Abstract
Cellular and leaf structural traits influence the regulation of leaf water balance, which in turn impacts leaf gas exchange, plant productivity and drought tolerance. Yet, the role of cell wall composition, and especially that of components that render cell walls flexible—such as pectin—in leaf water relations remains elusive. Here, we investigate the linkages among 26 traits, including cell wall composition, anatomy, and drought tolerance (described by pressure-volume curves) across 69 woody species from sub-tropical dry and wet forests. We find that the lower wilting point of species of dry forests relative to wet forests is associated with contrasting anatomy and wall composition. Pressure-volume traits correlate more strongly with wall composition, and particularly pectin concentration, in dry forests, and with anatomy in wet forests. Thus, pectin-enriched cell walls contribute to the ecological specialization of woody plants in dry versus wet forests. Our findings indicate that leaf hydraulic designs diverge according to two strategies: dry forest species vary in elastic and osmotic function via contrasting pectin concentration (“flexible cell wall” strategy), whereas wet forest species do so via contrasting palisade tissue investment (“stable leaf tissue” strategy). Overall, diversity in cell wall properties across species are strongly linked with drought tolerance.
Similar content being viewed by others
Introduction
Water is essential for the function of leaf tissues, including photosynthesis, and tissue dehydration leads to mortality during drought1,2. The cell wall surrounding plant cells provides structural rigidity and allows for the maintenance of cell volume and hydrostatic pressure necessary for fundamental physiological processes3,4. Thus, variation in cell wall structure has been hypothesized to affect leaf water relations and drought tolerance5,6,7. Indeed, across species, traits, such as smaller leaf size, smaller cells, and thicker cell walls are associated with greater drought tolerance5,6,8,9,10,11. However, the role of cell wall composition as a determinant of cell and leaf drought tolerance remains poorly understood12,13. This knowledge gap hinders an integrated understanding of drought tolerance from the cellular to ecosystem level, limiting the accuracy of predictions regarding plant responses to climate change.
Many leaf-level traits associated with drought tolerance are likely influenced by cell wall composition. The turgor loss point (ψtlp, i.e., the water potential at turgor loss), osmotic potential at full turgor (πo) and bulk modulus of elasticity (εsym, wall elasticity) (measured by pressure-volume (P-V) curves) are key drought tolerance traits driving plant adaptation to diverse habitats14. Specifically, πo is the major determinant of ψtlp; plants from drier habitats exhibit more negative πo and ψtlp14,15,16. Additionally, these dry-adapted plants might exhibit rigid cell walls (high ɛsym), which can mechanically constrain shrinkage and water loss in cells at very negative πo or ψtlp6,17. Indeed, some have proposed that wall elasticity is linked to cell wall composition (e.g., concentrations of cellulose, hemicellulose, lignin, and pectin)13,18,19,20,21. While cellulose and lignin are recognized as forming the mechanical basis of cell walls18,20, a role for pectin in modulating elastic and osmotic adjustments (εsym and πo) through Ca²⁺ chelation has also been suggested22,23,24,25, potentially enhancing environmental responsiveness24,26,27. However, quantitative evidence for the impact of specific cell wall components on physiological traits remains limited and often debated, and these impacts appear to vary depending on the species and environmental context.
The diversification of angiosperms across aridity gradients involved the evolution of diverse leaf drought tolerant strategies. These strategies arise from the coordinated evolution of physiological traits and leaf structural investments14,28,29,30. When plants first colonized land, they inhabited regions with abundant water availability. Leaf design placed minimal emphasis on water limitations, instead prioritizing investments in structural support and photosynthetic tissues, such as palisade mesophyll31,32,33. Consequently, species experiencing limited water stress typically maintain relatively stable water relations through the coordination of physiological traits with inherently static anatomical and/or biochemical structure (e.g., cellulose and lignin)34,35. The rigidity provides structural stability but limits flexibility in response to changes in soil water content36.
As plants diversified into drier soils, they evolved more flexible and sensitive water regulation mechanisms35,37. These adaptations involved two alternative cellular modifications: (1) smaller cells with thicker walls, which have reduced cell volume, resulting in higher solute accumulation per unit cell volume, and thus higher osmotic concentration11, and which would have greater reduction in osmotic potentials for a given cell water loss, while the thicker walls provide essential structural reinforcement10; (2) thinner and more elastic cell walls, contributing to increased cell water storage, and improved maintenance of hydration even after stomatal closure triggered by drought stress1,7,29,36,38. The latter is well exemplified by succulents and resurrection plants23,39,40,41. Empirical evidence suggests that the enhanced cell wall elasticity observed in these species is attributable to abundant pectin23,39, representing a cost-effective matrix with composition that can be adjusted according to environmental conditions22,42. Under water stress, sunflower (Helianthus annuus) exhibits a 60% increase in pectin concentration compared to well-watered controls19. Thus, increasing pectin concentration may serve as an evolutionary solution to increase physiological flexibility with relatively low biomass investment in water-stressed environments. Yet, an important but unresolved question is whether diversification in cell wall composition, such as pectin concentration alters the mechanisms that coordinate physiological and structural traits.
We develop a comprehensive framework integrating leaf water relations, cell wall traits, and leaf morphology to examine the impact of cell wall composition on leaf water relations across species and biomes, thereby clarifying the physiological mechanisms linking cell wall design and soil water availability. We hypothesize that cell wall composition, and particularly pectin concentration, enhances drought tolerance, and that these effects are modulated by water availability. To test this hypothesis, we measure 26 traits using standardized methods across 69 tree species (57 genera, 33 families) in four spatially independent sub-tropical forests. These forests–two wet and two dry–were selected for comparable climate but contrasting edaphic hydrology (Supplementary Fig. 1, Supplementary Table 1). Wet forests develop on deeply weathered lateritic soils (> 200 cm) over siliceous rock, characterized by high water retention capacity43,44. Conversely, dry forests occur on karst landscapes with exposed limestone bedrock and shallow calcareous soils (<45 cm), promoting rapid water drainage despite equivalent precipitation inputs45. This hydrological divergence is corroborated by higher leaf predawn water potential (ψpd) in wet forests (mean ± standard error = −0.05 ± 0.01 MPa) versus dry forests (−0.13 ± 0.02 MPa) during sampling in the growing season.
Based on our unique datasets, we specifically predict that: (ⅰ) trees in dry forests exhibit more negative ψtlp and πo, and higher εsym than those in wet forests; (ⅱ) dry and wet forests differ primarily in cell wall composition, particularly pectin concentration, rather than in relatively static anatomical structure; (iii) the coordination between cell wall composition, anatomical traits and P-V traits are environment-dependent. Specifically, we predict that in wet forests, εsym, πo, and ψtlp are associated with anatomical traits, while in dry forests, these physiological traits are associated with pectin concentration (Supplementary Fig. 2).
