Abstract
We aimed to determine the effect of semaglutide on patients with acute large vessel occlusion (LVO) receiving endovascular therapy (EVT). In this phase 2, investigator-initiated, multicenter, prospective, randomized, open-label, blinded endpoint trial conducted in China, we recruited patients with disabling LVO undergoing EVT. Patients were randomized to semaglutide therapy (0.5 mg subcutaneous semaglutide before and 1 week after EVT) or standard therapy. The primary outcome was defined as favorable neurological recovery (modified Rankin Scale 0–2 at 90 days). Between August 2023 and July 2024, 140 patients were randomized to semaglutide (n = 69) or standard therapy (n = 71). The primary outcome occurred in 39 (56.5%) in the semaglutide group and 39 (54.9%) in the standard therapy group (adjusted RR 1.05, 95% CI 0.95–1.15, p = 0.37). We observed treatment effect modification by intravenous thrombolysis (IVT) on semaglutide therapy (pinteraction = 0.02); thus we performed the following exploratory analyses: The primary outcome occurred in 22 (64.7%) in the semaglutide group and 15 (44.1%) in the standard therapy group (adjusted RR 1.18, 95% CI 1.02–1.36) in the no-IVT stratum (n = 68). The primary outcome was similar between two groups in the IVT-stratum. No severe adverse event was attributed to semaglutide treatment. This phase 2 trial suggested semaglutide was safe in patients with LVO and was associated with an improved neurological outcome in patients not receiving IVT. These preliminary observations should be confirmed in a phase 3 randomized trial (ClinicalTrials.gov Identifier: NCT05920889).
Similar content being viewed by others
Introduction
The glucagon-like peptide-1 (GLP-1) receptor is expressed in specialized neuronal subsets and glial cells widely distributed in the central nervous system1, modulating a broad range of physiological functions from food intake, glucose homeostasis and metabolism2,3,4,5, to systemic inflammation6. In rodent models of ischemic stroke, GLP-1 receptor agonists (GLP-1RAs) have been consistently shown to reduce infarct size, counteract reperfusion injury, and attenuate neuroinflammation7,8,9,10. These neuroprotective effects generalized across GLP-1RAs with variable blood-brain barrier (BBB) permeability (attributable to differences in properties such as molecular size, albumin binding11, and potentially receptor-mediated transport into the brain)12 and diverse experimental conditions7, including various rodent species and strains, animal models with or without diabetes mellitus, stroke induction methods, treatment doses and regimens (initiating prior to, during, or even 1–3 days after stroke induction)8,13, indicating a robust class effect.
Potential neuroprotective mechanisms of GLP-1RAs in acute ischemic stroke can be direct or indirect. Direct effects include attenuation of detrimental microglia activation8, promotion of M2 microglia polarization14, reduction of neuronal apoptosis and promotion of neurogenesis15, whereas indirect effects include amelioration of BBB breakdown16, stabilization of blood glucose, modulation of systemic inflammation6, and mimicking of ischemic preconditioning17. Collectively, these pleiotropic effects suggest that GLP-1RA use during the acute phase may offer neuroprotection in human ischemic stroke, aligning with the Stroke Treatment Academic Industry Roundtable X (STAIR X) recommendation for potential cytoprotective drugs18.
To date, one phase 2 randomized trial demonstrated that exenatide, a GLP-1RA, did not improve early neurological recovery at 7 days post-stroke in a study population mainly consisting of mild strokes, despite a significant reduction of hyperglycemic events19. The potential neuroprotective effects of GLP-1RAs remain unexplored in patients with a higher severity of stroke undergoing reperfusion therapy. In large vessel occlusion (LVO) stroke patients with salvageable penumbra, successful reperfusion by endovascular therapy (EVT) may enable the therapeutic potential of GLP-1RAs to be manifested by: 1) enabling better delivery of the drug and its secondary mediators to vulnerable brain tissues with ischemic injury (even though this is yet to be verified in animal models comparing drug distribution with and without reperfusion under otherwise identical experimental conditions), 2) counteraction of reperfusion injury and neuroinflammation once blood flow is restored, and 3) stabilization of the BBB. Apart from enhancing the rescue and protection of ischemic penumbra, these neurovascular benefits could also synergize with the systemic anti-inflammatory and glucose-stabilizing properties of GLP-1RAs to improve stroke outcomes20,21,22. Therefore, investigating the use of a potent GLP-1RA in the context of LVO strokes reperfused by EVT may uncover the neuroprotective effects of GLP-1RAs in human subjects.
To inform the design of a phase 3 trial, the GALLOP (Glucagon-Like Peptide-1 Receptor Agonist in Large Vessel Occlusion Stroke Treated by Reperfusion Therapy) randomized trial aimed to determine the safety and signals for efficacy of semaglutide – currently recognized as the most clinically potent GLP-1RA23—in patients with acute LVO stroke treated by EVT.
Results
Trial population
The GALLOP trial is a phase 2, investigator-initiated, prospective, randomized, open-label, blinded endpoint trial conducted at two thrombectomy centers in China, which also serve as the secondary referral sites from ten primary stroke centers. We compared the efficacy and safety of semaglutide plus reperfusion therapy, i.e., EVT with or without intravenous thrombolysis (IVT), versus reperfusion therapy alone in patients with disabling LVO. Main eligibility criteria include: (1) LVO at the M1 segment of middle cerebral artery (MCA) or terminal internal carotid artery, (2) last-known-well (LKW) within 12 h at presentation, (3) Alberta Stroke Program Early Computed Tomography Score (ASPECTS) ≥ 6, and (4) for patients with a LKW between 6 and 12 h at presentation, computed tomography (CT) perfusion demonstrating a significant clinical-radiological or core-penumbra mismatch according to the DAWN or DEFUSE3 criteria24,25, respectively (see Methods for details).
