Abstract
Transpiration cooling is a thermal management technique that utilizes the phase change of liquid coolant to effectively dissipate heat. Porous ceramic media play a crucial role in this cooling process by facilitating liquid transport and heat exchange; however, their intermittent capillary action and inter-grain phonon scattering significantly hinder rapid cooling. Here, we propose a strategy to create AlN-based nanofiber aerogel as a transpiration thermo-cooler, featuring vertically aligned channels and monocrystalline nanofibers by combining nanoengineering and multiscale structural assembly techniques. Benefiting from the unconstrained capillarity of aerogel channels, our thermo-coolers exhibit a fast liquid transport rate of up to 8.33 ± 0.026 mm s−1, surpassing that of state-of-the-art porous media by one to two orders of magnitude. In addition, the enhanced phonon conduction properties of single-crystal AlN nanofibers enables thermo-coolers to achieve a fast cooling rate of 156.8 °C s−1, outperforming advanced cooling materials by a factor of five and making them ideal for various thermal management applications.
Similar content being viewed by others
Introduction
Efficient thermal management, which prevents heat intrusion, is crucial in extreme thermal environments ranging from industrial and aerospace systems to nuclear plants1,2,3. Current thermal blocking techniques primarily rely on the inherent properties (i.e., insulation4,5, heat sink6, and radiation7) of materials, with their effectiveness constrained by the temperature tolerance of materials. However, with the ongoing exploration of high-altitude airspace and universe, aerodynamic heating generates temperatures over 3000 K, far beyond the limits of current materials8,9. Transpiration cooling technique utilizes porous media to deliver liquid coolant near the heated surface, where phase change occurs to efficiently dissipate heat, thereby preventing thermal failure of materials10. This technique has a theoretical maximum cooling capacity of up to 1.4 × 105 W cm−2, making it one of the most promising forms of active thermal protection11. By introducing coolant through porous media, it provides a direct heat dissipation mechanism that significantly improves cooling efficiency and reduces coolant flux, surpassing regenerative cooling, film cooling, and ablation cooling12. In this concept, porous media play a crucial role in coolant transport, while simultaneously enduring and transferring heat13. However, traditional high-temperature-resistant porous plates, composed of ceramic particle building blocks with intrinsically low solid-phase thermal conductivity (<30 W m−1 K−1), such as Al2O314, SiO215,16, and quartz17, exhibit poor fluid transport properties (<0.1 mm s−1) and restricted heat exchange capacity. These limitations result in an inefficient transpiration cooling rate (<30 °C s−1), rendering them inadequate for hypersonic thermal management even with complex pumping systems18. Additionally, the pumping systems not only increase system weight and energy consumption, but also cause faults such as delayed cooling response2,19. Hence, there is an urgent need to develop a porous material that cools rapidly and spontaneously.
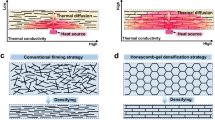
An optimal porous ceramic medium with rapid cooling performance requires two critical features: low-curvature interconnected channels, serving as fluid highways for the autonomous transport of liquid coolant with less capillary resistance (Supplementary Fig. 1), and building blocks with low-scattering phonon transport, which could exchange heat with the liquid simultaneously. Previous research has primarily focused on modifying pore diameters by regulating particle size in sintered porous ceramics, aiming to balance fluid resistance and capillary pressure20. However, the inherent intergranular pore structure, characterized by connections between particle-particle necks, results in significant capillary resistance and is prone to brittleness and structural instability21,22. Recent advancements have employed assisted hole formation (e.g., pore-forming agents23, additive manufacturing methods24, or organic templates25) to create porous media with interconnected large holes, such as SiC foam, carbon-ceramic plates, and composite materials. Despite the improved liquid transport performance of these materials, the high tortuosity of their channels still results in a low liquid transport rate, which will lead to uncontrolled phase-change migration and subsequent steam blockages. In addition, the polycrystalline building blocks of these materials exhibit inter-grain phonon scattering26,27. Phonon conduction is of paramount importance for achieving rapid heat exchange to avoid extreme situations such as heat accumulation or thermal runaway that could affect cooling stability or responsiveness28,29. Therefore, the design of an innovative porous material that possesses both ultrafast liquid transport and highly efficient heat exchange, if not impossible, remains a significant challenge.
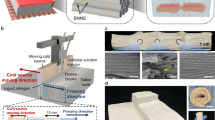
Herein, we present a nanoengineering and multiscale structural assembly strategy to develop a porous medium with a vertically aligned microchannel structure that is composed of monocrystalline aluminum nitride (AlN) nanofibers, which we term the ‘AlN-based fiber aerogel thermo-cooler’ (AFC). The advantage of the innovative design (Fig. 1) is that (ⅰ) high-purity single-crystal AlN nanofibers exhibit enhanced phonon conduction, achieving an intrinsic thermal conductivity of up to 120.0 ± 2.83 W m−1 K−1, which considerably exceeds that of current ceramics, thereby significantly enhancing heat exchange performance; (ⅱ) vertical channels with no tortuosity could promote unconstrained liquid capillarity, achieving a rapid liquid transport rate of 8.33 ± 0.026 mm s−1, which exceeds that of state-of-the-art porous media by one to two orders of magnitude; (ⅲ) nanofibers interwoven along the channel walls could form thousands of pores to provide an enlarged heat exchange area and multiple vapor pathways. Benefiting from the above-mentioned integrated coupling effect, the resulting AFC achieves a high cooling rate exceeding 156.8 °C s−1, which is five times higher than that of advanced cooling materials. In addition, our AFCs are lightweight (0.01 g cm−3) and flexible, exhibit a fast cooling response (within 1 s) and high cooling efficiency (96.7%), and are capable of sustained cooling without steam blockages or temperature oscillations, rendering them optimal for a wide range of thermal cooling applications. The design strategy of aerogel thermo-coolers may offer a transformative solution for sustainable extreme thermal protection.
Schemic of the monocrystalline sequence and multiscale structural assembly strategy applied for developing AFC with vertical channels composed of AlN nanofibers (AlN NF, teal), and its thermal protection mechanisms.
