Abstract
Artificial ionic diodes, inspired by biological ion channels, are typically realized by creating asymmetries in pore geometry or surface charge. Ferroelectric polarization, arising from the spontaneous alignment of electric dipoles within materials, provides an alternative strategy to achieve directional ion transport without additional geometry modification or surface functionalization. This work demonstrates that ultrathin freestanding ferroelectric crystalline bismuth ferrite symmetric-type single-nanopore membranes, with a thickness down to 30 nm, exhibit rectified ion currents driven by naturally spontaneous polarization. These nanopores exhibit reversible polarization switching with hysteretic ion current rectification in response to external pH stimuli, offering additional tunability in emulating the functions of biological ion channels. Furthermore, significant osmotic power generation is observed, enabled by current amplification through directional ion transport. These findings highlight the potential of utilizing ultrathin ferroelectric oxide membrane materials as ionic diodes with directional preferences and switchable polarizations for developing ionic circuits, mimicking biological membranes, and facilitating osmotic power conversion.
Similar content being viewed by others
Introduction
Biological ion channels are specialized proteins embedded within cell membranes that regulate the passage of ions, such as protons, potassium, sodium, calcium, and chloride ions, across the membranes1,2. These ion channels are essential for various physiological processes in living organisms, helping to maintain the electrochemical gradient and ionic balance3,4. Some ion channels demonstrate directional preferences analogously to electronic semiconductor p-n junctions, functioning as ionic diodes5,6. For instance, inward-rectifying potassium channels facilitate the flow of K+ into the cell, whereas other specific ion channels preferentially transport in the opposite direction7,8. Inspired by these natural functions, artificial ionic diodes are designed and fabricated asymmetrically using materials and techniques to enable selective filtering and rectifying capability2,9,10. These artificial ionic diodes, with directional transport preferences, serve as foundational building blocks in the development of ionic circuits and emulating biological membranes11.
The presence of asymmetry plays a pivotal role in enabling directional ion transport in ionic diodes, typically accomplished through the introduction of geometric or surface charge asymmetry12,13,14,15. Geometric asymmetry with one end narrower than the other induces directional ion flow16,17. Various shapes encompassing conical, funnel, and bullet geometries have been designed to tailor the degree of rectification12,15,18. On the other hand, surface charge asymmetry, including polarity or density, is induced by charge gradients along the channel, leading to an electrostatic potential difference19. This asymmetry is typically achieved through the modification of surface functional groups on membranes or the use of heterostructured membrane architectures19,20,21,22. In particular, single nanopores have been extensively studied to explore nanofluidic ion transport in confined nanospaces, aiming to fundamentally replicate the specialized ion transport observed in biological ion channels11,23. Polymer-based materials, such as polyethylene terephthalate and polyimide, are typically employed to fabricate single-nanopore ionic diodes, which demonstrate directional ion transport behaviors24,25,26,27. However, the membranes in these artificial ionic diodes have thicknesses of tens of micrometers, which are much thicker than the only a few nanometers of natural cell membranes. For more accurately emulating the functional characteristics of biological ion channels, the development of ultrathin single-nanopore membranes with directional ion transport is crucial and remains a significant challenge28,29,30.
Ferroelectric polarization, arising from the spontaneous alignment of electric dipoles within materials31, presents a compelling alternative strategy for constructing ionic diodes with directional ion transport and rectification. This intrinsic polarization alignment naturally generates an asymmetric surface charge distribution, offering a viable approach for achieving directional ion transport within an ultrathin membrane and eliminating the need for asymmetric geometric design or additional surface functionalization. The epitaxial growth of ferroelectric oxide thin films has garnered significant attention over the past decades, particularly due to the surge of interest in multiferroics, which possess coexisting ferroic orders32,33. These materials show great promise for nonvolatile components, exhibiting robust room-temperature ferroelectricity even in ultrathin films just a few unit cells thick34,35. Furthermore, recent developments in ultrathin freestanding ferroelectric oxide thin films, enabled by the use of a water-soluble sacrificial layer in epitaxial structures, have led to the realization of crack-free membranes extending up to several millimeters34,36,37. These breakthroughs in ultrathin freestanding thin films shed light on the development of innovative ionic diodes based on ultrathin single-nanopore ferroelectric membranes (FEMs).
This work demonstrates a freestanding single-crystalline FEM made of bismuth ferrite (BiFeO3, BFO), with an ultrathin thickness down to 30 nm, functioning as a single-nanopore ionic diode. By employing state-of-the-art nanofabrication techniques, a single “symmetric-type” BFO nanopore with pore dimensions of only a few nanometers is fabricated to exhibit distinct ion current rectification (ICR) behavior driven by the spontaneous polarization of the freestanding FEM. In addition, the single-nanopore BFO FEM exhibits hysteresis-like switchable rectification behavior under external pH regulation, establishing the membrane as a stimuli-adaptive system and offering additional control for mimicking biological functions. The single-nanopore BFO FEM ionic diodes also exhibit efficient osmotic power harvesting, benefiting from the significant current amplification of directional ion transport. These findings indicate that ultrathin ferroelectric oxide single-nanopore membranes offer a robust and controllable platform for investigating directional ion transport at the nanoscale, driven by intrinsic spontaneous polarization. Incorporating ferroelectric systems opens an avenue for artificial nanomembranes to more faithfully emulate the fundamental transport characteristics of biological membranes.
