Abstract
Whether a booster dose of hepatitis B vaccine is necessary for adolescents remains controversial. We conducted a retrospective cohort study from the Immunization Information System and The National Notifiable Disease Reporting System to evaluate the impact of hepatitis B virus (HBV) vaccination programs (universal infant hepatitis B immunization and adolescent booster) on HBV infection. The vaccine effectiveness (VE) of hepatitis B vaccine is 64.43% (95% CI 57.78% − 71.08%) within 10 years of birth, escalates to 95.36% (95% CI 94.84% − 95.88%) by 20 years, and decreases to 67.35% (95% CI 63.86% − 70.85%) by 30 years. Compared with Beijing’s birth-vaccinated-only group, the booster group showed a lower relative VE in the first 10 years but a slight increase thereafter. Compared with the national control group, the booster group maintained a relatively high relative VE with a smaller decline magnitude. This study demonstrates that adolescent HBV booster vaccination administered during junior high school improves hepatitis B surface antibody levels and contributes to a further reduction in the incidence of HBV infection.
Similar content being viewed by others
Introduction
Hepatitis B virus (HBV) infection is associated with the risk of chronicity, cirrhosis, hepatocellular carcinoma (HCC) and subsequent mortality. World Health Organization (WHO) reported that an estimated 254 million people were living with hepatitis B in 2022, with an HBV incidence of 2.2 million and 1.1 million deaths attributed to liver failure, cirrhosis, or HCC related to HBV infections1. Immunization with hepatitis B vaccines is widely recognized as the most effective strategy for preventing HBV infection and its progression2.
Hepatitis B vaccination is recommended by WHO for all children globally. A core standard for national immunization programs globally should be ensuring that all children receive at least three doses of the HBV vaccine3. Prior research has indicated that although the protective levels of hepatitis B surface antibody (HBsAb) induced by hepatitis B vaccination decrease gradually with time, they can persist for at least 15 years in most individuals4,5,6. Importantly, even when HBsAb becomes undetectable, immune memory stored in immune cells still provides effective defense against HBV7. However, some studies have reported that this vaccine-induced immune memory may fade after reaching a certain age, instead of persisting lifelong8,9,10. Since the protective efficacy of vaccines rarely reaches 100%, a lower protective effect is associated with a higher risk of vaccine breakthrough infections. Some vaccine breakthrough infections were observed among immunized children born to mothers with chronic HBV infection even when hepatitis B immune globulin was administered in a timely manner as they were growing up11,12. Studies have shown that the incidence of HBV vaccine breakthrough infections can reach approximately 25% by 20 years after completion of primary full-course HBV vaccination13,14,15. What’s more, differences in how individuals respond to vaccines, especially those with less-than-ideal immune responses, can also contribute to the occurrence of breakthrough infections in some cases.
Currently, the necessity of administering an HBV vaccine booster dose during adolescence (10–20 years of age) remains a topic of controversy12,16,17,18. However, given the observation of HBV infection in adolescents who received primary vaccination in infancy12, clinicians have put forward the recommendation that an HBV vaccine booster should be administered to this population during adolescence12,16. Among individuals with non-seroprotective levels of HBsAb, 88% exhibited a rapid increase in HBV-specific immunoglobulin G (IgG) titer following a booster dose. This finding indicates the presence of immunological memory and confirms that memory T-cells sustain protective immunity against HBV5. However, previous studies were either small-sample-size studies or focused solely on the short-term efficacy of HBV vaccines17,18. There were no studies with a large population and a long period supporting the necessity and the effectiveness of adolescent boosters against HBV infection.
The burden of hepatitis B infection has declined significantly over the past decades1. Data from four national seroepidemiological surveys showed that the prevalence of HBsAg in the Chinese population decreased from 9.72 in 1992 to 7.18% in 2006, and further decreased to 5.86% in 202019. Beijing started HBV vaccination in 1987, advocated it for routine hepatitis B vaccination of newborns in 1990, integrated it in the Expanded Program on Immunization (EPI) in 2002, and provided free boosters for the adolescents aged 13–14 years old in 2008. The hepatitis B vaccine immunization strategy in Beijing has undergone significant evolution over the past three decades (Fig. 1 and Supplementary Information files). In Beijing, the Hepatitis B Surface Antigen (HBsAg) prevalence has dramatically dropped from 6.03 in 1992 to 2.73% in 2014, when it was less than 1% in individuals under 25 years old20. Simultaneously, the incidence rate of hepatitis B has also decreased in Beijing. Beijing has implemented HBV vaccination strategies for over 30 years, and the administration of adolescent HBV booster doses has also been in practice for more than 15 years (Fig. 1 and Supplementary Information files). However, the long-term effectiveness of both primary hepatitis B vaccination and booster doses remains unevaluated. Additionally, the correlation between the reductions in HBsAg prevalence and hepatitis B incidence and the implementation of HBV vaccination programs, requires further investigation to be confirmed. Therefore, we conducted an evaluation to assess the effects of universal infant hepatitis B immunization and adolescent booster on the prevention and control of HBV infection in Beijing across the past 30 years.
