Abstract
The high ductility of Mg has posed major challenges for nanofabrication utilizing mechanical ball-milling. While the addition of organic solvents is effective, it has been unclear how they improve the ball-milling effect by modifying the material’s surface properties. Herein, we report that the solvent-mediated partial ionicity plays an important role in enhancing the nanosizing effect of Mg87.5Ni5.5Y7 alloy. This approach enables the Mg87.5Ni5.5Y7 particles to be 88 times smaller than those of the solvent-free procedure. The Mg87.5Ni5.5Y7 nanoparticles underwent complete dehydrogenation in 3 min at 300 °C and in 17 min at 240 °C, which can be stably cycled at least for 500 times. Solvent (THF) adsorption on Mg induces Mgδ+‒Mgδ− dipole structure. This increases the surface hardness of Mg-based alloy and maximizes the ball milling-driven structural deformation, thereby facilitating ion migration. Mg‒Mg bond breaking is caused by the resulting Coulombic repulsion between Mg atoms. These findings provide an affordable approach for nanoparticle fabrication of highly ductile materials.
Similar content being viewed by others
Introduction
Hydrogen, as a zero-carbon energy carrier, exhibits impressive characteristics of abundance, versatile application, and high energy conversion efficiency. Unfortunately, its widespread application is constrained by critical challenges in hydrogen storage and transportation sectors1. Magnesium (Mg) has emerged as a promising candidate, given its prominent theoretical hydrogen storage capacity (7.6 wt.% H2), environmental friendliness and abundant natural reserves2,3,4,5,6,7,8,9,10. Despite these advantages, Mg faces limitations due to its sluggish reaction kinetics, which stem from multistep reaction processes, including hydrogen adsorption, dissociation, diffusion and nucleation. These processes necessitate high operation temperature (~ 350 °C) and substantial energy loss11,12.
To enhance the kinetic processes, a variety of Mg-based alloys, such as Mg-Ni-RE (RE = rare earths), have been developed13,14,15,16. They undergo in situ decomposition after the activation process and generate Mg, Mg2Ni, and REHx nanoparticles (NPs). In it, Mg contributes hydrogen storage capacity, while REHx and Mg2Ni serve as catalyst phases for hydrogen adsorption and dissociation, thereby enhancing the rates of hydrogenation and dehydrogenation (de/hydrogenation)17,18,19,20,21. However, the hydrogen diffusion in the Mg phase is hardly accelerated by these catalyst phases, and persistent challenges of coarse particle size distribution and suboptimal morphology control are still unresolved22,23.
Nanostructuring strategies demonstrate transformative potential24,25. Nanoparticles increase their surface energies thanks to the unsaturated coordination number, increased grain boundaries and enriched reaction sites/defects, which accelerate hydrogen adsorption and dissociation26,27,28. Furthermore, nanoscale dimensions shorten hydrogen diffusion paths between surface and bulk regions, significantly ramping de/hydrogenation rates29. Combining Ni catalysts with nanostructured Mg simultaneously accelerates hydrogen adsorption, dissociation, diffusion and nucleation. Nanofabrication of Mg can drastically enhance de/hydrogenation rates at much milder reaction temperatures4,5,8,29,30,31,32. Room-temperature reversible hydrogen storage of Mg can even be achieved at particle sizes below ~ 5 nm33. While the current nanofabrication method can improve scalability, it predominantly relies on gas atomization and chemical reduction processes, which requires expensive precursors and complicated synthetic procedures, posing major challenges for industry utilization.
In contrast, mechanical ball milling offers a simpler and cost-effective alternative, and a variety of ball-milling studies on both Mg-based compounds, such as MgH2, have been reported34,35,36,37,38,39. However, unlike the brittle ceramic MgH2, the high ductility of metallic Mg fundamentally hinders its applicability due to severe agglomeration and cold welding40,41. Recent advances in solvent-assisted ball milling show promise. Zou et al.42 successfully synthesized nanoscale Mg via this approach, providing a scalable route for the nanoparticle production of Mg. However, the mechanistic role of solvents in enabling Mg nanosizing is still unclear. In addition, surface instability after ball-milling poses challenges for practical applications. These issues collectively impede the development of milder-temperature operatable Mg-based hydrogen storage materials.
Herein, we report that the partial ionicity induced by solvent adsorption on Mg-based alloy (Mg87.5Ni5.5Y7) can improve nanosizing effects during ball-milling. The solvent-assisted process yielded particles as small as ~ 511.87 nm, representing an 88-fold size reduction compared to solvent-free milling (~ 44.89 μm). The nanostructured Mg87.5Ni5.5Y7 exhibited dehydrogenation rates of 1.71 wt.% min‒1 at 300 °C and 0.46 wt.% min‒1 at 240 °C, surpassing the previously reported related Mg-based alloys. Density functional theory (DFT) calculations revealed that solvent (THF) adsorption on Mg surfaces induces electron redistribution. Mg atoms bonded to oxygen lose 0.56 e⁻, while adjacent Mg atoms gain partial electrons. It gives rise to partial ionicity in Mg (Mgδ+‒Mgδ‒ dipole structure), and as a result, the surface hardness (Vickers hardness) is ~ 22% enhanced by THF-adsorption. The ion migration during balling induces Coulombic repulsion between Mg atoms, maximizing stress-driven structural deformation and subsequent nanoparticle formation. The present data demonstrates the microscopic role of solvents on modifying the material’s surface electronic structure and mechanical properties, offering an affordable strategy for nanoparticle fabrication.
Results and discussion
Solvent-Assisted Ball-Milling of Mg87.5Ni5.5Y7
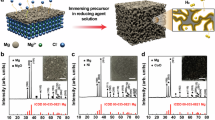
Figure 1a illustrates the powdering process achieved through mechanical impact, such as ball milling, of metallic and ionic compounds. Metallic materials are characterized by delocalized electrons, and structural deformation contributes a little to the metallic bond structure, resulting in more ductile properties. Meanwhile, ionic materials involve both positively and negatively charged ions. The slight modification of crystal deformation immediately gives rise to Coulombic repulsion between identically charged ions and generates cracks. Therefore, it is widely believed that ionic materials are more susceptible to mechanical impact and are easily nanosized by ball-milling. Mg87.5Ni5.5Y7 is one of the Mg-based alloys that is characterized by highly ductile properties. Prior to THF adsorption, ball-milling does not provide a powdering effect to the Mg87.5Ni5.5Y7 alloy, and the mean particle size was estimated to be 44.89 μm (Fig. 1b and Supplementary Figs. S1 and S2). Meanwhile, introducing 1 mL THF significantly enhanced the ball-milling effect. The THF-assisted milling achieved a mean particle size of 511.87 nm, an 88-fold reduction compared to the solvent-free process. Its BET area was measured as 3.72 m2·g‒1 (Fig. 1c). The particle size distribution analysis indicates that 78 % of the particulates fall within the range of 300 ~ 900 nm, highlighting the efficacy of THF-assisted milling in achieving uniform dispersion and considerable nanosizing during the ball-milling process.
