diastereomeric pyridoxal (R,S)-6a was less effective for the reaction, resulting in the product 3a with decreased yield and stereoselectivity (Fig. 2). Among the pyridoxals 6a-e examined, compound (S,S)-6a displayed the best performance regarding activity, diastereo-, and enantioselectivities (Fig. 2). Pyridoxals (R,S)-7 and (S,S)-7 possessing a lateral amide chain at the C2 position of the naphthyl ring were completely ineffective for the reaction (Fig. 2), indicating the side chain is important for the activity of the pyridoxal catalyst. Reaction condition investigations exhibited that DBU was the base of choice (Supplementary Table S1, entry 1 vs 12-16) and dichloromethane was the optimal solvent (Supplementary Table S1, entry 1 vs 17-20).
Reaction conditions: 1a (0.20 mmol), 2a (0.10 mmol), catalyst 6 (0.01 mmol, 10 mol%), and DBU (0.20 mmol) in DCM (0.5 mL) at -20 oC for 72 h. Isolated yields were based on 2a. The dr values were determined by 1H NMR analysis of the crude reaction mixtures. The ee values were determined by chiral HPLC analysis. iPr: isopropyl; tBu: tert-butyl; Ph: phenyl; Bn: benzyl; Ac, acetyl; DBU, 1,8-diazabicyclo[5.4.0]undec-7-ene; DCM, dichloromethane. aThe reaction time was 24 h.
Substrate scope
Under the optimal reaction conditions, substrate scope on benzylamines was investigated (Fig. 3). A variety of benzylamines 1 bearing different electron-donating and/or electron-withdrawing substituents at the ortho-, meta-, or/and para-positions of the benzene ring all smoothly underwent the direct asymmetric α C‒H allylic alkylation with MBH acetate 2a, producing the corresponding products 3b-m in good yields (60-77%) with excellent diastereo- and enantioselectivities ( > 20:1 dr and 96-99% ee) (Fig. 3a). The absolute configuration of the major diastereomer of product 3m was determined as (3S,4 R) by X-ray analysis (Fig. 3a). Delightfully, the reaction is not sensitive to the steric hindrance. The ortho-substituted benzylamine, such as o-tolylmethanamine (as for 3j), still displayed good reactivity with high stereoselectivity ( > 20:1 dr, 98% ee). Naphthyl substituted methanamines (as for 3n and 3o) as well as heteroarylmethanamines such as thiophen-3-ylmethanamine (as for 3p), pyridin-3-ylmethanamine (as for 3q), and (4-methoxypyridin-2-yl)methanamine (as for 3r) were also applicable for the reaction. The other reaction partner MBH acetates were also examined. Phenyl (as for 3s), substituted phenyl (as for 3t–ad) and heteroaromatic (as for 3ae–ah) MBH acetates with different electronic properties and substitution patterns on the benzene ring were proven to be effective substrates for the α-C allylic substitution, as they all exhibited good reactivities and led to the formation of products 3s–ah with uniformly high diastereomeric ratios ( > 20:1 dr) and high enantiopurities (94-98% ee) (Fig. 3b). The electronic property of the substituted phenyl groups seems to have little impact on the diastereo- and enantioselectivity. Alkenyl- and alkynyl-substituted MBH acetates are also reactive for the transformation, producing chiral γ-amino acid esters 3ai and 3aj with excellent diastereoselectivity (Fig. 3b). The relatively low ee value observed for 3aj is likely due to the linear geometry of the alkyne group, which lacks sufficient steric hindrance for effective enantiocontrol. Alkyl-substituted MBH acetates, such as tert-butyl 3-acetoxy-2-methylene-5-phenylpentanoate, are completely ineffective for the α-C allylic alkylation. Notably, when utilizing substrates containing a biologically active chiral moiety derived from D-glucose (as for 3ak), testosterone51 (as for 3al), or estrone (as for 3am), the reaction proceeded fluently to afford products 3ak-am in good yields with excellent diastereocontrol (Fig. 3c).