Results
Synthesis of monocrystalline AlN nanofibers
AlN nanofiber (AlN NF), a high thermal conductivity building block, was integrated on the basis of three primary criteria: (i) a monocrystalline structure with a highly ordered atomic arrangement and lower defect density to promote enhanced phonon conduction with minimal scattering; (ii) an extensive specific surface area, achieved through nanoscale diameters, to enhance contact with the coolant and increase the thermal exchange capacity; and (iii) a high aspect ratio, with ultralong fiber lengths (in the micron/centimeter range), supporting the assembly of stable 3D bulk aerogels. The first criterion was met through a facile chemical vapor deposition (CVD) process in which a high-purity aluminum source was used for direct nitridation to initiate AlN nucleation and growth. This source minimizes lattice and surface defects caused by aluminum-based impurities, oxygen, and byproducts. To achieve two other criteria, particularly the formation of ultralong AlN nanofiber clusters, a tandem catalyst nanoengineering strategy was constructed. This involved the use of chloride additives, which gasify and act as vapor transport agents at low temperatures, and metal mineralizers, which form droplets to dissolve vapor at high temperatures, enabling precise control over AlN nucleation and growth. The synthesis pathway, illustrated in Fig. 2a, began with the homogenization of a tandem catalyst mixture (AlCl3, NH4Cl, and Fe) and high-purity aluminum particles as starting materials for producing AlN NFs. As the temperature increased, AlCl3 and NH4Cl sequentially gasified and decomposed, generating NH3, HCl, and AlCl3 gases (Eq. (1))30. The generation of gas-induced porosity within the liquid aluminum inhibits droplet aggregation while facilitating the release of aluminum vapor (Supplementary Fig. 2a–c). Furthermore, HCl gas promotes spontaneous chlorination of liquid aluminum to form the intermediate AlClx (x = 1, 2, 3), which can interconvert, substantially enhancing the continuous release of aluminum vapor from the liquid phase (Supplementary Fig. 2d)31.
a Schematic illustration of the preparation process for the AlN NFs using a tandem catalyst nanoengineering strategy involving vapor–solid (VS) and vapor–liquid–solid (VLS) mechanisms. b Characterizations of the AlN NFs using scanning electron microscopy (SEM, upper left, scale bar = 20 μm), scanning transmission electron microscopy (STEM, upper right, scale bar = 300 nm), and energy-dispersive X-ray spectroscopy (EDS, bottom, scale bar = 300 nm). c Representative high-resolution transmission electron microscopy (HRTEM) images showing the monocrystalline structure of AlN NFs (scale bar = 5 nm). Inset: the lattice spacing (d) of the AlN NF crystallographic plane. d A single hexagonal wurtzite AlN NF exhibiting flexibility, bending under considerable strain while maintaining structural integrity (scale bar = 10 μm).
The formation process of AlN NFs involves three stages (illustrated by steps 1–3 in Fig. 2a): (1) supersaturated precipitation at nucleation sites, (2) primary growth, and (3) secondary growth of AlN NFs. Initially, Al and AlClx gases reached the substrate surface and reacted with NH3 gas (gasified from NH4Cl), which is the sole nitrogen source, to form AlN NF nuclei under supersaturated conditions (Eq. (2))32. This controlled nitridation prevents excessive nitriding of aluminum at low temperatures, which otherwise leads to AlN NFs appearing only as byproducts (Supplementary Fig. 2e). Subsequently, the continuous supply of gaseous Al, AlClₓ, and NH₃ facilitated the elongation of the AlN NFs by delivering aluminum to the nucleation sites at the fiber tips. This primary growth followed a vapor–solid (VS) mechanism, as indicated by the absence of droplets observed at the nanofiber tips (Supplementary Fig. 3)33. To facilitate further axial growth of the fibers, nitrogen gas was introduced, followed by an increase in temperature to promote Fe-assisted nucleation of AlN. During this process, Fe or FeCl3 alloy droplets established a liquid–solid interface at the tips of fibers, promoting Al–N dissolution in the droplets34. This prevents the formation of poor crystal morphology (e.g., tandems, deviations, and dendrites), which are caused by the uncontrolled, random growth of the crystal nuclei, thereby reducing phonon scattering. The continuous supply of nitrogen gas caused rapid enrichment of Al–N within the alloy droplets, further promoting nucleation and secondary growth at the fiber tips (Eq. (3)). This resulted in the formation of ultralong AlN NFs, with lengths reaching ~2 cm (Supplementary Fig. 4). Some nanoparticles observed at the tips of the AlN NFs (Supplementary Fig. 5a) provided evidence of the vapor–liquid–solid (VLS) process contributing to the secondary growth of the AlN NFs35. Furthermore, ultrasonication treatment completely removed these nanoparticles, suggesting that the catalysts did not integrate into the lattice and cause defects (Supplementary Fig. 5b). The specific reaction process and mechanism are detailed in the Supplementary Discussion and Supplementary equations (1)–(15).
Owing to the controlled growth process in our methodology, high yields of AlN NFs were readily achieved (Supplementary Fig. 6). SEM analysis revealed that the AlN NFs had a uniform diameter of ~418 nm, with a hexagonal wurtzite structure and a smooth surface without obvious defects or cracks (Fig. 2b, top and Supplementary Fig. 7), which are prerequisites for excellent structural continuity and flexibility. EDS elemental mapping (Fig. 2b, bottom) confirmed the high purity of the AlN NFs by revealing a homogeneous distribution of Al and N. X-ray photoelectron spectroscopy (XPS) further confirmed this finding, showing that the binding energies of N 1s and Al 2p were consistent with the reference values (Supplementary Discussion and Supplementary Fig. 8)36. HRTEM revealed that the monocrystalline nature of the AlN NFs, featuring a continuous crystallization zone virtually without defects and uniform growth in one direction (Fig. 2c). Additionally, the lattice spacing of 0.27 nm matches (001) crystallographic plane of hexagonal AlN, confirming that AlN NFs extend along the [001] crystal direction, which is orthogonal to this plane37. This resulted in the formation of ultralong AlN NFs with high aspect ratios, consistent with the prominent (100) plane diffraction peak observed in X-ray diffraction (XRD) analysis (Supplementary Discussion, Supplementary Figs. 9 and 10). Furthermore, the oxygen content of the AlN NFs is 0.387%, which is lower than the previously reported values for AlN fibers (over 0.5%)38. All results confirmed that the AlN NFs exhibited a highly ordered atomic arrangement and low defect density, which facilitated enhanced phonon conduction with minimal scattering39,40. Thus, AlN NFs have an inherently high thermal conductivity of over 120.0 ± 2.83 W m−1 K−1, as tested via the 3ω method (Supplementary Fig. 11 and Supplementary Note 2), which significantly surpasses those reported for existing ceramic fibers38. Furthermore, the flexibility of an individual monocrystalline hexagonal AlN NF was evaluated using focused ion beam (FIB)-SEM in situ bending tests. The results revealed that the nanofibers exhibited significant resilience, bending under substantial strain without fracturing (Fig. 2d). In summary, the AlN NFs obtained through tandem catalyst reactions feature an elongated structure, extensive surface area, exceptional monocrystalline purity, and remarkable flexibility, making them ideal candidates as building blocks in cooling materials. By using these AlN NFs, thermal exchange with coolants can be improved via an enlarged contact area and enhanced phonon conduction in single nanofibers.
