Abstract
Unresectable stage III NSCLC standard treatment is chemo-radiotherapy (CT-RT) followed by immunotherapy (IO) with durvalumab. We investigated adding an induction phase with chemo-immunotherapy (ChIO). APOLO was a multicentre, single-arm, phase 2 trial (NCT04776447). Non-resectable stage IIIA–IIIC NSCLC patients received induction ChIO (atezolizumab + carboplatin + paclitaxel, for 3 cycles), followed by concurrent CT-RT (3 cycles), and IO maintenance (atezolizumab for 16 cycles). Primary objective was 12-month progression-free survival (PFS). Secondary endpoints included 12- and 24-month overall survival (OS), ORR, first-site failure pattern, and safety. Exploratory endpoints included PD-L1, TMB, and ctDNA. 38 patients were enrolled. Median follow-up was 29.6 months (data cutoff, February 2024). PFS was 68.4% (95% CI, 51.1–80.7) at 12 months (statistically significant compared to null hypothesis of 55%) and 50.0% at 24 months; OS was 86.8% and 60.5%, respectively. After induction, 18 (47.4%) had partial response, 14 (36.8%) stable disease, and 5 (13.2%) progressive disease. Overall, 18 patients had disease progression: 8 local (44.4%) and 10 distant (55.6%). Grade ≥3 TRAEs occurred in 11 (28.9%) during induction, 10 (26.3%) during CT-RT, and 2 (5.3%) during maintenance. ChIO induction before CT-RT and IO maintenance showed activity and safety, warranting confirmation in larger studies.
Similar content being viewed by others
Introduction
At least one-third of patients with non-small cell lung cancer (NSCLC) are diagnosed with locally advanced disease (stage III)1. Stage III disease is highly heterogeneous, with ongoing debate regarding the optimal treatment approach, which may include surgery or chemoradiation in combination with neoadjuvant or adjuvant systemic therapy.
Until the approval of immune checkpoint inhibitors, the standard of care for patients with unresectable stage III NSCLC and good performance status was platinum-based doublet chemotherapy with concurrent radiotherapy (chemo-radiotherapy, CT-RT), which significantly improved survival and provided better locoregional disease control compared with radiotherapy alone2,3. However, survival rates in this setting remain poor.
In the PACIFIC study, maintenance therapy with durvalumab following CT-RT demonstrated progression-free survival (PFS) rates at 12 and 18 months of 55.9% (95% CI, 51.0–60.4) and 44.2% (95% CI, 37.7–50.5), respectively, compared with 35.3% (95% CI, 29.0–41.7) and 27.0% (95% CI, 19.9–34.5) in the placebo arm4. Despite these advances, other treatment regimens combining CT-RT with concurrent or consolidation immunotherapy have failed to improve PACIFIC trial results (GEMSTONE-3015, DETERRED6, NICOLAS7, KEYNOTE-7998, CheckMate-73L9 or PACIFIC-210).
However, the potential of immunotherapy as an induction treatment—capable of eliciting an earlier and more robust antitumor response11—remains largely unexplored in the setting of unresectable NSCLC12, despite its results in phase II trials (such as NADIM and NADIM II trials from our group13,14,15,16) and its approval based on phase III studies for the operable scenario17,18.
Furthermore, even less is known about the role of baseline biomarkers, such as PD-L1 expression or tumor mutational burden (TMB), as well as the potential utility of post-induction circulating tumor DNA (ctDNA) clearance19 in unresectable cases, which may be critical for guiding subsequent adjuvant therapy in the absence of pathological response data.
Here (APOLO trial) we show the activity and safety of chemo-immunotherapy induction before chemo-radiotherapy and immunotherapy maintenance in patients with unresectable stage III NSCLC.
Results
Patient characteristics
Between July 8, 2021, and March 16, 2022, 38 patients were evaluated for eligibility and enrolled across 17 centers (Supplementary Table 1). Of the 38 patients, 11 (28.9%) were female, 24 (63.2%) were stage IIIB or IIIC, 34 (89.5%) were clinical stage N0, N1 or N2, and 15 (39.5%) had a PD-L1 negative tumor (Table 1 and Supplementary Table 2).
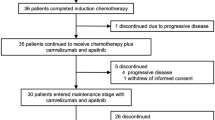
All 38 patients (100%) initiated induction treatment. Six (15.8%) received only one cycle and discontinued due to toxicity (n = 3, 7.9%), disease progression (n = 2, 5.3%), or investigator decision (n = 1, 2.6%). Thus, 32 (84.2%) were included in the per-protocol (PP) population, all of whom started concurrent CT-RT, receiving ≥60 Gy. Among these 32 patients, three (9.4%) did not complete CT-RT due to toxicity (n = 2, 6.3%) or disease progression (n = 1, 3.1%). Of the 29 patients (76.3% of the intention-to-treat [ITT] cohort) who initiated atezolizumab maintenance, 16 (55.2%) completed treatment, while 13 (44.8%) discontinued due to death (n = 1, 3.4%), toxicity (n = 3, 10.3%), disease progression (n = 7, 24.1%), or investigator decision (n = 2, 6.9%) (Supplementary Fig. 1).
