Abstract
The human utricle is a vestibular organ essential for balance, a function that declines with age. With the aging population projected to double to 2 billion by 2050 and no pharmaceutical or biological treatments available, balance disorders represent a significant unmet medical need. The utricle is composed of sensory and non-sensory cells, which are closely related. Non-sensory cells have limited capacity to regenerate sensory cells and, therefore, are a relevant therapeutic target. In this work, we profile the cellular and transcriptional landscape of the adult human utricle and its early response to ototoxic damage using bulk and single-cell RNA-sequencing of patient-derived samples. We identify six transcriptionally distinct non-sensory cell types, including a previously uncharacterized supporting cell-like population, demonstrating utricular heterogeneity. Following aminoglycoside-induced damage, we detect early transcriptional changes consistent with a capacity to respond to ototoxic damage within 24 hours and potentially initiate a regenerative response via an early-responding cell population, providing a foundation for regenerative strategies for balance recovery.
Similar content being viewed by others
Introduction
The utricle is the organ responsible for balance and facilitates the vestibulo-ocular reflex, one of the most critical human reflexes, which allows both high- and slow-velocity visual fixation. This function, together with other sensory inputs, enables us to navigate our environment. This balance organ is part of the vestibular system located within the inner ear (Fig. 1). The sensory epithelium of the utricle consists of mechanosensory hair cells (HCs), which detect movement and convert mechanical signals into electrical signals that are eventually processed by the vestibular cortex, as well as non-sensory supporting cells (SCs) that orchestrate the organ’s function, providing structural support for hair cells, mediating their survival, and helping to maintain extracellular homeostasis1,2. Defining the transcriptome of hair cells and supporting cells is essential for understanding physiological, pathological, and regenerative conditions of this understudied balance organ. Several factors, including aging, infection, and ototoxic drugs, can damage the cellular components within the utricle. Although limited hair cell turnover may occur spontaneously in adult human utricles3,4, this is not sufficient to recover function, resulting in progressive and permanent balance disorders as no treatments are available. Altogether, this leads to increased fall risk, morbidities associated with injuries, and considerably reduces quality of life5. Identifying cell types, markers, therapeutic candidates, and druggable targets in the adult human utricle is challenging as adult balance organ availability is limited to scarce cadaveric tissues and individuals with vestibular schwannoma. This results in a knowledge gap that hampers the investigation of potential solutions for this unmet medical need.
a Anatomical illustrations of the human inner ear encasing the utricle, a vestibular organ responsible for detecting head tilt and sensing linear acceleration, located within the vestibular region of the temporal bone. The utricular sensory epithelium contains sensory hair cells (HCs) and non-sensory supporting cells (SCs). Type I HCs are characterized by a narrow neck leading to a flask-shaped cell body, while type II HCs exhibit a cylindrical shape with the nucleus positioned higher in the cytoplasm. b H&E-stained utricle shows the organ structural integrity: HCs (eosin-stained pink cytoplasm), supporting cells nuclei (hematoxylin-stained, dark purple), and a small area with missing cells (white space between cells). Sensory nerve fibres are visible in the mesenchyme, n > 3. Scale bar = 100 μm. c Image showing the intactness of a dissected patient-derived utricle, representing the entire balance organ; inset shows sagittal axis (A-P, anterior-posterior) and frontal axis (M-L, medial-lateral), n > 3. d SEM micrograph of flat epithelium showing complete loss of HCs. e–g New/nascent HCs in the supporting cell layer of adult human utricles, which were previously reported4,18. In (e) SEM image shows four sensory cells: HC-1 without stereocilia, HC-2 with stereocilia rootlets on the surface, and HC-3 with kinocilium and short hair bundles as well as a new HC; f nascent hair cells MYO7A+ (green)/SOX2− (magenta), counterstained with DAPI (blue), are located at the basal region of the sensory epithelium, see also (h); (g) SEM image shows a new HC with short stereocilia. N = 3 for all SEM micrographs. Progressive numbers in the images reflect the numbers in the anatomical illustration in (a). h Immunofluorescence of MYO7A (green) and SOX2 (magenta) in a human utricle cross-section, counterstained with DAPI (blue). Inset (63x magnification of area outlined in white) highlighting the three main cell types: HC type I (MYO7A+/SOX2-), type II (MYO7A+/SOX2+), and supporting cell (SOX2+), n > 3. Schematics (created in BioRender. Luca, E. (2025), https://BioRender.com/zejtybk) illustrate the morphology of HCs and the expression of the subtype-specific nuclear marker SOX2 alongside cytoplasmatic marker MYO7A. Scale bars = 100 μm (b, e, h), 25 μm (inset in h), 1 mm (c), 50 μm (d), 10 μm (f, g).
Mounting evidence suggests that supporting cells are a potential target for endogenous regeneration as they survive after damage and hair cell loss; therefore, identifying the supporting cell transcriptome is crucial. In embryonic and newborn mouse utricles, supporting cells can regenerate hair cells by proliferation or direct SC-to-HC transdifferentiation during homeostasis and after ototoxic damage6,7,8, and this ability drastically declines with age6,9. Single-cell RNA-sequencing (scRNA-seq) of the mouse utricle has improved our understanding of utricle biology during development10,11,12, at maturity13,14, and after damage15,16. In humans, information on the utricle is minimal, despite the rise in balance disorders. A limited degree of spontaneous regeneration has been reported in adult utricles after ototoxic damage3,4 and via overexpression of ATOH1, a master regulator of hair cell development17,18,19. Until today, only three studies, one bulk20 and two single cell21,22, sequenced adult human utricles. Therefore, characterizing the cellular and transcriptional profiles of the adult human utricle sensory epithelium, unravelling supporting cell heterogeneity, and examining their response to damage, including any transcriptional changes related to their capacity to regenerate hair cells, is essential for designing regenerative strategies.
Here, we show transcriptional heterogeneity and damage-induced response by conducting bulk and scRNA-seq analyses on 12 patient-derived utricles, six for bulk analysis (control and gentamicin-treated; n = 3 per condition) and six for scRNA-seq (untreated, control, gentamicin-treated; n = 2 per condition); Supplementary Data 1a. Our study generates a single-cell atlas of the human utricle sensory epithelium, revealing, in addition to the already known two hair cell types, six types of supporting cells, leading to the discovery of SC-like cells. We also investigate the response to ototoxic damage, providing insight into the early transcriptional changes following 24 h gentamicin exposure and detecting an early-responding supporting cell population. We identify over 400 differentially expressed genes, including druggable targets and transcription factors, many associated with proliferation, survival, regeneration, and epithelial integrity. Cross-species comparisons between human and mouse utricles, identify human-specific or enriched genes as putative markers for sensory and non-sensory cells, highlighting species-specific differences in the balance organ.
Results
Transcriptional heterogeneity of the utricle sensory epithelium
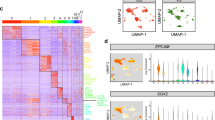
Before mapping the cellular and molecular features of the inner ear balance organ (Fig. 1a), we morphologically illustrated its structure via histology (Fig. 1b). We also show the intactness of the collected organs (Fig. 1c), and their features, demonstrating areas of flat epithelium with no sensory cells (Fig. 1d), with new hair cells (Fig. 1e–g) and their established cell type markers and hair cell shapes (Fig. 1h). Then, using the 10x Genomics Chromium system, we performed scRNA-seq and obtained 1,134 cells from one adult utricle sensory epithelium (a median of 31,163 reads per cell and a median of 991 genes per cell) collected from an individual with vestibular schwannoma (scUntr-2, Supplementary Data 1a). After quality control (Supplementary Table 1), we profiled the transcriptome and heterogeneity of 967 cells. Unsupervised cell clustering yielded eight transcriptionally distinct clusters, which were visualized using UMAP (Unsupervised Uniform Manifold Approximation and Projection, Fig. 2a, Supplementary Fig. 6a). We employed an additional dimensionality reduction method, SWNE (Similarity Weighted Nonnegative Embedding)23, to validate the UMAP output, and clustering was consistent. SWNE was also utilized for visualizing the local and global structure among cell types (Supplementary Fig. 1a). To annotate the eight clusters, in addition to the differentially expressed genes in each cluster (Fig. 2b, Supplementary Data 2a), we used known mammalian hair cell and supporting cell marker genes like MYO7A for hair cells and SOX2 for supporting cells and type II hair cells4 (Fig. 2c). Two groups of hair cells had specific expression for established markers: type I hair cells expressed subtype marker OCM24, and type II hair cells were marked by SOX2. To further confirm supporting cell populations, we also used the pan-SC marker OTOL125. Notably, Calb1, a mouse type I hair cell marker26, was specific for human type II hair cells (Fig. 2d).
a UMAP plot showing 967 cells of a utricular sensory epithelium (scUntr-2), where six supporting cells and two hair cell clusters are identified based on transcriptional differences and established markers; validated genes are labelled in grey. The proportional stacked bar graph inset indicates the composition of each cluster coloured by the original cell type. The number of cells in each cluster is indicated in brackets in the inset legend. b Heatmap of differential gene expression across cell clusters in (a), coloured by relative expression: -2 (magenta), 0 (black), and 2 (yellow). The top 10 marker genes per cluster highlight distinct transcriptional profiles; see Supplementary Data 2a for the full list. Colour bars represent each cell cluster. c UMAP and violin plots coloured by cell cluster display the expression of established markers used for cell annotation: MYO7A for sensory hair cells, and SOX2 for supporting cells and type II hair cells. d Dot plot illustrating the expression of representative marker genes (x-axis) for each main cell type (y-axis), along with established markers such as MYO7A, OCM, SOX2, and OTOL1; we also show the expression of specific markers for HC type II (CALB1) and SC-like cells (CRYAB and ANKRD1). Dot size corresponds to the percentage of cells within each cell type that express the marker, and colour represents the average gene expression (blue-low, yellow-high). e Dot plot showing the expression of SC validated genes, and validation of supporting cell populations using in situ hybridization; each dot represents a single transcript. Selected cluster-enriched genes CYR61 for SC-2, MANSC4 for SC-3, and PIGR for SC-5 were validated, n ≥ 3. High-magnification panels highlight the colocalization of the three cluster-defining genes (green) with SOX2 (magenta) in representative nuclei (blue), marked with a white circle in the images, low-magnification panels. Supporting cells expressing these cluster-enriched genes are interspersed in the sensory epithelium.
In this utricle, we identified five transcriptionally distinct supporting cell clusters and a previously uncharacterized SC-like cell population. All of the supporting cell clusters expressed established pan-SC markers like SOX2 and OTOL1, while the SC-like cells expressed cluster-specific genes such as CRYAB and ANKRD1 (Fig. 2d). To validate supporting cell cluster-defining genes (dot plot Fig. 2e, Supplementary Data 2a), we performed in situ hybridization using CYR61 for SC-2, MANSC4 for SC-3, and PIGR for SC-5 (Fig. 2e). Moreover, we investigated the expression patterns of vestibulopathy genes and genes associated with syndromic and nonsyndromic sensorineural hearing loss using a list of 158 genes21. Of the 68 vestibulopathy-related genes present in the list, 35% (24/68) were expressed in this utricle, 11 in supporting cells, including SC-like cells, and 13 in hair cells (Supplementary Fig. 1b). Similarly, for hearing loss genes, 30% (47/158) were expressed in our dataset, of which 36% (17/47) were in supporting cells and SC-like cells, and the remaining in hair cells (Supplementary Fig. 1c).
