Abstract
The critical progression of structural disorder, which governs the bulk glass-forming ability (GFA), can be elucidated as an ergodicity-breaking process. Understanding the atomic characteristics involved is imperative for establishing advanced glass design principles. However, conventional glasses present significant challenges due to their inherently complex and ambiguous disorder motifs, such as intricate and random atomic clusters or ring distributions. In this work, we synthesize a family of zero-dimensional hybrid metal halides with tunable short- to medium-range structural arrangements to form glasses with diverse GFAs. Through altering molecular shape and surface electrostatic potential of organic cations, their rotational order is able to be broken in a controlled manner. This leads to distinct phase space partitions of molecular movements, allowing for tunable GFAs. Our findings provide a fundamental design approach for synthesizing property-oriented glasses by controlling molecular rotational order, which can be applicable to a wide range of molecular glasses and amorphous solids.
Similar content being viewed by others
Introduction
Glasses, as non-periodic systems, possess inherent disorder and non-equilibrium states1,2,3, which are normally obtained through rapid cooling viscous melts, bypassing the crystallization process. During the liquid-glass transition, the atoms evolve from a non-localized state to a localized arrangement, while the disorder significantly remains4,5,6,7,8,9. From a perspective of dynamics, vitrification can be described as a process where atoms, experiencing any recurring states, are frozen to specific configurations. This is termed as the ergodicity breaking10, wherein structural disorder disperses across discrete phase space partitions, to allow the emergence of various localized motifs with similar energy11,12. Such a unique characteristic heavily depends on the formation dynamics, leading to elusive atomic arrangements in glasses and thus diverse properties13,14,15,16,17,18.
In the context of violating ergodicity, the sampling of adequate number of configurations of atoms and molecules, which determines the pathway of retarding nucleation and crystallization, would be frustrated. As this is strongly dependent on the motif of basic components and nature of atomic/molecular interactions19, how to design and manipulate the primary structural units is the key to engineer properties of glasses. However, the primitive ringed motifs in network-forming inorganic glasses have abundant varieties and are often intimately interwoven20,21,22. This complicates the confirmation of basic structural units and the possibility of modular control. In metallic glasses, the compositional clusters exhibit high diversity in both configurations and numbers of atoms23,24, so they normally only can be statistically described rather than quantitatively resolved25. In this context, selecting prototypical glass systems composed of definite components could provide insight into unravelling the structural and energetic evolution across ergodic-breaking, hence offering possibilities of engineering the formation abilities and properties of glasses.
Zero-dimensional (0D) hybrid metal halides, A2[MnBr4] (A = organic phosphonium cation), are a subclass of molecular solids, where the tetrahedral MnBr4 and pyramidal organic cations are associated via electrostatic interactions26,27,28. These materials form both crystals and glasses fairly easy, but with a wide range of temperatures both for melting and for the forming of the glass state on cooling the molten state (Tm and Tg respectively)29. In the crystalline phases, the length scales of [MnBr4]2− tetrahedra and the phosphonium cations are about 4.0 and 12.0–16.0 Å, respectively. The alternatively distributed [MnBr4]2− moieties show generally similar tetrahedra symmetry across all analogous compounds. The arrangement of the organic cations with distinct shape and polarity, spanning the short-range to medium-range order (MRO) scale within the phase space, is primarily responsible for the glass-forming ability (GFA, Fig. 1a). By thermally quenching the A2MnBr4 melts, the fast flexing and rotating phosphonium cations are frozen, and settled at discrete energy positions in a random manner, which breaks the rotational ordering to form glass. Therefore, controlling the glass formation of A2[MnBr4] can be achieved by manipulating the ergodicity breaking of the organic cation.
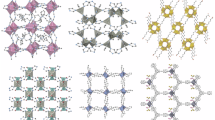
a Schematic patterns of the design strategy. Silver ball: organic components; Yellow ball: inorganic components. In the enlarged diagram, the molecular shape and surface are represented as the core and shell of the ellipsoid, respectively. b Diagram of glass formation through the melt-quenching process. The insets show the rotational order breaking behavior of molecular components during liquid-glass transition. The inorganic components are omitted for clarity. c Structural models for A2[MnBr4] glasses with diverse glass properties attributed to different molecular ergodicity breaking pathways. d Photographs of bulk [BuTPP]2[MnBr4] glass under sunlight and X-ray radiation. e SEM (top) and microscopic (bottom) images under UV light of [BPP]2[MnBr4] fibers.
In this work, nine A2[MnBr4] glasses were synthesized using the melt-quenched method (Fig. 1b, c and Supplementary Fig. 1). By applying molecular design principles, systems formed by different organic phosphonium cations show diverse GFAs. The organic cations, selected with different shapes and molecular electrostatic potential (ESP, describing the static distributions of charge around a molecule), give rise to distinct rotational disorder and broken ergodicity, hence versatile GFAs and properties. These glasses exhibit excellent processability, allowing for manufacturing in both the bulk (Fig. 1d and Supplementary Movie 1) and micron-sized fibers (Fig. 1e and Supplementary Fig. 2). Notably, the halide glasses are highly sensitive to X-ray radiation, making them promising scintillator materials with ease of manufacturing.
Results
Powder X-ray diffraction and pair distribution function (PDF) experiments were conducted before and after the melt-quenching to characterize the vitreous states of the formed glasses. In the case of [TPP]2[MnBr4] (TPP = tetraphenylphosphonium)30, where both the organic cations and MnBr4 anions are tetrahedral, the diffraction pattern of the melt-quenched sample closely resembles that of the parent phase, indicating no formation of glass (Supplementary Fig. 3). Conversely, other melt-quenched compounds, in which one phenyl group of TPP is substituted by aliphatic and benzyl side chains (denoted as Category I and II, Supplementary Data 1), show different behavior. Their diffraction patterns exhibit slight displacement and broadening upon heating (Supplementary Fig. 4), and do not show any significant Bragg peaks after quenching (Fig. 2a, Supplementary Fig. 3, and Supplementary Tables 1–3), indicating the absence of crystallinity. For the modified TPP derivatives, substituting one benzene ring with an aliphatic or benzyl group breaks the molecular symmetry. This modification introduces steric hindrance that prevents the structure from easily reordering into a crystalline phase during quenching. Although these side branches can rotate, their motion may be insufficient to restore the long-range translational symmetry, thereby suppressing the recrystallization process. This, in turn, facilitates the formation of a glassy phase. The observation is substantiated by the isotropic optical feature observed in the glassy samples under polarimetric imaging microscopy (Supplementary Fig. 5).
a The formation of [TPP]2[MnBr4] crystal and the replacement of TPP by other two categories of organic phosphonium cations according to their substituted side chains. b The representative PDF D(r) analysis of crystal and glass samples of [BuTPP]2[MnBr4], [BPP]2[MnBr4] and [BrBPP]2[MnBr4]. c RMC model of ag-[BrBPP]2[MnBr4]. d Tg, Tm and GFAs of A2[MnBr4] glasses extracted from DSC results. The Tg/Tm values of [BuTPP]2[MnBr4], [BPP]2[MnBr4] and [BrBPP]2[MnBr4] increase sequentially. Among the glass-forming compounds, [c-PrMeTPP]2[MnBr4] has the highest Tg/Tm of 0.811, and [BrBPP]2[MnBr4] exhibits the highest Tg (96.9 °C) and Tm (269.4 °C). For detailed information, please see Supplementary Table 4.
