Abstract
The non-radiative metallic core (MC) centered lowest excited states of most ligand-stabilized metallic clusters commonly quench radiative but high-lying ligand-centered excited states, e.g. intra-ligand charge transfer (LCT), which is one of the key issues limiting efficiencies of electroluminescent (EL) clusters. Herein, we realize the desired excited state reversal in a cubic [PXZDBFDP]2Cu4I4 (PXZDBFDP = 10-(4,6-bis(diphenylphosphino)dibenzo[b,d]furan-2-yl)−10H-phenoxazine) modified with strongly electron-donating phenoxazine (PXZ) to strengthen donor-acceptor (D-A) interactions and enhance LCT. Consequently, its thoroughly LCT-featured first singlet (S1) and triplet (T1) excited states are energetically lower than its Cu4I4-involved excited states. This case not only increases excited-state utilization through energy transfer from non-radiative MC to radiative LCT states, but also leads to balanced dual emission of thermally activated delayed fluorescence (TADF, 52%) and phosphorescence (PH, 48%) respectively from singlet and triplet LCT states. Therefore, compared to another congener [PhPXZDBFDP]2Cu4I4 (PhPXZDBFDP = 10-(4,6-bis(diphenylphosphino)dibenzo[b,d]furan-2-yl)−10H-phenylphenoxazine) with a D-π-A ligand and the normal low-lying MC states, [PXZDBFDP]2Cu4I4 achieves sevenfold increased photoluminescence quantum yield of ~90%, and the 13-fold increased maximum EL external quantum efficiency of 35.5%, which is the record-high value for EL homo-copper clusters. These results demonstrate the feasibility of accurate excited-state optimization for clusters through ligand engineering.
Similar content being viewed by others
Introduction
In recent decades, cluster-based light-emitting diodes (CLED) emerge rapidly, extending the applications of this kind of materials into optoelectronic field1. Compared to other electroluminescent (EL) systems including organic molecules2,3,4,5, complexes6,7,8, polymers9,10,11, quantum dots12,13,14,15 and perovskites16,17,18,19,20,21, cluster emitters combine the merits of organic ligands and inorganic metallic cores (MC) with respect to optical performances and stabilities. Discrete energy levels of cluster molecules further make emission modulation flexible. But, they simultaneously exhibit complicated excited states composed of not only efficiently radiative high-lying ligand-centered (LC), e.g., metal/counterion-ligand charge transfer (Mn/XLCT), intraligand (LCT) and interligand charge transfer (ILCT) and locally excited (LE) states, but also low-lying cluster-center (CC) components including metal-metal (MMCT) and metal-counterion charge transfer (MXCT) inferior in radiation22. Obviously, the competition between LC and CC components in exciton allocation directly determines luminescent properties of cluster emitters, giving rise to a big challenge in performance improvement through rational excited-state modulation.
Cuprous haloid clusters have the advantages of moderate Cu/X-ligand interactions, tunable excited state compositions and electrochemical stabilities, parts of whose merits for EL applications are already demonstrated by efficient organic light-emitting diodes based on CuX complexes23,24,25,26. Importantly, highly rigid and three-dimensional CunXm structures can intrinsically and effectively alleviate structural relaxation at excited states, e.g., John-Teller distortion of Cu+ ions in mononuclear27,28,29,30 and binuclear rhomboidal Cu2X2 complexes31,32,33, and provide flexible platforms for accurately modulating MC-ligand interactions, therefore emerging rapidly as emitters for CLEDs in most recent years34.
The excited states of CunXm clusters can be further modulated through: (i) MC optimization: geometric topologies and coordination environments of Cu+ ions can be modified to enhance molecular rigidities and adjust metal-metal/X and metal-ligand interactions, respectively reducing non-radiative energy loss and tuning ligand-MC charge transfer35,36,37. Consequently, chair-shape38, cubic39 and octahedral40,41 Cu4I4 and Cu4I642,43 with larger nuclei numbers, as well as heterobimetallic cores44, can enhance luminescence and improve efficiencies; (ii) Ligand engineering: functional modification of ligands can not only increase the ratios of LC components in excited states, but also influence metal-metal/X and MC-ligand interactions45. Our previous works demonstrated that introducing carbazole or acridine groups can effectively reduce the contributions of whole Cu4I4 cubic cores to excited states, leading to LC-predominant radiative excited states and photoluminescence (PL) quantum yields (PLQY, ηPL) reaching 90%46,47, but their half units, namely Cu2I2, were still involved in Mn/XLCT transitions. As a result, without external assistance, ϕEQE of CLEDs based on Cu4I4 cubes can hardly exceed 25%1,48.
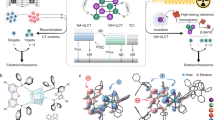
Actually, in most cases of clusters, one of the intrinsic issue is CC states energetically lower than LC states, therefore, it is difficult to completely prevent LC → CC energy transfer; meanwhile, the incorporation of Cu+ and I- in frontier molecular orbitals (FMO) inevitably induces the direct charge and exciton capture by Cu4I4 cores during EL processes. It means cluster involved charge transfer including not only CC states but also Mn/XLCT should be excluded from the lowest excited states, in turn making LCT and/or ILCT absolutely predominant in radiative transitions, which is the embodiment of the competition between MC and donor groups in intramolecular charge transfer to acceptor groups (Fig. 1a). However, it is undoubtedly a formidable challenge for thorough confinement of the lowest excited states and FMOs on organic ligands in low-valence metallic clusters.