To confirm the cluster-defining genes for the main clusters, we used a recently published dataset (vestibular schwannoma 2, VS-2)22 and integrated it with our scUntr-2 dataset (Supplementary Figs. 2a, 6b). We reannotated and colour-coded the integrated clusters in four main clusters: types I and II hair cells, supporting cells, and SC-like cells (Supplementary Fig. 2b). To evaluate the similarity between the two human datasets across the main clusters, we measured the Euclidean distances between these expression profiles. Distance values were zero for all four clusters, confirming the perfect concordance between them (Supplementary Fig. 2c). In addition, using their integrated datasets of human utricles (n = 6, including VS-2)22, we demonstrate that pan-HC markers, hair cell subtype-specific markers, and pan-SC markers are consistent across human datasets (Supplementary Fig. 2d, e).
Putative markers identified in the utricular transcriptome
To discover unknown and enriched genes, we compared sensory hair cells (types I and II) with non-sensory supporting cells (SC1-5, excluding SC-like cells); 473 differentially expressed genes were identified (Padj < 0.05, log2 FC > |1 |), including eight transcription factors for hair cells and 25 for supporting cells (Fig. 3a, Supplementary Data 2b). We found GFI1, IRX2, LHX3, and POU4F3 expressed among the known transcription factors for hair cells. We also found transcription factors like SMARCE1, RARB and three members of the zinc-finger family (ZNF385A, ZNF397, ZNF688) that are poorly characterized in the inner ear. Among the HC-enriched genes, we found previously undescribed HC-specific markers like secretoglobin (SCGB) 2A1 and MINDY4B (Fig. 3b, Supplementary Data 2b). Among the identified 187 SC-defining genes, in addition to well-known inner ear transcription factors such as SOXs, ELF3, XPA, and IDs, we found NFIB (nuclear factor I B) enriched in supporting cells. In line with the recently published integrated human datasets22 (Supplementary Fig. 2f, SWNE plot), we also detected ATOH1 (Supplementary Fig. 2f, UMAP plot), a master transcription factor essential for hair cell development, in 10.3% (9/87) hair cells and 1.4% (12/843) supporting cells (Supplementary Data 2b; P = 3.08 ×10-5, Fisher’s exact test).
a Volcano plot showing differentially expressed genes enriched in hair cells and supporting cells identified using a two-sided Wilcoxon rank-sum test. The top differentially expressed genes are labelled (Padj < 0.05, log2 FC > |1 |). b Dot plot showing the percentage of cells expressing representative markers for hair cells (teal, type I; pink, type II) and supporting cells (grey box), with the dot colour indicating average gene expression. c Enriched type II (pink) and type I (teal) hair cell genes are identified using a two-sided Wilcoxon rank-sum test. Arrowhead indicates the validated gene CALB1. d Dot plot illustrating the relative expression of a subset of marker genes across all clusters, indicating specificity for type I (teal) or II (pink) hair cells. Supplementary Data 2b, c contain the complete gene lists for (a, c). e UMAP plots of hair cell subtypes as well as CALB1 (magenta), SOX2 (green), and cells coexpressing both markers (white) in the type II hair cell cluster. Percentage of transcript counts are reported, proportions are calculated as the fraction of cells with normalized expression greater than zero relative to the total number of cells in each cluster shown in the inset in Fig. 2a; 19 is the total number of type II hair cells. Schematic of CALB1 expression was created with gEAR using a scalable vector graphic file derived from a cropped and inverted image of Fig. 1a. Immunofluorescences (20x magnification) in a human utricle section demonstrate the expression of hair cell type II-specific marker CALB1 (magenta). MYO7A (yellow) was used as a pan-HC marker, and SOX2 (green) was used to label hair cell type II and supporting cells. Some hair cells (MYO7A+) also express CALB1 and SOX2, visible in the merge panel, n > 3. Scale bar = 25 μm.
To profile the transcriptome of hair cell subtypes, including subtype-specific markers, we examined differentially expressed genes between type I and II hair cells (Padj < 0.05, log2 FC > |1|, Fig. 3c, Supplementary Data 2c). Among the 30 differentially expressed genes, we found known markers like OCM for type I and SOX2 for type II. We also found, among others, ADAM11, PPP1R27, ENC1 to be specific for type I, and ZNF385B, AC025244.1, ELFN1, and CXCL14 for type II (Fig. 3d). Furthermore, we validated the expression of CALB1 to be specific for human type II hair cell (Fig. 3e), and confirmed this subtype specificity in the human-integrated datasets available on gEAR22 (Supplementary Fig. 2d), as well as in the integrated scUntr-2 and VS-2 datasets (Supplementary Fig. 2g, h).
Cross-species comparisons highlighted differences in utricular genes
We performed three cross-species transcriptomic analyses to assess any gene expression divergences between human and mouse utricles and identify species-specific signature genes for hair cells and supporting cells. Using Reciprocal Principal Component Analysis (RPCA)27 we integrated our human utricle dataset (scUntr-2) with the adult mouse utricle dataset14 (Fig. 4a–d, Supplementary Figs. 3a, 6c, Supplementary Data 3a, b). To identify human-enriched hair cell and supporting cell genes, we performed differential expression analysis between human and mouse hair cell subtypes (Fig. 4e, f, Supplementary Data 3c, d) and supporting cells (Fig. 4g, Supplementary Data 3e, f). We used two external datasets for in silico validations. The first was a P6 dataset containing both hair cell and supporting cell information10 (Supplementary Figs. 3b–f, 7d, Supplementary Data 3g–i), while the second was a P100 dataset comprising only hair cell data. However, due to the limited number of adult cells analyzed (25 hair cells), we also included hair cell data from the next most mature stage (P12) available from the same study13. By combining both datasets, we obtained 76 hair cells for subsequent differential expression analysis with human hair cells. We found 248 differentially expressed genes for hair cell type I, including 15 transcription factors (Supplementary Fig. 3g, Supplementary Data 3j), and 273 differentially expressed genes for hair cell type II, including 23 transcription factors (Supplementary Fig. 3h, Supplementary Data 3k). Differentially expressed genes identified from hair cell and supporting cell comparisons showed a high degree of consistency across all the three datasets.
a, b UMAP plots of the integrated adult human (967 cells) and mouse (2273 cells) datasets (a) with reannotated cell identities (b). c UMAP plots show known cell markers for each main cluster, genes with darker colours indicating higher expression levels. Transitional epithelial cells (TEC, ISLR+) and SC-like cells (ANKRD1+) remain distinct. d Heatmap displaying the top 10 differential gene expression of main cell clusters in (b); see Supplementary Data 3a for the full list. e–g Volcano plots of differentially expressed genes between mouse and human for hair cell type I (e), type II (f), and supporting cells (g), identified using a two-sided Wilcoxon rank-sum test; the arrowhead indicates the validated HC-human-specific gene SCGB2A1 (e, f); inset in (e) shows its relative expression in the hair cell clusters. Top genes with Padj < 0.05, log2 FC > |2| are displayed, complete lists in Supplementary Data 3c–f. h UMAP plots display the specificity of SCGB2A1 in the hair cell clusters (purple). Relative transcript counts in type II hair cell cluster for SCGB2A1 (magenta), SOX2 (cyan), and their co-expression (white) are shown, 19 is the total number of cells in this cluster. i Fluorescent in situ hybridization validated SCGB2A1 (magenta) mRNA expression in type II hair cells, counterstained with SOX2 (cyan) and DAPI (blue) in a utricle cross-section, n ≥ 3. Insets depict the colocalization of SCGB2A1 with SOX2, a marker for HC type II, in a representative nucleus marked with a white circle in the image. j UMAP plots display human-specific gene SCGB2A1 (magenta) in the hair cell clusters with pan-HC marker MYO7A (green), along with relative transcript counts across these clusters, 87 is the total number of hair cells, as shown in the inset in Fig. 2a; immunofluorescences displaying the protein expression of both markers in whole-mount utricle (63x magnification), n = 3. k Validation of hair cell marker SCGB2A1 (magenta) along with established markers SOX2 (cyan) for hair cell type II and MYO7A (green) in utricle cross-section, n ≥ 3. Representative hair cell types I and II are marked in the sensory epithelium. Scale bars = 25 μm, 2 μm (inset in i).
Our results revealed significant SCGB2A1 expression, specifically in human hair cells (Fig. 4h). SCGBs are exclusive to mammals28, and SCGB2A1 encodes a secreted protein with an unknown function in the inner ear. Interestingly, the mouse orthologue of this gene, Scgb2a2, was not found to be expressed in hair cells of inner ear sensory epithelia and utricle datasets publicly available on gEAR29 or published, suggesting it is a gene unique to human utricle hair cells. We validated SCGB2A1 expression in patient-derived samples (n ≥ 3) at the transcript and protein levels using in situ hybridization (Fig. 4i) and immunofluorescence (Fig. 4j, k), respectively. SCGB2A1 is an uncharacterized human-specific hair cell gene, and the encoded protein was observed to localize either in the cytoplasm or the intercellular space. We also confirmed that SCGB2A1 was expressed in the integrated datasets recently published22 (Supplementary Fig. 2d) and in the human-integrated datasets, scUntr-2 and VS-2, where its expression was restricted to hair cells (Supplementary Fig. 3i, j). Altogether, these results indicate some divergence in the transcriptomes of sensory and non-sensory cells, and their subtypes, between mice and humans.
To further investigate species-specific hair cell markers, we analyzed the integrated scUntr-2 and VS-2 datasets, focusing on the expression of a hair cell marker previously established in mice. As we described earlier, Calb1, which marks type I hair cells in mice, is instead enriched in human type II hair cells. We confirmed that CALB1 expression was consistently specific to type II hair cells across both human datasets (Supplementary Fig. 2g, h). This finding highlights that the HC-subtype marker CALB1 exhibits species-specific divergence between mouse and human.
