Abstract
Dual-atom catalysts offer high atom utilization and synergistic inter-atom interactions, yet their use in high-temperature thermocatalysis remains largely unexplored due to challenges in achieving structurally homogeneous and robust active sites. Herein, we report a scalable coordinated bottom-up strategy for the synthesis of a Cu-Ni dual-atom catalyst supported on nitrogen-doped carbon (CuNi-DAC), featuring a well-defined N2Cu–N2–NiN2 configuration in which each metal atom is coordinated to four nitrogen atoms and bridged by two nitrogen atoms. Under reverse water-gas shift reaction conditions, CuNi-DAC achieves CO2 conversion approaching thermodynamic equilibrium with nearly 100% CO selectivity. Critically, CuNi-DAC maintains its atomic structure and catalytic performance up to 600 °C over repeated cycles, while reference catalysts including Cu-SAC and Ni-SAC experience severe deactivation along with metal sintering. Comprehensive ex-situ and in-situ characterizations, integrated with theoretical calculations, reveal that d–d orbital coupling and electronic polarization between adjacent Cu and Ni centers enhance selective CO2 reduction to CO product, while reinforcing metal–support interactions to mitigate sintering. The in-depth mechanistic insights and the scalable synthesis provide a blueprint for the rationally designing next-generation dual-atom catalysts with enhanced efficiency, stability, and tailored activity for target chemical transformations.
Similar content being viewed by others
Introduction
The conversion of carbon dioxide (CO2) into carbon monoxide (CO) via the reverse water-gas shift (RWGS) reaction is one of the most promising strategies for transforming this pervasive greenhouse gas into a valuable industrial feedstock1,2,3,4,5. Due to the high thermodynamic stability of CO2, the RWGS reaction typically requires high temperatures to overcome sluggish reaction kinetics, emphasizing the need for efficient catalysts to lower activation energy barriers and improve overall reaction efficiency3,4,5. Although noble metals exhibit outstanding catalytic performance, their high cost and limited availability have driven research toward alternative transition metal (TM) based alternatives, including iron (Fe), cobalt (Co), nickel (Ni), and copper (Cu), which offer comparable activity at a lower cost4,5,6,7,8,9. Extensive efforts have focused on developing TM-based catalysts for RWGS, including metal alloys, nanostructured metal, and metal/metal oxide composites2,3,4,5,6,7,8,9,10. However, these catalysts often suffer from undesired side reactions, particularly CO2 methanation, leading to CH4 formation and lower CO selectivity.
Nanoscaling metal particles can suppress methanation, but sintering-induced catalytic deactivation remains a significant concern under RWGS reaction conditions7,8,9,10. Recently, transition metal single-atom catalysts (SACs) have emerged to maximize atom utilization and exhibit unique selectivity toward RWGS reaction not observed in bulk or nanostructured catalysts7,11,12,13,14. Nonetheless, SACs often encounter stability issues in reducing atmospheres, where metal atoms tend to migrate, cluster, and ultimately deactivate the catalyst8,11,12,13,14,15. Emerging from recent advances in electrochemistry, dual-atom catalysts (DACs) have attracted significant attention as a promising strategy for stabilizing isolated metal sites and enhancing catalytic performance16,17. In contrast to SACs that rely on individual metal atoms, DACs feature adjacent metal centers that promote cooperative interactions, thereby modulating the local electron density, enhancing CO2 activation, and mitigating side reactions14,15,16,17,18,19. The introduction of a heteronuclear metal site disrupts linear scaling relationships, enabling more efficient catalytic pathways.
Although various heteroatom-based DACs, included NiZn, CoNi, NiCu, NiFe, TeCu, NiSn, and NiCd have shown improved kinetics and selectivity in electrochemical CO2 reduction19,20,21,22,23,24,25,26,27,28,29, their potential for thermochemical CO2 conversion remains largely unexplored4,12,30,31,32,33. Moreover, defining the atomic-scale structure of DACs through advanced characterization continues to pose a significant technical challenge. From the perspective of catalyst development, precisely controlling atomic configurations has been a major obstacle in establishing well-defined active sites in DACs19,20,21,22,34. Unlike conventional metal catalysts, DACs require tailored synthetic protocols not only to maintain atomic dispersion but also to achieve uniquely fine-tuned intermetallic interactions. Recent advances in bottom-up approaches, such as coordination chemistry, atomic layer deposition, and templated pyrolysis, have enabled synthesis of atomically dispersed catalysts35,36,37,38,39,40. Among various support materials, metal–nitrogen-doped carbon (M-N-C) frameworks stand out by providing strong metal–nitrogen coordination for anchoring isolated metal sites while retaining a high surface area24,25,26,27,39,40,41,42. However, the application of DACs in thermocatalytic reactions, a challenging and previously unexplored approach, necessitates meticulous atomic-level optimization to ensure robust high-temperature reactivity and durability. Additionally, even under the challenge of precisely controlling dual-atomic configurations, scalable and reproducible synthesis methods are essential for enabling practical applications39,40,41,42,43.
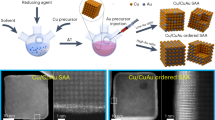
Herein, we developed a rational strategy to construct highly dispersed dual-atomic metal sites capable of efficiently activating CO2 molecules while delivering exceptional stability under thermocatalytic conditions. We synthesized an M-N-C based DAC with Cu-Ni atomic configuration (CuNi-DAC) through a coordinated bottom-up route involving Cu and Ni phthalocyanine precursors, a silica template, and a controlled pyrolysis process (Fig. 1a). To systematize structure and synthesis relationships, we benchmarked our coordinated bottom-up route against two controls (Fig. 1): (i) a non-coordinated bottom-up analogue using neat phthalocyanines with metal salts under identical templating/thermal conditions, and (ii) a conventional top-down route on a preformed carbon support loaded and hydrothermally reduced with metal and nitrogen precursors. The resulting catalyst from the coordinated bottom-up route features each Cu and Ni atom coordinated to four N atoms and interconnected by two bridging N atoms (N2Cu–N2–NiN2). This method affords gram-scale (~15 g) production of CuNi-DAC with uniform dual-atom sites, as verified by in-depth X-ray absorption spectroscopy and atomic-resolution electron microscopy. Under RWGS conditions, CuNi-DAC achieves CO2 conversion approaching thermodynamic equilibrium with ~100% CO selectivity while maintaining its structural integrity up to 600 °C over repeated cycles. A combined experimental and theoretical investigation reveals that a distinct Cu-Ni electronic structure not only facilitates CO desorption, driving superior CO selectivity, but also reinforces metal–support anchoring to mitigate sintering. These findings emphasize the importance of precisely engineered DACs within an M-N-C framework for advancing thermal catalysis and offer a scalable blueprint for next-generation CO2 reduction catalysts.
a Coordinated bottom-up strategy: pre-coordinated metal–phthalocyanine precursors are combined with a colloidal silica template, followed by pyrolysis and subsequent template etching. b Bottom-up control: metal salts with neat (non-coordinated) phthalocyanine precursors are processed under an otherwise identical protocol to (a). c Conventional top-down route: a preformed carbon support is first functionalized, then loaded and reduced hydrothermally with metal and nitrogen precursors.
Results
Strategic synthesis and characterization of CuNi-DAC
We developed a strategic synthesis of CuNi-DAC using Cu and Ni phthalocyanines (PC) as precursors through a coordinated bottom-up approach (CB-method) (Fig. 1a). The nitrogen linkages in the PC framework provide bridging sites that facilitate close coordination of Cu and Ni atoms, thereby enabling dual-atom site formation. Our CB-method involves pyrolysis of Cu-PC and Ni-PC within a silica composite template. The silica template helps mitigate restacking of PC precursors by interrupting π–π interactions in their two-dimensional form, thereby enhancing atomic dispersion and surface area (Supplementary Fig. 1 and Supplementary Table 1). Before pyrolysis, we performed an agitation step in a mixed-solvent system (DMF:ethanol = 3:1) to improve the molecular-level miscibility of Cu-PC, Ni-PC, and silica. By controlling the heating ramp rate (1.5–3.5 °C min−1) during pyrolysis, we optimized the decomposition and reconstruction of Cu-PC and Ni-PC, thus promoting the formation of closely positioned Cu and Ni atoms in the M-N-C lattice and achieving a highly dispersed dual-atom configuration (Supplementary Fig. 2a–c). Notably, having a highly dispersed DAC significantly influenced stable CO selectivity across a range of temperatures in the RWGS reaction (Supplementary Fig. 2d). To elucidate the roles of the silica template and etching process, we prepared two controls under otherwise identical conditions, a non-etched sample (CuNi-N/E) and a non-template sample (CuNi-N/T). As detailed in the Supplementary Information, these controls reveal that the silica-templated etching protocol not only suppresses π–π restacking but also mitigates metal–metal reaggregation during carbonization and serves as a sacrificial scaffold that opens porosity (Supplementary Figs. 3–7).
