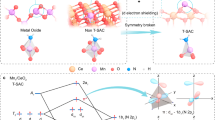
Abstract
Overcoming the trade-off between activity and selectivity has long been a challenge in catalytic reactions. Dual-atom catalysts (DACs) exhibit exceptional catalytic performance in cascade catalysis, owing to the synergistic effects of distinct active sites, which make them particularly promising for enhancing catalytic selectivity. Here, we present dual-atom Ir-Wx/CeO2 catalysts that integrate both oxidation (Ir) and reduction (W) sites for the selective catalytic oxidation of ammonia, a major precursor of air pollutants. Comprehensive characterizations revealed that Ir atoms were embedded on the CeO2 planes in single-atom form, while W sites were anchored on the CeO2 surface, forming Ir-W dimer structures. Operando studies and theoretical calculations demonstrated that NH3 was oxidized on Ir sites, producing NO, which then reacted with NH3 on W sites via selective catalytic reduction (SCR) to generate N2 and H2O. The synergistic effect of the Ir-W dual-atom dimer significantly enhanced low-temperature activity (≥ 92% at 200 °C) and high-temperature selectivity (≥ 92% at 300 °C) on the Ir-W7/CeO2 catalyst. Furthermore, this dual-atom strategy extends to Ir-Mo/CeO2 and Ir-Nb/CeO2 catalysts, demonstrating broad applicability. These findings highlight the potential of DACs for the rational design and application in various cascade catalytic reactions.
Similar content being viewed by others
Introduction
Single-atom catalysts (SACs), which consist of atomically dispersed metal atoms as active centers, have garnered significant attention due to their high atomic utilization efficiency, exceptional catalytic activity, and improved stability1,2,3,4,5. As a result, SACs have been widely applied in a range of catalytic processes, including thermochemical, electrochemical, and photochemical conversions6,7,8,9. However, their performance in complex reactions remains constrained by the linear relationship between the adsorption energies of reaction intermediates10,11,12,13, a limitation inherent to the isolated single-site nature, which struggles to stabilize multiple reactants simultaneously14,15. While increasing metal loading to create dense SACs could be a potential solution16,17,18, excessive metal density may lead to metal aggregation, ultimately causing significant catalyst deactivation19,20.
In contrast, dual-atom catalysts (DACs), consisting of paired homonuclear or heteronuclear metal sites21,22, offer spatially proximate yet isolated active centers capable of cooperative interactions23, enabling regulate reactant activation and intermediates formation/desorption as needed24,25. The synergistic interactions between the two metal atoms in DACs, facilitated by their sub-nanometer proximity, has been shown to significantly enhance catalytic performance, even in the absence of direct bonding26. By coupling two distinct active sites, DACs overcome the limitations of SACs, thus rendering them highly suitable for cascade catalysis, where they not only efficiently facilitate sequential intermediate steps but also simplify reaction processes26. This approach has enhanced activity and selectivity in various cascade reactions, including methane dry reforming27, selective hydrogenation28, simultaneous purification of hydrocarbon and nitrogen oxides (NOx)29, and three-way catalysis30.
Selective catalytic oxidation of ammonia (NH3-SCO) into N2 and H2O represents a promising approach for NH3 abatement in exhaust purification31,32. However, ammonia oxidation catalysts (AOCs) usually exhibit a ‘seesaw relationship’ between activity and selectivity, in which the enhancement of low-temperature activity often comes at the expense of high-temperature selectivity33,34. For example, noble metal catalysts excel in low-temperature activity but suffer from limited N2 selectivity due to over-oxidation35,36, while transition metal catalysts tend to achieve high N2 selectivity but lack sufficient activity at lower temperatures37,38. Researchers have explored numerous approaches to address this trade-off, including optimizing the metal-support interaction38, constructing dual-layer catalyst structures39, and synthesizing bimetallic catalysts containing both noble and transition metals40. Our recent work demonstrated the efficacy of a bifunctional Pt/Cu-SSZ-13 catalyst for ammonia catalytic oxidation, achieving high selectivity by integrating both an NH3 oxidation component and a selective catalytic reduction (SCR) component41. Therefore, coupling NH3 oxidation and NO reduction reactions represents an important and effective strategy to improve both the activity and selectivity of SCO catalysts.
Our previous studies have demonstrated that the single-atom Ir/CeO2 catalyst exhibits superior low-temperature activity in ammonia oxidation; however, it also produces significant amounts of NOx, which unfortunately, leads to reduced N2 selectivity34. In the present study, we designed and synthesized a dual-atom Ir–W/CeO2 catalyst, consisting of oxidation site (Ir–O–Ce) and reduction site (W–O–Ce), using a wet chemical method for efficient ammonia catalytic oxidation. On the Ir–Wx/CeO2 catalysts, Ir and W atoms formed highly dispersed dimer structures, which, through their synergistic interaction, facilitated the NH3–SCO process via a cascade mechanism. Specifically, NH3 was adsorbed and deeply oxidized on the single-atom Ir–O–Ce sites, resulting in the formation of N2 and NOx. The NOx then further reacted with adsorbed NH3 on the adjacent single-atom W–O–Ce sites, leading to the production of N2 and H2O via the SCR reaction, thereby achieving high activity and enhanced N2 selectivity. Additionally, the dual-atom strategy demonstrated broad applicability, as comparable performance improvements were achieved when other acidic metals, such as Nb and Mo, were employed as co-catalysts. This work highlights the significant potential of dual-atom catalysts in cascade catalysis, providing valuable insights that advance the design of high-performance DACs capable of overcoming the long-standing trade-off between activity and selectivity in conventional thermal catalysis, thereby contributing to the development of more efficient and sustainable catalytic processes.
Results
Structural characterizations
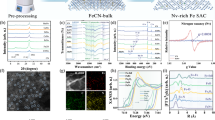
Defective CeO2 was prepared by calcining cerium nitrate at a relatively low temperature (350 °C), which induced the formation of abundant surface defects on the CeO2 support (Supplementary Fig. 1)42,43. These defects, in turn, facilitated the effective trapping and anchoring of single-atom metals, thereby enhancing the catalytic performance2,44. The Ir/CeO2 catalyst was then prepared using this defective CeO2 support through a wet chemical method, and the Ir–Wx/CeO2 were prepared similarly through the co-impregnation of Ir and W species. The defective CeO2 exhibited a Brunauer-Emmett-Teller (BET) specific surface area of 65.8 m2 g−1, and the introduction of Ir and W species did not affect the specific surface area (Supplementary Table 1). Inductively coupled plasma optical emission spectrometer (ICP-OES) analysis showed that the Ir ( ~ 0.8 wt%) and W contents on Ir–Wx/CeO2 catalysts were basically consistent with the design values (Supplementary Table 1). X-ray diffraction (XRD) patterns showed characteristic diffraction peaks of the fluorite-type CeO2 phase (PDF#43-1002) in all CeO2-supported samples with no distinct signals from Ir or W species, suggesting their high dispersion (Supplementary Fig. 2). High-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) images and corresponding inverse fast Fourier transform (IFFT) profiles showed that single Ir atoms were embedded on the CeO2 (111) planes of the Ir/CeO2 sample (Fig. 1a, Supplementary Fig. 3). After the incorporation of W species, numerous dual-atom dimers, were observed on the Ir–W3/CeO2, with each dimer consisting of two atoms, exhibiting an atomic distance ranging from 2.5 to 3.7 Å (Fig. 1b, Supplementary Figs. 4, 5). These dimers were mainly composed of Ir and W atoms, which appeared with relatively brighter and darker contrasts, respectively, due to their differing atomic weights. For the Ir–W7/CeO2 sample, which contained a higher W content, the number of dual-atom dimers increased significantly and remained clearly visible, with the average atomic distance maintained at 2.9 Å (Fig. 1c, Supplementary Figs. 6, 7). Notably, energy dispersive spectroscopy (EDS) provided direct visual evidence of Ir–W dimer formation on the CeO2 surface (Supplementary Figs. 5, 7). Additionally, the W/Ir mass ratios from EDS closely matched theoretical values based on ICP-OES analysis (Supplementary Table 2), further supporting the preferential formation of Ir–W dimers. Furthermore, no signals corresponding to WO3 lattice or WO3 nanoparticles were observed in any of these samples, indicating the W species were highly dispersed on the CeO2 surface. Notably, electron paramagnetic resonance (EPR) spectra revealed abundant Ce3+ defects in CeO2 (Supplementary Fig. 8)45,46. Ir incorporation markedly reduced the Ce3+ concentration in Ir/CeO2, whereas subsequent W addition had little further effect in Ir–W7/CeO2, indicating that Ir atoms preferentially occupied Ce defect sites, while W species were primarily anchored on the CeO2 surface.