EPI Expanded Program on Immunization.
Results
Universal infant hepatitis B immunization program
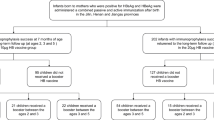
In this retrospective cohort study, the vaccinated cohort comprised 1,081,065 individuals born between 1993 and 2007, while the unvaccinated cohort included 415,994 individuals born between 1985 and 1987 (Fig. 2 and Supplementary Table 1). After linkage with the hepatitis B case database of the National Notifiable Disease Reporting System (NNDRS), the 10-year cumulative incidence of hepatitis B among individuals in the vaccinated group was 10.18/100,000 persons (95% CI 8.36/100,000−12.26/100,000). In contrast, the 10-year cumulative incidence of hepatitis B in the unvaccinated group was 79.66/100,000 persons (95% CI 71.72/100,000−87.60/100,000). Based on these incidence data, the VE of the hepatitis B vaccine within 10 years of birth was calculated to be 64.43% (95% CI 57.78−71.08%). The VE of the hepatitis B vaccine escalated to 95.36% (95% CI 94.84−95.88%) within 20 years after birth, declined moderately to 87.24% (95% CI 86.28−88.19%) within 25 years after birth, and further decreased to 67.35% (95% CI 63.86−70.85%) within 30 years after birth. Overall, the VE of hepatitis B vaccine exhibited a clear protection against HBV infection. Even with an observed decline of VE, the vaccine still reduced infection risk by more than two-thirds (67.35%) compared to no vaccination.
Group A, the unvaccinated group; Group B, the vaccinated group. The incidence rate of hepatitis B for Group A and Group B were calculated by 119/415,994 and 110/1081,065 within 10 years, 257/415,994 and 209/1081,065 within 15 years, 3624/415,994 and 309/764,323 within 20 years, 4824/415,994 and 690/466,184 within 25 years, and 5452/415,994 and 334/78056 within 30 years (Supplementary Table 1). The upper limit of 95% confidence interval (CI) is presented for the incidence rate of hepatitis B. The vaccine effectiveness (VE) and 95% CI are presented.
Booster hepatitis B vaccination
Within one year after administration of the hepatitis B vaccine booster dose, the incidence rate of HBV infection was lower in Group B1 than Group B2 (0.49/100,000 persons vs. 4.1/100,000 persons), with a relative vaccine effectiveness (rVE) of 88.05% (95% CI 64.32−96.07%) (Fig. 3A and Supplementary Table 2). While compared to Group C, the rVE of hepatitis B vaccine in Group B1 was 93.16% (95% CI 82.47−97.34%).
A hepatitis B incidence rate and relative vaccine effectiveness at 1 years, 5 years, 10 years, and 15 years after the booster dose. The numbers of hepatitis B virus (HBV) positive infections and total populations in the three groups of B1, B2 and C are presented in Supplementary Table 2. The upper limit of 95% confidence interval (CI) is presented for the incidence rate of hepatitis B. The vaccine effectiveness (VE) and 95% CI are presented. B vaccine effectiveness of hepatitis B vaccine booster dose at different periods. Group B1, the booster group; Group B2, the local control group; Group C, the national control group. The detailed numbers of hepatitis B and total population are presented in Supplementary Table 3. The relative VE and 95% CI are presented.
Within five years after administration of the hepatitis B vaccine booster dose, a lower incidence rate of HBV infection was observed in Group B1 (5.52/100,000 persons) compared to that in Group B2 (17.64/100,000 persons) or in Group C (85.89/100,000 persons) with the rVE of hepatitis B vaccine being 68.71% (95% CI 52.02−79.59%) and 93.58% (95% CI 91.25%−95.28%).
For the 10 years and 15 years after hepatitis B vaccine booster administration, higher HBV infection incidence rates were observed across all three groups. The rVE of Group B1 relative to Group B2 was 22.13% (95% CI 3.36−37.26%) at 10 years post-booster and 38.73% (95% CI 25.48−49.73%) at 15 years post-booster. In contrast, the rVE of Group B1 relative to Group C remained relatively high with 84.15% (95% CI 81.72−86.25%) at 10 years post-booster and 79.86% (95% CI 75.87−83.18%) at 15 years post-booster.
When compared to Group B2, the rVE of Group B1 declined sharply and remained at a low level 10 years after booster administration (Fig. 3B and Supplementary Table 3). In contrast, when compared to Group C, the rVE of Group B1 was consistently maintained at a relatively high level, with a much smaller magnitude of decline over the same period.