Fabrication and vertical channel structures of the AFCs
To design a spontaneous cooling device with superior performance, characterized by both rapid cooling response and sustained cooling, the aforementioned monocrystalline AlN NFs must be assembled into a 3D structure that enables rapid capillary flow and unobstructed evaporation. Considering these principles, the AFCs were developed and fabricated based on three fundamental criteria: (i) the nanofibers should have an interconnected cellular architecture with vertically aligned channels to ensure unconstrained capillary transport of coolant; (ii) the cellular walls of the capillary channels should feature pores to provide multiple pathways for steam escape; and (iii) the AFCs should be mechanically robust to guarantee long-term cooling efficiency. The first two criteria were achieved by assembling flexible AlN NFs into 3D AlN-based nanofiber aerogels (ANFAs) using a directional freeze-shaping method. To meet the last requirement, soluble phosphate Al(H2PO4)3 (AHP) was introduced, forming crosslinked 3D fibrous networks that impart long-term stability41. Figure 3a illustrates the multiscale structural assembly pathway for AFCs, including dispersion, freezing, freeze-drying, and calcination. The process began with the dispersal of nanofibers in an aqueous polyacrylamide (PAM) solution containing AHP to create a homogeneous nanofiber dispersion. The mixture was then frozen in a specific mold, degassed in a vacuum oven, and directionally freeze-dried to produce unbonded ANFAs. To strengthen the inter-fiber connections, the unbonded ANFAs underwent calcination at 800 °C for 30 min. This process induced the in-situ formation of robust aluminum phosphate bonds, forming a cohesive network (Al–O–P–O–Al) across the nanofibers, which significantly enhanced the structural stability of the ANFAs42. Throughout the assembly of ANFAs, the AlN NFs exhibited no detectable hydrolysis or oxidation, retaining their inherent structural integrity and excellent performance (Supplementary Fig. 12).
a Fabrication procedure of the AlN-based nanofiber (NF) aerogels (ANFAs). b Schematic diagram of the fibrous frame reconstruction mechanism. Ice crystals grow in the direction of the arrow and expel NFs in all directions, resulting in a higher concentration of NFs around the ice crystals (Ct, red) than that in the solution (C0, blue). c–e Transverse and longitudinal sectional SEM images of ANFAs at various magnifications. f and g Digital photographs demonstrating the ultralight nature and superhydrophilicity of ANFAs. h Optical images illustrating the flexibility of ANFAs, showcasing their ability to be rolled up or twisted without fracturing. i Compressive resilience of ANFAs demonstrated in water and under a butane blowtorch flame, showing no visible damage. Scale bars: c 300 μm (inset: 5 mm); d 40 μm; e 50 μm (inset: 5 mm); f 5 mm; g 500 μm (inset: 5 mm); h 1 cm; i 5 mm (upper) and 2 cm (bottom).
The formation of vertically aligned channel structures was directly influenced by the freezing process, prompting an investigation into the mechanism behind the 3D reconstruction of fibrous channels (Fig. 3b)43. During directional freezing, the water within the dispersion solution gradually solidified into an ice crystal template that grew upward from the bottom44. This process was driven by the significant contrast in thermal conductivity between the bottom cold plate and the lateral sides (Supplementary Figs. 13, 14 and Supplementary Table 1). During the phase change of water, the volume exclusion effect expels the nanofibers in the dispersions from the advancing solidification front, resulting in their accumulation at ice crystal boundaries45. This resulted in an increased local concentration of fibers around the vertical ice crystals, forming vertical channels with fiber network walls that contained pores, as observed in both the transverse and longitudinal sections in the SEM images of ANFAs (Fig. 3c–e). Furthermore, by controlling the freezing temperature of the nanofibrous dispersion from −20 to −196 °C, aerogels with varying diameters of vertical channels ranging from 99.49 ± 11.50 to 30.52 ± 5.47 μm were produced, referred to as VAC-1, VAC-2, and VAC-3, respectively (Supplementary Figs. 15, 16).
Benefiting from the simplicity of our assembly process, ANFAs could be shaped into a wide variety of forms, including hexagons or intricate designs, demonstrating their adaptability for constructing irregular and customized components (Supplementary Fig. 17). In dramatic contrast to conventional porous ceramics, ANFAs exhibited ultralight properties, featuring an extraordinary density of just 0.01 g cm−3, light enough to stand on the stamens of a flower (Fig. 3f, Supplementary Note 3 and Supplementary Fig. 18). This achievement represented significant progress toward the “double reduction” goal of minimizing weight wherever possible. Furthermore, the wettability of the ANFAs was assessed via contact angle testing and image capture with a high-speed camera. The results showed that the water contact angle was 0°, and that the droplets infiltrated ANFAs within 55 ms (Fig. 3g and Supplementary Fig. 19). This finding highlighted their superhydrophilic nature, which is beneficial for liquid absorption. Furthermore, AlPO4 served as both a protective layer, preventing AlN NFs from undergoing hydrolysis and oxidation, and a “nanoglue” that promoted the formation of crosslinked networks in ANFAs, thereby significantly enhancing their structural integrity and flexibility (Supplementary Fig. 20)41. As a result, the as-fabricated ANFAs exhibited remarkable bendability, allowing them to be easily rolled and twisted without fracturing (Fig. 3h). This flexibility was in stark contrast to the rigidity typically found in conventional porous media used in transpiration cooling applications. Meanwhile, as shown in Fig. 3i, the ANFAs demonstrated exceptional resilience, supported by rigid silica nanofibers, showing no signs of ignition or structural collapse when subjected to 30% compression underwater or high-temperature flames (~1100 °C). In addition, the ANFAs retained over 85% of their structural integrity following rigorous fatigue testing, which included 1000 bending cycles, 700 buckling cycles, and 1000 compression cycles, demonstrating exceptional structural robustness (Supplementary Fig. 21).
