Abstract
Copper-based catalysts are the premier choice for electrochemical reduction of CO2 (CO2RR) into hydrocarbons or oxygenates. However, the facilely structural reconstruction of copper sites during electrolysis poses significant challenges to the long-life electrolytic efficiency. Herein, we leverage the strong σ-π dative bonding between Cuδ+ and alkyne-based ligands to stabilize copper sites for the prolonged CO2RR. We demonstrate the feasibility of taming the electronic structures of copper sites through the tug of war between σ and π backbonding interactions. The optimal copper organic polymer with methoxy group functionalization (OMe-PhCu) exhibits a moderate charge density of copper sites and an intensified local asymmetric charge distribution of coordinative carbon, enhancing the selectivity of methane with a Faradaic efficiency of 68.8% and a partial current density of 324.5 mA cm−2 in acidic electrolyte. In situ spectra and density functional theory calculations reveal enhanced *CO adsorption and lowered energy barrier for CO2RR into methane over OMe-PhCu. Building upon such stable Cuδ+ sites, we further construct Cuδ+/Cu0 catalytic interfaces for the generally enhanced electrosynthesis of multi-carbons and ammonias. This synthetic chemistry paves the pathway for the design of stable catalytic active sites for renewable conversions.
Similar content being viewed by others
Introduction
Copper is one of the most promising catalysts for the generation of valuable chemicals beyond CO (for example, methane, ethylene, ethanol, and so on) through the electrochemical CO2 reduction reaction (CO2RR)1,2. Compared to alkaline electrolyte environments, the acidic systems have garnered significant interests due to their high single-pass CO2 conversion efficiency (SPCE)3,4,5. Among the various CO2 reduction products, methane possesses the highest heating value and serves as the primary component of natural gas, endowing it with significant energy value6. Furthermore, methane is also an important feedstock for producing syngas and numerous pharmaceutical chemicals. When integrated with renewable energy sources, the electrocatalytic methanation of CO2 represents a highly promising technological pathway.
However, drastic and unpredictable reconstruction, leaching, agglomeration or three-phase interface failure will simultaneously become more evident7,8 in acidic conditions. For instance, Cu+ species that function efficiently in alkaline environments will dissolve in acidic solutions and subsequently undergo disproportionation, forming dissolved Cu²⁺ species in the electrolyte9. Particularly in strongly acidic systems where halides serve as the supporting salt, even metallic copper suffers significant chemical corrosion and reconstruction, further transforming into dissolved Cu⁺ or Cu²⁺ ions within the electrolyte10. These critical issues highlight the urgent need for catalysts that are more active, selective, and stable—a requirement that remains challenging to fulfill for acidic CO2RR system.
Among copper species, partially oxidized Cuδ+ species play a vital role in stabilizing *CO and related intermediates, which are crucial for the generation of hydrocarbons or oxygenates11,12,13,14. However, preserving the active and stable geometry and electronic structures of Cuδ+ species under cathodic potentials is fundamentally challenging due to their intrinsic thermodynamic instability15,16. Unfortunately, effective methods to retain the catalyst structure during CO2RR remain scarce. A few studies have suggested heteroatom incorporation17,18, structural confinement19,20, molecular modifications21 or utilizing redox active substrate22. However, these stabilization strategies can only preserve a small amount of Cu+ species in most cases, and the catalyst bulk remains dominated by metallic copper during CO2RR process, which further obscures a precise understanding of the inherent structure-activity relationship of specified copper species. More critically, the underlying stabilization mechanism is still not as fully understood as in thermal CO2 hydrogenation reactions23,24. It is of fundamental importance to comprehend the chemistry limitations of existing methods and develop robust stabilization strategies for Cuδ+ catalysts in electrocatalytic environment. Further, flexible electronic structure tunning strategies are simultaneously needed in order to achieve catalysts with both high activity and selectivity.
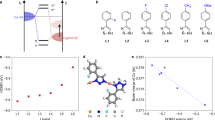
Herein, we report a class of Cuδ+ sites with tunable electronic structures through σ-π dative bonding interaction, capable of maintaining stable activity and meantime resisting structural reconstruction during CO2RR. Fundamentally different from previously reported electrophilic molecules or weak field ligands modification strategies (Fig. 1a) that usually work based upon limited interfacial electron transfer or weak σ bonding interaction21,25,26,27, phenylacetylene (R-Ph) based ligands, which possess π electrons and empty antibonding π* orbitals, were strategically utilized to coordinate with Cu(I) ions, resulting in the formation of metal-organic polymers (MOPs) featuring stable Cuδ+ sites. Within the coordination structure, according to Chatt-Dewar-Duncanson model28,29, alkyne bonds will donate π electrons to empty s orbital of Cu(I) and form strong σ bond, while antibonding π* orbitals of R-Ph accept electrons from d orbitals of Cu(I), and form d-p π-back-bonding due to their symmetry and energy alignment (Fig. 1b). The synergistic σ-π dative bonding enables enhanced structural stability of Cu(I) sites due to the increased copper-ligand bonding interaction. More importantly, the electronic structure of Cuδ+ sites can be manipulated by adjusting the relative strengths of σ and π backbonding, a strategy that has been seldom reported in the design of copper-based electrocatalysts.
a Typical molecular strategies for stabilizing Cuδ+ sites (blue: N, gray: C, yellow: S, white: H). b Schematic of the σ-π dative bonding between phenylacetylene and Cu+. c Formation processes of phenylacetylene based copper polymer with different functional groups. d Bader charge and Fermi level of alkynyl in MOPs. e Electrostatic potential near Cu centers in MOPs (isosurface = 0.05 electron/bohr3). Source data are provided as a Source Data file.
As outlined, by modulating the competition between σ and π back-donation, it is possible to fine-tune the electronic structures of copper sites. In this context, we selected electron-donating methoxyl and electron-withdrawing fluorine substituents to modify the phenylacetylene molecules (Fig. 1c), thereby indirectly controlling the Lewis basicity of the alkyne group through inductive and π-conjugation effects and their further bonding interactions with copper atoms. The MOPs without and with methoxyl and fluorine modification are donated as H-PhCu, OMe-PhCu and F-PhCu, respectively. The atomic coordinates of the optimized computational models are shown in Supplementary Data 1 file. More specifically, the charge density on alkynyl groups and copper atoms are affected by the functional groups on benzene ring (Fig. 1d and Supplementary Fig. 1). Additionally, the Fermi level (Ef) of MOPs shifts positively after methoxyl substitution (Fig. 1d), indicating that OMe-PhCu exhibits greater resistance to reduction under electrochemical scenarios. The electrochemical stability of MOPs is also proved by the positive free energy change of Cuδ+ reduction (Supplementary Fig. 2). Such ability is vital for the CO2RR catalysis in acid media, where metallic Cu worsens the selectivity to methane by accelerating the hydrogen evolution (Supplementary Fig. 3) and C-C coupling.