A unique SC-like cell population
To assess how distinct the SC-like cell, supporting cell, and hair cell populations are from one another, we calculated the average expression level of each gene within each cluster, and used the Euclidean distances to compare these average expression vectors. We found that SC-like cells had a more similar transcriptional profile to supporting cells than hair cells (Figs. 2a, and 5a). These results also confirm that SC-like cells differ from the rest of the supporting cell population (distance value of 14.53) as they present unique gene signatures and potential biological features. Among the 24 cluster-defining differentially expressed genes, one-third of the genes are involved in the organization or remodelling of the cytoskeleton, like keratins and thymosin family genes, VIM, RRAS, and PALLD. To further elucidate the transcriptomic landscape of the SC-like population, we compared it with the other five supporting cell clusters, identifying 21 differentially expressed genes (Fig. 5b; Supplementary Data 2d), including ATF3, a well-known transcription factor involved in cellular stress responses30 and a recently proposed candidate for leading supporting cell proliferation and likely hair cell regeneration in the avian inner ear31. In addition, this analysis revealed that SC-like cells also expressed KRT7 (Keratin-7) and SFRP4 (Fig. 5c), the secreted frizzled-related protein 4 that acts as an inhibitor of Wnt signalling through direct interaction, antagonizing both canonical and noncanonical Wnt pathways32,33, regulating cell growth and differentiation. We also found IL-32 (Fig. 5c), LGALS3 (galectin-3), and ANX family genes, ANXA1 and ANXA2, among the upregulated genes. IL-32 may contribute to the modulation of the inflammatory response by regulating intracellular Ca2+ in the inner ear34, increasing calcium levels. Galectin-3 has anti-apoptotic activity, and Annexin A1 and A2 are known to be involved in the anti-inflammatory response. Additionally, ANXA1 is a pro-resolving mediator. The four members of the S100 genes (A1, A6, A10, and A11) were also significantly upregulated in SC-like cells. These genes encode a family of proteins that contain two EF-hand (helix E-lop-helix F) calcium-binding motifs, and the S100A10 member is also the ligand of ANXA235.
a Heatmap displaying the Euclidean distances between hair cell, supporting cell, and SC-like cell clusters in the subspace of PC1:50. The colour gradient represents the distance between clusters, with darker colours indicating a closer relationship. b SC-like cell cluster-enriched genes are depicted in a volcano plot. Using Padj < 0.05, log2 FC > |1|, 21 differentially expressed genes were identified between SC-like cells and the other five supporting cell clusters using a two-sided Wilcoxon rank-sum test. Arrowheads indicate validated genes. See Supplementary Data 2d for the list of differentially expressed genes. c UMAP plots show significantly SC-like cell-enriched genes with darker colours indicating higher expression levels. d In situ hybridization on a human utricle cross-section showing the expression of ANKRD1 (magenta) and CRYAB (green), counterstained with DAPI (blue), n ≥ 3. The expression is mainly present in the sensory epithelium near areas of missing cells marked with asterisks and dashed lines in the lower inset (63x magnification of the area outlined in white). High-magnification insets highlight the colocalization of ANKRD1 and CRYAB within a representative nucleus (blue), outlined with a white circle near a region of absent cells (marked by an asterisk). The upper inset displays UMAP plots of the two target genes, showing their in silico transcript counts within the SC-like cell cluster (37 cells in total) and a high-magnification image illustrating their co-expression. Scale bars = 100 μm, 25 μm and 2 μm in the lower insets, and 10 μm in the upper inset.
Furthermore, our analysis highlighted the expression of cluster-specific genes like ANKRD1 (Ankyrin Repeat Domain 1) and CRYAB (Alpha-crystallin B), Fig. 5b–d. ANKRD1 is a downstream YAP signalling gene involved in the murine utricle’s development and is expressed from embryonic stages until P236. CRYAB is a small heat shock protein with neuroprotective (otoprotective) and anti-inflammatory activities37. In mouse utricles, it is expressed in supporting cells10 and found to be upregulated after heat shock15. It is involved in proliferation, regeneration, and regulation of intracellular apoptosis signalling with an anti-apoptotic function. CRYAB is a stem/progenitor cell marker involved in regeneration of human interlaminar astrocytes38 and induces osteogenesis differentiation of human bone marrow stem cells, stabilizing β-catenin and promoting Wnt signalling39. We validated this SC-like cell type in adult human utricles (n ≥ 3) using RNA probes for ANKRD1 and CRYAB (Fig. 5d). In accordance with our sequencing data, we observed co-localization of these transcripts in a subset of cells; both transcripts are enhanced near areas of missing cells (Fig. 5d, insets).
Furthermore, we conducted a cluster similarity analysis between SC-like cells in scUntr-2 and VS-2 datasets using ClusterFoldSimilarity40, a statistical method that computes similarity coefficients based on gene fold changes between clusters from different datasets. This analysis confirmed the high similarity between the two clusters and their presence across human utricles (Supplementary Fig. 2i).
Overall, our computational analyses, supported by experimental validation, demonstrate the adult human utricle cell heterogeneity, leading to the discovery of SC-like cells and enriched markers for human hair cells and supporting cell subtypes.
Bulk RNA-seq revealed early transcriptional changes after ototoxic damage
To examine the early response of the human utricle to ototoxic damage, we sequenced RNA from the sensory epithelia of six utricles. Briefly, we cultured whole organotypic utricles for 24 h, three with vehicle control and three treated with an aminoglycoside (gentamicin) to induce hair cell damage. Then, we isolated the sensory epithelium and performed bulk RNA-seq (Fig. 6a). We used principal component analysis (PCA) to visualize variance between the two groups and similarity within sample replicates. This analysis revealed two distinct clusters representing each group. Samples undergoing the same treatment clustered together and were well-separated along PC1, indicating that the difference in gentamicin treatment was the primary source of variance across all the samples (Fig. 6b).
a Schematic of the damage paradigm experimental design. Created in BioRender. Luca, E. (https://BioRender.com/kw809th). b PCA of bulk RNA-seq samples; control (C, teal) and gentamicin-treated (T, orange), n = 3 per condition. c Dendrogram illustrates the hierarchical relationship within the two groups and between the two conditions (control and gentamicin treated), Padj < 0.05. Each column represents an individual biological sample, and each row a gene. Euclidean distances were computed, and hierarchical cluster analysis was performed using Ward clustering. The heatmap displays 47 differentially expressed genes (DESeq2, FDR < 0.05) between control and gentamicin-treated utricles, including five highlighted transcription factors. Colour scale represents gene expression value (magenta-downregulation, yellow-upregulation). Arrowhead indicates validated gene SLPI. See Supplementary Data 4a for the gene list.
We profiled the transcriptome of the two sample groups (n = 3 per condition) and identified 47 differentially expressed genes, 28 upregulated and 19 downregulated in the treated group, including five transcription factors and 14 druggable targets (Fig. 6c, Supplementary Data 4a). Among the upregulated genes, we identified FDZ8 receptor for WNT proteins and WNT activation target genes, ATF3 and JUN, both of which are known to be upregulated in response to injury41; COCH, responsible for cochleo-vestibular dysfunction42,43 in Deafness Autosomal Dominant 9 (DFNA9) and DFNA31 nonsyndromic forms of hearing loss; TCERG1, a candidate gene for nonsyndromic human deafness in the interval of DFNA42 locus44 and within mapped deafness loci DFNA54, DNFB6045; and CSF1, a cytokine that regulates macrophage activation and immune responses in the inner ear, that is necessary for the survival of otic macrophages46, although the source(s) of CSF1 in the ear have not been identified. We also identified matrix metalloproteinase (MMP) ADAMTS4; AC058791.1, an uncharacterized long intergenic non-coding RNA (lincRNA), and lncRNAs EPB41L4A-AS1, SNHG15. Among the downregulated genes, we found and validated SLPI (Secretory Leukocyte Protease Inhibitor), which is known to be involved in the innate immune response47,48, transcription factors ZBTB8B and CSRNP3, MMP ERMP1, and lncRNA NBAT1. These results profiled the early response to damage of the human utricle, highlighting a transcriptional difference after 24 h of gentamicin exposure. In summary, the expression of early responding genes (including upregulated and downregulated genes involved in immune responses, metalloproteases, and lncRNAs) suggests that the utricular damage triggers multiple cellular responses.
Single-cell analysis uncovers detailed early damage response
To define the transcriptional landscape following ototoxic damage at the single-cell resolution, we cultured utricles for 24 h with either vehicle control or gentamicin and performing scRNA-seq, n = 2 per condition (Fig. 6a). In one control sample cultured for 24 h (scCtrl-1), we found type I and type II hair cells, SC-like cells, and six clusters of supporting cells (Fig. 7a, b, and Supplementary Fig. 6e, Supplementary Data 5a), similar to the uncultured utricle (scUntr-2). Next, we clustered and annotated the cells for one gentamicin-treated sample (scGent-2); we detected a cluster of damaged hair cells (type I and II clustered together), five transcriptionally distinct populations of supporting cells, one SC-like cell cluster, and a distinct cluster of early-responding SC (Fig. 7c, d, Supplementary Fig. 6f, Supplementary Data 5b).
a, b UMAP plot of 2,033 cells (a) and heatmap displaying the top 10 marker genes for each cluster in the control sample scCtrl-1 (b). c, d UMAP plot (c) and heatmap (d) of gentamicin-treated sample (scGent-2) showing 2670 cells and top 10 differentially expressed genes. Colour bars at the top represent the cell clusters, and the relative expression scale indicates the gene expression level (magenta-low, yellow-high). A complete list of these genes is found in Supplementary Data 5a, b. e Heatmap of 365 differentially expressed genes identified between control (teal) and gentamicin-treated (orange) utricles using a two-sided Wilcoxon rank-sum test. f Violin plots showing representative downregulated and upregulated differentially expressed genes, validated genes are shown in green. g, h In situ validation of SLPI (g) and ANKRD1 (h) in control (teal) and gentamicin-treated (orange) samples; scale bars = 25 μm. Middle insets show the quantification of SLPI and ANKRD1 transcripts per cell in control (teal) and gentamicin-treated (orange) sensory epithelia (n = 5) and depict significant downregulation of SLPI and upregulation of ANKRD1 following gentamicin treatment. * Indicates P < 0.05 by two-tailed Welch’s t-test (SLPI: P = 0.0423, t = 2.886, df=4.189, 95% confidence interval -30.31 to -0.8543; ANKRD1: P = 0.0143, t = 4.127, df=4.028, 95% confidence interval 1.269 to 6.446). Graphs show the mean with standard deviation. See Supplementary Data 5d for raw data counts.
We also performed differential expression analysis between these utricles. After 24 h of damage, we identified 365 genes with significant transcriptional changes (114 upregulated and 251 downregulated), including 103 druggable targets and seven transcription factors (Fig. 7e, Supplementary Data 5c). Among the downregulated genes (Fig. 7f, upper panel), we found SLPI, CRYAB, ELF3, a transcription factor regulator of mesenchymal to epithelial transition49, ID4, a transcriptional target of Notch signalling, and NFIB, which is involved in retinal neuron regeneration processes in zebrafish, chicks, and mice50. Among the upregulated genes (Fig. 7f, lower panel), we found ANKRD1 and transcription factors NR1D1, a circadian gene that suppresses inflammation51, and HMGA1, which encodes for a nonhistone architectural chromosomal protein involved in epigenetic regulation, cell differentiation and proliferation, and DNA repair52. We also found FZD7, a gene related to the activation of Wnt/β-catenin signalling, and MIR4435-2HG, an immune-related lncRNA53. CDH11 was also upregulated; cadherin-11 binds to PDGFRβ and enhances cell proliferation and tissue regeneration via the PDGFR-AKT signalling axis54. Some genes of the S100A family were upregulated (1 A, A2, and A14) and downregulated (A6, A11) after gentamicin treatment. Previous studies have shown that gentamicin can induce ferroptosis55,56,57, an iron-dependent non-apoptotic cell death. In the gentamicin-treated sample, the top three genes with the highest fold change are key iron metabolism-associated ferroptosis-resistant genes that encode for iron storage proteins FTH1 and FTL, and ion transporter LCN2: all downregulated. Also, TIMP1, an inhibitor of MMPs, was downregulated.
To validate some identified post-damage genes, we performed in situ hybridization using anti-SLPI and anti-ANKRD1 probes in control and damaged utricles. SLPI is a gene that was also significantly downregulated in the bulk dataset after damage (Fig. 6c). ANKRD1, in addition to being a SC-like cell cluster-defining gene, was significantly upregulated in the damaged utricle (Fig. 7f, lower panel). In situ hybridization, supported by statistical analysis, confirmed significant downregulation of SLPI transcripts in treated utricle vs control (Fig. 7g) and upregulation of ANKRD1 (Fig. 7h), confirming in silico analysis, n = 5 (Supplementary Data 5d). Altogether, these data suggest an intrinsic capacity of the adult human utricle to activate a transcriptional response within 24 h of gentamicin exposure.
