Abstract
Carbodications, bearing two positive charges on carbon atoms, are emerging as powerful superelectrophiles in organic synthesis. Traditionally formed through single-electron oxidation of overcrowded electron-rich alkenes or acid-mediated dehydration of diols, these species show high reactivity toward carbon-based nucleophiles. However, their reactivities with heteroatom nucleophiles like amines and water remain underexplored, largely due to difficulties in controlling oxidation and undesirable acid–base interactions. Here, we report that carbodication species, generated under mild electrochemical or photocatalytic conditions in the presence of heteroatom nucleophiles, display remarkable electrophilic reactivity toward primary amines and water. This enables an intermolecular umpolung [4 + 1] cycloaddition, proceeding via a cis-olefin-bridged dicationic intermediate, energetically favored over its trans-isomer, yielding a class of thioxanthene-based dispirocycles with 2,5-dihydro-1H-pyrrole or 2,5-dihydrofuran central cores. Theoretical studies of electrochemical cycloaddition with aniline suggest that hydrogen bonding between aniline and the fluorine atom of the nBu4NPF6 electrolyte stabilizes the transition state, promoting nucleophilic addition.
Similar content being viewed by others
Introduction
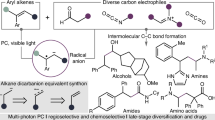
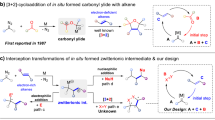
Dicationic species carrying two positive charges within a single molecule have garnered significant attention in synthetic chemistry and materials science due to their pronounced superelectrophilic reactivity and pronounced redox activity1,2,3,4,5,6,7,8,9,10,11. Among these, carbodications with spatially separated charges on two carbon atoms are particularly notable. Their stability is typically achieved through extended π-conjugation and incorporation of electron-donating groups, which not only enhance their electrophilic behavior in carbon–carbon bond-forming reactions but also impart distinctive electrochromic properties. For instance, the Suzuki group has isolated highly stable carbodications based on overcrowded ethylene scaffolds such as tetraaryl-9,10-anthraquinodimethane (Ar4AQD) and its bridged diol analog12,13. These carbodications exhibit reversible redox behavior and can be generated by either two-electron oxidation of electron-rich alkenes (e.g., with Magic Blue) or via Brønsted acid-promoted dehydration of diols (Fig. 1a). In parallel, our group and others have reported that fluorene end-capped 1,3-dienes (DFDPE) and 11,12-dihydroindeno[2,1-a]fluorene-11,12-diols ([2,1-a]-IFOH) undergo two-fold Friedel–Crafts cyclizations to form dispirocyclic π-systems (e.g., DSFIIF) and thiophene-fused polycycles (e.g., [2,1-a]-IF-Th), respectively (Fig. 1b)14,15. These transformations are proposed to proceed via superelectrophilic carbodication intermediates generated either through FeCl3-mediated two-step single-electron oxidations or Brønsted acid-promoted dehydration of diols. While these carbodications exhibit notable reactivity toward weak carbon-based nucleophiles, their reactivity with heteroatom nucleophiles such as amines or water remains largely unexplored. This limitation is likely due to competing oxidation of electron-rich dication precursors in the presence of heteroatom nucleophiles. In addition, unfavorable acid-base interactions between strong Brønsted acids and these heteroatoms suppress efficient diol dehydration process and also reduce the nucleophilicity of the heteroatoms. Furthermore, products sensitive to strongly acidic conditions may revert to stable carbodication intermediates, leading to undesirable side reactions. Consequently, developing methods for heteroatom-based bifunctionalization of carbodications remains a significant synthetic challenge.
a Selected examples for the formation of isolable carbodications. b Carbodication reactivity towards carbon nucleophiles. c Design of [4 + 1] cycloaddition between 1,3-dienes and heteroatom nucleophiles versus reported methods. d This study on electrochemical and photocatalytic generation of a cis-olefin-bridged carbodication intermediate for an umpolung [4 + 1] cycloaddition using primary amines and H2O. TMS: trimethylsilyl, EDG: electron-donating group, Nu: nucleophile, Ts: p-toluenesulfonyl, pin: pinacol, Gr: graphite.
Recently, mild oxidative strategies such as electrosynthesis and photocatalysis have emerged as promising alternatives, offering precise redox control and improved environmental compatibility16,17,18,19,20,21,22,23,24. We assumed that these methods could enable the selective formation of stable dicationic species in the presence of nucleophilic heteroatoms under neutral and mild oxidative conditions, thereby avoiding the harshness associated with traditional oxidants and strong acids. Indeed, previous studies have shown that carbodications can be generated via the cation pool method through oxidative C–C bond cleavage under low-temperature electrolysis in the absence of nucleophiles, and that the resulting carbodications display remarkable electrophilic reactivity toward various carbon nucleophiles24. As illustrated in Fig. 1b, our previous work on the formation of dispirocyclic DSFIIF revealed that its construction is facilitated by a trans-olefin-bridged carbodication conformer, which promotes intramolecular two-fold Friedel–Crafts cyclization14. Building on this, we considered whether a cis-olefin-bridged carbodication, thermodynamically more stable than its trans counterpart, could be selectively accessed via single-electron oxidation of electron-rich 1,3-dienes under electrochemical or photocatalytic conditions in the presence of heteroatom nucleophiles (Fig. 1c). If successful, this would enable an intermolecular umpolung [4 + 1] cycloaddition between electronically mismatched 1,3-dienes and simple heteroatom nucleophiles such as primary amines or water. Although [4 + 1] cycloadditions of 1,3-dienes with one-atom synthons such as CO, isocyanides, carbenes, nitrenes, or silylenes are well established, often via transition-metal catalysis (Fig. 1c)25,26,27,28,29,30, the use of primary amines or water as heteroatom nucleophiles in this context has not been reported to date31. Herein, we report an umpolung [4 + 1] cycloaddition of thioxanthene-end-capped 1,3-dienes with primary amines or water, acting as N1 or O1 synthons, respectively (Fig. 1d). This transformation proceeds via a cis-dicationic intermediate formed under mild oxidative conditions enabled by electrosynthesis or photocatalysis, ultimately affording structurally unique thioxanthene-based dispirocycles incorporating central 2,5-dihydro-1H-pyrrole or 2,5-dihydrofuran cores.