Results
Weakened coordination of osmotic and elastic function in dry forests
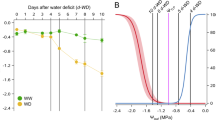
Across the 69 species examined, the turgor loss point (ψtlp), osmotic potential at full turgor (πo) and elastic modulus (ɛsym) showed 2.9-fold, 3.2-fold, and 4.4-fold variation, respectively (Supplementary Table 2). As expected, dry forest species had lower ψtlp than wet forest species (Fig. 1b; Cohen’s d = −0.59, p < 0.05, linear mixed-effects model (LMMs)), while other P-V traits, including ɛsym, πo, apoplastic water fraction (fapo), and capacitance (C*ft), exhibited no significant difference between two forest types (Fig. 1c; Supplementary Table 3). When considering the three species that occur in both forest types, the shift in ψtlp between forest types was stronger (Cohen’s d = 1.49, p = 0.03) compared to other P-V traits (Cohen’s d = 0.11–0.99, p = 0.15–0.88) (Supplementary Fig. 3a-e).
a the pressure-volume (P-V) traits for 35 tree species in wet forests are comparable to 34 tree species in dry forests. Each thin line represents the P-V curve per species plotting the inverse leaf potential (1/ψleaf) against relative water content (RWC). b,c trees in the wet and dry forests differ only in turgor loss point (ψtlp) (violin plot, Cohen’s d = −0.59, p = 0.02, LMMs, n = 69), while having similarity in other P-V traits like elastic modulus (ɛsym) (Cohen’s d = 0.53, p = 0.37). These results were derived using linear mixed model with forest type as a fixed effect and species as a random effect. The significance of the forest type effect was evaluated using a two-sided t-test (Methods). Solid points and lines represent marginal means and their 95% confidence intervals, respectively. d osmotic potential at full turgor (πo) was positively related with ψtlp between dry and wet forests. e,f unlike species from wet forests, those from dry forests showed a weaker coordination between wall elasticity (εsym) and osmotic-dependent traits (πo and ψtlp). Separate linear regression models were fitted for wet and dry forest species, respectively (d–f). The resulting regression lines are displayed as solid curves, accompanied by their 95% confidence intervals (shaded areas). Source data are available as a Source Data file.
The correlation between ψtlp and πo was consistently strong in both wet and dry forests (Fig. 1d; standardized regression coefficients β = 0.98 and 0.92, respectively, p < 0.001, linear model (LM)). However, the correlation between πo and εsym was significantly weaker in dry forests compared to wet forests (Fig. 1e; β = -0.57 and -0.85, respectively, p < 0.001; Fisher’s z = -2.5, p = 0.01). Notably, dry forests had a more limited range in πo (Fig. 1e); yet, wet forests maintained a significantly stronger πo versus εsym correlation even when the πo range was standardized to that of dry forests (r = -0.80 and -0.57, respectively, p = 0.04; two-sample bootstrap permutation test; Supplementary Fig. 4). After controlling for πo via partial correlation analysis, the initially non-significant or negative εsym-ψtlp relationships (Fig. 1f) became significantly positive in both forest types (partial r = 0.43 for wet forests, 0.55 for dry forests; p < 0.01).
Enriched cell wall pectin and calcium in dry forests
Wet and dry forests showed comparable variation in six anatomical traits, including cell wall thickness (Tcw), sizes of spongy and palisade cells (Sspongy and Spalisade), thickness of spongy and palisade tissue (Tspongy and Tpalisade), and airspace fraction in the leaf cross-section (AirF), whereas the palisade-to-spongy tissue thickness ratio (PSR) varied significantly more (11-fold) within wet forests (Fig. 2a–d; Fig. 5; Supplementary Table 2). Notably, in dry forests, pectin and Ca2+concentrations showed both significantly higher means (Cohen’s d = 1.66 and 1.46, respectively, p < 0.001, LMMs; Supplementary Table 3) and greater variation than in wet forests (Fig. 2f-g; Supplementary Fig. 4, Supplementary Table 2). Analyses of the three species that occurred in both forest types also revealed a markedly higher (approximately 2.4-fold) pectin concentrations in dry than wet forests (Supplementary Fig. 3).
a–c wet and dry forest species exhibited similar ranges of variation in most leaf anatomical traits and cell wall components, including the palisade cell size (Spalisade), spongy cell size (Sspongy), cell wall thickness (Tcw), and other seven functional traits (Supplementary Table 2). d in contrast, the palisade-to-spongy thickness ratio (PSR) showed greater variability in wet forests. We measured seven biochemical components of cell wall in leaves, with the key five components shown in panels e (referencing to Taiz et al. (2015)). We found that the cell wall pectin (f) and Ca2+ concentrations (g) showed significant differences between the two forest types, with greater variance in pectin and Ca2+ concentrations in dry forests. The p-value represents the significance level obtained from two-sided Levene’s test, confirming significant differences in variance (see Supplementary Table 2 for F-statistic and exact p value). Source data are provided as a Source Data file.
Distinct leaf drought tolerance strategies in dry versus wet forests
Drought tolerance strategies differed significantly between wet and dry forests. In wet forests, the P-V traits were strongly associated with PSR and spongy cell size (Sspongy) (Fig. 3a, PC1, 44%; Supplementary Table 4), and 86% of the explained variation in leaf drought tolerance was attributed to anatomical traits (Fig. 3c). By contrast, among species in dry forests, P-V traits were coupled with cell wall composition (Fig. 3b, PC1 35%; Supplementary Table 4, Supplementary Fig. 6), with cell wall pectin and Ca2+ concentrations explaining 78% of the variation in P-V traits (Fig. 3c).
a in wet forests, five pressure–volume (P-V) traits (gray) showed a decoupling from cell wall composition (orange) but a strong coupling with leaf anatomical traits (green). b in contrast, leaf drought tolerance in dry forests was associated with three cell wall components and was less influenced by leaf anatomical traits. Arrow length represents trait loadings (Supplementary Table 5). Gray arrows represent P-V traits: turgor loss point (ψtlp), osmotic potential at full turgor (πo), modulus of elasticity (ɛsym), capacitance (C*ft), and apoplastic fraction (fapo). Orange arrows are cell wall components: cellulose (Cellulosecw), protopectin (proPectincw), and soluble pectin (solPectincw). Green arrows represent the ratio of palisade to spongy thickness (PSR), cell wall thickness (Tcw), and spongy cell size (Sspongy). c, variation of P-V traits in wet forests was primarily explained by leaf anatomical traits, while in dry forests, drought tolerance was primarily explained by cell wall components, such as proPectincw, solPectincw, and Ca2+ (Supplementary Fig. 7-8). Values represent the relative importance of each explanatory variable. Tpalisade, palisade tissue thickness; Tspongy, spongy tissue thickness; Spalisade, palisade cell size; AirF, airspace fraction in leaf cross-section area; hemiCellulosecw, the hemicellulose content. Source data are provided as a Source Data file.