Treatment outcomes
As of the data cutoff (February 12, 2024), 16 patients (42.1%) had died—14 (36.8%) due to disease progression and 2 (5.3%) from other causes—while 22 (57.9%) remained in follow-up. Of the 38 patients, 16 (42.1%) completed the protocol-defined treatment. Among them, 1 (6.3%) died from disease progression, while 15 (93.8%) were alive, including 11 (68.8%) who remained recurrence-free.
The median follow-up was 29.6 months (95% CI, 28.8–29.8). In the ITT population, 18 of 38 patients (47.4%) experienced disease progression, including 14 (36.8%) who died and 4 (10.5%) who remained in follow-up. Two (5.3%) progressed in the induction phase, 1 (2.6%) in the concurrent CT-RT phase, 7 (18.4%) in the maintenance phase, and 8 (21.1%) in follow-up. The median duration of overall survival was not reached in the ITT population nor in the PP population.
The trial met its primary endpoint. PFS at 12 months in the ITT population, was 68.4% (95% CI, 51.1–80.7) (Fig. 1A), which is significantly higher than the null-hypothesis of 55% (z = 1.77, p = 0.038).
A Progression-free Survival in the intention-to-treat (ITT) population. B Overall Survival in the intention-to-treat population (ITT). Shaded areas represent the 95% confidence interval. Source data are provided as a Source Data file.
Secondary endpoints analyses showed that PFS at 24 months was 50.0% (95% CI, 33.4–64.5) (Fig. 1A). Overall-survival (OS) was 86.8% (95% CI, 71.2–94.3) at 12 months and 60.5% (95% CI, 43.2–74.0) at 24 months (Fig. 1B).
Overall Response Rate (ORR) according to RECIST Version 1.1. was assessed by investigators after receiving induction atezolizumab plus chemotherapy and after concurrent CT-RT. After induction, investigators identified in the ITT population: 18 (47.4%) patients with partial response (PR), 14 (36.8%) with stable disease (SD) and 5 (13.2%) with progressive disease (PD). After receiving the concurrent CT-RT, there were: 1 (2.6%) patient with complete response (CR), 23 (60.5%) with PR, 8 (21.1%) with SD and 1 (2.6%) with PD. Data were missing for induction response in 1 patient (2.6%) and for CT-RT response in 5 patients (13.2%). We observed that 9 patients (23.7%) improved their response following CT-RT, 23 patients (60.5%) maintained their response, and only 1 patient (2.6%) experienced a worsening of response.
Regarding the sites of first failure, of the 18 patients who had disease progression, 8 (44.4%) were local and 10 (55.6%) were distant. From 8 patients who had local progression, 2 (25.0%) progressed in the induction phase, 2 (25.0%) in the maintenance phase and 4 (50.0%) during follow-up. From 10 patients who had distant disease progression, 1 (10.0%) had disease progression in the concurrent CT-RT phase, 5 (50.0%) in the maintenance phase and 4 (40.0%) during follow-up. Total new lesions in the ITT population occurred in 14 cases (36.8%), with brain being the site with the highest incidence of new lesions (4 cases, 10.5%), followed lung (3 cases, 7.9%), bone and pleural effusion (2 cases each site, 5.2%) and pulmonary lymph nodes, liver and brain plus bone (1 case each site, 2.6%) (Supplementary Table 3).
Post-hoc survival analysis in the PP population (n = 32) showed PFS rates of 78.1% (95% CI, 59.5–88.9) at 12 months and 56.2% (95% CI, 37.5–71.3) at 24 months, with their corresponding OS rates of 90.6% (95% CI, 73.6–96.8) at 12 months and 68.7% (95% CI, 49.7–81.8) at 24 months (Supplementary Fig. 2).
In subsequent post-hoc evaluations, the PFS after induction, calculated for the 32 (84.2%) patients who completed treatment induction, was calculated according to ORR and no differences were found between them (log-rank p = 0.55): PFS after induction at 12 months was 76.5% (95% CI, 48.8–90.4), 66.7% (95% CI, 33.7–86.0) and 66.7% (95% CI, 5.4–94.5) for patients with PR, SD or PD, respectively (Fig. 2A).
A Progression-free Survival (PFS) after induction treatment by Overall Response Rate (ORR) in the intention-to-treat population. PR partial response, SD stable disease, PD progression disease. B Progression-free Survival “PACIFIC-related” in the intention-to-treat (ITT) population. Shaded areas represent the 95% confidence interval. Source data are provided as a Source Data file.
The PFS “PACIFIC-related”, calculated for all patients who started maintenance treatment (n = 29, 76.3%), was 68.9% (95% CI, 48.8–82.5%) at 12 months and 51.7% (95% CI, 32.5–67.9) at 24 months (Fig. 2B). We also evaluated PFS “PACIFIC-related” based on whether patients completed maintenance treatment (n = 16, 55.2%) or not (n = 13, 44.8%). At 24 months, PFS “PACIFIC-related” was significantly higher in those who completed treatment (68.8%, 95% CI, 40.5–85.6) compared to those who did not (30.8%, 95% CI, 9.5–55.4, p = 0.0056) (Supplementary Fig. 3A). No differences in PFS “PACIFIC-related” were found according to ORR after CT-RT period (Supplementary Fig. 3B)
Safety
All patients (38 cases, 100%) had at least one adverse effect (AE) of any cause and grade; 29 (76.3%) patients had grade 3 or 4 AEs (Supplementary Dataset 1). One (2.6%) patient had a grade 5 AE during maintenance phase that was not related to atezolizumab treatment.