Results and discussion
Ultrathin ferroelectric single-nanopore membrane
Given that well-regulated spontaneous polarization offers a promising route to controllable directional ion transport, high-quality single-crystalline FEMs with precise polarization control are essential. To this end, (001)-oriented BFO thin films were epitaxially grown on SrTiO3 (STO) single crystal substrate, buffered with Sr3Al2O6 (SAO) sacrificial layer (Step I, Fig. 1a). Here, BFO thin films with varying thicknesses of 30, 80, and 110 nm were prepared. The water-soluble SAO layer acted as a sacrificial layer, allowing the BFO layer to be released as a freestanding membrane without additional support (Step II). Following the release process of the freestanding membrane, the BFO thin film was transferred onto the target substrate for subsequent device fabrication and ionic transport measurements. Freestanding BFO single nanopores were prepared by initially employing a helium ion beam to drill an opening in the SiNx window, creating a hole with dimensions of approximately 100 nm × 200 nm (Step III, Supplementary Fig. 1). The BFO membranes were then transferred onto the SiNx window opening using a polymer-free transfer method (Supplementary Fig. 2)37. The resulting freestanding membranes preserved an uncontaminated surface, which is crucial for ensuring the functionality of ionic channels. Subsequently, single nanopores with various diameters were drilled into the BFO FEMs using an electron beam with TEM irradiation (Step IV).
a Schematic illustration of the fabrication process of the BFO nanopore, where the BFO is represented by yellow spheres. SrTiO3 and Sr3Al2O6 (blue spheres) are abbreviated as STO and SAO, respectively. b XRD pattern of L-scan (upper panel) and phi-scan (lower panel). c High-resolution TEM image and SAED pattern (inset) acquired along the surface normal. This experiment was repeated at least three times with similar results. d Single-point OP PFM hysteresis loop in piezoresponse amplitude and phase. e TEM image of an electron beam-drilled BFO nanopore with a pore size of 5 nm. This experiment was repeated at least three times with similar results. Source data are provided as a Source Data file.
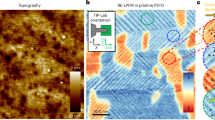
The X-ray diffraction (XRD) L-scan (upper panel) and phi-scan (lower panel) of the freestanding BFO, presented in Fig. 1b, show (00 L) reflections with no impurity peaks along the out-of-plane (OP) direction, and a distinct four-fold symmetry in the in-plane direction. These results confirm the high-quality single-crystalline nature of the BFO FEM. The high-resolution TEM image and selected-area electron diffraction (SAED) pattern (inset) shown in Fig. 1c confirm the preservation of the single-crystalline lattice structure in the BFO FEM after the freestanding process. The spontaneous ferroelectricity in BFO originates from the lone pair electrons and the displacement of the Bi3+ from their centrosymmetric positions relative to the distorted FeO6 octahedra, resulting in a large net polarization along the OP direction38,39. To verify the ferroelectricity of the freestanding BFO, scanning probe microscopy characterizations were performed. Fig. 1d presents the hysteresis loop of the BFO FEM, obtained through piezoresponse force microscopy (PFM). The result shows typical switchable ferroelectric behavior, as evidenced by a butterfly-shaped piezoresponse amplitude and 180° phase switching in response to the applied voltage. The switchable polarization enables the modulation of positively and negatively charged surface distributions, thereby allowing reversible control of surface potential, as further demonstrated by Kelvin probe force microscopy (KPFM) (Supplementary Fig. 4). Figure 1e presents the TEM image of a BFO single nanopore, featuring an effective diameter of 5 nm within a 30-nm-thick BFO FEM, which was produced using the aforementioned nanofabrication technique. Additionally, BFO single-nanopore FEMs with effective diameters of 3 nm and 9 nm were also fabricated, as shown in Supplementary Fig. 3.
Asymmetric ion transport in a symmetric-type nanopore
The ion transport characteristics of the single-nanopore BFO FEMs were investigated using a two-compartment electrochemical cell, as shown in Fig. 2a. Potassium chloride (KCl) was used as the electrolyte for all measurements due to the comparable mobilities and hydration radii of K⁺ and Cl− ions40. Prior to nanopore drilling, the integrity of the BFO FEM was verified by its near-zero, voltage-independent current response (Supplementary Fig. 5), confirming that the freestanding BFO FEM effectively blocks ion transport. Figure 2b presents the current–voltage (I–V) curves of 30-nm-thick BFO single nanopores with diameters of 3, 5, and 9 nm. For comparison, a single nanopore fabricated in a SiNx membrane with a similar thickness and a pore size of 3 nm was also created using the same nanofabrication process. The symmetric-type BFO nanopores exhibit non-linear asymmetric voltage-dependent ion transport behavior with a preferential transport direction, which is a characteristic feature of ICR observed in ionic diodes. The ICR ratio, which quantifies the degree of rectification, is defined as the ratio of current magnitudes at ±1 V (see “Methods” section). These 30-nm-thick BFO single nanopore FEMs, with pore sizes ranging from 3 to 9 nm, exhibit increasing ionic currents with increasing pore sizes. Their corresponding ICR ratios remain relatively consistent with values of 4.2 for 3 nm, 4.1 for 5 nm, and 4.3 for 9 nm at an electrolyte concentration of 10-3 M. As the pore size is further increased to 15 nm, the ionic current continues to rise, whereas the ICR ratio exhibits a slight decrease (Supplementary Fig. 6). This decline is attributed to the diminished influence of surface-governed effects in larger pores41. The pronounced rectification in single-pore BFO FEMs is primarily attributed to the asymmetric surface charge induced by ferroelectric polarization. In contrast, the symmetric-type SiNx nanopore, which lacks ferroelectricity, displays the expected linear and symmetric I–V response, as shown in the inset of Fig. 2b. In addition to pore size, the influence of membrane thickness on the ion transport behavior of single-nanopore BFO FEMs was examined by controlling the growth thickness of the membranes. Ferroelectric BFO nanopores with increased thicknesses of 80 and 110 nm exhibit enhanced ICR ratios of 7.8 and 13.7, respectively (Supplementary Fig. 7). These results indicate that, despite their symmetric-type geometry, the ferroelectric polarization in ultrathin single-nanopore BFO FEMs can effectively induce directional ion transport through the spontaneous alignment of electric dipoles, even at a thickness of only 30 nm. Typically, creating asymmetry in nanopore membranes is accomplished through methods such as asymmetric geometric fabrication, additional surface functionalization, or heterostructural architectures (see Supplementary Table 1). By contrast, the spontaneous ferroelectric polarization in single-nanopore FEMs provides a fundamentally distinct and inherently more straightforward approach to establishing asymmetry for directional ion transport, without the need for additional fabrication or modification techniques.
a Schematic illustration of ion transport through a single BFO nanopore, where the BFO is represented by yellow spheres. Red and blue spheres represent the cations (K+) and anions (Cl−), respectively. b I–V curves of BFO nanopores with a thickness of 30 nm and pore sizes of 3, 5, and 9 nm in 10−3 M KCl electrolyte. The inset shows the linear response of a SiNx nanopore with a pore size of 3 nm. c Ionic conductance of the 9-nm BFO nanopore at 1 V as a function of the KCl electrolyte concentration. The black dashed line represents the bulk transport behavior. d I–T response of the 9-nm BFO nanopore under alternating voltages of −1 V (blue) and +1 V (red) at 10−3 M. Each cycle lasts 5 min. The pH of the KCl electrolyte is 11. Source data are provided as a Source Data file.