Between August 11, 2023, and July 25, 2024, 149 patients were screened for study eligibility (Fig. 1), among which 140 patients were enrolled and randomly assigned to semaglutide (n = 69) or standard therapy (n = 71). All patients randomized to the semaglutide group received semaglutide injection before puncture, while 3 (4.3%) patients in the semaglutide group did not complete day-7 semaglutide injection due to mortality. The modified Rankin Scale (mRS) at 90 days was missing for 3 (4.3%) patients in the semaglutide group and 2 (2.8%) patients in the standard therapy group due to loss to follow-up (Table S2). There was no other missing data. Minor protocol violations occurred in 3 (2.1%) patients (Table S3). The trial enrolled to completion. The mean age of patients was 68.2 ± 10.9 years, with 94 (67.1%) male and 46 (32.9%) female subjects. The median (interquartile range) National Institutes of Health Stroke Scale (NIHSS) was 16 (12, 20) and the baseline ASPECTS was 8 (7, 10). The median time (interquartile range) for onset to puncture was 333 (194, 440) minutes. Intravenous thrombolysis (IVT), either by alteplase 0.9 mg/kg or tenecteplase 0.25 mg/kg, was given to 35 (50.7%) in the semaglutide group and 37 (52.1%) in the standard therapy group. No patients received intra-arterial thrombolysis. No patients had contrast extravasation on selective internal carotid artery angiography performed immediately after EVT. Baseline characteristics were balanced between the two groups (Table 1). All analyses were based on the intention-to-treat population, defined as all patients who were randomized (n = 140). Patients who were lost to follow-up were assigned with the worst possible score for an outcome measure. Complete case analysis (n = 135) was also performed for patients with no loss to follow-up as a sensitivity analysis. There were no violations of the statistical assumptions of the modified Poisson regression models and proportional odds assumption for the ordinal logistic regression models. A pre-specified interim safety analysis conducted after the first 69 patients completed the study suggested no indication of increased risk of intracranial hemorrhage, malignant brain edema, neurological deterioration (as measured by changes in NIHSS from baseline to day 3), or poor neurological recovery (mRS 4–6 at 90 days) with semaglutide therapy (Table S4).
This figure shows the overall patient flow in the trial. (BA basilar artery, CT computed tomography, IIT intention-to-treat, IV intravenous, NIHSS National Institutes of Health Stroke Scale).
Primary outcome
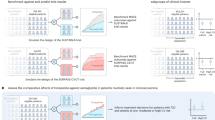
The primary outcome was efficacy, defined as achieving an mRS score of 0–2 at 90 days, a widely used scale to assess the degree of disability after stroke. This outcome occurred in 39 patients (56.5%) in the semaglutide group and 39 patients (54.9%) in the standard therapy group (adjusted risk ratio [RR] 1.05, 95% confidence interval [CI] 0.95–1.15, p = 0.37; Fig. 2A).
The event rates of the primary and binary secondary endpoints of semaglutide and standard therapy groups are shown in A the overall intention-to-treat sample (n = 140), B the no-IVT stratum (n = 68), and C the IVT stratum (n = 72). Data are presented as number (percentage) and risk ratios (95% CI). (CI confidence interval, IVT intravenous thrombolysis).
Secondary outcomes
Secondary outcomes are shown in Figs. 2A, 3A and Table 2. The composite safety outcome, which comprised death, malignant brain edema (parenchymal hypodensity of at least 50% of the MCA territory with signs of local brain swelling such as sulcal effacement and compression of the lateral ventricle, and midline shift of ≥ 5 mm at the septum pellucidum or pineal gland with obliteration of the basal cisterns)26 and intracranial hemorrhage (ICH, indicated by intracranial bleeding of Heidelberg Bleeding Classification of 2 or above)27, occurred in 16 (23.2%) in the semaglutide group and 17 (23.9%) in the standard therapy group (adjusted RR 0.99, 95% CI 0.89–1.11). Semaglutide use was associated with a lower risk of ICH (5.8% vs. 15.5%, adjusted RR 0.91, 95% CI 0.83–0.99), and a higher likelihood of mRS 0–1 at 90 days (39.1% vs. 29.6%, adjusted RR 1.18, 95% CI 1.01–1.37). mRS 0–3 at 90 days, death, malignant brain edema and ordinal shift of mRS at 90 days were similar between the two arms. Final infarct sizes were not different between semaglutide and standard therapy (13.1 [4.9, 66.4] mL vs. 21.8 [10.5, 41.7] mL). Unadjusted risk ratios of primary and secondary outcomes are reported in Table S7.
mRS scores are shown in A the overall intention-to-treat sample (n = 140), B the no-IVT stratum (n = 68), and C the IVT stratum (n = 72). Scores range from 0 to 6: 0 = no symptoms, 1 = symptoms without clinically significant disability, 2 = slight disability, 3 = moderate disability, 4 = moderately severe disability, 5 = severe disability, and 6 = death. (aOR adjusted odds ratio, CI confidence interval, IVT intravenous thrombolysis, mRS modified Rankin Scale).
Exploratory analyses
We looked for treatment effect modification by covariates pre-specified in the statistical model. We found that IVT significantly interacted with the treatment effect by semaglutide (pinteraction = 0.02). mTICI, and post-hoc analyses of diabetes mellitus and baseline blood glucose level did not have treatment effect modification with semaglutide.
