Abstract
The recycling of spent lithium-ion batteries is essential for sustainable development of clean energy industry. Traditional recycling methods face challenges such as high energy/chemical consumption and limited adaptability. This study introduces a ‘Three-in-One’ strategy that leverages mechanochemical (MC) treatment to enhance lithium recovery, transition metal upgrading, and CO2 sequestration. MC treatment induces micro-segregation of lithium and transition metals, resulting in a structurally disordered material with a Li-rich surface, facilitating selective lithium extraction. Subsequently, CO2 is used as the sole reagent and lithium leaching efficiency exceeding 95% is achieved by forming lithium bicarbonate. Conducted under ambient conditions without additional grinding aids or leaching reagents, this method minimizes environmental impact. Transition metals are simultaneously transformed into high-performance oxygen evolution reaction (OER) catalysts, demonstrating an overpotential of 322 mV at a current density of 10 mA cm-2. These catalysts maintain stability over 200 h of operation. This approach not only provides an efficient pathway for lithium recovery but also upcycles spent cathode materials into valuable catalysts, supporting sustainable energy conversion technologies. The strategy is particularly effective for high-Ni cathode systems, offering significant practical advantages.
Similar content being viewed by others
Introduction
The rapid application of lithium-ion batteries (LIBs) as essential clean energy storage devices in electric vehicles, portable electronics, and grid-scale storage is generating a growing stream of spent batteries1,2. The global volume of spent LIBs is projected to reach 381 million metric tons by 20503. Improper management of such waste streams may not only result in substantial waste of strategic metal resources such as lithium (Li)4, cobalt (Co), and nickel (Ni) but also pose major environmental pollution risks, which threaten ecosystems and human health. Consequently, the efficient recovery of critical metals from spent LIBs is crucial for economic, environmental, and social sustainability5,6.
Current recycling of critical metals from spent LIBs relies on three principal routes: pyrometallurgy, hydrometallurgy, and direct regeneration7. Pyrometallurgy uses high-temperature smelting (typically >1400 °C)8 to recover Co, Ni and Cu, while the recovery rates of lithium as well as Fe and P (in a LiFePO4 battery) are low. The process is inherent with stringent requirements for input materials and high energy consumption6. In contrast, hydrometallurgy offers improved recovery efficiency for different elements through chemical leaching with solvents such as mineral/organic acids or deep eutectic solvents9,10,11. Nevertheless, conventional hydrometallurgical processes often encounter challenges, such as high chemical consumption and lengthy purification sequences12. Direct regeneration is a promising alternative that employs shorter processing routes to restore degraded cathode materials (e.g., via solid-state, sol-gel, or hydrothermal Li replenishment)13,14,15 or increases cell capacity (e.g., using Li-compensating additives such as Li-Naph)16,17. However, its practical application is limited because it can only process less complex spent LIB streams18. Comparative analysis identifies hydrometallurgy as the most versatile and widely applicable approach, combining moderate energy requirements, adaptability to diverse battery chemistries, and tuneable metal separation processes.
Compared with impurity-sensitive conventional hydrometallurgy, selective leaching improves Li recovery and simplifies purification19,20. The mechanisms preferentially induce targeted phase transformations in spent cathodes, separating Li from transition metal (TM) phases, driven by strategies such as adding reductants or applying elevated temperature/pressure21. For example, carbothermic reduction converts spent cathodes into lithium carbonate (Li2CO3) and TM elements or their oxides20,22,23, and sulfation roasting yields lithium sulfate (Li2SO4) and TM oxides24,25,26. However, these methods often require substantial reagent inputs, produce hazardous emissions and secondary pollution, and generate leaching residues. These residues usually require acid leaching for conversion into salt solutions prior to reuse, which severely decreases their value. Consequently, optimizing resource utilization across the selective leaching chain requires greener, more efficient processes and solutions for managing low-value residues. Flash Joule heating27 can enable one-step selective Li extraction via second-long, high-temperature reactions. This instantaneous heating offers greener processing and uniquely induces structural transformations in spent cathodes, which could convert residual metals into self-assembled multi-metal hydroxide catalysts28. Electrochemical methods selectively leach Li at ambient temperature by using an applied potential. The resulting structurally defective, Li-depleted residue can serve directly as a functional material, such as an oxygen evolution reaction (OER) catalyst, but electrode reconstruction requirements hinder scale-up29,30,31. Mechanical methods selectively leach Li through induced room-temperature phase transitions32; however, grinding additives are associated with the risk of impurity contamination33,34. In summary, existing technologies have yet to optimally balance mild processing conditions, minimal chemical input, and the production of high-value outputs.
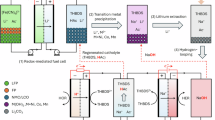
Here, we propose a “Three-in-One” strategy that leverages elemental micro-segregation in spent cathode materials, which is induced through controlled mechanochemical (MC) treatment. This innovative approach enables the simultaneous selective extraction of Li and the transformation of TM-based leaching products into OER catalysts using only CO2, as depicted in Fig. 1a. The entire procedure was conducted at ambient temperature without the use of grinding aids or leaching agents. Notably, CO2 fixation occurs simultaneously during the metal recovery process. Our strategy is inspired by prior work on the synthesis of Li-rich disordered rock salt cathode materials (DRX)35, which showed that ball-milling could alter the initial distribution of Li and TM elements within the cathode, creating Li-rich domains and increasing the charge–discharge capacity. The mechanical force in our study serves a similar purpose of disordering the arrangement of Li and TM elements to induce their micro-segregation, which endows these elements with varying leaching activities. Ultimately, this micro-segregation results in a Li leaching efficiency of >95% only with CO2—which could be prospectively derived entirely from industrial waste streams—thereby enhancing the environmental and economic benefits of the process. Moreover, the synergistic effects of MC treatment and selective Li extraction in a weakly acidic environment result in the creation of a catalyst with exceptional OER performance.
a Schematic diagram of the three-in-one strategy for recycling spent LiNixCoyMnzO2. b XRD results of P-NCM811 and MA-NCM811 for different activation time. c The FMWH of (003) and (104), and the value of I(003)/ I(104) in the XRD results of P-NCM811 and MA-NCM811 (inset: schematic of the mechanochemical transformation pathway of NCM811 after 0.5 h of activation). d The Rietveld XRD refinement of MA-NCM811-2h. e The relationship between activation time and amorphous phase ratio in MA-LTMO. f The effect of H2O and H+ on the leaching efficiency of Li in P-NCM811 and MA-NCM811. g The pH-pressure relationship profile (25 °C) of the CO2-H2O equilibrium system. And h effect of CO2 on the Li/TMs leaching from P-LTMO and MA-LTMO (activated for 2 h), and their Li leaching disparity. Data in (f) and (h) are presented as the mean ± standard deviation (SD) from three independent experiments.