HAADF-STEM images of Ir/CeO2 (a), Ir–W3/CeO2 (b), and Ir–W7/CeO2 (c) with the corresponding IFFT profiles. Normalized XANES at Ir L2-edge (d) and W L3-edge (g) for Ir–Wx/CeO2 catalysts. k2-weighted Fourier transform spectra from EXAFS at Ir L2-edge (e) and W L3-edge (h) for Ir–Wx/CeO2 catalysts. Wavelet transform (WT) contour plots of the k2-weighted Ir L2-edge (f) and W L3-edge (i) EXAFS for the Ir–W7/CeO2 catalyst.
X-ray absorption near-edge structure (XANES) spectra at the Ir L2-edge indicated that the white line of the Ir/CeO2 sample was close to that of IrO2, suggesting the Ir species were mainly present in the Ir4+ state (Fig. 1d). The introduction of W species did not significantly influence the oxidation state of the Ir species on the Ir–W3/CeO2 and Ir–W7/CeO2 samples (Fig. 1d, Supplementary Fig. 9a). Extended X-ray absorption fine structure (EXAFS) spectra without phase correction showed that, for the Ir/CeO2 sample, a primary peak at 1.55 Å corresponded to the first-shell Ir–O coordination, while a peak at 2.64 Å was attributed to Ir–O–Ce in the second shell. Notably, no Ir–Ir or Ir–O–Ir signals were detected in the Ir/CeO2 sample, further confirming that the Ir species existed in a single-atom state. For the Ir–W7/CeO2 sample, the Ir–O peak remained at 1.55 Å, but the Ir–O–Ce shifted slightly to 2.61 Å (Fig. 1e), and the same trend was also observed in the Ir–W3/CeO2 sample (Supplementary Fig. 9b)47. Wavelet transform (WT) EXAFS analysis further confirmed that the introduction of W species did not significantly influence the Ir–O and the Ir–O–Ce coordination on the Ir–W3/CeO2 and Ir–W7/CeO2, while slightly shortened the Ir–O–Ce distance (Fig. 1f, Supplementary Fig. 10), possibly due to the interaction between Ir and W dual-atom dimers.
XANES spectra at the W L3-edge showed that both the absorption edge position and white line intensity of Ir–W7/CeO2 closely resembled those of WO3 (Fig. 1g, Supplementary Fig. 11a). The linear combination fitting results showed that Ir–W7/CeO2 contained 95.1% W6+ species (Supplementary Fig. 11c, d), further indicating that W species were mainly present in the W6+ state. In both Ir–W3/CeO2 and Ir–W7/CeO2 samples, the EXAFS curves showed a prominent peak at 1.38 Å, which was assigned to the W–O coordination in the first shell48, while a peak at 2.73 Å was attributed to the W–O–Ce coordination in the second shell (Fig. 1h, Supplementary Fig. 11b)49. Additionally, EXAFS fitting showed that the W–O coordination number and bond distances in Ir–W7/CeO2 approximated those in WO3, further confirming the presence of W6+ species (Supplementary Table 3). Notably, no W–W or W–O–W signals were detected in both Ir–W3/CeO2 and Ir–W7/CeO2 samples, demonstrating the highly dispersion of W species. WT EXAFS analysis revealed a strong signal for W–O coordination and a weaker signal for W–O–Ce in Ir–Wx/CeO2 samples (Fig. 1i, Supplementary Fig. 12), further demonstrating the high dispersion of W species in the Ir–Wx/CeO2 catalysts. EXAFS fitting further revealed that no Ir–W scattering path was detected within the EXAFS fitting range. In contrast, Ir–W3/CeO2 showed a W–O–Ce coordination number of 2.4 at 2.91 Å and a contracted W–O distance of 1.81 Å relative to WO3 (Supplementary Fig. 13, Supplementary Table 3), indicating strong W–CeO2 interactions. Compared with Ir–W3/CeO2, Ir–W7/CeO2 exhibited an increased W–O coordination number (4.5) with a further contracted bond distance (1.75 Å), while the Ir–O bond was slightly elongated from 2.03 to 2.09 Å. Notably, the higher single-atom W density did not significantly change the coordination environment of Ir–O–Ce and W–O–Ce. These results indicate that increasing W single-atom density primarily influenced the local W coordination environment by contracting W-O bond distances and induced slight elongation of Ir--O bonds, while Ir–O–Ce and W–O–Ce coordination structures remained unchanged. These pronounced Ir– and W–CeO2 interactions suggested that both metals primarily bond to the CeO2 support rather than to each other, explaining the absence of an Ir–W scattering signal.
X-ray photoelectron spectroscopy (XPS) analysis showed that the Ir species in the Ir/CeO2 were mainly present in the Ir4+ state (62.4 eV, 87.9%) (Fig. 2a, Supplementary Table 4), with a minor fraction of Ir3+ (61.6 eV), and W existed primarily in the W6+ state (35.7 eV) in W7/CeO2 (Fig. 2b), consistent with XANES results. Additionally, the introduction of W or Ir species in the Ir–Wx/CeO2 samples did not significantly alter the oxidation state or peak position of Ir and W species, respectively (Supplementary Fig. 14), indicating that there was no significant electronic transfer between Ir and W species, consistent with the XAFS results. The Ce 3 d spectra showed that the pristine CeO2 contained Ce3+ (23.8%) and Ce4+ (76.2%) species37, indicating the defect-rich nature of CeO2 support (Fig. 2c). The introduction of Ir significantly reduced the amount of Ce3+ in Ir/CeO2 (19.8%), whereas the incorporation of W only slightly affected the Ce3+ content in W7/CeO2 (22.1%). The O 1 s spectra showed that CeO2 contained a considerable amount of surface oxygen species (Osurf, 531.8 eV, 38.5%), in addition to lattice oxygen species (Olatt, 529.5 eV, 61.5%). A decrease in Osurf content upon Ir addition (32.4% in Ir/CeO2) and a more pronounced reduction with W incorporation (26.0% in W7/CeO2), likely attributed to the coverage of the CeO2 surface by WO3, was observed. In Ir–Wx/CeO2, Osurf content decreased even further, reaching a ratio of 24.6% in the Ir–W7/CeO2 sample (Fig. 2d, Supplementary Fig. 14d). These results indicated a positive correlation between the content of Ce3+ and oxygen defects, since the presence of oxygen defects generally induces the formation of Ce3+ species. Furthermore, Raman spectra of Ir/CeO2 exhibited characteristic bands at 658 cm−1 and 721 cm−1, corresponding to Ir–O–Ce and Ir–O bonds vibrations, respectively, confirming the presence of Ir–O–Ce sites (Supplementary Fig. 15, Supplementary Note 1). Upon W incorporation, the W–O–Ce band at 887 cm−1 emerged, confirming the coexistence of Ir–O–Ce and W–O–Ce sites in Ir–Wx/CeO2 catalysts. Notably, with increasing W loading, the progressive enhancement of the W–O–Ce band intensity, accompanied by the weakening of the Ir–O–Ce band, indicated that Ir and W atoms were preferentially located in close proximity, thereby modifying their respective local coordination environment.