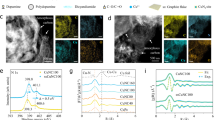
The morphology of CuNi-DAC was characterized via ultra-high-resolution high-angle annular dark-field (HAADF) scanning transmission electron microscopy (STEM) and energy-dispersive X-ray spectroscopy (EDS) (Fig. 2a, b and Supplementary Fig. 8). The resulting images revealed re-coordination of Cu and Ni sites with highly dispersed atomic moieties. We employed electron energy loss spectroscopy (EELS) to probe the local electronic environment (Fig. 2c and Supplementary Fig. 9). The Cu–Ni inter-atomic distance was found to be approximately 0.25 nm based on the Cu L-edge and Ni L-edge signals. Further examination by multi-channel ultra-high-resolution STEM/EDS/EELS showed that roughly 76% of the catalyst contains dual-atom sites (Fig. 2d, e and Supplementary Figs. 8–11). Kernel density distribution analysis indicated that the average Cu–Ni distance was about 2.5 ± 0.5 Å (Fig. 2e), matching the EELS measurements (Supplementary Figs. 9–11)22,44. The high fraction of dual-atom sites can be attributed to the binding energetics of the PC precursors. Dispersion-corrected density functional theory (DFT) calculations for π–π-coupled PCs reveal inter-precursor binding energies of −1.208 eV for CuPC/CuPC, −1.295 eV for NiPC/NiPC, and −1.301 eV for CuPC/NiPC, indicating thermodynamically robust precursor association with a modest energetic bias toward heteronuclear Cu-PC/Ni-PC pairing (Fig. 2f). This favorable interaction of PC precursors facilitates the formation of the preferentially arranged Cu-Ni dual-atom configuration at the atomic level. This strategic coordinated bottom-up method reproducibly yielded CuNi-DAC catalysts at a scalable batch size of 15 g (Fig. 2g). Through DFT calculations, we examined four types of CuNi-DAC configurations in an M-N-C planar framework (Fig. 2h), categorized by inter-atomic distances between Cu and Ni (2.380 Å, 2.557 Å, 4.088 Å, and 4.983 Å)22,34,37. Thermodynamic assessments at both 298 K and 773 K revealed that the structure featuring a 2.557 Å Cu–Ni spacing, linked by two bridging N atoms (N2Cu–N2–NiN2), was the most stable (binding energies of −3.7 eV at 298 K and −4.5 eV at 773 K). These theoretical results align well with EELS and STEM/EDS observations, and more details on the coordination structure analysis will be discussed in later sections.
a, b Ultra-high resolution Cs-corrected HAADF-STEM image (a) and EDS mapping (b) of the CuNi-DAC. Cu is shown in red, and Ni in cyan. c EELS mapping showing signal intensities as a function of spectral scanning vector, indicating the inter-atomic distance between Cu-L and Ni-L edges. d Percentage of DAC with an intermetallic distance of ~2.5 Å, compared to SAC. e Histogram of Cu-Ni inter-atomic distances observed via kernel density estimation based on STEM and EELS analyses. f DFT-calculated binding energies (EB) for interaction between metal phthalocyanine (PC) precursors. Square plots (gray line) represent Cu-Cu PCs interactions, circular plots (cyan line) represent Ni-Ni PCs interactions, and triangular plots (red line) represent Cu-Ni PCs interactions, showing the enhanced stability of heteronuclear conditions. g Scalable synthesis of CuNi-DAC ( ~ 15 g) through the coordinated bottom-up process. h DFT comparison of formation energies (EF) and relative structural stabilities of different CuNi-DAC coordination configurations at 298 K and 773 K.
As controls, we synthesized Cu-SAC and Ni-SAC using the same protocol but with only a single metal phthalocyanine precursor (Supplementary Figs. 12 and 13). To clarify the contribution of precursor coordination, we also implemented a non-coordinated bottom-up process (B-method) by co-pyrolyzing neat phthalocyanines with the corresponding metal salts under the same silica templating and etching and thermal program. The B-method was designed to produce a Cu–Ni single-atom/nanoparticle hybrid (CuNi-SA/NP) on an N-doped carbon, as well as single-metal analogues (Cu-SA/NP and Ni-SA/NP) (Fig. 1b and Supplementary Figs. 14–16). Additionally, to a provide nanoparticle benchmark without single-atom participation, we prepared CuNi nanoparticles (CuNi-NP) via a conventional top-down method (T-method). In this T-method, bulk graphite was exfoliated via Hummers’ method to form near-two-dimensional graphene oxide45, then reduced and doped with ionic Cu and Ni precursors alongside a nitrogen dopant through hydrothermal reduction (Fig. 1c and Supplementary Fig. 17). HAADF-STEM/EDS/EELS reveal in CuNi–SA/NP a mixed population of isolated M–N single-atom motifs and N-anchored nanoparticles that are more uniform and typically smaller (22.7avg ± 29.7 nm; 52.4max nm) than in the top-down CuNi–NP (45.9avg ± 40.5 nm; 86.4max nm) (Supplementary Fig. 18). These observations indicate that nitrogen functionalities from phthalocyanines assist nucleation and stabilization of particles but do not suppress M–M aggregation sufficiently to yield true Cu–Ni diatomic sites.
Coordination chemistry
To investigate the structural and compositional properties of CuNi-DAC, we conducted Raman spectroscopy, X-ray fluorescence spectroscopy (XRF), inductively coupled plasma atomic emission spectroscopy (ICP-AES), and X-ray photoelectron spectroscopy (XPS) analyses (Fig. 3a–d, Supplementary Figs. 19–26 and Supplementary Table 2). These techniques revealed distinct M-N-C structural features arising from the synthesis protocols: the CB-method that produces SAC and DAC, the B-method that provides mixed multi-SAC and NP, and the T-method that yields NP materials. In the Raman spectra, the defect (D) band at ~1350 cm−1 and the graphite (G) band at ~1580 cm−1 were observed in all M-N-C samples (Fig. 3a). The D-band corresponds to disordered graphitic carbon, whereas the G-band reflects the degree of graphitization. Notably, the intensity ratio (ID/IG) for CuNi-DAC (1.01) was significantly higher than that of CuNi-NP (0.24), implying a higher density of crystallographic defects introduced by atomic metal sites in CuNi-DAC. Catalysts prepared by the CB-method (CuNi-DAC, Cu-SAC, and Ni-SAC) and B-method (CuNi-SA/NP) exhibited comparable ID/IG ratios (1.01–1.18), with broader D and G peaks, an indication of distortion in the sp2 carbon lattice caused by nitrogen–carbon and metal–nitrogen bonding within the M-N-C framework36 (Fig. 3a and Supplementary Fig. 21). Additional Raman analyses for heterogeneity of detect sites are provided in the Supplementary Information (Supplementary Fig. 22).
Raman spectra (a) and XPS survey spectra in the Cu 2p–Ni 2p region (b) for CuNi-DAC, Ni-SAC, Cu-SAC, and CuNi-NP. HR-XPS spectra of Cu 2p3/2 (c) and Ni 2p3/2 (d) with deconvolution profiles. Normalized Cu K-edge XANES spectra (e) and Fourier-transformed (FT) k2-weighted EXAFS spectra in R-space (f) of CuNi-DAC, Cu-SAC, and reference samples. Normalized Ni K-edge XANES spectra (g) and FT k2-weighted EXAFS spectra in R-space (h) of CuNi-DAC, Ni-SAC, and reference samples. i FT k3-weighted EXAFS spectra at the Cu K-edge for CuNi-DAC, overlaid with fitted curves for four DAC models characterized by Cu–Ni inter-atomic distances of 2.380, 2.557, 4.088, and 4.983 Å. In the structural schematics, black, yellow, orange, and blue spheres represent carbon, nitrogen, copper, and nickel, respectively. j Wavelet-transform contour plots of Cu K-edge EXAFS for Cu foil, Cu-PC, Cu-SAC, and CuNi-DAC.
Elemental analyses by ICP, XRF, and XPS survey spectra confirmed the active metal concentration in the catalysts. ICP-AES results quantitatively confirmed that all samples exhibited a metal content trend within the range of 12–13 wt.% (Supplementary Table 2). Surface atomic concentrations of transition metals in all samples were obtained from XPS surveys, yielding the following values: 3.4 at.% Cu in Cu-SAC, 3.7 at.% Ni in Ni-SAC, 1.5 at.% Cu and 0.8 at.% Ni in CuNi-DAC, 3.6 at.% Cu and 0.9 at.% Ni in CuNi-SA/NP, and 1.5 at.% Cu and 4.3 at.% Ni in CuNi-NP, (Fig. 3b and Supplementary Fig. 23). Interestingly, XPS revealed that the surface concentration of Ni in CuNi-NP is significantly higher than its bulk value, suggesting that Ni may preferentially resides at the outer surface of the nanoparticle, consistent with HAADF-STEM and EDS analyses (Supplementary Fig. 17). In the high-resolution XPS (HR-XPS) N 1 s, the peak intensity of M–Nx in CuNi-DAC (10.31%) is enhanced compared to that in CuNi-NP (~0%) and CuNi-SA/NP (5.47%), indicating a higher concentration of metal–nitrogen moieties (Supplementary Fig. 19)6,11,23,24,25. This suggests that the CB-method synthesis effectively promotes the incorporation of atomically dispersed N-anchored metal sites within the M-N-C framework. As shown in Fig. 3c, d, HR-XPS Cu and Ni 2p spectrum further clarified the oxidation states of the active metal species. The Cu 2p3/2 peaks for CuNi-DAC and Cu-SAC appeared at binding energies between those of Cu1+/0 (932.1 eV) and Cu2+ (934.5 eV), indicating positive oxidation state for Cu15,21. Similarly, the binding energies of Ni 2p3/2 in CuNi-DAC and Ni-SAC are positioned between those of Ni0 (852.6 eV), Ni1+ (854.03 eV), and Ni2+ (854.6 eV), respectively, showing the positive Ni species6,46. Because the Cu1+ and Cu0 2p3/2 binding energies are closely spaced, the Cu LMM Auger spectrum was used to further differentiate Cu oxidation states (detailed in Supplementary Fig. 25). Quantitatively, CuNi-DAC exhibits a lower Cu2+ fraction (85.6%) than Cu-SAC (94.4% Cu2+), while the Ni2+ fraction is higher in CuNi-DAC (89.9%) than in Ni-SAC (75.3% Ni2+) (Supplementary Table 3). This directional change (Cu slightly reduced; Ni slightly oxidized) is consistent inter-site charge polarization inferred from followed DFT calculations in theoretical understanding section. In addition, CuNi-DAC, Cu-SAC, and Ni-SAC exhibit an extra N 1 s component at higher binding energy than free pyridinic-N (~398.0–398.7 eV) of the N-doped carbon support, attributable to M-Nx coordination and accounting for ~10.31% of the total N envelope in each catalyst (Supplementary Fig. 26). These cationic nature and N 1 s coordination signature result from the strong electron-withdrawing interactions between the metal centers and coordinated nitrogen atoms in the M-N-C framework21,47.