Notably, the four carbon atoms bonded to Cu exhibit different charge densities, leading to local asymmetry around copper sites. This asymmetry is supported by the calculated electrostatic potential (Fig. 1e) and Bader charge analysis (Supplementary Fig. 4 and Supplementary Table 1). Influenced by both the nature of the alkynyl groups and the local charge asymmetry, Cuδ+ sites in OMe-PhCu exhibit moderate charge density among the three samples. In general, the proposed design strategy offers an electronic and structural tunability of this class of copper-based electrocatalysts that is highly compatible with CO2RR systems.
Results
Synthesis and characterization of MOPs
The Cu2(OH)3OAc·H2O precursor was synthesized using copper acetate as the starting material, followed by gradual reduction with methanol and coordination with functionalized phenylacetylene to form the MOPs30. As discussed, PhCu MOPs with methoxyl group (OMe-PhCu) and another with fluorine group (F-PhCu) were chosen to investigate the impact of pendant group functionalization on the activity and selectivity of copper sites.
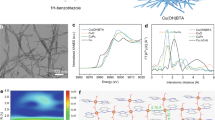
The MOPs are found to exhibit one-dimensional Cu arrays (Supplementary Fig. 1). The coordination reaction between Cu+ and phenylene diacetylene based ligands can be verified by infrared spectroscopy, as can be seen from the disappearance of -C ≡ C-H vibrations and stretching vibration shift of -C ≡ C- bonds after reaction31 (Supplementary Fig. 5). The MOPs show expected nanofiber structure due to the strong π-π stacking between phenylene diacetylene units (Fig. 2a, b and Supplementary Figs. 6–9). The corresponding high-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) and energy spectrum reveal that all the elements are evenly distributed within the nanofibers.
a Transmission electron microscopy (TEM) images of OMe-PhCu. b High-angle annular dark-field scanning TEM (HAADF-STEM) image and the corresponding energy dispersive spectroscopy mapping images. c Raman spectra of OMe-PhCu and corresponding phenylacetylene based ligands precursors. d Cu 2p XPS spectra of PhCu MOPs. e, f X-ray absorption spectroscopy (XAS) of PhCu MOPs and reference samples. e The Cu K-edge X-ray absorption near-edge structure (XANES) spectra. f Magnitude of Fourier-transformed Cu K-edge extended X-ray absorption fine structure spectroscopy (EXAFS) spectra. χ(k) is the extracted EXAFS signal, k is the photoelectron momentum and R is the radial distance without phase correction. Source data are provided as a Source Data file.
The crystal structure of copper organic polymer was investigated by powder X-ray diffraction (XRD) patterns (Supplementary Fig. 10). The XRD pattern of OMe-PhCu exhibits a broad hump between the 2θ range of 10–30°, suggesting the presence of poorly crystalline components. This may arise from the limitations of the synthetic conditions. And the triclinic cell parameters of OMe-PhCu was obtained by indexing peak positions of diffraction pattern (Supplementary Table 2) via Lebail method. Elemental analysis and inductively coupled plasma atomic emission spectroscopy (ICP-AES) (Supplementary Tables 3, 4) revealed a ~1:1 molar ratio between 1-ethynyl-4-methoxybenzene and copper, confirming the target stoichiometry.
As shown in Fig. 2c, the intense Raman lines at 2106 cm−1, corresponding to the -C ≡ C- triple bond vibration of 1-ethynyl-4-methoxybenzene30,32, shifts to lower frequency (1928 cm−1) upon coordination with Cu+. This shift suggests significant back-donation from the filled d orbitals of Cu⁺ to the antibonding orbitals of the ethynyl groups32, consistent with the theoretical analysis. Besides, there are three Raman spectrum peaks of OMe-PhCu at about 1165, 1195 and 1604 cm−1, assigned to the benzene ring33. Obviously, the band intensity within this range is relatively weak in the spectrum of OMe-PhCu. Other lines (479, 543 cm−1) may be ascribed to vibrations originating from copper and 4-methoxyphenylacetylene coordination interaction33. Similar behaviors were also observed for PhCu and F-PhCu catalysts (Supplementary Fig. 11).
No satellite peaks of Cu2+ are observed in the Cu 2p binding energy region (Fig. 2d), indicating that the Cu valence state in PhCu MOPs is approximately +1, further confirmed by the Cu Auger LMM spectra (Supplementary Fig. 12). F-PhCu exhibits a higher binding energy than OMe-PhCu and H-PhCu, revealing the tunable electronic structure of the centered copper. X-ray absorption fine structure (XAFS) spectroscopy was further employed to investigate the local environment of copper in MOPs. The Cu K-edge X-ray absorption near-edge structure (XANES) (Fig. 2e) and the first-order derivatives of the XANES spectra (Supplementary Fig. 13) show no evidence of Cu2+ or metallic copper, consistent with the XPS results. Furthermore, OMe-PhCu and F-PhCu MOPs exhibit slightly higher valence states compared to H-PhCu, likely due to the combined conjugated and inductive effects, as supported by the above Bader charge analysis (Supplementary Fig. 1). The extended X-ray absorption fine structure (EXAFS) spectra reveal the formation of a distinct Cu-C bond, accompanied by Cu-Cu interactions (Fig. 2f and Supplementary Fig. 14–17). The EXAFS fitting results based on this structure closely matched the Density Functional Theory (DFT) simulations.
