Abstract
For two molecules or atoms to react, they must first move towards each other and then, upon meeting, form new chemical bonds. Ultrafast spectroscopy and diffraction techniques have illuminated the bond-formation step for example by triggering reactions from weakly-bound precursors but not the initial approach, typically attributed to diffusion, although this is often the step that determines the reaction rate. Here, we measure and control the diffusion time for the reaction where a Li⁺ ion forms a complex with a benzene dimer, a textbook cation-π system, inside a liquid helium nanodroplet. Using femtosecond-timed Coulomb explosion, we find that Li⁺, initially at the droplet surface over 30 Å from the dimer, first solvates, then diffuses ballistically at 43 m/s and finally reacts. These results, rationalized by ring-polymer molecular dynamics simulations, pave the way for real-time imaging of stereodynamics in ion-molecule reactions.
Similar content being viewed by others
Introduction
Unimolecular processes such as bond-breaking and isomerization start from the well-defined geometry of a single parent molecule. Therefore, their kinetics and even the changing molecular structure are, in principle, straightforward to explore on the natural atomic time scale using femtosecond (fs) pump-probe methods. This is not the case for a bimolecular reaction between two atomic or molecular reactants A and B, because such a process requires that A and B first meet. In gases and liquids, this happens via diffusion, and since the distance between reactants varies, so does the moment they meet for any defined starting time. Consequently, diffusion prevents the femtosecond time resolution needed to follow the part of the reaction where chemical bonds actually change. In certain cases, this timing problem may be overcome if a weakly-bound, non-reactive A-B complex can be created and if a fs pump pulse can initiate the bimolecular reaction by photoexciting A or B1,2,3. Such a precursor complex eliminates diffusion and essentially turns the bimolecular reaction into a unimolecular process. Examples in gas phase and solvents have been reported, including proton transfer, vibrational dynamics and bond shortening4,5,6,7,8. Another approach, demonstrated for radical-molecule and photo-redox reactions in a solvent, is to increase the reactant concentration so much that the diffusion time becomes almost negligible9,10. It has also been shown that the intrinsic rate constant of e.g. bimolecular electron transfer reactions in solution can be determined when theoretical modelling of diffusion is applied to fs time-resolved experimental signals11.
Under most synthetic or natural conditions, the two reactants are, however, not in a close-proximity, precursor geometry. Rather, they are brought together via diffusion, which for many reaction types determines the overall reaction rate and may involve stereodynamical effects as the reactants approach each other. Insight into the progression of the entire bimolecular reaction therefore requires time-resolved techniques that also capture and possibly control the diffusion stage, a so far unmet challenge. Here, we introduce an experimental method that enables us to prepare two reactants, an ion and a molecule, at a controllable initial distance, large enough that diffusion must occur before they meet and react. The separation of the reactants is obtained using helium nanodroplets doped with an alkali atom, here Li, at its surface and a molecular system, here the benzene dimer Bz2, near its center. A fs pump pulse initiates the reaction at a precise time through selective ionization of the Li atom. Coulomb explosion, triggered by ionizing Bz2 with a delayed fs probe pulse, provides time-resolved information about both the initial solvation of the Li+ ion, its diffusion through the droplet and the final Li+Bz2 complex formation. Ring-polymer molecular dynamics (RPMD) simulations serve to interpret the experimental findings and benchmark the experimentally determined diffusion velocity.
Results
Atomistic modelling of the Li+ + Bz2 → Li+Bz2 reaction in a helium nanodroplet
First, we provide a theoretical reference for the reaction:
obtained through RPMD simulations12,13, details given in Methods and Supplementary Information. We consider a helium droplet containing 3600 atoms corresponding to a radius of 33 Å with Bz2 near the center and a Li atom residing ∼ 2 Å above the surface14. At time t = 0, the Li atom is instantly ionized. The ensuing dynamics, including the local environment of the Li+ ion, its distance to Bz2 and the spatial orientation of Bz2, are determined in real time. Figure 1a, illustrating the droplet and dopants at four selected times, shows that Li+ gradually moves towards the droplet center and eventually reaches Bz2 to form a Li+Bz2 complex. A movie of the RPMD trajectory is available as Supplementary Video 1.
a Illustration of the Li+ ion (blue circle) and Bz2 (white and red shapes), both enlarged for visualization, inside a 3600-atom helium nanodroplet at four selected times after Li+ is created. The droplet is shown as a continuous density medium after convoluting the He positions with a Gaussian broadening of 5 Å. In the lower two panels, the density outside the droplet region indicates helium evaporating14,18. Time dependence of b Li+−Bz2 (center-of-mass) distance, c distance between the benzene molecules and d coordination number of the Li+ ion. The grey shaded band marks the interval where Li+ binds to Bz2. a–d represent results from the same single RPMD trajectory. e Same as b but averaged over the trajectories from 10 independent simulations (due to computational constraints these trajectories were limited to t = 40 ps). The blue dash-dotted line, with a slope vdiffusion, is the best linear fit to the diffusion region, here 10 Å < r(Li+–Bz2) < 40 Å. The red dashed curve shows the distance of Li+ to the droplet center from the average of 10 independent simulations without Bz2. Source data are provided as a Source Data file.
The full curve in Fig. 1b, depicting the Li+–Bz2 distance, r(Li+–Bz2), for a single trajectory, as a function of time, shows that Li+ initially accelerates into the droplet, then decelerates and at t ∼ 10 ps settles at a constant velocity. These early dynamics, caused by the polarizability interaction between Li+ and the He atoms, see ref. 13. and Supplementary Information, are independent of the presence of Bz2, as seen by comparing results without Bz2 in Fig. 1e to those with Bz2. The latter represents the average over 10 trajectories corresponding to different initial orientations of Bz2 i.e., a situation more accurately reflecting the experimental conditions than the single trajectory in Fig. 1b. At longer times, Fig. 1b shows that the Li+ velocity remains constant until t ∼ 45 ps, where the ion accelerates and r(Li+–Bz2) drops from 13 Å to 3.5 Å at t = 52 ps. Thereafter, r(Li+–Bz2) does not change. We ascribe this final step, only observed when Bz2 is present, to the short-range Li+–Bz2 attraction and the ensuing formation of the Li+Bz2 complex, where Li+ binds to the outside of the dimer (Fig. 1a).