The atomic structure and local coordination environment of CuNi-DAC and other SACs were investigated through X-ray absorption near-edge structure (XANES) and extended X-ray absorption fine structure (EXAFS) measurements at Cu K-edge and Ni K-edge. As shown in Fig. 3e, the Cu K-edge XANES spectra for both CuNi-DAC and Cu-SAC lie between those of Cu foil (Cu0) and Cu-PC (Cu2+), while being closer to Cu2+. Similarly, the Ni K-edge XANES spectra are positioned between Ni foil (Ni0) and Ni-PC (Ni2+) (Fig. 3g). In addition, all three catalysts synthesized using the CB-method exhibited prominent white line peaks at the Cu and Ni K-edges, with intensities that were significantly higher than those of Cu foil and Ni foil, respectively. These observations indicate that the average oxidation state of isolated Ni and Cu atoms is between 0 and +2 in both SAC and DAC, consistent with HR-XPS deconvolution data (Fig. 3c, d). EXAFS spectra for CuNi-DAC and Cu-SAC reveal a principal peak at 1.5 Å, attributable to Cu–N coordination (Fig. 3f)22,36. No scattering peaks at 2.23 Å (Cu–Cu) or 2.79 Å (Cu–O–Cu), characteristic of Cu foil and CuO, were observed, confirming the absence of metallic Cu or CuOx nanoparticles (Supplementary Fig. 27b)10,47. In contrast, a peak at 2.45–2.54 Å in CuNi-DAC is attributed to Cu-Ni atomic coordination within the N2Cu–N2–NiN2 ensemble (Fig. 3i)22,30,48, while there is no corresponding signal in Cu-SAC at the same position (Fig. 3f). In the Ni K-edge EXAFS spectra, the dominant peak at 1.38 Å is assigned to the Ni−N coordination in CuNi-DAC and Ni-SAC18,19,20,21,22,23,48; the Cu–Ni peak at 2.48–2.52 Å in CuNi-DAC is similarly attributable to the Ni site (Fig. 3h). For the control CuNi-SA/NP, HR-XPS, XANES, and EXAFS data collectively indicate coexisting M–Nx single-atom sites and metallic M–M nanoparticles, distinct from the N2Cu–N2–NiN2 signature of CuNi-DAC; details are provided in the Supplementary Information (Supplementary Figs. 24 and 28).
The EXAFS fitting analysis was conducted to further determine the configuration of CuNi-DAC. The detailed fitting results, including metal−nitrogen/metal−metal path length and coordination numbers, are shown in Supplementary Fig. 29. Several structures of CuNi-DAC were proposed by DFT calculations, including N3Cu–NiN3, N2Cu–N2–NiN2, N2CuN2–N2NiN2, and N2CuN2–C2–N2NiN2 models, with different distances between Cu and Ni atoms (Supplementary Fig. 30a–d). It is suggested that the most plausible arrangement of DAC is the N2Cu–N2–NiN2 (2.557 Å) ensemble, where both Cu and Ni atoms are embedded in the nitrogen-doped carbon lattice by coordination with four N atoms each and colligated by the two bridged N atoms (Fig. 3i and Supplementary Fig. 29). This configuration is consistent with DFT calculations (Fig. 2h). Wavelet transform (WT) analysis of the EXAFS spectra also confirmed a predominant Cu–N coordination for both CuNi-DAC and Cu-SAC, distinct from that of CuNi-NP (Fig. 3j and Supplementary Fig. 31). Consistent with this assignment, no Cu–Cu contribution is observed for CuNi-DAC or Cu-SAC, in contrast to the signals present in Cu-foil and CuNi-NP. To verify that Cu and Ni are coordinated with N rather than O, the EXAFS spectra of the catalyst samples were compared with those of corresponding metal oxide reference samples (Supplementary Figs. 27 and 32)36,47,48,49. The results confirmed the presence of metal–nitrogen coordination in both DAC and SAC (Ni–N at 1.47 Å and Cu–N at 1.5 Å) samples. Collectively, the aforementioned characterizations of the as-prepared CuNi-DAC, particularly the HAADF-STEM and EXAFS analyses, align well with the proposed N2Cu–N2–NiN2 model, confirming consistency in electronic structure changes, local coordination environments, and intermetallic distances47,48,49,50.
Thermocatalytic CO2 reduction performance of CuNi-DAC
The RWGS activity of each catalyst was investigated under steady-state conditions by incrementally increasing the temperature from 300 °C to 600 °C in 50 °C intervals, then reversing the process back to 300 °C (Supplementary Fig. 33a–d). Notably, Ni-SAC and Cu-SAC exhibited a hysteresis in CO2 conversion and/or CO selectivity between the heating and cooling phases, suggesting catalyst deactivation. While Ni-SAC produced substantial amounts of the undesired byproduct CH4, Cu-SAC showed high CO selectivity but suffered from low CO2 conversion (Supplementary Fig. 33c, d). In contrast, the CuNi-DAC catalyst achieved outstanding performance, maintaining 100% CO selectivity and robust CO2 conversion across all temperatures tested (Supplementary Fig. 33a). To evaluate catalyst durability, a 3-cycle test extending over 100 h was carried out at a doubled weight hourly space velocity (WHSV) of 12,000 cc g-1 h-1 (Fig. 4a, c, e, g). Under these conditions, CuNi-NP revealed a dramatic decline in catalytic performance during the repeated test cycles, accompanied by severe sintering after reaction (Fig. 4a, b and Supplementary Fig. 34). Because these nanoparticles are not confined within the N-C framework, they coarsen via Ostwald ripening, leading to heterogeneous particle sintering on the carbon sheet8,9,10,43,51. Furthermore, DFT calculations indicated that the intrinsic instability of CuNi clusters substantially contributed to deactivation (Supplementary Fig. 35). Under the same steady-state protocol, CuNi-SA/NP shows the expected thermodynamic shift with higher CH4 at lower temperature and higher CO at higher temperature. (Fig. 4c, d). It maintains CO2 conversion more robustly than NP-only (CuNi-NP), which is consistent with its more uniform N-anchored nanoparticles and reduced sintering. The single-metal analogues (Cu-SA/NP and Ni-SA/NP) reaffirm the metal-dependent selectivity trends (Supplementary Fig. 36). Notably, CuNi-DAC sustained 100% CO selectivity and near-equilibrium CO2 conversion throughout all three cycles (Fig. 4e)52,53, while retaining its the atomic metal dispersion (Fig. 4f and Supplementary Fig. 37).
a, c, e, g, 3-cycle RWGS performance of CuNi-NP (a), CuNi-SA/NP (c), CuNi-DAC (e) and PM-SAC (a physical mixture of Cu-SAC and Ni-SAC) (g) measured at WHSV of 12,000 cc g−1 h−1 over 100 h. The test involved three thermal cycles (300 → 600 → 300 °C per cycle) under atmospheric pressure with a CO2:H2 feed ratio of 1:4. HAADF-STEM images of spent catalysts after the 3-cycle RWGS reaction for CuNi-NP (b), CuNi-SA/NP (d), CuNi-DAC (f), and PM-SAC (h). i DFT-calculated coordination formation energies (EF) for Ni-SAC (circle plots, blue line), Cu-SAC (square plots, orange line), and CuNi-DAC (triangle plot, red line), reflecting their intrinsic structural stabilities. j Catalytic performance map comparing the RWGS activity of CuNi-DAC with previously reported catalysts. Detailed experimental conditions of references are summarized in Supplementary Table 4.
To investigate the effect of inter-atomic proximity on catalytic behavior, a physically mixed sample of Cu-SAC and Ni-SAC (denoted as PM-SAC) was also evaluated (Fig. 4g, h). The improved conversion of the physical mixture (PM-SAC) in the first cycle arises from parallel reactions on separate CuN4 and NiN4 domains rather than true Cu–Ni dual-site synergy (Supplementary Fig. 33). Upon cycling, preferential aggregation/deactivation of Ni-rich domains removes the methanation pathway, yielding a more Cu-like ensemble with higher CO selectivity but lower overall conversion (Supplementary Fig. 38). Across repeated cycles, PM-SAC delivers ~10–20% lower CO2 conversion than CuNi-DAC. Consistent with this behavior, PM-SAC exhibited reduced CO2 conversion and significant CH4 production (up to 64.3% selectivity at 450 °C during the first cycle), along with severe metal sintering during the 3-cycle test. These results highlight the crucial role of a well-defined Cu-Ni atomic configuration in ensuring both performance and durability. Morphological analyses revealed severe sintering in the spent catalysts except CuNi-DAC, with the following average metal size distribution: CuNi-DAC (0.3avg ± 0.3 nm; 1.5max nm) <PM-SAC (6.9avg ± 7.3 nm; 26.8max nm) <CuNi-SA/NP (38.3avg ± 15.3 nm; 65.7max nm) <CuNi-NP (57.5avg ± 40.9 nm; 134.1max nm), consistent with the observed deactivation profiles (Supplementary Fig. 39). In addition, Raman spectra of fresh and spent CuNi-DAC retained an ID/IG ratio of 1.01 after reaction, indicating the stability of defective dual-atom active sites within the M-N-C framework under RWGS reaction conditions (Supplementary Fig. 40). Further corroborating these results, DFT calculations indicate that CuNi-DAC has a more favorable formation energy relative to the other SACs (Fig. 4i). Taken together, these findings underscore the importance of well-defined CuNi dual-atom sites for delivering both high catalytic selectivity and thermal stability in the RWGS reaction. Compared with previously reported RWGS catalysts, CuNi-DAC stands out by exhibiting superior CO2 conversion and CO selectivity (Fig. 4j, Supplementary Fig. 41 and Supplementary Table 4)5,6,7,8,9,10,11,12,13,32,54,55,56,57,58,59,60,61.