Rapid capillary flow and thermal stability
Unlike conventional porous media with narrow, high-tortuosity pores, vertical channels produced a stronger capillary effect, leading to faster liquid transport and greater elevation (Fig. 4a and Supplementary Fig. 22)46. As shown in Fig. 4b, AFCs with vertically aligned channels (VAC-x) absorbed liquid up to 3 cm (limited by their height) within 6 s, 3 s faster than the random porous (RP) structure. In contrast, the commercial felt material (CM) only reached a liquid absorption height of 2.3 cm. We hypothesized that the vertically aligned channels with low tortuosity were crucial to the exceptional capillary flow in AFCs, enabling a sustained liquid supply at extreme temperatures without steam intrusion. Thereby, the absorption data were subsequently fitted based on the Lucas–Washburn equation (Supplementary Note 4 and Supplementary Table 2)47. The calculated fit parameters were approximately 0.48 for vertical channels, aligning closely with the theoretical capillary structural value of 0.5. In contrast, random porous structures presented lower values (0.362 and 0.171), indicating the adverse influence of high tortuosity on absorption (Supplementary Discussion). To further elucidate the critical role of aligned channels in liquid transport, the numerical simulations of relative water pressure and flow speed were conducted (Supplementary Note 5 and Supplementary Fig. 23). VAC-1, which has the widest channels, exhibited the smallest negative relative water pressure. This indicates that VAC-1 has the strongest capillary pumping ability, resulting in faster continuous fluid flow to the upper surface. These results align with experimental findings showing that VAC-1 achieved the highest average absorption speed of 8.33 ± 0.026 mm s−1 and rate of 0.25 ± 0.001 mL cm−3 s−1, which are twice those of RP (Supplementary Fig. 24). In addition, for the aligned channels, the theoretical maximum absorption height exceeded 30 cm (Supplementary Table 3), ensuring complete liquid infiltration48. Aligned channels and excellent wettability enabled rapid penetration and distribution through the porous structures, despite slower speeds with more viscous liquids (Supplementary Figs. 25 and 26). VAC-1 maintained superior absorption across various liquids, outperforming RP with an enhancement ratio above 2 (Fig. 4c). Overall, vertically aligned channels combined with a superhydrophilic nanofiber network significantly enhanced spontaneous absorption by reducing tortuosity, outperforming other materials (Fig. 4d)14,16,24,46,49.
a Effect of tortuosity on the absorption capacity of AFCs with vertical channels and random pores. b Absorbed liquid height over time at a temperature of 25 ± 2 °C and a humidity of 45 ± 2%. The tested samples include vertically aligned channels with different channel widths (VAC-1, VAC-2, VAC-3), a random porous (RP), and a commercial felt material (CM). Each data point represents the mean value derived from three replicates, and the error bars indicate the standard deviation. c Comparison of the average absorption speed for vertical channels (vv) and random pores (vr), and the speed enhancement ratio (vv/vr) for liquids with varying viscosities (liquid viscosity: ηL, air viscosity: ηA). d Comparison of capillary performance between AFCs and conventional materials14,16,24,46,49. e Heatmap of the temperature distribution at different heights of the AFC and capillary flow curves over absorption time, under heating by a butane spray gun (~ 1100 °C). f Capillary transport mechanisms of the AFC under extreme temperatures. Rn, θn, and ∆pn represent the meniscus curvature radius, contact angle, and capillary pressure difference at different stages (n = 1, 2, 3), respectively, and r represents the channel radius. g Rapid capillary action responsiveness in AFCs under fluctuating temperatures. h Cooling power and capacity factor of AFCs at 1100 °C. i Cycling cooling power at 1100 °C, showing liquid mass changes in the 1st and 10th cycles. Source data are provided as a Source Data file.
Sustained liquid capillary flow is crucial for maintaining stable transpiration cooling. To evaluate the performance of AFCs under extreme temperatures (~1100 °C), we analyzed the behavior of liquid–vapor interfaces to investigate capillary flow dynamics, revealing three distinct stages (Fig. 4e, f and Supplementary Fig. 27). Prior to liquid absorption (t = 0 s), the AFCs exhibited a pronounced vertical temperature gradient: the upper surface (h = 3 cm) reached nearly 930 °C, while the lower surface remained at approximately 50 °C. During stage Ⅰ (h ≤ 1 cm), the liquid at the cooler base (<100 °C) was rapidly transported without evaporation, achieving an ultrafast absorption speed of 15.45 mm s−1, which surpasses that of reported porous media by two orders of magnitude, similar to that observed at room temperature. As the liquid front advanced to hotter regions (>100 °C, stage Ⅱ), phase change occurred, forming a dynamic liquid–gas interface. This vaporization reduced the contact angle and meniscus curvature radius, amplifying the capillary pressure difference24. Simultaneously, the localized cooling from the phase change established a thermal gradient that triggered Marangoni convection, promoting the fluid flow toward hotter regions with lower surface tension50. This self-regulating mechanism ensures the progressive cooling of the upper surface as the liquid advances, thereby maintaining an appropriate liquid-vapor interface. By t = 4 s, the liquid had reached the upper surface, cooling it to approximately 100 °C, while a protective liquid film formed to isolate the surface from the heat source. Importantly, this film maintained a stable evaporation-replenishment equilibrium (stage Ⅲ), sustaining uninterrupted capillary flow for up to 5 min without absorption delays or steam blockages. These results demonstrate the thermal stability and sustainability of spontaneous capillary flow in AFCs.
