Abstract
We learn not just to remember, but also to create. Yet, the neurocognitive foundations by which higher-order learning fosters subsequent creation remain elusive. After verifying the facilitating effects of creative exemplar-learning on subsequent creation, this study identified hippocampal and prefrontal encoding patterns during exemplar-learning that predict higher (HSC) or lower (LSC) subsequent creation. The HSC condition was characterized by reduced hippocampal representational dimensionality and increased hippocampal-ventrolateral prefrontal cortex (vlPFC) connectivity—effects independent of exemplar memory and consistent across textual and pictorial exemplar materials. Cross-phase analyses revealed that low-dimensional hippocampal and full-dimensional vlPFC representations transferred more effectively from learning to creating in HSC trials. Moreover, cross-phase decoding function in the vlPFC for discriminating HSC from LSC trials relied on low-dimensional hippocampal representations during learning. Together, these findings show that hippocampal–vlPFC orchestration abstracts and transfers exemplar information to support creativity, revealing a neurocognitive mechanism of higher-order learning distinct from memory acquisition.
Similar content being viewed by others
Introduction
Learning and creativity form the foundation of human intelligence, fueling individual and societal progress. Effective strategies for enhancing creativity through learning have long intrigued scholars, dating back to Confucius’s “enlightening teaching” and Socrates’s intellectual midwifery (500–400 BCE). One effective strategy is learning from others’ creative ideas to inspire one’s own creative ideation1,2,3,4, through imitation, abstraction, and generalization of thinking patterns5,6. This process represents a form of higher-order learning for creation, which goes beyond mere memorization or inference from the learned experience. Such learning strategies interact with effective memory formation to ensure that core knowledge is preserved, along with abstraction that supports generalization of knowledge, including flexible strategies applicable in novel situations. Although widely discussed in psychology and education, our understanding of the neurocognitive mechanisms underlying higher-order learning for creative ideation remains in its infancy.
A fundamental question in higher-order learning is how neural representations of learned knowledge are formed to foster individuals’ creativity. The hippocampus and prefrontal systems are recognized to play crucial roles in learning and memory across multiple scales in humans and animals, varying from regional activity to fine-tuned representations and large-scale brain network connectivity7,8,9. Moreover, recent advances have provided evidence for hippocampal and prefrontal contributions to abstraction and generalization in various learning strategies, which build on but go beyond memory encoding, retrieval, and retention10,11,12. Abstraction and generalization may also be fundamental to human creativity5,6, although creative original thoughts may exceed the scope of ordinary generalization. Thus, the hippocampal and prefrontal systems are likely to play a critical role in supporting creative generalization during higher-order learning.
According to influential cognitive theories and large language models for knowledge acquisition and generalization13,14, higher-order learning and creative generalization may require more compressed or abstract forms of knowledge representations—meaning they capture essential features and underlying structures while omitting irrelevant details5,15. Such compression can be estimated by information-based computational approaches such as representational dimensionality16, which quantifies the minimum number of dimensions required to explain maximal variability in neural population activity under given conditions17. This approach is proven to be effective in many cognitive domains, including but not limited to conceptual construction18, adaptation19, cognitive control17, and memory20. Recent studies on neural manifolds have demonstrated that high-dimensional neural activity data can be projected onto low-dimensional manifolds to represent the principal patterns of neural activity21,22. This process, known as dimensionality reduction23, extracts and retains the most informative features from inputs24,25. Dimensionality reduction is crucial for fostering the generalization ability in higher-level cognition and deep learning models26,27. Indeed, recent computational models suggest that the hippocampus may facilitate generalization by flexibly reconstructing memories from latent representations through dimensionality reduction enabled by memory replay28, thereby supporting imagination and episodic future thinking, which are closely related to creative ideation29,30. Low-dimensional hippocampal representations have also been found to be crucial for extracting prototype information from exemplars12,31 and for representing the abstracted geometries of episodic experiences32. These considerations suggest that hippocampal representational dimensionality reduction plays a critical role in higher-order learning for creative ideation.
In conjunction with the hippocampus, the prefrontal cortex is essential for mediating the abstraction and generalization of declarative memory representations12,33,34,35, by combining episodic mnemonic representations with schematic-based ones and exerting cognitive control over the abstraction and generation of strategies36. Hippocampal–prefrontal functional coordination has also been implicated in concept learning, updating37 and rule abstraction38, in which memories are integrated into the neocortex via crosstalk with the hippocampus39, suggesting that hippocampal–prefrontal connectivity may aid hippocampal dimensionality reduction by extracting flexible schemas from the specific features of learned exemplars40. Taken together, we hypothesize that lower-dimensional hippocampal representations and strong hippocampal–prefrontal connectivity during creative exemplar-learning will facilitate subsequent creative ideation.
Experimental designs
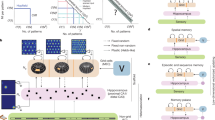
To test our hypotheses, we developed a higher-order learning paradigm for an event-related functional magnetic resonance imaging (fMRI) study aimed at identifying the neural encoding patterns during learning that predict subsequent creative ideation and remembrance. The paradigm comprised a creative exemplar-learning phase and a creative ideation phase. In the exemplar-learning phase, participants learned a set of creative exemplars for target objects. Thereafter, they were unexpectedly asked to generate their own creative ideas for the same set of target objects, either outside (Exp. 1) or inside (Exp. 2) the MRI scanner. Each generated idea was later evaluated by independent raters and assigned a creativity score, allowing individual-specific categorization of exemplar-learning trials into higher or lower subsequent creation (HSC/LSC) conditions. This design allowed us to directly examine how the hippocampal and prefrontal encoding during exemplar-learning affects subsequent creative outputs by contrasting HSC with LSC. Additionally, Exp. 2 employed pictorial rather than textual exemplars and combined neural recording across both learning and creating phases to examine cross-phase transfer of hippocampal and prefrontal encoding patterns.
We adopt an “output-based definition” of higher-order learning, in which learning is defined by its creative consequences. This contrasts with “intention-based” approaches (manipulating learner intentions, e.g., memorizing vs. creating) and “material-based” approaches (manipulating exemplar type, e.g., creative vs. conventional). Our framework enables critical contrasts between HSC and LSC while strictly controlling materials and intentions, paralleling the classic subsequent memory effect (SME)41,42,43, but instead focuses on the subsequent creation effect (SCE). Both SME and SCE classify the learning trials by subsequent memory or creation outcomes rather than by manipulated intentions or materials, although the conclusions drawn from our output-based framework may not fully generalize to paradigms involving such manipulations.
The similarities between SME and SCE paradigms make it possible to explore the relationship between neural encoding that benefits subsequent memory and supports creative ideation. Evidence suggests that episodic memory retrieval positively correlates with creative performance44,45, as both processes engage a shared network involving the hippocampus and prefrontal cortex46,47. Notably, hippocampal damage impairs both episodic memory and creative ideation30,48,49. However, creative ideation does not necessarily correlate with better memory retention50,51,52. Individuals who forgot more of the learned exemplars, for instance, may exhibit better subsequent creativity53, indicating a beneficial aspect of forgetting rather than remembering for creativity. To reconcile these seemingly contradictory findings, we conducted a supplementary fMRI Exp. S2 to directly examine hippocampal and prefrontal encoding during exemplar-learning that predicts subsequent memory retrieval. In addition, we conducted eight behavioral studies (Exp. S1a–c, S3–S7) aimed at: (1) verifying the facilitating effects of creative exemplar-learning on subsequent creative ideation, by comparing the creative performance after creative exemplar-learning (Exp. 1) with no exemplars-learning (Exp. S1a), ordinary (non-creative) exemplars-learning (Exp. S1b), and semantic priming with words drawn from exemplar pairs (Exp. S1c); and 2) exploring how manipulations of exemplar encoding and retention impact subsequent creation—including remembering vs. forgetting instruction during exemplar-learning (Exp. S3), exemplar retrieval with or without episodic details (Exp. S4), and incubation delays between exemplar-learning and self-creating (Exps. S5–S7, see Fig. 1 for an overview).
The study employed a higher-order learning paradigm comprising a creative exemplar-learning phase followed by a self-creating phase. During exemplar learning, participants underwent event-related fMRI while encoding creative exemplars for a set of target objects. Subsequently, they were unexpectedly instructed to generate novel creative ideas for the same objects—either outside the scanner (Exp. 1) or during scanning (Exp. 2). Exemplar-learning trials were retrospectively classified as higher subsequent creation (HSC) or lower subsequent creation (LSC) trials based on creativity ratings, enabling comparison of hippocampal representational dimensionality and hippocampal–prefrontal connectivity between trial types. In Exp. 2, joint analyses further quantified cross-phase transfer of hippocampal and prefrontal encoding from learning to creating. Supplementary experiments examined the promotive effects of exemplar-learning on creative performance (Exp. S1a–c); delineated hippocampal and prefrontal contributions to exemplar memory retrieval (Exp. S2), and examined the effects of exemplar remembering with or without episodic details (Exps. S3 and S4), and assessed the impact of incubation delays between learning and creating (Exps. S5–S7). HPC, hippocampus; IFG, inferior frontal cortex. Source data are provided as a Source Data file. Figure created in BioRender (Created in BioRender. Zhang, Z. 2025, https://BioRender.com/pgpmd7i).
Results
Behavioral effects of creative exemplar-learning on creativity and memory
In Exp. 1, participants learned a series of daily objects paired with exemplars of creative uses (e.g., “whisk—candlestick,” indicating that a whisk could serve as a candlestick) during fMRI scanning (phase 1), judging each object-usage pair as creative or uncreative via button press. They were then unexpectedly asked to generate their own creative ideas for the same object (e.g., generate creative uses for “whisk,” phase 2) outside the scanner. Finally, participants viewed each object again and recalled the exemplar use learned during phase 1 (e.g., recall “candlestick” for “whisk,” phase 3). Exemplar-learning trials were classified as HSC or LSC conditions based on each participant’s creativity scores; trials above the individual-specific median were labeled as HSC, while those below as LSC (Fig. 2A). Behavioral analyses revealed no significant difference in subjective exemplar creativity judgments during learning between HSC and LSC trials (t(26) = 1.71, p = 0.10, R2 = 0.10, 95% CI [−0.01, 0.14]; Fig. 2B, result. S1). However, HSC trials showed a significantly higher forgetting rate at cued-recall (t(26) = 3.63, p = 0.001, R2 = 0.34, 95% CI [0.08, 0.30]; Fig. 2C, result. S1). Regression and quartile-based analyses showed that the serial order of exemplar presentation (early or late) during the learning session did not affect subsequent creative ideation (result. S2, Fig. S1).
A During the exemplar-learning phase (scanning), participants learned 120 object–use pairs (100 creative exemplars and 20 uncreative fillers), each presented for 6 s with a 4–6 s fixation interval, and judged whether each pair was creative or not. Subsequently, participants were unexpectedly asked to generate creative ideas for the 100 target objects that had been paired with creative exemplars, outside the scanner (12 s per object, random order). Learning trials were retrospectively classified as higher (HSC) or lower (LSC) subsequent creation conditions according to whether each participant’s creative score for a given object fell above or below their individual-specific median across all objects, enabling within-subject analyses of the subsequent creation effect (SCE), defined as post-learning creative performance. B No significant difference was observed in subjective creative judgment during the learning phase between HSC and LSC trials (two-tailed paired t-test, t(26) = 1.71, p = 0.10, R2 = 0.10, 95% CI [−0.01, 0.14]). Each dot represents a participant (n = 27); gray lines connect paired values; half-violin plots show data distribution; box plots show the median (center), 25th–75th percentiles (bounds of box), and minima and maxima (whiskers). Black dots and error bars represent group means ± s.e.m.; dashed lines indicate mean differences. C Items associated with better creative performance in the creating phase (HSC) were more likely to be forgotten during cued-recall (two-tailed paired t-test, t(26) = 3.63, p = 0.001, R2 = 0.34, 95% CI [0.08, 0.30]). Plot features as in (B). D Creative exemplar learning (Exp. 1, n = 27; ‘Creative’ in plot) yielded significantly higher creativity scores than no exemplar-learning (Exp. S1a, n = 36; ‘None’), ordinary exemplar-learning (Exp. S1b, n = 40; ‘Ordinary’), or individual word semantic priming (Exp. S1c, n = 34; ‘Semantic’) controls (Kruskal–Wallis χ²(3) = 43.69, p = 1.8 × 10⁻⁹), with no significant differences among the three control groups (all p = 1.00). Half-violin plots depict score distributions; box plots show the median (center), 25th–75th percentiles (bounds of box), and minima and maxima (whiskers). Black points and error bars represent group means ± s.e.m.; colored dots indicate individual participant scores. *p < 0.05; ***p < 0.001; n.s., not statistically significant. Source data are provided as a Source Data file. Figure A created in BioRender (Zhang, Z. 2025, https://BioRender.com/wqyqyx3).