In situ DRIFTS analysis and 13CO2 isotope tracing for experimental mechanistic insights
To elucidate detailed mechanistic insights into CO2 hydrogenation during the RWGS reaction, we performed in situ diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) analyses on CuNi-DAC, Cu-SAC, Ni-SAC, and CuNi-NP. Sequential experiments were carried out at 500 °C and 600 °C, involving CO2 adsorption followed by hydrogenation for 30 min (Fig. 5a–e and Supplementary Figs. 42, 43). Distinct COOH* intermediates emerged prominently near 1400 cm−1 on the all Cu-containing catalysts (CuNi-DAC, Cu-SAC, and CuNi-NP)8,41,50; however, CuNi-DAC and Cu-SAC exhibited weaker COOH* signals than CuNi-NP, indicating lower accumulation of COOH* intermediate on atomically dispersed active sites (Fig. 5a, c, d). The presence of COOH* species was further corroborated by broad O–H stretching vibrations (v-OH)51 in the region of 3500–2800 cm−1. Conversely, Ni-SAC exhibited negligible COOH* bands but displayed pronounced adsorbed CO peaks (Fig. 5b). Detailed examination of adsorbed CO vibration (v-CO*) revealed the following order of peak intensities: Ni-SAC » CuNi-NP > Cu-SAC > CuNi-DAC, with Ni-SAC showing the strongest CO binding affinity (2175–2105 cm−1)2,18,48,49,50. In addition, PM-SAC exhibited Cu-SAC like features (v-COOH* near 1400 cm−1) together with Ni-SAC like signatures (linearly adsorbed CO* at 2175–2105 cm-1) (Supplementary Fig. 44). Importantly, other commonly reported intermediates such as formate (HCOO⁻; typically at 1590, 1516, 1395, 1375 and 1369 cm−1) were not detected on any catalyst62,63,64. This absence of formate intermediates, combined with clear COOH* signals, suggests a COOH*-mediated pathway for CO generation, with the subsequent potential for CO to undergo further hydrogenation to CH47,61.
a–d Time-resolved in situ DRIFTS spectra acquired during sequential gas-exposure experiments at 500 oC for Cu-SAC (a), Ni-SAC (b), CuNi-DAC (c), and CuNi-NP (d). Each experiment consisted of CO2 adsorption (30 min), N2 purging (30 min), and subsequent injection of 10 % H2/Ar; spectra were recorded continuously during the H2 injection (2–30 min) to monitor the evolution of adsorbed reaction intermediates. e Intensity mapping of DRIFTS spectra for the 2200–1200 cm-1 region, comparing COOH* intermediate accumulation and CO adsorption band across the catalyst samples. f Isotopic TPSR-MS ion current (m/z) profiles of CuNi-DAC using three feeds, 12CO2-only, 12CO2/H2 (1:4), and 13CO2/H2 (1:4).
As shown in Fig. 5b, e, the strong adsorption of CO on Ni-SAC promoted substantial hydrogenation to CH4, as evidenced by adsorbed hydrocarbon intermediates bands (CH2 at 2933 cm−1 and CH3 at 2853 cm−1)61. Conversely, CuNi-DAC showed no detectable ν-CO* band in DRIFTS (Fig. 5c, e and Supplementary Fig. 45), signifying minimal CO adsorption, as evidenced by complementary CO-temperature programmed desorption (TPD) results (Supplementary Fig. 46). Then, CO molecules formed from CO2 hydrogenation rapidly desorbed into the gas phase without subsequent surface reaction. These highlight the unique N2Cu–N2–NiN2 dual-atom structure inherently promotes efficient CO2 activation followed by facile CO release (Supplementary Figs. 47 and 48). Consequently, CuNi-DAC consistently maintained nearly 100% CO selectivity while suppressing CH4 formation. In comparison, the nanoparticle based CuNi-NP showed intensified v-CO* peaks (2175–2105 cm−1) along with substantial COOH* accumulation (Fig. 5d), reflecting a higher CO affinity on metallic CuNi nanoparticles conducive to CH4 production7,18. Additionally, a sharp ν-OH peak indicates uniform hydrogen bonding interactions, suggesting that COOH* species are bound at specific adsorption sites (e.g., nanoparticle surfaces) where they interact consistently51,52,53,61 (Supplementary Fig. 49). Conversely, the atomically dispersed catalysts (CuNi-DAC and Cu-SAC) exhibited weak and broad v-OH peaks, indicating diverse hydrogen bonding interactions within the N-doped carbon lattice. In addition, DFT results support that COOH* binds much more strongly on CuNi-NP (multi-bond configuration) than on Ni-SAC (single-bond), rationalizing the abundant COOH* signatures on NP versus weak COOH* on Ni-SAC (Supplementary Fig. 50).
To further validate these findings under conditions closer to actual catalysis, we performed in situ DRIFTS under a continuous CO2:H2 (1:4) flow while progressively increasing temperature from 300 °C to 600 °C (Supplementary Fig. 51). The intensity of gaseous CO signal at 2178 and 2109 cm−1 increased significantly with elevated temperature for CuNi-DAC, whereas gaseous CH4 peaks were absent (Supplementary Fig. 51b). The concurrent increase of the COOH* band further confirms their critical role as precursors to CO formation. Ni-SAC featured gaseous CH4 at 3118 cm−1, along with CHO* at 1836 cm–1 and adsorbed CO at 2064–2050 cm–1, indicating sequential hydrogenation of CO to CH4 (Supplementary Fig. 51c)7,61,65.
Finally, we performed temperature programmed surface reaction mass spectrometry (TPSR-MS) under RWGS conditions using three feeds, 13CO2/H2 (1:4), 12CO2/H2 (1:4), and 12CO2-only. With 13CO2/H2, a dominant 13CO signal at m/z = 29 was observed, while 12CO (m/z = 28) and 13CH4 (m/z = 17) remain at baseline (Fig. 5f). With 12CO2/H2, only 12CO (m/z = 28) appeared, while 12CH4 (m/z = 16) and 13CO (m/z = 29) were absent. Under 12CO2-only flow (no H2), no CO (m/z = 28 or 29) was detected, and only 12CO2-related fragments were present. These isotope-tracing results demonstrate that the detected CO originates exclusively from the RWGS reaction, and not from the oxidation of the carbon support.
Collectively, these results align closely with the catalytic performance data (Fig. 4e and Supplementary Fig. 33), strongly emphasizing that the precisely tailored N2Cu–N2–NiN2 architecture within CuNi-DAC effectively directs CO2 hydrogenation selectively toward CO, thus enabling exceptional catalytic selectivity and stability in the RWGS reaction.
DFT calculations for theoretical understanding of CuNi-DAC
To clarify the origin of the superior selectivity and structural stability of CuNi-DAC in RWGS reaction, we performed DFT calculations for CuNi-DAC, Cu-SAC, and Ni-SAC, focusing on the projected density of states (PDOS) for 3 d orbitals of the metal centers (Fig. 6a). These models were constructed in accordance with EXAFS data. In CuNi-DAC, the dxy and dx2-y2 orbitals of both Cu and Ni exhibit distinct hybridization features at −4 and −1 eV regions, respectively, as depicted red and blue arrows in Fig. 6a. Besides, peak broadening of dxz and dyz orbitals is observed for both metals in CuNi-DAC, which implies enhanced d-electron delocalization compared to SACs62,63,64,66,67,68,69. This electronic delocalization suggests significant d-d orbital coupling between Cu and Ni atoms, which contributes to the thermodynamic stability of the dual-atom configuration. Furthermore, this coupling is reinforced by d-p orbital hybridization with coordinated nitrogen atoms in the N-C frameworks, further anchoring the metal atoms within the M-N-C structure. Consistent with these PDOS trends, Cu–Ni d–d hybridization in CuNi-DAC broadens and downshifts the Cu d-manifold relative to Cu-SAC, reducing the Cu dz2 (and out-of-plane dxz/dyz) density near EF and thereby weakening σ-donation/π-backdonation with CO23. Concomitantly, the Ni dyz spectral weight nearest EF is attenuated by adjacent Cu, further disfavoring CO* adsorption at Ni within the diatomic ensemble (Supplementary Fig. 52).
a Projected density of states (PDOS) for the 3 d orbitals of Cu-SAC, Ni-SAC, CuNi-DAC, the black dashed line indicates Fermi level. b–d Charge density difference for Cu-SAC (b), Ni-SAC (c) and CuNi-DAC (d). Red and blue regions represent charge accumulation and depletion respectively. Black, yellow, orange, and cyan spheres correspond to carbon, nitrogen, copper, and nickel atoms. e–g Relative free energy profiles for the COOH* hydrogenation step on Cu-SAC (e), Ni-SAC (f), and CuNi-DAC (g). h Relative free energy profile for the subsequent hydrogenation of adsorbed CO*. i Schematic illustration of RWGS reaction in CuNi-DAC.