Multiple sample integration validated cluster-defining markers
To further investigate utricle heterogeneity across different treatment conditions at the single-cell level, we employed RPCA, to pool, integrate, and batch-correct all six samples (Supplementary Fig. 6g–i). We observed a robust alignment of cells across samples and experimental conditions (Supplementary Fig. 5a–f), indicating successful integration. Cluster markers (Supplementary Data 6a–c) remained consistent with non-integrated datasets, validating previous analyses (Figs. 2b, and 7b, d). Importantly, the consistency of cellular responses across diverse human samples subjected to the same conditions (untreated, 24 h control and gentamicin-treated) suggests shared underlying biological mechanisms. Although some degree of clustering by treatment was observed, this likely reflects treatment-induced transcriptional differences rather than residual batch effects. These results indicate that treatment-dependent transcriptional changes are a key driver of clustering, reinforcing the biological relevance of these differences.
An early-responding supporting cell population
Deciphering the immediate response to ototoxic damage, including any evidence for a regenerative response, is of fundamental importance for elucidating whether the adult human utricle has regenerative capacity. After only 24 h of gentamicin treatment, we detected a cluster of 122 supporting cells (Fig. 7c) with unique transcriptomic features (Fig. 8a, Supplementary Data 5b). We performed differential expression analysis between early-responding SC and supporting cell clusters, excluding SC-like cells, yielding 126 differentially expressed genes, finding 27 transcription factors, 21 upregulated and six downregulated (Fig. 8b, Supplementary Data 5e). Among the latter, we found NFIB, which maintains and restores quiescence, acting as a suppressor gene for proliferation and differentiation, and found to be downregulated immediately after damage50; representative transcription factors are shown (Fig. 8c). We also compared early-responding SC to SC-like cells and found 54 differentially expressed genes (Fig. 8d, Supplementary Data 5f). To determine the gene regulatory network of early-responding SC, we performed a network analysis using STRING (string-db.org58, P < 0.05, Fig. 8e, Supplementary Data 5e). The resulting network consisted of five subnetworks, including JUN and EGR families of transcription-regulatory factors; integrin superfamily members (ITGs) with THBS2 ligand; MMP9 and its inhibitor TIMP3. MMP9 participates in the shedding of membrane-anchored signalling molecules, like proHBEGF. When HBEGF is cleaved, it binds its receptor EGFR, the epidermal growth factor receptor, inducing cellular proliferation59,60. MMPs and TIMPs have a critical role in extracellular matrix (ECM) remodelling during regeneration of any tissue61,62,63,64. Loss of attachment to the ECM and neighbouring cells might be the first step of SC-to-HC transdifferentiation65.
a Dot plot displaying the top 30 early-responding SC-defining genes (Padj < 0.05) highlighted in yellow. b Volcano plot showing differentially expressed genes between early-responding SC and supporting cell clusters identified using a two-sided Wilcoxon rank-sum test, the top differentially expressed genes are labelled; Padj < 0.05, log2 FC > |1 | . c UMAP plots of representative downregulated and upregulated transcription factors in the early-responding supporting cell vs other supporting cell clusters. The colour gradient indicates statistically significant gene expression, with darker cells indicating higher gene expression. d Volcano plot with enriched SC-like cells vs early-responding SC genes, identified using a two-sided Wilcoxon rank-sum test. The top differentially expressed genes are labelled; Padj < 0.05, log2 FC > |1 | . e Gene network of selected differentially expressed genes (Supplementary Data 5e) between early-responding SC and supporting cells using STRING v12 (0.700 interaction score/high confidence). Markov Cluster Algorithm was used to group genes based on interaction strength indicated by line thickness (inflation parameter=2.4); dotted lines indicate edges between clusters. f Subclustering analysis of early-responding supporting cells reveals three distinct subpopulations: damage-responding, JUND+ (yellow); EGRs+ (violet); EDNRB+ MMPs+ (pink). g Slingshot v1.6.0 inferred pseudotime ordering, confirming the subclustering analysis (f). h UMAP plots, display validated genes EDNRB and MMP9 (both in green) for early-responding SC cluster (subcluster-3), along with relative transcript counts in ERSC cluster (122 cells in total as shown in the inset in Fig. 7c), SOX2 (magenta) is used as a pan-SC marker. Immunofluorescences show that cells expressing early-responding SC genes are located at the basal region of the sensory epithelium. High-magnification images highlight the colocalization of both cluster-defining genes (green) with SOX2 (magenta) within representative nuclei (blue), outlined in white in the low-magnification image (n = 3). Scale bars = 25 μm, 2 μm in the inset.
Furthermore, to enable pseudotemporal reconstruction, we first performed unsupervised clustering of early-responding SC and identified four distinct subclusters. We showed that subcluster-4, composed of 13 outlier cells, and one outlier cell from subcluster-3 were isolated from the other subclusters. Therefore, we excluded them from the trajectory analysis with Monocle 366 (Supplementary Fig. 4a). The remaining 108 cells were used to identify differentially expressed genes unique for each subcluster, discovering 60 transcription factors and 145 druggable targets (Fig. 8f, Supplementary Data 5g). We also conducted pseudotime trajectory analysis using Slingshot and examined the gene expression dynamics in early-responding SC, with subcluster-1, closest to supporting cells, set as the origin of the trajectory (Fig. 8g). In subcluster-1 (early genes), we detected early upregulated genes involved in the stress response, as well as JUND and JUN, also essential for proliferation, differentiation, and regeneration, including in the sensory organs67. Subcluster-2 (intermediate genes) exhibited upregulated genes such as EGR1, a downstream gene of JUN associated with tissue repair68. The upregulation of MMP9, EDNRB, GAP43, and APOE, among others, characterized subcluster-3 (late genes, Supplementary Fig. 4b). Mmp-9 regulates photoreceptor regeneration in adult zebrafish, promoting the survival of regenerated cones69; EDNRB (endothelin receptor) is a homolog of chicken EDNRB2, an early marker of SC-to-HC transdifferentiation in the avian cochlea70, a species where hair cells regenerate; GAP43 is a well-known gene involved in axonal growth and regeneration71; lastly, APOE promotes axonal regeneration in the mammalian central nervous system following injury, via the MAPK signalling pathway72. We carried out in situ hybridization to validate the ERSC cluster. We observed that early-responding supporting cells expressing EDNRB or MMP9 were located at the basal region of the sensory epithelium (Fig. 8h), adjacent to the mesenchymal tissue, where nascent hair cells (MYO7A+/SOX2-) were also present (Fig. 1f–h).
Discussion
In this study, we provided a comprehensive analysis of the adult human utricle transcriptome, revealing its cellular heterogeneity and early response to ototoxic damage. Using scRNA-seq, we generated a reference map of hair cells and supporting cells from patient-derived utricles. We discovered six transcriptionally distinct types of supporting cells, including a previously uncharacterized SC-like cell type. Additionally, we defined the early transcriptional changes occurring within 24 h of gentamicin exposure, uncovering a population of early-responding supporting cells. Our findings highlight key molecular features of the utricle’s sensory epithelium, including previously unreported genes and lists of druggable targets that could inform therapeutic strategies for balance disorders.
A major finding of this study is the identification of distinct supporting cell subtypes, underscoring a greater degree of heterogeneity than previously recognized. Transcriptionally distinct supporting cell subpopulations suggest functional diversification, with some subsets potentially primed for tissue damage repair and regeneration. Notably, the SC-like cell population expresses genes such as ANKRD1 and CRYAB, which are associated with cytoskeletal remodelling and stress response, indicating a possible role in cellular plasticity. Previous studies have shown that the transcription factor Ankrd1 is endogenously expressed in embryonic mouse utricles and is significantly downregulated by P236. ANKRD1 is, therefore, a utricle developmental marker, and the fact that it is expressed in the SC-like cells of the adult utricle suggests that this population may have some plasticity. Cryab was upregulated in supporting cells after reprogramming the cochlea (P1-P8) with hair-cell-specific transcription factors: Atoh1, Gfi1, and Pou4f373. The endogenous expression of ANKRD1 and CRYAB in adult human utricles has not been previously characterized, including their specific location within the sensory epithelium, where we showed expression in the surrounding area of missing cells. Furthermore, we identified ATOH1, a transcription factor that is both necessary and sufficient for hair cell formation74. Its expression may indicate the presence of immature hair cells, as we found in our cohort of adult human utricles, and that has been reported previously4,19,22. Altogether, our results raise important questions about whether specific supporting cell subtypes retain latent regenerative potential in the adult human utricle and how they might be harnessed for therapeutic purposes.
We also investigated the expression patterns of vestibulopathy genes and genes associated with syndromic and nonsyndromic sensorineural hearing loss in the scUntr-2 utricle using a previously published list21. Both classes of genes were expressed across all main cell types: hair cells, supporting cells, and SC-like cells. Similar results were obtained using different gene lists in Wang et al.22. Notably, a substantial portion of these genes was expressed in non-sensory cells, confirming their contributions to inner ear pathology. These analyses provide valuable insights into the cellular distribution of genes implicated in vestibular and auditory dysfunction and may help guide future studies on therapeutic targets for inner ear disorders.
We also performed cross-species comparisons using three mouse datasets ranging from P6 to adult stages, interrogating species-specific gene expression in hair cells and supporting cells. Our analysis highlighted conserved and divergent genes, as well as species-specific genes. We identified a discrepancy in the expression of some genes, leading to the discovery of SCGB2A1 as an HC-specific human gene. SCGB2A1 belongs to the secretoglobin family of proteins, primarily known for their roles in the respiratory system and has been associated with inflammation, tissue repair, and immune response. SCGB2A1 may also bind androgens and other steroids and be under the transcriptional regulation of steroid hormones28,75. Corticosteroids are the primary treatment for inner ear disorders characterized by hearing fluctuation or sudden changes in hearing, including sudden sensorineural hearing loss, Meniere’s disease, and autoimmune inner ear disease76. Therefore, it would be useful to characterize the function of this hair cell gene and the interplay with steroids in the human inner ear. Furthermore, CALB1 exhibited specificity for human type II hair cells, while in mouse, it is expressed in type I hair cells26. Understanding these distinctions is crucial for translating findings from mouse models to humans, as species differences may impact the efficacy and applicability of potential treatments for vestibular disorders. Consistent with our findings, this interspecies difference was documented in a recently published study22, which highlighted gene expression patterns between mouse and human utricles. These findings reinforce the need for human-based models to effectively translate research into potential clinical applications.
We also found that CXCL14 was significantly expressed in type II hair cells. In addition, our human/mouse cross-species comparisons between adult utricle type II hair cell indicate that this gene is significantly enriched in human type II HC. This gene is also expressed in mouse vestibular hair cells during development10,12,77 but without any distinction between murine HC subtypes at this stage. It was reported in HC of zebrafish lateral line neuromasts78, avian cochlea79, and was the most abundant transcript after damage and regenerative response in the avian utricle80. The enriched expression of CXCL14 in adult human type II hair cells and hair cells of species with spontaneous sensory cell regeneration, zebrafish and chicken, might warrant further investigation, especially given that regenerated hair cells in adult mice exhibit type II-like characteristics81.