We then examined the dependence of ionic conductance on electrolyte concentrations in a single-nanopore BFO FEM with a pore size of 9 nm and a thickness of 30 nm (Fig. 2c and Supplementary Fig. 8). As shown, the BFO nanopore exhibits a deviation from linear bulk behavior at low electrolyte concentrations, which is a typical signature of nanofluidic behavior42,43. As the salt concentration decreases, the electric double layer overlap becomes significant, and thus, the surface conductance, influenced by the surface charges on the pore walls, dominates the overall conductance behavior9,44. Furthermore, Fig. 2d demonstrates the stable operation of the single nanopore BFO FEM, highlighting its robustness as a potential ionic diode component, attributed to the inherent stability of metal oxides.
Polarization-switchable directional ion transport
In biological ion channels, activities, such as protonation or ion selectivity, are strongly influenced by the surrounding pH environment45. Similarly, the surface charge characteristics of artificial ion membranes, including charge density and polarity, can be modulated by the environmental pH, thereby affecting ion transport behavior23. Understanding the rectification responses of ionic diodes under varying pH conditions enables enhanced control of nanopore membranes for mimicking biological functionalities. In particular, the surface termination of metal oxides follows a general equilibrium expressed as
where M represents the metal element46. The equilibrium shifts to the right as pH increases and to the left as pH decreases, depending on the degree of hydroxyl dissociation when the metal oxide surface is in contact with an aqueous solution. By harnessing the pH-dependent surface termination chemistry of BFO thin films at the solid-liquid interface, polarization-switchable rectification behavior in response to environmental pH changes is demonstrated. This endows the membrane with pH-responsive adaptability and offers an additional level of control for emulating a biological ion channel. Fig. 3a shows the I–V characteristics of a 9-nm BFO single pore with a thickness of 30 nm measured at pH values of 11, 6, and 3. The I–V curves show current amplification at positive voltages and current suppression at negative voltages at pH 11 and 6. By contrast, the behavior reverses at pH 3, showing current amplification at negative voltages and suppression at positive voltages. The corresponding rectification ratios are 4.3 and 3.4 for pH 11 and 6, while a negative ratio of -3.2 is observed for pH 3. This shift from a positive to a negative ICR ratio indicates that the directional ionic transport behavior is responsive to environmental pH levels. The ferroelectric polarization directions of the BFO FEMs after exposure to the electrolytes with pH values of 11, 6, and 3 were examined through PFM. The resulting OP PFM phase images reveal a strong correlation between the polarization direction and the observed ionic rectification behavior, as indicated by the sign of the ICR ratio. As shown in Fig. 3b, upward polarization (Pup) for the BFO FEMs corresponds to positive ICR ratios at pH 11 and 6, while the downward polarization (Pdown) gives rise to a negative ratio at pH 3. Nevertheless, for a thicker BFO FEM with a thickness of 110 nm, the unchanged upward polarization direction leads to the consistently positive ICR ratios after exposure to electrolytes at pH 11 and 3 with no inversion observed (Supplementary Fig. 9). This underscores the critical role of polarization direction in governing directional ion transport in single-nanopore BFO FEMs, as reversing the polarization results in a corresponding inversion of the ICR.
a I–V of a 9-nm BFO nanopore in 10−3 M KCl at pH values of 11, 6, and 3. b OP PFM phase images of the BFO FEM after exposure to 10−3 M KCl electrolytes at different pH values. Scale bar, 300 nm. c Ion selectivity test under a large concentration gradient. Blue curves (K+ current) are measured by placing 1 M KCl on the front side and 10−5 M KCl on the back side of the BFO FEM, while red curves (Cl− current) are measured with an opposite electrolyte configuration. d Simulation model of the polarization-dependent BFO FEM (not to scale). e Simulated I–V response with respect to polarization direction. Pup and Pdown represent the upward and downward polarizations, respectively. Source data are provided as a Source Data file.
The polarization reversal in the 30-nm-thick BFO FEM is primarily driven by pH-varied surface termination of the metal oxide as the H⁺/OH⁻ concentration in aqueous solution changes. Initially, under alkaline conditions (pH 11), the surface is dominated by M–O- bonds, resulting in a negatively charged termination that generates an upward electric field aligned with the original upward polarization of the as-grown BFO FEM. At the neutral condition (pH 6), which is close to the point of zero charge for the BFO FEM surface47, the resulting weak electric field does not alter the polarization direction. In contrast, at acidic conditions (pH 3), the surface becomes enriched with M–OH2+ groups, creating a positively charged termination and a downward electric field that opposes the original polarization, thereby causing the polarization to switch direction. When H+ and OH- interact with O and Fe on the BFO surface, they form M–O–H bonds, leading to upward and downward displacements of Fe ions from their original positions48. This behavior is further supported by the X-ray photoelectron spectroscopy (XPS) result (Supplementary Fig. 10), which shows a significant increase in M–O–H surface species during the switching process as the environmental pH changes, indicating a strong interplay between the surface electrostatics and ionic interactions. However, when the BFO FEM thickness is increased to 110 nm, the electric field generated by surface termination becomes insufficient compared to the 30-nm-thick membrane despite having a similar surface charge density. These findings indicate that the polarization-switchable ion transport in response to pH stimuli is a distinct characteristic of the ultrathin single-nanopore BFO FEM, mimicking the behavior of biological ion channels.