The response time of AFCs under alternating thermal conditions was subsequently evaluated to investigate the dynamic stability of capillary flow by measuring surface temperatures and liquid consumption. Figure 4g illustrated that the surface temperature rose slightly as the heat source rapidly increased from 350 to 1100 °C, but it quickly decreased and stabilized due to the automatic adjustment of coolant consumption. The system responded to thermal changes under 1 s, demonstrating its ability to handle extreme temperature fluctuations effectively. To confirm that consumption adjustment was driven by capillary flow in the vertical channels, the cooling power was calculated on the basis of the evaporation rate51:
where qcool, J, hlv denote the cooling power (W m−2), evaporation rate (kg m−2 h−1), and enthalpy of evaporation of pure water (kJ kg−1), respectively. The evaporation rate of AFCs increased from 1.60 to 385.41 kg m−2 h−1 across an elevated temperature range of 50–1100 °C, whereas that of pure water increased from only 0.24 to 89.54 kg m−2 h−1 (Supplementary Fig. 28). This superior performance was attributed to the vertically aligned channels and porous walls of the AFCs. This ensured an ultrafast response and dynamic stability in capillary flow and enhanced heat transfer by increasing surface contact, promoting faster evaporation. At 1100 °C, the AFCs demonstrated an evaporation rate of nearly 500 kg m−2 h−1 and a cooling power of 1.34 MW m−2 within 15 min. This achievement is ~5 times greater than the cooling capacity of pure water and surpasses that of recent spontaneous transpiration cooling systems (Fig. 4h and Supplementary Fig. 29). Additionally, Fig. 4i illustrates the long-term stability of AFCs at 1100 °C, demonstrating that the cooling power remains at ~1.4 MW m−2 over 10 cycles. This durability was due to the stable capillary action maintained by the vertical channels to ensure efficient cooling without steam blockage, which withstood multiple heating cycles without discernible degradation or damage (Supplementary Fig. 30).
Transpiration cooling properties of AFCs
The extreme thermal management performance of the AFCs was assessed using tailored experimental devices (Fig. 5a), with water chosen as the coolant owing to its highest enthalpy of vaporization among all room-temperature liquids51. We applied a butane flame (~1100 °C) to an AFC sample that was 3 cm thick for 9 min. In the first 5 min without a water supply (Fig. 5b, top and Fig. 5c, left), passive thermal protection was observed, with the bottom of the AFC stabilizing at 130 °C, while the top surface reached ~930 °C. Upon water introduction, active thermal protection was initiated, rapidly reducing the top surface temperature to 297.9 °C within 4 s, achieving an ultrafast cooling rate of 156.8 °C s−1, which is highly advantageous for managing extreme environments (Fig. 5b, bottom and Fig. 5c, right). The inset of Fig. 5c demonstrated that the top surface temperature began to decrease within 1 s of water introduction, highlighting the instantaneous responsiveness of capillary cooling. Continuous cooling followed, with the surface temperature stabilizing at ~95 °C for ~4 min. This behavior suggests sustained capillary flow and a stable phase-change mechanism within the AFCs.
a Schematic and optical photographs of the testing apparatus for evaluating the extreme thermal management performance of AFCs. b, c Thermographic images and temperature variation curves over time under extreme temperature conditions, with and without water. The inset represents the immediate cooling response and fast cooling. d Surface temperature and cooling efficiency at different measurement points (x/L) on AFCs with a water supply, where x/L ranges from 0 at the top to 1 at the bottom (x: distance from top, L: total height). e Stability of the circulating cooling performance of AFCs, demonstrated through cyclic testing over 10 cycles. Thermal cooling simulations of f AFC and g conventional porous medium with random pores (CPM), including temperature and coolant liquid saturation (ranging from 0 of vapor phase to 1 of liquid phase to describe vapor–liquid phase distribution). h Schematic illustration of the factors contributing to the capillary cooling effect in AFCs. i Comprehensive performance comparison of AFCs with other porous media and cooling systems1,25,54,56. Source data are provided as a Source Data file.
The cooling performance of the AFCs was further evaluated by measuring their cooling efficiency (ηcool), which is defined as follows52:
Here, T∞, Tw, and Tc represent the spray gun nozzle temperature, the AFC test point temperature, and the coolant temperature, respectively. Figure 5d illustrates the temperature and cooling efficiency profiles at various measuring points on the AFC surfaces after 1 min of cooling, when exposed to an approximate temperature of 1100 °C. On the top surface of the AFC, the cooling efficiency reached 94.2%, gradually increasing along the surface to 96.7% and 97.3% in the upstream (x/L = 0.5) and downstream (x/L = 1) regions, respectively. As the AFC was exposed to extreme temperatures, the surface temperature steadily increased during the test. By the end of the test, the cooling efficiency in the top and upstream regions remained high, at 94.0% and 95.8%, respectively (Supplementary Fig. 31), indicating excellent long-time, sustained cooling performance of AFC. Importantly, the AFC remained fully intact without discernible degradation or structural damage throughout the testing period (Supplementary Fig. 32). These results demonstrated that the capillary cooling scheme effectively mitigated issues such as local overheating, uneven coolant distribution on the heated surface, and temperature oscillation. Additionally, when the water supply was reduced, the top surface temperature still reached 111.2 °C, with a cooling efficiency of 92.3% after 1 min of cooling (Supplementary Fig. 33). This meets the requirements for practical applications in hypersonic aircraft. To assess the cyclic capillary cooling performance, 5 mL of water was supplied during each cycle, and the results are shown in Fig. 5e. During the tenth cycle, the AFC retained robust cooling performance, achieving a response time of 1 s, a peak cooling rate of 158.1 °C s−1, and a cooling efficiency of 92.2%. This performance is comparable to that of the first cycle, which stems from the exceptional structural and chemical stability of the AFC (Supplementary Fig. 34). Notably, the bottom temperature of the AFCs remained below 95 °C throughout the testing period, even under water supply interruption, thanks to its passive protection mechanism. (Supplementary Discussion and Supplementary Fig. 35)53. This mechanism mitigates the risk of catastrophic internal thermal damage to internal components in the event of a failure in the capillary cooling system. Furthermore, the AFC demonstrated long-term stability, retaining its superior cooling functionality for 12 h after a storage period exceeding 6 months (Supplementary Fig. 36). These results suggest that the service life is theoretically permanent and is limited only by coolant depletion.
