Abstract
John B. Gurdon approached biology with characteristic clarity and patience, asking not only what happens in development, but how much of a molecule is present, and for how long. His nuclear-transfer experiments revealed that cellular identity is not fixed but can be reset, reshaping both biology and medicine. Through studies of oocytes, translational control, and the community effect, he showed that stability in living systems arises from persistence and interaction. His influence reached far beyond the bench: through generosity and curiosity, he nurtured a community of scientists who, like their mentor, enjoy asking how a cell knows what to be.
Similar content being viewed by others
Introduction
John Gurdon often spoke of biology in the language of gradients and time. In seminars, he would frequently return to a deceptively simple question, familiar to many of his colleagues: how many molecules are present, and at what concentration? He emphasized that both the abundance of a molecule and the duration of its activity together determine the outcome of a developmental process. A low-level signal, if sustained, could shape a fate as effectively as a transient, intense burst.
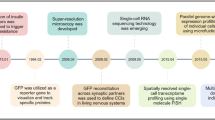
In many ways, John himself embodied this principle. In the cytoplasm of biology, he was something like a single molecule: constant, gentle in intensity, but enduringly present. Over decades, that committed, steady presence diffused through the field, influencing not only its direction but also its character. His long residence time in this medium induced development, not only through the discoveries he made, but also through the many scientists he mentored and inspired. His effect, like the molecular processes he studied, was the product of both concentration and time, and will be remembered for generations (Fig. 1).
Summer 2015.
One should always start with a question
In his most celebrated work, the question was whether the nucleus of a differentiated cell retains the capacity to form every cell type of an adult organism. This question was tackled by remarkable scientists before him, such as Briggs and King1, but remained a puzzle: do differentiated cells lose or permanently inactivate genes as they specialize? The answer required a definitive experiment.
That experiment was nuclear transfer: taking the nucleus from a differentiated intestinal cell and transplanting it into an enucleated Xenopus egg. When discussing experiments, John often asked his colleagues: What would be the best possible outcome? He valued experiments that would give a clear answer, much like this one. If the egg developed into a complete, fertile frog, it would mean the intestinal nucleus still contained the full set of genetic instructions for every cell type. Differentiation, it turned out, could be erased. A mature nucleus, when placed in the right cytoplasmic environment, in this case an enucleated egg, could indeed direct the formation of an entire organism2. The first adult cloned vertebrate was born: a frog. This revealed that the genome did not discard information during specialization; it merely used it differently.
When asked what set his success apart from the many failed attempts before him, John would attribute it humbly to “luck — and Xenopus. They’re very forgiving eggs.” Those who have spent days waiting for a cooperative batch of frog eggs might smile at that. John’s success, then, was unlikely to rest on luck alone, but rather on his persistence, precision, and a kind of craftsmanship that could turn even the most obstinate egg into an experiment worth keeping. The work demanded not only good eggs, but a trip to the hardware store, delicate technique, and the steady support of an equally persistent colleague. Depth micrometers were fastened to glass insulin syringes with tape and Parafilm. Pipettes were drawn and sharpened on home-built instruments that he maintained carefully in his office (Fig. 2). The permeabilization of the intestinal nucleus was refined step by step; UV exposure, delivered through a Hanovia lamp, was adjusted to the precise level that allowed the enucleated egg to survive the irradiation and the transplanted nucleus to respond. Each instrument, each condition, was tuned with patience. He remained throughout his career very much at the bench — often shaping the tools he needed himself, and continuing to maintain the microforge by hand into his nineties — though he allowed himself weekends away from the microscope.
A second station was set up for use by his colleagues. In the background, the needle polishing equipment can be seen. Most tools were fashioned by hand, often repurposing tools from a hardware store. November 2022.
The impact of that meticulous work extended far beyond the bench. By proving that a specialized cell could be reprogrammed to direct full development, John established a principle that would reshape modern biology. Decades later, Shinya Yamanaka and colleagues showed that a defined set of transcription factors could reprogram a somatic nucleus to pluripotency3. By introducing only a few transcription factors, they reprogrammed mature cells to a pluripotent state — creating stem cells without embryos.