a Investigation on benzylamines. b Investigation on MBH acetates. c Investigation on chiral substrates. Reaction conditions: 1 (0.40 mmol), 2 (0.20 mmol), (S,S)-6a (0.02 mmol, 10 mol%) and DBU (0.40 mmol) in DCM (1.0 mL) at −20 °C for 72 h. The isolated yields were based on 2. The dr values were determined by 1H NMR analysis of crude reaction mixtures. The ee values were determined by chiral HPLC analysis. The absolute configuration for 3m was determined as (3S,4R) by X-ray analysis, and those for 3a-3l, 3n-3am were tentatively assigned by analog. DBU, 1,8-diazabicyclo[5.4.0]undec-7-ene; DCM, dichloromethane.
Synthetic utility
To demonstrate the practical utility of the protocol, the reaction was carried out on a gram-scale. Chiral γ-amino acid ester 3h (1.051 g) was obtained in a comparable yield with the same diastereo- and enantioselectivies (Fig. 4a). The synthetic utility of the product was further investigated. Chiral γ-amino acid esters 3 can be facilely transformed into different derivatives with potential bioactivities (Fig. 4b). Deprotection of the tert-butyl moiety of the major diastereomer (3S,4R)-3h by means of HCl produced chiral γ-aminobutyric acid 8h in 95% yield while maintaining the same enantiopurity. As illustrated in Fig. 4b, cyclic α-methylene-γ-lactam 9a and 9h were successfully obtained in satisfactory yields with high enantiopurities via sequential deprotection and condensation. Notably, α-methylene-γ-lactam derivatives have been discovered to exhibit anti-inflammatory, phytotoxic, cytotoxic, and antimicrobial bioactivities30,31,32. Additionally, under Pd-catalyzed hydrogenation conditions, the reduction of 3h proceeded smoothly to afford a pair of chromatographically separable diastereomers (2R,3R,4R)-10h (46% yield) and (2S,3R,4R)-10h’ (32% yield) without any loss of enantiopurity, as depicted in Fig. 4b. Furthermore, compound 10h’ can be converted into cyclic γ-lactam 11h’ as presented in Fig. 4b. The absolute configuration of compounds 11h’ as well as 9a were confirmed by X-ray analysis (see Supplementary Information).
a Gram-scale reaction. b Synthetic transformations. Ac, acetyl; EDCl, 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride; HOBT, 1-hydroxybenzotriazole; DIPEA, N, N-Diisopropylethylamine; EA, ethyl acetate; DBU, 1,8-diazabicyclo[5.4.0]undec-7-ene; DCM, dichloromethane.
Mechanistic studies
The reaction was proposed to proceed through a carbonyl catalysis mechanistic pathway, in which the chiral pyridoxal catalyst served as a pivotal factor (Fig. 5a)35,36,37,38,39. The condensation between chiral pyridoxal catalyst 6a and arylmethanamine 1 results in the formation of imine 12, which activates the benzylic C–H bonds and remarkably increases the C–H acidity for further deprotonation to generate delocalized carbanion 1352,53. Computational pKa analysis reveals the benzylic C–H of imine 12 is more acidic than the phenolic O–H and N–H of the pyridoxal scaffold, thermodynamically favoring the formation of the carbanion via selective C–H deprotonation (see Supplementary Information for details). The carbanion 13 undergoes an asymmetric addition reaction to quaternary ammonium salt intermediate 14, which is generated via the nucleophilic addition of DBU to MBH acetate 2. The addition is accompanied by the expulsion of DBU, leading to the formation of species 16. Hydrolysis of compound 16 leads to the liberation of the SN2’-SN2’ α-C alkylation product 3 and the regeneration of the pyridoxal catalyst 6a, completing the catalytic cycle.
a Proposed reaction mechanism. b Kinetic isotope effect studies. c Computationally-optimized transition state. d Comparison of catalysts. DBU, 1,8-diazabicyclo[5.4.0]undec-7-ene; DCM, dichloromethane.