Results and discussion
Electrochemical [4 + 1] cycloaddition of thioxanthene end-capped dienes with primary amines
Building on our previous studies of the oxidative ring expansion of biphenyl-tethered methylene thioxanthenes to thiepine-embedded polyaromatics, we observed that the methylene moiety undergoes selective oxidation to a radical cation, owing to its high electron density32. Drawing from these findings and our prior work on carbodication reactivity (Fig. 1b)14, we designed and synthesized a thioxanthene end-capped 1,3-diene substrate, 1a, to explore a potential [4 + 1] cycloaddition pathway (Fig. 2). Cyclic voltammetry (CV) of 1a in a DCM/HFIP (5:1 v/v) mixture revealed a single oxidation wave and two distinct one-electron reduction peaks (Fig. 2a). To gain further insight into the oxidation process, differential pulse voltammetry (DPV) was conducted under identical solvent system. The DPV trace showed a slight separation of the oxidation wave into two closely spaced peaks (Fig. 2a, inset), suggesting that 1a undergoes a two-electron oxidation with nearly identical first and second oxidation potentials. These electrochemical features are consistent with density functional theory (DFT) calculations, which predict first and second oxidation potentials of 0.78 V and 0.59 V vs SCE, respectively, supporting the nearly simultaneous formation of the dicationic species 1a2+ (Table S4, SI). This behavior can be rationalized by the preferential two-electron oxidation to yield the thermodynamically stable aromatic thioxanthylium dication, rather than the less stable radical cation formed by single-electron oxidation. The stability of the cis-dication 1a2+ is further enhanced by intramolecular π–π interactions between the two thioxanthylium polyaromatic units. The large peak-to-peak separation ( ~ 0.5 V) between oxidation and reduction in the CV trace indicates the high stability of the dicationic intermediate12,13. Moreover, the oxidation potential of 1a (0.79 V vs Ag/AgNO3) is 0.15 V lower than that of aniline (0.94 V), confirming that 1a is more easily oxidized under anodic conditions. DFT analysis of 1a2+ in the presence of the PF₆‒ counteranion reveals that the cis conformer is 3.4 kcal/mol more stable than the trans form, supporting the proposed two-step electrophilic [4 + 1] cycloaddition pathway (Fig. 2b and Fig. S7, SI). In contrast, when the PF₆⁻ anion is omitted, the trans-dication becomes 3.6 kcal/mol more stable than the cis form. These findings suggest that the PF₆⁻ counteranion mitigates charge–charge repulsion between the two cationic centers by cation-anion interactions, thereby stabilizing the cis configuration. An electrostatic potential (MEP) map of the cis-dication indicates delocalization of positive charge across the individual bis(thioxanthylium) 14π-aromatic systems (Fig. 2c). Furthermore, non-covalent interaction (NCI) analysis confirms additional stabilization of the cis conformer through intramolecular π–π interactions between the parallel thioxanthylium π-surfaces (Fig. S8, SI)33,34.
a Cyclic voltammograms of 1a, aniline, and 2a in DCM/HFIP (5:1 v/v). Inset: differential pulse voltammetry (DPV) of 1a in DCM/HFIP (5:1 v/v). b DFT calculations of stabilization energy difference of cis- and trans-dication conformers of 1a. c DFT calculations of molecular electrostatic potential (MEP) of the cis-dication. Calculations were performed at the SMD(HFIP)/M06-2X/def2-SVP level.
Guided by these electrochemical and theoretical insights, we optimized the electrochemical [4 + 1] cycloaddition of 1a with aniline using an undivided electrochemical cell (Table 1). The optimal setup employed graphite electrodes (anode and cathode), a constant current of 3 mA, and nBu4NPF6 in DCM/HFIP (5:1) at room temperature for 3 h, yielding dispirocycle 2a in 98% yield (entry 1). The dispirocyclic structure of 2a was determined unambiguously by X-ray crystallography analysis. HFIP proved critical for product selectivity35; replacing it with isopropanol or omitting it led to formation of the reduced byproduct 1a′ instead of 2a (entries 2 and 3). Among various electrolytes, those containing fluorine, nBu4NPF6 (98%), nBu4NBF4 (97%), and nBu4NOTf (97%), outperformed nBu4NClO4 (86%) and nBu4NBr (75%), suggesting a beneficial role of fluorine (entries 1, 4–7). Switching the graphite cathode for platinum had minimal effect, but using platinum at both electrodes significantly reduced the yield, underscoring the importance of the graphite anode in the two-electron oxidation (entries 8 and 9)36. Lowering the current to 1 mA extended the reaction time to 9 h to maintain high yield (entry 10), while increasing the current to 5 mA led to decreased yield (entry 11), likely due to overoxidation and partial decomposition of 2a, which itself shows a relatively low oxidation peak at 0.94 V vs Ag/AgNO3 (Fig. 2a). No product formation was observed in the absence of electric current, confirming the necessity of the electrochemical approach. During optimization, it was noted that no dispirocyclic analogues such as DSFIIF (Fig. 1b)14 were detected.
