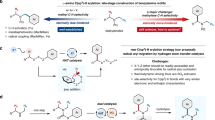
Abstract
Aliphatic amines, such as N,N-dialkyl secondary amines, represent important scaffolds in bioactive molecules, driving significant interest in their regio- and stereoselective C–H functionalisation. While hydrogen atom transfer (HAT) provides a powerful radical-based approach to elaborate such amines, achieving controllable, regio-divergent, and enantioselective functionalisation across different N-alkyl groups remains challenging. Herein, we report a Cu-catalyzed α’/β-regiodivergent and β-enantioselective cyanation of secondary amine-derived ureas through tunable 1,4’/1,5-HAT. The utilization of a sterically demanding ligand L14 enables the excellent α’-selective C–H cyanation at the N-methyl position, while two developed ligands (L24 and L41) promote the β-chirality construction at the other N-alkyl group. The approach is demonstrated for a broad scope of ureas with wide functional group compatibility. Experimental and computational studies reveal two distinct reaction pathways regarding the different reactive sites (α’/β) and the choice of ligands could significantly influence the selectivity in HAT process.
Similar content being viewed by others
Introduction
With an estimate of more than 60% of the top 200 small molecule pharmaceuticals by retail sales containing amine groups based on Njarðarson’s reports, which are often along with chiral centers, there is an increasing need to devise streamlined and efficient synthetic methods to access these motifs1,2. In this context, developing a modular synthetic strategy that enables the site-divergent and enantioselective functionalisation of C(sp3)–H bonds at different positions (α, β, γ, etc.) would be promising for the structural diversification of amines3,4,5,6. However, two or more different alkyl substituents on the amines makes the situation over double complicated compared to other compounds, albeit α-C–H bonds of amines might have higher reactivity for the appropriate bond dissociation energies (BDEs)7,8,9. By contrast to the α-site, reports concerning β-C–H bonds activation are rather less10,11,12,13,14, and of course the α/β regio-divergent functionalisation of acyclic amines are still largely unexplored15,16,17. In addition, current methods for asymmetric β-C(sp3)–H bond functionalisation of amines mainly focused on the intramolecular cycloamination18,19,20 and the functionalisation of more reactive cyclic amines11,16,21,22, while the enantioselective intermolecular C–H functionalisation at β-position of acyclic amines has met with limited success.
C(sp3)–H bond functionalisation of amines through intramolecular hydrogen atom transfer (HAT) process, known as Hofmann-Löffler-Freytag (HLF) reaction, has become a powerful tool to assemble elaborated amines23,24,25. Generally, the H-abstraction triggered by nitrogen-centered radical (NCR) usually occurs at the δ position via intramolecular 1,5-HAT from N(1) to C(5), given the stability of six-membered, chair-like conformational transition state26,27,28,29. Nevertheless, incorporation of auxiliary groups (e.g., urea or sulfonylurea) can alter the reaction sites to β/γ-positions, which as well undergo the preponderant 1,5/6-HAT (Fig. 1A(a))18,30,31,32. In these cases, the regioselectivity is dominated by intramolecular HAT step and is highly relied on the structure of substrate, rather than the reaction conditions20,33,34. Therefore, the radical transfer from N to other C-centered sites is of particularly difficult, especially for the rigid 1,4-HAT with coexisting of C(5) hydrogen35. Searching for suitable strategies that overcoming the intrinsic 1,5-HAT process is the key to unlock the α/β regio-divergent functionalisation of acyclic secondary amines.
A Site-selective functionalisation of aliphatic amines through radical pathways; B Ubiquitous α/β-nitrile amine structural motifs in pharmaceuticals and natural product; C This work: regiodivergent and enantioselective cyanation of ureas through ligand-regulated selective hydrogen atom transfer of α’/β-amino C(sp3)–H bonds.
Several elegant strategies have been developed for the site- and enantioselective C(sp3)–H functionalisation of molecules possessing multiple similar C–H bonds36,37,38. For instance, Rovis and co-workers reported the site-selective C(sp3)–H alkylation of tertiary amines at the more-crowded secondary α-position by turning the rate-determining step from hydrogen abstraction to radical trapping in the presence of a robust silylthiyl HAT catalyst (Fig. 1A(b))38. Alternatively, the groups of Liu and Lin also revealed that the Cu(II)-bound N-centered radical intermediates with a modular sulfonamide moiety and a specific bidentate ligand could be utilized as a tunable HAT reagent to discriminate the similar allylic C–H bonds in the same molecules39,40. Inspired by these precedents, we envisioned that the regiodivergent intramolecular HAT of secondary amines at different N-alkyl chains by NCRs could also be implemented by fine-tuning the ligand environment of the metal center.
On the other hand, the radical-mediated C–H cyanation has received great attention in recent years34,40,41,42,43,44,45,46. Given the importance of nitrile amines in medicine chemistry, the incorporation of a cyano-group onto amines at the different positions in a selective fashion is highly attractive in the discovery of drug molecules (Fig. 1B)47,48. Herein, we describe the ligand-controlled copper catalyzed regiodivergent and β-enantioselective cyanation of N-alkyl-N-methylamine-derived ureas by sliding 1,4’/1,5-HAT (Fig. 1C). Specifically, the more crowed ligand enabled the unusual 1,4’-HAT pathway to deliver α-amino radicals followed by kinetically favorable N-methyl cyanation while the sterically less demanding ligands promoted the β-cyanation through an entropically and enthalpically favored 1,5-HAT34,49,50. Moreover, the enantioselective β-cyanation was also achieved with two developed chiral ligands. The reaction features high regioselectivity and β-enantioselectivity, constituting an efficient pathway to access divergent cyano-substituted amines from the same set of starting materials. In addition, the mechanistic details have been elucidated by deuterium-labeling experiments and density functional theory (DFT) calculations.