a Molecular structures of [PXZDBFDP]2Cu4I4 and [PhPXZDBFDP]2Cu4I4 featuring donor-acceptor (D-A) and D-π-A structured ligands, in which phenoxazine (PXZ) and Cu4I4 metallic core (MC)-coordinated 4,6-bis(diphenylphosphaneyl)dibenzofuran (DBFDP) as donor and acceptor units. The predominance of intraligand charge transfer (LCT) or cluster centered (CC) charge transfer in the lowest excited states is based on the competition between PXZ and Cu4I4 core in electron donating. Phenyl between PXZ and DBFDP is a π-linker to weaken intraligand charge transfer and in turn strengthen cluster-centered charge transfer; b Molecular configurations of [PXZDBFDP]2Cu4I4 and [PhPXZDBFDP]2Cu4I4. P-Cu bond lengths and aromatic dihedral angles in ligands are highlighted; c Natural transition orbital analysis of the lowest LCT and CC excited states for [PXZDBFDP]2Cu4I4 and [PhPXZDBFDP]2Cu4I4. The main molecular orbitals involved in the excitations and the transition types are indicated. Contours of the occupied and unoccupied molecular orbitals for ground states (S0) and occupied (“hole”) and unoccupied (“electron”) nature transition orbitals for excited states are distinguished with blue and pink colors, respectively. Energy levels of frontier molecular orbitals and excited states are highlighted. fS refers to singlet oscillator strength.
In this contribution, a donor-acceptor (D-A) type biphosphine ligand named PXZDBFDP is constructed as 4,6-bis(diphenyl-phosphaneyl)dibenzofuran (DBFDP) substituted with strongly electron-donating phenoxazine (PXZ) (Fig. 1a). The corresponding cubic [PXZDBFDP]2Cu4I4 successfully achieves thoroughly ligand-confined FMOs and the LCT-featured first singlet (S1) and triplet (T1) excited states with occupied and unoccupied orbitals respectively localized on PXZ and dibenzofuran (DBF); while, its Cu4I4 core reversely contributes to the high-lying S3 and T3 states. This case of abnormal excited state reversal makes non-radiative CC states of [PXZDBFDP]2Cu4I4 can be converted to its LCT state for radiation. Compared to [PhPXZDBFDP]2Cu4I4 with D-π-A type ligands and weakened LCT, the superiority of [PXZDBFDP]2Cu4I4 in radiation is demonstrated with its fourfold larger singlet oscillator strength (fS) and sevenfold increased ϕPL to ~90%. [PXZDBFDP]2Cu4I4 further reveals the advantage in suppressing exciton accumulation induced quenching, since its LCT-featured S1 and T1 excited states support thermal equilibrium between intersystem crossing (ISC) and reverse ISC, giving rise to balanced dual emission with a thermally activated delayed fluorescence (TADF)/phosphorescence (PH) ratio of 52/48. Consequently, [PXZDBFDP]2Cu4I4 endowed its devices with 180-fold increased ηEQE beyond 35%, which is the record-high value for EL homo-copper clusters. These results demonstrate the feasibility of accurate excited-state optimization for cluster molecules through ligand engineering strategy.
Results
Cluster design and structures
A chelating motif DBFDP is used to form the cubic MC skeleton, namely [DBFDP]2Cu4I439. P→CuI coordination leads to Mn/XLCT from Cu4I4 to DBFDP under excitation, in which DBF actually serves as acceptor group withdrawing electron from Cu+. In this sense, LCT and ILCT components in LC excited states, involve in DBF as acceptor identical to that in Mn/XLCT, rendering the competition between them with respect to exciton allocation, let alone MMCT and MXCT in CC states. Obviously, donor substituents on DBFDP can enhance LCT and ILCT with DBF, in turn weakening MC-involved charge transfer (Fig. 1a). If the electron-donating effects of donor groups stronger than Cu4I4, it is of possibility to make LCT and/or ILCT energetically lower than CC states, leading to the reverse CC → LC energy transfer opposite to the common situations of Cu4I4 clusters, therefore making all the energies can be utilized through LCT/ILCT radiation. With this consideration, PXZ with accumulated electron-donating conjugation effects from O and N atoms can be competent as donor to strengthen interactions with DBF for the maximum. To evaluate the sensitivity of excited-state characteristics on electronic configurations of ligands, we designed two ligands PXZDBFDP and PhPXZDBFDP respectively having D-A and D-π-A structures for comparison. Obviously, D-A interactions between PXZ and DBF in PhPXZDBFDP is reduced by its phenylene as a π-linker, in turn enhancing MC-involved charge transfer.
PXZDBFDP and PhPXZDBFDP can be readily synthesized through Ullman and Suzuki coupling reactions with yields more than 80%, and then used to prepare Cu4I4 cubes with CuI in dichloromethane (Supplementary Figs 1–19). In contrast to the symmetric coordination skeleton of [PhPXZDBFDP]2Cu4I4, P-Cu bonds on the same sides of PXZ in [PXZDBFDP]2Cu4I4 are shorter than those on the other sides, accompanied by its slightly asymmetric Cu4I4 core, because the stronger electron-donating effect of PXZ on DBFDP unit increases electron cloud density of the P atom on the same side of PXZDBFDP (Fig. 1b and Supplementary Fig. 20 and Supplementary Table 1–4). Furthermore, for [PhPXZDBFDP]2Cu4I4, its phenylene and DBF groups are basically coplanar, inevitably inducing FMO extension and overlapping. On the contrary, PXZ groups in [PXZDBFDP]2Cu4I4 are almost orthogonal to DBF moieties, evidenced by their dihedral angles more than 70o, which can render FMO separation for PXZDBFDP, thereby enhancing PXZ → DBF charge transfer.
Density function theory (DFT) simulation on ground states (S0) of these two clusters shows their first three unoccupied molecular orbitals and highest occupied molecular orbitals (HOMO) are localized on DBF and PXZ groups, respectively (Fig. 1c and Supplementary Fig. 21). On the contrary to the situation of [PhPXZDBFDP]2Cu4I4, the HOMO-1 and HOMO-2 of [PXZDBFDP]2Cu4I4 are respectively contributed by PXZ and Cu4I4 core, reflecting the stronger D-A interactions. Consequently, [PXZDBFDP]2Cu4I4 exhibits the shallower HOMO and deeper lowest unoccupied molecular orbital (LUMO), compared to [PhPXZDBFDP]2Cu4I4, which are consistent with the HOMO and LUMO energy levels of −5.46 and −3.01 eV and −5.50 and −2.84 eV experimentally estimated with cyclic voltammetry (CV) analysis (Supplementary Figs 22 and 23 and Supplementary Table 5). Furthermore, the energy gap between the highest occupied molecular orbitals on PXZ and Cu4I4 core is 0.163 eV for [PXZDBFDP]2Cu4I4 (HOMO → HOMO-2), which is more than doubled in comparison to that of [PhPXZDBFDP]2Cu4I4 (HOMO → HOMO-1). In contrast to [PXZDBFDP]2Cu4I4, PXZ of [PXZDBFDP]2Cu4I4 is superior to Cu4I4 core with respect to electron donating effect, in addition to its stronger PXZ → DBF charge transfer.