The PDF D(r) patterns of the crystalline and glassy samples are broadly similar below 5 Å, while all peaks in the range of 5–30 Å show significant flat feature for the melt-quenched counterparts (Fig. 2b and Supplementary Figs. 6 and 7; the glass form is denoted as ag-A2[MnBr4]). Such phenomena reveal the breakdown of translational symmetry and corresponding loss of the MRO in the structure31. This implies that the disorder is attributed to the randomly-distributed organic cations resulting from the disruption of their freely-rotating molecular states. Additionally, reverse Monte Carlo (RMC) modeling based on experimental PDF data was conducted to elucidate the molecular arrangements of the phosphonium cations and [MnBr4]2− anions. The simulated and experimental D(r) and S(Q) are shown in Supplementary Figs. 8 and 9. The RMC models reveal that both the [MnBr4]2− and phosphonium cations maintain their molecular geometries, while displaying increased configuration and orientation diversity, indicative of the enhanced disorder upon vitrification (Fig. 2c and Supplementary Figs. 10 and 11). To quantify the rotational disorder, the orientational tensor Q was constructed from the RMC-derived models, and the maximum eigenvalue was extracted as the orientational order parameter Smax32:
where u is the unit vector along each P–C bond linking the phosphorus atom to the non-aromatic carbon atom, uT is the transpose of the orientation vector, and I is the identity matrix. Smax ranges from approximately 0.01 to 0.05, indicating pronounced rotational disorder (Supplementary Fig. 12). Lower Smax values, such as those of [c-PrMeTPP]2[MnBr4] and [BuTPP]2[MnBr4], correspond to broader orientational distributions and easier local reorientations. These features reflect weakened cooperative structural constraints, thus tending to reduce Tg while enhancing GFA. Conversely, higher Smax values (e.g., those of [BrBPP]2[MnBr4] and [MeTPP]2[MnBr4]) imply partial orientational alignment and stronger cation–anion interactions, leading to increased local rigidity, higher Tg, and reduced GFA.
The differential scanning calorimetry (DSC) results quantify the cation-dependent Tm and Tg of this family of halides (Fig. 2d, Supplementary Fig. 13, and Supplementary Table 4)33. For Category I halides, their Tg and Tm values lie in the range of 43.0–57.6 °C and 125.2–178.4 °C, respectively. The Tg/Tm ratios, which determine GFA according to the Kauzemann 2/3 law34, range from 0.705 for [EtTPP]2[MnBr4] (EtTPP = ethyl triphenyl phosphonium) to 0.806 for [c-PrMeTPP]2[MnBr4] (c-PrMeTPP = cyclopropyl triphenyl phosphonium). Notably, these Tg/Tm ratios show an overall trend towards easier glass-formation, correlating to the length of the A-site cations. Compared with Category I, the Tg (77.6–96.9 °C) and Tm (218.3–271.1 °C) values of Category II are much higher, whereas their Tg/Tm values (0.680–0.716) are significantly lower. Interestingly, the GFAs show a strong dependence on the “push-pull electronic effect” of the benzyl side chains, as manifested by the larger Tg/Tm index for [MeBPP]2[MnBr4] (0.716, MeBPP = methyl benzyl phosphonium) but lower Tg/Tm values for [FBPP]2[MnBr4] (0.706, FBPP = 6-fluorine benzyl phosphonium) and [BrBPP]2[MnBr4] (0.680, BrBPP = 6-bromine benzyl phosphonium), compared with that of the original [BPP]2[MnBr4] (0.714), respectively. This suggests the push-pull substituent effect adjusts the electron density of the cations and hence modulates the strength of the interactions between inorganic and organic molecular motifs. These electronic variations lead to pronounced differences in GFAs. Furthermore, the activation energies (Eg) derived from the Moynihan and Kissinger analyses exhibit consistent trends (Supplementary Figs. 13–17 and Supplementary Table 5), quantifying the structural relaxation kinetics near Tg35,36,37. Compounds containing rigid or polar substituents exhibit markedly higher Eg values, indicative of slower structural relaxation and stronger resistance to molecular rearrangement. This enhanced rigidity contributes to the elevated Tg observed in these glass systems.
UV–Vis absorption spectra of the crystalline and glassy materials show similar absorption at 320–350 nm (Supplementary Fig. 18). Meanwhile, the Tauc plots analysis reveals direct bandgap transitions for both states (Supplementary Fig. 19). The photoluminescence (PL) and photoluminescence excitation (PLE) spectra at 80 K display broad green emissions at 500–550 nm and excitation bands at 350–450 nm (Supplementary Fig. 20 and Supplementary Table 6), typical of the 4T1 → 6A1 transitions in tetrahedrally coordinated Mn2+. Subsequent luminescence measurements on the nine halide glasses at room temperature demonstrate that they maintained a green luminescence, albeit with significant broadening and a redshift after vitrification (Fig. 3a and Supplementary Figs. 21). This redshift is attributed to the increased depth of the self-trapped exciton state, arising from the shortened Mn–Br distance and the disordered environment around [MnBr4]2− (Supplementary Figs. 22). The redshift behavior agrees with previous reported glasses29,38,39,40,41,42, which were attributed to disorder-induced effects. Notably, the luminescent lifetimes of the glasses are significantly lower than those of the crystalline phases (Fig. 3b and Supplementary Table 7), implying a large increase of non-radiative loss. Compared with the crystalline parents, the luminescent peak widths of the glasses are larger, by 22% for [BuTPP]2[MnBr4] to 33% for [BrBPP]2[MnBr4] (Supplementary Fig. 23 and Supplementary Table 8). This could arise from the small and large increases in the number of [MnBr4]2− configurations during vitrification for [BuTPP]2[MnBr4] and [BrBPP]2[MnBr4], respectively. X-ray excited luminescence and detection limit measurements reveal excellent X-ray response and low detection limits of the A2[MnBr4] glasses, surpassing some prototypical commercial materials. Among them, ag-[MeBPP]2[MnBr4] exhibits an ultralow detection threshold of 23 nGyair s−1, highlighting its high X-ray sensitivity (Supplementary Figs. 24 and 25 and Supplementary Table 9).