Considering those Treatment-Related Adverse Events (TRAEs) with an incidence of at least ≥10% of patients, we observed that during induction phase 26 (68.4%) patients had TRAEs related to atezolizumab, 34 (89.5%) related to carboplatin and 36 (94.7%) related to paclitaxel (Table 2). The most common grade 3 or 4 AES related to atezolizumab were febrile neutropenia (2 [5.3%] of 38 patients) and fatigue (1 [2.6%]) (Supplementary Table 4).
Through concurrent CT-RT phase, 11 (28.9%) and 23 (60.5%) patients had AEs related to chemotherapy or radiotherapy, respectively (Supplementary Tables 5, 6).
During maintenance phase, 17 (44.7%) of 38 patients had AEs related to atezolizumab (Table 3 and Supplementary Table 7). Two (5.3%) of 38 patients had grade 3 or 4 AEs related to atezolizumab; the most common were pneumonitis (1 [2.6%]) of 38 patients, and anemia (1 [2.6%]).
Discontinuation of treatment due to AEs related to atezolizumab occurred in 3 (7.9%) patients in the induction phase and 2 (5.2%) in the maintenance; and serious TRAE occurred in 1 (2.6%) and 1 (2.6%), respectively. No deaths due to adverse events related to any treatment occurred during the trial (Tables 2 and 3).
Exploratory endpoints
Regarding exploratory endpoints, baseline clinical characteristics (i.e. sex, age, smoking history, stage, N3 disease, histology, ECOG, were not associated with PFS or OS in ITT population. (Supplementary Fig. 4).
Concerning molecular markers, no association was observed with PD-L1 levels using different cutoffs (Supplementary Fig. 5); however, some associations with genomic markers such as TMB or ctDNA were found.
For tissue TMB (tTMB), 26 of 38 (68.4%) patients included in the trial had tumor samples available for tTMB assessment, and 16 (42.1%) had valid next-generation sequencing data for tTMB calculation (Supplementary Data 2). Of the 7 patients with high tTMB (≥9,89 mut/Mb), only 1 experienced disease progression, and none died. In contrast, among the 9 patients with low tTMB (<9,89 mut/Mb), 5 had disease progression and 4 died (PFS Log-rank p = 0.089 and OS Log-rank p = 0.056, Supplementary Fig. 6A). For blood TMB (bTMB) at baseline, of the 36 samples analyzed, 27 (75%) were considered valid (Supplementary Data 2). Patients with low bTMB (<16,5 mut/Mb) had significantly lower OS compared to patients with high bTMB (≥16,5 mut/Mb) (HR: 5.6, 95% CI: 1.7–18.3, p = 0.013). A similar trend was observed for PFS (HR: 2.5, 95% CI: 0.9–7.3, p = 0.081) (Supplementary Fig. 6B). Finally, Baseline ctDNA levels (n = 27) were significantly associated with clinical stage (p = 0.013). However, neither baseline ctDNA levels nor clinical stage were prognostic for patient survival outcomes (Supplementary Fig. 7). Regarding baseline specific mutations in tissue, no mutations associated with poor prognosis to immunotherapy were identified, except for one patient (Patient 4) with KRAS p.Gly12Cys/STK11 p.Ser69Ter, who experienced progression at 6 months and died at 14 months. On the other hand, both BRAF-mutated patients (Patients 16 and 36, with p.Gly469Arg and p.Gly596Val, respectively) remain progression-free and alive at data cutoff (Supplementary Fig. 8). Prevalent mutations in these patients, such as KRAS (5 cases) and TP53 (8 cases), were not associated with poorer survival (Supplementary Fig. 9).
Regarding ctDNA monitoring, valid paired plasma samples from baseline and post-induction treatment were available for 17 patients. The induction phase alone, or including CT-RT, significantly reduced ctDNA levels (p = 0.035 and p = 0.0044, respectively; Supplementary Fig. 10A). In our cohort, a substantial reduction in ctDNA levels after induction (i.e a reduction of at least 92% of ctDNA; first quartile) was associated with improved PFS (p = 0.037, Supplementary Fig. 10C). Notably, none of these patients who exhibited a substantial reduction in ctDNA levels died or experienced disease progression. Similarly, fluctuations in ctDNA levels were concordant with clinical responses observed on imaging (Supplementary Fig. 10B). Similarly, a reduction of ctDNA between post-induction and post-CT-RT also identified patients with improved PFS (p = 0.022, Supplementary Fig. 10D), with survival estimated from the timepoint after completion of induction treatment. The treatment also led to a reduction in bTMB levels after induction, and a decrease in bTMB (Fold change <1) was associated with better prognosis (PFS Log-rank p = 0.013 and OS Log-rank p = 0.022) (Supplementary Fig. 11).