Natural transition orbital (NTO) analysis on S0 → S1 and S0 → T1 excitations of [PXZDBFDP]2Cu4I4 and [PhPXZDBFDP]2Cu4I4 reveals their opposite excited-state locations (Fig. 1c and Supplementary Figs 24 and 25). The “holes” and “electrons” of the S1 and T1 states for [PhPXZDBFDP]2Cu4I4 are respectively localized on Cu4I4 core and DBF groups, corresponding to nM/XLCT and CC states, which are identical to those of [DBFDP]2Cu4I4. Its lowest excited state involved in PXZ is the T6 state, whose “hole” and “electron” are localized on PXZ and dispersed on PXZ, DBF and phenylene, respectively, to form a hybrid “charger transfer + locally excited” state. On the contrary, the strong PXZ-DBF charge transfer in [PXZDBFDP]2Cu4I4 induces excited state reversal that its S1 and T1 states featuring LCT states with “holes” and “electrons” respectively localized on PXZ and DBF; while, its Cu4I4 core and DBF make the major contributions to “holes” and “electrons” of the S3 and T3 states characteristic of nM/XLCT and CC states.
Obviously, the HOMOs and LUMOs localized on PXZ and DBF groups make the ligands predominant in carrier injection during EL processes. However, the energy transfer from the high-lying LCT states of [PhPXZDBFDP]2Cu4I4 to its lowest CC states worsens the exciton quenching by the latter. The superiority of the LCT-featured S1 state for [PXZDBFDP]2Cu4I4 in radiation is directly verified by its oscillator strength (fS) more than three folds of its high-lying CC-featured S3 state, and four folds of that of CC-featured S1 state for [PhPXZDBFDP]2Cu4I4. More importantly, CC states of [PXZDBFDP]2Cu4I4 are ~0.25 eV higher than its LCT-featured S1 and T1 states, leading to abnormal “reverse” energy transfer from non-radiative CC to radiative LCT states. In this case, it is convincing that [PXZDBFDP]2Cu4I4 can theoretically harvest 100% electro-generated excitons confined on both ligands and MC for emission, therefore breaking through the efficiency bottleneck for EL copper clusters.
Photophysical properties
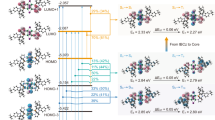
π → π* and n→π* transitions of the ligands peaked at ~240 and 280 nm are predominant in electronic absorption spectra of the clusters in dilute solutions (10−6 M in CH2Cl2), accompanied by long charge transfer absorption tails at 300–480 nm (Fig. 2a and Supplementary Table 1). In absorption spectra of spin-coated neat films, besides ligands-attributed bands, intermolecular interactions markedly enhance the charge transfer bands (Supplementary Fig. 26 and Supplementary Table 5). Nonetheless, it can be noted that CC band at 350–400 nm is predominant for [PhPXZDBFDP]2Cu4I4, but [PXZDBFDP]2Cu4I4 exhibits more distinct LCT band at 350–450 nm. Furthermore, excitation spectra of the neat films show the predominant contributions of MLCT, CC and LCT states to emission, which is consistent with NTO analysis results. [PXZDBFDP]2Cu4I4 and [PhPXZDBFDP]2Cu4I4 reveal the similar MLCT bands peaked at ~320 and 335 nm, accompanied with CC bands as shoulder peaks at ~340 and 370 nm, respectively. Importantly, in opposite to high-energy LCT band of [PhPXZDBFDP]2Cu4I4 at 275 nm, LCT band of neat [PXZDBFDP]2Cu4I4 film is the lowest energy band peaked at ~400 nm, experimentally demonstrating the abnormal excited-state reversal and the resulted exothermic CC → LCT energy transfer through fast and efficient internal conversion.
a Electronic absorption (abs., upper left) of [PXZDBFDP]2Cu4I4 and [PhPXZDBFDP]2Cu4I4 in dilute dichloromethane solution (10-6 mol L-1) and excitation (exc., below left) and photoluminescence (PL) spectra (right) of BCPO:x% [PXZDBFDP]2Cu4I4 and [PhPXZDBFDP]2Cu4I4 films (x = 100 for neat cluster films, and x = 20). BCPO is bis-4-(N-carbazolyl)phenyl)phenylphosphine oxide used as host matrix. MLCT refers to metal-ligand charge transfer. Prompt (PF) and delayed fluorescence (DF) and phosphorescence (PH) spectra at x = 20 were measured with time-resolved technology in the ranges of < 1 µs, 1–100 µs and > 200 µs, respectively. PL spectrum of neat [PhPXZDBFDP]2Cu4I4 film is fitted with dual peaks corresponding to MLCT and CC transitions, respectively highlighted with green and red fill colors; b Time decays (left) of BCPO:x% [PXZDBFDP]2Cu4I4 and [PhPXZDBFDP]2Cu4I4 films and dependence of PL quantum yields (PLQY) on doping concentrations of the films; c Temperature dependence of average lifetimes for [PXZDBFDP]2Cu4I4 and [PhPXZDBFDP]2Cu4I4 based films (left) and temperature-dependent fractional contributions of thermally activated delayed fluorescence (TADF) and PH to total emissions of [PXZDBFDP]2Cu4I4 based films (right); d Temperature-dependent time-resolved emission spectra (TRES) of BCPO:20% [PXZDBFDP]2Cu4I4 and [PhPXZDBFDP]2Cu4I4 films at 50 and 300 K.