a The typical green luminescence emission spectra of [BuTPP]2[MnBr4], [BPP]2[MnBr4], and [BrBPP]2[MnBr4], in the crystal and glass forms, which correspond to the 4T1–6A1 transition of tetrahedrally coordinated manganese ions under UV excitation. b Time-resolved emission spectra in the crystal and glass forms. c Raman spectra of [BuTPP]2[MnBr4], [BPP]2[MnBr4] and [BrBPP]2[MnBr4] in the crystal and glass forms at 20–500 cm−1. d The ratio of average luminescence lifetime τ of different A2MnBr4 glass and crystal systems, and Tg/Tm of different A2[MnBr4] glass systems.
To comprehensively evaluate the scintillation properties of [c-PrMeTPP]2[MnBr4] and ag-[c-PrMeTPP]2[MnBr4], a series of X-ray excited luminescence and kinetic tests were conducted (Supplementary Fig. 26). The emissions exhibit a linear dependence under dose rate of 2.94–366 mGyair s−1, suggesting stable X-ray responses. The detection limits, which are determined from a signal-to-noise of 3, are 131 and 110 nGyair s−1, respectively. Moreover, ag-[c-PrMeTPP]2[MnBr4] retains 99% of its original intensity after 3000 s of continuous irradiation and 50 cycles, demonstrating excellent radiation stability. ag-[c-PrMeTPP]2[MnBr4] also shows a spatial resolution of 12 lp mm−1, indicating its applicability for the X-ray imaging.
The glass transitions are confirmed by the Raman spectroscopy where several distinguishable peaks below 150 cm−1, corresponding to the vibrational modes of [MnBr4]2− tetrahedra, display a broad single peak after vitrification (Fig. 3c and Supplementary Fig. 27). FTIR analysis reveal that characteristic νC–H and νC=C (in the phenyl ring) vibration peaks roughly maintain their positions, while the C–H vibrations show significant broadening after vitrification (Supplementary Fig. 28). These results indicate the variations in chemical environments of the cations and emergence of disorder after the melt-quenching. Additional Raman peaks, for the symmetric and asymmetric C–H vibrations in the methylene and methyl groups, give similar results. To probe the evolution of molecular motion during vitrification, variable-temperature Raman spectra were analyzed for the Mn–Br stretching of [MnBr4]2− and the phenyl ring in-plane deformation of phosphonium cations (Supplementary Fig. 29). The peaks of Mn–Br stretching are located at 250–255 cm−1 (ν1) and 255–260 cm−1 (ν1’), while the vibration peak of the phenyl ring appears at 610–620 cm−1 (ν2) (Supplementary Fig. 30–37). The corresponding full widths at half maximum, fitted using the Lorentzian functions, are summarized in Supplementary Fig. 38. Both ν1 and ν2 bands exhibit gradual broadening with increasing temperature, and the spectra collected after quenching exhibit markedly broader features compared with those recorded before thermal annealing. Interestingly, the halide glasses with higher (or lower) GFAs give rise to less (or more) reduction of the lifetime as shown in the τglass/τcrystal plot (Fig. 3d). The low reduction in non-radiative decay of the high GFA glasses could arise from their approximate energy levels of the vibrational modes in their [MnBr4]2− configurations, which are relatively less altered compared with those in the melted phases. For the low GFA glasses, the [MnBr4]2− tetrahedra may experience subtle variations in their local environment after vitrification, leading to enhanced non-radiative decay. It is worth mentioning that the τglass/τcrystal curve is reminiscent of the trend of the Tg/Tm ratio. This implies the phase space distribution of molecular components would be more homogeneous in the high GFA glasses, and vice versa.
To elucidate the critical motion behavior governing the phase space characteristics of the melts, classical and ab initio molecular dynamic (MD) simulations (Supplementary Figs. 39–46, Supplementary Note 1, and Supplementary Movie 2) were conducted43,44. The crystal phases were heated up to 800 K to reach fully melted states, and then quenched into glass phases at 500 K (Supplementary Fig. 47). The MSD of the terminal C atom in the side chain was extracted to compare the dynamics of organic cations in different halide glasses (Supplementary Figs. 48 and 49). The MSD values do not show an obvious correlation with the Tg/Tm ratios, thereby excluding their direct influence on GFA. This could be attributed to the fact that the MSD metric includes both the molecular translation and rotation, which is inadequate for characterizing the rotational disorder crucial for assessing glass-formation. Therefore, the rotational correlation function (RCF) (Fig. 4a), C(t), a statistical measure to quantify the rotational motion of molecular moieties over time, was adopted:
where ξ is the integration variable, representing the initial time point; t is the delay time, denoting the time elapsed from the initial time point ξ; p is the orientational vector defined from the central P atom to the terminal C atom in the side chain; Pn(x) is the nth order Legendre polynomial, and n is set to 1 for determining the rotational disorder. The RCF plots of the organic moieties in different glass systems show a pronounced correlation with Tg/Tm (Fig. 4a), where the RCF values decay more rapidly in systems with higher GFAs. The rotational correlation time (\({\tau }_{{{\rm{c}}}}={\int }_{0}^{\infty }C\left(t\right){{\rm{d}}}t\)), derived from the RCF integration of the modified organic side chains in the glass state at 500 K also demonstrates a significant dependence on Tg/Tm. These results indicate the rotational energy landscape can provide an intuitive means to comprehend the features of phase space distribution affecting GFA. To capture the rotational energy landscape of phosphonium cations, metadynamics simulations based on angle and alphabeta CVs were employed, and the predominant characteristics of rotational phase space distribution were quantified45. The rotational potential wells in the phosphonium cations of [BuTPP]2[MnBr4], [BPP]2[MnBr4], and [BrBPP]2[MnBr4] glasses possess a gradually increasing depth, indicating an incremental hindrance to the cationic rotational behavior in these systems, as seen from the free-energy surfaces in Fig. 4b and Supplementary Fig. 50. The angles between primitive axes and the modified organic side chains are served as orientational indicators of phosphonium cations, and used as collective variables sx, sy and sz, for analyzing the energy landscape. Among the representative systems, [BuTPP]2[MnBr4] with a high GFA has a shallow potential well and a flat energy landscape, indicating a more uniform phase space distribution and pronounced disorder. In contrast, [BPP]2[MnBr4] and [BrBPP]2[MnBr4], as typical systems with higher Tm but lower GFAs, exhibits a substantially deeper potential well, which suggests the concentrated distribution in phase space and a reduced degree of disorder. The metadynamics simulations based on alphabeta CVs, featuring torsional indicator of the four side chains of the phosphonium cations, were also performed (Supplementary Fig. 51). The resulting free-energy profiles display the same trend as the angle-based CVs in these three glass systems, consistently confirming the distribution of energy landscapes.
