Abstract
Machine learning (ML) plays a pivotal role in extending the reach of quantum chemistry methods for simulating both molecules and materials. However, leveraging ML to overcome the limitations of human-designed density functional approximations (DFAs), the primary workhorse for quantum simulations, remains a major challenge due to their severely limited transferability to unseen chemical systems. Here, we demonstrate how transferability is achieved using real-space ML, where energies are learned point by point in space through energy densities. Central to our real-space learning strategy is the derivation and implementation of correlation energy densities from regularized perturbation theory. This enables two key advances toward constructing highly transferable DFAs, grounded in the Møller-Plesset adiabatic connection framework, for correlation energies defined with respect to the Hartree-Fock reference. First, we introduce the Local Energy Loss, whose data efficiency (expanding each system’s single energy into thousands of data points) dramatically enhances transferability when combined with a physically informed ML model. Second, we formulate a real-space, machine-learned, and regularized extension of Spin-Component-Scaled second-order Møller-Plesset perturbation theory, yielding transferable DFAs that effectively mitigate the self-interaction errors common to traditional DFAs.
Similar content being viewed by others
Introduction
Machine learning (ML) is driving a paradigm shift across scientific disciplines, including quantum chemistry (QC), where it reshapes the landscape of used methods1,2,3,4,5,6. The recent surge in ML has further increased the importance of density functional approximations (DFAs), already a cornerstone of quantum simulations in materials science and chemistry (see the recent review by von Lilienfeld and co-workers7). On one hand, DFAs generate vast amounts of data to train ML models7,8,9,10,11,12,13,14,15, greatly extending their reach in time and length scales16,17,18,19,20,21,22,23,24,25. On the other hand, ML techniques provide improved DFAs26,27,28,29,30,31,32,33,34,35,36,37,38 (DFAs), with the DM21 functional by DeepMind39 being a prominent example.
A remaining critical problem with ML in QC is their limited transferability: the ability to generalize to unseen data6,40,41,42. These limitations hinder the applicability of ML models in QC, making users cautious and often leading them to stick with well-established old-school methods43 over new-school ML counterparts. This situation has lead to a no man’s land between old-school and new-school DFAs44: the promised revolution of ML-based DFAs is hampered by the far broader applicability of old-school DFAs44, such as B3LYP45,46,47,48 or PBE49. For example, while DM21’s training on systems with fractional charges and spin addresses some limitations of old-school functionals39, its catastrophic transferability failures have recently become evident44. Namely, it has been shown that DM21 does not converge for certain transition metal (TM) atoms, a convergence task easily handled by reputable old-school functionals44. Thus, it is no wonder that organic chemists still prefer old-school DFAs over DM21 or other new-school models for TM-catalyzed reaction mechanisms50, despite major TM shortcomings of the former51,52.
To move from this no man’s land and leverage the power of ML for DFAs design, we need to solve the underlying transferability problem. Helping strategies are the use of physical constraints53,54,55,56 (a mix of old- and new-school methods), more diverse data in the training set40, or the engineering of new features57. Yet, an angle in ML of DFAs that requires more attention is the training data efficiency, particularly since feeding more data to ML models can become a never-ending game due to the data hunger in ML models and the vastness of chemical space2,3,5. The purpose of this work is to critically examine data efficiency in training DFAs and maximize it in order to embed transferability in ML of DFAs.
The primary goal when training DFAs is to learn how a given electronic density translates into energy. However, ML practices in QC currently treat energies as not very informative, following a 1 system = 1 energy data point approach (see refs. 30,31,32,34,37,39,54,58,59). For example, when learning force fields, forces (energy gradients) are much more informative than energies19,60. Similarly, recent works have shown that in current ML DFA practices, electronic densities are also far more informative than energies31,34,54,58,59 (each grid point is a density datapoint). While using electronic densities effectively enhances the transferability of ML-based DFAs, applying a point by point learning strategy to energies (typically the primary target of simulations) offers a distinct direction for improving ML-based DFAs. Identifying transferability as the key issue in ML DFAs, here we establish a framework for making energy training more data-efficient and address the challenges that must be overcome to leverage this data efficiency to embed transferability into ML DFAs.
In view of our objective to enhance data efficiency for energies in ML-based DFAs, we introduce real-space energy learning and apply it to machine-learn a DFA for correlation energy (a crucial target for DFAs). This approach expands each system’s single energy data point into thousands of energy data points for the training. During the learning of our DFAs, each point in space contributes to the loss function, which we call Local Energy Loss (LES). Crucially, LES penalizes error cancellations between energy contributions from different regions in space, thereby enhancing transferability. With LES, every system in the training set becomes an entire energy dataset, and we show here that a careful, physically-informed application of LES is essential to fully realize its data-efficiency potential and to embed transferability in ML of DFAs.
Applying LES for ML of correlation DFAs requires two crucial ingredients: (i) a well-defined correlation energy contributions at each point in space ec(r) (i.e., correlation energy density per particle, see Eq. (1) below) and (ii) a robust strategy for generating accurate ec(r) training data. To meet these requirements simultaneously, we develop here LES-based ML DFAs for correlation energies defined with respect to the Hartree-Fock61,62 (HF) reference. While historically DFA development has been tied to Kohn-Sham density functional theory63 (KS DFT), recent theoretical advances based on the Møller-Plesset adiabatic connection (MPAC) formally ground the developments of correlation DFAs evaluated on HF densities (see ref. 64). Constructing DFAs on fixed (HF) densities within the MPAC framework enables us to isolate and focus specifically on real-space energy learning strategies, complementing (see Discussion) existing real-space density learning approaches31,34,54,58,59. As this work employs local energy quantities, we note that these quantities provide valuable chemical insights when well-defined (see, e.g., refs. 65,66,67). Since ec(r) is not uniquely defined, here we adopt a physically transparent definition arising from the MPAC framework64 and demonstrate its advantages for LES.
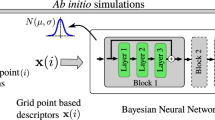
An overview of the key methods presented in this paper is given in Fig. 1. To demonstrate the power of the LES-based approach, and more generally real-space ML for DFAs, we first construct a robust proxy reference for ec(r) that preserves the original MPAC-based definition and efficiently implement it to enable direct training of our ML models for ec(r). This proxy reference ec(r) is built by combining second-order perturbation theory64 (PT2) [magenta circle in Fig. 1(a)] and the specific PT2 regularization68,69 [blue square in Fig. 1(a)], which is crucial for making our proxy reference sufficiently accurate. The effect of regularization on the PT2’s ec(r) for the helium dimer (going from the magenta circle to the blue square in Fig. 1(a) is displayed in Fig. 1(d, top). We implement a numerical data generator of this proxy reference ec(r) by leveraging modern Python libraries (e.g., JAX70). Input features tailored to the problem, such as Grimme’s real-space electronic correlation measures71,72, enable us to construct a robust neural network (NN) for ec(r) [Fig. 1(b)]. We then contrast the transferability of our LES strategy with the common global energy loss (GES) that adopts standard 1 system = 1 energy data point approach [cyan and maroon diamonds in Fig. 1(a)]. Keeping other factors in the DFA training the same allows us to isolate how the transferability is affected when we move from GES to LES [Fig. 1(c) shows how well the two models trained on small atoms transfer to the dissociation curve of BH within spin-restricted calculations]. Similar transferability tests reveal subtle yet crucial requirements for successful and robust LES applications: it should be defined in terms of ec(r) rather than alternative quantities (e.g., its density-weighted counterpart), and coupled with a physically-informed ML model trained on a physically-motivated ec(r) definition. We also derive the contributions at each point in space for different spin channel pairs of our proxy ec(r) [purple and orange circles or squares in Fig. 1(a)] and we show these spin-resolved interaction components for the helium dimer in Fig. 1(d, bottom). We then use spin-resolved energy densities per particle to build a real-space, machine-learned and regularized extension of spin-component-scaled73,74 (SCS) PT2 correlation energy (the performance of this ML strategy for the formic acid dimer is shown in Fig. 1(e, top)). The resulting model [green diamond in Fig. 1(a)] allows the construction of DFAs bridging the gap between our proxy reference correlation energies (regularized PT2) and their exact counterpart.
a Overview of methods and their connections. The correlation energy density per particle ec of second-order Møller-Plesset perturbation theory83 (MP2) is spin-resolved into opposite-spin (os) and same-spin (ss) parts, and κ-regularized (κMP2). Real-space machine-learning strategies presented here are energy densities from κMP2 (ML2) and extensions of spin-component-scaled κMP2 (MLS2). b Neural network illustration123 with real-space features defined in Supplementary Section S6 in the SI (reduced density gradient s, reduced density Laplacian q, regularized energy kinetic variable α and temperature-dependent fractional occupation number weighted densities71,72 (FOD) f1 and f2), and LES/GES strategies to produce machine-learned weights wc from Eq. (14). (c, top) Correlation energy density per particle of the Mg atom for GES-based and LES-based ML2, and κMP2 as a proxy reference. (c, bottom) Corresponding dissociation curve of the BH diatomic system. (d, top) Correlation energy density per particle of the helium dimer at an interatomic distance of 5.6 a.u., plotted along the principal axis of the system for different values of the κ-regularization. (d, bottom) Spin-resolved interaction correlation energy density of the helium dimer (dimer energy density minus that of atoms). (e, top) Errors in interaction energies (kcal mol−1) along the dissociation of the formic acid dimer (geometries and reference values are taken from the S22 × 5 database104) for Hartree–Fock61,62 (HF), MP2, κMP2 and MLS2. (e, bottom) Relative absolute correlation energy errors in log-scale of available systems from the W4-11 test dataset102 (see Supplementary Section S12 in the SI for a detailed list of test data points) for MP2, κMP2 and MLS2. The mean absolute relative errors (MArEs) are shown with dashed lines.