Electrocatalytic performance and structural stability during CO2RR
The CO2 electroreduction performances of MOP catalysts were evaluated in a 0.1 M K2SO4 acidic system (pH = 3 ± 0.05) using a flow cell. CH4, CO, H2, and C2H4 were identified and quantified as gaseous products via gas chromatography (Supplementary Figs. 18–19). Liquid products were analyzed by High Performance Liquid Chromatography (HPLC, Supplementary Fig. 20). Methane is found to be the primary electrolytic product for MOPs (Fig. 3a and Supplementary Fig. 21). With increasing current density, the Faradaic efficiency of OMe-PhCu for methane reaches 68.8% at 300.0 mA cm−2, much higher than those of F-PhCu and H-PhCu. This is because OMe-PhCu presents the most prominent local charge asymmetry around copper sites, which is advantageous for the adsorption of CO2 and optimization of the reaction pathway6. Besides, the FE of CH4 gradually decreases with the increased catalyst loading, while the FEs of multicarbon products (C2H4, C2H5OH etc.) increase with the increased catalyst loading (Supplementary Fig. 22). Generally, a lower catalyst loading would lead to sparser distribution of the copper active sites, which is therefore not conducive to C-C coupling process. Furthermore, the CO2RR performance of OMe-PhCu in more acidic electrolyte was investigated (Supplementary Fig. 23), showing decreased selectivity of methane.
a Faradaic efficiencies (FEs) of CH4 under different current densities over OMe-PhCu in 0.1 M K2SO4 acidic electrolyte (pH = 3 ± 0.05). b FEs of gaseous products (CH4, H2, CO, C2H4) over OMe-PhCu in 0.2 M K2SO4 acidic electrolyte (pH = 3 ± 0.05). c Partial current densities of CH4 vs. total CO2RR current densities. d FEs of CH4 under different CO2 flow rates and corresponding SPCE of CO2 (pH = 3 ± 0.05). e Comparison of the FE and partial current density of CH4 with those of state-of-the-art catalysts. f Schematic diagram of the proton exchange membrane (PEM) electrolyzer. g Cell voltage of the PEM electrolyzer and corresponding FE of CH4 under various current densities. h Stability test of CO2RR PEM system under 200 mA cm−2. The resistance of the PEM electrolyzer was 4.5 ± 0.1 Ω, the pH of catholyte was 3 ± 0.05, the voltage of the PEM electrolyzer was obtained with 95% iR-correction, and the electrode area is 1 cm2. The error bars represent standard error of the mean and are made based on three fully separate and identical measurements. Source data are provided as a Source Data file.
To further optimize its electrochemical performance, the current density of OMe-Ph is found to increase further in a 0.2 M K2SO4 acidic electrolyte, while the Faradaic efficiency for methane slightly decreases, achieving a maximum FE of 64.9% at 500.0 mA cm−2 (Fig. 3b). Notably, the maximum partial current density for methane over OMe-PhCu electrode reaches 324.5 mA cm−2 (Fig. 3c) in acid electrolyte, demonstrating a competitive level7,34,35,36 (Fig. 3e). Additionally, at the same current density, CO production increases slightly, whereas hydrogen production decreases, suggesting that a lower K+ concentration within a specific range aids in reducing the overpotential for CH4 formation. This is primarily because the ion concentration modulates the interfacial water structure and local electric field37,38,39, which facilitates the optimized *CO and *H coverage for CH4 production.
Furthermore, the single-pass carbon efficiency (SPCE) of the OMe-PhCu catalyst was evaluated under varied CO2 flow rates. The SPCE of CO2 increases to 24.1% when the flow rate is reduced to 3 mL min−1 at 500.0 mA cm−2, with a corresponding CH4 FE of 38.9% (Fig. 3d). In addition, we constructed a proton exchange membrane (PEM) electrolyzer with hydrogen oxidation (HOR) as the anode reaction rather than the common oxygen evolution reaction (OER) (Fig. 3f and Supplementary Fig. 24). This PEM system demonstrates lower cell voltage and good stability at 200.0 mA cm−2 by maintaining considerable CH4 FE (Fig. 3e). Notably, the performance of the electrode could largely recover in the initial stage after rest for 12 h without electrolysis. The degradation of the electrolyzer was found to be partly attributed to the water flooding and salt precipitation within the catalyst layer (Supplementary Figs. 25, 26). The carbon deposition on the catalyst surface during methane generation from CO2RR may also deactivate the active sites40. The electrochemical performance of OMe-PhCu was further evaluated in an alkaline electrolyte system, achieving a peak FE of 62.7% at 500.0 mA cm−2, along with a partial current density of 344.4 mA cm−2 (Supplementary Fig. 27), demonstrating a broad pH versatility. The electrochemical performance of CO reduction was also investigated (Supplementary Fig. 28), revealing that CH4 is still the main product, which indirectly indicates that CO is a crucial intermediate in CO2 electrochemical reduction process. This finding also confirms the feasibility of our strategy to enhance CO2 electroreduction activity by regulating *CO adsorption.
To elucidate the stability of the copper coordination polymer, we conducted a series of operando and ex situ characterizations on the electrode materials before and post CO2RR. As observed from the in situ Raman spectra, the OMe-PhCu electrodes show no obvious changes over a wide potential range, demonstrating its exceptional electrochemical stability (Fig. 4a and Supplementary Fig. 29). Additionally, XPS (Fig. 4b and Supplementary Fig. 30) and XANES (Fig. 4c, d) analyses further reveal that copper remains in about +1 oxidation state with no formation of metallic copper species after CO2RR. This indicates that the copper alkyne complex structure could withstand the harsh acidic electroreduction environment within a certain potential range.
a In situ Raman spectra of CO2RR over OMe-PhCu electrode. b Cu 2p XPS spectra. c Ex situ XANES spectra. d The first-order derivatives of the XANES spectra in (c). e FTIR spectra. f XRD patterns. The electrodes after CO2RR were obtained at 200 mA cm−2 with 1 h electrolysis. Source data are provided as a Source Data file.
For the purpose of comparison, we chose two common copper compounds to investigate their reconstruction behavior during CO2RR (Supplementary Figs. 31, 32): Cu2-xS and copper phthalocyanine that are chemically stable in acidic solution. The results demonstrate that, Cu2-xS completely transforms quickly into metallic copper (Supplementary Fig. 33). Although copper phthalocyanine does not exhibit significant phase changes (Supplementary Fig. 34), extensive studies have shown that it was easily converted into nano copper clusters during CO2RR process41. The most contrasting results emerge from the post-CO2RR TEM analysis. OMe-PhCu catalyst retains its initial nanofiber morphology with negligible changes (Supplementary Fig. 35). However, under the same condition, the as-synthesized Cu2-xS nanocages underwent complete reconstruction into cracked nanocrystals after CO2RR (Supplementary Fig. 36), and the nanoroad/plate like CuPc catalyst evolves into stacked nanosheets (Supplementary Fig. 37).