Given the remarkable electrochemical performance observed, a broad range of primary amines was evaluated under the optimized electrochemical conditions (Fig. 3a). Primary aryl amines with electron-donating substituents such as methyl, tert-butyl, and methoxy at the para-position of the phenyl ring showed excellent compatibility, delivering the corresponding dispirocycles 2b–2 d in high yields. Notably, the reaction between 1a and para-methoxy-substituted arylamine yielded product 2 d in high yield when 10 mol% nBu4NI was added as an additive, likely due to suppression of overoxidation of the electron-rich product 2 d. Aryl amines bearing electron-withdrawing groups such as Br, Cl and CF₃ also furnished the corresponding products 2e–2 g in good to high yields, despite their decreased nucleophilicity. The aryl amine with two methyl groups at the 3,5-positions of the phenyl ring, owing to its enhanced nucleophilicity, afforded dispirocycle 2 h in 86% yield. Primary alkyl amines such as 1-hexylamine and benzylamine were also compatible with the reaction conditions, providing dispirocycles 2i and 2j in good to high yields. However, substrates like p-tosylamide and tert-butyl carbamate, which possess electron-withdrawing substituents on nitrogen, failed to react due to their low nucleophilicity. Subsequently, the effect of substituents on the 1,3-diene moiety of substrates 1 was investigated (Fig. 3b). Aryl-substituted dienes with electron-donating groups at the para-position of the phenyl ring exhibited high reactivity, delivering the corresponding dispirocycles 2k–2n in good to high yields. Similarly, dienes bearing halogens such as fluorine or chlorine at the para-position gave products 2o and 2p in moderate to good yields, without any observable dehalogenation. For the sterically hindered 1,3-diene bearing a 3,5-dimethylphenyl group, an elevated temperature (50 °C) was required to drive the cycloaddition with aniline, resulting in a 65% yield of product 2q. Additionally, a 1,3-diene substituted with two n-butyl groups reacted smoothly, affording product 2r in good yield. Frontier molecular orbital (FMO) analysis of dispirocycle 2a revealed a clear spatial separation between the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) (Fig. S5, SI). The HOMO is primarily localized on the two electron-rich thioxanthene units and the nitrogen atom, whereas the LUMO is distributed over the two phenyl rings and the bridged C = C bond in the dihydro-1H-pyrrole core. The HOMO energy level of compound 2a, determined by CV analysis, was −5.09 eV, which correlates well with the calculated value of −5.29 eV.
a Substrate scope of primary amines. b Substrate scope of thioxanthene end-capped 1,3-dienes 1. Isolated yields are shown after silica gel chromatography. a Reaction conditions: 1 (0.1 mmol), nBu4NPF6 (0.3 mmol), amine (0.15 mmol), DCM/HFIP (0.03 M, v/v = 5/1) as solvent, graphite (Gr) used as anode and cathode electrodes in 5 mL ElectraSyn vial at constant current of 3 mA under Ar at room temperature for 3 h. b 10 mol% of nBu4NI was used as an additive.
Considering the pronounced selectivity and reactivity enhancement imparted by HFIP as a cosolvent in the electrochemical transformation (Table 1), we further investigated the oxidation potentials of aniline and compound 1a to elucidate HFIP’s role. CV analysis revealed that aniline exhibits an oxidation peak at 0.94 V (vs Ag/AgNO3) in a DCM/HFIP (5:1 v/v) solvent mixture, anodically shifted compared to its peak in pure DCM (0.83 V vs Ag/AgNO3) (Fig. 4a). This shift likely arises from hydrogen bonding interactions between aniline and HFIP. In contrast, compound 1a displays similar reversible redox behavior in both solvents; however, its oxidation wave undergoes a cathodic shift in DCM/HFIP (0.79 V vs Ag/ AgNO3) compared to DCM (0.83 V vs Ag/ AgNO3) (Fig. 4b). As a result, the oxidation potential gap between 1a and aniline increases by 0.15 V in the presence of HFIP, favoring the selective two-electron anodic oxidation of 1a during the electrochemical process. To obtain more precise oxidation information at the anode, we monitored the real-time anodic potentials of the reaction of 1a under standard electrolysis conditions using an Ag/AgNO3 reference electrode (Fig. 4c). The anodic potential gradually increased from 1.22 V to 1.48 V over 110 min, at which point product 2a was obtained in 92% yield, with 7% of unreacted 1a remaining. The potential then rose rapidly to 1.66 V, and after an additional 70 min, 2a was obtained in 98% yield. These applied potentials are higher than the oxidation potentials of both 1a and aniline, enabling efficient oxidation of both species. Moreover, the sharp increase in potential after 110 min suggests the onset of aniline oxidation, further supporting the selective anodic oxidation driven by the difference in oxidation potentials between 1a and aniline.
a Effect of HFIP on oxidation potentials of aniline in DCM and DCM/HFIP. b Effect of HFIP on oxidation potentials of 1a in DCM and DCM/HFIP. c Measurement of the real-time anodic potentials under standard electrolysis conditions using an Ag/AgNO3 reference electrode. d Generation of dication species 1a2+ and its redox property under standard electrolysis conditions in a divided cell. e Cycloaddition of 1a2+ with PhNH2 under current-free conditions. f Proposed electrochemical cycloaddition mechanism. g Theoretical study of transition states for the intermolecular nucleophilic addition of aniline to the cis-dication. h Theoretical study of transition states for the intramolecular nucleophilic addition. Calculations were performed at the SMD(HFIP)/M06-2X/def2-SVP level. 1H NMR yields are shown using CH2Br2 as an internal standard.