Results
Local and global energy loss (LES vs GES)
Distinguishing between LES and GES is a crucial point of this work when training ML DFAs. To define LES and GES generally, consider the reference (i.e. exact) energy
with a corresponding reference energy density57 per particle, eref(r), and electronic density ρ(r). An ML energy quantity defined in the same way is indicated using the ML superscript. Then, GES reads
In contrast, with LES, we consider the pointwise difference of the reference and ML energy densities per particle weighted by the density:
Minimizing LES, strictly defined in terms of the energy density per particle, instead of GES turns each point in space into an energy data point, and as we shall see, moving from GES to LES dramatically enhances the transferability of the underlying ML DFA even with very small training sets.
Improving and deriving PT2 correlation energy densities per particle
To demonstrate the difference between LES and GES, we will target correlation energy approximations. While correlation DFAs are typically developed within the KS DFT framework, recent work has shown that the Møller–Plesset adiabatic connection (MPAC) formally grounds the construction of DFAs mapping Hartree–Fock (HF) densities directly to correlation energies (defined here w.r.t. the HF energies). As detailed in ref. 64, this ground distinguishes the MPAC-based correlation DFAs from density-corrected DFT, where HF densities are introduced heuristically to improve DFAs developed within KS DFT75,76,77. Leveraging this MPAC formalism and its recently introduced correlation energy densities64, we construct DFAs as NN-based functionals of HF densities, enabling practical use of HF orbitals to compute all energy terms and input features for our NNs.
To briefly introduce our energy density per particle targets for LES, we define correlation energy as,
where Eref is the exact ground-state energy, \(\hat{H}\) is the corresponding exact Hamiltonian, and Φ is the HF wavefunction (a single Slater determinant minimizer of \(\hat{H}\), that yields ρHF(r) = ρ(r)). In Eq. (4), we distinguish the correlation energy density, \({\bar{e}}_{{{\rm{c}}}}({{\bf{r}}})={e}_{{{\rm{c}}}}({{\bf{r}}})\rho ({{\bf{r}}})\), from the correlation energy per particle, ec(r), as this distinction is crucial for our subsequent LES analysis. As ec(r) is not uniquely defined, we adopt here a specific definition (i.e., gauge) for ec(r), derived in ref. 64 from the MPAC theory. This gauge is designed as an MPAC-based analogue64 of the conventional DFT gauge for correlation energies, i.e., the electrostatic potential of the correlation hole78,79,80,81, known for transparent physical interpretation and advantages in DFA construction81,82. Since computing the exact ec(r) is costly64, we approximate it using its weakly interacting limit determined by second-order Møller-Plesset perturbation theory83 (MP2), preserving the original MPAC gauge,
where \({P}_{2}^{{\mbox{MP2}}}({{\bf{r}}},{{{\bf{r}}}}^{{\prime} })\) is the first-order MPAC correction to the pair density that yields the MP2 correlation energy (for its formal definition and derivation of Eq. (5), see Supplementary Section S1 in the SI). Crucially, \({P}_{2}^{{\mbox{MP2}}}({{\bf{r}}},{{{\bf{r}}}}^{{\prime} })\) isolates the correlation contribution to the pair density analogous to the DFT correlation hole64.
Expressing Eq. (5) in terms of HF orbitals yields,
where i, j are occupied, and a, b are virtual KS orbital (ϕ(r)) indices. Tijab are the partial MP2 doubles amplitude,
where ε are orbital energies, and Vijab(r) is the orbital potential,
As \({e}_{{\mbox{c}}}^{{\mbox{MP2}}}\,({{\bf{r}}})\) by Eq. (4) integrates to the MP2 correlation energy, it can be easily argued that it is not a sufficiently good proxy reference for LES-based applications given general MP2 limitations68,69,84, particularly for small orbital-gap systems85 (see the MP2 dissociation curve relevant to this work in Supplementary Fig. S1 in the SI). To address this, we apply Head-Gordon’s κ-regulizaration68,69 (κ ≥ 0) to \({e}_{{\mbox{c}}}^{{\mbox{MP2}}}\,({{\bf{r}}})\) of Eq. (5) by regularizing its partial MP2 doubles amplitudes,
The regularized \({e}_{{\mbox{c}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\) is obtained from Eq. (6) by the replacement \({T}_{ijab}\to {T}_{ijab}^{\kappa }\), which also regularizes the underlying pair density (see Supplementary Section S1 in the SI):
When κ = 0, \({e}_{{\mbox{c}}}^{{\kappa} {{\mbox{MP2}}}}({{\bf{r}}})=0\), and when κ → ∞, \({e}_{{\mbox{c}}}^{{\kappa} {{\mbox{MP2}}}}({{\bf{r}}})={e}_{{\mbox{c}}}^{{\mbox{MP2}}}\,({{\bf{r}}})\). In Fig. 1(d, top), we observe how \({e}_{{\mbox{c}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\) evolves to \({e}_{{\mbox{c}}}^{{\mbox{MP2}}}\,({{\bf{r}}})\) for the helium dimer as κ increases. Using the regularized \({e}_{{\mbox{c}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\) [blue square in Fig. 1(a)] instead of \({e}_{{\mbox{c}}}^{{\mbox{MP2}}}\,({{\bf{r}}})\) [magenta circle in Fig. 1(a)] as the proxy reference for LES-based learning is crucial, since the former significantly improves dissociation curves of diatomic systems, which are central to our LES vs. GES comparison. In these curves, κ-regularization removes the MP2 divergence at large bond lengths arising from small orbital energy gaps. Here we set κ = 2.0 to generate \({e}_{{\mbox{c}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\) proxy reference, as it better balances improvements over MP2 for stretched bonds while maintaining accuracy near equilibrium, compared to the originally proposed κ = 1.468 (see Supplementary Fig. S1 in the SI for a clarifying example of N2 dissociation).
After regularization, we perform the spin-resolution of \({e}_{{\mbox{c}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\) into same-spin (ss) and opposite-spin (os) components: \({e}_{{\mbox{c}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})={e}_{{\mbox{c,ss}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})+{e}_{{\mbox{c,os}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\), enabling a spin-resolved real-space analysis of electron correlation [purple and orange in Fig. 1(a)]. The more compact of the two, \({e}_{{\mbox{c,os}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\), we derive from Eq. (6) by considering only os electronic pairs:
Later, we will use ML for real-space scaling of \({e}_{{\mbox{c,os}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\) and \({e}_{{\mbox{c,ss}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\) separately to build a correction from κMP2 to the true Ec.
Central to our data generator for real-space ML of ec(r) are the κ-regularization and spin resolution of MP2 correlation energy densities, both efficiently implemented by combining density fitting86,87 with the power of JAX70 and modern tensor libraries88 for optimizing tensor contractions required to obtain \({e}_{{\mbox{c}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\). Further implementation details are provided in Methods and Supplementary Section S3 of the SI.
Regularized PT2 correlation energy densities for interaction energies
Now we move to a real-space analysis of the interaction correlation energy density, \(\Delta {\bar{e}}_{{{\rm{c}}}}({{\bf{r}}})\), defined as the total system’s \({\bar{e}}_{{{\rm{c}}}}({{\bf{r}}})\) minus the sum of the \({\bar{e}}_{{{\rm{c}}}}({{\bf{r}}})\) of the isolated subsystems (e.g., dimer minus monomers). A subtle point here is that while for ML purposes ec(r) is vastly superior to \({\bar{e}}_{{{\rm{c}}}}({{\bf{r}}})\) (see below), spatial visualization of interactions requires \(\Delta {\bar{e}}_{{{\rm{c}}}}({{\bf{r}}})\) instead of Δec(r), as the former directly integrates to ΔEc, while the latter lacks a clear density factor to do so.
From Eq. (10), \(\Delta {\bar{e}}_{{\mbox{c}}}^{{\mbox{MP2}}}\,({{\bf{r}}})\) is given by the electrostatic potential of \(\Delta {P}_{2}^{{\kappa} {{\mbox{MP2}}}}({{\bf{r}}},{{{\bf{r}}}}^{{\prime} })\),
where \(\Delta {P}_{2}^{{\kappa} {{\mbox{MP2}}}}({{\bf{r}}},{{{\bf{r}}}}^{{\prime} })\) is the interaction component of \({P}_{2}^{{\kappa} {{\mbox{MP2}}}}({{\bf{r}}},{{{\bf{r}}}}^{{\prime} })\). As \({P}_{2}^{{\kappa} {{\mbox{MP2}}}}({{\bf{r}}},{{{\bf{r}}}}^{{\prime} })\) isolates the correlation part of the underlying pair density, \(\Delta {P}_{2}^{{\kappa} {{\mbox{MP2}}}}({{\bf{r}}},{{{\bf{r}}}}^{{\prime} })\) further isolates how this quantity is deformed by the interaction between fragments, making this quantity crucial for describing the physics of weak interactions, particularly dispersion effects (see, e.g., refs. 89,90,91).