Building on the polarization-switchable directional ionic transport behavior of the ultrathin single-nanopore BFO FEM in various pH environments, we performed ion selectivity measurements as shown in Fig. 3c. Ion selectivity is assessed under a significant concentration gradient across the membrane, which allows for the evaluation of its selective ion transport characteristics and surface charge properties14. One side of the BFO FEM is in contact with 1 M KCl, while the opposite side is in contact with 10⁻⁵ M KCl. The contribution from the highly diluted side is considered negligible, enabling estimation of the individual ionic currents from K⁺ and Cl− by reversing the electrolyte configuration. At pH levels of 11 and 6, the BFO single nanopore exhibits cation-selective transport, with the K⁺ current being the dominant contribution. In contrast, at pH 3, the membrane shifts toward anion selectivity, where the Cl− current exceeds that of the cations. This switch in ion selectivity underscores the dynamic pH-responsive surface behavior of the BFO FEM for its ionic transport properties, which is closely linked to alterations in ferroelectric polarizations. Furthermore, the inversion of the ICR is quantitatively modeled using theoretical simulations based on the multi-ion Poisson–Nernst–Planck and Navier–Stokes (PNP-NS) model49, which incorporates surface chemical reactions on the pore wall, all ion species, and the effects of electroosmotic flow (see Methods). In the BFO nanopore model, the surface charge distribution is set to align with the direction of polarization, as illustrated in Fig. 3d and Supplementary Fig. 11. The simulated I–V curves, along with the corresponding ion concentration distributions, reproduce the observed ICR inversion with ion accumulation and depletion under opposite voltage polarities (Fig. 3e and Supplementary Fig. 11). It is worth noting that a discrepancy exists between the values of experimental and simulated ionic currents, arising from deviations in simulated parameters (e.g., induced surface charge density, nanopore dielectric constant, and surface site densities of functional groups) from the actual system. Nevertheless, the simulations qualitatively explain the experimental observations of rectification inversion under different polarization directions, thereby supporting the proposed polarization-switchable rectification.
Hysteresis-like rectification behavior
In addition to the switchable polarity of ionic transport behavior by adjusting H⁺ or OH⁻ concentrations near the BFO surface, the ultrathin 30-nm-thick BFO single nanopore exhibits an intriguing reversible ICR response to the changes in pH conditions. Fig. 4a, b show the evolution of the I–V curves to reflect ionic diode transport characteristics during pH cycling. A forward sweep was performed from pH 11 to pH 3, followed by a reverse sweep from pH 3 back to pH 11. We observed that an initially positive ICR ratio reverses to negative when exposed to acidic aqueous solutions, and subsequently returns to a positive value when re-exposed to basic aqueous solutions. However, the ICR ratio as a function of varying pH exhibits hysteresis-like behavior between the forward and reverse sweeps. Figure 4c summarizes the ICR ratios of the single-nanopore BFO FEMs during both sweeping directions under various pH values, revealing distinct rectification states at each pH value. For example, there are two sweep-dependent rectification states at pH 6, which are a positive ICR ratio of 3.4 during the forward sweep (also see the inset of Fig. 4a), and a negative ratio of −2.4 during the reverse sweep (also see the inset of Fig. 4b). The corresponding OP PFM phase images in Fig. 4c confirm upward and downward polarizations associated with the two distinct ICR states. This hysteresis-like rectification closely resembles the polarization hysteresis loop of the BFO FEM obtained from PFM measurements, as presented in Fig. 1d and Supplementary Fig. 12. The hysteresis in ferroelectric materials arises from a polarization response that depends on the history of the applied electric field, where polarization switching occurs only when the coercive field is exceeded. Because the directional ion transport in single-nanopore BFO FEMs is primarily governed by ferroelectric polarization, the resulting ICR ratio exhibits a hysteretic response under cyclic pH sweeps, mirroring the ferroelectric switching mechanism. These findings highlight that the rectified ion transport in single-nanopore BFO FEMs is predominantly controlled by ferroelectric polarization, with the ultrathin BFO nanopore functioning as a pH-regulated, polarization-switchable ionic diode capable of reversible switching.
I–V curves under a forward (from pH 11–3) and b reverse (from pH 3–11) sweep in 10−3 M KCl. Insets show the magnified curves at pH 6. c ICR ratio-pH hysteresis loop, with corresponding OP PFM phase images after exposure to electrolytes at varied pH values. Scale bar, 500 nm; vertical color bar, 180° for PFM phase images. Each data point represents the mean ± standard deviation obtained from ten samples. Pup and Pdown represent the upward and downward polarizations, respectively. Source data are provided as a Source Data file.
Ferroelectric single nanopores for osmotic power harvesting
Ionic diodes enable directional ion transport, allowing unidirectional current flow, preventing backflow, and reducing Joule heating-induced energy dissipation, thereby improving the efficiency of osmotic power conversion under salinity gradients50. The concentration gradients ranging from 10-fold to 1000-fold were introduced into the system to measure the osmotic power generation performance based on the ultrathin ferroelectric BFO single nanopore FEM as schematically shown in Fig. 5a. By measuring the I–V response of a BFO single nanopore with a thickness of 30 nm and pore size of 9 nm in a concentration gradient system (Supplementary Figs. 13−15 and Supplementary Table 2), we can determine the short-circuit current (Isc) at zero external bias and the osmotic potential derived from the open-circuit voltage (Voc) as shown in Fig. 5b. The pure osmosis-driven osmotic voltage (Vosm) and osmotic current (Iosm) are extracted from the voltage and current axis intercepts, respectively, after subtracting the inherent redox voltages (Vred) generated at the electrode-electrolyte interfaces due to concentration imbalances. Both Vosm and Iosm rise with increasing concentration gradient ratios. The Vosm follows an approximately linear trend, while the Iosm exhibits exponential growth with the concentration gradient, as shown in Fig. 5c, d. As a result, the BFO single nanopore demonstrates a high osmotic power (Posm, where Posm = 1/4 × Iosm × Vosm), as compared with those previously reported single-pore membranes (Supplementary Table 3). This superior osmotic power performance stems from the significant enhancement in the Iosm, enabled by the directional ion transport of the single-nanopore BFO FEM, which effectively amplifies ionic current despite its relatively lower surface charge density compared to state-of-the-art single-nanopore membranes (see Supplementary Figs. 16 and 17 for more details)12,42,44.
a Schematic of the experimental setup for osmotic power generation measurements. BFO FEM, cations (K+), and anions (Cl⁻) are represented by yellow, red, and blue spheres, respectively. b I–V curves of the BFO nanopore under the KCl concentration gradient of 1000. The dashed curve presents the pure osmotic contribution after subtracting the redox voltage (Vred). Voc, Vosm, Isc, and Iosm represent open-circuit voltage, osmotic voltage, short-circuit current, and osmotic current, respectively. c Osmotic voltage and d osmotic current of the BFO nanopore under concentration gradients. The base concentration is 10−3 M. Each data point represents the mean ± standard deviation obtained from ten samples. e Real output current and f output power generated when the BFO nanopore FEM is connected to a load resistor at a 1000-fold concentration gradient. The pH of the KCl electrolyte is 11. Source data are provided as a Source Data file.