To directly observe the dicationic species 1a2+, anodic oxidation of 1a was carried out under standard electrolysis conditions in the absence of amine using a divided cell (Fig. 4d). The resulting red solid was analyzed by 1H NMR spectroscopy in CD3CN, which showed complete consumption of 1a (Fig. S11, SI). High-resolution mass spectrometry (HRMS) detected signals at m/z 285.0732 and 715.1112, corresponding to 1a2+ and [1a2+]PF₆⁻, respectively (Fig. S12, SI). The 1H NMR spectrum of 1a²⁺ recorded in CD3CN displayed a single set of signals (Fig. S11, SI), indicating the formation of a single stereoisomer of the dicationic species. Furthermore, cathodic reduction of this red solid (1a2+) under identical electrolysis conditions in a divided cell regenerated 1a in 75% yield, as determined by 1H NMR analysis (Fig. S11, SI). Collectively, these spectroscopic and redox property provide strong evidence for the efficient generation of the dicationic species 1a2+ under anodic oxidation conditions.
To investigate the intrinsic reactivity of 1a2+ toward amines, the in situ generated 1a2+ was treated with aniline in the absence of applied current (Fig. 4e). The reaction afforded 2a in 63% yield, along with 23% recovered 1a (Fig. S13, SI). When the solvent was switched to DCM, 2a was obtained in 33% yield with 37% recovered 1a. These results demonstrate that 1a2+ is indeed a reactive intermediate capable of [4 + 1] cycloaddition with amines, even without electrolysis. The moderate efficiency observed under these conditions likely arises from the reducing nature of aniline toward 1a2+, as well as the generation of PhNH3+ species during product formation. In contrast, reactions in undivided cells proceed more efficiently, likely due to selective anodic oxidation of 1a and facilitated proton transfer enabled by aniline deprotonation and the cathodic reduction of PhNH3+ (Fig. 4f). Notably, during the investigation of substrate scope, no dispirocyclic analogues such as DSFIIFs (Fig. 1b), which are proposed to arise from a trans-dicationic intermediate (Fig. 2b), were detected. Together, these experimental observations and theoretical analysis suggest that the observed dicationic species 1a2+ adopts a cis-geometry.
To compare our electrochemical oxidation with a chemical approach, we tested molecular iodine (I2) (Figs. S14 and SI), which has been previously used to generate 1,2-ethylene dications2. When I2 (1.5 equiv) was used to the reaction of 1a with aniline in CH2Cl2 or CH2Cl2/HFIP (5:1), the desired product 2a was formed in only 55% and 52% yield, respectively, and a significant amount of 1a was recovered. In contrast, I2 was completely ineffective with alkylamines such as n-hexylamine, producing no detectable products, including the targeted cycloadduct 2i. These results demonstrate the superior oxidation efficiency of electrochemical method in promoting the [4 + 1] cycloaddition.
The proposed electrochemical cycloaddition mechanism is depicted in Fig. 4f. Initially, 1a undergoes a two-step, one-electron anodic oxidation at its 1,3-diene moiety, preferentially over aniline, yielding the cis-dication intermediate B (1a2+) via the cis-radical cation intermediate A. To gain insight into the subsequent intermolecular nucleophilic addition of aniline to dication B (1a2+), DFT calculations were conducted to identify energetically favorable transition states (TSs). These calculations indicate that the formation of an intermolecular N–H⋯F hydrogen bond between aniline and a fluorine atom from the nBu4NPF6 electrolyte significantly stabilizes transition state TSb–c, reducing the activation free energy (ΔG‡) to 3.4 kcal/mol (Fig. 4g). In the absence of this hydrogen bond, the corresponding TS′b–c has a substantially higher ΔG‡ of 7.5 kcal/mol. Similarly, the TS′c-d, stabilized by an intramolecular N–H⋯F hydrogen bond, shows a ΔG‡ of 6.8 kcal/mol, whereas TSc-d, lacking this interaction, exhibits a higher ΔG‡ of 13.8 kcal/mol (Fig. 4h). Despite this, the intermediates C′ and D′ that feature the hydrogen bonds are thermodynamically less stable than C and D, which do not involve such interactions. These computational results highlight the crucial role of hydrogen bonding, especially involving the electrolyte, in facilitating both the nucleophilic addition and cycloaddition steps under mild conditions. Finally, deprotonation of the cationic intermediate D or D′ by aniline produces the cycloadduct 2a along with PhNH3+. Subsequent cathodic reduction of PhNH3+ generates hydrogen gas and regenerates aniline.