To elucidate the heat loss mechanism in the thermo-cooler, structural models and thermal distribution of the AFC and a conventional porous medium with random pores (CPM) were constructed and simulated (Supplementary Note 6). As shown in Fig. 5f and g, the AFC demonstrated superior cooling performance to the traditional porous medium, achieving a lower temperature, faster liquid flow speed, and more efficient vapor diffusion (Supplementary Fig. 37). These results stem from multiscale heat loss mechanisms (Fig. 5h). At the macroscale, the vertically aligned channels accelerate capillary-driven fluid flow, resulting in the rapid and consistent formation of a continuous liquid film at the heating interface (Supplementary Fig. 38). This liquid film establishes a stable evaporation-replenishment equilibrium with nearly zero response delay. It effectively impedes heat penetration and prevents the temperature oscillations common in conventional media54. At the microscale, the nanofiber-interwoven channel walls contain numerous pores that enhance multipath diffusion for vapor generated by coolant evaporation. This structure prevents the liquid‒gas boundary from shifting downward due to the steam-blocking effect, ensuring reliable cooling performance55. At the nanoscale, heat exchange between nanofibers and coolant is significantly enhanced by nanoscale surface effects. Simultaneously, heat dissipates rapidly along monocrystalline fibers due to minimal lattice defect scattering and weak phonon anharmonicity, which enhances heat transfer to further optimize transpiration cooling. Together, these multiscale mechanisms enable the AFC to cool at a rate over seven times faster than commercial porous media (Supplementary Fig. 39) and 57% faster than silica nanofiber aerogel (SNA) with similar structural characteristics (Supplementary Fig. 40).
In summary, capillary transpiration cooling provided by AFCs effectively shielded the heated surface; mitigated challenges such as cooling delay, steam blockage, and unstable phase transitions; and minimized local overheating and uneven coolant distribution. Not only did the comprehensive capillary transpiration cooling performance of the AFCs exceed that of other cooling schemes, but they also offered flexibility and ultra-lightweight properties, with a density of just 0.01 g cm−3, compared to conventional cooling systems exceeding 0.3 g cm−3 (Fig. 5i and Supplementary Table 4)1,25,54,56. These attributes highlight the great potential of AFCs for application in extreme environments, particularly in the aerospace field, such as hypersonic deformable aircraft.
Discussion
Our innovative AlN-based nanofiber aerogel thermo-coolers address the perennial challenge of utilizing granular porous materials as cooling media in extreme thermal environments. This breakthrough was achieved through the independent optimization of heat transfer and liquid transport within aerogel, leveraging nanoengineering and multiscale structural assembly strategies. Specifically, the AFCs feature vertically aligned capillary channels composed of monocrystalline AlN nanofibers, enabling unconstrained liquid transport alongside enhanced phonon conduction. The coupled integration of efficient thermal exchange, unhindered liquid transport, and unrestricted vapor diffusion imparts AFCs with exceptional durability, outperforming existing thermal cooling systems and making them ideal for protection in extreme thermal environments. Alongside their excellent thermal cooling performance, AFCs are lightweight and flexible, redefining the possibilities for lightweight, sustained cooling protection materials and pioneering a new era of high-performance thermal management systems. Beyond this proof-of-concept, our study introduces a pioneering tactic to design customizable porous cooling materials, which are required for advanced aerospace applications such as cross-domain vehicles and hypersonic weapons.
Methods
Materials
Aluminum granules (Al, 99.999%, Zkynxc. Co., Ltd, China), Anhydrous aluminum chloride (AlCl3, ≥99.5%, Sinopharm Chemical Reagent Co., Ltd., China), Ammonium chloride (NH4Cl, ≥99.5%, Sinopharm Chemical Reagent Co., Ltd., China), Iron (Fe, 99.9%, Zkynxc. Co., Ltd, China), Polyacrylamide (PAM, Mw ~ 300,000, Aladdin, China), Aluminum dihydrogen phosphate (Al(H2PO4)3, Mw ~ 317.94, Henan Yulin Chemical Co., Ltd., China), silica nanofibers (SiO2 NFs, Supplementary Note 1), argon gas (Ar, 99.999%, Shanghai Wendong Chemical Co., Ltd., China), nitrogen gas (N2, 99.999%, Shanghai Wendong Chemical Co., Ltd., China).
Synthesis of monocrystalline AlN NFs
Al was chosen as the source, while AlCl3, NH4Cl, and Fe served as a tandem catalyst. The mixed powder was prepared by combining Al, AlCl3, NH4Cl, and Fe in a ratio of 6:3:4:10, then it was uniformly blended and transferred to a corundum crucible placed in a furnace. Initially, the furnace was filled with argon gas and then brought to a temperature of 1100 °C, which was maintained for 1 h. Subsequently, the temperature was raised to 1650 °C for half an hour, with the argon replaced by nitrogen gas, and then maintained for 5 h to complete the growth of AlN NFs. Finally, the AlN NFs were cleaned to remove surface impurities.
Fabrication of ANFAs
PAM used as a dispersant, Al(H2PO4)3 served as a binding agent, and SiO2 NFs provided rebound support without affecting other properties. To prepare a 0.01 g cm−3 ANFAs, a mixture of 1 g of AlN NFs and 99 g of a PAM solution (0.1 g in 100 g deionized water) was prepared by ultrasonic dispersion and stirring for 30 min. Then, 1 g of Al(H2PO4)3 solution and 0.1 g of SiO2 NFs were added and stirred for another 30 min. The resulting mixed dispersion was transferred to a custom mold and directionally frozen at various low temperatures (−20, −50, and −196 °C) until complete solidification. It was then freeze–dried for 48 hours, obtaining unbonded ANFAs. Subsequently, they were treated at 100 °C for 24 h, followed by calcination at 800 °C under flowing air for 30 min. Unless otherwise noted, all structural and property investigations were conducted using ANFAs with a density of 0.01 g cm−3.