a RCF of phosphonium cations in the nine different glass models simulated at 500 K. b The free-energy surfaces obtained from metadynamics simulations. The depth of the 3D map indicates the system energy with different molecular orientation. The contour map is the projection of the 3D map, which indicates the energy distribution. c Correlation matrix of glass properties and molecular descriptors. The descriptors include Iratio, \({\sigma }_{{{\rm{tot}}}}^{2}\), Smax, and τc. See the formula in Supplementary Note 2 and the full correlation matrix in Supplementary Figs. 53 and 54. d Schematic illustrations of the relationship between three representative structural motifs and glass properties with incremental ergodicity breaking characteristics. The ellipsoid with arrows denotes the possible configuration of molecular motifs, and the curve represents the rotational energy landscapes.
Since molecular rotation depends on both shape and ESP surface of the organic moieties, the Spearman and Pearson correlation coefficients46,47,48, along with linear regression protocol, were employed to further quantify the molecular characteristics determining GFA (Fig. 4c and Supplementary Figs. 52–54; see formula in Supplementary Note 2). Three descriptors of phosphonium cations dominating GFA were obtained using the density functional theory (DFT) calculations49,50,51 and MD analysis, which are the rotational anisotropy descriptor (Iratio), the overall variance of ESP (\({\sigma }_{{{\rm{tot}}}}^{2}\)), and τc. Iratio was defined as:
where Imax and Imin are the maximum and minimum moments of inertia along the principal axes. This descriptor was derived from the moment of inertia in a homogeneous ellipsoid. \({\sigma }_{{{\rm{tot}}}}^{2}\) was defined as52:
where \({V}_{{{{\bf{r}}}}_{i}}^{+}\) and \({V}_{{{{\bf{r}}}}_{j}}^{-}\) are the positive and negative ESP values on the van der Waals surface, and g and h are the respective positive and negative grid points. The Iratio, \({\sigma }_{{{\rm{tot}}}}^{2}\), and tc descriptors exhibit a significant positive correlation to Tg and Tm. Conversely, a negative correlation is observed with the Tg/Tm ratio, serving as efficient descriptors for assessing GFA. In addition, the orientational order parameter Smax, extracted from RMC modeling, exhibits a pronounced negative correlation with Tg/Tm, highlighting its reliability as an excellent indicator of GFA. These results demonstrate that the rotational disorder play a dominant role in governing glass formation. In terms of the molecular shape, it is noted that the mass distribution exerts a more pronounced influence in GFA than size alone. In addition, \({\sigma }_{{{\rm{tot}}}}^{2}\), which quantifies the electrostatic and van der Waals interactions, was found to significantly affect GFA. To further illustrate these primary characteristics, a disorder model of the glass structures is proposed (Fig. 4d). According to the molecular shape and surface ESP of the phosphonium cations, the nine glasses can be divided into three types. In the first type, the organic component exhibits a significantly uniform distribution of rotation and a weakly polar (or non-polar) ESP surface. This leads to a significant number of allowed configurations and flat energy landscapes, hence enabling high GFA and low Tg and Tm as observed from [c-PrMeTPP]2[MnBr4], [BuTPP]2[MnBr4], and [PrTPP]2[MnBr4] glasses. For the second type, the non-uniform distribution of the molecular rotation promotes the ergodicity breaking, which reduces the number of sampled configurations and therefore results in lowered GFAs in [MeTPP]2[MnBr4], [MeBPP]2[MnBr4], [BPP]2[MnBr4], and [EtTPP]2[MnBr4]). The lower GFA of [BrBPP]2[MnBr4] compared to [MeBPP]2[MnBr4] is attributed to its greater molecular polarity, which strengthens intermolecular interactions and increases structural frustration in the glassy state. The phosphonium cations in the last type have increased non-uniform distribution of rotation and surface ESP polarity, which further narrows the distribution of rotational phase space and gives rise to low GFA and high Tg and Tm ([FBPP]2[MnBr4] and [BrBPP]2[MnBr4]). In brief, compact and symmetrical cations with weakly polar or aliphatic substituents tend to exhibit uniform ESP surfaces and higher rotational freedom. This facilitates orientational disorder of the structural motifs and produces relatively low intermolecular potential barriers, thereby enhancing the GFA. Whereas cations with elongated shapes or strong polar substituents display anisotropic charge distributions and stronger intermolecular interactions, which restrict molecular conformations, create pronounced potential energy extrema, and ultimately lead to reduced GFA.
In summary, through modifying the molecular components, a family of 0D hybrid metal halide glasses, with diverse GFAs, have been synthesized. Their tunable GFAs are attributed to the differences in molecular rotational disorder and associated energetic landscapes of the structural unit during the melt-quenching process. Specifically, a more uniform distribution of molecular rotation and reduced variance of surface ESP contributes to a greater number of permissible configurations, and flatter potential energy surfaces in these 0D hybrid halide glasses. This implies an evolution of rotational ordering and an expansion of the phase space distribution, ultimately resulting in enhanced GFA, lower Tg, and Tm. These findings offer a pathway towards understanding the origins of glass formation from a more general perspective of disorder, which is applicable to a broad realm of hybrid organic-inorganic solids through molecular design. Given the existence of small atomic clusters in metallic glasses, this approach might be generalized for these systems too.
Methods
Chemicals and materials
All chemicals were purchased from commercial suppliers and used without further purification. Manganese bromide (MnBr2, 99.9%), tetraphenylphosphonium bromide (C24H20PBr, TPPBr, 99%), methyltriphenylphosphonium bromide (C19H18PBr, MeTPPBr, 98%), ethyltriphenylphosphonium bromide (C20H20PBr, EtTPPBr, 98%), propyltriphenylphosphonium bromide (C21H22PBr, PrTPPBr, 99%), methylenecyclopropyltriphenylphosphonium bromide (C22H22PBr, c-PrMeTPPBr, 98%), (n-butyl)triphenylphosphonium bromide (C22H24PBr, BuTPPBr, 98%), benzyltriphenylphosphonium bromide (C25H22PBr, BPPBr, 98%), (p-Xylyl)-triphenylphosphonium bromide (C26H24PBr, MeBPPBr, 98%), and (4-fluorobenzyl)triphenylphosphonium chloride (C25H21FPCl, FBPPCl, 98%), (4-bromobenzyl)triphenylphosphonium bromide (C25H21PBr2, BrBPPBr, 98%), were used for the synthesis of 0D hybrid metal halides. Ethanol (>99.5%), dichloromethane (>99.5%), hydrobromic acid (48%), and deionized water were used as solvents.
