Abstract
On-surface synthesis recently enabled access to generate a series of cyclo[n]carbons (Cn, up to C26) via delicately designed precursors. However, for larger cyclocarbons, synthesis of the corresponding precursors is being challenged. Here, we report another strategy for synthesizing larger cyclocarbons via the coupling and ring-opening reactions from smaller ones. We chose C10 as the precursor, and induce the coupling and ring-opening reactions via tip manipulation to successfully generate C20 and C30. Bond-resolved atomic force microscopy imaging unambiguously reveals the polyynic structure of C30, aligning with theoretical prediction. This approach establishes a new pathway to access larger cyclocarbons, and potentially enables controlled polymerization of cyclocarbons.
Similar content being viewed by others
Introduction
Cyclo[n]carbon, a family of carbon allotropes, has attracted substantial experimental and theoretical interest for decades owing to its remarkable properties1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17. However, the high reactivity of these molecules presents significant challenges for their synthesis. On-surface synthesis18,19,20,21,22 has recently emerged as a feasible approach for generating such reactive structures. Through tip manipulation23,24,25,26,27 of delicately designed precursors on insulating NaCl surfaces, several cyclocarbons (e.g., C628, C1029, C1230, C1331, C1429, C1632, C1833,34, C2030) have been synthesized.
Depending on the type of precursor molecules in these works, two strategies for synthesizing cyclocarbons can be classified: one is designing macrocyclic precursors and directly forming cyclocarbons by removing halogen atoms or CO molecules (e.g., C16, C18); the other is synthesizing fully halogenated precursors and then using dehalogenation and retro-Bergman ring-opening reactions to obtain cyclocarbons (C10, C12, C13, C14, C20).
While for n > 20, the synthesis of both types of precursors in solution becomes very challenging, thus limiting access to larger cyclocarbons32. Calculations predict that they may adopt a bicyclic or polycyclic configuration in the ground state rather than a monocyclic one35,36. Moreover, their aromaticity is expected to gradually diminish with increasing ring size11. Till now, the largest one generated on the surface is C26, which was obtained by tip-induced direct fusion from two C13Cl10 precursors31.
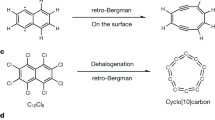
Here, we report another strategy for the synthesis of cyclocarbons, specifically targeting larger rings, i.e., via the coupling and ring-opening reactions from the smaller cyclocarbon precursor37,38. Using this strategy, we successfully generated C20 and C30 from C10. Individual C10 molecules were first generated on the NaCl surface using an established method29 (Fig. 1a). Subsequently, tip manipulation induced coupling of C10, forming dimers or trimers, followed by ring-opening reactions to yield C20 or C30 (Fig. 1b, c). Bond-resolved atomic force microscopy (AFM) imaging unambiguously resolved the polyynic structures in accordance with theoretical predictions. This work establishes a pathway for synthesizing larger cyclocarbons and inspires further studies for cyclocarbon polymerization4,39.
a The synthesis of cyclo[10]carbon via tip-induced dehalogenation and retro-Bergman reactions29. b Schematic illustration of the generation of cyclo[20]carbon via the tip-induced coupling and ring-opening reactions of two cyclo[10]carbon. c Schematic illustration of the generation of cyclo[30]carbon via the tip-induced coupling and ring-opening reactions of three cyclo[10]carbon.
Results and discussion
To generate cyclo[10]carbon precursors, we used the reported strategy via the scanning tunneling microscopy (STM) tip-induced dehalogenation and retro-Bergman reactions of a fully halogenated precursor, C10Cl8. All experiments were conducted at 2 ML NaCl/Au(111) surface at 4.7 K, with a precursor coverage of ~0.3 monolayer (ML).
Figure 2a shows two individual cyclo[10]carbons generated on the surface. To induce their coupling, typical voltage pulses (~4 V) were applied on the NaCl surface several nanometers from C10 molecules with a current of hundreds of fA. This process could couple two C10 molecules, yielding two typical intermediates (Int 1 and Int 2; Fig. 2b, c). For Int 1, AFM images (Fig. 2b II and b III) clearly show that two C10 molecules were bridged by a single C–C bond, forming a double-ring structure, in consistence with the simulated AFM image (Fig. 2b IV). For Int 2, two C10 were connected via a four-membered ring, as shown in Fig. 2c.
a I, a II AFM and Laplace-filtered AFM images of two individual cyclo[10]carbons. b I–b IV, c I–c IV, d I–d IV, e I–e IV Molecules, AFM images, Laplace-filtered AFM images, and simulated AFM images of intermediate (Int) 1, 2, 3, and final product cyclo[20]carbon. AFM tip offsets Δz: +0.4 Å, +0.4 Å, 0 Å, +0.3 Å, and +0.3 Å for a I, b II–e II. Reference set points of Δz: I = 0.5 pA, V = 0.3 V for all AFM images. The scale bar in (b II) applies to (b–e) images.