Figure 1c shows that the distance between the two Bz molecules, r(Bz–Bz), remains constant at ∼ 3.9 Å, the calculated equilibrium distance of Bz2, until t = 51 ps, where it drops to 3.2 Å within a few ps. This drop, highlighted by the grey shaded band, coincides with when Li+ makes the final approach to Bz2 and is another manifestation of the Li+Bz2 complex formation. Finally, Fig. 1d shows the coordination number for Li+, Nc(Li+), which is a measure of the number of He atoms closely bound to the ion. During the first ∼ 4 ps, Nc increases from 0 to 6, an effect ascribed to the solvation of Li+13,14. Thereafter, Nc increases slowly to about 8 and then, from t = 51 to 52 ps, it drops sharply to below 2 and subsequently recovers at 3. We interpret the sharp drop, occurring simultaneously with r(Bz–Bz) and r(Li+–Bz2) reaching their final values, as part of the solvation shell of Li+ being broken during its bonding to Bz2. The asymptotic value of 3 for Nc is the number of He atoms attached to the side of Li+ facing away from Bz2.
Initial solvation of the Li+ ion
The starting point for the experiments is also helium nanodroplets, each containing one Li atom, at the surface, and one benzene dimer, expected to be located near the droplet center. For experimental reasons, many of the droplets will, however, contain a Li atom and only a single benzene (Bz) molecule (see Methods). First, the doped droplets are irradiated by a 50-fs pump laser pulse that multiphoton ionizes Li and leaves Bz2 (or Bz) intact. This prompt creation of a Li+ ion sets a precise starting time for the reaction, Eq. (1), at a well-defined initial separation of the reactants determined by the radius of the droplets.
Next, the 60-fs probe pulse, sent at time t, ionizes Bz2 (or Bz), which leads to ejection of different ion species, notably Li+, Bz+ and Li+Bz, from the droplet. We detect the time-dependent velocity-resolved yields of these ions to obtain information about different stages of the reaction path. First, we focus on the Li+ ion yield. Figure 2a-f shows the covariance maps15,16 between the distributions of v(Li+) and v(Bz+) at six selected times, v(Li+) and v(Bz+) denoting the velocities of Li+ and Bz+, respectively.
a–f Covariance maps of the velocity distributions of Li+ and Bz+ ions at six selected times. g Filled circles: time dependence of the coincidence-filtered Li+ ion signal, i.e., the Li+ ions that, within the same laser shot, are detected along with a Bz+ ion having the opposite momentum. The full line connects the experimental points and serves to guide the eye. Note that the time scale is logarithmic. Details of the coincidence filtering are given in Methods. Source data are provided as a Source Data file.
The pronounced covariance signal centred at (3.0 km/s, 0.2 km/s), observed at t = 0.2 ps, shows that a Li+ ion with v around 3.0 km/s, corresponding to a kinetic energy, Ekin, of 0.33 eV, is correlated with a Bz+ ion with v around 0.2 km/s (Ekin ∼ 0.02 eV). This correlated ion-pair stems from the Coulomb repulsion between a Li+ and a Bz+ ion originating from the same droplet with Bz+ produced by dissociative ionization of Bz2 or by ionization of Bz. In either case, if Bz2 or Bz is located in the center of the droplets, with an expected average radius 〈R〉 of 35 Å, there will be a Coulomb energy of 0.37 eV between Li+ and Bz+, the moment Bz+ is created. Their repulsion causes the ejection of both ions from the droplet and momentum conservation gives the Li+ ion a final kinetic energy of 0.32 eV. The match with the measured Ekin corroborates that the initial Li+–Bz2-separation is to a good approximation given by the droplet radius. For details, see Supplementary Information.
At t = 0.6 ps, Fig. 2b, the covariance signal at (3.0 km/s, 0.2 km/s) is weaker and at t = 3.0 ps, Fig. 2c, it has essentially disappeared. Figure 2g shows the Li+ signal, coincidence-filtered with Bz+ ions17, as a function of time. The signal drops exponentially to zero within the first three ps, which we ascribe to Li+ solvating in the droplet, similar to recent observations of solvation of a single Na+ ion in a He droplet18,19. Details of the solvation dynamics of the Li+ ion are reported elsewhere20.
Diffusion and bond formation
Our main interest here is what happens at longer times. Figure 2g shows that after the initial decrease, the Li+ signal recovers and rises gradually until it reaches a plateau around 200 ps. We believe the reappearance of the Li+ signal occurs when the solvated Li+ ion moves sufficiently close to Bz2 (or Bz) that the probe-pulse-induced Coulomb repulsion strips off all He atoms in the solvation shell. The significantly higher v(Bz+) detected at longer times, Fig. 2d-f, compared to v(Bz+) for t < 1 ps, Fig. 2a-b, corroborates that Li+ has moved closer to Bz2 (or Bz). Thus, the results in Fig. 2 qualitatively show that Li+ gradually approaches and probably reacts with Bz2 (or Bz), but it is difficult to quantitatively determine when the Li+Bz2 complex forms.
Therefore, we turn to another experimental observable, namely Li+Bz ions detected in coincidence with Bz+ ions. This observable, termed Co(Li+Bz, Bz+), has two decisive advantages. Firstly, unlike the signals shown in Fig. 2, it only originates from droplets doped with a Li atom and a Bz dimer. Secondly, Li+Bz is only formed when Li+ has moved close to Bz2. Although this appears intuitively clear, we modelled the probe process and showed that formation of a Li+Bz–Bz+ ion pair only happens when r(Li+–Bz2) < 8 Å (Supplementary Information). According to Fig. 1b, r(Li+–Bz2) decreases from 8 Å to 3.5 Å, the value of the final Li+Bz2 complex, within ∼ 1 ps. Thus, we have an experimental observable that is nonzero only when Li+Bz2 has formed–or in the last ps before this happens.
Figure 3a displays the velocity distribution of the Li+Bz ions detected in coincidence with a Bz+ ion as a function of time. The figure shows that these ions first appear after about 5 ps and the distribution centres around 0.6 km/s, which we interpret as the final mean velocity of the Li+Bz ions after Coulomb repulsion from a Bz+ ion. Next, the time dependence of Co(Li+Bz, Bz+) is obtained by integrating the velocity distributions in Fig. 3a at each time step (Methods). The result, black circles in Fig. 3b, shows that the yield of Li+Bz2 complexes gradually increases from t ∼ 5 ps until t ∼ 100 ps where the signal flattens. This curve represents our measurement of the time-dependent yield of the reaction in Eq. (1).
a Time dependence of the velocity distribution of Li+Bz ions coincident with a Bz+ partner ion. The average droplet size 〈R〉 is 35 Å. b–e Black dots: Co(Li+Bz, Bz+) as a function of time for different 〈R〉 indicated on each panel. Blue curves: Best fit of the cumulative log-normal distribution function CDFLN to the data points. The dashed line shows the mean value, 〈ttotal〉, of the log-normal distribution of the total reaction time, ttotal. The uncertainty is calculated by propagating the 1σ uncertainties of µt and σt obtained by the fitting procedure of CDFLN. f Points: 〈ttotal〉 measured at 15 different 〈R〉. Dashed line: Best linear fit to the data points included. Methods includes an explanation of how 〈R〉 is estimated. Source data are provided as a Source Data file.