In a practical application, the BFO nanopore ionic diode was connected in series with an external electrical resistor to simulate power delivery to a load. Fig. 5e, f show the actual output current (Iout) and output power (Pout) as functions of load resistance (Rload) for the 9-nm BFO nanopore under a concentration gradient of 1000. With increasing Rload, Iout decreases, while the output power, Pout = Iout2 × Rload, reaches a local maximum when the load resistance matches the internal resistance of the nanopore9,40. Under these conditions, the BFO single nanopore produces a promising peak output power of 13.1 nW at a Rload of 1 MΩ. These results highlight the effectiveness of ferroelectric oxide membranes for energy harvesting, attributed to their unique polarization-induced directional ion transport behavior. In addition, the influence of surface charge on ion transport is known to become more significant as the pore diameter decreases. We evaluated the osmotic power generation performance of the 3-nm BFO nanopore and observed a higher osmotic voltage, which can be attributed to the enhanced electric double layer overlap at smaller pore sizes. However, the reduced pore size also led to a lower osmotic current, thereby limiting the overall power output. Because optimal osmotic power generation requires both high voltage and current, the 9-nm BFO pore, providing a superior osmotic current along with a sufficient voltage, achieves high power performance (also see Supplementary Fig. 18 for details).
In summary, we have demonstrated a polarization-switchable single-nanopore ionic diode fabricated from ultrathin freestanding ferroelectric BFO membranes. These BFO single symmetric-type nanopores enable directional ion transport due to their intrinsically asymmetrical surface charge arising from ferroelectric polarization. By harnessing the functionality of the pH-responsive surface termination of metal oxides, the ultrathin single-pore BFO FEM exhibits polarization-switchable and hysteresis-like reversible ionic rectification transport. Furthermore, a nanowatt-scale osmotic power is generated through a single BFO nanopore, benefiting from the amplification of directional ion currents. These artificial ionic diodes achieve exceptional directional ionic transport at nanometer-scale thicknesses without relying on traditional asymmetric geometry or surface functionalization. Such ultrathin ferroelectric metal oxide single-nanopore membranes act as promising foundational ionic components for future advancement in mimicking the functions of biological cell membranes for developing related ionic circuits and facilitating osmotic power conversion.
Methods
PLD growth
Single-crystalline freestanding BFO films were fabricated using a two-step process. First, epitaxial layers of SAO and BFO were deposited sequentially on STO single-crystal substrates via pulsed laser deposition (PLD), where the SAO layer served as an epitaxial sacrificial layer. The deposition of SAO was carried out at 740 °C under an oxygen pressure of 40 mTorr, while BFO was deposited at 700 °C under a higher oxygen pressure of 100 mTorr. Following the deposition, the samples were cooled to room temperature in an oxygen atmosphere maintained at 600 Torr to minimize oxygen vacancies.
Materials characterizations
High-resolution XRD L-scan measurement of the freestanding BFO membranes was performed using a synchrotron-based 6 + 2-circle diffractometer. The measurements utilized a photon energy of 10 keV and a beam size of 0.8 × 0.8 mm². These experiments were conducted at beamlines TPS-09A, TLS-13A1, and TLS-17B at the National Synchrotron Radiation Research Center (NSRRC), Taiwan. The L-scan and RSM measurements were acquired with an integration time of 2 s per data point and were presented in reciprocal lattice units (r.l.u.), normalized to the bulk lattice constant of BFO (1 r.l.u. = 2\(\pi\)/aBFO).
To examine the lattice structure of the freestanding BFO membranes, plane-view TEM images and diffraction patterns were acquired using a JEOL JEM-2100F transmission electron microscope equipped with a spherical aberration (Cs) corrector in STEM mode. These measurements were conducted at the Core Facility Center of National Cheng Kung University, Taiwan.
The PFM, KPFM, and AFM images of BFO were acquired using a commercial scanning probe system (Innova, Bruker; NanoDrive software, version 8.02). The PFM signal was collected in contact mode using the Pt/Ir-coated conductive Si tips (OSCM-PIT, Olympus) with a spring constant of 1–5 N m−1, driven by an AC voltage amplitude of 1 V. In KPFM, the same conductive tip was used to measure the surface potential. After the first topographic scan, the tip was lifted 100 nm above the surface for the potential measurement. The AFM image was acquired in tapping mode using Si tips (OLTESPA, Olympus) with a spring constant of 0.5–4.4 N m−1 and frequency at 60–100 kHz. The XPS spectra were acquired using the ULVAC PHI 5000 Versa Probe system equipped with an Al Kα monochromatic X-ray source. The zeta potentials of BFO thin film were measured using a streaming potential-based zeta potential analyzer (SurPASS 3, Anton Paar) in 10-2 M KCl.
Nanopore fabrication
To release the freestanding BFO membranes, the SAO sacrificial layer was selectively etched using deionized water, enabling the detachment of the BFO layer from the underlying STO substrate37. The combination of the mild etching condition and the strong mechanical robustness of the BFO membrane allows it to remain intact with a lateral size on the millimeter scale. The pre-fabricated SiNx aperture is intentionally fabricated at the center of the Si chip, the size of which is 1 cm, making it easy to handle. The floating BFO membrane can then be gently scooped up with the aperture chip, enabling the millimeter-sized BFO membrane to cover the central region of the SiNx aperture chip. The specifications of the SiNx substrate (Norcada, Canada) are as follows: a chip frame size of 10 mm by 10 mm, Si thickness of 200 μm, SiNx membrane thickness of 30 nm, and a membrane area of 50 μm by 50 μm. The SiNx hole was drilled using a helium ion beam microscope (ZEISS ORION NanoFab) with an acceleration voltage of 30 kV, beam current of 20 pA, spacing of 0.5 nm, dwell time of 1 μs for a total duration of 200 s. The secondary electron image of the drilled SiNx hole was also acquired. After the transfer, a BFO single nanopore was drilled using TEM (JEOL JEM-ARM300F2) equipped with double spherical aberration correctors. For comparison, a SiNx nanopore was also fabricated directly on the SiNx membrane using TEM drilling.