We also performed charge difference analysis to evaluate the electron transfer properties at the CuN4 and NiN4 sites (Fig. 6b–d). The SAC models exhibit relatively symmetric charge distributions around the CuN4 and NiN4 sites, where the CuN4 sites tend to gain electrons (electron-rich, red area) as compared to NiN4 sites that tend to lose electrons (electron-deficient, blue area). On the other hand, the CuNi-DAC structure exhibits a pronounced charge polarization, with relatively electron-rich CuN4 sites and electron-deficient NiN4 sites. This electronic asymmetry facilitates CO2 activation at the Ni center and promotes CO2 reduction and CO desorption at the Cu site. To elucidate how the electronic structure affects RWGS selectivity, we investigated hydrogenation pathways (Fig. 6e–h). In both SAC and DAC models, COOH* hydrogenation step is highly exothermic and proceeds with negligible activation barrier, suggesting favorable CO generation initially (Supplementary Fig. 53). However, only the DAC model enables spontaneous CO desorption upon COOH* hydrogenation, whereas in the SAC models, CO* remains adsorbed (Supplementary Fig. 54). Additionally, we examined homo-metal diatomic ensembles, Cu–Cu (CuCu-DAC) and Ni–Ni (NiNi-DAC), for comparison with the hetero-metal Cu–Ni pair (CuNi-DAC). The COOH* → CO* step is most exergonic on CuNi-DAC (ΔG ~ −2.00 eV), but significantly less favorable on CuCu- and NiNi-DAC (−0.59 and −0.48 eV, respectively), while the computed kinetic barriers for this step are similarly low the three motifs (Supplementary Fig. 55). Thus, thermodynamics rather than activation barriers differentiate these sites at this stage. We further evaluated the subsequent hydrogenation of CO* to CHO*, a key step toward CH4 formation. While Ni-SAC (0.17 eV) and Cu-SAC (0.34 eV) showed relatively low uphill for CO hydrogenation, but CuNi-DAC exhibited a significantly higher reaction energy of 0.52 eV, effectively suppressing the methanation pathway (Fig. 6h). We also assessed a direct CO2-dissociation route on CuNi-DAC and found it energetically disfavored relative to the COOH* pathway (Supplementary Fig. 56). Collectively, these results reveal two key factors underlying the high RWGS selectivity of CuNi-DAC: (1) facilitated CO desorption immediately following COOH* formation, and (2) a substantial kinetic barrier that inhibits further hydrogenation to CH4 (Fig. 6i). These mechanistic insights are consistent with experimental observations and demonstrate how the engineered electronic structure of the CuNi dual-atom ensemble governs catalytic performance in thermocatalytic CO2 conversion.
Discussion
In summary, we have developed a strategic coordinated bottom-up approach to synthesize a gram-scale CuNi-DAC in which each Cu and Ni center is anchored as an N2Cu–N2–NiN2 ensemble within an N-doped carbon matrix. XAFS measurements, corroborated by DFT calculations, identify this atomic configuration and identify it as the thermodynamically preferred Cu–Ni arrangement. Cooperative electronic coupling between the neighbouring Cu and Ni sites broadens the d-orbitals and polarizes the local charge distribution, accelerating COOH* reduction, suppressing further hydrogenation, and robustly binding the metal atoms to the support. Consequently, CuNi-DAC attains near-equilibrium CO2 conversion with ~100% CO selectivity in the RWGS reaction and preserves its atomic dispersion up to 600 °C over repeated cycles, superior to those of the mixed single-atom or nanoparticle benchmarks. By clarifying how heteronuclear pairing simultaneously optimizes reaction energetics and structural stability, this study provides a blueprint for designing multi-metal atomic architectures for CO2 upgrading and other energy-intensive transformations, thereby bridging fundamental coordination chemistry and practical heterogeneous catalysis.
Methods
Chemicals and materials
Copper (II) chloride dihydrate (CuCl2·2H2O), nickel (II) chloride hexahydrate (NiCl2·6H2O), Hydrochloric acid (HCl), Cu phthalocyanine (C32H16N8Cu), Ni phthalocyanine (C32H16N8Ni), metal-free phthalocyanine (Pure-PC, C32H18N8), Copper(II) nitrate trihydrate (Cu(NO3)2·3H2O), Nickel(II) nitrate hexahydrate (Ni(NO3)2·6H2O), Sodium hydroxide solution (NaOH), N,N-Dimethylformamide (DMF, HCON(CH3)2), Colloidal silica (SiO2, 40 wt.% suspension in H2O, LUDOXRAS-40 colloidal silica) from Sigma-Aldrich and Fisher Scientific for all of the experiments.
Preparation of CuNi-DAC, Cu-SAC, and Ni-SAC
For the synthesis of CuNi-DAC (coordinated bottom-up), 5.0 g of Cu phthalocyanine (Cu-PC) and 2.5 g of Ni phthalocyanine (Ni-PC), 112.5 ml ethanol and 37.5 ml DMF, and 12.5 g of colloidal silica were stirred at 200 rpm for about 1 h, and then slowly evaporated at 110 °C. The evaporation process was carried out only until about 10% solvent remains. Then, pyrolysis was performed at 800 °C for 2 h at a ramping rate of 3.5 °C min−1 under pyrolysis conditions in the presence of N2. To remove the colloidal silica present in the sample after pyrolysis, the sample after pyrolysis is finely ground with a mortar and pestle and 300 ml of 1 M NaOH solution was added to the beaker and stirred at 200 rpm and 90 °C for 1 h to perform the etching process. The process was then repeated twice by waiting for 30 min and decanting only the upper layer of the stirred solution to allow the residual SiO2 with low density to float in the NaOH. Sample washing was then performed using a centrifuge. Centrifugation (3350 × g for 15 min) and washing were performed once with 1.0 M NaOH and three times with D.I. water. Finally, 7–8 g of CuNi-DAC was obtained by drying step in a 110 °C oven for 12 h to remove residual moisture. For the scaled-up synthesis of CuNi-DAC at 15 g, the precursor input amounts in the coordinated bottom-up process were increased by 3.0 times, using 15.0 g of Cu-PC, 7.5 g of Ni-PC, 337.5 mL of ethanol, 112.5 mL of DMF, and 37.5 g of colloidal silica, while all other synthesis conditions remained unchanged. As a result of the scale-up synthesis, 13-15 g of CuNi-DAC was successfully obtained.
Preparation of CuNi-SA/NP, Cu-SA/NP, Ni-SA/NP, and NC
For the synthesis of CuNi-SA/NP, Cu-SA/NP and Ni-SA/NP (bottom-up samples), and NC (pyrolysis of Pure-PC), metal-free phthalocyanine (Pure-PC; C32H18N8, 514.55 g mol−1) was used as the carbon/nitrogen scaffold. To account for the absence of the central metal in Pure-PC relative to Cu-phthalocyanine (Cu-PC, 576.07 g mol−1) and Ni-phthalocyanine (Ni-PC, 571.22 g mol−1), the required copper and/or nickel were supplied as metal nitrate hydrates, Cu(NO3)2·3H2O and Ni(NO3)2·6H2O, so as to match the intended metal loadings. Pure-PC, the corresponding metal nitrate(s), ethanol (112.5 mL), DMF (37.5 mL), and colloidal silica (12.5 g) were combined, stirred at 200 rpm for 1 h, and slowly evaporated at 110 °C until ~10% of the solvent remained. The resulting gel was pyrolyzed at 800 °C for 2 h (ramp 3.5 °C min−1) under N2. After cooling, the solid was finely ground and etched in 300 mL of 1.0 M NaOH at 90 °C (200 rpm, 1 h); the suspension was allowed to stand for 30 min and the supernatant was decanted, and this etching step was repeated twice. The product was then washed by centrifugation at 3350 × g for 15 min (once with 1.0 M NaOH and three times with deionized water) and dried at 110 °C for 12 h. Using this single protocol, CuNi-SA/NP was obtained from Pure-PC (6.718 g), Cu(NO3)2·3H2O (2.096 g), and Ni(NO3)2·6H2O (1.272 g); Cu-SA/NP from Pure-PC (6.252 g) and Cu(NO3)2·3H2O (2.930 g); and Ni-SA/NP from Pure-PC (6.305 g) and Ni(NO3)2·6H2O (1.187 g). For the NC, Pure-PC (6.00 g) was directly pyrolyzed at 800 °C for 2 h (3.5 °C min-1) under N2.