To model and understand the experimental data, we note that according to the simulations, the total time, ttotal, to form Li+Bz2 from the Li+ and Bz2 reactants at their initial separated positions, i.e., the time we measure, has three contributions: 1) The solvation time, tsolvation, of Li+ including the initial acceleration and deceleration, i.e., the first ∼ 10 ps in Fig. 1b; 2) The diffusion time, tdiffusion, where Li+ moves with constant velocity, vdiffusion, i.e., ∼ 10 − 45 ps in Fig. 1b; 3) The bond formation time, tbond, where Li+ makes the final acceleration towards Bz2 and forms Li+Bz2, i.e., ∼ 45 − 52 ps in Fig. 1b. Thus,
Equation (2) is based on the simulation for a droplet with R = 33 Å, whereas the experimental results concern droplets with 33 Å ≤ 〈R〉 ≤ 49 Å. Currently, it is not computationally feasible to simulate larger droplets, but we expect that for a limited increase of R, tsolvation, tbond, and vdiffusion remain essentially unchanged. Therefore, we rewrite Eq. (2) as
and experimentally test its validity as described below.
The droplets are formed with a size distribution, well described by a log-normal distribution21. According to Eq. (3), we therefore expect a log-normal distribution of ttotal. However, Co(Li+Bz, Bz+) represents integration over ttotal, because the smallest droplets give a contribution at the earliest times and then the contributions from larger droplets gradually add at longer times. Thus, Co(Li+Bz, Bz+) should be given as the cumulative distribution function of ttotal, denoted CDFLN (see Methods). The best fit of CDFLN to the data points, blue curve in Fig. 3b, closely matches the experimental results. We repeated the measurements for 14 other droplet size distributions and the results for three of them are displayed in Fig. 3b–e. Again, CDFLN provides excellent fits to the data points, and it is seen that the reaction time increases as the droplet size increases consistent with a diffusion-limited reaction.
From each fit, we obtain the mean value of the log-normal distribution of ttotal. This value, denoted 〈ttotal〉, represents the average reaction time at the particular droplet size distribution. Figure 3f displays 〈ttotal〉 as a function of 〈R〉. It appears that for 33 Å ≤ 〈R〉 ≤ 42 Å, 〈ttotal〉 scales linearly with 〈R〉. The linear dependence is quantified by the dashed, blue line representing the best linear fit to the points in the 33 − 42 Å range. The linearity validates Eq. (3) and allows a determination of the average velocity, \(\left\langle {v}_{{\rm{diffusion}}}^{\exp }\right\rangle\), of Li+ in the diffusion region, as the inverse of the slope of the fitted line. We find \(\left\langle {v}_{{\rm{diffusion}}}^{\exp }\right\rangle\) = 43 ± 5 m/s (see Supplementary Information), i.e., larger than the simulated vdiffusion = 14 m/s, Fig. 1e.
Our simulations show that the linear motion results from the combined effect of Li+ being accelerated by the attractive interaction with the He atoms and Bz2 and decelerated by friction from the droplet (Supplementary Information). The simulations do, however, not account for the superfluidity that characterizes helium nanodroplets22,23 and therefore vdiffusion is expected to underestimate the real diffusion velocity. The found \(\left\langle {v}_{{\rm{diffusion}}}^{\exp }\right\rangle\) is close to the critical Landau velocity measured to be 56 m/s for Ag atoms in helium droplets23, indicating that the onset of friction on Li+ occurs when its speed is slightly below the Landau velocity. Similar to the findings in Ref.23, we believe this is caused by heating of the droplet–in our case due to the ion solvation process. Finally, Fig. 3f shows that for the larger droplets, the reaction time gradually increases compared to the extrapolated linear behaviour. This could be caused by increased delocalization of Bz2 away from the droplet center24, meaning Li+ not always moves straight towards Bz2, i.e., it may have to travel farther than the droplet radius to encounter the dimer. This effect is expected to be more pronounced for larger droplets in agreement with the observed behaviour of 〈ttotal〉. The excellent agreement between the fits of CDFLN and the experimental points suggests that it is unlikely that the delocalization becomes so pronounced that the Li+ ion does not meet Bz2 in the center – such an event would give a deviation from the log-normal distribution.
Discussion
Characterization of diffusion in fs time-resolved experiments of bimolecular reactions in classical liquids was addressed previously. For instance, transient absorption spectroscopy examined reactions between a chlorine radical and an alkane solvated in dichloromethane25. Smoluchowski diffusion modelling showed the reactions were diffusion-limited with rate constants of about 1×1010 M-1s-1. In comparison, for the diffusion-limited reaction explored here, we estimate the reaction constant based on the reaction time for a given droplet size and find 5×1012 M-1s-1 (Supplementary Information). Thus, diffusion of the Li+ ion in the He droplet is two orders of magnitude faster. This difference reflects two qualitatively different types of diffusion. In classical liquids, diffusion arises from Brownian motion, with diffusion time proportional to the mean square displacement26. In contrast, Li+ moves with constant velocity through the droplet, and diffusion time depends linearly on distance, see Eq. (3). The linear motion is an example of anomalous diffusion termed ballistic diffusion27,28,29, a phenomenon also encountered under widely different conditions, for instance in the transport of molecules in DNA model condensates30.
Currently, the time resolution of our technique is insufficient to capture details of the part of the reaction where new molecular bonds form. To explore this, it may be favourable to start from close-proximity precursor complexes. Helium droplets offer opportunities in this regard by enabling formation of diverse molecular and atomic complexes31,32, going beyond what can be accomplished using gas-phase samples or molecules in classical solvents. Starting from a close-proximity precursor misses, however, the stereodynamics of reactions, i.e., as two reactants approach, their mutual interaction may steer their relative orientation into a particular reaction geometry33,34,35,36. To explore this fundamental aspect of bimolecular reactions, the two reactants should start far apart, as ensured by the present method. In this connection, we point out that stereodynamics of bimolecular reactions have been explored extensively using crossed molecular beams37,38. In those works, typically addressing inelastic or reactive scattering between an atom and a molecule, the molecule was aligned or oriented along a laboratory-fixed axis by a static electric field sometimes preceded by quantum-state-selection or photoselection. Varying the molecular orientation or alignment with respect to the direction of the incoming atom and recording the total or differential cross section have provided deep insight into how steric effects influence chemical reactions.