While Eq. (12) represents just one possible gauge for \(\Delta {\bar{e}}_{{\mbox{c}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\), we emphasize that it encodes the physics of weak interactions through its direct link to \(\Delta {P}_{2}^{{\kappa} {{\mbox{MP2}}}}({{\bf{r}}},{{{\bf{r}}}}^{{\prime} })\), a fundamental quantity for describing the physics of weak interactions, such as dispersion effects89,90,91). To illustrate this for dispersion, we first consider a simple example: the helium dimer, for which spin-resolved interaction correlation energy densities \(\Delta {\bar{e}}_{{\mbox{c}}}^{{\mbox{MP2}}}\,({{\bf{r}}})\) are shown in Fig. 1 (d, bottom) along the internuclear axis. We now show their sum in Fig. 2(a), highlighting distinct binding (negative) and non-binding (positive) contributions for stretched He2. To illustrate the interaction physics encoded in \(\Delta {\bar{e}}_{{\mbox{c}}}^{{\mbox{MP2}}}\,({{\bf{r}}})\), we fix the reference electron at the position labeled by z0 in He2, and in the inset of Fig. 2(a) we show \(\Delta {P}_{2}^{{\mbox{MP2}}}\,({z}_{0},{z}^{{\prime} })\) along the internuclear axis as a function of the second electron position \({z}^{{\prime} }\). The plot reveals spatially nonlocal polarization in \(\Delta {P}_{2}^{{\mbox{MP2}}}\,\), characteristic of dispersion: when \({z}^{{\prime} }\) is near the second helium nucleus, \(\Delta {P}_{2}^{{\mbox{MP2}}}\,({z}_{0},{z}^{{\prime} })\) exhibits a negative accumulation closer to z0 and a corresponding positive region further away. As the negative part of \(\Delta {P}_{2}^{{\mbox{MP2}}}\,({z}_{0},{z}^{{\prime} })\) lies closer to z0 than the positive part, the resulting electrostatic potential, i.e., \(\Delta {\bar{e}}_{{{\rm{c}}}}({z}_{0})\), is negative. If the polarization pattern is reversed, i.e., the positive part of \(\Delta {P}_{2}^{{\mbox{MP2}}} \, ({z}_{0},{z}^{{\prime} })\) lies closer to z0 than the negative part, as when z0 is in the outer region, then \(\Delta {\bar{e}}_{{{\rm{c}}}}({z}_{0})\) becomes positive (see Supplementary Fig. S3 for additional plots in the SI). Still, the negative (i.e., binding) regions dominate, and MP2 correlation yields net binding in He2 (Supplementary Table S1 in the SI). In this way, Eq. (12) condenses information from \(\Delta {P}_{2}({{\bf{r}}},{{{\bf{r}}}}^{{\prime} })\), a key two-body quantity encoding interaction physics, into \(\Delta {\bar{e}}_{{{\rm{c}}}}({{\bf{r}}})\), a one-body correlation quantity for interaction energies. Thus, even though it is not unique, the specific gauge defined by Eq. (12) yields a physically interpretable local correlation energy quantity for describing weak interactions between fragments.
a Second-order Møller–Plesset perturbation theory (MP2) plot along the inter-nuclear axis of the helium dimer (position of the nuclei are denoted by the spheres) at an interatomic distance of 5.6 a.u.. Inset shows the corresponding interaction pair density \(\Delta {P}_{2}^{\,{\mbox{MP2}}\,}({{\bf{r}}},{{{\bf{r}}}}^{{\prime} })\) at r = z0 on the same axis. b MP2 volume slice plots for the benzene-CH4 complex85 along planes perpendicular and parallel to the benzene ring. Binding and non-binding regions are highlighted by negative (red) and positive (blue) \(\Delta {\bar{e}}_{{{\rm{c}}}}({{\bf{r}}})\) volumes. c Isosurface visualization of MP2 (rough mesh) and its κ-regularization (κMP2) with κ = 1.4 (solid surface) for the same complex at binding isovalue ( − 2.5e−5 (a.u.)). d Same as (c), but for the ss part.
Having established the direct connection between \(\Delta {\bar{e}}_{{\mbox{c}}}^{{\mbox{MP2}}}\,({{\bf{r}}})\) and dispersion physics, we now analyze \(\Delta {\bar{e}}_{{\mbox{c}}}^{{\mbox{MP2}}}\,({{\bf{r}}})\) for the benzene-methane complex in the remaining panels of Fig. 2. In Fig. 2(b), volume slices along planes perpendicular and parallel to the benzene ring distinguish regions between the fragments (typically binding regions) from those outside (typically non-binding). MP2 overbinds this complex, whereas κMP2 reduces this overbinding (Supplementary Table S2 in the SI). This reduction is visually reflected in Fig. 2(c), which compares MP2 and κMP2 \(\Delta {\bar{e}}_{{{\rm{c}}}}({{\bf{r}}})\) isosurfaces for the binding region, with the κMP2 isosurface confined within the MP2 counterpart. The spatial confinement is even more pronounced when focusing on just the ss component of \(\Delta {\bar{e}}_{{\mbox{c}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\) [the electrostatic potential of the ss component of the underlying \(\Delta {P}_{2}^{{\kappa} {{\mbox{MP2}}}}({{\bf{r}}},{{{\bf{r}}}}^{{\prime} })\)], as shown in Fig. 2(d).
ML2 model via machine-learning of regularized \({e}_{{\mbox{c}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\)
We will now present the ML2 model, a machine-learned correlation energy density based on the regularized MP2 proxy reference, which we obtain by mapping a set of pointwise features using neural networks (NNs) [going from the blue square to the cyan or maroon diamonds in Fig. 1(a)]. This mapping is illustrated in Fig. 1(b). In addition to the established features used in ML of DFAs31,56—the reduced density gradient s(r), the reduced density Laplacian q(r), and the regularized kinetic energy variable α(r) from the r2SCAN DFA92—we introduce Grimme’s electronic temperature-dependent fractional occupation number weighted density71,72 (FOD) as the crucial feature (a detailed list of features is given in Supplementary Section S6 in the SI). FOD differentiates strongly correlated and weakly correlated regions in molecules. While both \({e}_{{\mbox{c}}}^{{\kappa} {{\mbox{MP2}}}}({{\bf{r}}})\) and FOD provide insights into electronic correlation through the interaction between occupied and unoccupied orbitals, FOD is computationally much cheaper, making it an excellent feature for ML of the correlation energy density.
Even though ρ(r)-based features (e.g., the Wigner–Seitz radius) might seem like a natural choice, we intentionally omit them from the ML2 features. This is because we found that \({e}_{{\mbox{c}}}^{{\mbox{MP2}}}\,({{\bf{r}}})\) and its κ-counterpart are scaling invariant for a uniformly scaled93 density ρλ(r) = λ3ρ(λr),
A detailed derivation is given in Supplementary Section S7 in the SI. Following this scaling invariance, we construct \({e}_{{\mbox{c}}}^{{\mbox{ML2}}}\,({{\bf{r}}})\), the ML2 analog of \({e}_{{\mbox{c}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\), as
where ex(r) is the exchange energy (we use the same Python code to implement both \({e}_{{\mbox{c}}}^{{\mbox{MP2}}}\,({{\bf{r}}})\) and ex(r) on the same footing), and wc(r) are the ML2 weights obtained from the NN (see Fig. 1(b) for the illustration and Methods for NN architecture details). With the use of HF-based ingredients, the computational cost of ML2 is comparable to that of an HF calculation and cannot be lower. As we shall see later, embedding the physics into LES-based ML2 through Eq. (14) is crucial for the robustness of the model.
Using the same features, functional form of Eq. (14), and NN architecture for mapping the features at a given r to wc(r), we can now isolate the difference between using LES and GES for NN training of a DFA [cyan vs. maroon diamonds in Fig. 1(a)]. For simplicity and to create a challenging transferability test, we train our ML2-based NN only on eight small closed-shell atoms/ions (H−, He, Be, Ne, Mg, Ar, Ca, and Kr). The total loss is calculated as the mean over these eight datapoints, as detailed in Supplementary Section S8 in the SI. We validate our training on comparable small closed-shell atoms/ions (see Supplementary Fig. S4) in the SI.
In Fig. 1(c), we explore GES-based vs. LES-based ML2 results (cyan vs. maroon diamonds). Both energy densities are plotted against the \({e}_{{{\rm{c}}}}^{{{\rm{\kappa MP2}}}}({{\bf{r}}})\) reference in Fig. 1(c, top) for the Mg atom as one of the training datapoints. We can see that \({e}_{{\mbox{c}}}^{{\mbox{ML2}}}\,({{\bf{r}}})\) based on LES closely follows the \({e}_{{{\rm{c}}}}^{{{\rm{\kappa MP2}}}}({{\bf{r}}})\) (proxy) reference, whereas the GES-based \({e}_{{\mbox{c}}}^{{\mbox{ML2}}}\,({{\bf{r}}})\) completely misses the shape of the reference. But, can we, solely based on this observation, conclude that LES is better than GES? We need to be careful here, as these correlation energy densities are not observables even within our physically sound gauge (see previous section). Instead, they are used here to enhance energy training data efficiency in ML of DFAs, expanding from one to thousands of energy datapoints per system. Thus, what ultimately matters for judging the quality of GES vs. LES training are the integrated correlation energies (Eq. (1)) once we go outside of the training set.
For the Mg case in Fig. 1(c, top), all energy densities integrate to nearly the same correlation energy by Eq. (1). However, the correlation energies from our GES-based and LES-based ML2 models differ dramatically when tested for transferability. The first such test is shown in Fig. 1(c, bottom), where we see major differences in accuracy when applied to stretching the BH diatomic system along its dissociation curve. The LES-based model closely follows the \({e}_{{\mbox{c}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\) (proxy) reference result, while the GES-based model is not sufficiently accurate even at equilibrium and breaks down completely as the bond is stretched.
The advantages of LES-based learning process
After seeing in Fig. 1(c, bottom) that LES-based ML2 trained only on atomic systems successfully transfers to the BH diatomic along its dissociation curve, we now investigate this atom to diatomics transferability in more detail. Namely, a closer look at the learning process of the LES-based ML2 is given in Fig. 3, where panel (a) shows the absolute error in \({e}_{{\mbox{c}}}^{{\mbox{ML2}}}\,({{\bf{r}}})\) for the He atom (one of ML2’s training points) at different epochs. On the other hand, panel (b) focuses on the test and shows the absolute error in the ML2 correlation energy along the LiH dissociation curve for the same set of epochs. Overall, Fig. 3(a) shows that the pointwise improvement in \({e}_{{\mbox{c}}}^{{\mbox{ML2}}}\,({{\bf{r}}})\) for the He atom during training translates epoch by epoch into improved correlation energies for the unseen LiH system, as indicated by the decreasing errors with increasing epochs in panel (b). In contrast to that, the GES-based model yields no improvements of the transferability from atoms as the learning process progresses (see Supplementary Fig. S5 in the SI exposing poor transferability to diatomics, especially at large distances even at large epochs). Furthermore, we observe another crucial difference between GES-based and LES-based learning: when LES is used as the loss function, the learning process is much smoother and it has a much faster convergence with respect to learning steps (epochs) compared to GES (see Supplementary Fig. S6 in the SI).
a Absolute (local) error in correlation energy densities per particle for the helium atom. b Absolute global energy error for LiH along its dissociation curve.