PL spectrum of neat [PXZDBFDP]2Cu4I4 film is composed of a single-peak emission at 520 nm originated from LCT radiation (Fig. 2a and Supplementary Table 5). In contrast, neat [PhPXZDBFDP]2Cu4I4 film reveals two emission peaks at 522 and 626 nm originated from its high-energy MLCT and low-energy CC states with different geometrical reorganization (Supplementary Fig. 27)49,50, which is similar to donor-free [DBFDP]2Cu4I4 in accord with their identical CC-featured S1 and T1 states22,39. After 20% dispersed in bis-4-(N-carbazolyl)phenyl)phenylphosphine oxide (BCPO) matrix, the emission of [PXZDBFDP]2Cu4I4 is slightly blue-shifted to 508 nm, due to reduced inter-ligand interactions between adjacent cluster molecules. PL spectrum of BCPO:20% [PhPXZDBFDP]2Cu4I4 film is more markedly changed to single-peak emission at 498 nm, accompanied by a CC tail in 600–700 nm, because of suppressed quenching on MLCT state induced by inter-ligand interactions and lower radiative possibility for CC than MLCT.
On the basis of time-resolved technology, the nominal prompt (PF) and delayed fluorescence (DF) and PH spectra of BCPO:20% [PXZDBFDP]2Cu4I4 are similar but still gradually and slightly red shifted, different to completely overlapped spectra of [PhPXZDBFDP]2Cu4I4. According to absorption edges and 0–0 transitions of time-resolved PH spectra recorded after a delay of 200 µs, the S1 and T1 energy levels of [PXZDBFDP]2Cu4I4 and [PhPXZDBFDP]2Cu4I4 are 2.82 and 2.78 eV, and 2.90 and 2.88 eV, corresponding to singlet-triplet splitting energies (ΔEST) of 0.04 and 0.02 eV, respectively, which are highly consistent with NTO results (Supplementary Table 5).
Compared to neat films, time decays of BCPO:20% cluster films are also markedly elongated, owing to suppressed quenching (Fig. 2b). Nonetheless, [PXZDBFDP]2Cu4I4 doped films reveal markedly shorter lifetimes, which are beneficial to reduce collisional quenching. For the same reason, their lifetime dependence on doping concentration is limited. In contrast, lifetime of [PhPXZDBFDP]2Cu4I4 in BCPO matrix is elongated by about three folds, indicating its fragile excited states highly sensitive to quenching effect. Consequently, BCPO:x% [PXZDBFDP]2Cu4I4 films achieve markedly higher ϕPL reaching 90% at x = 20 for the maximum; while, ϕPL of [PhPXZDBFDP]2Cu4I4 based analogs also reaches the maximum value at x = 20 but as low as ~11%, which is only about one eighth of that of BCPO:20% [PXZDBFDP]2Cu4I4.
PL spectra of [PXZDBFDP]2Cu4I4 in film are almost unchanged in the temperature range of 20-300 K, further evidencing the radiation from single-component nLCT states (n = 1 and 3 for singlet and triplet, Supplementary Fig. 7a). In contrast, the intensities of CC-attributed long-wavelength bands for [PhPXZDBFDP]2Cu4I4 film are directly proportional to temperature, which is consistent with previously reported thermochromic copper iodide clusters, and commonly ascribed to MLCT → CC energy transfer through thermal relaxation (Supplementary Fig. 27b)51,52. Furthermore, although collisional quenching of neat [PhPXZDBFDP]2Cu4I4 film is worsened by increasing temperature and therefore induces gradually decrease of lifetime, with suppressed inter-cluster interactions, lifetime of BCPO:20% [PhPXZDBFDP]2Cu4I4 becomes directly proportional to temperature, in accord to the conversion from partially forbidden 3MLCT to forbidden 3CC state with smaller radiative rate constant (Supplementary Fig. 28). On the contrary, time decays of both neat and 20% doped films of [PXZDBFDP]2Cu4I4 are in reverse proportion to temperature (Supplementary Fig. 29). Considering its emission from single-component nLCT states, the lifetime decrease for [PXZDBFDP]2Cu4I4 verifies the gradual transition from forbidden 3LCT-attributed PH at low temperature to allowed 1LCT-attributed TADF at high temperature.
The PH/TADF ratios of the emissions from the clusters are quantitatively evaluated with the fractional PH and TADF intensities as functions of temperature, respectively (please see details in supporting information) (Fig. 2c)53. In consistence with NTO results, [PhPXZDBFDP]2Cu4I4 reveals pure PH emission with the nearly temperature-independent ratios more than 99%, which further manifests the elongated lifetime at high temperature is due to conversion between two triplet states, namely 3MLCT → 3CC through thermal relaxation54,55,56. On the contrary, despite ~100% PH at < 50 K, TADF intensities for [PXZDBFDP]2Cu4I4 rapidly increase at higher temperature, in accord with the thermal activation mechanism for triplet→singlet conversion. The balanced temperature at equal TADF and PH ratios of 50% is ~280 K. At ambient temperature, TADF proportion only slightly increases to 52%. In this sense, both the singlet and triplet LCT states of [PXZDBFDP]2Cu4I4 nearly equally contraibute to PH and TADF under a dual-emission mode, which can simultaneously reduce exciton-accumulation induced quenching and accelerate radiation.
Temperature-dependent time-resolved emission (TRES) spectra of BCPO:x% [PXZDBFDP]2Cu4I4 (x = 100 for neat film, and 20) reveal that emissions become concentrated, along with temperature increasing (Fig. 2d and Supplementary Fig. 30a). This variation tendency can be attributed to the accelerated radiation through both nLCT channels, especially the singlet one. The same situation for the neat film further indicates that the increased contribution of 1LCT state to radiation effectively alleviates the triplet quenching. TRES spectra of neat [PhPXZDBFDP]2Cu4I4 film show that the emissions are elongated when increasing temperature from 50 to 200 K, and then shortened at 300 K, corresponding to a transition to forbidden CC state in the first stage and CC quenching at the second stage (Fig. 2d and Supplementary Figs 30b and 31). BCPO matrix alleviates quenching of [PXZDBFDP]2Cu4I4, but transition from 3MLCT to 3CC still renders the emission elongation, due to slowed radiation from the latter.