Our method can address stereodynamics in two fundamentally different ways. Firstly, the molecule is not aligned or oriented by an external field defined in the laboratory frame. Instead, it is the approaching ion that induces alignment or orientation of the molecule through the interaction between its electrical properties and the electric field from the cation, see Supplementary Video 2. Secondly, our experimental method is time-resolved. In the Li+–Bz2 reaction, Bz2 gradually aligns the normal of its plane towards the trajectory of the incoming Li+ ion, (Supplementary Information). Since it has already been demonstrated that laser-induced Coulomb explosion can determine structure and alignment of molecules and molecular complexes inside He droplets39,40, we believe that our technique can be extended to provide real-time imaging of stereodynamics in ion-molecule reactions41,42,43,44.
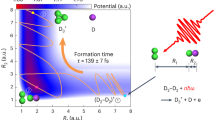
In conclusion, we have introduced an experimental method to explore, in real time, the bimolecular reaction where a Li+ ion diffuses towards a benzene dimer and forms a Li+Bz2 complex inside a liquid helium nanodroplet. In combination with atomistic RPMD simulations, the experimental results allow us to draw a picture of the progression of the entire reaction summarized in Fig. 4. The Li+Bz2 complex is an example of an ion-molecule system held together by noncovalent bonding referred to as cation-π interaction45. This interaction, which has not been explored previously by fs time-resolved techniques, plays an important role for e.g., molecular recognition in biological systems and in organic synthesis46. One opportunity opened by our work is to measure the stereodynamics of the approaching reactants using timed Coulomb explosion imaging. This should be possible for a variety of molecules reacting with atomic or molecular ions of alkali and alkaline earth atoms. Another opportunity is to explore interactions between alkali atoms or dimers at the droplet surface and fullerenes in the interior. The current method may provide insight into electron transfer in such systems – recently explored by electron ionization mass spectrometry47, helium density functional theory48 and path-integral molecular dynamics simulations49,50.
Upon sudden formation, by the pump-pulse-induced 4-photon ionization of a Li atom at the droplet surface, the Li+ ion solvates, then diffuses ballistically with a velocity of 43 m/s and finally binds to Bz2 near the droplet center to form Li+Bz2. In addition to the timescales of solvation, diffusion and bond-formation, the figure illustrates that the energy is lowered by ∼ 0.5 eV as Li+ solvates14 and by another ∼ 1.7 eV when it binds to Bz2 (Supplementary Information). The latter number expresses that the noncovalent cation- π interaction is almost as strong as a typical covalent bond. Energy levels and atomic sizes are not to scale.
Methods
Experimental details
An illustration of the experimental setup is shown in Supplementary Information. More details can be found in Ref. 19. A beam of liquid helium droplets is formed by subcritical expansion of 99.9999% purity 4He gas into vacuum through a 5 µm diameter orifice. The backing pressure of the He gas was 50 bar in all experiments, while the temperature of the expansion nozzle was varied between 13.5 K–20.5 K. The resulting droplet distributions had an average number of helium atoms between 3200 and 14600. The formed helium droplet beam passes through two doping cells. The first contains a vapor of benzene molecules, let into the cell through a leak valve. The second contains a gas of Li atoms obtained by heating a sample of solid Li in an oven. The set-point of the PID controller was set to 390 °C. The opening of the leak valve and the temperature of the Li metal were selected to maximize the likelihood of doubly doping with benzene and singly doping with Li. The doping process is Poissonian22, so other combinations of dopants are unavoidable. The case where a droplet is doped with one Bz molecule and one Li atom is addressed in the main text, and while other systems like LiBz3, Li2Bz, Li2Bz2 etc. are possibly formed, they are present to a much lesser extent and do not contribute noticeably to the signal.
The doped droplet beam enters the target vacuum chamber, that contains a velocity map imaging (VMI) spectrometer51,52. The VMI spectrometer is a three-plate setup, where the repeller (6000 V), extractor (3790 V) and ground (0 V) plate voltages are chosen to achieve velocity focusing. The droplets are irradiated by two pulsed, linearly polarized laser beams in the center of the VMI spectrometer. The two beams are spatially overlapped, and their linear polarization is in the plane of the detector at the end of the VMI spectrometer. The laser pulses are produced by a Coherent Astrella HE laser system (wavelength (nominal) λ0 = 800 nm, temporal FWHM τ = 35 fs, pulse energy Ep = 2 mJ, repetition rate frep = 5 kHz). The focused pump pulses used to ionize Li (τ = 50 fs, λ0 = 800 nm, Ep = 16 µJ, beam waist w0 = 41 µm) have a peak intensity of I0 = 1.1×1013 W/cm2, while the probe pulses used to ionize Bz (τ = 50 fs, λ0 = 400 nm, Ep = 18 µJ, w0 = 28 µm) have a peak intensity of I0 = 2.7×1013 W/cm2. The laser parameters were chosen to selectively ionize Li with the pump pulse and efficiently ionize Bz with the probe pulse. The probe pulse was delayed by time t using a linear delay stage.
The ions produced are projected by the VMI spectrometer onto a two-dimensional imaging detector. The detector consists of two micro-channel plates (MCP) and a phosphor screen (P47). The first MCP is a funnel-type plate, which increases the detection efficiency53. The front of the MCP stack was grounded, while the back was set to 2300 V. The middle of the stack was kept at 1150 V using a shim-ring and the phosphor screen was biased to 4900 V. The light produced by the phosphor screen was imaged by a TPX3Cam detector54,55,56. The TPX3Cam is event-based and records both the position and time-of-arrival of each lit-up pixel independently, which allows us to measure VMI images of multiple, different ions. The pixel data stream is time-walk corrected57,58, clustered and centroided using the DBSCAN algorithm, and time-walk corrected again to retrieve the time-of-flight (ToF) from the time-of-arrival. The x- and y-hit coordinates are calibrated to give the projected velocities, vx and vy, using velocities of Li+ ions coming from the Coulomb explosion of the 3Σu+ state of the Li2 dimer59. Mass-over-charge values (m/q) were obtained by calibrating the ToF-spectrum using known background ions (H2O+ and effusive Bz+). The final data stream then consists of tuples of values pertaining to each ion, including the laser shot number, (ls, m/q, vx, vy). The experiment was recorded for 15 different nozzle temperatures with a set of time delays between 1 ps and 1000 ps, where a trace was repeated between five and 20 times to achieve a reasonable signal-to-noise ratio.