Finishing off, by making energies more information-rich through LES in the training, we equip our models with a high level of transferability, ensuring that any information learned from atoms lead to better performance on molecules. LES reveals information completely washed away with GES, showing that combinations of features and corresponding energy densities per particle, even for atoms, are highly relevant for molecules. Thus, LES provides a powerful strategy for ML of transferable DFAs, and in what follows we will explore more subtle details linked to the LES-based training.
Uniqueness and robustness of our LES-based ML2 model
Building on the successful LES-based ML2 transferability from atoms to challenging BH and LiH dissociations, Fig. 4 shows dissociation curves for four additional diatomics, further confirming the LES’s advantage over GES, which can yield unphysical curves. The dissociation curves for closed-shell diatomics are obtained using a spin-restricted formalism to avoid artificial energy lowering from spin-symmetry breaking (see refs. 94,95,96 for discussions on the challenge of describing bond breaking without spin-symmetry breaking). We now present four analyses, each applied to a different curve from Fig. 4, to highlight distinct aspects of LES-based ML2 transferability. Importantly, the conclusions from each analysis are robust, consistently holding when cross-checked against other diatomics, as demonstrated in the SI (Supplementary Figs. S7–S11).
Shown are machine-learned results (ML2) from κ-regularized second-order Møller-Plesset perturbation theory (κMP2) using Global Energy Loss (GES) or Local Energy Loss (LES). a BH curves including ML2 results that employ LES with proxy correlation energy densities coming from different a − parameter dependent gauges (see Eq. (15)). b H2 curves including 'directly learned' model that learns \({\bar{e}}_{{{\rm{c}}}}({{\bf{r}}})\) from κMP2 based on a direct loss (see text). c BeH+ curves including GES- and LES-based ML2 results coming from different random seed initializations. d LiH curves including additional ML2 results coming from neural networks with different total number of neurons (see Supplementary Fig. S11 in the SI for the distribution of neurons per hidden layer).
We first test whether the superior transferability of LES over GES from atoms to diatomics arises purely from LES’s higher data efficiency. Specifically, panels (a) and (b) of Fig. 4 show that this transferability is lost if the LES-based ML2 model is constructed without the physics encoded in Eq. (3) (LES defined via energy densities per particle), Eq. (10) (specific ec(r) gauge), and Eq. (14) (physically constrained ML2 functional form).
To examine the sensitivity of ML2 training to the gauge choice of \({e}_{{\mbox{c}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\) (Eq. (5)), we introduce the following gauge transformation:
with \(q({{\bf{r}}})=[{\nabla }^{2}\rho ({{\bf{r}}})]/[4{(3{\pi }^{2})}^{2/3}\rho {({{\bf{r}}})}^{5/3}]\), and where the real parameter a does not affect the integrated correlation energy (Eq. (4)) for exponentially decaying densities. Instead of our original target, \({e}_{{\mbox{c}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\), we now repeat the LES-based ML2 training using \({e}_{{\mbox{c}}}^{a}({{\bf{r}}})\) as the target at various a values. In Fig. 4(a), we test the transferability of the underlying a-dependent model by using the BH dissociation curve previously shown in Fig. 1(c, bottom). These curves become worse as we move away from a = 0 (original LES-based ML2) and even become unphysical at larger a (the results for other diatomic dissociation curves follow similar trends as shown in Supplementary Fig. S7 in the SI). Observing \({e}_{{\mbox{c}}}^{a}({{\bf{r}}})\) for atoms (train) and BH (test) in Fig. S8 in the SI, we see that at a = 0, the range and shape of \({e}_{{\mbox{c}}}^{a}({{\bf{r}}})\) varies much less between train and test systems compared to larger a values, explaining why the original LES-based ML2 (a = 0) exhibits the best transferability. While Eq. (15) does not cover all possible gauges, our tests show that the transferability from atoms to diatomics achieved by our gauge is highly nontrivial, and that superior performance of LES over GES in our ML2 model is not solely due to higher data efficiency but critically depends on the gauge choice (Eq. (10)) for the \({e}_{{\mbox{c}}}^{{\mbox{ML}}}\,({{\bf{r}}})\) training target (observe how κMP2 is more amenable for ML in Supplementary Fig. S8 in the SI).
To further demonstrate that data efficiency alone is insufficient for ML2’s success, we compare dissociation curves for H2 from various models in Fig. 4(b). The figure highlights the importance of defining LES in terms of energy densities per particle (Eq. (3) and employing the physically constrained LES-based ML2 form (Eq. (14). If, instead of LES, the loss is defined directly via energy densities (not per particle)97,98,99, \({{{\mathcal{L}}}}_{{{\rm{direct}}}} \sim \int\left\vert \right.{\bar{e}}_{{\mbox{c}}}^{{\mbox{ref}}}\,({{\bf{r}}})-{\bar{e}}_{{\mbox{c}}}^{{\mbox{ML}}}\,({{\bf{r}}})\left\vert \right.d{{\bf{r}}}\), then the resulting model ("directly learned”) performs as bad as the GES-based model (see Fig. 4(b)). Additional examples of even more drastic failures of \({{{\mathcal{L}}}}_{{{\rm{direct}}}}\)-based learning, illustrating the subtle yet crucial importance of learning ec(r) rather than \({\bar{e}}_{{{\rm{c}}}}({{\bf{r}}})\), are shown in Supplementary Fig. S9 in the SI. The poor model’s transferability when direct loss is used (learning \({\bar{e}}_{{\mbox{c}}}^{{\mbox{ML}}}\,({{\bf{r}}})\)) in place of LES (learning \({e}_{{\mbox{c}}}^{{\mbox{ML}}}\,({{\bf{r}}})\)) is unsurprising, given that both ec(r) and the ML2 weights, wc(r) = ec(r)/(ex(r)ρ−1/3(r)) (Eq. (14), are far less sensitive to variations in system size than \({\bar{e}}_{{{\rm{c}}}}({{\bf{r}}})={e}_{{{\rm{c}}}}({{\bf{r}}})\rho ({{\bf{r}}})\). This comparison further emphasizes that LES-based ML2’s transferability success does not rely solely on higher data efficiency than GES, but critically depends on the synergy between this efficiency and the physics embedded in Eqs. (3), (10), and (14).
Finally, we demonstrate the robustness of LES-based ML2 with respect to NN training conditions: unlike GES, LES-based ML2 remains stable under variations in random initialization seeds (Fig. 4(c) for BeH+; additional examples in Supplementary Fig. S10 in the SI) and NN architecture, including the number of neurons (Fig. 4(d) for LiH; additional examples in Supplementary Fig. S11 in the SI). We also show in Supplementary Fig. S12 in the SI that the use of mean square-based LES (i.e. LES2 instead of the original absolute differences-based LES of Eq. (3)) has little effect on the ML2 results. While one may argue that training on only eight atomic systems leaves the GES model prone to overfitting and poor generalization, the good performance of LES with the same small number of systems is already its core advantage over GES in the low-data regime. Nevertheless, we also show that as the training set is progressively enlarged, LES remains more robust than GES for diatomic dissociation curves, even when additional diatomic systems are included in the GES training (see Supplementary Section S10 and Supplementary Fig. S13 in the SI). Overall, the robustness and uniqueness of the LES-based ML2 model demonstrated in this section further emphasize the advantages of LES-based ML2.
Spin-resolved and regularized modeling of the correlation energy density
In the previous section we have shown that LES enhances the transferability of ML DFAs. Yet, κMP2 correlation has been the proxy reference in place of its exact counterpart. In this section, we use our regularized PT2-based generator for a NN-based combination of the spin-resolved κMP2 energy densities to bridge the gap between κMP2 and true correlation energies. For this purpose, our ML model for correlation energy densities per particle is defined as
where wos(r) and wss(r) are machine-learned weights at every point in space. We call it MLS2, which represents a real-space, machine-learned and regularized extension of SCS MP273,74. Its construction is represented by the step from orange and purple squares to the green diamond in Fig. 1(a). By leveraging our implementation of spin-resolved \({{\mbox{w}}}_{{{\rm{os}}}}({{\bf{r}}}){e}_{{\mbox{c,os}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\) and \({{\mbox{w}}}_{{{\rm{ss}}}}({{\bf{r}}}){e}_{{\mbox{c,ss}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\), Eq. (16) yields a regularized and real-space extension of SCS MP2, thus opening up avenues for DFAs creation.
To obtain wos(r) and wss(r) in MLS2 using a NN, we employ a similar architecture as for the ML2 model (see Fig. 1(b)) with some crucial differences. First, unlike the MP2 correlation energy, the true correlation energy is generally not scale invariant93. This allows us contrary to ML2 (see Eq. (13) and the preceding paragraph) to incorporate also density-based features, specifically, the Wigner-Seitz radius. Second, a sigmoid activation function is applied to the NN’s output layer (more details on the activation functions between layers are given in Methods), constraining the MLS2 weights between 0 and 1. We scale the resulting NN weights wos(r) and wss(r) by a constant factor of 10 before applying them in Eq. (16). This scaling enables the MLS2 model to be accurate for the cases where the true correlation energy (in absolute terms) is much larger than what κMP2 predicts (see Supplementary Section S11 in the SI for more details). Finally, we use \({e}_{{\mbox{c}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\) and \({e}_{{\mbox{c,os}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\) quantities normalized by ρ−1/3(r)ex(r) as extra MLS2 dimensionless features (see Supplementary Section S6 in the SI for a detailed list of features).