Translational control: lessons from the oocyte
Once it was clear that a differentiated nucleus could be persuaded back into early embryogenesis, John looked instead at the cytoplasm — the environment that drove this persuasion.
In what became a series of straightforward experiments, John and his colleagues showed that mRNA injected into Xenopus laevis oocytes could be translated into functional proteins. The experiment was often recalled among colleagues when discussing experiments that seemed unlikely to succeed — few believed it would work, given the abundance of RNases in the egg that would degrade the RNA. Nevertheless, when globin mRNA was introduced by injection into oocytes, hemoglobin was produced4. The work demonstrated that the oocyte cytoplasm could direct translation of exogenous messages without difficulty, and it served as a reminder that sometimes experiments worth attempting are precisely those that everyone expects to fail.
This work showed that protein synthesis in the early embryo could be regulated independently of nuclear transcription and established the oocyte as a practical “test tube” for studying gene expression and protein function in living cells. The approach has since become a standard tool for analyzing protein activity, interactions, and regulation, with applications extending to therapeutic development and drug discovery.
The community effect
Later, Gurdon described what he called the “community effect.” Cells, he observed, rely on their neighbors to stabilize differentiation; an isolated cell, even when provided with the proper signals, may revert or die5. Differentiation, in other words, is a collaborative process.
John saw the humor in this. “Even cells,” he would remark, “seem to prefer good company.” The finding connected developmental biology to systems thinking — the idea that stability arises not from isolation, but from interaction.
His leadership philosophy reflected the same principles that characterized his experimental work. When he and Ron Laskey established the Wellcome/CRC Institute—later the Gurdon Institute—its organization was consciously modeled on Max Perutz’s MRC Laboratory in Cambridge. John spoke very highly of his time and colleagues at the MRC, crediting especially the importance of a good tearoom, where collegial discussions took place, including happy hours and Christmas parties. The structure of the MRC and Gurdon Institute was intentionally non-hierarchical, with minimal administrative barriers, and shared communal spaces designed to foster informal scientific exchange. The underlying conviction was that intellectual progress emerges most effectively from open dialogue and collective engagement. The same principle that governed his studies of cellular identity extended to the institutional level: stability was achieved not through rigidity, but through continuous conversation.
John’s concept of the community effect has had profound implications in tissue engineering and organoid development. Researchers have applied this principle to create complex tissue structures by ensuring that cells are cultured in environments that mimic their natural interactions. This approach has led to advancements in creating organoids that more accurately represent human tissues, enhancing the study of development and disease modeling.
Quantitative thinking in development
John was fascinated by how the dose and duration of signaling determine developmental outcomes: how a low-level morphogen signal, if sustained, can match the effect of a transient peak. He explored this experimentally in Xenopus animal caps, defining how cells read gradients over time to choose fates6. Building on his earlier investigations of morphogen gradients, he became interested in the occupancy and persistence of transcription factors at specific binding sites7. One factor in particular, ASCL1, frequently appeared in his conversations in those years, a reliable point of return, whether in a seminar question or over tea. Persistence, in both molecules and experiments, remained a recurring motif in his approach.
This perspective that developmental systems interpret continuous, quantitative information rather than operate through discrete binary states remains a foundational concept in developmental biology. It also reflected his personal temperament: measured, patient, and deliberate, with a preference for steady, sustained inquiry.
His quantitative approach inspired computational models that simulate how cells interpret signaling gradients. These models support both the understanding of pattern formation and the design of regenerative interventions.
Molecular mechanisms of cell fate reprogramming
A question John turned to in the latter years of his career concerned what stabilizes cellular identities and what is special about eggs and oocytes that allows them to reprogram differentiated nuclei. In many respects, he had returned to the questions that had guided his early work — now equipped with new methods and technologies that could address them.
John’s recent work on cellular reprogramming uncovered a broad spectrum of mechanisms that either permit or repress changes in transcriptional programs and cell identity. It encompassed DNA methylation, histone modifications, histone variants, chromatin organization, and the coordinated activity of transcriptional activators and repressors8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24. Together, these studies offered a more complete picture of how cells maintain stability, and how that stability can be transiently overcome to allow reprogramming.