It is convincing that the state-of-the-art ϕPL values of BCPO:x% [PXZDBFDP]2Cu4I4 films are ascribed to the radiative LCT-featured S1 and T1 states, reversed CC → LCT energy transfer and the balanced dual emission, giving rise to effective radiation acceleration and quenching suppression. Furthermore, with exciton state reversal, the direct carrier capture by MC of [PXZDBFDP]2Cu4I4 during EL process can not reduce the exciton utilization efficiencies, since carrier recombination on its MC would be followed with CC → LCT exciton transfer.
EL properties
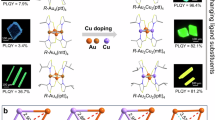
Encouraged by its ϕPL of 90% and dual-emissive characteristics, we used [PXZDBFDP]2Cu4I4 and [PhPXZDBFDP]2Cu4I4 as dopants in emitting layers (EML) of BCPO:x% clusters to fabricate spin-coated CLEDs with bilayer structures (Fig. 3a). With the shallower HOMOs and deeper LUMOs and lower S1 and T1 energy levels than those of BCPO hosts, the clusters can effectively confine the electro-generated excitons through both direct carrier capture and energy transfer. Therefore, despite the optimal x of 20 for the highest EL performances, all the devices exhibited green and yellowish green emissions from the clusters peaked at ~510 nm (Fig. 3b, and Supplementary Figs 32–33 and Supplementary Table 6). Nevertheless, profiles of EL spectra for [PXZDBFDP]2Cu4I4 based devices are markedly narrower than those of analogs based on [PhPXZDBFDP]2Cu4I4 by ~50 nm with respect to full width at half maximum (FWHM).
a Device configurations of ITO | PEDOT:PSS (40 nm)|BCPO:clusters (x%wt., 40 nm)|TmPyPB (50 nm)|LiF (1 nm)|Al (100 nm), chemical structures of BCPO and TmPyPB and device photos at 10 V; b EL spectra of the devices with x = 20 at 10 V, and the corresponding coordinates on CIE1931 chromaticity plane; c Current density (J)-Voltage-Luminance characteristics of CLEDs at x = 20; d Efficiencies-Luminance relationships of the devices at x = 20. PE, CE and EQE refer to power efficiency, current efficiency and external quantum efficiency, respectively. EQE values at the maximum, 100 and 500 nits are highlighted with arrows, respectively; e Comparison on the maximum EQE values of the representative EL clusters.
Owing to its stronger carrier injecting ability, [PXZDBFDP]2Cu4I4 effectively reduced driving voltages of its devices by 0.1–0.5 V for turn-on and 1.5–3.7 V at 100 cd m−2, in comparison to [PhPXZDBFDP]2Cu4I4 based analogs (Fig. 3c, Supplementary Figs 32–33 and Supplementary Table 6). Despite their comparable current densities (J), the maximum luminance of [PXZDBFDP]2Cu4I4 based devices was beyond 1500 nits, which was markedly increased by ten folds, compared to [PhPXZDBFDP]2Cu4I4 based analogs. Considering the similar J for these devices, the larger luminance of the former reflects the stronger exciton utilization ability of [PXZDBFDP]2Cu4I4 during EL process.
Inspiringly, BCPO:20% [PXZDBFDP]2Cu4I4 achieved the the state-of-the-art efficiencies with the maxima as high as 95.9 cd A-1 for current efficiency (CE, ηCE), 75.3 lm W-1 for power efficiency (PE, ηPE) and 35.5% for ηEQE, respectively (Fig. 3d, Supplementary Figs 32–33 and Supplementary Table 6), which dramatically improve the efficiency record of CLEDs based on copper clusters by about one fifth57. Actually, [PXZDBFDP]2Cu4I4 is one of the only two clusters reported so far can realize the maximum ηEQE beyond 35% (Fig. 3e and Supplementary Table 3). It is noteworthy that compared to the other one with sharply decreased ηEQE of ~10% at 500 nits, [PXZDBFDP]2Cu4I4 is more superior in efficiency stability at high luminance, evidenced by its ηEQE remaining 32.4% and 25.0% at 100 and 500 nits, corresponding to markedly reduced EQE roll-offs of 8.7% and 29.6%. It is noteworthy that EL efficiencies of [PXZDBFDP]2Cu4I4 are one order of magnitude larger than those of [PhPXZDBFDP]2Cu4I4 based analogs. At x = 20, the maximum ηEQE of the latter was only 2.7% at the same level of donor-free parent cluster [DBFDP]2Cu4I439, which is as low as one thirteenth of that of [PXZDBFDP]2Cu4I4 based devices.
Taking out-coupling ratio of indium tin oxide (ITO) glass as 0.30, [PXZDBFDP]2Cu4I4 based devices achieved 100% internal quantum (IQE, ηIQE) and exciton utilization efficiencies (EUE, ηEUE). It is noticed that ηEQE differences between these two clusters are about 1.6 folds of those between their ϕPL. Obviously, during EL process, MC-involved excited states of [PhPXZDBFDP]2Cu4I4 worsened exciton quenching through direct carrier capture; while, the reversed CC and LCT energy levels make highly radiative LCT-featured S1 and T1 states of [PXZDBFDP]2Cu4I4 can harvest 100% excitons generated on both ligands and Cu4I4 core. Furthermore, single-emission mode based on the triplet state of [PhPXZDBFDP]2Cu4I4 worsened triplet-triplet annihilation and triplet-polaron quenching. On the contrary, the balanced dual emissions from [PXZDBFDP]2Cu4I4 effectively alleviated exciton accumulation induced quenching at high luminance, therefore limiting efficiency roll-offs.