Kinetic isotope effect (KIE) studies were conducted with equimolar amounts of benzylamine 1a and α-deuterated counterpart 1a-d (Fig. 5b). The reaction led to the formation of compound 3a and the deuterated 3a-d, with a ratio of 7.69:1. The distinct kinetic isotope effect strongly suggests that the deprotonation of imine 12 to afford the active carbanion intermediate 13 is the rate-determining step for the entire reaction pathway.
To elucidate the origin of chirality, computational investigations have been conducted. Figure 5c depicts the optimized transition state (15) for the step involving the asymmetric addition of carbanion 13 to intermediate 14. While benzylamine 1 is bound by the catalyst via the formation of the imine with the pyridoxal moiety, the MBH acetate-derived intermediate 14 is activated by the side chain of the pyridoxal catalyst. This activation occurs via a hydrogen-bonding interaction54,55,56 between the NH group of the side chain and the carbonyl group of intermediate 14. To minimize steric repulsion, intermediate 14 adopts an orientation wherein both the bulky R group and the DBU moiety are directed away from the biaryl backbone of the pyridoxal catalyst. The carbanion originating from arylmethanamines approaches 14 from above to afford chiral γ-amino acid esters 3 with (3S,4R)-configuration from chiral pyridoxal (S,S)-6a.
The postulated transition state is supported by control experiments on the comparison of catalysts (Fig. 5d). Methylation of the amide N–H group on the lateral side chain of pyridoxal 6a resulted in a significant decline in activity and enantioselectivity. As shown in Fig. 5d, the reaction with catalyst 6f afforded a 10% yield, a diastereomeric ratio (dr) of >20:1, and an enantiomeric excess (ee) of 53%, in contrast to the 34% yield, >20:1 dr, and 92% ee obtained with catalyst 6e. The result implies that the amide N–H group likely participates in the catalytic process via hydrogen bonding, as proposed in transition state 15. The cooperative bifunctional activation accounts for the excellent performance of pyridoxals 6 bearing a C3 amide side chain in the reaction. This was further confirmed by the fact that pyridoxals 7, having a C2 amide side chain, are completely inactive for the reaction (Fig. 2). It is supposed that the side chain is too close to the aldehyde moiety to guarantee effective bifunctional activation during the catalysis.
Discussion
In summary, we have successfully developed a direct asymmetric α C−H allylic alkylation of benzylamines 1 with MBH acetates 2 by utilizing bifunctional chiral pyridoxal (S,S)-6a as the catalyst. This reaction furnishes diverse chiral γ-amino acid esters 3, which are of biological significance, in 40–77% yields with excellent diastereo- ( > 20:1 dr) and enantioselectivities (79–99% ee). This work exemplifies a remarkable illustration of organocatalyzed inert C–H bond functionalization under mild conditions, offering high stereocontrol without directing or protecting group manipulations. Moreover, it also highlights the distinctive exceptional capabilities of vitamin B657,58 based organocatalysts59,60,61 in organic synthesis.
Methods
General procedure for asymmetric α C(sp 3)‒H allylic alkylation of benzylamines with MBH acetates (Fig. 3)
To a 4 mL vial equipped with a magnetic stirrer bar were successively added chiral pyridoxal (S,S)-6a (0.0046 g, 0.010 mmol), DBU (0.0304 g, 0.20 mmol), DCM (0.3 mL) and benzylamine 1 (0.20 mmol). The mixture was stirred at −20 °C for 5 min, and a solution of MBH acetate 2 (0.10 mmol) in DCM (0.2 mL) was added in portions over 1 h. After the reaction mixture was stirred at −20 °C for 72 h, it was allowed to warm up to room temperature and concentrated via rotary evaporator to remove most of the solvent. Then it was dried under vacuum and submitted to 1H NMR analysis to determine the dr values. The product 3 was purified by column chromatography on silica gel (petroleum ether: ethyl acetate = 3:1). The dr values of products 3a-ak were determined by 1H NMR analysis of the crude reaction mixtures. The enantiomeric excesses (ee’s) of products 3a-ak were determined by chiral HPLC analysis.