Results
Mechanochemical treatment for selective extraction of Li
In the MC process, NCM811, for example, transforms into an amorphous phase, as evidenced by X-ray Diffraction (XRD) peak broadening36 (Fig. 1b, c). The initial increase followed by a sharp decrease in the I(003)/I(104) value (Fig. 1c) reveals two-stage structural evolution: mechanical energy first induces (003)-plane grain boundary slip in the R-3m layered structure, enhances (003) plane exposure, and then drives irreversible transformation to the disordered rock-salt phase (Fm-3m) through prolonged activation37,38. Rietveld refinement with internal standardization39 (Fig. 1d and Supplementary Fig. 1) revealed a 72.9% amorphous content after 2 h of activation (Fig. 1e), which subsequently decreased to 47.9% with extended MC treatment, confirming energy-dependent phase dynamics40,41. Amorphization initially dominated42, but the excess energy (activation time >2 h) surpassed the deformation barrier, inducing lattice reorganization, as evidenced by the unit cell information in Supplementary Fig. 1, which reduced the amorphous content43,44.
Systematic leaching experiments validated the correlation between mechanochemically induced amorphization and the leaching behaviors of Li and TMs. As shown in Fig. 1f, pristine NCM811 (P-NCM811) exhibited a mere 4.68% Li leaching efficiency in aqueous systems. In contrast, after mechanochemical activation (activation time ≥2 h), the Li leaching efficiency of mechanochemical activated NCM811 (MA-NCM811) exceeded 75% under identical conditions, with nearly 100% Li selectivity, confirming the formation of water-soluble Li phases during the MC process. Upon introducing a H+ source into the leaching system (Fig. 1f), H+ induces nonselective metal leaching in P-NCM811, whereas in MA-NCM811, it demonstrates superior selectivity for Li extraction with a Li leaching efficiency of >95% when the nH⁺/nLi⁺ ratio is 1. Experiments designed to elucidate this phenomenon (Supplementary Note 2) revealed a more critical role for the MC-induced amorphization of the crystal structure than for morphological changes such as particle size reduction. Moreover, a prolonged activation time (activation time >2 h) led to gradual dissolution of the TMs in acidic system. In terms of the pH endpoint in acidic systems (Supplementary Fig. 6a), the final pH first increased (activation time<2 h) but then decreased (activation time >2 h), indicating that excessive activation diminished the difference in leaching activity between Li and TMs, which could be attributed to recrystallization reducing their differences in kinetic leaching. This result maintains the solution pH within a lower value, thereby allowing for the dissolution of low-valence TMs in the TM-H2O system according to the E-pH diagrams (Supplementary Fig. 6b–d). Consequently, 2 h was chosen as the optimal activation time to maximize structural disorder while also maximizing the leaching selectivity between Li and TM. The sample prepared under these conditions was thus used as the starting material for subsequent leaching experiments.
This phenomenon facilitates a CO2-assisted leaching system, which relies solely on CO2 as the only H+ source through dissolution–dissociation equilibrium (Eq. 1). Under ambient pressure, CO2 could serve as a H+ source, selectively extracting 87% of Li from MA-NCM811 (Supplementary Fig. 7). However, the H+ concentration generated under these conditions was insufficient to completely dissolve the water-insoluble residual Li. Enhancing the dissolution-dissociation equilibrium of CO2 (e.g., by increasing the system pressure) effectively promoted H+ release (Fig. 1g)45. A systematic investigation of CO2 pressure and leaching time (Supplementary Fig. 7) revealed that insufficient pressure or an overly short duration leads to inadequate H+ supply, resulting in incomplete Li extraction (<90%). Conversely, excessively high pressure or a prolonged duration disrupts the TM-O framework, inducing codissolution of TMs and thereby reducing selectivity. Nevertheless, the CO32–/HCO3– species generated in situ during CO2 dissociation act as buffers and can complex with TM ions, thus limiting their dissolution and helping to maintain selectivity. Under the optimized conditions of 0.5 MPa CO2 pressure and 1 h leaching time, the system achieves a balance between H⁺ generation and buffering effects, ultimately enabling highly efficient Li extraction while effectively preserving the TM framework. With respect to other ternary cathode materials, this method can achieve excellent Li selective extraction efficiency. As shown in Fig. 1h and Supplementary Fig. 8, for NCM811, NCM622, NCM111 and spent layered lithium transition metal oxide (S-LTMO, where TM represents Ni, Co, and Mn), the Li leaching efficiencies were 95.43%, 91.56%, 88.53% and 93.13%, respectively, and the selectivities of Li were 94.63%, 97.04%, 99.37% and 98.84%, respectively. In contrast, the weak acidity of CO2 was insufficient to disrupt the lattice structure of pristine LTMOs (P-LTMOs), resulting in a relatively low Li leaching efficiency (<15%). Further exploration of the universality of the raw materials, as discussed in Supplementary Note 3, revealed that this process was not applicable to cathode materials with non-layered structures (e.g., LiFePO4 and LiMn2O4) under identical conditions. This inapplicability underscores that the effectiveness of the process is contingent upon a specific amorphization pathway. Another advantage of this process is the absence of cation (e.g., Na+, K+) and anion (e.g., Cl–, SO42–) impurities introduced into the system, as well as the elimination of any gaseous or liquid waste that would require subsequent treatment. The Li-rich leachate (LiHCO3) was converted directly into a Li2CO3 product with a purity of >98.5% (Supplementary Fig. 14 and Supplementary Table 2) by thermal decomposition46.
Leaching mechanisms of Li and characterization of leaching products
Additional characterization was used to further elucidate the leaching pathway of Li in MA-LTMO. The XRD patterns of MA-NCM811 and its leaching products in different systems are shown in Fig. 2a. The leaching product from the aqueous system (MA-NCM811-WL) exhibited a pattern highly similar to that of MA-NCM811, with no notable new peaks. In contrast, the leaching process in the CO2rich aqueous solution (MA-NCM811-CO2L) induced more significant phase changes, specifically showing a notable increase in the intensity of the characteristic peak at 18.9°, whereas the intensities of the remaining characteristic peaks were markedly reduced. On the basis of the earlier leaching results, it is evident that Li was primarily extracted from the amorphous regions in the aqueous system, which had minimal impact on the highly disordered residual crystalline regions of MA-NCM811. The CO2 system effectively disrupted the residual crystal structure, thereby releasing additional lattice Li. Depth-profiling X-ray Photoelectron Spectroscopy (XPS) analysis for Li 1s (Supplementary Fig. 15) revealed that the characteristic peaks related to Li completely disappeared on the surface of MA-NCM811-CO2L. A weak broad peak (attributed to Li-O-M bonding) gradually emerged at approximately 54 eV with increasing Ar+ etching depth47. This confirms that a small amount of residual lattice Li remains within the bulk phase of MA-NCM811-CO2L.
a XRD spectrums of MA-NCM811 and the leaching products of MA-NCM811 in aqueous system (MA-NCM811-WL) and CO2-assistedleaching system (MA-NCM811-CO2L). b FTIR results of P-NCM811-CO2L and MA-NCM811-CO2L. c Schematic diagram of Li leaching pathway in a CO2-rich aqueous solution. d HRTEM image of MA-NCM811-CO2L (green circles refer to disorder domains) with an expanded view and FFT pattern of a selected region (yellow square). e, f O 1 s and Ni 2p in-depth profile XPS of MA-NCM811-CO2L.