The different exciton kinetics in PL and EL processes were investigated with steady-state and time-resolved PL and EL spectra (Fig. 4a). BCPO:20% [PXZDBFDP]2Cu4I4 reveals identical PL and EL spectra with the same peak wavelengths and FWHM values. In this case, the composition and ratio of excited states incorporated in PL and EL of [PhPXZDBFDP]2Cu4I4 should be consistent, namely desired balanced radiations from nLCT states. On the contrary, EL peak of BCPO:20% [PhPXZDBFDP]2Cu4I4 are red shifted, in comparison to the PL spectra. More notably, its EL FWHM of 141 nm is about 1.5 folds of the PL value as 93 nm. Through dual-peak fitting, it is clear that different to photo-excitation, because of the statistical majority of electro-generated triplet excitons, the population of 3CC state for [PhPXZDBFDP]2Cu4I4 is markedly increased in EL process, rendering markedly enhanced long-wavelength emission and thereby worsening exciton quenching39.
a Comparison on temperature-dependent luminescent spectra (above) and time decays (below) of the clusters in PL and EL processes. EL spectrum of [PhPXZDBFDP]2Cu4I4 based devices at 300 K is fitted with dual peaks respectively attributed to MLCT and CC radiation; b Time-resolved EL emission spectra (TREES) and sliced TREES contours within 100 µs of the devices. Sliced TRES contours of corresponding films are included for comparison. The enhancement of 3CC band in EL process is highlighted with pink mark.
EL decays of BCPO:20% [PhPXZDBFDP]2Cu4I4 were elongated by at least one order of magnitude, in comparison to its PL decays (Fig. 4a and Supplementary Fig. 34). Obviously, besides exciton diffusion in the devices, largely decelerated exciton radiation verifies the conversion from MLCT to more forbidden CC state of [PhPXZDBFDP]2Cu4I4. In opposite, after offsetting time for carrier transportation, EL decays of BCPO:20% [PXZDBFDP]2Cu4I4 were still slightly shorter than its PL decays, which should be attributed to direct carrier capture and recombination by [PXZDBFDP]2Cu4I4. Furthermore, along with temperature increasing from 50 to 300 K, EL decays of BCPO:20% [PhPXZDBFDP]2Cu4I4 were slightly and gradually shortened, due to enhanced thermal motion. Differently, BCPO:20% [PXZDBFDP]2Cu4I4 based devices revealed almost unchanged decays at 50 and 100 K, but markedly shortened at 200 and 300 K, which are consistent with PL decays, and manifest the electricity-driven dual emission based on the balance between relatively forbidden 3LCT state and allowed 1LCT state of [PXZDBFDP]2Cu4I4 in exciton utilization.
Time-resolved EL emission spectra (TREES) further indicate that in contrast to BCPO:20% [PhPXZDBFDP]2Cu4I4 based analogs with 80% EL emissions dispersed in 20 µs-1 ms, 90% of EL emissions from BCPO:20% [PXZDBFDP]2Cu4I4 were concentrated within 20 µs, and the rest 10% were dispersed within 100 µs, which not only reflects more rapid exciton radiation through dual emission channels, but also alleviates exciton concentration quenching (Fig. 4b). Different to limited temperature dependence of TREES for BCPO:20% [PhPXZDBFDP]2Cu4I4, long tail in TREES at 50 K of [PXZDBFDP]2Cu4I4 attributed to forbidden transitions are suddenly disappeared at 200 K, leading to highly increased emission concentration, which further demonstrates the effectiveness of dual emission in balancing singlet and triplet radiation in the devices (Supplementary Fig. 35). In Consequence, [PXZDBFDP]2Cu4I4 endowed its devices with 100% exciton utilization efficiency, owing to the combined results of (i) the highly radiative LCT featured S1 and T1 states, (ii) reversed CC → LCT energy transfer and (iii) dual emission with suppressed quenching.
Discussion
[PXZDBFDP]2Cu4I4 are constructed by introducing a strong PXZ donor, to demonstrate the feasibility of excited state reversal in copper clusters through ligand engineering. In opposite to [PhPXZDBFDP]2Cu4I4 featuring the D-π-A ligand with weaker LCT and the resulted ordinary MC-centered lowest excited states, [PXZDBFDP]2Cu4I4 reveals the reversed excited state sequence of the low-lying highly radiative LCT-featured S1 and T1 states and high-lying non-radiative MC states, as well as dual emission characteristics, because of the strong D-A interactions in its ligands. Consequently, radiation acceleration and quenching suppression are simultaneously achieved by [PXZDBFDP]2Cu4I4, leading to its state-of-the-art ϕPL of ~90%, ηEQE beyond 35% and 100% ηEUE. These results demonstrate a feasible ligand engineering strategy to break through the bottleneck of cluster emitters in the non-radiative MC-centered lowest excited states, and further extend the space for developing high-performance cluster materials for optical applications including but not limited to information display, lighting, bio-optics and so on.
Methods
Electroluminescence analysis
The devices were layer-by-layer fabricated through firstly spin-coating BCPO: x% cluster layers, and then respectively vacuum evaporating electron-transporting/injecting layers and cathodes. A system composed of a Keithley 4200 source meter, a calibrated silicon photodiode and a PR655 colorimeter was used to measure voltage-current density-luminance characteristics and electroluminescence spectra. Transient and temperature-dependent electroluminescence spectra were recorded with FLS 1000 equipped with a Tektronix AFG3022G function generator.
Data availability
The authors declare that the data generated in this study are provided in Supplementary Information.
References
Zhang, X. & Xu, H. Electroluminescent clusters. Angew. Chem., Int Ed. 63, e202317597 (2024).
Kido, J., Kimura, M. & Nagai, K. Multilayer white light-emitting organic electroluminescent device. Science 267, 1332–1334 (1995).
Uoyama, H., Goushi, K., Shizu, K., Nomura, H. & Adachi, C. Highly efficient organic light-emitting diodes from delayed fluorescence. Nature 492, 234–238 (2012).