Exploratory analyses by stratification according to IVT status were subsequently performed. In patients who did not receive IVT, the primary outcome occurred in 22 (64.7%) patients in the semaglutide group (n = 34) and 15 (44.1%) patients in the standard therapy group (n = 34) (adjusted RR 1.18, 95% CI 1.02–1.36, Fig. 2B). In patients who received IVT, the primary outcome occurred in 17 (48.6%) patients in the semaglutide group and 24 (64.9%) patients in the standard therapy group (adjusted RR 0.96, 95% CI 0.85–1.08, Fig. 2C). Baseline characteristics of both arms in each stratum were similar, except for a higher systolic blood pressure on presentation and lower collateral score among the semaglutide recipients in the IVT stratum (Tables S5A and S6A). The LKW-to-puncture time (median [interquartile range]) was shorter in the IVT stratum compared to the no-IVT stratum (262 [171, 397] minutes vs. 359 [248, 521] minutes, p = 0.002). The proportional odds ordinal logistic regression suggested treatment effect modification on the ordinal mRS shift by IVT (Fig. 3A–C). An ordinal shift towards better functional recovery was observed with semaglutide therapy in the no-IVT stratum (adjusted odds ratio 2.92, 95% CI 1.21−7.2, Fig. 3B).
In the no-IVT stratum, we observed a higher proportion of mRS 0–1 at 90 days (44.1% vs. 26.5%, adjusted RR 1.18, 95% CI 1.02–1.37), mRS 0–3 at 90 days (73.5% vs. 55.9%, adjusted RR 1.14, 95% CI 1.00–1.30), greater reduction in day-3 NIHSS (−8 [−12, −4] vs. −3.5 [−6, −0.8]), and a lower day-3 blood glucose (6.4 ± 1.4 mmol/L vs. 7.8 ± 3.1 mmol/L) with semaglutide therapy (Table S5A–B). The change between day-3 and baseline blood glucose with semaglutide was modest (−0.4 ± 1.2 mmol/L vs. 0.2 ± 2.7 mmol/L). All secondary outcomes were not different between semaglutide and standard therapy in the IVT stratum (Figs. 2C, Fig. 3C and Table S6A–B).
Modified Poisson regression with restricted cubic spline found differing relationships between LKW-to-puncture time and risk ratios of achieving the primary outcome in the semaglutide and standard therapy arms (Fig. S1). In the standard therapy group, a longer LKW-to-puncture time was associated with a lower likelihood of the primary efficacy outcome, whereas this association was diminished in the semaglutide group.
Adverse events and sensitivity analysis
The rates of adverse events are listed in Table 3. Death occurred in 11 (15.9%) patients in the semaglutide group and 11 (15.4%) patients in the standard therapy group (unadjusted RR 1.00, 95% CI 0.9–1.11). Symptomatic ICH, defined as ICH with NIHSS increase of ≥ 4 points, occurred in 1 (1.4%) patient in the semaglutide group and 7 (9.9%) patients in the standard therapy group (unadjusted RR 0.92, 95%CI 0.85–0.98). No patients developed hypoglycemia. Complete case analysis showed similar findings (Tables S8–10).
Discussion
In this phase 2, investigator-initiated, prospective, randomized, open-label, blinded endpoint trial that investigated the use of semaglutide therapy in patients with LVO strokes treated by EVT, semaglutide was well-tolerated without signals of harm. Semaglutide treatment did not improve functional recovery in the overall population but was associated with a reduced risk of ICH. Furthermore, semaglutide treatment was associated with a better functional outcome at 90 days in patients who had not received IVT. These findings require confirmation in a larger trial.
Preclinical neuroprotective effects of GLP-1RAs were demonstrated in unilateral transient MCA occlusion animal models, showing reduced infarct volume, oxidative stress, neuroinflammation, neuronal loss and neurological deficit in both diabetic and non-diabetic rodents7. Notably, the potential neuroprotective effects of GLP-1RA were evident even with delayed drug administration. In normoglycemic mice, a two-week course of daily liraglutide initiated 24 h after stroke induction reduced infarct size, improved neurological recovery, and promoted angiogenesis13. In another study involving diabetic mice, delayed administration of GLP-1RA 3 days after stroke induction for a duration of 8 weeks led to improved motor recovery and normalization of micro-vessel density and pericyte coverage8. Preclinical evidence also suggests that GLP-1RA may enhance BBB integrity, which might have reduced ischemia- or reperfusion-related brain hemorrhage following EVT as observed in our study28,29. Systemically, GLP-1RA may reduce post-stroke stress hyperglycemia, which is a risk factor of BBB dysfunction and poor clinical outcomes20.
Clinical evidence exploring the use of GLP-1RA in acute ischemic stroke is scarce. Thus far, only the phase 2 TEXAIS (Treatment with Exenatide in Acute Ischemic Stroke) randomized trial has been conducted, with the primary outcome defined as NIHSS improvement ≥ 8 (or NIHSS scores 0–1) at 7 days post-stroke. Focusing on this outcome measure, TEXAIS did not demonstrate benefit with exenatide in patients with acute ischemic stroke within 9 h of onset, despite reporting a reduction in hyperglycemic events with exenatide19. The GALLOP trial differs from the TEXAIS in several aspects. First, we included only patients with severe ischemic stroke. This excluded mild stroke with a high likelihood of achieving excellent outcomes, which may offer only a small therapeutic window for the potential treatment effects of GLP-1RA to manifest. Second, we focused on patients eligible for EVT. The high rates of excellent angiographic outcomes may have created conditions more similar to those in transient MCA occlusion animal models. Thirdly, we chose semaglutide, which may be more efficacious than exenatide in terms of cardiovascular protection and regulation of systemic stress and inflammatory responses30,31, as reflected in post-stroke glucose stabilization.