Synthesis of metal halide crystal
The crystals of [TPP]2[MnBr4], [MeTPP]2[MnBr4], and [EtTPP]2[MnBr4] were prepared according to the previous literatures26,53,54.
Synthesis of [PrTPP]2[MnBr4] crystal: PrTPPBr (0.7706 g, 2 mmol) and MnBr2 (0.2147 g, 1 mmol) were dissolved in an open-top 10 mL vial using ethanol (>99.5%, 4 mL), then the vial was placed into a 250 mL sealed container containing isopropanol. Pale green crystals were obtained after three days.
Synthesis of [BuTPP]2[MnBr4] crystal: The synthesis process of this compound is identical to that of [PrTPP]2[MnBr4], except that PrTPPBr was replaced by BuTPPBr (0.8026 g, 2 mmol), then the vial was transferred into a 250 mL sealed container containing isopropanol. Pale green crystals were obtained after three days.
Synthesis of [c-PrMeTPP]2[MnBr4] crystal: The synthesis process of this compound is the same as that of [PrTPP]2[MnBr4], except that PrTPPBr was replaced by c-PrMeTPPBr (0.7985 g, 2 mmol).
Synthesis of [MeBPP]2[MnBr4] crystal: The synthesis process is the same as that of [PrTPP]2[MnBr4], except that PrTPPBr was replaced by MeBPPBr (0.8966 g, 2 mmol).
Synthesis of [BPP]2[MnBr4] crystal: BPPBr (0.8666 g, 2 mmol) and MnBr2 (0.2147 g, 1 mmol) were dissolved in dichloromethane (>99.5%, 5 mL). After two weeks of allowing the mixture to settle undisturbed, the crystals were obtained.
Synthesis of [FBPP]2[MnBr4] crystal: FBPPCl (0.8136 g, 2 mmol) and MnBr2 (0.2147 g, 1 mmol) were dissolved in HBr (48%, 5 mL) upon heating and continuous stirring at 110 °C, and green crystals were obtained by slowly cooling the saturated solution to room temperature.
Synthesis of [BrBPP]2[MnBr4] crystal: The synthesis process of this compound is the same as that of [PrTPP]2[MnBr4], except that PrTPPBr was replaced by BrBPPBr (1.0244 , 2 mmol). High-performance liquid chromatography analysis was performed using a SHIMADZU LC-20AD facility with commercial 5 µm, 4.6 × 150 mm columns, using a MeCN/H2O = 50/50 (vV/vV) at a flow rate of 1 mL min−1 and a detection wavelength of 254 nm. The results of BrBPPBr and [BrBPP]2[MnBr4] show that [BrBPP]2[MnBr4] possess two major peaks at retention times of 1.173 min and 3.526 min, corresponding to the organic cation and the inorganic species (Supplementary Fig. 55). Elemental analysis illustrates C and H contents of 47.94% and 3.47%, respectively, which is consistent with the theoretical values of 48.50% and 3.39%.
Synthesis of glasses
The synthesis of 0D metal halide glasses was performed on a Linkam LTS420 Heating and Freezing stage. The Category I crystalline samples were placed on the microscope slides and heated to 230 °C at a rate of 10 °C min−1 under N2 atmosphere. Glass samples were then obtained by cooling the halide melts to 30 °C at a rate of 10 °C min−1. The melt-quenching procedure of the Category II samples is similar to that described above, except that the target temperature was set to 260 °C for [BPP]2[MnBr4], [FBPP]2[MnBr4], and [MeBPP]2[MnBr4], and 280 °C for [BrBPP]2[MnBr4].
Synthesis of glass bulks and fibers
In the case of [BuTPP]2[MnBr4], for example, the crystal sample was placed in a silicone mold, heated to 190 °C in an oven until fully molten. Then the melt was cooled down naturally to form a glass bulk after removing mold. The fiber sample of [BPP]2[MnBr4] was synthesized using a homemade rotatable melting device.
Single crystal X-ray diffraction (SC-XRD) and Powder X-ray diffraction (PXRD) analysis
Single crystal X-ray diffraction experiments were performed on a Rigaku XtaLAB MM007 CCD facility using a mirror-monochromated Cu-Kα radiation (λ = 1.54178 Å) at 100 K. Data collection and reduction were processed using the Rigaku CrysAlisPro program. Structures were solved with the ShelXT package using the intrinsic phasing and refined with the Olex255 and ShelXL refinement package using the least squares minimization. The crystal information files were deposited in the Cambridge Crystallographic Data Centre with reference numbers 2241752–2241754, 2284544–2284546, and 2370641. Powder XRD patterns were collected using a Rigaku MiniFlex 600 diffractometer at 40 kV, 100 mA with a Cu-target tube and a graphite monochromator. The 2θ varied between 5 and 50° with a step rate of 5° min−1.
X-ray total scattering analysis and reverse Monte Carlo (RMC) simulation
X-ray total scattering data (λ = 0.208566 Å, 59.45 KeV) were collected at the Beijing Synchrotron Radiation Facility. Data of the samples and background were collected in the region of 0.5 < Q < 24.7 Å−1. Data process and background corrections were performed using the pdfgetx2 program56. RMC simulations of [BPP]2[MnBr4] were processed using the RMCProfile package57. A 5 × 4 × 3 supercell structure of [BPP]2[MnBr4] crystal was used as the initial structure, and the experimental S(Q) data of crystalline and glassy samples were used for fitting. The S(Q) data of 0.5–20.5 Å−1 were used to exclude interference during experiments. Due to the lack of constraints in dihedral angle potentials in the simulation package, the phenyl rings showed distorted configurations.
Thermogravimetric (TGA) and differential scanning calorimetry (DSC) analysis
Thermogravimetric curves were collected using a Rigaku Thermo Plus EVO2 analyzer under a constant airflow (20 mL min−1). Approximately 5 mg powder sample was heated from room temperature to 800 °C in an alumina crucible at a rate of 10 °C min−1. The TGA data were used to determine the decomposition temperatures (Td).