Procedure for synthesis of γ-amino acid ester 3 h in gram-scale (Fig. 3a)
To a 25 mL flask equipped with a magnetic stirrer bar were successively added chiral pyridoxal (S,S)-6a (0.144 g, 0.312 mmol), DBU (0.950 g, 6.250 mmol), DCM (9.35 mL) and arylmethanamine 1h (1.094 g, 6.250 mmol). The mixture was stirred at −20 °C for 5 min, and a solution of MBH acetate 2a (1.100 g, 3.130 mmol) in DCM (6.25 mL) was added in portions over 1 h. After the reaction mixture was stirred at −20 °C for 72 h, it was allowed to warm up to room temperature and concentrated via rotary evaporator to remove most of the solvent. Then it was dried under vacuum and submitted to 1H NMR analysis to determine the dr values. The crude reaction mixture was purified by column chromatography on silica gel (petroleum ether: ethyl acetate = 3:1) to afford compound (S,R)-3h (1.051 g, 72% yield, >20:1 dr, 96% ee) as a white solid.
Data availability
The authors declare that the data supporting the findings of this study are available within the article and Supplementary Information file, or from the corresponding author upon request. The X-ray crystallographic coordinates for structures reported in this study have been deposited at the Cambridge Crystallographic Data Center (CCDC), under deposition numbers of CCDC 2406669 [(3S,4R)-3m in Supplementary Fig. S8], CCDC 2406666 [(4S,5R)-9a in Supplementary Fig. S9] and CCDC 2406667 [(3S,4R,5R)-11h’ in Supplementary Fig. S10]. These data can be obtained free of charge from The Cambridge Crystallographic Data Center via https://www.ccdc.cam.ac.uk/structures/. Coordinates of the optimized structures are available from the Supplementary Data 1.
References
Rios, R. Organocatalytic enantioselective methodologies using Morita–Baylis–Hillman carbonates and acetates. Catal. Sci. Technol. 2, 267–278 (2012).
Liu, T.-Y., Xie, M. & Chen, Y.-C. Organocatalytic asymmetric transformations of modified Morita–Baylis–Hillman adducts. Chem. Soc. Rev. 41, 4101–4112 (2012).
Wei, Y. & Shi, M. Recent advances in organocatalytic asymmetric Morita–Baylis–Hillman/aza-Morita–Baylis–Hillman reactions. Chem. Rev. 113, 6659–6690 (2013).
Wang, T., Han, X., Zhong, F., Yao, W. & Lu, Y. Amino acid-derived bifunctional phosphines for enantioselective transformations. Acc. Chem. Res. 49, 1369–1378 (2016).
Guo, H., Fan, Y. C., Sun, Z., Wu, Y. & Kwon, O. Phosphine organocatalysis. Chem. Rev. 118, 10049–10293 (2018).
Pellissier, H. Asymmetric organocatalytic Morita−Baylis−Hillman reaction and asymmetric organocatalytic transformations of Morita−Baylis−Hillman adducts. An update. Tetrahedron 172, 134435 (2025).
Duan, X.-H., Du, H.-R. & Song, Y.-X. Recent advances in catalytic asymmetric reactions of Morita–Baylis–Hillman adducts. Org. Chem. Front. https://doi.org/10.1039/d4qo02212b (2025).
Cho, C.-W. & Krische, M. J. Regio- and stereoselective construction of γ-butenolides through phosphine-catalyzed substitution of Morita–Baylis–Hillman acetates: An organocatalytic allylic alkylation. Angew. Chem. Int. Ed. 43, 6689–6691 (2004).
Ramachandran, P. V., Madhi, S., Bland-Berry, L., Ram Reddy, M. V. & O’Donnell, M. J. Catalytic enantioselective synthesis of glutamic acid derivatives via tandem conjugate addition−elimination of activated allylic acetates under chiral PTC conditions. J. Am. Chem. Soc. 127, 13450–13451 (2005).
Jiang, Y.-Q., Shi, Y.-L. & Shi, M. Chiral phosphine-catalyzed enantioselective construction of γ-butenolides through substitution of Morita−Baylis−Hillman acetates with 2-trimethylsilyloxy furan. J. Am. Chem. Soc. 130, 7202–7203 (2008).