The treatment effect modification observed with IVT on semaglutide was unexpected. At present, there are no available preclinical or clinical studies that evaluated potential interactions between semaglutide and tenecteplase or alteplase32. Any potential pharmacokinetic interaction between GLP-1RA and tissue plasminogen activators should be excluded. On the other hand, GLP-1RA may inhibit the expression of plasminogen activator inhibitor-1 (PAI-1)33,34, a molecule that inhibits tissue plasminogen activators and the conversion of plasminogen to plasmin35. Downregulation of PAI-1 with co-administration of IVT may thus indirectly potentiate plasmin generation and undesired effects of thrombolytic agents, such as BBB disruption36, brain edema36, and neurotoxicity37,38. Nonetheless, these postulations require further confirmation. Importantly, the interaction between semaglutide and IVT could be confounded by time metrics, as IVT administration is a proxy of early presentation. As both neuroinflammation and reperfusion injury are time-dependent in ischemic stroke, the therapeutic benefits of GLP-1RAs in modulating or counteracting these processes may only become more apparent beyond the conventional 4.5-h time window for IVT administration, within which the maximum treatment efficacy is primarily determined by reperfusion therapy. Owing to the limited sample size inherited to the phase 2 trial design and the exploratory nature of this analysis, the possibility of type I error cannot be excluded. Thus, an adequately powered phase 3 study is needed to explore whether the potential neuroprotective effect of GLP-1RA is more pronounced in strokes with later presentations.
Our study has several limitations. First, the current phase 2 trial was not powered to draw definite conclusions regarding the efficacy of semaglutide in improving neurological outcomes in patients with LVO, but rather to estimate any potential treatment effects and evaluate the safety and tolerability of semaglutide treatment to inform the design for a large phase 3 trial. Second, as randomization only took place in two thrombectomy centers, future studies should consider evaluating GLP-1RA administration at primary stroke centers prior to secondary transfer for EVT. Expanding the number of participating centers and accounting for heterogeneity across sites are also necessary to provide a more accurate estimate of the treatment effect and enhance the generalizability of the findings. Third, whether similar observations could be reproduced with different dosages of semaglutide require further study. Fourth, the effect of semaglutide in LVO patients across different ages, LKW-to-presentation time, body weight, ASPECTS, collateral status, blood glucose level on presentation requires further analyses. Fifth, the original plan to assess BBB permeability as a radiological outcome and to conduct omics analyses was not implemented due to the lack of model validation and resource limitations, respectively. Future studies should incorporate BBB leakage-associated biomarkers (e.g., matrix metalloproteinase-9) to more effectively evaluate the impact of GLP-1RA on the BBB39. Last, placebo was not available, but we ensured that all operating neuro-interventionists and raters of functional outcomes were blinded from the knowledge of the treatment allocation. All radiological parameters were determined by the central core laboratory with human raters or computer algorithms blinded from treatment allocation and functional outcomes.
In conclusion, 0.5 mg semaglutide administration before and 1 week after EVT in patients with LVO onset within 12 h was safe and well-tolerated. Semaglutide treatment did not improve functional recovery in the overall population but was associated with lower risk of intracranial hemorrhage, and an improved neurological outcome in patients who did not receive IVT. These preliminary observations should be confirmed in a large phase 3 trial.
Methods
Study design and participants
The GALLOP trial is a phase 2, investigator-initiated, prospective, randomized, open-label, blinded endpoint (PROBE) trial conducted at two thrombectomy centers in China, which also serve as secondary referral sites from ten other primary stroke centers. We compared the efficacy and safety of semaglutide plus reperfusion therapy, i.e., EVT with or without intravenous thrombolysis (IVT), versus reperfusion therapy alone in patients with disabling LVO. Key inclusion criteria were adults age 18 years or older, LVO at the terminal internal carotid artery or M1 segment of the MCA, ASPECTS 6 to 10, NIHSS ≥ 10 at randomization, stroke onset or LKW ≤ 12 hours at randomization, and pre-stroke mRS ≤ 2. Patients who presented between 6 and 12 h from symptom onset or LKW required a CT perfusion scan to assess for clinical-core or perfusion-core mismatch according to the DAWN and DEFUSE3 criteria24,25. The DAWN criteria required a mismatch between clinical deficit and infarct core volume as determined by CT perfusion: patients aged ≥ 80 years needed an NIHSS score ≥ 10 and infarct core (volume of relative cerebral blood flow [rCBF] < 30%) < 21 mL; those < 80 years needed NIHSS ≥ 10 and core < 31 mL, or NIHSS ≥ 20 and core 31 – < 51 mL. The DEFUSE3 criteria required imaging evidence of a target mismatch profile: infarct core (volume of rCBF < 30%) < 70 mL, penumbral tissue (volume of Tmax > 6 s - volume of rCBF < 30%) ≥ 15 mL, and a mismatch ratio (volume of Tmax > 6 s: volume of rCBF < 30%) of ≥ 1.8. Full inclusion and exclusion criteria are enlisted in Table S1. The trial was registered with ClinicalTrials.gov (NCT05920889) and approved by the local institutional review boards (Joint CUHK-NTEC Clinical Research Ethics Committee Reference No.: 2023.026, Science Research Ethics Committee Linyi People’s Hospital Reference No.: YX200651). As the trial did not involve genetic information and materials, it was waived approval from China’s Ministry of Science and Technology related to the export of genetic information and materials. A detailed study protocol and statistical analysis plan can be found in the Study Protocol. The study protocol and statistical analysis plan had been revised once after commencement of study on August 1 2023 (see Supplementary Note 1, Study Endpoints and Statistical Analysis).