Ideally, with access to a robust data generator for the exact ec(r), we could use LES to train \({e}_{{\mbox{c}}}^{{\mbox{MLS2}}}\,({{\bf{r}}})\). However, due to the severe computational limitations of such a generator80,100 to very small systems and basis sets, we settle with a GES-based training of MLS2 (the resulting loss function is detailed in Supplementary Section S8 in the SI). Using GES here requires more data for training. Thus, we employ the eight atoms/ions training dataset from ML2 combined with 13 small closed-shell molecules, mainly dimers, and correlation energies of H2, N2 and Li2 at five geometries of large interatomic distances. In addition to these total energies, our MLS2 NN is also trained on interaction energies from the RG18 dataset101, including dispersion-bound complexes with noble gases. We elaborate in Supplementary Section S8 of the SI on how we combine the total energy-based GES and the interaction energy-based GES when training MLS2. Furthermore, a detailed MLS2’s list of all training data points is given in Supplementary Section S12 of the SI.
To test MLS2, we go back to Fig. 1(e, bottom), which includes results for 96 total correlation energies from the W4-11 database102 not present in the training set (see Supplementary Section S12 in the SI for a full list and how we obtain the underlying reference total correlation energies). Specifically, Fig. 1(e, bottom) shows the relative absolute correlation energy errors for κMP2, MP2 and MLS2. Note the log-scale in the y-axis and the dashed lines representing mean absolute relative errors (MArEs). Going from MP2 to κMP2 (from magenta circles to blue squares), we can see that the introduction of the κ = 2.0 regularization slightly increases the MP2 errors. On the other hand, our MLS2 model (green diamonds) yields here far more accurate correlation energies than MP2, with MArE reduced from ~ 10% to 1%.
In Fig. 5, we revisit the dissociation curve of BH to test MLS2 as the bond stretches and include additional methods beyond those shown in Fig. 1(c, bottom). Unsurprisingly, MP2 (magenta curve) fails to capture the correct physics of BH bond stretching due to the divergence of its correlation energies when the HOMO-LUMO orbital gap closes (see Eq. (7)). κMP2 (blue curve; Eq. (9)) eliminates this divergence, but its energies are much higher than the exact ones when the bond stretches. In contrast, MLS2 is more accurate than κMP2 not only when the unseen BH bond stretches but also at equilibrium (see also Supplementary Fig. S15 in the SI for training results of N2 and H2 bond stretches). This demonstrates the power of MLS2 to successfully dissociate covalent bonds without breaking spin symmetries, which is, as said, a crucial challenge for quantum chemistry methods94,95,96,103. In MLS2, this is achieved by first employing κMP2 to eliminate the divergence present in MP2, followed by real-space NN-based enhancements of its spin-resolved energy densities (see Eq. (16)).
Finally, revisiting Fig. 1(e, top) with the formic acid dimer interaction energy curve, we test the performance of MLS2 for hydrogen-bonded systems. The formic acid dimer is selected because of its two hydrogen bonds with a strong electrostatic component, making it a system that starkly contrasts the dispersion-bonded RG18 systems on which MLS2 was trained. Overall, MP2 overbinds the formic acid dimer, with κMP2 overbinding even more, while MLS2 outperforms both in predicting the dimer’s interaction energies.
We observe similar improvements of MLS2 over κMP2 for other dissociation curves of noncovalent systems from the S22 × 5 dataset104(see Supplementary Fig. S16 in the SI). These results further confirm MLS2’s transferability, as it successfully extrapolates from dispersion-bound systems (RG18) to distinctly different noncovalent interactions, such as hydrogen bonds.
To go beyond the closed-shell systems considered so far and demonstrate MLS2’s broader applicability, we retrain it on the W4-11 atomization energy dataset102 and test its performance on representative subsets of GMTKN55 (a large main-group database)105. We denote this variant as MLS2@W4. Since the MLS2@W4 training and test sets include open-shell systems, we supplement the existing MLS2 features (Supplementary Section S6 in the SI) with the spin polarization function ζ(r) (Supplementary Eq. (S12) in the SI). In Fig. 6, we summarize MLS2@W4 performance with the MAEs testing its transferability from W4-atomizations102 to unseen energy types (see Supplementary Section S13 in the SI for further details): reaction energies (W4-11RE) with ~ 11k reaction data points derived from the W4-11 total energies106, dissociation energies of small open-shell cationic dimers (SIE4 × 4)107, barrier heights (BH76) and reaction energies (BH76RC)105, and noncovalent interaction energies105(RG18). Results for MP2, κMP2, and state-of-the-art double-hybrid DFAs (ωB97M(2)108 and revDSD-PBEP86-D4109) are included for comparison. First, Fig. 6 shows that MLS2@W4 clearly outperforms MP2 and κMP2,demonstrating strong transferability from atomization energies alone to other energy types, even though it relies on a neural network with over 10k trainable parameters. In Fig. 6, translucent squares mark seen energy types (see Methods). While MLS2@W4 has only seen atomization energies, the two double hybrids have seen nearly all of the energy types shown, making the distinction between seen and unseen crucial for interpreting these results. Second, although the two double hybrids perform better for barrier heights and reaction energies (energy types seen by them and unseen by MLS2@W4), MLS2@W4 still achieves reasonable accuracy for these sets. For the SIE4 × 4 dataset, which is very difficult for the standard DFAs due to self-interaction errors105, with MAE slightly below 1 kcal mol−1, MLS2@W4 impressively outperforms both double hybrids by a factor of ~ 5. Overall, MLS2@W4 stands out as the most robust method considered here, being the only one that achieves MAE below 4 kcal mol−1 across all tested datasets. This further confirms that the general MLS2 framework combining ML with Eq. (16) is highly promising for developing future DFAs.
The real-space, machine-learned, and regularized extension of spin-component-scaled second-order Møller-Plesset perturbation theory (MLS2) model is trained on W4-11 atomization energies102 (MLS2@W4). MAEs of second-order Møller-Plesset perturbation theory (MP2), its κ-regularization (κMP2) and of two double-hybrid models (ωB97M(2)108 and revDSD-PBEP86-D4109) are shown for comparison. Translucent squares behind the markers denote seen energy types for the corresponding models (see Methods). The results for RG18 dataset101 are scaled by a factor of 10 for better visibility.
In view of the good MLS2 performance, it is important to note the regularizing role of Eq. (16). In addition to using a full amount of exact exchange (correlation here modeled relative to the HF reference), Eq. (16) ensures that MLS2 is exact for one-electron systems (\({E}_{c}^{{{\rm{MLS2}}}}=0\)). This is because, \({e}_{{\mbox{c}}}^{{\mbox{MLS2}}}\,({{\bf{r}}})={e}_{{\mbox{c,ss}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})={e}_{{\mbox{c,os}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})=0\) for N = 1, irrespective of the weights produced by the NN. This good property of MLS2 and likely its good transferability would be easily lost if other terms (e.g., exchange energy density or semilocal quantities multiplied by corresponding ML weights) are added to Eq. (16). Crucially, through the specific use of Eq. (16) and NNs, MLS2 provides a way of employing semilocal features [e.g., s(r)] while still satisfying the one-electron constraint, in contrast to double hybrids, which violate this constraint due to their way of employing semilocal features. Consequently, MLS2@W4 is not only exact for one-electron systems such as \({{{\rm{H}}}}_{2}^{+}\) (by contrast, the two double hybrids yield substantial errors upon stretching \({{{\rm{H}}}}_{2}^{+}\) as shown in Supplementary Fig. S17 in the SI), but also performs well for self-interaction cases involving more electrons given its excellent performance on SIE4 × 4.
Discussion
To address the urgent need for transferable ML DFAs, we introduce and analyze several key strategies based on real-space energy learning. By leveraging our regularized, spin-resolved PT2-based correlation energy density generator, we pursue two directions, each demonstrating a distinct aspect of the power of real-space ML for DFAs.
ML2 demonstrates the power of LES
The first direction, ML2, leverages \({e}_{{{\rm{c}}}}^{{{\rm{\kappa MP2}}}}({{\bf{r}}})\) (Eq. (5)) as a proxy reference for LES-based learning. While LES intrinsically expands a single energy datapoint of GES into thousands per molecule (each grid point becoming a distinct training datapoint), this data efficiency advantage is fully realized only when specific physical considerations are accounted for. These include the use of energy density per particle as the learning target (Eq. (3)), adopting the physically meaningful gauge (Eq. (5)), and employing a physically-informed ML model (Eq. (14)). When these conditions are met, LES provides significantly greater transferability compared to commonly used GES. In particular, our ML2 model trained solely on a small set of atoms effectively generalizes to diatomic dissociation curves. Moreover, under these physically-informed conditions, LES not only enhances transferability but also leads to smoother and faster learning convergence, as well as robustness with respect to variations in ML training conditions compared to GES.
MLS2 leverages our local quantities to develop transferable ML DFAs
In the second direction, MLS2, we employ our \({e}_{{\mbox{c}}}^{{\kappa} {{\mbox{MP2}}}} \, ({{\bf{r}}})\) and its decomposition into same-spin and opposite-spin channels to construct a real-space ML model for transferable DFAs. Specifically, MLS2 generalizes spin-component-scaled MP2 by scaling each spin channel locally with NN weights (Eq. (16)), combined with Head-Gordon’s κ-regularization. MLS2 improves over κMP2 across diverse systems, achieves competitive accuracy compared to modern double hybrids, and outperforms them for challenging systems affected by self-interaction errors. While the MLS2 ingredients [Eq. (16)] scale as their global (κMP2) counterparts, further algorithmic developments (e.g., ref. 110) will be required to reduce the cost to a level comparable with κMP2. Alternatively, ML2 surrogates of the MLS2 ingredients could be designed to lower the cost (see text below).