Complementary spectra analysis further corroborates these findings. The characteristic structural IR bands of OMe-PhCu remains intact after CO2RR (Fig. 4e). Also, there are no change of ex situ XRD patterns for both CO2RR and CORR (Fig. 4f). These results are consistent with the cyclic voltammetry (CV) tests with no reduction peaks observed for OMe-PhCu cathode (Supplementary Fig. 38). Similarly, the PhCu and F-PhCu samples also demonstrate stable structural composition during CO2RR (Supplementary Fig. 39). The SEM images of the electrodes before and post CO2RR further visually depict the structural stability of the MOPs materials (Supplementary Fig. 40). The resistance of OMe-PhCu to reconstruction during prolonged operation (30 h) was further confirmed by XPS (Supplementary Fig. 41) and TEM (Supplementary Fig. 42) characterizations.
Overall, the catalytic activity of PhCu MOPs is effectively regulated through substituents functionalization, and a vivid depiction of stable Cuδ+ sites during CO2 electroreduction emerges, highlighting the significant role of σ-π dative bonding interactions.
Electrochemical mechanism investigation
The observation that PhCu MOPs exhibit high activity for CO2 electroreduction, with a high selectivity for methane and remarkable stability against structural reconstruction, which is not the case for the Cu2-xS and CuPc, prompting us to further interrogate the underling electrocatalytic mechanism of PhCu MOPs and clarify how the geometry and electronic structures of copper sites influence the CO2RR activity.
DFT calculations were performed to gain insights into the CO2RR mechanism over PhCu MOPs. Following methoxyl substitution, the energy difference in the potential-determining step (CO2 to *COOH) on OMe-PhCu is reduced by 0.23 eV (Fig. 5a and Supplementary Figs. 43–45), indicating increased reactivity toward CO2 reduction. The selectivity of CO2RR toward CH4 largely depends on the adsorption behavior of *CO. Our findings indicate that *CO adsorption on PhCu MOPs is highly dependent on the functional groups present (Supplementary Fig. 46). Compared to pristine H-PhCu, methoxyl functionalization strengthens *CO adsorption, while F-PhCu weakens it significantly, generally triggering its desorption and preventing further reduction.
a Free energy diagram of CO2RR over PhCu MOPs. b Adsorption energy of the 1st and 2nd CO molecule on Cu and OMe-PhCu catalysts. c Comparison of free energy of C1 and C2 pathway. In situ Raman spectra of CO2RR over (d) OMe-PhCu and (e) F-PhCu under different cathodic potentials. Source data are provided as a Source Data file.
As further revealed in Fig. 5b, the adsorption of the first *CO is rather strong (Ead = −0.85 eV) on metallic copper, and the adsorption of the second *CO on neighboring site is almost unaffected (Ead = −0.76 eV). Such phenomenon suggested that high *CO coverage could be achieved on Cu, which favors C-C coupling on neighboring sites to produce C2+ products. Comparatively, on OMe-PhCu, while the adsorption of the first *CO remains strong, the adsorption of the second *CO on adjacent copper site is weakened by more than 40%, making it tough for C-C coupling. Additionally, the energy barrier for coupling two *CO molecules to form a C2 product on OMe-PhCu was calculated to be 2.87 eV (Fig. 5c), which is ~0.17 eV higher than the energy required for the hydrogenation of a single *CO. This suggests that the C1 pathway is more energetically favorable on PhCu MOPs.
In situ Raman and attenuated total reflectance infrared spectra (ATR-IR) spectroscopy were further employed to probe the interaction between *CO and copper polymer. The peak at 1976 cm−1 that emerges at −0.6 V vs. RHE in the Raman spectra of OMe-PhCu (Fig. 5d) is ascribed to the atop-adsorbed *CO, as observed in the ATR-IR spectra (Supplementary Figs. 47, 48). This observation is expected, given that OMe-PhCu presents quasi-continuous copper sites. In contrast, in the Raman spectra of F-PhCu electrode (Fig. 5e), this vibration band is blue-shifted to around 1990 cm−1, indicating weaker *CO binding, consistent with DFT calculations. The stronger CO binding affinity generally indicates enhanced Lewis acidity42,43,44, more clearly reflecting the modulation effect of functionalization on the electronic structures of copper sites. The spectral characterization results confirm that the CO2-to-CH4 conversion on PhCu MOPs proceeds with *CO as the major intermediate.
These results suggest that the quasi-continuous one-dimensional cuprous array in OMe-PhCu provides optimal sites for the conversion of CO2 to CH4, which aligns well with our experimental findings. Combined with DFT and empirical results, it is evident that the electronic structures of the copper sites are finely tuned through adjusting σ and d-π backbonding interaction, resulting in enhanced activity and selectivity for methane production from CO2RR.
Versatility of integrated catalyst design
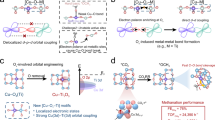
We further demonstrated the adaptability of our strategy for designing integrated catalysts with Cuδ+/Cu0 interface for other value-added chemicals. Our study results show that *COatop intermediate species mainly form over alkyne Cu(I) sites during CO2RR, leading to the formation of CH4 as the primary C1 product. In contrast, bridge or atop-adsorbed *CO has been found to generate on Cu(0) sites11. Thus, their composite may provide the opportunity to regulate the adsorption behavior of intermediates, which is crucial for multicarbon production14,45. Additionally, Cu-based materials have been developed for efficient nitrate electroreduction (NO3RR) to synthesize ammonia. For copper-based catalysts during NO3RR, nitrites (NO2−) tend to accumulate excessively, and the activation of water molecules thus becomes more critical for the hydronation of N-containing intermediates, which is closely related to the valence state of copper species46,47.