Subsequent voltage pulses can induce transformations from Int 1 or Int 2 to Int 3, a 12–10-membered ring structure (Fig. 2d), and generate the final product, cyclo[20]carbon, via ring-opening reaction (Fig. 2e). AFM imaging and simulated AFM images revealed ten triple bonds of C20, consistent with the literature30. Thus, we have achieved the formation of a C20 directly from the smaller precursor C10. Note that the intermediates with halogen atoms attached (e.g., C20Cl1, see Supplementary Fig. 1) can also undergo the same process characteristically of the final product cyclo[20]carbon.
Based on the successful synthesis of C20, we now target C30 via the coupling of three C10 molecules. As such, we generate a C10 trimer (Int 4, C30Cl1) with a chlorine atom attached (Fig. 3a). As noted in Supplementary Fig. 1, a Cl atom attached in intermediates does not affect the following reactions. Subsequent voltage pulses could induce the transformation from Int 4 to Int 5 (Fig. 3b), and finally the formation of cyclo[30]carbon (C30) (Fig. 3c, d). STM image (Fig. 3d I) revealed C30 as a distinct donut-shaped structure. AFM images at tip far and close distances clearly show a polyynic structure of C30 with fifteen characteristic bright features assigned to triple bonds. Based on the calculations (Supplementary Fig. 2), we verify that the aromatic C30 adopts a polyynic structure (Fig. 3c I) as the ground state, and features a BLA of 0.14 Å. The assignment of 15 triple bonds is also supported by AFM simulations.
a I–a IV, b I–b IV Molecules, AFM images, Laplace-filtered AFM images, and simulated AFM images of Int 4 (C30Cl1) and Int 5. c I–c IV Molecule, AFM image, Laplace-filtered AFM image, and simulated AFM image of the final product cyclo[30]carbon at tip far distance. d I–d IV STM image, AFM image, Laplace-filtered AFM image, and simulated AFM image of cyclo[30]carbon at tip close distance. AFM tip offsets Δz: +0.4 Å, +0.5 Å, +1.3 Å, +1.1 Å for (a II to d II). Reference set points of Δz: I = 0.5 pA, V = 0.3 V for all AFM images. The scale bar in (a II) applies to (a) and (b) images. The scale bar in c II applies to (c) and (d) images.
We have also observed other reaction pathways towards the formation of C20 and C30, as shown in Fig. 4. C10Cl8 precursors could also be fused together first without completely occurring retro-Bergman ring-opening reactions, leading to the formation of various intermediates, for example, Int 6 and Int 7. After applying voltage pulses, the Int 6 and Int 7 can also undergo dehalogenation and ring-opening reactions, ultimately, yielding cyclo[20]carbon and cyclo[30]carbon.
a I–a III, b I–b III, c I–c III, d I–d III Molecules, AFM images and Laplace-filtered AFM images with superimposed models of Int 6 (C20Cl2), C20, Int 7 (C30Cl4) and C30. AFM tip offsets Δz: +0.5 Å, +0.4 Å, +0.4 Å, +0.3 Å for (a II–d II). Reference set points of Δz: I = 0.5 pA, V = 0.3 V for all AFM images. The scale bar in a II applies to (a–d) images.
Synthesis towards cyclo[40]carbon is attempted to demonstrate the potential of the coupling strategy for accessing larger cyclocarbons. Figure 5a shows two adjacent molecules, i.e., a C20 and a C20Cl2 (Int 6). Applying voltage pulses could also induce their coupling and ring-opening reactions, leading to the formation of Int 8 (C40Cl2, Fig. 5b) and Int 9 (C40Cl2) with a larger-ring structure, characterized by 17 triple bonds as shown in AFM images (Fig. 5c). Though further voltage pulses sometimes result in the breakage of such a big ring, it is definitely promising to generate C40 and larger cyclocarbons by such a strategy.
a I–a III, b I–b III, c I–c III Molecules, AFM images and Laplace-filtered AFM images with superimposed models of a C20 with Int 6 (C20Cl2), Int 8 (C40Cl2), Int 9 (C40Cl2). AFM tip offsets Δz: +0.4 Å, +0.4 Å, +0.6 Å for (a II–c II). Reference set points of Δz: I = 0.5 pA, V = 0.3 V for all AFM images. The scale bar in a II applies to (a–c) images.