Going back to the demonstrated power of LES over GES in the proof-of-principle ML2 model based on the κMP2 reference calls for developing robust energy density generators at higher levels of theory. This will be a crucial objective in our future work, with the first step already taken in ref. 64, which enables obtaining ec(r) from full configuration interaction (FCI) wavefunctions, with the procedure being easily adaptable to other variational wavefunctions. To learn higher-level ec(r), one can adapt the LES-based ML2 model, currently designed for \({e}_{c}^{{\kappa} {{\mbox{MP2}}}}({{\bf{r}}})\), by adding rs in the features list and modifying Eq. (14). Nevertheless, we believe that a more controlled approach for ML of higher-level ec(r) can be achieved by integrating ML2 and MLS2 as follows. Using higher-level energy densities per particle, we can implement LES-based training for MLS2, while simultaneously replacing \({e}_{{\mbox{c,os}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\) and \({e}_{{\mbox{c,ss}}}^{{\kappa} {{\mbox{MP2}}}}\,({{\bf{r}}})\) of Eq. (16) with their ML2-based surrogates. This replacement avoids these two more expensive quantities in post-training calculations while leveraging existing ML2 physics (Eq. (14)).
Within the MPAC framework that we use here to demonstrate the advantages of LES64, the input density is fixed to HF, and the learning target is exclusively the energy, allowing us to clearly focus on the effects of LES as a real-space learning strategy for DFAs. At the same time, this does not imply that density learning, as a complementary real-space learning strategy, should be abandoned within frameworks where both densities and energies are learning targets. E.g., in DFA development within KS DFT, where both energies and densities are learning targets, loss functions can incorporate the LES term alongside density term (see, e.g., refs. 31,34), or even further sophisticate such loss functions by using the specific link between energy densities and correlation potential111. Such combined strategies would leverage strengths of both real-space approaches, which we plan to explore in future work.
While we demonstrate here several advantages of LES over GES, the strength of GES is that it can readily leverage existing global energy data from extensive chemical databases (e.g., GMTKN55). Computing high-level energy densities per particle for every system in such large databases would be impractical and likely unnecessary, particularly given the high transferability potential of LES demonstrated here using data from just eight atoms. Thus, a practical approach would be to design a loss function incorporating both GES and LES terms: employing GES for existing global energy data while strategically complementing it with LES-based training on carefully selected subsets (e.g., a dozen atoms and representative molecules from “Slim" subsets112 of GMTKN55) to leverage its power for embedding transferability into ML DFAs.
Methods
Computational details
All electronic structure calculations were performed using the PySCF 2.3.0 program package113,114 within the Python coding environment v3.11.4.
For the evaluations in Figs. 1(d), 2(a), and 6 and Supplementary Tables S1, S2, Supplementary Figs. S3, S17–S19, S20 in the SI, we have used def2-QZVPPD basis set115,116, while for the rest, unless specified otherwise, we have used def2-QZVP basis set115. Our implementation of the MP2 correlation energy density generator uses the Python package for optimizing tensor contractions88 together with JAX70 to enable parallelization and high-performance platform agnostic evaluation of the energy densities. For the energy density generation, we also employ the density fitting86,87 (DF) approximation for MP2 with the def2-QZVP(PD)-RI auxiliary basis set. We adapt the same DF code and combine it with the def2-universal-jkfit auxiliary basis set to calculate the exchange energy density. In Supplementary Section S3 in the SI, we discuss the effect on the accuracy from the numerical integration with respect to the DFT grid117 and from the use of DF.
For the regularized correlation energy density data generation, we set κ = 2 throughout this work unless otherwise specified.
Reference correlation energies are taken from specified references or obtained from CCSD(T) calculations in PySCF.
Neural Network training
The neural network training was performed with the Pytorch 2.1.2 deep learning library118. The input features (Supplementary Section S6 in the SI) were obtained in Python from the HF PySCF output of the given chemical system and pointwise evaluated on the DFT grid117. In particular, two FOD features at electronic temperatures T1 = 10,000K and T2 = 25,000K were employed for every system. Their implementation is based on the formula from ref. 71, which we divide by the density to obtain a dimensionless quantity. We employed the Adam optimizer119 in Pytorch for NN training with custom learning rates incorporating a warm-up period and a fixed number of training steps (epochs).
ML2 was trained on eight small closed-shell atoms/ions (H−, He, Be, Mg, Ne, Ar, Ca, and Kr). The architecture of the neural network for ML2 has three hidden layers, each consisting of 16 neurons. In ML2, we apply hyperbolic tangent (\(\tanh\)) activation functions to the NN output layers, bounding the ML2 weights between − 1 and 1 (for a concrete example illustrating the range of ML2 wc(r) weights that justifies this choice, see Supplementary Fig. S20 in the SI). For simplicity, the same \(\tanh\) activation is also used for all hidden layers. All models in this work are optimized by using either the LES-based or the GES-based loss function (see Supplementary Section S8 in the SI for specific details). In ML2, the learning rate follows an exponential decay that continues over 5000 epochs including the warm-up phase.
A detailed overview of the training set for the first MLS2 example is given in Supplementary Section S12 in the SI. For the architecture of the MLS2’s NN, we used four hidden layers with 64 neurons each. Following the ML2 architecture, in MLS2 we also use \(\tanh\) activation function for the hidden layers, while the sigmoid function is applied to the output layer to constrain the MLS2 weights between 0 and 1 before scaling them by a factor of 10 (see Supplementary Section S11 in the SI for further details on MLS2 construction). The total loss function contains total and interaction correlation energy errors, as detailed in Supplementary Section S8 in the SI. Here, the learning rate follows also an exponential decay over 1000 epochs.
The original MLS2 architecture (4 × 64) is also used for MLS2@W4, and in Supplementary Fig. S18 in the SI, we compare alternative NN architectures and show that 4 × 64 offers modest improvements in validation loss over smaller models, motivating its use. Additionally, we created ten MLS2@W4 NNs with different random seed initializations (see Supplementary Fig. S19 in the SI). From these ten models, the final MLS2@W4 NN shown in Fig. 6 was selected based on the lowest MAE on the entire W4-11 set used for training and validation. Since MLS2@W4 also involves open-shell systems, we have used unrestricted Hartree–Fock (UHF) orbitals. Following ref. 120 of Sim and co-workers, constrained-UHF (CUHF) was employed for reactions where UHF orbitals exhibited spin contamination following the criterion from ref. 121. In Fig. 6, translucent squares behind the markers denote seen energy types for a given model. The datasets shown correspond to the following energy types: atomization energies (W4-11), reaction energies (W4-11RE, BH76RC), noncovalent interaction energies (RG18), barrier heights (BH76), and the separate self-interaction error set (SIE4 × 4). While revDSD-PBEP86-D4 was trained on GMTKN55 (which includes W4-11, SIE4 × 4, BH76, BH76RC and RG18), ωB97M(2) was trained/validated on subsets of Head-Gordon’s large database, covering the same energy types with comparable datasets108 that are marked as seen for this model. For further MLS2@W4 training and testing details, see Supplementary Section S13 in the SI.
Data availability
Data supporting the key findings of this study are available from the manuscript and its supporting information. Additionally, a Source Data file is provided with this paper. Source data are provided with this paper.
Code availability
The Python code for the energy density generators used in this work is publicly available at Zenodo122 under the GNU General Public License v3.0.
References
Hush, M. R. Machine learning for quantum physics. Science 355, 580–580 (2017).
Ramakrishnan, R. & von Lilienfeld, O.A. Machine Learning, Quantum Chemistry, and Chemical Space. In Reviews in Computational Chemistry (eds Parrill, A.L. & Lipkowitz, K.B.) Vol. 30, Ch. 5 (John Wiley & Sons, Ltd), https://doi.org/10.1002/9781119356059.ch5 (2017).
von Lilienfeld, O. A. Quantum machine learning in chemical compound space. Angew. Chem. Int. Ed. 57, 4164–4169 (2018).
Schütt, K. T. et al. Unifying machine learning and quantum chemistry with a deep neural network for molecular wavefunctions. Nat. Commun. 10, 5024 (2019).
Tkatchenko, A. Machine learning for chemical discovery. Nat. Commun. 11, 4125 (2020).
Zheng, P. et al. Artificial intelligence-enhanced quantum chemical method with broad applicability. Nat. Commun. 12, 7022 (2021).
Huang, B. et al. The central role of density functional theory in the AI age. Science 381, 170–175 (2023).
Ramakrishnan, R. et al. Quantum chemistry structures and properties of 134 kilo molecules. Sci. Data 1, 140022 (2014).
Ramakrishnan, R., Dral, P., Rupp, M. & von Lilienfeld, A. O. Big data meets quantum chemistry approximations: the Δ-machine learning approach. J. Chem. Theory Comput. 11, 2087–2096 (2015).
Schütt, K. T., Sauceda, H. E., Kindermans, P.-J., Tkatchenko, A. & Müller, K.-R. SchNet - A deep learning architecture for molecules and materials. J. Chem. Phys. 148, 241722 (2018).
Chen, C., Ye, W., Zuo, Y., Zheng, C. & Ong, P. O. Graph networks as a universal machine learning framework for molecules and crystals. Chem. Mater. 31, 3564–3572 (2019).
Smith, J. S. et al. The ANI-1ccx and ANI-1x data sets, coupled-cluster and density functional theory properties for molecules. Sci. Data 7, 134 (2020).
Batatia, I., Kovacs, D. P., Simm, G., Ortner, C. & Csányi, G. MACE: Higher Order Equivariant Message Passing Neural Networks for Fast and Accurate Force Fields, 35, 11423–11436 (Curran Associates, Inc., 2022).
Merchant, A. et al. Scaling deep learning for materials discovery. Nature 624, 80–85 (2023).
Zhang, S. et al. Exploring the frontiers of condensed-phase chemistry with a general reactive machine learning potential. Nat. Chem. 16, 727–734 (2024).
Chandrasekaran, A. et al. Solving the electronic structure problem with machine learning. npj Comput. Mater. 5, 22 (2019).
Mills, K. et al. Extensive deep neural networks for transferring small scale learning to large scale systems. Chem. Sci. 10, 4129–4140 (2019).
von Lilienfeld, O. A. & Burke, K. Retrospective on a decade of machine learning for chemical discovery. Nat. Commun. 11, 4895 (2020).