Photocatalytic [4 + 1] cycloaddition of thioxanthene end-capped dienes with H2O
Encouraged by the remarkable reactivity of the cis-dication species toward primary amines, we next investigated its potential reactivity with water (H2O) as a hetero-nucleophile. We anticipated a comparable [4 + 1] cycloaddition, leading to the formation of a 2,5-dihydrofuran core. However, under previously optimized electrochemical conditions, the desired cycloaddition between 1a and H2O did not occur, as water was preferentially electrolyzed to produce H2 and O2 gases. To suppress this competing electrolysis of water, we examined the oxidative electrolysis of 1a in the presence of H2O under acidic conditions. When 0.01 M HCl or an aqueous KCl/HCl buffer (pH 2.0) was used under standard electrolysis conditions, no desired product 3a was detected, and 1a was largely recovered unchanged (Fig. S15, SI). To further overcome the competing electrolysis of water while still enabling generation of the active cis-dication intermediate, we turned to a photocatalytic strategy. After screening several conditions, we found that irradiation of a mixture containing Ru(bpy)3Cl2·6H2O (photocatalyst) and (NH4)2S2O8 (oxidant) in a 9:1 DCE/H2O solvent system under blue light (440 nm) at room temperature for 2 h afforded the desired dispirocycle 3a in 98% yield (Table 2, entry 1). The selected irradiation wavelength effectively excites the Ru-photocatalyst. Notably, compounds 1a and 3a have absorption onsets at 395 nm and 330 nm, respectively, and thus do not interfere with the excitation of the photocatalyst (Fig. 5a). No product formation was observed in the absence of (NH4)2S2O8 (entry 2), confirming its essential role as the oxidant. (entry 2). Alternative photocatalysts, including Ru(bpz)3PF6, Ir(ppy)3, and MesAcrMe+ClO4–, showed reduced or negligible activity in promoting 3a formation when paired with (NH₄)2S2O8 (entries 3–5). Replacing (NH₄)2S2O8 with K2S2O8 enabled the reaction, albeit with a reduced yield of 80% (entry 6), while other oxidants such as AgNO3 and H2O2 proved largely ineffective (entries 7 and 8), underscoring the unique role of the persulfate (S2O82–) anion. Control experiments further demonstrated that no product was formed in the absence of Ru(bpy)3Cl2·6H2O, either under light irradiation with (NH₄)2S2O8 or under thermal conditions at 55 °C without light (entries 9 and 10). Together, these results establish Ru(bpy)3Cl2·6H2O and (NH4)2S2O8 as an effective and synergistic photoredox system for generating the cis-dication intermediate and facilitating the [4 + 1] cycloaddition with water. Interestingly, when the previously described [4 + 1] cycloaddition of 1a with aniline was performed under these optimized photocatalytic conditions, only a low yield (24%) of product 2a was obtained. This outcome is likely due to oxidative degradation of aniline, which may act as a reductive quencher of the Ru(bpy)3Cl2·6H2O photocatalyst. For comparison with common chemical oxidants, I2 was used in the cycloaddition of 1a with H2O at room temperature (Fig. S14c, SI). However, no desired product 3a was obtained, and 1a was recovered in 72% yield. This result highlights the superior efficiency of the present photocatalytic oxidation in promoting the [4 + 1] cycloaddition.
a UV-vis absorption of 1a and 3a. b Substrate scope of thioxanthene end-capped 1,3-dienes 1. Isolated yields are shown after silica gel chromatography.
We next evaluated the substrate scope of this photocatalytic [4 + 1] cycloaddition using the optimized conditions. As shown in Fig. 5b, thioxanthene end-capped substrates bearing electron-donating aryl groups on the 1,3-diene moiety, such as tolyl and tert-butylphenyl groups, afforded the corresponding products 3b and 3c in high yields. In contrast, substrates with electron-rich alkoxy substituents (e.g., methoxy and decyloxy) gave lower yields of 3 d and 3e, likely due to partial decomposition via competing single-electron oxidation pathways facilitated by the high electron density of the alkoxy groups. Substrates bearing sterically bulky 3,5-dimethylphenyl groups afforded the corresponding spirocycle 3 f in 75% yield. Electron-withdrawing haloaryl groups such as fluorine and chlorine were also well tolerated, delivering products 3 g and 3 h in good yields with halogen substituents intact. Additionally, alkyl-substituted substrates containing n-butyl or bulky cyclohexyl groups underwent efficient [4 + 1] cycloaddition to form 3i and 3j in 73% and 68% yields, respectively. Structural confirmation of the spirocyclic framework was provided by X-ray crystallographic analysis of compound 3a. DFT calculations indicate that 3a possesses a similar electronic structure to 2a: its HOMO is delocalized over the two thioxanthene moieties and the oxygen atom, while the LUMO is localized over the two phenyl rings across the C = C bond of the 2,5-dihydrofuran ring (Fig. S5, SI). The HOMO energy of 3a is estimated to be –5.35 eV from CV analysis, which is slightly lower than that of 2a, likely due to the nitrogen atom present in the latter.