TG-MS results indicate that the MA-NCM811-CO2L releases H2O and O2 gases at 200 °C (Supplementary Fig. 16). In combination, the infrared spectroscopy result of MA-NCM811-CO2L (Fig. 2b) shows a broad peak at 3490 cm–1, a doublet peak in the range of 1485-1420 cm–1 and a sharp peak located at approximately 880 cm–1. These spectral features collectively demonstrate the presence of –OH and CO32– in the leaching products from CO2 system48. Real-time pH monitoring (Supplementary Fig. 17) revealed distinct evolution patterns in which the water leaching process pH increased from 7 to a peak and then decreased to 10.5, whereas the CO2–assisted leaching process pH increased from 4.2 to 6.1 and plateaued below 7. On the basis of the E-pH diagrams in Supplementary Fig. 18, the dissolution of TM2+ and subsequent complexation of TM2+ with CO32– may have occurred in this system. The absence of these phenomena in P-NCM811-CO2L also confirms that they originated solely from the pre-MC process (Fig. 2b and Supplementary Fig. 19). Comparative analysis of the Ni K-edge X-ray absorption near-edge structure (XANES) and extended X-ray absorption fine structure (EXAFS) spectra of MA-NCM811 and MA-NCM811-WL (Supplementary Fig. 20) revealed traces of H intercalation during the water leaching process. These findings demonstrate that the leaching mechanism of Li+ from MA-LTMO is not limited to a single pathway, even under aqueous conditions. J.R. Dahn et al.49. reported that during the washing of high-Ni LTMO, the ion exchange of Li⁺/H⁺ occurs more frequently in aqueous solutions with higher pH, whereas the dissolution of Li and TMs may occur in solutions with slightly lower pH.
In brief, the H+ introduced by CO2 may be involved in ion exchange reactions and acidic dissolution reactions within the residual insoluble Li in the lattice. The released Li+ from MA-LTMO in the CO2-assisted leaching may have three pathways: (i) dissolution in the soluble Li-rich phase (which is analogous to lithium oxide, Li2O) (Eq. 2), (ii) ion exchange of Li+ and H+ (Eq. 3), and (iii) acidic dissolution of lattice Li (Eq. 4), as shown in Fig. 2c. TMs may undergo dissolution followed by complexation under the weakly acidic influence of CO2 during this process (Eq. 5).
According to the preceding analysis, the leaching product is primarily composed of transition metal oxides (TMOs with trace amounts of residual lattice Li), oxyhydroxides (TMOOH), and complexation products of TMs. High-resolution Transmission Electron Microscopy (HR-TEM) (Fig. 2d) of the MA-NCM811-CO2L sample revealed that CO2-assisted leaching preserves the disordered structure of MA-LTMO. A 2 nm-thick layered phase covered the particle surface, with an interlayer spacing of 0.21 nm, which is consistent with the (105) plane of NiOOH50. In contrast, the core consisted of amorphous domains coexisting with spinel, rock-salt and layered phases. This core-shell structure is further supported by the variation in the hydroxyl oxygen (–OH) content observed by depth-profiling O1s XPS (Fig. 2e). Additionally, the concentration of oxygen vacancies on the surface was significantly greater than that in the bulk phase, as they are generated to maintain charge neutrality during anion extraction51 (Supplementary Note 4). These results suggest that the leaching products with disordered Li-deficient TMO cores and surfaces were enriched in TMOOH with oxygen vacancies. The Ni 2p XPS spectra (Fig. 2f) further revealed a higher proportion of Ni2+ species on the surface, which aligns with the proposed leaching pathway. Scanning electron microscopy (SEM) results (Supplementary Fig. 22) revealed that the primary particle size of the leaching product was between 50 and 100 nm. The combined features of the nanoscale dimensions and surface enrichment of oxygen vacancies and –OH endow the leaching product with a high catalytic surface area and abundant active sites, which are critical for enhancing the catalytic activity of TM-based materials52.
Micro-segregation of Li/TM in MA-LTMO
The reasons for the significant difference in the reactivity of Li and TMs after activation were further explored. Notably, MA-NCM811-2h developed lithium carbonate (Li2CO3) phases during ambient ageing (320 h), whereas its pristine counterpart demonstrated exceptional air stability with no detectable Li2CO3 formation under identical exposure conditions (Supplementary Fig. 23). This contrast in reactivity confirms that the activation process generates an amorphous Li-containing phase, which is highly reactive to spontaneous CO2 chemisorption (Li2O + CO2→Li2CO3, ΔGθ = − 176 kJ mol–1, as shown in Supplementary Fig. 24). The hysteresis observed in the CO2 adsorption-desorption isotherms of MA-NCM811 collected at 0 °C (Fig. 3a and Supplementary Fig. 25) also confirms irreversible CO2 adsorption sites. And MA-NCM811 had a peak specific surface area (107 m2 g–1) at 2 h of activation, which is consistent with the maximum CO2 uptake. Prolonged MC activation led to a reduction in the CO2 adsorption capacity, which was attributed to particle agglomeration, as evidenced by the SEM results of MA-NCM811 in Supplementary Fig. 2 (the specific surface area decreased by ~23.6%), eliminating partial CO2-chemisorption-active sites. This finding also indicates that the unique surface of the activated products, which can chemically adsorb CO2, warrants further exploration.
a CO2 adsorption and desorption isotherms of P-NCM811, MA-NCM811-2h and MA-NCM811-16h at 0 °C. (inset: surface specific areas of P-NCM811 and MA-NCM811 with different activation times). b TOF-SIMS mass spectrum of the surface in P-NCM811 and MA-NCM811 (Supplementary Figs. 26 and 27). c TOF-SIMS depth profile of Li+ in P-NCM811 and MA-NCM811, and the sputtering depth is calibrated by the SiO2 sputtering rate (Supplementary Fig. 28). d Li 1s, e O 1s and f Ni 2p in-depth profile XPS of MA-NCM811.