Incidence rate of HBV during 2005–2022
Between 2005 and 2022, the reported incidence rate of acute hepatitis B in Beijing was 6.12/100,000 persons in 2005, peaked at 6.34/100,000 persons in 2007, and declined to 0.43/100,000 persons in 2022 (Fig. 4A and Supplementary Table 4).
A The Join-point regression of annual incidence rates of hepatitis B infection in different age groups. B HBsAb seropositive rates in different stages in three surveys conducted in 2006, 2014 and 2020. C HBsAb titers in different stages in three surveys conducted in 2006, 2014 and 2020. The numbers of participants testing for HBsAb seropositive rates and antibody titers in the years of 2006, 2014 and 2020 was 193, 95 and 87 for the 8–12 years, 250, 223 and 19 for the 13–17 years, and 278, 607 and 53 for the 19–23 years. The HBsAb positive rate and the upper limit of 95% confidence interval were presented. The HBsAb geometric mean titer and the upper limit of 95% confidence interval were presented. Two-sided chi-square test is used to compare the HBsAb positive rates among three groups. Kruskal-Wallis H test is used to compare the HBsAb geometric means of the three groups. A P-value less than 0.02 was considered statistically significant after adjustments for three comparisons. HBsAb, hepatitis B surface antibody.
Joinpoint regression model showed that the incidence rate of acute hepatitis B in the age groups of 25−49 years old and ≥50 years old in Beijing increased prior to 2007, followed by a subsequent decrease. Two turning points of decline were observed in 2007 and 2013 for the group of 25−49 years (2007−2013: annual percent change [APC] = −25.47%, 95% CI −31.02 to −19.47%; 2013−2022: APC = −12.33%, 95% CI −17.33% to −7.04%). For the group of ≥50 years old, the turning points were in 2007 and 2011 (2007−2011: APC = −24.39%, 95% CI −32.47% to −15.34%; 2011−2022: APC = −8.56%, 95% CI −10.81% to −6.26%). While in the groups of 0−14 years old and 15 − 24 years old, the incidence rate of acute hepatitis B revealed a downward trend. The incidence rate of acute hepatitis B in the group of 15 − 24 years old showed a sharp decrease after the booster dose of hepatitis B vaccination (2005 − 2009: APC = −11.50%, 95% CI −17.46% to −5.11%; 2009−2015: APC = −37.80%, 95% CI −44.08% to −30.81%). The incidence rate of acute hepatitis B in the group of 0−14 years old showed a continuous decrease after the implementation of universal infant hepatitis B immunization (2005−2022: APC = −16.32%, 95% CI −23.63% to −8.32%).
HBsAb in three cross-sectional surveys
HBsAb seropositivity rates and antibody titers across different age groups were presented based on the three seroepidemiological surveys conducted in Beijing in 2006, 2014, and 2020 (Fig. 4B and C). In all three cross-sectional surveys, participants aged 8−12 years, 13−17 years and a subset of participants aged 19−23 years had received three doses of the universal infant hepatitis B immunization. In the 2014 and 2020 surveys, participants aged 13−17 years had additionally received a booster dose of the hepatitis B vaccine. In the 2020 survey alone, participants aged 19−23 years had received a booster dose of the hepatitis B vaccine. Statistical analyses revealed no significant differences in HBsAb seropositivity rates (P = 0.870) and HBsAb titers (P = 0.228) among the participants aged 8−12 years across the three surveys. In contrast, participants aged 13−17 years in the 2020 survey exhibited the highest HBsAb seropositivity rates (P = 0.022) and HBsAb antibody titers (P < 0.001). A consistent pattern was observed in the participants aged 19−23 years in the 2020 survey (P = 0.002 and P < 0.001).
Adverse events of Hepatitis B vaccine
The Adverse Events Following Immunization (AEFI) classification includes rare adverse reactions (such as allergic rash, urticaria and maculopapular rash), common adverse reactions (such as fever, erythema and induration in injection site), psychogenic reactions and coincidental events. Based on data from the AEFI surveillance system, the incidence of reported vaccine-related adverse events within 28 days after administration of a hepatitis B booster dose was significantly lower than that after each of the primary three doses (3.29/100,000 doses vs. 12.93/100,000 doses, P < 0.001) (Supplementary Table 5). We also analyzed the rare adverse reaction rate of hepatitis B vaccination, and the same phenomenon was also observed in the incidence of reported rare vaccine-related adverse events within 28 days of hepatitis B vaccine (0.94/100,000 doses vs. 4.22/100,000 doses, P < 0.001).
Discussion
This study described the dynamic changes in the prevalence and incidence of HBV infections following the introduction of hepatitis B vaccine in Beijing, and evaluated the protective effectiveness of hepatitis B vaccination and booster immunization at multiple time points post-administration. Results demonstrated that hepatitis B vaccine retained a protective efficacy for many years after primary immunization, while booster immunization further enhanced this protection, providing both superior and more durable efficacy against HBV infection.