The relationships between pectin concentration and P-V traits were environmentally dependent. In the wet forests, species with higher PSR have higher εsym (Fig. 4b; β = 0.52, R² = 0.29, p < 0.001) and lower ψtlp (Fig. 4c; β = -0.70, R² = -0.49, p < 0.001). In dry forests, species with greater soluble pectin concentrations (solPectincw) tend to have lower εsym (Fig. 4d; β = -0.70, R² = 0.28, p < 0.001), while species with greater protopectin concentrations (proPectincw) had higher ψtlp (Fig. 4e; β = 0.51, R² = 0.24, p < 0.01). These relationships were consistent when using phylogenetic mixed-effect model and phylogenetic independent contrasts (Supplementary Tables 6–8).
a,b modulus of elasticity (ɛsym) and turgor loss point (-ψtlp) in wet forests are positively related to PSR (R2 = 0.29 and 0.49, respectively, linear regression), suggesting leaves tend to have stable tissue organization and improve leaf physical rigidity, thus enhance the drought tolerance. c pectin content is negatively correlated with the family divergence time (p = 0.001, linear regression; n = 69), and displays a strong phylogenetic signal (Pagel’s λ = 0.72, p = 5.9e-05, Supplementary Table 5; Supplementary Fig. 6) d,e the ɛsym and -ψtlp in dry forests are negatively related to soluble pectin and protopectin concentrations (R2 = 0.28 and 0.24, respectively, linear regression), indicating that dry forest species tend to have flexible pectin-enriched leaves to cope with drought. All lines represent regression predictions with shaded 95% confidence intervals. Source data are provided as a Source Data file.
Diverse drought tolerance strategy in dry forests linked to cell wall pectin
Underlying the observed distinct drought tolerance strategies, a divergence in optimal leaf hydraulic design was identified (Fig. 5). In wet forests, PSR (a proxy for anatomical organization) was strongly associated with πo and ɛsym. However, in dry forests, this integration between anatomical traits and water relations differed. Thus, among dry forest species, the diversification in pectin concentration added an additional layer of complexity to the evolution of drought tolerance, as evidenced by a weaker linkage between πo and ɛsym and decoupling ɛsym and ψtlp, thereby promoting more diverse tolerance strategies.
Our path analysis identified the wet stable tissues integration versus dry flexible cell wall regulation in leaf drought tolerance. Wet forest species adapt to drought via by reinforcing the integrated relationship between tissue organization and P-V traits, whereas dry forest species employ flexible adjustments in soluble pectin and protopectin concentrations to alter leaf water relations. The piecewise structural equation model (left, Fisher’s C = 0.6, P = 0.97, and AIC = 16.5) explains 39% of the variance in ɛsym and 45% of the variance in πo in wet forests, while in dry forests, the model (right, Fisher’s C = 0.8, P = 0.93, and AIC = 12.8) explains the variance of 30% in ɛsym and of 24% in πo. A significant relationship between ψtlp and εsym was observed in wet forests but not in dry forests. The standardized path coefficient and associated p value (* p < 0.05, ** p < 0.01, *** p < 0.001) are given alongside arrows. Black and red arrows indicate positive and negative relationships, respectively, with arrow thickness proportional to the magnitude of the path coefficient; Double-headed arrows indicate strong associations between traits that are not directly linked; Dashed lines and R2 represent reduced major axis regression (SMA) fits between πo and ψtlp. Source data are provided as a Source Data file.
Discussion
Our findings suggest a fundamental role of cell wall pectin in influencing leaf water relations and drought tolerance. We propose that higher pectin concentration in cell walls may have acted as an important evolutionary mechanism for the adaptation of woody plants in dry habitats. Specifically, pectin-rich cell wall structures finely regulate leaf water relations through flexible adjustments in osmotic pressure and tissue elasticity, resulting from their high water-chelating and ion-regulating capacity.
Divergent regulatory mechanisms for leaf water relations
We show that the underlying mechanisms governing variation in drought tolerance traits, represented by P-V traits, differed strongly between wet and dry forest species (Fig. 1b-c; Supplementary Fig. 4, Supplementary Table 2). Indeed, consistent with previous studies, species in the dry forests had on average lower ψtlp than those from the wet forests, likely due to geological or climatic differences14,15,16. Yet, the contrasting associations of leaf drought tolerance with anatomical structure and cell wall composition (e.g., pectin) among species from the different forest types indicates convergence in leaf drought tolerance mediated by different mechanisms in the two forest types. These patterns highlight an important association of tissue anatomical organization2, or fine-scaled cellular structures (i.e. cell size, cell wall thickness, and composition)5,12 with leaf drought tolerance. Anatomical structure and biochemical composition may indirectly or directly impact leaf drought tolerance especially given their role in influencing photosynthetic physiology12,34,46. Additionally, in dry forests, we found a decoupling of ɛsym and ψtlp, which indicates the importance in drought tolerance of multiple strategies emerging from combinations of osmotic and elastic functions, leading to a predictable coordination of these physiological traits. The role of cell wall composition in regulating leaf water processes has been often overlooked in previous studies.
The key role of flexible pectin in cell walls in dry forests
Many dry forest species exhibited higher and more variable concentrations of pectin and Ca²⁺ in cell walls than wet forest species (Fig. 2f, g, Supplementary Table 3). This elevated pectin concentration may enhance drought responsiveness through chemically accessible domains that facilitate rapid cross-linking with signaling molecules24. Furthermore, the strong adjustment of pectin concentration for species of wet and dry forest through plasticity and/or ecotypic differentiation (2.4-fold increase in dry forests; Supplementary Fig. 3) supports its role as a dynamically responsive matrix. Our results thus show a compelling link between pectin and the diversification of trees with dry habitats. Notably, pectin can be classified into protopectin and soluble pectin. Protopectin, a structural pectin in cell walls, has a stronger association with ψtlp and πo in dry forest species than cell size or wall thickness (Figs. 3–5, Supplementary Tables 7-8). Protopectin would exert its influence on these physiological traits through several mechanisms:(1) enhanced apoplastic cation exchange capacity (CEC) via preferential Ca²⁺ chelation, establishing a dynamic ionic reservoir that enables rapid osmotic adjustment during stress3,47,48; (2) increased water retention capacity through hydrogel formation in the cell wall matrix, where bound water molecules create a hydration buffer23; and (3) activated aquaporin activity via pectin-Ca²⁺ signaling pathways to optimize membrane water permeability49,50. These properties would enable species with high pectin concentrations to reduce their reliance on intracellular solute accumulation, corresponding to less negative πo and ψtlp in these species. These findings suggest a ‘compensatory’ trade-off between physiological traits and biochemical composition within leaves51,52.