XPS spectra for the Ir 4 f orbitals (a), W 4 f orbitals (b), Ce 3 d orbitals (c), and O 1 s orbitals (d) of Ir–Wx/CeO2 catalysts. H2-TPR profiles (e) and NH3-TPD profiles (f) of Ir–Wx/CeO2 catalysts.
H2 temperature-programmed reduction (H2-TPR) experiment was performed to investigate the redox properties of Ir–Wx/CeO2 catalysts (Fig. 2e, Supplementary Table 5). CeO2 exhibited a broad peak ranging from 380 to 424 °C (800 μmol g−1), corresponding to the reduction of surface Ce4+ to Ce3+, along with a smaller sub-surface Ce4+ reduction peak at 500 °C (319 μmol g−1)50. For W7/CeO2, the hydrogen consumption for surface Ce4+ reduction decreased significantly (465 μmol g−1), possibly due to the WO3 coverage, while the sub-surface Ce4+ reduction peak shifted to a higher temperature of 524 °C. Moreover, no reduction for WOx species was observed due to the high temperature (above 600 °C) required for the reduction of both the bulk and dispersed WO3 species49,50. In Ir/CeO2, a small shoulder peak at 93 °C corresponded to the reduction of single-atom Ir (103 μmol g−1), while a sharp peak at 142 °C (769 μmol g−1) was attributed to the promoted reduction of surface Ce4+, facilitated by the H-spillover effect between Ir and CeO251. In the Ir–Wx/CeO2 samples, the surface Ce4+ reduction temperature gradually decreased, while the hydrogen consumption for the reduction of W6+ to W5+ increased with further addition of W content (Supplementary Fig. 16). For the Ir–W7/CeO2, the reduction peak for single-atom Ir shifted to 100 °C, the surface Ce4+ reduction peak appeared at 126 °C, and the W6+ reduction peak emerged at 177 °C. These results demonstrated that the strong metal-support interactions between Ir and CeO2 significantly enhanced the reducibility of the Ir–Wx/CeO2 catalysts, while surface coverage with WOx only slightly reduced the overall redox capability.
NH3 temperature-programmed desorption (NH3-TPD) analysis was conducted to investigate the acidic properties of Ir–Wx/CeO2 catalysts (Fig. 2f, Supplementary Fig. 17). NH3 desorption profiles could be deconvoluted into three peaks: a low-temperature peak ( < 150 °C, weak acidic sites), a mid-temperature peak (150–225 °C, moderate acidic sites), and a high-temperature peak ( > 225 °C, strong acidic sites). CeO2 exhibited abundant acidic sites, with adsorbed NH3 primarily desorbing at low and mid-temperatures (Supplementary Table 6). The total NH3 desorption on Ir/CeO2 was slightly decreased compared to CeO2, accompanied by a reduction in the desorption temperature52,53. Furthermore, Ir sites facilitated the reaction of NH3 with lattice oxygen in CeO2 to produce NO at low temperature ( ~ 200 °C), which possibly led to the decrease in NH3 desorption (Supplementary Fig. 18, Supplementary Table 7)34. Conversely, the introduction of W species resulted in an increase in surface acidity for W7/CeO2, specifically enhancing moderate acidic sites and leading to the emergence of strong acidic sites, along with a shift of desorption peaks to higher temperatures54. For the Ir–W7/CeO2, the surface acidity was higher than that of Ir/CeO2, with a further increase in both weak and moderate acidic sites as W content increased. Notably, NOx formation during NH3-TPD in the absence of gaseous O2 indicated that NH3 reacts with lattice oxygen in CeO2 via the Mars-Van Krevelen (MVK) mechanism.
Catalytic performance
During the NH3 selective oxidation reaction, CeO2 exhibited low activity, with moderate NH3 conversion only observed at high temperatures above 300 °C, and NO being the primary byproduct (Supplementary Fig. 19). W7/CeO2 showed no catalytic activity at temperatures below 225 °C, indicating that the W species itself contributed negligibly to the low-temperature NH3–SCO reaction. The introduction of Ir species significantly improved the low-temperature activity of Ir/CeO2, achieving 93% NH3 conversion at 200 °C (Fig. 3a). However, Ir/CeO2 exhibited poor N2 selectivity, which sharply decreased with rising temperature, reaching a minimum of 21% at 350 °C (Fig. 3b). Over the entire temperature range, NO and NO2 were the dominant byproducts (Fig. 3c). The introduction of a small amount of W in the Ir–W1/CeO2 catalyst slightly enhanced N2 selectivity by primarily reducing NOx byproducts, without negatively affecting its activity (Supplementary Fig. 20). As the W content increased in the Ir–Wx/CeO2 catalysts, N2 selectivity continued to improve by suppressing byproduct formation, while the low-temperature activity remained intact. Notably, the Ir–W7/CeO2 catalyst exhibited the best catalytic performance, maintaining 92% NH3 conversion at 200 °C and achieving a significant improvement in N2 selectivity across the entire temperature range. It showed a high N2 selectivity of 92% even at temperatures above 300 °C due to a significant reduction in NOx production (Fig. 3c), Additionally, Ir–W7/CeO2 exhibited a significantly lower activation energy (52.0 kJ/mol) than other representative catalysts reported in the literature, ranking among the most active and selective AOC catalysts (Supplementary Fig. 21, Supplementary Table 8). Further increasing the W loading to form Ir–W10/CeO2 catalyst improved N2 selectivity further; however, this benefit was accompanied by a slight decline in low-temperature activity, possibly due to the coverage of Ir sites by WO3 species. Notably, the W7/CeO2 catalyst maintained nearly 100% N2 selectivity over the entire temperature range, highlighting the crucial role of W in reducing byproduct formation. Additionally, the Ir–W7/CeO2 catalyst demonstrated excellent stability in a 100-h long-term durability test, at both high and low NH3 conversion (Fig. 3d, Supplementary Fig. 22), maintaining stability over three cycles (Supplementary Fig. 23). The presence of moisture slightly suppressed the low-temperature activity of the Ir–W7/CeO2 catalyst (Supplementary Fig. 24), possibly due to competitive adsorption.
NH3 conversion (a), N2 selectivity (b), and product selectivity on Ir/CeO2 (diagonal lines) and Ir–W7/CeO2 (blank) (c). Stability of Ir–W7/CeO2 in the NH3–SCO reaction at 200 °C (d). Catalytic performance of Ir–Mo4/CeO2 catalysts (e) and Ir–Nb3/CeO2 catalysts (f) in the NH3–SCO reaction. Reaction conditions: 1000 ppm NH3 and 5% O2 in N2 balance at a WHSV of 100,000 mL g−1 h−1.
To further investigate the mechanism behind the enhanced N2 selectivity of Ir–Wx/CeO2, an NH3 selective catalytic reduction (NH3–SCR) reaction was conducted (Supplementary Fig. 25). CeO2 exhibited poor NOx reduction activity, with a maximum NOx conversion of only 25% at 250 °C. In contrast, Ir/CeO2 displayed some NOx reduction activity only at low temperatures below 200 °C, possibly due to the direct oxidation of NH3 as the temperature increased. Notably, W7/CeO2 demonstrated exceptional NOx reduction activity above 200 °C ( > 90%), while maintaining nearly 100% N2 selectivity throughout the reaction, demonstrating the crucial role of W species in the NH3–SCR reaction. Furthermore, Ir–W7/CeO2 exhibited even higher activity at low temperatures, achieving 94% NOx conversion at 175 °C, a performance approximately equal to the sum of that on Ir/CeO2 and W7/CeO2. The gradual decline in NOx conversion at elevated temperatures could be attributed to the direct oxidation of NH3 on Ir sites. These results demonstrated that in the Ir–Wx/CeO2 catalysts, Ir sites primarily facilitated NH3 oxidation to produce N2, alongside byproducts such as NOx and N2O, while W sites promoted NOx reduction to N2 through the SCR reaction. The synergistic interaction between the Ir and W active sites endows Ir–Wx/CeO2 with bifunctional catalytic activity, combining both NH3 oxidation and NOx reduction capabilities.