Discussion
Our study tested the activity of chemo-immunotherapy induction followed by concurrent chemo-radiotherapy and subsequent consolidation with atezolizumab in unresectable stage III disease. This trial met its primary endpoint with a PFS of 68.4% (95% CI 51.1–80.6%) at one year and an OS of 60.5% (95% CI 43.3–74.0%) at 24 months, with a high maturity of the data. The PFS data after definitive combined treatment, which we have termed “PACIFIC-Related PFS”, are similar, with a 12-month PFS of 68.9% (95% CI: 48.8–82.4%). Although cross-trial comparisons must be interpreted with caution, these results are numerically higher than those reported by PACIFIC, which in its first publication reported a PFS at one year of 55.9% (95% CI 51.0–60.4%)4. Other phase II studies, such as DETERRED6, NICOLAS7, and KEYNOTE-7998, that aimed to improve outcomes in this scenario failed to do so closing the door to further studies that combine CT-RT and immunotherapy.
Recently, a phase II trial (AFT 16) with induction atezolizumab prior to CT-RT was published12, showing PFS at one and two year outcomes very similar to our results. However, their population was predominantly stage IIIA, whereas in our cohort, 63% of patients were stage IIIB/C. Notably, the decision on resectability varies by region and group. In our study, unresectability was determined by a multidisciplinary committee, and given that this trial runs parallel to NADIM II15 for potentially resectable N2 in the same hospitals, we believe our cohort has high homogeneity in locally advanced unresectable disease.
Another consequence of our study is the evidence of its feasibility. Compared to the AFT-16 study, we did not observe higher toxicity or more treatment discontinuations. On the contrary, the percentage of withdrawals before CT-RT was 29%, with 17% due to disease progression in the AFT-16 study12, compared to 16% in our study, with only 5% due to disease progression. Similarly, induction chemo-immunotherapy does not seem to compromise radical local treatment, with only 9% withdrawals during CT-RT, compared to 18% in the AFT-16 study. Moreover, early progression, prevents and selects a population that would never have benefited from the full radical treatment, as they would have progressed during it. One of the most concerning issues is the increase in pneumonitis after chemo-radiotherapy, which in our case was only 2.6%.
Furthermore, the OS data in the ITT population, with very high maturity, are very encouraging, with more than 86% remaining alive at 12 months, also more favorable than those published to date in similar studies4.
PD-L1 expression did not impact trial outcomes, supporting that PD-L1-negative patients can still achieve meaningful results with immunotherapy18,20. We observed a trend between high TMB and better prognosis, assessed both in tissue and blood, which methodologically strengthens our findings. High TMB has been linked to improved outcomes following CT-RT with durvalumab maintenance21, as well as to post-operative radiotherapy without immunotherapy22, making it a promising biomarker in this scenario that combines both therapies.
Tumor response assessed by RECIST criteria did not correlate with survival, reflecting a limitation similar to that observed in the resectable setting13,14,15. This limitation is even more crucial in the unresectable scenario, where the prognostic information provided by pathological response is absent19. In contrast, ctDNA clearance at the end of neoadjuvant therapy shows promise as a potential alternative to guide patient management in both unresectable and resectable stage III NSCLC neoadjuvant setting14,19.
The main limitations of this study stem from its design as a non-controlled single-arm phase II trial and its sample size. Consequently, comparisons with historical controls should be approached with caution. Additionally, the limited number of cases restricts the translational analysis to an exploratory nature, requiring all findings to be validated.
APOLO trial findings highlight the activity and safety of induction chemo-immunotherapy prior to chemo-radiotherapy and subsequent immunotherapy maintenance. Further randomized phase III studies are needed to clarify the role of neoadjuvant chemo-immunotherapy in the treatment of non-surgical stage III NSCLC.
Methods
Study design and patients
This study was conducted in accordance with EU Clinical Trial Directive and all relevant clinical trial regulations in Spain and also, the Declaration of Helsinki. The trial was authorized by the Spanish Agency for Medicines and Health Products (AEMPS) and approved by an Ethic Committee of Hospital Puerta de Hierro (Madrid, Spain; ref. 8/2021) acting as reference ethic committee. All patients provided written informed consent prior to any trial-related procedures. Patients did not receive financial compensation for their participation. The study was preregistered at ClinicalTrials.gov (NCT04776447; protocol registered on February 24, 2021). Original protocol, final protocol, summary of changes and statistical analysis plan are described in Supplementary Note.
This open-label, non-randomized, phase 2 trial was conducted at 17 hospitals in Spain. Patients were enrolled between July 8, 2021, and March 16, 2022. Eligible patients were aged 18–80 years, and had histologically or cytologically confirmed non-resectable (determined by a multidisciplinary tumor board at each site) Stage IIIA-IIIB-IIIC NSCLC with N2/3 disease (American Joint Committee on Cancer eighth edition criteria). Sex was considered in the study design. Self-reported sex information was collected for all participants and included in the clinical database. Analyses were conducted to explore potential differences in clinical outcomes between male and female patients. Patients were required to have an Eastern Cooperative Oncology Group (ECOG) performance status of ≤1, adequate lung function and no prior treatment with anti-neoplastic drugs or thoracic radiotherapy. Exclusion criteria were the presence of a known pathogenic alterations in EGFR, ALK, STK11, MDM2 or ROS1. PET-CT and brain CT or MRI were mandatory for patient inclusion. Complete details regarding the inclusion and exclusion criteria are available in the study protocol (Supplementary Note).