Furthermore, non-structural soluble pectin tends to increase wall elasticity (decreases ɛsym) (Figs. 2–4) by chelating and displacing Ca²⁺ from existing pectin-Ca²⁺ complexes53. This displacement weakens the load-bearing capacity of these complexes within the cellulose network, resulting in wall relaxation and enhanced flexibility, thereby facilitating water storage and turgor maintenance under stress. Succulents from phylogenetically diverse clades (e.g., Caryophyllales, Crassulaceae) and resurrection plants, which have leaves rich in soluble pectin, are good examples of this23,39.
Overall, a pectin-enriched cell wall allows quick adjustments in cell wall elasticity and osmotic pressure in sub-tropical dry forests. Thus, this “cell wall biochemistry” mechanism is key for understanding the leaf water processes in response to frequently drying soils.
Stable leaf tissue coordinates with drought tolerance under stable water supplies
Unlike in dry forests, leaf drought tolerance in wet forests is strongly associated with tissue organization, particularly the palisade to spongy tissue thickness ratio (PSR) (Figs. 3–4, Supplementary Table 2). A higher PSR correlated with more negative πo and ψtlp values, as well as increased elastic modulus (Figs. 4–5). This is likely due to the denser and more rigid palisade cells restricting cell volume flexibility, consequently increasing elastic modulus, and enabling species with a low πo and ψtlp to maintain cell volume with dehydration33,54. PSR is also closely linked with leaf dry mass content (LDMC) (Supplementary Fig. 11), which would contribute to a prolonged leaf lifespan for long-term revenue6,55. The high light competition and high water availability in these forests favors a high PSR in canopy species, maximizing light capture while minimizing outside-xylem hydraulic resistance2,56,57. The resulting combination of low ψtlp and high wall rigidity enhances long-distance water transport, facilitates cell water maintenance, and prevents tissue shrinkage6,58,59,60, providing a competitive advantage for tropical trees grown in wet forests.
The optimal designs of cell wall depend on the environments
Our study shows that pectin-mediated mechanisms may decouple leaf anatomical organization from leaf water relations and drought tolerance, enabling adaptation to water-deficit habitats. ψtlp is related to πo in both forest types, but the relationships among the modulus of elasticity, anatomical structure, and cell wall composition differ substantially. Specifically, we found a weaker linkage between πo and ɛsym and decoupling of ɛsym and ψtlp in dry forests (Figs. 1, 5), suggesting different leaf drought tolerance strategies in two forest types.
The distinct drought tolerance strategies likely result from an intricate interplay of construction costs, soil chemistry and water availability, and physiological requirements. Pectin, a semi-structural polysaccharide, is susceptible to degradation when exposed to sufficient water27. However, its relatively low construction cost (1.09 g glucose per gram of pectin) allows for flexibility and rapid responses to water-stressed environments24, contrasting with the higher costs of lignin (2.12 g glucose) and cellulose (1.22 g glucose)42. In alkaline soils, the combination of abundant Ca2+ and limited water optimizes the functionality of pectin-Ca2+ network61. The nearly 10-fold higher Ca²⁺ concentration in dry forest soils compared to wet forest soils, favors the formation of more extensive and diverse pectin-Ca²⁺ networks (Supplementary Fig. 2). Consequently, in high-Ca arid habitats, pectin, with low construction cost, high plasticity and flexibility would effectively regulate wall elasticity and support variation in osmotic potential, enhancing drought adaptation in xerophytes where rapid responses to fluctuating resource availability are rewarded.
Evolutionary and ecological implications of the biochemical composition of the cell wall
We propose that pectin enrichment constitutes a key biochemical adaptation enabling woody plants to thrive in dry forests. In mesic environments, stable leaf structure, with coordinated cell wall elasticity and osmotic potential, provides an adaptive advantage by optimizing light capture and water use efficiency. Under frequent water stress, flexible pectin-mediated cell walls enhance drought tolerance by modulating wall elasticity and osmotic pressure, decoupling structural constraints from cellular physiological processes. This biochemical flexibility may, in turn, facilitate functional diversification of drought tolerance across aridity gradients. Moderate phylogenetic conservation of pectin abundance (Fig. 4c) indicates that its evolutionary advantage may contribute to its persistence in dry habitats, or during radiations of species between wet and dry environments62.
Recent evidence confirms wide variation in pectin abundance across diverse plant lineages (e.g., mosses, ferns, gymnosperms, and angiosperms) and its correlation with photosynthetic capacity in non-crop angiosperms46. While pectin composition exhibits lineage-specific diversification35,63, our observations of strong interspecific divergences among angiosperms suggests that pectin abundance may represent a crucial functional dimension.
We identified two contrasting drought tolerance strategies: a ‘stable leaf tissue’ strategy in wet forest species, characterized by coordinated tissue organization, and leaf drought tolerance; and a ‘flexible cell wall’ strategy in dry forest species where pectin-rich leaves enable flexible shifts to fluctuating water availability via fine-tuning leaf physiology. These results indicate cell wall biochemistry as a key mediator of habitat specialization and functional diversification of leaf drought tolerance. Our findings also may inform the screening of drought-tolerant tree species for forestry and indicate the importance of linking genes to cell wall phenotypes in molecular breeding.
Overall, this study elucidated distinct mechanisms underlying drought tolerance across wet and dry forests, linking cell wall composition and anatomical structure with contrasting forest types and evolutionary trajectories. Our studies represent a crucial step towards directly linking specific cell wall composition to species’ drought tolerance. Future work should examine the generality of our findings among ecosystems globally, within and among lineages, and within species.
Methods
Study Sites and Species Selection
To examine the influence of cell wall structure on leaf water relations and its relationships with soil water availability, we compared two ecologically distinct wet (W1, W2) and two dry (D1, D2) subtropical forests across South China (21°37′N–26°23′N). These forests represent independent ecosystems, separated by hundreds of kilometers, sharing comparable climate conditions (mean annual precipitation, 1397–1532 mm; mean annual temperature, 17 °C–21 °C) but contrasting geological substrates and water availability (Supplementary Fig. 1, Supplementary Table 1). Two wet forests developed on deeply weathered lateritic soils, whereas two dry forests occupied limestone bedrock with shallow calcareous soils of poor water retention. This edaphic divergence corresponded with distinct ecological traits: wet forest trees were generally taller, and produced leaves with higher carbon but lower Ca concentrations; dry forests exhibited higher soil pH and Ca availability, though soil nitrogen and phosphorus levels remained similar between forest types.