Characterization
Macroscopic morphology was captured with a digital camera (Canon M50). Microstructural morphology was observed using FE-SEM (S-4800) and TEM (JEM-2100). Chemical composition and positional analysis were performed using EDS (Bruker Nano GmbH 610M) and XPS (Es-calab 250 Xi). The crystalline structure was examined via XRD (Bruker D8 Advance). An oxygen–nitrogen–hydrogen analyzer (EMGA-830) was used to test the oxygen content. A 3ω harmonic thermal property testing system (THSCB-01) was used to measure the thermal conductivity of each individual fiber (Supplementary Note 2). The flexibility of the nanofibers was assessed using FIB-SEM (Crossbeam340). Mechanical properties were tested by a DMA instrument (TA-Q850). The hot disk instrument (TPS2500S) and the laser flash apparatus (Netzsch LFA467) were employed to measure the effective thermal conductivity of the bulk material. Hydrophilicity was determined using a goniometer (Kino SL200B), a high-speed camera (Casio EX-F1), and a CCD industrial electron microscope (GP-L200). The capillary flow process was taped using a video camera (Sony CX405). Viscosity was determined with a rotational rheometer (HAAKE RS6000). Real-time temperature changes on the sample surfaces and freezing process were monitored using an infrared thermal imager (Fluke TiX560) and temperature collectors (MT500P).
Extreme thermal management tests
Surface temperature measurements were performed to evaluate the practical performance of AFCs under extreme thermal conditions. The experimental AFCs were positioned inside an alumina crucible and subjected to extreme temperatures, reaching ~1100 °C, using a butane spray gun. A fluid dispensing system supplied and controlled the coolant, which was delivered into the crucible via a syringe connected to a tube. Temperature probes were strategically placed at various measurement points on the AFCs and the spray gun nozzle for real-time monitoring. The response time for capillary cooling was calculated by measuring the time difference between the onset of top surface cooling and the water delivery, indicated by a sudden drop in the bottom temperature. Surface temperature distribution was captured via infrared thermography.
Data availability
The source data generated in this study are provided in the Source Data file. Source Data are provided with this paper. All the raw data relevant to the study are available from the corresponding author upon request. Source data are provided with this paper.
References
Jiang, M. et al. Inhibiting the Leidenfrost effect above 1000 °C for sustained thermal cooling. Nature 601, 568–572 (2022).
Goldstein, E. A., Raman, A. P. & Fan, S. Sub-ambient non-evaporative fluid cooling with the sky. Nat. Energy 2, 17143 (2017).
Xu, X. et al. Double-negative-index ceramic aerogels for thermal superinsulation. Science 363, 723–727 (2019).
Guo, J. et al. Hypocrystalline ceramic aerogels for thermal insulation at extreme conditions. Nature 606, 909 (2022).
Wu, M. et al. Biomimetic, knittable aerogel fiber for thermal insulation textile. Science 382, 1379–1383 (2023).
Kim, S. H., Heu, C. S., Mok, J. Y., Kang, S.-W. & Kim, D. R. Enhanced thermal performance of phase change material-integrated fin-type heat sinks for high power electronics cooling. Int. J. Heat. Mass Transf. 184, 122257 (2022).
Lin, C. et al. Pushing radiative cooling technology to real applications. Adv. Mater. 37, 2409738 (2025).
Peters, A. B. et al. Materials design for hypersonics. Nat. Commun. 15, 3328 (2024).
Smith, C. R. Aerodynamic heating in hypersonic flows. Phys. Today 74, 66–67 (2021).
Wang, D., Pan, K., Liu, Y., Wang, L. & Liu, Z. Research on the cooling performance of the discontinuous transpiration surface structure for the leading edge of a hypersonic vehicle. Appl. Therm. Eng. 241, 122324 (2024).
He, F. & Wang, J. Numerical investigation on critical heat flux and coolant volume required for transpiration cooling with phase change. Energ. Convers. Manag. 80, 591–597 (2014).
Li, Y., Zhang, B. & Fan, X. Numerical simulation of the effects of double blocking regions on transpiration cooling in directional porous structures. Phys. Fluids 37, 017126 (2025).
Kong, R. et al. Enhancing data center cooling efficiency and ability: a comprehensive review of direct liquid cooling technologies. Energy 308, 132846 (2024).
Al-Sharabi, M. et al. Investigating the effect of sintering rate and solvent type on the liquid transport kinetics of α-alumina powder compacts. Chem. Eng. Sci. 284, 119414 (2024).
van Foreest, A. et al. Transpiration cooling using liquid water. J. Thermophys. Heat Transf. 23, 693–702 (2009).
Ren, Y. et al. Effects of the particle size on the microstructures and liquid absorbency of silica porous ceramics. J. Mater. Eng. Perform. 33, 2209–2218 (2024).
Zhang, P., Pan, Y., Yang, R., Manthar, A. & Zhou, X. Investigation on a CMC-aided multilayer thermal structure and the functionality of active cooling. Appl. Therm. Eng. 245, 122834 (2024).
Mi, Q., Yi, S. H., Gang, D. D., Lu, X. G. & Liu, X. L. Research progress of transpiration cooling for aircraft thermal protection. Appl. Therm. Eng. 236, 121360 (2024).
Li, X., Liao, Z., Li, H., Jiang, P. & Xu, R. Phase-change transpiration cooling in heterogeneous composite porous plates: Heat transfer characteristics and their prediction. Int. J. Heat Mass Transf. 224, 125290 (2024).
Feng, S. et al. Three-dimensional capillary ratchet-induced liquid directional steering. Science 373, 1344–1348 (2021).
Liu, Y.-Q., Jiang, P.-X., Xiong, Y.-B. & Wang, Y.-P. Experimental and numerical investigation of transpiration cooling for sintered porous flat plates. Appl. Therm. Eng. 50, 997–1007 (2013).
Zhang, X., Yu, J. & Si, Y. Programmable shape-morphing enables ceramic meta-aerogel highly stretchable for thermal protection. Adv. Mater. 37, 2412962 (2025).
Guo, X., Cai, X., Zhu, L. & Yang, H. Preparation and pore structure characteristics of SiC honeycomb ceramics with macroporous walls. Ceram. Int. 40, 6339–6343 (2014).
Xu, R. et al. Nanoengineering-enhanced capillary cooling achieves sustained thermal protection for ultra-high heat flux and temperature. Adv. Mater. 36, 2312765 (2024).
Yang, F., Xie, W. & Meng, S. Study on transpiration cooling performance of SiC porous ceramics. Appl. Therm. Eng. 229, 120542 (2023).
Sheng, P. & Zhou, M. Y. Heat-conductivity of amorphous solids-simulation results on model structures. Science 253, 539–542 (1991).
Gelin, S., Tanaka, H. & Lemaitre, A. Anomalous phonon scattering and elastic correlations in amorphous solids. Nat. Mater. 15, 1177–1181 (2016).