Data analysis for extraction of Co(Li+Bz, Bz+)
The main observable in the experiment is the yield of Li+Bz ions detected in coincidence with Bz+ ions, denoted Co(Li+Bz, Bz+). The experimental data acquired in the experiment consists of tuples of values from the TPX3Cam containing the shot number, mass-over-charge, velocity x-component and velocity y-component, (ls, m/q, vx, vy). The data analysis employed to extract Co(Li+Bz, Bz+) from these tuples is shown in Supplementary Fig. 10, for a dataset recorded with \(\left\langle R\right\rangle=35\) Å. First, Li+Bz ions and Bz+ ions are assigned in the m/q-spectrum, Supplementary Fig. 10a. VMI images are then constructed by calculating the 2D (vx, vy) histogram of the selected ion hits across all laser shots. A VMI image of Li+Bz is shown in Supplementary Fig. 10b for a time-resolved experiment, where images from each time delay are summed. The image shows a strong circular feature with a radius of around ~ 0.4 km/s. There is also a dark line in the center of the image, which is attributed to a burn-in on the MCP detector. The VMI image shows that Li+Bz ions indeed are formed in the experiment and the majority of these have very low kinetic energy.
To gain further information about the time-resolved dynamics, we employ covariance map analysis15,16,60,61 of the velocities and recoil directions of Li+Bz and Bz+ ions. Covariance maps between the radial-velocity distributions, v(Li+Bz) and v(Bz+), of the corresponding VMI images are shown in Supplementary Fig. 10c, similarly summed across a time-resolved experiment. The covariance map shows that Li+Bz ions can be observed in coincidence with Bz+ ions and that these ions have a well-defined kinetic energy that is larger than the dominating background signal observed in the VMI image in Supplementary Fig. 10b. To further corroborate this analysis, we also consider the covariance map between the angular distributions, \({\theta }_{{{\rm{L}}}{{{\rm{i}}}}^{+}{{\rm{Bz}}}}\) and \({\theta }_{{{\rm{Bz}}}}\), shown in Supplementary Fig. 10d. The covariance map shows a 180° angular relation between the two ions, which is characteristic of a Coulomb explosion between two charges62. The angular covariance map is very wide compared to gas-phase experiments62, which is attributed to non-axial recoil effects coming from collisions with He atoms inside the droplet as the ions leave. The joint analysis of the velocities and recoil directions in Supplementary Fig. 10c-d shows that Li+Bz ions with a relation to Bz+ ions representative of a Coulomb explosion process can be observed and that these ions have a kinetic energy that is larger than what can be seen in the VMI image of Li+Bz, see Supplementary Fig. 10b. The covariance maps are then used to coincidence filter the Li+Bz ions17. The filtering parameters used are:
The tolerances are chosen to fully cover the covariance maps shown in Supplementary Fig. 10c-d. The relation between the two velocities, v(Li+Bz) and v(Bz+), is not defined by the mass ratio between the species but is found empirically. This discrepancy is attributed to the ions having different interactions with the helium droplet as they leave the droplet. The filtered VMI image is shown in Supplementary Fig. 10e for Li+Bz. There is still a significant signal in the center of the image, but a second channel now appears in the image with a radius of ~ 0.8 km/s. To analyze this new feature, we extract the full velocity distribution, P(vr). The velocities reported so far have been the projected velocities in the plane of the detector, vx and vy, but the full velocity distribution can be reconstructed by Abel inverting the image, here using the MEVIR algorithm63. The normalized P(vr) distributions for the raw and filtered VMI images are shown in Supplementary Fig. 10f. By applying coincidence filtering, a broad peak in the velocity distribution is revealed, which is suppressed by background signal in the raw image. This peak is attributed to the Coulomb explosion between Li+Bz and Bz+ ions inside the droplet. Co(Li+Bz, Bz+) is then extracted by radially integrating the velocity distributions. In the experiment, this analysis is performed for each time delay. The resulting Co(Li+Bz, Bz+) trace for \(\left\langle R\right\rangle=35\) Å is shown in Supplementary Fig. 10g and is identical to Fig. 3b. The panel also includes a fit of the cumulative-distribution function of a log-normal distribution, CDFLN. The cumulative-distribution function is defined as:
where A, c, \({\mu }_{t}\) and \({\sigma }_{t}\) are fitted to Co(Li+Bz, Bz+). A and c are parameters used to scale and offset the distribution. 〈ttotal〉 can then be calculated as the mean of the log-normal distribution:
Theoretical details of ring-polymer molecular dynamics
Simulations of the ionization, diffusion and reaction processes were conducted at the atomistic level of detail, using the same approach previously implemented for the alkali-xenon pair of dopants inside helium nanodroplets19. The system is prepared in the neutral state with the alkali atom sitting outside the droplet and the molecular dopant Bz2 sitting at the center of the droplet. Thermostatted path-integral molecular dynamics (PIMD) were carried out before sudden ionization of the lithium atom takes place, at a temperature of 2 K and using time a step of 0.5 fs and a Trotter discretization number of 32, which is sufficient to ensure a liquid droplet. After 10 ps of equilibration, ring-polymer molecular dynamics (RPMD) simulations20 were subsequently integrated up to 50 ps, using the same time step of 0.5 fs but without thermostatting, assuming the potential energy surface now corresponds to Li+. The details of the interaction potentials for the neutral and cationic systems are described in the Supplementary Information. Different initial conditions for the trajectories were generated by supplementing the potential used in the equilibrated PIMD simulations with a simple biasing potential, detailed in the Supplementary Information. The dynamics of the Li+-Bz2 complex formation process was characterized using several descriptors that were monitored as a function of time, namely the distance r(Li+−Bz2) between the lithium cation and the Bz dimer, instantaneously measured at the dimers center of mass, the distance r(Bz−Bz) between the two benzene molecules, instantaneously measured as the minimum distance between all atomic pairs of the two different molecules, and the coordination number Nc(Li+) of helium atoms around Li+, defined by summing a cut-off function over all Li+-He pairs with value 1 for distances below 3.8 Å, 0 for distances above 4.2 Å, and smoothly interpolated between these values using a fifth-order polynomial. These values were chosen to yield a strictly vanishing coordination number for the neutral lithium atom sitting above the droplet. All the structural properties above were further averaged over the individual real-space beads in the path-integral description.