Results
Reaction optimization
Current methods for asymmetric C(sp3)–H bond functionalisation of amines via HLF process mainly relies on the initial generation of NCRs through homolytic cleavage of N-F34,45,51,52,53,54 or N-O55 precursors in the presence of transition metal catalysts or photoinduced catalysis. However, the analogous process triggered by homolytic cleavage of the readily-accessible N-Cl compounds have rarely been harnessed in these transformations, probably due to the fact that easy trapping of in-situ generated C-centered radicals by another N-Cl amine would usually lead to the side chlorination reaction or a subsequent intramolecular amination32,33,56,57. However, it has also been reported that the HLF process could be intercepted through the radical trapping by a Cu(II) intermediate and thereby results in the selective C(sp3)–H bond functionalisation58. Given the well explored Cu-catalyzed radical-mediated C–H bond cyanation, we envisioned that chloroamide could be employed as a directing group for the regioselective C–H bond cyanation of amines via intercepted HLF process in the presence of copper/chiral bisoxazoline ligands catalytic systems42,43,46,59. With this consideration in mind, we commenced our investigation by firstly testing the Cu(CH3CN)4BF4 catalysed α’/β-regioselective C–H bond cyanation of a N-Cl urea derivative of N-Me phenylethylamine (1a) with TMSCN in the presence of different N-containing ligands (Fig. 2A). The pyridine derived achiral ligands usually provided poor regioselectivity and only the utilization of di(2-pyridyl) ketone (L4) afforded the β-cyanation product 3a with good regioselectivity (α’/β = 1:9). The chiral BOX and diamine ligands usually offered satisfactory β-siteselectivity, albeit with poor β-enantioselectivity (L5−L10). Among them, diamine ligand L8 performed best, affording β-cyanation product 3a in 77% isolated yield with excellent selectivity. Intriguingly, the use of ligand L11 gave a mixture of 3a and 2a (62%/23% yield, respectively), and a promising enantioselectivity of 3a was obtained (82.5:17.5 er). This result encouraged us to attempt a broader set of such ligands based on L11. To our surprise, the sterically more demanding analogous BOX ligands could give higher α’-selectivity and the ligand L14 bearing bulky bis-4-tBuBn group afforded the desired N-methyl cyanation product 2a in 69% isolated yield with excellent 30:1 site-selectivity. Subjecting 1a with racemic ligand (obtained by mixing (1R,2S)-L14 and (1S,2R)-L14) gave a comparative yield and selectivity (Fig. 2B, entry 2). Further screening on various copper catalysts did not give better results for the selective formation of 3a (Fig. 2B, entries 3–6). Next, we tried to enhance both the regio- and enantioselectivity for the β-cyanation of 3a using L11 as the ligand. After extensive evaluation of various reaction parameters (Fig. 2B, entries 7–12), CuSCN was identified as the optimal catalyst and CH3CN was selected as the solvent, 3a was obtained in good β-selectivity and enantioselectivity (75:5 rr and 91.5:8.5 er, entry 11). To further improve the enantio- and regioselectivity, a careful investigation of diverse chiral ligands under condition B was performed (Fig. 2C). It was found that the alkyl substituents at the central methylene position of BOX ligands could significantly influence the regio- and enantioselectivity probably through the modulation of bidentate bite angles34. For instance, the utilization of mono-alkyl (e.g., iPr, cyclopentyl, cyclohexyl) substituted BOX ligands promoted the enantioselectivity to around 95:5 er (L18−L20). On the basis of these results, several designed BOX ligands bearing substituted cyclohexyl groups were prepared and tested (L21−L24). L24 that possessing a 4,4-dimethylcyclohexyl group at the methylene position was identified as the optimized ligand, which afforded 3a with both excellent enantioselectivity (97:3 er) and site-selectivity (25:1 β-selectivity).
[a]Condition A: 1a (26.9 mg, 0.1 mmol), TMSCN (29.7 mg, 0.3 mmol), Cu(CH3CN)4BF4 (1.6 mg, 5 mol%), L (12 mol%), DCM (2 mL), N2, rt, 0.5 h; [b]Condition B: 1a (26.9 mg, 0.1 mmol), TMSCN (29.7 mg, 0.3 mmol), CuSCN (0.36 mg, 3 mol%), L* (7.5 mol%), CH3CN (2 mL), N2, rt, 0.5 h; [c]Isolation yield is in parentheses. The yields and regioselectivity ratios (rr, 2a/3a) were determined by GC-MS using dodecane as internal standard, and the enantiomeric ratios (er) were determined by HPLC analysis. A Ligand screening for α’/β-regioselective cyanation; B Optimization of other parameters for α’-regioselective and β-enantioselective cyanation; C Ligand screening for β-enantioselective cyanation.