To further probe the role of subjective exemplar judgments (phase 1)—potentially reflecting subjective prediction errors54—we examined their relationships with subsequent creation (phase 2) and exemplar memory (phase 3). We also assessed the similarity between generated ideas and their corresponding exemplars, as an index of imitation. Although overall imitation levels were low in both conditions (<1.5 on a 5-point scale), the imitation score was significantly lower in HSC than in LSC trials (p = 0.002). Combined analyses revealed that subjective prediction errors positively predicted exemplar imitation (p = 0.033) and memory (p = 0.011), but not creative performance (p = 0.543). However, imitation itself positively predicted exemplar memory (p < 0.001) but negatively correlated with creative performance (p < 0.001), showing a possible trade-off between imitation and independent creative ideation55. Moreover, prediction error significantly increased the tendency toward imitation in this “imitation-creation trade-off” (p = 0.026), calculated as the ratio of exemplar imitation to creative performance on each creative idea (see results. S3; Figs. S2–S4; methods. S1). Together, these findings reveal a behavioral trade-off between memory retention and creative ideation, implying that the neural mechanisms underlying successful memory encoding (SME) and those supporting creative expression (SCE) may be distinct. We evaluate this divergence directly in the neuroimaging analyses.
Promoting effects of creative exemplar-learning on subsequent creative ideation
To evaluate the effect of exemplar-learning on subsequent creative ideation, we conducted three supplementary behavioral experiments. In Exp. S1a, participants completed the Alternative Uses Task (AUT, same as Exp. 1) without prior exposure to creative exemplars. In Exp. S1b, participants first learned the ordinary uses of the same objects (as in Exp. 1) before performing the AUT. In Exp. S1c, participants processed single words derived from the creative exemplars in Exp. 1 (e.g., “newspaper–lampshade” presented separately as “newspaper” and “lampshade”; they were randomly mixed with words taken from other exemplars) to conduct semantic priming of individual words. Results indicated that participants in the creative exemplar-learning condition (Exp. 1) exhibited significantly higher creative performance than those in any of the three control conditions (χ² = 43.69, df = 3, p < 0.001), whereas no significant differences were observed among the control groups (Fig. 2D; results. S4).
Lower hippocampal dimensionality in textual exemplar-learning predicts better subsequent creation
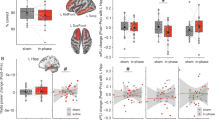
To test our key hypothesis that reduced hippocampal dimensionality supports higher-order learning, we compared multivoxel hippocampal representational dimensionality between HSC and LSC trials during the exemplar-learning phase of Exp. 1. Principal component analysis (PCA) was performed on trial-wise activation pattern matrices, with a 1000-sample bootstrap procedure to control for minor differences in trial numbers (mean: HSC = 42.83, LSC = 46.13). Dimensionality was defined as the number of principal components (PCs) explained 70–80% of the variance, averaged across this range (Fig. 3A). In the left hippocampus, representational dimensionality was significantly lower for HSC than for LSC trials (paired t-test, t(22) = −2.47, pbonferroni = 0.04, R2 = 0.22, 95% CI [−1.27, −0.11]; Fig. 3B, C), no significant difference was observed in the right hippocampus (t(22) = −1.64, pbonferroni = 0.24, R2 = 0.11, 95% CI [−0.56, 0.06]). To further examine whether this effect reflects structured information compression, we analyzed variance explained by the earliest, information-rich PCs (e.g., 1st to 3rd PCs), and found that the dimensionality difference was primarily attributable to these early components but not later ones (e.g., 4th to 6th PCs, result. S5, Fig. S5). Additional analyses using the standard 80% variance56,57,58 threshold corroborated these findings (result. S6). No HSC vs. LSC difference was observed in the visual control region (Neurosynth meta-analysis; p = 0.28), and a 10,000-iteration bootstrap confirmed the effect was significantly greater in the left hippocampus than in the visual control region (95% CI [0.18, 1.55]; result. S7, method. S4).
A Principal component analysis (PCA) was performed on trial-wise activation pattern matrices for higher (HSC) and lower (LSC) subsequent creation conditions. Dimensionality was defined as the number of principal components (PCs) required to explain 70–80% of the variance. B In the left hippocampus, representational dimensionality was significantly lower for HSC than LSC trials (two-tailed paired t-test, t(22) = –2.47, pbonferroni = 0.04, R2 = 0.22, 95% CI [−1.27, −0.11]); no significant difference was found in the right hippocampus (two-tailed paired t-test, t(22) = −1.64, pbonferroni = 0.24, R2 = 0.11, 95% CI [−0.56, 0.06]). Each dot represents a participant (n = 23); gray lines connect paired values; half-violin plots show data distribution; box plots show the median (center), 25th–75th percentiles (bounds of box), and minima and maxima (whiskers). Black dots and error bars represent group means ± s.e.m.; dashed lines indicate mean differences. C Left hippocampal dimensionality remained consistently lower for HSC than LSC conditions across the 70–80% explained variance range; shaded regions indicate s.e.m. D Global pattern similarity in the left hippocampus was higher in HSC than LSC trials (two-tailed paired t-test, t(22) = 2.34, pbonferroni = 0.04, R2 = 0.20, 95% CI [0.0006, 0.01]); no significant difference was observed in the right hippocampus (t(22) = 0.18, pbonferroni = 0.86, R2 = 0.002, 95% CI [−0.005, 0.004]). Plot features as in (B). E Pearson correlations between the representational dissimilarity matrices of the left hippocampus and the left inferior frontal gyrus (IFG) were computed under HSC and LSC conditions. F Hippocampus-IFG representational connectivity in HSC condition is significantly higher than LSC condition (two-tailed paired t-test, t(22) = 2.61, pbonferroni = 0.03, R2 = 0.24, 95% CI [0.006, 0.050]). Plot features as in (B). G A significant negative correlation was observed between hippocampal–IFG representational connectivity and hippocampal representational dimensionality (Pearson’s r = −0.49, two-tailed, p = 0.01). The black line represents the mean regression fit, and the gray shaded area indicates the 95% CI around the mean. Marginal distributions for representational connectivity and representational dimensionality are shown along the top (red) and right (blue) axes, illustrating the distribution of each variable. Bonferroni corrections were applied to account for comparisons between the left and right hippocampus. *p < 0.05; n.s., not statistically significant. Source data are provided as a Source Data file. Figures A and E created in BioRender (Zhang, Z. 2025, https://BioRender.com/4koh8ae; https://BioRender.com/kdgepur).
We next calculated global pattern similarity in the hippocampus to measure the similarity of activation patterns for each exemplar-learning trial relative to all other trials59, where higher similarity reflects greater overlap among neural representations and a more uniform engagement of cognitive processes across trials60. HSC trials exhibited significantly higher global pattern similarity in left hippocampus (t(22) = 2.34, pbonferroni = 0.04, R2 = 0.20, 95% CI [0.0006, 0.01]) but not right hippocampus (t(22) = 0.18, pbonferroni = 0.86, R2 = 0.002, 95% CI [−0.005, 0.004]; Fig. 3D).
Enhanced hippocampal–prefrontal connectivity predicts better subsequent creation
To investigating how the prefrontal cortex—especially the inferior frontal gyrus (IFG, also referred to as the vlPFC), which is critically involved in semantic selection61 and creative idea generation62—interacts with the hippocampal encoding to guide the extraction of essential exemplar information while filtering out irrelevant details63, we conducted a psychophysiological interaction (PPI) analysis using the left hippocampus as a seed. This analysis identified a significant cluster in the left IFG (x = −52, y = 36, z = 6; k = 138; FWE-corrected p < 0.05, cluster-forming threshold p < 0.001), showing stronger hippocampus-vlPFC connectivity for the HSC condition. We further conducted a representational connectivity analysis between the anatomically defined left hippocampus and left IFG/vlPFC regions (automated anatomical labeling, AAL atlas64) by correlating their representational dissimilarity matrices (RDMs) for HSC and LSC trials (Fig. 3E). Higher representational connectivity was again observed between the left hippocampus and the left VLPFC during HSC trials (t(22) = 2.61, p = 0.016, R2 = 0.24, 95% CI [0.006, 0.050]; Fig. 3F). Additionally, robust regression revealed that between-condition differences in hippocampal–vlPFC connectivity negatively predicted hippocampal dimensionality, suggesting the role of this connectivity in hippocampal representation compression (r = –0.49, p = 0.01; Fig. 3G; result. S8, Tables S1 and S2).
Robustness and control analyses
To ensure robustness and specificity of our primary neural findings, we conducted a series of control analyses addressing potential confounds. Across all tests, the HSC–LSC differences in hippocampal representational dimensionality and hippocampal–prefrontal connectivity––though not global hippocampal pattern similarity—remained reliable after controlling for the following factors (result. S9; Figs. S6–S8):
(1) Univariate activation: Bilateral univariate hippocampal activation was significantly lower in HSC than LSC trials (left hippocampus: t(22) = 2.46, pbonferroni = 0.04; right hippocampus: t(22) = 2.72, pbonferroni = 0.02]. After statistically controlling for these differences, both the reduced hippocampal representational dimensionality (F(1,21) = 6.06, p = 0.02) and the enhanced hippocampal–lPFC connectivity (F(1,21) = 6.33, p = 0.02) in the HSC condition remained significant. Further analyses excluding voxels sensitive to univariate activation yielded consistent results (hippocampal dimensionality: HSC < LSC, t(22) = 2.72, p = 0.04; hippocampal–vlPFC connectivity: HSC > LSC, t(22) = 3.26, p = 0.004).
2) Imitation and exemplars copying: To assess the extent to which participants imitated or copied learned exemplars, we evaluated two possibilities. First, we examined the similarity between self-generated ideas and the corresponding learned exemplar for the same object; as noted earlier, similarity ratings were generally low (all <1.5). Second, we identified and excluded items in which a generated idea closely resembled any learned function from other exemplars (e.g., after learning “paper clip–hairpin,” reporting “pencil as hairpin” during creation). Even after excluding these items, the main neural effects persisted: hippocampal dimensionality remained lower for HSC than LSC trials (mean trial counts: HSC = 44.61, LSC = 46; HSC < LSC, t(22) = −4.14, p < 0.001) and hippocampal–prefrontal connectivity (HSC > LSC, t(22) = 3.26, p = 0.004].
(3) Item-specific creative potential: To control for exemplar-specific differences in the potential to elicit creative ideation, we excluded items that were too consistently classified as HSC (n = 14) or LSC (n = 16) across participants. The reduced left hippocampal dimensionality (mean trial counts: HSC = 29.91, LSC = 31.48; HSC < LSC, t(22) = −2.12, p = 0.04) and enhanced hippocampal–vlPFC connectivity (HSC > LSC, t(22) = 2.42, p = 0.02) remained significant.
(4) Exemplar memory: We also considered cued-recall performance. As memory retrieval (phase 3) occurred after the creative ideation task (phase 2), this sequence may induce “problem-solving-induced forgetting”53. Thus, failure to recall a specific exemplar in phase 3 does not necessarily imply forgetting during phase 2 but could reflect the cognitive demands of generating novel ideas immediately beforehand. Nevertheless, exemplars successfully recalled in phase 3 can be regarded as reliably remembered, even if this approach underestimates the total number actually retained during creation. We therefore restricted analyses to items with successfully recalled exemplars, thereby controlling for potential memory confounds. Results remained consistent: after bootstrapping to match trial counts (mean trial counts: HSC = 8.83, LSC = 11.26), remembered HSC trials exhibited significantly lower hippocampal dimensionality (t(11) = –2.21, p = 0.04) and greater hippocampal–vlPFC connectivity (t(11) = 2.24, p = 0.04) than remembered LSC trials. Additionally, activation in Default Mode Network (DMN) regions—often linked to subsequent forgetting—did not differ between HSC and LSC conditions (all p > 0.30), ruling out attentional lapses or mind-wandering as alternative explanations for reduced hippocampal engagement in HSC.
(5) Conditional definition criteria: To mitigate potential bias from participant-specific HSC/LSC classifications, we redefined the two conditions using a fixed threshold across all participants (trials with creativity scores ≥ the group mean were classified as HSC). This reclassification again yielded consistent effects: hippocampal dimensionality remained lower for HSC than LSC trials (mean trials: HSC = 50.87, LSC = 49.13; t(22) = 2.37, p = 0.03) and hippocampal–vlPFC connectivity remained higher for HSC than LSC (t(22) = 2.35, p = 0.03).
Differential hippocampal and prefrontal encoding for subsequent memory and creation
In Exp. 1, the creative ideation task preceding the final memory test likely interfered with exemplar memory53, limiting precise assessment of SME. To directly explore hippocampal and prefrontal contributions to exemplar memory, we conducted a supplementary fMRI study (Exp. S2), employing the same exemplar-learning procedure and materials as Exp. 1 but replacing the self-creating phase (phase 2) with a cued-recall test (Fig. S9). Exemplar-learning trials were classified as “remembered” or “forgotten” based on participants’ ability to recall the exemplar use of each object when cued by its name. Remembered items elicited greater hippocampal univariate activation (t(22) = 2.22, pbonferroni = 0.04) and enhanced hippocampal global pattern similarity (t(22) = 4.33, pbonferroni < 0.001) than forgotten. However, no significant differences were observed in left hippocampal representational dimensionality (t(22) = 0.94, p = 0.36) or hippocampal–vlPFC representation connectivity (t(22) = 0.17, p = 0.87; result. S10, Table S3). These results demonstrate that the neural correlates supporting exemplar memory (SME) and creative ideation (SCE) may diverge, reflecting the multifaceted relationship between SCE and SME.