The hepatitis B vaccination provides substantial long-term benefits globally. One study conducted in Italy confirmed that the first 30 years of implementing universal hepatitis B vaccination remained a cost-saving public health strategy, which was associated with significant reductions in HBV-related diseases (an 82% decrease in the number of HBV infections, chronic HBV cases, and hepatocellular carcinoma cases) and related healthcare costs (a 67% reduction during the immunization period)21. A 37-year extended follow-up study demonstrated that hepatitis B vaccine offers 72% (95% CI 30−89%) protection against liver cancer in China22. Another study showed that among individuals born after 1991, hepatitis B vaccine achieved an overall VE of 58%, with the VE reaching 85% for those followed for ≥20 years, whereas no protective effect was observed in individuals born before 199023. Notably, in the era of universal infant HBV vaccination, hepatitis B vaccine was associated with reduced risks of all-cause mortality and cancer-related mortality, and it remained highly effective against HBV infection, providing up to 20 years of protection. Consistent with these global and national findings, the Beijing Municipal Health Commission has implemented a series of strategies to eliminate HBV transmission. In our study, individuals born after the implementation of the universal hepatitis B vaccination program and booster immunization had a lower hepatitis B incidence rate than those born before the program’s implementation. Specifically, the VE was 95.36% within 20 years of birth, and remained at 67.35% even 30 years after birth. When compared to the birth-vaccinated-only group, the rVE of the booster HBV vaccine exhibited a notable decline followed by a marginal increase between 10 and 15 years after the booster administration. This may be related to the fact that in the non-booster group, over 15 years after their last vaccination, antibody titers have stabilized or slightly decreased, with relatively constant vaccine protective effect. For the booster group, the booster vaccination induces a significant rise in antibody titers, greatly improving protective effect; however, its vaccine protective effect declines more sharply than that in the non-booster group after 10 years, leading to a lower rVE. The previous study has shown that the HBV carrier rate in the population aged over 20 years has started to rise sharply due to various factors such as lifestyle behaviors19. The booster group exhibits a better protective effect compared to the non-booster group, contributing to the increase of rVE after 10 years of the booster vaccination. In Iran, the estimated long-term vaccine effectiveness against hepatitis B infection was 29% after 25 years24, much lower than the 87.24% observed in Beijing. This discrepancy may be associated with difference in hepatitis B vaccination policies and vaccine types. Overall, the long-term protection effectiveness of hepatitis B vaccine is critical for further controlling HBV prevalence and achieving the 2030 global hepatitis elimination goals. It should be noted that the decline in hepatitis B incidence cannot be attributed solely to vaccination. It may also be associated with improved population health literacy fueled by economic development, which supports the implementation of transmission-limiting measures, such as better management of chronic infections and avoidance of unsafe healthcare practices. These measures, in turn, are made possible by both increased financial resources and enhanced capacity to access, interpret, and act on health-related information.
Whether an adolescent or young adult hepatitis B vaccine booster dose is necessary remains controversial. Booster vaccination is generally deemed unnecessary, even when HBsAb levels decline to less than 10 mIU/mL5,23,25. The booster dose is not recommended by international guidelines including that of the WHO3. In contrast, other studies have supported adolescent booster for certain groups such as individuals born to HBsAg-positive mothers18,26. Further health economic evaluation has also indicated that this booster strategy yields additional Quality-Adjusted Life Years and is cost-effective27. In our study, the adolescent booster group exhibited a lower incidence rate of HBV infection and significantly higher rVE compared to non-booster group. These findings provide evidence that administering an HBV vaccine booster to 13−14 years old enhances protection against HBV infection relative to the 3-dose primary vaccination series alone. This aligns with results from a 2024 meta-analysis, which demonstrated that HBV vaccine booster doses effectively reduce HBV infection compared to non-booster populations28. Our study also reviewed Beijing’s HBV immunization strategies, population-level incidence levels, and HBsAb seroprevalence over 30 years since the initiation of universal infant hepatitis B immunization. It reported that adolescent booster was associated with higher HBsAb titers and lower incidence than the 3-dose universal infant hepatitis B vaccine series alone. Meanwhile, adolescent booster did not increase the incidence of reported AEFI following immunization. Consequently, the provision of optional booster doses, when implemented under suitable circumstances, holds the potential to enhance the effectiveness of hepatitis B prevention and control strategies. Collectively, these results indicate that providing optional HBV vaccine booster doses, when administered under appropriate conditions, has the potential to enhance the effectiveness of HBV prevention and control initiatives. However, more in-depth assessments of its health economic value are still needed to fully evaluate the long-term feasibility and overall public health impact of this strategy.