Fong, K. Y. et al. Phonon heat transfer across a vacuum through quantum fluctuations. Nature 576, 243–247 (2019).
Zhang, Y., Lei, C., Wu, K. & Fu, Q. Fully organic bulk polymer with metallic thermal conductivity and tunable thermal pathways. Adv. Sci. 8, 2004821 (2021).
Zheng, M., Zhu, S., Jia, Q., Jia, G. & Liu, X. Synthesis and growth mechanism of aluminum nitride nanowires via a chloride-assisted chemical vapor reaction method. Ceram. Int. 45, 4520–4525 (2019).
Zheng, M., Jia, Q., Zhu, S. & Liu, X. Large scale synthesis and photoluminescent property of ultra-long AlN nanowires via a NH4Cl assisted chemical vapor reaction method. Ceram. Int. 44, 7267–7272 (2018).
Li, W., Ye, L., Chen, F., Qiu, W. & Zhao, T. Effect of nitriding atmosphere on the morphology of AlN nanofibers from solution blow spinning. Ceram. Int. 47, 706–715 (2021).
Jiang, G. J. et al. Morphologies and growth mechanisms of aluminum nitride whiskers by SHS method—Part 2. J. Mater. Sci. 35, 63–69 (2000).
Matsumoto, M. et al. Morphology of AlN whiskers grown by reacting N2 gas and Al vapor. J. Cryst. Growth 468, 576–580 (2017).
Wan, S. et al. Hot-pressing induced alignment of AlN whiskers in polymer matrix leading to enhanced in-plane thermal conductivity. Ceram. Int. 49, 35094–35103 (2023).
Motamedi, P. & Cadien, K. XPS analysis of AlN thin films deposited by plasma enhanced atomic layer deposition. Appl. Surf. Sci. 315, 104–109 (2014).
Hao, X. et al. Carbothermal synthesis of high-aspect-ratio AlN whiskers using graphite felt as carbon source. Ceram. Int. 48, 9842–9847 (2022).
Bolt, J. D. & Tebbe, F. N. Aluminum nitride fibers from a thermoplastics organoaluminum precursor. Mater. Res. Soc. Symp. Proc. 108, 337–344 (1988).
Koh, Y. R. et al. Bulk-like intrinsic phonon thermal conductivity of micrometer-thick AlN films. ACS Appl. Mater. Interfaces 12, 29443–29450 (2020).
Zhang, W. et al. Atomic layer deposited high quality AlN thin films for efficient thermal management. J. Mater. Chem. A 11, 21846–21856 (2023).
Zhang, X. et al. Ultrastrong, superelastic, and lamellar multiarch structured ZrO2−Al2O3 nanofibrous aerogels with high-temperature resistance over 1300 °C. ACS Nano 14, 15616–15625 (2020).
Gao, B., Sun, X., Yao, C. & Mao, L. A new strategy to chemically transform waste PET plastic into aerogel with high fire resistance and mechanical strength. Polymer 254, 125074 (2022).
Hua, M. et al. Strong tough hydrogels via the synergy of freeze-casting and salting out. Nature 590, 594–599 (2021).
Liu, H. et al. Building-envelope-inspired, thermomechanically robust all-fiber ceramic meta-aerogel for temperature-controlled dominant infrared camouflage. Adv. Mater. 36, 2313720 (2024).
Jiang, T. et al. Superporous sponge prepared by secondary network compaction with enhanced permeability and mechanical properties for non-compressible hemostasis in pigs. Nat. Commun. 15, 5460 (2024).
Cui, Y. et al. Smart sponge for fast liquid absorption and thermal responsive self-squeezing. Adv. Mater. 32, 1908249 (2020).
Cai, J. et al. Lucas−Washburn equation-based modeling of capillary-driven flow in porous systems. Langmuir 37, 1623–1636 (2021).
Tian, L. et al. Tailoring centripetal metamaterial with superelasticity and negative Poisson’s ratio for organic solvents adsorption. Sci. Adv. 8, eabo1014 (2022).
Du, X. et al. Microchannelled alkylated chitosan sponge to treat noncompressible hemorrhages and facilitate wound healing. Nat. Commun. 12, 4733 (2021).
Buffone, C., Sefiane, K. & Easson, W. Marangoni-driven instabilities of an evaporating liquid–vapor interface. Phys. Rev. E 71, 056302 (2005).
Li, R., Wang, W., Shi, Y., Wang, C.-T. & Wang, P. Advanced material design and engineering for water-based evaporative cooling. Adv. Mater. 36, 2209460 (2024).
Duwez, P. & Wheeler, H. L. Experimental study of cooling by injection of a fluid through a porous material. J. Aeronaut. Sci. 15, 509–521 (1948).
Xu, Z. et al. Ceramic meta-aerogel with thermal superinsulation up to 1700 °C constructed by self-crosslinked nanofibrous network via reaction electrospinning. Adv. Mater. 36, 2401299 (2024).
Luan, Y., He, F., Wang, J., Wu, Y. & Zhu, G. An experimental investigation on instability of transpiration cooling with phase change. Int. J. Therm. Sci. 156, 106498 (2020).
Chen, K., Xu, R.-N. & Jiang, P.-X. Evaporation enhancement of microscale droplet impact on micro/nanostructured surfaces. Langmuir 36, 12230–12236 (2020).
Han, X. et al. Slippery damper of an overlay for arresting and manipulating droplets on nonwetting surfaces. Nat. Commun. 12, 3154 (2021).
Acknowledgements
This work is supported by the National Natural Science Foundation of China (No. 92371110 to Y.S., 52373281 to Y.S., and 92271201 to X.L.), the Ministry of Science and Technology of China (No. 2023YFC3011705 to Y.S.), and the Fundamental Research Funds for the Central Universities (No. 2232023Y-01 to Y.S.).
Author information
Authors and Affiliations
Contributions
Y.S. and S.M. conceived and designed the study. Q.Z. and S.M. organized the data. H.W. and S.M. wrote the manuscript. X.L. and J.Y. supervised the project. All authors discussed the results and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Lei Su and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Meng, S., Wang, H., Zhao, Q. et al. AlN-based aerogel thermo-cooler enabled by enhanced phonon conduction and unconstrained liquid capillarity. Nat Commun 16, 11068 (2025). https://doi.org/10.1038/s41467-025-65983-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-65983-7