Differential scanning calorimetry (DSC) measurements were performed on a TA DSC 25 facility with the heating/cooling rates of 10 °C min−1 under N2 protection. For each measurement, approximately 5 mg sample was sealed into an aluminum crucible. It took two up-scans to determine the melting points (Tm) and glass transition temperatures (Tg). Tm was determined from the onset of the endothermic peak in the first heating run of the DSC curve, and Tg was determined from the midpoint of the step change in heat flow in the second heating run. To quantify the kinetic behavior of the system, DSC measurements were conducted at diverse heating and cooling rates. Owing to the absence of distinct crystallization peaks in the compounds, the activation energies associated with the glass transition were estimated using the Moynihan and Kissinger methods. The corresponding formulas are as follows:
where β is the heating or cooling rate, Ea is the activation energy related to the structural relaxation.
Photoluminescence (PL) and X-ray scintillation properties measurements
PL and time-resolved luminescence spectra were measured on a FS5 spectrofluorometer with a monochromatized Xe lamp. X-ray emission spectra were measured on a FS5 spectrofluorometer with a radiation source (40 kV, 150 μA). X-ray imaging was performed using a homemade X-ray imaging system. The object and scintillator glass were placed under X-ray radiation, and a camera was used to take pictures of the object.
Fourier transform infrared spectrometer (FTIR) and Raman measurements
FTIR spectra were recorded using a Thermo Scientific Nicolet iS20 facility in the range of 400–4000 cm−1. All the samples were fully dried, mixed with KBr, and then pressed into pellets approximately 1 mm thick. Raman spectra at room temperature were measured on a HORIBA Scientific LabRAM HR Evolution spectrometer. All samples were excited with a 785 nm laser beam. Three cycles of data collection were implemented to reduce the effect of background noise. Variable temperature Raman spectra were measured on an Edinburgh RMS1000 spectrometer and the samples were excited with a 625 nm laser beam.
Density functional theory (DFT) calculations
DFT calculations were performed using the ORCA 5.0.3 package to obtain geometry information and surface ESP information50,51. The geometry optimization of phosphonium cations and [MnBr4]2− anions was performed using a PBE0 functional58 and def2-TZVP basis sets59 with the Grimme’s DFT-D3(BJ) dispersion corrections60,61, following with frequency calculations. The optimized structures were used for calculating molecular descriptors with the Multiwfn package49. The dipole moments were calculated using the ma-def2-TZVP basis sets62. As a comparison, the molecular descriptors of phosphonium cations based on geometries in the crystal structures were calculated using the same methodology mentioned above. The heavy atoms were fixed and the positions of hydrogen atoms were optimized.
Classical molecular dynamics (MD) simulations
To investigate the behaviors of phosphonium cations and [MnBr4]2− tetrahedra in the solid and liquid phases, and elucidate the difference between different halide glasses, classical MD simulations were carried out using the GROMACS 2022 package44. The geometries of phosphonium cations and [MnBr4]2− anions was obtained from DFT calculations, and the full systems were built using PACKMOL, including 400 cations and 200 anions63. The topology files were generated using the Sobtop package64. The Generalized Amber Force Field (GAFF) was used as the initial parameters to the simulation system, with Mn and Br atoms assigned Universal Force Field atomic types. Bond and angle parameters were derived from DFT frequency calculations using the modified Seminario method (ORCA), and the remaining parameters were taken from GAFF65,66. The atomic charges were calculated with the Multiwfn using the restrained ESP method. During the production simulations, we used a leap-frog Verlet integrator with a 0.5 fs time interval, a Velocity-Rescale thermostat, and a Parrinello-Rahman barostat. To improve the performance, the bonds with H atoms were constrained. Since [MnBr4]2− anions in [MeTPP]2[MnBr4] and [EtTPP]2[MnBr4] models inclined to dimerize due to the small cation size, we modified the atomic charge distribution of [MnBr4]2− in these two models to prevent dimerization. Energy minimization and equilibration were performed under isothermal-isobaric ensembles (NPT), that is, the number of particles, the temperature and pressure are fixed, for 1 ns and 100 ns at 800 K, respectively. Then, the whole systems were annealed from 600 to 300 K to determine the Tg. The constant temperature simulations were performed at 500, 600, and 800 K for 40 ns, and the last 10 ns of the trajectories was used to calculate the root mean square deviation, mean square displacement (MSD), radial distribution function, and root mean square fluctuation. In addition, for the calculations of RCF, the simulations were conducted at 500 K for 100 ~ 300 ns. All trajectories were analyzed and drawn in the Visual Molecular Dynamics67.
Ab initio molecular dynamics (AIMD) and metadynamics simulations
To probe the dynamics of A2[MnBr4] at different temperatures, AIMD simulations at 300, 600, and 800 K were performed using the CP2K program43. In these simulations, the PBE functional68 and Grimme’s DFT-D3(BJ) dispersion corrections were used in combination with the DZVP-MOLOPT-SR-GTH basis sets and Goedecker-Teter-Hutter pseudopotentials69. The CUTOFF and REL_CUTOFF were set to 600 Ry and 40 Ry, respectively, and the Canonical Sampling through Velocity Rescaling thermostat and Martyna–Tobias–Klein barostat were used. All AIMD models were pre-equilibrated and run for 10 ps with a time interval of 0.5 fs in the NPT ensemble. In addition, a frequency calculation was performed to confirm the IR/Raman peak contributions using CP2K.
The metadynamics simulations based on angle and alphabeta CVs were performed using the CP2K program70. The angle CVs are defined using the angles between the primitive vectors a, b, c, and directional vectors defined by modified organic side chains. Note that only 2 of 3 collective variables (sx and sy) were used to plot the free-energy landscapes for clarity. The alphabeta CVs are defined as:
where sAB is the alphabeta CV value, ϕi is the instantaneous torsional angle, ϕiRef is the corresponding reference torsion angle. Four torsional angles between the principle axes and each of the four side chains of the phosphonium cation are combined into a single alphabeta CV, ranging from 0 to 4. Note that the angle CVs used GFN1-xTB method with CP2K built-in kernel, while the alphabeta CVs used density-functional tight binding method with Plumed package71,72,73.
Data availability
All data are available in the Article or its Supplementary information, and available from the corresponding authors upon request. The raw data have been deposited in a Figshare repository (https://doi.org/10.6084/m9.figshare.30880172). Source data are provided with this paper.
References
Angell, C. A. Formation of glasses from liquids and biopolymers. Science267, 1924–1935 (1995).
Debenedetti, P. G. & Stillinger, F. H. Supercooled liquids and the glass transition. Nature410, 259–267 (2001).