As in the nuclei he studied, John appreciated that change took time and the right conditions. He would pose a question, then let it settle, giving students space to explore, fail, and learn. Small, careful interventions — a suggestion here, a demonstration there — often produced results that initially seemed unlikely, reflecting the same steady, persistent influence he uncovered in reprogramming experiments.
The impact of this work has been profound. By dissecting the molecular rules that govern cellular identity, John’s later studies provided a framework for understanding how epigenetic and transcriptional regulators interact to stabilize or alter cell fate. This knowledge has informed research into regenerative medicine, cellular reprogramming, and the development of strategies to manipulate cell identity for therapeutic purposes.
Legacy and character
John’s legacy is both conceptual and communal. He demonstrated that cellular identity is not fixed. Biology, it seems, is unusually generous with second chances. At school, he was deemed so hopeless at science that he was asked to abandon it entirely and instead read classics at Oxford. On arrival, he discovered there was no place for him in classics, and the only available course was in biological sciences. Later, intending to pursue entomology, he was again redirected, this time to Michail Fischberg’s laboratory, where space and frogs were available. These early course corrections, rather than deterring him, hinted at the persistence and independent thought that would define his career.
Those who trained with him remember a laboratory shaped by calm respect and quiet determination. He was an exceptional mentor, though he would never have claimed the title. “When we joined his lab, we became his colleagues,” one former student said. Conversations flowed over tea and digestive biscuits at 10:30 am in the tearoom. He encouraged young scientists to think critically, to design simple and decisive experiments, and to view setbacks as part of discovery rather than evidence of failure. Praise was freely given, while criticism, when needed, was thoughtful and respectful. He guided rather than directed, helping students choose a question worth asking and then giving them the confidence and space to enjoy the pursuit. He made young researchers feel capable, not by grand statements but through a quiet belief that their ideas mattered. Many remember that he could make you believe the experiment would work, and often, somehow, it did.
In the lab, when John wanted to speak, he would first make his presence known with a knock on your bench, or leave a small yellow sticky note on your desk carrying a question, an idea, or sometimes even an invitation. Successes were celebrated at formal dinners, and each year he and his wife, Jean, welcomed the whole lab to their home for lunch (Fig. 3). Tennis matches were a regular fixture, spirited and fiercely contested. John was also known for his subtle, clever humor. Though he rarely watched films, he enjoyed “The Sting” and liked to playfully trick his colleagues in the same clever, understated way, with only a slight twitch of his eye revealing the joke. His humor was matched by his humility. Asked in later years about his motivation, he said, “I just wanted to understand how a cell knows what to be. And I never got bored of frogs.”
Summer 2017.
His scientific contributions, from nuclear reprogramming to translational control and the community effect, revealed that development is both autonomous and cooperative, molecular and social. Through his leadership, he showed that scientific communities thrive under similar principles. Shared curiosity, respectful exchange, and the confidence that even a faint signal, if sustained, can shift the course of a field.
John’s presence, sharp and warm and quietly formidable, will be deeply missed. Yet his influence continues in the many scientists he trained and in the experiments he set in motion. His legacy reminds us that in science, as in life, one should remain humble, patient, and genuinely curious - interested in what might happen next.
References
Briggs, R. & King, T. J. Transplantation of Living Nuclei From Blastula Cells into Enucleated Frogs’ Eggs. Proc. Natl. Acad. Sci. USA. 38, 455–463 (1952).
Gurdon, J. B., Elsdale, T. R. & Fischberg, M. Sexually mature individuals of Xenopus laevis from the transplantation of single somatic nuclei. Nature 182, 64–65 (1958).
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
Gurdon, J. B., Lane, C. D., Woodland, H. R. & Marbaix, G. Use of frog eggs and oocytes for the study of messenger RNA and its translation in living cells. Nature 233, 177–182 (1971).
Gurdon, J. B. A community effect in animal development. Nature 336, 772–774 (1988).
Gurdon, J. B. & Bourillot, P.-Y. Morphogen gradient interpretation. Nature 413, 797–803 (2001).
Gurdon, J. B., Javed, K., Vodnala, M. & Garrett, N. Long-term association of a transcription factor with its chromatin binding site can stabilize gene expression and cell fate commitment. Proc. Natl. Acad. Sci. 117, 15075–15084 (2020).