Preparation of CuNi-NP
For the synthesis of CuNi-NP (top-down), 5 g of graphite was added to 30% concentrated sulfuric acid solution (50 ml) and stirred (200 rpm) at 35 oC for 24 h, and another 20 ml of 30% hydrogen peroxide solution was added and stirred (200 rpm) for 1 h. Then sonicate for 2 h to exfoliate the graphite layer. Washed the solution three times with a hydrochloric acid solution (1.0 M HCl) and deionized (D.I.) water using a centrifuge (3350 × g for 15 min), and then dried it to obtain exfoliated graphite. Combined 3 g of exfoliated graphite obtained from the previous steps with 2 g of urea (nitrogen dopant), 0.5 g of copper (II) chloride dihydrate (CuCl2·2H2O), 0.25 g of nickel (II) chloride hexahydrate (NiCl2·6H2O), and 300 ml of D.I. water in an autoclave. Maintain the mixture at 180 oC for 12 h. After hydrotreatment, the mixture evaporated to 110 oC to remove the solvent, and proceeded with the pyrolysis in an inert environment (nitrogen atmosphere) of the insulation system. The pyrolysis conditions involved heating at a rate of 3 oC min−1 until reaching 800 oC, for 2 h, and then cooling down to room temperature (25 oC) at the same rate of 3 oC min−1. The powder was washed three times with a 1.0 M hydrochloric acid (HCl) solution using a centrifuge at 3350 × g for 15 min. Subsequently, the washed powder was purified in a vacuum oven at 60 oC for 12 h, resulting in the final product, CuNi-NP.
Material characterization
X-ray photoelectron spectroscopy (XPS) was performed using a standard Al K source (K-ALPHA+) by AXIS SUPRA (NFEC-2020-09-265067). The binding energy (B.E.) was measured in the basis of carbon contamination utilizing C 1 s peak centered at 284.8 eV as reference. To estimate the atomic concentration ratios, a standard procedure was followed, which involved utilizing atomic sensitivity factors and subtracting the Shirley background. Spectral lines were then fitted using a symmetric pseudo-Voigt function referred to as GL30 with the CasaXPS software. XPS deconvolution was confirmed whether the catalyst before and after the reaction was structurally changed. Analysis was performed through changes in the metal-state of the active metal, changes in atomic concentration, and differences in C 1 s, N 1 s, Cu 2p and Ni 2p regions. Inductively coupled plasma atomic emission spectroscopy (ICP-AES) was performed for measuring the concentration of inorganic elements in the sample by detecting an intrinsic wavelength from the energy emitted by electrons using atomic emission spectroscopy by Thermo Fisher/iCAP-Pro XP. X-ray fluorescence spectroscopy (XRF) was performed using a Mechelle 5000 Spectrograph instrument with simultaneous recording of a wide wavelength range (200–975 nm). XRF analysis was performed for quantitative analysis of the ratio of heavy to light active metal atoms. Quantitative analysis of the samples was performed by determining the mass ratio of the elements according to the exact mass of 0.5 g of the substance and the mass percentage according to the fluorescence spectrum K-alpha of each element. Raman spectra analysis was performed by Nanophoton-Ramanforce Raman spectrometer under the following conditions: Laser wavelength: 532 nm, Detector Loaded: cooled CCD (1340 × 400 pixels), focal length: 550 mm, substrate: Glass. N2 physisorption analysis was performed at 77 K using a Tristar instrument after sample pretreatment at 120 °C in a vacuum for 24 h. Surface areas were calculated using the Brunauer-Emmett-Teller (BET) and pore volumes were calculated using the Barrett-Joyner-Halenda (BJH) methods. Electron energy loss spectroscopy (EELS) was performed by same Titan-TEM 50–300 microscopy analyzer and the data of distance range-spectrum was gained by Gatan 3.0.0 and GMS software provided from AMETEK.
X-ray absorption fine structure (XAFS)
Energy shifts in the extended X-ray absorption fine structure (EXAFS) spectra were calculated by summing the contributions from all distinct coordination sites of each transition metal (Cu and Ni K-edges). Normalization and background subtraction of calculated EXAFS were carried out using ATHENA. Wavelet transform (WT) contour plot was calculated from the JWT-EXAFS software and Origin matrix mapping software. XAFS data were collected at the 6D beamline of Pohang Accelerator Laboratory PLS-II (UNIST-PAL CSS) and 10 C beamline in Republic of Korea. EXAFS data processing was carried out by removing the post-edge background and normalizing the spectra to the edge-jump. The Cu and Ni K-edge χ(k) data, weighted by k2- and k3, were Fourier-transformed (FT) from k-space to R-space using a Hanning window function to isolate contributions from distinct coordination shells. Quantitative structural parameters were extracted by fitting the FT(k3χ(k)) spectra in R-space using the ARTEMIS module within the IFEFFIT software package. The theoretical scattering amplitudes and phase shifts for each fitting path were computed using FEFF calculations.
Ultra-high-resolution STEM/EDS
Scanning transmission electron microscopy (STEM) and energy-dispersive X-ray spectrometry (EDS) were performed using a FEI Titan Cubed G2 (Spectra Ultra-Thermo Fisher) 60–300 from KARA (KAIST Analysis center Research Advancement). This ultra-high-resolution microscope, operated at 300 kV, features double Cs correctors (probe and image), a monochromator, and an ultra-stable high brightness electron gun (X-FEG). EDS analysis was conducted using multi-channel detectors.
In situ diffuse reflection infrared Fourier transform spectroscopy
In situ diffuse reflection infrared Fourier transform spectroscopy (DRIFTS) spectra were recorded using a Fourier-transform (FT) infrared FTIR spectrometer (Nicolet iS10, Thermo Fisher Scientific) equipped with a commercial DRIFTS cell (PIKE, Diffuse IR), ZnS window for considering H2O and an MCT detector cooled by liquid N2. The data were collected in the Kubelka-Munk format with 32 scans at a resolution of 4 cm-1. Sequential in situ DRIFTS measurements were carried out at 500 °C with stepwise gas exposure (10% CO2/Ar → N2 → 10% H2/Ar). After 30 min of CO2 adsorption and purging, DRIFTS spectra were collected during 10% H2/Ar flow from 2 to 30 min to monitor surface species evolution. In situ DRIFTS via feed gas, under reaction-relevant conditions was conducted using a mixed feed gas (CO2:H2 = 1:4 v/v), following the same pretreatment protocol applied during catalytic performance evaluation. The measurements were performed over a temperature range of 300–600 °C, with a heating rate of 10 °C min−1. At each target temperature, spectra were collected over a 30 min interval to monitor surface species evolution.
Temperature programed desorption analysis
Temperature-programmed desorption (TPD) experiments were conducted using an Autochem II 2920 system (Micromeritics) to investigate the thermochemical adsorption/desorption characteristics of CO and CO2 gases on M-N-C samples. For both CO2- and CO-TPD analyses, 0.15 g of each sample was first pretreated under a He atmosphere at 120 °C for 1 h, followed by reduction using 10% H2/He gas while ramping to 700 °C, and maintained at that temperature for 1 h. Afterward, the samples were cooled to 50 °C and exposed to either CO2 or CO gas for 1 h. Subsequently, He purging was performed at the same temperature for 1 h, followed by a stabilization step at 100 °C for 10 min. Finally, the samples were heated to 700 °C under He flow, and the desorbed CO or CO2 signals were analyzed. All heating and cooling steps were conducted at a constant ramping rate of 10 °C min-1.
Evaluation of thermo-catalytic RWGS performance
To measure the CO2 conversion and CO/CH4 selectivity of the catalysts for RWGS, a temperature-dependent performance evaluation of the catalysts was performed using a continuous fixed bed reactor. 0.5 g of the as-prepared catalyst was loaded into a half-inch quartz reactor, flowing at a flow rate of 10 standard cc per minute (SCCM) of hydrogen (H2, 99.9999%) and 90 SCCM of nitrogen (N2, 99.9999%), and pre-treatment was carried out under normal pressure conditions, ramping at 10 °C min-1 and maintaining the temperature at 300 °C for 60 min. Subsequently, at the same temperature, nitrogen (N2, 99.9999%) was flowed at a flow rate of 100 SCCM for 60 min to prevent the pre-treatment gas from intervening in the catalytic reaction before the reaction gas injection. At WHSV of 6000 cc g−1 h−1, hydrogen (H2, 99.9999%, 32 SCCM), carbon dioxide (CO2, 99.9999%, 8 SCCM), and nitrogen (N2, 99.9999%, 10 SCCM) were injected into the reactor as reaction gases. The reaction temperature was varied at 50 °C intervals with a ramp-up rate of 10 °C min-1 in the temperature range from 300 °C to 600 °C (1-cycle), and the performance was measured at each reaction temperature for 100 min. Then, the reaction temperature was varied at 50 oC intervals by cooling to 300 oC, and the catalyst performance was measured by maintaining each reaction temperature for 100 min. The composition of the gaseous products after the reaction was analyzed using gas chromatography (GC) equipped with a packed column connected to the reaction unit. The 3-cycle hysteresis performance was performed harsher than the 1-cycle performance, that total reaction time (100 h) was varied for 3-cycle under the conditions of 0.25 g of catalyst and twice high space velocity at WHSV 12,000 cc g−1 h−1. The reaction temperature was increased or decreased by every 50 oC in the range of 300–600 oC, and reaction was carried out for a total of 37 steps at each reaction temperature for 160 min. Data on CO2 conversion and CO selectivity in the 1-cycle and 3-cycle hysteresis evaluations were assessed through the last reaction point of the 100 min performed at each reaction temperature. Total reaction evaluation of 3-cycle performance was conducted for 100 h.
RWGS reaction performance evaluation metrics
The CO2 conversion rate (Eq. 1), CO and CH4 selectivity (Eqs. 2 and 3), and the yield per unit catalyst mass of CO (STYCO) (Eq. 4) were calculated as shown in the equation below.