Electrical measurement
All electrical measurements were conducted using a customized setup consisting of two PMMA reservoirs and holed PDMS buffer layers to sandwich the nanopore samples securely. Electrodes were prepared by electroplating silver wires (2 mm in diameter and 70 mm in length) in a 0.1 M KCl solution (without adjusting pH) at 2 V against a graphite rod, which served as the counter electrode, for 15 min. In the measurements, the electrode in the left reservoir, positioned at the rear side of the nanopore sample, served as the reference electrode, while the electrode in the right reservoir, located at the front side, functioned as the working electrode. The pH values of the KCl electrolytes were measured and adjusted beforehand using a pH meter (6177 M, Jenco). A Keithley 6487 sourcemeter is used to apply voltage and record current. The measurement was controlled using the LabVIEW program (NI LabVIEW 2019). A variable resistor (RS-200W, IET Labs) with resistance ranging from 0 to 108 Ω is used for real output performances. To minimize electromagnetic interference, the measurement setup is enclosed in a Faraday cage (Ivium Technologies) with a common ground with all the measurement equipment.
ICR ratio
The ICR ratio in this work is defined as the ratio of current magnitudes measured at ±1 V, and is expressed as follows:
The sign of the ICR ratio value indicates the rectification direction observed.
Theoretical simulation
The influence of pH on the ion transport behaviors of a single cylindrical BFO nanopore was also investigated through numerical simulation (COMSOL Multiphysics, version 4.3a). The multi-ion Poisson–Nernst–Planck (PNP) and Navier–Stokes (NS) equations were adopted to describe the ion transport behaviors. The electrical potential is expressed by the Poisson equation:
where ϕ is the electric potential, ρe is the electric charge density of electrolyte ions, εf is the relative permittivity of the electrolyte solution, and Cj and zj are the concentration and valence of jth ionic species (j = 1 for K+, j = 2 for Cl−, j = 3 for H+, and j = 4 for OH−), respectively.
The ion transport is expressed by the Nernst-Planck equation:
where Jj and Dj are the flux and diffusivity of jth ionic species, respectively; F, R, and T are the Faraday constant, universal gas constant, and absolute temperature, respectively; u is the fluid velocity.
The flow field is described by the continuity and Navier–Stokes equations:
where p and μ are the hydrodynamic pressure and the fluid viscosity, respectively. Since the Reynolds numbers in the system are very small, the inertial terms in the Navier–Stokes equation can be neglected.
Subsequently, the calculation of the ionic current involves integrating the total ionic flux over the cross-sectional area, which can be expressed as follows:
where S and n represent the cross-sectional area and unit outer normal vector, respectively.
We assumed the following boundary conditions to solve the above governing Eq. (1). We used the cylindrical coordinate system (r, z) with the origin at the center of the nanopore to describe a cylindrical nanopore with length (L = 30 nm) and radius (R = 3 nm) connecting two big reservoirs. The symmetric boundary condition was specified along the axis of the nanopore. (2) The reservoirs at both ends were sufficiently large, ensuring that the ion concentrations remained unaffected by the charged channel or the reservoir on the opposite side, allowing both ends to maintain the bulk concentration value (\({C}_{j}={C}_{j0}\)). (3) To determine the potential across the system, the electrical potential at the reservoir boundary on the upper side was set to the applied bias voltage (\(\phi={V}_{{app}}\)), while the boundary on the lower side of the reservoir was grounded (\(\phi=0\)). (4) There was no external pressure gradient (∆p = 0) within the system. (5) The side walls of two reservoirs were ion-impermeable (\(n\cdot {{{\bf{J}}}}_{{{\boldsymbol{j}}}}=0\)) and nonslip, and were set to be uncharged (\(-{{\bf{n}}}\cdot ({\varepsilon }_{f}\nabla \phi )=0\)). (6) The walls of the BFO region were also ion-impermeable (\({{\bf{n}}}\cdot {{{\bf{J}}}}_{j}=0\)) and nonslip, with a surface charge density \((\sigma )\). (7) We set an induced surface charge density \(({{{\rm{\sigma }}}}_{p}=1{{\rm{C}}}{{{\rm{m}}}}^{-2})\) to describe the extra effect of the pH value. In pH 11, surface charge density was set to \(({{\rm{\sigma }}}-{{{\rm{\sigma }}}}_{p})\) at the BFO region boundary near the bias voltage, and surface charge density was set to \(({{\rm{\sigma }}}+{{{\rm{\sigma }}}}_{p})\) at the BFO region boundary near the grounded. In pH 3, the opposite surface charge density was applied. (8) Since the dielectric material of the membrane wall, when in contact with an aqueous solution, exhibited a charge-regulated nature. Its surface charge density \((\sigma )\) could be determined by the following deprotonation/protonation reactions with equilibrium constants KA and KB:
In the above reactions, \({10}^{{-{{\rm{p}}}K}_{{{\rm{A}}}}}\,=\,{\Gamma }_{{{{\rm{MO}}}}^{-}}{[{{{\rm{H}}}}^{+}]}_{{{\rm{S}}}}/{\Gamma }_{{{\rm{MOH}}}}\) and \({10}^{{-{{\rm{p}}}K}_{{{\rm{B}}}}}\,=\,{\Gamma }_{{{{\rm{MOH}}}}_{2}^{+}}/({\Gamma }_{{{\rm{MOH}}}}{\left[{{{\rm{H}}}}^{+}\right]}_{{{\rm{S}}}})\), with pKj = -[log Kj] (j = A and B),\(\,{\Gamma }_{{{\rm{MOH}}}}\), \({\Gamma }_{{{{\rm{MO}}}}^{-}}\), and \({\Gamma }_{{{{\rm{MOH}}}}_{2}^{+}}\) were the surface site densities of MOH, MO–, and MOH2+, respectively, and \({\left[{{{\rm{H}}}}^{+}\right]}_{{{\rm{S}}}}\) was the molar concentration of H+ ions at the membrane wall/liquid interface. The total number of site density of dissociable MOH molecules on the membrane wall surface was \({\Gamma }_{{{\rm{total}}}}={\Gamma }_{{{\rm{MOH}}}}+{\Gamma }_{{{{\rm{MO}}}}^{-}}{+\Gamma }_{{{{\rm{MOH}}}}_{2}^{+}}\). The surface charge density of the membrane wall was expressed as
For the BFO, we assumed the parameters Γtotal, pKA, and pKB were 0.1 nm–2, 5, and -2, respectively.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All data that support the findings of this study are presented in the Manuscript and Supplementary Information or are available from the corresponding author upon request. Source data are provided with this paper.