Randomization and masking
All potentially eligible patients underwent computed tomography angiography (CTA) to confirm the LVO. We obtained written informed consent from patients or their legal representatives upon confirmation of study eligibility by two investigators. Patients were then randomized in a 1:1 ratio to receive semaglutide plus EVT or EVT alone in the emergency department. Permuted blocked randomization was employed in the study to ensure the balance of subjects throughout the trial setting40. Randomly generated block sizes of 2, 4, and 6 were adopted to avoid possible mid-block inequality caused by larger blocks. Two blocks with unbalanced treatment distribution were generated at the start and middle of the list. The randomization process was performed using the blockrand package (v1.5) in R studio (v4.4.1, R Project for Statistical Computing, RStudio Team 2022). Treatment allocation was concealed until study eligibility was confirmed by two investigators and a written informed consent had been obtained.
Procedures
All consecutive patients with LVO who planned to undergo EVT were screened for study eligibility. In addition, study participants underwent screening for intravenous thrombolysis (IVT) by alteplase or tenecteplase before the randomization process. In general, patients with an LKW-to-presentation time of ≤ 4.5 h were considered for IVT unless otherwise contraindicated41. Patients in the semaglutide arm received subcutaneous semaglutide 0.5 mg before arterial puncture and 7 days after EVT. Semaglutide administration was documented using the computerized systems at the participating centers, either in the stroke unit or rehabilitation unit. The regimen was chosen based on observational studies that suggested an increase in early BBB permeability was associated with worse functional outcomes42, while a subacute BBB permeability increase beyond 10 days after ischemic stroke was associated with good functional outcomes43. Operating neurointerventionalists were masked from the treatment allocation. EVT devices were deployed at the discretion of treating interventionists. All study participants were transferred to the stroke unit or neuro-intensive care unit after the procedure, where they received stroke rehabilitation and secondary stroke prevention according to the Chinese national guidelines44.
We prospectively collected patients’ demographic characteristics, medical history, laboratory results and stroke-related parameters including the NIHSS on admission, LKW-to-puncture time and periprocedural details (mode of anesthesia, procedure time, modified Thrombolysis in Cerebral Infarction [mTICI] score). All patients received a follow-up NIHSS reassessment three days after the procedure. Plain CT brain was repeated 4 to 7 days or during clinical deterioration to detect any intracranial hemorrhage (ICH) or malignant brain edema (MBE). Brain magnetic resonance imaging (MRI) was obtained 14 to 21 days of randomization for quantification of final infarct size. Clinical follow-up was arranged 90 days after randomization for evaluation of functional recovery by mRS. A telephone or video conferencing follow-up was arranged for patients who were unable to attend an in-person follow-up. All certified assessors for NIHSS and mRS were blinded from the treatment allocation and were not the operating neuro-interventionists of the study subjects.
Radiological parameters
Images were processed in the central core image processing laboratory. ASPECTS, volumes of infarct core and total ischemic territory on CT perfusion, and final infarct size on MRI brain were determined by in-house automated image-processing algorithms validated with commercially available software. Collateral score45, mTICI score46, and hemorrhagic complications were determined by two raters (BI, JA) with more than 10 years of experience27. Disparities were resolved by a third rater (SM). Raters and computer algorithms were blinded from treatment allocation and clinical outcomes.
Study endpoints
The primary outcome was efficacy, defined as mRS of 0 to 2 at 90 days. The original definition of mRS of 0 to 3 at 90 days was not used after the steering committee discussion on 15 April 2024 as most clinical trials adopted mRS 0 to 2 as the definition for good neurological recovery for anterior circulation LVO (Supplementary Note 1)47,48. In the study protocol, a primary composite safety endpoint comprising death, MBE, and ICH was defined. This endpoint was analyzed as a secondary outcome, as no statistical power had been prespecified for its assessment. Other secondary outcomes were the ordinal shift in mRS at 90 days, mRS 0 to 3 at 90 days, mRS 0 to 1 at 90 days, death, final infarct size on MRI 14–21 days after randomization, MBE (parenchymal hypodensity of at least 50% of the MCA territory with signs of local brain swelling such as sulcal effacement and compression of the lateral ventricle, and midline shift of ≥ 5 mm at the septum pellucidum or pineal gland with obliteration of the basal cisterns)26, and ICH (hemorrhage occupying more than 30% of the infarcted tissue with mass effect, and/or hemorrhage outside the infarcted brain tissue, or any intracerebral-extracerebral hemorrhage, such as hematoma in parenchymal tissue remote from the infarcted area, intraventricular hemorrhage, subarachnoid hemorrhage, or subdural hemorrhage)27. Secondary exploratory outcomes included changes between day-3 and baseline NIHSS and day-3 and baseline blood glucose level.
Sample size estimation
No human trials had evaluated the efficacy and safety of GLP-1RA on EVT-eligible patients. We hypothesized that semaglutide had a mild-to-moderate effect size, corresponding to a standardized difference of 0.15–0.25, in achieving good functional outcome, i.e., the primary efficacy outcome of mRS 0–2 at 90 days, in patients eligible for the study. Considering a rate of 2.5% suboptimal scan qualities, 5% of loss-to-follow-up and 10% of suboptimal recanalization according to the track records of participating centers, 140 participants were required for 90% power and significance level of 0.05 for the main trial49. Details of the sample size estimation are described in the Study Protocol.