Kennedy, D. & Norman, C. What don’t we know? Science309, 75 (2005).
Santen, L. & Krauth, W. Absence of thermodynamic phase transition in a model glass former. Nature405, 550–551 (2000).
Greaves, G. N. & Sen, S. Inorganic glasses, glass-forming liquids and amorphizing solids. Adv. Phys.56, 1–166 (2007).
Bennett, T. D. et al. Structure and properties of an amorphous metal-organic framework. Phys. Rev. Lett.104, 115503 (2010).
Yang, Y. et al. Determining the three-dimensional atomic structure of an amorphous solid. Nature592, 60–64 (2021).
Swallen, S. F. et al. Organic glasses with exceptional thermodynamic and kinetic stability. Science315, 353–356 (2007).
Ma, N. & Horike, S. Metal-organic network-forming glasses. Chem. Rev.122, 4163–4203 (2022).
Liu, L. R. et al. Ergodicity breaking in rapidly rotating C60 fullerenes. Science381, 778–783 (2023).
Thirumalai, D., Mountain, R. D. & Kirkpatrick, T. R. Ergodic behavior in supercooled liquids and in glasses. Phys. Rev. A39, 3563–3574 (1989).
Hedges, L. O., Jack, R. L., Garrahan, J. P. & Chandler, D. Dynamic order-disorder in atomistic models of structural glass formers. Science323, 1309–1313 (2009).
Stillinger, F. H. & Debenedetti, P. G. Glass transition thermodynamics and kinetics. Annu. Rev. Condens. Matter Phys.4, 263–285 (2013).
Ma, E. Tuning order in disorder. Nat. Mater.14, 547–552 (2015).
Frankberg, E. J. et al. Highly ductile amorphous oxide at room temperature and high strain rate. Science366, 864–869 (2019).
Wang, W. H. The elastic properties, elastic models and elastic perspectives of metallic glasses. Prog. Mater. Sci.57, 487–656 (2012).
Fang, W. et al. Organic-inorganic covalent-ionic molecules for elastic ceramic plastic. Nature619, 293–299 (2023).
Yang, Z. et al. ZIF-62 glass foam self-supported membranes to address CH4/N2 separations. Nat. Mater.22, 888–894 (2023).
Chen, Y. et al. Microscopic ergodicity breaking governs the emergence and evolution of elasticity in glass-forming nanoclay suspensions. Phys. Rev. E102, 042619 (2020).
Rino, J. P. et al. Structure of rings in vitreous SiO2. Phys. Rev. B47, 3053–3062 (1993).
Salmon, P. S., Martin, R. A., Mason, P. E. & Cuello, G. J. Topological versus chemical ordering in network glasses at intermediate and extended length scales. Nature435, 75–78 (2005).
Zhou, Q. et al. Experimental method to quantify the ring size distribution in silicate glasses and simulation validation thereof. Sci. Adv.7, eabh1761 (2021).
Inoue, A. Stabilization of metallic supercooled liquid and bulk amorphous alloys. Acta Mater.48, 279–306 (2000).
Sheng, H. W. et al. Atomic packing and short-to-medium-range order in metallic glasses. Nature439, 419–425 (2006).
Li, M.-X. et al. Data-driven discovery of a universal indicator for metallic glass forming ability. Nat. Mater.21, 165–172 (2021).
Wrighton, M. & Ginley, D. Excited state decay of tetrahalomanganese(II) complexes. Chem. Phys.4, 295–299 (1974).
Balsamy, S., Natarajan, P., Vedalakshmi, R. & Muralidharan, S. Triboluminescence and vapor-induced phase transitions in the solids of methyltriphenylphosphonium tetrahalomanganate(II) complexes. Inorg. Chem.53, 6054–6059 (2014).
Li, M. & Xia, Z. Recent progress of zero-dimensional luminescent metal halides. Chem. Soc. Rev.50, 2626–2662 (2021).
Luo, J. B. et al. A melt-quenched luminescent glass of an organic–inorganic manganese halide as a large-area scintillator for radiation detection. Angew. Chem. Int. Ed.62, e202216504 (2023).
Xu, L.-J. et al. Green-light-emitting diodes based on tetrabromide manganese(II) complex through solution process. Adv. Mater.29, 1605739 (2017).
Dove, M. T. & Li, G. Review: pair distribution functions from neutron total scattering for the study of local structure in disordered materials. Nucl. Anal.1, 100037 (2022).
Selinger, J. V. Introduction to the Theory of Soft Matter: From Ideal Gases to Liquid Crystals, Ch. 5 (Springer, 2016).
Zheng, Q. et al. Understanding glass through differential scanning calorimetry. Chem. Rev.119, 7848–7939 (2019).
Turnbull, D. Under what conditions can a glass be formed? Contem. Phys.10, 473–488 (1969).
Kissinger, H. E. Reaction kinetics in differential thermal analysis. Anal. Chem.29, 1702–1706 (1957).
Moynihan, C. T., Easteal, A. J., Wilder, J. & Tucker, J. Dependence of the glass transition temperature on heating and cooling rate. J. Phys. Chem.78, 2673–2677 (1974).
Vyazovkin, S. Kissinger method in kinetics of materials: things to beware and be aware of. Molecules25, 2813 (2020).
Yin, M. et al. Crystal–glass phase transition enabling reversible fluorescence switching in zero-dimensional antimony halides. Chem. Commun.59, 11361–11364 (2023).
Zhang, Z.-Z. et al. Organic–inorganic hybrid Mn-based transparent glass for curved X-ray scintillation imaging. Adv. Opt. Mater.12, 2302434 (2024).
Dong, C. et al. Organic–inorganic hybrid glasses of atomically precise nanoclusters. J. Am. Chem. Soc.146, 7373–7385 (2024).
Feng, T. et al. Large-area transparent antimony-based perovskite glass for high-resolution X-ray imaging. ACS Nano18, 16715–16725 (2024).
He, Z.-L. et al. A universal strategy toward two-component organic–inorganic metal-halide luminescent glasses and glass–crystal composites. Sci. Adv.11, eadu1982 (2025).
Hutter, J., Iannuzzi, M., Schiffmann, F. & VandeVondele, J. CP2K: atomistic simulations of condensed matter systems. Wiley Interdiscip. Rev.-Comput. Mol. Sci.4, 15–25 (2014).
Abraham, M. J. et al. GROMACS: high performance molecular simulations through multi-level parallelism from laptops to supercomputers. SoftwareX1–2, 19–25 (2015).