These findings are reminiscent of our developed alkyne-Cuδ+ catalysts. Accordingly, we modified alkyne-Cuδ+ sites on Cu0 surface to get the Cuδ+/Cu0 composite (Supplementary Fig. 49) for the electrosynthesis of multi-carbon chemicals and ammonias (Fig. 6a). To confirm the successful incorporation of alkyne-Cuδ+ sites onto the nano copper catalyst, XRD and FTIR spectra were collected. As shown in the XRD patterns (Supplementary Fig. 50), the nano copper catalyst consists of cuprous oxide (Cu2O) and metallic copper. The defective Cu+ species within nano copper would easily coordinate with alkyne groups to form the final metal organic polymer. Although no diffraction peaks corresponding to OMe-PhCu were observed, FTIR spectroscopy revealed characteristic signals of OMe-PhCu species on the obtained material (Supplementary Fig. 51). These results indicate the successful grafting of a small amount of alkyne-Cuδ+ sites onto the nano copper catalyst.
a Schematic illustration of the construction of alkyne-Cuδ+/Cu0 interfaces for C2+ and ammonia production. b FEs of ethylene over Cu0 and Cuδ+/Cu0 catalysts in 1 M KOH electrolyte (pH = 13.8 ± 0.1). c FE of NH3 over Cu0 and Cuδ+/Cu0 catalysts in 0.5 M K2SO4/0.1 M KNO3 electrolyte (pH = 6.5 ± 0.1, potential without iR-correction), the electrode area was 1 cm2. The error bars represent standard error of the mean and are made based on three fully separate and identical measurements. Source data are provided as a Source Data file.
As shown in Fig. 6b and Supplementary Fig. 52, the alkyne-Cuδ+/Cu0 catalyst exhibits higher C2H4 selectivity in an alkaline environment, which was also proved through DFT calculations (Supplementary Figs. 53, 54). The FE of NH3 was also increased in a neutral electrolyte (Fig. 6c and Supplementary Fig. 55). We found that the modification of Cuδ+ species effectively facilitated the dissociation of H2O to *H (Supplementary Figs. 56, 57), as evidenced by the lowered energy barrier. The enhanced yields of ethylene and ammonia convincingly demonstrate the synergy between Cuδ+ and Cu0 sites, which can well extend to other electro-hydrogenation or chemically catalytic reactions that are sensitive to copper oxidation states.
Discussion
In this work, we reported an anti-reconstructing strategy for Cu-based catalysts with tunable activity for CO2RR through σ-π dative bonding. The alkyne-Cuδ+ sites demonstrate highly selective CO2-to-methane conversion in acidic electrolyte, with a competitive CH4 FE of 68.8% and a partial current density of 324.5 mA cm−2. The increased covalent binding interaction between Cuδ+ and alkyne ligands leads to a good reconstruction-resistant ability during CO2RR. We demonstrate that intentional manipulation of the Lewis basicity of coordination ligands flexibility regulates the electronic structure of copper sites through the tug-of-war between σ and π backbonding interactions. Furter, the expanded integration of Cu0 and alkyne-Cuδ+ interface is quite general for the electrochemical reduction of carbon dioxide into multi-carbons and nitrate into ammonias. Our catalyst design principle frames a guideline to construct stable and active copper electrocatalysts and beyond.
Methods
Chemicals and materials
Cupric Acetate Monohydrate (Cu(OAc)2 H2O, 99%), sodium hydroxide (NaOH, 96%), potassium hydroxide (KOH, 85%), potassium bicarbonate (K2SO4, 99%) and copper(II) nitrate trihydrate (Cu(NO3)2 3H2O, 99%) were purchased from Sinopharm Chemicals Co., Ltd. Phenylacetylene (97%), 4-ethynylanisole (99%), 2-ethynyl-1,3-difluoro-benzene (97%), and methanol (CH3OH, 99.5%) were purchased from Shanghai Aladdin Biochemical Technology Co., Ltd. Sulfuric acid (H2SO4, 95 ~ 98%) and hydrochloric acid (HCl, 35 ~ 37%) were purchased from Xilong Scientific Co., Ltd. All the above reagents were in analytical grade and used without further purification. Deionized water with a resistivity of 18.2 MΩ cm was acquired from Milli-Q ultrapure water system. Quaternary ammonia poly(N-methyl-piperidine-co-p-terphenyl) (QAPPT) anion exchange membrane (AEM) was purchased from Huizhou EVE Hydrogen Energy Co., LTD. Ti felt (250 μm), and iridium oxide (IrO2, 99.9%) were purchased from Ningbo Zhongkeke Innovative Energy Technology Co., Nafion 211 membrane (25.4 μm) and PtRu/C alloy (40 wt% Pt, 20 wt% Ru) catalyst was purchased from Shanghai Hesen Import&Export Co., Ltd.
Synthesis of Cu2(OH)3OAc·H2O
Cu(OAc)2⋅H2O (4.00 g, 20.1 mmol) was dissolved in 200 mL of water and stirred at room temperature. NaOH (200 mL, 0.1 M) was then slowly added with vigorous stirring. The mixture was stirred for 7 days, then centrifuged and washed five times at 5000 rpm for 5 min with deionized water. This process was repeated once with ethanol to yield the product as a pale green solid.
Synthesis of MOPs
0.06 g Cu2(OH)3OAc·H2O was first dispersed in 10 mL methanol in a 15 mL glass pressure bottle, and then phenylacetylene (0.08 g) was added to the above solution. Cu2+ would be slowly reduced to Cu+, which then coordinated with phenylacetylene to form the final polymer. The reaction was continued at 32 °C for 48 h. After reaction, the product was centrifuged (5000 rpm, 5 min) and washed four times with ethanol and chloroform respectively, then vacuum drying at 60 °C for 12 h. OMe-PhCu and F-PhCu were prepared in a similar way except changing phenylacetylene to 4-ethynylanisole and 2-ethynyl-1,3-difluoro-benzene, respectively.
Physical characterization
The crystal phases of the samples were determined using an X-ray diffractometer (Rigaku Smartlab) with Cu Kα radiation. The Raman spectra was obtained via a Renishaw inVia Raman microscope with a 633 nm laser. The Infrared Spectroscopy was acquired on Nicoletls10 (Thermo Nicolet Corporation). Transmission electron microscopy (TEM) images were obtained using a Hitachi H-7700 microscope operated at an acceleration voltage of 100 kV. Scanning electron microscope (SEM) images were acquired with a Hitachi S-4800 microscope at 5.0 kV. High-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) and elemental mapping were conducted using a JEOL ARM-200F microscope equipped with a spherical aberration corrector, operated at 200 kV. X-ray photoemission spectroscopy was measured on a Thermo Scientific ESCALAB 250Xi, and the binding energies of the samples were calibrated by aligning Au foil 4 f 7/2 to 84.0 eV. Cu K-edge XAS spectra were collected at the BL14W1 beamline of the Shanghai Synchrotron Radiation Facility (SSRF, 3.5 GeV, 350 mA, Si (311) double-crystal) using a Lytle detector. The acquired data was processed using the ATHENA module of the IFEFFIT software package. Calibration, background subtraction, and normalization were performed following standard procedures. The quantification of copper and carbon/hydrogen (C/H) elements was performed using inductively coupled plasma optical emission spectroscopy (ICP-OES) and elemental analysis (EA), respectively.