Statistical analyses
We expressed normally distributed continuous variables as mean ± standard deviation and non-normally distributed continuous variables as median (interquartile range [IQR]). Categorical variables were expressed as number (percentage). We compared baseline continuous variables of semaglutide versus standard therapy by independent sample t-test or Wilcoxon rank-sum test, and categorical variables by Chi-squared or Fisher’s exact test as appropriate.
We used the modified Poisson regression models (Poisson regression with a robust error variance estimator to correct standard errors for binary data) to evaluate the risk ratios (RR) of the binary primary and secondary outcomes of semaglutide versus standard therapy50. The original plan of multivariable logistic regression was not used as modified Poisson regression could provide a more unbiased estimation of risk ratios in the setting of common event occurrence compared to logistic regression (Supplementary Note 1)51. The goodness-of-fit of the modified Poisson regression models was assessed using the normalized sum of square test52. Proportional odds ordinal logistic regression was used to compare the ordinal shift of mRS between the two groups. The proportional odds assumption was tested with the Brant test53. Cox regression was used to compare mortality at 90 days. Analysis of covariance (ANCOVA) was used to compare continuous outcomes. Comparisons of the primary efficacy outcome, safety outcome, ordinal shift of mRS, mRS 0–3 and mRS 0–1 at 90 days were adjusted for a pre-specified set of covariates, including age, premorbid mRS, IVT status, NIHSS on presentation, LKW-to-puncture time, mTICI, and baseline ASPECTS. Comparisons of other secondary outcomes were adjusted for NIHSS on presentation. All analyses were performed for the intention-to-treat population, defined as patients who underwent randomization, regardless of treatment received. Deceased patients were considered having an NIHSS of 42. Missing outcomes were imputed as the worst possible score for an outcome measure, i.e., an mRS of 6 and an NIHSS of 42. Complete case analyses for the outcomes were performed in patients without missing data. We performed post-hoc analyses by stratifying IVT status due to the evidence of treatment effect modification by IVT treatment on semaglutide therapy. Modified Poisson regression with restricted cubic spline was used to depict the relationship between LKW-to-puncture and risk ratios of achieving the primary outcome. All secondary outcomes and post-hoc analyses should be considered exploratory. All statistical analyses were performed using R studio (v4.4.1, R Project for Statistical Computing, RStudio Team 2022).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data generated in the figures have been deposited in the Figshare database (https://doi.org/10.6084/m9.figshare.28089197). Anonymized data, including age, sex, admission NIHSS, premorbid mRS, treatment allocation, and study outcomes will be made available by requesting the corresponding authors (Bonaventure Y. Ip, email: ipyiuming@gmail.com, or Ho Ko, email: ho.ko@cuhk.edu.hk) from qualified investigators for academic purposes, beginning 3 months and ending 5 years following publication. The corresponding authors will reply to the request within 2 months, subject to the approval of the Joint Chinese University of Hong Kong-New Territory East Cluster and the Linyi People’s Hospital Clinical Research Ethics Committees. Source data are provided with this paper.
References
Cork, S. C. et al. Distribution and characterisation of Glucagon-like peptide-1 receptor expressing cells in the mouse brain. Mol. Metab. 4, 718–731 (2015).
Holst, J. J. The physiology of glucagon-like peptide 1. Physiol. Rev. 87, 1409–1439 (2007).
Drucker, D. J. Mechanisms of action and therapeutic application of glucagon-like peptide-1. Cell Metab. 27, 740–756 (2018).
Gupta, T. et al. Investigating the glucagon-like peptide-1 and its receptor in human brain: distribution of expression, functional implications, age-related changes and species specific characteristics. Basic Clin. Neurosci. 14, 341–353 (2023).
Merchenthaler, I., Lane, M. & Shughrue, P. Distribution of pre-pro-glucagon and glucagon-like peptide-1 receptor messenger RNAs in the rat central nervous system. J. Comp. Neurol. 403, 261–280 (1999).
Wong, C. K. et al. Central glucagon-like peptide 1 receptor activation inhibits Toll-like receptor agonist-induced inflammation. Cell Metab. 36, 130–143.e135 (2024).
Maskery, M. P. et al. Glucagon-like peptide-1 receptor agonists as neuroprotective agents for ischemic stroke: a systematic scoping review. J. Cereb. Blood Flow. Metab. 41, 14–30 (2021).
Augestad, I. L. et al. Normalisation of glucose metabolism by exendin-4 in the chronic phase after stroke promotes functional recovery in male diabetic mice. Br. J. Pharm. 179, 677–694 (2022).
Li, Y. et al. GLP-1 receptor stimulation preserves primary cortical and dopaminergic neurons in cellular and rodent models of stroke and Parkinsonism. Proc. Natl. Acad. Sci. USA 106, 1285–1290 (2009).
Darsalia, V. et al. Glucagon-like peptide−1 receptor activation reduces ischaemic brain damage following stroke in Type 2 diabetic rats. Clin. Sci. (Lond.) 122, 473–483 (2012).
Smits, M. M. & Holst, J. J. Endogenous glucagon-like peptide (GLP)-1 as alternative for GLP-1 receptor agonists: could this work and how?. Diabetes Metab. Res. Rev. 39, e3699 (2023).
Imbernon, M. et al. Tanycytes control hypothalamic liraglutide uptake and its anti-obesity actions. Cell Metab. 34, 1054–1063.e1057 (2022).