To further probe this relationship, we conducted additional behavioral experiments. First, because HSC trials exhibited weaker hippocampal activation during exemplar-learning than LSC trials—an effect typically linked to subsequent forgetting—we tested whether instructing participants to forget vs. remember exemplars during learning would influence subsequent creativity (Exp. S3). Items instructed to forget were recalled less accurately (t(32) = 4.27, p < 0.001) but yielded superior creative ideation (t(32) = 2.36, p = 0.02) than items instructed to remember (result. S11, Fig. S10A and B). Second, given that HSC trials were characterized by lower hippocampal representational dimensionality, we examined whether “Know” (K) vs. “Remember” (R) responses—distinguished by the absence or presence of vivid episodic detail during recognition65—influenced subsequent creative ideation (Exp. S4). No significant difference emerged between K- and R-responses (t(34) = 1.21, p = 0.23; result. S12, Fig. S10C), indicating that the loss of episodic richness per se does not impact creative ideation. Third, as temporal delays post-learning may induce forgetting while promoting abstraction and generalization, we examined the effects of incubation delays between exemplar-learning and self-creating (Exps. S5–S7). Subsequent creativity was significantly enhanced after an incubation delay (F(1,37) = 4.99, p < 0.05; Exp. S5), especially when the delay was prolonged (t(31) = 3.86, p < 0.001; Exp. S6) and coupled with a low-load working memory task (t(35) = 3.01, p = 0.005; Exp. S7; result. S13, Fig. S11). Taken together, Exp. S3 indicated that instructing participants to forget during learning enhanced subsequent creativity, whereas Exp. S4 showed that, once exemplars were successfully retrieved, the presence or absence of episodic detail had no effect on creative ideation. Together with the findings from Exp. 1 that reduced hippocampal encoding dimensionality in HSC relative to LSC persisted even when exemplars were equally remembered, these results suggest that dimensionality reduction is not simply a consequence of memory loss but rather reflects a higher level of abstraction. Consistent with this view, Exps. S5–S7 demonstrated that incubation-induced abstraction and generalization further boosted creative performance.
Lower hippocampal dimensionality in pictorial exemplar-learning predicts better subsequent creative ideation
To replicate the main findings of Exp. 1, Exp. 2 employed the same experimental paradigm as Exp. 1 but replaced textual exemplars with pictorial stimuli in the learning phase. fMRI data were collected across both the learning and creating phases, with a sparse-sampling protocol implemented during the creating phase to enable accurate capture of participants’ oral reports (Fig. 4A). Behavioral results are reported in result. S14. Consistent with Exp. 1, we observed lower representational dimensionality in the left hippocampus during exemplar-learning in HSC compared to LSC trials (t(38) = −2.21, pbonferroni = 0.03, R2 = 0.11, 95% CI [−0.31, −0.01]; Fig. 4B), alongside enhanced left hippocampal–vlPFC (same IFG region as in Exp. 1) representational connectivity (t(38) = 4.07, p = 0.0002, R2 = 0.30, 95% CI [-0.150, 0.049]; for additional replication analyses, see result. S15, Fig. S12). Given that the anterior hippocampus is preferentially associated with coarse-grained contextual representations, while the posterior hippocampus is linked to fine-grained local representations66,67,68, we conducted a hippocampal segmentation analysis and found reduced representational dimensionality in the left anterior hippocampus in the HSC condition (result. S16, Fig. S13).
A Participants performed exemplar-learning and creating tasks during MRI scanning. In the exemplar-learning phase, they learned a list of pictorial creative exemplars along with uninterested fillers. Subsequently, participants independently generated creative uses for these items; sparse fMRI sampling allowed accurate recording of oral responses. B In the left hippocampus, representational dimensionality was significantly lower for HSC than LSC trials (one-tailed paired t-test, t(38) = –2.21, pbonferroni = 0.03, R2 = 0.11, 95% CI [−0.31, −0.01]), with no significant difference in the right hippocampus (one-tailed paired t-test, t(38) = –1.48, p = 0.15, R2 = 0.06, 95% CI [–0.24, 0.04]). Each dot represents a participant (n = 39); gray lines connect paired values; half-violin plots show data distribution; box plots show the median (center), 25th–75th percentiles (bounds of box), and minima and maxima (whiskers). Black dots and error bars represent group means ± s.e.m.; dashed lines indicate mean differences. C Across combined data from Exps. 1 and 2 (n = 62), representational dimensionality in the left hippocampus (HPC, red) and left inferior frontal gyrus (IFG, blue) negatively correlated with creative performance (HPC: Pearson’s r = −0.42, two-tailed, p < 0.001; IFG: Pearson’s r = −0.31,two-tailed, p = 0.02). Each dot represents a participant; lines and shaded areas denote the linear regression fit and 95% CI, respectively. Marginal histograms show the distribution of each variable. D Schematic of the analytic framework quantifying the cross-phase transfer of exemplar representations from learning to creating. Hippocampal and IFG activity patterns were extracted from both phases and compared using cross-phase representational similarity measures, applied in both compressed (low-dimensional, via principal component analysis [PCA]) and uncompressed (full-dimensional) representational spaces. This approach enables evaluation of how neural representations established during learning are reactivated and utilized during creative ideation. *p < 0.05; n.s., not statistically significant. Source data are provided as a Source Data file. Figure A created in BioRender (Zhang, Z. 2025, https://BioRender.com/ssx9c3z).
Critically, combining data from Exps. 1 and 2 and treating creative performance as a continuous variable in a between-subject analysis, we observed significant negative correlations between post-learning creative performance and the representational dimensionality in both the left hippocampus (r = –0.42, p < 0.001) and left IFG (r = −0.31, p = 0.02; Fig. 4C, result. S17) during exemplar-learning, with no such effects in visual control regions. Finally, to assess whether exemplar memory exerted significant interference with independent creative ideation, we compared activation in regions implicated in cognitive conflict processing (dorsolateral prefrontal cortex [DLPFC], anterior cingulate cortex [ACC])69,70 and in the integration of internally and externally generated information (insula) between HSC and LSC conditions during the creating phase. No significant differences were observed (result. S18), suggesting that any interference from exemplar memory with creative ideation was too weak to be detected at the neural level.
Lower-dimensional hippocampal representational transfer from learning to creating
We assessed the transfer of exemplar representations from learning to creating phases using cross-phase representational similarity analyses on regional encoding patterns in both compressed (low-dimensional, abstracted) and uncompressed (full-dimensional) spaces (Fig. 4D). First, we examined how low-dimensional hippocampal representations during learning relate to subsequent creative processing. Using PCA, we mapped hippocampal activity patterns from both the learning and creating phases into a shared low-dimensional representational space, enabling a comparison of latent variables between the HSC and LSC conditions (Fig. 5A). The results showed that compressed left hippocampal representations transferred more effectively from learning to creating in the HSC compared to the LSC condition, as indicated by a greater similarity between the latent variables (paired t-test: t(38) = 2.12, p = 0.04, R2 = 0.34, 95% CI [0.002, 0.08]; Fig. 5B). No significant difference was observed in the visual control area (t(38) = 0.43, p = 0.67, R2 = 0.07, 95% CI [−0.25, 0.38]). To investigate the role of neural compression in category-specific coding18, we mapped the cross-phase hippocampal encoding patterns from both conditions into their respective PCA spaces (Fig. 5C). In the HSC condition, the left hippocampus exhibited higher similarity in its low-dimensional information representations from learning to creating (t(38) = 2.41, p = 0.02, R2 = 0.39, 95% CI [0.06, 0.71]; Fig. 5D). This effect remained consistent regardless of whether the learning phase was fitted first and the creating phase subsequently transformed, or vice versa (result. S19). No significant differences were found in the visual control region (t(38) = −0.45, p = 0.65, R2 = 0.07, 95% CI [−0.39, 0.24]), and a 10,000-iteration bootstrap confirmed the hippocampal effect was significantly stronger than in the control region (95% CI [−0.96, −0.01]).
A Trial-wise activity patterns were extracted from the left hippocampus for both higher (HSC, red) or lower (LSC, blue) subsequent creation conditions during the learning and creating phases. These patterns were projected into a shared principal component analysis (PCA) space, and cross-phase pattern similarity was computed based on PCs to quantify the transfer of low-dimensional neural representations between conditions. B Cross-phase similarity as a function of variance explained by retained PCs (70–90%). HSC trials (red) consistently showed higher similarity than LSC trials (blue), reflecting more effective transfer of low-dimensional hippocampal representations. The inset boxplots display average similarity across the 70–90% range, confirming a significant HSC > LSC effect (n = 39 independent participants; two-tailed paired t-test: t(38) = 2.12, p = 0.04, R2 = 0.34, 95% CI [0.002, 0.08]). Curves indicate mean similarity, with shaded areas representing ±s.e.m. Box plots show the median (center), 25th–75th percentiles (bounds of box), and minima and maxima (whiskers). C Trial-wise hippocampal activity patterns from HSC and LSC conditions were separately projected into condition-specific PCA spaces. Within each condition, cross-phase similarity was computed between learning and creating patterns, and the resulting similarity indices were then compared across conditions at the group level. D HSC trials (red) again exhibited higher cross-phase similarity of low-dimensional representations than LSC trials (blue) across the 70–90% explained variance range (n = 39 independent participants; two-tailed paired t-test: t(38) = 2.41, p = 0.02, R2 = 0.39, 95% CI [0.06, 0.71]), plot features as in (B). *p < 0.05. Source data are provided as a Source Data file.
Uncompressed prefrontal representational transfer from learning to creating
We also examined the cross-phase transfer of regional neural encoding in their uncompressed, full-dimensional form (Fig. 6A). The hippocampus, when examined as a whole, yielded no reliable HSC–LSC difference in uncompressed representational transfer (t(38) = 1.00, p = 0.32, R2 = 0.03, 95% CI [−0.16, 0.48]). By contrast, the anterior hippocampus—associated with pattern completion and gist formation66––exhibited significantly greater transfer in HSC trials (t(38) = 2.72, pbonferroni = 0.02, R2 = 0.44, 95% CI [0.10, 0.76]; Fig. 6B, results. S20). Similarly, the left vlPFC showed enhanced learning-to-creating similarity in uncompressed representational space for HSC trials (t(38) = 2.17, p = 0.04, R² = 0.11, 95% CI [0.02, 0.67], Fig. 6C), but not in the compressed, low-dimensional space (t(38) = −0.77, p = 0.45, R² = 0.02, 95% CI [−0.44, 0.19]). These findings suggest that both the vlPFC and anterior hippocampus—regions implicated in abstraction and generalization—may contribute to higher-order learning by directly transferring uncompressed neural codes from learning into creative ideation.
A Schematic demonstration of analysis on cross-phase transfer of uncompressed regional representations from learning to creating. For each participant and region (i.e., hippocampus and vlPFC), Pearson correlations were computed between the representational dissimilarity matrices from the learning and creative phases, separately for higher (HSC) or lower (LSC) subsequent creation conditions. Comparisons reveal differences in cross-phase neural encoding transfer between conditions for each region. B Learning-to-creating pattern similarity (cross-phase representational transfer) in the left anterior hippocampus was significantly higher for HSC (red) than LSC (blue) trials in its uncompressed information representation space (two-tailed paired t-test: t(38) = 2.72, pbonferroni = 0.02, R2 = 0.44, 95% CI: [0.10, 0.76]). Each dot represents a participant (n = 39); gray lines connect paired values; half-violin plots show data distribution; box plots show the median (center), 25th–75th percentiles (bounds of box), and minima and maxima (whiskers). Black dots and error bars represent group means ± s.e.m.; dashed lines indicate mean differences. C The same kind of difference as in B was also found in the left vlPFC region (HSC > LSC, two-tailed paired t-test: t(38) = 2.17, p = 0.04, R2 = 0.11, 95% CI [0.02, 0.67]). D In the compressed representational space, hippocampus-vlPFC representational connectivity for HSC trials was significantly higher than for LSC trials in both the learning (two-tailed paired t-test: t(38) = 5.37, pbonferroni = 8.40 × 10−⁶, R2 = 0.43, 95% CI [0.16, 0.36]) and creating phases (two-tailed paired t-test: t(38) = 4.37, pbonferroni = 1.88 × 10−⁴, R2 = 0.33, 95% CI [0.12, 0.32]), plot features as in (B). E In the uncompressed representational space, HSC trials exhibited higher hippocampus–vlPFC representational connectivity during learning phase (two-tailed paired t-test: t(38) = 4.07, pbonferroni = 0.0004, R2 = 0.30, 95% CI [0.04, 0.15]), but this difference was not significant during the creating phase (two-tailed paired t-test: t(38) = −0.33, pbonferroni = 1.50, R2 = 0.002, 95% CI [−0.08, 0.06]), plot features as in (B). F Schematic of the cross-phase decoding approach. Each axis represents a voxel activation in multi-dimensional feature space, with points indicating activation patterns for higher (HSC, red) and lower (LSC, blue) subsequent creative conditions during the learning and creating phases. A linear classifier is trained to distinguish HSC from LSC in the learning phase and tested on data from the creating phase, assessing the region’s ability to generalize decoding from learning to subsequent creating. G Decoding accuracy within the left vlPFC was positively correlated with an individual’s creative performance (n = 39 independent participants; Pearson’s r = 0.34, two-tailed, p = 0.03). Lines and shaded areas indicate linear regression fits and 95% CIs; marginal density plots show variable distributions. ***p < 0.001; *p < 0.05; n.s., not significant. Source data are provided as a Source Data file. Figures A and F created in BioRender (Zhang, Z. 2025, https://BioRender.com/4fc2m2s; https://BioRender.com/qjudj0y).