Estimation of mean droplet radius 〈R〉
A crucial parameter used in the analysis to determine the diffusion velocity of Li+ in liquid helium droplets is the mean droplet radius, 〈R〉. Direct measurements of the helium droplet size distributions typically require a dedicated experimental setup21,64 and is therefore not currently possible in our experiment. Experimentally recorded droplet sizes have, however, been reported for conditions similar to ours also using a 5 µm orifice and similar stagnation pressures and temperatures22. These measurements form the basis for estimating the size of the droplets used in our experiment. To this aim, we first consider an undoped droplet size distribution characterized by a log-normal distribution65:
where N is the number of helium atoms and \({\delta }_{{{\rm{N}}}}\) and \({\mu }_{{{\rm{N}}}}\) are parameters that characterize the distribution. For the expansion conditions in our experiment, the two parameters are given as65:
which are uniquely determined by the mean number of helium atoms, \(\left\langle N\right\rangle\). The mean radius, \(\left\langle R\right\rangle\), can then be found assuming that the droplets are hard spheres with uniform density as:
Estimated values for \(\left\langle N\right\rangle\) and \(\left\langle R\right\rangle\), based on previously reported droplet sizes22, are shown in Supplementary Table 2 for the nozzle temperatures used in the experiment. The droplets of interest in our experiment, which give rise to our observable Co(Li+Bz, Bz+), are those that are doubly doped with benzene molecules and singly doped with lithium atoms. To estimate the size distributions of these droplets, we must then also include the doping probability of the molecules and atoms. The doped droplet size distribution is determined as the renormalized product of the two distributions:
where \(P(k;N,{p}_{{{\rm{d}}}})\) is the probability of doping k particles at pressure pd. This probability is described by a Poissonian distribution:
where k is the number of doped particles, pd is the doping pressure, N is the number of He atoms and \({\beta }_{{{\rm{N}}}}\) is a collection of factors:
Here, \({L}_{{{\rm{d}}}}\) is the length of the doping cell, \({\sigma }_{{{\rm{D}}}}(N)\) is the cross-section of the droplets, \({{{\rm{k}}}}_{{{\rm{B}}}}\) is the Boltzmann constant and \({T}_{{{\rm{d}}}}\) is the dopant temperature. The correction factor \({F}_{{{\rm{a}}}0}\) accounts for the movement of the droplets and dopant particles, and includes the most probable speed of the dopants (\(\widehat{{v}_{{{\rm{d}}}}}\)) and the speed of the droplets (\({v}_{{{\rm{D}}}}\)):
with \({v}_{{{\rm{D}}}}=\sqrt{\frac{5{T}_{{{\rm{nozzle}}}}{{{\rm{k}}}}_{{{\rm{B}}}}}{{m}_{{{\rm{He}}}}}}\), \(\widehat{{v}_{{{\rm{d}}}}}=\sqrt{\frac{2{T}_{{{\rm{d}}}}{{{\rm{k}}}}_{{{\rm{B}}}}}{{m}_{{{\rm{d}}}}}}\) and erf(x) the Gaussian error function. The temperatures of the dopants were \({T}_{{{\rm{Bz}}}}=298 \; {{\rm{K}}}\) and \({T}_{{{\rm{Li}}}}=628 \; {{\rm{K}}}\) and the doping cells had lengths of \({L}_{{{\rm{Bz}}}}=7.7\) cm and \({L}_{{{\rm{Li}}}}=1.7\) cm. Based on the experimental data given in reference65, the droplet cross section can be written as:
The last parameters required to estimate the doped droplet distribution are the vapor pressures of the samples. The optimal vapor pressures of benzene and lithium were found in the following way. We performed a pump-probe experiment at t = 500 ps, where Co(Li+Bz, Bz+) was maximized by varying the leak valve opening for \({T}_{{{\rm{nozzle}}}}=\) 17 K, see Supplementary Fig. 11. This leak valve position was then used throughout the experiment for other nozzle temperatures. The maximum of Co(Li+Bz, Bz+) must coincide with the vapor pressure of benzene where the doping of two molecules is most likely. This vapor pressure was then found by maximizing the overlap of the two distributions, \({\int }_{0}^{\infty }P\left(N\right)\cdot P\left(k;N,{p}_{{{\rm{d}}}}\right){dN}\), as a function of \({p}_{{{\rm{d}}}}\). The vapor pressure was found to be \({p}_{{{\rm{Bz}}}}=1.2\cdot {10}^{-5}\) mbar. The vapor pressure of the Li sample was similarly estimated as the pressure which optimizes the likelihood of doping one lithium atom for the 17 K droplet size distribution, \({p}_{{{\rm{Li}}}}=1.99\cdot {10}^{-5}\) mbar, which corresponds to a temperature of \({T}_{{{\rm{Li}}}}=628 \; {{\rm{K}}}\)66\(.\) Note that this is not entirely consistent with the vapor pressure of Li metal at the set temperature of the oven, which indicates that the temperature measured on the outside of the oven is not exactly the same as the temperature of the sample. The final doped droplet distribution was then found by first estimating the undoped droplet distribution, using values in Supplementary Table 2, finding the doped droplet distribution for \(k=2\) with benzene and then finding the doubly doped droplet distribution for \(k=1\) with lithium. The resulting droplet size distribution was then transformed from N to R and the numerical average \(\left\langle R\right\rangle\) was computed. The effect of including the doping probabilities on \(\left\langle R\right\rangle\) is shown in Supplementary Fig. 12 for the experimental conditions along with the undoped distributions.
Data availability
The data generated in this study are provided in the Supplementary Information/Source Data file. Source data are provided with this paper and are available at (https://doi.org/10.6084/m9.figshare.30344179). Source data are provided with this paper.
References
Scherer, N. F., Khundkar, L. R., Bernstein, R. B. & Zewail, A. H. Real-time picosecond clocking of the collision complex in a bimolecular reaction: the birth of OH from H+CO2. J. Chem. Phys. 87, 1451–1453 (1987).
Scherer, N. F., Sipes, C., Bernstein, R. B. & Zewail, A. H. Real-time clocking of bimolecular reactions: Application to H+CO2. J. Chem. Phys. 92, 5239–5259 (1990).