References
Shen, J., Liu, G., Han, Y. & Jin, W. Artificial channels for confined mass transport at the sub-nanometre scale. Nat. Rev. Mater. 6, 294–312 (2021).
Mo, R. J. et al. Regulating ion affinity and dehydration of metal-organic framework sub-nanochannels for high-precision ion separation. Nat. Commun. 15, 2145 (2024).
Xu, Z., Hueckel, T., Irvine, W. T. M. & Sacanna, S. Transmembrane transport in inorganic colloidal cell-mimics. Nature 597, 220–224 (2021).
Didier, M. E. P., Tarun, O. B., Jourdain, P., Magistretti, P. & Roke, S. Membrane water for probing neuronal membrane potentials and ionic fluxes at the single cell level. Nat. Commun. 9, 5287 (2018).
Zhang, Y. et al. Sustainable power generation for at least one month from ambient humidity using unique nanofluidic diode. Nat. Commun. 13, 3484 (2022).
Guan, W., Fan, R. & Reed, M. A. Field-effect reconfigurable nanofluidic ionic diodes. Nat. Commun. 2, 506 (2011).
Kubo, Y., Baldwin, T. J., Jan, Y. N. & Jan, L. Y. Primary structure and functional expression of a mouse inward rectifier potassium channel. Nature 362, 127–133 (1993).
Ketchum, K. A., Joiner, W. J., Sellers, A. J., Kaczmarek, L. K. & Goldstein, S. A. N. A new family of outwardly rectifying potassium channel proteins with two pore domains in tandem. Nature 376, 690–695 (1995).
Wang, J. et al. Unlocking osmotic energy harvesting potential in challenging real-world hypersaline environments through vermiculite-based hetero-nanochannels. Nat. Commun. 15, 608 (2024).
Xin, W. et al. Biomimetic KcsA channels with ultra-selective K+ transport for monovalent ion sieving. Nat. Commun. 13, 1701 (2022).
Zhang, Z., Wen, L. & Jiang, L. Bioinspired smart asymmetric nanochannel membranes. Chem. Soc. Rev. 47, 322–356 (2018).
Gao, M., Tsai, P.-C., Su, Y.-S., Peng, P.-H. & Yeh, L.-H. Single mesopores with high surface charges as ultrahigh performance osmotic power generators. Small 16, 2006013 (2020).
Zhu, X. et al. Unique ion rectification in hypersaline environment: a high-performance and sustainable power generator system. Sci. Adv. 4, eaau1665 (2018).
Zhang, Z. et al. Engineered asymmetric heterogeneous membrane: a concentration-gradient-driven energy harvesting device. J. Am. Chem. Soc. 137, 14765–14772 (2015).
Xiao, K. et al. Enhanced stability and controllability of an ionic diode based on funnel-shaped nanochannels with an extended critical region. Adv. Mater. 28, 3345–3350 (2016).
Kubeil, C. & Bund, A. The role of nanopore geometry for the rectification of ionic currents. J. Phys. Chem. C 115, 7866–7873 (2011).
Ramirez, P., Apel, P. Y., Cervera, J. & Mafe, S. Pore structure and function of synthetic nanopores with fixed charges: tip shape and rectification properties. Nanotechnology 19, 315707 (2008).
Laucirica, G. et al. Shape matters: enhanced osmotic energy harvesting in bullet-shaped nanochannels. Nano Energy 71, 104612 (2020).
Ding, L. et al. Bioinspired Ti3C2Tx MXene-based ionic diode membrane for high-efficient osmotic energy conversion. Angew. Chem. Int. Ed. 61, e202206152 (2022).
Zhang, X. et al. Asymmetric electrokinetic proton transport through 2D nanofluidic heterojunctions. ACS Nano 13, 4238–4245 (2019).
Zhao, C. et al. Effect of anion species on ion current rectification properties of positively charged nanochannels. ACS Appl. Mater. Interfaces 12, 28915–28922 (2020).
Zhang, M. et al. Controllable ion transport by surface-charged graphene oxide membrane. Nat. Commun. 10, 1253 (2019).
Hao, J., Wu, R., Zhou, J., Zhou, Y. & Jiang, L. Regulation of bioinspired ion diodes: from fundamental study to blue energy harvesting. Nano Today 46, 101593 (2022).
Lu, J. et al. Ultrafast rectifying counter-directional transport of proton and metal ions in metal-organic framework–based nanochannels. Sci. Adv. 8, eabl5070 (2022).
Lu, J. et al. Efficient metal ion sieving in rectifying subnanochannels enabled by metal-organic frameworks. Nat. Mater. 19, 767–774 (2020).
Sun, Y. et al. A highly selective and recyclable NO-responsive nanochannel based on a spiroring opening-closing reaction strategy. Nat. Commun. 10, 1323 (2019).
Xie, G. et al. A bioinspired switchable and tunable carbonate-activated nanofluidic diode based on a single nanochannel. Angew. Chem. Int. Ed. 54, 13664–13668 (2015).