a Schematic illustration of mechanical impact to the metallic and ionic compounds. The light blue region represents the distributed free electrons. b SEM image and mean particle size distribution of dry and THF-assisted ball-milled Mg87.5Ni5.5Y7 alloy. The solid lines represent a Gaussian fit. The error bars were given utilizing the standard deviation of 147 particle data. c Adsorption isotherm of dry and THF-assisted ball-milled Mg87.5Ni5.5Y7 alloy. d HRTEM images and corresponding EDS mapping of THF-assisted ball-milled Mg87.5Ni5.5Y7 alloy. The inset represents the (102) lattice plane of Mg. The red, light-blue and green dots represent Mg, Ni and Y elements, respectively. e–g XPS spectra of Mg87.5Ni5.5Y7 NPs. The dots and solid lines represent experimental data and fitting results. During the fitting of the Mg 1 s, Ni 2p, and Y 3 d core-level spectra, the full width at half maximum (FWHM) was constrained to be identical for multiple peaks belonging to the same chemical state. Their area ratios (shaded region) were also fixed with the theoretical ratio.
High-resolution TEM (HRTEM) characterization of the Mg87.5Ni5.5Y7 alloy (Fig. 1d) reveals characteristic lattice fringes with measured d-spacings of 0.19 nm, which corresponds to the (102) plane of hexagonal close-packed Mg. It agrees well with the XRD data, in which Mg and Mg2Ni were observed as major and minor phases, respectively (Supplementary Fig. S3). EDS elemental mapping verifies homogeneous distribution of Mg and Y across the matrix, whereas Ni demonstrates preferential segregation within the Mg2Ni domains. These findings are further supported by XPS analysis of the electronic states of the elements within Mg87.5Ni5.5Y7 NPs. Specifically, Mg displays a Mg0 peak at 1303.5 eV, metallic Ni 2p3/2 signatures at 852.7 eV in Mg2Ni phases, and the Y 3 d spectrum reveals spin-orbit resolved doublets where the 3d5/2 component at 156.8 eV is assigned to Yδ+ (Fig. 1e–g). These XPS characteristics are consistent with Mg-Ni-Y alloys reported elsewhere43,44, demonstrating that the Mg87.5Ni5.5Y7 NPs are not oxidized during the nanosizing process.
It is emphasized that not all solvents can enhance the nanosizing effect of ball-milling. As shown in Supplementary Fig. S4, the addition of cyclohexane exhibits little contribution to ball-milling efficiency, and the observed mean particle size is largely consistent with that of the dry-milled samples. The mechanism difference of THF- and cyclohexane-assisted nanosizing effect will be discussed later.
Hydrolysis corrosion properties
While nanosizing enhances the hydrogen storage capability of Mg-based hydrogen storage materials, it also increases their surface areas, thereby worsening the material’s chemical stability. This is because the increased surface area provides more reactive sites for impurities such as water to form Mg(OH)2. The degradation of Mg2Ni further exacerbates the surface corrosion rates45. To tackle the contradiction between hydrogen storage performance and chemical stability of Mg87.5Ni5.5Y7 NPs, we applied PMMA coatings. In this study, we synthesized Mg87.5Ni5.5Y7 NPs with 0, 0.1, 0.5, and 1.0 wt.% PMMA content and successfully suppressed the surface corrosion rates (Fig. 2a). Uncoated nanoparticles exhibited severe hydrogen corrosion with a rapid hydrogen evolution at 51.0 mL·g‒1·min‒1 during the first 3 minutes (Fig. 2b). Meanwhile, the H2 production rate of Mg87.5Ni5.5Y7 NPs with 0.1, 0.5, and 1.0 wt.% PMMA content were estimated to be 14.7 mL·g‒1·min‒1, 7.0 mL·g‒1·min‒1 and 5.6 mL·g‒1·min‒1, respectively, representing a 71.2–89.0 % reduction compared to the PMMA-free system (Fig. 2b and Supplementary Figs. S5 and S6). The suppression of H2 evolution is interpreted by the polymer’s (e.g., PMMA) protection effect that suppress direct contact between Mg87.5Ni5.5Y7 NPs and water, which is consistent with previous studies4,22,46,47,48,49.
a Kinetic hydrogen generation via hydrolysis measured at room temperature. b Hydrogen generation rate in alloys with varied PMMA additions. The inset shows actual photographs of tested Mg87.5Ni5.5Y7 NPs coated with different PMMA coatings. The samples were placed on weighing paper with a size of 9.8 cm × 9.8 cm. c, d compare the XRD data of uncoated and 0.1 wt.% PMMA coated Mg87.5Ni5.5Y7 NPs after water and after two weeks of exposure to air. The reference peaks of Mg and Mg2Ni were shown as gray and blue solid lines. The club symbol represents the peak position of Mg(OH)2.
Figure 2c shows the XRD pattern of water-exposed uncoated and 0.1 wt.% PMMA coated Mg87.5Ni5.5Y7 NPs. While the uncoated samples exhibit major Mg(OH)2 peaks, the corresponding peaks are much suppressed for the 0.1 wt.% PMMA-coated samples. To further confirm the surface protection effect of PMMA, we exposed the uncoated and 0.1 wt.% PMMA-coated Mg87.5Ni5.5Y7 NPs under an ambient atmosphere for two weeks (Fig. 2d). Again, a notable Mg(OH)2 peak was confirmed for the uncoated samples, due to the reaction with air moisture. Meanwhile, the XRD is largely unchanged for 0.1 wt.% PMMA-coated Mg87.5Ni5.5Y7 NPs, and both Mg(OH)2 and MgO peaks were not identified. These data demonstrated improved surface stability by the PMMA coating.
Hydrogen storage performances
To investigate the effects of nanosizing and surface modification on the dehydrogenation kinetics of Mg87.5Ni5.5Y7, we conducted dehydrogenation experiments on samples with and without PMMA coating. Prior to the kinetics analysis, all of the samples were activated utilizing de/hydrogenation cycles. While Mg87.5Ni5.5Y7 NPs (THF-assisted ball-milled samples) are easily activated, the dry-milled samples cannot be activated under the same conditions (Supplementary Fig. S7). After the activation, the Mg87.5Ni5.5Y7 NPs transform to MgH2‒Mg2Ni‒YH3 nanocomposite, whose BET surface area is 9.51 m2·g‒1 (Supplementary Fig. S8). In it, both Mg2Ni and YHx exhibit improved hydrogen adoption and dissociation capability, which serve as catalytically active phases (Supplementary Fig. S9). In YH3, hydrogen deficiencies are required to serve as a catalytically active phase. The de/hydrogenation kinetics of dry-milled sample is fiarly worse than that of Mg87.5Ni5.5Y7 NPs (Supplementary Fig. S7). The dehydrogenation kinetics were analyzed using the JMAK model and the Arrhenius equation to determine the apparent activation energy, and the isothermal kinetics were further explored using the Sharp and Jones method (Supplementary Table S1).