Hepatitis B infection, as a major infectious disease, is one of the key targets for infectious diseases prevention and control in China. Over the past 30 years, China has implemented a comprehensive prevention and control strategy centered on HBV vaccination, supplemented by integrated prevention and treatment measures. This strategy has yielded remarkable outcomes in HBV control: the prevalence of HBsAg in the Chinese population has declined consistently, from 9.72 in 1992 to 5.86% in 202019. In particular, the most substantial reduction has been observed in children under five years old, with HBsAg prevalence dropping to 0.1% in 2022, ranking China among the countries with the lowest childhood HBsAg prevalence globally29. Beijing is the only city in China that has implemented an adolescent HBV vaccine booster program continuously. Beijing’s adolescent booster plays significant positive role for HBV control and was associated with higher HBsAb titers and lower incidences.
This study had several limitations that should be acknowledged. First, when calculating HBV infection incidence, the analysis did not account for population migration (both inflow and outflow). Second, the observation end point for hepatitis B cases was set in 2022. Given the study’s focus on long-term VE, continued surveillance of HBV infection and vaccine breakthrough infections is urgently needed to further characterize the duration of HBV vaccine protection and update VE estimates over extended follow-up periods. Third, vaccine types were not stratified in the analysis. During the universal infant vaccination era in Beijing (1995–2007), a 10-5-5 microgram hepatitis B vaccine dosage schedule was used, whereas other regions in China adopted a 5-5-5 microgram schedule. This dosage difference may have led to overestimation of the immunization’s VE in Beijing when compared to the national control group, as the higher initial dosage in Beijing could independently contribute to stronger baseline protection, which might also explain the large discrepancy between rVE of the booster group compared to the local control group and the national control group over time. Fourth, vaccination data from IIS system was only complete after 2001. Before 2001, vaccination records relied solely on paper-based registration (electronic systems were unavailable). Although these paper records were later retroactively entered into the electronic IIS, the manual data transfer process may have introduced omissions. Consequently, some children’s vaccination histories before 2001 may be incomplete or missing from the database, potentially biasing analyses of vaccine exposure. Fifth, the 2020 seroepidemiological survey data used in this study was part of a national survey. While national surveys typically aim for broad representativeness, the sampling framework may not fully align with the specific demographic and epidemiological characteristics of Beijing’s population, potentially introducing sampling bias that may affect the validity of serological indicators. Sixth, the incomplete data of general hepatitis B cases in NNDRS, which may be caused by the national system’s criteria for reporting hepatitis B cases and the different criteria of numerous involved institutions, can lead to bias in the results. The hepatitis B vaccine has an obvious and undoubted preventive and control effect, especially among adolescents. So, it can be speculated that the proportion of hepatitis B virus-infected individuals or unreported hepatitis B patients in the vaccinated group is far lower than that in the unvaccinated group. In addition, as there is good monitoring system, early high hepatitis B vaccine coverage, high economic level, quality education contributing to low under reporting rates among children and adolescents in Beijing City, it may be lower than that in national data outside Beijing. These situations may lead to a certain underestimation of the effect of the hepatitis B vaccine booster dose for adolescents. The universal infant hepatitis B immunization initiative implemented in Beijing has achieved success over a 30-year follow-up period, with a reduction in HBV infection incidence. Furthermore, the adolescent HBV vaccine booster program administered during junior high school in the real-world setting of Beijing, may have played a role in elevating antibody titer among adolescents, concomitantly contributing to a further decline in the incidence rate. These outcomes highlight the necessity of sustaining and further expanding this component of Beijing’s immunization strategy to enhance public health protection against hepatitis B. The implementation of viable long-term hepatitis B vaccination strategies holds significance in not only forestalling HBV-related complications but also attaining the WHO cascade-of-care targets.
Methods
Study design
The study adopted a retrospective cohort design, utilizing data extracted from Immunization Information System (IIS) and NNDRS to evaluate the impact of HBV vaccination programs including universal infant hepatitis B immunization and adolescent booster on HBV infection in Beijing, China. Furthermore, three serological cross-sectional studies were conducted to assess HBsAb positivity rate and antibody titers among residents who had resided in Beijing for at least six months, thereby providing additional evidence for the long-term effectiveness of these vaccination strategies. The study protocol was approved by the Ethics Review Committee of Beijing Center for Disease Prevention and Control (BJCDC2024015). Informed consent was waived for this study. For the three serological cross-sectional studies, informed consent was obtained from the legal guardians or primary caregivers of participating children, as well as from all adult participants, prior to conducting the interviews and collecting blood samples. The 2006 and 2014 surveys were approved by the Beijing Center for Disease Control and Prevention20,30, and the 2020 survey received ethical approval from the Ethical Review Committee of the Chinese Center for Disease Control and Prevention19.