Yang, Z. et al. Recent advances in organic thermally activated delayed fluorescence materials. Chem. Soc. Rev. 46, 915–1016 (2017).
Liu, Y., Li, C., Ren, Z., Yan, S. & Bryce, M. R. All-organic thermally activated delayed fluorescence materials for organic light-emitting diodes. Nat. Rev. Mater. 3, 18020 (2018).
Reineke, S. et al. White organic light-emitting diodes with fluorescent tube efficiency. Nature 459, 234–238 (2009).
McCarthy, M. A. et al. Low-voltage, low-power, organic light-emitting transistors for active matrix displays. Science 332, 570–573 (2011).
Xu, H. et al. Recent progress in metal–organic complexes for optoelectronic applications. Chem. Soc. Rev. 43, 3259–3302 (2014).
Berggren, M. et al. Light-emitting diodes with variable colours from polymer blends. Nature 372, 444–446 (1994).
Niikura, H. et al. Probing molecular dynamics with attosecond resolution using correlated wave packet pairs. Nature 421, 823–826 (2003).
Gong, S., Yang, C. & Qin, J. Efficient phosphorescent polymer light-emitting diodes by suppressing triplet energy back transfer. Chem. Soc. Rev. 41, 4797–4807 (2012).
Shirasaki, Y., Supran, G. J., Bawendi, M. G. & Bulović, V. Emergence of colloidal quantum-dot light-emitting technologies. Nat. Photonics 7, 13–23 (2012).
Kim, T. et al. Efficient and stable blue quantum dot light-emitting diode. Nature 586, 385–389 (2020).
Xiang, H., Wang, R., Chen, J., Li, F. & Zen, H. Research progress of full electroluminescent white light-emitting diodes based on a single emissive layer. Light Sci. Appl 10, 206–222 (2021).
Liu, Y. et al. Efficient blue light-emitting diodes based on quantum-confined bromide perovskite nanostructures. Nat. Photonics 13, 760–764 (2019).
Cao, Y. et al. Perovskite light-emitting diodes based on spontaneously formed submicrometre-scale structures. Nature 562, 249–253 (2018).
Lin, K. et al. Perovskite light-emitting diodes with external quantum efficiency exceeding 20 per cent. Nature 562, 245–248 (2018).
Luo, J. et al. Efficient and stable emission of warm-white light from lead-free halide double perovskites. Nature 563, 541–545 (2018).
Matsushima, T. et al. High performance from extraordinarily thick organic light-emitting diodes. Nature 572, 502–506 (2019).
Fakharuddin, A. et al. Perovskite light-emitting diodes. Nat. Electron 5, 203–216 (2022).
Kim, J. S. et al. Ultra-bright, efficient and stable perovskite light emitting diodes. Nature 611, 688–694 (2022).
Ford, P. C., Cariati, E. & Bourassa, J. Photoluminescence properties of multinuclear copper(I) compounds. Chem. Rev. 99, 3625–3648 (1999).
Hashimoto, M. et al. Highly efficient green organic light-emitting diodes containing luminescent three-coordinate copper(I) complexes. J. Am. Chem. Soc. 133, 10348–10351 (2011).
Hamze, R. et al. Eliminating nonradiative decay in Cu(I) emitters: >99% quantum efficiency and microsecond lifetime. Science 363, 601–606 (2019).
Ying, A., Ai, Y., Yang, C. & Gong, S. Aggregation-dependent circularly polarized luminescence and thermally activated delayed fluorescence from chiral carbene-CuI-amide enantiomers. Angew. Chem., Int Ed. 61, e202210490 (2022).
Wang, H.-J. et al. A configurationally confined thermally activated delayed fluorescent two-coordinate cui complex for efficient blue electroluminescence. Angew. Chem., Int Ed. 62, e202217195 (2023).
Martin, A. et al. Mechano-stimulus and environment-dependent circularly polarized TADF in chiral copper(I) complexes and their application in OLEDs. J. Am. Chem. Soc. 145, 4438–4449 (2023).
Zhang, Q. et al. Harnessing of cooperative Cu⋅⋅⋅H interactions for luminescent low-coordinate copper(I) complexes towards stable OLEDs. Angew. Chem., Int Ed. 64, e202419290 (2024).
Tang, R. et al. Highly robust CuI-TADF emitters for vacuum-deposited OLEDs with luminance up to 222 200 cd m−2 and device lifetimes (LT90) up to 1300 hours at an initial luminance of 1000 cd m−2. Angew. Chem., Int Ed. 61, e202203982 (2022).
Zhang, J. et al. Balanced dual emissions from tridentate phosphine-coordinate copper(I) complexes toward highly efficient yellow OLEDs. Adv. Mater. 28, 5975–5979 (2016).
Zink, D. M. et al. Heteroleptic, dinuclear copper(I) complexes for application in organic light-emitting diodes. Chem. Mater. 25, 4471–4486 (2013).
Zhang, Q. et al. Triplet exciton confinement in green organic light-emitting diodes containing luminescent charge-transfer Cu(I) complexes. Adv. Funct. Mater. 22, 2327–2336 (2012).
Li, X. et al. Bluish-green Cu(I) dimers chelated with thiophene ring-introduced diphosphine ligands for both singlet and triplet harvesting in OLEDs. ACS Appl Mater. Interfaces 11, 3262–3270 (2019).
Wang, J.-J. et al. High efficiency warm-white light-emitting diodes based on copper–iodide clusters. Nat. Photonics 18, 200–206 (2024).
Bizzarri, C., Spuling, E., Knoll, D. M., Volz, D. & Bräse, S. Sustainable metal complexes for organic light-emitting diodes (OLEDs). Coord. Chem. Rev. 373, 49–82 (2018).
Liu, Y., Yiu, S.-C., Ho, C.-L. & Wong, W.-Y. Recent advances in copper complexes for electrical/light energy conversion. Coord. Chem. Rev. 375, 514–557 (2018).