Substrate scope
With the optimized reaction conditions in hand, the generality of developed strategy for the synthesis of α/β-nitrile amines was firstly investigated. Of note, despite various methods have been developed for α-C(sp3)–H cyanation of secondary and tertiary amines, the regioselectivity among two or three side chains possibly cannot be circumvented given the co-existing of similar α-C–H bonds60,61,62,63,64,65. To our delight, the α-cyanation took place selectively at the N-methyl position of diverse urea protected N-methyl amines under the optimized condition A, as shown in Fig. 3, left. A variety of N-methyl phenylethylamine derivatives involving electronically diverse substituents (e.g., Me, Ph, halo, CF3 et al.) at para, meta, and ortho positions of the aromatic ring were readily accommodated to the reaction, affording the corresponding α-nitrile amines in good yields and α’-selectivity (2a and 2b, 2d−2m). A larger gram-scale transformation of 1a (5 mmol) also took place smoothly to afford the product 2a in 57% yield. In the case of β-naphthylethylamine substrate, the product 2n was obtained in 65% yield with 18:1 rr (α’/β). For di-substituted phenylethylamines, the reaction also proceeded well with moderate yield and decent site-selectivity (2o and 2p). Besides the aryl group, the β-alkynyl substituent was also tolerated well in this C–C coupling process, delivering the desired products in moderate yield and good α’-selectivity (2r). Substrate bearing two phenyl groups at the β-position was compatible for α’-cyanation, despite the significant stability of possible β-benzylic radical (2s). Moreover, linear γ-phenylpropamine also survived the reaction condition, albeit with slightly decreased α’-selectivity (9:1), indicating that the L14-ligated Cu(II) intermediate could differentiate the subtle difference in steric environments of primary and common secondary C–H bonds (2t). In the case of N-Me benzylamine substrate that containing a competing highly reactive benzylic α-C–H bond, the α’-cyanation product 2u could also be obtained by overriding the intrinsic reactivity en route 2u’ (rr = 1:1). Besides, the reaction of α-naphthylethylamine delivered product 2v in a considerable yield with good selectivity at the primary α’-site over many other possible C–H bonds. Furthermore, functional groups such as, cyclohexyl (2w), and thienyl (2x) groups that tethered at the side chain of N-methyl amines were all tolerated without the loss in reaction efficiency or selectivity control. Notably, substrates that derived from pharmaceutical or bioactive molecules e.g., Isoxepac, Ibuprofen, and Nortriptyline could be well accommodated to the N-methyl C–H cyanation, allowing the valuable late-stage C–H functionalisation of complex molecules (2z, 2aa and 2aj). Subsequently we explored the scope of β-cyanation of the same set of starting materials by simply changing the ligand L14 to ligand L8 (Fig. 3, right). Generally, the electron-donating and withdrawing substituents tethered at different positions of aromatic ring were all tolerated under the reaction conditions and had negligible influence on the reaction efficiency and regioselectivity (3a and 3b, 3c−3l). The synthetic protocol for the β-cyanation also proved robust and readily scalable, with isolated 1.02 g of 3a on a 5 mmol scale. Moreover, substrates bearing 2-naphthalene, 1,3-benzodioxole, 2,3-dihydrobenzofuran, and thienyl groups were compatible as well (3n−3p, 3x). The late-stage cyanation of structurally complex substrates containing bioactive moieties, such as Tryptamine, Isoxepac, and Ibuprofen, took place smoothly with exclusive β-selectivity (3y−3aa). Furthermore, the ferrocene group was also tolerated to the reaction, affording 3ab in 62% yield. These results further demonstrate that the regio-divergency could be well regulated in the presence of different ligands.
Condition A: 1 (0.2 mmol), Cu(CH3CN)4BF4 (3.1 mg, 5 mol%), L (12 mol%), TMSCN (59.5 mg, 0.6 mmol), DCM (4 mL), N2, rt, 0.5 h; isolated yield; the regioselectivity ratios (rr) were determined by GC-MS or HPLC. [a]All rr (2/3) < 1:20.
In light of the lack of adaptive strategy for constructing β-chiral centers of amines via intermolecular C–H conversion11, we next explored the applicability of these asymmetric β-cyanation protocol. Generally, a suite of N-Me phenylethylamines were site-specifically transformed into the optical pure β-nitrile amines with comparable yields, excellent regio- and enantioselectivity (Fig. 4). Both electron-donating (3a*−3d*) and electron-withdrawing (3e*−3i*) substituents at para-position were tolerated to the reaction with satisfactory results (er > 95:5 in most cases). To demonstrate the practical utility and scalability of this methodology, a large-scale (5 mmol) synthesis of the chiral compound 3a* was carried out and a comparable result was obtained. Moreover, meta-substituted phenylethylamines were subjected to the reaction conditions and the corresponding cyanation products 3j*−3l* were formed with intact yields and selectivity. It is worth mentioning that substrate bearing ortho-F group delivered the product 3 m* in 65% yield, along with excellent 99:1 er and over 20:1 β-selectivity. Moreover, the reaction also exhibited perfect compatibility with di-substituted substrates at both 3,4-positions of aromatic ring (3n*−3q*). Varying the phenyl derivatives to thiophene did not hamper the reactivity of the reaction, albeit with slightly decreased yield and selectivity (3x*). Importantly, several complex bioactive relevant molecules were compatible as well and the corresponding chiral β-cyanation products were successfully obtained with excellent regio- and enantioselectivity (3y*−3aa*). Intriguingly, the protocol could also provide rapid access to both enantiomers by simply alternating the configuration of chiral ligand (3z* & ent-3z*). In addition, this methodology also worked smoothly with ferrocene-containing substrate, which gave 3ab* with excellent enantioselectivity and site-selectivity (95:5 er, 18:1 rr). The absolute configuration of 3a* was determined to be S by comparing the optical rotation and the chiral HPLC diagram of a 3a* derivative with that of an authentic chiral sample (See Section 6 in Supplementary Information for details). That of the other cyanation products was assigned accordingly.