The results revealed that Mg87.5Ni5.5Y7 NPs achieved 5.15 wt.% H2 released within 3 minutes at 300 °C, reaching 95 % of its theoretical capacity, and released 4.85 wt.% H2 in 15 min at 240 °C (Fig. 3a). The analysis indicated its apparent activation energy of 69.9 kJ·mol‒1, significantly lower than 158.0 kJ·mol−1 reported for bulk MgH2 (Fig. 3g)50. The isothermal kinetic analysis of Mg87.5Ni5.5Y7 NPs showed that the experimental data were well fitted to the A2 model, suggesting that nucleation-and-growth of Mg/MgH2 phases is the rate-determining step, consistent with previously reported Mg-TM-RE (TM = transition metals, RE=rare earths) hydrogen storage alloys (Supplementary Table S2 and Supplementary Fig. S10)19. These findings demonstrate that nanosizing Mg87.5Ni5.5Y7 notably improves the dehydrogenation kinetics by increasing surface reactivity and reducing diffusion barriers, compared with the reported Mg-TM-RE systems (Fig. 3h and Supplementary Table S3)51,52,53,54,55.
a–c Isothermal dehydrogenation kinetic curves of the nanostructured Mg87.5Ni5.5Y7 alloy with three different PMMA protective layer contents. The isothermal dehydrogenation measurements were conducted under the initial pressure of 0.01 bar. d–f JMAK plots of corresponding kinetic data. g Arrhenius’s plots obtained from JMAK plots. h Hydrogen desorption performance comparison at 280 °C between the nanostructured Mg87.5Ni5.5Y7 alloy developed in this work and other reported Mg-based alloys51,52,53,54,55. The data of dry-milled and THF-assisted samples were drawn utilizing blue bars, while the reference data were illustrated utilizing light-blue bars, respectively. The red dots illustrate the mean particle size of the compared samples.
To explore the impacts of PMMA coating on dehydrogenation kinetics, we synthesized Mg87.5Ni5.5Y7 NPs with varying PMMA thicknesses and tested their dehydrogenation capabilities from 240 °C and 300 °C. At 300 °C, 0.1-PMMA@Mg87.5Ni5.5Y7 released 4.85 wt.% H2, while 0.5-PMMA@Mg87.5Ni5.5Y7 showed reduced capacity of 3.65 wt.% H2, with 83.5 kJ·mol‒1 and 81.3 kJ·mol‒1, respectively (Fig. 3b–g). This temperature-dependent trend persisted at 240 °C, with desorption capacities decreasing to 4.46 wt.% and 3.61 wt.% H2, respectively, maintaining the inverse correlation between PMMA loading and hydrogen release efficiency. The kinetic model was fitted to the R2 model (Supplementary Table S4 and Supplementary Fig. S11), indicating that interfacial chemical reactions are the rate-determining step. Accordingly, the PMMA layer on the alloy surface impeded hydrogen molecule access to active sites, thereby suppressing the interfacial reaction kinetics. The suppressed dehydrogenation kinetics corroborate this mechanism observed for 1.0-PMMA@Mg87.5Ni5.5Y7. It only releases 1.94 wt.% H2 at 300 °C and 1.76 wt.% H2 at 240 °C. The excessive coating of 1.0 wt.% PMMA caused a 63.1% hydrogen capacity loss and a 28.0 ~ 52.8 % increase in the activation energy (Supplementary Fig. S12). Supplementary Fig. S13 shows the equilibrium pressures of uncoated and 0.1-PMMA@Mg87.5Ni5.5Y7. The reaction enthalpy obtained from van’t Hoff equation shows the nanosizing effect affect little to the thermodynamic properties.
To investigate the effects of different PMMA concentrations on the microstructure and surface chemical states of dehydrogenated Mg87.5Ni5.5Y7 alloys, we systematically analyzed the dehydrogenated samples. All the samples predominantly consist of Mg, Mg2Ni, and YH3 phases after dehydrogenation (Supplementary Fig. S14). The YH3 phase, formed during hydrogen absorption, remains stable without decomposition during the dehydrogenation process. SEM images and EDS elemental mappings of dehydrogenated alloys confirm that the modified samples with varying PMMA concentrations maintain a fine particulate morphology after dehydrogenation (Supplementary Fig. S15). These results indicate that the Mg87.5Ni5.5Y7 alloy maintains a relatively stable phase composition after dehydrogenation, and the different PMMA concentrations have minimal impact on the phase distribution.
HRTEM-EDS mapping (Supplementary Fig. S16) reveals uniform dispersion of Mg and Y elements, whereas Ni predominantly segregates as Mg2Ni clusters within the alloy matrix after dehydrogenation. XPS analysis of uncoated and 0.1 wt.% PMMA-coated samples reveals that the Mg 1 s spectra resolves dual peaks, i.e., a metallic magnesium component at 1303.5 eV and an oxidized magnesium species at 1306.3 eV (Supplementary Fig. S17). This indicates that partial surface oxidation exists. Meanwhile, the 0.5 and 1.0 wt.% PMMA samples show a single Mg 1 s peak at 1303 eV, verifying complete dehydrogenation without detectable oxidation. The Ni 2p spectra consistently displays spin-orbit doublets (2p3/2 and 2p1/2) accompanied by two characteristic satellite peaks across all compositions. The dominant 2p3/2 component at 852.5 eV corresponds to Mg‒Ni intermetallic bonding, confirming the structural integrity of intermetallic phases during dehydrogenation. The Y 3 d spectra reveals spin-orbit split 3d5/2 and 3d3/2 doublets, where the 3d5/2 peak at 157.2 eV is assigned to Y‒H bonding configurations. Optimizing the concentration of PMMA is critical to balance surface protection with hydrogen storage performance. Based on our preliminary analysis of samples with varying PMMA contents, 0.1-PMMA@Mg87.5Ni5.5Y7 exhibited an optimal combination of high hydrogen storage capacity and surface stability. Hence, we selected it for in-depth characterization to further elucidate its cyclic hydrogen storage performance and structural stability.