Wang, L., Prabhudas, B. & Clive, D. L. J. Formation of carbocycles by intramolecular conjugate displacement: scope and mechanistic insights. J. Am. Chem. Soc. 131, 6003–6012 (2009).
Zhang, Q. & Yang, L. & Tong, X. 2-(Acetoxymethyl)buta-2,3-dienoate, a versatile 1,4-biselectrophile for phosphine-catalyzed (4 + n) annulations with 1,n-bisnucleophiles (n = 1, 2). J. Am. Chem. Soc. 132, 2550–2551 (2010).
Peng, J., Huang, X., Cui, H.-L. & Chen, Y.-C. Organocatalytic and electrophilic approach to oxindoles with C3-quaternary stereocenters. Org. Lett. 12, 4260–4263 (2010).
Furukawa, T. et al. Asymmetric allylic monofluoromethylation and methylation of Morita–Baylis–Hillman carbonates with FBSM and BSM by cooperative cinchona alkaloid/FeCl2 catalysis. Angew. Chem. Int. Ed. 50, 9684–9688 (2011).
Liu, C. et al. Chiral biscinchona alkaloid promoted asymmetric allylic alkylation of 3-substituted benzofuran-2(3H)-ones with Morita–Baylis–Hillman carbonates. J. Org. Chem. 76, 5838–5845 (2011).
Zhong, F., Luo, J., Chen, G.-Y., Dou, X. & Lu, Y. Highly enantioselective regiodivergent allylic alkylations of MBH carbonates with phthalides. J. Am. Chem. Soc. 134, 10222–10227 (2012).
Yang, W. et al. Direct asymmetric allylic alkenylation of N-itaconimides with Morita–Baylis–Hillman carbonates. J. Org. Chem. 77, 6600–6607 (2012).
Mao, H. et al. Construction of enantiomerically enriched diazo compounds using diazo esters as nucleophiles: chiral Lewis base catalysis. Angew. Chem. Int. Ed. 52, 6288–6292 (2013).
Han, X. et al. Asymmetric synthesis of spiropyrazolones through phosphine-catalyzed [4+1] annulation. Angew. Chem. Int. Ed. 53, 5643–5647 (2014).
Zhang, L. et al. Phosphine-catalyzed highly enantioselective [3 + 3] cycloaddition of Morita–Baylis–Hillman carbonates with C,N-cyclic azomethine imines. J. Am. Chem. Soc. 137, 4316–4319 (2015).
Chen, P. et al. Phosphine-catalyzed asymmetric umpolung addition of trifluoromethyl ketimines to Morita–Baylis–Hillman carbonates. Angew. Chem. Int. Ed. 55, 13316–13320 (2016).
Ni, C. & Tong, X. Amine-catalyzed asymmetric (3 + 3) annulations of β′-acetoxy allenoates: enantioselective synthesis of 4H-pyrans. J. Am. Chem. Soc. 138, 7872–7875 (2016).
Hu, Y. et al. Organocatalytic asymmetric C(sp2)−H allylic alkylation: enantioselective synthesis of tetrasubstituted allenoates. Angew. Chem. Int. Ed. 59, 19820–19824 (2020).
Hou, C. et al. Catalytic asymmetric α C(sp3)–H addition of benzylamines to aldehydes. Nat. Catal. 5, 1061–1068 (2022).
Nascimento, A. A., Pereira-Figueiredo, D., Borges-Martins, V. P., Kubrusly, R. C. & Calaza, K. C. GABAergic system and chloride cotransporters as potential therapeutic targets to mitigate cell death in ischemia. J. Neurosci. Res. 102, e25355 (2024).
Laribi, M., Chaouali, N., Jaballah, S., Amira, D. & Hedhili, A. Drug abuse of pregabalin-state of the situation, risks and means of control. Ann. Pharm. Fr. 81, 419–424 (2023).