Mechanochemically induced amorphization may promote elemental segregation by perturbing the spatially ordered distribution of elements in crystal structures53. Time-of-Flight Secondary Ion Mass Spectrometry (TOF-SIMS) depth profiling (Fig. 3b, c) first revealed a pronounced chemical gradient in MA-NCM811 in which the Li+ abundance decreased sharply from the surface inward, whereas the TM+ abundance increased, confirming that the Li-rich surface layer was approximately 20 nm thick. XPS depth-etching (Fig. 3d–f) further resolved the chemical states of the surface elements, showing that the surface was dominated by LiOH/Li2CO3, which may have been formed by the adsorption of atmospheric H2O and CO2 onto the Li-rich amorphous phase, identified by O 1s at 531.5 eV54 and Li 1s at 55 eV54,55,56. From the Ni 2p spectrum of MA-NCM811 (Fig. 3f and Supplementary Fig. 29), the intensity of the Ni 2p peaks on the surface was significantly lower than that in the bulk phase, and the Ni2+/Ni3+ ratio decreased with depth but exceeded 14% in the outer regions. On the basis of the XPS etching results and the element concentration inflection points from TOF-SIMS, the MC process disrupts the spatially ordered distribution of elements and thereby drives the segregation of Li and TMs. This mechanism induces the formation of such a reconstruction layer (~20 nm) in the near-surface region of MA-LTMO, which is essentially a depth-gradient transition zone dominated by a Li-rich amorphous phase. With increasing depth, the concentration of active Li sites capable of reacting with atmospheric components decreases, while the valence state distribution of Ni gradually changes, and its concentration shows an opposite trend to that of Li.
Causes for the Micro-Segregation of Li/TM
The Li-rich domain generated from elemental segregation is water soluble, which primarily leads to the selective extraction of Li from MA-LTMOs. However, the origin of elemental segregation caused by MC treatment remains unresolved. Prior studies suggest that high-valent Ni ions undergo charge redistribution during electrochemical processes, releasing oxygen and reducing Ni oxidation states57,58. Here, the Ni K-edge XANES spectrum (Fig. 4a) revealed a downwards shift in the Ni K-edge energy (~1.5 eV) in MA-NCM811, confirming the reduction in Ni. The results from the EXAFS spectra (Fig. 4b) revealed an increase in the interatomic distances of Ni-O and Ni-M after mechanochemical activation, with a gradual weakening of the Ni-M bond strength, indicating that the short-range structure was significantly disrupted. The Raman spectra shown in Fig. 4c and Supplementary Fig. 30 demonstrate that P-NCM811 exhibits typical layer-like anisotropic vibration modes59. After MC treatment, the A1g peak60 showed broadening and a pronounced low-frequency shift (31.3 cm–1), along with attenuation and eventual disappearance of the Eg peak. This behavior is similar to that of the disordered rock salt structure61, indicating a fundamental transformation from an ordered layered structure to a highly disordered configuration, with the loss of distinct in-plane and out-of-plane vibrational modes. Further analysis combining EXAFS spectra and HR-TEM of MA-NCM811 (Fig. 4d) revealed the coexistence of multiple phases (layered, rock salt and spinel phases)62, reflecting the basic composition of its highly disordered structure. These changes in the chemical state and coordination environment of Ni can be attributed to mechanochemically facilitated Li/TM cation migration, as evidenced by the XRD refinement of MA-NCM811 (Supplementary Fig. 1). It showed significant evolution in the Li/Ni antisite mixing ratio, which is a key parameter for structural integrity63. The mixing ratio first increased to 45.6% after 2 h, reflecting structural evolution driven by atomic displacement.
a Normalized Ni K-edge XANES (inset: expanded view of the absorption edge region), and b EXAFS spectra of P-NCM811 and MA-NCM811 for different activation times. c Raman results, and e Mn 2p in XPS results of P-NCM811 and MA-NCM811. d HRTEM images of P-NCM811 and MA-NCM811 with selected regions (yellow squares) alongside their corresponding FFT patterns. f The schematic graphic for micro-segregation of Li/TM in MA-LTMO.
Collectively, the structural evolution observed in MA-NCM811 fundamentally originates from mechanochemical coupling effects (Fig. 4f). High-valent Ni ions undergo reduction to Ni2+ by accepting electrons from lattice oxygen and partially from Mn4+, as evidenced by XPS-detected Mn7+ (Fig. 4e) and Mn-dependent purple leachate colouration (Supplementary Fig. 31). Moreover, marked Li/Ni antisite disordering further triggers phase micro-segregation, resulting in the formation of distinct Li-rich domains and TM-concentrated regions. On the basis of previous analysis of the phase distribution characteristics of the activated product, we found that MC treatment creates a highly amorphous and defect-rich structure. Within this structure, Li/TM micro-segregation occurs; specifically, Li becomes enriched in the amorphous surface reconstruction layer, whereas TM remains stabilized within the TM-O framework composed of alternating tetrahedral and octahedral coordination, with only a small amount of lattice Li embedded in the framework. This unique structure, resulting from the synergistic effect of macroscopic grain refinement and microscopic interelement electron transfer and atomic displacement, ultimately converts the Li locked in the original layered structure into a state that is highly reactive towards H2O and CO2. This mechanism effectively overcomes the inherent limitations encountered when H2O and CO2 are used directly to leach pristine cathode materials.
In-situ Upcycling Leaching Products for OER catalysts
The unpaired d-orbital electrons of TMs play a pivotal role in facilitating redox reactions, while their multivalent characteristics endow them with high performance in catalytic processes64. These intrinsic properties render TMs ideal catalysts for diverse electrochemical reactions, with Ni and Co standing out as particularly prominent candidates65,66. Notably, the increase in catalytic activity depends not solely on elemental properties but also critically on the rational regulation of microstructural features. Prior studies have demonstrated that introducing structural disorder can significantly increase catalytic activity67,68, forming a critical strategy for modifying nonprecious metal catalysts. The MC process has been found to impart favorable structural disorder, whereas the nondestructive Li extraction process preserved this disorder and concurrently established approximate core–shell heterointerfaces on the surfaces of the leaching products. These interfaces are hypothesized to lower the oxygen vacancy formation energy65, thereby enhancing the catalytic performance of the OER.