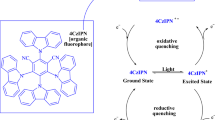
To elucidate the mechanism of the present photocatalytic cycloaddition, the redox potentials of the photocatalysts and reactants were analyzed. The oxidation potential of compound 1a is estimated to be 1.01 V (vs. SCE), based on CV data (Fig. 4b). Therefore, 1a can be oxidized by the Ru(bpy)33+ species (E1/2Ⅲ/Ⅱ = 1.29 V vs SCE)21,22,23,37. In contrast, the excited-state Ru(bpy)32+* species (E1/2Ⅱ*/Ⅰ = 0.77 V vs SCE) lacks sufficient oxidative ability to effectively oxidize 1a. The persulfate ion (S2O82⁻) is known to oxidatively quench the excited Ru(bpy)32+* species, yielding Ru(bpy)33+ and generating sulfate ions (SO42⁻) along with highly reactive sulfate radical anions (SO4•⁻), which are strong oxidants (E⁰ = 2.5–3.1 V vs. SCE) and may also oxidize 1a38,39. Photocatalytic on/off experiments for the reaction of 1a with H2O revealed that no product formation occurred during periods without light irradiation (Fig. 6a). This observation indicates that the Ru(bpy)32+/S2O82⁻ system is not an effective oxidant in the dark. Instead, the reactive oxidant species formed under light irradiation are short-lived and highly reactive, underscoring the essential role of photoactivation in enabling the cycloaddition process. A plausible reaction mechanism is proposed in Fig. 6b. Upon blue light irradiation, the Ru(bpy)32+ catalyst is excited to its photoactive state, Ru(bpy)32+*22,23. This excited species undergoes oxidative quenching by S2O82⁻, producing Ru(bpy)33+, SO42–, and SO4•⁻. The substrate 1a undergoes a two-step, one-electron oxidation, mediated by either Ru(bpy)33+ and/or SO4•⁻, to form the cis-radical cation intermediate A, which is further oxidized to the cis-dication intermediate B. These oxidation steps proceed smoothly and concomitantly, as illustrated in Fig. 2a, with concurrent regeneration of the Ru(bpy)32+ catalyst. Subsequently, water undergoes an intermolecular nucleophilic addition to the cis-dication B, yielding the cationic intermediate C. This intermediate then undergoes an intramolecular nucleophilic cyclization to afford the dispirocyclic product 3a, which features a 2,5-dihydrofuran central core.
a Light on/off experiments for the reaction of 1a with H2O. The reaction yields of 3a at various time intervals were measured by 1H NMR analysis (see SI in detail) and determined using dibromomethane as an internal standard. b Plausible photocatalysis mechanism.
In conclusion, we have demonstrated that a carbodication species, generated through either electrosynthesis or photocatalysis, exhibits high electrophilic reactivity toward heteroatom nucleophiles such as primary amines and water. This reactivity enables an intermolecular umpolung [4 + 1] cycloaddition reaction that has not been developed to date. The key to this transformation lies in the formation of a reactive cis-olefin-bridged dicationic intermediate derived from thioxanthene end-capped 1,3-dienes. This intermediate selectively undergoes cycloaddition with primary amines under electrochemical conditions and with water under photoredox catalytic conditions, yielding a class of dispirocyclic π-molecules that feature 2,5-dihydro-1H-pyrrole and 2,5-dihydrofuran cores, respectively. Given the established optoelectronic performance of structurally related dispirocyclic π-systems as hole-transporting and light-emitting materials40,41,42,43, these newly synthesized compounds are expected to exhibit high potential in future optoelectronic applications. Further studies in this direction are currently underway in our laboratory. This work highlights the synthetic potential of carbodicationic intermediates in enabling selective and mild incorporation of heteroatom nucleophiles, broadening the scope of dication chemistry and offering effective strategies for constructing complex and valuable heterocyclic frameworks.
Methods
General procedure for the electrochemical [4 + 1] cycloaddition with primary amines
To an undivided ElectraSyn cell (5 mL) vial equipped with a stirring bar was added 1a (57 mg, 0.1 mmol) and nBu4NPF6 (116.2 mg, 0.3 mmol, 0.1 M). The cell was equipped with (+) graphite/ (–) graphite electrodes and sealed with a cap. The cell was charged with argon via a balloon. A mixture of DCM/HFIP (v/v = 5:1, 3.0 mL) and aniline (14 mg, 0.15 mmol) were added to the cell via syringe. The reaction mixture was subjected to the programmed electrolysis at room temperature for 3 h at a constant current of 3 mA and a stirring speed of 400 rpm. Upon completion, the electrodes were washed with DCM, which was combined with the reaction mixture. After concentration in vacuo, the residue was purified by flash column chromatography using hexane/DCM (5/1) as an eluent to give desired product 2a in 98% yield (64.8 mg, 0.098 mmol) as white solid.
General procedure for the photocatalytic [4 + 1] cycloaddition with H2O
To a transparent test tube (Ø15 ×05 mm) was added substrate 1a (57 mg, 0.1 mmol), Ru(bpy)3Cl2·6H2O (3.7 mg, 5.0 mol%), anhydrous (NH4)2S2O8 (45.6 mg, 0.2 mmol) and anhydrous DCE/H2O (v/v = 9:1, 2 mL, 0.05 M). The test tube was stirred for 2 h under an Ar atmosphere under irradiation of a Kessil lamp (440 nm, 100% intensity) placed 5–7 cm away from the test tube. A regular fan was equipped to maintain the ambient temperature at 25 °C. Upon completion, the reaction mixture was diluted with DCM (10 mL), poured into water (20 mL) and extracted with DCM (2 × 10 mL). The combined organic layer was washed with brine (3 × 10 mL) and dried over MgSO4. After filtration and concentration in vacuo, the residue was purified by flash column chromatography using hexane/DCM (5/1) as an eluent to give desired product 3a in 98% yield (57.4 mg, 0.098 mmol) as white solid.