To further investigate the synergistic effect of Ir and W atoms, a series of CeO2-supported Ir–W catalysts was prepared involving the successive anchoring of Ir and W atoms. Specifically, Wx/Ir–CeO2 catalysts were synthesized by impregnating W atoms onto a single-atom Ir/CeO2 support, while Ir/Wx–CeO2 catalysts were obtained by impregnating Ir onto a Wx/CeO2 support (Supplementary Note 2). Notably, both the W3/Ir–CeO2 and Ir/W3–CeO2 catalysts exhibited catalytic performances similar to that of Ir–W3/CeO2, with significantly improved N2 selectivity compared to Ir/CeO2 (Supplementary Fig. 26). Similarly, at higher W loadings, the W7/Ir–CeO2, Ir/W7–CeO2, and Ir–W7/CeO2 catalysts also displayed comparable activity and selectivity (Supplementary Fig. 27). These results indicated that the anchoring order of the metals had no significant effect on the catalytic performance of the CeO2-supported Ir–W catalysts, suggesting that Ir and W species were anchored at distinct sites on the CeO2 surface.
To demonstrate the universality of the DAC strategy of combining redox and acidic sites, Mo and Nb were introduced to construct analogous Ir–M (M representing acidic transition metals) dual-atom structures. Similar to the Ir–Wx/CeO2 catalysts, the introduction of Mo progressively enhanced the N2 selectivity of Ir–Mox/CeO2 with increasing Mo content (Supplementary Fig. 28). Among them, Ir–Mo4/CeO2 exhibited the best catalytic performance, maintaining 100% NH3 conversion at 225 °C while achieving significantly improved N2 selectivity (Fig. 3e). Similarly, Nb species substantially enhanced the N2 selectivity of Ir–Nbx/CeO2, with Ir–Nb3/CeO2 showing the best catalytic performance within the Ir–Nbx/CeO2 catalysts (Fig. 3f, Supplementary Fig. 29). Notably, the best-performing samples—Ir–W7/CeO2, Ir–Mo4/CeO2, and Ir–Nb3/CeO2—shared similar M: Ir atomic ratios (W = 7.3, Mo = 8.0, Nb = 6.2). This observation indicated that single Ir atoms paired with multiple acidic metal atoms on the Ir–Mx/CeO2 catalysts maximized synergy, although excessive acidic metal atoms may lead to the coverage of Ir sites, thereby reducing catalytic availability.
Reaction mechanism
Operando diffuse reflectance Fourier transform infrared spectroscopy coupled with mass spectrometry (Operando DRIFTS-MS) experiment was performed to investigate the reaction mechanism of cascade ammonia oxidation on the dual-atom Ir–Wx/CeO2 catalysts (Fig. 4). To further investigate the acidic sites on Ir–Wx/CeO2 catalysts, DRIFTS experiments of NH3 adsorption were performed at 100 °C (Fig. 4a). The DRIFTS spectra of NH3 adsorption showed peaks corresponding to NH3 coordinated on Ce-related Lewis acid sites (1139 and 1279 cm−1), as well as negative band (1218 cm−1) attributed to residual nitrate species on the CeO2 support (Fig. 4a, Supplementary Table 9)34. On Ir/CeO2, NH3 was coordinated on Ir–related Lewis acid sites, which caused a shift in the NH3 adsorption peaks (1163 and 1293 cm−1) and an enhancement of the negative bands (1034 and 1218 cm−1). For W7/CeO2, several peaks were observed, including those related to NH3 coordinated on W-related Lewis acid sites (980 and 1172 cm−1), NH4+ coordinated on Brønsted acid sites (948 and 1412 cm−1), and a negative band from the weakening of the W = O bond signal due to NH3 adsorption (1003 cm−1)46. On Ir–W7/CeO2, several peaks due to NH3 adsorption were observed, including NH3 coordinated on Ir sites (1163 cm−1) and W sites (980 cm−1), and NH4+ coordinated to Brønsted acid sites (948 and 1412 cm−1). Notably, at higher temperature (200 °C), a significant amount of NH3 remained adsorbed on the Ir sites of Ir/CeO2, indicating strong NH3 binding at these sites (Supplementary Fig. 30). Additionally, NH3 could react directly with Ir–W7/CeO2 and Ir/CeO2 to produce a considerable amount of N2 ( ~ 80 ppm) in the absence of gaseous O2 (Supplementary Fig. 31). This observation was consistent with the NH3–TPD experiment (Supplementary Fig. 18), further confirming that Ir sites facilitated NH3 reacted with lattice oxygen via the MVK mechanism.
DRIFTS spectra of NH3 adsorption on Ir–Wx/CeO2 catalysts at 100 °C (a). Reactivity of pre-adsorbed NH3 on Ir/CeO2 toward O2 at 200 °C (b). Reactivity of pre-adsorbed NH3 on W7/CeO2 toward NO at 200 °C (c). Reactivity of pre-adsorbed NH3 on Ir–W7/CeO2 toward O2 (e) and NO (f) at 200 °C. N2 formation in the corresponding reactions (d). Typical feed composition: 1000 ppm NH3, 1000 ppm NO, and 5% O2 in Ar balance at a WHSV of 60,000 mL g−1 h−1.
To investigate the reaction mechanism of NH3 oxidation and NO reduction on Ir–Wx/CeO2 catalysts, the reactivity of adsorbed NH3 toward O2 and NO was evaluated, respectively (Fig. 4, Supplementary Table 9). On CeO2, the adsorbed NH3 species exhibited negligible reactivity toward either O2 or NO, producing minimal N2 (Supplementary Fig. 32). In contrast, exposure to NO led to the formation of substantial nitrate species (1100, 1158, and 1557 cm−1). On Ir/CeO2, NH3 adsorption generated a significant amount of NO+ species (2226 cm−1)55, predominantly via reaction with lattice oxygen. Additionally, NH3 interacted with residual nitrate species from CeO2, yielding negative nitrate bands. Upon O2 exposure, NH3 adsorbed on Ir sites (1163 cm−1) reacted rapidly (Fig. 4b), producing significant N2 within 3 min (Fig. 4d). Subsequent formation of surface nitrate species (1215 and 1530 cm−1) occurred without further N2 formation, indicating that adsorbed NH3 could react with gaseous O2 to form inert nitrate species. Critically, the pre-formed NO+ species remained largely unchanged, confirming their exclusive origin from NH3-lattice oxygen reactions and their inertness toward gaseous O2. Moreover, NH3 adsorbed on Ir/CeO2 (1163 cm−1) reacted only slightly with NO, producing limited N2 and demonstrating poor NH3-SCR activity (Supplementary Fig. 33).
On W7/CeO2, NH3 adsorbed at W sites (980, 1172 cm−1) reacted rapidly with NO within 5 min (Fig. 4c), producing substantial N2 (824 ppm), and demonstrating efficient NH3–SCR activity. As the NH3 at W sites were consumed, a progressive depletion of NH4+ coordinated on Brønsted acid sites (958 and 1412 cm−1) was observed. Critically, NH3 adsorption on W7/CeO2 did not generate NO+ species, indicating that NH3 oxidation occurred exclusively on Ir sites, whereas W sites served as selective reduction centers. After the complete consumption of adsorbed NH3, a small amount of adsorbed NO+ species (2226 cm−1) emerged, reflecting competitive adsorption between NH3 and NO. Additionally, the NH3-SCR reaction caused an initial increase in Brønsted acid sites (958 cm−1), likely due to the transformation between W-related Lewis acid sites and Brønsted acid sites induced by water vapor (Supplementary Fig. 34, Note 3). Besides, NH3 adsorbed on W7/CeO2 exhibited no reactivity toward O2 (Supplementary Fig. 35), consistent with the observed catalytic behavior.