Preparation of working and counter electrodes
Typically, 20 mg of prepared catalyst and 40 μL of Nafion D520 perfluorinated resin (5 wt%, Dupont) solution were mixed and sonicated in 10 mL of absolute ethanol. The catalyst ink was then airbrushed onto the gas diffusion layer (AvCarb GDS5130) to achieve a catalyst loading of approximately 0.01 mg cm−2. The catalyst loading was maintained at approximately 0.5 mg cm−2 for post-CO2RR cathode characterizations to get clear signal, unless stated otherwise. The counter electrode was similarly prepared using a Ti fiber substrate, with an IrO2 loading of approximately 2 mg cm−2. For all ex situ tests, the geometric area of the cathode was fixed at 1 cm2.
Electrochemical measurements
All the electrochemical measurements were performed on a high-current electrochemical station (Lvium CompactStat) using a flow cell under ambient pressure and room temperature (25 °C). Hg/Hg2SO4 and Hg/HgO electrodes were used as reference electrodes in acidic and alkaline electrolytes, respectively. The potential versus the reversible hydrogen electrode (vs. RHE) were calibrated using the following formula: E (RHE) = E (Hg/Hg2SO4 + 0.656 + 0.059 pH), E (RHE) = E (Hg/HgO) + 0.098 + 0.059 pH. An IrO2/Ti fiber was used as the counter electrode. An anion-exchange membrane (QAPPT, 25 μm) was used to separate the cathode and anode chambers. The membrane was soaked in 1 M KOH solution at 60 °C for 24 h before use. Unless stated otherwise, all CO2 electroreduction (CO2RR) and CO electroreduction (CORR) measurements were conducted in an electrochemical flow cell setup. The inlet flow rate of CO2/CO was controlled at 35 sccm by a mass flow meter. 100 mL 0.1 M K2SO4 (pH = 3 ± 0.05) was used as the catholyte. The outlet flow rate was measured by a soap film flowmeter. 100 mL 0.5 M K2SO4 (pH = 1 ± 0.05) was adopted as the anolyte. The electrolyte was freshly prepared before testing by dissolving specific chemicals into deionized water, and then the pH was adjusted with concentrated H2SO4 under monitoring by a pH meter (Mettler-Toledo, FE28-Standard). The electroreduction of nitrate was conducted in a H-type cell with 0.5 M K2SO4/0.1 M KNO3 solution as catholyte (pH = 6.4 ± 0.1).
Construction of proton exchange membrane (PEM) electrolyzers
The CO2RR catalytic activity was further investigated using a home-made PEM cell with a cathode area of 1 cm × 1 cm. The cathode was prepared with the same method as described above. In order to increase the cathode stability, Vulcan XC 72 R carbon black was added into the catalyst ink, and this may lead to a slight difference of the selectivity of the products. Specifically, 20 mg of prepared catalyst, 40 mg Vulcan XC 72 R carbon black and 120 μL of Nafion D520 perfluorinated resin (5 wt%, Dupont) solution were mixed and sonicated in 10 mL of absolute ethanol. The catalyst ink was then airbrushed onto the gas diffusion layer (AvCarb GDS5130) to achieve a total loading of 0.1 mg cm−2, the area of the electrode was fixed at 1 cm2. A thin PTFE membrane (15 μm) was hot-pressed onto the backside of the GDL to increase its hydrophobicity. PtRu/C (40 wt% Pt, 20 wt% Ru) was used as anode catalyst. Typically, 10 mg PtRu/C and 100 μL of Nafion D520 perfluorinated resin (5 wt%, Dupont) solution were mixed and sonicated in 5 mL of absolute ethanol. The catalyst ink was then airbrushed onto the gas diffusion layer (JNT30-A6H) to achieve a total catalyst loading of about 0.2 mg cm−2. The Nafion 211 membrane (24.5 μm) was pretreated by heating in 5 wt% H2O2 at 80 °C for 1 h, rinsing with deionized water for 30 min, heating again in 5 wt% H2SO4 at 80 °C for 1 h, and finally soaking in deionized water for 30 min prior to use. Then the anode GDL was hot pressed onto the Nafion membrane to get the membrane electrode. 500 mL 0.2 M K2SO4 (pH = 3 ± 0.05) was adopted as the catholyte with a flow rate of 4 mL min−1 and refreshed every hour. Humidified hydrogen was obtained from the hydrogen generator. The flow rate of CO2 and H2 were kept at 20 sccm and 40 sccm, respectively. The electrochemical tests of the PEM electrolyzer were conducted on a Linear power supply (A-BF, SS-L303SPD). The cell voltage of the PEM system was obtained via proper iR corrected. The resistance was determined by electrochemical work station (CHI660E).
Determination of CO2 reduction products
Gaseous products were detected using online gas chromatography (GC 2014, Shimadzu, Japan). The flame ionization detector (FID) was employed to detect CH4, C2H4, and CO, while the thermal conductivity detector (TCD) was used for H2 detection. The liquid products were analyzed by High Performance Liquid Chromatography (HPLC, Waters Alliance 2695) equipped with a refractive index detector. An Xtimate Sugar-H column (5 μm, 7.8 × 300 m) was used for product separation. 5 mM H2SO4 was used as the mobile phase with a constant flow rate of 0.6 mL min−1 at 60 °C. Product identification and concentration calculations were performed using calibration curves derived from standard solutions. The reported faradaic efficiency of the product is based on the mean value for three independent measurements if error bar is labeled.
The Faradaic efficiency (FE) is calculated by the following equation:
Here, z represents the number of electrons transferred in the reaction, n denotes the number of moles of product formed, F is the Faraday constant, and Q is the total charge passed during CO2RR. The partial current density was calculated by multiplying the Faradaic efficiency by the total current density.
The SPCE of CO2RR was calculated according to the following equation:
where j is the partial current density of a specific product from CO2RR, z represents the number of electrons transferred in the reaction and F is the Faradaic constant.
In situ spectra measurements
At low current densities, acidic electrolytes result in extremely low CO2 electroreduction efficiency in H-type cells. As the local environment would still rapidly shifts to alkaline conditions during effective CO2RR in acidic electrolytes. Therefore, we conducted in situ Raman and attenuated total reflectance infrared (ATR-IR) measurements in CO2-saturated 0.5 M KHCO3 (pH = 7.2 ± 0.1) to simulate real-world acidic reaction conditions and study the adsorption behavior of reaction species. The reference electrode was Ag/AgCl electrode. The potential was calibrated to Reversible Hydrogen Electrode (RHE) obtained without iR correction.