The electrocatalytic performance of MA-LTMO-CO2Ls was evaluated for the OER in 1.0 M KOH through electrochemical measurements. Commercial Co3O4 was used as a contrast sample. As shown by the linear sweep voltammetry (LSV) curves in Fig. 5a, compared with commercial Co3O4, MA-LTMO-CO2Ls possess superior electrocatalytic activity, as evidenced by their significantly lower overpotentials, which are required to achieve a current density of 10 mA cm–2. Among them, MA-NCM811-CO2L demonstrated the best performance, with an overpotential of 322 mV, which is considerably lower than 367 mV required by Co3O4. The fitted Tafel slopes of MA-NCM811-CO2L (90 mv dec–1) and MA-NCM622-CO2L (127 mV dec–1) were lower than that of commercial Co3O4 (140 mV dec-1), indicating that the catalytic kinetics of high-Ni leaching products for the OER are more favorable. This finding is consistent with the results of the electrochemical impedance spectroscopy (EIS) results presented in Fig. 5c, which revealed that the impedance arcs of the high-Ni leaching products were smaller than those of their counterparts and commercial Co3O4, suggesting that they have the lowest charge transfer resistance and exhibit the fastest OER kinetics. High-Ni systems exhibited superior catalytic synergies, which could be attributed to their elevated Ni3+ content, which facilitates the adsorption–desorption of OH→O intermediates66,69. In addition, to comprehensively evaluate the OER performance, the electrochemical surface area (ECSA) was determined via double-layer capacitance (Cdl) measurements from CV scans in the nonfaradaic region at various scan rates (Fig. 5d and Supplementary Fig. 32). The leaching products, especially MA-NCM811-CO2L, demonstrated a superior Cdl of 10.01 mF cm–2, markedly exceeding that of Co3O4 (6.52 mF cm–2), indicating a higher density of OER active sites likely originating from the pre-MCM process. Critically, the operational stability was validated by chronopotentiometry testing (Supplementary Fig. 33), which revealed a minimal potential increase over the course of 24 h. Notably, the leaching product derived from spent cathode materials (MA-S-LTMO-CO2L) also exhibited excellent stability over 200 h of operation under identical testing conditions (Fig. 5e). This observation strongly suggests that the structural and compositional defects (e.g., trace impurities) introduced into the cathode waste during long-term electrochemical cycling and insufficient pretreatment did not compromise the catalytic performance of the final leaching product. In fact, the performance continued to improve during operation. It was further examined by LSV (Fig. 5e, inset) and EIS (Supplementary Fig. 34), which revealed a more favorable polarization curve after 200 h of operation than before the stability test. The outstanding stability underscores the robustness of the catalytic sites formed in this waste-to-catalyst process. Benchmarking against state-of-the-art TM-based OER electrocatalysts (Fig. 5f and Supplementary Table 3), our catalyst demonstrated competitive η10 values, with the performance advantage stemming from tailored interfacial engineering and disordered state modulation. Collectively, these findings establish a sustainable upcycling pathway that transforms battery waste into high-performance OER catalysts, bridging resource circularity and energy conversion technology.
a LSV polarization curves, b corresponding Tafel plots, c EIS spectrums of MA-LTMO-CO2Ls and Co3O4. d Double layer capacitance (Cdl) of MA-LTMO-CO2Ls and Co3O4, derived from linear regression fits with R2 > 0.995. e Chronopotentiometry curve (10 mA cm–2, 200 h, without iR compensation) for MA-S-LTMO-CO2L, with the arrow marking the electrolyte replenishment (inset: LSV polarization curves of MA-S-LTMO-CO2L before and after long-term stability). And f the overpotentials at a current density of 10 mA cm–2 for MA-LTMO-CO2Ls and other reported TM-based OER catalysts (Supplementary Table 3).
Discussion
In summary, we present a mechanochemistry-driven upcycling paradigm enabling concurrent Li recovery (>95%) and direct upcycling of spent LIB cathodes into high-performance OER catalysts using CO2 as the sole processing reagent. Studies reveal that mechanochemically induced Li/TM directional micro-segregation endows Li and TM with differential reactivities, while H+ supplied by CO2 achieve selective Li extraction through three pathways: (i) soluble Li-rich phase dissolution, (ii) Li+/H+ exchange, and (iii) acidic dissolution of lattice Li. The leaching products inherit structurally disordered Li-deficient TMO bulk phases with oxygen vacancies and OH-enriched surfaces, resulting in high OER activity (η10 = 322 mV) surpassing that of conventional TM-based catalysts. This work provides fundamental insights into cationic disordering mechanisms with the practical development of green battery recycling technologies, establishing an energy-efficient closed-loop route from waste electrodes to sustainable energy conversion systems. Notably, this strategy results in superior Li extraction efficiency and enhanced catalytic activity in high-Ni cathode systems. Owing to their electrochemical instability, high-Ni cathode materials develop complex failure structures that are difficult to regenerate directly. In contrast, the MC-based “divide-and-conquer” recycling approach, which prioritizes Li extraction while simultaneously constructing catalytically active structures, offers greater practical advantages and application potential. Future research may further focus on integrating industrial waste gas containing low concentrations of CO2 into the process to enhance both the environmental benefits and the economic feasibility of this technology. This upcycling paradigm holds great promise for supporting the new energy industry in achieving closed-loop resource management and carbon reduction goals.
Methods
Materials
The cathode materials, including LiNi0.33Co0.33Mn0.33O2 (NCM111), LiNi0.6Co0.2Mn0.2O2 (NCM622), LiNi0.8Co0.1Mn0.1O2 (NCM811), LiCoO2 (LCO), LiMn2O4 (LMO) and LiFePO4 (LFP) were purchased from MTI Corporation. The spent cathode materials were provided by Huayou Company, and their compositions are shown in Supplementary Table 1 as determined by Inductively Coupled Plasma-Optical Emission Spectrometry (ICP-OES). Sulfuric acid (H2SO4, analytical pure) was purchased from Beijing Chemical Plant. CO2 (≥99.99%) used in this study was provided from a high-purity gas cylinder sourced from Beijing Huan Yu Jing Hui Jing Cheng Gas Technology Co., Ltd. Potassium hydroxide (KOH, analytical pure) was purchased from Shanghai Hushi Chemical Reagent Co., Ltd. The aqua regia solution was prepared by mixing concentrated nitric acid (HNO3, analytical pure) and concentrated hydrochloric acid (HCl, analytical pure) in a volume ratio of 1:3. HNO3 and HCl were purchased from Beijing Chemical Plant. The standard solution of Li. Ni, Co, Mn, Al, Fe, Cu, Na, K, Ca, P, Zn and Mg used for ICP were purchased from Guobiao (Beijing) Testing & Certification Co., Ltd. Nafion (~5%) was purchased from Shanghai Macklin Biochemical Co., Ltd.