The cycle stability of uncoated and 0.1-PMMA coated Mg87.5Ni5.5Y7 NPs is presented in Fig. 4a. To each sample, cycle stability was evaluated up to 500 cycles. Both samples exhibit excellent cycling stability, while the uncoated Mg87.5Ni5.5Y7 exhibits persistent degradation in terms of hydrogen storage capacity and reaction rates (Fig. 4a and b). After experiencing 500 cycles, the hydrogen storage capacity changed from 4.86 wt.% to 4.52 wt.%. Notable degradation of hydrogen storage capacity and reaction rates were not identified for the 0.1-PMMA coated Mg87.5Ni5.5Y7 (Fig. 4a and b). The 0.1 wt.% PMMA-coated Mg87.5Ni5.5Y7 material outperforms previously reported Mg-based systems in both de/hydrogenation kinetics and cycling stability (Fig. 4c)56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80. These results collectively confirm that the PMMA-coated nanocrystalline alloy powder possesses exceptional chemical stability and remarkable cyclic durability under repeated hydrogen cycling conditions.
a Cycle stability of uncoated and 0.1-PMMA coated Mg87.5Ni5.5Y7 NPs. The hydrogenation and dehydrogenation reactions were conducted at 280 °C under 30 bar and 0.01 bar, respectively. The inset emphasizes the hydrogen storage capacity degradation tendency between PMMA uncoated (blue dots) and 0.1 wt.% coated (green dots) samples. Evolution of their de/hydrogenation kinetics are shown in (b). The (c) compares the cycle stability with other Mg-based hydrogen storage materials56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80.
Figure 5a shows the Mg, Ni and Y XPS data of 50 and 500 times cycled uncoated Mg87.5Ni5.5Y7 NPs. The Mg 1 s spectrum exhibits a metallic Mg0 peak centered at 1303.5 eV, indicating minimal surface oxidation. The Ni 2p spectrum was deconvoluted into Mg‒Ni alloy components with two satellite peaks, confirming the presence of Mg‒Ni intermetallic bonding. The Y 3 d spectrum revealed a Y‒H bonding doublet, suggesting the presence of YH3 species. These are consistent with XRD data of cycled samples (Supplementary Figs. S14 and S18), showing that the Mg, Mg2Ni and YH3 are the major phases for the dehydrogenated samples. It should be highlighted that the XPS data is largely unchanged between the 50th and 500th samples for the uncoated Mg87.5Ni5.5Y7 NPs (Fig. 5a). These suggest that the degradation of hydrogen storage capability is likely associated with the structural or morphological evolution of materials. Figure 5b, c show the TEM images of uncoated and 0.1 wt.% PMMA-coated Mg87.5Ni5.5Y7 NPs after 500 cycles. While the chemical composition keeps relatively uniform, clear particle growth can be identified for the uncoated Mg87.5Ni5.5Y7 NPs relative to the 50th cycled sample (Supplementary Fig. S19), leading to the degraded cycle properties. Meanwhile, the 0.1 wt.% PMMA coated sample kept its finer particle morphology even after the long-cycle experiment (Fig. 5c). These data show that PMMA coating not only improves chemical stability but also helpful to keep particle morphology during long-cycle operation.
a The XPS data of 50 and 500 times cycled Mg87.5Ni5.5Y7 NPs. The dots and solid lines represent experimental data and data fitting. During the fitting of the Mg 1 s, Ni 2p, and Y 3 d core-level spectra, the full width at half maximum (FWHM) was constrained to be identical for multiple peaks belonging to the same chemical state. Their area ratios (shaded region) were also fixed to the theoretical value. b, c TEM-EDS data of 500-cycled uncoated Mg87.5Ni5.5Y7 and 0.1-PMMA@Mg87.5Ni5.5Y7 NPs. The red, light-blue and green dots in the EDS represent Mg, Ni and Y signals, respectively.
Solvent-mediated partial ionicity
To elucidate the formation mechanism of nanostructures, we calculated the adsorption energies of THF at four distinct sites on the Mg (0001) surface, namely FCC-hollow, Bridge, HCP-hollow and on-top sites. Figure 6a shows that the most stable adsorption configuration occurs at the on-top site above Mg atoms, with an adsorption energy of ‒0.48 eV. Bader charge analysis (Fig. 6b and Supplementary Fig. S20) combined with charge distribution difference calculations reveal that upon THF adsorption, the Mg atom coordinating with the oxygen loses 0.56 electrons (manifested as positive charge states in blue), while neighboring Mg atoms acquire 0.03-0.08 electrons, giving rise to the Mgδ+‒Mgδ‒ dipole structure. Additionally, Y-doping induces charge redistribution in proximal Mg atoms, with electron transfer quantities as quantified (Supplementary Fig. S21). Different views of the THF adsorption structure at the on-top site of Mg and the Y-doped structure are shown in Supplementary Fig. S22. Indeed, XPS analysis on the as milled samples show the presence of both positively and negatively charged Mg, consistent with the DFT calculations (Supplementary Fig. S23). The synergistic interplay between THF adsorption and Y-doping induces localized charge redistribution in adjacent Mg atoms, giving rise to the partial ionicity between Mg atoms.
a Calculated adsorption energies of THF at four different sites on the Mg (0001) surface. b Bader charge analysis and charge density difference analysis, along with the differential charge density analysis, with accompanying visualizations of the top view of THF on Mg and the top view of the Y-doped structure. c The microstructure of studied Mg87.5Ni5.5Y7 alloy before and after Vickers hardness test. Both clean surface and THF-adsorbed surface were tested. d The summary of the Vickers hardness test. For each sample, 120 tests were performed and utilized for statics evaluation. The box plot depicts the median (solid line), mean (dashed line), the 25th and 75th quartiles (box), respectively. Error bars were derived from the standard deviation. e Proposed solvent-assisted mechanical nanosizing mechanism. It is considered that the adsorbed THF gives rise to electron distribution on Mg surface. It resulted in harder surface properties and made the Mg-based alloy being more susceptible to mechanical impact.
Subsequently, we discuss the impact of cyclohexane adsorption on Mg-based alloy. Unlike THF, cyclohexane is characterized by less polarized molecules and exhibits much weaker interaction (Supplementary Figs. S24 and S25). As a result, adsorption of cyclohexane on Mg-alloy does not cause drastic charge redistribution as was observed for THF, and this is why cyclohexane does not obviously contribute to mechanical nanosizing efficiency (Supplementary Fig. S4).
Such partial ionicity should suppress the ductility of Mg metal and maximize the ball milling-driven structural deformation that facilitates ion migration. The modified surface mechanical properties are indeed demonstrated utilizing Vickers hardness measurements. As shown in Fig. 6c and d, the notable enhancement of Vickers hardness was identified for the THF-adsorbed sample. It achieved an average hardness of 103.1 HV, representing 21.9 % enhancement compared to the untreated surface (84.6 HV) (Supplementary Table S5). These results indicate that the Mg surface becomes less ductile and thus more susceptible to mechanical impact, leading to enhanced ball-milling efficiency. Based on the above-mentioned results, we propose the mechanism of solvent-assisted mechanical nanosizing processes in Fig. 6e. First, solvent molecules, such as THF, adsorb on the metal surface. It gives rise to an electron distribution that enhances surface hardness, making the metal powder more susceptible to mechanical impact. Subsequently, the cracks are easier to generate, and the solvent molecules can be adsorbed on the fresh surface, further enabling mechanical nanosizing.