13CO2 isotopic tracing temperature programmed surface reaction-mass spectrometry (TPSR-MS)
Isotopic TPSR-MS to verify RWGS-derived CO and 100% CO selectivity over CuNi-DAC. To confirm that CO originates from gas-phase RWGS chemistry rather than from the carbon matrix of the CuNi-DAC catalyst, isotopic temperature-programmed surface reaction–mass spectrometry (TPSR–MS) was conducted at atmospheric pressure. A 0.25 g charge of CuNi-DAC was loaded in a half-inch quartz fixed-bed reactor held at 600 °C. Three feed regimens were employed: (i) 12CO2-only: 12CO2 (99.9999%, 40 SCCM); (ii) RWGS (12CO2/H2): 12CO2 (99.9999%, 10SCCM, 10 SCCM) with H2 (99.9999%, 40 SCCM) (12CO2:H2 = 1:4); and (iii) RWGS (13CO2/H2): 13CO2 (99%; <2% 18O, 10 SCCM) with H2, (99.9999%, 40 SCCM) (13CO2:H2 = 1:4). Prior to reaction, the line and bed were stabilized by flowing Ar for 4 h at room temperature, followed by heating at 10 °C min−1 to 600 °C, a 2 h H2 pretreatment, and an additional 4 h Ar hold at 600 °C to ensure ion-current stability. Product ion-currents (m/z) were then recorded for 12 h under, sequentially, the 12CO2-only condition and the two RWGS conditions (12CO2/H2 and 13CO2/H2). TPSR–MS measurements were performed on an RT-STA2 quadrupole MS (REALTEK) coupled to a BELCAT II unit (Microtrac-BEL Corp.).
Computational details and density functional theory (DFT) calculation
Spin-polarized density functional theory calculations were conducted by using Vienna ab-initio simulation package (VASP) version 5.4.4 code62,66,67. RPBE parameterization of GGA exchange-correlation functional was adopted for whole calculations with 400 eV plane wave energy cutoff. To obtain binding energies between combinations of Cu-PC and Ni-PC, we constructed cubic cell with 30 Å lattice length to avoid the unphysical interaction between periodic images. All structures were fully optimized in 1 × 1 × 1 k-mesh until all atomic forces are less than 0.01 eV Å−1 with van der Waals correction by Tkatchenko-Scheffler method68. We calculated the binding energy of each structures by the Eq. 5, where Ecomb, ECu-PC/Ni-PC and nCu-Pc/Ni-Pc denotes total energy of given PC molecules, total energy of isolated Cu-PC/Ni-PC molecule, and number of Cu-PC/Ni-PC molecule in given PC molecules, respectively.
To model appropriate SAC and DAC configurations, we adopted graphene model with lattice parameter a = 17.22 Å, b = 12.78 Å for 2D surface structure which is extended along a- and b-directions. Also, vacuum region greater than 20 Å is considered to avoid the unphysical interaction between periodic images. Whole structures were fully optimized in 2 × 2 × 1 k-mesh until all atomic forces are less than 0.01 eV Å−1. For density of states and charge density difference calculations, 8 × 8 × 1 k-mesh was adopted. The charge density difference plots were calculated by the Eq. 6, where ρtot, ρG, and ρCu/Ni corresponds to charge density of total system, N-doped graphene frameworks, and Cu/Ni atoms, respectively. We note that charge density of Cu and Ni atoms was calculated individually when we obtain charge density difference of DAC configuration.
We calculated the formation energy of each structures by Eq. 7 and Eq. 8, where Etot., EGN, and nCu/Ni denote total energy of given system, N-doped carbon framework, and number of 3 d metal elements, respectively. The chemical potential of 3 d metal elements, μCu/Ni, were calculated by dividing number of atoms in Cu/Ni bulk unit cell with total energy of Cu/Ni bulk. For chemical potential of oxygen, μO, oxygen molecule was considered and divided by number of atoms in molecule with total energy of oxygen molecule. Thermochemical properties to obtain μCu/Ni and μO in 293 K, 773 K were taken from NIST Chemistry Webbook database69. To calculate transition state energy, Nudged Elastic Band (NEB) method was employed.
Data availability
The authors declare that all data supporting the findings of this study are available in the paper and Supplementary Information. All data are available from the corresponding authors upon request.
References
Xin, H. et al. Reverse water gas-shift reaction product driven dynamic activation of molybdenum nitride catalyst surface. Nat. Commun. 15, 3100 (2024).
Kattel, S., Liu, P. & Chen, J. G. Tuning selectivity of CO2 hydrogenation reactions at the metal/oxide interface. J. Am. Chem. Soc. 139, 9739–9754 (2017).
Pahija, E. et al. Experimental and computational synergistic design of Cu and Fe catalysts for the reverse water−gas shift: a review. ACS Catal. 12, 6887–6905 (2022).
Wang, H. et al. Synergistic interactions of neighboring platinum and iron atoms enhance reverse water−gas shift reaction performance. J. Am. Chem. Soc. 145, 2264–2270 (2023).
Liu, H.-X. et al. Partially sintered copper‒ceria as excellent catalyst for the high-temperature reverse water gas shift reaction. Nat. Commun. 13, 867 (2022).
Wen, C. F. et al. Nitrogen-stabilized low-valent Ni motifs for efficient CO2 electrocatalysis. ACS Catal. 10, 1086–1093 (2020).
Li, Y. et al. Single-atom Co-N-C catalysts for high-efficiency reverse water-gas shift reaction. Appl. Catal. B Environ. 324, 122298 (2023).
Visser, N. L. et al. Direct observation of Ni nanoparticle growth in carbon-supported nickel under carbon dioxide hydrogenation atmosphere. ACS Nano 17, 14963–14973 (2023).
Wang, X., Shi, H. & Szanyi, J. Controlling selectivities in CO2 reduction through mechanistic understanding. Nat. Commun. 8, 513 (2017).
Jiang, Y. et al. Single-atom molybdenum-N3 sites for selective hydrogenation of CO2 to CO. Angew. Chem. 134, e202203836 (2022).
Hu, X. et al. Hybrid catalyst coupling Zn single atoms and CuNx clusters for synergetic catalytic reduction of CO2. Adv. Funct. Mater. 33, 2214215 (2023).
Park, D. et al. Insights into the synergy effect in dual single-atom catalysts on defective CeO2 under CO2 hydrogenation. Appl. Catal. B Environ. Energy 365, 124987 (2025).
Rabee, A. I. M. et al. CeO2 supported single-atom Cu catalysts modified with Fe for RWGS reaction: deciphering the role of fe in the reaction mechanism by in situ/Operando spectroscopic techniques. ACS Catal. 14, 10913–10927 (2024).
Shan, J. et al. Metal-metal interactions in correlated single-atom catalysts. Sci. Adv. 8, eabo0762 (2022).
Yang, J. et al. Dynamic behavior of single-atom catalysts in electrocatalysis: identification of Cu‑N3 as an active site for the oxygen reduction reaction. J. Am. Chem. Soc. 143, 14530–14539 (2021).
He, T. et al. Atomically dispersed heteronuclear dual-atom catalysts: a new rising star in atomic catalysis. Small 18, 2106091 (2022).
Li, R. & Wang, D. Superiority of dual-atom catalysts in electrocatalysis: one step further than single-atom catalysts. Adv. Energy Mater. 12, 2103564 (2022).
Hai, X. et al. Scalable two-step annealing method for preparing ultra-high-density single-atom catalyst libraries. Nat. Nanotechnol. 17, 174–181 (2022).
Li, Y. et al. Synergistic effect of atomically dispersed Ni–Zn pair sites for enhanced CO2 electroreduction. Adv. Mater. 33, 2102212 (2021).
He, Q. et al. Electrochemical conversion of CO2 to syngas with controllable CO/H2 ratios over Co and Ni single-atom catalysts. Angew. Chem. Int. Ed. 59, 3033–3037 (2020).
Cheng, H. et al. Atomically dispersed Ni/Cu dual sites for boosting the CO2 reduction reaction. ACS Catal. 11, 12673–12681 (2021).
Yao, D. et al. Inter-metal interaction with a threshold effect in NiCu dual-atom catalysts for CO2 electroreduction. Adv. Mater. 35, 2209386 (2023).
Zeng, Z. et al. Orbital coupling of hetero-diatomic nickel-iron site for bifunctional electrocatalysis of CO2 reduction and oxygen evolution. Nat. Commun. 12, 4088 (2021).
Ju, W. et al. Understanding activity and selectivity of metal-nitrogen-doped carbon catalysts for electrochemical reduction of CO2. Nat. Commun. 8, 944 (2017).
Jiang, Z. et al. Interfacial assembly of binary atomic metal-Nx sites for high-performance energy devices. Nat. Commun. 14, 1822 (2023).
Yao, Z. et al. Hydrogen radical-boosted electrocatalytic CO2 reduction using Ni-partnered heteroatomic pairs. Nat. Commun. 15, 9881 (2024).
Liang, X.-M. et al. Controlled synthesis of a Ni2 dual-atom catalyst for synergistic CO2 electroreduction. Appl. Catal. B Environ. 322, 122073 (2023).
Jiao, J. et al. Constructing asymmetric double-atomic sites for synergistic catalysis of electrochemical CO2 reduction. Nat. Commun. 14, 616 (2023).
Xie, W. et al. NiSn atomic pair on an integrated electrode for synergistic electrocatalytic CO2 reduction. Angew. Chem. Int. Ed. 60, 7382–7388 (2021).
Tang, B. et al. A Janus dual-atom catalyst for electrocatalytic oxygen reduction and evolution. Nat. Synth. 3, 878–890 (2024).
Hursán, D. et al. CO2 conversion on N‑doped carbon catalysts via thermo- and electrocatalysis: role of C−NOx Moieties. ACS Catal. 12, 10127–10140 (2022).