Procedures
During the induction phase, patients received intravenous atezolizumab (1200 mg), paclitaxel (200 mg/m²), and carboplatin (area under the curve [AUC] 5 mg/mL/min) on day 1 of a 3-week cycle. After three cycles, patients proceeded to concurrent chemo-radiotherapy according to local guidelines. A platinum-based doublet regimen was recommended (carboplatin-paclitaxel, carboplatin-vinorelbine, or carboplatin/cisplatin-pemetrexed), along with radiotherapy at a total dose of 66 Gy, delivered in 33 once-daily fractions. Preferably, radiotherapy was initiated on day 1 of cycle 1 of concurrent CT-RT. Following the third cycle of concurrent CT-RT (i.e., the sixth cycle of chemotherapy), maintenance therapy with intravenous atezolizumab (1200 mg) was initiated for 12 months, administered every 3 weeks for a total of 16 cycles.
For all patients, tumor response was assessed locally until disease progression using a CT scan, MRI or full-body PET at baseline and at approximately every 9 weeks from cycle 1 day 1 during treatment. Progression-free survival was centrally reviewed.
All adverse events were assessed at baseline, continuously while on treatment, at the treatment discontinuation visit, and at the safety follow-up visit. Adverse events were graded in accordance with the US National Cancer Institute Common Terminology Criteria for Adverse Events (NCI-CTCAE), version 5.0. Patients had laboratory blood tests at baseline visit and within 3 days prior to day 1 administration of each cycle. Adverse events were recorded and followed from the day of enrolment to 30 days from last dose of administration.
Patient withdrawal criteria included intolerable toxicities, symptom worsening from disease progression, severe inter-current illnesses impacting assessments, patient decision, or non-adherence to protocol.
End points
The primary endpoint was to assess the activity of the treatment in terms of Progression Free Survival (PFS) at 12 months according to RECIST Version 1.1. PFS was defined as the time from inclusion until objective tumor progression or death.
Secondary endpoints included: PFS at 24 months from the inclusion, Overall Survival (OS) at 12 and 24 months (time from inclusion until death from any cause), Overall Response Rate (ORR) as measured by investigator-assessed according to RECIST v1.1, the sites of first failure, and the safety and tolerability of atezolizumab in combination with chemotherapy and atezolizumab as maintenance treatment.
Exploratory endpoints included the prognostic PFS and OS value of molecular markers related to immune response at diagnosis (i.e. PD-L1 TPS, TMB, and specific mutations), ctDNA levels, and ORR upon induction therapy.
PD-L1
Formalin-fixed paraffin-embedded (FFPE) tissue samples were obtained from primary tumor or metastatic sites at diagnosis. PD-L1 expression was quantitatively assessed at each center according to clinical practice guidelines. PD-L1 protein expression was determined by using Tumor Proportion Score (TPS, percentage of viable tumor cells showing partial or complete membrane staining at any intensity). A tissue sample was considered adequate for assay interpretation if it contained at least 50 viable tumor cells. PD-L1 TPS was categorized using different thresholds for statistical analysis (<1% vs ≥1%; <1% vs 1–49% vs ≥50%; <50% vs ≥50%).
Tissue TMB and specific mutations
For tissue TMB 26 of 38 (68%) patients included in the trial had tumor samples available for TMB assessment, and 16 (42%) had valid next-generation sequencing data for TMB calculation. Library generation and sequencing of samples was performed on an Ion Chef System and S5 Sequencer (ThermoFisher, Palo Alto, CA). DNA was extracted from 10-µm-thick paraffin sections using the truXTRAC® FFPE total Nucleic Acid (Covaris). Extracted DNA was quantified using the Qubit® dsDNA HS Kit in combination with a Qubit® 2.0 fluorometer (Thermo Fisher Scientific). To remove deaminated bases before target amplification, 20 ng of FFPE DNA was treated with heat labile Uracil-DNA Glycosylase (UDG). Library preparation was performed, on an Ion Chef™ System (Thermo Fisher Scientific), using 20 ng of input DNA and the Oncomine Tumor Mutation Load Assay (Thermo Fisher Scientific). The panel covers 1.7 megabases of 409 genes with known cancer associations. The final barcoded libraries were pooled and adjusted to a final concentration of 50 pM. Eight samples were loaded onto an Ion 540 chip. Template preparation and chip loading were carried out on an Ion Chef System. Finally, the Ion 540 chips were sequenced on an Ion S5 Sequencer. Reads were aligned to hg19 using Torrent Suite 5.20 and BAM files were transferred to Ion Reporter 5.20 for variant calling. TMB was computed using the TMB filter chain and the TMB algorithm 3.4 (Thermo Fisher Scientific) and secondary analysis including TMB calculation. Briefly, germline variants were filtered out using a germline filter-chain based on population databases: variant alleles present from the 1000 Genome Project, NHLBI GO Exome Sequencing Project (ESP), and ExAC. Different empirical cut points for TMB were evaluated for PFS studies.