In summary, we successfully prepared Mg87.5Ni5.5Y7 NPs utilizing solvent-assisted mechanical ball-milling. It was found that the adsorption of solvents, such as THF, induces weak positive and negative charge distribution on the Mg surface. This partial ionicity enhances the surface hardness of Mg-based alloys, thereby enhancing mechanical nanosizing effect by the factor of 88. The nanostructured Mg87.5Ni5.5Y7 alloy exhibited exceptional dehydrogenation kinetics, releasing 5.22 wt.% H2 within 5 min at 280 °C and 4.85 wt.% H₂ in 15 min even at 240 °C, exceeding previously reported Mg-based hydrogen storage alloys. The Mg87.5Ni5.5Y7 NPs also exhibit improved chemical and cycle stability, which is further improved by PMMA coating. Notable kinetic and capacity degradation were not identified for the PMMA-coated Mg87.5Ni5.5Y7 NPs, at least for 500 cycles. The present data reveals how the charge rearrangement caused by metal-solvent interfaces affect surface mechanical properties and improve nanofabrication efficiency of ball-milling, which is even applicable to ductile Mg-based alloys, offering an affordable and scalable approach for nanofabrication of inorganic materials.
Methods
Sample preparation
The Mg87.5Ni5.5Y7 alloy was synthesized via vacuum induction melting under a high-purity argon atmosphere. Nanostructured alloy powder was fabricated via solvent-assisted ball milling with the following protocol. In it, 0.25 g of alloy powder was weighed into a 100 ml ball milling jar, followed by uniform dispersion of 1 mL tetrahydrofuran (THF) onto the powder surface using a calibrated micropipette. Ball milling was performed for 20 hours under an argon atmosphere. During the ball-milling, ball-to-powder ratio was set to 40:1 with the rotational speed of 400 rpm. 10 min pause was introduced between every 10 min milling to prevent overheating.
Three polymethyl methacrylate (PMMA, Mw ~ 120,000, Aladdin, Shanghai) solutions with distinct concentrations (0.00025 g/mL, 0.00125 g/mL, 0.0025 g/mL) were prepared by dissolving 0.005 g, 0.025 g, and 0.050 g PMMA in 20 mL anhydrous tetrahydrofuran (99.5%, Macklin). The mixtures were magnetically stirred in argon-protected containers for 12 h to ensure complete dissolution. Three gradient-concentration PMMA solutions were mixed with the Mg87.5Ni5.5Y7 NPs for PMMA coating. The experimental parameters are summarized in Supplementary Table S6. For the test of cyclohexane-assisted ball-milling the solvent was changed to cyclohexane (99.9 %, Macklin) while other conditions were retained.
Material characterization
The phase composition of the alloy was characterized by X-ray diffraction (XRD, Rigaku, Cu Kα radiation) at a step size of 0.02° and a scanning rate of 5°·min‒1. The morphological characteristics and elemental distribution of the samples were analyzed before and after hydrogen storage performance tests using environmental scanning electron microscopy (ESEM, Quattro S) and transmission electron microscopy (TEM; Talos F200S). The error bars of particle distribution were estimated utilizing Gaussian fit with statistics from n = 147. The elemental composition and stoichiometry of the samples were determined by X-ray photoelectron spectroscopy (XPS, Thermo K-Alpha, Thermo Fisher Scientific). The C 1 s peak (284.8 eV) was used as the reference. During the fitting of the Mg 1 s, Ni 2p, and Y 3 d core-level spectra, the full width at half maximum (FWHM) was constrained to be identical for multiple peaks belonging to the same chemical state, and their area ratios were fixed. For the Ni 2p and Y 3 d doublet, the area ratio and splitting set at reference values, thereby ensuring the accuracy of the results.
Vickers hardness testing using a 300 g load and a holding time of 10 s was carried out on a Micro Vickers hardness testing system (UHV-1000F). For each sample, 120 tests were performed and utilized for statics estimation, such as median/mean values, the 25th/75th quartiles and standard deviation. Prior to the Vickers hardness test, the alloys surface was polished. THF was then sprayed to the surface, and the excess THF was dried under an ambient condition.
Hydrolysis corrosion experiments
The hydrolysis corrosion reaction was tested in a hydrolysis H2 generation device based on the drainage method. The equipment consists of a reaction device and a data logger. The amount of gas released was measured by the mass of water discharged from the gas scrubber bottle, which was weighed with a balance throughout. The weight of deionized water was obtained automatically by an electronic balance connected to a computer, which stored data and reaction times. Then, the generation rate-time curve corresponding to the hydrolysis H2 generation process was obtained. For hydrolysis corrosion reaction, in a typical experiment, ~ 0.05 g of the sample was loaded in a 100 ml flask bottle, and 50 ml deionized water was added to start the reaction at room temperature and under ambient atmosphere. The flask was immersed in a circulating water bath to control reaction temperature and put over a magnetic stirrer to agitate the reactions. The background flow (0.15 ml·min‒1) was subtracted from the data.
Hydrogen storage performance measurements
The isothermal hydrogenation and dehydrogenation characteristics of the samples, including cycling curves, were investigated using a commercial Sieverts-type apparatus (PCT Pro, Setaram and H-Sorb 4600PCT PRO, Ciqtec). The background hydrogen pressures were set to 30 bar for hydrogenation and 0.01 bar for dehydrogenation processes, respectively. Prior to hydrogen absorption/desorption kinetics testing, the alloy samples were subjected to 5 activation cycles at 300 °C.
To better understand the dehydrogenation kinetic behavior, the equations associated with Sharp’s and Jone’s method model (equation 1 and 2) were used to fit the isothermal dehydrogenation curves at different temperatures81,82
where dα is the differential of conversion extent, dt is the differential of time, k is the reaction rate constant, f(α) is a function that depends on a specific kinetic mechanism, A represents a constant related to the kinetic mechanism, and t0.5 represents the time when a equals to 0.5. The kinetic model was obtained by plotting the experimental value (t/t0.5)exp against the theoretical value (t/t0.5)theo.
The PCT curves of samples in different cycle stages were tested at a temperatures of 280 °C, 300 °C, 320 °C, 340 °C and hydrogen pressures ranging from 0.02 to 3 MPa. The reaction enthalpy (ΔH) was then calculated utilizing van’t Hoff equation.
where PH2 denotes the equilibrium pressure of the Mg/MgH2 reaction, ΔH represents the enthalpy change, ΔS is the entropy change, T is the testing temperature, R is the gas constant [8.314 J/(mol·K)], and P0 the standard atmospheric pressure (~ 1.01 × 105 Pa).