On the dual-atom Ir–W7/CeO2 catalyst, NH3 adsorbed on Ir sites (1163 cm−1) and W sites (980 cm−1) were gradually consumed in the presence of O2 (Fig. 4e), producing substantial N2 (347 ppm) within 10 min (Fig. 4d), whereas NH3 on Brønsted acid sites (958, 1412 and 1660 cm−1) remained largely inert. The emergence of weakly adsorbed NO (1931 cm−1) on Ir sites, alongside the absence of nitrate formation, suggested that NH3 oxidation proceeded via lattice oxygen replenished by gaseous O2 rather than direct reaction with O2 to form nitrates. Notably, adsorbed NH3 on Ir (1163 cm−1) and W sites (980 cm−1) could also rapidly react with NO (Fig. 4f), generating a large amount of N2 (354 ppm) through the NH3–SCR pathway (Fig. 4d). After the consumption of NH3 on Lewis acid sites, NH3 on Brønsted acid sites (958, 1412 and 1660 cm−1) continued to react with NO to produce a small amount of N2 over 30 min, accompanied by the appearance of weakly adsorbed NO (1886 and 1920 cm−1)55. Step-response experiments (Supplementary Fig. 36) confirmed the absence of NO+ species (2226 cm−1) under either NH3/O2 or NO/NH3/O2, indicating that the reaction of NO+ with NH3 was faster than its formation. Steady-state NH3 oxidation produced approximately 125 ppm N2, while the introduction of NO led to a marked increase in N2 formation (330 ppm), demonstrating superior NH3–SCR performance. The delayed emergence of weakly adsorbed NO after NH3 consumption suggested preferential NH3 adsorption. Collectively, these observations confirmed that NH3–SCO on the dual-atom Ir–Wx/CeO2 catalysts followed a cascade mechanism, involving both NH3 oxidation and NO reduction pathways. The synergistic effect between Ir and W dual-atom sites enabled the simultaneous removal of NH3 and NOx, significantly enhancing the selectivity of ammonia oxidation.
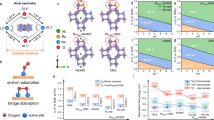
Density functional theory (DFT) calculations were performed to further investigate the reaction pathway and energy barriers for ammonia selective oxidation over dual-atom Ir–Wx/CeO2 catalysts. Three computational models were constructed to determine the optimal structure of single-atom Ir/CeO2 and dual-atom Ir–W/CeO2 catalysts (Supplementary Figs. 37, 38, Supplementary Note 4). Based on the formation energy and previous characterization results, the single-atom Ir/CeO2 and dual-atom Ir–W/CeO2 models were constructed by embedding an Ir atom on the CeO2(111) planes, with a W atom positioned adjacent to the Ir site on the CeO2 surface (Supplementary Fig. 39)34. The anchoring of W exhibited a strong interaction with the adjacent O atoms in the CeO2 lattice, resulting in a significantly shortened W–O bond (1.84 Å) and an elongation of the Ce–O bond (3.37 Å) within the W–O–Ce structure (Supplementary Tables 10, 11). This interaction likely facilitated the formation of unsaturated Ce3+ species, consistent with the XPS results. Additionally, this strong interaction induced the elongation of the Ir–O bond (3.66 Å) within the W-O-Ir structure, which may explain the absence of Ir–O–W bonds in the EXAFS spectra.
DFT calculation revealed that the adsorption energies of NH3 on the Ce, Ir, and W sites were −0.829 eV, −2.13 eV, and −0.643 eV, respectively (Supplementary Fig. 40). In the first stage of NH3 oxidation (Fig. 5a, Supplementary Fig. 41), NH3 adsorbed on the Ir site underwent N–H bond cleavage, with the transfer of a hydrogen atom to the Ir–O–Ce interface, resulting in the formation of *OH and *NH2 (TS1), with an energy barrier of 0.55 eV (Supplementary Tables 12, 13). The *NH2 then underwent dehydrogenation to form *NH, while the hydrogen atom reacted with *OH to produce *H2O (TS2, 0.88 eV). This dehydrogenation step was followed by the desorption of *H2O, which generated an oxygen vacancy that was subsequently replenished by O2 adsorption, leading to the formation of active oxygen species. Hence, lattice oxygen in CeO2 participated in this reaction, confirming the involvement of the MVK mechanism. The *NH species then reacted with the active oxygen species, resulting in the formation of *NHO (TS3, 0.58 eV). Finally, *NHO decomposes into *NO and *OH with a low energy barrier (TS4, 0.03 eV), after which *NO was desorbed. Notably, the dehydrogenation of *NH2 (TS2) was identified as the rate-determining step in this stage, with an energy barrier of 0.88 eV (Supplementary Fig. 42).
Energy barriers for cascade NH3 oxidation (a) and NO reduction (b) on dual-atom Ir–W/CeO2 catalyst. The asterisk indicates the adsorption state, and ‘TS’ denotes the transition state. Atomic color codes: Ir (cyan), O (red), Ce (yellow), W (brown), N (blue), and H (white).
In the second stage of NO reduction (Fig. 5b, Supplementary Fig. 43), DRIFTS-MS experiments and catalytic tests confirmed that the SCR reaction predominantly occurred on W and Ce sites. Specifically, *NH3 adsorbed on the W site underwent dehydrogenation, forming *NH2 and *OH. The *NO species adsorbed on Ce then reacted with *NH2 to generate the critical intermediate *NH2NO (TS5, 0.77 eV), which subsequently underwent dehydrogenation to form *NHNO and *OH (TS6, 0.76 eV). *NHNO then underwent further dehydrogenation to yield *NN and *OH (TS7, 0.40 eV), with the released O atom filling an oxygen vacancy. Finally, *NN dissociated into N2, and two *OH species combined to form a H2O molecule, with the O atom refilling another oxygen vacancy. The dehydrogenation of *NH2NO, which exhibited the highest energy barrier (0.77 eV), was identified as the rate-determining step of the second stage (Supplementary Fig. 44, Supplementary Table 16). Therefore, DFT calculations confirmed that the cascade NH3–SCO reaction occurring on the Ir–W dual-atom catalysts involved the oxidation of NH3 on Ir sites to generate NO, which subsequently reacted with the NH3 adsorbed on W sites, leading to the production of N2 and H2O (Supplementary Tables 14, 15).
The long-standing trade-off between activity and selectivity in catalytic reactions has been a key challenge in catalysis. Previous studies have demonstrated the efficiency of single-atom catalysts for ammonia oxidation but often suffer from poor selectivity, owing to limitations in controlling reaction pathways34,49. In contrast, this work successfully engineered the atomic-level synergy between Ir and W, optimizing the original pathway on single-atom Ir/CeO2 catalysts into a cascade reaction mechanism on Ir–Wx/CeO2 catalysts. This dual-atom configuration effectively overcomes the ‘seesaw relationship’ between activity and selectivity, which has long been a challenge in ammonia oxidation catalysts. The ability to decouple the reaction sequence from individual sites and implement a cascade mechanism across dual sites represents a major advancement in catalytic design, resulting in significantly improved performance. By enabling fine-tuned control over reaction pathways and decoupling the reaction sequence, DACs achieve a significant enhancement in catalytic performance. This work provides compelling evidence for the immense potential of DACs in overcoming the traditional activity-selectivity trade-off, not only in the NH3–SCO reaction but also in a wide array of catalytic processes. The findings presented here offer a universal framework for the rational design of advanced catalysts with specific functionalities, particularly for reactions involving multiple steps or intermediates. Future investigations will systematically explore diverse metal combinations to further enrich the theory of dual-atom catalysis across a broader catalytic process.