Across wet and dry forests, we studied a total of 69 tree species from 57 genera and 33 families, covering key clades of common species in southern China; the sample included 34 species from dry forests, 35 from wet forests, with three overlapping species (Supplementary Figs. 3, 9); these species were indigenous and dominant in the canopy and sub-canopy layers in the natural communities and vary in their drought tolerance within and between communities45. Data collection within each forest was completed within two weeks during the peak growing season (July and August) between 2019 and 2021 to minimize temporal variation between physiological traits and cell wall composition, although the two-week window varied slightly among forests.
Leaf sampling and preparation
For each species, we selected 5–7 individuals and collected mature, sunlit leaves from their canopies using a combination of crane, climbing, and fishing rod sampling methods. This allowed us to obtain mean trait values for each species. From each selected individual, we collected one leafy branch after sunset to minimize transpiration-induced water potential fluctuations. From each harvested branch, we selected one fully expanded, undamaged leaf for P-V curve measurements. This sampling strategy, common in comparative physiology9, allowed us to balance the need to resolve statistical differences across species with the time-consuming nature of P-V curve measurements. The remaining leaves (approximately 15–30) on each branch were used for morphological/structural trait measurements. Before measurement, branches were re-cut under water and rehydrated overnight. This standard pre-treatment made all measurements comparable at forest sites with different water availability64. Additionally, we collected leaves at predawn (4:30–5:30 am) for three analyses: i) predawn water potential (n = two leaves of five individuals per species); ii) cell wall composition (n = 3 individuals per species, immediately placed in a cooler to minimize the effect of leaf starch content on wall composition, Carriquí et al. 2020); iii) and leaf anatomical preparation, stored in FAA solution (37% formaldehyde-glacial acidic acid, 95% ethanol in deionized water) (n = 5 individuals per species).
Trait measurements
Pressure-volume curves describe the interplay between leaf water potential (including osmotic and pressure potential) and cell water content and volume, serving as a crucial physiological method for assessing leaf drought tolerance. With the bench drying method65, we measured five critical P-V curve parameters for each species, using 5–7 leaves per species. These parameters were: turgor loss point (ψtlp), osmotic potential at full turgor (πo), bulk modulus of elasticity (ɛsym), leaf mass-specific capacitance at full turgor (C*ft), and apoplastic fraction (fapo). Water potential was measured in situ using a portable pressure chamber (model 1050D-EXP, PMS Instrument Company). To quantify cell wall composition, we first extracted cell wall dry mass (alcohol-insoluble residue, AIR) following the general protocol outlined by ref. 19, followed by trifluoroacetic acid hydrolysis to obtain a pellet and supernatant fractions for the quantification of cellulose and hemicellulose, respectively, using the anthrone-sulfuric acid colorimetric method66. Lignin was quantified using the acetyl bromide method67. We employed the method provided by ref. 68 to extract and determine soluble pectin and protopectin of cell wall. We also measured cell wall Ca and Mg using inductively coupled plasma atomic emission spectrometry (ICP-AES, Spectro, Germany), as these cations influence cell wall elasticity, cell water exchange and osmosis3,47. Leaf anatomical traits were obtained through photographic measurement of tissue sections69. Transverse sections were made for one leaf from each of 5 individuals (Supplementary Fig. 12). Seven key leaf anatomical traits measured were: palisade and spongy mesophyll tissue thickness (Tpalisade and Tspongy), ratio of palisade to spongy tissue thickness (PSR), fraction of intercellular air spaces (AirF), the volume of palisade and spongy mesophyll cell (approximated as cylinder and sphere, respectively) (Spalisade and Sspongy), and the averaged cell wall thickness of palisade to spongy mesophyll (Tcw). Specific measurement protocols for P-V traits, cell wall composition, and anatomical traits are outlined in Supplementary Method S1.
Species and phylogeny
We constructed phylogenetic trees of our species using the R package ‘V.PHYLOMAKER2’, with the GBOTB phylogeny as the backbone70. Species names in the phylogenetic tree were corrected according to The Plant List (http://www.theplantlist.org/). We calculated parametric Blomberg’s K71 using the ‘Picante’ package in R language and evaluated the strength of the phylogenetic signal for each trait72; a larger K value indicates a greater phylogenetic conservatism for the given trait. We also performed the Pagel’s λ test73 to detect phylogenetic signals of each trait. For the three species that occurred in both forest types, we used mean trait values to calculate total phylogenetic signals. We calculated phylogenetically independent contrast74 using the ‘ape’ package to control for the influence of common ancestry on trait relationships75. Following previous analyses, we defined the divergence time of a plant family using the earliest diverging genus within that family76,77.
Data analyses
Across wet and dry forests, we examined the equality of variance in all leaf traits using Levene′s test and differences in mean leaf traits using linear mixed effects model (lmer function in R package lme4)78. We treated forest type as the fixed effect and species as a random effect to account for non-independence of data points due to three shared species (n = 35 and 34 for wet and dry forests, respectively). We examined the normality of residuals using the Shapiro-Wilk test. Data were log-transformed as necessary to meet this assumption; Importantly, after applying log-transformations where necessary, only three out of 26 traits (apoplastic fraction, Airspace fraction and leaf thickness) still failed to fully meet the normality assumption. We therefore performed Mann-Whitney U test for these traits. To determine whether forest type shifts consistently influenced leaf traits within and across species, we conducted paired t-test for each trait-species combination in three species found in both forest types. We reported effect sizes as Cohen’s d for LMMs and t-test, and Cliff’s σ for Mann-Whitney U test79,80.
To examine relationships among P-V traits in wet and dry forests, we performed linear regression and partial correlation analyses. Specifically, we used partial correlation to assess the relationship between ψtlp and ɛsym, controlling for πo. This isolated the direct relationship between ψtlp and ɛsym, independent of πo. To test for significant differences in the πo-ɛsym correlation between forest types, we compared their Pearson correlation coefficients using Fisher’s z81. Finally, to determine if a limited πo range weakened the πo-ɛsym correlation in dry forests, we performed a bootstrap analysis. We iteratively (1000 iterations) subsampled the wet forest data (n = 10 species per iteration) to match the πo range observed in dry forests, calculating the Pearson coefficient for πo-ɛsym in each subsample. We then compared the resulting distribution of coefficients to the observed dry forest correlation using percentile-based confidence intervals and permutation testing.