Cross-phase hippocampal–vlPFC coupling: alignment and misalignment in compressed and uncompressed representations
To further characterize hippocampal–vlPFC interactions across learning and creating, we examined cross-phase coupling in both compressed and uncompressed representational spaces. In compressed spaces, representational connectivity was significantly higher in HSC than LSC, both during learning (t(38) = 5.37, pbonferroni < 0.001, R² = 0.43, 95% CI [0.16, 0.36]) and during the creating phase (t(38) = 4.37, pbonferroni < 0.001, R² = 0.33, 95% CI [0.12, 0.32]), Fig. 6D. In uncompressed spaces, HSC trials continued to show greater hippocampus–vlPFC coupling during learning (t(38) = 4.07, pbonferroni < 0.001, R² = 0.30, 95% CI [0.04, 0.15]); however, this advantage disappeared in the creating phase and even showed a non-significant trend in the opposite direction (t(38) = −0.33, p = 0.75, R² = 0.002, 95% CI [−0.08, 0.06]; Fig. 6E).
Generalizable decoding in vlPFC depends on compressed hippocampal representations
We next examined whether the vlPFC can act as a generalizable decoder from learning to creating to discriminate HSC and LSC trials. A whole-brain searchlight decoding analysis was conducted using a support vector machine (SVM) classifier, trained on multivoxel activation patterns from the learning phase and tested on the creating phase (Fig. 6F). Statistical significance was determined via permutation testing (Statistical nonparametric Mapping, SnPM13; 10,000 simulations, voxel-wise threshold p = 0.001; cluster-wise FDR correction p < 0.05). This analysis identified a significant cluster in the left vlPFC (MNI: x = –38, y = 16, z = 28; k = 18), where cross-phase decoding accuracy was significantly above chance (M = 54.28%, SD = 0.04). Notably, cross-phase decoding accuracy within this vlPFC region (10 mm spherical region of interest, ROI) was found to be positively correlated with subsequent creative performance (r = 0.34, p = 0.03; Fig. 6G), suggesting a direct link between the cross-phase generalizable decoding function of left vlPFC and post-learning creative output. To determine whether this generalizable decoding capacity of vlPFC depends on its access to low-dimensional hippocampal representations formed during learning, we retested this decoding analysis after regressing out the compressed hippocampal encoding and found that, once this hippocampal component was removed, cross-phase classification accuracy in the left vlPFC was no longer significantly above chance (M = 51.19%, SD = 0.04, p > 0.05), suggesting that hippocampal encoding compression supported by the vlPFC, in turn, enables vlPFC-based cross-phase generalization. Control analysis showed the cross-phase decoding performance within the left hippocampus itself did not exceed the chance (M = 48.99%, SD = 0.04, p > 0.05).
Discussion
The present study systematically investigated how the hippocampus and prefrontal cortex encode exemplar knowledge and readily generalize it to support subsequent creative ideation. Behaviorally, exposure to creative exemplars robustly enhanced later creative performance (Exp. S1a–c). Neurally, higher-order exemplar-learning that facilitated subsequent creating was marked by reduced representational dimensionality in the left hippocampus, coupled, and potentially driven, by increased hippocampal–vlPFC connectivity (Exps. 1 and 2). Cross-phase analyses further revealed that low-dimensional hippocampal representations and full-dimensional vlPFC representations transferred more from learning to creating in the HSC condition. Notably, the vlPFC exhibited above-chance cross-phase decoding accuracy in distinguishing HSC from LSC trials, and this decoding ability was positively correlated with creative performance and was likely supported by hippocampal encoding compression during learning (Exp. 2). Additionally, supplemental experiments revealed that reduced hippocampal dimensionality and enhanced hippocampal–prefrontal connectivity were independent of remembering or forgetting of the exemplars (Exp. S2), while forgetting instructions during exemplar-learning (Exp. S3) and incubation delays between learning and creating (Exps. S5–S7)—but not diminished episodic recollective experiences (Exp. S4)—contributed to improved creative output. Our main findings were reproducible with both textual (Exp. 1) and pictorial (Exp. 2) stimuli and remained robust after controlling for potential confounds. Together, these findings suggest a neurocognitive mechanism by which the hippocampal and prefrontal systems orchestrate to construct and transfer neural representations of creative exemplars to support subsequent creation.
The behavioral results revealed multiple facets of higher-order learning. First, Exp. 1 and Exp. S1a–c confirmed that creative exemplar-learning significantly improved subsequent creative performance, compared with multiple control conditions. This finding is consistent with previous studies showing that creative exemplar-learning facilitates self-generated creativity2,3,4, and argues against a purely interference-based account—which would predict an overall detrimental effect of exemplar-learning on subsequent creativity in Exp. 1, with effects expected to be more prominent in LSC than in HSC. Second, Exp. 1 also found that HSC was associated with greater forgetting of learned exemplars, and Exp. S3 found that participants instructed to forget during exemplar-learning outperformed those instructed to remember—aligning with prior studies linking exemplar forgetting to enhanced creativity53. Further analyses indicated that prediction errors induced by exemplar creativity positively predicted both imitation and memory for learned exemplars, but did not directly influence creative performance. Nevertheless, given the negative correlation between exemplar imitation and creative output, the prediction errors may ultimately shift the trade-off between imitation and creation toward imitation, thereby constraining creative ideation. These findings illustrate the “double-edged sword” effect of creative exemplar-learning: while it stimulates creative ideation, it can also induce mental sets that constrain originality4,71,72. In this context, weakening memory details may be beneficial. By contrast, Exp. S4 showed that, for successfully retrieved exemplars, whether episodic details were retained or lost did not affect creative output, suggesting that mere loss of memory details is insufficient to boost creativity. However, Exps. S5–S7 found that introducing an incubation delay between learning and creating—particularly when combined with a prolonged, low-load working memory task—further enhanced creative performance, underscoring the role of incubation-induced optimization of exemplar memory in higher-order learning.
Our neuroimaging results shed light on how the brain mediates the “double-edged sword” effects of exemplar-learning on subsequent creativity. Specifically, we show highly abstracted exemplar encodings—manifested as lower-dimensional hippocampal representations and enhanced hippocampal–vlPFC connectivity—optimize the balance between retaining essential features of exemplar-learning and avoiding overfitting to specific learning experiences. The observation of lower-dimensional hippocampal encoding in higher-order exemplar learning aligns with previous evidence that neural compression supports memory generalization in domains such as spatial reasoning73, concept learning18, associative inference67,74, and reversal learning75. Our results extend this mechanism to creative ideation. Notably, this effect was lateralized to the left hippocampus, a lateralization that appears to be independent of the verbal (Exp. 1) or graphic (Exp. 2) presentation of exemplars. A prior model of hippocampal lateralization in creative cognition proposes that the right hippocampus is involved in the emotion-driven processing of novel features in creative ideas, whereas the left hippocampus is responsible for the more precise cognitive integration of novelty with relevance or appropriateness, often in conjunction with the prefrontal cortex76. Therefore, the left hippocampus may facilitate subsequent creativity through the evaluation and integration of the novelty and appropriateness of exemplars during the learning process. Reducing the representational dimensionality of hippocampal encoding of learned exemplars may weaken nonessential exemplar details24,25, thereby minimizing potential interference during creative tasks61, while preserving the essential features of creative exemplar-learning. Moreover, enhanced hippocampal–vlPFC connectivity in higher-order learning is consistent with previous studies highlighting the vlPFC’s role in semantic selection77, abstract rule construction78, and hippocampal–prefrontal interactions in extracting cross-event patterns and assimilating new concepts into memory79,80. We speculate that these neural indices reflect the extraction of high-level heuristic rules from creative exemplars, facilitating creative ideation by enabling the formation of adaptable associative knowledge networks and by capturing essential similarities across diverse learning episodes34,81.
Additional consideration suggests that these two neural markers of higher-order learning do not simply reflect weakened or forgotten exemplar memories. First, lower-dimensional hippocampal representations and enhanced hippocampus–vlPFC connectivity found in HSC were independent of univariate hippocampal activation that is typically associated with successful memory encoding82. Second, while Exp. 1 (targeting SCE of HSC–LSC difference) revealed significant HSC–LSC differences in both neural indices; no such effects were observed in Exp. S2 (targeting the SME of remember-forget difference). Although the lack of hippocampal representational dimensionality differences in SME appears inconsistent with the previous study linking higher hippocampal encoding dimensionality with successful associative memory20, such discrepancies may result from variations in task parameters and trial counts. Notably, previous studies employed memory tasks requiring fine-grained, item-specific distinctions (e.g., face-name pairs) that favor higher-dimensional encoding59, whereas our paradigm—emphasizing creative combinations of objects and novel uses—relies on semantic integration and schema abstraction, likely reflected by enhanced global hippocampal pattern similarity rather than increased encoding dimensionality36,60. Third, restricting the HSC vs. LSC contrast to successfully recalled exemplars confirmed persistent differences in hippocampal dimensionality and hippocampal–prefrontal connectivity. Collectively, these findings suggest that exemplar-learning exerts dual effects on creative ideation: providing essential strategic knowledge for creativity while potentially constraining originality through overreliance on exemplars. Forming generalizable, abstract representations—via lower-dimensional hippocampal encoding and enhanced hippocampal–prefrontal connectivity—may offer an optimal solution, balancing insight acquisition with flexibility, and enabling the transformation of learned experiences into novel creative outcomes83.
The finding that compressed, lower-dimensional hippocampal representations showed enhanced cross-phase transfer from learning to creating in HSC trials further supports the proposed mechanism. This aligns with prior evidence that hippocampal integration facilitates concept generalization, either by constructing prototypes from exemplars81 or by abstracting shared structures across events to support rapid inference84. Additionally, both the anterior hippocampus and vlPFC—regions involved in pattern completion, gist formation66,85,86, and rule abstraction78—exhibited greater cross-phase transfer in HSC, but in the form of uncompressed representations. This indicates that the exemplar representations carried forward from learning to creation were abstracted rather than verbatim. Notably, the vlPFC, which encodes higher-level AUT-related information, also demonstrated above-chance cross-phase decoding accuracy in distinguishing HSC from LSC trials, consistent with previous findings that left prefrontal patterns decode the integration of overlapping memories and predict inference performance87,88,89. Given that creative ideation depends on the flexible manipulation of prior experiences90, the vlPFC’s role in semantic retrieval is critical for both exemplar-learning and generative creativity, supporting its function as a generalizable cross-phase decoder. Further analysis revealed that this decoding function depended on low-dimensional hippocampal encoding during learning, suggesting that compressed representations are selectively accessed or transformed by the vlPFC and subsequently reinstated during creative ideation.
Moreover, hippocampal–vlPFC coupling analyses revealed a dissociation between compressed and uncompressed representational spaces. In the compressed space, hippocampal–vlPFC coupling was higher in HSC during both learning and creating, suggesting that higher-order learning relies on the efficient utilization of abstracted, task-relevant features represented by this compressed coupling. In contrast, in the uncompressed space, hippocampal–vlPFC coupling was higher in HSC during learning but not during creating. One possible interpretation is that enhanced coupling during learning facilitates effective abstraction of exemplar representations—particularly within the hippocampus—whereas reduced coupling during creating may be advantageous for ideation, as it allows each region to contribute more independently. Specifically, the hippocampus supports associative memory processes, while the vlPFC mediates cognitive control and semantic selection48,77,91, two complementary functions essential for divergent thinking.
One limitation of our study is that we investigated the neural correlates of higher-order learning primarily from an output-based perspective, retrospectively categorizing exemplar-learning trials by subsequent creative performance, thus limiting causal inferences about how exemplar properties or learning intentions influence creativity. Future studies could directly manipulate exemplar creativity (e.g., highly vs. less creative) and learning intentions (e.g., to create vs. to memorize) to test their effects. A second limitation concerns the relatively short duration allowed for creative idea generation (12 or 16 s). Although this design was intended to capture the immediate influence of exemplar-learning on subsequent creation, it may restrict creative output, as high-quality ideas often require extended reflection92. Future research should examine how creative ideation unfolds over longer durations and how this interacts with neural encoding during exemplar-learning. A third limitation is the narrow range of exemplar creativity—used to maintain material homogeneity—which reduced variability in prediction errors. Combined with the low imitation levels, likely driven by instructions discouraging imitation, this may limit the generalizability of findings on the trade-off between creativity and imitation. Future work should introduce more varied exemplars, manipulate imitation demands, and directly assess prediction error dynamics to reveal their impact on subsequent creative ideation.