DFT studies
All structural relaxations and electronic structure calculations were carried out using density functional theory (DFT) as implemented in the Vienna Ab initio Simulation Package (VASP)83. Based on the optimized bulk lattice parameters, a 6 × 6 Mg(0001) surface model was constructed for the simulations, with a vacuum layer of 15 Å introduced to eliminate interlayer interactions. The supercell consists of five atomic layers, with the bottom three layers fixed to mimic the bulk behavior and improve computational efficiency. During geometry optimization, the plane-wave cutoff energy was set to 450 eV, and a Monkhorst-Pack k-point mesh of 5 × 5 × 1 was used. The convergence criteria for electronic self-consistent iteration and ionic relaxation were set to 10−6 eV per atom for the total energy and 0.01 eV·Å−1 for the maximum atomic force, respectively. The exchange-correlation functional was treated within the generalized gradient approximation (GGA) using the Perdew-Burke-Ernzerhof (PBE) formulation84,85,86. The electron charge density was analyzed using the Bader charge analysis method combined with charge density difference calculations. For the transition state analysis of the hydrogen dissociation reaction, the climbing image nudged elastic band (CI-NEB) was employed to identify saddle points and minimum energy paths (MEPs)87,88. The number of CI-NEB images was determined using the VASP-VTST978 script, based on the interatomic distances between the initial and final states. All transition-state models were fully optimized until the energy and force convergence thresholds of 1 × 10−6 eV and 0.01 eV·Å−1, respectively, were reached. The adsorption energy Ead(X) of X (X = tetrahydrofuran or cyclohexane) on the surface is defined as Ead(X) = Etot(X/sub)-E(sub)-E(X), where Etot(X/sub) is the total energy of the optimized configuration of the X molecule adsorbed on the substrate, E(sub) is the total energy of the clean substrate, and E(X) is the total energy of an isolated X molecule.
Data availability
The data generated in this study are presented in the main text, Supplementary Information and Source Data file. Source data are provided in this paper.
References
Allendorf, M. D. et al. Challenges to developing materials for the transport and storage of hydrogen. Nat. Chem. 14, 1214–1223 (2022).
Guan, H., Liu, J., Li, Q., Lu, Y. & Pan, F. Chemically stable TMO @Ti-MgO (TM = Mn and Cu) catalyst enhanced De/hydrogenation kinetics of Mg/MgH2. J. Magnes. Alloys https://doi.org/10.1016/j.jma.2025.02.032 (2025).
Guan, H. et al. Titanium-nickel dual active sites enabled reversible hydrogen storage of magnesium at 180 °C with exceptional cycle stability. Adv. Mater. 37, 2500178 (2025).
Jeon, K.-J. et al. Air-stable magnesium nanocomposites provide rapid and high-capacity hydrogen storage without using heavy-metal catalysts. Nat. Mater. 10, 286–290 (2011).
Kim, H. et al. Facile synthesis of nanoporous Mg crystalline structure by organic solvent-based reduction for solid-state hydrogen storage. Nat. Commun. 15, 10800 (2024).
Larsson, P., Araújo, C. M., Larsson, J. A., Jena, P. & Ahuja, R. Role of catalysts in dehydrogenation of MgH2 nanoclusters. Proc. Natl. Acad. Sci. USA 105, 8227–8231 (2008).
Li, Q. et al. Thermodynamics and kinetics of hydriding and dehydriding reactions in Mg-based hydrogen storage materials. J. Magnes. Alloys 9, 1922–1941 (2021).
Wang, H. et al. Thermodynamic and kinetic regulation for Mg-based hydrogen storage materials: challenges, strategies, and perspectives. Adv. Funct. Mater. 34, 2406639 (2024).
Zhang, X. et al. Atomic reconstruction for realizing stable solar-driven reversible hydrogen storage of magnesium hydride. Nat. Commun. 15, 2851 (2024).
Zhao, X. et al. Tunable metal-hydrogen bonding in Cu-Ru catalysts enables selective hydrogen storage reactions in Mg-based composite. Adv. Funct. Mater. 35, 2505352 (2025).
Han, Z., Wu, Y., Yu, H. & Zhou, S. Location-dependent effect of nickel on hydrogen dissociation and diffusion on Mg (0001) surface: Insights into hydrogen storage material design. J. Magnes. Alloys 10, 1617–1630 (2022).
Xinglin, Y., Xiaohui, L., Jiaqi, Z., Quanhui, H. & Junhu, Z. Progress in improving hydrogen storage properties of Mg-based materials. Mater. Today Adv. 19, 100387 (2023).
Chen, Y. et al. Hydrogen Storage in Mg-Ni-Type Alloys with La and Sm Incorporation. ACS Appl. Energy Mater. 7, 8858–8868 (2024).
Liang, G. Y., Wu, D. C., Li, L. & Huang, L. J. A discussion on decay of discharge capacity for amorphous Mg-Ni-Nd hydrogen storage alloy. J. Power Sources 186, 528–531 (2009).
Liu, J. W., Zou, C. C., Wang, H., Ouyang, L. Z. & Zhu, M. Facilitating de/hydrogenation by long-period stacking ordered structure in Mg based alloys. Int. J. Hydrogen Energy 38, 10438–10445 (2013).
Yin, J., Yamada, T., Yoshinari, O. & Tanaka, K. Improvement of hydrogen storage properties of Mg-Ni alloys by rare-earth addition. Mater. Trans. 42, 712–716 (2001).
Li, Y., Gu, Q., Li, Q. & Zhang, T. In-situ synchrotron X-ray diffraction investigation on hydrogen-induced decomposition of long period stacking ordered structure in Mg-Ni-Y system. Scr. Mater. 127, 102–107 (2017).
Luo, Q. et al. Achieving superior cycling stability byin situforming NdH2-Mg-Mg2Ni nanocomposites. J. Mater. Chem. A 6, 23308–23317 (2018).
Sun, X. et al. Hot extrusion-induced Mg-Ni-Y alloy with enhanced hydrogen storage kinetics. J. Mater. Sci. Technol. 202, 119–128 (2024).
Spassov, T., Rangelova, V. & Neykov, N. Nanocrystallization and hydrogen storage in rapidly solidified Mg-Ni-RE alloys. J. Alloys Compounds 334, 219–223 (2002).
Tanaka, K. et al. Improvement of hydrogen storage properties of melt-spun Mg-Ni-RE alloys by nanocrystallization. J. Alloys Compounds 293-295, 521–525 (1999).
Li, K., Huang, T., Gao, Y. & Zhou, C. Enhancing antioxidant properties of hydrogen storage alloys using PMMA coating. Int. J. Hydrogen Energy 48, 4339–4348 (2023).
Ranjbar, A. et al. Comparison of hydrogen storage properties of Mg-Ni from different preparation methods. Mater. Chem. Phys. 127, 405–408 (2011).
Yu, X., Tang, Z., Sun, D., Ouyang, L. & Zhu, M. Recent advances and remaining challenges of nanostructured materials for hydrogen storage applications. Prog. Mater. Sci. 88, 1–48 (2017).