Condition B: 1 (0.2 mmol), CuSCN (0.7 mg, 3 mol%), L* (7.5 mol%), TMSCN (59.5 mg, 0.6 mmol), CH3CN (4 mL), N2, rt, 0.5 h; isolated yield; the regioselectivity ratios (rr) were determined by GC-MS analysis or HPLC, and the enantiomeric ratios (er) were determined by HPLC analysis. [a]Cu(OTf)2 (3 mol%) was used.
Next, inspired by Liu’s work on the asymmetric allylic C–H bond cyanation40, we continued to investigate the asymmetric allylic β-C–H cyanation of amines under the developed condition B. However, a series of oxazoline-derived chiral ligands including L24 gave negative results. Gratifyingly, after extensive exploration of diverse ligands, it was found that the elaborately modified chiral diamine ligand (1S,2S)-L41, which was designed by incorporating two particular bulky groups as “long spreading arms” at side chains66, could provide decent enantioselectivity with exclusive β-selectivity (see Table S6 in Supplementary Information for more details). Under these conditions, various N-methyl homoallylic amines were efficiently converted into the stereochemically pure cyanides with comparable enantioselectivity (3ac*−3af*). Furthermore, the cyanation of 1,1-disubstitutied olefins also took place smoothly and only the β-nitrile chiral amines were obtained with good enantioselectivity (3ag*−3aj*). Beyond aryl alkylene variants, cycloalkenes were also tested, furnishing the target products with excellent regioselectivity and acceptable enantioselectivity (3ak* and 3al*).
Mechanistic studies
To get further insight into the reaction mechanism, several control experiments and isotope labeling experiments were conducted. It was previously found that the reaction of 1u, a substrate having both N-methyl and N-benzyl moieties, under the condition A led the formation of a mixture of 2u/2u’ (Fig. 3). However, running the reaction in the absence of L14 exclusively provided the benzylic cyanation product 2u’ (Fig. 5A). It is in line with the fact that benzyl C–H bond is more likely to undergo intramolecular 1,4-HAT than methyl because of the stabilized benzylic radical67. These results also suggest that the ligand should play a pivotal role in regioselectivity control and could even reverse the intrinsic reactivity. To elucidate the different radical species involved in the selective cyanation mechanism, a series of radical experiments were conducted (Fig. 5B). The addition of radical inhibitor 2,2,6,6-tetramethyl-1-piperdinyloxy (TEMPO) to the standard conditions of either condition A or condition B completely shut down the reactions, suggesting that these two reactions should proceed through radical pathways (Fig. 5B(a)). However, the α’-radical adduct product 2a-BHT was not observed in the presence of butylated hydroxytoluene (BHT) under condition A, probably due to the instability of primary methyl radical. In contrast, the formation of the β-radical was verified by the identification of its trapping product 3a-BHT under condition B (Fig. 5B(b)). Overall, these results firmly establish the involvement of radical pathways in the selective cyanation mechanism. Moreover, the non-linear effect study of 1a revealed a linear relationship between the ee value of 3a* and ligand (1R,2S)-L24, implying that a single chiral ligand-ligated copper intermediate should be involved in the enantio-determining transition state (Fig. 5C). Next, a series of deuterium-labeling experiments were performed to probe the nature of C–H bond cleavage steps. The cyanation of deuterated substrate 1a-[α’]d3 under condition B provided the β-nitrile product 3a*-[α’]d3 while the N-CD3 remained intact (Fig. 5D(a)). In sharp contrast, treating 1a-[α’]d3 with condition A resulted in the formation of a 1:1 mixture of 2a-[α’]d2 and 3a*-[α’]d3, in which no H/D distribution between α’ and β positions were observed (Fig. 5D(b)). These results unambiguously demonstrate that the site-divergent C–H cyanation undergoes straightforward 1,5-HAT or 1,4’-HAT, rather than reversible HAT process36,38 or radical transfer process (e.g., α’-C radical to β-C radical through 1,4-HAT)67,68. Furthermore, the successful regioselectivity control partially relied on the similar BDE of β-benzylic and α’ hydrogens (~89 kcal/mol vs ~93 kcal/mol)7, since a poor regioselectivity (1:1) was obtained when increasing the BDE of α’-C–H bonds by replacing N-CH3 with N-CD3 (~89 kcal/mol vs ~96 kcal/mol, Fig. 5D(b)). Finally, several kinetic isotopic effect (KIE) experiments were conducted between different 1a-dn (n = 0, 2–5) in mixed and parallel modes. The obtained KIE values of α’-cyanation from intermolecular competition (KIE = 2.2) and parallel reactions (KIE = 2.1) under condition A suggested that the 1,4’-HAT process might be the rate-determining step (Fig. 5E). Moreover, large primary KIE values (kH/kD > 2.5) were observed either in the intermolecular/intramolecular β-H/D competing reaction, or two parallel reactions under condition B, indicating that the 1,5-HAT process is probably involved in the rate-determining steps in the presence of (1R,2S)-L24 (Fig. 5F)69.