In conclusion, our study shows that functional coordination between the hippocampus and prefrontal cortex is critical for higher-order exemplar-learning that predicts enhanced subsequent creative ideation. This is reflected in reduced hippocampal dimensionality and increased hippocampal–vlPFC connectivity during exemplar-learning, and the transfer of these neural encoding patterns into the creating phase. These processes likely represent the abstraction of strategies that optimize the balance between learning from exemplars and avoiding overfitting on learning, thereby facilitating creative performance. Our findings highlight a distinct hippocampal–prefrontal functional organization that supports information representation and knowledge generalization in higher-order learning, setting it apart from mechanisms underlying ordinary memory formation. This dissociation between the neural bases of SME and SCE offers new insights into the neurocognitive foundations of creativity and suggests potential avenues for enhancing creative learning in educational settings.
Methods
Participants
All participants were right-handed, native Chinese speakers with normal or corrected-to-normal vision. Sex was determined by self-report. For the fMRI studies (Exps. 1, 2, and S2), initial recruitment included 30, 40, and 27 individuals, respectively. In Exp. 1, four participants were excluded due to equipment malfunction (fMRI data not available), and three others were excluded for extremely poor performance in the creating phase (defined as more than two SD below the group mean). In Exp. 2, one participant withdrew midway due to being physically sick. In Exp. S2, data from four participants were excluded from analysis due to poor performance in the cued-recall test (remembering fewer than ten items out of 100 trials). The final sample sizes were as follows: Exp. 1, n = 23 for fMRI data (15 females, mean age = 23.17 ± 1.64 years) and n = 27 for behavioral data (16 females, mean age = 23.33 ± 1.57 years); Exp. 2, n = 39 (24 females, mean age = 22.13 ± 3.14 years); Exp. S2, n = 23 (16 females, mean age = 22.87 ± 1.92 years). No participant took part in more than one experiment. All received 150 RMB for participation. For the behavioral Exp. S1a–c, S3–S7, the sample sizes were as follows: Exp. S1a, n = 36 (23 females, mean age = 21.42 ± 0.73 years); Exp. S1b, n = 40 (20 females, mean age = 21.20 ± 2.08 years); Exp. S1c, n = 34 (15 females, mean age = 21.97 ± 2.38 years); Exp. S3, n = 33 (23 females, mean age = 23.30 ± 2.76 years); Exp. S4, n = 35 (28 females, mean age = 22.00 ± 2.98 years); Exp. S5, n = 38 (23 females, mean age = 22.70 ± 2.61 years); Exp. S6, n = 32 (23 females, mean age = 22.53 ± 2.29 years); and Exp. S7, n = 36 (23 females, mean age = 22.16 ± 2.50 years). No participants overlapped across experiments. Compensation ranged from 30 to 60 RMB depending on study duration. No sex- or gender-based analysis was conducted, as the study was not designed or powered to detect sex-related effects.
Experimental procedures
The procedures of Exp. 1
Experimental materials comprised 100 creative exemplars of everyday objects, drawn from a previously established AUT dataset, and presented as item-use pairs (e.g., “whisk-candlestick”). Exemplars were selected based on pre-experimental ratings to ensure moderate-to-high creativity (M = 3.43, SD = 0.27) and novelty (M = 3.85, SD = 0.33), while maintaining high understandability (M = 4.53, SD = 0.28). All ratings were made on a 5-point scale (method. S2). This selection was guided by previous showing that exposure to creative exemplars robustly enhances subsequent creative performance and associated neural activity compared to common materials1,3. To minimize material-related confounds, between-item variability was carefully controlled, with coefficients of variation for creativity (CV = 0.08), novelty (CV = 0.09), and understandability (CV = 0.06) all below 0.1. An additional 20 non-creative exemplars (M = 2.20, SD = 0.46) served as filler items in the learning phase only, preventing participants from exclusively encoding creative prototypes. The experiment comprised three consecutive phases, each preceded by standardized instructions and practice trials.
Exemplar-learning (phase 1): The phase 1 involved an event-related fMRI design distributed over four runs. Each run comprised 25 creative exemplars and 5 uncreative fillers, presented in random order. During each trial (Fig. 2A), an object-usage pair was displayed for 6 s. Participants were asked to attentively comprehend the exemplar and judge its creativity via button press (“yes” for creative, “no” for uncreative). Importantly, participants were not informed about the subsequent self-generated creating (phase 2) or cued-recall (phase 3) tasks to avoid any relevant attentive processing. Each decision was followed by a jittered fixation (4–6 s) to maintain attention and prepare for the next trial.
Self-generated creating (phase 2): After the exemplar-learning phase, participants were taken to a quiet room for behavioral testing. A surprise AUT was administered: in each trial, a random object (from the 100 learned creative exemplars) was presented for 12 s, during which participants were asked to generate and verbally report novel and original uses for the object—distinct from any previously learned exemplars. Responses were audio-recorded, and the experimenter also transcribed them. Trials were separated by a 2 s fixation. After the task, participants confirmed the accuracy of the recorded responses.
Cued-recall (phase 3): In phase 3, participants completed a cued-recall test for the exemplars learned in phase 1. Object names from the creative object-use pairs were presented in random order as cues (maximum 4 s each). For each cue, participants indicated whether they could recall the associated creative use; if so, they typed their answer; otherwise, the next object was presented. Each trial was separated by a 2 s fixation. Notably, the self-generated creating task (phase 2) preceded this recall test to allow an accurate assessment of the impact of exemplar-learning on creative output independently of memory retrieval effects.
The procedures of Exps. S1–S7
Exp. S1a–c
To systematically examine the effect of creative exemplar-learning on subsequent creative ideation, we conducted three control experiments (Exp. S1a–c). In Exp. S1a, participants completed the AUT with the same set of 100 target objects as in Exp. 1, but without any prior exemplar-learning. In Exp. S1b, a separate group learned the conventional uncreative uses of the 100 target objects as adopted in Exp. 1 (e.g., “newspaper–reading news”) in random order before performing the AUT. In Exp. S1c, another group learned 200 single words derived from the creative object-use pairs in Exp. 1 (e.g., “newspaper–lampshade” presented separately as “newspaper” and “lampshade”, randomly mixed with other words taken from other pairs), each displayed for 3 s during which participants judged whether it was a real word, followed by the same creative ideation task as in phase 2 of Exp. 1. Across all three experiments, the creative ideation procedures were identical to those in Exp. 1, and none included a cued-recall phase.
Exp. S2
This experiment aimed to identify the neural correlates of successful memory encoding for creative exemplars—referring to as SME82. The exemplar-learning materials and procedures were identical to those in phase 1 of Exp. 1. Following the learning phase, participants exited the MRI scanner and were taken to a testing room, where they first completed a cued-recall task for the learned exemplars (identical to phase 3 of Exp. 1). Subsequently, they performed a recognition memory test, in which 220 object–usage pairs were presented sequentially for old–new memory judgments. The test set comprised 100 targets (objects paired with their original learned creative uses), 100 lures (objects paired with creative uses that were slightly different from the correct answer), and 20 entirely new object–usage pairs. For each item, participants indicated whether it matched the originally learned exemplar and rated their confidence on a five-point scale (1 = low confidence, 5 = high confidence; see Fig. S9A).
Exp. S3
This behavioral experiment utilized a directed forgetting paradigm to detect the relationship between subsequent forgetting of learned exemplars and creative ideation for the same target object. Participants were presented with 52 creative exemplars (object-use pairs) of moderately high creativity, selected from the same AUT Creative Uses Dataset as in Exp. 1. For each participant, half of the items were randomly assigned to the directed remembering (DR) condition and the other half to the directed forgetting (DF) condition. During the exemplar-learning phase, each object-use pair was displayed for 4 s, followed by a 0.3 s blank screen. A cue indicator (“√“ for remember; “ד for forget) then appeared for 2 s, instructing participants either to intentionally remember or forget the just-presented exemplar. In the test phase, the names of the 52 target objects were presented in random order. For each object, participants first attempted to recall the previously learned creative alternate use (cued recall; 4 s per item) and then generated their own creative ideas for using the same object (self-generated creation; 8 s verbal response). Responses were recorded, and the task structure is illustrated in Fig. S10A.
Exp. S4
This experiment examined whether the presence or absence of recollective experiences during exemplar-learning influenced subsequent creative ideation. Exp. S4 comprised three phases: exemplar-learning, exemplar recognition with Remember/Know (R/K) judgments, and self-generated creation. During the exemplar-learning phase, participants viewed the same 100 creative exemplars (object–use pairs) as in phase 1 of Exp. 1 in random order. Each exemplar was displayed for 4 s, with a 1 s inter-trial interval, during which participants judged its creativity. Following a 7-min numerical distraction task, participants completed the recognition phase, which consisted of 200 trials in which object–use pairs (100 targets and 100 lures) were presented sequentially in random order. For each trial (maximum 6 s), participants judged whether the presented pair matched an exemplar learned earlier. If they responded “yes,” they were prompted to make an R/K judgment: an “R” (remember) response indicated retrieval of vivid episodic details (e.g., visual appearance, thoughts, or feelings at encoding), whereas a “K” (know) response indicated recognition without specific episodic content65. This secondary judgment was also allotted up to 6 s, separated by a 0.5 s interval (Fig. S10C). Finally, participants were presented with the 100 target objects and instructed to generate their own creative uses following the same procedure as in Exp. 1.
Exps. S5–S7
Exps. S5–S7 explored the effect of learning-creating incubation delay on creative ideation. Across these three experiments, procedures and materials were closely matched, with critical modifications described below.
Exp. S5 involved 40 moderately high-creative exemplars that served as the exemplar material. The set was divided into four sub-groups, each assigned to one of four conditions: (1) exemplar-learning with incubation (EI); (2) exemplar-learning without incubation (E); (3) incubation without exemplar-learning (I); (4) a control condition (C), with 10 items per group. Statistical analysis confirmed no significant differences among the four sub-groups in novelty, appropriateness, or creativity (all ps > 0.2). Assignment of materials to conditions was counterbalanced across participants, and the presentation order of conditions was determined using a Latin square design. Each trial in all four conditions followed a classic “pretest–incubation–posttest” structure. The trial began with an initial AUT (AUT1), in which participants generated as many creative uses as possible for the target object, without a time limit. Participants then experienced one of the four experimental conditions before completing a second AUT (AUT2), in which they again generated creative uses for the same target object within 20 s.
(1) EI condition (exemplar-learning + incubation delay): Before AUT2, participants learned a creative exemplar (object-usage pair) for 4 s, with instructions to attend carefully to its content. This was followed by a 40 s incubation period in which numbers were presented sequentially at a rate of one per second, interspersed with a 1 s inter-trial interval. During this period, participants performed a 0-back working memory (WM) task, pressing the enter key whenever a number other than “3” appeared. The incubation phase was designed to provide a brief cognitive distraction, thereby facilitating incubation and potentially enhancing creative performance in AUT2.
(2) E condition (exemplar-learning only): This condition mirrored the EI condition, except that the 40 s incubation task was omitted. Participants learned the creative exemplar for 4 s and then proceeded immediately to AUT2, allowing assessment of the effect of exemplar-learning in the absence of incubation.
(3) I condition (incubation delay only): Participants completed the same 40 s 0-back WM incubation task as in the EI condition, but without any prior exemplar-learning. This condition isolated the effect of incubation delay on creative performance, independent of exemplar-learning.
(4) C condition (control): Neither exemplar-learning nor incubation was included. Instead, a 6 s fixation period separated AUT1 and AUT2, providing a baseline for evaluating the independent and combined effects of exemplar-learning and incubation (Fig. S11A).
Exp. S6
Exp. S6 utilized the same materials and procedures as Exp. S5 but compared only two conditions: E and EIlong. The E condition was identical to the E condition in Exp. S5, while the EIlong condition was identical to the EI condition in Exp. S5 except that the incubation interval (0-back WM task) was extended from 40 to 180 s, allowing examination of the effect of prolonged incubation on creative performance (because no significant difference was found between the E and EI conditions in Exp. S5; Fig. S11C).
Exp. S7
Exp. S7 further examined the impact of WM load during the incubation period by expanding on the EIlong condition from Exp. S6, with the hypothesis that high vs. low WM load would respectively produce stronger interference or greater incubation-induced optimization of learned exemplar knowledge. Two incubation conditions were implemented to manipulate WM load. In Low-load incubation (LI), participants performed a 0-back WM task, the same as in the EIlong condition of Exp. S6. In High-load incubation (HI), participants engaged in a high-load 2-back WM task. Here, numbers were presented sequentially, and participants were required to compare each number with the one presented two trials earlier, responding when the current number matched the number two positions back (Fig. S11E).