We conducted principal component analysis (PCA) to investigate trait coordination and identify the key anatomical and cell wall biochemical traits influencing leaf water relations (Fig. 2). Prior to PCA, all data were standardized using a zero-mean approach to ensure each variable has a unit variance82. To enhance the interpretability of our PCA results, we first retained traits exhibiting significant Pearson correlations between P-V traits and anatomical/biochemical traits in both wet and dry forest (Supplementary Tables 6-7). These traits included PSR, spongy cell size, spongy and palisade tissue thickness, pectin and cell wall Ca. We then used the stepwise approach described by Weigelt et al. (2023)83 to incorporate additional traits, such as cell wall thickness and cellulose, which have direct biological linkage with leaf water relations12,13. Finally, we simplified the analysis by removing variables with high collinearity (e.g., PSR with spongy and palisade tissue thickness) and those with non-significant loadings on the first two principal components using the ‘PCAtest’ package84.
We used multivariate linear models to assess the effects of anatomical traits and cell wall composition on five P-V traits. We first applied a backward stepwise regression (dredge function in ‘MuMIn’ package)85 to screen for the best set of predictors for each P-V trait based on minimizing the corrected Akaike Information Criterion (AICc). Next, we pooled these predictors together and assessed their individual effects on all P-V traits by performing hierarchical partitioning (HP) using the ‘rdacca.hp’ package86. Following Lai et al. (2022), we addressed multicollinearity by removing predictors with negative contributions. The relative importance of each predictor as the ratio of its explained variation to the total explained variation, expressed as a percentage. Finally, to improve visualization, we focused on those predictors with a relative importance of at least 5%. To cross-validate the HP results, random forest analyses with permutation tests, implemented using the ‘rfPermute’ package87, were used to assess the relative importance of anatomical traits and cell wall composition in determining specific P-V traits. We grew 1000 trees and conducted 1000 permutations for each random forest. Predictor importance was quantified as the percentage increase in mean squared error (%IncMSE) after permuting each predictor.
Building on the random forest results, which highlighted the importance of PSR and pectin, we used linear regression to specifically analyze their relationships with three P-V traits (ɛsym, πo, and ψtlp) (Supplementary Fig. 7). We also implemented phylogenetic mixed effect models with the phylogenetic relatedness matrix as a random effect, to further assessing the same relationships using the relmatLmer function in ‘lme4qtl’ package88. Given the strong phylogenetic signal and high variation observed in pectin content, we tested the relationship between pectin and divergence time of major taxa using linear regression analysis. All LM and LMMs standardized regression coefficients (β), confidential interval (CI) and p value were reported.
We employed piecewise structural equation modeling (SEM), combined with multivariate linear regression, to explore the relationships among anatomical, biochemical, and P-V traits in wet and dry forests separately, using the ‘piecewiseSEM’ package89. A basic model was constructed with all possible paths (Supplementary Fig. 2). Following Bartlett et al. (2012), we allowed ɛsym and πo to be interdependent in this model. Recognizing that including both πo as a predictor of the ψtlp can lead to inflated path coefficients, we chose to fit this relationship using standardized major axis (SMA) regression instead of including a direct path in the SEM. We used all-subsets regression (dredge function in ‘MuMIn’ package) to both select relevant traits guided by lowest AICc and remove non-significant paths. This iterative procedure continued until a parsimonious model with acceptable was achieved. Model fit was evaluated using Fisher’C, P value and Akaike Information Criterion (AIC)89. We reported the standardized path coefficients and R2 for the response variables (ɛsym and πo).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The datasets analyzed in this study are archived on figshare (https://doi.org/10.6084/m9.figshare.30000388). Source data are provided with this paper.
Code availability
R scripts are deposited together with associated data on figshare (https://doi.org/10.6084/m9.figshare.30000388).
References
Nadal, M. et al. Incorporating pressure–volume traits into the leaf economics spectrum. Ecol. Lett. 26, 549–562 (2023).
Sack, L. & Frole, K. Leaf structural diversity is related to hydraulic capacity in tropical rain forest trees. Ecology 87, 483–491 (2006).
Taiz L., Zeiger E., Møller I. M., & Murphy A. Plant Physiology and Development. (Sinauer Associates Incorporated, 2015).
Preston, R. D. Polysaccharide conformation and cell wall function. Annu. Rev. Plant Physiol. 30, 55–78 (1979).
Tyree M. T. & Jarvis P. G. Water in tissues and cells. In: Physiological Plant Ecology II: Water Relations and Carbon Assimilation (eds Lange O. L., Nobel P. S., Osmond C. B., Ziegler H.). (Springer, 1982).
Niinemets, Ü Global-scale climatic controls of leaf dry mass per area, density, and thickness in trees and shrubs. Ecology 82, 453–469 (2001).
Nadal, M., Flexas, J. & Gulias, J. Possible link between photosynthesis and leaf modulus of elasticity among vascular plants: a new player in leaf traits relationships? Ecol. Lett. 21, 1372–1379 (2018).
Wright, I. J. et al. Global climatic drivers of leaf size. Science 357, 917–921 (2017).
Baird, A. S. et al. Developmental and biophysical determinants of grass leaf size worldwide. Nature 592, 242–247 (2021).
Peguero-Pina, J. J., Sancho-Knapik, D. & Gil-Pelegrín, E. Ancient cell structural traits and photosynthesis in today’s environment. J. Exp. Bot. 68, 1389–1392 (2017).
Cutler, J. M., Rains, D. W. & Loomis, R. S. The importance of cell size in the water relations of plants. Physiol. Plant. 40, 255–260 (1977).
Flexas, J. et al. Cell wall thickness and composition are involved in photosynthetic limitation. J. Exp. Bot. 72, 3971–3986 (2021).
Cosgrove, D. J. Building an extensible cell wall. Plant Physiol. 189, 1246–1277 (2022).
Bartlett, M. K., Scoffoni, C. & Sack, L. The determinants of leaf turgor loss point and prediction of drought tolerance of species and biomes: a global meta-analysis. Ecol. Lett. 15, 393–405 (2012).
Maréchaux, I. et al. Drought tolerance as predicted by leaf water potential at turgor loss point varies strongly across species within an Amazonian forest. Funct. Ecol. 29, 1268–1277 (2015).
Zhu, S. et al. Leaf turgor loss point is correlated with drought tolerance and leaf carbon economics traits. Tree Physiol. 38, 658–663 (2018).
Cheung, Y. N. S., Tyree, M. T. & Dainty, J. Water relations parameters on single leaves obtained in a pressure bomb and some ecological interpretations. Can. J. Bot. 53, 1342–1346 (1975).