Data availability
Details of the experimental procedures, spectral data, and full characterizations available within the paper and Supplementary Information. The X-ray crystallographic coordinates for structures reported in this study have been deposited at the Cambridge Crystallographic Data Centre (CCDC) under deposition numbers CCDC 2453063 (2a) and 2453062 (3a). These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif. All data are available from the corresponding author upon request. The coordinates of the optimized structures in this study are provided in the Source Data file. Source data are provided with this paper.
References
Olah, G. A. Carbocations and Electrophilic Reactions. Angew. Chem. Int. Ed. 12, 173–254 (1973).
Olah, G. A. & Klumpp, D. A. Superelectrophiles and Their Chemistry (John Wiley & Sons, Inc., 2008).
Naredla, R. R. & Klumpp, D. A. Contemporary carbocation chemistry: applications in organic synthesis. Chem. Rev. 113, 6905–6948 (2013).
Tan, G. & Wang, X. Isolable Bis(triarylamine) dications: analogues of thiele’s, chichibabin’s, and Müller’s Hydrocarbons. Acc. Chem. Res. 50, 1997–2006 (2017).
Klumpp, D. A. & Kennedy, S. Superelectrophiles in ring-forming reactions. Arkivoc ii, 215–232 (2018).
Klumpp, D. A. & Anokhin, M. V. Superelectrophiles: recent advances. Molecules 25, 3281 (2020).
Reddy, V. P. & Prakash, G. K. S. Recent studies of persistent carbodications. Adv. Phys. Org. Chem. 43, 219–260 (2009).
Ishigaki, Y. et al. Redox-active tetraaryldibenzoquinodimethanes. Chem. Commun. 57, 7201–7214 (2021).
Suzuki, T., Higuchi, H., Ohkita, M. & Tsuji, T. Dual-mode electrochromism switched by proton transfer: dynamic redox properties of bis(diarylmethylenium)-type dyes. Chem. Commun. 1574−1575 (2001).
Ishigaki, Y. et al. 9,10-dihydrophenanthrene with two Spiro(dibenzocycloheptatriene) units: a highly strained caged hydrocarbon exhibiting reversible electrochromic behavior. Molecules 22, 1900 (2017).
Harimoto, T. & Ishigaki, Y. Recent advances in NIR-switchable multi-redox systems based on organic molecules. Chem. Eur. J. 31, e202403273 (2025).
Ishigaki, Y., Hashimoto, T., Sugawara, K., Suzuki, S. & Suzuki, T. Switching of Redox Properties Triggered by a Thermal Equilibrium between Closed-Shell Folded and Open-Shell Twisted Species. Angew. Chem. Int. Ed. 59, 6581–6584 (2020).
Ishigaki, Y., Hayashi, Y. & Suzuki, T. Photo- and thermal interconversion of multiconfigurational strained hydrocarbons exhibiting completely switchable oxidation to stable dicationic dyes. J. Am. Chem. Soc. 141, 18293–18300 (2019).
Zhao, J. et al. FeCl3-mediated oxidative spirocyclization of difluorenylidene diarylethanes leading to dispiro[fluorene-9,5’-indeno[2,1-a]indene-10’,9”-fluorene]s. Angew. Chem. Int. Ed. 55, 259–263 (2016).
Wei, Y. et al. Superelectrophilic-initiated C–H functionalization at the β-position of thiophenes: a one-pot synthesis of trans-stereospecific saddle-shaped cyclic compounds. J. Org. Chem. 84, 10701–10709 (2019).
Hawkins, B. C., Chalker, J. M., Coote, M. L. & Bissember, A. C. Electrochemically generated carbocations in organic synthesis. Angew. Chem. Int. Ed. 63, e202407207 (2024).
Yoshida, J.-I., Kataoka, K., Horcajada, R. & Nagaki, A. Modern strategies in electroorganic synthesis. Chem. Rev. 108, 2265–2299 (2008).
Yan, W., Kawamata, Y. & Baran, P. S. Synthetic organic electrochemical methods since 2000: on the verge of a renaissance. Chem. Rev. 117, 13230–13319 (2017). 2017.
Zhu, C., Ang, N. W. J., Meyer, T. H., Qiu, Y. & Ackermann, L. Organic electrochemistry: molecular syntheses with potential. ACS Cent. Sci. 7, 415–431 (2021).
Okumura, Y., Sato, E., Mitsudo, K. & Suga, S. Electrochemical synthesis of heterocyclic compounds via carbon–heteroatom bond formation: direct and indirect electrolysis. Chem. Lett. 53, upae146 (2024).
Prier, C. K., Rankic, D. A. & MacMillan, D. W. C. Visible light photoredox catalysis with transition metal complexes: applications in organic synthesis. Chem. Rev. 113, 5322–5363 (2013).
Wang, C.-S., Dixneuf, P. H. & Soulé, J.-F. Photoredox Catalysis for Building C–C Bonds from C(sp2)–H Bonds. Chem. Rev. 118, 7532–7585 (2018).
Tay, N. E. S., Lehnherr, D. & Rovis, T. Photons or Electrons? A critical comparison of electrochemistry and photoredox catalysis for organic synthesis. Chem. Rev. 122, 2487–2649 (2022).
Okajima, M., Suga, S., Itami, K. & Yoshida, J.-I. “Cation Pool” method based on C−C bond dissociation. effective generation of monocations and dications. J. Am. Chem. Soc. 127, 6930–6931 (2005).
Chen, J.-R., Hu, X.-Q., Lu, L.-Q. & Xiao, W.-J. Formal [4+1] annulation reactions in the synthesis of carbocyclic and heterocyclic systems. Chem. Rev. 115, 5301–5365 (2015).