Zhou, C. et al. In situ formation of nanocrystalline MgH2 through room temperature hydrogenation. Mater. Des. 218, 110729 (2022).
Rolison, D. R. et al. Multifunctional 3D nanoarchitectures for energy storage and conversion. Chem. Soc. Rev. 38, 226–252 (2009).
Xu, C. et al. Graphene-based electrodes for electrochemical energy storage. Energy Environ. Sci. 6, 1388–1414 (2013).
Zhou, Z.-Y., Tian, N., Li, J.-T., Broadwell, I. & Sun, S.-G. Nanomaterials of high surface energy with exceptional properties in catalysis and energy storage. Chem. Soc. Rev. 40, 4167–4185 (2011).
Zhu, W. et al. Nanoconfined and in situ catalyzed MgH2 self-assembled on 3D Ti3C2 MXene folded nanosheets with enhanced hydrogen sorption performances. ACS Nano 15, 18494–18504 (2021).
Li, W., Li, C., Ma, H. & Chen, J. Magnesium nanowires: enhanced kinetics for hydrogen absorption and desorption. J. Am. Chem. Soc. 129, 6710–6711 (2007).
Norberg, N. S., Arthur, T. S., Fredrick, S. J. & Prieto, A. L. Size-Dependent hydrogen storage properties of Mg nanocrystals prepared from solution. J. Am. Chem. Soc. 133, 10679–10681 (2011).
Qin, J. et al. Construction of Mg/Zr superlattice structure to achieve efficient hydrogen storage via atomic-scale interaction in Mg-Zr modulation films. Acta Materialia 263, 119470 (2024).
Zhang, X. et al. Realizing 6.7 wt% reversible storage of hydrogen at ambient temperature with non-confined ultrafine magnesium hydrides. Energy Environ. Sci. 14, 2302–2313 (2021).
Banrejee, S., Kumar, A., Ruz, P. & Sudarsan, V. Improvement of hydrogen storage characteristics of catalyst free magnesium nanoparticles prepared by wet milling. Int. J. Energy Res. 45, 17597–17608 (2021).
Cui, J. et al. Remarkable enhancement in dehydrogenation of MgH2 by a nano-coating of multi-valence Ti-based catalysts. J. Mater. Chem. A 1, 5603–5611 (2013).
Gao, G. et al. Improvement on hydrogen storage performance of MgH2 by THF-promoted nano-crystallization under low-speed ball milling. Rare Metals 44, 6366–6374 (2025).
Paik, B. et al. Evolution of microstructure in MgH2 powder particles during high energy ball milling and hydrogen cycling. J. Alloys Compounds 492, 515–520 (2010).
Porcheddu, A., Cincotti, A. & Delogu, F. Kinetics of MgH2 formation by ball milling. Int. J. Hydrogen Energy 46, 967–973 (2021).
Schimmel, H. G. et al. Structural information on ball milled magnesium hydride from vibrational spectroscopy and ab-initio calculations. J. Alloys Compounds 393, 1–4 (2005).
Tian, M. & Shang, C. Nano-structured MgH2 catalyzed by TIC nanoparticles for hydrogen storage. J. Chem. Technol. Biotechnol. 86, 69–74 (2011).
Varin, R. A., Czujko, T., Chiu, C. & Wronski, Z. Particle size effects on the desorption properties of nanostructured magnesium dihydride (MgH2) synthesized by controlled reactive mechanical milling (CRMM). J. Alloys Compounds 424, 356–364 (2006).
Zou, R. et al. One-step synthesis of light metal nanoparticle from metastable complex. Small 19, 2206518 (2022).
Zhang, T., Song, W., Kou, H. & Li, J. Surface valence transformation during thermal activation and hydrogenation thermodynamics of Mg-Ni-Y melt-spun ribbons. Appl. Surface Sci. 371, 35–43 (2016).
Zhou, H. et al. Enhancement of hydrogen storage properties from amorphous Mg85Ni5Y10 alloy. J. NonCrystalline Solids 605, 122167 (2023).
Guan, H. et al. Dipole Coupling Accelerated H2O Dissociation by Magnesium-Based Intermetallic Catalysts. Angew. Chem. Int. Ed. 63, e202400119 (2024).
Hashimoto, T. & Notomi, M. Hydrogen storage properties of Mg-based multilayer films. Mech. Eng. J. 3, 16-00228 (2016).
Shen, C. & Aguey-Zinsou, K.-F. Electrochemical deposited Mg-PPy multilayered film to store hydrogen. Int. J. Hydrogen Energy 43, 22385–22390 (2018).
Yao, L. et al. Improved dehydriding property of polyvinylpyrrolidone coated Mg-Ni hydrogen storage nano-composite prepared by hydriding combustion synthesis and wet mechanical milling. Prog. Nat. Sci. Mater. Int. 28, 7–14 (2018).
Yuan, J. G., Zhu, Y. F., Li, L. Q., Wu, Y. & Zhou, S. X. Preparation and hydrogen storage property of Mg-based hydrogen storage composite embedded by polymethyl methacrylate. Int. J. Hydrogen Energy 42, 22366–22372 (2017).
Guan, H. et al. Microscopic Scaling Relation of Ti-Based Catalysts in De/Hydrogenation Reactions of Mg/MgH2. ACS Catal.14, 17159–17170 (2024).
Chen, Y. et al. Investigation of microstructure characteristics, kinetics, and thermodynamics of Mg-Ni-RE (RE = Y and RE = Ce) hydrogen storage alloys. Int. J. Hydrogen Energy 69, 1329–1340 (2024).
Lin, M. et al. Hydrogen storage properties of Mg95-xNi5Ndx (x=0, 1, 3, 5) alloys. Int. J. Hydrogen Energy 97, 11–24 (2025).
Yin, Y., Li, B., Yuan, Z., Qi, Y. & Zhang, Y. Enhanced hydrogen storage performance of Mg-Cu-Ni system catalyzed by CeO2 additive. J. Rare Earths 38, 983–993 (2020).
Yong, H. et al. In-situ influence of TiF3 catalyst on the hydrogen storage behavior of Mg-Ni-Y based alloy. J. Energy Storage 77, 110022 (2024).
Yuan, Z. et al. Influence of CeO2 nanoparticles on microstructure and hydrogen storage performance of Mg-Ni-Zn alloy. Mater. Charact. 178, 111248 (2021).
Chen, Y. et al. Mn nanoparticles enhanced dehydrogenation and hydrogenation kinetics of MgH2 for hydrogen storage. Trans.Nonferrous Met.Soc. China 31, 3469–3477 (2021).
Ding, S. et al. A novel carbon-induced-porosity mechanism for improved cycling stability of magnesium hydride. J. Magnes. Alloys 13, 1341–1352 (2025).