Coen, E. & Cosgrove, D. J. The mechanics of plant morphogenesis. Science 379, eade8055 (2023).
Roig-Oliver, M., Bresta, P., Nikolopoulos, D., Bota, J. & Flexas, J. Dynamic changes in cell wall composition of mature sunflower leaves under distinct water regimes affect photosynthesis. J. Exp. Bot. 72, 7863–7875 (2021).
Zhang, Y. et al. Molecular insights into the complex mechanics of plant epidermal cell walls. Science 372, 706–711 (2021).
Gall, H. L. et al. Cell wall metabolism in response to abiotic stress. Plants 4, 112–166 (2015).
Du, J., Anderson, C. T. & Xiao, C. Dynamics of pectic homogalacturonan in cellular morphogenesis and adhesion, wall integrity sensing and plant development. Nat. Plants 8, 332–340 (2022).
Moore, J. P. et al. Response of the leaf cell wall to desiccation in the resurrection plant Myrothamnus flabellifolius. Plant Physiol. 141, 651–662 (2006).
Vaahtera, L., Schulz, J. & Hamann, T. Cell wall integrity maintenance during plant development and interaction with the environment. Nat. Plants 5, 924–932 (2019).
Daher, F. B. & Braybrook, S. A. How to let go: pectin and plant cell adhesion. Front. Plant Sci. 6, 523 (2015).
Jarvis, M., Briggs, S. & Knox, J. Intercellular adhesion and cell separation in plants. Plant Cell Environ. 26, 977–989 (2003).
Tibbits, C. W., MacDougall, A. J. & Ring, S. G. Calcium binding and swelling behaviour of a high methoxyl pectin gel. Carbohydr. Res. 310, 101–107 (1998).
Mitchell, P. J., Veneklaas, E. J., Lambers, H. & Burgess, S. S. O. Leaf water relations during summer water deficit: differential responses in turgor maintenance and variation in leaf structure among different plant communities in south-western Australia. Plant Cell Environ. 31, 1791–1802 (2008).
Sack, L., Cowan, P., Jaikumar, N. & Holbrook, N. The ‘hydrology’of leaves: co-ordination of structure and function in temperate woody species. Plant Cell Environ. 26, 1343–1356 (2003).
John, G. P., Henry, C. & Sack, L. Leaf rehydration capacity: associations with other indices of drought tolerance and environment. Plant Cell Environ. 41, 2638–2653 (2018).
Fu, P. et al. The contrasting leaf functional traits between a karst forest and a nearby non-karst forest in south-west China. Funct. Plant Biol. 46, 907–915 (2019).
Xing, D. et al. Translocation and utilization mechanisms of leaf intracellular water in karst plants Orychophragmus violaceus (L.) O. E. Schulz and Brassica napus L. Horticulturae 8, 1082 (2022).
Binks, O. et al. Plasticity in leaf-level water relations of tropical rainforest trees in response to experimental drought. N. Phytol. 211, 477–488 (2016).
Carriquí, M. et al. Cell wall composition strongly influences mesophyll conductance in gymnosperms. Plant J. 103, 1372–1385 (2020).
Sarkar, P., Bosneaga, E. & Auer, M. Plant cell walls throughout evolution: towards a molecular understanding of their design principles. J. Exp. Bot. 60, 3615–3635 (2009).
Lambers H. & Oliveira R. S. Plant water relations. In: Plant Physiological Ecology. (Springer International Publishing, 2019).
Lo Gullo, M. A. & Salleo, S. Different strategies of drought resistance in three Mediterranean sclerophyllous trees growing in the same environmental conditions. N. Phytol. 108, 267–276 (1988).
Xiong, D. & Nadal, M. Linking water relations and hydraulics with photosynthesis. Plant J. 101, 800–815 (2020).
Fradera-Soler, M. et al. Are cell wall traits a component of the succulent syndrome? Front. Plant Sci. 13, 1043429 (2022).
Gotsch, S. G. et al. Trade-offs between succulent and non-succulent epiphytes underlie variation in drought tolerance and avoidance. Oecologia 198, 645–661 (2022).
Ogburn R. M. & Edwards E. J. The ecological water-use strategies of succulent plants. In: Advances in Botanical Research (eds Kader J. C., Delseny M.). (Academic Press, 2010).
Poorter H. Construction costs and payback time of biomass: a whole plant perspective. 111-127 (Utrecht University, 1994).
Chen, Y. et al. Water-use advantage for lianas over trees in tropical seasonal forests. N. Phytol. 205, 128–136 (2015).
Jiang, P. et al. Linking reliance on deep soil water to resource economy strategies and abundance among coexisting understorey shrub species in subtropical pine plantations. N. Phytol. 225, 222–233 (2020).
Cao, K., Fu, P., Chen, Y., Jiang, Y. & Zhu, S. Implications of the ecophysiological adaptation of plants on tropical karst habitats for the ecological restoration of desertified rocky lands in southern China. Sci. China Life Sci. 44, 238–247 (2014).
Roig-Oliver, M., Flexas, J., Clemente-Moreno, M. J. & Carriquí, M. Cell wall composition in relation to photosynthesis across land plants’ phylogeny: crops as outliers. New Phytol. 246, 2384–2391 (2025).
White, P. J. & Broadley, M. R. Calcium in plants. Ann. Bot. 92, 487–511 (2003).
White, P. J., Broadley, M. R., El-Serehy, H. A., George, T. S. & Neugebauer, K. Linear relationships between shoot magnesium and calcium concentrations among angiosperm species are associated with cell wall chemistry. Ann. Bot. 122, 221–226 (2018).
Tyerman S., Niemietz C. & Bramley, H. Plant aquaporins: multifunctional water and solute channels with expanding roles. Plant Cell Environ. 25, 173–194 (2002).
Zhao, X., Sucoff, E. & Stadelmann, E. J. Al3+ and Ca2+ alteration of membrane permeability of Quercus rubra root cortex cells. Plant Physiol. 83, 159–162 (1987).
Agrawal A., Conner J. & Rasmann S. Tradeoffs and negative correlations in evolutionary ecology. In: Evolution after darwin: the first 150 years). (Sinauer Associates, 2010).
Garland, T., Downs, C. J. & Ives, A. R. Trade-offs (and Constraints) in organismal biology. Physiol. Biochem. Zool. 95, 82–112 (2022).
Peaucelle, A., Braybrook, S. & Höfte, H. Cell wall mechanics and growth control in plants: the role of pectins revisited. Front. Plant Sci. 3, 121 (2012).
Zhang, Q. et al. Effects of low-phosphorus stress on use of leaf intracellular water and nutrients, photosynthesis, and growth of Brassica napus L. Horticulturae 10, 821 (2024).