In conclusion, we have developed a flexible strategy for synthesizing larger cyclocarbons (e.g., C20 and C30) from a smaller one (C10) based on tip-induced coupling and ring-opening reactions. C30 was characterized via bond-resolved AFM imaging, resolving the polyynic structure. This method demonstrates great potential for generating a series of cyclocarbons without being challenged to synthesize complicated precursors, and may stimulate further research into controlled coupling and polymerization of carbon nanorings.
Methods
Experimental details for STM and AFM measurements
STM and AFM measurements were carried out in a commercial (Createc) low-temperature system operated at 4.7 K with a base pressure better than 1 × 10−10 mbar. The single-crystalline Au(111) surface was cleaned by several sputtering and annealing cycles. The NaCl films were obtained by thermally evaporating NaCl crystals onto a clean Au(111) surface at room temperature, resulting in islands of one and two monolayers (ML) thickness. Octachloronaphthalene (C10Cl8, purchased from Aladdin, >99 %) molecules were deposited on a cold NaCl/Au(111) surface by thermal sublimation from a molecular evaporator. In this work, all experiments were conducted at a 2 ML NaCl/Au(111) surface. CO molecules for tip modification40 were dosed onto the cold sample via a leak valve. We used qPlus sensors41 with a resonance frequency f0 = 29.49 kHz, quality factor Q ≈ 45,000 and a spring constant k ≈ 1800 N/m operating in frequency-modulation mode42. The bias voltage V was applied to the sample with respect to the tip. AFM images were acquired in constant-height mode at V = 0 V and an oscillation amplitude of A = 1 Å. The tip-height offsets Δz for constant-height AFM images are defined as the offset in tip-sample distance relative to the STM set point at the NaCl surface. The positive (negative) values of Δz correspond to the tip-sample distance increased (decreased) with respect to a STM set point. Further discussions on the strategy can be found in Supplementary Figs. 3–6.
Density functional theory calculations
Density functional theory (DFT) calculations were carried out in the gas phase using the Gaussian 16 program package43. ωB97XD exchange-correlation functional44 in conjunction with def2-TZVP45 basis sets was used for calculations in the gas phase.
The AFM simulations were conducted by the PP-AFM code provided by Hapala et al.46. The detailed parameters are listed below. The lateral spring constant for the CO tip was 0.2 N/m, and a quadrupole-like charge distribution at the tip apex was used to simulate the CO tip with q = −0.1 e (e is the elementary charge and refers to |e|, and q is the magnitude of the quadrupole charge at the tip apex). The amplitude was set as 1 Å. The difference in probe height between “sim. far” and “sim. close” corresponded to the respective difference between “AFM far” and “AFM close”.
Data availability
All data are available from the corresponding author upon request. Source data are provided with this paper.
References
Parent, D. C. & McElvany, S. W. Investigations of small carbon cluster-ion structures by reactions with hydrogen cyanide. J. Am. Chem. Soc. 111, 2393–2401 (1989).
Van Orden, A. & Saykally, R. J. Small carbon clusters: spectroscopy, structure, and energetics. Chem. Rev. 98, 2313–2357 (1998).
Grutter, M. et al. Electronic absorption spectra of linear C6, C8 and cyclic C10, C12 in neon matrices. J. Chem. Phys. 111, 7397–7401 (1999).
Diederich, F. Carbon scaffolding: building acetylenic all-carbon and carbon-rich compounds. Nature 369, 199–207 (1994).
Pitzer, K. S. & Clementi, E. Large molecules in carbon vapor. J. Am. Chem. Soc. 81, 4477–4485 (1959).
Parasuk, V., Almlof, J. & Feyereisen, M. W. The [18] all-carbon molecule: cumulene or polyacetylene?. J. Am. Chem. Soc. 113, 1049–1050 (1991).
Torelli, T. & Mitas, L. Electron correlation in C4N+2 carbon rings: aromatic versus dimerized structures. Phys. Rev. Lett. 85, 1702–1705 (2000).
Arulmozhiraja, S. & Ohno, T. CCSD calculations on C14, C18, and C22 carbon clusters. J. Chem. Phys. 128, 114301 (2008).