Stratmann, K., Burgoyne, D. L., Moore, R. E., Patterson, G. M. L. & Smith, C. D. Hapalosin, a cyanobacterial cyclic depsipeptide with multidrug-resistance reversing activity. J. Org. Chem. 59, 7219–7226 (1994).
Guckian, K. M. et al. Bicyclic aryl sphingosine 1-phosphate analogs, WO patent 2,010,051,030 A1 (2010).
Urquilla, A., Merrer, D. C., Sumner, R. & Denton, R. W. Synthesis and biological activity of 2-(2-amino-2-phenylethyl)-5-oxotetrahydrofuran-2-carboxylic acid: a microwave-assisted 1,3-dipolar cycloaddition approach. Synlett 32, 1735–1740 (2021).
Konaklieva, M. I. & Plotkin, B. J. Bioisosters of β-lactams as anti-infectives. Curr. Med. Chem. - Anti-Infect. Agents 2, 287–302 (2003).
Cardellina, J. H. & Moore, R. E. The structures of pukeleimides A, B, D, E, F, and G. Tetrahedron Lett. 20, 2007–2010 (1979).
Tchissambou, L., Bénéchie, M. & Khuong-Huu, F. Alcaloïdes imidazoliques IV. Synthèse de la DL-isoanantine et de la DL-anantine. Tetrahedron Lett. 19, 1801–1802 (1978).
Li, D., Li, J. & Jia, X. Regio- and stereoselective synthesis of α,β-dehydro-β-amino esters. J. Chem. Res. 2008, 434–436 (2008).
Ghosh, S., Dey, R., Chattopadhyay, K. & Ranu, B. C. Water-promoted highly regio- and stereoselective synthesis of α-dehydro-β-amino esters and nitriles from Baylis–Hillman acetates. Tetrahedron Lett. 50, 4892–4895 (2009).
Chen, J. et al. Carbonyl catalysis enables a biomimetic asymmetric Mannich reaction. Science 360, 1438–1442 (2018).
Li, S., Chen, X.-Y. & Enders, D. Aldehyde catalysis: new options for asymmetric organocatalytic reactions. Chem 4, 2026–2028 (2018).
Wang, Q., Gu, Q. & You, S.-L. Enantioselective carbonyl catalysis enabled by chiral aldehydes. Angew. Chem. Int. Ed. 58, 6818–6825 (2019).
Xiao, X. et al. Biomimetic asymmetric catalysis. Sci. China Chem. 66, 1553–1633 (2023).
Xiao, X. & Zhao, B. Vitamin B6-based biomimetic asymmetric catalysis. Acc. Chem. Res. 56, 1097–1117 (2023).
Wen, W. & Guo, Q.-X. Chiral aldehyde catalysis-enabled asymmetric α-functionalization of activated primary amines. Acc. Chem. Res. 57, 776–794 (2024).
Ma, J. et al. Asymmetric α-allylation of glycinate with switched chemoselectivity enabled by customized bifunctional pyridoxal catalysts. Angew. Chem. Int. Ed. 134, e202200850 (2022).
Ma, J. et al. Enantioselective synthesis of pyroglutamic acid esters from glycinate via carbonyl catalysis. Angew. Chem. Int. Ed. 60, 10588–10592 (2021).
Cheng, A. et al. Efficient asymmetric biomimetic aldol reaction of glycinates and trifluoromethyl ketones by carbonyl catalysis. Angew. Chem. Int. Ed. 60, 20166–20172 (2021).
Ji, P. et al. Direct asymmetric α-C−H addition of N-unprotected propargylic amines to trifluoromethyl ketones by carbonyl catalysis. Angew. Chem. Int. Ed. 61, e202206111 (2022).
Zhang, R. et al. Direct enantioselective α-C–H conjugate addition of propargylamines to α,β-unsaturated ketones via carbonyl catalysis. J. Am. Chem. Soc. 146, 25927–25933 (2024).
Xu, B. et al. Catalytic asymmetric direct α-alkylation of amino esters by aldehydes via imine activation. Chem. Sci. 5, 1988–1991 (2014).