Wright, I. J. et al. The worldwide leaf economics spectrum. Nature 428, 821–827 (2004).
Buckley, T. N., John, G. P., Scoffoni, C. & Sack, L. How does leaf anatomy influence water transport outside the xylem? Plant Physiol. 168, 1616–1635 (2015).
Johnson, D. M., Smith, W. K., Vogelmann, T. C. & Brodersen, C. R. Leaf architecture and direction of incident light influence mesophyll fluorescence profiles. Am. J. Bot. 92, 1425–1431 (2005).
Nardini, A. Hard and tough: the coordination between leaf mechanical resistance and drought tolerance. Flora 288, 152023 (2022).
Scoffoni, C., Vuong, C., Diep, S., Cochard, H. & Sack, L. Leaf shrinkage with dehydration: coordination with hydraulic vulnerability and drought tolerance. Plant Physiol. 164, 1772–1788 (2013).
Steudle, E. Water flow in plants and its coupling to other processes: an overview. Methods Enzymol. 174, 183–225 (1989).
Fink, S. Unusual patterns in the distribution of calcium oxalate in spruce needles and their possible relationships to the impact of pollutants. N. Phytol. 119, 41–51 (1991).
Donoghue, M. J. A phylogenetic perspective on the distribution of plant diversity. Proc. Natl. Acad. Sci. USA 105, 11549–11555 (2008).
Atmodjo, M. A., Hao, Z. & Mohnen, D. Evolving views of pectin biosynthesis. Annu. Rev. Plant Biol. 64, 747–779 (2013).
Bartlett, M. K. et al. Rapid determination of comparative drought tolerance traits: using an osmometer to predict turgor loss point. Methods Ecol. Evol. 3, 880–888 (2012).
Sack L., Pasquet-Kok J. & PrometheusWiki. Leaf pressure-volume curve parameters. http://prometheuswiki.publish.csiro.au/tiki-index.php?page=Leaf%20pressure-volume%20curve%20parameters (2011).
Dubois, M., Gilles, K. A., Hamilton, J. K., Rebers, P. A. & Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 28, 350–356 (1956).
Moreira-Vilar, F. C. et al. The acetyl bromide method is faster, simpler and presents best recovery of lignin in different herbaceous tissues than Klason and thioglycolic acid methods. PLoS One 9, e110000 (2014).
Sun, Q. et al. Melatonin promotes ripening and improves quality of tomato fruit during postharvest life. J. Exp. Bot. 66, 657–668 (2015).
John, G. P. et al. The anatomical and compositional basis of leaf mass per area. Ecol. Lett. 20, 412–425 (2017).
Jin, Y. & Qian, H. V. PhyloMaker2: an updated and enlarged R package that can generate very large phylogenies for vascular plants. Plant Divers. 44, 335–339 (2022).
Blomberg, S. P., Garland Jr, T. & Ives, A. R. Testing for phylogenetic signal in comparative data: behavioral traits are more labile. Evolution 57, 717–745 (2003).
Kembel, S. W. et al. Picante: R tools for integrating phylogenies and ecology. Bioinformatics 26, 1463–1464 (2010).
Pagel, M. Inferring the historical patterns of biological evolution. Nature 401, 877–884 (1999).
Felsenstein, J. Phylogenies and the comparative method. Am. Nat. 125, 1–15 (1985).
Paradis, E. & Schliep, K. ape 5.0: an environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 35, 526–528 (2018).
Wikström, N., Savolainen, V. & Chase, M. W. Evolution of the angiosperms: calibrating the family tree. Proc. Biol. Sci. 268, 2211–2220 (2001).
Bansal, M. et al. Southeast Asian Dipterocarp origin and diversification driven by Africa-India floristic interchange. Science 375, 455–460 (2022).
Bates, D., Mächler, M., Bolker, B. & Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 67, 1–48 (2015).
Cohen J. Chapter 2 - The t test for means. In: Statistical power analysis for the behavioral sciences (ed Cohen J.). (Academic Press, 1977).
Macbeth, G., Razumiejczyk, E. & Ledesma, R. Cliff’s Delta Calculator: A non-parametric effect size program for two groups of observations. Universitas Psychologica. 10, 545–555 (2011).
David, F. N. The moments of the z and F distributions. Biometrika 36, 394–403 (1949).
Lever, J., Krzywinski, M. & Altman, N. Principal component analysis. Nat. Methods 14, 641–642 (2017).
Weigelt, A. et al. The importance of trait selection in ecology. Nature 618, E29–E30 (2023).
Camargo, A. PCAtest: testing the statistical significance of principal component analysis in R. PeerJ 10, e12967 (2022).
Bartoń K. MuMIn: multi-model inference, R package (version 0.12.0.) (2009).
Lai, J., Zou, Y., Zhang, J. & Peres-Neto, P. R. Generalizing hierarchical and variation partitioning in multiple regression and canonical analyses using the rdacca.hp R package. Methods Ecol. Evol. 13, 782–788 (2022).
Archer E. rfPermute: estimate permutation p-values for random forest importance metrics. R package (version 2.5.4.) (2025).
Ziyatdinov, A. et al. lme4qtl: linear mixed models with flexible covariance structure for genetic studies of related individuals. BMC Bioinf. 19, 68 (2018).
Lefcheck, J. S. piecewiseSEM: piecewise structural equation modelling in r for ecology, evolution, and systematics. Methods Ecol. Evol. 7, 573–579 (2016).
Acknowledgements
We thank the National Forest Ecosystem Research Station at Xishuangbanna, Xishuangbanna Tropical Botanical Garden, Chinese Academy of Sciences (CAS), and the Puding Karst Ecosystem Research Station (CAS) for their support. We thank Zhizhao Xu and Hong Chen for their assistance in field sampling and lab experiment. We acknowledge funding support from the National Natural Science Foundation of China (32192432 and 31971633), the National Key Research and Development Program of China (2020YFA0608102), the Key Research Program of Frontier Sciences CAS Grant (ZDBS-LY-DQC023), and the Youth Cross-disciplinary Team of the Chinese Academy of Sciences (2023000126).
Author information
Authors and Affiliations
Contributions
Z.M. and M.D. conceived the study. M.D. performed the experiment, conducted the analysis, and created visualizations. M.D. and Z.M. wrote the original draft. L.S. and A.B. contributed significantly to writing and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks David Clark and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Duan, M., Sack, L., Baird, A.S. et al. Cell wall pectin reshapes leaf drought tolerance in dry forests. Nat Commun 17, 1529 (2026). https://doi.org/10.1038/s41467-025-68251-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-68251-w