ctDNA measurement and blood TMB
Peripheral whole-blood samples were collected in two 8.5-mL PPT™ tubes (Becton Dickinson) before and after induction treatment. Plasma was separated from the cellular fraction through two consecutive centrifugations: first at 1600 × g for 10 min and then at 6000 × g for 10 min. The samples were then divided into four 2.0 mL aliquots and sent to the central laboratory for analysis. Circulating free DNA (cfDNA) was isolated using the QIAamp Circulating Nucleic Acid Kit (QIAGEN, Valencia, CA, USA). Libraries were prepared from at least 30 ng of cfDNA using the hybrid capture-based TruSight Oncology 500 ctDNA next-generation sequencing (NGS) assay, following the manufacturer’s instructions. The libraries were subsequently pooled, denatured, and diluted to the appropriate loading concentration. Six libraries were sequenced per lane on an S4 flow cell, allowing for a total of 24 samples per run, with a read length of 2 × 151 bp. Data analysis was performed using the DRAGEN TSO 500 ctDNA Analysis Software v1.2, utilizing the TSO 500 pipeline. Briefly, sequencing reads were aligned to the hg19 reference genome. For liquid tumor mutational burden (TMB) calculation, germline variants were filtered out using public databases, including GnomAD exome and genome databases, as well as the 1000 Genomes Project database. A post-database filtering strategy was employed that used allele frequency information and considered variants in close proximity. Variants eligible for TMB computation were those in the coding region with a minimum variant allele frequency (VAF) ≥ 0.2% and below 40%, and with coverage ≥1000X. Mutations in TET2, TP53, DNMT3A, and CBL were excluded, as they were categorized as mutations derived from clonal hematopoiesis (CH). Multiple nucleotide variants were also excluded from the analysis. A receiver operating characteristic (ROC) curve was used to select the best cutoff value for mortality status (high bTMB defined as ≥16.5 mut/Mb). ctDNA was quantified using the mean of the maximum somatic variant allele frequency (VAF) (MaxSomaticVAF), which was calculated as the 4th highest variant allele frequency among the remaining variants after excluding germline and CH-derived mutations. ctDNA clearance were calculated as the log2 ratio of MaxSomaticVAF after induction to MaxSomaticVAF at baseline for the paired plasma samples.
Statistical analysis
It was estimated that the study would have a power of 80% to detect a PFS at 12 months of 75% (p1), corresponding to an extension of PFS of 20% from null hypothesis (p0, 55%), based on a log-rank test with one-sided significance level of 5%. Assuming a 15% of withdrawals a sample size of 37 patients was estimated. Informative censoring was not observed in the study data.
The intention-to-treat (ITT) population included all enrolled patients, while the per-protocol (PP) population comprised those who received at least two induction cycles or underwent the first tumor response evaluation. Safety population included all patients that were exposed to study treatment (atezolizumab).
To assess the primary and secondary objectives Kaplan-Meier method was used to estimate survival functions at 12 and 24 months, with 95% CIs and to draw the corresponding survival curves. A Z-test rejected the null hypothesis if z ≥ 1.645, where z = (Kaplan–Meier estimate at 12 months − null hypothesis value [55%])/standard error. Descriptive analysis was performed for first failure sites, ORR, and safety outcomes. Median follow-up was estimated using the reverse Kaplan–Meier method. Survival curves were compared using the log-rank test.
Post-hoc analyses included PFS and OS in the PP population, PFS after induction, and “PACIFIC-related” PFS (i.e., PFS after definitive combined treatment) at 12 and 24 months.
PD-L1 TPS was categorized using different published thresholds (<1% vs ≥1%; <1% vs 1–49% vs ≥50%; and <50% vs ≥50%). For TMB thresholds determination, ROC curves for exitus status were used to select the best cutoff value in survival analysis. Baseline ctDNA was categorized using 0.01 Max MAF threshold and ctDNA clearer patients were defined as those in the upper quartile for ctDNA clearance.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
De-identified individual participant data, along with a data dictionary, will be accessible under restricted access following the publication of this study for research purpose only in compliance with patient consent for data sharing. Researchers wishing to access the data should submit a research proposal to the corresponding author. The proposal will be reviewed by the Spanish Lung Cancer Group and the ethics committee for clinical investigation. A response to requests will be provided within 4–6 weeks. The remaining data are available within the Article, Supplementary Information, Supplementary Data, or Source Data file. Source data are provided with this paper.
References
Aupérin, A. et al. Meta-analysis of concomitant versus sequential radiochemotherapy in locally advanced non - small-cell lung cancer. J. Clin. Oncol. 28, 2181–2190 (2010).
Schaake-Koning, C. et al. Effects of concomitant cisplatin and radiotherapy on inoperable non-small-cell lung cancer. N. Engl. J. Med. 326, 524–30 (1992).
Jeremic, B., Shibamoto, Y., Acimovic, L. & Djuric, L. Randomized trial of hyperfractionated radiation therapy with or without concurrent chemotherapy for stage III non-small-cell lung cancer. J. Clin. Oncol. 13, 452–458 (1995).
Antonia, S. J. et al. Durvalumab after chemoradiotherapy in stage III non–small-cell lung cancer. N. Engl. J. Med. 377, 1919–1929 (2017).
Zhou, Q. et al. Sugemalimab versus placebo after concurrent or sequential chemoradiotherapy in patients with locally advanced, unresectable, stage III non-small-cell lung cancer in China (GEMSTONE-301): interim results of a randomised, double-blind, multicentre, phase 3 trial. Lancet Oncol. 23, 209–219 (2022).
Liu, Y. et al. Final efficacy outcomes of atezolizumab with chemoradiation for unresectable NSCLC: The phase II DETERRED trial. Lung Cancer 174, 112–117 (2022).