Discussion
This study introduces a dual-atom strategy to overcome the long-standing activity-selectivity trade-off in heterogeneous catalysis, demonstrated through the NH3–SCO reaction. By incorporating highly dispersed Ir and W atoms into dual-atom structures on Ir–Wx/CeO2 catalysts, the synergistic interaction between these sites enables a cascade mechanism that integrates both catalytic oxidation and selective catalytic reduction pathways. This synergy not only significantly enhances N2 selectivity but also preserves the exceptional low-temperature activity of Ir–W7/CeO2 catalyst in ammonia selective oxidation. Importantly, this effect is consistent across Ir–Mo and Ir–Nb dual-atom catalysts, highlighting the broad applicability of the dual-atom approach. These findings provide new insights into cascade mechanisms within dual-atom catalysts, offering a rational framework for the design of advanced heterogeneous catalysts with controlled product selectivity. This strategy has broad implications for developing highly efficient catalysts for a range of selective catalytic processes.
Methods
Chemicals and materials
The following reagents were used without further purification: cerium (III) nitrate hexahydrate (Ce(NO3)3·6H2O, 99.95% Ce basis, Aladdin), iridium (III) chloride (IrCl3, 99.95%, 64% Ir basis, Innochem), ammonium metatungstate (VI) hydrate ((NH4)6H2W12O40·xH2O, 90% WO3 basis, Aladdin), ammonium molybdate (VI) tetrahydrate (H24Mo7N6O24·4H2O, 99.9% Mo basis, Aladdin), and niobium (V) oxalate hydrate (C10H5NbO20·xH2O, 98% Nb basis, Innochem).
Catalysts preparation
Ir–Wx/CeO2 catalysts were prepared via a wet chemical method. Initially, defective CeO2 was prepared by calcining cerium (III) nitrate hexahydrate in air at 350 °C for 2 h. The CeO2 support was then mixed with deionized water to form a suspension under continuous stirring. Subsequently, the Ir precursor (IrCl3) and acidic metal precursors (ammonium metatungstate for W) were added to the suspension, which was stirred for an additional 2 h. Afterward, excess water was removed under vacuum using a rotary evaporator at 60 °C to obtain a dry product. The dried samples were subsequently heated at 80 °C for 12 h, followed by calcination in air at 400 °C for 3 h. The catalysts were denoted as Ir–Wx/CeO2, where “x” represents the weight content of the acidic metal component based on the respective acidic metal precursors. Unless otherwise specified, the Ir loading on all Ir–containing samples was 1 wt.%. For comparison, reference samples, such as Ir/CeO2 and W7/CeO2, were prepared using the same procedure.
Ir–Mox/CeO2 and Ir–Nbx/CeO2 catalysts were synthesized using a similar method. Initially, CeO2 was suspended in deionized water under continuous stirring. The Ir precursor (IrCl3) and the corresponding acidic metal precursors—ammonium molybdate for Ir–Mox/CeO2 and niobium oxalate for Ir–Nbx/CeO2—were then added to the suspension, which was stirred for an additional 2 h. Excess water was removed under vacuum using a rotary evaporator at 60 °C, yielding a dry product. The dried samples were subsequently heated at 80 °C for 12 h, followed by calcination in air at 400 °C for 3 h.
Wx/Ir–CeO2 and Ir/Wx–CeO2 catalysts were synthesized using a stepwise wet chemical method with a reverse loading sequence. For the Wx/Ir–CeO2, Ir/CeO2 was first prepared as described above, followed by the loading of varying W contents (3 and 7 wt%) onto the as-synthesized Ir/CeO2 to obtain Wx/Ir–CeO2 catalysts. Conversely, for the Ir/Wx–CeO2 catalysts, Wx/CeO2 catalysts (prepared similarly to the standard W7/CeO2 sample, but with 3 and 7 wt% W loading) were initially synthesized, after which 1 wt% Ir was loaded onto the as-synthesized Wx/CeO2 to yield Ir/Wx–CeO2 catalysts.
Catalyst characterizations
N2 adsorption measurements were conducted using a physical adsorption instrument (Micromeritics, ASAP 2460), and the specific surface area was calculated based on the Brunauer-Emmett-Teller (BET) method. The contents of Ir, W and Ce were determined by inductively coupled plasma-optical emission spectrometry (ICP-OES, Agilent 5110). X-ray diffraction (XRD) experiments were performed on a D8-ADVANCE diffractometer (Bruker) using Cu Kα radiation (40 kV, 40 mA). High-angle annular dark field-scanning transmission electron microscopy (HAADF-STEM) images were collected on a transmission electron microscope (JEM-ARM200F). HAADF-STEM images and energy dispersive X-ray spectroscopy (EDS) mapping were performed on a FEI Titan Cubed G2 300 transmission electron microscope. Electron paramagnetic resonance (EPR) analysis was performed by a Bruker (EMXplus-6/1) operating at the X band (∼9.8 GHz) at 295 K and 100 kHz. X-ray absorption fine structure (XAFS) spectra were collected using Si(111) crystal monochromators at the BL14W1 beamlines at the Shanghai Synchrotron Radiation Facility (SSRF). The X-ray absorption near-edge structure (XANES) data were background-corrected using the Athena module in the IFFEFIT software package56. During data fitting, parameters including bond length (R), Debye-Waller factor(σ2), amplitude factor (\({{\mbox{S}}}_{0}^{2}\)) and energy shift (ΔE0) were optimized for precision. For wavelet transform (WT) analysis, the χ(k) exported from Athena was imported into the Hama Fortran code57,58. The parameters were set with kweight at 2, deploying the Morlet function, specified with κ = 10 and σ = 1. X-ray photoelectron spectroscopy (XPS) analysis was conducted on an AXIS Supra spectrometer from KRATOS Analytical, utilizing Al Kα radiation. Spectra were collected with a step size of 0.10 eV and calibrated using the C 1 s peak (B.E. = 284.8 eV). Raman spectra were recorded on a Renishaw inVia confocal microprobe Raman system with a 532 nm excitation laser. All Raman spectra were baseline-corrected to remove background interference.
H2-TPR experiments were performed on a chemical adsorption instrument (Micromeritics, AutoChem II 2920) equipped with a thermal conductivity detector34,59. A 100 mg sample (40–60 mesh) was placed in a U-shaped quartz cell and pretreated in 10% O2/He (50 ml min−1) at 300 °C for 30 min. After pretreatment, the sample was cooled to 20 °C, purged with Ar for 20 min, and then exposed to 10% H2/Ar (50 ml min−1) for 30 min. Once the baseline stabilized, the temperature was increased to 800 °C at a heating rate of 20 °C min−1, under a 50 ml min−1 flow of 10% H2/Ar. NH3-TPD experiments were performed using a fixed-bed flow reactor with a 100 mg sample41. Prior to measurement, the sample was pretreated in a flow of 5% O2/N2 (500 ml min−1) at 300 °C for 30 min, followed by cooling to 50 °C, after which the gas flow was switched to N2 for a 30-min purge. The catalyst was then exposed to 1000 ppm NH3/N2 at 30 °C for 30 min, followed by N2 purge for 1 h. During the TPD experiments, the desorption of NH3 and the formation of byproducts were continuously monitored by an online infrared spectrometer (Nicolet iS 50) as the temperature was ramped to 600 °C at a heating rate of 10 °C min−1.