Unke, O. T. et al. Machine learning force fields. Chem. Rev. 121, 10142–10186 (2021).
Nandy, A. et al. Computational discovery of transition-metal complexes: from high-throughput screening to machine learning. Chem. Rev. 121, 9927–10000 (2021).
Pederson, R., Kalita, B. & Burke, K. Machine learning and density functional theory. Nat. Rev. Phys. 4, 357–358 (2022).
Kulik, H. J. What’s left for a computational chemist to do in the age of machine learning? Isr. J. Chem. 62, e202100016 (2022).
Fiedler, L., Shah, K., Bussmann, M. & Cangi, A. Deep dive into machine learning density functional theory for materials science and chemistry. Phys. Rev. Mater. 6, 040301 (2022).
Rackers, J. A. A recipe for cracking the quantum scaling limit with machine learned electron densities. Mach. Learn. Sci. Technol. 4, 015027 (2023).
Fiedler, L. et al. Predicting electronic structures at any length scale with machine learning. npj Comput. Mater. 9, 115 (2023).
Tozer, D. J., Ingamells, V. E. & Handy, N. C. Exchange-correlation potentials. J. Chem. Phys. 105, 9200–9213 (1996).
Snyder, J. C., Rupp, M., Hansen, K., Müller, K.-R. & Burke, K. Finding density functionals with machine learning. Phys. Rev. Lett. 108, 253002 (2012).
Li, L., Baker, T. E., White, S. R. & Burke, K. Pure density functional for strong correlation and the thermodynamic limit from machine learning. Phys. Rev. B 94, 245129 (2016).
Schmidt, J., Benavides-Riveros, C. L. & Marques, M. A. L. Machine learning the physical nonlocal exchange-correlation functional of density-functional theory. J. Phys. Chem. Lett. 10, 6425–6431 (2019).
Dick, S. & Fernandez-Serra, M. Machine learning accurate exchange and correlation functionals of the electronic density. Nat. Commun. 11, 3509 (2020).
Nagai, R., Akashi, R. & Sugino, O. Completing density functional theory by machine learning hidden messages from molecules. npj Comput. Mater. 6, 43 (2020).
Bogojeski, M. et al. Quantum chemical accuracy from density functional approximations via machine learning. Nat. Commun. 11, 5223 (2020).
Kalita, B., Li, L., McCarty, R. J. & Burke, K. Learning to approximate density functionals. Acc. Chem. Res. 54, 818–826 (2021).
Li, L. et al. Kohn-sham equations as regularizer: building prior knowledge into machine-learned physics. Phys. Rev. Lett. 126, 036401 (2021).
Margraf, J. T. & Reuter, K. Pure non-local machine-learned density functional theory for electron correlation. Nat. Commun. 12, 344 (2021).
Bystrom, K. & Kozinsky, B. CIDER: an expressive, nonlocal feature set for machine learning density functionals with exact constraints. J. Chem. Theory Comput. 18, 2180–2192 (2022).
Ruth, M., Gerbig, D. & Schreiner, P. R. Machine learning for bridging the gap between density functional theory and coupled cluster energies. J. Chem. Theory Comput. 19, 4912–4920 (2023).
Riemelmoser, S., Verdi, C., Kaltak, M. & Kresse, G. Machine learning density functionals from the random-phase approximation. J. Chem. Theory Comput. 19, 7287–7299 (2023).
Kirkpatrick, J. Pushing the frontiers of density functionals by solving the fractional electron problem. Science 374, 1385–1389 (2021).
Gould, T., Chan, B., Dale, S. G. & Vuckovic, S. Identifying and embedding transferability in data-driven representations of chemical space. Chem. Sci. 15, 11122–11133 (2024).
Shimakawa, H., Kumada, A. & Sato, M. Extrapolative prediction of small-data molecular property using quantum mechanics-assisted machine learning. npj Comput. Mater. 10, 11 (2024).
Li, K. et al. Probing out-of-distribution generalization in machine learning for materials. Commun. Mater 6, 9 (2025).
Sherrill, C. D., Manolopoulos, D. E., Martinez, T. J. & Michaelides, A. Electronic structure software. J. Chem. Phys. 153, 070401 (2020).
Zhao, H., Gould, T. & Vuckovic, S. Deep mind 21 functional does not extrapolate to transition metal chemistry. Phys. Chem. Chem. Phys. 26, 12289–12298 (2024).
Becke, A. D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. A 38, 3098–3100 (1988).
Lee, C., Yang, W. & Parr, R. G. Development of the colle-salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 37, 785–789 (1988).
Becke, A. D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, 5648–5652 (1993).
Stephens, P. J., Devlin, F. J., Chabalowski, C. F. & Frisch, M. J. Ab initio calculation of vibrational absorption and circular dichroism spectra using density functional force fields. J. Phys. Chem. 98, 11623–11627 (1994).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Butera, V. Density functional theory methods applied to homogeneous and heterogeneous catalysis: a short review and a practical user guide. Phys. Chem. Chem. l Phys. 26, 7950–7970 (2024).
Cramer, C. J. & Truhlar, D. G. Density functional theory for transition metals and transition metal chemistry. Phys. Chem. Chem. Phys. 11, 10757 (2009).
Liu, F., Duan, C. & Kulik, H. J. Rapid detection of strong correlation with machine learning for transition-metal complex high-throughput screening. J. Phys. Chem. Lett. 11, 8067–8076 (2020).
Hollingsworth, J., Li, L., Baker, T. E. & Burke, K. Can exact conditions improve machine-learned density functionals? J. Chem. Phys. 148, 241743 (2018).
Dick, S. & Fernandez-Serra, M. Highly accurate and constrained density functional obtained with differentiable programming. Phys. Rev. B 104, L161109 (2021).
Nagai, R., Akashi, R. & Sugino, O. Machine-learning-based exchange correlation functional with physical asymptotic constraints. Phys. Rev. Res. 4, 013106 (2022).
Pokharel, K. Exact constraints and appropriate norms in machine-learned exchange-correlation functionals. J. Chem. Phys. 157, 174106 (2022).
Vuckovic, S. & Bahmann, H. Nonlocal functionals inspired by the strongly interacting limit of dft: exact constraints and implementation. J. Chem. Theory Comput. 19, 6172–6184 (2023).
Kasim, M. F. & Vinko, S. M. Learning the exchange-correlation functional from nature with fully differentiable density functional theory. Phys. Rev. Lett. 127, 126403 (2021).
Casares, P. A. M. GradDft. A software library for machine learning enhanced density functional theory. J. Chem. Phys. 160, 062501 (2024).
Mohanty, S., Yoo, S., Kang, K. & Cai, W. Evaluating the transferability of machine-learned force fields for material property modeling. Comput. Phys. Commun. 288, 108723 (2023).
Hartree, D. R. The wave mechanics of an atom with a non-coulomb central field. Part I. Theory and methods. Math. Proc. Camb. Philos. Soc. 24, 89–110 (1928).
Fock, V. Näherungsmethode zur Lösung des quantenmechanischen Mehrkörperproblems. Z. f.ür. Phys. 61, 126–148 (1930).
Kohn, W. & Sham, L. J. Self-consistent equations including exchange and correlation effects. Phys. Rev. 140, A1133–A1138 (1965).
Daas, K. J., Zhao, H., Polak, E. & Vuckovic, S. Exact Møller-Plesset adiabatic connection correlation energy densities. J. Chem. Theory Comput. 21, 5501–5513 (2025).
Szarek, P. & Tachibana, A. The field theoretical study of chemical interaction in terms of the rigged QED: new reactivity indices. J. Mol. Model. 13, 651–663 (2007).
Szarek, P., Sueda, Y. & Tachibana, A. Electronic stress tensor description of chemical bonds using nonclassical bond order concept. J. Chem. Phys. 129, 094102 (2008).
Tachibana, A. Energy density concept: a stress tensor approach. J. Mol. Structure 943, 138–151 (2010).
Lee, J. & Head-Gordon, M. Regularized orbital-optimized second-order Møller-Plesset perturbation theory: a reliable fifth-order-scaling electron correlation model with orbital energy dependent regularizers. J. Chem. Theory Comput. 14, 5203–5219 (2018).
Shee, J., Loipersberger, M., Rettig, A., Lee, J. & Head-Gordon, M. Regularized second-order Møller-Plesset theory: a more accurate alternative to conventional MP2 for noncovalent interactions and transition metal thermochemistry for the same computational cost. J. Phys. Chem. Lett. 12, 12084–12097 (2021).
Bradbury, J. et al. Jax: composable transformations of Python+NumPy programs. http://github.com/google/jax (2018).
Grimme, S. & Hansen, A. A practicable real-space measure and visualization of static electron-correlation effects. Angew. Chem. Int. Ed. 54, 12308–12313 (2015).
Bauer, C. A., Hansen, A. & Grimme, S. The fractional occupation number weighted density as a versatile analysis tool for molecules with a complicated electronic structure. Chem. Eur. J. 23, 6150–6164 (2017).
Grimme, S. Improved second-order Møller-Plesset perturbation theory by separate scaling of parallel- and antiparallel-spin pair correlation energies. J. Chem. Phys. 118, 9095–9102 (2003).
Grimme, S., Goerigk, L. & Fink, R. F. Spin-component-scaled electron correlation methods. WIREs Comput. Mol. Sci. 2, 886–906 (2012).
Kim, M.-C., Sim, E. & Burke, K. Understanding and reducing errors in density functional calculations. Phys. Rev. Lett. 111, 073003 (2013).
Vuckovic, S., Song, S., Kozlowski, J., Sim, E. & Burke, K. Density functional analysis: the theory of density-corrected dft. J. Chem. Theory Comput. 15, 6636–6646 (2019).
Song, S. et al. Extending density functional theory with near chemical accuracy beyond pure water. Nat. Commun. 14, 799 (2023).
Cruz, F. G., Lam, K.-C. & Burke, K. Exchange-correlation energy density from virial theorem. J. Phys. Chem. A 102, 4911–4917 (1998).