Participants
The unvaccinated group (Group A) comprised individuals born in Beijing between 1985 and 1987, and those having retrievable records in the IIS were excluded from this group. The vaccinated group (Group B) was further divided into two subgroups: the vaccinated at birth and adolescent booster group (Group B1) and the birth-vaccinated-only group (Group B2). For the retrospective cohort study, the inclusion criteria for Group B were defined as follows: (1) individuals born in Beijing between 1993 and 2007, with the first dose of hepatitis B vaccine administered in an obstetrical department of a medical institution in Beijing; (2) individuals who received the third dose of hepatitis B vaccine before reaching one year of age. Individuals without valid identification numbers were excluded from Group B. Group B1 included individuals born in Beijing between 1996 and 2007 who received both the 3-dose primary hepatitis B vaccine series during infancy and a 1-dose booster vaccination in the seventh grade (typically corresponding to early adolescence, approximately 12–13 years of age). Group B2 comprised individuals in Beijing born between 1993 and 1995 who received only a 3-dose primary hepatitis B vaccine series during infancy. Additionally, the national control group (Group C) was defined as individuals born between 1996 and 2007 in regions of China other than Beijing, who received only the 3-dose primary HBV vaccination series during infancy.
For the three cross-sectional studies included in this analysis, participants were restricted to Beijing residents who had resided in the city for more than half a year. The target age ranges for these surveys were 1–59 years in 200630, 1–59 years in 201420, and 1–69 years in 202019. Hepatitis B seroepidemiological surveys in 2006 and 2014 were not part of the sampling for the national survey. Instead, these two surveys constituted independent hepatitis B seroepidemiological surveys among the population of Beijing, conducted independently by the Beijing Center for Disease Control and Prevention (Beijing CDC). The 2020 survey was part of the national serological survey. The sampling strategies utilized in the 2006, 2014, and 2020 surveys were detailed comprehensively in prior publications19,20,30.
Data source and collection
Hepatitis B vaccination information and vaccine coverage rates were retrieved from the IIS of Beijing, with detailed data specifications provided in the Supplementary Information files. Hepatitis B case data were obtained from NNDRS. NNDRS is a hospital-based, passive national surveillance system that covers all county-level hospitals across 31 provinces in China, with additional system details available in the Supplementary Information files. The diagnosis of hepatitis B was confirmed in accordance with the national diagnostic criteria for viral hepatitis B (Standard No.: WS 299–2008). Epidemiological characteristics of hepatitis B cases, including name, gender, identification number, date of birth and date of onset, were acquired from NNDRS.
Procedure
Data on hepatitis B vaccination and HBV infection in the vaccinated and unvaccinated groups were extracted from the IIS and NNDRS. From the IIS, the following information was retrieved for each individual: unique identifiers (either identification number or name combined with date of birth), gender, number of HBV vaccine doses administered, maternal HBsAg status, and vaccine administration dates. Individuals born between 1993 and 2007 who could not be matched to records in the IIS were excluded. For those who could be matched by either identification number or name plus date of birth) across both the IIS and NNDRS, they were confirmed as HBV cases in the vaccinated group, and the date of symptom onset for these cases was recorded. The total number of individuals born in Beijing between 1985 and 1987 was obtained from the Beijing Municipal Commission of Health. Individuals in this birth cohort were also matched to records in NNDRS to identify HBV cases in the unvaccinated group, with the date of symptom onset for these cases similarly recorded. A multistage cluster sampling was used for the three serological cross-sectional studies to explore the HBsAb titers and positivity in residents in 200630, 201420 and 202019 in Beijing. Specifically, the sampling process proceeded in three stages. Firstly, for district-level sampling, five urban districts (Dongcheng, Xicheng, Xuanwu, Chaoyang, and Haidian) and five suburban districts (Changping, Tongzhou, Fangshan, Miyun, and Huairou) were selected as survey districts in 2006 and 2014. In 2020, the district-level sampling was adjusted to one urban district (Dongcheng) and one suburban district (Tongzhou). Secondly, for communities/villages-level sampling, two sampling communities/villages were randomly selected in each selected survey district. Thirdly, for individual-level sampling, individuals aged ≥1 year within each sampling community/villages were selected using a random number table.
Basic demographic information of participants was collected via a questionnaire designed specifically for this study. Venous blood samples were obtained from each participant to measure HBsAb levels (including titer quantification and positivity determination).