Our hypothesis posits that the human utricle’s regenerative response is linked to its cellular heterogeneity and cell populations that can respond to damage. Although mice remain the mainstay for vestibular research82, particularly for ototoxicity studies, there is an increasing need for experiments on human vestibular tissues. Our study used high-throughput technologies to uncover ototoxic-induced transcriptomic responses in human utricles via bulk and scRNA-seq. We generated a cell-type-specific profile of the utricular transcriptional response after 24 h of gentamicin exposure. Combining the outcome of bulk and single-cell sequencing approaches, we detected over 400 differentially expressed genes. Notably, we found an early-responding SC population that upregulated the EDNRB gene in response to ototoxic damage, a homolog of the avian Ednrb2 gene involved in SC-to-HC transdifferentiation. Mutation in this gene causes Waardenburg Shah syndrome, featuring sensorineural hearing loss and pigmentary defects83. EDNRB can modulate several cellular responses, including cell proliferation, differentiation, survival, and regeneration84,85. Additionally, MMP9 and other matrix metalloproteinases were upregulated, indicating early extracellular matrix remodelling, a process critical for tissue repair. We found that early-responding SCs are situated at the base of the sensory epithelium, where nascent hair cells were also identified. These findings suggest that the adult human utricle retains some capacity to trigger a transcriptional program prone to repair and likely regenerative processes. Gaining a deeper understanding of the biological aspects of the early-responding SC will further enhance these efforts. Overall, characterizing the early stages of the utricle damage response is crucial for developing targeted therapeutics.
This exploratory study has certain limitations. The scRNA-seq datasets, while providing valuable insights into the cellular composition and damage response of the adult human utricle, include samples with relatively low median number of genes detected per cell. Reduced detection rates are not uncommon in studies involving adult human tissues, particularly from the inner ear; however, they may limit the depth of transcriptomic profiling and the potential for detailed comparative or integrative analyses. In this study, samples with the lowest mean/median number of detected genes per cell were used exclusively for in silico validation and not for primary analyses. Furthermore, the core biological findings were supported by orthogonal validation methods, including in situ hybridization and immunofluorescence, which reinforced our conclusions. Future studies with greater sequencing depth and larger sample sizes per group will further refine the transcriptional landscape and strengthen the reference framework for human vestibular research.
In summary, our study advances our knowledge of the human balance organ at the cellular and molecular levels in both homeostatic and ototoxic conditions, establishing a foundation for regenerative research and potential therapies to ameliorate inner ear diseases, including hearing loss and balance disorders.
Methods
Sample cohort, collections, and dissection
We enrolled 46 patients (21 to 75 y.o.) for this study. Patient cohort includes 57% (26/46) females (average age of 43.4 years) and 43% (20/46) males (average age of 48.6 years); Supplementary Data 1a. Samples ID, age, sex, affected side, and clinical information regarding hearing and vestibular symptoms were reported (Supplementary Data 1a). Utricles were collected from individuals with vestibular schwannomas undergoing surgical tumour resection via a translabyrinthine approach. A thorough lateral exposure was achieved to ensure clear visualization of the utricle. The utricle and its ampullary attachments were detached from the elliptical recess using a curved Rosen needle and a curved Fisch dissector. Then, tissue was carefully removed with fine forceps and transferred into 45 mL cold Tis-U-Sol® solution (Baxter Healthcare Corporation, Deerfield, IL 60015 USA) for bulk RNA-seq, or HBSS+/+ supplemented with 10 mM HEPES for scRNA-seq. The time between collecting and receiving the tissue in our laboratory was less than 10 minutes. Utricles were dissected in a black sylgard-coated dish immersed in the above solutions. Any bone fragments, residual blood, or otoconia were gently flushed away using a 1 mL syringe/30 G 1/2’’ needle filled with the above solutions.
Organ culture, treatments, and tissue preparation
Utricles were cultured free-floating for 24 h in DMEM/F-12 GlutaMAXTM medium (Thermo Fisher Scientific, 10565042) supplemented with 10% FBS (Invitrogen, 12484028), 10 mM HEPES (Gibco, 15630-080), 2.5 µg/mL fungizone (Gibco, 15290018), 10 µg/mL ciprofloxacin (Sigma-Aldrich, 17850-5G-F) at 37 °C in a humidified incubator with 5% CO2. Tissues were treated with 2 mM gentamicin (Gibco 15750-060 Gentamicin Reagent Solution, 50 mg/mL) or distilled water as vehicle control. After 24 h, samples were washed in HBSS+/+ supplemented with 10 mM HEPES, then incubated for 5-10 min at 37 °C in a glass vial with Rubber-Lined Cap, 1 dram, 15x45mm (Fisher, 03-339-25B) containing an enzymatic solution, 2 mL of HBSS+/+ with 1 mg/mL of thermolysin (Sigma-Aldrich, P1512), pre-heated for 10 min at 37 °C. For RNA-seq, sensory epithelia were isolated from the underlying mesenchymal stroma using forceps and a 1 mL insulin syringe/30 G 1/2’’ needle.
For hematoxylin and eosin staining, immunofluorescence, and in situ hybridization, after dissection, whole utricles were immediately fixed with freshly prepared 4% formaldehyde in 1x PBS – derived from a 16% (w/v) solution of paraformaldehyde in distilled/deionized water – overnight (o.n.) at 4 °C; then processed for sectioning or used as whole mounts. For cryosectioning, tissues were washed in 1x PBS, underwent a serial sucrose gradient (10%, 20%, 30%), cryo-embedded in Tissue Plus O.C.T. compound (Fisher Health Care), and stored at -80 °C in a sealed plastic bag. After cryopreservation, 10-μm-thick sections were sectioned and stored at -80 °C in a sealed box with desiccant (Humidity Sponge, VWR 61161-319).
Bulk RNA sequencing and analysis
Six human utricle samples were processed for bulk RNA-seq, comprising three biological replicates per condition (control: n = 3; gentamicin-treated: n = 3). RNA from delaminated sensory epithelia was extracted using Quick-RNATM Microprep kit according to the manufacturer’s guidelines. Libraries were prepared following ribosomal RNA depletion using the Illumina Truseq Stranded mRNA Library Prep kit and then sequenced with the HiSeq 2000 platform (Illumina) using the 100-bp paired-end strategy. Quality control of raw reads was performed using RNA-SeQC v1.1.786 for all six RNA-seq samples, and adapters were trimmed with cutadapt v1.9.187. Reads were then aligned to the appropriate reference genome (Human GRCh38, GENCODE v32/Ensembl98) using STAR v2.5.288. Gene abundances were quantified with RSEM v1.3.089 using default settings. All estimated gene abundances were imported into R v4.1.1 with tximport, and genes with total counts < 10 across all samples were filtered out before downstream analyses. The R package DESeq2 v1.28.190 was used for variance stabilizing transformation, normalization (DESeq2’s median of ratios method), and differential gene expression. Confounding effects (i.e., sex and age) were accounted for in the generalized linear model. Differential genes were defined using an FDR (false discovery rate) threshold of 0.05. Variance-stabilized counts were used for principal component analysis and sample-to-sample distance assessments to evaluate batch effects and sample separation.
Sensory epithelia dissociation for scRNA-seq
Six human utricle samples were processed for scRNA-seq, comprising two biological replicates per condition (untreated: n = 2; control: n = 2; gentamicin-treated: n = 2). Utricles were transferred with a mini perforated spoon (FST) in 1 mL cold DMEM/F-12 GlutaMAXTM (Thermo Fisher Scientific, 10565042) supplemented with 10% FBS for a washing step. Sensory epithelia were delaminated using fine Dumont Forceps (FST) and a 1 mL syringe/30 G 1/2’’ needle in a clean sylgard-coated dish with the solution as above. Then, using a spoon micro curette (FST), they were transferred to the bottom of a low binding/DNase-free tube with 200 µL of cold DMEM/F-12 GlutaMAXTM supplemented with 10% FBS. Media was removed, and a fresh-prepared dissociating mix was added: 200 µL (10 mg/mL) Native Bacillus licheniformis Protease–Subtilisin, (Creative Enzymes NATE-0633); 1 µL (125 U/mL) Deoxyribonuclease I from bovine pancreas (DNase I - Sigma-Aldrich D5025-15KU); 1 µL (10 µg/mL) N-Acetyl-L-cysteine (Sigma-Aldrich A9165-5G); 1 µL (10 µg/mL) 2-Phospho-L-ascorbic acid trisodium salt (Sigma-Aldrich 49752-10 G). Tissues were incubated for 30–40 min at 4 °C and triturated every 5 min with a P200 pipette set at 50 µL (wide-bore tip) until complete dissociation. Vertex Filter Tips, Clear, Graduated, and NoStick®, were used for the entire procedure. 500 µL of DMEM/F-12 GlutaMAXTM medium supplemented with 10% FBS and 1% MACS® BSA Stock Solution (Miltenyi Biotech) was added to the enzymatic dissociation mix. Cell viability for each sample is reported in Supplementary Table 1.
Microfluidic capture of single cells and generation of cDNA
Sample preparation and cell capture were performed using the 10x Genomics Chromium Single Cell 3’ v3 Reagent Kits user guide (10x Genomics, Pleasanton, CA). Briefly, two washes were done to exchange sample buffer for one recommended for loading on the Chromium Controller. Samples were spun at 0.3 G for 6 min at 4oC and resuspended in 250 µL RPMI-1640 Medium with 0.04% BSA and 5 U/µL RNase Inhibitor. Cell viability was assessed via Trypan Blue (Thermo Fisher) and using a hemocytometer (Thermo Fisher). A second spin was performed under identical conditions, and cells were resuspended in 55 µL final volume. Following counting, cell suspension (46.6 µL) was loaded onto the Chromium Single Cell B Chip Kit. After droplet generation, samples were transferred onto a pre-chilled 96-well plate (Eppendorf), heat-sealed, and incubated overnight in a Veriti 96-well thermal cycler (Thermo Fisher) for the reverse transcriptase reaction. Following this reaction, cDNA was recovered using the Recovery Agent provided by 10x Genomics and subsequently cleaned up using a Silane DynaBead (Thermo Fisher) as outlined by the user guide. Purified cDNA was amplified as follows: 12 cycles for control utricles and 15 cycles for uncultured and gentamicin-treated utricles before cleaning up using SPRIselect beads (Beckman). Samples were run neat on a Bioanalyzer (Agilent Technologies) to determine cDNA concentration. cDNA libraries were prepared as outlined by the Single Cell 3’ Reagent Kits v3 user guide with 16 cycles of PCR amplification based on the calculated cDNA concentration. A final clean-up followed this with SPRIselect beads (Beckman). Sample quantity was assessed by NanoDrop (Thermo Scientific) and normalized to ~5 ng/µL.
scRNA-seq library construction and sequencing
The molarity of each library was calculated based on library size as measured by BioAnalyzer (Agilent Technologies), qPCR amplification data (Sigma) or Illumina MiSeq clustering data (Illumina). Details for each sample are listed in Supplementary Table 2. Briefly, samples were normalized, then diluted using elution buffer (Qiagen) with 0.1% Tween® 20 (Sigma-Aldrich), and each pool denaturated at RT. Library pools were further diluted before being diluted to a final loading concentration. 100 µL from each pool were loaded into each well of an 8-well strip tube and loaded onto a cBot (Illumina) for cluster generation. All samples were sequenced with the following run parameters: read 1–28 cycles, read 2–91 cycles, and index 1–8 cycles. A median sequencing depth of 60,000 reads/cell was targeted for each sample.