Remya, K. & Suresh, C. H. Carbon rings: a DFT study on geometry, aromaticity, intermolecular carbon-carbon interactions and stability. RSC Adv. 6, 44261–44271 (2016).
Baryshnikov, G. V., Valiev, R. R., Kuklin, A. V., Sundholm, D. & Ågren, H. Cyclo[18]carbon: Insight into electronic structure, aromaticity, and surface coupling. J. Phys. Chem. Lett. 10, 6701–6705 (2019).
Baryshnikov, G. V. et al. Aromaticity of even-number cyclo[n]carbons (n = 6-100). J. Phys. Chem. A 124, 10849–10855 (2020).
Charistos, N. D. & Muñoz-Castro, A. Induced magnetic field in sp-hybridized carbon rings: analysis of double aromaticity and antiaromaticity in cyclo[2N]carbon allotropes. Phys. Chem. Chem. Phys. 22, 9240–9249 (2020).
Brémond, E., Pérez-Jiménez, A. J., Adamo, C. & Sancho-García, J. C. Stability of the polyynic form of C18, C22, C26, and C30 nanorings: a challenge tackled by range-separated double-hybrid density functionals. Phys. Chem. Chem. Phys. 24, 4515–4525 (2022).
Li, M. et al. Potential molecular semiconductor devices: cyclo-Cn (n = 10 and 14) with higher stabilities and aromaticities than acknowledged cyclo-C18. Phys. Chem. Chem. Phys. 22, 4823–4831 (2020).
Liu, Z., Lu, T. & Chen, Q. An sp-hybridized all-carboatomic ring, cyclo[18]carbon: Bonding character, electron delocalization, and aromaticity. Carbon 165, 468–475 (2020).
Baryshnikov, G. V. et al. Odd-number cyclo[n]carbons sustaining alternating aromaticity. J. Phys. Chem. A 126, 2445–2452 (2022).
Fowler, P. W., Mizoguchi, N., Bean, D. E. & Havenith, R. W. Double aromaticity and ring currents in all-carbon rings. Chem. Eur. J. 15, 6964–6972 (2009).
Mishra, S. et al. Observation of fractional edge excitations in nanographene spin chains. Nature 598, 287–292 (2021).
Sun, K. et al. On-surface synthesis of disilabenzene-bridged covalent organic frameworks. Nat. Chem. 15, 136–142 (2022).
Song, S. et al. Highly entangled polyradical nanographene with coexisting strong correlation and topological frustration. Nat. Chem. 16, 938–944 (2024).
Riss, A. et al. Imaging single-molecule reaction intermediates stabilized by surface dissipation and entropy. Nat. Chem. 8, 678–683 (2016).
Cai, J. et al. Atomically precise bottom-up fabrication of graphene nanoribbons. Nature 466, 470–473 (2010).
Pavliček, N. et al. Polyyne formation via skeletal rearrangement induced by atomic manipulation. Nat. Chem. 10, 853–858 (2018).
Pavliček, N. et al. On-surface generation and imaging of arynes by atomic force microscopy. Nat. Chem. 7, 623–628 (2015).
Zhong, Q. et al. Constructing covalent organic nanoarchitectures molecule by molecule via scanning probe manipulation. Nat. Chem. 13, 1133–1139 (2021).
Sun, L., Guo, Y., Xiang, W., Zhao, M. & Xu, W. The cumulenic linear C5 and its coupling-reaction products. Nat. Commun. 16, 9222 (2025).
Sun, L. et al. Tuning aromaticity of cyclocarbons by heteroatom doping: C12S and C12N. Natl. Sci. Rev. https://doi.org/10.1093/nsr/nwaf472 (2025).
Sun, L., Guo, Y., Xiang, W. & Xu, W. On-surface synthesis and characterization of linear and cyclic C6. Nat. Synth. 4, 940–946 (2025).
Sun, L. et al. On-surface synthesis of aromatic cyclo[10]carbon and cyclo[14]carbon. Nature 623, 972–976 (2023).
Sun, L. et al. On-surface synthesis and characterization of anti-aromatic cyclo[12]carbon and cyclo[20]carbon. Nat. Commun. 15, 7649 (2024).
Albrecht, F. et al. The odd-number cyclo[13]carbon and its dimer, cyclo[26]carbon. Science 384, 677–682 (2024).
Gao, Y. et al. On-surface synthesis of a doubly anti-aromatic carbon allotrope. Nature 623, 977–981 (2023).