Outcomes
The primary outcome of this study was the annual incidence rate of HBV infection, with all HBV infection cases confirmed via the NNDRS. Annual incidence rate of HBV infection between 2005 and 2022 was reported. Additionally, VE was estimated based on comparisons of HBV infection incidence rates across three groups: the vaccinated group versus the unvaccinated group, and the vaccinated at birth and adolescent booster subgroup (Group B1) versus the vaccinated at birth only subgroup (Group B2).The incidence rate of HBV infection in any given group over a specific period was calculated using the formula: the incidence rate = number of new HBV infection cases during the study period/total persons at risk. For the primary outcome (assessment of HBV infection status), HBsAg detection was performed using an enzyme-linked immunosorbent assay (ELISA) platform, in compliance with the National Diagnostic Criteria for Viral Hepatitis B (Standard No. WS 299-2008).
The second outcome was the quantification of HBsAb titers and the determination of positivity which are newly collected and analyzed for this study. HBV serological markers were tested using Architect i2000 (Chemiluminescence Microparticle Immunoassay, Abbott, Chicago, USA). Positive cut-off value for HBsAb was defined as ≥10 mIU/L.
The third outcome was the incidence of reported vaccine-related adverse events within 28 days following each dose of HBV vaccination.
Statistical analysis
Descriptive epidemiological methods and Joinpoint regression model were used to analyze the epidemiological characteristics (including temporal trends) of hepatitis B in Beijing City from 2005 to 2022, based on data from the NNDRS. Joinpoint software 5.0.2 was employed to calculate the APC in the reported incidence of acute clinical hepatitis B in Beijing during this period. Categorical variables were compared using the chi-square test (χ 2 test) or Fisher’s exact test. Relative risk (RR) and 95% CI were used to assess the association between different vaccine doses and HBsAb seropositive rates in different groups or birth cohorts by Logistic regression model. VE was estimated by comparing the risk of HBV infection between vaccinated and unvaccinated populations31, using the formula: VE (%) = [1 - (Risk of HBV infection in the vaccinated group/Risk of HBV infection in the unvaccinated group)] × 100. Statistical significance was defined as a two-tailed P < 0.05. All statistical analyses were conducted using Stata 17 software (Stata CorpLP, College Station, TX, USA).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data that support the findings of this study are available from the corresponding author (Luodan Suo: suoluodan2004@163.com and Qing-Bin Lu: qingbinlu@bjmu.edu.cn) upon request and upon approval from the local ethics committee. The data are not publicly available due to ethical restrictions related to participant confidentiality and consent. Additional summary data and the study information are provided in the Supplementary Information. Source data are provided with this paper.
References
World Health Organization. Global hepatitis report 2024: action for access in low- and middle-income countries, <https://www.who.int/publications/i/item/9789240091672> (2024).
Pattyn, J., Hendrickx, G., Vorsters, A. & Van Damme, P. Hepatitis B Vaccines. J. Infect. Dis. 224, S343–S351 (2021).
World Health Organization Hepatitis B vaccines: WHO position paper, July 2017 - Recommendations. Vaccine 37, 223–225 (2019).
European Consensus Group on Hepatitis B Immunity Are booster immunisations needed for lifelong hepatitis B immunity? European Consensus Group on Hepatitis B Immunity. Lancet 355, 561–565 (2000).
Bruce, M. G. et al. Antibody levels and protection after hepatitis B vaccine: results of a 30-year follow-up study and response to a booster dose. J. Infect. Dis. 214, 16–22 (2016).
McMahon, B. J. et al. Antibody levels and protection after hepatitis B vaccine: results of a 22-year follow-up study and response to a booster dose. J. Infect. Dis. 200, 1390–1396 (2009).
Chaves, S. S. et al. Persistence of long-term immunity to hepatitis B among adolescents immunized at birth. Vaccine 30, 1644–1649 (2012).
Lu, C. Y. et al. Waning immunity to plasma-derived hepatitis B vaccine and the need for boosters 15 years after neonatal vaccination. Hepatology 40, 1415–1420 (2004).
Mahmood, S., Shah, K. U. & Khan, T. M. Immune persistence after infant Hepatitis-B vaccination: a systematic review and meta-analysis. Sci. Rep. 8, 12550 (2018).
Stramer, S. L. et al. Nucleic acid testing to detect HBV infection in blood donors. N. Engl. J. Med. 364, 236–247 (2011).
Shahmoradi, S. et al. High prevalence of occult hepatitis B virus infection in children born to HBsAg-positive mothers despite prophylaxis with hepatitis B vaccination and HBIG. J. Hepatol. 57, 515–521 (2012).
Wu, T. W., Lin, H. H. & Wang, L. Y. Chronic hepatitis B infection in adolescents who received primary infantile vaccination. Hepatology 57, 37–45 (2013).
Chen, H. L. et al. Effects of maternal screening and universal immunization to prevent mother-to-infant transmission of HBV. Gastroenterology 142, 773–781.e772 (2012).
Miao, N., Zheng, H., Sun, X., Zhang, G. & Wang, F. Protective effect of vaccinating infants with a 5 microg recombinant yeast-derived hepatitis B vaccine and the need for a booster dose in China. Sci. Rep. 10, 18155 (2020).