scRNA-seq data analysis
Quality control metrics for all FASTQ reads were obtained using RNA-SeQC v1.1.7. The CellRanger v3.0.2 pipeline was used for primary data processing and the production of gene-barcode matrices, with settings as recommended by the manufacturer (10x Genomics). The raw gene-barcode matrices for all groups (Uncultured, Control, and Damaged; two samples per group) were processed as follows: first, cell quality was determined by examining the distribution of library sizes, the number of expressed genes across cells, and the fraction of reads mapped to mitochondrial (MT) genes. Then, we filtered out doublets using DoubletFinder v2.091. Cells with log-library sizes that were more than three median absolute deviations (MADs) below the median log-library size were also filtered out92. The MAD approach was also used to determine mitochondrial expression cutoffs for each group (Supplementary Table 1). Genes detected in less than three cells were removed. The total cell counts after QC are provided in Supplementary Table 1. Batch effects were regressed from the integrated data using Seurat’s (v5.1.0) reciprocal principal component analysis. In brief, each sample was individually normalized and scaled with Seurat’s SCTransform function. The top 3000 highly variable genes (HVGs) were used as the integration features for each sample (nfeatures = 3000), SelectIntegrationFeatures function. RPCA integration anchors were estimated by setting k.anchor to 25, and all samples were integrated using the IntegrateData function with k-nearest neighbors (k.weight) set to 56. This integrated RPCA object was then used for downstream analysis. Clusters were identified using an iterative SNN-Cliq-inspired clustering approach (Seurat’s FindCluster function), with dims=PC1:PC18 for the integrated data. Optimal clustering resolution values were decided using the Clustree93 R package (Supplementary Fig. 6a–i), as well as by exploring the biological significance of the differentially expressed genes between all clusters (using Seurat’s FindMarkers function; two-sided Wilcoxon rank-sum test, with a Bonferroni adjusted p-value threshold of 0.05). We identified the transcription factors among the differentially expressed genes using AnimalTFDB94 and druggable targets using DGIdb95. The RunUMAP function was used to generate UMAP plots. UMAP was selected as the primary projection technique due to its superior preservation of the data’s global structure96. Clusters were visualized by plotting the reduction values on a Cartesian graph. The following well-characterized marker genes were used to identify known cell types: GFI1, MYOSIN (6, 7 A, 15 A), OCM, OTOF, POU4F3, and TMC1 for hair cells; GFAP, KREMEN1, OTOL1, PLP1, SOX2, and SPARCL1 for supporting cells. Cell-type identification was also facilitated by using the differentially expressed genes (upregulated marker genes) in each cluster. We calculated the Euclidean distances between the clusters (HC, SC and SC-like cell) in the subspace of PC1:PC50 when annotating the SC-like cell cluster.
Trajectory analysis
Trajectory analysis for the early-responding supporting cell (ERSC) cluster in the gentamicin-treated sample was performed using Monocle 3 v1.3.466 and Slingshot v1.6.097, setting subcluster-1 as the starting point. To gain deeper insights into the cellular dynamics across the clusters, we visualized the expression changes of marker genes for each subcluster (P < 0.05; log2 FC > 0.25) along with the trajectory.
Human datasets integration and comparisons
For human-specific comparisons, the datasets in Wang et al.22, were used. We employed the Seurat (v5.0.3) scRNAseq workflow for normalization (SCTransform method), data integration using RPCA (IntegrateLayers function), clustering, and creating visualizations for the two patient-derived datasets scUntr-2 (967 cells) and VS-2 (1124 cells). Clustree v0.5.1, in addition to the differentially expressed genes between clusters, was used to select the optimal clustering resolution, with a final value of 0.6 (Supplementary Fig. 6b). Canonical markers were found using the FindAllMarkers function using the following parameters: returning all positive markers, return.thresh = 1, and min.pct = 0.25. Correlation analysis between clusters was performed using ClusterFoldSimilarity v1.0.0. We also used an integrated dataset of human utricles (n = 6), which was recently published22 and available on gEAR, and performed cluster-defining gene comparisons for hair cells, their subtypes, and supporting cells.
Comparison between human and mouse datasets
For cross-species comparison, three scRNA-seq datasets for mouse utricles were downloaded from NCBI Gene Expression Omnibus (GEO): GSM 771038314; GSE 11593413; GSE 15596610. For GSM 7710383 cells with >20% mitochondrial UMIs or >25,000 total UMIs were excluded during quality control. Data were normalized using SCTransform v2. Clusters representing the main sensory epithelium cell types (HCs, SCs, TECs) were identified using canonical markers (EPCAM, MYO7A, SOX2, ISLR). To enable cross-species integration, mouse gene symbols were matched to human orthologs using OrthoIntegrate98. The renamed adult mouse dataset (909 hair cells – 470 type I, 439 type II, 923 supporting cells, 441 transitional epithelial cells) was merged with human scUntr-2 dataset (87 hair cells – 68 type I, 19 type II, 843 supporting cells, 37 SC-like cells). Integration was performed using Seurat v5’s IntegrateLayers with RPCA and k.anchor = 25 and clustering resolution of 0.1. Cell types were annotated based on marker expression, and cluster-specific differentially expressed genes were identified using FindAllMarkers and FindMarkers using MAST test. Conserved markers across species were determined with FindConservedMarkers. The lists of differentially expressed genes were filtered using the Housekeeping Transcript Atlas to remove housekeeping genes, and low-expression genes using pct_diff ≤0.25. Mouse genes without human orthologues retained their original gene symbols. Top markers per cluster were used to generate heatmaps and volcano plots highlighting species-specific differences. For GSE115934 (P12 + P100 datasets), we generated the gene count matrix from the FASTQ files using kallisto v0.46.099 (reference transcriptome: mm10 RefSeq genes). For GSE155966 (P6 dataset), we utilized the processed data downloaded from GEO. Next, we applied quantile normalization100 to both mouse datasets. Afterward, to compensate for sequencing depth differences, we normalized human and mouse datasets using SCTransform v0.3.3101 in the Seurat v4.1.027 workflow. To convert mouse gene symbols to corresponding human ortholog gene symbols, we first identified orthologs between human and mouse based on the vertebrate ortholog list from the Mouse Genome Informatics database (informatics.jax.org), downloaded in June 2022. If multiple orthologs were mapped between species, we chose the genes expressed in the human dataset. If multiple orthologs were present in the human dataset, we did not use those orthologs. Then, we selected the top 3000 common variable genes and integrated the human dataset and either of the two mouse datasets based on the RPCA method27. We visualized integrated datasets using PCA and UMAP to find the similarities and differences of cell types between species. We performed clustering with the resolution parameter of 0.4 determined by the Clustree R package. Then, we annotated the new clusters based on the proportion of original cell types. We applied log-normalization to raw count matrices for differential expression analysis and performed a two-sided Wilcoxon rank-sum test between species, with a Bonferroni Padj < 0.05, log2 FC > |2| . Results were visualized using the EnhancedVolcano R package (v1.12.0).
Hematoxylin and eosin (H&E) staining and immunofluorescence
H&E staining of 10 μm-thick cross-sections was conducted using standard protocol for hematoxylin Gill II (Leica, 3801522), slides were mounted with Eukitt® mounting medium (Sigma-Aldrich, 03989), n > 3. For immunofluorescence, utricle cross-sections were permeabilized with 1x PBS with 0.5% Triton X-100 (Fisher #BP151-500) for 30 min, followed by 30 min of antigen blocking with 10% donkey serum in 1x PBS 0.5% Triton X-100 containing 0.3 M glycine. Primary antibodies were incubated o.n. at 4 °C (Supplementary Table 2); we performed heat-induced epitope retrieval using ACD (Advanced Cell Diagnostics) Target Retrieval Reagent for SCGB2A1. Species-specific Alexa-conjugated secondary antibodies (1:1000; Invitrogen) and DAPI (1 µg/mL; Invitrogen #D1306) were applied for 1 h at RT in 1x PBS with 0.5% Triton X-100. ProLongTM Gold Antifade Mountant was used for mounting the slides.
Fluorescent in situ hybridization
RNAscope® assay (ACD) validated the candidate marker for hair cells and cluster-specific supporting cell markers. Briefly, antigen retrieval was performed at 99 °C for 10 min, followed by protease III treatment for 20 min at 40 °C. Probe hybridizations and amplification steps were carried out following the manufacturer’s protocol (RNAscope Multiplex Fluorescent Reagent kit v2 Assay, 323100-USM). Positive and negative control probes were used with every sample during every run. Sections were counterstained with DAPI and mounted, n ≥ 3.
Transcripts quantification and statistical analysis
For each condition, control and gentamicin-treated samples (n = 5, Supplementary Data 5d), a 200 µm segment of the sensory epithelium was selected for quantifying SLPI and ANKRD1 transcripts as well as nuclei. Nuclei counts were performed manually, and automated transcript quantification was conducted using Fiji software and ACD protocol to analyze RNAscope data. Briefly, the fluorescent image of the target probe was converted to a binary image (“Image > Adjust > Threshold”) using Otsu’s method to isolate the probe signal. The area of a single transcript (“Analyze > Measure”) was used as the lower threshold for automated particle analysis (“Analyze > Analyze Particles”). Clusters of probe signals were quantified by dividing the cluster area by the area of a single transcript. Data were normalized to the number of nuclei. Statistical analysis was performed with GraphPad Prism software. Comparisons between conditions were conducted using a two-tailed Welch’s t-test, with significance set to P < 0.05. Data are presented as the mean ± standard deviation.
Scanning electron microscopy
Dissected utricles were fixed (2.5% glutaraldehyde in 0.1 M cacodylate buffer, pH 7.4) o.n. at 4 °C. Tissues were washed in PBS for 15 min and immersed in 1% buffered OsO4 for 1.5 h at RT, washed again and dehydrated using a series of ethanol with increasing concentration (30%, 50%, 70%, 90%, 95%, and 100%) in a rotating rack. Utricles were fully immersed in ethanol during this process, as exposing the samples to the air can damage the architectural structures. Then, samples were transferred into microporous capsules for sample protection during critical point drying, filled with 100% ethanol. Samples were rinsed three times in a CO2 aqueous solution and maintained at a pressure of 800 psi. When the pressure increased to 1500 psi at 42 °C, samples were critical-point dried. Then, utricles were mounted on stubs covered with adhesive carbon sheets, gold-sputtered for 2 min, and stored at 37 °C until imaging; n = 3.
Imaging
Imaging was carried out using a Leica DIM6000 SP8 Lightning Confocal/STED microscope, with gating function engaged, and a Nikon A1 confocal. For each sample, positive and negative controls were used to set the background threshold, laser power, and gain. These settings were then applied to capture pictures using 20x/0.75 NA, 40x/1.3 NA and 63x/1.4 NA oil immersion objectives. Images were captured using z-projection from a stack covering the entire depth of the utricle section. Z-stack step size was 0.35 μm and 0.2 μm for SCGB2A1. For whole-mount utricle, Z-stack step size was 0.15 μm. For the analysis of SLPI and ANKRD1 transcripts in control and gentamicin-treated samples, representative images from in situ experiments were captured using a Leica DMi8 inverted microscope with a 40x/0.95 DRY objective. H&E stained image was acquired using a Nikon Eclipse Ti microscope equipped with a DS-Ri2 camera and a 20×/0.75 NA objective under brightfield illumination and tiling mode. Scanning electron micrographs were acquired with Hitachi S-3400N microscope. Image processing was performed using Fiji.