Lithium extraction process
The recycling of Li is divided into two steps, which are the mechanochemical (MC) process and the leaching process. The MC is conducted in the ball mill (Pulverisette 7, FRITSCH, Germany). If there is no additional mark, the rotation conditions are 600 rpm, rotation 20 min and pause 10 min, and the ball-to-powder mass ratio is 20:1. The leaching process is allowed to occur in a hydrothermal reaction vessel. If there is no additional mark, the solid-liquid ratio is 20 g L–1, the leaching temperature is 25 °C, the leaching pressure is 0.5 MPa, and the leaching time is 1 h. After leaching, the final lixivium was separated from the leaching product (MA-LTMO-CO2L) through filtration. The leachate was purified by modulating pH with 1.0 M LiOH solution and then prepared for Li2CO3 product through thermal decomposition, and the decomposition conditions are temperature of 95 °C, time of 1.5 h, and stirring speed of 300 rpm.
To choose an appropriate recycling route, the leaching efficiency (Ri) was determined as shown in Eq. 6, and the selectivity (Si) was calculated according to Eq. 7, where miL represents the mass of the element “i” in the lixivium; miR represents the mass of the element “i” in the leaching product; “i” is Li, Co, Ni, or Mn; RAll indicates the sum of leaching efficiency of all the metal elements.
Materials characterization
The compositions of the materials used in this article are analyzed through an indictively coupled plasma optical emission spectrometer (ICP-OES, ICAP 6300Radial, Thermo Scientific). The materials are firstly dissolved by aqua regia solution, and then the contents of metals in the solution and the leachate are quantified by ICP-OES. The structures of the materials or leaching products were characterized by an X-ray diffractometer (XRD, SmartLab 9 kW, Rigaku) with a Cu Kα radiation source. The proportion of the amorphous phase in MA-LTMO was determined by the XRD internal standard method. The Highscore5.3 software was used to fit the XRD patterns, and the Rietveld quantitative analysis without standards was employed to calculate the relative contents of LTMO and Si, as present in Supplementary Note 1. The microstructure of MC products and leaching products was characterized using a JEOL JEM-F200 field-emission transmission electron microscope (TEM) equipped with selected-area electron diffraction, with the specimens prepared by depositing an ultrasonically dispersed suspension onto a TEM grid and drying. The Raman spectra were characterized by a Raman spectrometer (inVia, Renishaw). The valence states of the elements were analyzed by X-ray photoelectron spectroscopy (XPS, ESCALAB 250Xi, Thermo Scientific). Depth-profiling was performed using an argon ion (Ar+) etching gun, with 4 etching cycles (120 s per cycle, corresponding to 8 nm per cycle as calibrated on a Ta2O5 standard), to track the variation of the valence states from the surface to the bulk. Synchrotron radiation X-ray absorption spectroscopy (XAS, at the Ni K-edge) was recorded in the transmission mode at the XRD station of the beamline 4B9A of the Beijing Synchrotron Radiation Facility. The specific surface area of the activated product was determined by CO2 adsorption at 0 °C and calculated using the BET model. The elemental distribution was characterized by Time-of-Flight Secondary Ion Mass Spectrometry (TOF-SIMS, TOF.SIMS 5, ION-TOF GmbH), and the sputtering depth is calibrated by the SiO2 sputtering rate. The thermal decomposition behavior of the leaching product was detected by a high-temperature thermogravimetric analyzer (TG-DSC-MS, LABSYS EVOT), with the test run in a nitrogen atmosphere from 30 °C to 800 °C at a heating rate of 10 °C min–1.
Electrocatalytic measurements
All the electrocatalytic measurements were conducted using a CHI760E electrochemical workstation under ambient conditions, which used a three-electrode cell system. The leaching residues obtained from CO2-assisted extraction system (MA-LTMO-CO2Ls) were used as the working electrode, Hg/HgO and Pt served as reference and counter electrode, respectively. The catalyst ink was prepared with 2 mg active materials, 0.5 mg acetylene black, 25 μL 5 wt% Nafion solution, and 375 μL isopropanol/H2O (v:v = 1:1) mixed solvent, followed by a 30 min ultrasonic dispersion process. 23 μL catalyst ink was dropped onto a carbon cloth with the mass loading of 0.38 mg cm–2. The OER was measured in O2-saturated 1.0 M KOH solution at the rotating speed of 600 rpm with the scan range of 10 mV s–1. All potentials were calibrated versus the reversible hydrogen electrode (vs. RHE) using Eq. 8:
All LSV curves were measured at a scan rate of 10 mV s−1, and then the potential was corrected by 90% IR compensation to remove the solution resistance effect. The overpotential (η) was determined by Eq. 9:
The LSV curves were further used to construct the Tafel plots, which plot overpotential (η) as a function of the current density log ( j).
The ECSA was measured by scanning the CV in a non-Faradaic region. Then, the ECSA was calculated using the following Eq. 10:
Where Cdl is the double-layer capacitance derived from the slope of the linear regression (R2 > 0.995) of the current density at the midpoint potential versus the CV scan rate.; Cs is the specific capacitance of a flat surface (40 μF·cm−2).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data supporting the findings of the study are included in the main text, supplementary information and source data files. Source data are provided with this paper.
References
Bai, Y. et al. Energy and environmental aspects in recycling lithium-ion batteries: concept of Battery Identity Global Passport. Mater. Today 41, 304–315 (2020).
Harper, G. et al. Recycling lithium-ion batteries from electric vehicles. Nature 575, 75–86 (2019).
Xu, C. et al. Electric vehicle batteries alone could satisfy short-term grid storage demand by as early as 2030. Nat. Commun. 14, 119 (2023).
Fang, J., Wan, G., Zheng, M., Liu, T. & Lu, J. Recycling of spent lithium-ion batteries in view of lithium, advanced energy. Materials 15, 2501318 (2025).
Armand, M. & Tarascon, J. M. Building better batteries. Nature 451, 652–657 (2008).
Fan, E. et al. Sustainable recycling technology for Li-ion batteries and beyond: challenges and future prospects. Chem. Rev. 120, 7020–7063 (2020).
Saju, D., Ebenezer, J., Chandran, N. & Chandrasekaran, N. Recycling of lithium iron phosphate cathode materials from spent lithium-ion batteries: a mini-review. Ind. Eng. Chem. Res. 62, 11768–11783 (2023).
Zhou, M., Li, B., Li, J. & Xu, Z. Pyrometallurgical technology in the recycling of a spent lithium ion battery: evolution and the challenge. ACS EST Eng. 1, 1369–1382 (2021).