Zhang, Q.-C., Xiao, H., Zhang, X., Xu, L.-J. & Chen, Z.-N. Luminescent oligonuclear metal complexes and the use in organic light-emitting diodes. Coord. Chem. Rev. 378, 121–133 (2019).
Zhang, L. & Li, B. Room-temperature pure blue-emitting phosphorescent multinuclear Cu(I)-based emitters. J. Electrochem Soc. 156, J174–J178 (2009).
Xie, M., Han, C., Zhang, J., Xie, G. & Xu, H. White electroluminescent phosphine-chelated copper iodide nanoclusters. Chem. Mater. 29, 6606–6610 (2017).
Lu, X. et al. Efficient doped and non-doped light-emitting diodes based on a TADF-emitting Cu4Br4 cluster. Inorg. Chem. Front 11, 2775–2783 (2024).
Sun, J. et al. Ligand-mediate exciton allocation enables efficient cluster-based white light-emitting diodes via single and heavy doping. Nat. Commun. 15, 4997–5008 (2024).
Liu, W. et al. All-in-one: achieving robust, strongly luminescent and highly dispersible hybrid materials by combining ionic and coordinate bonds in molecular crystals. J. Am. Chem. Soc. 139, 9281–9290 (2017).
Zhu, K. et al. A new type of hybrid copper iodide as nontoxic and ultrastable LED emissive layer material. ACS Energy Lett. 6, 2565–2574 (2021).
Ma, X.-H. et al. Carbene-stabilized enantiopure heterometallic clusters featuring EQE of 20.8% in circularly-polarized OLED. Nat. Commun. 14, 4121–4132 (2023).
Zhang, N. et al. Sky blue and yellow cluster light-emitting diodes based on asymmetric Cu4I4 nanocubes. Research 2022, 0005 (2022).
Xie, M. et al. Highly efficient sky blue electroluminescence from ligand-activated copper iodide clusters: Overcoming the limitations of cluster light-emitting diodes. Sci. Adv. 5, eaav9857 (2019).
Zhang, N. et al. Overcoming efficiency limitation of cluster light-emitting diodes with asymmetrically functionalized biphosphine Cu4I4 Cubes. J. Am. Chem. Soc. 144, 6551–6557 (2022).
Zhong, C., Zhang, X., Gong, Z. & Xu, H. Recent advances in electroluminescent metallic nanoclusters: from materials to devices. Nano Lett. 24, 9415–9428 (2024).
Kyle, K. R., Ryu, C. K., Ford, P. C. & DiBenedetto, J. A. Photophysical studies in solution of the tetranuclear copper(I) clusters Cu4I4L4 (L = pyridine or substituted pyridine). J. Am. Chem. Soc. 113, 2954–2965 (1991).
Ford, P. C. & Vogler, A. Photochemical and photophysical properties of tetranuclear and hexanuclear clusters of metals with d10 and s2 electronic configurations. Acc. Chem. Res 26, 220–226 (1993).
Perruchas, S. et al. Mechanochromic and thermochromic luminescence of a copper iodide cluster. J. Am. Chem. Soc. 132, 10967–10969 (2010).
Ma, Y. et al. Allochroic cluster light-emitting diodes based on unique μ3-tetraphosphine Cu3X3 crowns with tunable excited states. Sci. Adv. 10, eadk3983 (2024).
Hofbeck, T., Monkowius, U. & Yersin, H. Highly efficient luminescence of Cu(I) compounds: thermally activated delayed fluorescence combined with short-lived phosphorescence. J. Am. Chem. Soc. 137, 399–404 (2015).
Fan, X. et al. Structural isomerization in Cu(I) clusters: tracing the Cu thermal migration paths and unveiling the structure-dependent photoluminescence. CCS Chem. 5, 350–360 (2023).
Deng, C.-L. et al. Hierarchical homochiral assembly of polyhedral cage-type nanoclusters. CCS Chem. 6, 2537–2548 (2024).
Zhao, Z.-H. et al. Buckling cluster-based H-bonded icosahedral capsules and their propagation to a robust zeolite-like supramolecular framework. Nat. Commun. 15, 9401–9410 (2024).
Zhang, N. et al. Cluster light-emitting diodes containing copper iodine cube with 100 % exciton utilization using host-cluster synergy. Angew. Chem. Int Ed. 62, e202305018 (2023).
Acknowledgements
H.X. acknowledges the support from National Natural Science Foundation of China (NSFC) (Grant No. 22325502, 92461304, W2412073, 22305073 and 52273173).
Author information
Authors and Affiliations
Contributions
H.X. conceived, designed, and supervised the project. Y.M., C.L., J.F., and Y.Y. synthesized clusters. Y.M., Y.W., J.Z. and C.H. performed photophysical analyses. Y.M. and C.D. fabricated devices and measured electroluminescence properties. Y.M. and H.X. wrote the manuscript. All authors participated in the discussion and analysis of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial and non-financial interests.
Peer review
Peer review information
Nature Communications thanks Hong-Bin Yao and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Meng, Y., Liu, C., Fei, J. et al. Excited state reversal in copper iodide clusters enables 100% exciton radiation. Nat Commun 17, 933 (2026). https://doi.org/10.1038/s41467-025-67664-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-67664-x


![Fig. 2: Photophysical properties of [PXZDBFDP]2Cu4I4 and [PhPXZDBFDP]2Cu4I4.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs41467-025-67664-x/MediaObjects/41467_2025_67664_Fig2_HTML.png)
![Fig. 3: Electroluminescence performances of [PXZDBFDP]2Cu4I4 and [PhPXZDBFDP]2Cu4I4 based devices.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs41467-025-67664-x/MediaObjects/41467_2025_67664_Fig3_HTML.png)
![Fig. 4: Exciton kinetics of [PXZDBFDP]2Cu4I4 and [PhPXZDBFDP]2Cu4I4 based devices.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs41467-025-67664-x/MediaObjects/41467_2025_67664_Fig4_HTML.png)