Wen, W. et al. Chiral aldehyde catalysis for the catalytic asymmetric activation of glycine esters. J. Am. Chem. Soc. 140, 9774–9780 (2018).
Chen, L., Luo, M.-J., Zhu, F., Wen, W. & Guo, Q.-X. Combining chiral aldehyde catalysis and transition-metal catalysis for enantioselective α-allylic alkylation of amino acid esters. J. Am. Chem. Soc. 141, 5159–5163 (2019).
Liu, J.-H. et al. Catalytic asymmetric Tsuji–Trost α−benzylation reaction of N-unprotected amino acids and benzyl alcohol derivatives. Nat. Commun. 13, 2509 (2022).
Zhu, F. et al. Chiral aldehyde/palladium catalysis enables asymmetric branched-selective ring-opening functionalization of methylenecyclopropanes with amino acid esters. J. Am. Chem. Soc. 147, 2315–2322 (2025).
Luberti, F. R. & Carré, J. M. Testosterone’s role in modulating human behaviors relevant to mating and parenting. Front. Neuroendocr. 72, 101112 (2024).
Crugeiras, J., Rios, A., Riveiros, E. & Richard, J. P. Substituent effects on electrophilic catalysis by the carbonyl group: anatomy of the rate acceleration for PLP-catalyzed deprotonation of glycine. J. Am. Chem. Soc. 133, 3173–3183 (2011).
Tang, S., Zhang, X., Sun, J., Niu, D. & Chruma, J. J. 2-Azaallyl anions, 2-azaallyl cations, 2-azaallyl radicals, and azomethine ylides. Chem. Rev. 118, 10393–10457 (2018).
Taylor, M. S. & Jacobsen, E. N. Asymmetric catalysis by chiral hydrogen-bond donors. Angew. Chem. Int. Ed. 45, 1520–1543 (2006).
Doyle, A. G. & Jacobsen, E. N. Small-molecule H-bond donors in asymmetric catalysis. Chem. Rev. 107, 5713–5743 (2007).
Lu, L.-Q., An, X.-L., Chen, J.-R. & Xiao, W.-J. Dual activation in organocatalysis: design of tunable and bifunctional organocatalysts and their applications in enantioselective reactions. Synlett 2012, 490–508 (2012).
Breslow, R. Biomimetic chemistry and artificial enzymes: catalysis by design. Acc. Chem. Res. 28, 146–153 (1995).
Chen, J., Liu, Y. E., Gong, X., Shi, L. & Zhao, B. Biomimetic chiral pyridoxal and pyridoxamine catalysts. Chin. J. Chem. 37, 103–112 (2019).
Dalko, P. I. & Moisan, L. Enantioselective organocatalysis. Angew. Chem. Int. Ed. 40, 3726–3748 (2001).
List, B. Introduction: organocatalysis. Chem. Rev. 107, 5413–5415 (2007).
MacMillan, D. W. C. The advent and development of organocatalysis. Nature 455, 304–308 (2008).
Acknowledgments
We are grateful for the generous financial support from the National Key R&D Program of China (2023YFA1506402, B.Z.), National Natural Science Foundation of China (NSFC) (22271192, B.Z.; 22271191, W.C.), the Shanghai Municipal Science and Technology Major Project, the Shanghai Municipal Committee of Science and Technology (23ZR1446300, W.C.; 24ZR1456200, S.L.), and the Shanghai Engineering Research Center of Green Energy Chemical Engineering (18DZ2254200, B.Z.).
Author information
Authors and Affiliations
Contributions
B.Z. conceived and directed the project and wrote the paper. W.C. co-directed the project and co-wrote the manuscript. J.C. and Y.Y. performed the experiments and most of the Supplementary Documents. S.L. and T.C. conducted the DFT calculations. W.L. and R.Z. prepared part of the Supplementary Documents.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, J., Yang, Y., Liu, S. et al. Direct asymmetric α C(sp3)‒H alkylation of benzylamines with MBH acetates enabled by bifunctional pyridoxal catalysts. Nat Commun 16, 10645 (2025). https://doi.org/10.1038/s41467-025-65648-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-65648-5