Condition A: Cu(CH3CN)4BF4 (5 mol%), L14 (12 mol%), TMSCN (3.0 equiv), DCM (0.05 M), N2, rt; Condition B: CuSCN (3 mol%), (1R,2S)-L24 (7.5 mol%), TMSCN (3.0 equiv), CH3CN (0.05 M), N2, rt. More details see SI. A Control experiments for 1 u with/without L14; B Radical inhibition and trapping experiments under condition A/B; C Non-linear effect study of β-cyanation under condition B; D Deuteration experiments; E KIE experiments of α’-cyanation under condition A; F KIE experiments of β-cyanation under condition B.
To gain insight into the origin of selectivity, DFT calculations were performed (See Section 8 in Supplementary Information for details). The free energy profiles for C–H cyanation at the β- and α’-positions in the Cu(CN)(L24) system are shown in Figs. S16–S1870,71. DFT results indicate that the HAT step serves as the rate-determining step, consistent with the kinetic isotope effect experiments, whereas the enantioselectivity in β-C–H cyanation is established during the radical recombination and reductive elimination steps. The steric repulsion between the substrate and the L24 ligand was found to play a crucial role in the observed enantioselectivity (Fig. S19). For the Cu(CN)(L14) system, the regioselectivity calculations were focused on the HAT step, which was identified as irreversible. The computed transition states show a small energetic preference for the pathway leading to the experimentally observed α’-cyanation product (Fig. S21). Additional analysis suggests that weak noncovalent interactions may contribute to stabilizing the transition state (Fig. S22).
Discussion
In this work, we have achieved the regio-divergent α’/β-C(sp3)–H bonds cyanation of secondary N-alkyl-N-methyl amines through ligand-controlled selective 1,4’/1,5-HAT. The reaction indicated that the presence of bulky ligand could facilitated the α’-selective C−H cyanation of secondary amines at the N-methyl moiety, while the less sterically demanding diamine ligand enabled the β-cyanation. Furthermore, two chiral ligands were developed that successfully mediated the enantioselective C–H cyanation of secondary amines at β-benzylic and allylic positions. This protocol facilitates rapid cyanation of various secondary amine-derived ureas under mild conditions, demonstrating broad substrate compatibility, excellent α’/β-regioselectivity, and remarkable β-enantioselectivity. Mechanistic studies suggest that the copper-bounded NCR species with different ligand environments plays a critical role in site selectivity during the irreversible HAT step, and the following two distinct reaction models would eventually lead to the regiodivergent C−H cyanation of secondary amines at different sites. In addition, the origin of the high site- and enantioselectivity has also been clarified by the DFT calculations. This study not only provides the alternative strategy for the regiodivergent and enantioselective C−H functionalisation of amines but also deepens the mechanistic understanding in the controllable HAT process. The pursue of ligand-controlled diverse functionalisation of dialkylamines with other reagents through selective HAT is on the way in our laboratory.
Methods
Regioselective for the α’/β-C(sp3)–H cyanation (Condition A)
In a dried sealed 10 mL Schlenk tube equipped with a stir bar, Cu(CH3CN)4BF4 (0.01 mmol, 5 mol%), L14/L8 (0.024 mmol, 12 mol%) and N-chloro substrate 1 (0.2 mmol) were dissolved in degassed DCM (4 mL) under N2 atmosphere, then TMSCN (0.6 mmol) was added. The tube was sealed with Teflon septum and stirred at room temperature for 0.5 h. After completed, the mixture was concentrated under reduced pressure, and the residue was purified by column chromatography or preparative thin-layer chromatography (PE/EA = 10/1) to provide desired products 2/3.
Enantioselective for the β-C(sp3)–H cyanation (Condition B)
In a dried sealed 10 mL Schlenk tube equipped with a stir bar, CuSCN (0.006 mmol, 3 mol%), (1R,2S)-L24/(1S,2S)-L41 (0.015 mmol, 7.5 mol%) and N-chloro substrate 1 (0.2 mmol) were dissolved in degassed CH3CN (4 mL) under N2 atmosphere, then TMSCN (0.6 mmol) was added. The tube was sealed with Teflon septum and stirred at room temperature for 0.5 h. After completed, the mixture was concentrated under reduced pressure, and the residue was purified by column chromatography or preparative thin-layer chromatography (PE/EA = 10/1) to provide desired products 3*.
Data availability
The experimental data generated in this study are provided in the Supplementary Information. The computational coordinate data generated in this study are provided in the Source Data, The X-ray crystallographic coordinates for structures reported in this study have been deposited at the Cambridge Crystallographic Data Centre (CCDC), under deposition numbers of CCDC 2382322 (2d), and CCDC 2382282 (3k). These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif. All data are available from the corresponding author upon request. Source data are provided with this paper.
References
Hu, S. et al. Ni-catalyzed enantioselective reductive cyclization/amidation and amination of 1,6-enynes and 1,7-enynes. Angew. Chem. Int. Ed. 63, e202413892 (2024).
Flick, A. C. et al. Synthetic approaches to the new drugs approved during 2019. J. Med. Chem. 64, 3604–3657 (2021).