Catalytic evaluations
The catalytic test was performed in a fixed-bed flow reactor (i.d. = 6 mm)60. The feed composition for the NH3–SCO reaction consisted of 1000 ppm NH3, 5% O2, and 5% H2O (when used), with N2 as the balance gas, at a total flow rate of 500 mL min−1. A 300 mg sample (40–60 mesh) was used in the catalytic test, equaling a weight hourly space velocity (WHSV) of 100,000 mL g−1 h−1. The reactants and products (NH3, N2O, NO, and NO2) were monitored using an online infrared spectrometer (Nicolet iS 50). The NH3 conversion and N2 selectivity for the NH3–SCO reaction were calculated as follows:
For the NH3–SCR reaction, the feed composition consisted of 550 ppm NH3, 500 ppm NO, and 5% O2 in N2 balance, with a WHSV of 100,000 mL g−1 h−1. The NOx (NOx = NO + NO2) conversion and N2 selectivity were calculated as follows:
where []in and []out represent the inlet and outlet concentrations of reactants and products.
Operando DRIFTS-MS
Operando DRIFTS-MS experiments were conducted using an FTIR spectrometer (Nicolet iS 50) equipped with a Harrick Scientific cell and an online mass spectrometry (InProcess Instruments, GAM 200)59,61. The typical gas composition consisted of 1000 ppm NH3, 1000 ppm NO, 3% H2O, and 5% O2 in Ar balance, at a total flow rate of 100 mL min−1. A ~ 100 mg sample was used, corresponding to a WHSV of 60,000 mL g−1 h−1. Spectra were recorded with a resolution of 4 cm−1 (100 scans) and were represented in Kubelka−Munk units. Prior to the experiment, the sample was pre-treated in an atmosphere of 5%O2/Ar at 400 °C for 30 min. The sample was then cooled to the target temperature for the background spectrum collection. The reaction products, including N2 (m/z = 28), H2O (m/z = 18), NO (m/z = 30), and N2O (m/z = 44), were monitored by the online mass spectrometer.
DFT calculations
First-principles density functional theory (DFT) calculations were performed using the Vienna ab initio simulation package (VASP 6.5.0)62, with the projector augmented wave (PAW) method63. The exchange functional was treated within the generalized gradient approximation (GGA) employing the Perdew-Burke-Ernzerhof (PBE) functional64. The long-range van der Waals interactions are accounted for through the DFT-D3 approach65. A plane wave basis set with an energy cutoff of 500 eV was employed, and the geometry relaxation was performed until the forces on each atom were below 0.03 eV Å−1. The Brillouin zone was sampled using 2 × 2 × 1 k-point grid. Self-consistent calculations were conducted with an energy convergence threshold of 10−5 eV. To prevent interactions between periodic structures, a vacuum region of 15 Å was added along the z direction. Both the AIMD and Slow-growth approach simulations were sampled within the canonical (NVT) ensemble by Nosé-Hoover thermostats with a time step of 1.0 fs at a finite temperature of 298.15 K. The free energy of the intermediates is calculated:
where ΔEDFT, ΔZPE, and ΔS are the changes of the reaction energy obtained from DFT calculations, zero-point energy, and the changes of entropy from the initial state to the final state, respectively. T is temperature, and the T of 298.15 K was used in all computations.
Data availability
All data supporting the findings of this study are available within the paper, Supplementary Information files, and source data files. All raw data generated during the current study are available from the corresponding authors upon request. Source data are provided with this paper.
References
Qiao, B. et al. Single-atom catalysis of CO oxidation using Pt1/FeOx. Nat. Chem. 3, 634–641 (2011).
Jones, J. et al. Thermally stable single-atom platinum-on-ceria catalysts via atom trapping. Science 353, 150–154 (2016).
Shi, Y. et al. Single-atom catalysis in mesoporous photovoltaics: the principle of utility maximization. Adv. Mater. 26, 8147–8153 (2014).
Li, T. et al. Maximizing the number of interfacial sites in single-atom catalysts for the highly selective, solvent-free oxidation of primary alcohols. Angew. Chem. Int. Ed. 57, 7795–7799 (2018).
Lang, R. et al. Non defect-stabilized thermally stable single-atom catalyst. Nat. Commun. 10, 234 (2019).
Wang, F. et al. Resolving the puzzle of single-atom silver dispersion on nanosized γ-Al2O3 surface for high catalytic performance. Nat. Commun. 11, 529 (2020).
Beniya, A. & Higashi, S. Towards dense single-atom catalysts for future automotive applications. Nat. Catal. 2, 590–602 (2019).
Zhao, Y. et al. Stable iridium dinuclear heterogeneous catalysts supported on metal-oxide substrate for solar water oxidation. Proc. Natl Acad. Sci. 115, 2902–2907 (2018).
Wang, A., Li, J. & Zhang, T. Heterogeneous single-atom catalysis. Nat. Rev. Chem. 2, 65–81 (2018).
Calle-Vallejo, F., Loffreda, D., Koper, M. T. M. & Sautet, P. Introducing structural sensitivity into adsorption–energy scaling relations by means of coordination numbers. Nat. Chem. 7, 403–410 (2015).
Bakandritsos, A. et al. Mixed-valence single-atom catalyst derived from functionalized graphene. Adv. Mater. 31, 1900323 (2019).
Sun, G. et al. Breaking the scaling relationship via thermally stable Pt/Cu single atom alloys for catalytic dehydrogenation. Nat. Commun. 9, 4454 (2018).
Pérez-Ramírez, J. & López, N. Strategies to break linear scaling relationships. Nat. Catal. 2, 971–976 (2019).
Li, Z. et al. Well-defined materials for heterogeneous catalysis: from nanoparticles to isolated single-atom sites. Chem. Rev. 120, 623–682 (2020).
Kaiser, S. K., Chen, Z., Faust Akl, D., Mitchell, S. & Pérez-Ramírez, J. Single-atom catalysts across the periodic table. Chem. Rev. 120, 11703–11809 (2020).
Yoo, M. et al. A tailored oxide interface creates dense Pt single-atom catalysts with high catalytic activity. Energy Environ. Sci. 13, 1231–1239 (2020).
Kumar, P. et al. High-density cobalt single-atom catalysts for enhanced oxygen evolution reaction. J. Am. Chem. Soc. 145, 8052–8063 (2023).
Chang, J. et al. Synthesis of ultrahigh-metal-density single-atom catalysts via metal sulfide-mediated atomic trapping. Nat. Synth. 3, 1427–1438 (2024).
Zhang, L., Zhou, M., Wang, A. & Zhang, T. Selective hydrogenation over supported metal catalysts: from nanoparticles to single atoms. Chem. Rev. 120, 683–733 (2020).
Wang, Y. et al. Catalysis with two-dimensional materials confining single atoms: concept, design, and applications. Chem. Rev. 119, 1806–1854 (2019).
Chen, Y., Lin, J., Jia, B., Wang, X., Jiang, S. & Ma, T. Isolating single and few atoms for enhanced catalysis. Adv. Mater. 34, 2201796 (2022).
Paolucci, C. et al. Dynamic multinuclear sites formed by mobilized copper ions in NOx selective catalytic reduction. Science 357, 898–903 (2017).
Chen, S. et al. Dehydrogenation of ammonia borane by platinum-nickel dimers: regulation of heteroatom interspace boosts bifunctional synergetic catalysis. Angew. Chem. Int. Ed. 61, e202211919 (2022).
Hai, X. et al. Geminal-atom catalysis for cross-coupling. Nature 622, 754–760 (2023).
Hao, Q. et al. Nickel dual-atom sites for electrochemical carbon dioxide reduction. Nat. Synth. 1, 719–728 (2022).
Liang, X., Fu, N., Yao, S., Li, Z. & Li, Y. The progress and outlook of metal single-atom-site catalysis. J. Am. Chem. Soc. 144, 18155–18174 (2022).
Tang, Y. et al. Synergy of single-atom Ni1 and Ru1 sites on CeO2 for dry reforming of CH4. J. Am. Chem. Soc. 141, 7283–7293 (2019).