Wittig, C., Sharpe, S. & Beaudet, R. A. Photoinitiated reactions in weakly bonded complexes. Acc. Chem. Res. 21, 341–347 (1988).
Rini, M., Magnes, B.-Z., Pines, E. & Nibbering, E. T. J. Real-time observation of bimodal proton transfer in acid-base pairs in water. Science 301, 349–352 (2003).
Kim, K. H. et al. Direct observation of bond formation in solution with femtosecond X-ray scattering. Nature 518, 385–389 (2015).
Kim, J. G. et al. Mapping the emergence of molecular vibrations mediating bond formation. Nature 582, 520–524 (2020).
Mi, Y. et al. D3+ formation through photoionization of the molecular D2–D2 dimer. Nat. Chem. 15, 1224–1228 (2023).
Zhou, L. et al. Ultrafast formation dynamics of D3+ from the light-driven bimolecular reaction of the D2–D2 dimer. Nat. Chem. 15, 1229–1235 (2023).
Orr-Ewing, A. J. Taking the plunge: chemical reaction dynamics in liquids. Chem. Soc. Rev. 46, 7597–7614 (2017).
Koyama, D., Dale, H. J. A. & Orr-Ewing, A. J. Ultrafast observation of a photoredox reaction mechanism: photoinitiation in organocatalyzed atom-transfer radical polymerization. J. Am. Chem. Soc. 140, 1285–1293 (2018).
Rosspeintner, A., Angulo, G. & Vauthey, E. Bimolecular photoinduced electron transfer beyond the diffusion limit: the Rehm–Weller experiment revisited with femtosecond time resolution. J. Am. Chem. Soc. 136, 2026–2032 (2014).
Craig, I. R. & Manolopoulos, D. E. Quantum statistics and classical mechanics: real time correlation functions from ring polymer molecular dynamics. J. Chem. Phys. 121, 3368–3373 (2004).
Calvo, F. Concurrent processes in the time-resolved solvation and Coulomb ejection of sodium ions in helium nanodroplets. J. Chem. Phys. 161, 121101 (2024).
García-Alfonso, E., Barranco, M., Halberstadt, N. & Pi, M. Time-resolved solvation of alkali ions in superfluid helium nanodroplets. J. Chem. Phys. 160, 164308 (2024).
Frasinski, L. J. Covariance mapping techniques. J. Phys. B: Mol. Optical Phys. 49, 152004 (2016).
Vallance, C., Heathcote, D. & Lee, J. W. L. Covariance-map imaging: a powerful tool for chemical dynamics studies. J. Phys. Chem. A 125, 1117–1133 (2021).
Albrechtsen, S. H., Christensen, J. K., Tanyag, R. M. P., Kristensen, H. H. & Stapelfeldt, H. Laser-induced Coulomb explosion of heteronuclear alkali-metal dimers on helium nanodroplets. Phys. Rev. A 109, 043112 (2024).
Albrechtsen, S. H. et al. Observing the primary steps of ion solvation in helium droplets. Nature 623, 319–323 (2023).
Albrechtsen, S. H. et al. Femtosecond-and-atom-resolved solvation dynamics of a Na+ ion in a helium nanodroplet. J. Chem. Phys. 162, 174309 (2025).
Christensen, J. K. et al. Time-resolved solvation dynamics of Li+, Na+ and K+ ions in liquid helium nanodroplets. Phys. Chem. Chem. Phys. 27, 24184–24194 (2025).
Lewerenz, M., Schilling, B. & Toennies, J. P. A new scattering deflection method for determining and selecting the sizes of large liquid clusters of 4He. Chem. Phys. Lett. 206, 381–387 (1993).
Toennies, J. P. & Vilesov, A. F. Superfluid helium droplets: a uniquely cold nanomatrix for molecules and molecular complexes. Angew. Chem. Int. Ed. 43, 2622–2648 (2004).
Brauer, N. B. et al. Critical Landau velocity in helium nanodroplets. Phys. Rev. Lett. 111, 153002 (2013).
Lehmann, K. K. Potential of a neutral impurity in a large 4He cluster. Mol. Phys. 97, 645–666 (1999).
Sheps, L., Crowther, A. C., Elles, C. G. & Crim, F. F. Recombination dynamics and hydrogen abstraction reactions of chlorine radicals in solution. J. Phys. Chem. A 109, 4296–4302 (2005).
Einstein, A. Über die von der molekularkinetischen Theorie der Wärme geforderte Bewegung von in ruhenden Flüssigkeiten suspendierten Teilchen. Ann. der Phys. 322, 549–560 (1905).
Saxton, M. J. & Jacobson, K. SINGLE-PARTICLE TRACKING: applications to membrane dynamics. Annu. Rev. Biophys. 26, 373–399 (1997).
Metzler, R. & Klafter, J. The random walk’s guide to anomalous diffusion: a fractional dynamics approach. Phys. Rep. 339, 1–77 (2000).
Huang, R. et al. Direct observation of the full transition from ballistic to diffusive Brownian motion in a liquid. Nat. Phys. 7, 576–580 (2011).
Chen, W. et al. Ballistic diffusion fronts in biomolecular condensates. Nat. Nanotechnol. 20, 1062–1070 (2025).
Choi, M. Y. et al. Infrared spectroscopy of helium nanodroplets: novel methods for physics and chemistry. Int. Rev. Phys. Chem. 25, 15 (2006).
Mauracher, A. et al. Cold physics and chemistry: collisions, ionization and reactions inside helium nanodroplets close to zero K. Phys. Rep. 751, 1–90 (2018).
Levine, R. D. & Bernstein, R. B. Dynamical stereochemistry of activationless bimolecular reactions of ions and neutrals. J. Phys. Chem. 92, 6954–6958 (1988).
Pan, H., Wang, F., Czakó, G. & Liu, K. Direct mapping of the angle-dependent barrier to reaction for Cl + CHD3 using polarized scattering data. Nat. Chem. 9, 1175–1180 (2017).
Ploenes, L. et al. Collisional alignment and molecular rotation control the chemi-ionization of individual conformers of hydroquinone with metastable neon. Nat. Chem. 16, 1876–1881 (2024).
Zou, J. et al. Ne*(3P2,0) + CO chemi-ionization reactions: atomic alignment and molecular orientation effects. J. Chem. Phys. 163, 054301 (2025).
Brooks, P. R. Reactions of oriented molecules. Science 193, 11–16 (1976).