Chen, L. et al. Photocatalytic carboxylation of C−N bonds in cyclic amines with CO2 by consecutive visible-light-induced electron transfer. Angew. Chem. Int. Ed. 62, e202217918 (2023).
Ni, S.-F. et al. Recent advances in γ-C(sp3)–H bond activation of amides, aliphatic amines, sulfanilamides and amino acids. Coord. Chem. Rev. 455, 214255 (2022).
Wu, X. & Zhu, C. Radical functionalization of remote C(sp3)–H bonds mediated by unprotected alcohols and amides. CCS Chem. 2, 813–828 (2020).
He, C., Whitehurst, W. G. & Gaunt, M. J. Palladium-catalyzed C(sp3)–H bond functionalization of aliphatic amines. Chem 5, 1031–1058 (2019).
Le, C., Liang, Y., Evans, R. W., Li, X. & MacMillan, D. W. C. Selective sp3 C–H alkylation via polarity-match-based cross-coupling. Nature 547, 79–83 (2017).
Loh, Y. Y. et al. Photoredox-catalyzed deuteration and tritiation of pharmaceutical compounds. Science 358, 1182–1187 (2017).
Shaw, M. H., Shurtleff, V. W., Terrett, J. A., Cuthbertson, J. D. & MacMillan, D. W. C. Native functionality in triple catalytic cross-coupling: sp3 C−H bonds as latent nucleophiles. Science 352, 1304–1308 (2016).
Chen, J., Tan, C., Rodrigalvarez, J., Zhang, S. & Martin, R. Site-selective distal C(sp3)−H bromination of aliphatic amines as a gateway for forging nitrogen-containing sp3 architectures. Angew. Chem. Int. Ed. 63, e202406485 (2024).
Chang, Y. et al. Enantioselective synthesis of N-alkylamines through β-amino C–H functionalization promoted by cooperative actions of B(C6F5)3 and a chiral lewis acid co-catalyst. J. Am. Chem. Soc. 143, 2441–2455 (2021).
Sarver, P. J. et al. The merger of decatungstate and copper catalysis to enable aliphatic C(sp3)–H trifluoromethylation. Nat. Chem. 12, 459–467 (2020).
Huang, Z., Wang, C. & Dong, G. A hydrazone-based exo-directing-group strategy for β C−H oxidation of aliphatic amines. Angew. Chem. Int. Ed. 55, 5299–5303 (2016).
McNally, A., Haffemayer, B., Collins, B. S. L. & Gaunt, M. J. Palladium-catalysed C–H activation of aliphatic amines to give strained nitrogen heterocycles. Nature 510, 129–133 (2014).
Rumo, C. et al. An artificial metalloenzyme based on a copper heteroscorpionate enables sp3 C–H functionalization via intramolecular carbene insertion. J. Am. Chem. Soc. 144, 11676–11684 (2022).
Lin, W., Zhang, K.-F. & Baudoin, O. Regiodivergent enantioselective C–H functionalization of Boc-1,3-oxazinanes for the synthesis of β2- and β3-amino acids. Nat. Catal. 2, 882–888 (2019).
Millet, A., Dailler, D., Larini, P. & Baudoin, O. Ligand-controlled α- and β-arylation of acyclic N-boc amines. Angew. Chem., Int. Ed. 53, 2678–2682 (2014).
Zhou, Z. et al. Enantioselective ring-closing C–H amination of urea derivatives. Chem 6, 2024–2034 (2020).
Lang, K., Torker, S., Wojtas, L. & Zhang, X. P. Asymmetric induction and enantiodivergence in catalytic radical C–H amination via enantiodifferentiative H-atom abstraction and stereoretentive radical substitution. J. Am. Chem. Soc. 141, 12388–12396 (2019).
Yang, Y., Cho, I., Qi, X., Liu, P. & Arnold, F. H. An enzymatic platform for the asymmetric amination of primary, secondary and tertiary C(sp3)–H bonds. Nat. Chem. 11, 987–993 (2019).
Kuroda, Y. et al. An iridium/aluminum cooperative strategy for the β-C(sp3)-H borylation of saturated cyclic amines. Angew. Chem. Int. Ed. 62, e202300704 (2023).
Smalley, A. P., Cuthbertson, J. D. & Gaunt, M. J. Palladium-catalyzed enantioselective C–H activation of aliphatic amines using chiral anionic binol-phosphoric acid ligands. J. Am. Chem. Soc. 139, 1412–1415 (2017).
Lv, X., Yang, Y., Zhou, L. & Zeng, X. Copper-catalyzed fluoroamide-directed remote C(sp3)-H bond functionalization through intramolecular hydrogen atom transfer. Eur. J. Org. Chem. 27, e202400027 (2024).
Zhang, Z., Chen, P. & Liu, G. Copper-catalyzed radical relay in C(sp3)–H functionalization. Chem. Soc. Rev. 51, 1640–1658 (2022).
Muñoz-Molina, J. M., Belderrain, T. R. & Pérez, P. J. Recent advances in copper-catalyzed radical C–H bond activation using N–F reagents. Synthesis 53, 51–64 (2021).
Sinha, S. K. et al. Toolbox for distal C–H bond functionalizations in organic molecules. Chem. Rev. 122, 5682–5841 (2022).
Sarkar, S., Cheung, K. P. S. & Gevorgyan, V. C–H functionalization reactions enabled by hydrogen atom transfer to carbon-centered radicals. Chem. Sci. 11, 12974–12993 (2020).