Chen, Y., Zhang, X., He, J., Xie, Y. & Yang, Y. Delayed administration of the glucagon-like peptide 1 analog liraglutide promoting angiogenesis after focal cerebral ischemia in mice. J. Stroke Cerebrovasc. Dis. 27, 1318–1325 (2018).
Darsalia, V. et al. Exendin-4 reduces ischemic brain injury in normal and aged type 2 diabetic mice and promotes microglial M2 polarization. PLoS One 9, e103114 (2014).
Yang, X. et al. The diabetes drug semaglutide reduces infarct size, inflammation, and apoptosis, and normalizes neurogenesis in a rat model of stroke. Neuropharmacology 158, 107748 (2019).
Shan, Y. et al. The glucagon-like peptide-1 receptor agonist reduces inflammation and blood-brain barrier breakdown in an astrocyte-dependent manner in experimental stroke. J. Neuroinflamm. 16, 242 (2019).
Nizari, S. et al. Glucagon-like peptide-1 (GLP-1) receptor activation dilates cerebral arterioles, increases cerebral blood flow, and mediates remote (pre)conditioning neuroprotection against ischaemic stroke. Basic Res. Cardiol. 116, 32 (2021).
Savitz, S. I., Baron, J. C. & Fisher, M. Stroke treatment academic industry roundtable X: brain cytoprotection therapies in the reperfusion era. Stroke 50, 1026–1031 (2019).
Bladin, C. F. et al. Management of poststroke hyperglycemia: results of the TEXAIS randomized clinical trial. Stroke 54, 2962–2971 (2023).
Kawai, N., Keep, R. F., Betz, A. L. & Nagao, S. Hyperglycemia induces progressive changes in the cerebral microvasculature and blood-brain barrier transport during focal cerebral ischemia. Acta Neurochir. Suppl. 71, 219–221 (1998).
Skrobucha, A., Pindlowski, P., Krajewska, N., Grabowski, M. & Jonik, S. Anti-inflammatory effects of glucagon-like peptide-1 (GLP-1) in coronary artery disease: a comprehensive review. Front Cardiovasc Med 11, 1446468 (2024).
Ren, Y. et al. The effect of GLP-1 receptor agonists on circulating inflammatory markers in type 2 diabetes patients: a systematic review and meta-analysis. Diabetes Obes. Metab. 27, 3607–3626 (2025).
Liu, L. et al. Comparative efficacy and safety of glucagon-like peptide-1 receptor agonists in children and adolescents with obesity or overweight: a systematic review and network meta-analysis. Pharmaceuticals (Basel) 17 https://doi.org/10.3390/ph17070828 (2024).
Albers, G. W. et al. Thrombectomy for stroke at 6 to 16 hours with selection by perfusion imaging. N. Engl. J. Med 378, 708–718 (2018).
Nogueira, R. G. et al. Thrombectomy 6 to 24 hours after stroke with a mismatch between deficit and infarct. N. Engl. J. Med. 378, 11–21 (2018).
Huang, X. et al. Predictors of malignant brain edema after mechanical thrombectomy for acute ischemic stroke. J. Neurointerv Surg. 11, 994–998 (2019).
von Kummer, R. et al. The Heidelberg bleeding classification: classification of bleeding events after ischemic stroke and reperfusion therapy. Stroke 46, 2981–2986 (2015).
Zhao, L. et al. Pharmacologically reversible zonation-dependent endothelial cell transcriptomic changes with neurodegenerative disease associations in the aged brain. Nat. Commun. 11, 4413 (2020).
Chen, F. et al. The glucagon-like peptide-1 receptor agonist exendin-4 ameliorates warfarin-associated hemorrhagic transformation after cerebral ischemia. J. Neuroinflamm. 13, 204 (2016).
Sabina, M. & Alsamman, M. M. Pulse of progress: a systematic review of glucagon-like peptide-1 receptor agonists in cardiovascular health. Cardiol. Res 15, 1–11 (2024).
Kaneko, M. & Narukawa, M. Assessment of cardiovascular risk with glucagon-like peptide 1 receptor agonists in patients with type 2 diabetes using an alternative measure to the hazard ratio. Ann. Pharmacother. 52, 632–638 (2018).
Tan, Z., Li, X., Kelly, K. A., Rosen, C. L. & Huber, J. D. Plasminogen activator inhibitor type 1 derived peptide, EEIIMD, diminishes cortical infarct but fails to improve neurological function in aged rats following middle cerebral artery occlusion. Brain Res. 1281, 84–90 (2009).
Nauck, M. et al. Long-term efficacy and safety comparison of liraglutide, glimepiride and placebo, all in combination with metformin in type 2 diabetes: 2-year results from the LEAD-2 study. Diabetes Obes. Metab. 15, 204–212 (2013).
Liu, H., Dear, A. E., Knudsen, L. B. & Simpson, R. W. A long-acting glucagon-like peptide-1 analogue attenuates induction of plasminogen activator inhibitor type-1 and vascular adhesion molecules. J. Endocrinol. 201, 59–66 (2009).
Gong, L. et al. Crystal structure of the Michaelis complex between tissue-type plasminogen activator and plasminogen activators inhibitor-1. J. Biol. Chem. 290, 25795–25804 (2015).
Niego, B. & Medcalf, R. L. Plasmin-dependent modulation of the blood-brain barrier: a major consideration during tPA-induced thrombolysis?. J. Cereb. Blood Flow. Metab. 34, 1283–1296 (2014).
Lee, T. W., Tsang, V. W. & Birch, N. P. Physiological and pathological roles of tissue plasminogen activator and its inhibitor neuroserpin in the nervous system. Front Cell Neurosci. 9, 396 (2015).