Peters, S. et al. Progression-Free and Overall Survival for Concurrent Nivolumab With Standard Concurrent Chemoradiotherapy in Locally Advanced Stage IIIA-B NSCLC: Results From the European Thoracic Oncology Platform NICOLAS Phase II Trial (European Thoracic Oncology Platf. J. Thorac. Oncol. 16, 278–288 (2021).
Jabbour, S. K. et al. Pembrolizumab Plus Concurrent Chemoradiation Therapy in Patients With Unresectable, Locally Advanced, Stage III Non-Small Cell Lung Cancer: The Phase 2 KEYNOTE-799 Nonrandomized Trial. JAMA Oncol. 7, 1351–1359 (2021).
De Ruysscher, D. et al. CheckMate 73L: A Phase 3 Study Comparing Nivolumab Plus Concurrent Chemoradiotherapy Followed by Nivolumab With or Without Ipilimumab Versus Concurrent Chemoradiotherapy Followed by Durvalumab for Previously Untreated, Locally Advanced Stage III Non-Small-Cell Lung Cancer. Clin. Lung Cancer 23, e264–e268 (2022).
Bradley, J. D. et al. LBA1 Durvalumab in combination with chemoradiotherapy for patients with unresectable stage III NSCLC: Final results from PACIFIC-2. ESMO Open 9, 102986 (2024).
Provencio, M., Calvo, V., Romero, A., Spicer, J. D. & Cruz-Bermúdez, A. Treatment Sequencing in Resectable Lung Cancer: The Good and the Bad of Adjuvant Versus Neoadjuvant Therapy. Am. Soc. Clin. Oncol. Educ. B. 1–18 https://doi.org/10.1200/EDBK_358995 (2022).
Ross, H. J. et al. Atezolizumab before and after Chemoradiation for Unresectable Stage III Non-Small Cell Lung Cancer: A Phase II Nonrandomized Controlled Trial. JAMA Oncol. 60607, 1–8 (2024).
Provencio, M. et al. Neoadjuvant chemotherapy and nivolumab in resectable non-small-cell lung cancer (NADIM): an open-label, multicentre, single-arm, phase 2 trial. Lancet Oncol. 21, 1413–1422 (2020).
Provencio, M. et al. Overall Survival and Biomarker Analysis of Neoadjuvant Nivolumab Plus Chemotherapy in Operable Stage IIIA Non-Small-Cell Lung Cancer (NADIM phase II trial). J. Clin. Oncol. JCO2102660 https://doi.org/10.1200/JCO.21.02660 (2022).
Provencio, M. et al. Perioperative Nivolumab and Chemotherapy in Stage III Non–Small-Cell Lung Cancer. N. Engl. J. Med. 389, 504–513 (2023).
Provencio, M. et al. Perioperative chemotherapy and nivolumab in non-small-cell lung cancer (NADIM): 5-year clinical outcomes from a multicentre, single-arm, phase 2 trial. Lancet Oncol. 1453–1464 https://doi.org/10.1016/S1470-2045(24)00498-4 (2024).
Forde, P. M. et al. Neoadjuvant Nivolumab plus Chemotherapy in Resectable Lung Cancer. N. Engl. J. Med. 386, 1973–1985 (2022).
Cascone, T. et al. Perioperative Nivolumab in Resectable Lung Cancer. N. Engl. J. Med. 390, 1756–1769 (2024).
Deutsch, J. S. et al. Association between pathologic response and survival after neoadjuvant therapy in lung cancer. Nat. Med. https://doi.org/10.1038/s41591-023-02660-6 (2023).
Girard, N. et al. Treatment Characteristics and Real-World Progression-Free Survival in Patients With Unresectable Stage III NSCLC Who Received Durvalumab After Chemoradiotherapy: Findings From the PACIFIC-R Study. J. Thorac. Oncol. 18, 181–193 (2023).
Lebow, E. S. et al. Analysis of Tumor Mutational Burden, Progression-Free Survival, and Local-Regional Control in Patents with Locally Advanced Non-Small Cell Lung Cancer Treated with Chemoradiation and Durvalumab. JAMA Netw. Open 6, E2249591 (2023).
Shaverdian, N. et al. Effects of Tumor Mutational Burden and Gene Alterations Associated with Radiation Response on Outcomes of Postoperative Radiation Therapy in Non-Small Cell Lung Cancer. Int. J. Radiat. Oncol. Biol. Phys. 113, 335–344 (2022).
Acknowledgements
The trial’s design and data analysis were conducted by the Spanish Lung Cancer Group (Fundación GECP) and the APOLO trial investigators. The study funders had no involvement in the study design, data collection, analysis, interpretation, or the writing of the manuscript. This study was supported by Roche; the European Union Horizon 2020 Research and Innovation program (European Commission) under grant agreement number 875160; Instituto de Salud Carlos III (ISCIII) (grant nos. PI21/01500, PI22/01223, PI23/01054 [co-funded by the European Regional Development Fund and European Social Fund “A way to make Europe”/“Investing in your future” from the European Commission]); and the Ministry of Science and Innovation (grant no. RTC2019-007359-1 [BLI-O] and CPP2022-009545 [STRAGEN-IO]). C.M-T is supported by a Comunidad de Madrid predoctoral contract (PIPF-2022/SAL-GL-25283) granted to M.P. P.M is supported by an ISCIII predoctoral contract PFIS (FI22/00321) granted to A.R. A.C.-B. is supported by an ISCIII-“Miguel Servet” contract (CP23/00044). We thank the patients, their families, all the participating clinical teams and all the staff at the Spanish Lung Cancer Group (Fundación GECP) for making this study possible.