Poovorawan, Y. et al. Persistence and immune memory to hepatitis B vaccine 20 years after primary vaccination of Thai infants, born to HBsAg and HBeAg positive mothers. Hum. Vaccin Immunother. 8, 896–904 (2012).
Chinchai, T. et al. Long-term humoral and cellular immune response to hepatitis B vaccine in high-risk children 18-20 years after neonatal immunization. Viral Immunol. 22, 125–130 (2009).
Li, Y. et al. Booster vaccination in infancy reduces the incidence of occult HBV infection in maternal HBsAg-positive children. J. Clin. Transl. Hepatol. 11, 661–669 (2023).
Wang, Y. et al. Adolescent booster with hepatitis B virus vaccines decreases HBV infection in high-risk adults. Vaccine 35, 1064–1070 (2017).
Zheng, H. et al. New progress in HBV control and the cascade of health care for people living with HBV in China: evidence from the fourth national serological survey, 2020. Lancet Reg. Health West Pac. 51, 101193 (2024).
Gao, P. et al. A sero-epidemiological study of hepatitis B among general population in Beijing. Chin. J. Epidemiol. 37, 658–662 (2016).
Boccalini, S. et al. The First 30 Years of the Universal Hepatitis-B Vaccination-Program in Italy: A Health Strategy with a Relevant and Favorable Economic-Profile. Int. J. Environ. Res. Public Health 19, 16365 (2022).
Cao, M. et al. Long term outcome of prevention of liver cancer by hepatitis B vaccine: Results from an RCT with 37 years. Cancer Lett. 536, 215652 (2022).
Le, M. H. et al. Prevalence of Hepatitis B vaccination coverage and serologic evidence of immunity among US-Born children and adolescents from 1999 to 2016. JAMA Netw. Open 3, e2022388 (2020).
Moghadami, M., Dadashpour, N., Mokhtari, A. M., Ebrahimi, M. & Mirahmadizadeh, A. The effectiveness of the national hepatitis B vaccination program 25 years after its introduction in Iran: a historical cohort study. Braz. J. Infect. Dis. 23, 419–426 (2019).
Gara, N. et al. Durability of antibody response against hepatitis B virus in healthcare workers vaccinated as adults. Clin. Infect. Dis. 60, 505–513 (2015).
Qu, C. et al. Efficacy of neonatal HBV vaccination on liver cancer and other liver diseases over 30-year follow-up of the Qidong hepatitis B intervention study: a cluster randomized controlled trial. PLoS Med. 11, e1001774 (2014).
Wang, Y. et al. Cost-effectiveness analysis of hepatitis B vaccine booster in children born to HBsAg-positive mothers in rural China. Int. J. Infect. Dis. 78, 130–139 (2019).
Qiu, J. et al. Efficacy and safety of hepatitis B vaccine: an umbrella review of meta-analyses. Expert Rev. Vaccines 23, 69–81 (2024).
He, W. Q., Guo, G. N. & Li, C. The impact of hepatitis B vaccination in the United States, 1999-2018. Hepatology 75, 1566–1578 (2022).
Wu, J. et al. A sero-epidemiological study on hepatitis B among general population in Beijing. Chin. J. Epidemiol. 28, 555–557 (2007).
Farrington, C. P. Estimation of vaccine effectiveness using the screening method. Int. J. Epidemiol. 52, 14–21 (2023).
Acknowledgements
This work was funded by The Government of Beijing Municipality. We acknowledge all the personnel from the District Center for Disease Control and Prevention who took part in the collection of samples in 2006, 2014 and 2020 surveys. We acknowledge Professor Fuzhen Wang from China Center for Disease Control and Prevention and Li Wang from Chinese Academy of Medical Sciences and Peking Union Medical College for giving valuable advice to this paper. We also acknowledge Shi-Yi Lu for drawing the syringe.
Author information
Authors and Affiliations
Contributions
Huai Wang, Qing-Bin Lu and Luodan Suo had full access to all of the data in the study and took responsibility for the integrity of the data and the accuracy of the data analysis and manuscript. Weixin Chen was responsible for the hepatitis B serological experiments. Pei Gao, Qinyi Ma, Qianli Yuan and Zhiqiang Cao was responsible for data extraction and drafted the figures. Jiang Wu critically reviewed and edited the manuscript. All the authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The author declare no competing interests
Peer review
Peer review information
Nature Communications thanks Peyton Thompson who co-reviewed with Nefer Batsuli; Jan Drobeniuc and Yu Wang for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, H., Chen, W., Gao, P. et al. Retrospective cohort study of hepatitis B immunization strategy effects in Beijing across 30 years. Nat Commun 17, 1523 (2026). https://doi.org/10.1038/s41467-025-68243-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-68243-w