Fu, J. et al. Synergistic effects for enhanced catalysis in a dual single-atom catalyst. ACS Catal. 11, 1952–1961 (2021).
Chu, P. et al. Regulation of lattice oxygen mobility via dual single atoms for simultaneously enhancing VOC oxidation and NOx reduction. Environ. Sci. Technol. 58, 17475–17484 (2024).
Tan, Z., Haneda, M., Kitagawa, H. & Huang, B. Slow synthesis methodology-directed immiscible octahedral PdRh1−x dual-atom-site catalysts for superior three-way catalytic activities over Rh. Angew. Chem. Int. Ed. 61, e202202588 (2022).
Chmielarz, L. & Jabłońska, M. Advances in selective catalytic oxidation of ammonia to dinitrogen: a review. RSC Adv. 5, 43408–43431 (2015).
Xu, G. et al. Advances in emission control of diesel vehicles in China. J. Environ. Sci. 123, 15–29 (2023).
Wang, F. et al. Nanosize effect of Al2O3 in Ag/Al2O3 catalyst for the selective catalytic oxidation of ammonia. ACS Catal. 8, 2670–2682 (2018).
Liu J. et al. Reverse effect of metal-support interaction on platinum and iridium catalysts in ammonia selective oxidation. Chem Catal. 101229 (2024).
Dann, E. K. et al. Structural selectivity of supported Pd nanoparticles for catalytic NH3 oxidation resolved using combined operando spectroscopy. Nat. Catal. 2, 157–163 (2019).
Lan, T. et al. Isolated Pt atoms embedded in CuO nanocatalysts for selective oxidation of ammonia. ACS Catal. 13, 14070–14079 (2023).
Sun, H., Wang, H. & Qu, Z. Construction of CuO/CeO2 catalysts via the ceria shape effect for selective catalytic oxidation of ammonia. ACS Catal. 13, 1077–1088 (2023).
Peng, L. et al. Ammonia abatement via selective oxidation over electron-deficient copper catalysts. Environ. Sci. Technol. 56, 14008–14018 (2022).
Ghosh, R. S. et al. Enhanced selective oxidation of ammonia in a Pt/Al2O3@Cu/ZSM-5 core–shell catalyst. ACS Catal. 10, 3604–3617 (2020).
Chen, L. et al. Tuning the selectivity of NH3 oxidation via cooperative electronic interactions between platinum and copper sites. Nat. Commun. 16, 26 (2025).
An, Q. et al. Designing a bifunctional Pt/Cu-SSZ-13 catalyst for ammonia-selective catalytic oxidation with superior selectivity. ACS Catal. 13, 6851–6861 (2023).
Nie, L. et al. Activation of surface lattice oxygen in single-atom Pt/CeO2 for low-temperature CO oxidation. Science 358, 1419–1423 (2017).
Esch, F. et al. Electron localization determines defect formation on ceria substrates. Science 309, 752–755 (2005).
Alcala, R. et al. Atomically dispersed dopants for stabilizing ceria surface area. Appl. Catal. B 284, 119722 (2021).
Li, Y. et al. Boron doping-induced ultrahigh Ce3+ ratio in amorphous CeO2/GO catalyst for low-concentration CO2 photoreduction. Angew. Chem. Int. Ed. 64, e202505668 (2025).
Rakhmatullin, R. M. et al. EPR study of ceria nanoparticles containing different concentrations of Ce3+ ions. Mater. Chem. Phys. 219, 251–257 (2018).
Wang, Y. et al. Single-atom Ir1 supported on rutile TiO2 for excellent selective catalytic oxidation of ammonia. J. Hazard. Mater. 432, 128670 (2022).
Chen, W. et al. Single tungsten atoms supported on MOF-derived N-doped carbon for robust electrochemical hydrogen evolution. Adv. Mater. 30, 1800396 (2018).
Liu, J. et al. Introducing tin to develop ternary metal oxides with excellent hydrothermal stability for NH3 selective catalytic reduction of NOx. Appl. Catal. B 291, 120125 (2021).
Zhan, S. et al. Efficient NH3-SCR removal of NOx with highly ordered mesoporous WO3(χ)-CeO2 at low temperatures. Appl. Catal. B 203, 199–209 (2017).
Guo, Y. et al. Low-temperature CO2 methanation over CeO2-supported Ru single atoms, nanoclusters, and nanoparticles competitively tuned by strong metal–support interactions and H-spillover effect. ACS Catal. 8, 6203–6215 (2018).
Xu, W. et al. Insight into hydroxyl groups in anchoring Ir single-atoms on vacancy-deficient rutile TiO2 supports for selective catalytic oxidation of ammonia. Appl. Catal. B: Environ. Energy 345, 123684 (2024).
Wang, Y. et al. Effects of IrO2 nanoparticle sizes on Ir/Al2O3 catalysts for the selective catalytic oxidation of ammonia. Chem. Eng. J. 437, 135398 (2022).
Cai, Y. et al. Shielding ceria-based catalysts from SO2 poisoning in NH3–SCR reaction: Modification effect of acid metal oxides. Appl. Catal. B 342, 123424 (2024).
Khivantsev, K. et al. Achieving atomic dispersion of highly loaded transition metals in small-pore zeolite SSZ-13: high-capacity and high-efficiency low-temperature CO and passive NOx adsorbers. Angew. Chem. Int. Ed. 57, 16672–16677 (2018).
Newville, M. IFEFFIT: interactive XAFS analysis and FEFF fitting. J. Synchrotron Radiat. 8, 322–324 (2001).
Funke, H., Scheinost, A. C. & Chukalina, M. Wavelet analysis of extended x-ray absorption fine structure data. Phys. Rev. B 71, 094110 (2005).
Funke, H., Chukalina, M. & Scheinost, A. C. A new FEFF-based wavelet for EXAFS data analysis. J. Synchrotron Radiat. 14, 426–432 (2007).
Xu, G. et al. Unraveling the mechanism of ammonia selective catalytic oxidation on Ag/Al2O3 catalysts by operando spectroscopy. ACS Catal. 11, 5506–5516 (2021).
An, Q. et al. Tandem reaction on Ru/Cu-CHA catalysts for ammonia elimination with enhanced activity and selectivity. Environ. Sci. Technol. 59, 2849–2860 (2025).
Xu, G., Wang, H., Yu, Y. & He, H. Role of silver species in H2-NH3-SCR of NOx over Ag/Al2O3 catalysts: operando spectroscopy and DFT calculations. J. Catal. 395, 1–9 (2021).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Blöchl, P. E., Jepsen, O. & Andersen, O. K. Improved tetrahedron method for Brillouin-zone integrations. Phys. Rev. B 49, 16223–16233 (1994).
Perdew, J. P. et al. Erratum: atoms, molecules, solids, and surfaces: Applications of the generalized gradient approximation for exchange and correlation. Phys. Rev. B 48, 4978–4978 (1993).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Acknowledgments
This work was supported by the National Key R&D Program of China (2022YFC3704400, G.X.) and the National Natural Science Foundation of China (22422609 and 22276203, G.X.; and 52225003, Q. W.).
Author information
Authors and Affiliations
Contributions
G.X. conceived the idea for the project, designed the experiments, analysed the data, and revised the manuscript. T.C. carried out the experiments, performed the data analysis, and drafted the manuscript. D.L. performed the DFT theoretical calculations, analysed the data, and revised the manuscript. M.Z., Y.H., L.Z., and Y.W. helped with the experiments and data analysis. G.X., Q.W., and H.H. supervised the project and revised the manuscript. All authors contributed to the revision of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Feng Ryan Wang and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, T., Liu, D., Zhang, M. et al. Cascade catalysis on dual-atom iridium-tungsten catalysts for enhanced ammonia selective oxidation. Nat Commun 16, 11179 (2025). https://doi.org/10.1038/s41467-025-66144-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-66144-6