Aoiz, F. J. et al. A new perspective: imaging the stereochemistry of molecular collisions. Phys. Chem. Chem. Phys. 17, 30210–30228 (2015).
Schouder, C. et al. Laser-induced Coulomb explosion imaging of (C6H5Br)2 and C6H5Br–I2 dimers in helium nanodroplets using a Tpx3Cam. J. Phys. B: ., Mol. Optical Phys. 54, 184001 (2021).
Schouder, C. A., Chatterley, A. S., Pickering, J. D. & Stapelfeldt, H. Laser-induced coulomb explosion imaging of aligned molecules and molecular dimers. Annu. Rev. Phys. Chem. 73, 323–347 (2022).
Willitsch, S., Bell, M. T., Gingell, A. D., Procter, S. R. & Softley, T. P. Cold reactive collisions between laser-cooled ions and velocity-selected neutral molecules. Phys. Rev. Lett. 100, 043203 (2008).
Mikosch, J. et al. Imaging nucleophilic substitution dynamics. Science 319, 183–186 (2008).
Chang, Y.-P. et al. Specific chemical reactivities of spatially separated 3-aminophenol conformers with cold Ca+ Ions. Science 342, 98–101 (2013).
Carrascosa, E., Meyer, J. & Wester, R. Imaging the dynamics of ion–molecule reactions. Chem. Soc. Rev. 46, 7498–7516 (2017).
Ma, J. C. & Dougherty, D. A. The Cation−π Interaction. Chem. Rev. 97, 1303–1324 (1997).
Mahadevi, A. S. & Sastry, G. N. Cation−π interaction: its role and relevance in chemistry, biology, and material science. Chem. Rev. 113, 2100–2138 (2013).
Renzler, M. et al. Communication: dopant-induced solvation of alkalis in liquid helium nanodroplets. J. Chem. Phys. 145, 181101 (2016).
Hauser, A. W. & Lara-Castells, M. P. D. Spatial quenching of a molecular charge-transfer process in a quantum fluid: the Csx–C60 reaction in superfluid helium nanodroplets. Phys. Chem. Chem. Phys. 19, 1342–1351 (2017).
de Lara-Castells, M. P., Hauser, A. W. & Mitrushchenkov, A. O. Ab initio confirmation of a harpoon-type electron transfer in a helium droplet. J. Phys. Chem. Lett. 8, 4284–4288 (2017).
Castillo-García, A. et al. A Path integral molecular dynamics simulation of a harpoon-type redox reaction in a helium nanodroplet. Molecules 26, 5783 (2021).
Chandler, D. W. & Houston, P. L. Two-dimensional imaging of state-selected photodissociation products detected by multiphoton ionization. J. Chem. Phys. 87, 1445–1447 (1987).
Eppink, A. T. J. B. & Parker, D. H. Velocity map imaging of ions and electrons using electrostatic lenses: Application in photoelectron and photofragment ion imaging of molecular oxygen. Rev. Sci. Instrum. 68, 3477–3484 (1997).
Fehre, K. et al. Absolute ion detection efficiencies of microchannel plates and funnel microchannel plates for multi-coincidence detection. Rev. Sci. Instrum. 89, 045112 (2018).
Fisher-Levine, M. & Nomerotski, A. TimepixCam: a fast optical imager with time-stamping. J. Instrum. 11, C03016 (2016).
Nomerotski, A. Imaging and time stamping of photons with nanosecond resolution in Timepix based optical cameras. Nucl. Instrum. Methods Phys. Res. Sect. A: Accelerators, Spectrometers, Detect. Associated Equip. 937, 26–30 (2019).
Zhao, A. et al. Coincidence velocity map imaging using Tpx3Cam, a time stamping optical camera with 1.5 ns timing resolution. Rev. Sci. Instrum. 88, 113104 (2017).
Pitters, F. et al. Time resolution studies of Timepix3 assemblies with thin silicon pixel sensors. J. Instrum. 14, P05022–P05022 (2019).
Bromberger, H. et al. Shot-by-shot 250 kHz 3D ion and MHz photoelectron imaging using Timepix3. J. Phys. B: ., Mol. Optical Phys. 55, 144001 (2022).
Kristensen, H. H. et al. Quantum-state-sensitive detection of alkali dimers on helium nanodroplets by laser-induced coulomb explosion. Phys. Rev. Lett. 128, 093201 (2022).
Hansen, J. L. et al. Control and femtosecond time-resolved imaging of torsion in a chiral molecule. J. Chem. Phys. 136, 204310 (2012).
Christiansen, L. et al. Laser-induced Coulomb explosion of 1,4-diiodobenzene molecules: studies of isolated molecules and molecules in helium nanodroplets. Phys. Rev. A 93, 023411 (2016).
Shepperson, B. et al. Strongly aligned molecules inside helium droplets in the near-adiabatic regime. J. Chem. Phys. 147, 013946 (2017).
Dick, B. Inverting ion images without Abel inversion: maximum entropy reconstruction of velocity maps. Phys. Chem. Chem. Phys. 16, 570–580 (2014).
Gomez, L. F., Loginov, E., Sliter, R. & Vilesov, A. F. Sizes of large He droplets. J. Chem. Phys. 135, 154201 (2011).
Harms, J., Toennies, J. P. & Dalfovo, F. Density of superfluid helium droplets. Phys. Rev. B 58, 3341 (1998).
Alcock, C. B., Itkin, V. P. & Horrigan, M. K. Vapour pressure equations for the metallic elements: 298–2500K. Can. Metall. Q. 23, 309–313 (1984).
Acknowledgements
We thank Jan Thøgersen for expert help in the laboratory. The benzene molecules in Fig. 4 were rendered using Mol*. H.S. acknowledges support from Villum Fonden through a Villum Investigator Grant number 25886.
Author information
Authors and Affiliations
Contributions
J.K.C., C.E.P., S.H.A. and J.G. designed and built the experimental set-ups, maintained the experimental apparatus and carried out the experiments. J.K.C., C.E.P. and S.H.A. and H.S. analyzed the experimental data. F.C. designed, wrote the code for and carried out the molecular dynamics simulations and all electronic structure calculations. All authors discussed and interpreted the data. H.S., J.K.C. and F.C. wrote the manuscript and supplementary text and all authors contributed to the editing and reviewing.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Christensen, J.K., Petersen, C.E., Albrechtsen, S.H. et al. Real-time observation of the diffusion-limited formation of a cation-molecule complex. Nat Commun 17, 1249 (2026). https://doi.org/10.1038/s41467-025-68008-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-68008-5