The procedures of Exp. 2
The procedure of Exp. 2 closely paralleled that of Exp. 1, comprising three phases: exemplar-learning (phase 1), self-generated creating (phase 2), and cued-recall (phase 3). The key distinction was the use of pictorial materials to illustrate creative uses of everyday objects, with both the learning and creating phases conducted during MRI scanning (Fig. 4A). This design enabled in-depth investigation of hippocampal and prefrontal representations acquired during exemplar-learning and their transfer to creative ideation. To address a limitation of Exp. 1—in which the creating phase was conducted outside the scanner due to technical challenges in recording speech93—Exp. 2 employed a sparse-sampling fMRI protocol94 during the creating phase to enable clear in-scanner audio recording of participants’ verbal responses. During phase 1, participants learned a list of pictorial creative exemplars (object–use pairs) from the established database (method. S3), which were rated highly for novelty (M = 5.78, SD = 0.58), usefulness (M = 5.41, SD = 0.57), and creativity (M = 5.13, SD = 0.54) on a 1-7 point rating scale. To prevent habituation, additional non-creative pictorial exemplars were included but not analyzed. Each trial presented an object–use pair for 6 s, during which participants judged its creativity (“yes”/“no” via keypress), followed by a jittered fixation (4–6 s). In phase 2, participants generated creative ideas for the previously learned objects while undergoing fMRI. A sparse-sampling protocol (2 s scanning + 5 s silent gap) ensured accurate capture of verbal responses: each object was displayed for 16 s, during which participants silently conceived creative uses, followed by a 5 s yellow-screen cue (synchronous with scanning silence) during which they verbally reported their ideas. Trials were separated by a 7 s fixation cross. Finally, in phase 3, participants completed a recall test for the learned exemplars, using the same procedure as in Exp. 1.
Image acquisition and preprocessing
Participants in Exps. 1, 2, and S2 underwent MRI scans at the Center for Biomedical Imaging Research, Tsinghua University, using a 3T Philips Achieva 3.0T TX MRI scanner with a 32-channel head coil. Foam padding was employed to minimize head movement during scans. The acquisition parameters for T1*-weighted anatomical images included: 180 slices encompassing the whole brain, repetition time (TR) = 7.65 ms, echo time (TE) = 3.73 ms, flip angle (FA) = 8°, field of view (FOV) = 230 mm × 230 mm, voxel size = 1 mm × 1 mm × 1 mm, 1 mm thickness. For T2*-weighted functional images, the parameters were: TR = 2000ms, TE = 35 ms, FA = 90°, FOV = 200 mm × 200 mm, 64 × 64 matrix, voxel size = 2.5 × 2.5 × 4 mm, and 30 slices covering the whole brain with 4 mm thickness and no inter-slices gap. Phase 2 of Exp. 2 employed a sparse sampling acquisition technique, introducing 5-s silent intervals between the acquisitions of 2-s MR volumes. This modification facilitated the collection of overt participant responses during phase 2 without the interference of scanner noise. Visual stimuli were delivered using E-prime 2.0 software and projected onto a screen within the participant’s view via a mirror system.
fMRI data preprocessing was conducted using fMRIPrep 21.0.0 (https://fmriprep.org/en/stable), a software built on Nipype 1.6.1 (https://nipype.readthedocs.io/en/latest/). This comprehensive preprocessing pipeline addressed several key aspects of data correction and standardization: it corrected for slice timing discrepancies, realigned all images to a mean reference image, and registered the data to the MNI152 template at a resolution of 2 mm isotropic voxels. Spatial smoothing was intentionally omitted to preserve the original spatial properties of the data. For a detailed account of the preprocessing steps and results, please refer to the HTML reports generated by fMRIPrep, accessible via the fMRIPrep Documentation.
Data analysis
Behavioral data analysis
Creativity assessment
The creativity of self-generated AUT ideas was evaluated using the Consensus Assessment Technique95. Three independent raters—master’s students specializing in creative psychology research with prior experience evaluating AUT responses—independently assessed each idea offline. Ratings were based on both the novelty and appropriateness of the idea in determining its degree of creativity. In Exp. 1, participants generated a total of 3430 ideas in phase 2 across 100 target objects. Each idea was rated, yielding a high inter-rater reliability (ICC) of 0.85 across the three raters. For the other experiments, ICCs were as follows: Exp. 2 = 0.83, Exp. S1a = 0.80, Exp. S1b = 0.74, Exp. S1c = 0.88, Exp. S3 = 0.82, Exp. S4 = 0.81, Exp. S5 = 0.88, Exp. S6 = 0.82, and Exp. S7 = 0.93.
HSC/LSC trial classification (Exps. 1 and 2)
For each trial in phase 2, the creativity score was calculated as the mean rating across the three raters. Trials in which participants either failed to produce a response or simply repeated the learned exemplar were assigned a creativity score of zero. For trials with one or more creative responses, the highest-scoring idea was used as the trial’s creativity index, reflecting the common practice of taking a person’s most creative work as representative of their creative ability in real-world contexts. An individual-specific median split was then applied to each participant’s trial list in the exemplar-learning phase, classifying trials as higher subsequent creation (HSC; above-median creativity score) or lower subsequent creation (LSC; below-median creativity score). To further ensure the robustness of results, we also conducted analyses using the mean creativity scores across all participants as an alternative criterion for HSC/LSC categorization.
Detailed behavioral data analysis
Comprehensive procedures and statistical approaches for behavioral data analysis across all experiments are provided in the Supplementary Information. Specifically, see results. S1–S3 and methods. S1 for Exp. 1; result. S4 for Exp. S1a–c; results S11–S13 for Exps. S3–S7; and result. S14 for Exp. 2.
fMRI data analysis
Single-item response estimation (Exps. 1 and 2)
To investigate the fixed effects of each regressor across participants, general linear model (GLM) analyses were utilized. We adopted a least squares all (LSA) model, which delineates each trial of each condition of interest as a distinct entity within the design matrix. Researchers have advocated for the superiority of LSA over other single-item estimation methods, such as least squares separate (LSS), particularly in scenarios of substantial trial variability96. Given the variability inherent in the stimuli presented—novel usages of different objects—the LSA method was selected for its robustness. Regressors were convolved with the canonical hemodynamic response function to model the expected physiological changes. Each GLM also incorporated six head-motion parameters derived from head-motion correction as nuisance regressors. To mitigate low-frequency signal drift, a 128-s high-pass filter was applied to each run.
Definition of ROIs (Exps. 1 and 2)
Our analysis focused on the encoding features of the hippocampus and its interaction with the inferior frontal gyrus (IFG; ventrolateral prefrontal cortex, vlPFC) in fostering creative ideation. Both the hippocampus and IFG were delineated using the AAL atlas64. To segment the hippocampus into anterior and posterior sections, we applied a MNI coordinate-based segmentation method. This division was based on the location of the uncal apex within the MNI space, specifically at a y-coordinate of −21 mm97. Additionally, vision-related brain areas were identified utilizing the Neurosynth meta-analytic database by searching for the keyword “vision”98. The association test map for “vision” from Neurosynth was thresholded at a q < 0.01 FDR-corrected and obtained as the mask to generate the mask for these areas.
Estimation of low-dimensional representation (Exps. 1 and 2)
We hypothesized that hippocampal representational dimensionality reduction during exemplar-learning correlates with enhanced performance in subsequent creation. To quantify the extent of this neural compression, we measured the representational dimensionality of hippocampal neural activity patterns within PCA space, based on unsmoothed functional data. Activation patterns for each exemplar-learning trial were estimated individually, and activation vectors spanning all voxels within the hippocampus were extracted for both HSC and LSC trials. As shown in Fig. 3A, representational dimensionality for the HSC and LSC conditions was estimated separately using the following procedure: To ensure robust dimensionality estimation—given that the number of HSC and LSC trials for each participant may differ slightly (k1 ≠ k2)—a 1000-sample bootstrap procedure was employed. An equal number of trials, determined by the smaller trial count across conditions, were randomly sampled from each condition to construct an n × p (voxels × trials) activity matrix. PCA was then performed to estimate the number of principal components (PCs) required to explain each variance threshold from 70% to 80% in 1% increments. The number of PCs at each threshold was averaged across bootstrap samples, and the final dimensionality estimate for each condition was obtained by averaging across the 70–80% explained variance range99. Dimensionality estimates were compared between HSC and LSC conditions using a group-level paired t-test. Given that 80% explained variance remains a widely used criterion in neural dimensionality studies56,57—for its effective balance between capturing task-relevant signals and minimizing noise—we also estimated hippocampal representational dimensionality as the number of PCs required to explain 80% of the variance in the activation patterns. This estimation was performed using the same bootstrap approach described above to ensure statistical robustness.
Estimation of global pattern similarity (Exps. 1 and 2)
To assess the relationship between the neural representations of different exemplars, pairwise Pearson correlations were calculated. For each exemplar i (denoted as Ai), its activation pattern was compared to the activation pattern of each other exemplar j in the set of all exemplars (ktotal) learned during phase 1. This method was systematically repeated for each exemplar, providing a comprehensive matrix of correlation coefficients, according to the formula:
To normalize the distribution of similarity scores, we transformed these values into Fisher’s z-scores. These transformed scores were then averaged to calculate the global pattern similarity value for each trial type (HSC and LSC). These global pattern similarity values for each trial type were then averaged and statistically contrasted at the individual level to investigate differences in neural representation. For group-level analysis, we utilized a random-effects model. This approach was chosen to accommodate inter-subject variability, ensuring robust statistical inference across the study population.
Psychophysiological interaction (PPI) analysis (Exp. 1)
We conducted a PPI analysis100 with the left hippocampus as the seed region. The analysis workflow commenced with smoothing each subject’s preprocessed fMRI data using a Gaussian kernel with a full width at half-maximum (FWHM) of 6 mm. The BOLD signals were then extracted from the seed region to construct the PPI design matrix. This matrix included the seed region’s time series (physiological variable), the task-related time series (psychological variable), and their interaction term. The interaction term was derived by element-wise multiplication of the seed region’s time course with the task time course, representing a specific task condition (HSC vs. LSC), and subsequently convolved with the canonical HRF. Additionally, a high-pass filter with a cut-off of 128-s was applied, and six motion parameters obtained from realignment were incorporated as nuisance regressors. The PPI model estimation and statistical map generation were then performed for each subject individually. These subject-level statistical maps were entered into a second-level group analysis. This correction was applied with a cluster-wise FWE correction set at p < 0.05 and a cluster-forming threshold at p < 0.001, using SPM 12.
Representational connectivity analysis (Exps. 1 and 2)
To evaluate the degree to which the left hippocampus and the left IFG/vlPFC encode similar information, we compared the RDMs of these two regions. RDMs were constructed separately for trials under HSC and LSC conditions in both the hippocampus and IFG (anatomical ROI). The similarity between the RDMs for the two regions was quantified by calculating the Pearson correlation coefficient (r) between the lower triangles of the respective matrices (see Fig. 3E). For each participant, correlation coefficients were converted to Fisher’s z-scores to normalize the distribution. Group-level t-tests were then performed to assess differences in representational similarity between the HSC and LSC conditions.
Regression analysis (Exp. 1)
We employed a regression analysis using the robust regression toolbox (available at https://github.com/canlab/RobustToolbox) to identify voxels in the neocortex that are coupled with the left hippocampus and contribute to neural compression. The searchlight-based representational connectivity analysis yielded searchlight maps for two conditions (HSC and LSC). We generated representational connectivity difference maps by subtracting LSC maps from HSC maps. Within the left hippocampus, the representations dimensionality score for the LSC condition was subtracted from the representations dimensionality score for the HSC condition, each calculated at 80% explained variance, resulting in the representations dimensionality difference score. We then regressed the voxel values from the representational connectivity difference maps onto these representations’ dimensionality difference scores. Leveraging the representational connectivity analysis outcomes, we applied a small volume correction (SVC) procedure, focusing on the bilateral prefrontal, and subsequently extended this analysis to encompass the entire brain. Significant clusters were identified using voxel-wise FDR correction (q < 0.05), which was the default setting for multiple comparisons correction in the robust regression toolbox.
Comparison of cross-phase low-dimensional representational transfer (Exp. 2)
We used PCA to analyze the low-dimensional representational pattern similarity between learning and creating phases. Let the neural data matrix be:
where n is the number of voxels and p is the number of trials within the left hippocampus. PCA decomposes X as
where \({{{\bf{V}}}}\in {{{{\bf{R}}}}}^{p\times k}\) contains the first k principal axes (loadings), and \({{{\bf{Z}}}}\in {{{{\bf{R}}}}}^{n\times k}\) contains the corresponding PC scores, ordered by explained variance. The number of retained PCs (k) was chosen to capture a fixed proportion of explained variance (70–90%).