Scriven, L. M. et al. Synthesis of cyclo[18]carbon via debromination of C18Br6. J. Am. Chem. Soc. 142, 12921–12924 (2020).
Kaiser, K. et al. An sp-hybridized molecular carbon allotrope, cyclo[18]carbon. Science 365, 1299–1301 (2019).
Jones, R. O. Density functional study of carbon clusters C2n (2⩽n⩽16). I. Structure and bonding in the neutral clusters. J. Chem. Phys. 110, 5189–5200 (1999).
Manna, D. & Martin, J. M. What are the ground state structures of C20 and C24? An explicitly correlated ab Initio approach. J. Phys. Chem. A 120, 153–160 (2016).
Lei, X., Fang, T., Liu, H. & Li, S. Cyclo[6]carbon-connected isomers of cyclo[18]carbon: exploring the electronic structure, aromaticity and antiaromaticity. Phys. Chem. Chem. Phys. 27, 17019–17030 (2025).
Brzyska, A., Panczyk, T. & Wolinski, K. From cyclo[18]carbon to the novel nanostructures—theoretical predictions. Int. J. Mol. Sci. 23, 12960 (2022).
Diederich, F. & Rubin, Y. Synthetic approaches toward molecular and polymeric carbon allotropes. Angew. Chem. Int. Ed. 31, 1101–1123 (1992).
Gross, L., Mohn, F., Moll, N., Liljeroth, P. & Meyer, G. The chemical structure of a molecule resolved by atomic force microscopy. Science 325, 1110–1114 (2009).
Giessibl, F. J. High-speed force sensor for force microscopy and profilometry utilizing a quartz tuning fork. Appl. Phys. Lett. 73, 3956–3958 (1998).
Albrecht, T. R., Grütter, P., Horne, D. & Rugar, D. Frequency modulation detection using high-Q cantilevers for enhanced force microscope sensitivity. J. Appl. Phys. 69, 668–673 (1991).
Frisch, M. J. et al. Gaussian 16 Rev. (C.01; Gaussian, Inc., Wallingford, CT, 2016).
Chai, J. D. & Head-Gordon, M. Long-range corrected hybrid density functionals with damped atom-atom dispersion corrections. Phys. Chem. Chem. Phys. 10, 6615–6620 (2008).
Weigend, F. & Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: design and assessment of accuracy. Phys. Chem. Chem. Phys. 7, 3297–3305 (2005).
Hapala, P. et al. Mechanism of high-resolution STM/AFM imaging with functionalized tips. Phys. Rev. B 90, 085421 (2014).
Acknowledgements
The authors acknowledge the financial support from the National Natural Science Foundation of China (22125203 to W.Xu, 22402149 to L.S.), the National Key R&D Program of China (2023YFE0101900 to W. Xu), the Ministry of Science and Technology of the People’s Republic of China, and the Shanghai Science and Technology Program (24ZR1470000 to L.S.).
Author information
Authors and Affiliations
Contributions
W. Xu conceived the research, L.S., Y.G., Y.Y., W. Xiang, and G.X. performed the STM/AFM experiments, L.S. and Y.G. carried out the DFT calculations, all authors contributed to writing the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Ji Ma and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Guo, Y., Yun, Y., Xiang, W. et al. On-surface synthesis of cyclo[20]carbon and cyclo[30]carbon from cyclo[10]carbon. Nat Commun 16, 11507 (2025). https://doi.org/10.1038/s41467-025-66650-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-66650-7


![Fig. 1: Synthesis of cyclo[20]carbon and cyclo[30]carbon.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs41467-025-66650-7/MediaObjects/41467_2025_66650_Fig1_HTML.png)
![Fig. 2: The coupling and ring-opening reactions of two cyclo[10]carbons.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs41467-025-66650-7/MediaObjects/41467_2025_66650_Fig2_HTML.png)
![Fig. 3: The coupling and ring-opening reactions of three cyclo[10]carbons.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs41467-025-66650-7/MediaObjects/41467_2025_66650_Fig3_HTML.png)
![Fig. 4: Other reaction pathways towards cyclo[20]carbon and cyclo[30]carbon.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs41467-025-66650-7/MediaObjects/41467_2025_66650_Fig4_HTML.png)
![Fig. 5: Attempts towards cyclo[40]carbon.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs41467-025-66650-7/MediaObjects/41467_2025_66650_Fig5_HTML.png)