Lopez-Atalaya, J. P. et al. Toward safer thrombolytic agents in stroke: molecular requirements for NMDA receptor-mediated neurotoxicity. J. Cereb. Blood Flow. Metab. 28, 1212–1221 (2008).
Kollikowski, A. M. et al. MMP-9 release into collateral blood vessels before endovascular thrombectomy to assess the risk of major intracerebral haemorrhages and poor outcome for acute ischaemic stroke: a proof-of-concept study. EBioMedicine 103, 105095 (2024).
Zhao, W., Ciolino, J. & Palesch, Y. Step-forward randomization in multicenter emergency treatment clinical trials. Acad. Emerg. Med 17, 659–665 (2010).
Powers, W. J. et al. 2018 Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 49, e46–e110 (2018).
Nadareishvili, Z., Simpkins, A. N., Hitomi, E., Reyes, D. & Leigh, R. Post-stroke blood-brain barrier disruption and poor functional outcome in patients receiving thrombolytic therapy. Cerebrovasc. Dis. 47, 135–142 (2019).
Bernardo-Castro, S. et al. The evolution of blood-brain barrier permeability changes after stroke and its implications on clinical outcome: a systematic review and meta-analysis. Int J. Stroke 18, 783–794 (2023).
Liu, L. et al. Chinese Stroke Association guidelines for clinical management of ischaemic cerebrovascular diseases: executive summary and 2023 update. Stroke Vasc. Neurol. 8, e3 (2023).
Yeo, L. L. et al. Assessment of intracranial collaterals on CT angiography in anterior circulation acute ischemic stroke. AJNR Am. J. Neuroradiol. 36, 289–294 (2015).
Goyal, M. et al. 2C or not 2C: defining an improved revascularization grading scale and the need for standardization of angiography outcomes in stroke trials. J. Neurointerv Surg. 6, 83–86 (2014).
Saver, J. L. et al. Stent-retriever thrombectomy after intravenous t-PA vs. t-PA alone in stroke. N. Engl. J. Med 372, 2285–2295 (2015).
Goyal, M. et al. Endovascular thrombectomy after large-vessel ischaemic stroke: a meta-analysis of individual patient data from five randomised trials. Lancet 387, 1723–1731 (2016).
Teare, M. D. et al. Sample size requirements to estimate key design parameters from external pilot randomised controlled trials: a simulation study. Trials 15, 264 (2014).
Zou, G. A modified Poisson regression approach to prospective studies with binary data. Am. J. Epidemiol. 159, 702–706 (2004).
Chen, W., Qian, L., Shi, J. & Franklin, M. Comparing performance between log-binomial and robust Poisson regression models for estimating risk ratios under model misspecification. BMC Med. Res. Methodol. 18, 63 (2018).
Hagiwara, Y. & Matsuyama, Y. Goodness-of-fit tests for modified Poisson regression possibly producing fitted values exceeding one in binary outcome analysis. Stat. Methods Med. Res. 33, 1185–1196 (2024).
Brant, R. Assessing proportionality in the proportional odds model for ordinal logistic regression. Biometrics 46, 1171–1178 (1990).
Acknowledgements
Non-author contributions to Data collection, Analysis, or Writing/editing assistance: The authors thank the contributions from Dr. Robert Dan (MBBS, The Chinese University of Hong Kong), Mrs. Sophia Dan (BBS, MH, JP, The Chinese University of Hong Kong), Ms. Anki Miu (MSc, The Chinese University of Hong Kong), Mr. Ka Hung Li (MSc, Hong Kong Baptist University) and Ms. Maggie Lau (MSc, The Chinese University of Hong Kong). Dr. Robert Dan provided general advice for the study. Mrs. Sophia Dan provided general advice for the study. Ms. Anki Miu provided clerical assistance for the study. Mr. Ka Hung Li provided general advice for the study. Ms. Maggie Lau provided general advice for the study. Access to data and data analysis: Bonaventure Y. Ip and Ho Ko have full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Author information
Authors and Affiliations
Contributions
H.W. conducted the study, collected the data, and critically revised the manuscript for intellectual content. T.W.L., J.H., J.S., Y.Liang, J.Z., Q.C., W.Z., Y.Li, S.H.M., W.T.L., J.C., C.Chan, J.W., A.J.K., K.M., F.F., A.C., V.I., H.Leung., Y.S., K.T.W., B.Lai, C.Chu, H.S.L., A.H., T.C., J.A., S.H.L., L.C., J.Y., L.L., T.H. and S.F.T. conducted the study and collected the data. H.Li. verified the data. S.P. and T.Y. conducted statistical analysis. X.L., B.Lam, V.C.T.M., R.H.M.C., T.N.N., W.H., F.C. critically revised the manuscript for intellectual content. H.K. and B.Y.I. created the study concept and design, supervised the study, conducted statistical analysis, verified the data, wrote and critically revised the manuscript for intellectual content.
Corresponding authors
Ethics declarations
Competing interests
T.N.N. reports Associate Editorship of Stroke; Advisory boards of Brainomix and Aruna Bio; Speaker for Genentech and Kaneka; Consulting for Medtronic. All other authors report no disclosures relevant to the manuscript.
Peer review
Peer review information
Nature Communications thanks Ming Wei, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, H., Ko, H., Leung, T.W. et al. Glucagon-like peptide-1 receptor agonist in large vessel occlusion treated by reperfusion therapy—a phase 2 randomized trial. Nat Commun 16, 11274 (2025). https://doi.org/10.1038/s41467-025-66167-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-66167-z