Zhang, Z.-H., Wei, H., Li, Z.-L. & Liu, X.-Y. Recent advances in radical-involved alkynylation of unactivated C(sp3)–H bonds by hydrogen atom abstraction. Synlett 32, 362–369 (2020).
Stateman, L. M., Nakafuku, K. M. & Nagib, D. A. Remote C–H functionalization via selective hydrogen atom transfer. Synthesis 50, 1569–1586 (2018).
Simons, R. T. et al. Directed photochemically mediated nickel-catalyzed (hetero)arylation of aliphatic C–H bonds. J. Am. Chem. Soc. 145, 3882–3890 (2023).
Bafaluy, D., Georgieva, Z. & Muñiz, K. Iodine catalysis for C(sp3)–H fluorination with a nucleophilic fluorine source. Angew. Chem. Int. Ed. 59, 14241–14245 (2020).
Short, M. A., Shehata, M. F., Sanders, M. A. & Roizen, J. L. Sulfamides direct radical-mediated chlorination of aliphatic C–H bonds. Chem. Sci. 11, 217–223 (2020).
Nanjo, T., Matsumoto, A., Oshita, T. & Takemoto, Y. Synthesis of chlorinated oligopeptides via γ- and δ-selective hydrogen atom transfer enabled by the N-chloropeptide strategy. J. Am. Chem. Soc. 145, 19067–19075 (2023).
Zhang, Z., Zhang, X. & Nagib, D. A. Chiral piperidines from acyclic amines via enantioselective, radical-mediated δ C–H cyanation. Chem 5, 3127–3134 (2019).
He, Y., Tian, C., An, G. & Li, G. β-C(sp3)−H chlorination of amide derivatives via photoinduced copper charge transfer catalysis. Chin. Chem. Lett. 35, 108546 (2024).
Wang, M., Huang, Y. & Hu, P. Terminal C(sp3)−H borylation through intermolecular radical sampling. Science 383, 537–544 (2024).
Xu, J. et al. Site-selective α-C(sp3)–H arylation of dialkylamines via hydrogen atom transfer catalysis-enabled radical aryl migration. Nat. Commun. 15, 6791 (2024).
Shen, Y., Funez-Ardoiz, I., Schoenebeck, F. & Rovis, T. Site-selective α-C–H functionalization of trialkylamines via reversible hydrogen atom transfer catalysis. J. Am. Chem. Soc. 143, 18952–18959 (2021).
Li, J. et al. Mechanistic insights into copper-catalyzed asymmetric cyanation of allylic C–H bonds. J. Am. Chem. Soc. 147, 14756–14768 (2025).
Li, J. et al. Site-specific allylic C–H bond functionalization with a copper-bound N-centred radical. Nature 574, 516–521 (2019).
Chen, K. et al. Functional-group translocation of cyano groups by reversible C–H sampling. Nature 620, 1007–1012 (2023).
Cai, C.-Y. et al. Photoelectrochemical asymmetric catalysis enables site- and enantioselective cyanation of benzylic C–H bonds. Nat. Catal. 5, 943–951 (2022).
Wang, F., Chen, P. & Liu, G. Copper-catalysed asymmetric radical cyanation. Nat. Synth. 1, 107–116 (2022).
Bao, X., Wang, Q. & Zhu, J. Dual photoredox/copper catalysis for the remote C(sp3)−H functionalization of alcohols and alkyl halides by N-alkoxypyridinium salts. Angew. Chem. Int. Ed. 58, 2139–2143 (2019).
Wang, C.-Y. et al. Enantioselective copper-catalyzed cyanation of remote C(sp3)-H bonds enabled by 1,5-hydrogen atom transfer. iScience 21, 490–498 (2019).
Zhang, W. et al. Enantioselective cyanation of benzylic C–H bonds via copper-catalyzed radical relay. Science 353, 1014–1018 (2016).
Qian, S., Lazarus, T. M. & Nicewicz, D. A. Enantioselective amino- and oxycyanation of alkenes via organic photoredox and copper catalysis. J. Am. Chem. Soc. 145, 18247–18252 (2023).
Duan, S. et al. Catalytic chemo- and regioselective radical carbocyanation of 2-azadienes for the synthesis of α-amino nitriles. Angew. Chem. Int. Ed. 62, e202300605 (2023).
Bower, J. K. et al. C(sp3)–H cyanation by a formal copper(III) cyanide complex. Chem. Sci. 14, 1301–1307 (2023).
Ratani, T. S., Bachman, S., Fu, G. C. & Peters, J. C. Photoinduced, copper-catalyzed carbon–carbon bond formation with alkyl electrophiles: cyanation of unactivated secondary alkyl chlorides at room temperature. J. Am. Chem. Soc. 137, 13902–13907 (2015).
Wang, W. et al. Copper-catalyzed asymmetric remote C(sp3)–H alkylation of N-fluorocarboxamides with glycine derivatives and peptides. J. Am. Chem. Soc. 146, 6307–6316 (2024).
Liu, L. et al. Copper-catalyzed intermolecular enantioselective radical oxidative C(sp3)−H/C(sp)−H cross-coupling with rationally designed oxazoline-derived N,N,P(O)-ligands. Angew. Chem. Int. Ed. 60, 26710–26717 (2021).