Kaur, T., Wadhwa, P., Bagchi, S. & Sharma, A. Isocyanide based [4+1] cycloaddition reactions: an indispensable tool in multi-component reactions (MCRs). Chem. Commun. 52, 6958–6976 (2016).
Zhou, Y.-Y. & Uyeda, C. Catalytic reductive [4+1]-cycloadditions of vinylidenes and dienes. Science 363, 857–862 (2019).
Yang, Y., Li, H.-X., Zhu, T.-Y., Zhang, Z.-Y. & Yu, Z.-X. Rh-Catalyzed [4+1] reaction of cyclopropyl-capped dienes (but not common dienes) and carbon monoxide: reaction development and mechanistic study. J. Am. Chem. Soc. 145, 17087–17095 (2023).
Wu, Q., Hu, J., Ren, X. & Zhou, J. An efficient, overall [4+1] cycloadditon of 1,3-dienes and nitrene precursors. Chem. Eur. J. 17, 11553–11558 (2011).
Ohmura, T., Masuda, K., Takase, I. & Suginome, M. Palladium-Catalyzed Silylene-1,3-Diene [4+1] cycloaddition with use of (aminosilyl)boronic esters as synthetic equivalents of silylene. J. Am. Chem. Soc. 131, 16624–16625 (2009).
Xu, J., Wang, G., Ding, K. & Wang, X. Dirhodium-palladium dual-catalyzed [1+1+3] annulation to heterocycles using primary amines or H2O as the Heteroatom Sources. J. Am. Chem. Soc. 147, 2000–2009 (2025).
Zhang, S., Matsuyama, H., Nakamura, I., Terada, M. & Jin, T. Synthesis of curved polycyclic arenes embedded with oxepine and thiepine using ring expansion strategy. Org. Lett. 25, 5027–5032 (2023).
Lu, T. & Chen, F. Multiwfn: A multifunctional wavefunction analyzer. J. Comput. Chem. 33, 580–592 (2012).
Lu, T. A comprehensive electron wavefunction analysis toolbox for chemists. Multiwfn. J. Chem. Phys. 161, 082503 (2024).
Marset, X., Montilla-Verdú, S., Rico, E. & Guijarro, N. Beyond conventional organic electrosynthesis: the role of fluorinated solvents. ACS Electrochem. 1, 3–19 (2025).
Zhang, J. et al. First-principles investigation of the unique role of anode surfaces in organic electrochemical reactions. J. Phys. Chem. C 127, 15215–15226 (2023).
Kalyanasundaram, K. Photophysics, photochemistry and solar energy conversion and its analogues. Coord. Chem. Rev. 46, 159–244 (1982).
Minisci, F. & Citterio, A. Electron-transfer processes: peroxydisulfate, a useful and versatile reagent in organic chemistry. Acc. Chem. Res. 16, 27–32 (1983).
Dai, C., Meschini, F., Narayanam, J. M. R. & Stephenson, C. R. J. Friedel–Crafts Amidoalkylation via Thermolysis and Oxidative Photocatalysis. J. Org. Chem. 77, 4425–4431 (2012).
Romain, M. et al. ortho-, meta-, and para-dihydroindenofluorene derivatives as host materials for phosphorescent OLEDs. Angew. Chem. Int. Ed. 54, 1176–1180 (2015).
Gao, K. et al. Di-spiro-based hole-transporting materials for highly efficient perovskite solar cells. Adv. Energy Mater. 8, 1800809 (2018).
Yu, W. et al. A dispiro-type fluorene-indenofluorene-centered hole transporting material for efficient planar perovskite solar cells. Sol. RRL 2, 1800048 (2018).
Sakamaki, T. et al. Doubly spiro-conjugated chiral carbocycles exhibiting SOMO−HOMO inversion in persistent radical cations. J. Am. Chem. Soc. 146, 12712–12722 (2024).
Acknowledgements
This research was supported by a Grant-in-Aid for Transformative Research Areas (A) “Green Catalysis Science for Renovating Transformation of Carbon-Based Resources” (JP23H04908, M.T.) from MEXT, Japan and a Grant-in-Aid for Scientific Research (S) (JP22H04969, M.T.) from the JSPS. The computation was performed using Research Center for Computational Science, Okazaki, Japan (Project: 25-IMS-C110).
Author information
Authors and Affiliations
Contributions
T.J. and M.T. conceived and designed the project and wrote the manuscript with the assistance of other authors. H.M. and K.Y. conducted experiments. T.S. performed theoretical calculations. All the authors analyzed the data and discussed the results.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Debajit Maiti, and the other, anonymous, reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Matsuyama, H., Yokoyama, K., Sato, T. et al. Electrochemical and photocatalytic generation of cis-olefin-bridged carbodication for umpolung [4+1] cycloaddition. Nat Commun 17, 2270 (2026). https://doi.org/10.1038/s41467-026-68836-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-68836-z





![Fig. 5: Study of photoredox catalytic [4 + 1] cycloaddition of thioxanthene end-capped 1,3-dienes 1 with H2O.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs41467-026-68836-z/MediaObjects/41467_2026_68836_Fig5_HTML.png)
![Fig. 6: Reaction mechanism for the photocatalytic [4 + 1] cycloaddition with H2O.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs41467-026-68836-z/MediaObjects/41467_2026_68836_Fig6_HTML.png)