In situ Raman measurements
In situ Raman measurements were performed using a Renishaw inVia Raman microscope with a 633 nm laser at 10% intensity in a customized H-type cell. The working electrode was prepared by applying the catalyst ink onto a circular Ti electrode (6 mm diameter). Each spectrum was acquired by integrating twice for 60 seconds per integration. The applied potentials ranged from open circuit potential to −1.1 V vs. RHE. Spectra were collected under CO2 flow at a rate of 20 sccm.
In situ attenuated total reflectance infrared spectra (ATR-IR)
ATR-IR measurements were performed using a Bruker Vertex 70 v spectrometer equipped with an MCT detector cooled by liquid nitrogen. The catalyst, with a loading of 1 mg mL−1, was drop-casted onto an Au-coated circular Si prism (1.2 cm diameter) to enhance the signal. IR spectra were collected in a CO2-saturated 0.5 M KHCO3 solution at potentials ranging from open circuit potential to −1.7 V vs. RHE.
DFT calculations
The electronic and geometric properties of MOPs, as well as CO2RR thermodynamics were calculated using DFT. The DFT calculations on MOPs were performed by using the Vienna ab initio simulation program (VASP), with projected augmented wave (PAW)48 pseudopotentials (energy cutoff of 400 eV) and the Perdew-Burke-Ernzerhof (PBE) functional49. All structures were modeled using single layer of MOPs31 periodicity in the x and y directions (18.1 × 14.9 × 30.6 Å), and were visualized using VESTA software50. Neighboring slabs were separated by a vacuum of 20 Å to avoid spurious self-interactions. The convergence criteria for self-consistent calculation were 10−5 eV energy difference, and 0.03 eV Å−1 force change per atom. Gamma-only grid was used for these models All of the electronic energies were corrected for zero-point energy (ZPE) contributions.
O2, H2, H2O, CO, and CO2 molecules are considered to be in gas phase at room temperature and under ambient pressure. As indicated earlier51, in this free energy analysis, “H+ + e−” has been assumed to be in equilibrium with 1/2 H2, at pH = 0 and 0 V potential in SHE.
The change in Gibbs free energy (ΔG) for the elementary steps was calculated by the following equation
where ΔE is the change in calculated energies from DFT; ΔZPE and ΔS are the change in zero-point energy and entropy obtained from vibrational frequencies; T is 298.15 K; n represents the number of transferred electrons, and U is the electrochemical potential.
Data availability
All the data supporting the findings of this study are available within the article and its Supplementary Information files. Source data are provided with this paper.
References
Dinh, C. T. et al. CO2 electroreduction to ethylene via hydroxide-mediated copper catalysis at an abrupt interface. Science 360, 783–787 (2018).
Overa, S. et al. Enhancing acetate selectivity by coupling anodic oxidation to carbon monoxide electroreduction. Nat. Catal. 5, 738–745 (2022).
Huang, J. E. et al. CO2 electrolysis to multicarbon products in strong acid. Science 372, 1074–1078 (2021).
Chen, Y. et al. Efficient multicarbon formation in acidic CO2 reduction via tandem electrocatalysis. Nat. Nanotechnol. 19, 311–318 (2023).
Zhao, Y. et al. Conversion of CO2 to multicarbon products in strong acid by controlling the catalyst microenvironment. Nat. Synth. 2, 403–412 (2023).
Dong, J. et al. Continuous electroproduction of formate via CO2 reduction on local symmetry-broken single-atom catalysts. Nat. Commun. 14, 6849 (2023).
Fan, M. et al. Single-site decorated copper enables energy- and carbon-efficient CO2 methanation in acidic conditions. Nat. Commun. 14, 3314 (2023).
Li, X. et al. Confinement of an alkaline environment for electrocatalytic CO2 reduction in acidic electrolytes. Chem. Sci. 14, 5602–5607 (2023).
Kim, D. et al. Acid-stable Cu cluster precatalysts enable high energy and carbon efficiency in CO2 electroreduction. J. Am. Chem. Soc. 146, 27701–27712 (2024).
Cui, H. et al. Effect of surface corrosion on the zeta potential of copper in acidic solutions. Surf. Interf. 54, 105291 (2024).
Chou, T.-C. et al. Controlling the oxidation state of the Cu electrode and reaction intermediates for electrochemical CO2 reduction to ethylene. J. Am. Chem. Soc. 142, 2857–2867 (2020).
Cheng, D. et al. The nature of active sites for carbon dioxide electroreduction over oxide-derived copper catalysts. Nat. Commun. 12, 395 (2021).
Kortlever, R., Shen, J., Schouten, K. J. P., Calle-Vallejo, F. & Koper, M. T. M. Catalysts and reaction pathways for the electrochemical reduction of carbon dioxide. J. Phys. Chem. Lett. 6, 4073–4082 (2015).
Zhan, C. et al. Revealing the CO coverage-driven C-C coupling mechanism for electrochemical CO2 reduction on Cu2O nanocubes via operando Raman spectroscopy. ACS Catal. 11, 7694–7701 (2021).
Lum, Y. & Ager, J. W. Stability of residual oxides in oxide-derived copper catalysts for electrochemical CO2 reduction investigated with 18O labeling. Angew. Chem. Int. Ed. 57, 551–554 (2018).
Yang, Y. et al. Operando studies reveal active Cu nanograins for CO2 electroreduction. Nature 614, 262–269 (2023).
Okatenko, V. et al. Alloying as a strategy to boost the stability of copper nanocatalysts during the electrochemical CO2 reduction reaction. J. Am. Chem. Soc. 145, 5370–5383 (2023).
Zhou, Y. et al. Dopant-induced electron localization drives CO2 reduction to C2 hydrocarbons. Nat. Chem. 10, 974–980 (2018).
Albertini, P. P. et al. Hybrid oxide coatings generate stable Cu catalysts for CO2 electroreduction. Nat. Mater. 23, 680–687 (2024).
Yang, P.-P. et al. Protecting copper oxidation state via intermediate confinement for selective CO2 electroreduction to C2+ fuels. J. Am. Chem. Soc. 142, 6400–6408 (2020).