(1) Within a unified PCA-space: We combined activity patterns of HSC and LSC trials within the left hippocampus during both the learning and creating phases into a single matrix Xall, PCA was performed on Xall:
From the resulting score matrix Z, we extracted trial-wise PC scores for HSC-learning, HSC-creating, LSC-learning, and LSC-creating. For each PC i, Spearman correlations were computed between learning-phase and creating-phase scores within each condition. The similarity score was defined as
where LHSC/LSC\(\left(:,i\right)\) and CHSC/LSC\(\left(:,i\right)\) denote the PC scores of learning and creating trials for condition HSC or LSC, respectively. Loading similarity scores were computed across explained-variance thresholds (70–90%) and compared between HSC and LSC using a paired t-test (Fig. 5A).
(2) Within category-specific PCA-space: Based on the category-specific view of neural representation abstraction18, we also utilized PCA to project the activation patterns of HSC and LSC conditions into their respective PCA spaces, then calculated the pattern similarity within each condition and subsequently compared. For each condition (HSC or LSC), hippocampal activation during the learning phase (X1) was decomposed as
The first k PCs were retained, yielding a score matrix:
where each column L:,i represents the trial-wise scores of the ith PC in the learning phase.
The creating phase data (X2) were then projected onto the same set of principal axes (V1) that had been derived from the learning phase, resulting in:
where each column C:,j represents the trial-wise scores of the jth PC in the creating phase.
To quantify low-dimensional representational transfer from learning to creating, we computed neural pattern similarity (NPS) scores. Specifically, for each PC i from the learning phase, we calculated the correlation between its score vector L:,i and the score vectors of all k PCs from the creating phase (C:,j):
This procedure ensures that each learning-phase PC is not only compared to its “matched” PC in the creating phase, but also to all possible PCs, thereby capturing a more generalizable measure of exemplars’ low-dimensional representational overlap between learning and creating. Finally, the overall learning–creating similarity index (LC-NPS) was obtained by averaging across the k PCs:
Thus, LC-NPS reflects the mean cross-phase similarity between the low-dimensional representations formed during learning and those engaged during creating. LC-NPS values were calculated for multiple explained-variance thresholds (70–90%), and the averaged similarity scores across thresholds were statistically compared between HSC and LSC conditions using paired t-tests (Fig. 5C).
Comparison of cross-phase full-dimensional representational connectivity and transfer (Exp. 2)
To examine the cross-phase differences in representational connectivity between the hippocampus and IFG/vlPFC under HSC and LSC conditions, we conducted cross-phase representational similarity analysis in uncompressed (full-dimensional) spaces. RDMs were computed separately for HSC and LSC conditions during both the exemplar-learning and creating phases. Pearson correlation coefficients were then calculated between RDMs from the learning and creating phases within each condition. These coefficients were Fisher z-transformed and subjected to group-level t-tests (Fig. 6A). Cross-phase representational similarity between HSC and LSC conditions in the uncompressed space was also compared, allowing us to directly assess the transfer of exemplar representations from learning to creative ideation in both the hippocampus and IFG.
Cross-phase multivoxel pattern decoding and the contribution of low-dimensional hippocampal representations (Exp. 2)
We first trained a linear decoder to distinguish HSC and LSC trials during the exemplar-learning phase, and tested its classification performance on the creating phase, which was previously unseen by the classifier (Fig. 6F). For each participant, we implemented a whole-brain searchlight analysis to assess local activation patterns associated with HSC and LSC trials across both phases. Within each searchlight (spherical mask, radius ~100 voxels), a linear support vector machine (SVM) classifier was trained to discriminate HSC from LSC trials in the learning phase. The trained classifier was then tested on data from the creating phase to evaluate cross-phase generalization. Classification accuracy was defined as the proportion of correctly predicted labels in the creating phase, with above-chance performance determined by subtracting the empirical chance level (50%):
where nc denotes the total number of trials in the creating phase, \({x}_{i}^{{{{\rm{c}}}}}\) is the multivoxel activation pattern for the ith creating-phase trial, \({y}_{i}^{{{{\rm{c}}}}}\) is the ground-truth class label (i.e., HSC or LSC), f (⋅) represents the classifier trained on learning phase data, and I (⋅) is the indicator function returning 1 if the predicted label matches the true label, and 0 otherwise. This metric quantifies the fraction of creating-phase trials that were correctly classified based on neural representations learned during the exemplar-learning phase.
To address potential imbalances in HSC and LSC trials, the dataset was partitioned into multiple balanced subsets using the cosmo_balance_partitions function in the CoSMoMVPA toolbox, ensuring equal representation of each label and comprehensive inclusion of all trials. Classification accuracies were averaged across all subsets. Individual classification maps were subjected to a group-level random-effects analysis (one-sample t-test), with multiple comparison correction performed using cluster-level FDR (q < 0.05) as implemented in SnPM13 (10,000 permutations). At the group level, we identified a peak in the left vlPFC (x = –38, y = 16, z = 28) showing robust cross-phase decoding performance. For each participant, mean classification accuracy was extracted from a 10 mm spherical ROI centered on this peak, and these values were correlated with post-learning creativity scores to assess behavioral relevance.
To determine whether low-dimensional hippocampal representations formed during the learning phase support vlPFC cross-phase decoding ability, we conducted the following analyses. For each participant, low-dimensional hippocampal features were extracted using PCA, retaining PCs explaining 80% of the variance. These signals were regressed out from trial-wise vlPFC activation patterns (within the same ROI) by fitting, for each voxel j, the linear model:
where Pj is the activation vector for voxel j across trials, HPC is the matrix of retained hippocampal low-dimensional representations (trials × PCs), βj is the regression coefficient vector, and εj denotes the residuals. The residuals (vlPFC activity patterns not explained by hippocampal low-dimensional signals) were then used for cross-phase decoding. A linear SVM classifier was again trained on residual vlPFC activity patterns from the learning phase and tested on the creating phase. The same cross-phase decoding procedure was also applied within the left hippocampus (anatomical ROI). All decoding accuracies were statistically compared to chance (50%) using permutation tests. All analyses were conducted using the CoSMoMVPA toolbox, custom MATLAB (2020a) scripts, and Python (3.8) packages, including scikit-learn (version 0.24.1) and nilearn (version 0.8.1).
Ethic approval
All experiments received approval from the Institutional Review Board of Capital Normal University and/or the Ethics Committee of the Center for Biomedical Imaging Research at Tsinghua University. All participants provided informed consent in accordance with the guidelines approved by the Institutional Review Board.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All data supporting the findings of this study are available at the Science Data Bank (https://doi.org/10.57760/sciencedb.14791). Source data are provided with this paper.
Code availability
Custom code used in this study is available at GitHub (https://github.com/psy-luolab).
References
Agogué, M. et al. The impacts of examples on creative design: explaining fixation and stimulation effects. In: Proceedings of the 18th International Conference on Engineering Design (ICED 11), Copenhagen, Denmark (2011).
Fink, A. et al. Enhancing creativity by means of cognitive stimulation: evidence from an fMRI study. NeuroImage 52, 1687–1695 (2010).
Fink, A. et al. Stimulating creativity via the exposure to other people’s ideas. Hum. Brain Mapp. 33, 2603–2610 (2011).
Sio, U. N., Kotovsky, K. & Cagan, J. Fixation or inspiration? A meta-analytic review of the role of examples on design processes. Des. Stud. 39, 70–99 (2015).
Ward, T. B., Patterson, M. J. & Sifonis, C. M. The role of specificity and abstraction in creative idea generation. Creat. Res. J. 16, 1–9 (2004).
Welling, H. Four mental operations in creative cognition: the importance of abstraction. Creat. Res. J. 19, 163–177 (2007).
O’Keefe, J. & Nadel, L. The Hippocampus as a Cognitive Map (Clarendon Press, 1978).
Squire, L. R. Memory and the hippocampus: a synthesis from findings with rats, monkeys, and humans. Psychol. Rev. 99, 195–231 (1992).
Brincat, S. L. & Miller, E. K. Frequency-specific hippocampal–prefrontal interactions during associative learning. Nat. Neurosci. 18, 576–581 (2015).
Kumaran, D. & McClelland, J. L. Generalization through the recurrent interaction of episodic memories: a model of the hippocampal system. Psychol. Rev. 119, 573–616 (2012).
Saez, A., Rigotti, M., Ostojic, S., Fusi, S. & Salzman, C. D. Abstract context representations in primate amygdala and prefrontal cortex. Neuron 87, 869–881 (2015).
Bernardi, S. et al. The geometry of abstraction in the hippocampus and prefrontal cortex. Cell 183, 954–967.e21 (2020).
Garner, K. G. & Dux, P. E. Knowledge generalization and the costs of multitasking. Nat. Rev. Neurosci. 24, 98–112 (2022).
Anil, C. et al. Exploring length generalization in large language models. Adv. Neural Inf. Process. Syst. 35, 38546–38556 (2022).
Ezzat, H., Agogué, M., Le Masson, P., Weil, B. & Cassotti, M. Specificity and abstraction of examples: opposite effects on fixation for creative ideation. J. Creat. Behav. 54, 115–122 (2018).
Roads, B. D. & Love, B. C. The dimensions of dimensionality. Trends Cogn. Sci. 28, 1118–1131 (2024).
Badre, D., Bhandari, A., Keglovits, H. & Kikumoto, A. The dimensionality of neural representations for control. Curr. Opin. Behav. Sci. 38, 20–28 (2021).
Mack, M. L., Preston, A. R. & Love, B. C. Ventromedial prefrontal cortex compression during concept learning. Nat. Commun. 11, 46 (2020).
Adibi, M., McDonald, J. S., Clifford, C. W. G. & Arabzadeh, E. Adaptation improves neural coding efficiency despite increasing correlations in variability. J. Neurosci. 33, 2108–2120 (2013).
Sheng, J. et al. Higher-dimensional neural representations predict better episodic memory. Sci. Adv. 8, eabm3829 (2022).
Chung, S. & Abbott, L. F. Neural population geometry: an approach for understanding biological and artificial neural networks. Curr. Opin. Neurobiol. 70, 137–144 (2021).
Gallego, J. A., Perich, M. G., Miller, L. E. & Solla, S. A. Neural manifolds for the control of movement. Neuron 94, 978–984 (2017).
Williamson, R. C., Doiron, B., Smith, M. A. & Yu, B. M. Bridging large-scale neuronal recordings and large-scale network models using dimensionality reduction. Curr. Opin. Neurobiol. 55, 40–47 (2019).
Cunningham, J. P. & Yu, B. M. Dimensionality reduction for large-scale neural recordings. Nat. Neurosci. 17, 1500–1509 (2014).
Hinton, G. E. & Salakhutdinov, R. R. Reducing the dimensionality of data with neural networks. Science 313, 504–507 (2006).
Fusi, S., Miller, E. K. & Rigotti, M. Why neurons mix: high dimensionality for higher cognition. Curr. Opin. Neurobiol. 37, 66–74 (2016).
Zhang, C., Bengio, S., Hardt, M., Recht, B. & Vinyals, O. Understanding deep learning (still) requires rethinking generalization. Commun. ACM 64, 107–115 (2021).
Spens, E. & Burgess, N. A generative model of memory construction and consolidation. Nat. Hum. Behav. 8, 526–543 (2024).
Addis, D. R., Wong, A. T. & Schacter, D. L. Remembering the past and imagining the future: common and distinct neural substrates during event construction and elaboration. Neuropsychologia 45, 1363–1377 (2007).
Hassabis, D. & Maguire, E. A. Deconstructing episodic memory with construction. Trends Cogn. Sci. 11, 299–306 (2007).
Bowman, C. R. & Zeithamova, D. Abstract memory representations in the ventromedial prefrontal cortex and hippocampus support concept generalization. J. Neurosci. 38, 2605–2614 (2018).
Nieh, E. H. et al. Geometry of abstract learned knowledge in the hippocampus. Nature 595, 80–84 (2021).
Eichenbaum, H. Prefrontal–hippocampal interactions in episodic memory. Nat. Rev. Neurosci. 18, 547–558 (2017).
Preston, A. R. & Eichenbaum, H. Interplay of hippocampus and prefrontal cortex in memory. Curr. Biol. 23, R764–R773 (2013).
Zeithamova, D., Dominick, A. L. & Preston, A. R. Hippocampal and ventral medial prefrontal activation during retrieval-mediated learning supports novel inference. Neuron 75, 168–179 (2012).
van Kesteren, M. T. R. et al. Differential roles for medial prefrontal and medial temporal cortices in schema-dependent encoding: From congruent to incongruent. Neuropsychologia 51, 2352–2359 (2013).
Mack, M. L., Love, B. C. & Preston, A. R. Dynamic updating of hippocampal object representations reflects new conceptual knowledge. Proc. Natl. Acad. Sci. USA 113, 13203–13208 (2016).
Opitz, B. & Friederici, A. D. Interactions of the hippocampal system and the prefrontal cortex in learning language-like rules. NeuroImage 19, 1730–1737 (2003).
Kumaran, D., Hassabis, D. & McClelland, J. L. What Learning systems do intelligent agents need? Complementary learning systems theory updated. Trends Cogn. Sci. 20, 512–534 (2016).
Cortese, A., De Martino, B. & Kawato, M. The neural and cognitive architecture for learning from a small sample. Curr. Opin. Neurobiol. 55, 133–141 (2019).