Burke, K., Cruz, F. G. & Lam, K.-C. Unambiguous exchange-correlation energy density. J. Chem. Phys. 109, 8161–8167 (1998).
Vuckovic, S., Irons, T. J. P., Savin, A., Teale, A. M. & Gori-Giorgi, P. Exchange-correlation functionals via local interpolation along the adiabatic connection. J. Chem. Theory Comput. 12, 2598–2610 (2016).
Vuckovic, S., Irons, T. J. P., Wagner, L. O., Teale, A. M. & Gori-Giorgi, P. Interpolated energy densities, correlation indicators and lower bounds from approximations to the strong coupling limit of dft. Phys. Chem. Chem. Phys. 19, 6169–6183 (2017).
Vuckovic, S., Levy, M. & Gori-Giorgi, P. Augmented potential, energy densities, and virial relations in the weak- and strong-interaction limits of dft. J. Chem. Phys. 147, 214107 (2017).
Møller, C. & Plesset, M. S. Note on an approximation treatment for many-electron systems. Phys. Rev. 46, 618–622 (1934).
Vuckovic, S., Fabiano, E., Gori-Giorgi, P. & Burke, K. MAP: An MP2 accuracy predictor for weak interactions from adiabatic connection theory. J. Chem. Theory Comput. 16, 4141–4149 (2020).
Jurečka, P., Šponer, J., Černý, J. & Hobza, P. Benchmark database of accurate (MP2 and CCSD(T) complete basis set limit) interaction energies of small model complexes, dna base pairs, and amino acid pairs. Phys. Chem. Chem. Phys. 8, 1985–1993 (2006).
Dunlap, B. I. Robust and variational fitting. Phys. Chem. Chem. Phys. 2, 2113–2116 (2000).
Werner, H. J., Manby, F. R. & Knowles, P. J. F. Fast linear scaling second-order Møller-Plesset perturbation theory (MP2) using local and density fitting approximations. J. Chem. Phys. 118, 8149–8160 (2003).
Smith, D. G. A. & Gray, J. opt_einsum - a python package for optimizing contraction order for einsum-like expressions. J. Open Source Softw. 3, 753 (2018).
Piris, M., Lopez, X. & Ugalde, J. M. Correlation holes for the helium dimer. J. Chem. Phys. 128, 134102 (2008).
Gritsenko, O. & Baerends, E. J. A simple natural orbital mechanism of “pure” van der waals interaction in the lowest excited triplet state of the hydrogen molecule. J. Chemi. Phys. 124, 054115 (2006).
Via-Nadal, M., Rodríguez-Mayorga, M. & Matito, E. Salient signature of van der waals interactions. Phys. Rev. A 96, 050501 (2017).
Furness, J. W. Accurate and numerically efficient r2 SCAN meta-generalized gradient approximation. J. Phys. Chem. Lett. 11, 8208–8215 (2020).
Levy, M. & Perdew, J. P. Hellmann-Feynman, virial, and scaling requisites for the exact universal density functionals. Shape of the correlation potential and diamagnetic susceptibility for atoms. Phys. Rev. A 32, 2010–2021 (1985).
Shi, Y., Shi, Y. & Wasserman, A. Stretching bonds without breaking symmetries in density functional theory. J. Phys. Chem. Lett. 15, 826–833 (2024).
Su, N. Q., Li, C. & Yang, W. Describing strong correlation with fractional-spin correction in density functional theory. Proc. Natl Acad. Sci. USA 115, 9678–9683 (2018).
Vuckovic, S., Wagner, L. O., Mirtschink, A. & Gori-Giorgi, P. Hydrogen molecule dissociation curve with functionals based on the strictly correlated regime. J. Chem. Theory Comput. 11, 3153–3162 (2015).
Nudejima, T. Machine-learned electron correlation model based on correlation energy density at complete basis set limit. J. Chem. Phys. 151, 024104 (2019).
Ikabata, Y. Machine-learned electron correlation model based on frozen core approximation. J. Chem. Phys. 153, 184108 (2020).
Han, R., Rodríguez-Mayorga, M. & Luber, S. A machine learning approach for MP2 correlation energies and its application to organic compounds. J. Chem. Theory Comput. 17, 777–790 (2021).
Irons, T. J. P. & Teale, A. M. The coupling constant averaged exchange-correlation energy density. Mol. Phys. 114, 484–497 (2015).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Karton, A., Daon, S. & Martin, J. M. L. W4-11: a high-confidence benchmark dataset for computational thermochemistry derived from first-principles W4 data. Chem. Phys. Lett. 510, 165–178 (2011).
Vuckovic, S. Density functionals from the multiple-radii approach: analysis and recovery of the kinetic correlation energy. J. Chem. Theory Comput. 15, 3580–3590 (2019).
Gráfová, L., Pitoňák, M., Rezáč, J. & Hobza, P. Comparative study of selected wave function and density functional methods for noncovalent interaction energy calculations using the extended S22 data set. J. Chem. Theory Comput. 6, 2365–2376 (2010).
Görigk, L. A look at the density functional theory zoo with the advanced GMTKN55 database for general main group thermochemistry, kinetics and noncovalent interactions. Phys. Chem. Chem. Phys. 19, 32184–32215 (2017).
Margraf, J. T., Ranasinghe, D. S. & Bartlett, R. J. Automatic generation of reaction energy databases from highly accurate atomization energy benchmark sets. Phys. Chem. Chem. Phys. 19, 9798–9805 (2017).
Goerigk, L. & Grimme, S. A general database for main group thermochemistry, kinetics, and noncovalent interactions – assessment of common and reparameterized (meta-)GGA density functionals. J. Chem. Theory Comput. 6, 107–126 (2010).
Mardirossian, N. & Head-Gordon, M. Survival of the most transferable at the top of Jacob’s ladder: defining and testing the ωB97M(2) double hybrid density functional. J. Chem. Phys. 148, 241736 (2018).
Santra, G., Sylvetsky, N. & Martin, J. M. L. Minimally empirical double-hybrid functionals trained against the GMTKN55 database: revDSD-PBEP86-D4, revDOD-PBE-D4, and DOD-SCAN-D4. J. Phys. Chem. A 123, 5129–5143 (2019).
Bahmann, H. & Kaupp, M. Efficient self-consistent implementation of local hybrid functionals. J. Chem. Theory Comput. 11, 1540–1548 (2015).
Giarrusso, S., Vuckovic, S. & Gori-Giorgi, P. Response potential in the strong-interaction limit of density functional theory: analysis and comparison with the coupling-constant average. J. Chem. Theory Comput. 14, 4151–4167 (2018).
Gould, T. & Vuckovic, S. “Slim” benchmark sets for faster method development. J. Chem. Theory Comput. 6, 6517–6527 (2025).
Sun, Q. et al. PySCF: the Python-based simulations of chemistry framework. WIREs Comput. Mol. Sci. 8, e1340 (2018).
Sun, Q. Recent developments in the PySCF program package. J. Chem. Phys. 153, 024109 (2020).
Weigend, F., Furche, F. & Ahlrichs, R. Gaussian basis sets of quadruple zeta valence quality for atoms H-Kr. J. Chem. Phys. 119, 12753–12762 (2003).
Rappoport, D. & Furche, F. Property-optimized Gaussian basis sets for molecular response calculations. J. Chem. Phys. 133, 134105 (2010).
Lebedev, V. I. & Laikov, D. N. A quadrature formula for the sphere of the 131st algebraic order of accuracy. Dokl. Math. 59, 477–481 (1999).
Ansel, J. Pytorch 2: faster machine learning through dynamic python bytecode transformation and graph compilation. In Proc. 29th ACM International Conference on Architectural Support for Programming Languages and Operating Systems, 2, 929–947 (ACM, 2024).
Kingma, D.P. & Ba, J. Adam: a method for stochastic optimization. Preprint at http://arxiv.org/abs/1412.6980 (2014).
Yu, H., Song, S., Nam, S., Burke, K. & Sim, E. Density-corrected density functional theory for open shells: how to deal with spin contamination. J. Phys. Chem. Lett. 14, 9230–9237 (2023).
Song, S., Vuckovic, S., Sim, E. & Burke, K. Density-corrected DFT explained: questions and answers. J. Chem. Theory Comput. 18, 817–827 (2022).
Polak, E., Zhao, H. & Vuckovic, S. Real-space machine learning of correlation density functionals. Repository name: Energy Density Generator v1.0. https://doi.org/10.5281/zenodo.17390040 (2025).
LeNail, A. NN-SVG: publication-ready neural network architecture schematics. J. Open Source Softw. 4, 747 (2019).
Mentel, Ł. M., van Meer, R., Gritsenko, O. V. & Baerends, E. J. The density matrix functional approach to electron correlation: dynamic and nondynamic correlation along the full dissociation coordinate. J. Chem. Phys. 140, 214105 (2014).
Acknowledgements
We thank for fruitful discussions and helpful comments from Kimberly Daas and Tim Gould. We also thank Prof. Eunji Sim for giving us access to their code120, which we have used to calculate CUHF orbitals in PySCF. The authors acknowledge technical support for the energy density generator from the EPICURE Support team (Emmanuel Kieffer, Frederic Pinel, Alban Rousset and Francesco Bongiovanni) within the EuroHPC project (EHPC-DEV-2024D97-076). The authors also acknowledge funding from the SNSF Starting Grant project to S.V. (TMSGI2_211246).
Author information
Authors and Affiliations
Contributions
E.P. ran all ML simulations. The electronic structure dataset was generated by H.Z. S.V. and E.P. did the data analysis and co-wrote the paper. S.V. supervised the project and conceptualized the idea together with E.P.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Ryosuke AKASHI and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Polak, E., Zhao, H. & Vuckovic, S. Real-space machine learning of correlation density functionals. Nat Commun 16, 11306 (2025). https://doi.org/10.1038/s41467-025-66450-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-66450-z