Choi, J. W., Kim, J., Kim, S. K. & Yun, Y. S. Simple, green organic acid-based hydrometallurgy for waste-to-energy storage devices: recovery of NiMnCoC2O4 as an electrode material for pseudocapacitor from spent LiNiMnCoO2 batteries. J. Hazard. Mater. 424, 127481 (2021).
Li, P. et al. Optimization of synergistic leaching of valuable metals from spent lithium-ion batteries by the sulfuric acid-malonic acid system using response surface methodology. ACS Appl. Mater. Interfaces 14, 11359–11374 (2022).
Wang, S., Zhang, Z., Lu, Z. & Xu, Z. A novel method for screening deep eutectic solvent to recycle the cathode of Li-ion batteries. Green. Chem. 22, 4473–4482 (2020).
Xiao, J., Niu, B., Song, Q., Zhan, L. & Xu, Z. Novel targetedly extracting lithium: an environmental-friendly controlled chlorinating technology and mechanism of spent lithium ion batteries recovery. J. Hazard. Mater. 404, 123947 (2020).
Jing, Q. et al. Direct regeneration of spent LiFePO4 cathode material by a green and efficient one-step hydrothermal method. ACS Sustain. Chem. Eng. 8, 17622–17628 (2020).
Yang, T., Luo, D., Yu, A., & Chen, Z. Enabling future closed-loop recycling of spent lithium-ion batteries: direct cathode regeneration. Adv. Mater. 35, e2203218 (2023).
Nie, H. et al. LiCoO2: recycling from spent batteries and regeneration with solid-state synthesis. Green. Chem. 17, 1276–1280 (2015).
Chen, S. et al. External Li supply reshapes Li deficiency and the lifetime limit of batteries. Nature 638, 676–683 (2025).
Ogihara, N. & Okayama, S. et al. Direct capacity regeneration for spent Li-ion batteries. Joule 8, 1364–1379 (2024).
Jia, K. et al. Degradation mechanisms of electrodes promote direct regeneration of spent Li-ion batteries: a review. Adv. Mater. 36, 2313273 (2024).
Zheng, H. et al. Selective extraction of lithium from spent lithium batteries by functional ionic liquid. ACS Sustain. Chem. Eng. 9, 7022–7029 (2021).
Lin, T. et al. A high-efficiency and low-carbon strategy for selective lithium recovery from spent lithium-ion batteries: combining mechanochemical activation with biomass reduction roasting. Sep. Purif. Technol. 338, 126458 (2024).
Yuan, X. et al. Magnetization roasting combined with multi-stage extraction for selective recovery of lithium from spent lithium-ion batteries. Sep.Purif. Technol. 338, 126349 (2024).
Zhu, X. H. et al. Recycling valuable metals from spent lithium-ion batteries using carbothermal shock method. Angew. Chem. Int. Ed. 62, e202300074 (2023).
Wang, Y., Zheng, X., Lv, W., Li, L., & Sun, Z. Towards a low-carbon process for lithium recovery from spent lithium-ion batteries by a carbon conversion control strategy of carbothermic reduction. Green Chem. 24, 9734–9744 (2022).
Lin, J. et al. Conversion mechanisms of selective extraction of lithium from spent lithium-ion batteries by sulfation roasting. ACS Appl. Mater. Interfaces 12, 18482–18489 (2020).
Lin, J. et al. Environmentally benign process for selective recovery of valuable metals from spent lithium-ion batteries by using conventional sulfation roasting. Green. Chem. 21, 5904–5913 (2019).
Chang, D. et al. Efficient separation and stepwise recovery of lithium and transition metals from spent ternary lithium-ion battery powders. Chem. Eng. J. 522, 167321 (2025).
Yu, H. et al. Toward Joule heating recycling of spent lithium-ion batteries: a rising direct regeneration method. J. Energy Chem. 105, 501–513 (2025).
Gou, C. et al. Fully upcycling spent ternary cathodes by simultaneously extracting lithium and constructing high-performance oxygen evolution catalysts. Resour. Conserv. Recycl. 222, 108466 (2025).
Wang, W. et al. Electrochemical lithium recycling from spent batteries with electricity generation, Nature. Sustainability 8, 287–296 (2025).
Liu, S. et al. Paving the way for electrochemical recycling of spent lithium-ion batteries: targeting the direct regeneration of de-lithiated materials. Chem. Eng. J. 479, 147607 (2024).
Yang, L. et al. Direct electrochemical leaching method for high-purity lithium recovery from spent lithium batteries. Environ. Sci. Technol. 57, 4591–4597 (2023).
Ardila-Fierro, K. J., & Hernández, J. G. Intermediates in Mechanochemical Reactions. Angew. Chem. Int. Ed. 63, e202317638 (2024).
Wang, M. et al. Converting spent lithium cobalt oxide battery cathode materials into high-value products via a mechanochemical extraction and thermal reduction route. J. Hazard. Mater. 413, 125222 (2021).
Wang, M., Tan, Q., Liu, L., & Li, J. Selective regeneration of lithium from spent lithium-ion batteries using ionic substitution stimulated by mechanochemistry. J. Clean. Prod. 279, 123612 (2021).
Cai, Z. et al. In situ formed partially disordered phases as earth-abundant Mn-rich cathode materials, Nature. Energy 9, 27–36 (2023).
Sharma, S. M. & Sikka, S. K. Pressure-induced amorphization of materials. Prog. Mater. Sci. 40, 1–77 (1996).
Zhang, H. et al. Rock-salt growth-induced (003) cracking in a layered positive electrode for Li-ion batteries. ACS Energy Lett. 2, 2607–2615 (2017).
Qiu, L. et al. Deciphering the degradation discrepancy in Ni-rich cathodes with a diverse proportion of [003] crystallographic textures. Carbon Energy 5, e298 (2022).
De La Torre, A. G., Bruque, S. & Aranda, M. A. G. Rietveld quantitative amorphous content analysis. J. Appl. Crystallogr. 34, 196–202 (2001).
Hinuma, Y., Meng, Y. S., Kang, K. & Ceder, G. Phase transitions in the LiNi0.5Mn0.5O2 system with temperature. Chem. Mater. 19, 1790–1800 (2007).
Zhao, P., Liu, X., De La Torre, A. G., Lu, L. & Sobolev, K. Assessment of the quantitative accuracy of Rietveld/XRD analysis of crystalline and amorphous phases in fly ash, analytical. Methods 9, 2415–2424 (2017).
Huang, S., Zhang, S., Belytschko, T., Terdalkar, S. S. & Zhu, T. Mechanics of nanocrack: fracture, dislocation emission, and amorphization. J. Mech. Phys. Solids 57, 840–850 (2009).