Wagner, A. D. et al. Building memories: remembering and forgetting of verbal experiences as predicted by brain activity. Science 281, 1188–1191 (1998).
Fernández, G. & Tendolkar, I. Integrated brain activity in medial temporal and prefrontal areas predicts subsequent memory performance: human declarative memory formation at the system level. Brain Res. Bull. 55, 1–9 (2001).
Paller, K. A. & Wagner, A. D. Observing the transformation of experience into memory. Trends Cogn. Sci. 6, 93–102 (2002).
van Genugten, R. D. I., Beaty, R. E., Madore, K. P. & Schacter, D. L. Does episodic retrieval contribute to creative writing? An exploratory study. Creat. Res. J. 34, 145–158 (2021).
Madore, K. P., Szpunar, K. K., Addis, D. R. & Schacter, D. L. Episodic specificity induction impacts activity in a core brain network during construction of imagined future experiences. Proc. Natl. Acad. Sci. USA 113, 10696–10701 (2016).
Beaty, R. E., Thakral, P. P., Madore, K. P., Benedek, M. & Schacter, D. L. Core network contributions to remembering the past, imagining the future, and thinking creatively. J. Cogn. Neurosci. 30, 1939–1951 (2018).
Madore, K. P., Thakral, P. P., Beaty, R. E., Addis, D. R. & Schacter, D. L. Neural mechanisms of episodic retrieval support divergent creative thinking. Cereb. Cortex 29, 150–166 (2017).
Duff, M. C., Kurczek, J., Rubin, R., Cohen, N. J. & Tranel, D. Hippocampal amnesia disrupts creative thinking: creative thinking and hippocampus. Hippocampus 23, 1143–1149 (2013).
Thakral, P. P., Madore, K. P., Kalinowski, S. E. & Schacter, D. L. Modulation of hippocampal brain networks produces changes in episodic simulation and divergent thinking. Proc. Natl. Acad. Sci. USA 117, 12729–12740 (2020).
Thakral, P. P., Madore, K. P., Devitt, A. L. & Schacter, D. L. Adaptive constructive processes: an episodic specificity induction impacts false recall in the Deese–Roediger–McDermott paradigm. J. Exp. Psychol.: Gen. 148, 1480–1493 (2019).
Thakral, P. P., Devitt, A. L., Brashier, N. M. & Schacter, D. L. Linking creativity and false memory: common consequences of a flexible memory system. Cognition 217, 104905 (2021).
Thakral, P. P., Barberio, N. M., Devitt, A. L. & Schacter, D. L. Constructive episodic retrieval processes underlying memory distortion contribute to creative thinking and everyday problem solving. Mem. Cogn. 51, 1125–1144 (2022).
Storm, B. C. & Patel, T. N. Forgetting as a consequence and enabler of creative thinking. J. Exp. Psychol.: Learn. Mem. Cogn. 40, 1594–1609 (2014).
Becker, M., Sommer, T. & Cabeza, R. Insight predicts subsequent memory via cortical representational change and hippocampal activity. Nat. Commun. 16, 4341 (2025).
Legare, C. H. & Nielsen, M. Imitation and innovation: The dual engines of cultural learning. Trends Cogn. Sci. 19, 688–699 (2015).
Jazayeri, M. & Ostojic, S. Interpreting neural computations by examining intrinsic and embedding dimensionality of neural activity. Curr. Opin. Neurobiol. 70, 113–120 (2021).
Zhou, J. et al. Evolving schema representations in orbitofrontal ensembles during learning. Nature 590, 606–611 (2020).
Zhou, J. et al. Complementary task structure representations in hippocampus and orbitofrontal cortex during an odor sequence task. Curr. Biol. 29, 3402–3409.e3 (2019).
LaRocque, K. F. et al. Global similarity and pattern separation in the human medial temporal lobe predict subsequent memory. J. Neurosci. 33, 5466–5474 (2013).
Davis, T., Xue, G., Love, B. C., Preston, A. R. & Poldrack, R. A. Global neural pattern similarity as a common basis for categorization and recognition memory. J. Neurosci. 34, 7472–7484 (2014).
Kenett, Y. N. & Faust, M. A semantic network cartography of the creative mind. Trends Cogn. Sci. 23, 271–274 (2019).
Mayseless, N. & Shamay-Tsoory, S. G. Enhancing verbal creativity: modulating creativity by altering the balance between right and left inferior frontal gyrus with tDCS. Neuroscience 291, 167–176 (2015).
Collins, A. & Koechlin, E. Reasoning, learning, and creativity: frontal lobe function and human decision-making. PLoS Biol. 10, e1001293 (2012).
Tzourio-Mazoyer, N. et al. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. NeuroImage 15, 273–289 (2002).
Gardiner, J. M. & Java, R. I. Recollective experience in word and nonword recognition. Mem. Cogn. 18, 23–30 (1990).
Kjelstrup, K. B. et al. Finite scale of spatial representation in the hippocampus. Science 321, 140–143 (2008).
Courellis, H. S. et al. Abstract representations emerge in human hippocampal neurons during inference. Nature 632, 841–849 (2024).
Poppenk, J. & Moscovitch, M. A hippocampal marker of recollection memory ability among healthy young adults: contributions of posterior and anterior segments. Neuron 72, 931–937 (2011).
Wittfoth, M., Schardt, D. M., Fahle, M. & Herrmann, M. How the brain resolves high conflict situations: double conflict involvement of dorsolateral prefrontal cortex. NeuroImage 44, 1201–1209 (2009).
Fan, J., Hof, P. R., Guise, K. G., Fossella, J. A. & Posner, M. I. The functional integration of the anterior cingulate cortex during conflict processing. Cereb. Cortex https://doi.org/10.1093/cercor/bhm125 (2008).
Linsey, J. S. et al. A study of design fixation, its mitigation and perception in engineering design faculty. J. Mech. Design 132, 041003 (2010).
Gerver, C. R., Griffin, J. W., Dennis, N. A. & Beaty, R. E. Memory and creativity: a meta-analytic examination of the relationship between memory systems and creative cognition. Psychon. Bull. Rev. 30, 2116–2154 (2023).
Muhle-Karbe, P. S. et al. Goal-seeking compresses neural codes for space in the human hippocampus and orbitofrontal cortex. Neuron 111, 3885–3899.e6 (2023).
Vaidya, A. R. & Badre, D. Abstract task representations for inference and control. Trends Cogn. Sci. 26, 484–498 (2022).
Samborska, V., Butler, J. L., Walton, M. E., Behrens, T. E. J. & Akam, T. Complementary task representations in hippocampus and prefrontal cortex for generalizing the structure of problems. Nat. Neurosci. 25, 1314–1326 (2022).
Ren, J. et al. Functional lateralization of the medial temporal lobe in novel associative processing during creativity evaluation. Cereb. Cortex 33, 1186–1206 (2023).
Thompson-Schill, S. L., D’Esposito, M., Aguirre, G. K. & Farah, M. J. Role of left inferior prefrontal cortex in retrieval of semantic knowledge: a reevaluation. Proc. Natl. Acad. Sci. USA 94, 14792–14797 (1997).
Wallis, J. D., Anderson, K. C. & Miller, E. K. Single neurons in prefrontal cortex encode abstract rules. Nature 411, 953–956 (2001).
Doeller, C. F., King, J. A. & Burgess, N. Parallel striatal and hippocampal systems for landmarks and boundaries in spatial memory. Proc. Natl. Acad. Sci. USA 105, 5915–5920 (2008).
Bein, O., Reggev, N. & Maril, A. Prior knowledge promotes hippocampal separation but cortical assimilation in the left inferior frontal gyrus. Nat. Commun. 11, 4590 (2020).
Bowman, C. R., Iwashita, T. & Zeithamova, D. Tracking prototype and exemplar representations in the brain across learning. eLife 9, e55369 (2020).
Kim, H. Neural activity that predicts subsequent memory and forgetting: a meta-analysis of 74 fMRI studies. NeuroImage 54, 2446–2461 (2011).
Maguire, E. A. & Hassabis, D. Role of the hippocampus in imagination and future thinking. Proc. Natl. Acad. Sci. USA 108, E39 (2011).
Morton, N. W., Schlichting, M. L. & Preston, A. R. Representations of common event structure in medial temporal lobe and frontoparietal cortex support efficient inference. Proc. Natl. Acad. Sci. USA 117, 29338–29345 (2020).
Nadel, L., Hoscheidt, S. & Ryan, L. R. Spatial cognition and the hippocampus: the anterior–posterior axis. J. Cogn. Neurosci. 25, 22–28 (2013).
Gutchess, A. H. & Schacter, D. L. The neural correlates of gist-based true and false recognition. NeuroImage 59, 3418–3426 (2012).
Zeithamova, D. & Preston, A. R. Temporal proximity promotes integration of overlapping events. J. Cogn. Neurosci. 29, 1311–1323 (2017).
Richter, F. R., Chanales, A. J. H. & Kuhl, B. A. Predicting the integration of overlapping memories by decoding mnemonic processing states during learning. NeuroImage 124, 323–335 (2016).
Chanales, A. J. H., Dudukovic, N. M., Richter, F. R. & Kuhl, B. A. Interference between overlapping memories is predicted by neural states during learning. Nat. Commun. 10, 5363 (2019).
Gilhooly, K. J., Fioratou, E., Anthony, S. H. & Wynn, V. Divergent thinking: strategies and executive involvement in generating novel uses for familiar objects. Br. J. Psychol. 98, 611–625 (2007).
Beaty, R. E. The neuroscience of musical improvisation. Neurosci. Biobehav. Rev. 51, 108–117 (2015).
Beaty, R. E. & Silvia, P. J. Why do ideas get more creative across time? An executive interpretation of the serial order effect in divergent thinking tasks. Psychol. Aesthet. Creat. Arts 6, 309–319 (2012).
Birn, R. M., Bandettini, P. A., Cox, R. W. & Shaker, R. Event-related fMRI of tasks involving brief motion. Hum. Brain Mapp. 7, 106–114 (1999).
Palmer, E. D. et al. An event-related fMRI study of overt and covert word stem completion. Neuroimage 14, 182–193 (2001).
Baer, J. & McKool, S. Assessing creativity using the consensual assessment technique. In Handbook of Research on Assessment Technologies, Methods, and Applications in Higher Education 65–77 (IGI Global, 2009).
Abdulrahman, H. & Henson, R. N. Effect of trial-to-trial variability on optimal event-related fMRI design: implications for Beta-series correlation and multi-voxel pattern analysis. NeuroImage 125, 756–766 (2016).
Poppenk, J., Evensmoen, H. R., Moscovitch, M. & Nadel, L. Long-axis specialization of the human hippocampus. Trends Cogn. Sci. 17, 230–240 (2013).
Yarkoni, T., Poldrack, R. A., Nichols, T. E., Van Essen, D. C. & Wager, T. D. Large-scale automated synthesis of human functional neuroimaging data. Nat. Methods 8, 665–670 (2011).
Principal components as a small number of interpretable variables: some examples. In Principal Component Analysis (ed. Jolliffe, I. T.) 63–77 (Springer, 2002).
Friston, K. J. et al. Psychophysiological and modulatory interactions in neuroimaging. NeuroImage 6, 218–229 (1997).
Acknowledgements
This work was supported by the Scientific and Technological Innovation 2030—Major Project (Grant No. 2021ZD0200500 to S.Z.Q.), the National Natural Science Foundation of China (Grant No. 32271097 to J.L., Grant No. 32130045 to S.Z.Q., Grant No. 32361163611 to S.Z.Q., and Grant No. 82021004 to S.Z.Q.), the Major Project of the National Social Science Foundation of China (Grant No. 19ZDA363 to S.Z.Q. and Grant No. 20&ZD153 to S.Z.Q.), the Fundamental Research Funds for the Central Universities (Grant No. 2024-JYB-XJSJJ020 to S.Z.Q.), and the National Education Science Planning Project (Grant No. EBA250433 to Z.Z.). We thank Ying Zhang, Yanting Zou, Yuechen Wang, Qingjing Luo, and Yue Li for their assistance in experimental material preparation and behavioral data collection.
Author information
Authors and Affiliations
Contributions
Conceptualization: J.L., S.Z.Q., and Z.Z.; Methodology: J.L., S.Z.Q., Z.Z., Z.Y.L., G.W.W., and R.D.; Investigation: Z.Z. and J.Q.W.; Visualization: Z.Z., S.Z.Q., and J.L.; Funding acquisition: J.L., S.Z.Q., and Z.Z.; Project administration: Z.Z., J.L., and S.Z.Q.; Supervision: J.L. and S.Z.Q.; Writing—original draft: Z.Z., J.L., and S.Z.Q.; Writing—review and editing: Z.Z., J.L., S.Z.Q., Z.Y.L., X.X.X., G.W.W., and R.D.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, Z., Li, Z., Ding, R. et al. Hippocampal-prefrontal orchestration supports higher-order learning for creative ideation. Nat Commun 17, 464 (2026). https://doi.org/10.1038/s41467-025-67157-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-67157-x