Fu, H. et al. Synergistic effect of a facilely synthesized MnV2O6 catalyst on improving the low-temperature kinetic properties of MgH2. ACS Appl. Mater. Interfaces 14, 33161–33172 (2022).
Guo, F., Zhang, T., Shi, L. & Song, L. Hydrogen absorption/desorption cycling performance of Mg-based alloys with in-situ formed Mg2Ni and LaHx (x=2, 3) nanocrystallines. J. Magnes. Alloys 11, 1180–1192 (2023).
Guo, Z. et al. Catalytic phase engineering for enhanced hydrogen storage kinetics in Mg-Ni-Y-Si alloys via solid-solution and reprecipitation. J. Alloys Compounds 1030, 180903 (2025).
Kajiwara, K., Sugime, H., Noda, S. & Hanada, N. Fast and stable hydrogen storage in the porous composite of MgH2 with Nb2O5 catalyst and carbon nanotube. J. Alloys Compounds 893, 162206 (2022).
Lan, Z. et al. Roles of in situ-formed NbN and Nb2O5 from N-doped Nb2C MXene in regulating the re/hydrogenation and cycling performance of magnesium hydride. Chem. Eng. J. 431, 133985 (2022).
Li, B. et al. Synchronously upgrading of hydrogen storage thermodynamic, kinetics and cycling properties of MgH2 via VTiMn catalyst. Sep. Purif. Technol. 355, 129760 (2025).
Li, H. et al. Effect of element substitution on hydrogen storage properties of La-Mg-Ni-based alloys prepare by heat treatment. Intermetallics 184, 108861 (2025).
Li, J. et al. In situ formed MgTi2O4 from MgO improving the cycling stability of MgH2. Chem. Eng. J. 470, 144259 (2023).
Nyahuma, F. M. et al. Significantly improved hydrogen storage behaviors in MgH2 with Nb nanocatalyst. Int. J. Miner. Metall. Mater. 29, 1788–1797 (2022).
Peng, C., Li, Y. & Zhang, Q. Enhanced hydrogen desorption properties of MgH2 by highly dispersed Ni: The role of in-situ hydrogenolysis of nickelocene in ball milling process. J. Alloys Compounds 900, 163547 (2022).
Qin, Y. et al. Constructing VO/V2O3 interface to enhance hydrogen storage performance of MgH2. J. Magnes. Alloys 12, 4877–4886 (2024).
Ren, L. et al. Boosting hydrogen storage performance of MgH2 by oxygen vacancy-Rich H-V2O5 nanosheet as an excited H-Pump. NanoMicro Lett. 16, 160 (2024).
Sun, S. et al. Tailoring hydrogen storage properties in cold rolled Mg-Ni-Si alloys: Microstructural evolution and low-temperature performance optimization. Chem. Eng. J. 521, 166822 (2025).
Tan, D., Peng, C. & Zhang, Q. Microstructural characteristics and hydrogen storage properties of the Mg-Ni-TiS2 nanocomposite prepared by a solution-based method. Int. J. Hydrogen Energy 48, 16756–16768 (2023).
Tanaka, H. et al. Cycle durability of Ca-Mg-Ni alloys and factors which cause degradation of hydrogen storage capacity. Mater. Sci. Eng. B 108, 81–90 (2004).
Verma, S. K., Abu Shaz, M. & Yadav, T. P. Introducing 2D layered WS2 and MoS2 as an active catalyst to enhance the hydrogen storage properties of MgH2. Int. J. Hydrogen Energy 87, 1035–1046 (2024).
Xian, K. et al. A unique nanoflake-shape bimetallic Ti-Nb oxide of superior catalytic effect for hydrogen storage of MgH2. Small 18, 2107013 (2022).
Yang, J. et al. MXene-supported V2O5 nanocatalysts: Boosting hydrogen storage efficiency in MgH2 through synergistic catalysis. J. Energy Storage 130, 117474 (2025).
Zhang, H. et al. Engineering the oxygevacancies in Na2Ti3O7 for boosting its catalytic performance in MgH2 hydrogen storage. ACS Sustain. Chem. Eng. 10, 363–371 (2021).
Zhang, J. et al. Single-atom Ni supported on TiO2 for catalyzing hydrogen storage in MgH2. J. Am. Chem. Soc. 146, 10432–10442 (2024).
Zhang, Z. et al. Improving the hydrogen storage property of Sm6.5Mg93.5-TiMn2 by accelerating H2 dissociation/recombination and H diffusion. ACS Sustain. Chem. Eng. 12, 17702–17712 (2024).
Zhou, X. et al. The anionic Tx defects of Nb2CTx MXene as the effective catalytically active center for the Mg-based hydrogen storage materials. J. Magnes. Alloys 13, 571–582 (2025).
Zou, R. et al. Enhancing the cycling stability of MgH2 using nitrogen modified titanate. J. Mater. Chem. A 11, 11748–11754 (2023).
Jones, L. F., Dollimore, D. & Nicklin, T. Comparison of experimental kinetic decomposition data with master data using a linear plot method. Thermochimica Acta 13, 240–245 (1975).
Sharp, J. H., Brindley, G. W. & Achar, B. N. N. Numerical data for some commonly used solid state reaction equations. J. Am. Ceramic Soc. 49, 379–382 (1966).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Kresse, G. D. J. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. Lett. 59, 1758–1775 (1999).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Henkelman, G. & Jónsson, H. Improved tangent estimate in the nudged elastic band method for finding minimum energy paths and saddle points. J. Chem. Phys. 113, 9978–9985 (2000).
Henkelman, G., Uberuaga, B. P. & Jónsson, H. A climbing image nudged elastic band method for finding saddle points and minimum energy paths. J. Chem. Phys. 113, 9901–9904 (2000).
Acknowledgements
This work was supported by the National Key R&D Program of China (2023YFB3809101). Q.L. acknowledges the support of the National Natural Science Foundation of China (U23A20128) and the Chongqing Science and Technology Commission of China (CSTC2024YCJH-BGZXM0041). We appreciate the Analytical and Testing Center of Chongqing University for their technical support. We are grateful to Yijian Chen for his assistance on DFT calculations.
Author information
Authors and Affiliations
Contributions
Y.L. proposed the idea behind the research. Y.L. and Qi.L. co-supervised the project. T.S. performed the synthesis, characterization, property measurements and DFT calculations. X.S. and Qu.L. conducted Vickers hardness tests. T.S. and Y.Z. analyzed SEM and TEM data. Z.T. and J.L. conducted a long-cycle stability test. T.S., J.L., and Y.L. co-wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Wenjie Song and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Sun, T., Tang, Z., Liu, J. et al. Solvent-mediated partial ionicity enhances mechanical nanosizing effect of Mg-based hydrogen storage alloys. Nat Commun 17, 1688 (2026). https://doi.org/10.1038/s41467-026-68390-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-68390-8