Li, B. Y., Li, A. C., Zhao, S. & Meyers, M. A. Amorphization by mechanical deformation. Mater. Sci. Eng. R Rep. 149, 100673 (2022).
Huang, K., Marthinsen, K., Zhao, Q. & Logé, R. E. The double-edge effect of second-phase particles on the recrystallization behaviour and associated mechanical properties of metallic materials. Prog. Mater. Sci. 92, 284–359 (2018).
Xu, C. et al. Integrated process of CO2 sequestration and recycling spent LiFePO4 batteries. Energy Storage Mater. 60, 102819 (2023).
Wang, J. & Hu, H. Microbubble-assisted pressure carbonation for preparation of high-purity lithium carbonate. J. Mater. Res. Technol. 9, 9498–9505 (2020).
Wang, M., Tan, Q. & Li, J. Unveiling the role and mechanism of mechanochemical activation on lithium cobalt oxide powders from spent Lithium-ion batteries. Environ. Sci. Technol. 52, 13136–13143 (2018).
Buijs, K. & Schutte, C. J. H. The infra-red spectra and structures of Li2CO3 and anhydrous Na3CO3. Spectrochim. Acta 17, 927–932 (1961).
Hamam, I., Zhang, N., Liu, A., Johnson, M. B., & Dahn, J. R. Study of the reactions between Ni-rich positive electrode materials and aqueous solutions and their relation to the failure of Li-ion cells. J. Electrochem. Soc. 167, 130521 (2020).
Ji, P. et al. Ultra-fast in Situ reconstructed nickel (oxy)hydroxide nanoparticle crosslinked structure for super-efficient alkaline water electrolysis by sacrificing template strategy. Small Struct. 4, 2300013 (2023).
Xiao, M. et al. Surface defects induced by acid etching for promoting Ni-rich cathode regeneration, Energy Storage. Materials 79, 104307 (2025).
Kuznetsov, D. A. et al. Tuning redox transitions via inductive effect in metal oxides and complexes, and implications in oxygen. Electrocatal. Joule 2, 225–244 (2018).
Varga, T. & Wilkinson, A. P. Zirconium coordination change upon the pressure-induced amorphization of cubic ZrW2O8 and ZrMo2O8. Phys. Rev. B 79, 224119 (2009).
Kosova, N. V. et al. Electronic state of cobalt and oxygen ions in stoichiometric and nonstoichiometric Li1+xCoO2 before and after delithiation according to XPS and DRS. J. Power Sources 119-121, 669–673 (2003).
Yang, Q., Yao, Z., Lai, C. & Li, C. Pre-pulverizing Ni-rich layered oxide cathodes via “liquid explosive” infiltration toward highly endurable 4.5 V lithium batteries, Energy Storage. Materials 50, 819–828 (2022).
Wood, K. N. & Teeter, G. XPS on Li-battery-related compounds: analysis of inorganic sei phases and a methodology for charge correction, Acs Applied Energy. Materials 1, 4493–4504 (2018).
Park, N.-Y. et al. Degradation mechanism of Ni-rich cathode materials: focusing on particle interior. ACS Energy Lett. 7, 2362–2369 (2022).
Pan, R., Jo, E., Cui, Z. & Manthiram, A. Degradation pathways of cobalt-free LiNiO2 cathode in lithium batteries. Adv. Funct. Mater. 33, 2211461 (2022).
Flores, E., Novák, P., & Berg, E. J. In situ and operando Raman Spectroscopy of layered transition metal oxides for Li-ion battery cathodes. Front. Energy Res. 6, 82 (2018).
Ghanty, C. et al. Li+-Ion Extraction/Insertion of Ni-Rich Li1+x(NiyCozMnz)wO2(0.005<x<0.03;y:z=8:1,w≈1) Electrodes: In Situ XRD and Raman Spectroscopy Study. ChemElectroChem 2, 1479–1486 (2015).
Wang, Y. et al. Over-stoichiometric metastabilization of cation-disordered rock salts. Adv. Mater. 35 e2306396 (2023).
Jo, S. et al. Solid-state reaction heterogeneity during calcination of lithium-ion battery cathode. Adv. Mater. 35, e2207076 (2022).
Lin, Q. et al. Ni–Li anti-site defect induced intragranular cracking in Ni-rich layer-structured cathode. Nano Energy 76, 105021 (2020).
Zhang, K., & Zou, R. Advanced transition metal-based oer electrocatalysts: current status, opportunities, and challenges. Small 17, e2100129 (2021).
Sun, Y. et al, Navigating surface reconstruction of spinel oxides for electrochemical water oxidation. Nat. Commun. 14, 2467 (2023).
Olowoyo, J. O. & Kriek, R. J. Recent progress on bimetallic-based spinels as electrocatalysts for the oxygen evolution reaction. Small 18, e2203125 (2022).
Chen, Q. et al. Significantly enhanced photocatalytic co2 reduction by surface amorphization of cocatalysts. Small 17, e2102105 (2021).
Kim, T. et al. oxyhydroxide electrocatalyst wet gel for highly efficient water electrolysis in alkaline media. Nano Energy 124, 109428 (2024).
Sun, H. & Song, S. Nickel Hydroxide-Based Electrocatalysts for Promising Electrochemical Oxidation Reactions: Beyond Water Oxidation. Small 20, e2401343 (2024).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 52525408, Z.S.), the Strategic Priority Research Program of Chinese Academy of Sciences (No. XDA0430105, Z.S.), the National Natural Science Foundation of China (No.52404431, W.L.), the Beijing Natural Science Foundation (No. 2244081, W.L.) and the 14th Five-year Informatization Plan of Chinese Academy of Sciences, Construction of Scientific Data Center System (WX145XQ07-12, H.C.). The authors would like to thank Minting Luo and Mingming He from Institutional Center for Shared Technologies and Facilities, Institute of Process Engineering, Chinese Academy of Sciences for the XRD and SEM measurement and analysis. The authors are also grated to the facility support of the 4B9A beamline of the Beijing Synchrotron Radiation Facility (BSRF).
Author information
Authors and Affiliations
Contributions
Y.W., X.Z., W.L., and Z.S. designed the research; Y.W. and W.L. conducted the experiments and analysis; Y.W. visualized the data and drafted the manuscript; L.L., H.C., and Z.S. supervised the research; and Y.W., X.Z., W.L., L.L., H.C., and Z.S. wrote, reviewed and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Huayi Yin and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, Y., Zheng, X., Lv, W. et al. A three-in-one strategy for lithium recovery and upcycling of spent cathode materials. Nat Commun 17, 1153 (2026). https://doi.org/10.1038/s41467-025-67912-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-67912-0