Statistics & reproducibility
For this exploratory study, no statistical method was used to predetermine sample size, and sex- and gender-based analyses were not performed. However, the findings apply to both sexes, and sex information was collected via self-report. All experiments included multiple replicates and, unless otherwise specified, were performed in at least triplicate. Statistical analyses are detailed in the relevant sections. A P-value < 0.05 was considered statistically significant. Samples were randomly assigned to experimental groups, ensuring representation across both sexes and a range of ages (21–75 years). Investigators were not blinded to group allocation during experiments or outcome assessment. Data excluded from the analyses were limited to poor-quality cells identified and removed during scRNA-seq quality control filtering.
Ethics statements
This study (Project Identification Number: 1711) was approved by the Research Ethics Board of Sunnybrook Health Science Centre. The Sunnybrook Department of Otolaryngology—Head & Neck Surgery assessed participants’ hearing and balance function and obtained written informed consent from all participants. No compensation was provided for participation in this study. Two expert otolaryngologists (VL and JC) collected utricles during translabyrinthine surgeries between March 2016 and February 2024, informed consent was obtained from all.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
For cross-species comparisons, the following datasets were used: GSM7710383; GSE115934; and GSE155966. For human-specific comparisons, dataset GSE207817 was used. The RNA-seq data generated in this study have been deposited in the NCBI GEO database under accession code GSE244056. Datasets are available for viewing and analysis through the gEAR Portal in the datasets “Adult human utricle - Bulk RNA-seq and scRNA-seq (Luca/Dabdoub, 2023)” at umgear.org/p?l=human-utricle-2023, and Trailmaker repository, entitled “Adult human utricle - scRNA-seq (Luca/Dabdoub, 2023)” at app.trailmaker.parsebiosciences.com/repository. All raw and processed data generated in this study are provided in the Supplementary Information and Supplementary Data files. Samples used for RNA-seq were destroyed during processing. Researchers interested in accessing future collected samples can contact the corresponding authors.
References
Monzack, E. L. & Cunningham, L. L. Lead roles for supporting actors: critical functions of inner ear supporting cells. Hear Res 303, 20–29 (2013).
Wan, G., Corfas, G. & Stone, J. S. Inner ear supporting cells: rethinking the silent majority. Semin. Cell Dev. Biol. 24, 448–459 (2013).
Huang, Y., Mao, H. & Chen, Y. Regeneration of hair cells in the human vestibular system. Front. Mol. Neurosci. 15, 854635 (2022).
Taylor, R. R. et al. Characterizing human vestibular sensory epithelia for experimental studies: new hair bundles on old tissue and implications for therapeutic interventions in ageing. Neurobiol. Aging 36, 2068–2084 (2015).
Agrawal, Y., Carey, J. P., Della Santina, C. C., Schubert, M. C. & Minor, L. B. Disorders of balance and vestibular function in US adults: data from the National Health and Nutrition Examination Survey, 2001–2004. Arch. Intern Med. 169, 938–944 (2009).
Burns, J. C., Cox, B. C., Thiede, B. R., Zuo, J. & Corwin, J. T. <em>In Vivo</em> proliferative regeneration of balance hair cells in newborn mice. J. Neurosci. 32, 6570–6577 (2012).
Wang, T. et al. Lgr5+ cells regenerate hair cells via proliferation and direct transdifferentiation in damaged neonatal mouse utricle. Nat. Commun. 6, 6613 (2015).
Lin, V. et al. Inhibition of Notch activity promotes nonmitotic regeneration of hair cells in the adult mouse utricles. J. Neurosci. 31, 15329–15339 (2011).
Gao, Z. et al. Spatial and age-dependent hair cell generation in the postnatal mammalian utricle. Mol. Neurobiol. 53, 1601–1612 (2016).
Jan, T. A. et al. Spatiotemporal dynamics of inner ear sensory and non-sensory cells revealed by single-cell transcriptomics. Cell Rep. 36, 109358 (2021).
Burns, J. C., Kelly, M. C., Hoa, M., Morell, R. J. & Kelley, M. W. Single-cell RNA-Seq resolves cellular complexity in sensory organs from the neonatal inner ear. Nat. Commun. 6, 8557 (2015).
You, D. et al. The heterogeneity of mammalian utricular cells over the course of development. Clin. Transl. Med. 12, e1052 (2022).
McInturff, S., Burns, J. C. & Kelley, M. W. Characterization of spatial and temporal development of Type I and Type II hair cells in the mouse utricle using new cell-type-specific markers. Biol. Open 7, bio038083 (2018).
Wang, T. et al. Precise genetic control of ATOH1 enhances maturation of regenerated hair cells in the mature mouse utricle. Nat. Commun. 15, 9166 (2024).
Sadler, E. et al. Cell-specific transcriptional responses to heat shock in the mouse utricle epithelium. Front Cell Neurosci. 14, 123 (2020).
Jen, H. I. et al. Transcriptomic and epigenetic regulation of hair cell regeneration in the mouse utricle and its potentiation by Atoh1. Elife 8, e44328 (2019).
Warchol, M. E., Lambert, P. R., Goldstein, B. J., Forge, A. & Corwin, J. T. Regenerative proliferation in inner ear sensory epithelia from adult guinea pigs and humans. Science 259, 1619–1622 (1993).
Forge, A., Li, L., Corwin, J. T. & Nevill, G. Ultrastructural evidence for hair cell regeneration in the mammalian inner ear. Science 259, 1616–1619 (1993).
Taylor, R. R. et al. Regenerating hair cells in vestibular sensory epithelia from humans. eLife 7, e34817 (2018).
Schrauwen, I. et al. A comprehensive catalogue of the coding and non-coding transcripts of the human inner ear. Hear Res 333, 266–274 (2016).
van der Valk, W. H. et al. A single-cell level comparison of human inner ear organoids with the human cochlea and vestibular organs. Cell Rep. 42, 112623 (2023).
Wang, T. et al. Single-cell transcriptomic atlas reveals increased regeneration in diseased human inner ear balance organs. Nat. Commun. 15, 4833 (2024).
Wu, Y., Tamayo, P. & Zhang, K. Visualizing and interpreting single-cell gene expression datasets with similarity weighted nonnegative embedding. Cell Syst. 7, 656–666.e654 (2018).
Simmons, D. D., Tong, B., Schrader, A. D. & Hornak, A. J. Oncomodulin identifies different hair cell types in the mammalian inner ear. J. Comp. Neurol. 518, 3785–3802 (2010).
Deans, M. R., Peterson, J. M. & Wong, G. W. Mammalian Otolin: a multimeric glycoprotein specific to the inner ear that interacts with otoconial matrix protein Otoconin-90 and Cerebellin-1. PLoS One 5, e12765 (2010).
Stone, J. S., Pujol, R., Nguyen, T. B. & Cox, B. C. The transcription factor Sox2 is required to maintain the cell type-specific properties and innervation of type ii vestibular hair cells in adult mice. J. Neurosci. 41, 6217–6233 (2021).
Hao, Y. et al. Integrated analysis of multimodal single-cell data. Cell 184, 3573–3587.e3529 (2021).
Jackson, B. C. et al. Update of the human secretoglobin (SCGB) gene superfamily and an example of ‘evolutionary bloom’ of androgen-binding protein genes within the mouse Scgb gene superfamily. Hum. Genomics 5, 691–702 (2011).
Orvis, J. et al. gEAR: gene expression analysis resource portal for community-driven, multi-omic data exploration. Nat. Methods 18, 843–844 (2021).
Hai, T., Wolfgang, C. D., Marsee, D. K., Allen, A. E. & Sivaprasad, U. ATF3 and stress responses. Gene Expr. 7, 321–335 (1999).
Benkafadar, N. et al. An essential signaling cascade for avian auditory hair cell regeneration. Dev. Cell 59, 280–291.e5 (2024).
Kawano, Y. & Kypta, R. Secreted antagonists of the Wnt signalling pathway. J. Cell Sci. 116, 2627–2634 (2003).
Muley, A. et al. Secreted frizzled-related protein 4: an angiogenesis inhibitor. Am. J. Pathol. 176, 1505–1516 (2010).
Jeong, H. J., Han, N. R., Moon, P. D., Kim, M. H. & Kim, H. M. Intracellular calcium level is upregulated by interleukin-32 in auditory cells. Cytokine 53, 153–157 (2011).
Choi, S. et al. Encyclopedia of Signaling Molecules. (Springer New York, 2017).
Gnedeva, K., Jacobo, A., Salvi, J. D., Petelski, A. A. & Hudspeth, A. J. Elastic force restricts growth of the murine utricle. Elife 6, e25681 (2017).
Erni, S. T. et al. Anti-inflammatory and Oto-Protective Effect of the Small Heat Shock Protein Alpha B-Crystallin (HspB5) in Experimental Pneumococcal Meningitis. Front. Neurol. 10, 570 (2019).
Falcone, C. et al. Cortical interlaminar astrocytes are generated prenatally, mature postnatally, and express unique markers in human and nonhuman primates. Cereb. Cortex 31, 379–395 (2020).
Zhu, B., Xue, F., Li, G. & Zhang, C. CRYAB promotes osteogenic differentiation of human bone marrow stem cells via stabilizing β-catenin and promoting the Wnt signalling. Cell Prolif. 53, e12709 (2020).
González-Velasco, Ó. Identifying similar populations across independent single cell studies without data integration. NAR Genom. Bioinform. 7, lqaf042 (2025).
Kiryu-Seo, S. & Kiyama, H. The nuclear events guiding successful nerve regeneration. Front Mol. Neurosci. 4, 53 (2011).
Robertson, N. G. et al. Mutations in a novel cochlear gene cause DFNA9, a human nonsyndromic deafness with vestibular dysfunction. Nat. Genet 20, 299–303 (1998).
Usami, S. -i et al. Mutations in the COCH gene are a frequent cause of autosomal dominant progressive cochleo-vestibular dysfunction, but not of Meniere’s disease. Eur. J. Hum. Genet. 11, 744–748 (2003).
Sajan, S. A., Warchol, M. E. & Lovett, M. Toward a systems biology of mouse inner ear organogenesis: gene expression pathways, patterns and network analysis. Genetics 177, 631–653 (2007).
Shen, J., Scheffer, D. I., Kwan, K. Y. & Corey, D. P. SHIELD: an integrative gene expression database for inner ear research. Database (Oxf.) 2015, bav071 (2015).
Manickam, V. et al. Macrophages promote repair of inner hair cell ribbon synapses following noise-induced cochlear synaptopathy. J. Neurosci. 43, 2075–2089 (2023).
Velarde, M. C., Parisek, S. I., Eason, R. R., Simmen, F. A. & Simmen, R. C. The secretory leukocyte protease inhibitor gene is a target of epidermal growth factor receptor action in endometrial epithelial cells. J. Endocrinol. 184, 141–151 (2005).