Wang, C.-Y. et al. Enantioselective copper-catalyzed remote C(sp3)–H alkynylation of linear primary sulfonamides. Org. Lett. 22, 4006–4009 (2020).
Zhang, Z.-H. et al. Copper-catalyzed enantioselective Sonogashira-type oxidative cross-coupling of unactivated C(sp3)−H bonds with alkynes. Nat. Commun. 10, 5689 (2019).
Chen, H., Jin, W. & Yu, S. Enantioselective remote C(sp3)–H cyanation via dual photoredox and copper catalysis. Org. Lett. 22, 5910–5914 (2020).
Liu, R.-Z. et al. Generation and reactivity of amidyl radicals: manganese-mediated atom-transfer reaction. Angew. Chem. Int. Ed. 59, 4428–4433 (2020).
Qin, Q. & Yu, S. Visible-light-promoted remote C(sp3)–H amidation and chlorination. Org. Lett. 17, 1894–1897 (2015).
Zeng, X. et al. Copper-catalyzed, chloroamide-directed benzylic C–H difluoromethylation. J. Am. Chem. Soc. 141, 19941–19949 (2019).
Fan, W. et al. Electrophotocatalytic decoupled radical relay enables highly efficient and enantioselective benzylic C–H functionalization. J. Am. Chem. Soc. 144, 21674–21682 (2022).
Yusan, B., Amuti, A., Xu, X. J. & Wusiman, A. NIS/TEMPO-promoted oxidative cyanation and nitro-mannich reaction of tertiary amines. Eur. J. Org. Chem. 27, e202400144 (2024).
Grundke, C. et al. Photochemical α-aminonitrile synthesis using Zn-phthalocyanines as near-infrared photocatalysts. J. Org. Chem. 87, 5630–5642 (2022).
Xu, F. et al. Iron(III)-catalyzed α-cyanation and carbonylation with 2-pyridylacetonitrile: divergent synthesis of α-amino nitriles and tetrahydroisoquinolinones. Org. Biomol. Chem. 20, 7031–7035 (2022).
Lennox, A. J. J. et al. Electrochemical aminoxyl-mediated α-cyanation of secondary piperidines for pharmaceutical building block diversification. J. Am. Chem. Soc. 140, 11227–11231 (2018).
Suzuki, K., Tang, F., Kikukawa, Y., Yamaguchi, K. & Mizuno, N. Visible-light-induced photoredox catalysis with a tetracerium-containing silicotungstate. Angew. Chem. Int. Ed. 53, 5356–5360 (2014).
Yamaguchi, K., Wang, Y. & Mizuno, N. A widely applicable regioselective aerobic α-cyanation of tertiary amines heterogeneously catalyzed by manganese oxides. ChemCatChem 5, 2835–2838 (2013).
Wang, F.-L. et al. Mechanism-based ligand design for copper-catalysed enantioconvergent C(sp3)–C(sp) cross-coupling of tertiary electrophiles with alkynes. Nat. Chem. 14, 949–957 (2022).
Shimizu, D., Kurose, A. & Nishikata, T. Remote nucleophilic substitution at a C(sp3)–H bond of α-bromocarboxamides via 1,4-hydrogen atom transfer to access N-acyl-N,O-acetal compounds. Org. Lett. 24, 7873–7877 (2022).
Xie, J. et al. New catalytic radical process involving 1,4-hydrogen atom abstraction: asymmetric construction of cyclobutanones. J. Am. Chem. Soc. 143, 11670–11678 (2021).
Simmons, E. M. & Hartwig, J. F. On the interpretation of deuterium kinetic isotope effects in C–H bond functionalizations by transition-metal complexes. Angew. Chem. Int. Ed. 51, 3066–3072 (2012).
Chen, X. et al. Mechanistic insights into copper-mediated benzylic C(sp3)–H bond trifluoromethylation. Org. Biomol. Chem. 22, 8480–8487 (2024).
Fang, C. et al. Mechanistically guided predictive models for ligand and initiator effects in copper-catalyzed atom transfer radical polymerization (Cu-ATRP). J. Am. Chem. Soc. 141, 7486–7497 (2019).
Acknowledgements
This work was supported by National Natural Science Foundation of China (No. 22301275 for Y.-J.M., 22371256 for Y.-F.Y.), the Research Foundation for Advanced Talents from ZJUT (No. 2021101016929 for S.-J.L.), Zhejiang Provincial Natural Science Foundation of China (LMS25B020004 for S.-J.L., LR25B020002 for Y.-F.Y.), Beidou Scientific Research Project of Zhejiang University of Technology (D.-Q.X.), and the Fundamental Research Funds for the Provincial Universities of Zhejiang (RF-C2022006 for Y.-F.Y.).
Author information
Authors and Affiliations
Contributions
S.-J.L., D.-Q.X., and Z.-Y.X. conceived and designed the project. Y.-J.M. developed the reaction and prepared the supplementary information. Y.-F.Y. and X.C. designed and performed the DFT calculations. Q.P., H.-L.L., and K.Z. performed the experiments and collected the data. Y.-J.M. and S.-J.L. prepared the manuscript with feedback from all co-authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Shuichi Nakamura and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mao, YJ., Chen, X., Li, HL. et al. Ligand-controlled regiodivergent and enantioselective C–H cyanation of secondary amines. Nat Commun 17, 1869 (2026). https://doi.org/10.1038/s41467-026-68598-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-68598-8