Bussi, G. & Laio, A. Using metadynamics to explore complex free-energy landscapes. Nat. Rev. Phys.2, 200–212 (2020).
Pearson, K. Note on regression and inheritance in the case of two parents. Proc. R. Soc. Lond.58, 240–242 (1895).
Spearman, C. The proof and measurement of association between two things. Am. J. Psychol.15, 72–101 (1904).
de Siqueira Santos, S., Takahashi, D. Y., Nakata, A. & Fujita, A. A comparative study of statistical methods used to identify dependencies between gene expression signals. Brief Bioinform.15, 906–918 (2014).
Lu, T. & Chen, F. Multiwfn: a multifunctional wavefunction analyzer. J. Comput. Chem.33, 580–592 (2012).
Neese, F., Wennmohs, F., Becker, U. & Riplinger, C. The ORCA quantum chemistry program package. J. Chem. Phys.152, 224108 (2020).
Neese, F. Software update: the ORCA program system—Version 5.0. Wiley Interdiscip. Rev.-Comput. Mol. Sci.12, e1606 (2022).
Murray, J. S. & Politzer, P. Statistical analysis of the molecular surface electrostatic potential: an approach to describing noncovalent interactions in condensed phases. J. Mol. Struc-Theochem.425, 107–114 (1998).
Lin, J. et al. Narrowing the band of green emission in manganese hybrids by reducing the hydrogen bond strength and structural distortion. J. Mater. Chem. C10, 16773–16780 (2022).
Li, B. et al. Zero-dimensional luminescent metal halide hybrids enabling bulk transparent medium as large-area X-ray scintillators. Adv. Opt. Mater.10, 2102793 (2022).
Dolomanov, O. V. et al. OLEX2: a complete structure solution, refinement and analysis program. J. Appl. Crystallogr.42, 339–341 (2009).
Qiu, X., Thompson, J. W. & Billinge, S. J. L. PDFgetX2: a GUI-driven program to obtain the pair distribution function from X-ray powder diffraction data. J. Appl. Crystallogr.37, 678–678 (2004).
Tucker, M. G. et al. RMCProfile: reverse Monte Carlo for polycrystalline materials. J. Phys.-Condes. Matter19, 335218 (2007).
Adamo, C. & Barone, V. Toward reliable density functional methods without adjustable parameters: the PBE0 model. J. Chem. Phys.110, 6158–6170 (1999).
Weigend, F. & Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: design and assessment of accuracy. Phys. Chem. Chem. Phys.7, 3297–3305 (2005).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys.132, 154104 (2010).
Grimme, S., Ehrlich, S. & Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem.32, 1456–1465 (2011).
Papajak, E. et al. Perspectives on basis sets beautiful: seasonal plantings of diffuse basis functions. J. Chem. Theory Comput.7, 3027–3034 (2011).
Martínez, L., Andrade, R., Birgin, E. G. & Martínez, J. M. PACKMOL: a package for building initial configurations for molecular dynamics simulations. J. Comput. Chem.30, 2157–2164 (2009).
Lu T. Sobtop, Version 1.0 (dev3.1). http://sobereva.com/soft/Sobtop.
Wang, J., Wang, W., Kollman, P. A. & Case, D. A. Automatic atom type and bond type perception in molecular mechanical calculations. J. Mol. Graph.25, 247–260 (2006).
Wang, J. et al. Development and testing of a general amber force field. J. Comput. Chem.25, 1157–1174 (2004).
Humphrey, W., Dalke, A. & Schulten, K. VMD: visual molecular dynamics. J. Mol. Graph.14, 33–38 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett.77, 3865–3868 (1996).
Goedecker, S., Teter, M. & Hutter, J. Separable dual-space Gaussian pseudopotentials. Phys. Rev. B54, 1703–1710 (1996).
Grimme, S., Bannwarth, C. & Shushkov, P. A robust and accurate tight-binding quantum chemical method for structures, vibrational frequencies, and noncovalent interactions of large molecular systems parametrized for all spd-block elements (Z = 1-86). J. Chem. Theory Comput.13, 1989–2009 (2017).
Elstner, M. et al. Self-consistent-charge density-functional tight-binding method for simulations of complex materials properties. Phys. Rev. B58, 7260–7268 (1998).
Cui, M., Reuter, K. & Margraf, J. T. Obtaining robust density functional tight-binding parameters for solids across the periodic table. J. Chem. Theory Comput.20, 5276–5290 (2024).
Bonomi, M. et al. PLUMED: a portable plugin for free-energy calculations with molecular dynamics. Comput. Phys. Commun.180, 1961–1972 (2009).
Acknowledgements
We acknowledge the financial support from the National Key Research and Development Program of China (No. 2022YFA1503301 to W.L.), the National Natural Science Foundation of China (Nos. 22035003 to X.H.B., 22375105 to W.L., and 12174274 to M.T.D.), and the State Key Laboratory of Silicate Materials for Architectures, Wuhan University of Technology (No. SYSJJ2022-02 to W.L.).
Author information
Authors and Affiliations
Contributions
Z.-Y.L., R.F., W.L., and X.-H.B. conceived the idea and led the research. Z.-Y.L. and R.F. contributed equally to this work. Z.-Y.L. and R.F. performed the experiments and collected the data. Z.-Y.L. synthesized the materials and performed XRD, SEM, FTIR, PL, polarization, DSC, TGA, and Raman tests. R.F. performed some PL, FTIR, and Raman tests. R.F. performed the theoretical calculations. C.S., X.L., and L.Z. performed X-ray total scattering measurements. W.L., X.-H.B., Z.C., Z.-Y.L., R.F., M.T.D., N.L., and Z.-G.L. evaluated and discussed data. Z.-Y.L., R.F., F.W., W.L., and X.-H.B. contributed to writing the manuscript. S.-S.H. helped to revise the manuscript. All authors reviewed and discussed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, ZY., Feng, R., Li, ZG. et al. Glass formation in hybrid metal halides via breaking molecular rotational order. Nat Commun 17, 1850 (2026). https://doi.org/10.1038/s41467-026-68563-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-68563-5


![Fig. 1: Design and synthesis of hybrid A2[MnBr4] glasses.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs41467-026-68563-5/MediaObjects/41467_2026_68563_Fig1_HTML.png)
![Fig. 2: Structural and thermal properties of A2[MnBr4] glasses.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs41467-026-68563-5/MediaObjects/41467_2026_68563_Fig2_HTML.png)
![Fig. 3: The luminescence and vibrational properties of A2[MnBr4] glasses.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs41467-026-68563-5/MediaObjects/41467_2026_68563_Fig3_HTML.png)