Wu, M., Xia, M., Li, W. & Li, H. Single-cell sequencing applications in the inner ear. Front Cell Dev. Biol. 9, 637779 (2021).
Sengez, B. et al. The transcription factor Elf3 is essential for a successful mesenchymal to epithelial transition. Cells 8, 858 (2019).
Hoang, T. et al. Gene regulatory networks controlling vertebrate retinal regeneration. Science 370, eabb8598 (2020).
Wang, Z. et al. Clock Gene Nr1d1 alleviates retinal inflammation through repression of Hmga2 in microglia. J. Inflamm. Res 14, 5901–5918 (2021).
Chiefari, E. et al. Transcriptional regulation of glucose metabolism: the emerging role of the HMGA1 chromatin factor. Front. Endocrinol. 9, 357 (2018).
Jiang, H., Sun, J., Liu, F., Wu, X. & Wen, Z. An immune-related long noncoding RNA pair as a new biomarker to predict the prognosis of patients in breast cancer. Front Genet 13, 895200 (2022).
Liu, Y., Lei, P., Row, S. & Andreadis, S. T. Cadherin-11 binds to PDGFRβ and enhances cell proliferation and tissue regeneration via the PDGFR-AKT signaling axis. Faseb j. 34, 3792–3804 (2020).
Hu, Z. et al. Emerging role of ferroptosis in acute kidney injury. Oxid. Med. Cell. Longev. 2019, 8010614 (2019).
Chen, X., Yu, C., Kang, R. & Tang, D. Iron Metabolism in Ferroptosis. Front. Cell. Dev. Biol. 8, 590226 (2020).
Dixon, S. J. et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 149, 1060–1072 (2012).
Szklarczyk, D. et al. The STRING database in 2023: protein-protein association networks and functional enrichment analyses for any sequenced genome of interest. Nucleic Acids Res 51, D638–d646 (2023).
Jain, C. V. et al. Trophoblast survival signaling during human placentation requires HSP70 activation of MMP2-mediated HBEGF shedding. Cell Death Differ. 24, 1772–1783 (2017).
Dao, D. T., Anez-Bustillos, L., Adam, R. M., Puder, M. & Bielenberg, D. R. Heparin-binding epidermal growth factor–like growth factor as a critical mediator of tissue repair and regeneration. Am. J. Pathol. 188, 2446–2456 (2018).
Yong, V. W. Metalloproteinases: mediators of pathology and regeneration in the CNS. Nat. Rev. Neurosci. 6, 931–944 (2005).
Almalki, S. G. & Agrawal, D. K. Effects of matrix metalloproteinases on the fate of mesenchymal stem cells. Stem Cell Res. Ther. 7, 129 (2016).
Kasper, G. et al. Mesenchymal stem cells regulate angiogenesis according to their mechanical environment. Stem Cells 25, 903–910 (2007).
Mott, J. D. & Werb, Z. Regulation of matrix biology by matrix metalloproteinases. Curr. Opin. Cell Biol. 16, 558–564 (2004).
Burns, J. C. et al. Reinforcement of cell junctions correlates with the absence of hair cell regeneration in mammals and its occurrence in birds. J. Comp. Neurol. 511, 396–414 (2008).
Cao, J. et al. The single-cell transcriptional landscape of mammalian organogenesis. Nature 566, 496–502 (2019).
He, Y. et al. Effect of JNK inhibitor SP600125 on hair cell regeneration in zebrafish (Danio rerio) larvae. Oncotarget 7, 51640–51650 (2016).
Bhattacharyya, S., Fang, F., Tourtellotte, W. & Varga, J. Egr-1: new conductor for the tissue repair orchestra directs harmony (regeneration) or cacophony (fibrosis). J. Pathol. 229, 286–297 (2013).
Silva, N. J. et al. Inflammation and matrix metalloproteinase 9 (Mmp-9) regulate photoreceptor regeneration in adult zebrafish. Glia 68, 1445–1465 (2020).
Matsunaga, M. et al. Stepwise fate conversion of supporting cells to sensory hair cells in the chick auditory epithelium. iScience 26, 106046 (2023).
Doster, S. K., Lozano, A. M., Aguayo, A. J. & Willard, M. B. Expression of the growth-associated protein GAP-43 in adult rat retinal ganglion cells following axon injury. Neuron 6, 635–647 (1991).
Yin, C., Guo, Z.-D., He, Z.-Z., Wang, Z.-Y. & Sun, X.-C. Apolipoprotein E affects in vitro axonal growth and regeneration via the MAPK signaling pathway. Cell Transplant. 28, 691–703 (2019).
Iyer, A. A. et al. Cellular reprogramming with ATOH1, GFI1, and POU4F3 implicate epigenetic changes and cell-cell signaling as obstacles to hair cell regeneration in mature mammals. eLife 11, e79712 (2022).
Mulvaney, J. & Dabdoub, A. Atoh1, an essential transcription factor in neurogenesis and intestinal and inner ear development: function, regulation, and context dependency. J. Assoc. Res. Otolaryngology: JARO 13, 281–293 (2012).
Xiao, F., Mirwald, A., Papaioannou, M., Baniahmad, A. & Klug, J. r. Secretoglobin 2A1 is under selective androgen control mediated by a peculiar binding site for Sp family transcription factors. Mol. Endocrinol. 19, 2964–2978 (2005).
Missner, A. A., Johns, J. D., Gu, S. & Hoa, M. Repurposable drugs that interact with steroid responsive gene targets for inner ear disease. Biomolecules 12, 1641 (2022).
Wilkerson, B. A. et al. Novel cell types and developmental lineages revealed by single-cell RNA-seq analysis of the mouse crista ampullaris. Elife 10, e60108 (2021).
Long, Q., Quint, E., Lin, S. & Ekker, M. The zebrafish scyba gene encodes a novel CXC-type chemokine with distinctive expression patterns in the vestibulo-acoustic system during embryogenesis. Mech. Dev. 97, 183–186 (2000).
Janesick, A. et al. Cell-type identity of the avian cochlea. Cell Rep. 34, 108900 (2021).
Ku, Y.-C. et al. The transcriptome of utricle hair cell regeneration in the avian inner ear. J. Neurosci. 34, 3523–3535 (2014).
González-Garrido, A. et al. The differentiation status of hair cells that regenerate naturally in the vestibular inner ear of the adult mouse. J. Neurosci. 37, 3127–3120 (2021).
Ohlemiller, K. K., Jones, S. M. & Johnson, K. R. Application of mouse models to research in hearing and balance. J. Assoc. Res Otolaryngol. 17, 493–523 (2016).
Chandra Mohan, S. L. N. Case of Waardenburg Shah syndrome in a family with review of literature. J. Otol. 13, 105–110 (2018).
Bondurand, N., Dufour, S. & Pingault, V. News from the endothelin-3/EDNRB signaling pathway: Role during enteric nervous system development and involvement in neural crest-associated disorders. Developmental Biol. 444, S156–S169 (2018).
Kruger, G. M. et al. Temporally distinct requirements for endothelin receptor B in the generation and migration of gut neural crest stem cells. Neuron 40, 917–929 (2003).
DeLuca, D. S. et al. RNA-SeQC: RNA-seq metrics for quality control and process optimization. Bioinformatics 28, 1530–1532 (2012).
Martin, M. et al. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet. J., [S.l.]. 17, 10–12 (2011).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Li, B. & Dewey, C. N. RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinforma. 12, 323 (2011).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
McGinnis, C. S., Murrow, L. M. & Gartner, Z. J. Doubletfinder: doublet detection in single-cell RNA sequencing data using artificial nearest neighbors. Cell Syst. 8, 329–337 e324 (2019).
Lun, A. T., Bach, K. & Marioni, J. C. Pooling across cells to normalize single-cell RNA sequencing data with many zero counts. Genome Biol. 17, 75 (2016).
Zappia, L. & Oshlack, A. Clustering trees: a visualization for evaluating clusterings at multiple resolutions. Gigascience 7, giy083 (2018).
Hu, H. et al. AnimalTFDB 3.0: a comprehensive resource for annotation and prediction of animal transcription factors. Nucleic Acids Res. 47, D33–D38 (2018).
Freshour, S. L. et al. Integration of the drug–gene interaction database (DGIdb 4.0) with open crowdsource efforts. Nucleic Acids Res. 49, D1144–D1151 (2020).
McInnes, L., Healy, J., Saul, N. & Großberger, L. UMAP: uniform manifold approximation and projection. J. Open Source Softw. 3, 861 (2018).
Street, K. et al. Slingshot: cell lineage and pseudotime inference for single-cell transcriptomics. BMC Genomics 19, 477 (2018).
Jurado, M. R. et al. Improved integration of single-cell transcriptome data demonstrates common and unique signatures of heart failure in mice and humans. Gigascience 13, giae011 (2024).
Bray, N. L., Pimentel, H., Melsted, P. & Pachter, L. Near-optimal probabilistic RNA-seq quantification. Nat. Biotechnol. 34, 525–527 (2016).
Townes, F. W. & Irizarry, R. A. Quantile normalization of single-cell RNA-seq read counts without unique molecular identifiers. Genome Biol. 21, 160 (2020).
Choudhary, S. & Satija, R. Comparison and evaluation of statistical error models for scRNA-seq. Genome Biol. 23, 27 (2022).
Acknowledgements
We are grateful to the sample donors. We also thank the Princess Margaret Genomics Centre, the Bioinformatics and High Performance Computing Core, and the Ottawa Bioinformatics Core Facility, for providing high-throughput sequencing assistance and bioinformatics services; Compute Ontario (computeontario.ca) and the Digital Research Alliance of Canada (alliancecan.ca) for providing hardware, storage and computing resources, and the imaging facilities at Sunnybrook Research Institute and SickKids. We extend our thanks to Eng. G. Luca for illustrations in Fig. 1, gEAR’s team–especially D. Lesperance–for their support, and Drs. O. Bermingham-McDonogh, L.V. Goodrich, and M. Warchol for their comments on an earlier version of the manuscript. This work was supported by grant funding from the Canadian Institutes of Health Research – CIHR PJT-183579 (EL and AD); Hearing Health Foundation’s Hearing Restoration Project (ML and AD); the University of Toronto, Department of Otolaryngology, Harry Barberian Research Fund (EL); Japan Society for the Promotion of Science – JSPS Postdoctoral Fellowship (RY); Canadian Graduate Scholarship – Masters Program from CIHR, Ontario Graduate Scholarship - Doctoral Award, and Raymond H.W. Ng Graduate Scholarship (WL), and support from the Michael and Sonja Koerner Charitable Foundation (AD).
Author information
Authors and Affiliations
Contributions
Conceptualization and experimental design: E.L., M.L., and A.D.; sample collections: V.L. and J.C.; generation of bulk and scRNA-seq datasets: M.L. and E.L., respectively; experiments: E.L., W.L., D.B., and A.D.; computational analysis: N.I. and R.Y.; data analysis: E.L., N.I., R.Y., W.L., and A.D.; writing – original draft: E.L.; writing – review & editing: E.L., N.I., R.Y., W.L., and A.D.; funding acquisition: E.L., R.Y., W.L., M.L., and A.D.; supervision: A.D. All authors read and accepted the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Luca, E., Ibeh, N., Yamamoto, R. et al. Revealing heterogeneity and damage response in the adult human utricle. Nat Commun 17, 9 (2026). https://doi.org/10.1038/s41467-025-66358-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-66358-8