Author information
Authors and Affiliations
Contributions
M.P. conceived and supervised the study, performed investigation and formal analysis, obtained funding and drafted the manuscript. A.C.-B. contributed to study conceptualization, investigation, formal analysis, supervision, funding acquisition, and writing – original draft and reviewed the manuscript. A.R. participated in investigation, formal analysis, supervision and funding acquisition and reviewed the manuscript. C.M.-T. and P.M. contributed to investigation, formal analysis and review. B.C., M.G., L.V., R.G.C., M.D., S.V.E., A.F., M.A.S., A.L.O., A.B., A.I., M.C.A., I.S., R.L., V.C., D.R.-A., J.B.-B., A.L.-M., R.M., L.T., K.M., J.G., M.F.B. and E.S.S. participated in investigation and manuscript review. All authors had full access to study data, contributed to the final version of the manuscript, and approved submission.
Corresponding author
Ethics declarations
Competing interests
M.P. reports grants, consulting fees and non-financial support from BMS; grants, consulting fees and non-financial support from Roche; grants, consulting fees and non-financial support from Astra Zeneca; consulting fees from MSD; consulting fees from Takeda; outside the submitted work. B.C. reports payment or honoraria from BMS, Astra Zeneca, Novartis, Amgen; support for attending meetings or travels from Roche, Astra Zeneca, GSK, Lilly; and advisory board from Roche and Astra Zeneca. R.G.-C. reports payment or honoraria from AstraZeneca, BMS, Janssen, Lilly, MSD, Novartis, Pfizer, Roche, Sanofi, and Takeda; support for attending meetings or travel from AstraZeneca, Roche, Pfizer, BMS, MSD, and Janssen; and advisory board participation with AstraZeneca and Janssen. M.D. reports payment or honoraria from Janssen, Bristol Myers Squibb, AstraZeneca, MSD, Guardant; support for attending meetings or travels from Janssen; advisory Board participation with Bristol Myers Squibb. S.V-E. reports consulting fees from AstraZeneca; payment or honoraria from AstraZeneca; support for attending meetings or travels from AstraZeneca. A.F. reports payment or honoraria from AstraZeneca; support for attending meetings or travels from Merck, MSD, Pfizer. A.B. reports consulting fees from AZ; payment Honoraria from Regeneron; support for attending meetings or travels from Roche Pharma. A.I. reports payment or honoraria from Amgen, BMS, Regeneron, Roche, Takeda; support for attending meetings or travel: Roche, Pfizer, MSD, Takeda; and advisory board participation with AstraZeneca, Roche, Pfizer, Takeda. M.C-A. reports payment or honoraria: Roche, Bristol, MSD, AstraZeneca, Janssen, Pfizer, Takeda, Regeneron; support for attending meetings and/or travel: Roche, Bristol, MSD, AstraZeneca, Janssen, Pfizer, Takeda, Regeneron. R.L. reports payment or honoraria from Takeda, AstraZeneca, Amgen, Bristol-Myers Squibb, Roche, Pierre-Fabre; support for attending meetings or travel from Roche, AstraZeneca, MSD, Takeda, Merck Serono. V.C. reports consulting fees from Roche, AstraZeneca, MSD, BMS, Takeda, Sanofi, Amgen, GSK, Boehringer Ingelheim, Janssen; payment or honoraria from Roche, AstraZeneca, MSD, BMS, Takeda, Sanofi, Amgen, Pfizer, Janssen, Beigene; support for attending meetings or travels: AstraZeneca, Roche, MSD, Takeda, Janssen. D.R-A. reports honoraria for lectures from MSD, Roche, BMS, Novartis, Takeda, Lilly, AstraZeneca. Support for attending meetings or travels from Roche, MSD, Novartis, Sanofi; advisory board participation with Merck Sharp Dohme, Regeneron, BMS, GSK, Lilly. J.B-B. reports consulting fees from Roche; payment or honoraria from BMS, Roche, AstraZeneca, Regeneron, MSD, Pfizer, Amgen; support for attending meetings or travels from MSD, Roche, Takeda. A.L-M. reports payment or honoraria from Pfizer, Lilly, Novartis; support for attending meetings or travels from MSD, AMGEM, Takeda; support for attending meetings or travels from MSD, Roche, Takeda. A.R. reports grants from EMQN; consulting fees from Johnson and Johnson, ThermoFisher; payment or honoraria from Illumina, Health in Code, ThermoFisher; support for attending meetings or travels from ThermoFisher, Bristol Myers Squibb; and Advisory Board participation with Takeda. All other authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Michel van den Heuvel and the other anonymous reviewer(s) for their contribution to the peer review of this work. [A peer review file is available].
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Provencio, M., Campos, B., Guirado, M. et al. Induction chemo-immunotherapy followed by chemo-radiotherapy and immunotherapy maintenance in stage III NSCLC (APOLO): a phase 2 trial. Nat Commun 16, 10124 (2025). https://doi.org/10.1038/s41467-025-66097-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-66097-w