Wu, H. et al. Improved electrochemical conversion of CO2 to multicarbon products by using molecular doping. Nat. Commun. 12, 7210 (2021).
Zhao, B. et al. C60-stabilized Cu+ sites boost electrocatalytic reduction of CO2 to C2+ products. Adv. Energy Mater. 13, 2204346 (2023).
Liu, K. et al. Atomic overlayer of permeable microporous cuprous oxide on palladium promotes hydrogenation catalysis. Nat. Commun. 13, 2597 (2022).
Zheng, J. et al. Ambient-pressure synthesis of ethylene glycol catalyzed by C60-buffered Cu/SiO2. Science 376, 288–292 (2022).
Wu, H. et al. Selective and energy-efficient electrosynthesis of ethylene from CO2 by tuning the valence of Cu catalysts through aryl diazonium functionalization. Nat. Energy 9, 422–433 (2024).
Luo, M. et al. Coordination polymer electrocatalysts enable efficient CO-to-acetate conversion. Adv. Mater. 35, 2209567 (2023).
Kong, S. et al. Delocalization state-induced selective bond breaking for efficient methanol electrosynthesis from CO2. Nat. Catal. 6, 6–15 (2023).
Braunschweig, H., Damme, A., Dewhurst, R. D. & Vargas, A. Bond-strengthening π backdonation in a transition-metal π-diborene complex. Nat. Chem. 5, 115–121 (2013).
Frenking, G. & Fröhlich, N. The nature of the bonding in transition-metal compounds. Chem. Rev. 100, 717–774 (2000).
Wei, Z. et al. Self-assembled polymer phenylethnylcopper nanowires for photoelectrochemical and photocatalytic performance under visible light. Appl. Catal. B Environ. 226, 616–623 (2018).
Liang, Q., Chang, X., Su, Y. -q, Mugo, S. M. & Zhang, Q. Mechanistic investigation on copper–arylacetylide polymerization and sensing applications. Angew. Chem. Int. Ed. 60, 18014–18021 (2021).
Krikor, H., Rotti, M. & Nagels, P. Highly conducting iodine-doped poly(copper-yne) from phenylacetylene. Synth. Met 21, 353–359 (1987).
Garbusova, I. A., Alexanjan, V. T., Leites, L. A., Golding, I. R. & Sladkov, A. M. Vibrational spectra of phenylethynylcopper(I). J. Organomet. Chem. 54, 341–344 (1973).
Xu, K. et al. Favoring CO intermediate stabilization and protonation by crown ether for CO2 electromethanation in acidic media. Angew. Chem. Int. Ed. 62, e202311968 (2023).
Zhu, Z. et al. Covalent organic framework ionomer steering the CO2 electroreduction pathway on Cu at industrial-grade current density. J. Am. Chem. Soc. 146, 1572–1579 (2024).
Xu, Z. et al. Steering the selectivity of electrochemical CO2 reduction in acidic media. ChemCatChem 14, e202200052 (2022).
Li, J. et al. Constraining CO coverage on copper promotes high-efficiency ethylene electroproduction. Nat. Catal. 2, 1124–1131 (2019).
Zhang, H., Gao, J., Raciti, D. & Hall, A. S. Promoting Cu-catalysed CO2 electroreduction to multicarbon products by tuning the activity of H2O. Nat. Catal. 6, 807–817 (2023).
Gu, J. et al. Modulating electric field distribution by alkali cations for CO2 electroreduction in strongly acidic medium. Nat. Catal. 5, 268–276 (2022).
DuanMu, J.-W. et al. Investigation and Mitigation of Carbon Deposition over Copper Catalyst during Electrochemical CO2 Reduction. Precis. Chem. 2, 151–160 (2024).
Weng, Z. et al. Active sites of copper-complex catalytic materials for electrochemical carbon dioxide reduction. Nat. Commun. 9, 415 (2018).
Zhou, H. et al. Engineering the Cu/Mo2CTx (MXene) interface to drive CO2 hydrogenation to methanol. Nat. Catal. 4, 860–871 (2021).
Cox, D. F. & Schulz, K. H. Interaction of CO with Cu+ cations: CO adsorption on Cu2O(100). Surf. Sci. 249, 138–148 (1991).
Chen, A. et al. Structure of the catalytically active copper-ceria interfacial perimeter. Nat. Catal. 2, 334–341 (2019).
Li, F. et al. Molecular tuning of CO2-to-ethylene conversion. Nature 577, 509–513 (2019).
Zhang, L.-H. et al. Dopant-induced electronic states regulation boosting electroreduction of dilute nitrate to ammonium. Angew. Chem. Int. Ed. 62, e202303483 (2023).
Feng, T. et al. Selective electrocatalytic reduction of nitrate to dinitrogen by Cu2O nanowires with mixed oxidation-state. Chem. Eng. J. 433, 133495 (2022).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B Condens. Matter 50, 17953–17979 (1994).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Momma, K. & Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 44, 1272–1276 (2011).
Nørskov, J. K. et al. Origin of the overpotential for oxygen reduction at a fuel-cell cathode. J. Phys. Chem. B 108, 17886–17892 (2004).
Acknowledgements
This study was financially supported by the Science and Technology Innovation Foundation of Laoshan Laboratory (LSKJ202205400), the National Science Fund for Distinguished Young Scholars (No. 52025133), National Key R&D Program of China (No. 2022YFE0128500), Young Scientists Fund of the National Natural Science Foundation of China (No. 22409004). The authors thank the photoemission photoendstations BL14W1 in the Shanghai Synchrotron Radiation 466 Facility (SSRF) for the help with characterizations.
Author information
Authors and Affiliations
Contributions
S.J.G. conceived and supervised the project. Z.Y.Q. and Y.J.T. designed and performed the experiments, collected and analyzed the data. G.K.H. carried out the DFT simulation. N.Y., S.G.W., and Z.L. helped to perform the Operando Raman measurements. Q.Z.H. and F.L. carried out the SEM measurements. Y.G., L.L., K.W., H.Y.G., and M.C.L. participated in part of the mechanism analysis. C.S.S. conducted some TEM measurements. Z.Y.Q. prepared the manuscript. All authors took part in the discussion of the results and assisted during manuscript preparation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Xinlong Tian and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Qian, Z., Han, G., Tan, Y. et al. σ-π dative bond stabilizing copper active site drives CO2 electrocatalysis to hydrocarbon. Nat Commun 16, 11265 (2025). https://doi.org/10.1038/s41467-025-66140-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-66140-w