Oikawa, M. et al. Epigenetic homogeneity in histone methylation underlies sperm programming for embryonic transcription. Nat. Commun. 11, 3491 (2020).
Jullien, J. et al. Gene Resistance to Transcriptional Reprogramming following Nuclear Transfer Is Directly Mediated by Multiple Chromatin-Repressive Pathways. Mol. Cell 65, 873–884.e8 (2017).
Jullien, J. et al. Hierarchical Molecular Events Driven by Oocyte-Specific Factors Lead to Rapid and Extensive Reprogramming. Mol. Cell 55, 524–536 (2014).
Jullien, J. et al. HIRA dependent H3.3 deposition is required for transcriptional reprogramming following nuclear transfer to Xenopus oocytes. Epigenet. Chromatin 5, 17 (2012).
Jullien, J., Åstrand, Halley-Stott, C., Garrett, R. P. & Gurdon, N. J. B. Characterization of somatic cell nuclear reprogramming by oocytes in which a linker histone is required for pluripotency gene reactivation. Proc. Natl. Acad. Sci. 107, 5483–5488 (2010).
Halley-Stott, R. P., Jullien, J., Pasque, V. & Gurdon, J. Mitosis gives a brief window of opportunity for a change in gene transcription. PLoS Biol. 12, e1001914 (2014).
Hörmanseder, E. et al. H3K4 methylation-dependent memory of somatic cell identity inhibits reprogramming and development of nuclear transfer Embryos. Cell Stem Cell 21, 135–143.e6 (2017).
Miyamoto, K., Pasque, V., Jullien, J. & Gurdon, J. B. Nuclear actin polymerization is required for transcriptional reprogramming of Oct4 by oocytes. Genes Dev. 25, 946–958 (2011).
Teperek, M. et al. Sperm is epigenetically programmed to regulate gene transcription in embryos. Genome Res 26, 1034–1046 (2016).
Lavagnolli, T. et al. Initiation and maintenance of pluripotency gene expression in the absence of cohesin. Genes Dev. 29, 23–38 (2015).
Simeoni, I., Gilchrist, M. J., Garrett, N., Armisen, J. & Gurdon, J. B. Widespread transcription in an amphibian oocyte relates to its reprogramming activity on transplanted somatic nuclei. Stem Cells Dev. 21, 181–190 (2012).
Narbonne, P. & Gurdon, J. B. Amphibian interorder nuclear transfer embryos reveal conserved embryonic gene transcription, but deficient DNA replication or chromosome segregation. Int. J. Dev. Biol. 56, 975–986 (2012).
Pasque, V., Halley-Stott, R. P., Gillich, A., Garrett, N. & Gurdon, J. B. Epigenetic stability of repressed states involving the histone variant macroH2A revealed by nuclear transfer to Xenopus oocytes. Nucleus 2, 533–539 (2011).
Pasque, V., Gillich, A., Garrett, N. & Gurdon, J. B. Histone variant macroH2A confers resistance to nuclear reprogramming: MacroH2A restricts reprogramming. EMBO J. 30, 2373–2387 (2011).
Ng, R. K. & Gurdon, J. B. Epigenetic memory of active gene transcription is inherited through somatic cell nuclear transfer. Proc. Natl. Acad. Sci. 102, 1957–1962 (2005).
Byrne, J. A., Simonsson, S., Western, P. S. & Gurdon, J. B. Nuclei of adult mammalian somatic cells are directly reprogrammed to oct-4 stem cell gene expression by amphibian oocytes. Curr. Biol. 13, 1206–1213 (2003).
Simonsson, S. & Gurdon, J. DNA demethylation is necessary for the epigenetic reprogramming of somatic cell nuclei. Nat. Cell Biol. 6, 984–990 (2004).
Author information
Authors and Affiliations
Contributions
Authors E.H., V.P., R.H.-S., and J.J. conceptualized, wrote, and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Halley-Stott, R.P., Hörmanseder, E., Jullien, J. et al. Reversibility, regulation, and the community of development: the legacy of Sir John B. Gurdon. Nat Commun 16, 10899 (2025). https://doi.org/10.1038/s41467-025-67023-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-67023-w