Wang, Y., Chen, J., Chen, L. & Li, Y. Breaking the linear scaling relationship of the reverse water−gas−shift reaction via construction of dual-atom Pt−Ni pairs. ACS Catal. 13, 3735–3742 (2023).
Koshy, D. M. et al. Bridging thermal catalysis and electrocatalysis: catalyzing CO2 conversion with carbon-based materials. Angew. Chem. Int. Ed. 60, 17472–17480 (2021).
Jiang, S. et al. Visualization of the distance-dependent synergistic interaction in heterogeneous dual-site catalysis. J. Am. Chem. Soc. 146, 29084–29093 (2024).
Zhang, L. et al. Atomic layer deposited Pt-Ru dual-metal dimers and identifying their active sites for hydrogen evolution reaction. Nat. Commun. 10, 4936 (2019).
Hu, Y. et al. Entropy-engineered middle-in synthesis of dual single-atom compounds for nitrate reduction reaction. ACS Nano 18, 23168–23180 (2024).
Chen, Y. et al. Inter-metal interaction of dual-atom catalysts in heterogeneous catalysis. Angew. Chem. Int. Ed. 62, e202306469 (2023).
Yan, H. et al. Bottom-up precise synthesis of stable platinum dimers on graphene. Nat. Commun. 8, 1070 (2017).
Liang, X., Fu, N., Yao, S., Li, Z. & Li, Y. The progress and outlook of metal single-atom-site catalysis. J. Am. Chem. Soc. 144, 18155–18174 (2022).
Jia, C. et al. Challenges and opportunities for single-atom electrocatalysts: from lab-scale research to potential industry-level applications. Adv. Mater. 36, 2404659 (2024).
Chen, C. et al. Asymmetrically coordinated Cu dual-atom-sites enables selective CO2 electroreduction to ethanol. Adv. Mater. 36, 2409797 (2024).
Liu, W. et al. A durable nickel single-atom catalyst for hydrogenation reactions and cellulose valorization under harsh conditions. Angew. Chem. Int. Ed. 57, 7071–7075 (2018).
Zhu, Y. et al. A cocoon silk chemistry strategy to ultrathin N-doped carbon nanosheet with metal single-site catalysts. Nat. Commun. 9, 3861 (2018).
Rao, P. et al. Precise synthesis of dual-single-atom electrocatalysts through pre-coordination-directed in situ confinement for CO2 reduction. Angew. Chem. Int. Ed. 64, e202415223 (2025).
Hummers, W. S. & Offeman, R. E. Preparation of graphitic oxide. J. Am. Chem. Soc. 80, 1339 (1958).
Grosvenor, A. P., Biesinger, M. C., Smart, R. St.C. & Mclntyre, N. S. New interpretations of XPS spectra of nickel metal and oxides. Surf. Sci. 600, 1771–1779 (2006).
Zhao, X., Fang, R., Wang, F., Kong, X. & Li, Y. Dual-metal single atoms with dual coordination for the domino synthesis of natural flavones. J. Am. Chem. Soc. Au. 3, 185–194 (2023).
Li, Z. et al. Geometric and electronic engineering of atomically dispersed copper-cobalt diatomic sites for synergistic promotion of bifunctional oxygen electrocatalysis in zinc–air batteries. Adv. Mater. 35, 2300905 (2023).
Wang, X. et al. Precisely designing asymmetrical selenium-based dual-atom sites for efficient oxygen reduction. Nat. Commun. 16, 470 (2025).
Wang, S. et al. Manipulating C-C coupling pathway in electrochemical CO2 reduction for selective ethylene and ethanol production over single-atom alloy catalyst. Nat. Commun. 15, 10247 (2024).
Winter, M. et al. Defining reactivity of solid sorbents: what is the most appropriate metric? Chem. Mater. 21, 2367–2374 (2009).
Bown, R. M., Joyce, M., Zhang, Q., Reina, T. R. & Duyar, M. S. Identifying commercial opportunities for the reverse water gas shift reaction. Energy Technol. 9, 2100554 (2021).
Bobadilla, L. F., Santos, J. L., Ivanova, S., Odriozola, J. A. & Urakawa, A. Unravelling the role of oxygen vacancies in the mechanism of the reverse water−gas shift reaction by operando DRIFTS and ultraviolet−visible spectroscopy. ACS Catal. 8, 7455–7467 (2018).
Gandara-Loe, J. et al. Design of full-temperature-range RWGS catalysts: impact of alkali promoters on Ni/CeO2. Energy Fuels 36, 6362–6373 (2022).
Wang, C. et al. Product selectivity controlled by nanoporous environments in zeolite crystals enveloping rhodium nanoparticle catalysts for CO2 hydrogenation. J. Am. Chem. Soc. 141, 8482–8488 (2019).
Zhang, Q., Bown, M., Pastor-Pérez, L., Duyar, M. S. & Reina, T. R. CO2 conversion via reverse water gas shift reaction using fully selective Mo−P multicomponent catalysts. Ind. Eng. Chem. Res. 61, 12857–12865 (2022).
Zhao, X. et al. Monodisperse metal–organic framework nanospheres with encapsulated core–shell nanoparticles Pt/Au@Pd@{Co2(oba)4(3-bpdh)2}4H2O for the highly selective conversion of CO2 to CO. ACS Appl. Mater. Interfaces 10, 15096–15103 (2018).
Yan, B. et al. Tuning CO2 hydrogenation selectivity via metal-oxide interfacial sites. J. Catal. 374, 60–71 (2019).
Jiang, Y. et al. Tuning the selectivity of catalytic carbon dioxide hydrogenation over iridium/cerium oxide catalysts with a strong metal–support interaction. Angew. Chem. 129, 10901–10905 (2017).
Wang, L., Liu, H., Chen, Y. & Yang, S. Reverse water–gas shift reaction over co-precipitated Co–CeO2 catalysts: Effect of Co content on selectivity and carbon formation. Int. J. Hydrog. Energy 42, 3682–3689 (2017).
Lorber, K. et al. Non-oxidative calcination enhances the methane dry reforming performance of Ni/CeO2−x catalysts under thermal and photo-thermal conditions. J. Mater. Chem. A. 12, 19910 (2024).
Hammer, B. & Nørskov, J. K. Theoretical surface science and catalysis-calculations and concepts. Adv. Catal. 45, 71–129 (2000).
Choi, S.-J. et al. Catalytic behavior of metal catalysts in high-temperature RWGS reaction: In-situ FT-IR experiments and first-principles calculations. Sci. Rep. 7, 41207 (2017).
Zhao, Z. et al. Atomically dispersed Pt/CeO2 catalyst with superior CO selectivity in reverse water gas shift reaction. Appl. Catal. B Environ. 291, 120101 (2021).
Zhu, S., Jiang, B., Cai, W.-B. & Shao, M. Direct observation on reaction intermediates and the role of bicarbonate anions in CO2 electrochemical reduction reaction on Cu surfaces. J. Am. Chem. Soc. 139, 15664–15667 (2017).
Kresse, G. & Furthmuller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Hammer, B., Hansen, L. B. & Nørskov, J. K. Improved adsorption energetics within density-functional theory using revised Perdew-Burke-Ernzerhof functionals. Phys. Rev. B. 59, 7413 (1999).
Tkatchenko, A. & Scheffler, M. Accurate molecular van der Waals interactions from ground-state electron density and free-atom reference data. Phys. Rev. Lett. 102, 073005 (2009).
Linstrom, P. J. & Mallard, W. G. The NIST Chemistry WebBook: a chemical data resource on the internet. J. Chem. Eng. Data. 46, 1059–1063 (2001).
Acknowledgements
This work was supported by the Institutional Research Program of KRICT (KK2513-30 and KK2513-10, H.-T.K.), the National Research Foundation (NRF) of Korea (Korea government MSIT program, No. RS-2023-00282392, RS-2023-00259994, and the STEAM BRIGDGE R&D program, 2022M3C1C3095083, H.-T.K., Y.-J.K., and S.-J.K.). Experiments at PLS-II were supported in part by MSIT and POSTECH. Computation resources were supported by KISTI (Grants No. KSC-2024-CRE-0201 and No. KSC-2024-CRE-0482, G.L.). We also appreciate KARA (KAIST Analysis center for Research Advancement) for characterization equipment for XPS and Ultra-high-resolution STEM/EDS.
Author information
Authors and Affiliations
Contributions
K.-M.K. and J.M.: These authors contributed equally to this work. H.-T.K., Y.J.K., S.-J.K. and G.L. designed and supervised this project. H.-T.K., Y.J.K., S.-J.K., G.L., K.-M.K. and J.M. wrote manuscript. K.-M.K. carried out all experiments. J.M. performed the DFT calculation. K.-M.K., G.-N.Y. and Y.-W.Y. contributed to the XAFS simulation. K.-M.K., J.H.P. and J.H.L. performed the synthesis and characterization. K.-M.K. carried out the thermochemical measurements. K.-M.K., J.S., H.S., J.K., S.K. and T.-H.K. contributed to the XANES and XAFS measurements. K.-M.K., S.H.K. and Y.K.K. performed the electron microscopy analyses. K.-M.K. and Y.-S.B. carried out the in situ DRIFT analysis. All of the authors discussed the results and assisted during manuscript preparation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Shengjie Xia and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kim, KM., Mun, J., Yun, GN. et al. Rational synthesis of dual-atom catalysts for optimized thermochemical CO2 reduction. Nat Commun 16, 11617 (2025). https://doi.org/10.1038/s41467-025-66608-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-66608-9