Ishizaki-Betchaku, Y., Kumakura, N., Yamamoto, S., Nagano, S. & Mitsuishi, M. Ultrathin ionic diodes with electrostatically heterogeneous hybrid interfaces of nanoporous SiO2 nanofilms and polymer layer-by-layer multilayers. Small 20, 2404306 (2024).
Wu, Y., Jiang, L. & Wen, L. Nanofluidic membrane for confined ion transport: from uniform to composite strategy. Mater. Today 65, 189–206 (2023).
Cao, E. & Siwy, Z. S. Heterogeneous nanopore arrays - selective modification of nanopores embedded in a membrane. Adv. Mater. 37, e2418987 (2025).
Li, W. et al. Emergence of ferroelectricity in a nonferroelectric monolayer. Nat. Commun. 14, 2757 (2023).
Fiebig, M., Lottermoser, T., Meier, D. & Trassin, M. The evolution of multiferroics. Nat. Rev. Mater. 1, 16046 (2016).
Ramesh, R. & Schlom, D. G. Creating emergent phenomena in oxide superlattices. Nat. Rev. Mater. 4, 257–268 (2019).
Ji, D. et al. Freestanding crystalline oxide perovskites down to the monolayer limit. Nature 570, 87–90 (2019).
Wang, H. et al. Direct observation of room-temperature out-of-plane ferroelectricity and tunneling electroresistance at the two-dimensional limit. Nat. Commun. 9, 3319 (2018).
Zhang, J. et al. Super-tetragonal Sr4Al2O7 as a sacrificial layer for high-integrity freestanding oxide membranes. Science 383, 388–394 (2024).
Butcher, T. A. et al. Ptychographic nanoscale imaging of the magnetoelectric coupling in freestanding BiFeO3. Adv. Mater. 36, 2311157 (2024).
Wang, J. et al. Epitaxial BiFeO3 multiferroic thin film heterostructures. Science 299, 1719–1722 (2003).
Zhao, T. et al. Electrical control of antiferromagnetic domains in multiferroic BiFeO3 films at room temperature. Nat. Mater. 5, 823–829 (2006).
Yang, J. et al. Advancing osmotic power generation by covalent organic framework monolayer. Nat. Nanotechnol. 17, 622–628 (2022).
Ai, Y., Zhang, M., Joo, S. W., Cheney, M. A. & Qian, S. Effects of electroosmotic flow on ionic current rectification in conical nanopores. J. Phys. Chem. C 114, 3883–3890 (2010).
Feng, J. et al. Single-layer MoS2 nanopores as nanopower generators. Nature 536, 197–200 (2016).
Siria, A. et al. Giant osmotic energy conversion measured in a single transmembrane boron nitride nanotube. Nature 494, 455–458 (2013).
Liu, T.-R. et al. Single-layer hexagonal boron nitride nanopores as high-performance ionic gradient power generators. Small 20, 2306018 (2024).
Liu, Z., Wang, W., Xie, R., Ju, X. J. & Chu, L. Y. Stimuli-responsive smart gating membranes. Chem. Soc. Rev. 45, 460–475 (2016).
Yan, R., Liang, W., Fan, R. & Yang, P. Nanofluidic diodes based on nanotube heterojunctions. Nano Lett. 9, 3820–3825 (2009).
Li, S. et al. Remarkably enhanced photocatalytic activity of laser ablated Au nanoparticle decorated BiFeO3 nanowires under visible-light. Chem. Commun. 49, 5856–5858 (2013).
Tian, Y. et al. Water printing of ferroelectric polarization. Nat. Commun. 9, 3809 (2018).
Su, Y.-S., Hung, W.-H., Fauziah, A. R., Siwy, Z. S. & Yeh, L.-H. A pH gradient induced rectification inversion in asymmetric nanochannels leads to remarkably improved osmotic power. Chem. Eng. J. 456, 141064 (2023).
Macha, M., Marion, S., Nandigana, V. V. R. & Radenovic, A. 2D materials as an emerging platform for nanopore-based power generation. Nat. Rev. Mater. 4, 588–605 (2019).
Acknowledgements
This work is financially supported by the National Science and Technology Council (NSTC), Taiwan (Grant Nos. 112-2124-M-002-010 and 113-2124-M-002-007, C.-W.C.; Grant Nos. 113-2628-E-011-002, 113-2124-M-011-002, 113-2628-E-011-005-MY3, and 112-2923-E-011-003-MY3, L.-H.Y.; Grant Nos. 112-2112-M-006-020-MY3 and 113-2124-M-006-010, J.-C.Y.). The financial supports from the Ministry of Education in Taiwan (Grant No. 111L900801 by the Center of Atomic Initiative for New Materials (AI-Mat), National Taiwan University, from the Featured Areas Research Center Program within the framework of the Higher Education Sprout Project, C.-W.C.; Sustainable Electrochemical Energy Development Center (SEED) project, L.-H.Y.) are also acknowledged. Technical support from the Advanced Materials Characterization Lab, Inst. Atomic and Molecular Sciences (IAMS), iMATE program, Academia Sinica, is also acknowledged.
Author information
Authors and Affiliations
Contributions
C.-W.C. conceived and guided the project. C.-H.C. performed the nanopore fabrication, electrical measurements, PFM and KPFM measurements, data analysis, and manuscript writing with C.-W.C. C.-C.W., L.-S.W., Y.-L.H., and J.-C.Y. carried out the PLD growth, freestanding membrane fabrication, XRD characterization, and ferroelectric measurements. P.-C.K., T.-R.L., and J.S. performed the nanopore fabrication and TEM imaging. W.-L.H. and L.-H.Y. assisted with the electrical measurements and simulation data collection. C.-A.L. conducted the simulations. Y.-C.L. performed the PFM and KPFM measurements. Z.-W.C. produced the schematic illustrations. J.-C.Y., L.-H.Y., and C.-W.C. revised the manuscript and discussed the results.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Yuya Ishizaki-Betchaku, Zuhuang Chen, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chiang, CH., Wei, CC., Kuo, PC. et al. Single symmetric nanopores in ultrathin crystalline ferroelectric BiFeO3 as polarization-switchable ionic diodes. Nat Commun 17, 1542 (2026). https://doi.org/10.1038/s41467-025-68262-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-68262-7